Feb 26, 2026

You have done the research. You have read the studies. You have heard the success stories from people who lost 15, 20, even 50 pounds on semaglutide. And now you want in. But the path from "interested" to "actually getting a prescription" feels like navigating a maze with no map. BMI thresholds, insurance requirements, medical evaluations, blood work, prior authorizations. The list goes on. And every source you read seems to give slightly different answers about who actually qualifies.
Here is the truth. Qualifying for semaglutide is more straightforward than most people think, but only if you understand exactly what providers and insurance companies are looking for. The criteria are specific. The process has defined steps. And the requirements vary depending on whether you are pursuing brand-name Wegovy or Ozempic, compounded semaglutide, or an off-label prescription.
This guide breaks down every qualification pathway available. You will learn the exact BMI requirements, which medical conditions strengthen your case, how the prescription process works from first appointment to first injection, and what disqualifies people entirely. Whether you are working with your primary care physician, exploring telehealth options, or considering compounded alternatives, SeekPeptides has mapped out the complete process so you can walk into your appointment prepared and confident.
No guessing. No conflicting information. Just the clear, specific criteria that determine whether semaglutide is an option for you.
BMI requirements for semaglutide
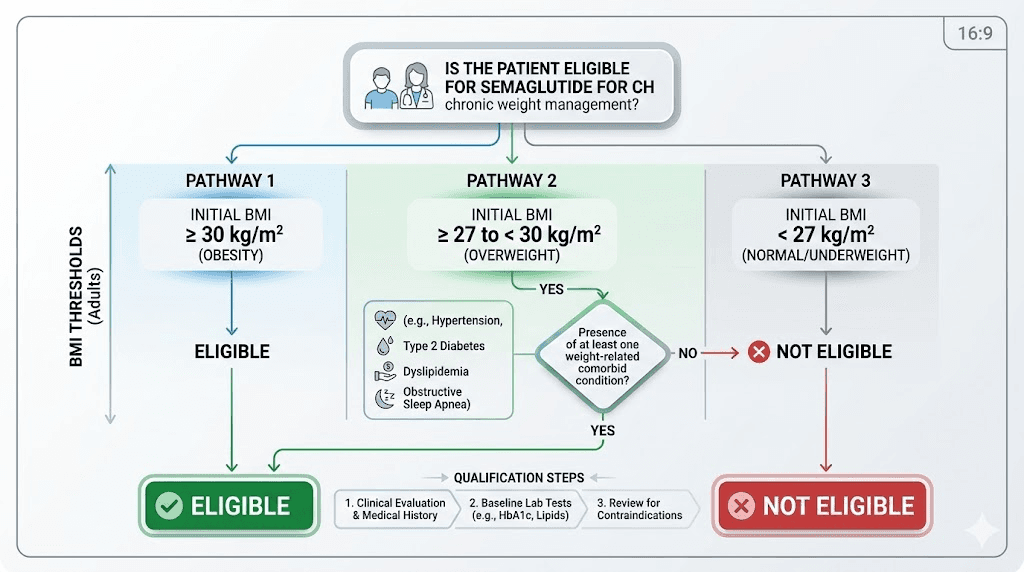
Body mass index is the single most important number in the semaglutide qualification process. Every prescriber, every insurance company, and every telehealth platform starts here. Your BMI determines which door opens for you, and understanding the exact thresholds can mean the difference between approval and denial.
BMI is calculated by dividing weight in kilograms by height in meters squared. For most adults, the number falls somewhere between 18.5 and 40+. The FDA and healthcare providers use specific BMI cutoffs to determine who qualifies for semaglutide treatment.
BMI of 30 or higher (obesity classification)
If your BMI is 30 or above, you meet the primary qualification threshold for semaglutide weight loss treatment. Full stop. This is the cleanest path to a prescription because you do not need any additional qualifying conditions. The FDA approved Wegovy specifically for adults with a BMI of 30 or higher, recognizing obesity as a chronic medical condition that warrants pharmacological intervention.
What does a BMI of 30 look like in practice? A person who stands 5 feet 6 inches tall would need to weigh approximately 186 pounds or more. At 5 feet 10 inches, the threshold is around 209 pounds. These numbers might surprise you. Many people who consider themselves "a little overweight" actually fall into the obesity BMI category, which means they qualify without needing additional medical conditions on their chart.
Providers who prescribe semaglutide for weight management generally approve patients with a BMI of 30+ without extensive justification. The semaglutide dosage calculator can help you understand what your starting protocol might look like once approved.
BMI of 27 to 29.9 with weight-related conditions
This is where qualification gets more nuanced. A BMI between 27 and 29.9 puts you in the "overweight" category, which alone does not qualify you for semaglutide. However, if you have at least one weight-related medical condition, you become eligible.
The qualifying comorbidities include type 2 diabetes, hypertension (high blood pressure), dyslipidemia (high cholesterol or triglycerides), obstructive sleep apnea, and cardiovascular disease. Having just one of these conditions alongside a BMI of 27+ opens the door. Many adults in this BMI range have at least one of these conditions without even knowing it, which is why blood work during the qualification process is so important.
Understanding how semaglutide works over time can help you set realistic expectations if you qualify through this pathway. Results at the lower BMI threshold may look different than results for patients starting at a BMI of 35 or 40, but one-month semaglutide results show meaningful progress across all qualifying BMI ranges.
How to calculate your BMI accurately
Before your appointment, know your number. The formula is straightforward.
Take your weight in pounds. Multiply it by 703. Then divide by your height in inches squared. A person who weighs 200 pounds and stands 5 feet 8 inches (68 inches) would calculate: 200 times 703 equals 140,600. Then 68 squared equals 4,624. Divide 140,600 by 4,624, and you get a BMI of approximately 30.4. That qualifies.
If math is not your thing, the peptide calculator tools at SeekPeptides can help with various calculations related to peptide protocols. For BMI specifically, nearly every healthcare provider has a calculator built into their intake system. You can also use the NIH BMI calculator online before your appointment so you know exactly where you stand.
One important note. BMI is an imperfect measurement. It does not account for muscle mass, bone density, or body composition. A muscular athlete might have a BMI of 30 without being obese. Most providers understand this limitation and will consider your overall health picture, not just the number. However, for insurance and prescription purposes, BMI remains the gatekeeping metric. If you are borderline, being weighed in heavier clothing or at a time of day when you typically weigh more can make a legitimate difference.
BMI thresholds comparison table
BMI Range | Classification | Semaglutide Eligible? | Additional Requirements |
|---|---|---|---|
Below 25 | Normal weight | Generally no | Not eligible for standard prescription |
25 to 26.9 | Overweight | Rarely | Some compounded providers may consider with conditions |
27 to 29.9 | Overweight | Yes, with conditions | Must have at least one weight-related comorbidity |
30 to 34.9 | Obesity Class I | Yes | No additional conditions required |
35 to 39.9 | Obesity Class II | Yes | No additional conditions required |
40+ | Obesity Class III | Yes | No additional conditions required, highest priority |
Understanding where you fall on this chart is the first step. The next step is knowing which specific medical conditions can qualify you if your BMI falls in that 27-to-29.9 zone.

