Feb 8, 2026

The math behind mixing 10mg of semaglutide
You have a 10mg vial of semaglutide sitting in front of you. A bottle of bacteriostatic water beside it. And a question that has stopped thousands of researchers in their tracks. How much water goes in? Get it wrong and you are either overdosing or underdosing every single injection for weeks. Get it right and every dose becomes predictable, accurate, and effective.
This is not a small detail. It is the foundation of your entire protocol.
The amount of bacteriostatic water you add determines the concentration of your solution, which determines how many units you draw for each dose, which determines whether your results match the research or fall flat. One miscalculation here cascades through every injection. And with semaglutide research protocols running 16 to 20 weeks, a reconstitution error on day one means weeks of inaccurate dosing before you even realize something is off. The semaglutide dosage calculator on SeekPeptides can help verify your math, but understanding the principles behind it matters just as much as getting the number right.
This guide breaks down every reconstitution scenario for a 10mg semaglutide vial, with exact water volumes, concentration charts, unit conversions for insulin syringes, and step-by-step mixing instructions that protect peptide integrity. Whether you are starting your first vial or switching from a 5mg setup, the calculations here will make every dose precise.
Why bacteriostatic water and not sterile water
Before measuring anything, you need the right diluent. This choice affects how long your reconstituted semaglutide stays viable, how many doses you can safely draw from one vial, and whether bacterial contamination becomes a risk. The wrong water type can ruin an entire vial before you finish using it.
Bacteriostatic water contains 0.9% benzyl alcohol as a preservative. This is critical. Every time you insert a needle through the vial stopper to draw a dose, you introduce a potential pathway for bacteria. The benzyl alcohol in bacteriostatic water inhibits bacterial growth, keeping the solution safe for multi-dose use over approximately 28 days. Without it, you would need to use the entire vial in a single session or risk contamination.
Sterile water for injection lacks any preservative. It is pure, sterilized water and nothing else. Fine for single-use applications. Dangerous for multi-dose vials. A 10mg semaglutide vial contains enough peptide for weeks of dosing at standard escalation protocols. Using sterile water means every needle puncture after the first one increases your contamination risk with zero bacterial defense in the solution.
The practical difference is simple. Bacteriostatic water lets you reconstitute once and draw doses for up to 28 days. Sterile water would require either using all 10mg immediately or risking every subsequent dose. For a peptide reconstitution that needs to last, bacteriostatic water is the only rational choice.
What about sodium chloride solutions
Some protocols mention normal saline (0.9% sodium chloride) as a reconstitution option. While technically possible, bacteriostatic water remains the standard for semaglutide. The peptide dissolves readily in bacteriostatic water without needing the osmolality adjustment that saline provides. Unless specifically directed by a prescribing clinician, stick with bacteriostatic water for consistency and simplicity. The bacteriostatic water guide covers everything you need to know about selecting and using the right diluent for peptide mixing.
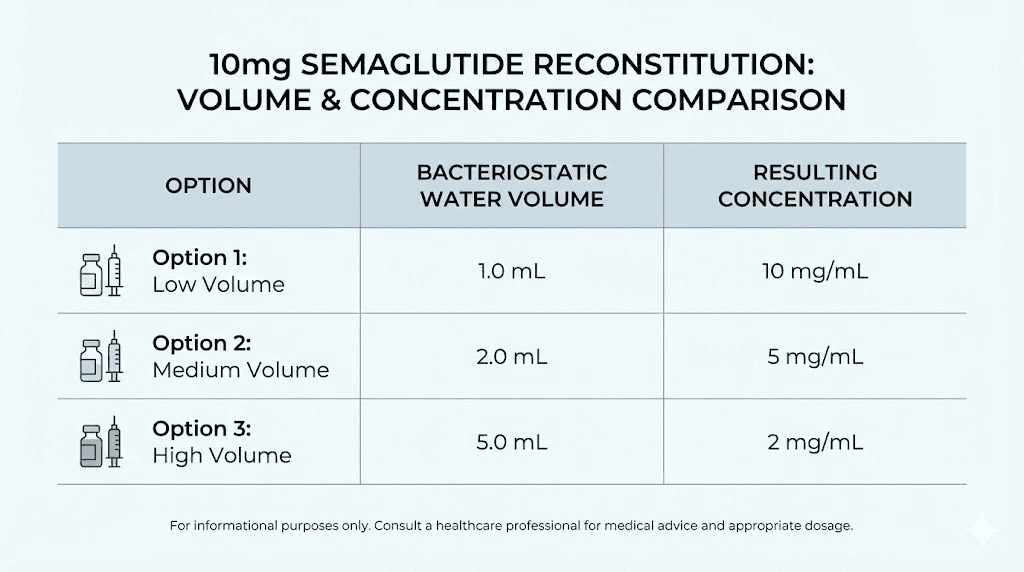
The three standard reconstitution volumes for 10mg semaglutide
There is no single "correct" amount of bacteriostatic water for a 10mg vial. The right volume depends on your target concentration, your syringe type, and your dosing protocol. Three volumes have become standard in research settings, each creating a different concentration that suits different needs.
Option 1: 2mL of bacteriostatic water (5mg/mL concentration)
Adding 2mL creates the highest practical concentration at 5mg per milliliter. This means each 0.1mL (10 units on a U-100 insulin syringe) contains 0.5mg of semaglutide. The advantage here is small injection volumes, which matters for subcutaneous comfort. The disadvantage is that small dosing errors become magnified. If you are targeting 0.25mg (the standard starting dose), you would draw just 5 units on an insulin syringe. That is a tiny amount where even a one-unit error represents a 20% dosing mistake.
