Feb 25, 2026

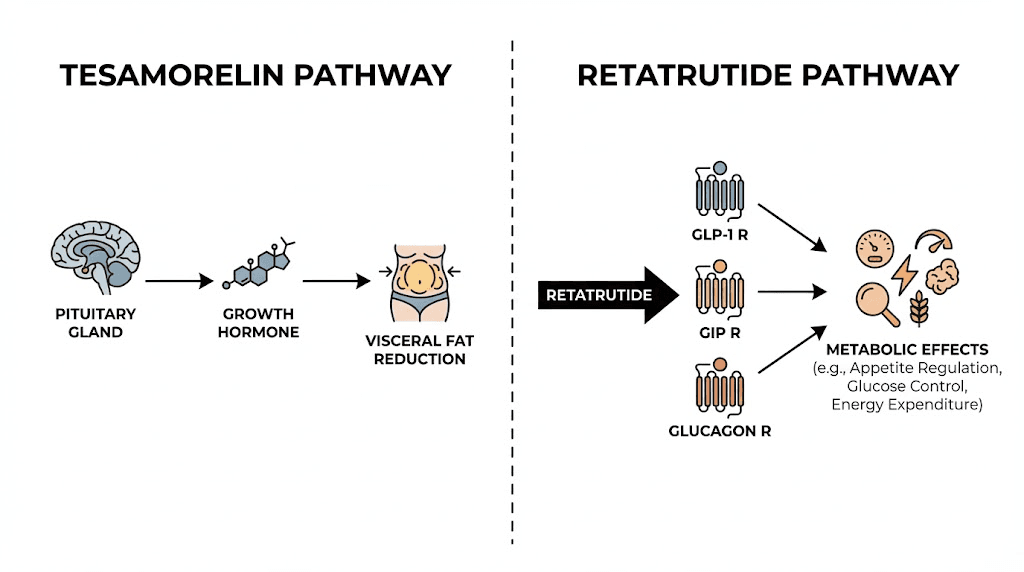
At the cellular level, tesamorelin and retatrutide trigger two entirely different cascades. One pushes the pituitary gland to release growth hormone in natural pulses. The other activates three metabolic receptors simultaneously, suppressing appetite, improving insulin sensitivity, and ramping up energy expenditure. They do not compete. They do not overlap. They work on parallel tracks that, in theory, could produce something neither compound achieves alone.
That theory is exactly what makes this question so compelling for researchers exploring peptide-based fat loss strategies. Tesamorelin, an FDA-approved GHRH analog, has spent years proving its ability to reduce visceral fat through growth hormone pathways. Retatrutide, a triple-receptor agonist still in Phase 3 clinical trials, has posted some of the most impressive weight loss numbers ever recorded in obesity research, hitting 24.2% body weight reduction in 48 weeks. Together, their complementary mechanisms suggest a theoretical synergy that targets different fat depots through entirely different biological pathways.
But theory and practice are not the same thing. No clinical trial has ever tested this combination. No regulatory body has approved it. The potential benefits come packaged with real risks, from insulin resistance concerns to unknown long-term interactions that no researcher can currently predict. This guide breaks down everything known about both compounds, examines the theoretical case for combining them, maps the risks you need to understand, and provides the monitoring framework that any serious researcher would require before considering this stack. If you are looking for a comprehensive peptide stacking resource, this is where the science meets practical application.
Understanding tesamorelin: the growth hormone pathway
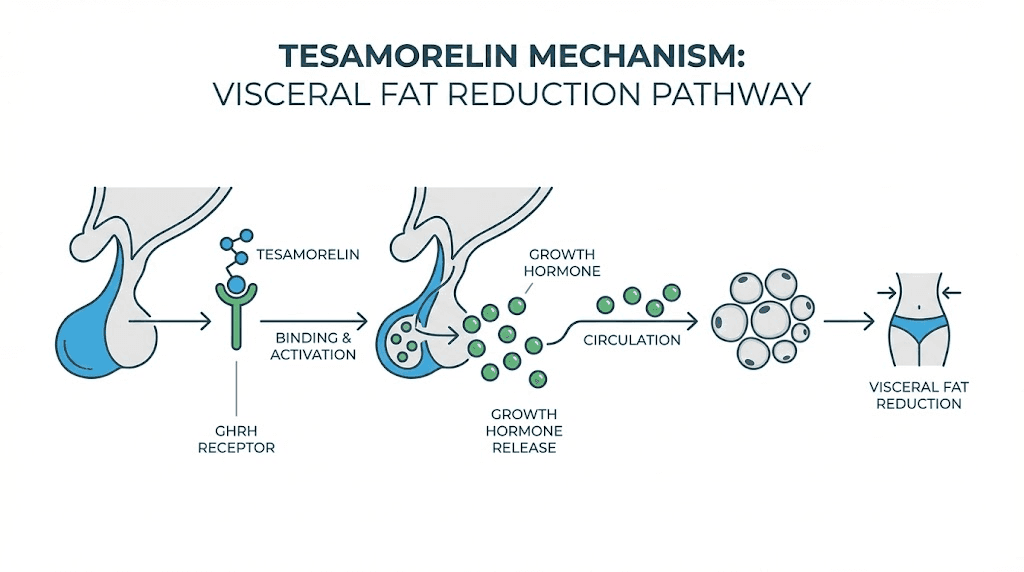
Tesamorelin is not a growth hormone. That distinction matters more than most people realize. It is a synthetic analog of growth hormone-releasing hormone (GHRH), a 44-amino acid peptide that mimics what the hypothalamus naturally produces to signal the pituitary gland. When tesamorelin binds to GHRH receptors on pituitary somatotroph cells, it triggers the adenylate cyclase-cAMP signaling cascade, which stimulates the synthesis and secretion of endogenous growth hormone in pulsatile bursts that mirror the body natural rhythm.
This pulsatile release pattern is critical. Direct growth hormone injections flood the system with supraphysiological levels that bypass feedback loops. Tesamorelin works through the existing regulatory architecture, meaning the body retains its ability to modulate output. When growth hormone levels rise too high, somatostatin release increases to bring them back down. The feedback mechanism stays intact.
The downstream effects follow a predictable cascade. Growth hormone acts directly on adipose tissue through lipolytic pathways, breaking down stored triglycerides. It also stimulates the liver to produce insulin-like growth factor 1 (IGF-1), which promotes tissue growth, supports protein synthesis, and contributes to overall metabolic regulation. For researchers studying IGF peptide biology, tesamorelin represents one of the cleanest ways to elevate IGF-1 without direct exogenous administration.
FDA approval and clinical evidence
Tesamorelin carries FDA approval under the brand name Egrifta, specifically for reducing excess visceral abdominal fat in HIV-positive patients with lipodystrophy. That approval came with substantial clinical evidence. Studies demonstrated significant reductions in visceral adipose tissue (VAT) within 6 to 12 weeks of consistent daily administration, with effects maintained for the duration of treatment.
A key study published in the New England Journal of Medicine showed that tesamorelin reduced trunk fat by approximately 15% compared to placebo over 26 weeks. More importantly for researchers considering stacking scenarios, the compound increased lean body mass without altering subcutaneous fat distribution. It specifically targets the metabolically dangerous fat surrounding organs. That selectivity is what makes researchers interested in combining it with compounds that target different fat depots.
The safety profile from clinical trials revealed manageable side effects including joint pain, muscle aches, peripheral edema, and symptoms resembling carpal tunnel syndrome. These effects relate directly to growth hormone elevation and typically resolve with dose adjustment. For those tracking growth hormone peptide side effects, tesamorelin produces a milder profile than direct GH administration precisely because it works through the natural pulsatile release system.
