Feb 20, 2026

Before you order another vial from any compounding pharmacy, stop. Read what follows. The story behind BPI Labs and the tirzepatide formulations they produce is more complicated than most researchers expect. And the details buried in FDA inspection reports, court documents, and patent filings could change how you evaluate every compounded tirzepatide source on your list.
BPI Labs is not just another 503B outsourcing facility. This Florida-based operation runs as a subsidiary of Belcher Pharmaceuticals, an FDA-registered drug manufacturer with a history stretching back decades. That dual registration, as both a manufacturer and a compounder, gives BPI Labs capabilities most pharmacies cannot match. Robotic aseptic filling systems. In-house analytical laboratories. AI-powered visual inspection technology. The infrastructure is genuinely impressive by any standard.
But impressive technology does not tell the whole story.
The corporate history behind BPI Labs includes fraud convictions tied to the parent company, multiple FDA Form 483 observations, whistleblower lawsuits, and a regulatory landscape for tirzepatide that changed dramatically when the FDA drug shortage ended. This guide covers everything informed researchers need to know before making decisions. The manufacturing quality. The product formulations. The FDA compliance record. The Belcher Pharmaceuticals backstory. The ongoing patent battle with Eli Lilly. And how BPI Labs compares to other compounders in a market where due diligence is no longer optional, it is essential. SeekPeptides researchers know that understanding your source matters as much as understanding the compound itself.
What is BPI Labs?
BPI Labs LLC operates from Largo, Florida as both an FDA-registered drug manufacturer and a 503B outsourcing facility. That dual designation is rare. Most compounding pharmacies hold one registration or the other. BPI Labs holds both, which means their operations fall under stricter regulatory requirements than a standard compounder. They are licensed in 49 states and comply with 21 CFR Part 210 and 211, the current Good Manufacturing Practice regulations that govern pharmaceutical production in the United States.
The company headquarters sits at 12393 Belcher Road South, Suite 450, Largo, FL 33773. From this facility, BPI Labs services hospitals, clinics, and telehealth platforms across the country. Their client base includes major names in the direct-to-consumer health space, and their production capabilities span both sterile and non-sterile compounded medications.
Belcher Pharmaceuticals and the BPI Labs origin story
Understanding BPI Labs requires understanding Belcher Pharmaceuticals. BPI Labs LLC is a wholly owned subsidiary of Belcher Pharmaceuticals LLC, a company that has manufactured branded and generic injectable medications for years. The relationship is not just financial. Belcher Pharmaceuticals provides the manufacturing infrastructure, the regulatory framework, and the corporate leadership that shapes how BPI Labs operates.
Belcher Pharmaceuticals itself has a complicated corporate lineage. The company connects back to GeoPharma Inc., a publicly traded pharmaceutical entity that experienced significant regulatory and legal challenges over its history. That lineage matters, and researchers evaluating BPI Labs tirzepatide compounds should understand the full corporate picture before making sourcing decisions.
The connection between BPI Labs and Belcher is not hidden. BPI Labs openly identifies as a Belcher subsidiary on their website and in regulatory filings. This transparency is worth noting, even as the implications of that connection deserve careful examination.
Manufacturing location and nationwide licensing
Operating from a single facility in Largo, Florida, BPI Labs maintains licensing in 49 of 50 states. That near-universal licensing means clinics and telehealth providers across nearly all of the United States can source compounded tirzepatide and other formulations from BPI Labs without state-level regulatory barriers.
The Florida location is strategic. The state has become a hub for compounding pharmacies and pharmaceutical manufacturing, partly because of favorable regulatory environments and proximity to raw material supply chains. Several other major compounders, including facilities that produce compounded semaglutide, operate from the same region.
BPI Labs manufacturing and quality infrastructure
The manufacturing capabilities at BPI Labs represent a genuine step above what most 503B outsourcing facilities offer. That is not marketing language. It reflects the physical infrastructure, the technology investments, and the in-house capabilities that come from operating under both FDA drug manufacturer and 503B registrations simultaneously.
Robotic aseptic filling technology
BPI Labs uses fully automated, human-less robotic aseptic filling systems operating in an ISO5 environment. This is the same classification level required for commercial pharmaceutical manufacturing, not just compounding. The robotic systems handle the filling process without human intervention in the critical zone, which dramatically reduces contamination risk during production of sterile injectables like tirzepatide injections.
Why does this matter? Because most compounding pharmacies rely on manual aseptic technique, where trained technicians fill vials by hand inside laminar flow hoods. Manual filling introduces variability. Every human movement inside a clean room creates potential contamination events. Robotic filling eliminates that variable entirely.
The distinction between manual and robotic filling is not trivial. For researchers concerned about sterility and product integrity, the filling technology used by a compounder should rank high on the evaluation checklist. BPI Labs claims to be the only FDA-registered drug manufacturer with a 503B pharmacy producing GLP-1 medications using this level of automation.