This concentration works best for experienced researchers who are comfortable with precision syringe work and who prefer minimal injection volumes. It is not ideal for beginners.
Option 2: 3mL of bacteriostatic water (3.33mg/mL concentration)
This is the most commonly recommended volume. Adding 3mL to a 10mg vial gives you a concentration of approximately 3.33mg per milliliter. At this concentration, a 0.25mg dose equals about 7.5 units on a U-100 insulin syringe. A 0.5mg dose equals about 15 units. These volumes are much easier to measure accurately, reducing the chance of dosing errors while keeping injection volumes manageable.
The 3mL reconstitution has become the default recommendation for good reason. It strikes the optimal balance between concentration (keeping injection volumes reasonable) and accuracy (making dose measurement practical on standard syringes). For most researchers following a standard semaglutide protocol, this is the volume to use.
Option 3: 4mL of bacteriostatic water (2.5mg/mL concentration)
Adding 4mL creates a 2.5mg/mL solution. A 0.25mg dose becomes 10 units exactly, and a 0.5mg dose becomes 20 units. The math is clean and the measurements are straightforward. This volume is excellent for beginners or anyone who prioritizes dosing accuracy over injection volume. The tradeoff is larger injection volumes at higher doses. At the maintenance dose of 2.4mg, you would need to draw 96 units, which is nearly a full 1mL syringe. That is a significant subcutaneous injection.
Some researchers prefer this option during the initial low-dose escalation phase, then switch to a higher concentration for maintenance dosing.
Complete dosing chart for every concentration
Once you have chosen your reconstitution volume, you need to know exactly how many units to draw for each dose throughout your escalation protocol. The standard semaglutide dosing schedule follows a gradual escalation from 0.25mg to 2.4mg over 16 to 20 weeks. Here is exactly what that looks like at each concentration.
Dosing chart with 2mL water (5mg/mL)
At this concentration, every unit on a U-100 insulin syringe equals 0.05mg of semaglutide.
0.25mg dose: Draw to 5 units
0.5mg dose: Draw to 10 units
1.0mg dose: Draw to 20 units
1.7mg dose: Draw to 34 units
2.0mg dose: Draw to 40 units
2.4mg dose: Draw to 48 units
Total volume in vial: 2mL (200 units). At the starting dose of 0.25mg weekly, one vial provides 40 weekly doses, though reconstituted semaglutide should be used within 28 days for optimal potency.
Dosing chart with 3mL water (3.33mg/mL)
At this concentration, every unit equals approximately 0.033mg of semaglutide. This is the most popular option and works well with the peptide calculator for verification.
0.25mg dose: Draw to 7.5 units (round to 7 or 8)
0.5mg dose: Draw to 15 units
1.0mg dose: Draw to 30 units
1.7mg dose: Draw to 51 units
2.0mg dose: Draw to 60 units
2.4mg dose: Draw to 72 units
These unit measurements are easier to read on a standard insulin syringe, which is why the 3mL reconstitution has become the default recommendation across most research communities.
Dosing chart with 4mL water (2.5mg/mL)
At this concentration, every unit equals 0.025mg of semaglutide. The cleanest math of all three options.
0.25mg dose: Draw to 10 units
0.5mg dose: Draw to 20 units
1.0mg dose: Draw to 40 units
1.7mg dose: Draw to 68 units
2.0mg dose: Draw to 80 units
2.4mg dose: Draw to 96 units
Notice how each dose falls on a round number. No rounding needed. No approximation. If dosing accuracy is your top priority and you do not mind slightly larger injection volumes, the 4mL option eliminates guesswork entirely.
Which concentration should you choose
For most researchers, 3mL is the answer. It provides the best balance of accuracy and injection comfort. The unit measurements land in a range that is easy to read on standard syringes without creating unnecessarily large injection volumes at maintenance doses. Researchers using our peptide reconstitution calculator can verify their specific concentration and dosing calculations before mixing.
Choose 2mL if you have extensive experience with precision dosing and prefer minimal injection volumes. Choose 4mL if you are new to peptide injections and want the simplest possible math.
Step-by-step reconstitution process
Knowing the right volume is only half the equation. How you add the water matters just as much. Poor technique can denature the peptide, create foam that wastes product, or introduce contamination that ruins the entire vial. Follow these steps precisely.
Gather your supplies
Before opening anything, have everything ready on a clean surface. You need your 10mg semaglutide vial, a bottle or ampule of bacteriostatic water, an appropriately sized mixing syringe (a 3mL or 5mL syringe works well for drawing and transferring the water), alcohol swabs, and a clean work surface. The getting started with peptides guide covers workspace preparation in detail.
Wash your hands thoroughly. This seems obvious. It gets skipped constantly.
Prepare the vials
Remove the flip-off cap from both the semaglutide vial and the bacteriostatic water container. Swab the rubber stoppers on both with alcohol pads. Let the alcohol dry completely, about 30 seconds. Do not blow on them to speed drying. Your breath introduces bacteria.
If your semaglutide vial has been refrigerated (which it should be for proper peptide storage), let it sit at room temperature for about 10 minutes before reconstituting. Cold powder dissolves more slowly and can lead to incomplete dissolution if you rush the process.
Draw the bacteriostatic water
Using your mixing syringe, draw the exact amount of bacteriostatic water you have chosen. For the recommended 3mL reconstitution, draw exactly 3mL. Precision here sets the foundation for every subsequent dose. A syringe with 0.1mL graduations makes this easier to measure accurately than trying to use an insulin syringe with its smaller volume markings.
