Feb 25, 2026

Week one. Nothing much happens. Maybe a slight dip in hunger, maybe not. Week four. The scale begins moving, slowly at first, in small increments that barely register. Week twelve. Something shifts. The numbers drop faster now, and the mirror starts reflecting what the data already showed. Week twenty-four. You are down 17% of your starting body weight, and the trajectory has not even begun to flatten. This is not wishful thinking or supplement hype. This is what phase 2 clinical trial data published in the New England Journal of Medicine actually demonstrated for retatrutide, the triple-receptor agonist that produced weight reduction numbers researchers had never seen before in obesity pharmacotherapy.
But clinical trial averages tell you what happened across hundreds of participants. They do not tell you what will happen to you, specifically, in your first week or your first month. And that is the question most people actually want answered when they search for how long retatrutide takes to work. Not the abstract data. The practical, week-by-week reality of what this peptide does inside the body, when appetite suppression kicks in, when the weight starts dropping, and when the results become undeniable.
That is exactly what this guide covers. Drawing from published clinical data, mechanism-of-action research, and the emerging body of real-world observations, this is the most comprehensive timeline available for understanding retatrutide results from first injection to peak efficacy. Whether you are considering retatrutide, already on it, or switching from another GLP-1 medication, this is the reference you will want bookmarked.
How retatrutide works differently from other weight loss peptides
Understanding why retatrutide works the way it does, and why the timeline looks the way it does, starts with understanding what makes this molecule fundamentally different from semaglutide and tirzepatide. Those medications activate one or two receptors respectively. Retatrutide activates three. This is not a small distinction.
Retatrutide is a 39-amino-acid synthetic peptide engineered from a GIP peptide backbone. It simultaneously activates three hormone receptors: the glucagon-like peptide-1 receptor, the glucose-dependent insulinotropic polypeptide receptor, and the glucagon receptor. Each receptor triggers distinct metabolic pathways, and the combined effect produces results that no single-receptor or dual-receptor agonist can match.
The GLP-1 receptor activation enhances glucose-stimulated insulin secretion, slows gastric emptying, and promotes satiety. This is the primary appetite suppression mechanism. The GIP receptor activation facilitates additional insulin secretion in a glucose-dependent manner and plays a critical role in lipid metabolism, contributing to reductions in fat deposition and improvements in overall energy balance. The glucagon receptor activation, which is the unique component missing from both semaglutide and tirzepatide, promotes energy expenditure and modulates hepatic glucose production, leading to increased thermogenesis and lipid mobilization.
Here is why this matters for the timeline question. The receptor potencies are not equal. Research shows the following EC50 values for retatrutide: GIP receptor at 0.0643 nM (most potent), GLP-1 receptor at 0.775 nM, and glucagon receptor at 5.79 nM (least potent). This means retatrutide activates the GIP receptor most aggressively, the GLP-1 receptor at moderate strength, and the glucagon receptor at the lowest level.
That hierarchy matters because it explains the onset pattern.
GIP receptor effects kick in quickly, influencing insulin dynamics and lipid handling from early doses. GLP-1 effects, including the appetite suppression most people notice first, activate at moderate potency but become more pronounced as doses increase during titration. Glucagon receptor effects, particularly the increased energy expenditure and thermogenesis, build gradually as the body adjusts to higher doses. This layered activation pattern is why retatrutide results follow a specific curve rather than appearing all at once.
The half-life of retatrutide is approximately six days, achieved through acylation with a fatty diacid moiety. This allows for convenient once-weekly subcutaneous injection, and it means each dose builds on the previous one. Steady-state blood levels develop over the first few weeks, and the full pharmacological effect at any given dose takes approximately four to five half-lives to establish, roughly three to four weeks.