In-house testing and analytical capabilities
BPI Labs maintains in-house analytical laboratories, in-house microbiological testing facilities, and an in-house research and development department. All batch testing happens on-site rather than being outsourced to third-party contract laboratories. This includes potency testing, sterility testing, endotoxin testing, and impurity analysis.
In-house testing offers several advantages for tirzepatide dosage accuracy and safety. Results come back faster, allowing quicker release of batches. The testing protocols remain under direct control of BPI Labs quality assurance teams. And there is no reliance on third-party timelines or quality standards that may differ from internal expectations.
Additionally, BPI Labs uses automated visual inspection powered by artificial intelligence technology. Every vial passes through visual inspection systems that check for particulate matter, fill volume accuracy, and container integrity. This automated approach catches defects that human visual inspection might miss, particularly during high-volume production runs.
Production scale and batch capacity
BPI Labs can handle batch sizes up to 500 liters. That production capacity positions them as a high-volume compounder capable of supplying large telehealth platforms and hospital networks simultaneously. For context, a 500-liter batch of tirzepatide at 10mg per mL concentration would produce tens of thousands of individual vials.
The facility also manufactures its own sterile water for injection on-site, using a dedicated sterile water manufacturing plant. This vertical integration means BPI Labs controls another critical input in the compounding process rather than sourcing it externally.
One additional infrastructure detail worth noting: BPI Labs uses single-use, disposable pathways throughout production. Disposable pathways eliminate cross-contamination risk between batches and remove the need for cleaning validation between production runs. This approach is standard in commercial pharmaceutical manufacturing but less common in compounding facilities. For researchers tracking their tirzepatide dosing for weight loss, knowing that each batch starts with completely fresh contact surfaces provides additional confidence in product consistency.
BPI Labs tirzepatide product overview
BPI Labs produces tirzepatide in a formulation that differs from standalone compounded tirzepatide available from other pharmacies. Their product combines tirzepatide with Vitamin B6, specifically pyridoxine hydrochloride. This combination formulation is available as a prescription-only sterile injectable and comes in multiple strengths and vial configurations.
The tirzepatide with Vitamin B6 formulation
The primary product from BPI Labs is tirzepatide combined with Vitamin B6 at a concentration of 10mg per mL for both the tirzepatide and the pyridoxine components. This is a ready-to-use formulation, meaning healthcare providers do not need to perform any additional reconstitution before administration. The vials arrive pre-mixed and ready for injection.
For comparison, many other compounders produce tirzepatide as a lyophilized powder requiring reconstitution with bacteriostatic water before use. The ready-to-use format from BPI Labs eliminates reconstitution errors, which represent one of the most common sources of dosing inaccuracy in compounded peptide protocols. No measuring bacteriostatic water volumes. No calculating concentrations. No risk of contamination during the mixing process.
This distinction matters enormously for researchers following precise compounded tirzepatide dosage protocols. A ready-to-use formulation removes an entire category of potential error from the process.
Available concentrations and vial sizes
BPI Labs offers their tirzepatide with B6 formulation in at least three strength configurations. The product is available in both 1mL and 3mL vial sizes, giving healthcare providers flexibility in prescribing based on individual patient protocols and tirzepatide dose requirements.
The 3mL vial at 10mg per mL concentration contains 30mg of tirzepatide total. At standard titration schedules, a single 3mL vial could last several weeks depending on the prescribed dose level. Researchers familiar with tirzepatide 30mg dosage charts will recognize this as a common vial size for multi-week protocols.
Higher concentration formulations, including 20mg per mL options, have also been referenced in distribution materials. These higher concentrations reduce the injection volume needed for larger doses, which can improve comfort and compliance. Check our tirzepatide 20mg/mL dosing chart for detailed unit calculations at this concentration.
Storage requirements and beyond use dating
One of the notable features of BPI Labs tirzepatide formulations is the 12-month beyond use date (BUD). That is significantly longer than many compounded tirzepatide products from other pharmacies, which frequently carry 90-day or 180-day beyond use dates. A 12-month BUD provides more flexibility for clinics managing inventory and for patients on longer treatment timelines.
BPI Labs also ships their tirzepatide products without requiring refrigeration during transit. This non-refrigerated shipping approach simplifies logistics and reduces the risk of temperature excursions during delivery. Once received, standard tirzepatide storage guidelines should be followed. For detailed information on how temperature affects compounded tirzepatide, see our guide on how long tirzepatide lasts in the fridge and what happens when tirzepatide is left out of the fridge.
The extended beyond use date likely reflects the stability data BPI Labs has generated through their in-house testing capabilities. Longer BUD periods require supporting stability studies demonstrating that the product maintains potency, sterility, and overall quality throughout the claimed shelf life. For researchers concerned about tirzepatide expiration, a 12-month BUD represents a meaningful advantage over shorter-dated alternatives.
Why Vitamin B6 is added to tirzepatide formulations
The inclusion of Vitamin B6 in BPI Labs tirzepatide products serves both a clinical purpose and, arguably, a regulatory one. Understanding both dimensions helps researchers evaluate whether this formulation approach matters for their specific needs.
