Feb 18, 2026

You are throwing money away. Right now, sitting in your refrigerator or medicine cabinet, there is a vial or pen of semaglutide that may have already lost a significant portion of its potency. Or worse, you may have discarded a perfectly good dose because you were not sure whether it was still safe to use. This is not a small problem. A single Ozempic pen can cost hundreds of dollars without insurance, and compounded semaglutide vials represent a real investment in your health goals. Every dose that degrades unnecessarily is money wasted, progress delayed, and frustration compounded.
The question of how long semaglutide stays good is not as straightforward as checking a single expiration date. It depends on the formulation you have, whether the container has been opened, how it has been stored, and what temperature conditions it has been exposed to throughout its life. Brand-name pens follow different rules than compounded semaglutide vials. Lyophilized powder follows different rules than pre-mixed solutions. And the guidelines your pharmacy prints on the label may be more conservative, or more generous, than the actual science supports.
Getting this wrong has real consequences. Use degraded semaglutide and you will not get the appetite suppression or blood sugar control you expect. Throw away good semaglutide and you waste money you did not need to spend. This guide covers every formulation, every storage scenario, and every sign of degradation so you never have to guess again. Whether you are using an Ozempic pen, a Wegovy pen, a compounded vial, oral semaglutide drops, or Rybelsus tablets, you will find the exact shelf life timelines and storage rules you need right here.
Quick reference shelf life chart
Before diving into the details, here is a quick reference chart covering every major semaglutide formulation. Bookmark this section. You will come back to it.
Formulation | Unopened shelf life | Storage (unopened) | After opening/first use | Storage (opened) |
|---|---|---|---|---|
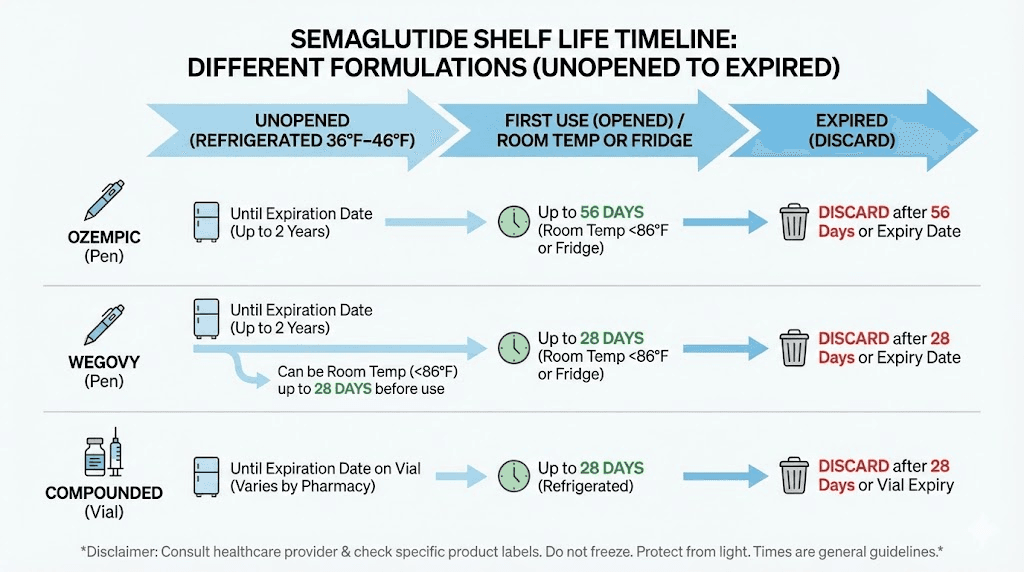
Ozempic pen | Until expiration date (24-36 months) | Refrigerated 36-46F (2-8C) | 56 days | Refrigerated or room temp 59-86F |
Wegovy pen | Until expiration date (24-36 months) | Refrigerated 36-46F (2-8C) | 28 days | Room temp 59-86F |
Compounded vial (pre-mixed) | Per pharmacy BUD (28-120 days) | Refrigerated 36-46F (2-8C) | 28-90 days (pharmacy dependent) | Refrigerated 36-46F (2-8C) |
Lyophilized powder | 24-36 months | Below -18C (desiccated) | 2-7 days after reconstitution | Refrigerated 2-8C |
Rybelsus tablets | Until expiration date (24 months) | Room temp 68-77F (20-25C) | N/A (single dose tablets) | Keep in original packaging |
Reconstituted with BAC water | N/A | N/A | 28-30 days | Refrigerated 36-46F (2-8C) |
This table gives you the overview. But the details matter enormously, and the difference between proper and improper storage can mean the difference between full potency and a useless solution. Keep reading for the complete breakdown of each formulation, including the nuances that most guides miss entirely. If you are also managing tirzepatide dosing alongside semaglutide, understanding the shelf life differences between these two GLP-1 medications prevents costly storage mistakes.
If you need help calculating your semaglutide dosage before your next injection, tools like dosage calculators can help ensure you are drawing the correct amount from whatever formulation you are using. Accurate dosing paired with proper storage is the foundation of consistent results.
Unopened semaglutide shelf life by formulation
Unopened semaglutide has the longest shelf life of any stage. The sealed container protects the peptide from contamination, light exposure, and the chemical reactions that accelerate once air enters the equation. But "unopened" does not mean "indestructible." Storage conditions still matter. A lot.
Ozempic pens (unopened)
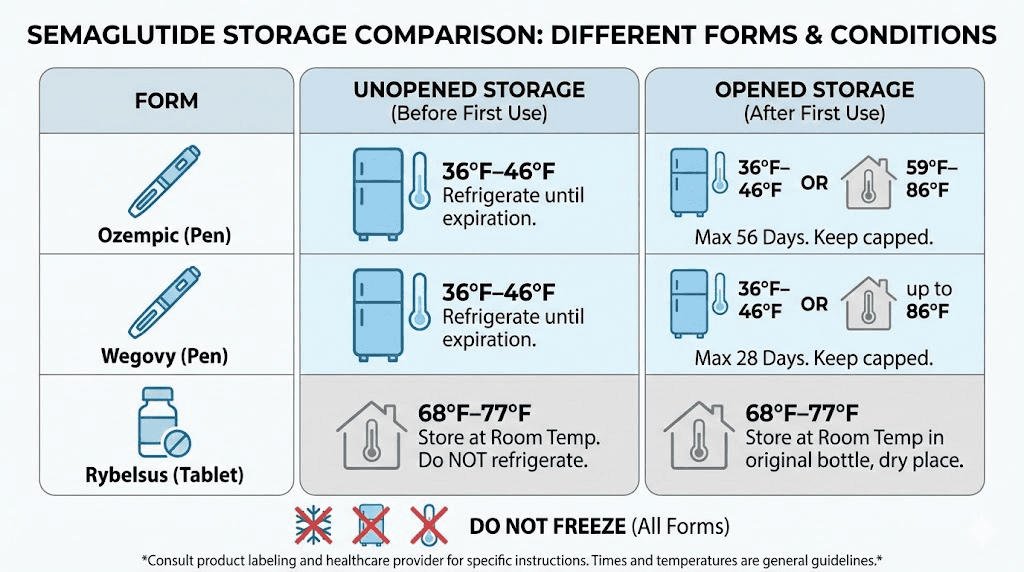
Ozempic pens contain pre-mixed semaglutide solution in a multi-dose delivery device. When stored properly in a refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius), an unopened Ozempic pen remains good until the manufacturer expiration date printed on the packaging. That date is typically 24 to 36 months from the date of manufacture.
Do not freeze Ozempic. Freezing destroys the peptide structure and renders the medication useless, even if it thaws and looks normal afterward. The solution may appear fine visually, but the molecular damage from ice crystal formation cannot be reversed. If your Ozempic has been frozen, even briefly, discard it. This is a consistent rule across all injectable peptide formulations.
There is an important distinction here that many people overlook. The expiration date assumes continuous refrigeration. If your unopened Ozempic pen sat on a counter at room temperature for a few hours during transit or while you were organizing your refrigerator, that is generally fine. But extended room temperature exposure before the first use will reduce the overall shelf life, even though Novo Nordisk does not provide specific guidance on cumulative room temperature exposure for unopened pens. The conservative approach is to keep unopened pens refrigerated at all times until you are ready to use them.
For those comparing injectable formulations, understanding the differences between injectable and oral peptides can help you make informed decisions about which delivery method works best for your situation.