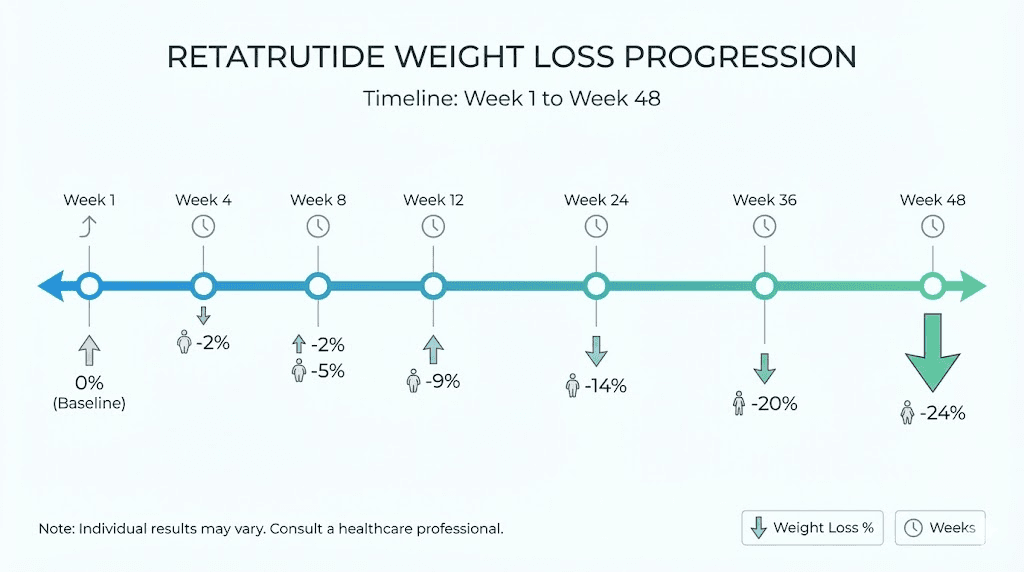
The complete week-by-week retatrutide timeline
The following timeline draws from phase 2 trial data involving 338 adults published in the New England Journal of Medicine, phase 3 data from Eli Lilly announcements, and the published qualitative study on participant experiences during the clinical trial. Dose references follow the typical starting dose protocol of beginning at 1-2 mg and titrating upward.
Week 1-2: the adjustment window
Do not expect dramatic changes during the first two weeks. The starting dose of retatrutide, typically 1-2 mg, is deliberately conservative. It exists to let the body adjust to triple-receptor activation without overwhelming the gastrointestinal system.
What happens physiologically during this window is subtle but important. GIP receptor activation begins influencing insulin dynamics almost immediately. Some people notice mild changes in hunger patterns within 24-72 hours of the first injection. Others feel nothing different at all. Both responses are normal.
In the phase 2 trial, the 1 mg dose group showed minimal weight change at early time points. This is expected. The starting dose is not designed to produce weight loss. It is designed to establish baseline tolerance and begin the process of metabolic adjustment that makes higher doses effective and manageable.
Common experiences during weeks 1-2 include mild nausea (reported by approximately 14% of participants at the 1 mg dose level), slight changes in appetite timing, increased awareness of fullness cues, and occasional fatigue as the body processes a new metabolic signal. If you experience no noticeable effects during this period, that does not mean the medication is not working. The dose is simply too low to produce obvious clinical effects in most people.
Week 3-4: early signals emerge
By weeks three and four, two things have changed. First, if the titration protocol calls for a dose increase at week two or four, the higher dose amplifies all three receptor pathways. Second, retatrutide has reached closer to steady-state concentration in the blood at the initial dose level.
This is when many people notice the first real appetite changes. Food does not disappear from the mind entirely, but portions naturally shrink. The desire for between-meal snacking decreases. Some people report that the sheer volume of food they previously consumed now feels physically uncomfortable.
Published qualitative research from the phase 2 trial found that 31 out of 36 retatrutide-treated participants reported meaningful changes in eating behaviors within the first eight weeks of treatment. That means the majority were noticing something by this stage. The changes described included eating smaller portions, feeling satisfied sooner during meals, reduced cravings for calorie-dense foods, and decreased interest in habitual or emotional eating.
Weight-wise, the scale may begin showing small decreases. At the 4 mg dose level in the phase 2 trial, participants averaged approximately 3-5% body weight reduction by week four to six. This might translate to 6-12 pounds for someone starting at 250 pounds. Not dramatic, but consistent. And consistency is what separates medications that work from those that do not.
Use the retatrutide dosage calculator to understand your specific dose progression, and track your results carefully during this period.
Week 5-8: building momentum
The five-to-eight-week window is where retatrutide begins separating itself from other weight loss peptides. By this point, most protocols have titrated to 4 mg or are approaching 8 mg. The triple-receptor mechanism is now running at meaningful potency across all three pathways.
Appetite suppression becomes more pronounced and consistent. The glucagon receptor activation begins contributing noticeably to energy expenditure, creating a metabolic environment where the body burns more calories at rest than it did before treatment. This is a feature unique to retatrutide among the current generation of weight loss medications.
The gastrointestinal side effects that appeared during dose increases typically begin resolving. Nausea, which affected 18-36% of participants at the 4 mg dose and 17-60% at the 8 mg dose in clinical trials, usually peaks during the first week of each new dose level and then subsides as the body adapts. Most people report that by week six to eight, the side effects have diminished significantly while the appetite suppression and metabolic benefits continue building.
