Feb 25, 2026

Tired of conflicting information about compounded tirzepatide shelf life? One pharmacy says 30 days. Another says 90. The label on your vial might say something entirely different from what you read online last night. And every time you search for a straight answer, you get buried under vague disclaimers and contradictory timelines that leave you more confused than when you started.
Here is the truth. Compounded tirzepatide does not come with the same standardized shelf life as FDA-approved pens like Mounjaro or Zepbound. The shelf life of tirzepatide from a compounding pharmacy depends on the formulation type, the preservatives used, the storage conditions maintained, and the specific protocols that pharmacy follows. That creates real variation. Real confusion. And real risk if you get it wrong.
This guide covers everything you need to know about how long your compounded tirzepatide stays safe and effective. You will learn the exact timelines for refrigerated and room temperature storage, the difference between lyophilized and liquid formulations, what the beyond use date on your vial actually means, the science behind peptide degradation, and what to do if something goes wrong with your storage. Whether you are three weeks into your first vial or planning a trip with your medication, you will find the specific answers you need. SeekPeptides has compiled the most comprehensive breakdown available, drawing from USP guidelines, compounding pharmacy standards, and peptide stability research.
The short answer: how long compounded tirzepatide actually lasts
Most compounded tirzepatide lasts between 28 and 90 days when stored properly in the refrigerator. That is the quick version. But the real answer depends on several factors that make a significant difference in whether your vial is still good at week four, week eight, or week twelve.
The timeline breaks down like this.
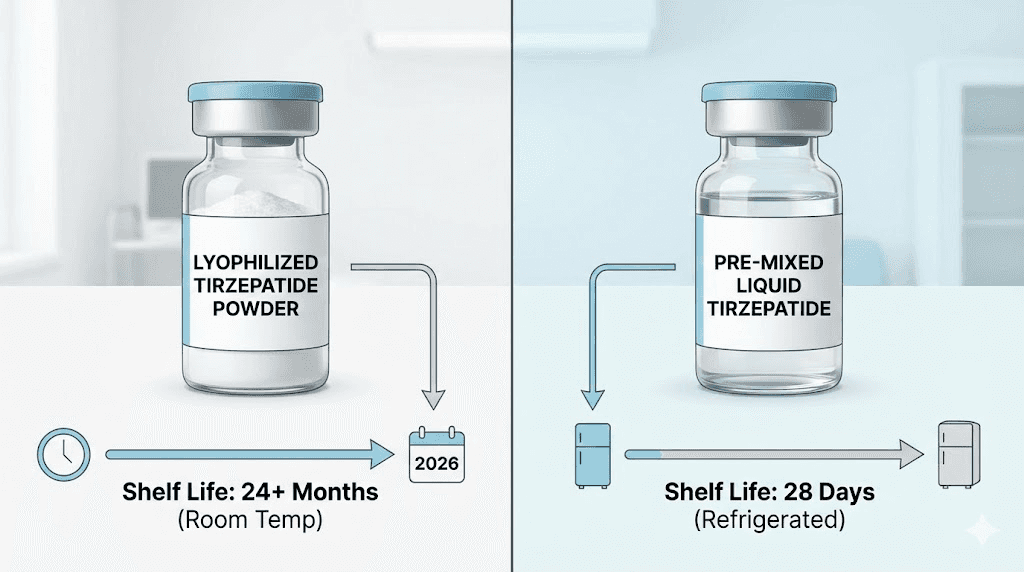
Lyophilized (freeze-dried powder) tirzepatide can remain stable for up to 24 months before you reconstitute it, as long as it stays refrigerated and sealed. Once you add bacteriostatic water and mix it into a liquid solution, that clock starts ticking much faster. Reconstituted lyophilized tirzepatide typically needs to be used within 28 to 30 days when kept in the refrigerator at 2 to 8 degrees Celsius.
Pre-mixed liquid compounded tirzepatide, the kind that arrives ready to inject from most compounding pharmacies, generally carries a beyond use date of 28 to 90 days from the date of preparation. The specific timeline depends on the pharmacy, the preservatives in the formulation, and whether the vial has been punctured.
Multi-dose vials that have been punctured carry a stricter timeline. Once you insert a needle into a multi-dose vial, most pharmacies set the beyond use date at 28 days regardless of what the original label says. This protects against bacterial contamination that can occur each time the rubber stopper is pierced. If your pharmacy includes bacteriostatic preservatives in the formulation, you may get the full BUD printed on the label. Without preservatives, 28 days after first puncture is the standard.
At room temperature, the window shrinks dramatically. Compounded tirzepatide can safely remain outside the refrigerator for up to 21 days, but only if the ambient temperature stays below 86 degrees Fahrenheit (30 degrees Celsius). Exceed that threshold, even briefly, and the degradation accelerates. You cannot simply refrigerate it again and reset the clock. For a deeper look at temperature guidelines, see our guide on how long compounded tirzepatide can be out of the fridge.
And freezing? Never. Freezing compounded tirzepatide irreversibly damages the peptide structure. If your vial has frozen, even partially, discard it. There is no recovery from freeze damage at the molecular level.
Understanding the beyond use date on your vial
The beyond use date, often abbreviated as BUD, is the single most important number on your compounded tirzepatide vial. It is not the same thing as a manufacturer expiration date. Understanding the difference will save you from using degraded medication and wasting money on vials you stored incorrectly.
A manufacturer expiration date applies to FDA-approved products like Mounjaro and Zepbound. These dates are determined through extensive, long-term stability testing conducted under controlled conditions. The manufacturer can prove, with clinical data, that the medication remains safe and effective through that date. Those tests often cover 12 to 24 months of storage.
A beyond use date is different. Compounding pharmacies assign BUDs based on United States Pharmacopeia (USP) guidelines, specifically USP Chapter 797 for sterile preparations. The BUD represents the last date the pharmacy can guarantee that the compounded preparation meets acceptable standards for sterility, potency, and stability. It is typically much shorter than a manufacturer expiration date because compounded medications do not undergo the same extensive testing.
Think of it this way. The manufacturer expiration date says we tested this for two years and it was still good. The BUD says based on the standards we follow and the conditions we compound in, we can guarantee quality through this date.
Both dates matter. But the BUD on a compounded vial is more conservative by design, which means you should take it seriously. Using compounded tirzepatide past its BUD means the pharmacy can no longer vouch for what is inside that vial. The peptide may have lost potency. The preservatives may have degraded. Bacterial contamination becomes a genuine concern, especially in multi-dose vials that have been punctured multiple times.