Wegovy pens (unopened)
Wegovy works the same way as Ozempic, both contain semaglutide, but the dosing and delivery differ. Unopened Wegovy pens should be stored refrigerated at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius) and are good until the expiration date on the packaging, typically 24 to 36 months from manufacture.
The same freezing warnings apply. Never freeze Wegovy. And like Ozempic, brief temperature excursions during handling are acceptable, but prolonged room temperature storage before opening will compromise shelf life. General peptide storage principles apply consistently across all injectable GLP-1 formulations.
One thing worth noting about Wegovy specifically: the pen comes in single-dose presentations at various strengths (0.25 mg, 0.5 mg, 1 mg, 1.7 mg, and 2.4 mg). Because each pen is designed for a single use, the concept of "after opening" means something slightly different than with multi-dose Ozempic pens. We will cover that distinction in the next section. If you are curious about how semaglutide compares to tirzepatide in terms of both side effects and storage requirements, the differences extend beyond just the active ingredient.
Compounded semaglutide vials (unopened)
This is where things get more complicated.
Compounded semaglutide does not come with a standard manufacturer expiration date the way brand-name products do. Instead, compounding pharmacies assign a beyond-use date (BUD) based on their specific formulation, stability testing, and the standards set by USP Chapter 797. The shelf life of an unopened compounded semaglutide vial in the fridge varies dramatically depending on the pharmacy that produced it.
Conservative pharmacies assign a 28-day BUD. This is the default when stability testing has not been performed or when the compounding environment does not support longer dating. Other pharmacies, particularly 503B outsourcing facilities that have conducted rigorous stability testing under controlled conditions, may assign BUDs of 60, 90, or even 120 days.
The key factor is whether the pharmacy has performed stability-indicating testing on their specific formulation. Without that testing, you should assume the shorter timeframe. The vial should be refrigerated at 36 to 46 degrees Fahrenheit at all times, and most pharmacies will clearly state "refrigerate" on the label. If you are using compounded semaglutide and the label does not include a beyond-use date, contact the pharmacy immediately. Never guess. You can explore compounded semaglutide dose charts to understand the concentrations typically used across different pharmacies.
Lyophilized semaglutide powder (unopened)
Lyophilized (freeze-dried) semaglutide powder represents the most stable form of the peptide. When stored desiccated (dry) at temperatures below minus 18 degrees Celsius (roughly 0 degrees Fahrenheit), lyophilized semaglutide can maintain potency for 24 to 36 months. Some research suggests even longer when stored at minus 80 degrees Celsius, though that requires specialized laboratory freezer equipment.
The stability of lyophilized peptides comes from removing water from the equation. Most degradation pathways for peptides involve hydrolysis, which requires water molecules. Remove the water, and you dramatically slow the chemical breakdown. This is why lyophilized peptides generally outlast liquid formulations by a significant margin.
If you are working with lyophilized semaglutide, proper storage is critical. It needs to remain sealed, desiccated, and frozen. Exposure to moisture, even the humidity in ambient air, begins the degradation process before you ever add bacteriostatic water. Keep the vial sealed until you are ready to reconstitute. Understanding how long peptides last in powder form gives you a broader perspective on stability across different peptide types.
Rybelsus tablets (unopened)
Rybelsus is oral semaglutide in tablet form, and its storage requirements differ completely from injectable formulations. Tablets should be stored at controlled room temperature, 68 to 77 degrees Fahrenheit (20 to 25 degrees Celsius), with brief excursions permitted between 59 and 86 degrees Fahrenheit.
Here is the critical detail most people miss with Rybelsus: the tablets are sensitive to light and moisture. They must remain in the original blister packaging until the moment of use. Do not transfer them to a pill organizer. Do not remove them from the foil backing the night before. The protective packaging is not optional, it is part of the stability design.
Unopened Rybelsus in its original packaging has a shelf life of approximately 24 months from manufacture. The expiration date on the package reflects this timeline. Unlike injectable semaglutide, Rybelsus does not need refrigeration, and in fact, refrigeration could introduce moisture condensation that damages the tablets.
Opened and in-use shelf life by formulation
Once you open a semaglutide product, the clock starts ticking faster. The protective seal has been broken. Air, bacteria, light, and temperature fluctuations now have direct access to the peptide. Every formulation handles this differently, and the timelines matter. Missing them means you are either wasting good medication or, worse, injecting degraded solution.
Ozempic pen after first use
After the first injection from an Ozempic pen, the pen remains good for 56 days. That is eight weeks. This timeline applies whether you keep the opened pen refrigerated or store it at room temperature between 59 and 86 degrees Fahrenheit (15 to 30 degrees Celsius).
Fifty-six days is generous compared to most injectable peptide products, and it reflects the preservative system built into the Ozempic formulation. The pen contains antimicrobial preservatives that prevent bacterial growth during the multi-dose use period. However, there are conditions. Always replace the pen cap after each use. Never leave the needle attached between injections, as this can allow air bubbles and contaminants to enter. And never share your pen with another person, even if the needle is changed.
A common question people have is whether it matters if they keep the opened pen in the fridge or at room temperature. The answer, per the manufacturer guidance, is that either is acceptable within that 56-day window. Some people prefer refrigeration because the injection feels smoother with a cold solution. Others prefer room temperature because cold injections can sting. Both approaches maintain potency during that 56-day use period, so it comes down to personal preference and comfort during injection. For guidance on timing your semaglutide injections, the time of day you choose can influence both comfort and effectiveness.
After 56 days from first use, discard the Ozempic pen. Even if solution remains. Even if it looks perfectly clear. The 56-day limit exists for safety, not because the medication instantly becomes dangerous on day 57, but because potency and sterility can no longer be guaranteed beyond that point.
Wegovy pen after first use
Wegovy pens have a shorter in-use window than Ozempic. After removing the pen cap for the first time, Wegovy is good for 28 days when stored at room temperature between 59 and 86 degrees Fahrenheit. Each Wegovy pen is designed for a single dose, so the "after first use" scenario primarily applies to situations where the full dose was not delivered or where the pen was uncapped but not immediately used.
Why is Wegovy shorter than Ozempic? The formulations differ slightly, and the single-dose design means the preservative system is optimized for a different use pattern. Novo Nordisk tested stability under specific conditions and determined that 28 days at room temperature is the validated timeframe.
If you are transitioning between different semaglutide products, or considering a switch to tirzepatide, understanding these shelf life differences helps with planning and minimizing waste. The semaglutide to tirzepatide conversion chart can help if you are making that transition.
Compounded semaglutide vial after first use
Once you puncture the rubber stopper on a compounded semaglutide vial with a needle, the beyond-use date becomes the governing timeline. Most compounding pharmacies specify that punctured vials should be used within 28 days when kept refrigerated. Some pharmacies with more robust stability testing allow 60 to 90 days after first puncture.
There are several factors that affect how long your compounded vial remains viable after opening:
Preservative content: Vials containing bacteriostatic water (with benzyl alcohol preservative) have longer in-use dates than those mixed with sterile water
Number of punctures: Each needle insertion through the stopper creates a microscopic pathway for contamination, so minimizing punctures extends usable life
Technique: Swabbing the stopper with alcohol before each draw and using a new sterile needle each time reduces contamination risk significantly
Storage consistency: Keeping the vial refrigerated without temperature fluctuations helps maintain stability throughout the use period
If your compounded semaglutide comes from a 503B outsourcing facility, the labeling should include a clear beyond-use date. If you obtained it from a 503A compounding pharmacy, the BUD may be shorter due to different regulatory requirements. Either way, refrigeration is essential for compounded semaglutide after first use. No exceptions.
Understanding proper semaglutide reconstitution is just as important as storage. If the vial was reconstituted incorrectly from the start, storage will not save it.
Rybelsus tablets after opening the packaging
Rybelsus tablets are individually sealed in blister packs within a bottle. Once you open the bottle, the tablets inside their individual blister packs remain protected. The shelf life does not change from the original expiration date as long as each tablet stays in its sealed blister until use.
However, once you pop a tablet out of its blister pack, it should be taken immediately. The tablet absorbs moisture rapidly once exposed to air, and the absorption enhancer (SNAC) that enables oral bioavailability is sensitive to environmental conditions. Do not remove a tablet and save it for later. Do not handle tablets with wet hands. Take it out, swallow it with a small amount of plain water, and wait 30 minutes before eating or drinking anything else.