Weight loss during this period accelerates. The dosage chart for weight loss shows a steepening curve starting around week six. In the phase 2 trial, the combined 8 mg group was already showing approximately 10-12% body weight reduction by week eight to ten. For a 250-pound individual, that represents 25-30 pounds lost.
This is also the phase where people start receiving comments from others. Clothing fits differently. Energy levels shift. The changes become visible and undeniable, not just to you but to the people around you.
Week 9-16: accelerated progress
Weeks nine through sixteen represent the steepest part of the weight loss curve for most retatrutide users. By this point, many people have reached or are approaching the 8 mg to 12 mg dose range where the clinical data shows the most dramatic results.
All three receptor pathways are now operating at high capacity. The combined effect creates a metabolic state where appetite is significantly reduced, gastric emptying is slowed (meaning food stays in the stomach longer and meals feel more satisfying), insulin dynamics are optimized for fat mobilization rather than fat storage, and resting energy expenditure is elevated through glucagon-mediated thermogenesis.
The weight loss data from this period is striking. In the phase 2 trial, participants on the 12 mg dose had already achieved approximately 15-17% body weight reduction by week sixteen. The 8 mg group was tracking close behind at approximately 13-15%. These numbers exceed what semaglutide produces over its entire 68-week treatment period.
Metabolic improvements beyond weight loss also become measurable during this window. Clinical trial participants showed improvements in systolic and diastolic blood pressure, triglycerides, LDL cholesterol, total cholesterol, HbA1c, and fasting glucose and insulin levels. For people with type 2 diabetes or metabolic syndrome, these changes can be clinically significant.
One finding that particularly stands out: liver fat reduction. In a phase 2a trial specifically examining metabolic dysfunction-associated steatotic liver disease, the 8 mg retatrutide group showed 81.7% mean relative reduction in liver fat, and the 12 mg group showed 86% reduction. By week sixteen, 89% of participants on 8 mg and 93% on 12 mg had achieved normal liver fat levels below 5%. These are extraordinary numbers that no other obesity medication has matched. Understanding how peptides work at the cellular level helps explain why triple-receptor activation produces these multi-system benefits.
Week 17-24: the half-year milestone
Week twenty-four was the primary endpoint of the phase 2 trial, and the results at this mark were what made the medical world take notice. The least-squares mean percentage change in body weight from baseline was -7.2% in the 1 mg group, -12.9% in the 4 mg group (starting at 2 mg), -17.1% in the combined 4 mg groups, -17.5% in the combined 8 mg groups, and -17.5% in the 12 mg group, compared with -1.6% in the placebo group.
Let those numbers sink in.
At the highest dose, participants lost an average of 17.5% of their body weight in just 24 weeks. For someone starting at 250 pounds, that represents approximately 44 pounds. For someone at 300 pounds, roughly 52 pounds. And unlike crash diets or stimulant-based approaches, this weight loss occurred through sustainable metabolic mechanisms, reduced appetite, improved fat oxidation, and increased energy expenditure, rather than through deprivation or artificial stimulation.
The 24-week mark also reveals something important about the dose-response relationship. The 4 mg and 8 mg groups showed very similar results at 24 weeks, suggesting that moderate doses can produce excellent outcomes. Not everyone needs the maximum 12 mg dose to achieve clinically meaningful weight loss. This is important context for people working with their researchers to find the right dosage that balances efficacy with tolerability.
By this point, appetite suppression is well established and feels like a new baseline rather than a temporary effect. Many people describe it not as feeling like they cannot eat, but as simply not thinking about food the way they used to. The psychological relationship with food shifts. Meals become functional rather than compulsive. This represents genuine behavioral change driven by the peptide pharmacology, not willpower.
Week 25-36: continued transformation
The period from month six to month nine is where retatrutide demonstrates one of its most impressive characteristics: the weight loss does not plateau the way it does with many other interventions.
With semaglutide, weight loss tends to slow significantly after 20-30 weeks, approaching a plateau by week 40-50. With tirzepatide, the trajectory is similar but extends somewhat further. With retatrutide, the curve continues descending through this entire period, driven in part by the ongoing glucagon receptor activation that other medications lack.
The combined 8 mg group was tracking approximately 20-22% body weight reduction by week 30-36 in the phase 2 trial. The 12 mg group was approaching 22-24%. These are numbers that were considered impossible through pharmacotherapy just five years ago.
During this phase, the body composition changes become increasingly dramatic. People who started with significant visceral fat see major reductions in waist circumference. The relationship between visceral fat loss and metabolic health means that many of the cardiometabolic improvements continue advancing even as the rate of weight loss begins to gradually slow. Lipid panels improve further. Inflammatory markers decrease. Insulin sensitivity continues normalizing.