Pyridoxine and gastrointestinal tolerability
Vitamin B6, or pyridoxine, is one of the most well-established anti-nausea compounds in medicine. Obstetricians have prescribed pyridoxine for pregnancy-related nausea for decades. The mechanism involves pyridoxine serving as a cofactor in neurotransmitter synthesis, particularly serotonin and dopamine, which play direct roles in the signaling pathways that trigger nausea.
GLP-1 receptor agonists like tirzepatide are notorious for gastrointestinal side effects, especially during the early titration phases. Nausea, vomiting, and general stomach discomfort are among the most commonly reported issues. Research into semaglutide vs tirzepatide side effects consistently shows that GI symptoms represent the primary reason researchers discontinue protocols or fail to titrate to effective doses.
By combining pyridoxine directly with tirzepatide in a single formulation, BPI Labs aims to provide built-in nausea management from the first injection. The theory is straightforward. Rather than requiring separate anti-nausea medication, the B6 component arrives with each dose automatically. For researchers who have experienced tirzepatide GI side effects, this approach offers potential convenience.
The question of effectiveness is worth examining honestly. While pyridoxine has strong evidence for pregnancy nausea, the evidence for its effectiveness against GLP-1 induced nausea specifically is less robust. The dose of B6 included in the formulation (10mg per mL) falls within standard supplemental ranges, which provides some theoretical basis. But researchers should not assume the B6 addition will eliminate GI symptoms entirely. Many people experience tirzepatide headaches, fatigue, and body aches regardless of B6 supplementation.
Other compounders have taken similar approaches with different additives. Tirzepatide with B12 is another common combination, as is tirzepatide with glycine and tirzepatide with niacinamide. Each additive targets slightly different aspects of tolerability and effectiveness. The tirzepatide glycine B12 combination has gained particular attention in research communities.
The regulatory angle of combination formulations
There is a second dimension to the B6 addition that cannot be ignored. When the FDA removed tirzepatide from the drug shortage list in October 2024 and subsequently ended enforcement discretion for compounding, pharmacies needed to demonstrate that their compounded products were not "essentially a copy" of the commercially available branded version (Mounjaro/Zepbound).
Adding Vitamin B6 to the tirzepatide formulation creates a product that differs from what Eli Lilly sells. The commercial products contain tirzepatide alone. A tirzepatide plus B6 formulation is, by definition, a different product. Whether this difference satisfies the FDA definition of "not essentially a copy" remains legally contested.
The Alliance for Pharmacy Compounding has suggested that formulations or dosage strengths not commercially available are not considered copies. Others, including the FDA and Eli Lilly, have argued that adding commonly available vitamins to avoid the "essentially a copy" restriction does not constitute a legitimate clinical modification. The legal landscape here is genuinely unsettled. Researchers should understand that the B6 addition serves both a potential clinical benefit and a regulatory strategy simultaneously.
FDA inspection history and compliance
No evaluation of a compounding pharmacy is complete without examining their FDA inspection record. BPI Labs has been inspected multiple times, and the results provide important context for researchers evaluating their tirzepatide products. Understanding what these inspection findings mean requires some background on how FDA oversight actually works.
The 2021 Form 483 findings
In 2021, the FDA inspected BPI Labs and issued a Form 483 with multiple observations. The findings included failure to use proper equipment, failure to adequately vet suppliers, and failure to thoroughly assess the risks associated with injectable drug products. These are not minor observations. They touch on fundamental elements of pharmaceutical manufacturing quality.
Equipment issues can affect everything from fill accuracy to sterility. Supplier vetting failures raise questions about the quality of raw materials, including the active pharmaceutical ingredients used in compounds like tirzepatide and semaglutide. And incomplete risk assessments for injectable products represent a potentially serious gap in quality management.
BPI Labs has stated that this audit was "successfully closed" and that the FDA issued a satisfactory Establishment Inspection Report (EIR) afterward. In FDA terminology, a closed inspection with an EIR means the agency reviewed the corrective actions taken and found them adequate. This does not erase the original findings, but it indicates the issues were addressed to FDA satisfaction at that time.
February 2025 inspection results
More recently, on February 21, 2025, the FDA issued another Form 483 to BPI Labs. This inspection found that compounded drug labels lacked mandatory information required under Section 503B, specifically the statement "Office Use Only." The labeling omission affected multiple sterile injectable batches, including semaglutide and tirzepatide vials.
A labeling deficiency might sound minor compared to equipment or supplier issues. But in pharmaceutical manufacturing, labeling is a critical compliance area. Incorrect or incomplete labels can lead to improper use, regulatory enforcement actions, and questions about overall quality system effectiveness. The "Office Use Only" designation is not optional for 503B products, it is a federal requirement that distinguishes outsourcing facility products from individually prescribed 503A compounds.