Where do you find the BUD on your vial? It should be printed directly on the pharmacy label, typically near the preparation date. If your vial does not have a clear BUD printed on it, contact your compounding pharmacy immediately. Every legitimate compounding pharmacy is required to include this information. If you are comparing compounding options, our guide on affordable compounded tirzepatide covers what to look for in a reputable source.
One critical point that many people miss. The BUD on your label assumes proper storage from the moment the vial was prepared. If your vial spent three days in transit without proper cold-chain shipping, or sat on your counter for a week before you put it in the refrigerator, the effective shelf life is shorter than what the label indicates. The BUD is a best-case scenario, not a guarantee regardless of handling.
Lyophilized powder vs pre-mixed liquid: shelf life differences
Compounded tirzepatide comes in two primary formulations, and the shelf life difference between them is dramatic. Understanding which one you have changes everything about storage expectations and planning.
Lyophilized (freeze-dried) tirzepatide
Lyophilized tirzepatide arrives as a dry powder in a sealed vial. This is the same freeze-drying process used across pharmaceutical manufacturing to extend the stability of sensitive peptide compounds. The water is removed under vacuum at low temperatures, leaving behind a stable cake or powder that resists degradation far better than a liquid solution.
Before reconstitution, lyophilized tirzepatide can remain stable for 12 to 24 months when stored in the refrigerator. Some sources cite stability at room temperature for shorter periods, but refrigeration is always the safer choice for maximum longevity. This makes lyophilized formulations ideal for people who want to buy in bulk or who will not start their protocol immediately. For a complete breakdown of the differences between these formulation types, read our comparison of lyophilized vs liquid peptides.
The catch comes after reconstitution. Once you add bacteriostatic water or sterile water to the powder, the clock starts. Reconstituted lyophilized tirzepatide should be used within 28 to 30 days when refrigerated at 2 to 8 degrees Celsius. Some pharmacies may allow up to 60 days if bacteriostatic water was used and proper sterile technique was maintained during reconstitution. But 28 days is the conservative, widely accepted standard.
If you are working with lyophilized tirzepatide, the reconstitution process matters enormously for shelf life. Rough handling, contamination during mixing, or using sterile water instead of bacteriostatic water will all shorten the usable window. Check our reconstituting tirzepatide chart and the peptide reconstitution calculator for precise mixing ratios.
Pre-mixed liquid tirzepatide
Most compounding pharmacies now ship tirzepatide as a pre-mixed liquid in multi-dose vials. This formulation is ready to draw and inject, with no reconstitution required. Convenient. But less shelf-stable than unreconstituted powder.
Pre-mixed liquid compounded tirzepatide carries a BUD of 28 to 90 days from the date of preparation. The variation comes from differences in compounding environment, preservative systems, and the specific stability testing each pharmacy has conducted. A pharmacy operating under USP 797 Category 3 conditions with validated stability data can assign longer BUDs. A pharmacy working under Category 1 or 2 conditions will assign shorter ones.
The convenience of pre-mixed liquid comes with a trade-off. You are working with a shorter total shelf life from the day the pharmacy prepared it. If the pharmacy compounded your vial on January 1st and assigned a 60-day BUD, you have until March 2nd regardless of when you received it or opened it. Transit time counts against your window.
Which formulation is better for your situation?
If you want maximum flexibility and the longest possible shelf life before starting your protocol, lyophilized is the clear winner. You can store the unreconstituted powder for months and only mix what you need when you need it. This is also the better option for researchers who purchase larger quantities at once.
If you prefer simplicity and do not want to deal with reconstitution, pre-mixed liquid is more convenient. Just be aware that the clock started ticking the day the pharmacy made it, not the day you received it. Plan your dosing schedule accordingly to use the full vial before the BUD expires. For help calculating your doses, use the compounded tirzepatide dosage calculator.
Refrigeration rules that protect your tirzepatide
Refrigeration is not optional for compounded tirzepatide. It is the foundation of everything related to shelf life, potency, and safety. Get the temperature right, and your medication stays effective through the full beyond use date. Get it wrong, and you are injecting something that may no longer work as intended.
The ideal temperature range
Store compounded tirzepatide at 2 to 8 degrees Celsius, which translates to 36 to 46 degrees Fahrenheit. This is the standard pharmaceutical cold storage range, and it applies to virtually all injectable peptide formulations. Most household refrigerators maintain temperatures between 35 and 38 degrees Fahrenheit, which falls squarely within the acceptable range.
Temperature consistency matters as much as the average temperature. A refrigerator that cycles between 30 and 50 degrees Fahrenheit may average out to 40 degrees, but those swings at either extreme accelerate degradation. The peptide bonds in tirzepatide are sensitive to thermal fluctuation. Repeated warming and cooling cycles, even within the broadly acceptable range, cause cumulative damage that adds up over weeks.
Where to place the vial in your refrigerator
The middle shelf is the best location. Not the door, which experiences the most temperature fluctuation every time you open it. Not the back wall, where temperatures can drop close to freezing. And definitely not the vegetable crisper drawer, which can trap moisture and sometimes runs colder than the main compartment.
Keep the vial in its original packaging or in an opaque container. Light degrades tirzepatide. Fluorescent refrigerator lights, while not as damaging as sunlight, still contribute to photodegradation over weeks of exposure. If your vial came in a box, leave it in the box. Simple protection that costs nothing but makes a real difference over a 60 or 90-day BUD period.
For more detailed storage guidance across all peptide types, our peptide storage guide covers best practices. And if you are also storing semaglutide, the rules are similar but not identical, as detailed in our guide on compounded semaglutide refrigeration.
The freezing danger
This deserves its own emphasis because the consequences are severe and irreversible. If compounded tirzepatide freezes, the peptide structure is permanently damaged. Ice crystal formation physically disrupts the molecular bonds that give tirzepatide its biological activity. A frozen and thawed vial may look perfectly fine, clear solution, no particles, normal color, but the peptide inside has lost an unpredictable amount of its potency.
Freezing risk is highest in these situations: placing the vial against the back wall of a cold refrigerator, storing it in a mini-fridge with poor temperature control, leaving it in a car during winter, and shipping without adequate insulation during cold months. If you suspect your vial may have frozen, even partially, the safest course is to discard it and contact your pharmacy for a replacement.
This same principle applies to all peptide-based medications. For broader context, see how long peptides last in the fridge and the general rules around storing peptides after reconstitution.
Room temperature storage and the 21-day rule
Sometimes refrigeration is not immediately possible. You are traveling. Your pharmacy shipped without a cold pack. You left the vial on the counter overnight. Life happens. The question becomes: is your tirzepatide still good?