Weight-related medical conditions that qualify you
If your BMI sits between 27 and 29.9, one qualifying medical condition is all you need. But even if your BMI is above 30, having documented comorbidities strengthens your case with insurance companies and can expedite the prior authorization process. Here is a detailed look at each qualifying condition.
Type 2 diabetes
This is the most straightforward qualifying condition because semaglutide was originally developed and FDA-approved for type 2 diabetes management. Ozempic, the diabetes formulation, received FDA approval in 2017 specifically for improving glycemic control. If you have a type 2 diabetes diagnosis, you qualify for semaglutide regardless of whether your primary goal is blood sugar management or weight loss.
An A1C level of 6.5% or higher confirms a diabetes diagnosis. Fasting blood glucose above 126 mg/dL also qualifies. If your A1C falls between 5.7% and 6.4%, you are in the prediabetes range, which some providers will accept as a qualifying condition, particularly when combined with other risk factors. The connection between semaglutide appetite suppression and improved blood sugar control makes this medication uniquely valuable for diabetic patients.
High blood pressure (hypertension)
Hypertension affects nearly half of all American adults, making it one of the most common qualifying conditions. A blood pressure reading consistently at or above 130/80 mmHg meets the diagnostic criteria. Many people with elevated BMI have undiagnosed hypertension, which is why blood pressure measurement during your qualification appointment can actually work in your favor.
If you are already taking blood pressure medication, that counts. You do not need to have uncontrolled hypertension. The fact that you have a diagnosis and are managing it pharmaceutically is sufficient documentation. Studies show that semaglutide treatment often leads to blood pressure improvements as weight decreases, making it a particularly logical treatment for patients with both obesity and hypertension.
High cholesterol (dyslipidemia)
Abnormal cholesterol levels qualify you for semaglutide when combined with an overweight BMI. The specific thresholds include total cholesterol above 200 mg/dL, LDL cholesterol above 130 mg/dL, HDL cholesterol below 40 mg/dL for men or below 50 mg/dL for women, and triglycerides above 150 mg/dL.
Like hypertension, if you are already taking statins or other cholesterol-lowering medications, the existing diagnosis qualifies you. Your provider does not need to see actively elevated numbers on your lab work. The prescription history in your medical records serves as documentation of the condition.
Obstructive sleep apnea
Sleep apnea is strongly correlated with excess weight, and a diagnosis can qualify you for semaglutide treatment. If you use a CPAP machine, have had a sleep study confirming sleep apnea, or have a documented diagnosis in your medical records, this counts as a qualifying comorbidity.
Many people with undiagnosed sleep apnea experience symptoms like persistent fatigue, morning headaches, loud snoring, and excessive daytime sleepiness. If you experience these symptoms but have not been formally diagnosed, mentioning them to your provider during the qualification process could lead to screening, potentially adding a qualifying condition to your chart. Weight loss through semaglutide treatment often improves or even resolves mild sleep apnea.
Cardiovascular disease
A history of cardiovascular events or diagnosed cardiovascular disease represents a strong qualifying condition. This includes coronary artery disease, previous heart attack, stroke, peripheral arterial disease, and heart failure. The Wegovy FDA label was actually expanded in March 2024 to include cardiovascular risk reduction as an approved indication, based on the SELECT trial showing a 20% reduction in major adverse cardiovascular events.
If you have established cardiovascular disease, semaglutide is not just a weight loss tool. It becomes a cardioprotective intervention. This dual benefit makes qualification through cardiovascular disease particularly compelling for both providers and insurance companies.
Other qualifying conditions
Several additional conditions may qualify you depending on your provider and insurance plan. These include nonalcoholic fatty liver disease (now called metabolic dysfunction-associated steatohepatitis or MASH), polycystic ovary syndrome (PCOS), osteoarthritis of weight-bearing joints, and metabolic syndrome. The broader metabolic effects of GLP-1 medications make them relevant across multiple conditions connected to excess weight.
Some providers also consider prediabetes, insulin resistance confirmed by lab work, and a strong family history of obesity-related diseases as supporting factors, even if they do not technically meet the strict comorbidity definition for every insurance plan.
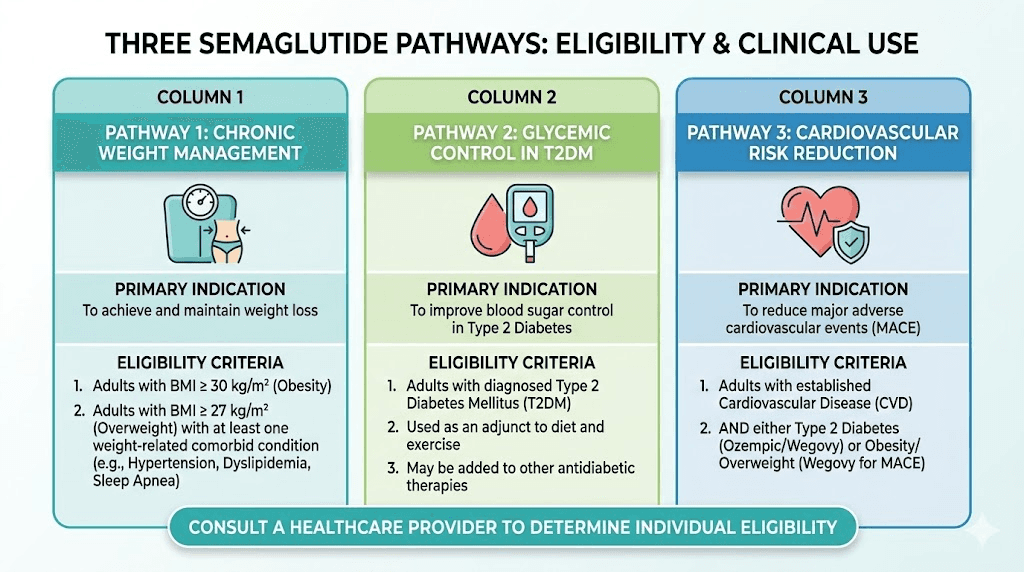
Wegovy vs Ozempic vs compounded semaglutide eligibility
Not all semaglutide is created equal when it comes to qualification criteria. The three main pathways, Wegovy, Ozempic, and compounded formulations, each have different eligibility requirements, different approval processes, and different cost structures. Understanding which pathway fits your situation can save you months of frustration.
Wegovy (FDA-approved for weight management)
Wegovy is the gold standard for semaglutide weight loss treatment. FDA-approved specifically for chronic weight management, it carries the most straightforward eligibility criteria for weight loss purposes. Adults with a BMI of 30+ or BMI of 27+ with at least one weight-related comorbidity qualify for Wegovy. Children ages 12 and older with obesity (BMI at or above the 95th percentile for age and sex) also qualify.
Wegovy reaches a maximum maintenance dose of 2.4 mg per week, which is higher than the maximum Ozempic dose of 2 mg. This higher ceiling means potentially greater weight loss results. Clinical trials showed average weight loss of approximately 15% of body weight over 68 weeks at the 2.4 mg dose.
The challenge with Wegovy is availability and cost. Supply shortages have been common since its launch. Without insurance, a monthly supply costs approximately $1,300 to $1,600. Insurance coverage varies wildly, which is why understanding the insurance landscape for GLP-1 medications is critical before pursuing this pathway.
