Feb 20, 2026

You are wasting your semaglutide right now. Maybe not all of it. Maybe not today. But every time that vial sits on a warm counter, every time the ice packs melt during shipping, every time you leave it in a hot car for even twenty minutes, molecules are breaking apart. Silently. Invisibly. And by the time you notice something is wrong, by the time your appetite suppression weakens or your weight loss stalls, you have already injected doses of degraded medication that did nothing.
Here is the number that should concern you. Semaglutide is a 31-amino-acid peptide with a specific three-dimensional structure that allows it to bind to GLP-1 receptors in your body. That structure depends on hydrogen bonds and hydrophobic interactions that are extremely sensitive to heat. Once those bonds break, the peptide unfolds. Once it unfolds, it cannot refold correctly. Once it cannot refold, you are injecting expensive water. And the worst part? You cannot see any of this happening. The solution looks identical whether it contains fully active semaglutide or a vial full of denatured, useless peptide fragments.
This guide covers everything you need to know about semaglutide and temperature. The exact thresholds where damage begins. The differences between compounded semaglutide and branded formulations like Ozempic and Wegovy. How to tell if your medication has been compromised. What to do when accidents happen. And how to store, travel with, and handle your semaglutide so that every single dose delivers its full therapeutic effect. Because understanding how long semaglutide stays good is not optional knowledge. It is essential.
The science behind semaglutide and heat damage
Semaglutide is not a simple chemical compound. It is a biologic, a complex molecule built from amino acids arranged in a precise sequence and folded into a specific three-dimensional shape. That shape is everything. Without it, the molecule cannot dock with GLP-1 receptors, cannot trigger the signaling cascades that suppress appetite, and cannot produce the metabolic effects that make the medication work.
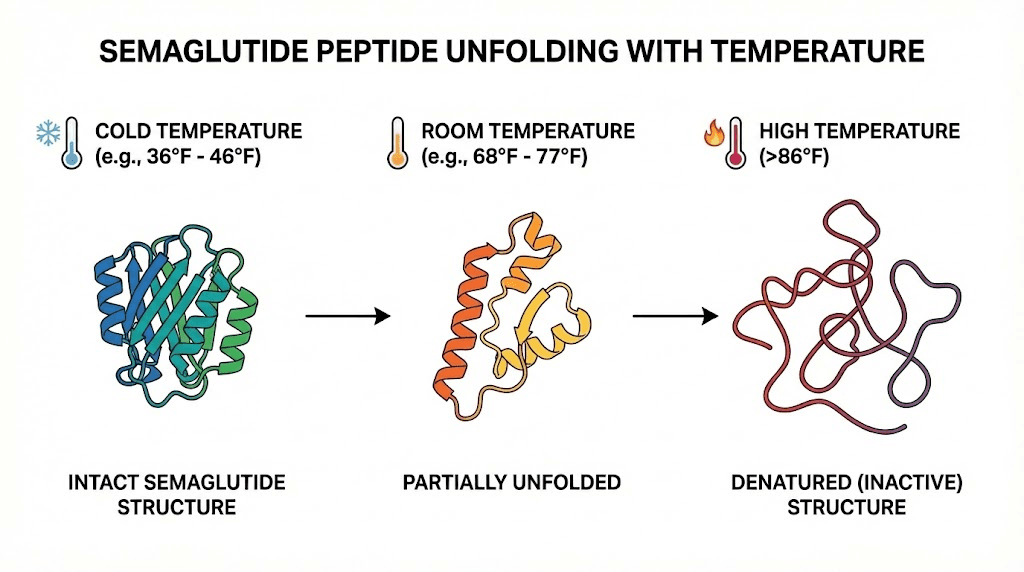
Heat disrupts this shape through a process called denaturation.
Think of a protein like a carefully folded piece of origami. Each fold is held in place by weak chemical bonds, hydrogen bonds between amino acid residues, hydrophobic interactions that keep water-hating segments tucked inside the structure, and van der Waals forces that maintain the overall configuration. When temperature rises, these bonds begin to vibrate more intensely. At a certain point, they break.
The peptide unfolds. And unlike that origami, it does not refold correctly when cooled back down.
Research published in the Journal of Peptide Science examined semaglutide stability across multiple temperatures. At 25 degrees Celsius (77 degrees Fahrenheit), the peptide remained relatively stable for extended periods. At 40 degrees Celsius (104 degrees Fahrenheit), degradation accelerated significantly. And at 80 degrees Celsius (176 degrees Fahrenheit), semaglutide survived only about 3 hours before substantial breakdown occurred.
But raw survival is not the same as therapeutic potency. A peptide can appear physically intact while losing enough structural integrity to reduce its binding affinity. This means your semaglutide dose might contain technically present but functionally impaired molecules, delivering less medication than you calculated.
Chemical degradation pathways
Denaturation is only one way heat damages semaglutide. Several chemical degradation pathways also accelerate with rising temperatures.
Oxidation attacks specific amino acid residues, particularly methionine and cysteine. Heat accelerates oxidative reactions by increasing molecular collision rates. Oxidized semaglutide shows reduced receptor binding and altered pharmacokinetics, meaning it clears from your system differently than intact medication.
Hydrolysis breaks peptide bonds themselves. Water molecules attack the backbone of the amino acid chain, literally cutting the semaglutide molecule into fragments. These fragments have no therapeutic activity. At elevated temperatures, hydrolysis rates can increase dramatically, turning what would be months of slow degradation into days or even hours of rapid breakdown.
Aggregation happens when partially unfolded semaglutide molecules stick together. Research from Biomacromolecules found that semaglutide in aqueous solution forms oligomeric micelles and short fibrils over time. Heat accelerates this process significantly. Aggregated semaglutide not only loses potency but can potentially trigger immune responses at the injection site.