The general rule for compounded tirzepatide at room temperature is 21 days maximum at temperatures not exceeding 86 degrees Fahrenheit (30 degrees Celsius). This comes from stability data on the FDA-approved formulation of tirzepatide (Mounjaro and Zepbound), which compounding pharmacies use as a reference point for their own formulations. Some compounding pharmacies set an even more conservative limit of 7 to 14 days at room temperature for their specific formulations.
Critical temperature thresholds
Below 77 degrees Fahrenheit (25 degrees Celsius): This is standard room temperature. Degradation occurs slowly. The 21-day window applies with reasonable confidence for most formulations.
Between 77 and 86 degrees Fahrenheit (25 to 30 degrees Celsius): Degradation accelerates. The 21-day window is the absolute maximum. Use the vial as quickly as practically possible and return it to the refrigerator between uses.
Above 86 degrees Fahrenheit (30 degrees Celsius): The 21-day rule no longer applies. Peptide degradation becomes rapid. Even brief exposure, like leaving a vial in a hot car for a few hours, can compromise potency significantly. At temperatures above 104 degrees Fahrenheit (40 degrees Celsius), degradation can occur within hours. For related temperature concerns, see what happens if tirzepatide gets warm.
Can you re-refrigerate after room temperature exposure?
This is one of the most commonly asked questions, and the answer depends on who you ask. Some sources say no, absolutely not, that once removed from the refrigerator the vial must be used within the room temperature window and then discarded. Others say brief room temperature exposure followed by prompt refrigeration is acceptable as long as you account for the cumulative time out of the fridge.
The conservative and safest approach: track the total time your vial spends outside the refrigerator. If that cumulative total reaches 21 days, discard the vial regardless of the BUD. Each hour at room temperature counts against your total budget. And any exposure above 86 degrees should prompt serious consideration about whether the vial is still viable.
For reference on how similar GLP-1 medications handle room temperature exposure, see our article on semaglutide storage in the fridge and what happens if semaglutide gets warm.

The science behind tirzepatide degradation
Understanding why compounded tirzepatide degrades helps you make smarter storage decisions. This is not just abstract chemistry. These degradation mechanisms directly explain why the 28 to 90-day BUD exists, why temperature control matters so much, and why visual inspection alone cannot tell you whether your tirzepatide is still fully potent.
Chemical degradation pathways
Tirzepatide is a 39-amino-acid peptide that acts as a dual GIP and GLP-1 receptor agonist. Like all peptides, its biological activity depends on maintaining a precise three-dimensional structure. Several chemical processes can disrupt that structure over time.
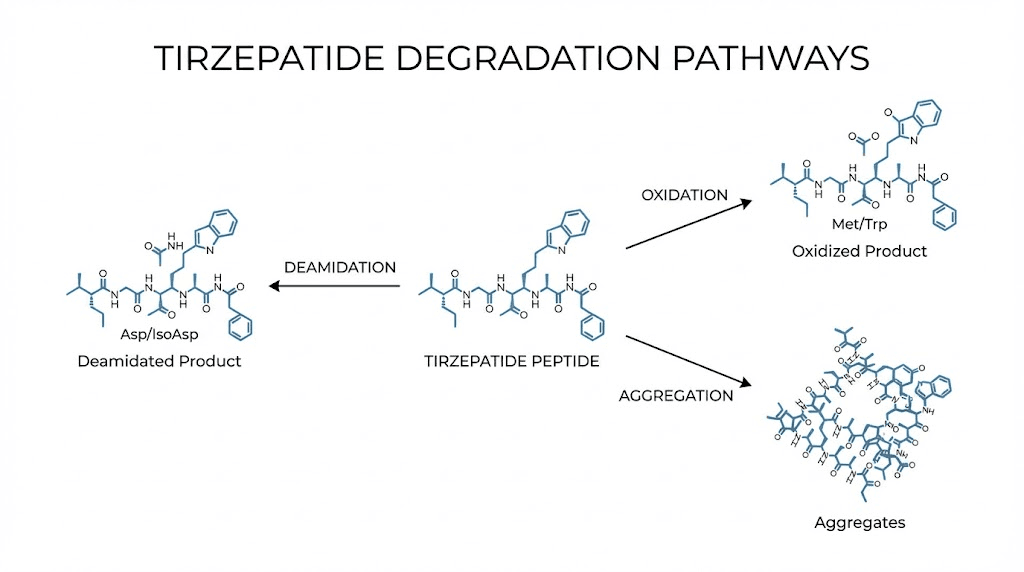
Deamidation is the most common degradation pathway for peptides containing asparagine and glutamine residues. Water molecules attack these amino acids, converting them into aspartic acid or glutamic acid derivatives. This changes the charge and shape of the peptide, reducing its ability to bind to GIP and GLP-1 receptors effectively. Deamidation accelerates at higher temperatures and at pH values outside the optimal range of 4 to 5 for storage formulations.
Oxidation targets methionine and cysteine residues in the peptide chain. Exposure to light, dissolved oxygen in the solution, and trace metal contaminants all promote oxidation. This is why keeping the vial in its original packaging and away from light matters, and why some pharmacies include antioxidant excipients in their formulations.
Hydrolysis involves the breaking of peptide bonds by water molecules. All peptides in aqueous solution are subject to hydrolysis, but the rate depends heavily on temperature and pH. At refrigeration temperatures, hydrolysis proceeds slowly enough to preserve potency for weeks to months. At room temperature or above, the rate increases significantly.
For more on how peptides function at the molecular level, see our guide on how peptides work and the broader overview of what peptides are.
Physical degradation: aggregation and precipitation
Beyond chemical changes, tirzepatide can undergo physical degradation that alters its structure without breaking covalent bonds.
Aggregation occurs when individual tirzepatide molecules clump together, transitioning from their normal alpha-helix structure to beta-sheet formations. These aggregates can be soluble (invisible to the eye) or insoluble (visible as cloudiness or particles). Soluble aggregates are particularly concerning because the solution looks normal but the peptide has lost significant biological activity.
Aggregation is promoted by temperature fluctuations, mechanical agitation (shaking the vial vigorously), and freeze-thaw cycles. This is one reason gentle handling is emphasized. When drawing from the vial, rotate it gently between your palms. Do not shake it. The research on peptide stability consistently shows that physical agitation accelerates aggregate formation.
Surface adsorption is another physical process where tirzepatide molecules stick to the glass walls of the vial or the rubber stopper. This reduces the concentration of active peptide in solution without any visible change. Quality compounding pharmacies add surfactants like polysorbate 80 to their formulations to minimize surface adsorption.
The role of pH in stability
Research indicates that tirzepatide stability is optimized in a pH range of 4 to 5 for storage formulations, though some studies suggest broader stability up to pH 8.5 depending on the specific buffer system used. Formulations outside the optimal pH range degrade faster, even under proper refrigeration.
