Feb 24, 2026

You checked the vial. The date has passed. And now you are standing in your kitchen, syringe in hand, wondering whether this expired tirzepatide will still work or whether it could actually hurt you. That question hits different when you have been paying out of pocket, waiting weeks for refills, and finally building momentum on a protocol that was changing your body composition.
Here is the honest answer. Expired tirzepatide will not poison you. Not typically. But what it will do is far more insidious, because tirzepatide does expire, and the consequences of using it past that date range from mildly frustrating to genuinely problematic for your health outcomes. The active peptide degrades silently. You cannot see potency loss. You cannot taste it. You inject what you believe is your full dose, but your body receives something less, something different, something that may trigger side effects without delivering the therapeutic benefits you are counting on.
This guide covers everything researchers need to know about expired tirzepatide, from the molecular degradation pathways that destroy potency to the specific visual signs that your medication has gone bad. We will walk through the real risks, the actual timelines, and the concrete steps you should take if you discover your tirzepatide has expired. Whether you are using brand-name Mounjaro, Zepbound, or compounded tirzepatide, the science of expiration affects you. SeekPeptides members understand that proper storage and timing are just as important as the peptide itself, and this guide will show you exactly why.
Why tirzepatide has an expiration date in the first place
Every medication carries an expiration date. But tirzepatide is not aspirin. It is not a simple chemical compound sitting stable in a tablet. Tirzepatide is a 39-amino-acid peptide with a C20 fatty diacid moiety attached, making it one of the most complex GLP-1 receptor agonists ever developed. That molecular complexity means it is inherently fragile.
The expiration date represents the last day the manufacturer guarantees the medication retains at least 90% of its labeled potency when stored under specified conditions. This is not arbitrary. Pharmaceutical companies conduct accelerated stability studies and real-time testing to determine exactly when a drug falls below that 90% threshold. For tirzepatide, those studies account for temperature fluctuations, light exposure, physical agitation, and the natural tendency of peptide bonds to break down over time.
The FDA requires this testing for every approved medication. The Shelf Life Extension Program, originally developed for the Department of Defense, tested over 3,000 lots representing 122 drug products and found that 88% of conventional medications retained potency well beyond their labeled dates, sometimes by 66 months on average. But here is the critical distinction that most articles about expired medications miss completely.
Peptides are not conventional medications.
Small-molecule drugs like ibuprofen or metformin have simple, stable chemical structures. They can sit in a medicine cabinet for years and retain most of their potency. Peptide drugs like tirzepatide are protein-based. They are susceptible to degradation pathways that small molecules simply do not face, including deamidation, oxidation, aggregation, and hydrolysis. The SLEP findings about conventional drugs do not apply to peptide therapeutics, and anyone telling you otherwise is dangerously oversimplifying the science.
The expiration date on your tirzepatide is not a suggestion. It is not pharmaceutical companies trying to sell you more medication. It represents real science about when the peptide begins losing its ability to do what you are paying it to do.
How tirzepatide degrades after expiration
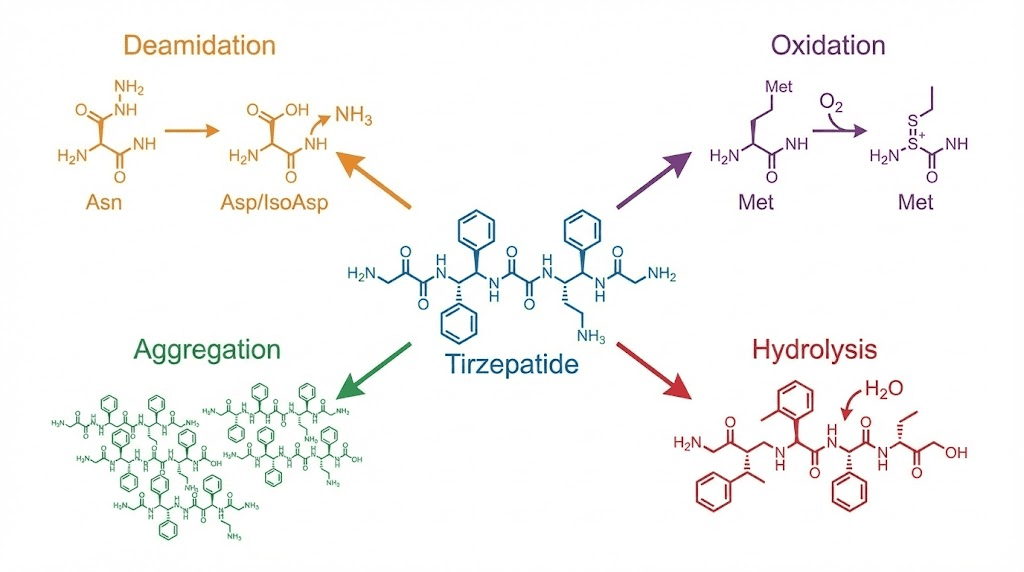
Understanding what actually happens at the molecular level helps explain why expired tirzepatide behaves differently than fresh medication. Four primary degradation pathways affect tirzepatide, and each one chips away at the peptide in a different way.