The visceral fat connection
Visceral fat is not just cosmetically undesirable. It is metabolically active tissue that secretes inflammatory cytokines, disrupts insulin signaling, and contributes to cardiovascular disease risk. Traditional weight loss approaches, including caloric restriction and exercise, reduce both visceral and subcutaneous fat simultaneously. Tesamorelin offers something unusual: preferential reduction of visceral fat while preserving or even increasing lean mass.
This specificity comes from growth hormone direct lipolytic action on visceral adipocytes, which express higher concentrations of growth hormone receptors than subcutaneous fat cells. The result is targeted mobilization of the most dangerous fat compartment. Clinical data shows reductions in VAT beginning within 1 to 2 weeks, with substantial changes evident by 8 to 12 weeks of daily 2mg subcutaneous injections.
Understanding this mechanism is essential for anyone considering peptides for visceral fat reduction. Tesamorelin addresses what many weight loss peptide approaches miss entirely: the deep abdominal fat that drives metabolic syndrome.
Dosing and administration
The standard clinical protocol calls for 2mg administered subcutaneously once daily, typically in the abdominal region with injection sites rotated regularly. A newer formulation (Egrifta WR) delivers 1.28mg daily with equivalent efficacy through improved bioavailability. The injection is straightforward for anyone familiar with subcutaneous peptide injection techniques.
Tesamorelin has a half-life of approximately 26 minutes after subcutaneous injection, though the growth hormone release it triggers persists for hours. This is not a weekly injection like retatrutide. It demands daily consistency. Miss several days and the growth hormone pulsatility that drives fat loss diminishes. Researchers exploring this compound need to commit to the daily protocol or expect suboptimal results.
Reconstitution follows standard peptide preparation methods. The lyophilized powder requires bacteriostatic water, and researchers familiar with peptide reconstitution procedures will find the process identical to other injectable peptides. Storage requires refrigeration at 2 to 8 degrees Celsius, consistent with standard peptide storage guidelines.
Understanding retatrutide: the triple-receptor approach
Retatrutide represents something genuinely new in metabolic pharmacology. While semaglutide activates one receptor (GLP-1) and tirzepatide activates two (GLP-1 and GIP), retatrutide activates three: GLP-1, GIP, and glucagon receptors simultaneously. This triple-agonist approach creates metabolic effects that exceed what any single or dual-receptor compound achieves alone.
The peptide consists of 39 amino acids engineered from a GIP peptide backbone, modified to stimulate all three target receptors. Compared to natural ligands, retatrutide shows lower potency at GLP-1 and glucagon receptors but higher potency at GIP receptors. This deliberate tuning of receptor affinity allows the compound to produce balanced activation across all three pathways without overwhelming any single one.
For researchers who have followed the evolution of retatrutide dosing protocols, the compound represents the cutting edge of incretin-based therapy. It builds on the foundation laid by semaglutide and tirzepatide but adds the glucagon receptor component that may explain its superior weight loss outcomes.
The three receptors explained
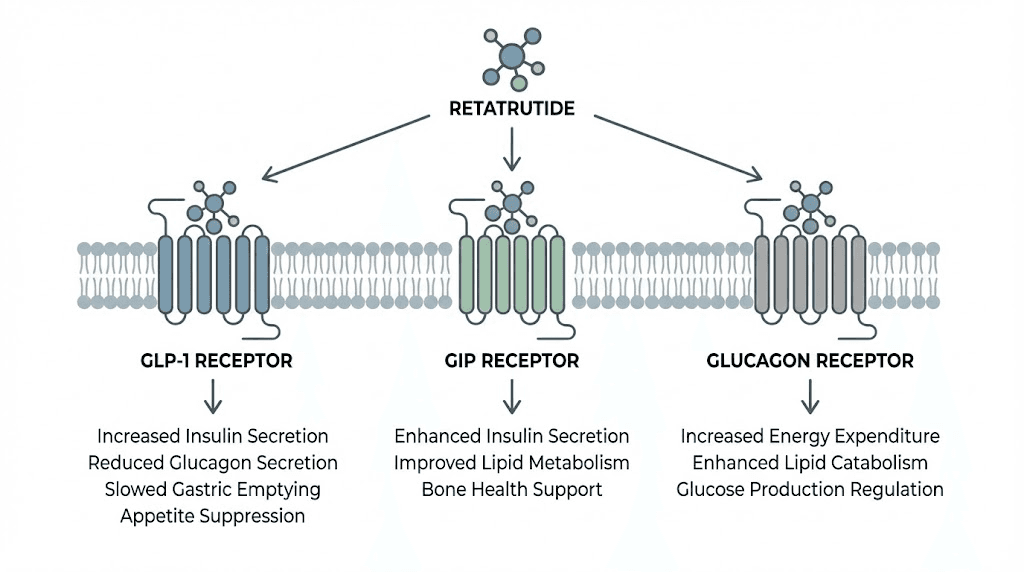
GLP-1 receptor activation suppresses appetite, slows gastric emptying, and enhances insulin secretion in response to food. This is the mechanism behind semaglutide, tirzepatide, and every other GLP-1 agonist on the market. Retatrutide includes this pathway but does not rely on it exclusively.
GIP receptor activation complements GLP-1 effects by enhancing insulin sensitivity and potentially improving fat metabolism. The addition of GIP agonism is what separated tirzepatide from pure GLP-1 drugs, and researchers tracking the progression from semaglutide to tirzepatide to retatrutide can see how each additional receptor adds another layer of metabolic benefit.
Glucagon receptor activation is where retatrutide diverges most dramatically from its predecessors. Glucagon promotes hepatic glycogenolysis, stimulates gluconeogenesis, and critically, increases energy expenditure and thermogenesis. While GLP-1 agonists primarily work through appetite suppression (the input side of the energy equation), glucagon activation works on the output side, increasing how many calories the body burns at rest. This dual-sided approach, reducing intake while boosting expenditure, helps explain the exceptional weight loss numbers.
Clinical trial results
Phase 2 trial data published in the New England Journal of Medicine showed that participants receiving the highest dose of retatrutide (12mg weekly) achieved 24.2% body weight reduction over 48 weeks. That number surpasses anything recorded with semaglutide (approximately 15-16%) or tirzepatide (approximately 20-22%). The placebo group lost only 2.1% over the same period.
Beyond raw weight loss, the metabolic improvements were remarkable. Researchers tracking retatrutide dosing for weight loss should note that 72% of participants with prediabetes at baseline reverted to normal blood sugar levels. HbA1c improved by up to 2.2% in participants with type 2 diabetes. Waist circumference, blood pressure, fasting glucose, insulin levels, and lipid profiles all showed significant improvement.
Perhaps most relevant to the tesamorelin combination question, retatrutide demonstrated extraordinary liver fat reduction. At the 12mg dose, liver fat decreased by 82.4% from baseline at 24 weeks, with 86% of participants achieving normal liver fat levels below 5%. This hepatic effect operates through glucagon receptor activation and has implications for anyone studying metabolic syndrome comprehensively.
Side effects and tolerability
Gastrointestinal side effects dominate the retatrutide safety profile. Nausea, vomiting, diarrhea, and constipation affected 60 to 80% of participants at higher doses, though most symptoms were mild to moderate in severity. The fatigue associated with GLP-1 compounds appeared in retatrutide trials as well, likely related to reduced caloric intake during the adaptation phase.
Gradual dose titration significantly reduced side effect severity. Participants who started at 1mg and escalated slowly over weeks experienced substantially fewer GI issues than those assigned directly to higher doses. GI symptom rates nearly doubled when participants skipped the titration phase, making the proper retatrutide dose schedule a critical factor in tolerability.