If you have not yet explored the comparison between semaglutide vs tirzepatide vs retatrutide, this mid-treatment window is where the differences become most apparent.
Week 37-48: approaching peak results
The 48-week endpoint of the phase 2 trial represents the longest published clinical data for retatrutide. And the numbers are remarkable.
At 48 weeks, the least-squares mean percentage change in body weight was -8.7% in the 1 mg group, -17.1% in the combined 4 mg group, -22.8% in the combined 8 mg group, and -24.2% in the 12 mg group, compared with -2.1% in the placebo group.
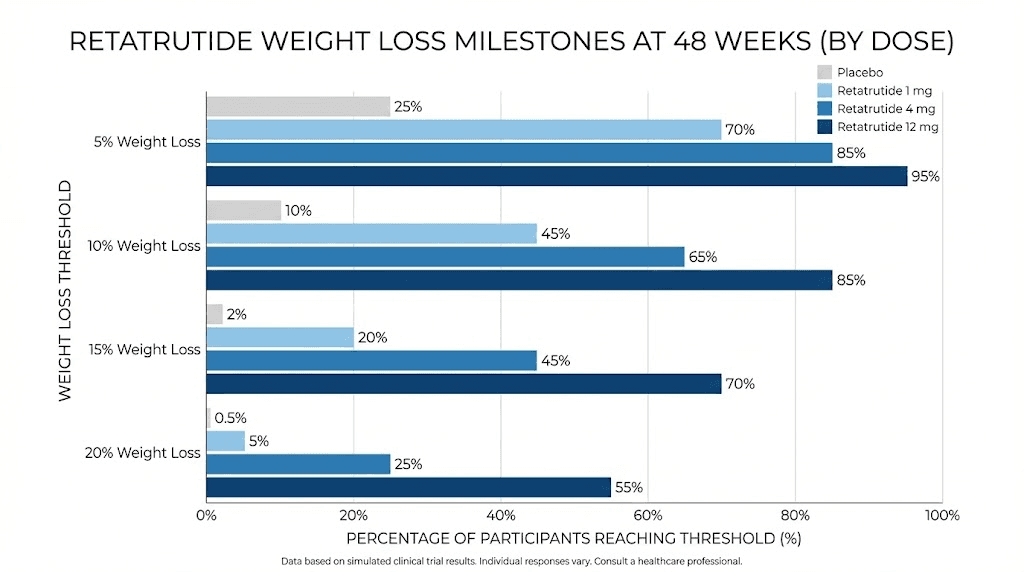
The 12 mg group lost an average of 24.2% of their body weight, which translates to approximately 57.8 pounds or 26.2 kg. Even more telling is the distribution of results. At 48 weeks on the 12 mg dose, 100% of participants achieved at least 5% weight reduction, 93% achieved at least 10%, and 83% achieved at least 15%. These response rates are unprecedented in obesity pharmacotherapy.
The phase 3 TRIUMPH trial results, announced more recently, showed even longer-term data. At 68 weeks, the treatment-regimen estimand results were -20.0% (9 mg dose, approximately 50.5 pounds) and -23.7% (12 mg dose, approximately 60 pounds). In the osteoarthritis trial, participants lost up to an average of 71.2 pounds.
The weight loss trajectory at week 37-48 is beginning to decelerate, which is physiologically normal and expected. The body reaches a new equilibrium point where reduced body mass means lower caloric needs, and the metabolic effects of the medication balance against the reduced energy requirements of a lighter body. This is not a failure of the medication. It is the natural endpoint of what pharmacotherapy can achieve, and the results represent a transformation that most people could never sustain through diet and exercise alone.
For anyone tracking their own journey, the reconstitution chart and mixing guide are essential references throughout treatment.
When does appetite suppression actually start on retatrutide
Appetite suppression is usually the first noticeable effect, and it is often what people mean when they ask whether retatrutide is "working." The timeline for this specific effect varies, but the published data provides useful benchmarks.