This is one of the invisible quality differences between compounding pharmacies. A pharmacy that properly buffers its tirzepatide formulation to maintain optimal pH throughout the shelf life will produce a product that actually lasts through the full BUD. A pharmacy that cuts corners on formulation quality may produce a product that starts degrading well before the labeled date. This is why choosing a reputable compounding source matters enormously. Our review of Empower Pharmacy tirzepatide and our guide on affordable tirzepatide options can help you evaluate your choices.
Alt: Tirzepatide peptide degradation mechanisms diagram showing chemical breakdown pathways
Signs your compounded tirzepatide has gone bad
Visual inspection is your first line of defense, but it is not your only one. Some signs of degradation are obvious. Others are subtle enough to miss if you do not know what to look for. And some forms of degradation are completely invisible, which is why the BUD matters even when the solution looks fine.
Visual indicators to check every time
Before every injection, hold the vial up to a light source and examine the solution carefully.
Cloudiness or turbidity. Compounded tirzepatide should be clear. If the solution appears hazy, milky, or cloudy, that indicates protein aggregation or bacterial contamination. Do not use it. Discard the vial and contact your pharmacy.
Particles or floaters. Any visible particles floating in the solution, whether they look like tiny specks, fibers, or crystalline fragments, indicate degradation or contamination. Even a single visible particle is grounds for discarding the vial.
Color changes. Plain compounded tirzepatide should be clear and colorless to slightly yellow. Tirzepatide compounded with B12 may appear light yellow to light pink, which is normal. Any shift outside these expected ranges, such as darkening, browning, or unexpected color changes, signals chemical breakdown. For more on color expectations with B12 formulations, see what color tirzepatide with B12 should look like.
Vial damage. Cracks in the glass, a loose or damaged stopper, or any sign that the seal has been compromised means the vial is no longer sterile. Discard immediately.
Physical indicators during use
Pay attention to how the medication feels and behaves during and after injection.
Increased injection site reactions. Unusual burning, stinging, or redness beyond what you normally experience could indicate degraded peptide or bacterial contamination. Some injection site reactions are normal, but a sudden change from your usual experience warrants attention. See our guide on injection site reaction treatment for distinguishing normal from abnormal responses.
Loss of appetite suppression. If your compounded tirzepatide was effectively suppressing your appetite and you suddenly notice that effect diminishing without changing your dose, degraded medication is one possible explanation. This is tricky because metabolic adaptation and plateau effects can cause similar symptoms. But if the timing coincides with a vial that has been stored improperly or is approaching its BUD, degradation should be considered. For more on distinguishing between plateau and medication issues, see tirzepatide not working anymore and why you might not be losing weight on tirzepatide.
The invisible degradation problem
Here is the uncomfortable truth. A significant amount of peptide degradation is invisible. Soluble aggregates do not cause cloudiness. Early-stage deamidation does not change the color. Partial oxidation does not produce particles. Your tirzepatide can look completely normal and still have lost 20, 30, or even 50 percent of its potency.
This is exactly why the BUD exists. It is not a guess. It is the pharmacy saying: beyond this date, we can no longer guarantee that the peptide inside meets the minimum standards for potency and sterility, even if it looks fine to the naked eye.
Respect the date. If your vial has passed its BUD, discard it regardless of appearance. For context on what happens if you use expired medication, see our articles on what happens if you use expired tirzepatide and whether tirzepatide expires.
Why pharmacy BUDs vary so much
If you have ordered compounded tirzepatide from different pharmacies, you have probably noticed that the beyond use dates can differ by weeks or even months. One pharmacy stamps 28 days on the label. Another gives you 60 days. A third claims 90 days. All for what appears to be the same medication. This is not arbitrary. There are real reasons for the variation.
Compounding environment category
Under USP 797, the standard that governs sterile compounding in the United States, pharmacies are classified into categories based on the environmental controls in their compounding areas. The category determines the maximum BUD they can assign.
Category 1 preparations are compounded in less controlled environments, typically segregated compounding areas without full ISO-classified cleanrooms. These preparations receive the shortest BUDs, often 12 to 24 hours at room temperature or 24 hours refrigerated, though multi-dose vials with preservatives may receive longer dates.
Category 2 preparations come from pharmacies with more controlled environments and may receive BUDs of up to 4 days or longer depending on specific conditions.
Category 3 preparations are produced in ISO-classified cleanroom environments with extensive environmental monitoring, personnel qualification, and process validation. These can receive BUDs of 60 to 180 days depending on whether the preparation was aseptically processed or terminally sterilized, and whether sterility testing was performed.
The bottom line: a pharmacy operating under Category 3 conditions with validated stability data can legitimately assign a 90-day BUD to their compounded tirzepatide. A pharmacy operating under less stringent conditions cannot. The longer BUD reflects better quality control, not marketing exaggeration.
Preservative systems
The preservatives included in the formulation significantly impact shelf life. Bacteriostatic preservatives like benzyl alcohol inhibit bacterial growth in multi-dose vials, which is essential for maintaining sterility through repeated needle punctures over weeks of use.
Formulations without preservatives must be used more quickly, often within days rather than weeks, because each puncture introduces potential contamination with no chemical defense against bacterial growth. Most compounding pharmacies include preservatives in their multi-dose tirzepatide vials, but the specific preservative system and concentration vary, which contributes to BUD variation.
Stability testing
Some compounding pharmacies invest in proprietary stability testing for their specific formulations. They submit samples to independent laboratories that test potency, purity, sterility, and particulate matter at regular intervals over months. This real-world stability data allows them to assign longer, evidence-based BUDs with confidence.
Other pharmacies rely solely on the default BUDs prescribed by USP 797 based on their compounding category, without conducting product-specific stability testing. There is nothing wrong with this approach, as it follows the regulatory framework. But it typically results in shorter, more conservative BUDs.
When evaluating compounding pharmacies, ask whether they have conducted stability testing specific to their tirzepatide formulation. This is one of the most meaningful quality indicators available to consumers. For guidance on evaluating compounding sources, our reviews of ProRX tirzepatide, Orderly Meds tirzepatide, and Southend Pharmacy tirzepatide include pharmacy quality indicators.
USP 797 and compounding standards explained
USP 797 is the regulatory framework that governs how sterile medications, including compounded tirzepatide, are prepared, stored, and assigned beyond use dates in the United States. Understanding the basics of this standard helps you evaluate the quality and reliability of your compounded medication.
What USP 797 actually requires
The revised USP 797 standard, which became official on November 1, 2023, establishes requirements for compounding personnel qualifications, facility design and environmental monitoring, preparation and handling procedures, quality assurance processes, and beyond use dating. It applies to all organizations that compound sterile preparations, including hospitals, outpatient pharmacies, and 503B outsourcing facilities.