Reconstituted semaglutide shelf life
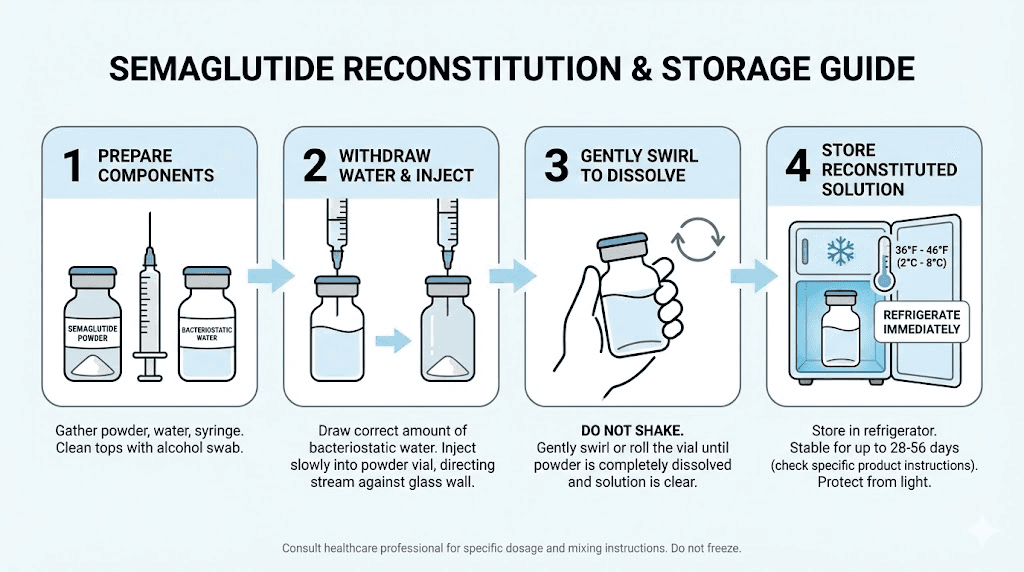
Reconstitution is the process of adding a liquid (usually bacteriostatic water) to lyophilized semaglutide powder to create an injectable solution. This is common with compounded semaglutide and research-grade peptides. The shelf life of reconstituted semaglutide is significantly shorter than the powder form, and understanding the specifics can save you from wasting expensive product.
Reconstituted with bacteriostatic water
When semaglutide powder is reconstituted with bacteriostatic water (water containing 0.9% benzyl alcohol as a preservative), the resulting solution is generally good for 28 to 30 days when stored refrigerated at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). The benzyl alcohol provides antimicrobial protection that prevents bacterial growth during this period.
This 28 to 30 day window is the standard recommendation, and it aligns with general guidance for reconstituted peptides stored with bacteriostatic water. Some researchers report maintaining potency beyond this window, but without formal stability testing on your specific formulation, the conservative 28-day rule is the safest approach.
The peptide reconstitution calculator can help you determine the correct amount of bacteriostatic water to add based on your vial size and desired concentration. Getting the ratio right from the start affects both dosing accuracy and solution stability. For specific semaglutide reconstitution guidance, the semaglutide reconstitution chart provides detailed mixing ratios.
Reconstituted with sterile water (no preservative)
If semaglutide is reconstituted with plain sterile water (no preservative), the shelf life drops dramatically. Without antimicrobial protection, the solution should be used within 24 to 48 hours when refrigerated. Some guidelines suggest up to 72 hours, but this carries increased risk.
This is why bacteriostatic water is strongly preferred for multi-dose vials. If you are only taking one or two doses from a vial, sterile water may be acceptable. But for vials intended to last weeks, bacteriostatic water is not just preferred, it is necessary. You can find detailed mixing instructions in the 5 mg semaglutide mixing guide and the 10 mg semaglutide mixing guide.
Research-grade lyophilized semaglutide after reconstitution
Research-grade semaglutide that has been reconstituted follows shorter timelines than pharmaceutical-grade products. Once reconstituted and stored at 4 degrees Celsius (standard laboratory refrigerator temperature), research-grade semaglutide solution typically maintains potency for 2 to 7 days.
For longer storage needs, researchers often prepare aliquots, dividing the reconstituted solution into smaller single-use portions and freezing them at minus 20 degrees Celsius or minus 80 degrees Celsius. Frozen aliquots can maintain stability for several months. The key is to avoid repeated freeze-thaw cycles, as each cycle causes peptide aggregation and degradation. Thaw an aliquot once, use it, and discard any remainder.
This approach is also relevant for people working with 5 mg semaglutide vials who want to maximize the usable life of their reconstituted product. The broader principles of storing peptides after reconstitution apply across all peptide types, not just semaglutide.
Storage requirements that determine shelf life
Storage is not a minor detail. It is the single most important factor determining whether your semaglutide maintains full potency or degrades into an expensive, useless solution. Temperature, light, agitation, and contamination control all play roles, and getting any one of them wrong can shorten shelf life dramatically.
Temperature is everything
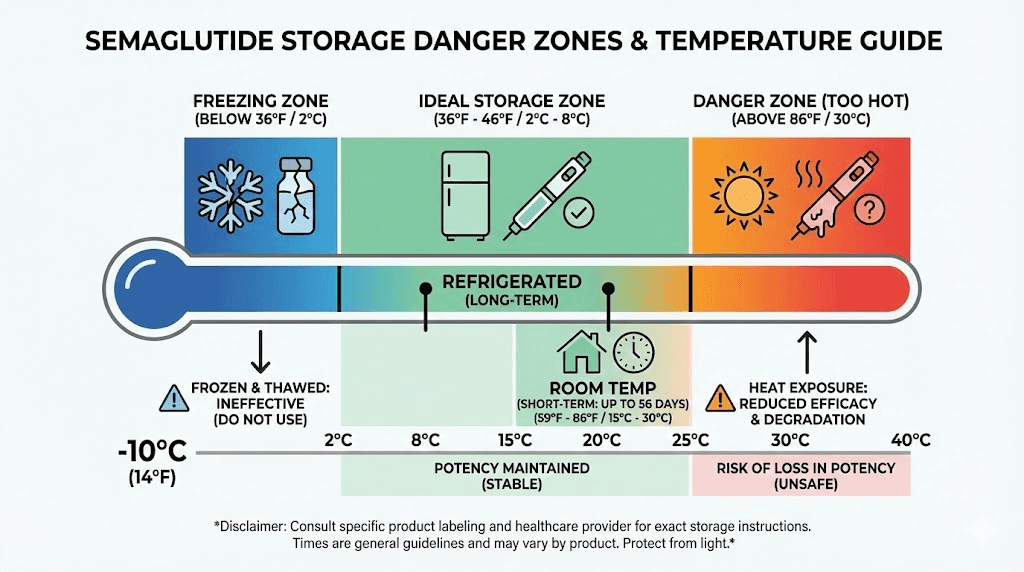
Temperature controls the rate of chemical degradation. For every 10 degrees Celsius increase in storage temperature, the rate of peptide degradation roughly doubles. This is not a vague guideline. It is basic chemistry (the Arrhenius equation), and it applies to semaglutide as it does to every other peptide.
The ideal storage temperature for most semaglutide formulations is 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius), which is standard refrigerator temperature. Your household refrigerator typically operates in this range, but placement within the refrigerator matters. Do not store semaglutide in the door, where temperatures fluctuate with every opening. Do not store it against the back wall where it might freeze. The middle shelf toward the front is usually the most temperature-stable location.
Investing in a small refrigerator thermometer costs a few dollars and provides peace of mind that your storage conditions are within range. This simple step can help you determine whether your semaglutide is maintaining the stability it should. Those managing multiple peptide products benefit from reading the complete peptide storage guide for a comprehensive overview of proper handling practices.
Light exposure degrades semaglutide
Semaglutide, like many peptides, is sensitive to light. Ultraviolet light and even prolonged exposure to fluorescent lighting can accelerate the oxidation of amino acid residues in the peptide chain, particularly tryptophan and methionine. This oxidation reduces biological activity.
Brand-name products account for this with opaque or amber-colored packaging. Ozempic and Wegovy pens come in protective cases that limit light exposure. But compounded semaglutide often arrives in clear glass vials. If your vial is clear, store it in the original box or wrap it in aluminum foil. Never leave a clear vial sitting on a countertop or windowsill.
Rybelsus tablets are particularly light-sensitive, which is one reason they must remain in the original blister packaging until use. The foil backing provides a light barrier that protects the tablet formulation.