Researchers should note that a Form 483 is not the same as a Warning Letter. Form 483 observations represent findings noted during inspection. Warning Letters represent formal regulatory action with potential consequences including facility shutdown. As of the most recent available information, neither BPI Labs nor Belcher Pharmaceuticals has received an FDA Warning Letter. The distinction matters for accurate risk assessment.
Understanding what Form 483 observations mean
It is important to provide context about Form 483 findings. Receiving Form 483 observations during an FDA inspection is not uncommon. Many legitimate, high-quality pharmaceutical manufacturers receive observations that they subsequently address. The FDA inspection process is designed to identify areas for improvement, and observations alone do not indicate that products are unsafe or ineffective.
That said, the nature and pattern of observations matters. Equipment and supplier vetting issues in 2021 followed by labeling issues in 2025 suggest that compliance requires ongoing attention at the facility. For researchers evaluating compounded tirzepatide sources, the FDA inspection record should be one data point among many, not the sole determining factor.
Comparing inspection records across compounders provides better perspective. Empower Pharmacy and other major 503B facilities have their own inspection histories. No large-scale pharmaceutical operation operates without occasional regulatory findings. The question is whether findings indicate systemic issues or isolated correctable items.
The Belcher Pharmaceuticals corporate history
This section covers information that is publicly available through court records, SEC filings, Department of Justice press releases, and investigative journalism. Researchers deserve access to this context when evaluating any pharmaceutical supplier, and BPI Labs is no exception.
GeoPharma origins and early regulatory issues
The corporate lineage behind BPI Labs traces back to GeoPharma Inc., a publicly traded pharmaceutical company. GeoPharma served as a parent entity for Belcher Pharmaceuticals, which in turn created BPI Labs as a subsidiary. The GeoPharma story includes several notable episodes.
In 2004, GeoPharma issued a press release that the SEC later characterized as misleading. The company claimed FDA approval for a prescription chemotherapy side-effect treatment, but the product had only received FDA clearance as a medical device, not as a drug. The distinction between device clearance and drug approval is significant, and the press release allegedly caused a stock price increase based on investor misunderstanding of the regulatory status.
In 2011, GeoPharma filed for bankruptcy. Shareholders subsequently filed lawsuits alleging that before the collapse, company insiders transferred Belcher Pharmaceuticals to a shell company they controlled at a suspiciously low valuation. These allegations paint a picture of a corporate entity with a turbulent history.
The 2020 fraud conviction and sentencing
In November 2020, Mihir Taneja, the former CEO of GeoPharma, pleaded guilty to "introduction of misbranded drugs into interstate commerce with the intent to defraud and mislead." The Department of Justice characterized the scheme as part of a "Nationwide Telemedicine Pharmacy Health Care Fraud Conspiracy."
The specifics of the conviction involved paying kickbacks to physicians and repackaging over-the-counter medicines and vitamins as prescription-only drugs to inflate pricing. Taneja received a sentence of 10 months in federal prison plus one year of supervised release and was ordered to pay approximately 21 million dollars in restitution.
These are facts from federal court records. They do not directly involve BPI Labs operations or their current tirzepatide production. But they are part of the corporate history of the parent entity, and informed researchers should know this context exists.
Whistleblower allegations
A former Chief Science Officer named Darren Rubin filed a whistleblower lawsuit in 2020 alleging a widespread fraud scheme targeting Medicare and military TRICARE. The allegations specifically mentioned Belcher Pharmaceuticals and Taneja family members. The lawsuit included claims about BPI Labs launching 503B injectable products and alleged concerns about production practices.
The DOJ case alleged over 4,000 fraudulent TRICARE claims totaling approximately 19 million dollars. Reports indicate the parties were heading toward settlement. The timing of the settlement reportedly coincided with business developments involving BPI Labs supply partnerships.
Again, allegations in lawsuits do not constitute proven facts unless resolved through judgment or admission. But the pattern of legal issues in the corporate family tree deserves attention. Researchers comparing BPI Labs to alternatives like Priority Meds, Orderly Meds, or Peptide Sciences may want to investigate the corporate histories of those organizations as well.
How this context applies to BPI Labs today
Corporate history provides context, not conclusions. BPI Labs operates with its own leadership, its own quality systems, and its own regulatory relationships. The fraud convictions involved a former CEO of the parent company predecessor, not current BPI Labs management. The manufacturing technology and quality infrastructure that BPI Labs uses today did not exist during the GeoPharma era.
However, corporate culture and governance practices can persist through organizational transitions. Researchers should weigh this historical context alongside the manufacturing capabilities, FDA inspection records, and product quality evidence when making decisions. The BPI Labs semaglutide products undergo the same scrutiny, and researchers evaluating either product line should consider the full picture.
The tirzepatide patent battle with Eli Lilly
BPI Labs is not just manufacturing tirzepatide compounds. They are actively challenging Eli Lilly patent protection on the molecule itself. This legal battle has significant implications for the future of compounded tirzepatide availability and could reshape the market if successful.