Deamidation breaks the backbone
Asparagine and glutamine residues in the tirzepatide sequence are vulnerable to deamidation, a chemical reaction where the amide group converts to a carboxylic acid. This creates a mass shift of just +1 Dalton, tiny in molecular terms but enough to alter how the peptide interacts with both the GLP-1 receptor and the GIP receptor. Deamidation accelerates at higher temperatures and higher pH levels, which is why proper refrigeration matters so much.
The rate of deamidation roughly doubles for every 10 degrees Celsius increase in temperature. A vial sitting on your counter at room temperature degrades at roughly four times the rate of one stored properly at 2-8 degrees Celsius. After expiration, this process does not suddenly stop. It continues, and it accelerates if storage conditions have been anything less than perfect.
Oxidation damages key residues
Tirzepatide contains tryptophan and methionine residues that are particularly susceptible to oxidation. When these amino acids oxidize, the peptide structure changes in ways that can reduce receptor binding affinity. Oxidation is triggered by exposure to light, air, and certain metals. Even trace amounts of metal ions from the rubber stopper or glass vial can catalyze oxidation reactions over time.
This is one reason why manufacturers specify dark storage conditions. Light, particularly UV light, accelerates oxidation dramatically. If your tirzepatide vial has been sitting on a windowsill or under fluorescent lights, oxidation damage may have occurred well before the printed expiration date.
Aggregation creates potentially harmful clumps
Perhaps the most concerning degradation pathway for expired tirzepatide is aggregation. Tirzepatide has an amphiphilic nature, meaning part of the molecule is water-loving and part is water-fearing (the C20 fatty acid chain). This dual nature makes it prone to forming aggregates, clusters of peptide molecules that clump together in solution.
Aggregation is dangerous for two reasons. First, aggregated peptide cannot bind to receptors properly, reducing or eliminating the therapeutic effect. Second, and more importantly, protein aggregates can trigger immune responses. Your body may recognize these clumps as foreign invaders and mount an antibody response, which could reduce the effectiveness of tirzepatide not just now but in future doses as well.
You might notice aggregation as cloudiness or particles in your solution. But early-stage aggregation is invisible. The peptide molecules are clustering at a submicroscopic level, reducing potency without any visible change. This is why understanding tirzepatide shelf life matters even when the medication looks perfectly clear.
Hydrolysis breaks peptide bonds
The peptide bonds holding tirzepatide together are susceptible to hydrolysis, a reaction with water that literally breaks the molecule apart. Hydrolysis is temperature-dependent and pH-dependent. The formulation buffer is designed to minimize this, but over time, especially after expiration, hydrolysis fragments accumulate. These fragments have no therapeutic value and represent pure potency loss.
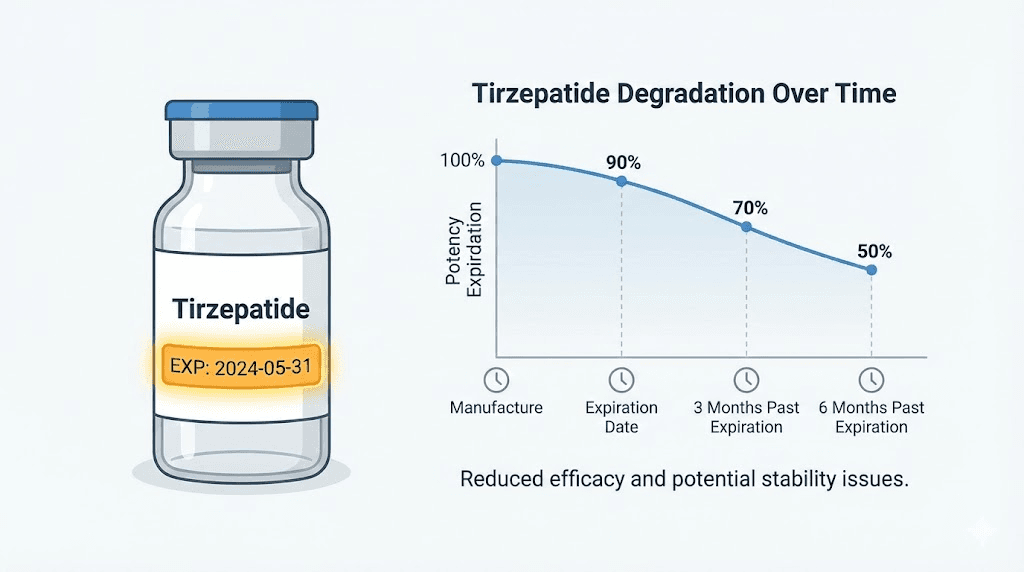
Research tracking peptide degradation kinetics has found that tirzepatide stored at proper refrigeration temperatures loses roughly 1-2% potency per month after the expiration date. That sounds small until you do the math. Three months past expiration, you could be down 3-6%. Six months, 6-12%. A year past expiration, your 5mg dose might deliver the equivalent of 4.4mg or less. For a medication where precise dosing determines outcomes, that gap matters enormously.