Handling and contamination control
Every time you insert a needle through a vial stopper, you introduce a small risk of contamination. Proper technique minimizes this risk, but the cumulative effect of multiple punctures over weeks adds up. This is one reason compounded vials have finite beyond-use dates even when refrigerated continuously.
Best practices for handling:
Wash hands thoroughly before handling any semaglutide product
Swab vial stoppers with 70% isopropyl alcohol before each needle insertion
Use a new sterile needle for every draw
Never touch the needle tip or allow it to contact any non-sterile surface
Replace Ozempic pen caps immediately after use
Store vials upright to minimize stopper contact with the solution
These steps might seem excessive. They are not. Contamination is the fastest way to render your semaglutide unsafe, and contaminated solutions do not always look different from clean ones. Understanding proper peptide injection technique is just as important as proper storage. The GLP-1 injection guide covers technique specifics that minimize both waste and contamination risk. If you are using an injection pen, the handling protocols differ slightly from vial-and-syringe methods.
Never freeze liquid semaglutide
This bears repeating because it is the single most common mistake that destroys semaglutide. Freezing liquid (pre-mixed) semaglutide causes ice crystals to form within the solution. These ice crystals physically disrupt the three-dimensional structure of the peptide through a process called cryoconcentration, where the peptide becomes concentrated in unfrozen pockets and denatures.
The damage from freezing is invisible. The solution will look normal after thawing. It will draw into a syringe the same way. But the molecular structure has been compromised, and biological activity is reduced or eliminated entirely. There is no way to reverse this damage.
If your refrigerator runs cold or if you suspect your semaglutide may have frozen, look for subtle signs: ice crystals on the outside of the vial when removed, a slightly different clarity than before, or (in worst cases) visible particulate matter that was not there previously. When in doubt, err on the side of caution and replace it. The broader question of peptide expiration applies here, because frozen-and-thawed peptides may technically not be "expired" by date, but they are functionally compromised.
Room temperature stability explained
Room temperature storage is sometimes necessary. Travel, power outages, forgotten doses on the counter, these situations happen. Knowing how long your semaglutide can safely remain at room temperature helps you make informed decisions about whether a dose is still safe to use.
Ozempic at room temperature
Ozempic is the most forgiving of the injectable formulations when it comes to room temperature exposure. After the first use, an Ozempic pen can be stored at room temperature (59 to 86 degrees Fahrenheit, or 15 to 30 degrees Celsius) for up to 56 days. This means the 56-day use period applies regardless of whether you keep it in the fridge or on the counter.
This is good news for people who travel frequently. You do not need a cooler pack for your Ozempic pen during a week-long vacation, assuming the ambient temperature stays below 86 degrees Fahrenheit. In hot climates or during summer months, however, temperatures can easily exceed this threshold, and at that point refrigeration or an insulated travel case becomes necessary.
An unopened Ozempic pen that has been at room temperature can still be used, but the total room temperature time should not exceed 56 days. If an unopened pen sat on a counter for 30 days, then was refrigerated, and then opened, you would have less than the full 56-day use period remaining. The manufacturer guidance focuses on cumulative room temperature exposure.
Wegovy at room temperature
Wegovy has a shorter room temperature tolerance. After the cap is removed, Wegovy can be stored at room temperature (59 to 86 degrees Fahrenheit) for up to 28 days. This is half the Ozempic timeframe, reflecting differences in the formulation and delivery system.
For people comparing Ozempic and Wegovy, this storage difference is worth considering. If you frequently forget to refrigerate your pen, or if you travel often, Ozempic provides a wider margin of error. Of course, the clinical dosing and indications differ between the two products, so storage convenience should not be the only factor in your decision.
Compounded semaglutide at room temperature
Compounded semaglutide should generally remain refrigerated at all times. Unlike brand-name pens that have been extensively tested at room temperature, most compounded formulations have not undergone similar stability testing outside of refrigerated conditions.
Brief room temperature exposure, such as the time it takes to draw a dose, is acceptable. But leaving a compounded vial on a counter for hours, or storing it at room temperature routinely, is not recommended. If you need to transport your compounded semaglutide, use an insulated bag with an ice pack, keeping the vial cool without allowing it to freeze. The travel with semaglutide guide covers practical solutions for maintaining cold chain during transit.
There is a reason room temperature stability for peptides is such a frequently searched topic. The reality is that most peptides, semaglutide included, degrade faster at higher temperatures. Refrigeration is your best defense against premature potency loss.
Rybelsus at room temperature
Rybelsus tablets are specifically designed for room temperature storage. The recommended storage is 68 to 77 degrees Fahrenheit (20 to 25 degrees Celsius), with brief temperature excursions between 59 and 86 degrees Fahrenheit being acceptable.
Unlike injectable semaglutide, you should not refrigerate Rybelsus. The concern with refrigeration is condensation. When a cold blister pack is brought to room temperature, moisture can form on and around the tablet, potentially compromising the SNAC absorption enhancer and reducing the already limited bioavailability of oral semaglutide. Keep it in a medicine cabinet or drawer at room temperature, away from direct sunlight and moisture.
Traveling with semaglutide
Traveling introduces unique challenges for semaglutide storage. Temperature control, security screening, altitude changes, and simply being out of your normal routine all create opportunities for mistakes. Planning ahead prevents waste.
Air travel considerations
The Transportation Security Administration (TSA) allows injectable medications through security checkpoints. You do not need to remove your semaglutide from your carry-on for screening, though declaring it can speed up the process. Keep medication in its original packaging with the pharmacy label visible.
Never pack semaglutide in checked luggage. Cargo holds are not temperature controlled, and temperatures can drop well below freezing at altitude. Your semaglutide will freeze and be destroyed. Always carry it on the plane in an insulated travel case.
For flights, an insulated pouch with a gel ice pack (not frozen solid, slightly thawed) keeps your semaglutide in the appropriate temperature range for several hours. Avoid placing the vial or pen in direct contact with the ice pack, as this can cause localized freezing. A small cloth or paper towel between the product and the ice pack provides adequate insulation.
Road trips and car travel
Cars present temperature extremes. In summer, a car interior can reach 150 degrees Fahrenheit or more. In winter, overnight temperatures can freeze anything left in the vehicle. Never leave semaglutide in a car for any extended period.
Use an insulated travel bag or cooler. Bring it inside with you at every stop. Treat it like a living thing that cannot survive extreme temperatures, because biochemically, that is exactly what it is.
If you are planning extended travel and want to understand the full picture, the complete travel with semaglutide guide covers everything from international regulations to hotel refrigerator considerations. For those managing multiple peptide products during travel, the peptide storage guide provides a broader framework.
International travel
Different countries have different regulations regarding importing medications, including peptides. Carry a copy of your prescription or a letter from your healthcare provider. Keep medication in original packaging with clear labeling. Some countries require specific documentation for importing injectable medications, so research your destination requirements well in advance.
Time zone changes can affect your injection schedule. If you normally inject on Friday mornings and fly across eight time zones, when is your next dose? The semaglutide timing guide can help you navigate scheduling adjustments without compromising your dosing consistency. Knowing where to inject GLP-1 medications during travel also helps maintain proper technique when you are away from your normal routine.
Signs your semaglutide has gone bad
Knowing what degraded semaglutide looks like is essential. Some signs are obvious. Others are subtle. And some forms of degradation produce no visible signs at all, which is why following storage guidelines and expiration dates matters even when the product looks fine.
Visual signs of degradation
Semaglutide solution should be clear and colorless. Any deviation from this is a red flag:
Cloudiness or haziness: Normal semaglutide solution is water-clear. Any cloudiness indicates protein aggregation or particulate contamination. Discard immediately.
Particles floating in solution: Hold the vial up to a light source and gently tilt it. If you see any particles, specks, or fibers floating in the solution, it is contaminated or degraded. Do not use it.
Color change: Semaglutide solution is colorless. Yellowing, browning, or any tint indicates chemical degradation, typically oxidation of amino acid residues. Once color change is visible, significant potency loss has already occurred. The GLP-1 and hair loss guide discusses how to distinguish between medication side effects and product quality issues.
Thickened or grainy texture: If the solution appears more viscous than normal or has a grainy consistency, aggregation has occurred. The peptide molecules have clumped together and lost their functional structure.
One important caveat: the absence of visual signs does not guarantee potency. A solution can look perfectly clear and still have lost significant biological activity through chemical degradation pathways that produce no visible changes. This is why date-based expiration matters, you cannot rely on visual inspection alone to determine if semaglutide is still effective.
Functional signs of degradation
Sometimes the first sign of degraded semaglutide is not something you see but something you feel, or more accurately, something you stop feeling.