The pH of the solution also matters. Studies show that pH is a key factor in thermal degradation of semaglutide, with certain pH ranges accelerating breakdown at elevated temperatures. This is why branded formulations use carefully optimized buffer systems, while compounded versions may vary in their thermal stability depending on the pharmacy formulation.
Why you cannot see the damage
This is the most dangerous aspect of heat-damaged semaglutide. There are no visible indicators of degradation.
The solution stays clear. It stays colorless. It looks exactly the same as perfectly stored medication. Unlike food that smells bad when spoiled or milk that curdles when expired, semaglutide gives you no warning signs until degradation becomes extreme.
Only at advanced stages of breakdown might you notice cloudiness, particles floating in the solution, or color changes. By that point, the medication is severely compromised and should have been discarded long ago.
The practical signs are clinical. Your appetite suppression weakens. Your blood glucose levels rise if you are using it for diabetes management. Your weight loss stalls without explanation. And you might blame yourself, your diet, or your dosing protocol when the real problem is sitting in your refrigerator, a vial of medication that looks perfect but has lost its potency.
Temperature thresholds: the exact numbers
Not all heat exposure is equal. Understanding the specific temperature thresholds helps you assess whether your semaglutide has been compromised and what action to take. These numbers come directly from manufacturer data, FDA guidelines, and published stability research.
The safe zone: 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius)
This is optimal storage temperature. Your refrigerator. At these temperatures, semaglutide remains stable for its full shelf life, typically until the manufacturer expiration date for branded products or the beyond-use date for compounded formulations.
Every moment your semaglutide spends in this range is a moment of zero degradation. This is where it should live when not in active use. If you are managing your dosing schedule properly, the vial leaves the refrigerator only briefly for injection preparation.
The acceptable zone: 46 to 86 degrees Fahrenheit (8 to 30 degrees Celsius)
Room temperature. This is where the differences between branded and compounded semaglutide become critical.
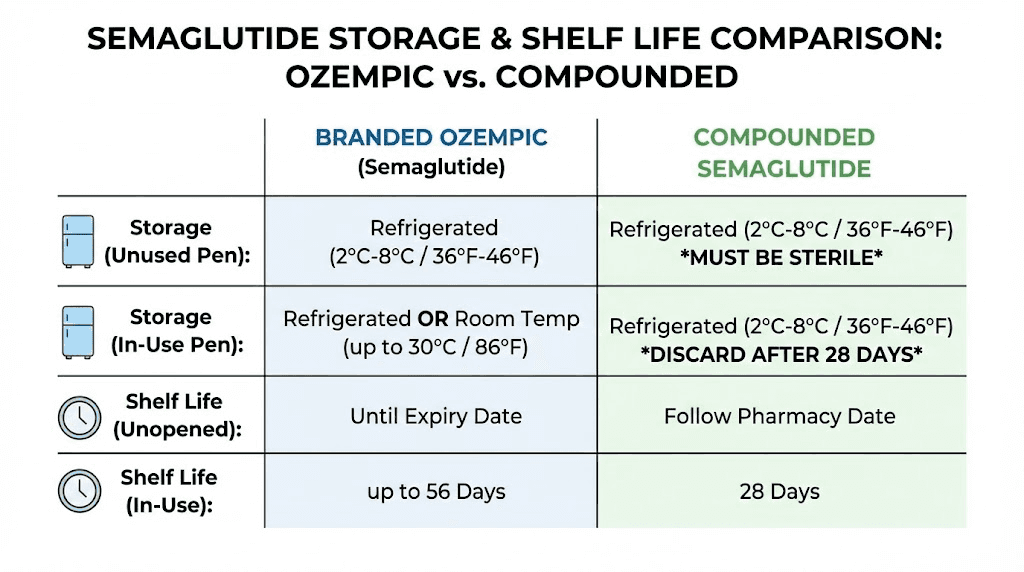
For Ozempic (branded semaglutide injection for diabetes), once the pen is in use, it can be stored at room temperature up to 86 degrees Fahrenheit for up to 56 days. Novo Nordisk conducted extensive stability testing to validate this window. After 56 days at room temperature, the pen must be discarded regardless of remaining medication.
For Wegovy (branded semaglutide for weight management), the window is shorter. Unopened pens can be kept at room temperature up to 86 degrees Fahrenheit for up to 28 days before first use. This shorter window reflects the different pen design and concentration.
For Rybelsus (oral semaglutide tablets), room temperature storage between 68 and 77 degrees Fahrenheit is actually the standard recommendation, with temporary excursions permitted up to 86 degrees.
For compounded semaglutide, the rules are stricter. Most compounding pharmacies require continuous refrigeration with a beyond-use date of 28 to 30 days after first puncture. Some formulations from pharmacies conducting rigorous stability testing may allow up to 120 days in the refrigerator. But room temperature tolerance is generally limited, and you should follow whatever your specific pharmacy specifies. When in doubt, check our guide on whether compounded semaglutide needs refrigeration.
The danger zone: 86 to 104 degrees Fahrenheit (30 to 40 degrees Celsius)
Above 86 degrees, degradation accelerates meaningfully. This is the temperature range of a hot car interior, a warm shipping container, or a sunny kitchen counter in summer.
Manufacturer guidelines are clear. Semaglutide exposed to temperatures above 86 degrees Fahrenheit should be evaluated carefully. Brief excursions of minutes may not cause significant damage. Extended exposure of hours begins to measurably reduce potency.
There is no published data on exactly how much potency is lost per hour at these temperatures because degradation depends on multiple variables: the specific formulation, preservatives used, pH of the solution, whether the vial has been previously opened, and the exact temperature profile during exposure.