Remove air bubbles from the syringe by holding it needle-up and tapping gently, then pushing the plunger slightly to expel trapped air. Verify your water volume after removing bubbles, as expelled air can change the measured amount.
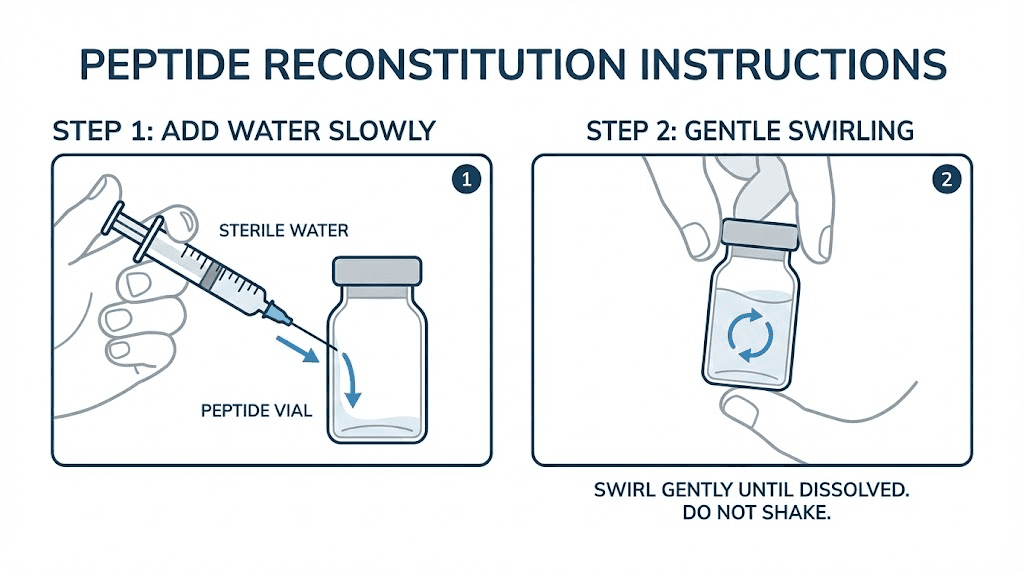
Add water to the semaglutide vial
This is where most reconstitution mistakes happen. Insert the needle through the rubber stopper of the semaglutide vial at a slight angle. Do not aim the needle directly at the lyophilized powder cake at the bottom.
Depress the plunger slowly. Very slowly.
Let the water run down the inside wall of the vial. This is critical. Shooting water directly onto the powder creates foam, wastes peptide that gets trapped in bubbles, and can damage the molecule through mechanical stress. The water should trickle down the glass wall and gently reach the powder from the side, not blast into it from above. Understanding proper peptide mixing technique prevents these common errors.
Take at least 30 seconds to add all the water. Patience here protects your investment.
Dissolve the peptide
Once all the water is in the vial, remove the syringe and set it aside. Now gently swirl the vial by rolling it between your palms. Do not shake it. Do not invert it violently. Do not vortex it. Semaglutide is a relatively stable peptide, but aggressive agitation creates shearing forces that can degrade any protein-based molecule.
Roll the vial gently for one to two minutes. The powder should dissolve completely, leaving a clear, colorless solution. If small particles remain, set the vial down and wait five minutes, then roll again. Some reconstitutions take a few minutes for complete dissolution, especially if the vial was cold.
If the solution appears cloudy, discolored, or contains particles that do not dissolve after 10 minutes of gentle rolling, something has gone wrong. A properly reconstituted semaglutide solution is always clear and colorless. Discard cloudy or discolored solutions.
How many doses does a 10mg vial provide
This depends entirely on your dosing protocol and which phase of escalation you are in. A standard semaglutide weight management protocol runs through five escalation phases before reaching maintenance dosing. Understanding how far your vial goes helps you plan purchases and reconstitution timing.
Escalation phase breakdown
The standard protocol starts low and increases every four weeks to minimize gastrointestinal side effects that rapid dose escalation can cause.
Weeks 1 through 4: 0.25mg per week (1mg total used over 4 weeks)
Weeks 5 through 8: 0.5mg per week (2mg total used over 4 weeks)
Weeks 9 through 12: 1.0mg per week (4mg total used over 4 weeks)
Weeks 13 through 16: 1.7mg per week (6.8mg total used over 4 weeks)
Week 17 onward: 2.4mg per week (maintenance dose)
During the first four weeks at 0.25mg weekly, you use only 1mg from your 10mg vial. Through week eight, you have used a cumulative 3mg. Through week twelve, 7mg total. This means a single 10mg vial can theoretically cover the first 12 to 13 weeks of a standard escalation protocol, using roughly 7mg through that period.
But here is the critical caveat. Reconstituted semaglutide should be used within 28 days of mixing. A single 10mg vial reconstituted all at once cannot safely last 12 weeks.
The practical solution
Many researchers address this by reconstituting only what they need for roughly four weeks of dosing. With a 10mg vial, you could reconstitute with a proportional amount of water to create a solution you will use entirely within the 28-day window. Or you can reconstitute the full 10mg and accept that doses drawn after 28 days may have reduced potency.
At the 0.25mg starting dose, four weeks requires only 1mg of peptide. That means 90% of your reconstituted 10mg vial sits in the fridge unused for most of its shelf life. This is where understanding how long compounded semaglutide lasts in the fridge becomes important. Some researchers report usable potency beyond 28 days when stored properly, though 28 days remains the standard safety recommendation.
The most practical approach for many protocols involves starting with a 5mg vial for the early escalation phases and switching to a 10mg vial once weekly doses are higher. This minimizes waste from the 28-day use window.