The inter partes review filing
On August 4, 2025, BPI Labs filed an inter partes review (IPR) petition with the US Patent Trial and Appeal Board targeting Eli Lilly US Patent No. 9,474,780. The case number is IPR2025-01346. This patent is one of the foundational intellectual property protections covering tirzepatide, the active ingredient in Eli Lilly branded products Mounjaro and Zepbound.
An inter partes review is a formal proceeding where a petitioner presents evidence that an existing patent should not have been granted because the claimed invention was obvious based on prior publications. It is one of the most powerful tools available for challenging pharmaceutical patents outside of full federal litigation.
Key legal arguments and prior art claims
BPI Labs contends that the claims in Lilly patent are obvious based on prior publications by researchers including Alsina-Fernandez, DiMarchi, and Lau. The challenge introduces prior art regarding peptide design methods that enhance stability and reduce immunogenicity, capabilities that BPI Labs argues were already established before Lilly filed the patent application.
This legal strategy targets the novelty of the patent claims themselves. If the PTAB agrees that the prior art makes Lilly claims obvious, the patent could be invalidated, which would remove one of the key legal barriers to compounded tirzepatide production. For researchers monitoring tirzepatide affordability and access, this case has real implications.
BPI Labs filed this challenge in collaboration with Empower Pharmacy, another major 503B outsourcing facility that has been at the center of the tirzepatide compounding debate. Empower was sued by Eli Lilly in early 2024 for alleged patent infringement through their tirzepatide production. The collaboration between BPI Labs and Empower on the patent challenge suggests a coordinated legal strategy by major compounders.
Current status and what comes next
As of the most recent available information, the USPTO Director acknowledged BPI Labs request for Director Review following an initial denial from the PTAB, and Lilly was granted five business days to respond. The outcome of this review process will determine whether the inter partes review proceeds to a full hearing.
Meanwhile, the US Patent and Trademark Office has officially declined to review a separate petition from Empower Pharmacy seeking to invalidate the same foundational Lilly patent. That denial does not automatically affect the BPI Labs petition, as different legal arguments and prior art references may yield different outcomes. But it does indicate that challenging these patents faces significant hurdles.
Regardless of the patent challenge outcome, the legal proceedings generate valuable public information about tirzepatide formulation science, manufacturing standards, and the competitive dynamics between branded pharmaceutical companies and compounding pharmacies. Researchers interested in the broader semaglutide vs tirzepatide landscape should follow these developments.
Tirzepatide compounding after the FDA shortage ended
The regulatory environment for compounded tirzepatide changed dramatically in late 2024 and early 2025. Understanding this context is critical for anyone evaluating BPI Labs or any other tirzepatide source, because the legal basis for compounding tirzepatide shifted fundamentally when the shortage ended.
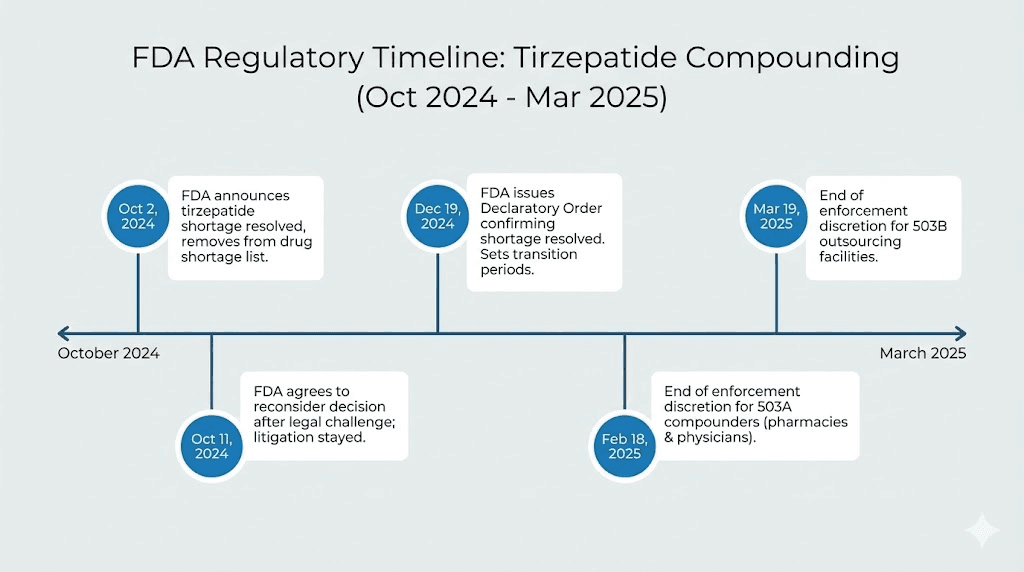
The enforcement discretion timeline
The FDA removed tirzepatide from the drug shortage list on October 2, 2024. This triggered a countdown on enforcement discretion periods that had allowed pharmacies to compound tirzepatide while the branded product was in short supply.
For 503A pharmacies, those that compound individual prescriptions based on specific patient prescriptions, the enforcement discretion period ended on February 18, 2025. For 503B outsourcing facilities like BPI Labs, the period ended on March 19, 2025. After these dates, compounding tirzepatide without meeting specific legal requirements became a significantly riskier proposition from a regulatory standpoint.