What we do know is that this range marks the transition from acceptable storage to active degradation. If you are traveling with semaglutide, keeping it below this threshold is essential.
The critical zone: above 104 degrees Fahrenheit (40 degrees Celsius)
At 40 degrees Celsius and above, degradation becomes rapid. This temperature is easily reached inside a closed car on a warm day, in direct sunlight, or near heat sources. Stability studies show that semaglutide at 40 degrees Celsius degrades significantly faster than at 25 degrees, with measurable potency loss occurring within hours rather than days.
If your semaglutide reached this temperature for any extended period, discarding it is the safest course of action.

Branded vs compounded semaglutide: different storage rules
One of the most common mistakes people make is assuming all semaglutide products follow the same storage rules. They do not. The differences are significant and can mean the difference between effective medication and wasted doses.
Branded semaglutide (Ozempic, Wegovy, Rybelsus)
Branded products benefit from Novo Nordisk proprietary formulation technology. The company invests millions in stability testing, optimizing buffer systems, preservatives, and delivery mechanisms to maximize shelf life under various conditions.
Ozempic pens:
Unopened: refrigerate at 36 to 46 degrees Fahrenheit until expiration date
In use: room temperature (up to 86 degrees Fahrenheit) or refrigerator for 56 days
Never freeze
Never expose above 86 degrees Fahrenheit
Protect from direct light
Wegovy pens:
Unopened: refrigerate at 36 to 46 degrees Fahrenheit
Can tolerate room temperature (46 to 86 degrees Fahrenheit) for up to 28 days before use
After first use: use within 28 days
Never freeze
The longer room-temperature tolerance of branded products comes from their proprietary formulation. The excipients, buffers, and preservative systems are specifically designed to maintain peptide stability outside the refrigerator. This is why branded products cost more but offer more flexibility in storage and travel situations.
Compounded semaglutide
Compounded semaglutide from 503A and 503B pharmacies follows different rules. Understanding these differences matters enormously for anyone using compounded formulations.
Storage requirements:
Refrigerate at 36 to 46 degrees Fahrenheit at all times
Beyond-use date (BUD): typically 28 to 30 days after first puncture
Some pharmacies with rigorous stability testing: up to 120 days refrigerated from compounding date
Room temperature tolerance: limited and pharmacy-specific
Never freeze
The key difference is the beyond-use date system. The FDA assigns expiration dates to approved medications based on extensive stability testing. Compounding pharmacies assign BUDs based on USP (United States Pharmacopeia) standards, which are generally more conservative.
This means compounded semaglutide expires approximately 2 to 4 times faster than branded Ozempic after first use. It also means compounded formulations have less room for temperature error. A compounded vial left out on the counter for several hours loses a larger proportion of its remaining usable life than an Ozempic pen in the same situation.
If you are using compounded semaglutide, pay extra attention to your storage routine. Keep the vial in the refrigerator immediately after each injection. And understand that the shelf life in the fridge is already shorter than branded alternatives.
Lyophilized (freeze-dried) vs liquid semaglutide
Some compounded semaglutide comes in lyophilized (freeze-dried) form that requires reconstitution before use. This form has a major advantage for heat resistance.
Lyophilized peptides are dramatically more stable at elevated temperatures than liquid formulations. Without water present, the chemical reactions that drive degradation, hydrolysis, oxidation, and aggregation, proceed much more slowly. A lyophilized vial that experiences a temperature excursion during shipping may be far less damaged than a pre-mixed liquid vial exposed to the same conditions.
However, once you reconstitute the peptide with bacteriostatic water, the clock starts ticking. The liquid solution becomes just as vulnerable to heat as any other liquid semaglutide product. Store it in the refrigerator immediately after mixing. You can learn more about the differences in our guide to lyophilized vs liquid peptides.
Common scenarios: what happens in real life
Theory is useful. But most people searching for information about warm semaglutide are dealing with a specific situation right now. Here are the most common scenarios, what actually happened to your medication, and what to do about each one.
Scenario 1: left on the counter overnight
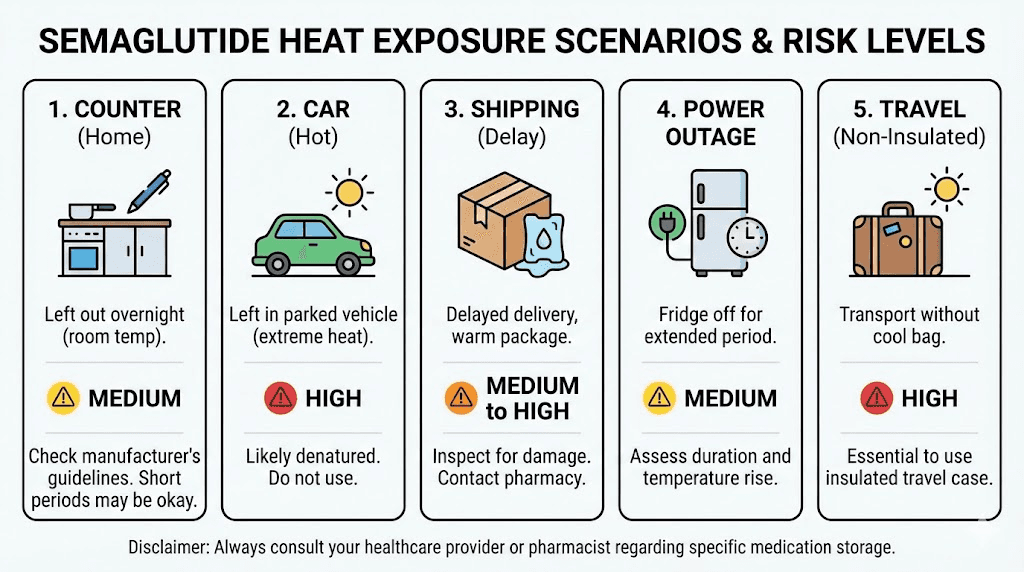
You injected your dose before bed and forgot to put the vial back in the refrigerator. You wake up and find it sitting on the kitchen counter. Eight hours at room temperature.