The real risks of using expired tirzepatide
Now that you understand what happens chemically, let us translate that into what happens practically when you inject expired tirzepatide into your body.
Reduced effectiveness is the primary concern
The biggest risk is not that expired tirzepatide will make you sick. It is that it will not work. You inject your dose expecting a certain level of appetite suppression, a certain impact on blood glucose, a certain contribution to your weight loss timeline. Degraded tirzepatide delivers less of all three.
For people using tirzepatide for type 2 diabetes management, this is particularly dangerous. Reduced potency means inadequate glycemic control. Blood sugar levels rise. HbA1c creeps upward. Over time, poorly controlled blood sugar increases the risk of cardiovascular disease, nephropathy, retinopathy, and neuropathy. You might not notice the subtle shift for weeks, and by then the damage compounds.
For weight management, reduced potency means your appetite suppression wanes. You feel hungrier than expected. Cravings return. The metabolic advantages of the GIP/GLP-1 dual agonism diminish. You might attribute this to a plateau, to your body adapting, to needing a higher dose. But the real problem is degraded medication. People sometimes wonder why tirzepatide is not working anymore, and expired or improperly stored medication is one of the most overlooked causes.
Unpredictable dosing creates protocol chaos
When you use expired tirzepatide, you have no way of knowing how much active ingredient remains. There is no at-home test. No color change. No taste difference. Laboratory analysis using HPLC (high-performance liquid chromatography) is the only reliable method, and you do not have one in your kitchen.
This means every injection becomes a guess. Maybe today your vial still has 85% potency. Maybe it is down to 70%. You cannot tell. This unpredictability wreaks havoc on your protocol. If you are following a tirzepatide dosing schedule for weight loss, the entire framework depends on consistent, known doses. Remove that consistency and your results become unreliable, your side effect profile changes, and your ability to make informed decisions about dose adjustments disappears.
Some researchers try to compensate by injecting more, figuring that extra volume will offset potency loss. This is risky. If the medication has degraded less than expected, you are effectively overdosing. If it has degraded more, you are still underdosing. The math never works in your favor.
Degradation byproducts and side effects
Degraded tirzepatide does not just become weaker. It becomes something different. The deamidation products, oxidation products, and aggregation fragments are new chemical entities that your body was never meant to receive. While the risk of severe adverse reactions from these byproducts is considered low in properly sealed medication, it is not zero.
Some users report increased gastrointestinal side effects when using older medication, including nausea that seems disproportionate to the dose. Others notice injection site reactions that they did not experience with fresh tirzepatide, redness, swelling, or itching at the injection site. Injection site redness and itching can signal that degradation products are triggering a local inflammatory response.
Aggregated peptides pose a particular concern. As mentioned, protein aggregates can provoke immune responses. If your body develops anti-drug antibodies against tirzepatide, the medication may become less effective even when you switch to a fresh supply. This is one of the less-discussed but potentially significant risks of routinely using degraded peptide medications.
Wasted money and lost momentum
Beyond the biological risks, there is a practical cost. Tirzepatide is not cheap. Whether you are paying for brand-name Mounjaro, Zepbound, or compounded tirzepatide, every wasted dose is money thrown away. But the real cost is lost momentum.
Weight loss and metabolic improvement are cumulative processes. Weeks of suboptimal dosing do not just mean slower progress. They can mean regression. Appetite returns. Blood sugar fluctuates. The psychological toll of feeling like the medication "stopped working" can derail your entire commitment to the protocol. All because the peptide in your syringe was not what you thought it was.
Compounded vs. brand-name tirzepatide expiration differences
Not all tirzepatide is created equal when it comes to shelf life and expiration timelines. The source of your medication significantly affects how long it lasts and how quickly it degrades.