This timeline matters because it explains why BPI Labs and other compounders have pursued alternative formulations like the tirzepatide plus B6 combination, patent challenges, and other legal strategies to maintain their ability to produce tirzepatide products.
503A vs 503B and why the distinction matters
The difference between 503A and 503B pharmacies is not just regulatory classification. It reflects fundamentally different manufacturing approaches, quality requirements, and distribution models.
A 503A pharmacy compounds medications based on individual prescriptions for specific patients. They operate under state pharmacy board oversight and are generally not subject to FDA inspections or cGMP requirements. A 503B outsourcing facility like BPI Labs can produce larger batches without individual prescriptions, is FDA-registered, must follow current Good Manufacturing Practices, and is subject to FDA inspection.
For researchers evaluating compounded tirzepatide sources, the 503B designation generally provides more quality assurance. FDA oversight, cGMP compliance, adverse event reporting requirements, and regular inspections create accountability mechanisms that do not exist for most 503A pharmacies. The trade-off is that 503B facilities face stricter regulatory scrutiny, which is precisely what recent tirzepatide enforcement actions have targeted.
What "not essentially a copy" means in practice
Under Section 503B, outsourcing facilities cannot compound drugs that are "essentially a copy" of a commercially available FDA-approved product. When tirzepatide came off the shortage list, this provision became the central regulatory question for every compounder producing tirzepatide, including BPI Labs.
The addition of Vitamin B6 to BPI Labs tirzepatide formulations creates a product that is literally different from Eli Lilly commercial offerings. But whether that difference satisfies the "not essentially a copy" standard is contested. The FDA and Eli Lilly have argued that adding commonly available vitamins does not create a clinically meaningful modification. Compounders argue that any formulation difference, particularly one with clinical rationale like nausea management, should qualify.
Similar approaches have been adopted by pharmacies using B12, glycine, and other additives. The legal status of all these combination products remains in flux. Researchers should understand that regulatory risk is currently inherent in the entire compounded tirzepatide market, not just at BPI Labs.
How BPI Labs compares to other tirzepatide compounders
Evaluating BPI Labs in isolation is not particularly useful. Researchers need to understand how this facility compares to other options in the market. Several major 503B outsourcing facilities produce tirzepatide formulations, each with different manufacturing approaches, quality records, and regulatory strategies.
Empower Pharmacy comparison
Empower Pharmacy operates from Houston, Texas as a licensed 503B and 503A facility under cGMP standards. They are arguably the most well-known tirzepatide compounder due to their high profile legal battle with Eli Lilly and their extensive telehealth partnerships. Empower offers tirzepatide in various formulations and concentrations, including their own dosage chart options.
Key differences between BPI Labs and Empower include manufacturing technology (BPI Labs robotic filling vs Empower manual processes), scale of operations, and corporate structure. Both facilities face similar regulatory challenges post-shortage, and both are involved in challenging Eli Lilly tirzepatide patents. For detailed dosing comparisons, see the Empower tirzepatide dosing chart alongside BPI Labs offerings.
Other major 503B facilities
The compounded tirzepatide market includes several other notable players. Olympia Pharmacy offers both semaglutide and tirzepatide compounds with their own dosing protocols. The Olympia tirzepatide dosage chart provides another reference point for researchers comparing sources.
Lavender Sky is another pharmacy that researchers frequently evaluate alongside BPI Labs. Each compounder brings different strengths. Some excel in customer service. Others offer lower pricing. Still others provide more comprehensive testing documentation. The right choice depends on individual priorities, and researchers should evaluate multiple options rather than defaulting to any single source.
For researchers focused on finding the most affordable tirzepatide options, comparing across multiple pharmacies is essential. Price alone should never drive the decision, but it is a legitimate factor when quality and regulatory compliance are comparable. Our guide to cheap compounded tirzepatide covers how to balance cost with quality considerations.
A quality comparison framework
Rather than declaring one compounder "best," researchers benefit more from a systematic comparison framework. Consider these factors across all potential sources:
Regulatory status: 503A vs 503B vs FDA-registered manufacturer
Inspection record: Form 483 history, Warning Letters, corrective actions
Testing capabilities: In-house vs outsourced, scope of testing
Manufacturing technology: Manual vs automated filling systems
Corporate history: Parent companies, legal issues, leadership background
Product specifications: Concentrations, vial sizes, beyond use dating
Formulation approach: Standalone vs combination products
Documentation: Certificates of analysis availability, batch testing results
BPI Labs scores well on manufacturing technology, testing capabilities, and production scale. They face more scrutiny on corporate history and FDA inspection findings. Every compounder has strengths and weaknesses. The goal is understanding the full picture.
Evaluating any compounding pharmacy for tirzepatide
Whether you choose BPI Labs or any other source, the evaluation process should follow the same rigorous approach. SeekPeptides provides comprehensive resources to help researchers navigate these decisions with confidence.