Understanding insulin syringe markings for semaglutide dosing
Insulin syringes are the standard tool for drawing and administering reconstituted semaglutide. But they were designed for insulin, not peptides, and this creates a translation challenge that confuses many researchers. Understanding the markings is essential for accurate dosing.
How insulin syringe units work
A standard U-100 insulin syringe is calibrated so that 100 units equals 1mL of liquid. The "units" marked on the syringe are volume measurements, not dose measurements. When used for insulin, 1 unit equals 1 international unit of insulin because insulin is formulated at 100 IU per mL. But when used for semaglutide, each "unit" simply represents 0.01mL of whatever solution you have in the syringe.
This distinction matters enormously. The amount of semaglutide in each "unit" depends entirely on your reconstitution concentration. Drawing 10 units from a 5mg/mL solution gives you 0.5mg. Drawing 10 units from a 2.5mg/mL solution gives you 0.25mg. Same syringe, same marking, completely different doses. The units to milligrams conversion depends on your specific reconstitution.
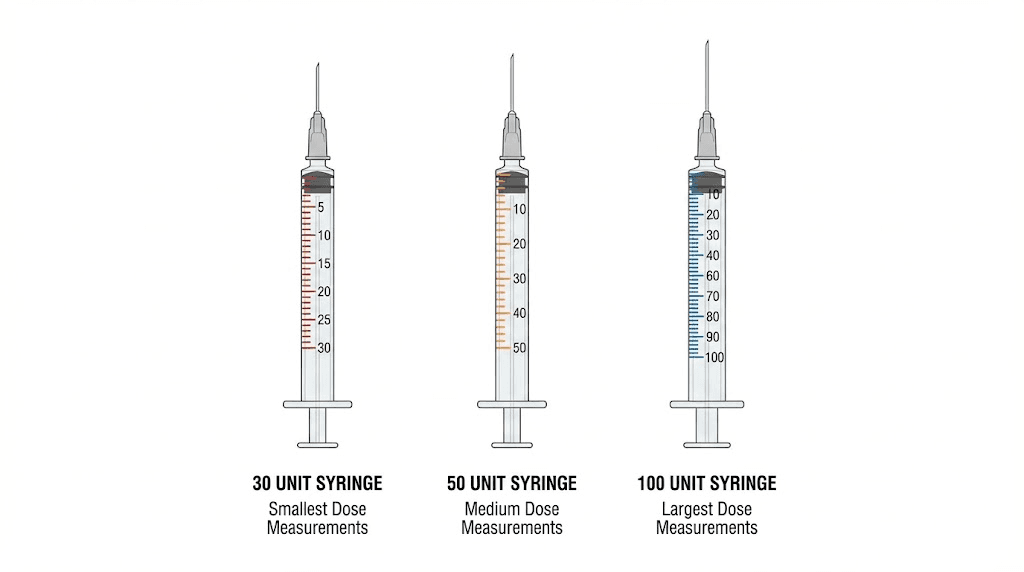
Choosing the right syringe size
Insulin syringes come in three common sizes: 0.3mL (30 units), 0.5mL (50 units), and 1.0mL (100 units). For semaglutide dosing, the best syringe depends on your dose and concentration.
During the early escalation phase at 0.25mg, you are drawing very small volumes regardless of concentration. A 0.3mL (30 unit) syringe provides the finest graduations and easiest reading for these small amounts. Each mark represents 0.5 units, making it much easier to hit precise targets like 7.5 units compared to reading the same mark on a larger syringe.
At maintenance doses of 2.0mg or 2.4mg, the volume increases significantly. With a 3.33mg/mL concentration, a 2.4mg dose requires 72 units, which needs a 1.0mL (100 unit) syringe. With a 2.5mg/mL concentration, that same dose requires 96 units. Still within the 100-unit syringe range, but just barely. Understanding how semaglutide units convert to milligrams eliminates confusion during dose escalation.
Consider starting with 30-unit syringes for the first eight weeks of escalation (0.25mg and 0.5mg doses), then switching to 100-unit syringes when doses increase beyond what the smaller syringes can accommodate.
The complete unit conversion formula
Rather than memorizing dosing charts, understanding the conversion formula lets you calculate any dose at any concentration. This is useful when switching between vial sizes, adjusting reconstitution volumes, or verifying pre-calculated charts.
The formula
Units to draw = (desired dose in mg / concentration in mg per mL) x 100
Let us work through examples at each standard concentration.
Example 1: You want 0.5mg from a 3.33mg/mL solution. Units = (0.5 / 3.33) x 100 = 15 units. Draw to the 15-unit mark on your insulin syringe.
Example 2: You want 1.0mg from a 5mg/mL solution. Units = (1.0 / 5.0) x 100 = 20 units. Draw to the 20-unit mark.
Example 3: You want 2.4mg from a 2.5mg/mL solution. Units = (2.4 / 2.5) x 100 = 96 units. Draw to the 96-unit mark.
The formula works for any concentration. If you reconstitute with an unusual volume, say 2.5mL for a 4mg/mL concentration, the formula still gives you the exact unit measurement. The peptide dosage calculation guide walks through more complex scenarios, including accounting for dead space in syringes and multi-peptide calculations.
Accounting for syringe dead space
Every syringe has a small amount of "dead space," the volume of liquid that remains in the needle hub and syringe tip after you push the plunger all the way down. For most insulin syringes, this is approximately 0.5 to 1 unit of volume. Over dozens of injections from a single vial, this dead space adds up.