Brand-name tirzepatide (Mounjaro, Zepbound)
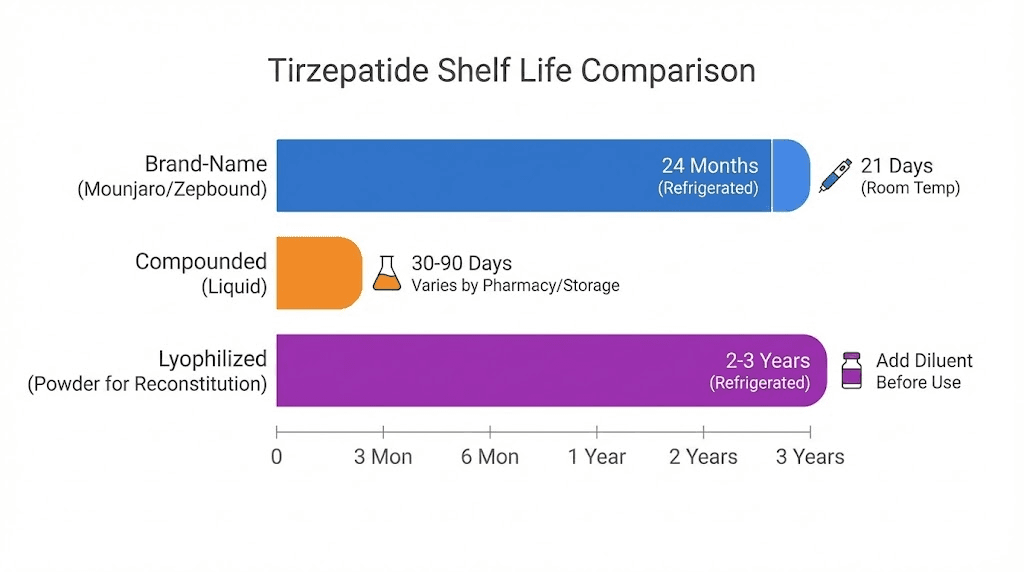
Eli Lilly manufactures brand-name tirzepatide in prefilled injection pens with extensive stability testing. These pens carry manufacturer-determined expiration dates based on real-time and accelerated stability data. When stored properly at 2-8 degrees Celsius, they retain potency until the printed date, which is typically 18-24 months from manufacture.
Once removed from refrigeration, brand-name tirzepatide pens can be stored at room temperature (below 30 degrees Celsius or 86 degrees Fahrenheit) for a maximum of 21 days. After 21 days at room temperature, the medication should be discarded regardless of the printed expiration date. This room-temperature limit exists because degradation accelerates significantly outside refrigeration.
Compounded tirzepatide has shorter timelines
Compounded medications follow different rules. Instead of an expiration date determined by the manufacturer, compounded tirzepatide carries a beyond-use date (BUD) assigned by the compounding pharmacy based on United States Pharmacopeia guidelines.
Typical BUD ranges for compounded tirzepatide include:
Unopened, refrigerated (2-8 degrees Celsius): 45-90 days depending on the pharmacy
Unopened, room temperature: 14-45 days
After first puncture (multi-dose vials): 28 days regardless of storage method
These timelines are significantly shorter than brand-name medication because compounding pharmacies operate under different regulatory frameworks and testing requirements. The BUD is conservative by design, intended to ensure sterility and potency within a validated window.
If you are using compounded tirzepatide, the 28-day post-puncture rule is critical. Once you push a needle through that rubber stopper, the clock starts ticking. Every entry introduces microscopic amounts of air and potential contaminants. After 28 days, the risks of using the remaining medication increase substantially, not just from potency loss but from potential microbial contamination.
SeekPeptides members often ask about the differences between compounded and brand-name timelines. The key takeaway is simple: compounded tirzepatide has less room for error. Shorter shelf life means tighter adherence to storage protocols.
Lyophilized (freeze-dried) tirzepatide
Some compounding pharmacies and research suppliers provide tirzepatide in lyophilized (freeze-dried) powder form. Before expiration or beyond-use date, this form is actually more stable than liquid formulations because the absence of water dramatically slows degradation reactions. Lyophilized tirzepatide stored properly can maintain potency for months.
However, once reconstituted with bacteriostatic water, the clock starts immediately. Reconstituted tirzepatide follows the same degradation pathways as any liquid formulation, and the 28-day rule applies. Using expired reconstituted tirzepatide carries all the same risks discussed above, plus additional contamination concerns if the bacteriostatic water preservative has degraded.
How to tell if your tirzepatide has gone bad
Visual inspection is not a substitute for laboratory testing. But it can catch the most obvious signs of degradation. Think of it as a first-pass filter, if your tirzepatide fails the visual test, it has definitely gone bad. If it passes, it might still have degraded invisibly.