Hair loss emerged as a concern in some participants, consistent with what researchers observe across the GLP-1 agonist class. Anyone monitoring for retatrutide-related hair loss should understand that this typically relates to rapid weight loss rather than direct drug toxicity, and it generally resolves as weight stabilizes.
Dosing protocols
Phase 2 clinical trials used a titration schedule starting at 0.5mg weekly, escalating through 1mg, 2mg, 4mg, 8mg, and reaching maintenance doses of up to 12mg weekly over several months. The gradual escalation serves two purposes: it allows the body to adapt to GLP-1 receptor activation (reducing nausea), and it permits glucagon receptor effects to build progressively without overwhelming metabolic pathways.
Retatrutide is administered as a once-weekly subcutaneous injection. This stands in stark contrast to tesamorelin daily dosing requirement, creating a practical consideration for anyone evaluating the combination. Researchers familiar with retatrutide injection techniques know that the weekly schedule offers convenience, but the starting dose selection and titration pace determine both efficacy and tolerability.
Current Phase 3 trials (expected to report results in 2027) are exploring doses ranging from 4mg to 12mg weekly, with some protocols testing up to 16mg. The retatrutide dosage calculation for individual researchers depends on body weight, metabolic goals, and tolerance to GI side effects during titration.
Comparing mechanisms side by side
The fundamental appeal of combining tesamorelin and retatrutide comes down to mechanism separation. These compounds do not work through the same pathways. They do not compete for the same receptors. They produce their effects through entirely independent biological systems that happen to converge on a shared outcome: improved body composition.
Factor | Tesamorelin | Retatrutide |
|---|---|---|
Primary mechanism | GHRH receptor agonist | GLP-1/GIP/glucagon triple agonist |
Target receptors | Pituitary GHRH receptors | GLP-1R, GIPR, GCGR |
Primary fat target | Visceral adipose tissue | Total body fat (subcutaneous and visceral) |
Weight loss mechanism | GH-mediated lipolysis | Appetite suppression plus energy expenditure |
Muscle effects | Preserves and may increase lean mass | May preserve muscle during weight loss |
Dosing frequency | Daily (2mg subcutaneous) | Weekly (titrated to 8-12mg) |
FDA status | Approved (HIV lipodystrophy) | Investigational (Phase 3) |
Liver fat effects | Modest reduction through GH pathways | 82% reduction at 12mg dose |
Key hormone affected | Growth hormone and IGF-1 | Insulin, glucagon, GLP-1, GIP |
Half-life | 26 minutes (GH effects persist hours) | Approximately 6 days |
The table reveals why researchers find this combination theoretically interesting. Every major mechanism differs. Tesamorelin works through growth hormone. Retatrutide works through incretin and glucagon pathways. Tesamorelin targets visceral fat preferentially. Retatrutide reduces total body fat with particular effectiveness for liver fat. One preserves muscle through anabolic GH effects. The other supports muscle preservation through mechanisms that are still being studied.
For researchers building effective peptide stacks for weight loss, mechanism separation is the gold standard. Stacking two GLP-1 agonists makes no sense because they compete for the same receptors. Stacking a GLP-1 agonist with a GHRH analog creates genuinely parallel pathways, each contributing independently to the end goal. The peptide stacking calculator can help researchers understand how different compounds interact when combined.
Where the mechanisms could synergize
Three areas of potential synergy stand out in the theoretical analysis.
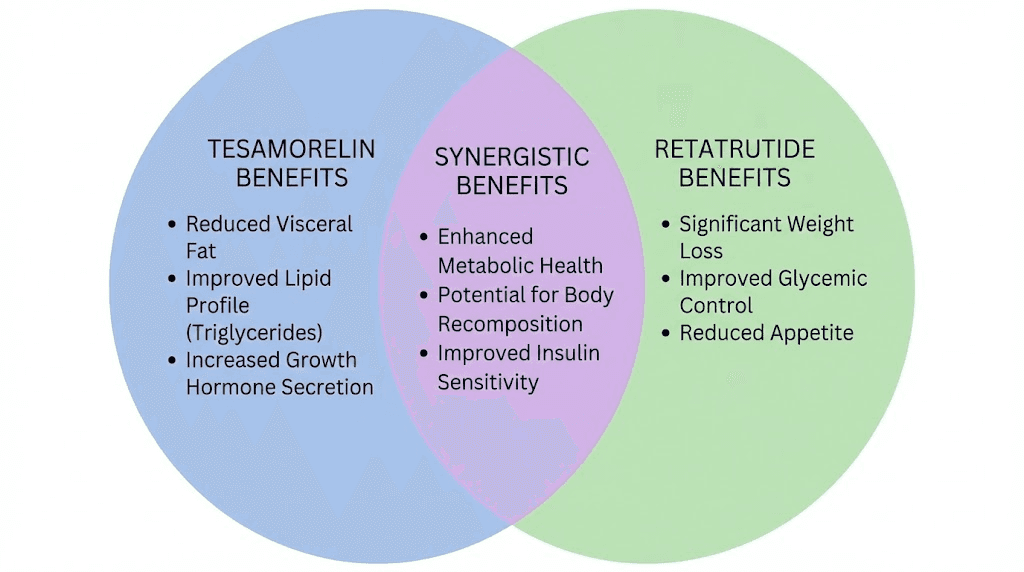
First, fat depot coverage. Retatrutide produces substantial overall weight loss but does not preferentially target visceral fat the way tesamorelin does. Adding tesamorelin to a retatrutide protocol could theoretically accelerate visceral fat reduction beyond what retatrutide achieves alone. For researchers specifically concerned about stubborn abdominal fat, this dual approach covers both superficial and deep fat compartments.
Second, muscle preservation. Rapid weight loss from GLP-1 agonists comes with a well-documented risk of muscle loss. Tesamorelin elevates growth hormone and IGF-1, both of which support protein synthesis and lean tissue maintenance. The combination could theoretically protect against the muscle wasting that sometimes accompanies aggressive caloric restriction. Researchers studying simultaneous fat loss and muscle preservation will recognize this as addressing the most common complaint about GLP-1 therapy.
Third, metabolic scope. Retatrutide improves glucose metabolism, lipid profiles, and liver fat through incretin and glucagon pathways. Tesamorelin improves body composition through growth hormone pathways that also support bone density, skin quality, and recovery capacity. The combined metabolic footprint would be broader than either compound alone, touching more systems through more pathways.
Where the mechanisms could conflict
Potential conflicts deserve equal attention. The most significant concern involves insulin and glucose regulation.
Growth hormone is inherently diabetogenic. It promotes insulin resistance by interfering with insulin signaling in peripheral tissues. Tesamorelin mitigates this through pulsatile release (avoiding the sustained supraphysiological GH levels that direct injection creates), and clinical trials in type 2 diabetic patients showed no significant changes in glucose control. But adding tesamorelin growth hormone elevation to retatrutide metabolic milieu creates an interaction that has never been studied.
Retatrutide improves insulin sensitivity through GLP-1 and GIP receptor activation. In theory, this could counterbalance any insulin resistance from tesamorelin GH elevation. In practice, nobody knows. The interaction between growth hormone pathway activation and triple-receptor incretin agonism has zero clinical data supporting its safety or efficacy.
Researchers considering complex peptide protocols need to understand that theoretical mechanism separation does not guarantee practical safety. Biological systems interact in ways that isolated pathway analysis cannot fully predict.
The theoretical case for combining tesamorelin and retatrutide
Researchers interested in this combination are not being reckless. They are responding to a genuine gap in what either compound achieves independently. Retatrutide produces remarkable overall weight loss but may not optimally target the visceral fat that drives the most dangerous metabolic consequences. Tesamorelin excels at visceral fat reduction but produces modest overall weight loss. The combination addresses both limitations simultaneously.