With a 3.33mg/mL concentration, losing 1 unit of dead space per injection wastes about 0.033mg per dose. Over 12 weekly doses, that is roughly 0.4mg of lost peptide. Not catastrophic, but worth noting. Low dead space syringes are available and reduce this waste significantly. They cost slightly more but preserve more of your peptide over the life of the vial.
How 10mg reconstitution differs from 5mg
Many researchers start with 5mg vials and transition to 10mg as their protocol advances. The reconstitution principles are identical, but the practical details change in ways worth understanding.
Concentration differences at the same water volume
If you add 2mL to a 5mg vial, you get 2.5mg/mL. The same 2mL added to a 10mg vial gives you 5mg/mL, double the concentration. This means the same unit measurement on your syringe delivers twice the dose. Failing to account for this when switching vial sizes is one of the most common and dangerous reconstitution errors. Always recalculate your dosing when changing vial sizes, even if you use the same water volume.
The general bacteriostatic water guide covers the math for all common vial sizes. For semaglutide specifically, here is how the two vial sizes compare at each water volume:
2mL water + 5mg vial = 2.5mg/mL vs. 2mL water + 10mg vial = 5.0mg/mL
3mL water + 5mg vial = 1.67mg/mL vs. 3mL water + 10mg vial = 3.33mg/mL
4mL water + 5mg vial = 1.25mg/mL vs. 4mL water + 10mg vial = 2.5mg/mL
Notice the pattern. At every water volume, the 10mg vial produces exactly double the concentration of the 5mg vial. This makes mental math straightforward once you understand the relationship, but it also means dosing errors are doubled if you accidentally use the wrong chart.
When to use 10mg versus 5mg vials
A 5mg vial makes more sense during early dose escalation (0.25mg to 0.5mg weekly) because you will use less total peptide over the 28-day reconstitution window. A 10mg vial becomes more economical at higher doses (1.0mg and above) where weekly consumption increases significantly. Many researchers following the standard semaglutide protocol start with 5mg vials for the first two months and transition to 10mg vials when their weekly dose reaches 1mg or higher.
From a cost perspective, 10mg vials typically offer better value per milligram than two separate 5mg vials. The peptide cost calculator can help compare per-dose costs across different vial sizes and vendors.
Storage after reconstitution
Mixing your semaglutide correctly is only valuable if you store it properly afterward. Improper storage degrades the peptide, reducing its effectiveness with each passing day. The rules here are non-negotiable.
Temperature requirements
Reconstituted semaglutide must be refrigerated at 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit). This is the standard pharmaceutical cold chain range and matches what most household refrigerators maintain. Do not store it in the door compartment where temperature fluctuates every time the door opens. Place it on a middle shelf toward the back where temperatures remain most stable.
Do not freeze reconstituted semaglutide. Freezing creates ice crystals that can physically damage the peptide structure. If your refrigerator tends to freeze items at the back wall, place the vial slightly forward or use a small container to insulate it. Our complete guide to peptide refrigeration covers optimal placement and temperature monitoring in detail.
Light protection
Semaglutide degrades when exposed to light, particularly UV light. Store the vial in its original box if possible. If the box is discarded, wrap the vial in aluminum foil or place it in an opaque container. Even refrigerator lights, which turn on briefly each time you open the door, contribute to cumulative light exposure over 28 days.
The 28-day rule
Standard guidance recommends using reconstituted semaglutide within 28 days of mixing. After this point, both peptide degradation and bacterial contamination risk increase beyond acceptable thresholds, even with bacteriostatic water as the diluent. Label your vial with the reconstitution date immediately after mixing. Do not rely on memory. Understanding whether expired semaglutide is still usable helps you make informed decisions when vials approach their use-by date.
Some researchers report that properly stored reconstituted semaglutide maintains acceptable potency for 30 to 45 days. The research literature supports 28 days as the conservative recommendation with a reasonable safety margin. Going beyond this introduces increasing uncertainty about both potency and sterility.
Signs your reconstituted semaglutide has degraded
Watch for these indicators that something has gone wrong:
Color change: Fresh semaglutide solution is clear and colorless. Any yellow, brown, or hazy appearance indicates degradation. Discard immediately.
Particles: Floating particles, fibers, or cloudiness suggest either contamination or protein aggregation. Neither is acceptable. Discard.
Unusual odor: Reconstituted semaglutide should have no noticeable smell beyond a very faint odor from the benzyl alcohol in bacteriostatic water. Strong or unusual odors indicate bacterial growth.
Reduced effectiveness: If your typical dose seems less effective than previous weeks from the same vial, potency may have decreased. This is harder to quantify but worth noting.
The peptide expiration guide covers degradation timelines for multiple peptide types, including semaglutide stability data under various storage conditions.
Common reconstitution mistakes and how to avoid them
After analyzing hundreds of questions from researchers about semaglutide reconstitution, certain errors appear again and again. Most are preventable with awareness. All of them compromise your results.
Mistake 1: Shooting water directly onto the powder
This is the most common error. Researchers aim the needle at the powder cake and push the plunger quickly, blasting water directly into the lyophilized peptide. This creates foam, traps peptide in bubbles, and generates mechanical stress that can denature protein molecules.
The fix is simple. Angle the needle toward the glass wall. Let the water trickle down the side. Be patient. The extra 30 seconds this takes protects every dose you will draw from that vial.
Mistake 2: Shaking the vial to dissolve
Vigorous shaking creates shearing forces between liquid layers that can damage peptide bonds. It also generates foam that makes it impossible to verify whether the powder has fully dissolved. Proteins are more fragile than most people realize. Even stable peptides like semaglutide can degrade under aggressive mechanical agitation.