Ozempic (FDA-approved for type 2 diabetes)
Ozempic shares the same active ingredient as Wegovy but carries an FDA approval specifically for type 2 diabetes. To qualify for an on-label Ozempic prescription, you need a type 2 diabetes diagnosis. Period. BMI is not a requirement for Ozempic when prescribed for diabetes management.
Here is where things get interesting. Many providers prescribe Ozempic off-label for weight loss in patients who do not have diabetes. Off-label prescribing is legal and common in medicine. However, insurance companies are much less likely to cover Ozempic when prescribed off-label for weight loss. If your provider writes an off-label Ozempic prescription, expect to pay out of pocket in most cases.
The relationship between semaglutide and tirzepatide is worth understanding here. Both are GLP-1 receptor agonists with weight loss benefits, but they have different mechanisms and eligibility pathways. If you do not qualify for one, you might qualify for the other.
Compounded semaglutide (alternative access pathway)
Compounded semaglutide exists because brand-name supply has not kept up with demand. Compounding pharmacies create semaglutide formulations that are typically more affordable than brand-name options, though they are not FDA-approved products. The complete guide to compounded semaglutide covers the nuances in detail.
Eligibility requirements for compounded semaglutide vary by provider but are generally more flexible than brand-name requirements. Many telehealth platforms that prescribe compounded semaglutide will consider patients with a BMI as low as 25 if they have weight-related health conditions. Some even consider patients with a BMI above 22 if they have a history of obesity and previous GLP-1 use within the past 12 months.
Important caveat. Compounded semaglutide can only legally be prescribed when the brand-name versions (Ozempic and Wegovy) are on the FDA Drug Shortage List. This status has been fluctuating, so availability is not guaranteed long-term. When pursuing this pathway, understanding storage requirements and shelf life becomes essential for maintaining medication effectiveness.
Many compounded formulations include additional ingredients like vitamin B12, glycine, or methylcobalamin to enhance absorption or reduce side effects. These combination formulations may have slightly different eligibility considerations.
Feature | Wegovy | Ozempic | Compounded Semaglutide |
|---|---|---|---|
FDA-approved indication | Weight management | Type 2 diabetes | Not FDA-approved |
BMI requirement | 30+ or 27+ with conditions | No BMI requirement (diabetes diagnosis needed) | Varies by provider, often 25+ |
Maximum dose | 2.4 mg/week | 2 mg/week | Varies by pharmacy |
Insurance coverage | Limited but growing | Good for diabetes | Not covered |
Approximate monthly cost (without insurance) | $1,300-$1,600 | $900-$1,200 | $150-$500 |
Availability | Supply shortages common | Generally available | Dependent on shortage status |
The qualification process step by step
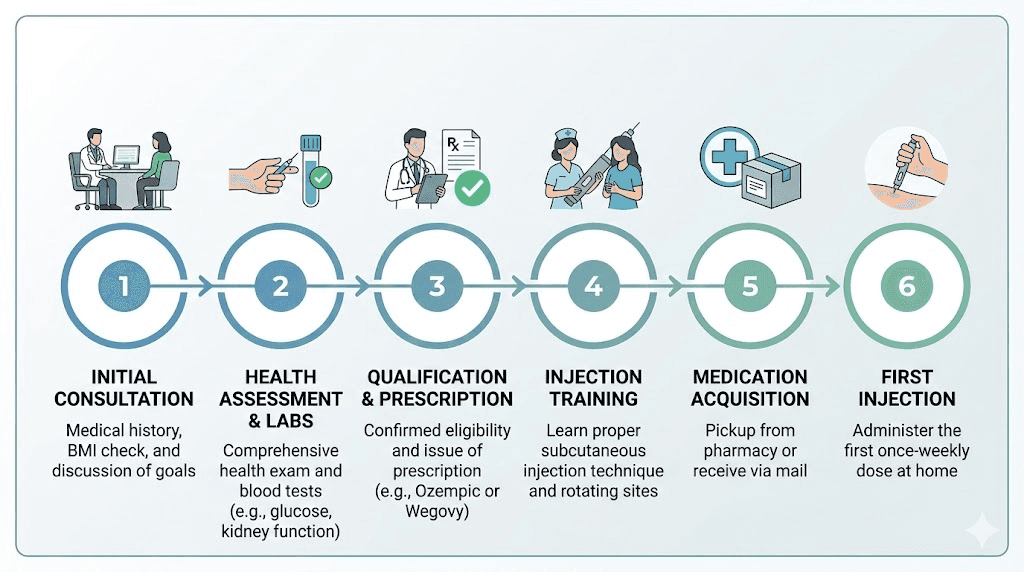
Knowing you meet the criteria and actually getting a prescription are two different things. The qualification process involves specific steps, and being prepared for each one dramatically increases your chances of approval. Whether you are seeing your primary care doctor or using a telehealth platform, here is what to expect.
Step 1: Choose your provider pathway
You have three main options for obtaining a semaglutide prescription. Your primary care physician, an obesity medicine specialist or endocrinologist, or a telehealth weight loss platform. Each has advantages and drawbacks.
Primary care physicians know your full medical history, which can expedite the qualification process if you have documented comorbidities. However, some PCPs are uncomfortable prescribing GLP-1 medications or unfamiliar with the qualification criteria. If your PCP seems hesitant, knowing what to say when requesting GLP-1 options can make the conversation more productive.
Obesity medicine specialists and endocrinologists are the most knowledgeable about semaglutide prescribing. They handle these prescriptions daily and are well-versed in navigating insurance requirements. The downside is wait times for appointments can stretch weeks or months, and you may need a referral.
Telehealth platforms offer the fastest path to a prescription. Companies like Ro, Hims/Hers, and others can complete the entire qualification process in days rather than weeks. They typically prescribe compounded semaglutide or, in some cases, brand-name medications. The trade-off is less personalized care and limited physical examination capabilities.
Step 2: Gather your medical documentation
Before your appointment, collect everything that supports your case. This includes a list of current medications (prescription and over-the-counter), your weight history over the past several years, documentation of previous weight loss attempts (diets, exercise programs, other medications), records of any weight-related medical conditions, and recent lab work if available.
The previous weight loss attempts piece is particularly important. Many insurance companies require documentation that you have tried and failed lifestyle modifications before approving pharmacological treatment. Having records of past dieting efforts, gym memberships, nutritionist visits, or previous weight loss programs strengthens your application considerably.
Step 3: Initial consultation and health assessment
During the actual appointment, your provider will take vital signs including blood pressure, heart rate, and current weight. They will calculate your BMI and review your medical history in detail. This is where your preparation pays off.
Be honest and thorough about your weight history, eating habits, physical activity level, and how excess weight affects your daily life. Providers need to understand the full picture to make an appropriate prescribing decision. Mention any symptoms that might indicate undiagnosed conditions, such as snoring (sleep apnea), frequent headaches, or joint pain from carrying excess weight.
Your provider will also assess your mental health. Some providers require screening for eating disorders and depression before prescribing GLP-1 medications, as these conditions can affect treatment safety and outcomes.
Step 4: Blood work and laboratory testing
Most providers order baseline blood work before prescribing semaglutide. Standard panels include fasting blood glucose and A1C to assess diabetes status, a comprehensive metabolic panel to check kidney and liver function, a lipid panel for cholesterol and triglyceride levels, and thyroid function tests (TSH at minimum).