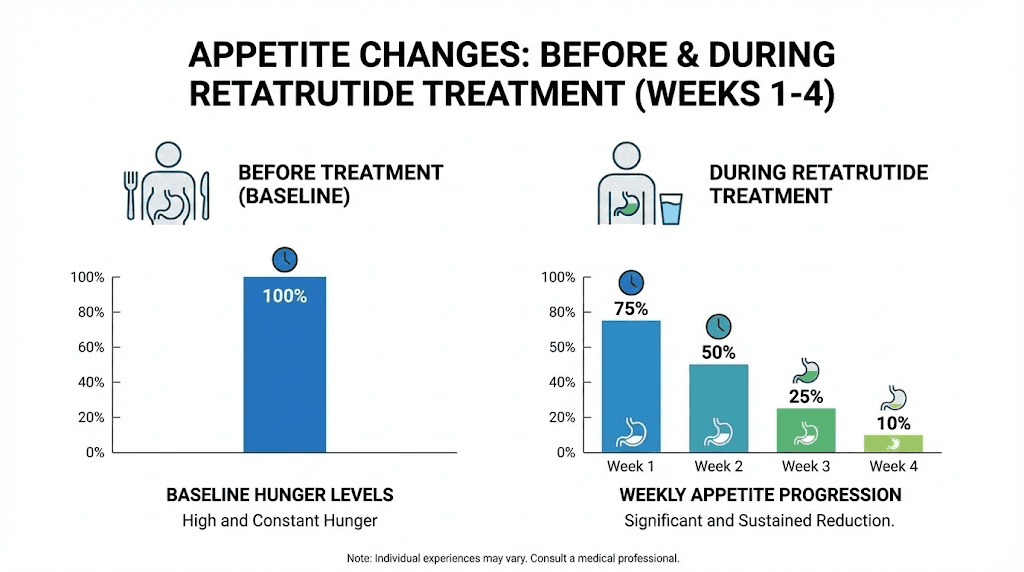
A published study examining appetite and eating behaviors during retatrutide treatment in the phase 2 trial found that 31 of 36 retatrutide-treated participants (86%) reported changes in eating behaviors within the first eight weeks of treatment. That is a high response rate, but it also means 14% did not notice significant changes during that window.
The breakdown of when appetite suppression typically begins follows a pattern. Within 24-72 hours of the first injection, some people notice a subtle decrease in hunger. This is not universal, and the effect at starting doses is mild. Between days 3 and 14, approximately 60-70% of users report some degree of noticeable appetite change. This might be as simple as leaving food on the plate for the first time or forgetting about a snack they would normally have eaten.
By weeks 2-4, as the medication reaches steady-state levels and as dose titration increases the pharmacological impact, appetite suppression becomes more consistent and more noticeable. For most people, this is when the effect transitions from "I think something might be different" to "I am definitely eating less and it requires no effort."
By weeks 4-8, particularly as doses reach 4 mg or higher, the appetite effect is typically well established. Meals naturally shrink. Cravings for calorie-dense foods diminish. The constant background noise of hunger that many people with obesity experience, what researchers call hedonic hunger, quiets down significantly.
One important nuance: appetite suppression from retatrutide is not the same as having no appetite. Most people still experience hunger at meal times. What changes is the intensity and duration of that hunger, and the amount of food required to satisfy it. The sensation of fullness arrives sooner and lasts longer. This is primarily driven by the GLP-1 receptor-mediated slowing of gastric emptying and the central appetite signaling effects that retatrutide shares with semaglutide and tirzepatide.
If you are three to four weeks into retatrutide and noticing no appetite changes at all, that does not necessarily mean the medication will not work. Some people are slower responders. The dose may need further titration. And factors like stress, sleep disruption, and medication interactions can all blunt the appetite-suppressing effect temporarily. Consult your researcher before making any dose adjustments, and review the dose schedule to understand the expected progression.
Factors that influence how quickly retatrutide works
No two people will have identical timelines on retatrutide. The clinical trial averages provide useful benchmarks, but individual results depend on several modifiable and non-modifiable factors.
Dose and titration speed
This is the single biggest variable. Someone titrating slowly (starting at 1 mg, increasing by 1-2 mg every four weeks) will see a slower onset than someone who starts at 2 mg and increases by 2 mg every two weeks. The phase 2 trial tested different starting dose strategies and found that slower titration reduced side effects but also delayed the onset of maximum weight loss. Finding the right balance between tolerability and efficacy is a conversation to have with your researcher using the retatrutide dosage chart as a reference.
Starting body weight and composition
People with higher starting body weights tend to lose more absolute weight but may show lower percentage changes in the early weeks. This is partly because the same dose of retatrutide is distributed across more body mass, producing a lower effective concentration per kilogram. Conversely, someone starting at a lower body weight may reach clinically meaningful percentage changes faster but may find that the absolute numbers feel less impressive. Body composition matters too. People with higher proportions of visceral fat, which is more metabolically active and responsive to hormonal signals, often see more dramatic early changes than those carrying primarily subcutaneous fat. The BMI requirements and starting weight influence the entire trajectory.