Reduced appetite suppression is the most commonly reported functional sign. If you have been using semaglutide consistently and suddenly notice that your appetite control has weakened significantly, degraded medication is one possible explanation. Of course, tolerance, dietary changes, and other factors can also affect appetite suppression, so degraded medication is not always the cause. But if the timing coincides with a product that has been stored longer than recommended or exposed to temperature extremes, degradation should be high on the suspect list.
Similarly, reduced blood sugar control in people using semaglutide for diabetes management can indicate a potency issue. If blood glucose readings begin trending upward without changes in diet, activity, or other medications, check whether the semaglutide has exceeded its use-by date or been improperly stored. For those wondering about how quickly semaglutide suppresses appetite, understanding baseline expectations makes it easier to spot when something is off.
For anyone wondering whether appetite suppression should be immediate, the answer is nuanced. But a sudden loss of previously established appetite control is a clearer signal that something has changed, whether in dosing, storage, or the product itself.
The smell test
Semaglutide solution should have no noticeable odor. Bacteriostatic water has a faint preservative smell from the benzyl alcohol, but it should not be strong or unpleasant. A sour, musty, or otherwise off odor indicates bacterial contamination. Do not inject a solution that smells wrong. Trust your senses.
What happens if you use expired or degraded semaglutide
This is the question everyone wants answered. Is expired semaglutide dangerous? The answer is more nuanced than a simple yes or no.
Reduced potency is the primary risk
The main consequence of using expired or degraded semaglutide is reduced effectiveness. As the peptide breaks down, its ability to bind GLP-1 receptors and exert biological effects decreases. You might get 80% of the expected effect, or 50%, or 10%. There is no way to know without laboratory testing.
For people using semaglutide for weight management, reduced potency means slower progress, increased appetite, and frustration. For people using it for blood sugar control, reduced potency can have more serious implications, including blood glucose spikes that affect overall metabolic management. If you are not seeing results after several weeks, degraded medication is one factor worth investigating alongside dosing accuracy and lifestyle factors.
Contamination is the real danger
While the degradation products of semaglutide itself are generally not toxic, bacterial contamination of expired or improperly stored solutions is genuinely dangerous. Understanding GLP-1 related fatigue versus contamination symptoms helps you identify the source of any adverse reactions. Injecting a contaminated solution can cause:
Injection site infections (redness, swelling, warmth, pain)
Abscess formation requiring medical treatment
Systemic infection in severe cases
Endotoxin reactions (fever, chills, nausea)
This is why beyond-use dates exist. They reflect the period during which both potency and sterility can be reasonably assured. Using a product beyond its BUD is not just a potency gamble, it is a safety gamble. For a deeper analysis of whether using expired semaglutide is worth the risk, the article on using expired semaglutide covers the topic in detail.
Aggregation products
When peptides degrade, they can form aggregates, clumps of misfolded protein that the immune system may recognize as foreign. Injecting aggregated peptide products can trigger immune reactions ranging from mild injection site responses to more significant allergic-type reactions in rare cases. This is another reason why clear, particle-free solution is non-negotiable for safe injection.

Compounded versus brand-name shelf life comparison
The shelf life differences between compounded and brand-name semaglutide are significant, and understanding them helps you plan your purchases, schedule your doses, and avoid waste.
Why brand-name products last longer
Brand-name products like Ozempic and Wegovy are manufactured in facilities with extensive environmental controls, use pharmaceutical-grade excipients specifically selected for stability, and undergo rigorous accelerated stability testing before receiving FDA approval. Their expiration dates reflect years of data from controlled stability studies.
Compounded semaglutide, by comparison, is prepared in pharmacy settings with variable levels of environmental control. The excipients may differ from the brand-name formulation. And the stability testing, while required, may not be as extensive as what large pharmaceutical manufacturers conduct. This does not mean compounded semaglutide is inferior during its stated shelf life, it simply means the validated timeframe is shorter.
Many people choose compounded semaglutide for cost or accessibility reasons, and that is a valid decision. The complete guide to compounded semaglutide covers the differences between brand and compounded formulations in detail. Understanding how GLP-1 medications relate to each other provides additional context for making informed choices.
503A versus 503B pharmacies
Not all compounding pharmacies are the same. The distinction between 503A and 503B facilities has direct implications for shelf life:
503A pharmacies compound medications pursuant to individual patient prescriptions. They typically operate under state pharmacy board oversight and follow USP 797 guidelines. Beyond-use dates from 503A pharmacies tend to be conservative, often 28 days for sterile preparations, because extensive stability testing is not always performed.
503B outsourcing facilities operate under FDA oversight and can compound without individual prescriptions in anticipation of demand. They must follow current good manufacturing practice (cGMP) requirements and typically conduct more extensive stability testing. As a result, 503B facilities may assign longer beyond-use dates, sometimes 60, 90, or even 120 days.
When evaluating compounded semaglutide dose options, ask your pharmacy whether they are a 503A or 503B facility. This single question tells you a lot about the stability data behind the beyond-use date on your vial. Specific pharmacy guides for Empower Pharmacy, Olympia Pharmacy, and Direct Meds can help you understand what different pharmacies offer.
Compounded semaglutide with additives
Some compounded semaglutide formulations include additives like vitamin B12 (methylcobalamin), glycine, or other ingredients. These additives can affect shelf life in ways that are not always intuitive.
For example, semaglutide with B12 formulations may have different stability profiles than semaglutide alone, because methylcobalamin has its own sensitivity to light and temperature. The B12 semaglutide dosage chart accounts for these combination formulations. Similarly, semaglutide with glycine may have stability characteristics that differ from the base formulation.
Always follow the beyond-use date specific to your exact formulation. A BUD for plain semaglutide does not necessarily apply to a semaglutide-B12 combination from the same pharmacy.
USP 797 and beyond-use dates explained
If you use compounded semaglutide, understanding USP 797 helps you make sense of the beyond-use dates on your vials. This chapter of the United States Pharmacopeia governs how compounded sterile preparations are made, tested, and dated.
What USP 797 requires
USP 797 establishes standards for compounding sterile preparations in pharmacies and healthcare settings. It defines requirements for:
Personnel training and competency assessment
Cleanroom environments and air quality standards
Equipment maintenance and calibration
Procedures for compounding, labeling, and storage
Quality assurance including sterility testing
Beyond-use date assignment
The beyond-use date (BUD) is different from an expiration date. An expiration date is assigned by a manufacturer based on extensive stability studies. A beyond-use date is assigned by a compounder based on available stability data, professional judgment, and USP 797 default timeframes.
Default beyond-use dates under USP 797
When a pharmacy has not performed stability testing on a specific formulation, USP 797 provides default BUDs based on the compounding environment:
For preparations compounded in a segregated compounding area (lower environmental controls), the default BUD is 12 hours at room temperature or 24 hours refrigerated. For preparations compounded in a classified cleanroom (higher environmental controls), default BUDs extend to 28 days refrigerated.
These defaults are conservative by design. They assume worst-case scenarios for contamination and degradation. Pharmacies that conduct stability-indicating testing can assign longer BUDs that more accurately reflect the actual shelf life of their specific formulation.
How this affects your semaglutide
If your compounded semaglutide vial has a 28-day BUD, it likely means the pharmacy is using USP 797 default dating without product-specific stability testing. This does not mean the semaglutide degrades on day 29. It means the pharmacy cannot guarantee potency and sterility beyond that point based on the data they have.
If your vial has a 90 or 120-day BUD, the pharmacy has conducted stability testing demonstrating that the formulation maintains potency and sterility for that extended period under specified conditions. This is a good sign, it means more rigorous quality control.
Either way, always store and handle the product exactly as directed on the label. The BUD is only valid under the specified storage conditions. Understanding the specifics of your semaglutide formulation helps you ask the right questions at your pharmacy.
How to maximize semaglutide shelf life
You cannot extend semaglutide beyond its validated shelf life. But you can absolutely prevent premature degradation by following these practices. Most people lose potency not because the product expired, but because they stored it poorly.
Temperature control best practices
Keep injectable semaglutide refrigerated at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius) whenever possible. Even if room temperature storage is permitted (as with opened Ozempic pens), refrigeration slows degradation and maximizes potency throughout the use period.
Use a refrigerator thermometer. Verify your refrigerator is actually maintaining the correct temperature. Household refrigerators can vary by several degrees depending on location, how often the door is opened, and the age of the appliance. A thermometer costs a few dollars and eliminates guesswork.