These labs serve dual purposes. First, they can reveal qualifying comorbidities you did not know you had. It is remarkably common for overweight patients to discover elevated blood sugar, high cholesterol, or thyroid abnormalities during this testing. Second, they establish baseline values that your provider will monitor throughout treatment to ensure safety.
Some providers also check amylase and lipase levels (pancreatic enzymes) as a baseline, since pancreatitis is a rare but serious potential side effect. Understanding the full spectrum of semaglutide side effects helps you have informed conversations with your provider during this process.
Step 5: Discussion of goals and treatment plan
If your labs and examination support semaglutide use, your provider will discuss treatment goals and expectations. This typically includes target weight loss (realistic expectations are 10-15% of body weight over the first year), lifestyle modifications you will implement alongside the medication, the dosing titration schedule, potential side effects and how to manage them, and follow-up appointment frequency.
Understanding the semaglutide dosing structure before this conversation helps you ask informed questions. Most patients start at a low dose (0.25 mg weekly for Wegovy) and gradually increase over 16-20 weeks to minimize gastrointestinal side effects. Your provider should explain this titration schedule in detail.
This is also the time to discuss how long you will stay on semaglutide. Treatment is typically long-term, as weight regain is common after discontinuation. Having clear expectations about the timeline helps you commit to the process.
Step 6: Prescription and prior authorization
If your provider determines semaglutide is appropriate, they will write a prescription. For brand-name Wegovy or Ozempic, this often triggers a prior authorization process with your insurance company. Prior authorization can take anywhere from 24 hours to several weeks depending on your insurer.
During prior authorization, your insurance company reviews the medical justification for the prescription. They check your BMI, documented comorbidities, history of lifestyle modification attempts, and whether you meet their specific coverage criteria. Having all documentation in order before this step dramatically reduces delays and denials.
If you are pursuing compounded semaglutide, there is typically no prior authorization required. The provider prescribes directly to a compounding pharmacy, and you can often begin treatment within days. Understanding how to reconstitute semaglutide and proper injection technique becomes important at this stage.
Who should not take semaglutide
Not everyone who meets the BMI and comorbidity criteria can safely take semaglutide. Several conditions represent absolute contraindications, meaning the medication should never be used. Others are relative contraindications where the decision requires careful risk-benefit analysis. Understanding these exclusions before pursuing qualification saves time and protects your health.
Personal or family history of medullary thyroid carcinoma
This is the most significant absolute contraindication. In rodent studies, semaglutide caused thyroid C-cell tumors, including medullary thyroid carcinoma (MTC). While the relevance to humans is not established, the FDA placed a black box warning on all GLP-1 receptor agonists regarding this risk.
If you or a first-degree relative (parent, sibling, child) have been diagnosed with MTC, semaglutide is off the table. No exceptions. Your provider will ask about thyroid cancer history during the qualification process, and any indication of MTC risk will result in automatic disqualification.
Multiple endocrine neoplasia syndrome type 2 (MEN 2)
MEN 2 is a genetic condition that dramatically increases the risk of developing MTC along with other endocrine tumors. If you have MEN 2, semaglutide carries an unacceptable risk. This condition is relatively rare, affecting approximately 1 in 35,000 people, but your provider will screen for it during the qualification process.
History of pancreatitis
Pancreatitis, inflammation of the pancreas, represents a serious concern with GLP-1 medications. Reports of acute pancreatitis have been documented in patients taking semaglutide, though the causal relationship is still being studied. If you have a history of pancreatitis, most providers will either decline to prescribe semaglutide or require extensive additional monitoring.
Symptoms of pancreatitis include severe abdominal pain that radiates to the back, nausea, vomiting, and elevated pancreatic enzymes. If these symptoms develop during treatment, semaglutide should be discontinued immediately. Understanding potential gastrointestinal effects helps you distinguish between common side effects and warning signs that require medical attention.
Pregnancy and breastfeeding
Semaglutide is contraindicated during pregnancy. Animal studies have shown adverse fetal effects at clinically relevant doses. The FDA recommends discontinuing semaglutide at least two months before a planned pregnancy. This is especially important to understand given the hormonal effects semaglutide can have on the menstrual cycle, including increased fertility in some women.
For breastfeeding mothers, semaglutide is also not recommended. There is insufficient data on whether semaglutide passes into breast milk or what effects it might have on nursing infants. If you are of childbearing age, your provider will discuss contraception options as part of the qualification process.
Type 1 diabetes
Semaglutide is not approved for and should not be used to treat type 1 diabetes. Type 1 diabetes involves autoimmune destruction of insulin-producing beta cells, which is fundamentally different from the insulin resistance that characterizes type 2 diabetes. Using semaglutide in type 1 diabetes could lead to diabetic ketoacidosis, a potentially life-threatening complication.
Severe gastrointestinal conditions
Gastroparesis (delayed stomach emptying), inflammatory bowel disease, and severe gastroesophageal reflux disease can all be worsened by semaglutide. The medication slows gastric emptying as part of its mechanism of action, which can exacerbate existing GI conditions. If you have a diagnosed gastrointestinal condition, your provider will evaluate whether semaglutide is safe for your specific situation.
Common side effects like nausea, dizziness, and constipation are distinct from serious GI contraindications. Most patients experience mild GI effects during the titration phase that resolve within a few weeks, while pre-existing severe GI conditions represent a more fundamental concern.
Kidney and liver disease
Severe kidney impairment (eGFR below 15 mL/min) or end-stage renal disease typically disqualifies patients from semaglutide treatment. The medication is not dialyzable, and impaired kidney function affects drug clearance. Moderate kidney impairment may be acceptable with dose adjustments and close monitoring.
Similarly, severe liver disease or cirrhosis is generally a disqualifier. Mild to moderate liver disease, including fatty liver, is usually acceptable and may actually benefit from semaglutide-induced weight loss.
Complete contraindications summary
Contraindication | Type | Action |
|---|---|---|
Personal/family MTC history | Absolute | Do not prescribe under any circumstances |
MEN 2 syndrome | Absolute | Do not prescribe under any circumstances |
Current pregnancy | Absolute | Discontinue immediately, wait 2+ months before conceiving |
Type 1 diabetes | Absolute | Do not prescribe, risk of diabetic ketoacidosis |
History of pancreatitis | Relative | Prescribe with extreme caution, enhanced monitoring |
Severe kidney disease | Relative | Generally avoid, case-by-case evaluation |
Severe liver disease | Relative | Generally avoid, case-by-case evaluation |
Gastroparesis | Relative | Prescribe with caution, monitor GI symptoms closely |
Breastfeeding | Relative | Not recommended, insufficient safety data |
Active eating disorder | Relative | Requires mental health clearance first |
Insurance coverage and prior authorization
Qualifying medically for semaglutide is only half the battle. The other half involves navigating insurance coverage, a process that can feel more complicated than the medical evaluation itself. Coverage varies dramatically between plans, employers, and states, making it essential to understand your specific situation before assuming anything about cost.