Roll the vial gently between your palms instead. If the powder does not dissolve within two minutes of rolling, set it down for five minutes and try again. Patience dissolves more peptides than force ever will.
Mistake 3: Using the wrong water volume
Some researchers add bacteriostatic water by estimation rather than precise measurement. "About 3mL" is not 3mL. If you add 2.5mL instead of 3mL, your concentration jumps from 3.33mg/mL to 4mg/mL. Every dose you calculate from that point forward is wrong by roughly 20%. Use a syringe with clear mL markings, not guesswork. The common peptide mistakes guide covers this and other errors that undermine research outcomes.
Mistake 4: Forgetting to sanitize
Skipping alcohol swabbing of vial stoppers introduces bacteria directly into your peptide solution. Even "clean-looking" surfaces harbor microorganisms. Swab every stopper, every time, with a fresh alcohol pad. Let it dry completely. This 30-second step prevents contamination that could make days of reconstituted peptide unusable.
Mistake 5: Mixing at the wrong temperature
Reconstituting an ice-cold vial straight from the refrigerator slows dissolution significantly. The lyophilized powder dissolves best at room temperature. Let the vial warm naturally for about 10 minutes before adding water. But do not heat it. Do not use warm water. Do not place it near a heat source. Room temperature means room temperature, around 20 to 25 degrees Celsius.
Mistake 6: Not labeling the vial
Without a reconstitution date on the vial, you have no way to track the 28-day use window. Without a concentration label, you risk dosing errors if you mix multiple peptides or use different reconstitution volumes across vials. Write the date, concentration, and peptide name directly on the vial with a permanent marker or adhesive label. It takes 10 seconds and prevents potentially serious errors.
Advanced reconstitution considerations
Beyond the basics, several nuances can further optimize your reconstitution results. These details separate good protocols from excellent ones.
Air pressure equalization
Lyophilized peptide vials are often sealed under reduced pressure or vacuum. When you push the needle through the stopper, you may notice the water getting pulled in by the vacuum rather than requiring you to push the plunger. Alternatively, if the vial has positive pressure, pushing water in becomes difficult because you are compressing the air inside.
The solution is to equalize pressure before adding water. Draw air into your mixing syringe equal to the volume of water you plan to add. Insert the needle into the vial and inject the air first. This equalizes the pressure, making the water addition smooth and controlled. Without equalization, you fight against pressure differences that make precise measurement difficult.
Multiple draw technique for large volumes
If you are adding 4mL or 5mL of bacteriostatic water but only have a 3mL syringe, you will need two draws. The process stays the same, just repeated. Add the first portion (say 3mL), remove the syringe, draw the remaining portion, and add it the same way, down the glass wall, slowly. Some researchers worry that the two-step process agitates the powder unnecessarily. It does not, as long as you maintain gentle technique throughout.
Dead volume accounting
Every vial has dead volume, the small amount of solution trapped below the needle tip when the vial appears empty. For most standard vials, this is approximately 0.05 to 0.1mL. With a 3.33mg/mL concentration, 0.1mL of dead volume represents about 0.33mg of unrecoverable semaglutide. Over multiple vials, this adds up. Some researchers add an extra 0.1mL of bacteriostatic water to compensate, accepting a slightly lower concentration (3.23mg/mL instead of 3.33mg/mL) in exchange for recovering nearly all the peptide. The comprehensive peptide dosing guide discusses dead volume strategies across different vial and syringe types.
Semaglutide dose escalation timeline with a 10mg vial
Planning how your 10mg vial fits into a full dose escalation protocol requires understanding both the weekly dosing schedule and the 28-day reconstitution shelf life. Here is a practical framework that balances efficiency with safety.
Week-by-week plan using 3mL reconstitution (3.33mg/mL)
Vial 1 reconstitution (covers weeks 1 through 4):
Reconstitute full 10mg with 3mL bacteriostatic water. Draw 7.5 units weekly for 0.25mg doses. Total semaglutide used in 4 weeks: 1mg. Remaining in vial: approximately 9mg. Vial expires 28 days after reconstitution.
Here is the challenge. You use only 1mg out of 10mg within the 28-day window. That leaves 9mg potentially beyond its safe use date. This is where some researchers deviate from the one-vial approach.
Alternative approach (more economical):
Start your first 8 weeks with a 5mg vial instead. At 0.25mg for 4 weeks then 0.5mg for 4 weeks, you use 3mg total from the 5mg vial. The remaining 2mg falls within the 28-day window more efficiently. Then switch to a 10mg vial for weeks 9 through 16, where weekly doses of 1mg and 1.7mg consume the vial much faster.
For researchers who want optimal results from their semaglutide protocol, vial efficiency matters because degraded peptide means inconsistent dosing, which means inconsistent outcomes.
Managing multiple vials during escalation
As your dose increases, you may go through vials faster than expected. At the 2.4mg maintenance dose, a 10mg vial lasts only 4.16 weeks, which aligns almost perfectly with the 28-day reconstitution window. This is actually the sweet spot for 10mg vials. At maintenance dosing, you reconstitute a fresh vial roughly every 4 weeks and use nearly all of it before the next reconstitution is needed.
Keep track of which vial you are drawing from, especially if you have multiple peptides in your research program. Mixing up vials between different peptides or different reconstitution dates can lead to serious dosing errors. The peptide cycle planning guide provides organizational strategies for multi-peptide protocols.
Comparing semaglutide reconstitution to other GLP-1 peptides
If you have experience reconstituting other peptides, semaglutide follows the same general principles but has some specific characteristics worth noting.