Position matters. Store semaglutide on a middle shelf, not in the door (temperature fluctuations) and not against the back wall (freezing risk). A dedicated spot that you use consistently reduces the chance of accidental temperature exposure. Applying the same discipline to all your peptides, whether semaglutide or BPC-157, keeps everything organized and properly stored.
Minimize exposure events
Every time you take your semaglutide out of the refrigerator, the clock is ticking on room temperature exposure. Minimize this by having your injection supplies ready before removing the vial or pen. Draw your dose, inject, and return the product to the refrigerator promptly. Do not leave it sitting on a counter while you eat breakfast or get ready for work.
For multi-dose vials, use proper aseptic technique to minimize contamination risk with each draw. The fewer contaminants that enter the vial, the longer the solution remains viable. Using a peptide calculator before drawing helps you get the correct dose on the first attempt, reducing the number of times you need to access the vial.
Use the right reconstitution volume
If you are reconstituting lyophilized semaglutide, the volume of bacteriostatic water you add affects how quickly you use the vial and, consequently, how long the reconstituted solution needs to last. Adding too much water creates a dilute solution that requires larger injection volumes per dose but gives you more total doses from the vial, extending the time it takes to use it up.
If it will take you more than 28 days to use the vial at your current dose, consider adding less water to create a more concentrated solution. This means smaller injection volumes but faster use of the vial, keeping you within the safe reconstituted shelf life window. The semaglutide mixing chart provides detailed guidance on water volumes for different concentrations. You can also consult the 5 mg mixing chart or the 10 mg mixing chart for vial-specific ratios.
Track your dates
Write the date of first use directly on the vial or pen with a permanent marker. Do not rely on memory. After a few weeks, you will not remember whether you opened it on a Tuesday or a Thursday, and that ambiguity leads to either premature disposal or risky use of potentially expired product.
For compounded vials, write both the beyond-use date and the date you first punctured the stopper. The shorter of these two dates governs when you should discard the product. Some people keep a simple log, a note on their phone or a sticky note on the refrigerator, tracking all their medication dates.
Common mistakes that shorten semaglutide shelf life
Knowing what to do is important. Knowing what not to do is equally valuable. These are the most common mistakes that cause premature semaglutide degradation, and nearly all of them are preventable.
Leaving semaglutide out overnight
It happens. You inject before bed, set the vial on the bathroom counter, and forget to put it back in the refrigerator. By morning, it has been at room temperature for 8 to 10 hours. Is it ruined?
For Ozempic pens, a single overnight room temperature exposure within the 59 to 86 degree range is unlikely to cause significant degradation. The pen is rated for 56 days at room temperature, so one night will not destroy it. But it does chip away at the overall stability budget.
For compounded semaglutide, the answer depends on your specific formulation and the room temperature. One night at 70 degrees Fahrenheit is different from one night at 85 degrees. As a general rule, a single overnight exposure is recoverable if you promptly return the vial to the refrigerator. Multiple overnight exposures accumulate damage. If this happens regularly, expect reduced potency before the beyond-use date.
Storing in the refrigerator door
The refrigerator door is the warmest and most temperature-variable location in the unit. Every time you open the door, warm air rushes in and directly contacts items stored there. Temperature can swing by 10 degrees or more with each door opening. Over weeks, these repeated temperature fluctuations accelerate peptide degradation.
Move your semaglutide to the main body of the refrigerator. The middle shelf, away from the back wall and away from the door, provides the most stable temperature environment.
Using non-sterile equipment
Drawing from a vial with a previously used needle, touching the needle tip, or skipping alcohol swabs on the stopper introduces microorganisms into the solution. Once contaminated, degradation accelerates rapidly regardless of temperature control. Bacterial enzymes break down peptides far faster than chemical degradation alone.
Always use new, sterile needles and syringes for every draw. The semaglutide syringe dosage chart helps you identify the correct syringe size for accurate dosing. If you are unsure about proper injection equipment, the peptide injection overview covers the fundamentals.
Shaking the vial or pen
Vigorous shaking creates foam and air bubbles in the solution. This introduces air interfaces where the peptide molecules can denature (unfold). The agitation plus air exposure combination is particularly damaging.
If you need to mix a compounded vial (for example, after reconstitution), gently swirl or roll the vial between your palms. Never shake. The same applies to Ozempic and Wegovy pens, do not shake them. If you see air bubbles in an Ozempic pen, tap the pen gently with the needle pointing up and push the air bubble out before injection.
Exposing to direct sunlight
Even 30 minutes of direct sunlight can cause measurable degradation in semaglutide solutions. UV radiation breaks chemical bonds in the peptide chain. Keep your semaglutide in its original box, in the refrigerator, away from windows. When transporting, use an opaque insulated bag.
Splitting vials into multiple containers
Some people try to extend usability by transferring semaglutide from the original vial into multiple smaller containers. This is risky for several reasons. Each transfer introduces contamination risk. The new containers may not be pharmaceutical grade. The solution contacts additional surfaces that can catalyze degradation. Unless you are working in a certified cleanroom with proper aseptic technique, keep the semaglutide in its original container.
If you have more semaglutide than you can use within the shelf life window, the 5 mg reconstitution chart and 10 mg reconstitution chart can help you calculate a reconstitution volume that matches your usage timeline.

Semaglutide shelf life and cost efficiency
Understanding shelf life is not just about safety. It is about getting the most value from every dollar you spend on semaglutide. Wasted doses add up quickly, especially for self-pay patients or those using compounded formulations.
Calculating your true cost per dose
The cost per dose depends on how many usable doses you actually get from a vial or pen before it expires. If a compounded vial has a 28-day BUD and your dosing schedule only uses 60% of the vial in that time, your effective cost per dose is nearly double the theoretical cost.
The peptide cost calculator can help you determine actual cost per dose based on your specific vial size, concentration, and dosing schedule. This information helps you choose the most cost-effective vial size and reconstitution volume for your needs.
For example, if you are on a low starting dose and using a large vial, you may not finish the vial before the BUD expires. In that case, a smaller vial (like a 5 mg vial) might be more cost-effective than a 10 mg vial, even though the per-milligram price is higher. The math depends on your dose and your vial shelf life.
Timing your orders
Do not stockpile semaglutide. Order what you need for the next 30 to 60 days and no more. Compounded vials sitting in your refrigerator accumulate age from the date of compounding, not the date you open them. A vial that arrives with 90 days of BUD remaining and sits unopened for 60 days only has 30 days left when you start using it.
Ask your pharmacy about the BUD when you order. If possible, request the most recently compounded vial available. Some pharmacies will note the compounding date on the label, which gives you full visibility into the remaining shelf life.
Dose titration and shelf life planning
Semaglutide protocols typically involve dose escalation, starting at a lower dose (often 0.25 mg per week) and gradually increasing. During the low-dose phase, you use less product per week, meaning a vial lasts longer. If your vial BUD is 28 days and your starting dose only uses a quarter of the vial in that time, three-quarters of your product will be wasted.
Planning your reconstitution volume to match your dosing phase helps. The semaglutide dosage chart in mL and dosage chart in units help you calculate exactly how much you will use per week at each dose level. Cross-reference this with your vial shelf life to find the optimal balance.
SeekPeptides provides tools and guides specifically designed to help with these calculations, including the semaglutide dosage calculator that accounts for vial concentration, dose amount, and frequency to tell you exactly how many doses you will get from each vial. Members access detailed protocols that cover semaglutide dosage in units, making it easier to calculate use timelines.
Specific formulation storage guides
This section consolidates the storage rules for each specific formulation into quick-reference guides. If you know exactly which product you are using, jump to that section for all the information you need in one place.
Ozempic storage guide
Before first use: Refrigerate at 36-46F (2-8C). Good until expiration date. Do not freeze.
After first use: 56 days at room temperature (59-86F) or refrigerated. Replace cap after each use. Remove needle after each injection. Discard after 56 days regardless of remaining medication.
What to avoid: Freezing, direct sunlight, temperatures above 86F, leaving uncapped, sharing with others.
The Ozempic dosing chart in units helps you track how many doses remain in your pen so you can plan accordingly and avoid waste. For long-term semaglutide use, consistent storage practices become second nature over time.
Wegovy storage guide
Before first use: Refrigerate at 36-46F (2-8C). Good until expiration date. Do not freeze.
After first use: 28 days at room temperature (59-86F). Each pen is single-dose, use promptly after removing cap. Discard used pens properly.