Private insurance coverage landscape
Coverage for GLP-1 medications has been expanding but remains inconsistent. For Ozempic prescribed for type 2 diabetes, most commercial insurance plans provide coverage, often with a specialty tier copay ranging from $25 to $150 per month. For Wegovy prescribed for weight management, coverage is more limited. Many employers specifically exclude weight loss medications from their benefit plans.
Recent legislative pressure and the growing recognition of obesity as a chronic disease have been pushing more insurers to cover GLP-1 medications for weight loss. The Blue Cross Blue Shield GLP-1 coverage guide provides specific information for one of the largest insurers, but coverage details change frequently.
Before starting the qualification process, call your insurance company directly and ask three specific questions. First, does your plan cover Wegovy or Ozempic? Second, what are the specific coverage criteria (BMI thresholds, required comorbidities, required prior weight loss attempts)? Third, is prior authorization required, and if so, what documentation is needed?
Medicare coverage updates
Medicare coverage of GLP-1 medications has been a rapidly evolving area. Historically, Medicare Part D did not cover medications prescribed solely for weight loss. However, Wegovy gained a cardiovascular risk reduction indication, which opened a pathway for Medicare coverage when prescribed for that purpose.
In 2025, the Centers for Medicare and Medicaid Services (CMS) selected Ozempic, Rybelsus, and Wegovy for Medicare drug price negotiations. Negotiated prices take effect in 2027, with expected costs around $274 for a 30-day supply. This represents a significant reduction from current retail pricing and will dramatically improve access for Medicare beneficiaries.
Additionally, the TREAT Act (Treat and Reduce Obesity Act) has been introduced in Congress to expand Medicare Part D coverage of FDA-approved anti-obesity medications. While not yet passed, this legislation signals growing political momentum toward broader coverage.
Step therapy and prior authorization requirements
Many insurance plans require step therapy before approving semaglutide. This means you must try and fail less expensive treatments before the insurer will cover GLP-1 medications. Common step therapy requirements include documented attempts at diet and exercise programs (typically 3-6 months), trial of older weight loss medications like phentermine or orlistat, enrollment in a behavioral modification program, and evidence of supervised weight loss attempts.
Understanding how phentermine compares to semaglutide can help you navigate step therapy requirements. Some patients find that completing the phentermine step quickly, even if it does not produce significant results, expedites their access to semaglutide.
Prior authorization typically requires your provider to submit clinical documentation including your BMI, documented comorbidities, previous weight loss attempts, and current lab work. The process can take 48 hours to several weeks. If denied, you have the right to appeal, and many initial denials are overturned on appeal with additional documentation.
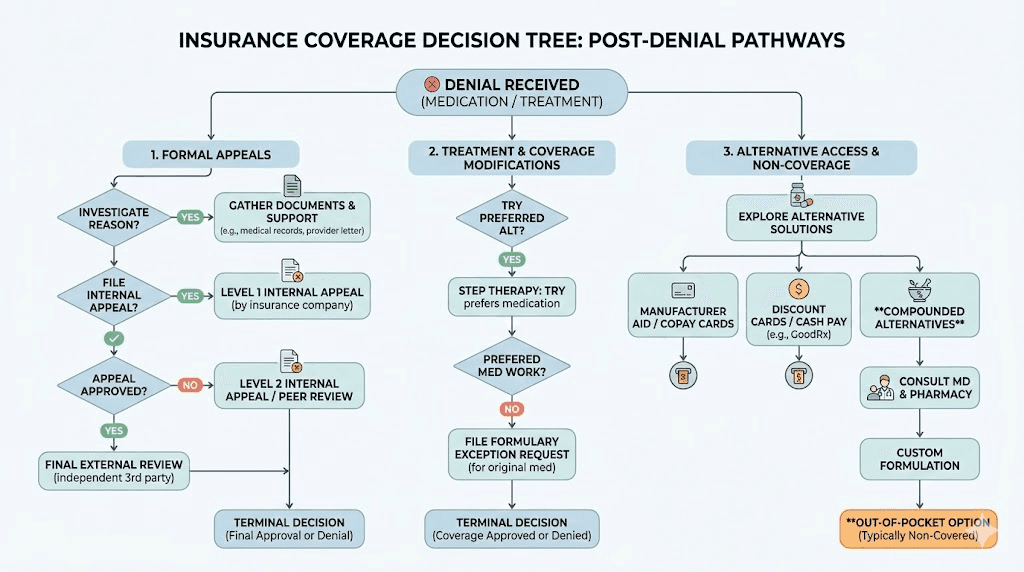
What to do if denied coverage
Insurance denial is not the end of the road. Several strategies can help.
First, appeal the decision. Request a peer-to-peer review where your prescribing physician speaks directly with the insurance company medical director. This personal advocacy often succeeds where paperwork fails.
Second, ask your provider to revise the diagnosis codes or add additional qualifying conditions that were not included in the initial submission. Sometimes a simple coding change resolves the issue.
Third, explore manufacturer savings programs. Novo Nordisk offers the Wegovy savings card program for commercially insured patients, potentially reducing out-of-pocket costs significantly.
Fourth, consider compounded semaglutide as an alternative. The compounded formulation typically costs $150 to $500 per month without insurance, making it more accessible than brand-name options. Many providers who prescribe compounded semaglutide can start the process quickly, and the dosing charts are straightforward to follow.
Fifth, look into clinical trials. Ongoing semaglutide research studies sometimes provide the medication at no cost to qualifying participants.
Telehealth and online qualification options
The rise of telehealth has transformed semaglutide access. What once required multiple in-person visits, lab orders, and weeks of waiting can now happen in days through online platforms. For many people, telehealth represents the fastest and most convenient path to a semaglutide prescription.
How telehealth consultations work
The typical telehealth qualification process follows a streamlined version of the in-person pathway. You complete a detailed medical questionnaire covering your weight history, current medications, medical conditions, and weight loss goals. A licensed healthcare provider reviews your information and conducts a consultation via video call, phone, or secure messaging.
Most platforms require you to submit recent lab work or will order labs through a partner laboratory. Some accept labs completed within the past 6-12 months if they include the necessary panels. Others require fresh labs regardless of recency. The specific requirements depend on the platform and the prescribing provider state regulations.
If the provider determines you qualify, they prescribe semaglutide and send the prescription to a pharmacy. For compounded semaglutide, the medication typically ships directly to your door. For brand-name prescriptions, you may pick up from a local pharmacy or receive it through a specialty pharmacy mail service.
What telehealth providers look for
Online platforms generally assess the same criteria as in-person providers but may have slightly different thresholds. Most telehealth weight loss companies require a minimum BMI of 25 to 30 depending on the platform, adults aged 18 or older, no absolute contraindications, willingness to participate in lifestyle modifications, and the ability to complete required lab work.
Some platforms are more aggressive in their prescribing criteria, accepting lower BMI thresholds or requiring fewer documented comorbidities. Others maintain strict adherence to FDA guidelines. Research the specific platform before investing time in their intake process.
The consultation itself typically involves discussing your diet and nutrition plans, understanding your expectations for the first weeks of treatment, and establishing a monitoring plan for side effects and progress.
Advantages and limitations of online qualification
The advantages are speed and convenience. Many patients go from initial questionnaire to medication delivery in under a week. There is no need to take time off work for in-person appointments. No sitting in waiting rooms. No scheduling difficulties.