What happened: If your home stays below 77 degrees Fahrenheit (25 degrees Celsius) overnight, very little degradation occurred. Room temperature exposure for 8 to 12 hours within this range is unlikely to cause clinically significant potency loss for branded products, which are tested to remain stable at room temperature for weeks.
For branded Ozempic/Wegovy: Put it back in the refrigerator. Note the date. This counts toward the 56-day (Ozempic) or 28-day (Wegovy) room temperature clock. If the pen was already in use and within that window, it is almost certainly fine.
For compounded semaglutide: Return it to the refrigerator immediately. The exposure is more concerning because compounded formulations have less room-temperature stability testing. Contact your compounding pharmacy for guidance specific to your formulation. Most pharmacies will tell you that a single overnight exposure at normal room temperature is acceptable, but repeated incidents compound the degradation.
If you are concerned about whether your semaglutide is still working effectively, monitor your appetite suppression and weight trends over the next few days.
Scenario 2: left in a hot car
You had your semaglutide in a bag and left it in the car. Depending on the outdoor temperature and how long it sat there, this is potentially serious.
Car interiors can exceed 120 degrees Fahrenheit (49 degrees Celsius) on a warm day, even with windows cracked. A car parked in direct sunlight with an outdoor temperature of 80 degrees can reach 109 degrees Fahrenheit within 20 minutes and climb higher from there.
What happened: At these temperatures, semaglutide degrades rapidly. The exact damage depends on duration and peak temperature, but any exposure above 104 degrees Fahrenheit for more than 30 minutes warrants serious concern.
What to do: If the exposure was brief (under 15 minutes) and the car was not in direct sunlight, the medication may be acceptable. If the exposure was longer, or the car interior was clearly hot to the touch, discard the medication. The cost of a replacement vial is less than the cost of weeks of ineffective treatment.
This scenario reinforces why proper travel storage for semaglutide is so important. Insulated cases with gel packs prevent this exact situation.
Scenario 3: arrived warm from shipping
Your semaglutide was delivered, but the ice packs in the box are melted and the vial or pen feels warm.
This is one of the most common and most frustrating scenarios. Shipping temperature excursions are unpredictable. The package may have sat on a hot delivery truck, on a sun-baked porch, or in a warm distribution center.
What happened: Without knowing the exact temperature profile during transit, it is impossible to determine precisely how much degradation occurred. Some compounded semaglutide formulations can tolerate temperatures up to 95 degrees Fahrenheit for up to 4 days and temperatures up to 77 degrees Fahrenheit for 15 days without impacting efficacy.
What to do: Contact the pharmacy or supplier immediately. Describe the condition of the ice packs and the warmth of the medication. Many pharmacies and compounding providers will replace shipments that experienced temperature excursions. Some manufacturers and pharmacy benefit managers have specific policies for heat-damaged deliveries.
Refrigerate the medication immediately while you wait for a response. If the pharmacy confirms it is safe, use it. If they cannot confirm, request a replacement. Do not assume it is fine just because it looks normal.
Scenario 4: power outage
Your electricity went out and the refrigerator warmed up. How long do you have?
A closed refrigerator maintains safe temperatures for approximately 4 hours during a power outage. If you did not open the door frequently, your semaglutide stayed in the safe zone for most of that time. Even after the refrigerator warms to room temperature, branded products have the room-temperature tolerance windows mentioned above.
What to do: Note the approximate duration of the outage. If it was under 4 hours, your medication is almost certainly fine. If it was longer, check the ambient temperature inside the refrigerator (if you have a thermometer) and apply the temperature threshold guidelines above. For extended outages, consider transferring your semaglutide to a cooler with ice packs.
Scenario 5: vacation or travel mishap
You brought your semaglutide on vacation but the hotel refrigerator was not cold enough, or you forgot to put it away after your weekly injection.
Travel creates the highest risk for temperature excursions because your normal storage routine is disrupted. Every change in environment, from airport security to hotel rooms to beach days, introduces a new opportunity for the medication to get warm.
Our complete guide to traveling with semaglutide covers this in detail. The short version: always use an insulated travel case, always carry it in your hand luggage, and always verify hotel refrigerator temperatures with a thermometer.
How to tell if your semaglutide has been compromised
The honest answer is that you often cannot tell visually. But there are both physical and clinical indicators that suggest degradation has occurred.
Physical signs to check
Cloudiness or haziness. Fresh semaglutide solution should be clear and colorless. Any cloudiness suggests protein aggregation has occurred. This is an advanced sign of degradation, meaning significant potency has already been lost.
Particles or floaters. Visible particles in the solution indicate that peptide molecules have aggregated into clumps large enough to see. This medication should be discarded immediately. Injecting particulate matter can cause injection site reactions and delivers no therapeutic benefit.
Color changes. Semaglutide should be colorless. Any yellowing, browning, or other discoloration suggests chemical degradation. Discard immediately.
Unusual viscosity. If the solution feels thicker or thinner than normal when drawing into a syringe, the formulation may have been altered by heat exposure.
However, the absence of these signs does not guarantee potency. Semaglutide can lose substantial therapeutic activity while remaining visually indistinguishable from fresh medication. This is why tracking your storage conditions matters more than visual inspection.
Clinical signs of reduced potency
These signs develop over days to weeks of using compromised medication.