Cloudiness or haziness
Fresh tirzepatide solution should be completely clear and colorless. Hold the vial up to a light source and look through it. Any cloudiness, haziness, or milky appearance indicates protein aggregation. The peptide molecules have clumped together, and the medication should not be used. This is the most reliable visual indicator of significant degradation.
Visible particles or floaters
Tilt and roll the vial gently (never shake it). Look for particles floating in the solution or settled at the bottom. These could be aggregated peptide, precipitated buffer components, or contaminants from repeated needle punctures. Any visible particulate matter means the vial should be discarded. The presence of particles indicates the solution has lost integrity.
Color changes
Tirzepatide should be colorless to very slightly yellow at most. Any significant yellowing, browning, or other discoloration suggests oxidation damage. Tirzepatide compounded with B12 will have a pink or reddish tint from the B12, so this test applies primarily to plain tirzepatide formulations.
Unusual odor
Standard tirzepatide solution has no significant odor. If you detect any unusual smell when drawing up the medication, this could indicate bacterial contamination or chemical breakdown. Discard the vial immediately.
Resistance when drawing or injecting
If the solution seems thicker than usual, requires more pressure to draw into the syringe, or feels different during injection, aggregation may have increased the viscosity. Normal syringe draw should feel smooth and easy. Any change in viscosity warrants suspicion.
The invisible degradation problem
The frustrating reality is that early-stage degradation produces no visible changes. Your tirzepatide can lose 5-10% of its potency while still looking perfectly clear, colorless, and particle-free. This is why the expiration date and beyond-use date exist, they tell you when degradation has likely progressed to a meaningful degree even if you cannot see it.
If you are experiencing diminished results from tirzepatide and everything else in your protocol is consistent, check your vial dates first. Degradation is one of the most common and least suspected causes of reduced effectiveness.
Temperature exposure and accelerated expiration
The printed expiration date assumes proper storage. Temperature excursions, periods where your medication was outside the recommended range, effectively move the expiration date forward. Think of it this way: your tirzepatide ages faster when it is warm.
The temperature-degradation relationship
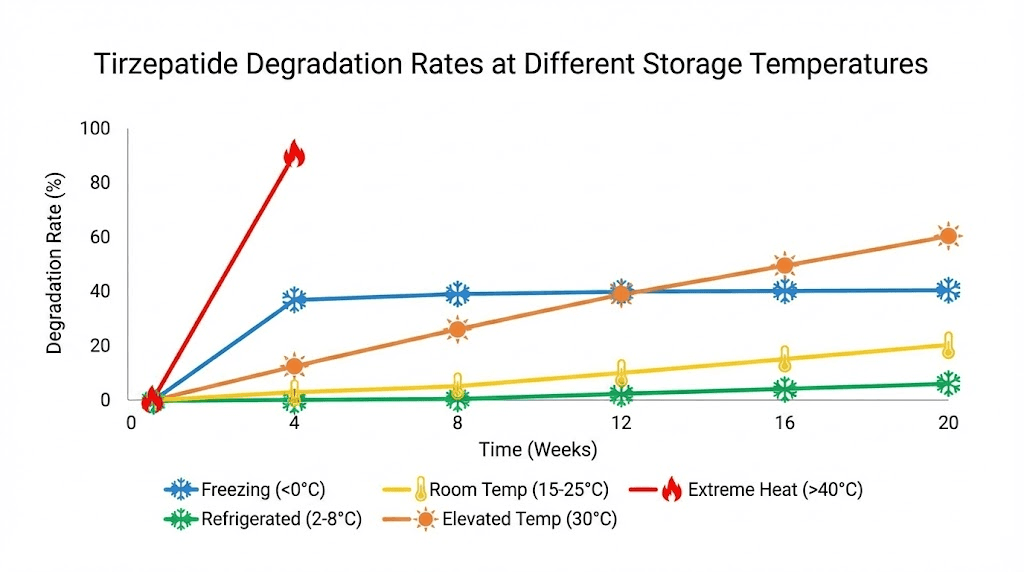
Chemical reaction rates roughly double for every 10 degrees Celsius increase. At room temperature (25 degrees Celsius), tirzepatide degrades approximately four times faster than at refrigerated temperatures (2-8 degrees Celsius). At body temperature (37 degrees Celsius), degradation is roughly eight times faster. At extreme heat (40 degrees Celsius or above), the peptide can denature rapidly and irreversibly.