Diet and physical activity
Retatrutide is not a magic override for unlimited caloric intake. The medication reduces appetite and increases energy expenditure, but the degree of weight loss depends partly on what and how much you eat in the reduced-appetite state. People who use the appetite suppression as an opportunity to improve dietary quality, choosing nutrient-dense foods, adequate protein, and appropriate fiber, tend to see better and faster results than those who simply eat smaller portions of the same calorie-dense foods.
Physical activity amplifies retatrutide results by preserving muscle mass during weight loss (critical for maintaining metabolic rate), increasing the caloric deficit beyond what appetite suppression alone achieves, and improving insulin sensitivity, which supports the metabolic effects of the medication. A combination of resistance training and moderate cardiovascular exercise, scaled to individual capacity, produces the best outcomes. Protein supplementation becomes particularly important when combining exercise with GLP-1 therapy.
Previous GLP-1 medication experience
People transitioning from semaglutide or tirzepatide to retatrutide may experience different onset patterns than treatment-naive individuals. The body has already adapted to GLP-1 receptor activation, which can mean less intense initial side effects but potentially a delayed perception of "new" appetite suppression. The additional glucagon receptor activation is what provides the novel mechanism for these individuals, and its effects build more gradually. The switching timeline from tirzepatide to retatrutide and the dosage schedule after high-dose tirzepatide require careful planning with a researcher.
Individual metabolic variation
Genetics influence receptor density, receptor sensitivity, gut microbiome composition, metabolic rate, and fat distribution patterns, all of which affect how quickly any medication works. This is why clinical trials report averages with wide confidence intervals. Some people in the 12 mg retatrutide group lost more than 30% of their body weight. Others in the same group lost less than 15%. Both are valid responses to the same medication. Understanding your own response pattern requires patience and consistent tracking.
How retatrutide compares to semaglutide and tirzepatide for speed of results
One of the most practical questions people ask is whether retatrutide works faster than the alternatives. The answer requires comparing data across different trials, which involves some caveats about different study populations and designs. But the numbers paint a clear picture.
Semaglutide (single GLP-1 receptor agonist): In the STEP 1 trial, participants on semaglutide 2.4 mg achieved approximately 14.9% body weight reduction at 68 weeks. The trajectory shows meaningful weight loss beginning around weeks 4-8, with most of the reduction occurring by week 40-50 before plateauing. At 24 weeks, participants had lost approximately 10-11%. Learn more about the speed of semaglutide results.
Tirzepatide (dual GLP-1/GIP receptor agonist): In the SURMOUNT-1 trial, participants on the highest tirzepatide dose (15 mg) achieved approximately 22.5% body weight reduction at 72 weeks. The onset of meaningful weight loss was slightly faster than semaglutide, with approximately 15% reduction by week 24. The full comparison of how fast tirzepatide works provides additional context.
Retatrutide (triple GLP-1/GIP/glucagon receptor agonist): At 24 weeks, the 12 mg dose achieved 17.5% body weight reduction. At 48 weeks, it reached 24.2%. At 68 weeks in the phase 3 trial, 23.7%. The critical comparison is speed: retatrutide achieved comparable or superior weight loss in approximately half the time required by semaglutide, and in roughly two-thirds the time required by tirzepatide.
Medication | Receptors | ~24 week loss | Peak loss | Time to peak |
|---|---|---|---|---|
Semaglutide | GLP-1 | ~10-11% | ~14.9% | ~60-68 weeks |
Tirzepatide | GLP-1 + GIP | ~15% | ~22.5% | ~60-72 weeks |
Retatrutide | GLP-1 + GIP + Glucagon | ~17.5% | ~24.2% | ~48 weeks |
The speed advantage comes primarily from the glucagon receptor activation, which adds a calorie-burning component that the other medications lack. While semaglutide and tirzepatide work primarily through appetite reduction (eat less), retatrutide adds genuine metabolic acceleration (burn more). This dual approach, decreased input plus increased output, creates a steeper weight loss trajectory.
A network meta-analysis published recently confirmed this pattern, finding that retatrutide demonstrated greater absolute weight reduction compared to tirzepatide (retatrutide: MD -16.34 kg vs tirzepatide: MD -11.82 kg). The Ozempic vs retatrutide comparison and the broader semaglutide vs tirzepatide comparison pages provide detailed breakdowns of each medication for different use cases.
Side effects timeline: what to expect and when
Understanding the side effect timeline is inseparable from understanding when retatrutide starts working, because the side effects and the therapeutic effects share overlapping mechanisms. Gastrointestinal symptoms in particular are directly related to the GLP-1-mediated slowing of gastric emptying that also produces appetite suppression.