Complementary fat loss pathways
Retatrutide reduces caloric intake through appetite suppression (GLP-1 and GIP) while increasing caloric expenditure through thermogenesis (glucagon). This creates a substantial energy deficit that drives overall fat loss. But the fat loss is distributed across all adipose depots based on their metabolic activity and receptor density.
Tesamorelin adds a targeted intervention. Growth hormone direct lipolytic effect on visceral adipocytes, which express more GH receptors than subcutaneous fat cells, creates preferential mobilization of the most metabolically dangerous fat. In a combined protocol, retatrutide would drive overall weight reduction while tesamorelin would accelerate visceral fat clearance beyond what general weight loss achieves.
For context, consider the clinical data separately. Retatrutide at 12mg weekly produced 24.2% total body weight loss over 48 weeks. Tesamorelin at 2mg daily produced approximately 15% visceral fat reduction over 26 weeks. Neither compound produces both effects optimally. A theoretical combination targets the complete fat loss picture, both the total and the visceral.
Researchers exploring the most effective peptide approaches for fat loss will recognize this as a comprehensive strategy rather than a single-pathway approach.
The muscle preservation argument
Weight loss from GLP-1 agonists typically includes 25 to 40% lean tissue loss. That means for every 10 pounds lost, 2.5 to 4 pounds comes from muscle. This ratio concerns researchers focused on long-term metabolic health, since muscle mass is a primary driver of basal metabolic rate and functional capacity.
Tesamorelin addresses this directly. Growth hormone promotes protein synthesis, supports nitrogen retention, and stimulates IGF-1 production, all of which counteract the catabolic signals that caloric deficit creates. Clinical studies show tesamorelin increases lean body mass even while reducing visceral fat. Adding this anabolic support to a retatrutide-driven caloric deficit could theoretically shift the composition of weight loss toward greater fat loss and less muscle loss.
This consideration becomes especially relevant for male researchers and women over 40 who face accelerated muscle loss during weight reduction. The growth hormone pathway that tesamorelin activates supports the very tissues that aggressive weight loss strategies tend to sacrifice.
The combination also has potential relevance for muscle growth protocols where researchers want to recompose body tissue rather than simply lose weight.
Broader metabolic coverage
Metabolic syndrome involves multiple interconnected dysfunctions: insulin resistance, dyslipidemia, hypertension, visceral obesity, and chronic inflammation. No single compound addresses all of these simultaneously. But the tesamorelin-retatrutide combination, at least in theory, comes closer than most approaches.
Retatrutide contribution includes improved insulin sensitivity (GLP-1 and GIP), reduced liver fat (glucagon), improved lipid profiles (all three receptors), and reduced blood pressure (weight loss effect). These improvements span the cardiovascular risk factors that metabolic syndrome creates.
Tesamorelin adds visceral fat reduction (directly addressing the obesity component), improved body composition (supporting metabolic rate), and potential cardiovascular benefits through reduction of the inflammatory visceral fat depot. The GH and IGF-1 elevation also supports bone density and connective tissue integrity, which become increasingly important as researchers age.
For those studying peptides for anti-aging and longevity, this broader metabolic coverage represents a multi-system approach rather than a single-target intervention.
Potential risks and safety concerns
The theoretical benefits of this combination come with substantial unknowns. No human clinical trial has ever tested tesamorelin and retatrutide together. Every risk assessment relies on extrapolation from individual compound data and theoretical pharmacological analysis. That level of uncertainty demands serious attention.
The insulin resistance paradox
This is the primary safety concern, and it deserves detailed explanation.
Growth hormone is diabetogenic. It reduces peripheral insulin sensitivity by interfering with insulin receptor signaling, promoting hepatic gluconeogenesis, and increasing free fatty acid levels. These effects are well-documented with exogenous GH administration and represent the main metabolic risk of elevated growth hormone levels.
Tesamorelin partially mitigates this risk through pulsatile GH release that preserves feedback mechanisms. Clinical trials in type 2 diabetic patients showed no significant worsening of glycemic control over 12 weeks of treatment. IGF-1 levels increased by approximately 181 micrograms per liter but remained within normal physiological ranges. The feedback inhibition of IGF-1 on further GH secretion stayed intact, preventing the GH excess that drives insulin resistance with direct injection.
But those studies tested tesamorelin in isolation. Adding retatrutide changes the metabolic environment significantly. Retatrutide improves insulin sensitivity through GLP-1 and GIP receptor activation, potentially counterbalancing any tesamorelin-related insulin resistance. However, the glucagon receptor activation in retatrutide independently promotes hepatic glucose output, which could compound with GH effects on gluconeogenesis to create unpredictable blood sugar fluctuations.
Nobody knows which force wins in this interaction. The insulin-sensitizing effects of GLP-1/GIP agonism versus the insulin-desensitizing effects of GH elevation, modulated by glucagon-driven hepatic glucose output. Researchers who track their metabolic peptide protocols carefully will recognize this as the kind of variable that demands close monitoring rather than assumption.
Gastrointestinal burden
Retatrutide already produces significant GI side effects. Nausea affects the majority of users, particularly during dose titration. Vomiting, diarrhea, and constipation follow in frequency. These symptoms relate primarily to GLP-1 receptor activation slowing gastric motility and affecting appetite centers in the brain.
Tesamorelin can increase appetite through growth hormone effects on hunger signaling. In an isolated context, this might seem beneficial for someone experiencing retatrutide-induced appetite suppression. In practice, the combination could create conflicting signals: GLP-1 suppressing appetite while GH promotes it, potentially worsening nausea or creating unpredictable eating patterns.
Additionally, tesamorelin side effects include joint pain, muscle aches, and peripheral edema. When combined with retatrutide GI effects and potential fatigue, the cumulative side effect burden may significantly impact quality of life during the initial weeks of combined use. Managing GLP-1 related headaches becomes more complex when growth hormone-related symptoms overlap.
IGF-1 elevation concerns
Tesamorelin raises IGF-1 levels. In clinical studies, the increase was significant (approximately 181 micrograms per liter above baseline) but remained within physiological norms. IGF-1 is a growth factor, and chronically elevated levels carry theoretical concerns about promoting the growth of pre-existing neoplasms.
This concern appears in the prescribing information for Egrifta and represents a known risk of any intervention that elevates growth hormone or IGF-1. For researchers considering long-term use, regular IGF-1 monitoring becomes essential. The risk profile changes when tesamorelin is combined with retatrutide, which through its metabolic effects could alter the hormonal environment in ways that affect IGF-1 clearance or activity.
Researchers interested in the biology of IGF-1 and its implications should understand that this is not a trivial concern. While clinical data does not show increased cancer risk with tesamorelin specifically, the absence of evidence from combination protocols means the risk cannot be quantified.
Unknown long-term interactions
The most honest assessment of combining tesamorelin and retatrutide is simply this: nobody knows what happens. Individual compound safety profiles exist. Theoretical pharmacological analysis can identify potential interaction points. But the actual outcome of combining a daily GHRH analog with a weekly triple-receptor agonist in a living human body remains entirely unknown.
Biological systems are not additive. Two compounds that are safe individually can produce emergent effects when combined, effects that neither compound would produce alone. Enzyme induction, receptor cross-talk, altered clearance rates, and unexpected feedback loop modifications are all possibilities that theoretical analysis cannot reliably predict.
This uncertainty is not a reason to dismiss the combination entirely. It is a reason to approach it with extreme caution, comprehensive monitoring, and the involvement of qualified medical supervision. Self-experimentation without medical oversight is dangerous with either compound alone. With the combination, it becomes genuinely reckless.