This means if your tirzepatide sat on a hot delivery truck for 8 hours at 35 degrees Celsius, it experienced the equivalent of roughly 32 hours of degradation at refrigerated temperatures. If it spent a weekend on your kitchen counter at 25 degrees Celsius (48 hours), that is equivalent to roughly 192 hours (8 days) of refrigerated degradation.
The math matters. Cumulative temperature excursions accelerate the path to expiration. If your vial has experienced multiple warm periods, even brief ones, the printed expiration date overestimates the remaining potency.
What happens when tirzepatide gets too warm
Temperature excursions beyond 30 degrees Celsius are particularly damaging. Above this threshold, denaturation begins. The tertiary structure of the peptide, the 3D shape that allows it to bind to GIP and GLP-1 receptors, starts to unfold. Once denatured, the peptide cannot refold correctly. This damage is permanent and irreversible, no amount of re-refrigeration will restore potency lost to heat denaturation.
This is especially relevant for people who order tirzepatide from various sources where shipping conditions may not be perfectly controlled. If your package arrives warm to the touch, the cold chain may have been broken.
Freezing is equally destructive
On the opposite extreme, freezing damages tirzepatide just as surely as overheating. When the solution freezes, ice crystals form that physically shear peptide molecules apart. The freeze-thaw cycle also concentrates solutes in unfrozen regions, creating localized conditions that accelerate chemical degradation.
A frozen and thawed vial of tirzepatide should be discarded even if it has not reached its expiration date. The damage from freezing is immediate and irreversible. If your refrigerator has a tendency to freeze items near the back wall or cooling element, store your tirzepatide on a middle shelf or in the door where temperatures are more consistent.
The semaglutide comparison
For context, similar degradation principles apply to other GLP-1 agonists. Accidentally leaving semaglutide out overnight creates comparable potency concerns, and using expired semaglutide carries similar risks. The broader principle is universal across peptide medications: temperature control is potency control.
Proper storage protocols that maximize tirzepatide shelf life
Prevention is better than gambling with expired medication. Proper storage does not just maintain potency until the expiration date. It ensures you get every microgram of active peptide you paid for.
Refrigeration is non-negotiable
Store tirzepatide at 2-8 degrees Celsius (36-46 degrees Fahrenheit) at all times when not in active use. This is the single most important factor in preserving potency. A dedicated medication refrigerator or a consistent spot in your regular refrigerator (not the door, not the back wall) works best.
Use a refrigerator thermometer to verify your storage temperature. Consumer refrigerators can vary by several degrees depending on location, loading, and door-opening frequency. Knowing your actual storage temperature gives you confidence in your medication potency.
Protect from light
Store tirzepatide in its original packaging or in an opaque container. Light, especially UV light, accelerates oxidation reactions. If your vial comes in a box, keep it in the box until you need it. If it does not, wrap it in aluminum foil or store it in a dark section of the refrigerator.
Minimize temperature excursions
Take your tirzepatide out of the refrigerator only when you are ready to inject. Let it warm to room temperature for 5-10 minutes if you prefer (cold injections can sting more), then return the vial promptly. Do not leave it on the counter for hours while you go about your day.
For injection preparation, keep your total out-of-fridge time under 30 minutes per use. This minimizes cumulative temperature exposure and preserves potency for the full duration of the vial.
Track your vial timeline
Write the date of first puncture on every multi-dose vial. Use a piece of tape or a marker directly on the vial. This gives you a clear reference point for the 28-day post-puncture limit. Do not rely on memory. After 28 days from first puncture, discard the vial regardless of remaining volume.
Some researchers create a simple tracking system in their phone with calendar reminders set for 28 days after opening each vial. This removes any ambiguity about when the medication needs to be replaced.
Travel requires extra precautions
Traveling with tirzepatide demands a medical-grade cooler bag with gel ice packs or a portable medication refrigerator. Standard cooler bags with ice can drop below freezing temperature, which is just as damaging as overheating. Position the medication away from direct contact with ice packs and monitor temperature with a portable thermometer.
Understanding how long tirzepatide can be out of the fridge is essential for travel planning. Brand-name pens allow up to 21 days at room temperature. Compounded vials may have shorter tolerances. Plan your travel around these windows and carry only enough medication for the trip duration plus a small buffer.
The reconstitution factor
If you are working with lyophilized tirzepatide, the reconstitution process introduces its own set of stability considerations. Use the correct volume of bacteriostatic water at the proper ratio. Swirl gently, never shake, as vigorous agitation promotes aggregation. After reconstitution, refrigerate immediately and start counting the 28-day window.
Tirzepatide reconstitution charts provide the exact water-to-powder ratios for different concentrations. Getting this right from the start means your solution has the best possible foundation for maintaining potency through its usable window.