For tirzepatide specifically, the key provisions relate to environmental controls during compounding, preservative requirements for multi-dose containers, and the criteria pharmacies must meet to assign extended BUDs. These are not voluntary guidelines. They carry regulatory weight, and pharmacies found out of compliance face enforcement action.
503A vs 503B pharmacies
Compounding pharmacies in the United States generally fall into two categories under federal law, and the distinction matters for your tirzepatide quality.
503A pharmacies are traditional compounding pharmacies that prepare medications pursuant to individual patient prescriptions. They are regulated primarily by state boards of pharmacy and must comply with USP 797 standards.
503B outsourcing facilities operate under more stringent federal oversight by the FDA. They can compound without individual prescriptions and must comply with current Good Manufacturing Practice (cGMP) requirements, which impose additional quality controls beyond USP 797. Tirzepatide from a 503B facility generally benefits from more rigorous quality assurance, which can translate to more reliable shelf life through the stated BUD.
When ordering compounded tirzepatide, knowing whether your pharmacy is a 503A or 503B facility gives you additional context for the reliability of the BUD on your vial. For those researching the grey market tirzepatide landscape, understanding these regulatory categories is essential for distinguishing legitimate sources from questionable ones.
How to maximize the shelf life of your compounded tirzepatide
You cannot change the BUD printed on your vial. But you can ensure that your tirzepatide actually remains viable through the full date by following evidence-based storage and handling practices. Many people unknowingly shorten their medication shelf life through small, easily correctable mistakes.
Temperature management
Maintain a consistent 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit) from the moment you receive your vial. If your pharmacy ships with cold packs, transfer the vial to the refrigerator immediately upon delivery. Do not leave the package sitting on your porch or kitchen counter while you tend to other things.
Consider purchasing a small refrigerator thermometer (available for a few dollars) and placing it near where you store your vials. This lets you verify that your refrigerator actually maintains the appropriate temperature range. Older refrigerators, mini-fridges, and units with frequent door openings can drift outside the safe range without any external indication.
Light protection
Keep the vial in its original box or in an opaque bag inside the refrigerator. Tirzepatide undergoes photodegradation when exposed to light, including the light inside your refrigerator. This is a slow process, but over a 60 or 90-day BUD period, cumulative light exposure can meaningfully reduce potency.
Sterile technique during use
Every time you draw from a multi-dose vial, you introduce a tiny risk of contamination. Minimize this risk by cleaning the rubber stopper with an alcohol swab before every draw, using a new sterile needle and syringe each time, not touching the needle to any surface before inserting it into the vial, and drawing the dose in a clean environment (not a dusty garage or outdoor setting).
Proper injection technique also matters for your overall protocol success. Our guides on how to inject tirzepatide in the stomach, where to inject GLP-1, and best injection sites cover technique in detail.
Minimize room temperature exposure
Remove the vial from the refrigerator only long enough to draw your dose. Do not leave it sitting on the counter while you prepare the injection site, check your phone, or get distracted by other tasks. Two minutes out of the fridge is fine. Twenty minutes is unnecessary exposure. Two hours is the start of cumulative time that reduces your effective storage window.
Track your cumulative room temperature time if you are methodical about it. Some people keep a small note on the vial box recording each time they remove it and how long it stayed out. This is especially useful if you are working with a 90-day BUD and want to ensure the vial remains fully viable through the end.
Plan your dosing to use the full vial
One of the most common sources of waste is ordering a vial size that contains more doses than you can use within the BUD period. If your vial contains 8 doses and your BUD is 28 days, you need to inject at least twice per week to finish it in time.
Use the compounded tirzepatide dosage calculator to determine exactly how many doses your vial contains at your current dose level. Then compare that against your BUD to ensure you will not have leftover medication that expires before you can use it. Our tirzepatide dose chart and compound dosage chart can help with planning.
SeekPeptides members get access to protocol planning tools that help optimize dose scheduling around vial sizes and BUD timelines, ensuring nothing goes to waste.
Traveling with compounded tirzepatide
Travel introduces every storage challenge simultaneously: temperature fluctuations, extended time without refrigeration, physical jostling, and unpredictable environments. But with proper planning, you can travel safely with your compounded tirzepatide without compromising its potency.
Short trips (1 to 3 days)
For short trips, an insulated medication travel case with a gel ice pack is sufficient. These cases maintain temperatures in the acceptable range for 12 to 24 hours depending on ambient conditions. Pack the case in your carry-on luggage, not in checked baggage where temperatures in the cargo hold can drop below freezing or exceed safe limits.
The 21-day room temperature rule gives you plenty of buffer for a weekend trip. Even without active cooling, the vial will remain viable for several days at normal indoor temperatures below 86 degrees Fahrenheit. Just return it to the refrigerator as soon as you reach your destination.
Longer trips (4+ days)
For extended travel, you need a reliable cooling solution. Medical-grade travel coolers designed for insulin and injectable medications work well for tirzepatide. These use either gel packs that are replaced daily or battery-powered active cooling systems that maintain precise temperatures.
When staying in hotels, use the in-room mini-fridge with caution. Hotel mini-fridges are notoriously inconsistent in temperature, and many run warmer than a standard household refrigerator. Place the vial in the main compartment (not the door), and consider wrapping it in an insulating layer to buffer against temperature swings when the unit cycles on and off.
If you are also traveling with semaglutide, the storage considerations are similar but not identical. Our comprehensive traveling with semaglutide guide covers parallel tips that apply to tirzepatide travel as well.
Air travel specifics
Compounded tirzepatide in vials is permitted through TSA security as a medically necessary liquid. Carry a copy of your prescription or a letter from your prescriber to smooth the screening process. Keep the medication in its original pharmacy-labeled vial, as this serves as documentation. Syringes require a prescription label as well.
Place the travel cooler and medication in your carry-on bag. Do not check it. Cargo holds on commercial aircraft can experience temperatures ranging from below freezing to over 100 degrees Fahrenheit depending on the aircraft type and route. A single flight in checked luggage could destroy your entire supply.
International travel considerations
If traveling internationally, research the destination country regulations on importing injectable medications. Most countries permit travelers to bring personal medication supplies with proper documentation (prescription, pharmacy label, prescriber letter). Some countries have restrictions on importing compounded medications specifically. For those sourcing from other countries, our guide on tirzepatide in Mexico covers cross-border considerations.
Compounded vs FDA-approved tirzepatide: storage comparison
If you are transitioning between compounded and FDA-approved tirzepatide, or deciding between them, understanding the storage differences helps you plan your protocol and manage expectations.