Reduced appetite suppression. If your semaglutide has been reliably controlling your appetite and suddenly stops working as well at the same dose, heat damage to the medication is a possible explanation. Before assuming the medication has stopped working, consider whether a storage incident occurred.
Increased hunger between doses. Semaglutide works by slowing gastric emptying and signaling satiety to the brain. If you notice hunger returning sooner than usual after your weekly injection, the dose you received may have contained less active peptide than expected. Your appetite suppression timeline should remain consistent if the medication is properly stored.
Weight loss stalling. Unexpected weight plateaus can have many causes, including metabolic adaptation and dietary changes. But if you have ruled out other factors and suspect a storage issue, compromised medication may be contributing. Check our guide on why you are not losing weight on semaglutide to evaluate all possible causes.
Blood glucose changes. For people using semaglutide for type 2 diabetes management, unexplained rises in blood glucose despite consistent medication use may indicate reduced potency from heat damage.
Increased fatigue and other side effect changes. Some people notice that side effects like GI symptoms decrease when medication potency drops, because there is less active compound producing those effects. Paradoxically, a sudden improvement in side effects without a dose change could indicate degraded medication.
What to do if your semaglutide got warm: the action plan
If you discover that your semaglutide has been exposed to elevated temperatures, follow this step-by-step assessment.
Step 1: assess the exposure
Determine three things as accurately as possible.
How warm did it get? Was it room temperature (around 72 degrees Fahrenheit), a warm room (80 to 86 degrees Fahrenheit), a hot car (potentially 100 degrees or more), or direct sunlight?
How long was it exposed? Minutes, hours, or days? The longer the exposure, the more degradation has occurred. A 30-minute exposure at 90 degrees is very different from a 12-hour exposure at the same temperature.
What formulation is it? Branded Ozempic has more room-temperature tolerance than compounded semaglutide. Know what you have.
Step 2: apply the temperature threshold guidelines
Use the thresholds outlined earlier in this guide.
Below 86 degrees Fahrenheit for a short time: Likely fine. Return to refrigerator. Note the date and duration.
Below 86 degrees Fahrenheit for extended time: Probably fine for branded products within their room-temperature windows. More concerning for compounded. Contact your pharmacy for guidance.
Above 86 degrees Fahrenheit: Evaluate carefully. Brief exposure (under 30 minutes) at moderate temperatures may be acceptable. Longer exposure warrants replacement.
Above 104 degrees Fahrenheit: Discard. The degradation risk is too high to justify continued use.
Step 3: inspect the medication
Check for the physical signs described above: cloudiness, particles, color changes, or unusual viscosity. Remember that passing visual inspection does not guarantee potency, but failing it means immediate discard.
Step 4: contact your provider
If you are uncertain, call your pharmacy, healthcare provider, or the manufacturer. Provide specific details about the exposure duration and estimated temperature. They can advise based on the specific formulation you are using.
Many providers and pharmacies have replacement policies for heat-damaged medication. Compounding pharmacies like Empower and Olympia often have customer service teams experienced with these situations.
Step 5: monitor your response
If you decide to continue using the medication after a mild temperature excursion, pay close attention to your clinical response over the next 1 to 2 weeks. Track your appetite levels, weight changes, and any shifts in side effects. If you notice reduced efficacy, the medication was likely compromised and should be replaced.
It is important to understand that heat-damaged semaglutide does not typically become toxic or dangerous. The primary risk is therapeutic failure, not adverse reactions. You will not get sick from injecting degraded semaglutide. You simply will not get the therapeutic benefits you expected.
Proper semaglutide storage from day one
Prevention is cheaper than replacement. Setting up proper storage habits from the beginning protects your investment and ensures every dose works as intended.
Refrigerator placement matters
Not all spots in your refrigerator maintain the same temperature. The ideal range is 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius).
Best locations: The middle shelf toward the back of the refrigerator maintains the most consistent temperature. This area is less affected by door openings and stays within the optimal range.
Worst locations: The door shelves experience the largest temperature swings every time you open the refrigerator. The back wall of the refrigerator can sometimes drop below freezing, especially in older models, which brings its own set of problems (more on freezing below). The crisper drawers can also run colder than expected.
A dedicated spot. Designate a specific location for your semaglutide and always return it there. Consistency in placement reduces the chance of leaving it out accidentally. Some people keep their vial in a small labeled container to make it visually distinct from food items.
Temperature monitoring
For researchers and patients serious about medication integrity, a refrigerator thermometer provides peace of mind. Digital versions with alarm features alert you if temperatures drift outside the safe range. This is especially valuable during power outages, when you might not realize the refrigerator has warmed up.
You do not need expensive laboratory equipment. A basic digital thermometer with min/max recording costs under $15 and sits right next to your semaglutide. Check it weekly.
The injection routine
Develop a consistent routine that minimizes time outside the refrigerator.
Remove the vial or pen from the refrigerator
Prepare your injection supplies
Draw the dose (use your semaglutide dosage calculator for accurate measurement)
Administer the injection following proper GLP-1 injection technique
Return the vial or pen to the refrigerator immediately
The entire process should take 5 to 10 minutes. Do not leave the vial out while you clean up, have a conversation, or get distracted. The best time to take your semaglutide is whenever you can consistently follow this routine without interruption.
Reconstituted semaglutide: extra care required
If you use lyophilized semaglutide that requires reconstitution, the storage requirements become even more critical after mixing. The dry powder is relatively stable. The liquid solution is not.
After reconstituting with the correct amount of bacteriostatic water, refrigerate immediately. The beyond-use date for reconstituted peptides is typically 28 to 30 days, sometimes shorter depending on the specific formulation. Never leave reconstituted semaglutide at room temperature for more than the few minutes required for injection preparation.
For detailed mixing instructions by vial size, see our 5mg vial dosage chart or 10mg dosage chart.