Dosing considerations for a theoretical combined protocol
No established protocol exists for combining tesamorelin and retatrutide. What follows is theoretical framework based on individual compound dosing data and pharmacological principles. This is not medical advice. This is analysis of how individual dosing parameters might inform a combined approach.
Tesamorelin dosing in a combination context
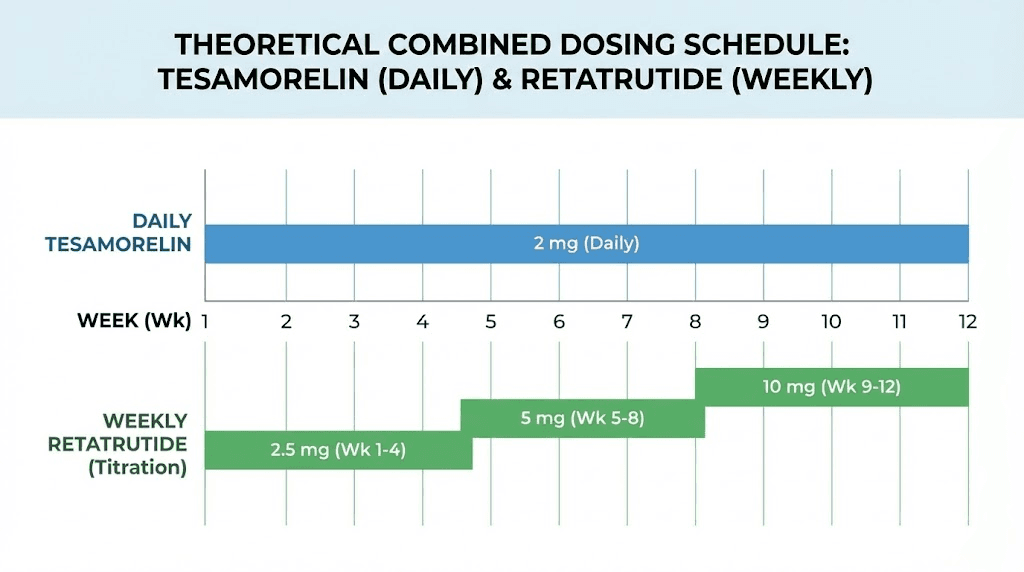
The standard tesamorelin dose is 2mg subcutaneously once daily. This dose has been validated in clinical trials for efficacy and safety. In a combination protocol, some researchers theorize that starting at a lower dose (1mg daily) and assessing tolerance before escalating to the full 2mg might reduce the risk of compounding side effects with retatrutide.
Timing matters. Tesamorelin is typically administered in the morning on an empty stomach, as food intake can blunt the GH response. Since retatrutide is a weekly injection, the daily tesamorelin schedule would continue independently. Spacing the weekly retatrutide injection and any tesamorelin dose adjustment by at least 24 hours could help researchers isolate which compound is responsible for any symptoms that emerge.
For researchers already tracking their peptide dosing with calculators, tesamorelin dosing does not require weight-based adjustment. The fixed 2mg dose was effective across body weight ranges in clinical trials.
Retatrutide dosing in a combination context
Retatrutide demands careful titration regardless of combination status. The standard Phase 2 trial schedule began at 0.5 to 1mg weekly, escalating every 4 weeks through 2mg, 4mg, 8mg, and up to 12mg. In a combined protocol, even slower titration makes theoretical sense to monitor for interactions.
A conservative approach might extend each titration step to 6 weeks rather than 4, allowing more time to assess the cumulative effects of both compounds before increasing the retatrutide dose. The standard retatrutide dosage chart provides the baseline, but researchers combining with tesamorelin would likely need to adjust based on individual response.
The injection itself follows standard subcutaneous technique. Researchers can refer to retatrutide injection site guidance for proper administration. The weekly schedule means that most days involve only the tesamorelin injection, with one day per week including both compounds (injected at different sites).
Reconstitution and storage
Both compounds require reconstitution from lyophilized powder using bacteriostatic water. Standard peptide reconstitution procedures apply, and the peptide reconstitution calculator can determine exact water volumes for desired concentrations.
Critical storage note: both compounds require refrigeration after reconstitution. They should be stored separately at 2 to 8 degrees Celsius. Never mix the two compounds in the same vial. Researchers who need guidance on proper storage can review post-reconstitution storage protocols and peptide refrigeration timelines for detailed guidance.
For retatrutide specifically, reconstitution requires careful attention to volume. Researchers using the correct bacteriostatic water ratio for retatrutide will find that standard concentrations make weekly dose measurement straightforward with an insulin syringe.
Monitoring and bloodwork requirements
Any researcher considering this combination without comprehensive monitoring is making a serious mistake. The interaction between growth hormone pathway activation and triple-receptor incretin agonism creates metabolic variables that must be tracked consistently. The monitoring framework below represents the minimum standard for responsible research.
Baseline bloodwork before starting
Before initiating either compound, establish baseline values for every marker you plan to track. This includes a complete metabolic panel, fasting insulin, HbA1c, fasting glucose, lipid panel (complete with LDL particle count if available), IGF-1, growth hormone levels (random and stimulated if possible), liver enzymes (ALT, AST, GGT), kidney function markers (creatinine, BUN, eGFR), complete blood count, and thyroid panel.
Body composition assessment should use DEXA scan or equivalent for accurate visceral fat, subcutaneous fat, and lean mass quantification. Waist circumference and waist-to-hip ratio provide supplementary tracking metrics. These baseline measurements become the reference against which all changes are evaluated.
For researchers who already monitor their protocols through structured peptide dosage tracking, this monitoring framework integrates into existing systems. The key is consistency: same lab, same time of day, same fasting conditions for every draw.
Critical markers during combined use
IGF-1 levels: Check every 4 weeks for the first 12 weeks, then every 8 weeks if stable. Tesamorelin elevates IGF-1, and this elevation must remain within normal physiological range (roughly 100 to 300 ng/mL depending on age). If IGF-1 exceeds the upper normal limit, tesamorelin dose reduction or discontinuation should be considered immediately.
Fasting glucose and insulin: Check every 2 weeks for the first 8 weeks, then monthly. This is the primary safety metric for the insulin resistance concern. Any trend toward higher fasting glucose or declining insulin sensitivity should trigger protocol reassessment. The combination of growth hormone elevation and glucagon receptor activation creates two potential forces pushing glucose upward, and only GLP-1/GIP agonism pushes it down.
HbA1c: Check at baseline, 6 weeks, 12 weeks, and quarterly thereafter. This provides the medium-term view of glucose control that spot fasting glucose measurements can miss. A rising HbA1c despite retatrutide use suggests that the tesamorelin GH effect is overwhelming the incretin benefits.
Liver enzymes: Check monthly. Both compounds affect liver metabolism through different pathways. Retatrutide reduces liver fat dramatically. Tesamorelin affects hepatic IGF-1 production and gluconeogenesis. Monitoring liver enzymes ensures neither compound is causing hepatotoxicity.
Body composition: DEXA scan at baseline, 12 weeks, and 24 weeks minimum. This objectively quantifies changes in visceral fat, subcutaneous fat, and lean mass, allowing researchers to evaluate whether the combination is producing the hypothesized benefits (preferential visceral fat loss with muscle preservation).
Red flags that should stop the protocol
Certain findings should trigger immediate discontinuation of one or both compounds:
IGF-1 levels exceeding the age-appropriate upper normal limit on two consecutive tests
Fasting glucose rising above 126 mg/dL or HbA1c above 6.5% (indicating new-onset diabetes)
Liver enzymes (ALT or AST) exceeding three times the upper limit of normal
Persistent severe nausea, vomiting, or abdominal pain suggesting gastroparesis or pancreatitis
New or worsening carpal tunnel syndrome symptoms
Peripheral edema that does not resolve with fluid management
Any sign of an allergic or hypersensitivity reaction
These red flags are not exhaustive. Any unexpected symptom that develops after initiating the combined protocol should be evaluated in the context of potential compound interactions.