Factor | Compounded tirzepatide | FDA-approved (Mounjaro/Zepbound) |
|---|---|---|
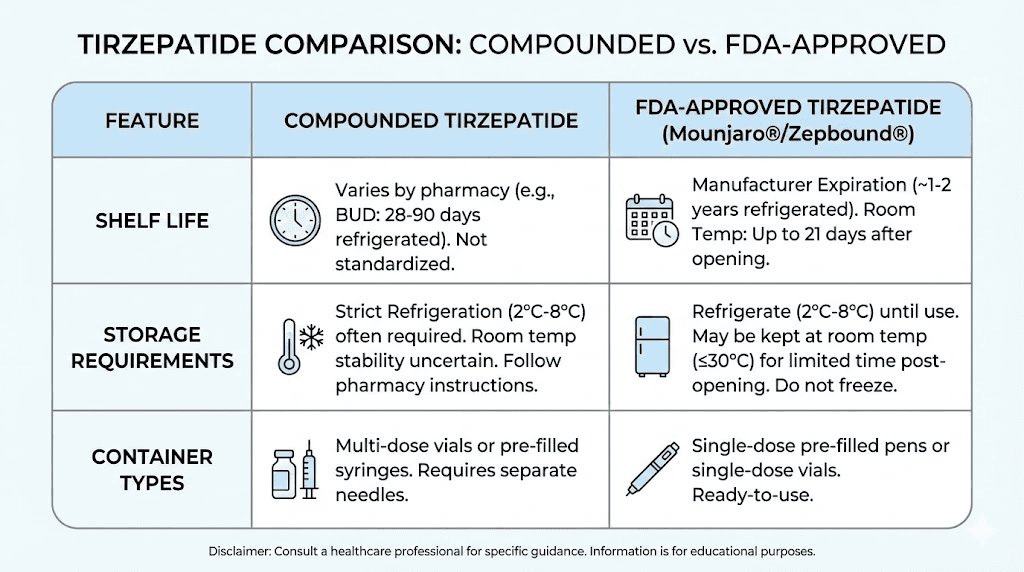
Shelf life unopened | 28 to 90 days (BUD varies by pharmacy) | Up to 24 months (manufacturer tested) |
Refrigeration temp | 2 to 8 degrees C (36 to 46 degrees F) | 2 to 8 degrees C (36 to 46 degrees F) |
Room temp duration | Up to 21 days below 86 degrees F | Up to 21 days below 86 degrees F |
After first use | 28 days for multi-dose vials (typical) | 21 days for pens (per manufacturer) |
Freeze tolerance | None, discard if frozen | None, discard if frozen |
Container type | Multi-dose vials (usually) | Single-dose pre-filled pens |
Stability testing | USP 797 based, some pharmacy-specific | Extensive manufacturer testing (ICH guidelines) |
Preservatives | Varies by pharmacy formulation | Standardized manufacturer formulation |
The most significant practical difference is the shelf life window. FDA-approved tirzepatide pens can sit in your refrigerator for months before first use. Compounded vials start counting down from the day the pharmacy prepared them, giving you a much tighter window. Plan accordingly.
For a broader comparison of compounded vs branded GLP-1 options, see our detailed semaglutide vs tirzepatide comparison and our guide on compounded semaglutide.
Common mistakes that shorten shelf life
Most compounded tirzepatide that goes bad before its BUD does so because of user handling errors, not pharmacy quality issues. These are the most common mistakes and how to avoid them.
Shaking the vial vigorously
This is perhaps the most widespread mistake. People shake the vial to mix the contents before drawing a dose, especially if they notice anything settled at the bottom. Vigorous shaking creates mechanical stress that promotes aggregation, the clumping of peptide molecules that reduces potency. Instead, gently roll the vial between your palms or invert it slowly a few times. The goal is gentle mixing, not a cocktail shaker.
Leaving the vial out during dose preparation
It takes five minutes to prepare an injection. Maybe ten if you are careful about cleaning the injection site, drawing the dose, and checking for air bubbles. But many people take the vial out of the refrigerator and leave it on the counter for 30 minutes or an hour while they go about their routine. Each extended exposure accumulates. Over weeks of this pattern, your vial spends hours at room temperature that eat into the 21-day cumulative limit.
Storing in the refrigerator door
The door shelf experiences the most dramatic temperature swings, cycling between cold and ambient temperatures every time you open the refrigerator. For items with long shelf lives, like condiments, this does not matter. For a sensitive peptide formulation, it does.
Ignoring the preparation date
The BUD counts from the date of preparation, not the date you received the vial. If your pharmacy prepared the vial two weeks before shipping it, you have already lost two weeks of your storage window before you ever opened the package. Always check the preparation date on the label and plan your usage accordingly.
Using non-sterile technique
Touching the rubber stopper without alcohol cleaning, reusing needles, or drawing doses in unclean environments introduces bacteria into the vial. Preservatives can inhibit bacterial growth, but they are not a substitute for sterile technique. Contaminated vials can become dangerous regardless of the BUD.
For proper technique guidance, review our peptide injection guide and the step-by-step instructions for giving injections with a syringe.
Not keeping the vial in its box
The original pharmacy packaging is not just for shipping. It protects the vial from light exposure during storage. Removing the vial and placing it bare in the refrigerator subjects it to the light inside the unit every time the door opens, accelerating photodegradation over weeks of storage.
Using vials from questionable sources
Compounded tirzepatide from unregulated sources may not follow USP 797 standards, may not include proper preservatives, may not be stored correctly before shipping, and may not carry accurate BUD information. The shelf life stamped on a vial is only as reliable as the pharmacy that produced it. Stick with verified, reputable compounding pharmacies. Our guide on grey market tirzepatide explains the risks in detail.
What to do if your tirzepatide was stored incorrectly
Mistakes happen. The vial sat on the counter overnight. The cold pack in your shipping box thawed during transit. Your mini-fridge malfunctioned while you were away for the weekend. Here is how to assess the situation and decide whether your medication is still safe to use.
Step 1: Assess the exposure
Determine what happened as precisely as possible. How long was the vial at room temperature? What was the approximate ambient temperature? Did the vial get warm (above 86 degrees F) or potentially freeze? The specifics matter enormously.
Step 2: Visual inspection
Examine the solution for cloudiness, particles, color changes, or any deviation from its normal appearance. If anything looks off, discard it. No exceptions.
Step 3: Apply the temperature-time rules
If the vial was at room temperature (below 86 degrees F) for less than 24 hours: it is likely still viable. Return it to the refrigerator and add that time to your cumulative room temperature total.