Semaglutide versus tirzepatide reconstitution
Semaglutide and tirzepatide are both GLP-1 receptor agonists, but their reconstitution profiles differ. Tirzepatide is a dual GIP/GLP-1 agonist with a slightly different molecular structure. Both dissolve readily in bacteriostatic water. Both use similar reconstitution volumes. The key difference is in dosing, as tirzepatide escalation starts at 2.5mg rather than 0.25mg, which means much larger weekly doses and faster vial consumption.
Researchers transitioning between the two should recalculate everything. The tirzepatide dosing guide covers its specific reconstitution and unit calculations, which differ substantially from semaglutide despite using the same type of syringe and diluent.
Semaglutide versus BPC-157 reconstitution
If your first peptide experience was with BPC-157, you are already familiar with the basic process. Both use bacteriostatic water, both require gentle swirling rather than shaking, and both need refrigerated storage after mixing. The main difference is concentration. BPC-157 vials typically contain 5mg and are reconstituted for twice-daily dosing at microgram-level amounts. Semaglutide vials contain 5 to 10mg and are dosed weekly at milligram-level amounts. The math is simpler for semaglutide because the doses are larger and easier to measure on standard syringes.
The BPC-157 dosage calculator handles the more complex calculations that sub-milligram dosing requires, which is less of a concern with semaglutide and its relatively larger weekly doses.
Reconstitution stability comparison
Semaglutide is considered one of the more stable peptides once reconstituted. Its molecular structure includes a fatty acid side chain that improves stability and gives it its long half-life of approximately 7 days. This is why once-weekly dosing works. Compare this to peptides like GHK-Cu or TB-500, which may require more frequent dosing due to shorter half-lives. Semaglutide and its stability profile is one reason why the 28-day reconstituted shelf life is generally achievable without significant potency loss. The peptide stability guide compares shelf life and degradation profiles across commonly used peptides.
Troubleshooting reconstitution problems
Even with perfect technique, issues occasionally arise. Here is how to diagnose and address the most common reconstitution problems.
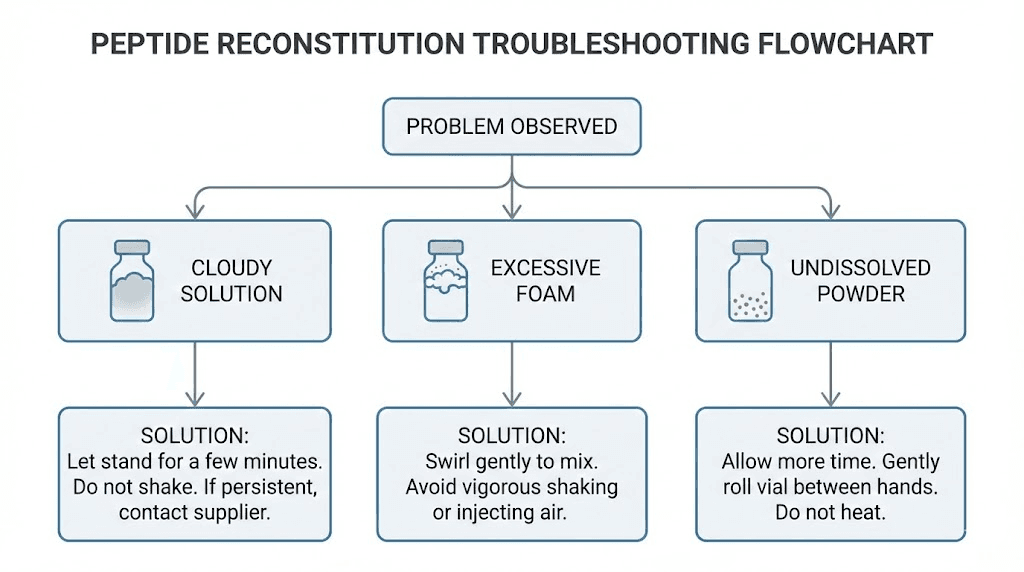
The powder will not dissolve
If gentle rolling does not dissolve the powder within five to ten minutes, the issue is usually temperature. Cold powder dissolves slowly. Remove the vial from the refrigerator and let it sit at room temperature for 15 to 20 minutes before trying again. Do not add heat. If the powder still does not dissolve after extended room-temperature rolling, the lyophilized product may have been damaged during shipping or storage. Contact the vendor for a replacement.
Excessive foam after adding water
Foam indicates that water was added too forcefully or that the vial was agitated too aggressively. Small amounts of foam are normal and will dissipate within a few minutes if you let the vial sit undisturbed. Large amounts of foam mean peptide is trapped in air-liquid interfaces, reducing the actual concentration of your solution. Prevention is better than cure here. Add water slowly down the vial wall and never shake the vial.
Difficulty pushing water into the vial
If you encounter resistance when trying to push the plunger, the vial has positive internal pressure. Inject an equal volume of air into the vial before adding water to equalize pressure. This is standard technique for sealed vials and should be done routinely, not just when you encounter resistance.
Solution turns cloudy after initial clarity
A solution that was clear after reconstitution but turns cloudy during storage typically indicates protein aggregation due to temperature excursion (the vial got too warm or too cold) or bacterial contamination.
Either way, the solution should be discarded. Check your refrigerator temperature and ensure the vial is not near the freezer compartment or exposed to temperature fluctuations. Understanding how peptides perform at room temperature helps identify whether a temperature excursion may have occurred.
Safety considerations for reconstituted semaglutide
Working with reconstituted peptides carries responsibilities beyond just getting the math right. Safety protocols protect both the integrity of the peptide and the researcher.