What to avoid: Freezing, heat above 86F, removing from packaging prematurely, moisture exposure.
Compounded semaglutide vial storage guide
Before first use: Refrigerate at 36-46F (2-8C). Follow pharmacy BUD (28-120 days depending on facility). Do not freeze.
After first puncture: Continue refrigerating. Use within pharmacy-specified BUD from date of first puncture or compounding date (whichever is shorter). Swab stopper before each use. Use new sterile needle each time.
What to avoid: Room temperature storage, freezing, contaminated needles, multiple rapid temperature changes, shaking, sunlight exposure.
Resources like the Strive Pharmacy semaglutide guide and the IVIM semaglutide dosing chart provide pharmacy-specific information that can help you understand the products from these particular providers.
Lyophilized semaglutide storage guide
Before reconstitution: Store desiccated at -18C or below. Good for 24-36 months. Keep sealed and protected from moisture.
After reconstitution with bacteriostatic water: Refrigerate at 36-46F (2-8C). Use within 28-30 days. Swirl gently to mix, never shake.
After reconstitution with sterile water: Refrigerate. Use within 24-48 hours. Preferably single-use.
For extended storage: Prepare single-use aliquots and freeze at -20C or -80C. Thaw once, use, discard remainder. Never refreeze.
The reconstitution calculator helps you determine the exact water volume needed for your desired concentration. For a step-by-step walkthrough, the general peptide reconstitution guide covers the fundamentals, and the free reconstitution calculator guide explains how to use online tools for accurate mixing.
Rybelsus tablet storage guide
Storage: Room temperature 68-77F (20-25C). Excursions to 59-86F permitted. Do not refrigerate.
Packaging: Keep in original blister pack until moment of use. Do not transfer to pill organizer. Protect from moisture and light.
Shelf life: Until expiration date (approximately 24 months from manufacture).
What to avoid: Refrigeration, moisture, light exposure, removing from blister pack early, handling with wet hands.
Semaglutide alternatives and their shelf life comparison
Understanding how semaglutide shelf life compares to alternatives helps you make informed decisions, especially if shelf life is a practical concern for your situation.
Tirzepatide shelf life comparison
Tirzepatide (Mounjaro/Zepbound) has similar storage requirements to semaglutide. Unopened, it is stored refrigerated and good until the expiration date. After first use, Mounjaro pens are good for 21 days at room temperature (shorter than Ozempic at 56 days but similar to Wegovy at 28 days).
For those choosing between the two, the semaglutide versus tirzepatide comparison covers not just shelf life but dosing, side effects, and effectiveness differences. The dosage comparison chart helps with understanding equivalent doses. You can also explore the question of tirzepatide expiration and tirzepatide refrigeration requirements in dedicated guides.
Compounded tirzepatide follows similar rules to compounded semaglutide. Beyond-use dates depend on the pharmacy and their stability testing. The tirzepatide refrigerated shelf life and tirzepatide room temperature stability guides provide specific timelines. If you are working with powdered tirzepatide, the tirzepatide reconstitution guide covers the process step by step.
Other GLP-1 peptides and shelf life
Other GLP-1 related peptides and combinations have their own shelf life profiles. Cagrilintide and semaglutide combinations represent newer formulations with their own storage requirements. Retatrutide, a triple agonist, is another option being explored with different stability characteristics.
For those looking at Ozempic alternatives, understanding the shelf life of each option is part of the overall cost-benefit analysis. Shorter shelf life means more potential waste, higher effective cost, and more frequent ordering.
The broader landscape of peptides for weight loss includes multiple options beyond semaglutide, and the best peptides for weight loss guide compares them on multiple factors including stability and storage requirements. Understanding peptides for fat loss more broadly provides context for where semaglutide fits in the larger peptide landscape.
Semaglutide dosing schedules and shelf life planning
Your dosing schedule directly impacts how shelf life affects you. A higher-dose user will finish a vial faster and may never encounter shelf life limits. A lower-dose user needs to plan more carefully.
Standard dose escalation and vial usage
The typical semaglutide dose escalation for weight management follows this pattern:
Weeks 1-4: 0.25 mg per week
Weeks 5-8: 0.5 mg per week
Weeks 9-12: 1.0 mg per week
Weeks 13-16: 1.7 mg per week
Week 17 onward: 2.4 mg per week (maintenance)
At the starting dose of 0.25 mg per week, a 5 mg vial would theoretically last 20 weeks, far beyond any reasonable shelf life for reconstituted solution. This is why vial size selection and reconstitution volume planning matter so much in the early phases. The 5 mg semaglutide dosage chart and 5 mg/2 mL dosage chart help you map out usage at different doses.
At maintenance doses of 1.7 to 2.4 mg per week, a 5 mg vial would last roughly 2 to 3 weeks, well within the 28-day minimum BUD. At this phase, shelf life is rarely a concern. The 5 mg/mL concentration chart and 10 mg dosage chart provide dose-specific calculations for maintenance-phase users.
Unit conversions and shelf life tracking
Many people using compounded semaglutide measure doses in units (on an insulin syringe) rather than milligrams. Understanding the conversion between units and milligrams is essential for tracking how quickly you are using your vial and whether you will finish it before the BUD.
The semaglutide unit conversion chart and syringe dosage conversion chart make these calculations straightforward. Common conversions people search for include how many mg is 40 units, how many mg is 50 units, 10 units in mg, 20 units in mg, and 100 units in mg. Knowing these conversions helps you calculate exactly how many weeks of use your vial provides. You can also reference the 20 units in mL guide for volume-based measurements.
Accurate dosing eliminates waste. The peptide dosage calculation guide provides the mathematical framework for these conversions, and the peptide dosage chart offers a quick visual reference. Tools like the peptide stack calculator can also help if you are managing multiple peptides alongside your semaglutide protocol.
Managing semaglutide side effects and shelf life concerns
Sometimes what people attribute to degraded semaglutide is actually a side effect of the medication working as intended. And sometimes what people dismiss as a normal side effect is actually a sign of product degradation. Knowing the difference helps you avoid unnecessary waste while staying safe.
Normal side effects versus degradation signs
Normal side effects that are NOT signs of degradation:
Constipation (common, especially at higher doses)
Nausea (most common side effect, usually temporary)
Fatigue (reported by some users, especially early in treatment)
Injection site reactions (mild redness, itching)
Headache (common in early weeks)
Signs that suggest degraded product rather than normal side effects:
Sudden loss of previously established appetite suppression
Blood sugar readings trending upward without dietary changes
Increased injection site reactions (larger area, more redness than previously experienced)
Unusual pain or burning at injection site not experienced with fresh product
If you are experiencing difficulty losing weight on semaglutide, product degradation is one of many factors to consider. The semaglutide withdrawal symptoms guide can help you distinguish between reduced medication effect and true withdrawal if you are transitioning between products.
Diet and lifestyle interactions with shelf life planning
Your diet can influence how you respond to semaglutide, which in turn affects whether you attribute changes to the medication or to product degradation. Understanding which foods to eat while on semaglutide helps maintain consistent results that make it easier to spot when something is off with the product itself.
Similarly, alcohol consumption on semaglutide affects both tolerance and perceived effectiveness. If your results are inconsistent, addressing lifestyle factors first before suspecting product degradation is the logical approach. The semaglutide diet plan provides a structured framework for nutrition while on the medication.
Some users combine semaglutide with other medications like phentermine. The phentermine versus semaglutide comparison covers the differences between these approaches. Others explore supportive peptides to take alongside semaglutide for complementary benefits.
Energy and effectiveness over time
Questions about whether semaglutide affects energy levels are common, and changes in energy during a treatment course can sometimes be confused with product degradation. If you notice a shift in energy or effectiveness, consider where you are in the dose escalation process, how long you have been at your current dose, and whether your vial is within its shelf life before drawing conclusions.
The timeline for semaglutide effects provides realistic expectations for when to expect changes, which serves as a baseline for evaluating product quality over time. Understanding this timeline helps separate normal adaptation from degradation-related potency loss.
Switching between semaglutide formulations
People switch between semaglutide formulations for various reasons: cost, availability, preference, or insurance changes. Each switch requires understanding the shelf life rules for the new formulation.
Brand-name to compounded
Switching from Ozempic or Wegovy to compounded semaglutide means adapting to shorter, more variable shelf life expectations. Where Ozempic gave you 56 days of room temperature stability, your compounded vial may require strict refrigeration and a 28-day use window.