The limitations are real, though. Physical examinations are limited to what can be assessed visually through a camera. Complex medical histories may require in-person evaluation. Some states have restrictions on telehealth prescribing for controlled substances or specific medication categories. And the ongoing monitoring may be less comprehensive than what an in-person provider offers.
For straightforward cases, someone with a clear BMI of 30+ and no complex medical history, telehealth is often the optimal pathway. For patients with multiple comorbidities, a history of adverse medication reactions, or borderline qualification criteria, an in-person evaluation provides more thorough assessment.
What to expect after qualifying
Getting the prescription is the beginning, not the end. Understanding what comes next helps you maximize the medication effectiveness and stay on track with your treatment plan. The first several weeks set the tone for your entire semaglutide journey.
Starting dose and titration schedule
Every semaglutide protocol begins with a low dose that gradually increases over time. This titration approach minimizes gastrointestinal side effects and allows your body to adjust to the medication. For Wegovy, the standard titration schedule spans 16-20 weeks.
The semaglutide dosage chart in units breaks down the exact amounts at each stage. Typically, you start at 0.25 mg weekly for four weeks, increase to 0.5 mg for four weeks, then 1 mg for four weeks, followed by 1.7 mg for four weeks, before reaching the maintenance dose of 2.4 mg. Your provider may adjust this schedule based on your tolerance and response.
Understanding unit-to-milligram conversions is important if you are using compounded semaglutide from a vial rather than a pre-filled pen. The semaglutide dosage calculator can help ensure accurate dosing throughout the titration process.
Lifestyle requirements
Semaglutide is not a standalone solution. Every prescribing guideline specifies that it should be used in conjunction with a reduced-calorie diet and increased physical activity. This is not just a box to check. Patients who combine semaglutide with genuine lifestyle changes consistently achieve better results than those who rely on the medication alone.
Understanding which foods to avoid on semaglutide and the best foods to eat makes a significant difference in both tolerability and results. High-fat, greasy foods tend to worsen nausea. Protein-rich meals support muscle preservation during weight loss. The semaglutide diet plan provides detailed nutritional guidance tailored to people on GLP-1 medications.
Exercise does not need to be extreme. Walking 150 minutes per week, roughly 20-30 minutes daily, meets the baseline recommendation. Some patients find that semaglutide actually makes exercise more enjoyable as excess weight decreases and energy levels improve. Understanding whether semaglutide affects energy levels and whether you can lose weight without exercise helps you set realistic expectations.
Follow-up appointments and monitoring
After starting semaglutide, expect follow-up appointments at regular intervals. Most providers schedule check-ins at 4 weeks, 8 weeks, 12 weeks, and then quarterly. These appointments typically include weight measurement and progress assessment, blood pressure monitoring, review of side effects and tolerability, lab work (usually at 3 months and 6 months), and dose adjustment discussions.
If you are not seeing expected results, do not panic. Lack of weight loss in the first four weeks is not uncommon, especially during the low-dose titration phase. Most patients do not see significant movement on the scale until reaching higher doses. The troubleshooting guide for semaglutide weight loss plateaus covers the most common reasons for slow progress and how to address them.
A semaglutide plateau can happen at any stage of treatment. Knowing that this is normal and having strategies to push through can keep you committed to the process when progress stalls temporarily.
Managing common side effects
The most common side effects during the early weeks of treatment are gastrointestinal. Nausea affects approximately 40-45% of patients, though severity varies widely. Constipation, dizziness, and fatigue are also reported. Most side effects improve within the first 4-8 weeks as your body adjusts.
Practical strategies help. Eat smaller meals. Stay hydrated. Avoid lying down immediately after eating. Ginger tea and peppermint can ease nausea naturally. Some patients find that taking semaglutide at a specific time of day reduces side effects. If constipation becomes an issue, increasing fiber intake and water consumption usually resolves it.
The question of alcohol use while on semaglutide comes up frequently. Alcohol tolerance often decreases significantly on GLP-1 medications, and alcohol can worsen nausea and GI effects. Most providers recommend limiting or eliminating alcohol, especially during the titration phase.
Common reasons people get disqualified
Even when you think you meet all the criteria, certain factors can trip up the qualification process. Knowing these pitfalls in advance helps you avoid them or address them proactively.
BMI falls below the threshold
The most common disqualification is simply not meeting the BMI requirement. If your BMI is 26.5 and you do not have documented comorbidities, you will not qualify through standard pathways. Some people are surprised when their measured BMI comes in lower than expected, particularly if they weighed themselves at home using an inaccurate scale or measured their height incorrectly.
If you are borderline, timing matters. BMI naturally fluctuates throughout the day, across the menstrual cycle, and with hydration status. Being weighed in the afternoon rather than first thing in the morning, or after a meal rather than fasting, can legitimately shift your BMI by a point or more. This is not gaming the system. It is simply recognizing that a single measurement represents a snapshot, not a fixed value.
Conflicting medications
Certain medications create interaction concerns with semaglutide. Insulin and other diabetes medications may need dose adjustments when combined with semaglutide, potentially complicating the prescribing decision. Oral medications that require precise absorption timing can be affected by semaglutide-induced delayed gastric emptying.
If you are currently taking phentermine, some providers will not prescribe semaglutide simultaneously, while others consider this combination acceptable. The interaction between berberine and semaglutide is another common question. Discussing all current medications and supplements transparently with your provider prevents surprises during the qualification process.
Insufficient documentation of prior weight loss attempts
Insurance companies frequently deny coverage because the submitted documentation does not adequately demonstrate previous weight loss efforts. A verbal statement of "I have tried dieting" is not sufficient. They want records. Gym memberships, nutritionist visits, WeightWatchers enrollment, documented calorie-restricted diet plans, and records of previous prescription weight loss medications all serve as acceptable documentation.
If you lack formal documentation, start creating it now. Schedule an appointment with a nutritionist. Join a structured weight loss program. Document everything. Even a few months of documented lifestyle intervention can satisfy this requirement for future semaglutide qualification attempts.
Uncontrolled medical conditions
Having qualifying comorbidities helps, but if those conditions are severely uncontrolled, some providers will want to stabilize them before adding semaglutide. For example, wildly uncontrolled diabetes with an A1C above 10% might need insulin stabilization first. Severe uncontrolled hypertension might require blood pressure management before GLP-1 initiation.
This is not a permanent disqualification. It is a "get this under control first, then we will add semaglutide" situation. Working with your provider to manage acute conditions positions you for successful semaglutide qualification once the immediate health concerns are addressed.
Mental health concerns
Active eating disorders, particularly bulimia and anorexia, are relative contraindications for semaglutide. The appetite-suppressing effects of the medication can exacerbate disordered eating patterns. Some providers also screen for severe depression and suicidal ideation, as there have been reports (still under investigation) of potential mood effects with GLP-1 medications.
If you have a history of eating disorders but are currently in recovery and working with a mental health provider, many prescribers will still consider you for semaglutide with appropriate monitoring. Transparency about your mental health history helps your provider make the safest decision.
Tips to increase your chances of qualifying
The qualification process is not just about meeting criteria. It is about presenting your case effectively. These strategies can tip the scales in your favor.