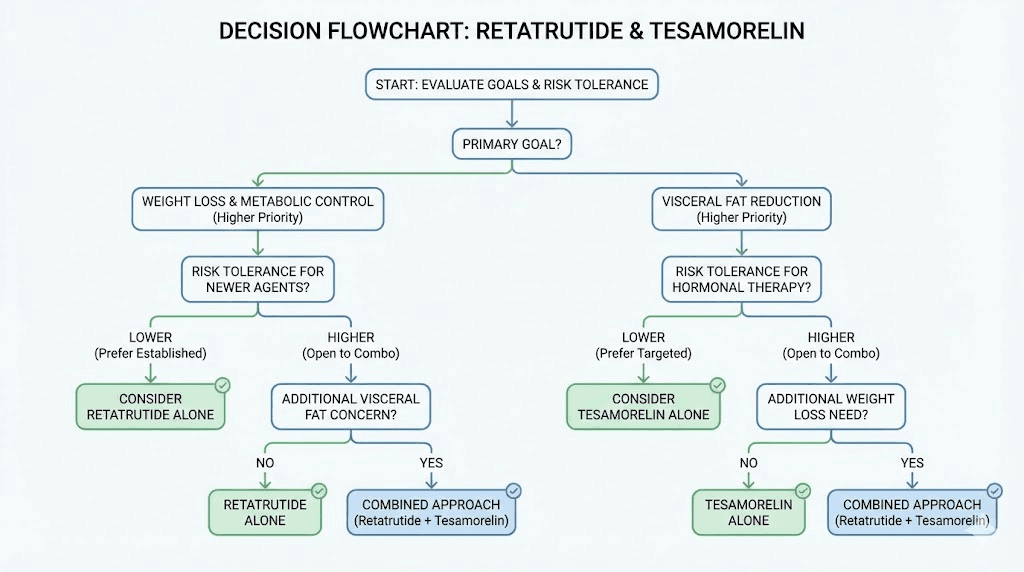
Alternative approaches to consider
Before committing to an untested combination, researchers should consider whether simpler approaches might achieve similar goals with known safety profiles.
Retatrutide alone
The 24.2% weight loss observed with retatrutide at 12mg weekly is extraordinary by any standard. Combined with 82% liver fat reduction, improved insulin sensitivity, and comprehensive metabolic improvements, retatrutide alone may accomplish what researchers seek from the combination. The question becomes whether the additional visceral fat targeting and muscle preservation from tesamorelin adds enough value to justify the unknown risks.
For researchers primarily focused on weight loss, determining the right retatrutide dose and following proper titration may produce results that make tesamorelin addition unnecessary. The comparison between retatrutide and semaglutide demonstrates how much more effective the triple-agonist approach already is compared to single-receptor compounds.
Tesamorelin alone
For researchers whose primary concern is visceral fat reduction rather than overall weight loss, tesamorelin alone may be the more appropriate choice. It has FDA approval, a well-characterized safety profile, and proven efficacy for its specific target. The standard peptide protocols for tesamorelin are well-established and carry far less uncertainty than any combination approach.
Tesamorelin also offers muscle preservation benefits through GH and IGF-1 pathways, making it valuable for researchers prioritizing safe muscle maintenance during body recomposition.
Retatrutide plus exercise and nutrition optimization
Adding structured resistance training and optimized protein intake to a retatrutide protocol may address the muscle preservation concern without the risks of adding tesamorelin. Research consistently shows that resistance exercise during GLP-1 agonist-induced weight loss significantly reduces lean mass loss, potentially achieving the same protective effect that tesamorelin growth hormone elevation provides.
The protein supplementation strategies for GLP-1 users and dietary optimization during peptide protocols offer practical guidance for maximizing muscle retention without pharmacological intervention.
Other peptide stacking alternatives
Researchers interested in multi-compound approaches might consider alternatives with more established safety data. CJC-1295 (another GHRH analog) combined with ipamorelin provides growth hormone elevation with extensive community experience, though less clinical data than tesamorelin. The ipamorelin and CJC-1295 comparison and CJC-1295 dosage calculator provide starting points for these alternative GH-elevating approaches.
AOD-9604, a modified fragment of growth hormone, offers fat-loss effects without the full growth hormone elevation that creates insulin resistance concerns. Researchers studying AOD-9604 dosing protocols and AOD-9604 combined with incretin agonists may find a safer combination pathway that addresses similar goals with less unknown risk.
For those exploring cagrilintide combined with retatrutide, the amylin analog pathway offers another approach to appetite modulation and weight loss enhancement with different risk profiles.
What the current research actually says
Honesty matters more than enthusiasm when evaluating an untested combination. Here is exactly what the research supports and where it falls silent.
What we know with confidence
Tesamorelin reduces visceral fat in HIV-positive patients with lipodystrophy. Multiple clinical trials confirm this with consistent results. The compound elevates GH and IGF-1 within physiological ranges when administered at 2mg daily. Side effects are manageable and well-characterized. This data comes from randomized, placebo-controlled trials published in peer-reviewed journals.
Retatrutide produces significant weight loss in individuals with obesity and type 2 diabetes. Phase 2 trial data shows up to 24.2% body weight reduction at the 12mg dose over 48 weeks. The compound dramatically reduces liver fat, improves insulin sensitivity, and enhances metabolic markers across multiple domains. This data comes from randomized trials published in the New England Journal of Medicine and Nature Medicine.
Both compounds have individual safety profiles that are well-documented within their respective trial populations. Neither compound showed unexpected serious adverse events in isolation during clinical trials lasting up to 48 weeks.
What we do not know
No clinical trial has tested tesamorelin and retatrutide together. Zero. Not in animal models. Not in human subjects. The combination exists only in theoretical pharmacological analysis and, likely, in unsupervised self-experimentation that produces no publishable data.
We do not know whether the insulin-sensitizing effects of GLP-1/GIP agonism adequately counterbalance the insulin-desensitizing effects of GH elevation when both occur simultaneously. We do not know whether IGF-1 clearance changes in the presence of triple-receptor activation. We do not know whether liver fat reduction from retatrutide alters the hepatic response to tesamorelin-stimulated GH. We do not know the long-term safety of combining these pathways.
This level of uncertainty places the combination firmly in the experimental category. Researchers proceeding despite these unknowns must accept that they are operating without the safety data that clinical trials are designed to generate. Understanding the difference between research and pharmaceutical-grade compounds becomes critical in this context.
What the regulatory landscape looks like
Tesamorelin is FDA-approved but only for HIV-associated lipodystrophy. Any other use is off-label. Retatrutide is not approved anywhere in the world for any indication. It remains an investigational compound available only through clinical trial participation. Accessing retatrutide outside of clinical trials typically involves research chemical suppliers whose products lack the quality assurance of pharmaceutical manufacturing.
The current availability of retatrutide remains limited, and researchers should understand the sourcing considerations outlined in the retatrutide purchasing guide. Quality verification becomes even more important when combining compounds, since impurities or inaccurate dosing in one product amplify risks when another compound is added to the protocol.
For those comparing their options, understanding retatrutide pricing and sourcing reliability helps inform practical decisions about whether a combination protocol is even feasible.
Practical timeline expectations
For researchers who decide to proceed with a combined protocol despite the unknown risks, realistic timeline expectations help set appropriate milestones for evaluation.