Traveling with semaglutide: keeping it cool on the go
Travel presents the biggest challenge for semaglutide storage. Your normal refrigerator routine breaks down completely when you are moving between airports, hotels, and destinations. Yet maintaining the cold chain is just as important away from home as it is in your kitchen.
Essential travel gear
Insulated medication travel case. Medical-grade insulated cases designed for injectable medications are the minimum requirement. These cases use phase-change gel packs that maintain temperatures in the 36 to 46 degree range for 12 to 48 hours depending on the quality of the case and ambient conditions.
Gel packs (not ice). Use gel packs designed for medication transport, not regular ice. Ice melts into water that can damage packaging and creates temperature inconsistency. Pre-freeze gel packs according to manufacturer directions. Wrap the semaglutide in a cloth barrier so the gel pack does not make direct contact, which could cause localized freezing.
Portable thermometer. A small digital thermometer inside the travel case lets you verify temperatures at any point during your journey.
Flying with semaglutide
Always carry in hand luggage. Never check semaglutide in your luggage. Cargo holds can experience extreme temperature variations, from below freezing at altitude to extremely warm on the tarmac. Hand luggage stays in the climate-controlled cabin.
TSA allows injectable medications through security with proper documentation. Carry the original pharmacy label or a letter from your prescriber. For a complete walkthrough, see our semaglutide travel guide.
Hotel room storage
Hotel mini-bars and mini-fridges are unreliable for medication storage. They often run at 45 to 50 degrees Fahrenheit, which is within the acceptable range but not optimal. Some run even warmer.
If possible, request a room with a full-size refrigerator. If that is not available, verify the mini-fridge temperature with your thermometer before trusting it with your medication. As a backup, the hotel front desk can often store medication in their kitchen refrigerator, which maintains proper food-safety temperatures.
Road trips and summer travel
The car is the single biggest risk for heat exposure during travel. Never leave semaglutide in a parked car, even for a few minutes. Car interiors can reach dangerous temperatures within 10 to 15 minutes on a warm day.
Keep your insulated case with you at all times. If you stop for a meal or activities, bring the case inside. If you are traveling long distances by car, refresh the gel packs at regular intervals using hotel ice machines or convenience store ice.
Freezing: the other temperature extreme
While this guide focuses primarily on heat exposure, freezing is equally destructive to semaglutide and deserves attention. Many people who carefully avoid heat exposure do not realize that cold can be just as damaging.
What happens when semaglutide freezes
Freezing causes ice crystals to form within the solution. These crystals physically tear through the peptide structure, causing denaturation at a mechanical level rather than a thermal one. The ice crystals act like microscopic blades, cutting through hydrogen bonds and disrupting the three-dimensional configuration of the molecule.
Once frozen, semaglutide is permanently damaged. Even if it thaws and looks normal, the structural damage has occurred. The peptide chains have been mechanically disrupted and cannot reassemble correctly. Manufacturer labeling for all semaglutide products explicitly warns against freezing and recommends immediate discard of any frozen medication.
How freezing happens accidentally
Refrigerator cold spots. The back wall of many refrigerators can drop below 32 degrees Fahrenheit, especially near the cooling element. Placing semaglutide directly against the back wall risks freezing. Use a thermometer to map your refrigerator temperatures and identify safe zones.
Shipping in winter. Just as summer shipping risks heat exposure, winter shipping in cold climates risks freezing. If your compounded semaglutide arrives with frozen gel packs touching the vial directly, the medication may have experienced freezing temperatures during transit.
Car storage in winter. Overnight temperatures in many regions drop below freezing. Semaglutide left in a car overnight during winter is likely frozen and should be discarded.
Improper travel cooling. Placing semaglutide directly on an ice pack or frozen gel without a barrier cloth can cause localized freezing of the solution closest to the cold source, even if the overall case temperature is acceptable.
The freeze-thaw rule
The rule is absolute. If your semaglutide has frozen at any point, discard it. Do not thaw and use it. Do not assume it is fine because it looks clear after thawing. The damage is invisible but real. Contact your pharmacy or insurance for a replacement.
This applies equally to all formulations, branded Ozempic, Wegovy, and every variety of compounded semaglutide.
Semaglutide stability by formulation type
Different semaglutide formulations handle temperature stress differently. Understanding your specific formulation helps you make better decisions about storage and temperature excursion management.
Semaglutide acetate vs semaglutide sodium
Compounding pharmacies produce semaglutide in different salt forms. Semaglutide acetate is the most common compounded form, while the branded products use proprietary formulations. The salt form can affect stability characteristics, particularly at elevated temperatures.
The buffer system in which the semaglutide is dissolved also plays a role. Different pH levels affect thermal degradation rates, and compounding pharmacies may use different buffer systems than the branded manufacturer. This is one reason why compounded semaglutide storage rules can vary between pharmacies.
Semaglutide with additives
Many compounded semaglutide formulations include additives like vitamin B12, methylcobalamin, or glycine. These additives can interact with the thermal stability profile of the formulation.
Glycine, for example, is sometimes added as a stabilizer that may actually improve the thermal stability of the peptide in solution. B12 and methylcobalamin additions change the chemical environment of the solution but their impact on semaglutide thermal stability has not been extensively studied in the compounding context.
If you are using a multi-ingredient compounded formulation, follow the storage guidelines from your specific pharmacy rather than general semaglutide storage advice. The pharmacy should provide storage instructions based on stability testing of their specific formulation, not just the semaglutide component alone.
Oral semaglutide formulations
Oral semaglutide drops and sublingual semaglutide have different stability profiles than injectable formulations. Oral formulations contain absorption enhancers and protective compounds that alter the overall stability of the product.