Document everything before your appointment. Create a written weight history covering the past 5-10 years. List every diet, exercise program, and weight loss medication you have tried, with approximate dates and outcomes. Bring records of gym memberships, nutritionist visits, or Weight Watchers enrollment. The more documentation you provide, the stronger your case becomes, especially for insurance approval.
Get comprehensive lab work done in advance. If your primary care provider will order labs before your semaglutide consultation, do this first. Labs revealing previously undiagnosed conditions like prediabetes, high cholesterol, or elevated liver enzymes can add qualifying comorbidities to your chart. Knowledge is leverage in the qualification process.
Choose the right provider. Obesity medicine specialists and endocrinologists approve semaglutide prescriptions at higher rates than general practitioners simply because they are more familiar with the medication and its qualification criteria. If your PCP seems uncertain or resistant, requesting a referral to a specialist can change the outcome entirely.
Be specific about how excess weight affects your life. During your consultation, go beyond "I want to lose weight." Describe how your weight impacts your daily function. Mention joint pain, breathlessness with physical activity, poor sleep, reduced mobility, and emotional effects. These specific descriptions help your provider document medical necessity comprehensively.
Ask about all available formulations. If Wegovy is not covered by your insurance, ask about Ozempic (especially if you have diabetes or prediabetes). If brand-name options are too expensive, discuss compounded semaglutide. If semaglutide does not work out, explore whether tirzepatide might be an alternative. Flexibility in which formulation you are willing to use dramatically expands your options.
Consider the timing of your weigh-in. If you are borderline on BMI, small details matter. Weigh yourself at the same time of day your appointment is scheduled. Wear normal clothing, not workout gear. Do not dehydrate yourself, but be aware that afternoon weigh-ins typically show 2-4 pounds higher than morning weigh-ins. This natural variation is real and significant when you are sitting at a BMI of 29.5.
Prepare for the insurance fight. If you anticipate insurance complications, ask your provider to include all relevant diagnosis codes on the prior authorization submission. Request a letter of medical necessity. Keep copies of all submitted documentation. And know your appeal rights before you receive a denial, so you can respond immediately rather than starting from scratch.
The members at SeekPeptides have navigated these exact challenges. With comprehensive protocol guides, dosing resources, and community support, the platform helps you approach the entire process with confidence and clarity.

Understanding the different semaglutide formulations
The semaglutide landscape has expanded beyond simple weekly injections. Multiple delivery methods exist, each with different qualification considerations and practical implications. Knowing your options helps you have a more productive conversation with your prescribing provider.
Injectable semaglutide
The standard delivery method, and the one with the most clinical data behind it. Injectable semaglutide is administered subcutaneously once weekly, typically in the abdomen, thigh, or upper arm. The best injection sites for semaglutide and proper injection technique are straightforward to learn.
Brand-name injectable options come in pre-filled pens (Wegovy and Ozempic) that make dosing simple. Compounded injectable semaglutide comes in vials that require drawing up the correct dose with a syringe. If you are using vials, understanding reconstitution procedures and mixing charts is essential for accurate dosing.
Whether you are working with a 5mg vial or a 10mg vial, the dosing mathematics need to be precise. Converting between units and milligrams can be confusing at first. Resources like the syringe dosage chart and understanding how many milligrams are in common unit measurements, whether that is 20 units, 40 units, or 50 units, make the process manageable.
Oral semaglutide
Rybelsus is the oral tablet form of semaglutide, FDA-approved for type 2 diabetes. It is taken daily rather than weekly. The bioavailability of oral semaglutide is significantly lower than the injectable form, which is why it requires higher doses to achieve similar effects.
For patients who are needle-averse, oral semaglutide offers a viable alternative. However, it comes with strict administration requirements. It must be taken on an empty stomach with no more than 4 ounces of plain water, and you must wait at least 30 minutes before eating, drinking, or taking other medications. The oral semaglutide complete guide covers the specific protocols in detail.
Qualification criteria for Rybelsus mirror Ozempic since both are approved for type 2 diabetes. Off-label use for weight loss follows the same considerations as off-label Ozempic prescribing.
Sublingual semaglutide
Some compounding pharmacies offer sublingual semaglutide, which is placed under the tongue for absorption. This delivery method aims to combine the convenience of oral administration with better bioavailability than traditional tablets. Qualification criteria for sublingual formulations follow the same general principles as other compounded semaglutide products.
Proper storage after qualifying
Once you have your semaglutide prescription filled, proper storage becomes critical. Understanding refrigeration requirements, knowing how long semaglutide lasts in the fridge, and learning what happens if the medication gets warm protects your investment. The shelf life guide covers all storage scenarios.
If you travel with semaglutide, maintaining cold chain storage requires planning. Insulated travel cases with gel packs keep the medication at proper temperature during flights and road trips.
Semaglutide vs alternative GLP-1 medications
If semaglutide qualification proves difficult through your preferred pathway, understanding alternative GLP-1 medications helps you find the right fit. The GLP-1 class has expanded significantly, and different medications have different eligibility criteria.
Tirzepatide (Mounjaro/Zepbound)
Tirzepatide is a dual GIP/GLP-1 receptor agonist that has shown even greater weight loss in clinical trials compared to semaglutide. Zepbound is the weight loss formulation (similar to how Wegovy is the weight loss formulation of semaglutide), while Mounjaro is the diabetes formulation.
Qualification criteria for Zepbound mirror Wegovy: BMI of 30+ or BMI of 27+ with comorbidities. The detailed comparison between semaglutide and tirzepatide covers the differences in efficacy, side effects, and cost. Some patients who cannot access semaglutide find easier pathways to tirzepatide, and vice versa.
If you are considering switching between semaglutide and tirzepatide, the conversion chart helps providers determine equivalent dosing. Understanding how quickly tirzepatide works and reviewing before and after results can inform your decision.
Retatrutide
A newer triple agonist (GLP-1, GIP, and glucagon receptor) that has shown remarkable weight loss results in clinical trials, with some participants losing over 24% of their body weight. While not yet FDA-approved, retatrutide is available through research channels. The retatrutide dosing guide and comparison with semaglutide help you understand whether this newer option might be worth discussing with your provider.
Phentermine
While not a GLP-1 medication, phentermine deserves mention because it is often the first weight loss medication insurance requires you to try before approving GLP-1s. Phentermine is a sympathomimetic amine that suppresses appetite through a different mechanism. It is less expensive and more widely covered by insurance but carries its own set of side effects and is typically only prescribed for short-term use (12 weeks).
Understanding how phentermine compares to GLP-1 medications broadly helps you navigate the step therapy process more effectively.
Special qualification considerations
Certain populations face unique challenges and considerations when pursuing semaglutide qualification. These special circumstances deserve their own attention.
Qualification after bariatric surgery
Patients who have undergone bariatric surgery can and do qualify for semaglutide, particularly if they experience weight regain. The combination of surgical intervention and GLP-1 medication is increasingly recognized as a viable approach for patients who have not achieved or maintained their weight loss goals through surgery alone.
However, the altered GI anatomy from surgery, particularly gastric bypass, can affect semaglutide absorption and tolerability. Providers experienced with post-bariatric patients will adjust monitoring and dosing accordingly. Some insurers that previously denied GLP-1 coverage for post-surgical patients have updated their policies as evidence supporting this combination grows.