Weeks 1 through 4: establishment phase
Most theoretical frameworks suggest starting tesamorelin first, alone, for 2 to 4 weeks. This establishes baseline GH and IGF-1 response, identifies any tesamorelin-specific side effects, and provides clean pre-combination bloodwork. During this phase, administer tesamorelin at 1mg daily (half the standard dose) to assess tolerance.
Baseline bloodwork should be completed before the first injection. Repeat at week 2 to capture initial IGF-1 and metabolic response. If markers remain stable, proceed to introducing retatrutide in week 3 or 4.
Weeks 4 through 8: introduction phase
Begin retatrutide at the lowest available dose (0.5 to 1mg weekly) while continuing tesamorelin. This phase focuses on monitoring the interaction between compounds. Bloodwork every 2 weeks tracks glucose, insulin, and IGF-1. GI symptoms from retatrutide introduction are expected and should be managed with dietary adjustments, smaller meals, and adequate hydration.
Increase tesamorelin to 2mg daily during this phase if the initial 1mg dose was well-tolerated. Do not increase both compounds simultaneously. Change one variable at a time to isolate which compound is responsible for any changes in symptoms or blood markers.
Weeks 8 through 16: titration phase
Gradually increase retatrutide dose following standard titration (2mg, then 4mg, then 8mg at 4-week intervals). Continue tesamorelin at 2mg daily. Monthly bloodwork tracks all key markers. Body composition assessment at week 12 provides the first objective measure of whether the combination is producing the hypothesized benefits.
This is the critical evaluation window. If fasting glucose is trending upward, if IGF-1 exceeds normal ranges, or if side effects are becoming unmanageable, this is the point to reassess the protocol. Many researchers find that one compound alone, paired with proper nutrition and exercise, achieves 80% of the theoretical benefit with substantially less risk.
Weeks 16 through 24 and beyond: maintenance phase
If the combination has been well-tolerated through the titration phase, maintenance involves continuing both compounds at their established doses while reducing bloodwork frequency to monthly. DEXA scan at 24 weeks compared to baseline quantifies the body composition changes.
Long-term use beyond 6 months enters truly uncharted territory. Even the individual compound data becomes thinner at longer timeframes. Quarterly comprehensive bloodwork becomes the minimum monitoring standard for ongoing combined use.
Comparing this combination to other peptide stacks
Context helps researchers evaluate whether the tesamorelin-retatrutide combination offers advantages over better-established stacking approaches.
Tesamorelin plus semaglutide
This combination has more community experience than the retatrutide version, though still lacks clinical trial data. Semaglutide activates only the GLP-1 receptor compared to retatrutide triple-agonist approach, producing less weight loss but with a longer safety track record. For researchers who want the GH-plus-incretin combination with less pharmacological novelty, semaglutide may be the more conservative partner compound.
Understanding semaglutide onset timelines and discontinuation considerations helps researchers who might transition between single and combined protocols.
Tesamorelin plus tirzepatide
Tirzepatide dual-agonist approach (GLP-1 plus GIP) sits between semaglutide and retatrutide in both efficacy and novelty. It has FDA approval for type 2 diabetes and obesity, providing a stronger regulatory basis than retatrutide. Some researchers prefer the tirzepatide-tesamorelin combination as a middle ground: more potent than semaglutide-tesamorelin but less experimental than retatrutide-tesamorelin.
The three-way comparison between these incretin compounds helps researchers understand where each sits on the efficacy-safety spectrum.
Retatrutide plus BPC-157 or TB-500
For researchers concerned about tissue integrity during rapid weight loss, healing peptides like BPC-157 and TB-500 offer a different complementary approach than tesamorelin. Rather than elevating growth hormone systemically, these peptides provide localized tissue support. The BPC-157 versus TB-500 comparison and injury recovery peptide guide explore these options in detail.
Retatrutide plus cagrilintide
Cagrilintide, an amylin analog, combined with GLP-1 agonists produces weight loss comparable to tirzepatide. Adding cagrilintide to retatrutide creates a four-receptor approach (GLP-1, GIP, glucagon, and amylin). The cagrilintide-retatrutide combination guide explores this alternative stacking strategy. The cagrilintide safety profile and weight loss data inform whether this approach might suit researchers better than the tesamorelin combination.
Cost and practical considerations
Beyond the science, practical factors influence whether this combination makes sense for individual researchers.
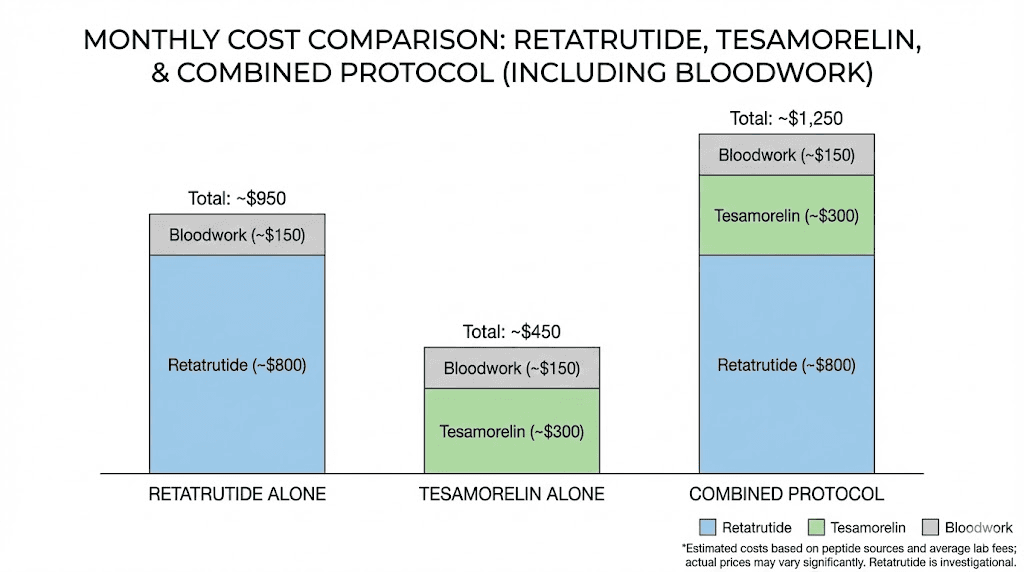
Financial commitment
Running both compounds simultaneously involves significant ongoing expense. Tesamorelin at 2mg daily requires approximately 60mg per month. Retatrutide at maintenance doses of 8 to 12mg weekly requires 32 to 48mg per month. Both compounds come from specialized suppliers at research-grade pricing that varies considerably by source and quality.
The peptide cost calculator can help estimate monthly expenses based on current pricing. Add the cost of regular bloodwork (comprehensive panels every 2 to 4 weeks initially), body composition scans, and any medical consultation, and the total monthly investment becomes substantial.
For researchers evaluating cost-effectiveness, retatrutide alone at an appropriate dose may deliver 80 to 90% of the fat loss benefit at roughly half the compound cost and a fraction of the monitoring expense. The retatrutide cost analysis provides current pricing context.
Daily logistics
The combined protocol involves daily tesamorelin injections (morning, fasting) plus weekly retatrutide injections (any time, rotated sites). Both compounds require refrigerated storage after reconstitution. Both have finite shelf lives once reconstituted, typically 4 to 6 weeks refrigerated.
Researchers managing multiple reconstituted vials need organized storage and clear labeling. Cross-contamination between vials, while unlikely with proper technique, represents a risk that increases with protocol complexity. Following proper reconstitution procedures and using the reconstitution calculator minimizes preparation errors.
Travel creates additional challenges. Maintaining cold chain for two separate compounds, carrying injection supplies, and managing daily versus weekly injection schedules while traveling requires planning. Researchers who travel frequently may find the peptide travel guide helpful for logistics, though it was written for single-compound protocols.