Quality indicators that matter
Start with verifiable credentials. Is the pharmacy FDA-registered? Can you confirm their 503B status on the FDA Outsourcing Facility database? Do they provide certificates of analysis (COAs) for each batch? Are those COAs from in-house testing or third-party laboratories?
Next, examine their product specifications. What are the exact concentrations? What is the beyond use date? How is the product shipped? What temperature controls are maintained during transit? These details directly affect whether the tirzepatide you receive will perform as expected.
Manufacturing technology matters too. Robotic filling is not universally better than manual filling, but it does reduce certain categories of risk. Understanding the production process helps researchers make informed comparisons. Use the peptide calculator to verify that the concentrations and volumes you receive match expected dosing requirements.
Red flags that demand caution
Some warning signs should immediately raise concerns about any compounding pharmacy, BPI Labs or otherwise:
Inability or unwillingness to provide certificates of analysis. Vague or missing information about manufacturing processes. No verifiable FDA registration. Pricing dramatically below market averages without explanation. Claims of proprietary formulations without published supporting data. Aggressive marketing language promising specific clinical outcomes.
If a compounding pharmacy cannot answer basic questions about their testing protocols, their raw material sourcing, or their FDA inspection history, that silence is itself informative. Legitimate operations have nothing to hide from informed researchers.
Questions to ask before ordering
Before sourcing tirzepatide from any pharmacy, researchers should ask direct questions:
What is your current FDA registration status? When was your last FDA inspection, and what were the findings? Can you provide a certificate of analysis for the specific batch I would receive? What is the beyond use date, and what stability data supports it? What raw material suppliers do you use, and how are they vetted? What happens if I experience an adverse event, and how are adverse events reported?
These questions are not adversarial. They reflect responsible research practices. Any pharmacy that welcomes these questions and provides transparent answers deserves more confidence than one that deflects or provides vague responses. For those managing ongoing protocols, our guide on tirzepatide effectiveness helps troubleshoot beyond sourcing issues.
Managing compounded tirzepatide effectively
Regardless of the source, compounded tirzepatide requires careful management to achieve optimal results. Sourcing from BPI Labs or any other pharmacy is only the first step. How you handle, store, dose, and monitor your protocol matters equally.
Dosing considerations
BPI Labs tirzepatide products arrive pre-mixed and ready to use, which simplifies the dosing process. But researchers still need to calculate correct injection volumes based on their prescribed dose and the vial concentration. A 10mg per mL vial requires different volumes than a 20mg per mL vial for the same target dose.
Standard tirzepatide titration typically starts at 2.5mg and increases every four weeks through established dose levels. Use our compounded tirzepatide dosage calculator to determine exact injection volumes based on your specific vial concentration. For unit-based dosing references, see our guides on how many units is 2.5mg of tirzepatide, how many units is 5mg, and how many units is 7.5mg.
Researchers who are transitioning from semaglutide to tirzepatide should review the semaglutide to tirzepatide conversion chart before starting their BPI Labs protocol. The dose equivalences are not straightforward, and improper conversion can lead to underdosing or excessive side effects.
Side effect management
Even with the Vitamin B6 addition in BPI Labs formulations, side effects are a normal part of tirzepatide protocols, especially during titration. The most common issues include nausea, constipation, decreased appetite, and injection site reactions. Less common but reported effects include anxiety, insomnia, and muscle pain.
Dietary management plays a significant role in tolerability. Our guides on foods to avoid on tirzepatide and what to eat on tirzepatide provide evidence-based nutritional strategies that complement any compounded tirzepatide protocol. Many researchers find that following a structured tirzepatide diet plan significantly reduces GI symptoms.
For those who want targeted nutritional support alongside their tirzepatide protocol, our guide to supplements to take with tirzepatide covers evidence-based options beyond the B6 already included in BPI Labs formulations.
Proper storage and handling
BPI Labs tirzepatide formulations ship without refrigeration requirements, but proper storage after receipt follows standard tirzepatide refrigeration guidelines. Keep vials refrigerated between 2 and 8 degrees Celsius for optimal stability. Brief periods at room temperature for dosing preparation are acceptable, but extended exposure to heat can degrade the peptide.
The 12-month beyond use date assumes proper storage conditions are maintained. Storing vials outside recommended temperature ranges will reduce effective shelf life regardless of the date printed on the label. For researchers who travel or have variable storage conditions, our guide on how long compounded tirzepatide can be out of the fridge provides practical guidance.
Injection technique also matters. Proper subcutaneous injection practices maximize absorption and minimize injection site reactions. For detailed guidance, see our guides on how to inject tirzepatide in the stomach and where to inject GLP-1 medications for optimal results.
The bigger picture for tirzepatide research
BPI Labs operates at the intersection of pharmaceutical innovation and regulatory complexity. Their manufacturing technology is genuinely advanced. Their product formulations offer real conveniences like ready-to-use formats and extended shelf life. And their willingness to challenge Eli Lilly patents demonstrates commitment to maintaining compounded tirzepatide access.