Qualification for older adults
Older adults (65+) face additional considerations. Sarcopenia (age-related muscle loss) is already a concern, and the weight loss from semaglutide includes some lean mass loss. Providers typically emphasize protein intake (at least 1.0-1.2 grams per kilogram of body weight daily) and resistance training to preserve muscle.
Medicare coverage considerations also come into play for older adults. As discussed earlier, the coverage landscape is evolving, but navigating Medicare Part D formularies requires specific knowledge of plan-level coverage decisions.
Qualification for adolescents
Wegovy is FDA-approved for adolescents aged 12 and older with a BMI at or above the 95th percentile for age and sex. Pediatric qualification requires involvement of both the adolescent and a parent/guardian, along with a comprehensive assessment by a provider experienced in pediatric obesity management.
The qualification process for adolescents includes additional screening for growth and development considerations, mental health assessment, and family-based behavioral intervention requirements. Most insurers have specific pediatric protocols for GLP-1 coverage that differ from adult criteria.
Qualification around surgical procedures
If you have an upcoming surgery, the qualification timeline matters. Many surgeons require patients to stop semaglutide 1-3 weeks before elective surgery due to concerns about delayed gastric emptying and aspiration risk during anesthesia. Understanding when you can resume semaglutide after surgery helps you plan appropriately.
If you are in the process of qualifying for semaglutide and have upcoming surgery, discuss the timing with both your prescribing provider and your surgeon. It may make sense to delay starting treatment until after your surgical recovery is complete.
Maintaining your prescription long-term
Qualifying once is not the end. Many insurance plans require ongoing documentation to continue covering semaglutide. Understanding these maintenance requirements prevents unexpected gaps in treatment.
Reauthorization requirements
Most insurance plans that cover GLP-1 medications require prior authorization renewal every 6-12 months. These renewals typically require documented weight loss progress (usually at least 5% of body weight within the first 6 months), continued compliance with lifestyle modifications, ongoing medical necessity, and updated lab work showing continued benefit.
If you hit a weight loss plateau before your reauthorization date, work with your provider to document other benefits, improved A1C, better blood pressure, reduced cholesterol, improved sleep apnea scores, that demonstrate continued medical value beyond the scale.
What happens if you stop qualifying
Weight loss itself can paradoxically threaten your qualification. If you lose enough weight to drop your BMI below 27 and your comorbidities resolve, some strict interpretations of the criteria suggest you no longer "qualify" for the medication. This creates a frustrating catch-22 where the treatment success becomes the reason for treatment discontinuation.
Most providers navigate this by emphasizing that obesity is a chronic disease requiring ongoing management, similar to hypertension that resolves with medication, you do not stop the medication that is working. Additionally, documenting the risk of weight regain upon discontinuation supports continued prescribing. Research on semaglutide withdrawal and weight regain provides evidence for continued treatment.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
The real cost of not qualifying the first time
Failed qualification attempts cost more than just time. Each denied insurance claim, each wasted appointment copay, each month of delayed treatment represents both financial loss and continued health consequences from untreated obesity. Understanding this calculus helps you approach the process with the preparation it deserves.
A single prior authorization denial can delay treatment by 4-6 weeks while you appeal. During that time, someone with a BMI of 35 continues carrying excess weight that stresses joints, elevates blood pressure, disrupts sleep, and increases cardiovascular risk. The compounding health effects of each month without treatment are real and measurable.
Financial losses add up quickly too. An initial consultation copay of $40-$75. Lab work costing $100-$300 depending on insurance coverage. A second consultation to address the denial. Time off work for appointments. The emotional toll of rejection when you have already built up the courage to seek help. These costs multiply with each failed attempt.
This is precisely why preparation matters so much. Walking into your first appointment with complete documentation, recent lab work, a clear understanding of your insurance requirements, and knowledge of alternative pathways dramatically increases first-attempt success rates. The strategies outlined in this guide exist to help you qualify efficiently, whether that happens through traditional insurance channels, telehealth platforms, or compounded alternatives.
The data supports aggressive preparation. Patients who bring documentation of previous weight loss attempts to their first appointment receive prescriptions at significantly higher rates than those who arrive without records. Patients who have already confirmed their insurance coverage details before the visit avoid the most common administrative delays. And patients who understand the full landscape of options, from semaglutide versus tirzepatide to brand-name versus compounded formulations, can pivot immediately when one pathway closes rather than starting over from scratch.
Frequently asked questions
What is the minimum BMI to qualify for semaglutide?
The standard minimum BMI is 30 (obesity) without any additional conditions. If your BMI is 27-29.9, you can qualify with at least one weight-related health condition such as type 2 diabetes, high blood pressure, or high cholesterol. Some compounded semaglutide providers may consider patients with a BMI as low as 25 with documented health conditions. The complete BMI guide for GLP-1 eligibility covers all thresholds in detail.
Can I qualify for semaglutide without diabetes?
Yes. Wegovy is FDA-approved specifically for weight management in non-diabetic patients who meet the BMI criteria. You do not need a diabetes diagnosis to qualify for semaglutide for weight loss. However, Ozempic is only approved for type 2 diabetes, so using it without diabetes would be off-label.
How long does the qualification process take?
Through telehealth platforms, the process can take as little as 3-7 days from initial questionnaire to medication delivery. Through a primary care physician, expect 2-4 weeks including lab work turnaround and appointment scheduling. If insurance prior authorization is required, add an additional 1-4 weeks. The timeline varies significantly based on your chosen pathway.
Will my insurance cover semaglutide for weight loss?
Coverage varies dramatically by plan. Approximately 40-50% of commercial insurance plans now offer some coverage for GLP-1 weight loss medications, though many include significant restrictions. Call your insurance company directly and ask about coverage for Wegovy specifically. If denied, explore appeal options and compounded semaglutide as an affordable alternative.
What blood tests do I need before qualifying?
Standard labs include fasting blood glucose, A1C, comprehensive metabolic panel (kidney and liver function), lipid panel (cholesterol and triglycerides), and thyroid function tests (TSH). Some providers also order amylase, lipase, and a complete blood count. Having recent labs (within 6-12 months) can expedite the qualification process.
Can I qualify if I only need to lose 10-15 pounds?
If your BMI is below 27, qualifying through standard channels is unlikely. Semaglutide is approved for patients with clinical obesity or overweight with comorbidities, not for cosmetic or minor weight loss goals. Some compounded semaglutide providers have more flexible criteria, but most still require a BMI of at least 25 for consideration.
What happens if I get denied?
You have options. Appeal the insurance decision with additional documentation. Ask your provider to submit a peer-to-peer review. Try a different insurance pathway (Ozempic instead of Wegovy if you have diabetes). Explore compounded semaglutide which does not require insurance approval. Or consider alternative GLP-1 medications like tirzepatide that might have different coverage under your plan.
Do I need to keep qualifying or is it a one-time process?
For insurance coverage, reauthorization is typically required every 6-12 months. You will need to demonstrate continued medical necessity, weight loss progress, and ongoing compliance with lifestyle modifications. For compounded semaglutide prescribed through telehealth, ongoing prescriptions usually require periodic follow-up consultations but the reauthorization process is less formal.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your qualification process stay smooth, your prescriptions stay approved, and your results stay consistent.