Rybelsus (branded oral semaglutide) is actually stored at room temperature as its standard condition, demonstrating that the tablet formulation provides significant thermal protection to the active ingredient. However, compounded oral and sublingual formulations may not share this stability advantage.
The real cost of heat-damaged semaglutide
Beyond the direct cost of replacing a vial, heat-damaged semaglutide creates hidden costs that add up over time.
Direct replacement costs
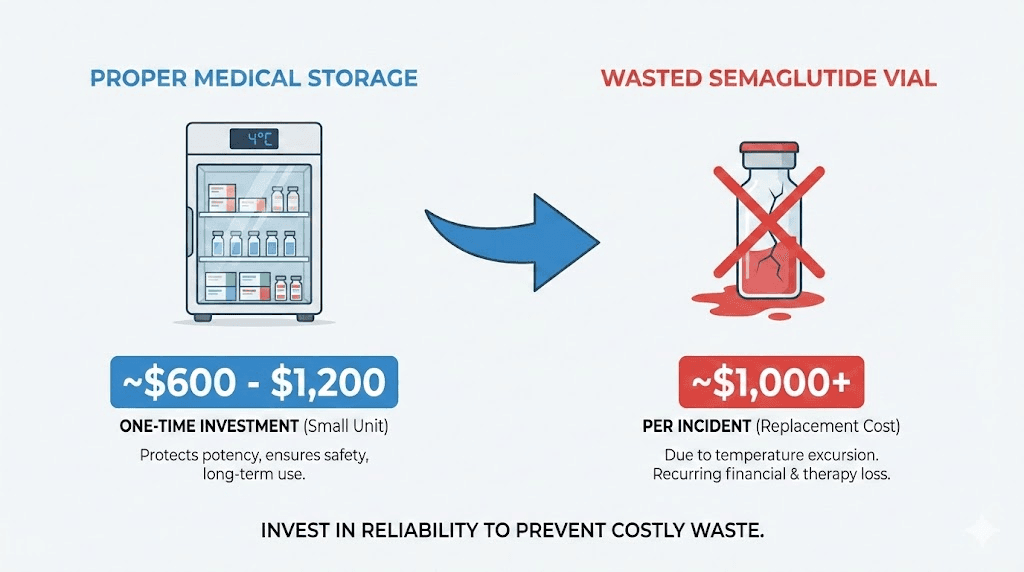
A single vial of compounded semaglutide represents a significant investment. Branded Ozempic and Wegovy are even more expensive. Every vial discarded due to heat damage is money lost.
If you are on a tight budget, which many people are given the cost of GLP-1 medications, a single storage mistake can set you back financially. An insulated storage case and a refrigerator thermometer together cost less than a single replacement vial.
Treatment interruption costs
When you discard a vial, you may face a gap in treatment while waiting for a replacement. Depending on your pharmacy and insurance, this gap could be days or even weeks.
During treatment gaps, the effects of stopping semaglutide can include appetite returning, weight regain beginning, and blood glucose levels rising. Restarting treatment after a gap may require dose re-titration, adding more time before you return to your therapeutic dose.
Hidden potency loss costs
Perhaps the most insidious cost is using partially degraded medication without realizing it. You are paying full price for a dose but receiving less active compound than expected. Your results slow down. You blame the medication, your body, or your compliance. You might escalate your dose unnecessarily, or consider switching to a different medication when the real problem was storage all along.
Proper storage from day one eliminates this entire category of problems. It is the cheapest, simplest way to ensure you get full value from every injection.
Special considerations for different situations
Semaglutide during summer months
Summer creates persistent storage challenges. Higher ambient temperatures mean the refrigerator works harder. Power outages from storms are more common. And the temptation to leave medication in the car or bag during warm-weather activities increases.
During summer months, be extra vigilant about your storage routine. Double-check that your refrigerator is maintaining proper temperatures. Keep backup gel packs in the freezer for power outage emergencies. And never leave your semaglutide in any uncontrolled environment, even briefly.
If you are managing your weight on semaglutide during summer, consistency matters. The dietary plan you follow and the foods you eat during warm weather affect your results alongside medication potency.
Multiple medication management
If you are using semaglutide alongside other medications that require refrigeration, managing your cold chain becomes more complex but also more standardized. Designate a specific shelf or container for all refrigerated medications. Use the same temperature monitoring for all of them.
Some people combine semaglutide with other peptides or supplements. If you are taking phentermine alongside semaglutide or using additional peptide protocols, each product may have its own storage requirements. Keep them organized but separate to avoid confusion.
Pregnancy and semaglutide storage
While semaglutide is not recommended during pregnancy, some people discover pregnancy while using the medication. In this situation, proper storage becomes even more critical during the wind-down period when you are finishing existing doses under medical supervision. See our article on GLP-1 medications and breastfeeding for related guidance.
How semaglutide compares to other GLP-1 medications for heat sensitivity
Semaglutide is not the only GLP-1 receptor agonist affected by heat. Understanding how different medications in this class compare helps you manage storage expectations, especially if you are considering switching medications or using multiple products.
Semaglutide vs tirzepatide storage
Tirzepatide (Zepbound, Mounjaro) follows similar storage principles but with some differences. Like semaglutide, tirzepatide requires refrigeration before first use and has a limited room-temperature window after opening.
Both medications are peptide-based and vulnerable to the same degradation mechanisms. If you are managing tirzepatide refrigeration, the principles in this guide apply equally. The main difference is in the specific room-temperature tolerance windows, which vary by product. For detailed comparison, see our semaglutide vs tirzepatide comparison.
Compounded tirzepatide faces the same shortened shelf life challenges as compounded semaglutide. If you want to know how long tirzepatide lasts in the fridge or how long it can stay out, the same conservative approach applies.