Sourcing quality concerns
With retatrutide not yet FDA-approved, sourcing comes exclusively from research chemical suppliers. Quality varies enormously. Underdosed products, contaminated batches, and incorrectly labeled compounds all exist in the research peptide market. When combining two compounds, the risk of quality issues multiplies because each product introduces its own sourcing risk.
Researchers should consider established supplier reviews and, when possible, verify product identity and purity through third-party testing. The bulk purchasing guide discusses quality indicators that help distinguish reliable sources from unreliable ones.
For those navigating the grey market peptide landscape, understanding what separates quality research compounds from questionable products becomes even more important in a multi-compound protocol where each additional variable increases overall risk.
SeekPeptides provides comprehensive guidance on peptide sourcing, quality evaluation, and protocol development. Members access detailed supplier analyses, community-verified sourcing recommendations, and tools that help researchers navigate the complex research peptide landscape with more confidence.
Who should and should not consider this combination
Not every researcher exploring peptide-based body composition strategies needs a dual-compound approach. The combination of tesamorelin and retatrutide, despite its theoretical appeal, suits a narrow profile of researchers with specific goals and risk tolerance.
The combination might make sense for
Researchers with significant visceral adiposity who have already optimized single-compound approaches and found them insufficient for visceral fat specifically. Individuals with metabolic syndrome features that span multiple domains (visceral obesity, insulin resistance, dyslipidemia) who want comprehensive metabolic intervention. Those with access to regular medical monitoring, including frequent bloodwork and body composition assessment. Researchers with prior experience running individual peptide protocols who understand their personal response patterns.
Prior experience with weight loss peptides and muscle growth peptides separately provides the baseline understanding needed to evaluate combination effects. First-time peptide users should not begin with a dual-compound protocol.
The combination is not appropriate for
Individuals with diabetes or prediabetes (the insulin resistance interaction risk is too poorly understood). Anyone with a history of or current malignancy (IGF-1 elevation concern). Researchers without access to regular bloodwork and medical oversight. First-time peptide users who have not established tolerance to either compound individually. Anyone seeking a shortcut who has not optimized diet, exercise, and sleep first.
Those with pre-existing GI conditions, including gastroparesis, inflammatory bowel disease, or chronic pancreatitis, should avoid retatrutide entirely. Adding tesamorelin to the picture does not mitigate the GI risks that GLP-1 receptor activation creates.
For researchers in these categories, exploring single-compound fat loss approaches or the best peptides for metabolic support provides safer starting points.
Frequently asked questions
Is it safe to combine tesamorelin and retatrutide?
No clinical trial has ever tested this combination, so safety cannot be confirmed. Theoretical analysis suggests complementary mechanisms with potential insulin resistance concerns. Any combined use should involve comprehensive medical monitoring, including regular bloodwork for IGF-1, glucose, insulin, and liver enzymes. Researchers exploring peptide stacking approaches should understand that untested combinations carry inherently higher risk than established protocols.
What makes tesamorelin and retatrutide theoretically complementary?
Tesamorelin works through growth hormone pathways to target visceral fat specifically, while retatrutide activates GLP-1, GIP, and glucagon receptors to reduce overall body weight and liver fat. These mechanisms do not overlap or compete. Tesamorelin also supports muscle preservation through GH and IGF-1 elevation, potentially counteracting the lean mass loss associated with aggressive weight loss peptide protocols.
How should I dose tesamorelin if combining it with retatrutide?
Theoretical frameworks suggest starting tesamorelin at 1mg daily for 2 to 4 weeks before adding retatrutide, then escalating to the standard 2mg daily once tolerance is established. The peptide dosage calculator can help determine volumes for injection. Never adjust both compounds simultaneously, as this makes it impossible to identify which compound is responsible for any changes in symptoms or blood markers.
Can tesamorelin counteract the muscle loss from retatrutide?
Theoretically yes. Tesamorelin elevates growth hormone and IGF-1, both of which support protein synthesis and lean tissue maintenance. Clinical data shows tesamorelin increases lean body mass even during visceral fat reduction. Whether this effect persists when combined with the substantial caloric deficit that retatrutide creates has not been studied. Resistance training and adequate protein intake remain the evidence-based approaches to muscle preservation during peptide-based fat loss.
What bloodwork should I monitor during combined use?
At minimum: IGF-1 every 4 weeks, fasting glucose and insulin every 2 weeks initially (monthly once stable), HbA1c every 6 weeks, liver enzymes monthly, and comprehensive metabolic panel quarterly. DEXA body composition scans at baseline, 12 weeks, and 24 weeks provide objective tissue composition data. Any researcher using this combination without regular monitoring is accepting unknown risks without the data needed to identify problems early.
Is retatrutide alone sufficient without adding tesamorelin?
For most researchers, yes. Retatrutide at 12mg weekly produced 24.2% body weight loss in Phase 2 trials, which is the highest recorded for any single compound in obesity research. The retatrutide weight loss dosing guide details the titration schedule. Adding tesamorelin may provide incremental benefits for visceral fat and muscle preservation, but those benefits come with significant unknown risks.
How does this combination compare to retatrutide plus cagrilintide?
Both combinations target different aspects of the same problem. Tesamorelin adds growth hormone pathway activation for visceral fat and muscle preservation. Cagrilintide adds amylin receptor activation for additional appetite suppression and metabolic modulation. The cagrilintide-retatrutide combination stays within the incretin and satiety hormone family, while tesamorelin-retatrutide combines two entirely different hormonal systems.
What happens if I stop one compound but continue the other?
Discontinuing tesamorelin while continuing retatrutide would remove the growth hormone elevation. IGF-1 levels return to baseline within 2 weeks of stopping tesamorelin. Any visceral fat reduction benefits specific to the GH pathway would gradually diminish, but overall weight loss from retatrutide would continue. Stopping retatrutide while continuing tesamorelin removes the appetite suppression and metabolic rate enhancement, potentially leading to weight regain while visceral fat reduction continues. The GLP-1 agonist discontinuation experience provides context for what retatrutide cessation might involve.
Where can I find reliable retatrutide for research?
Retatrutide is not FDA-approved and is only available through research chemical suppliers. Quality varies significantly between sources. Researchers should review the complete retatrutide sourcing guide and consider third-party testing to verify product identity and purity. SeekPeptides members access supplier reviews, community-verified sourcing recommendations, and quality assessment tools that help navigate this complex landscape.
Can I use oral retatrutide with tesamorelin instead of injectable?
Oral formulations of triple-agonist peptides are in very early development and are not commercially available in any reliable form. The clinical trial data that supports retatrutide efficacy was generated using subcutaneous injection. Any oral product claiming to be retatrutide should be viewed with extreme skepticism. For background on oral versus injectable peptide delivery, the injectable versus oral peptide comparison provides useful context.
External resources
Efficacy and safety of retatrutide: systematic review and meta-analysis (PMC)
Triple hormone receptor agonist retatrutide for obesity: Phase 2 trial (NEJM)
Tesamorelin clinical pharmacology and safety (NCBI LiverTox)
Safety and metabolic effects of tesamorelin in type 2 diabetes (PMC)
Tesamorelin prescribing information and side effects (Mayo Clinic)
Retatrutide for metabolic dysfunction-associated steatotic liver disease (Nature Medicine)
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Members access detailed stacking guides, dosing calculators, and expert-reviewed protocol frameworks that take the guesswork out of complex research decisions.
In case I do not see you, good afternoon, good evening, and good night. May your growth hormone pulses stay physiological, your metabolic markers stay favorable, and your research protocols stay methodical.