What to do if you have already used expired tirzepatide
If you injected expired tirzepatide before reading this guide, take a breath. The situation is almost certainly manageable. Here is your step-by-step response plan.
Do not panic
A single dose of recently expired tirzepatide stored under proper conditions is unlikely to cause harm. The peptide does not suddenly become toxic at the expiration date. Degradation is gradual. If the medication was only days or even a few weeks past expiration and was stored properly refrigerated the entire time, the potency loss is likely minimal.
Monitor for unusual symptoms
Pay attention to any changes in the 24-48 hours after injection. Watch for:
Unusual injection site reactions (excessive redness, swelling, pain, or injection site reactions beyond normal)
Gastrointestinal symptoms that seem disproportionate to your dose (unexpected diarrhea, severe nausea, headaches)
Signs of inadequate blood sugar control (for diabetes patients)
Allergic-type reactions (hives, difficulty breathing, rapid heartbeat)
Most people who use recently expired tirzepatide experience nothing more than slightly reduced effectiveness. Serious adverse reactions from degradation products in properly sealed medication are rare.
Check your blood sugar
If you use tirzepatide for diabetes management, check your blood glucose more frequently for the next few days. Reduced potency from expired medication may mean your glucose levels are higher than expected. Adjust your monitoring schedule and be prepared to contact your healthcare provider if levels are consistently elevated.
Assess your remaining supply
Look at your other vials or pens. Are they also expired? Check dates on everything. If you have a systemic issue with expired medication, whether from a delayed shipment, overstocking, or irregular use patterns, you need to address the root cause.
Dispose of expired medication properly
Do not flush tirzepatide down the toilet or throw it in the regular trash. The FDA recommends using pharmacy take-back programs or community drug disposal events. If neither is available, mix the medication with an undesirable substance (like coffee grounds or cat litter), place it in a sealed container, and dispose of it in household trash. Remove any personal information from the vial or pen before disposal.
Resume with fresh medication
Replace your expired tirzepatide with a fresh supply as soon as possible. If you are between refills, contact your pharmacy or provider about an early refill. Explain the situation. Most pharmacies and insurance plans accommodate early refills for expired or damaged medication.
Frequently asked questions about expired tirzepatide
Can expired tirzepatide make you sick?
Expired tirzepatide is unlikely to cause acute illness if it was stored properly and is only recently past its expiration date. The primary risk is reduced effectiveness rather than toxicity. However, medication that has been stored improperly (left at high temperatures, frozen, or exposed to light) may contain degradation products that could cause increased side effects like body aches, injection site reactions, or gastrointestinal symptoms beyond what you normally experience.
How long past the expiration date is tirzepatide still effective?
There is no definitive answer because it depends entirely on storage conditions. Under perfect refrigeration (2-8 degrees Celsius), tirzepatide loses approximately 1-2% potency per month after expiration. This means it retains roughly 90% potency for 5-10 months past expiration under ideal conditions. However, no manufacturer or regulatory body recommends using it past the expiration date, and most healthcare providers advise against it because the actual potency in your specific vial is unknowable without laboratory testing.
Is expired compounded tirzepatide more dangerous than expired brand-name?
Compounded tirzepatide generally has less stability testing behind its beyond-use date compared to brand-name products. This means the safety margin beyond the BUD may be narrower. Additionally, compounded formulations with additives like B12, glycine, or niacinamide introduce additional variables that can affect degradation rates. When in doubt, compounded medication past its BUD should be treated with more caution than brand-name medication slightly past its expiration date.
Should I increase my dose if I think my tirzepatide has lost potency?
No. Absolutely not. You have no way to determine how much potency has been lost, which means you cannot calculate an appropriate dose adjustment. Injecting more expired medication could mean you are getting your normal dose (if degradation was minimal) or you could still be underdosed (if degradation was significant). The only safe response to suspected potency loss is to replace the medication with a fresh supply. Consult your tirzepatide dosing guide and resume at your normal dose with fresh medication.
Does tirzepatide expire faster once opened?
Yes. Once a vial is punctured, the sterile seal is broken. Repeated needle entries introduce microscopic amounts of air and potential contaminants. Brand-name tirzepatide pens and compounded vials in the fridge should be used within 28 days of first puncture, regardless of the printed expiration date. This 28-day limit applies even if the expiration date is months away.
What if my tirzepatide was left out overnight but is not expired?
A single overnight room-temperature excursion (approximately 8-12 hours at 20-25 degrees Celsius) is unlikely to cause significant degradation if the medication is otherwise within its expiration date and has been properly stored. Return it to the refrigerator immediately. However, the 21-day room temperature limit still applies cumulatively, so this overnight episode reduces your remaining room-temperature allowance. If this happens frequently, read our guide on warm tirzepatide for a more detailed analysis.