If the vial was at room temperature for 1 to 7 days: it is probably still usable, but subtract those days from the 21-day room temperature maximum. Use the remaining medication promptly and keep it refrigerated between uses.
If the vial was at temperatures above 86 degrees F for any significant period (more than a few hours): the risk of meaningful degradation is high. Consider discarding it, especially if you are in the early stages of your protocol where consistent dosing matters most. For related scenarios, see what to do if you accidentally left semaglutide out overnight, as the same principles apply.
If the vial may have frozen: discard it. There is no way to assess freeze damage visually, and partially frozen, thawed tirzepatide may have lost significant potency while appearing normal.
Step 4: Contact your pharmacy
Most reputable compounding pharmacies have policies for medication that may have been compromised during shipping or storage. Contact them, explain the situation, and ask whether a replacement is warranted. Many pharmacies will replace vials that were damaged during transit at no additional cost.
Step 5: Monitor your response
If you decide to continue using the vial after a storage incident, pay close attention to your body response over the next one to two weeks. Reduced appetite suppression, unexpected side effects, or injection site reactions that differ from your normal experience could indicate that the medication has degraded. Track your weight loss progress and how your body responds to identify any changes that might correlate with the storage incident.
When in doubt, err on the side of caution. Replacing a vial is far less costly than weeks of ineffective treatment because you continued using degraded medication. For help managing the financial side, our guides on affordable compounded tirzepatide and the peptide cost calculator can help you budget effectively.
Additional formulation considerations
Compounded tirzepatide does not only come in standard injectable form. Several formulation variants exist, each with slightly different shelf life characteristics.
Tirzepatide with B12 or glycine
Many compounding pharmacies offer tirzepatide combined with vitamin B12 (methylcobalamin), glycine, or both. These tirzepatide with B12 formulations and tirzepatide glycine B12 compounds generally follow the same BUD as standard compounded tirzepatide, since the additional ingredients do not significantly alter the primary storage requirements. However, B12 can change the expected color of the solution (making it slightly pink or yellow), which means your visual inspection baseline is different from plain tirzepatide.
For detailed information on these formulation additions, see our guides on tirzepatide with B12, tirzepatide with glycine, tirzepatide with niacinamide, and tirzepatide with methylcobalamin.
Oral and sublingual formulations
Oral tirzepatide drops and sublingual formulations are emerging alternatives to injectable compounded tirzepatide. These formulations have different storage characteristics because they are not sterile preparations and may use different preservative systems. The BUD for tirzepatide drops and orally disintegrating tablet formulations varies significantly from injectable versions. Always follow the specific storage instructions provided by the pharmacy for non-injectable formulations. Our comparison of oral vs injectable tirzepatide and tablets vs injections covers the practical differences in detail.
Compounded vs research-grade peptides
Some researchers work with research-grade tirzepatide rather than pharmacy-compounded formulations. Research-grade peptides, typically sold as lyophilized powder, have different storage expectations and are not assigned BUDs under the same regulatory framework. For information on how long peptides last in powder form and general peptide expiration timelines, see our dedicated guides.
Optimizing your protocol around vial shelf life
Smart protocol planning accounts for your vial BUD from the beginning, not as an afterthought when you realize you have leftover medication approaching expiration.
Matching vial size to your dosing schedule
If your compounding pharmacy offers multiple vial sizes, choose one that matches your weekly dose and your BUD. For example, if you inject 5 mg once weekly and your vial contains 30 mg with a 28-day BUD, you have enough medication for 6 weeks but only 4 weeks of guaranteed stability. Either use a smaller vial or increase your injection frequency if appropriate for your protocol.
Discuss vial sizing with your prescriber and pharmacy. Many pharmacies can customize the concentration or volume to match your needs. The dosage calculator helps determine what vial configuration works best for your dose level. For those starting out, see our guide on compounded tirzepatide starting dose.
Dose escalation and shelf life planning
Tirzepatide protocols typically involve dose escalation over weeks or months, starting at lower doses and gradually increasing. This means your per-injection volume changes over time, which affects how quickly you use each vial. Plan your vial orders to align with your current dose level, not the dose you expect to be on in two months.
For those using microdosing protocols, the math is different. Lower doses mean less medication drawn per injection, which means each vial lasts longer in terms of dose count, but the BUD does not change. A microdosing protocol on a 28-day BUD vial may result in significant waste if the vial contains more doses than you can use in four weeks.
SeekPeptides members can access detailed protocol planning tools and syringe dosage guides that account for vial sizes, BUD timelines, and dose escalation schedules to minimize waste and maximize effectiveness.
Long-term storage and bulk purchasing considerations
If you purchase compounded tirzepatide in quantities larger than a single vial, proper storage planning becomes even more important.
Storing multiple vials
Keep unopened vials in the refrigerator at 2 to 8 degrees Celsius. If you receive lyophilized vials, do not reconstitute them until you are ready to begin using them. Unreconstituted lyophilized tirzepatide has a dramatically longer shelf life (up to 24 months) compared to reconstituted liquid (28 to 30 days). Reconstitute one vial at a time and keep the rest as powder.
For pre-mixed liquid vials, the BUD is set at the time of compounding. If you order three vials simultaneously, they all have the same preparation date and the same BUD. You cannot stockpile pre-mixed liquid compounded tirzepatide for months, because all your vials will expire around the same time. Stagger your orders so each vial arrives as you need it, with fresh BUD dates.
The cost-shelf life balance
Larger orders often come at lower per-unit costs, but the savings evaporate if you cannot use all the medication before it expires. Use the peptide cost calculator to compare the actual cost per usable dose, accounting for any waste from expired medication. Sometimes a smaller, more frequent order is more economical than a bulk purchase with 30 percent waste.
For insights on getting the best value, see our guides on affordable tirzepatide and cheap compounded tirzepatide options.
Supporting your protocol while managing storage
Proper storage is just one piece of a successful tirzepatide protocol. Your results depend on consistent dosing, appropriate lifestyle factors, and understanding how the medication works in your body.
Diet matters significantly during tirzepatide use. Understanding which foods to avoid, following a structured diet plan, and knowing what to eat on tirzepatide can enhance your results and reduce side effects like constipation and digestive discomfort.
Side effects are a normal part of the adjustment process. Common experiences include fatigue, headaches, body aches, and sleep disturbances. These typically improve as your body adjusts to the medication. For a complete picture, see our breakdown of semaglutide vs tirzepatide side effects and general peptide safety information.
Supplements can support your protocol as well. Our guide on supplements to take with tirzepatide covers evidence-based additions that complement the medication effects without interfering with its stability or efficacy.
For those exploring broader peptide options alongside tirzepatide, see our guides on best peptides for weight loss, peptides for weight loss overview, and the comparison of GLP-1 effectiveness timelines.