Sterile technique fundamentals
Every interaction with the vial is a potential contamination event. Minimize the number of times you puncture the stopper. Use a fresh needle for each draw. Swab the stopper with alcohol before every needle insertion, not just the first time. Work in a clean, low-traffic area away from open windows, fans, or air vents that could carry airborne particles onto your work surface.
Some researchers use a designated clean space or even a small laminar flow hood for reconstitution. While not strictly necessary for personal research use, these measures reduce contamination risk significantly. The peptide safety guide covers sterile technique in comprehensive detail.
Needle safety and disposal
Used syringes and needles are sharps waste. Never throw them in regular trash. Use a proper sharps disposal container. Many pharmacies accept sharps containers for free disposal. Some local governments offer mail-back programs for sharps waste. Proper injection safety includes responsible disposal of all materials.
When to consult a healthcare provider
Semaglutide is a prescription medication in its FDA-approved forms (Ozempic, Wegovy, Rybelsus). Compounded versions used in research settings should still be approached with appropriate medical guidance. Consult a healthcare provider before beginning any semaglutide protocol to discuss appropriate dosing, potential interactions with other medications, and contraindications that may apply to your specific health situation. The peptide therapy guide discusses how to find qualified providers who understand peptide protocols.
Frequently asked questions
Can I use sterile water instead of bacteriostatic water for 10mg semaglutide?
Technically yes, but it is strongly discouraged for multi-dose vials. Sterile water lacks preservatives, meaning each needle puncture increases contamination risk. Since a 10mg vial provides weeks of doses, bacteriostatic water with its benzyl alcohol preservative is the only safe choice for multi-dose reconstitution. The bacteriostatic water guide explains why this distinction matters.
How do I know if my reconstituted semaglutide has gone bad?
Look for visual changes. A properly reconstituted semaglutide solution is clear, colorless, and free of particles. If the solution turns yellow, cloudy, or develops floating particles, it has degraded or become contaminated. Also watch for reduced effectiveness at your usual dose, which may indicate potency loss. Discard any vial that shows visible changes. Check the expired semaglutide guide for detailed degradation indicators.
What happens if I add too much bacteriostatic water?
Adding more water than intended creates a lower concentration, which means each unit on your syringe delivers less semaglutide than planned. Your doses will be smaller than intended unless you recalculate. The solution is to determine your actual concentration (10mg divided by actual mL added) and use the unit conversion formula (dose in mg / concentration in mg per mL x 100) to recalculate your correct draw volume. The reconstitution calculator can help with this recalculation.
Can I add more bacteriostatic water later if the solution is too concentrated?
Yes. If you initially added 2mL and want a less concentrated solution, you can add additional bacteriostatic water. Simply calculate your new total volume and divide 10mg by that number to get your new concentration. For example, adding 1mL more to an existing 2mL reconstitution gives you 3mL total, yielding a 3.33mg/mL concentration. Make sure to re-label the vial with the new concentration.
How long can I leave reconstituted semaglutide out of the fridge?
Minimize room temperature exposure. Brief periods of 30 to 60 minutes for dose preparation are fine. Extended room temperature storage (hours or days) accelerates degradation. If your vial has been at room temperature for more than 2 hours, return it to the refrigerator immediately and monitor for changes in appearance over the next few days. Our guide on peptide stability at room temperature provides specific timeframes for various peptides.
Is 10mg of semaglutide the same regardless of the vendor?
The labeled amount should be consistent, but actual peptide content can vary between vendors, especially with compounded or research-grade products. Reputable vendors provide certificates of analysis (COAs) showing actual peptide content, purity, and endotoxin testing results. Always verify COAs before reconstituting. The peptide testing labs guide explains how to read and verify these certificates.
Do I need to use a filter needle for reconstitution?
Filter needles are not typically required for standard semaglutide reconstitution from sealed vials. They are more relevant when drawing from glass ampules that may shed glass particles when broken open. Vials with rubber stoppers and flip-off caps do not pose the same glass fragment risk. However, some researchers prefer filter needles for an extra layer of particulate protection. It is a reasonable precaution, not a strict requirement.
Can I reconstitute semaglutide and freeze it for later use?
Freezing reconstituted semaglutide is generally not recommended. Ice crystal formation can damage the peptide structure through physical disruption of molecular bonds. Some researchers report success with single-freeze protocols (reconstituting, aliquoting into single-dose volumes, and freezing once), but this is not standard practice and may affect potency unpredictably. Refrigeration at 2 to 8 degrees Celsius remains the recommended storage method after reconstitution.
Quick reference summary
Recommended setup for most researchers
Add 3mL of bacteriostatic water to your 10mg semaglutide vial. This creates a 3.33mg/mL concentration. Use U-100 insulin syringes. For 0.25mg, draw 7.5 units. For 0.5mg, draw 15 units. For 1.0mg, draw 30 units. Store refrigerated at 2 to 8 degrees Celsius. Use within 28 days. The SeekPeptides peptide calculator can verify these calculations instantly.
The formula you need to remember
Units to draw = (desired dose in mg / concentration in mg per mL) x 100
That single formula, combined with your known concentration, handles every dosing scenario you will encounter across your entire protocol.
For researchers serious about optimizing their semaglutide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, accurate calculators, proven protocols, and a community of thousands who have navigated these exact questions. Members access detailed reconstitution guides, dosing calculators, and expert-reviewed protocols that take the guesswork out of peptide research.
External resources
NIH PubMed Central - Once-Weekly Semaglutide for Weight Management
Drugs.com - Semaglutide Uses, Dosage, and Side Effects
In case I do not see you, good afternoon, good evening, and good night. May your reconstitutions stay clear, your calculations stay accurate, and your protocols stay consistent.