Can I tell from my results whether my tirzepatide has degraded?
Sometimes. If you notice a sudden change in effectiveness, increased hunger, reduced appetite suppression, or stalled weight loss, and nothing else in your protocol has changed, degraded medication should be on your list of suspects. But many other factors affect results too, including diet, food choices, stress, sleep, and natural plateaus. Check your vial dates and storage history before assuming the medication is to blame.
Does the FDA SLEP program apply to tirzepatide?
The FDA Shelf Life Extension Program tested primarily small-molecule drugs in solid dosage forms (tablets, capsules) for the military. Peptide biologics like tirzepatide were not included in SLEP testing. The finding that 88% of medications retained potency well beyond expiration dates does not apply to peptide drugs, which are inherently less stable than conventional small molecules. Do not use SLEP data to justify using expired tirzepatide.
How expired tirzepatide compares to expired semaglutide
If you have experience with semaglutide (Ozempic, Wegovy) and tirzepatide, you might wonder whether expiration affects them differently. The answer is nuanced.
Both are peptide-based GLP-1 receptor agonists susceptible to the same degradation pathways. However, tirzepatide has a larger, more complex structure (39 amino acids with a C20 fatty acid moiety vs. semaglutide 31 amino acids with a C18 fatty acid chain). More complexity generally means more potential degradation sites.
Semaglutide follows similar expiration rules. Semaglutide has a 28-day post-opening limit and requires proper refrigeration for compounded formulations. Semaglutide shelf life in the fridge is comparable to tirzepatide when stored properly.
The practical implication is that if you have been cavalier about expiration dates with one peptide, you should reconsider your approach with all of them. The same storage discipline that protects tirzepatide protency also applies to compounded semaglutide in the fridge, semaglutide potency windows, and any other peptide in your protocol.
For researchers considering a switch between medications, understanding these storage parallels is important. Whether you are switching from tirzepatide to semaglutide or converting from semaglutide to tirzepatide, the same storage vigilance applies to both.
The cost of cutting corners on expiration
We understand the temptation. Tirzepatide is expensive. The thought of throwing away a half-full vial because it passed a date on the label feels wasteful. And in some cases, recently expired, properly stored medication probably retains most of its potency. But the math rarely works in your favor over the long run.
The hidden cost calculation
Consider this scenario. You have a vial that expired two months ago. Based on typical degradation rates, it might retain 85-90% potency. You inject your normal 5mg dose but actually receive 4.25-4.5mg. Over a month of weekly injections, you receive 3-5mg less total tirzepatide than planned.
That 3-5mg shortfall means reduced appetite suppression, less metabolic benefit, and slower progress. If it adds even one extra week to your timeline, the cost of that week (continued medication, lost time, potential rebound effects) likely exceeds the cost of replacing the expired vial. The false economy of using degraded medication rarely saves money in the aggregate.
Buying patterns that prevent waste
The best approach is preventing expiration in the first place. Order only what you will use within the shelf life window. For compounded tirzepatide with a 90-day BUD, do not stockpile more than a 90-day supply at a time. Coordinate your refill schedule with your actual usage rate so that vials arrive when you need them, not months in advance.
If you use a tirzepatide dosage calculator to plan your protocol, you can predict exactly how many vials you need for a given period. This prevents over-ordering and reduces the risk of medications expiring before use. SeekPeptides provides tools to help members calculate their exact supply needs, reducing waste and ensuring every dose delivers full potency.
When in doubt, throw it out
The medical community has a simple maxim about expired medications: when in doubt, throw it out. For tirzepatide, this principle applies with extra force because peptide degradation is invisible, unpredictable without laboratory testing, and can affect not just potency but also your body immune response to the medication.
Your health outcomes depend on consistent, predictable dosing. Every expired injection introduces an unknown variable that undermines the precision your protocol requires. Whether you are tracking your tirzepatide weight loss timeline, managing blood sugar, or optimizing your dosage progression, you need to trust that what is in the syringe matches what is on the label.
If your tirzepatide is expired, past its beyond-use date, or has been exposed to conditions outside the recommended range, replace it. The cost of a new vial is always less than the cost of compromised results, wasted time, and the nagging uncertainty of not knowing what you actually injected.
For researchers serious about getting the most from their tirzepatide protocols, SeekPeptides offers comprehensive storage guides, dosage calculators, and evidence-based protocol frameworks that help you maximize every dose from the moment you open the vial to the last injection before replacement.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your vials stay potent, your storage stay cold, and your protocols stay consistent.