Newer GLP-1 formulations
Emerging medications like retatrutide and other next-generation GLP-1 receptor agonists will likely face similar storage challenges. As a triple agonist, retatrutide may have different stability characteristics, but all peptide-based medications share fundamental vulnerability to heat denaturation.
The GLP-1 patch delivery systems being developed may eventually offer better temperature stability because the peptide is embedded in a solid matrix rather than dissolved in solution. But for now, injectable and oral formulations dominate the market, and proper cold-chain management remains essential.
Long-term storage strategies
Batch management for multi-vial users
If you purchase multiple vials of compounded semaglutide at once, proper long-term storage becomes important. Keep unopened vials in the coldest stable area of your refrigerator (but not against the back wall where freezing can occur). Use vials in order of their beyond-use dates, oldest first.
Track each vial with a simple label noting the compounding date, the beyond-use date, and the date you first punctured the seal. This simple tracking prevents accidentally using expired medication and helps you identify if storage issues affected a specific vial.
Backup storage options
For maximum security, consider having a backup cooling option available.
A small, dedicated mini-fridge set to the correct temperature range provides a backup if your main refrigerator fails. This is especially valuable if you have an expensive multi-month supply of medication on hand.
Pre-frozen gel packs in your freezer serve as emergency cooling if the power goes out. Place them around the semaglutide vials in an insulated bag or cooler to maintain temperatures for hours.
A battery-powered temperature alarm for your refrigerator provides early warning of temperature drift before it reaches dangerous levels.
Record keeping
Maintaining a simple log of your semaglutide storage can help troubleshoot issues later. Note any temperature excursions, power outages, travel dates, and when you open new vials. If you ever experience reduced efficacy and need to investigate whether storage was a factor, this log provides the data you need.
This level of tracking might seem excessive. But if you are investing hundreds of dollars per month in medication, a 30-second log entry after each injection is cheap insurance against waste.
For researchers serious about optimizing their peptide protocols, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, storage protocols, and a community of thousands who have navigated these exact questions. SeekPeptides members access detailed protocol guides, dosage calculators, and expert-reviewed storage recommendations that account for every formulation type.
Frequently asked questions
Can I use semaglutide that was left out overnight?
It depends on the temperature and formulation. Branded Ozempic that was left at room temperature (below 86 degrees Fahrenheit) overnight is likely fine, as it is tested for up to 56 days at room temperature. Compounded semaglutide is more sensitive, so contact your pharmacy for guidance specific to your formulation. Return it to the refrigerator immediately and note the date.
How can I tell if heat damaged my semaglutide?
Unfortunately, you often cannot tell visually. Check for cloudiness, particles, or color changes, but these only appear with severe degradation. The most reliable indicator is monitoring your clinical response. If your appetite suppression weakens or weight loss stalls unexpectedly, compromised medication may be the cause.
Will my semaglutide work if it got warm during shipping?
Contact your pharmacy immediately with details about the shipping conditions. Some formulations tolerate temperatures up to 95 degrees Fahrenheit for up to 4 days without significant potency loss. Many pharmacies and compounding providers will replace shipments that experienced temperature excursions. Refrigerate the medication while awaiting their response.
Is it safe to inject semaglutide that got warm?
Heat-damaged semaglutide does not typically become toxic or dangerous. The main risk is reduced therapeutic effect, not adverse reactions. However, if you see particles, cloudiness, or discoloration, do not inject it. These visible changes indicate significant degradation that could cause injection site reactions.
Can I freeze semaglutide to make it last longer?
No. Never freeze semaglutide. Freezing causes ice crystal formation that permanently destroys the peptide structure. Even if it looks normal after thawing, the medication is damaged and should be discarded. This applies to all formulations, including branded and compounded versions.
How long can Ozempic sit at room temperature?
An in-use Ozempic pen can be stored at room temperature up to 86 degrees Fahrenheit (30 degrees Celsius) for up to 56 days. After 56 days, discard the pen regardless of remaining medication. Unopened pens should remain refrigerated until first use. For comprehensive shelf life information, see our complete storage guide.
Does compounded semaglutide need to stay refrigerated at all times?
Most compounding pharmacies recommend continuous refrigeration for their formulations. Brief removal for injection preparation (5 to 10 minutes) is fine. Extended room temperature exposure should be avoided. For complete guidance, read our article on compounded semaglutide refrigeration requirements.
What temperature is too hot for semaglutide?
Temperatures above 86 degrees Fahrenheit (30 degrees Celsius) exceed the manufacturer recommended storage range. Brief exposure (under 30 minutes) at moderate temperatures above this threshold may not cause significant damage, but prolonged exposure above 86 degrees or any exposure above 104 degrees Fahrenheit (40 degrees Celsius) warrants discarding the medication.
Can I take my semaglutide to the beach or pool?
Only with proper temperature protection. Use a medical-grade insulated case with frozen gel packs. Never leave semaglutide in direct sunlight, in a hot bag, or unattended in any warm environment. Temperatures at beaches and pools can easily exceed 100 degrees in direct sun. Our travel guide covers portable storage solutions in detail.
My semaglutide pen feels warm but was in the fridge. Is it okay?
Semaglutide always feels slightly cool when removed from the refrigerator but warms to the touch quickly because the vial or pen has low thermal mass. Feeling "warm" after 5 to 10 minutes outside the fridge is normal and does not indicate a problem. However, if the pen feels warm immediately when you take it from the refrigerator, check your fridge temperature, it may be running too warm.
External resources
PubMed: Influence of Buffering Capacity, pH, and Temperature on Semaglutide Stability
PMC: Factors Affecting Physical Stability of Peptide Therapeutics
In case I do not see you, good afternoon, good evening, and good night. May your vials stay cold, your peptides stay potent, and your results stay consistent.