Week 1-2: initial adjustment
Mild nausea is the most common early side effect. At the 1 mg starting dose, approximately 14% of trial participants experienced nausea. This typically peaks on days 1-3 after the first injection and resolves within 4-7 days. Other possible effects include mild headache, fatigue, and slight digestive changes. Most are mild and self-limiting.
Week 3-6: dose escalation effects
Each dose increase can trigger a temporary resurgence of gastrointestinal symptoms. Nausea rates increased significantly with higher doses in the trial: 18-36% at 4 mg, 17-60% at 8 mg, and 45% at 12 mg. The wide ranges at 8 mg reflect different starting dose strategies, with participants who started at a higher initial dose experiencing more GI symptoms.
Diarrhea, vomiting, and constipation also appeared during this phase, though less frequently than nausea. These effects were dose-related and mostly mild to moderate in severity. The clinical trial data showed that using a lower starting dose (2 mg vs 4 mg initial) significantly reduced the incidence and severity of these symptoms.
This is important practical knowledge. If you are experiencing significant side effects, the solution is often not to stop the medication but to slow the titration. Adding an extra week or two at each dose level allows the body more time to adapt. Review peptide safety considerations for additional management strategies.
Week 7-12: adaptation phase
For most people, gastrointestinal side effects diminish substantially by weeks seven to twelve, even as they continue titrating to higher doses. The body adapts to the delayed gastric emptying. Nausea becomes less frequent and less intense. Bowel patterns normalize.
Some less common effects that may appear during this phase include increased heart rate (a known class effect of GLP-1 receptor agonists), skin hyperesthesia in rare cases, and temporary elevations in liver enzymes (alanine aminotransferase). These were monitored in the clinical trial and did not lead to significant safety concerns at the doses studied.
Week 12 and beyond: steady state
By week twelve and beyond, most participants in the clinical trials reported minimal ongoing side effects. The body has adapted to the medication. Appetite suppression continues without the initial gastrointestinal disruption. Energy levels typically normalize or improve. The metabolic benefits continue building without the tolerability challenges of the early weeks.
Discontinuation rates from the phase 2 trial due to side effects were generally low, though the broader literature on GLP-1 receptor agonists notes that discontinuation rates across the class can reach 20-50% within the first year. Many of these discontinuations occur early, during the adjustment phase, which reinforces the importance of proper titration and avoiding common mistakes that new users make.
For a comprehensive understanding of side effects management, the GLP-1 fatigue guide, GLP-1 headache guide, and hair loss guide cover specific concerns in detail.
Signs retatrutide is working even before the scale moves
Weight is the most tracked metric, but it is also the most misleading in the early weeks. Water retention, bowel contents, hormonal fluctuations, and time of day can swing the scale by 3-5 pounds in either direction regardless of actual fat loss. This leads many people to prematurely conclude that retatrutide is not working when, in fact, it is working exactly as designed.
Here are the early signals that indicate retatrutide is active and building toward results.
Appetite changes. Feeling full sooner during meals. Forgetting about snacks. Not thinking about food between meals. Finding that a portion that used to feel normal now feels like too much. These are the earliest and most reliable indicators of GLP-1 and GIP receptor activation.
Taste and food preference shifts. Many GLP-1 class medications alter food preferences. Calorie-dense, highly palatable foods like fried foods, sweets, and fast food become less appealing. Some people describe it as the food looking good but not tasting as satisfying as it used to. This reflects central appetite circuit modulation in the brain.
Digestive changes. Slower digestion, occasional mild nausea, a feeling of food sitting in the stomach longer than usual. These are direct evidence of GLP-1-mediated gastric emptying delay. They are temporary inconveniences that confirm the mechanism is active.
Energy fluctuations. Some people experience mild fatigue in the first 1-2 weeks as their caloric intake decreases and the body adjusts to new metabolic signaling. Others notice improved energy as insulin dynamics stabilize. Both patterns indicate the medication is pharmacologically active.
Blood glucose changes. If you monitor blood glucose, improvements may appear before any weight change. Retatrutide directly improves glucose regulation through GLP-1-mediated insulin secretion and GIP-mediated glucose-dependent insulin effects. Fasting glucose drops and post-meal glucose spikes flatten. In the clinical trial of participants with type 2 diabetes, HbA1c reductions of -1.88% to -2.02% were observed at the 8 mg and 12 mg doses by 24 weeks.
Reduced cravings. The specific, intense desire for certain foods, particularly carbohydrate-heavy and sugar-heavy options, diminishes. This is different from general appetite reduction. It reflects modulation of reward pathways in the brain that drive hedonic eating behavior.