Understanding whether your medication is working as expected requires realistic timeline expectations. Our guides on how fast tirzepatide works, before and after results, and metabolic effects help you distinguish between normal progress and signs that something, including storage issues, may need attention.
For researchers serious about maximizing every aspect of their peptide protocols, SeekPeptides provides the most comprehensive resource available. Members access evidence-based storage guides, protocol optimization tools, detailed pharmacology breakdowns, and a community of thousands who have navigated these exact questions. From dosing guides to meal planning to complete peptide dosing references, the platform covers everything you need for safe, effective peptide research.
When to discard and reorder: a practical decision framework
Knowing when to discard a vial and order a replacement is just as important as knowing how to store it. Here is a practical framework for making that decision without overthinking it.
Discard immediately if:
The solution is cloudy, contains particles, or has changed color. The vial has passed its beyond use date, even if the solution looks perfectly normal. The vial may have frozen at any point during storage or transit. The rubber stopper appears compromised, damaged, or loose. You have drawn from the vial more than 10 times (some pharmacies set lower limits).
Consider discarding if:
The vial has spent more than 14 cumulative days at room temperature, even if the 21-day limit has not been reached. Your appetite suppression or weight loss results have noticeably decreased without any other explanation. The vial was exposed to temperatures above 86 degrees Fahrenheit for more than a few hours. You cannot confirm the cold chain was maintained during shipping.
Probably still fine if:
The vial was at room temperature for less than 8 hours and is within its BUD. The solution is clear, colorless (or appropriately tinted for B12 formulations), and free of particles. The vial has been consistently refrigerated except for brief removal during dose preparation. Your clinical response remains consistent with previous injections from the same vial.
Reordering strategy
Do not wait until your current vial is empty to order a replacement. Account for pharmacy preparation time (1 to 5 days), shipping time (1 to 3 days), and potential delays. Ideally, place your next order when you have 7 to 10 days of medication remaining. This prevents gaps in your protocol that can reduce effectiveness and cause rebound appetite increase.
If you are on a structured dosing protocol, maintaining consistency is critical for results. Gaps between vials allow the medication to clear your system partially, which can lead to temporary return of appetite and other symptoms. Our compound dosage charts can help you plan orders around your specific dose schedule.
For those managing costs, planning ahead also allows you to take advantage of bulk pricing or subscription discounts that some pharmacies offer. The cost calculator helps you compare options and find the most economical approach without sacrificing quality or freshness.
Frequently asked questions
Can I use compounded tirzepatide after the beyond use date?
No. The beyond use date represents the last day the compounding pharmacy guarantees the sterility, potency, and safety of the preparation. Using it after this date carries risks of reduced effectiveness and potential contamination. Discard any vial that has passed its BUD and order a fresh supply. For more detail, see our guide on tirzepatide expiration.
Does compounded tirzepatide go bad faster than Mounjaro or Zepbound?
Compounded tirzepatide typically has a shorter guaranteed shelf life than FDA-approved versions because it does not undergo the same extensive stability testing. However, when stored properly and used within the BUD, compounded tirzepatide should deliver equivalent potency during its stated window. The key difference is the length of that window, not the quality within it.
My vial was left out overnight. Is it still good?
Most likely yes, if the ambient temperature was below 86 degrees Fahrenheit and the total room temperature exposure time (including this incident) has not exceeded 21 days. Return it to the refrigerator immediately and make a note of the exposure. For a detailed assessment, see how long tirzepatide can be out of the fridge.
How do I know if my pharmacy is following proper compounding standards?
Ask your pharmacy whether they are a 503A or 503B facility, whether they comply with current USP 797 standards, and whether they have conducted stability testing specific to their tirzepatide formulation. Legitimate pharmacies will answer these questions readily. Reluctance to discuss their compounding standards is a red flag.
Can I freeze compounded tirzepatide to extend its shelf life?
Absolutely not. Freezing destroys the peptide structure irreversibly. Ice crystal formation causes physical damage to the molecular bonds that give tirzepatide its biological activity. A frozen and thawed vial should be discarded entirely, even if the solution appears normal after thawing.
Why does my compounded tirzepatide have a 28-day BUD when others get 90 days?
BUD variation reflects differences in compounding environment quality, preservative systems, and whether the pharmacy has conducted product-specific stability testing. A 28-day BUD from a pharmacy using basic compounding conditions is equally valid as a 90-day BUD from a pharmacy with advanced cleanroom facilities and stability data. The shorter date is more conservative, not less legitimate.
Does tirzepatide with B12 expire at a different rate?
The addition of B12 (methylcobalamin) does not significantly change the shelf life of compounded tirzepatide. The BUD assigned by the pharmacy accounts for all ingredients in the formulation. However, B12 formulations have a different normal color range (light yellow to light pink), so adjust your visual inspection expectations accordingly. See our tirzepatide with B12 guide for details.
Is lyophilized tirzepatide better than pre-mixed liquid for long-term storage?
Yes, if long-term storage is your priority. Lyophilized (freeze-dried) tirzepatide can remain stable for up to 24 months before reconstitution, compared to 28 to 90 days for pre-mixed liquid. Once reconstituted, however, the shelf life drops to approximately 28 to 30 days, making it comparable to pre-mixed formulations. Lyophilized is the better choice for anyone who purchases multiple vials at once or who may not start their protocol immediately. For a full comparison, see our guide on lyophilized vs liquid peptides.
How do I track cumulative room temperature time for my vial?
Keep a small piece of tape or a note on the vial box and write down each time you remove it from the refrigerator, along with the approximate duration. Most injection preparation takes 2 to 5 minutes, which is negligible. The concern is longer exposures: the vial sitting out for an hour during meal prep, overnight on the counter, or extended periods during travel. Add up the total minutes and hours over the life of the vial. Once the cumulative total approaches 21 days (or your pharmacy specific limit), plan to finish the vial quickly or discard any remaining medication.
What should I do with expired compounded tirzepatide?
Do not throw expired medication in household trash or flush it down the drain. Use a drug take-back program if available in your area, or follow FDA guidelines for safe disposal of injectable medications. Your compounding pharmacy may also accept returned expired medication for proper disposal. For more on using expired medication, see what happens if you use expired tirzepatide.
External resources
USP General Chapter 797: Pharmaceutical Compounding, Sterile Preparations
Designing Formulation Strategies for Enhanced Stability of Therapeutic Peptides, PMC
Factors Affecting Physical Stability of Peptide Therapeutics, Royal Society
In case I do not see you, good afternoon, good evening, and good night. May your vials stay cold, your peptides stay potent, and your protocols stay effective.