At the same time, the corporate history behind Belcher Pharmaceuticals, the FDA inspection findings, and the unsettled regulatory status of combination formulations all warrant careful consideration. No compounding pharmacy is perfect. The question is whether a given facility strengths outweigh its weaknesses for your specific needs and risk tolerance.
The tirzepatide compounding market continues to evolve rapidly. Patent challenges, FDA enforcement actions, state-level regulations, and new formulation approaches are all in flux. What is true today may change in weeks or months. Staying informed requires ongoing attention, not a one-time evaluation.
For researchers interested in alternative delivery methods, BPI Labs injectable formulations represent one approach, but the market also includes oral tirzepatide, tirzepatide drops, and tablet formulations. Each delivery method carries its own set of considerations regarding bioavailability, convenience, and cost. And for those comparing GLP-1 options beyond tirzepatide, our analyses of survodutide vs tirzepatide, mazdutide vs tirzepatide, and orforglipron vs tirzepatide provide comprehensive comparisons.
SeekPeptides members access detailed vendor analyses, dosing protocols, safety databases, and community insights that help navigate these complex sourcing decisions. For researchers serious about optimizing their peptide protocols, having access to comprehensive, continuously updated research resources is not a luxury, it is a necessity. SeekPeptides provides the most thorough evidence-based guidance available, with protocols, comparison tools, and a community of experienced researchers who have navigated these exact decisions.
Frequently asked questions
Is BPI Labs FDA approved?
BPI Labs is FDA-registered as both a drug manufacturer and a 503B outsourcing facility. This is different from FDA approval of specific products. FDA registration means the facility meets regulatory requirements for operation and is subject to FDA inspection. Individual compounded products from 503B facilities are not FDA-approved in the same way that brand-name drugs like Mounjaro receive approval. The distinction matters for understanding the regulatory framework around compounded tirzepatide.
What strengths of tirzepatide does BPI Labs offer?
BPI Labs produces tirzepatide with Vitamin B6 in multiple strength configurations, including formulations at 10mg per mL concentration. The product is available in both 1mL and 3mL vial sizes. Higher concentration options may also be available. Check our tirzepatide dosage chart in mL for help calculating injection volumes across different concentrations.
Does BPI Labs tirzepatide need to be refrigerated?
BPI Labs ships their tirzepatide products without refrigeration during transit. After receipt, standard refrigeration between 2 and 8 degrees Celsius is recommended for optimal stability. The product carries a 12-month beyond use date when stored properly. For detailed storage guidance, see our complete guide on tirzepatide refrigeration requirements.
Why does BPI Labs add Vitamin B6 to tirzepatide?
Vitamin B6 (pyridoxine) is included to help manage nausea, one of the most common side effects of GLP-1 receptor agonists like tirzepatide. Pyridoxine has established anti-nausea properties, particularly through its role in neurotransmitter synthesis. The addition also creates a formulation that differs from commercially available branded tirzepatide, which has regulatory implications under the "not essentially a copy" provision. Other compounders use similar strategies with B12 or glycine additions.
How does BPI Labs compare to Empower Pharmacy for tirzepatide?
Both are 503B outsourcing facilities producing compounded tirzepatide. BPI Labs differentiates with robotic aseptic filling technology and dual FDA registration as both a manufacturer and compounder. Empower Pharmacy has a larger public profile and more extensive telehealth partnerships. Both face similar regulatory challenges and are both involved in challenging Eli Lilly tirzepatide patents. For detailed Empower tirzepatide dosing comparisons, see our dedicated guide.
Can I still get compounded tirzepatide legally?
The legal landscape for compounded tirzepatide is complex and evolving. The FDA ended enforcement discretion for tirzepatide compounding in early 2025 after the drug shortage ended. However, pharmacies may still compound tirzepatide under certain conditions, such as when a physician determines a patient requires a significantly different formulation. The combination with Vitamin B6 represents one approach to meeting this requirement. Researchers should consult healthcare providers about current tirzepatide dosing options and legal availability in their state.
What is BPI Labs connection to Belcher Pharmaceuticals?
BPI Labs LLC is a wholly owned subsidiary of Belcher Pharmaceuticals LLC. Belcher provides the manufacturing infrastructure and regulatory framework under which BPI Labs operates. The corporate lineage also connects to GeoPharma Inc., a former parent entity with a complex regulatory and legal history. These connections are publicly documented and should be considered as part of any vendor evaluation.
Has BPI Labs received FDA warning letters?
As of the most recent available information, neither BPI Labs nor Belcher Pharmaceuticals has received an FDA Warning Letter. BPI Labs has received Form 483 observations during FDA inspections in 2021 and 2025. Form 483 observations are findings noted during inspection, while Warning Letters represent formal regulatory action. The distinction is important for accurate assessment of compliance status. For broader context on evaluating compounding pharmacies, see our guide comparing GLP-1 options and safety.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your sources stay verified, your formulations stay potent, and your protocols stay informed.