The switching guide covers transitions between GLP-1 medications, including practical storage adjustments. For dose equivalence during transitions, the semaglutide B12 dosage chart helps if your new compounded formulation includes B12.
Injectable to oral
Moving from injectable semaglutide to Rybelsus tablets represents a complete change in storage requirements. The injectable versus oral comparison covers the broader differences, but from a storage perspective, the transition eliminates the need for refrigeration while introducing light and moisture sensitivity concerns.
Oral bioavailability of semaglutide is significantly lower than injectable, which means the dose numbers look different. The oral semaglutide drops guide covers alternative oral delivery methods beyond tablets. This does not reflect a shelf life issue, it is a fundamental difference in how the two formulations deliver the active ingredient to the bloodstream.
Long-term storage for peptide researchers
Researchers working with semaglutide often need to store larger quantities for extended periods. The strategies differ from clinical use and involve more specialized equipment and techniques.
Bulk powder storage
Lyophilized semaglutide powder, when stored properly desiccated at -20C or below, maintains stability for extended periods. Researchers purchasing in bulk should divide the powder into smaller aliquots in a dry, controlled environment, seal each aliquot under nitrogen or argon gas to displace oxygen, and store at the coldest available temperature.
At -80C, lyophilized peptides can maintain stability far beyond the standard 24 to 36 month window. The investment in a -80C freezer pays for itself many times over if you are working with expensive peptides in significant quantities. For general guidance on peptide powder stability, the powder form shelf life guide provides detailed timelines across different peptide types.
Working solution preparation
Rather than reconstituting an entire vial and hoping to use it within 28 days, researchers often reconstitute only what they need for a specific period. This minimizes waste and ensures every dose comes from freshly reconstituted solution.
For research-grade semaglutide, reconstitute a portion sufficient for 5 to 7 days of use, keeping the remainder as dry powder. This approach maximizes the potency of every dose while taking advantage of the long-term stability of the lyophilized form. The reconstituted peptide refrigerator life guide provides timelines for different reconstitution approaches. Understanding peptide refrigerator stability in general helps frame expectations for any reconstituted product.
The list of injectable peptides provides context for how semaglutide storage compares across the broader peptide landscape. Many of the same principles apply whether you are working with semaglutide, BPC-157, or any other research peptide. Building comprehensive storage protocols using the peptide stacks guide helps manage multiple products simultaneously.
The role of peptide quality in shelf life
Not all semaglutide starts from the same baseline. The purity and quality of the starting material affects how long the final product remains stable and effective.
Purity matters
Higher purity semaglutide generally has a longer effective shelf life. Impurities in the peptide, synthesis byproducts, truncated sequences, oxidized residues, act as catalysts for further degradation. A product that starts at 98% purity will degrade faster than one starting at 99.5% purity because the impurities themselves promote chemical breakdown.
Resources like the peptide dosage calculation guide emphasize the importance of starting with high-quality products. This is one reason brand-name products from large pharmaceutical manufacturers tend to have longer shelf lives. Their manufacturing processes achieve and verify extremely high purity levels. Compounded products may vary in purity depending on the API (active pharmaceutical ingredient) source and the compounding process.
Excipient quality
The inactive ingredients (excipients) in a semaglutide formulation affect stability. Buffers that maintain optimal pH, preservatives that prevent microbial growth, and stabilizers that protect the peptide structure all contribute to shelf life. Brand-name formulations use excipients that have been specifically optimized through extensive formulation development. Compounded products use standard pharmaceutical excipients that provide adequate but potentially not equivalent protection.
For those exploring different peptide options and their relative stability profiles, the copper peptide refrigeration guide demonstrates how storage requirements vary based on formulation chemistry. Different peptide types require different approaches, and understanding these differences prevents applying one-size-fits-all storage rules to products that need individualized handling.
Using tools and trackers to manage shelf life
Managing semaglutide shelf life does not need to be complicated, but it does need to be systematic. A few simple tools and habits prevent most waste and safety issues.
Phone reminders and calendar alerts
When you open a new vial or pen, immediately set a phone reminder for the discard date. If your Ozempic pen has 56 days, set a reminder for day 50 giving you a week to use remaining doses. For a compounded vial with a 28-day BUD, set reminders at day 21 (one week remaining) and day 27 (final day).
Dose tracking
Track how much medication you have used from each vial. This helps you estimate whether you will finish the vial before the BUD, or whether you need to adjust your reconstitution volume next time. Simple tracking can be a note on your phone or a small chart taped to the refrigerator.
SeekPeptides offers tools that simplify this tracking, including the GLP-1 plotter for visualizing your dosing schedule and the GLP-1 companion tool for tracking your overall GLP-1 medication management. These resources take the guesswork out of managing both dosing and shelf life simultaneously.
Temperature monitoring
A refrigerator thermometer with min/max recording capability shows you the temperature range your semaglutide has been exposed to. If the max temperature reading exceeds 46F (8C) or the min drops below 36F (2C), you know the storage conditions have been compromised and can adjust accordingly.
More advanced options include wireless temperature monitors that send alerts to your phone if the refrigerator temperature goes out of range. These are especially useful during travel or if you store semaglutide in a secondary refrigerator that you do not check regularly.
Frequently asked questions
Can I use semaglutide after the expiration date?
Using semaglutide after the expiration date is not recommended. Potency decreases over time, meaning you may not get the intended therapeutic effect. More importantly, sterility cannot be guaranteed beyond the expiration or beyond-use date. For a thorough analysis of the risks and realities, the expired semaglutide guide covers this topic in depth.
Does semaglutide need to be refrigerated at all times?
Compounded semaglutide should be refrigerated at all times. Ozempic pens can be stored at room temperature (59-86F) for up to 56 days after first use. Wegovy pens allow 28 days at room temperature after opening. Rybelsus tablets are stored at room temperature and should not be refrigerated. The compounded semaglutide refrigeration guide provides specific guidance for compounded formulations.
What should I do if my semaglutide was left out of the fridge overnight?
For Ozempic pens, a single overnight exposure at room temperature is within the allowed usage window and should not cause significant issues. For compounded semaglutide, return it to the refrigerator immediately. A single overnight exposure at normal room temperature is unlikely to cause complete degradation, but repeated exposures will reduce potency and shorten the effective shelf life.
How can I tell if my semaglutide has gone bad?
Check for cloudiness, floating particles, color changes (yellowing), unusual thickness, or off odors. However, degraded semaglutide can also look normal while having reduced potency. If it is past the expiration or beyond-use date, discard it regardless of appearance. Reduced appetite suppression or blood sugar control can also indicate product degradation.
Is frozen semaglutide still good?
No. Once liquid semaglutide has frozen, the peptide structure is damaged by ice crystal formation. Even if the solution looks normal after thawing, the biological activity is compromised. This applies to all liquid semaglutide formulations. The only form that can be frozen is lyophilized (dry) powder, which is designed for frozen storage. Understanding the broader principles of peptide expiration reinforces why freeze-thaw damage is irreversible.
Why does compounded semaglutide have a shorter shelf life than Ozempic?
Brand-name products like Ozempic undergo years of stability testing with optimized formulations designed for maximum shelf life. Compounded semaglutide uses different excipients, is prepared in different environments, and has less extensive stability data. The result is shorter validated shelf life, though the semaglutide molecule itself is identical. 503B outsourcing facilities with rigorous stability testing may offer longer beyond-use dates than standard 503A pharmacies.
Can I freeze reconstituted semaglutide in aliquots for later use?
This is a research-grade approach that works under specific conditions. Divide reconstituted semaglutide into single-use aliquots immediately after reconstitution and freeze at -20C or colder. Thaw each aliquot once, use it completely, and discard any remainder. Never refreeze a thawed aliquot. This method extends usable life well beyond the 28-day refrigerated window, but proper technique is essential to maintain potency.
How long is semaglutide good for in a syringe?
Semaglutide drawn into a syringe should be injected within a few hours. Syringes are not designed for storage. They lack the preservative environment of a sealed vial, and the plastic and rubber components can interact with the peptide over time. Draw your dose and inject promptly. If you are using an appropriate injection site, the process from drawing to injecting takes only minutes.
External resources
USP Chapter 797 Pharmaceutical Compounding, Sterile Preparations
FDA Information on Compounded GLP-1 Receptor Agonist Products
For researchers serious about optimizing their peptide protocols and managing their semaglutide with precision, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, storage protocols, dosing calculators, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your semaglutide stay potent, your storage stay cold, and your results stay consistent.