Track these signals during the first month. If you are experiencing any of them, the medication is working. The scale will catch up. And if you want to understand the broader experience of how these peptides feel in the early stages, that guide provides useful context.
How to maximize your retatrutide results
The clinical trial participants who achieved the best results were not simply taking the medication and waiting. While retatrutide does the heavy lifting through pharmacology, several strategies can amplify and accelerate the results.
Prioritize protein intake
During rapid weight loss, preserving muscle mass is critical. Muscle is metabolically active tissue, meaning it burns calories at rest. Losing muscle along with fat reduces metabolic rate and creates the conditions for weight regain. Aim for 0.7-1.0 grams of protein per pound of ideal body weight daily. This is challenging when appetite is suppressed, which is why protein shakes formulated for GLP-1 users become valuable tools. Prioritize protein at every meal. Plan meals around the protein source first, then add vegetables and complex carbohydrates around it.
Maintain consistent injection timing
The six-day half-life of retatrutide means the medication maintains relatively stable blood levels throughout the week. However, injecting at the same time on the same day each week optimizes this stability. Many people find that injecting in the evening reduces the impact of any nausea, since they sleep through the peak absorption window. Choose your injection site carefully and follow proper injection technique.
Resistance training over cardio
While any exercise is better than none, resistance training provides disproportionate benefits during GLP-1-mediated weight loss. It directly counters the muscle loss that occurs during caloric deficit, improves insulin sensitivity beyond what the medication alone achieves, increases resting metabolic rate, and improves body composition even when the scale number stalls. Three to four sessions per week of full-body resistance training, progressive in nature, creates the best synergy with retatrutide pharmacology. The muscle growth guide and athletic performance overview provide complementary information.
Manage hydration and electrolytes
Reduced food intake means reduced intake of water-containing foods and electrolyte-rich foods. This can lead to headaches, fatigue, dizziness, and muscle cramps that get misattributed to the medication itself. Increase water intake deliberately, targeting at least 80-100 ounces daily. Supplement sodium, potassium, and magnesium if needed. Many of the early "side effects" people report are actually dehydration and electrolyte imbalance rather than direct pharmacological effects.
Follow the titration schedule faithfully
The urge to increase doses faster to get faster results is understandable but counterproductive. Rapid titration increases side effects, which increases the likelihood of missed doses or discontinuation. A steady, protocol-driven titration gives the body time to adapt at each level and produces better long-term adherence and results. Use the retatrutide dosage calculator to plan your titration path and reference the detailed dose schedule for standard protocols.
Track beyond the scale
Weekly weigh-ins are useful but insufficient. Measure waist circumference, hip circumference, and if possible body fat percentage at regular intervals. Take progress photos monthly under consistent lighting and conditions. Track energy levels, sleep quality, and mood. These metrics often show improvement before the scale moves and provide motivation during inevitable plateaus.
What to do if retatrutide does not seem to be working
Some people reach week four, six, or even eight without seeing the results they expected. Before concluding that the medication has failed, work through this systematic assessment.
Check the dose. Are you still on the starting dose? Results at 1-2 mg are minimal for most people. The clinical data shows that meaningful weight loss typically requires doses of 4 mg or higher. If you have not yet titrated beyond the starting dose, it is too early to evaluate effectiveness.
Verify injection technique. Subcutaneous injection requires the needle to reach the fatty tissue beneath the skin but not the muscle below. Injecting too shallow (intradermal) or too deep (intramuscular) can affect absorption rates and pharmacokinetics. Review proper injection technique to ensure correct administration.
Assess dietary intake honestly. The appetite suppression from retatrutide is real, but it does not eliminate eating entirely. If you are consuming calorie-dense foods even in smaller quantities, the caloric deficit may not be sufficient for measurable weight loss. Liquid calories from alcohol, sugary beverages, and cream-heavy coffee drinks can bypass the satiety signals entirely. Consider foods to avoid on GLP-1 therapy.
Evaluate sleep and stress. Cortisol from chronic stress and poor sleep directly opposes the metabolic effects of retatrutide. Elevated cortisol promotes fat storage, increases insulin resistance, and stimulates appetite through pathways that partially bypass GLP-1 signaling. If you are sleeping fewer than seven hours per night or experiencing significant chronic stress, these factors may be blunting the medication response.
Rule out medication interactions. Certain medications can interfere with GLP-1 class drug effects. Corticosteroids, some antidepressants, insulin-secreting medications, and others may alter the metabolic environment in ways that reduce retatrutide effectiveness. Discuss your complete medication list with your researcher.
Consider the timeline. If you are fewer than eight weeks into treatment and have not yet reached at least 4 mg, you are in the adjustment phase. The medication is building in your system and the dose is not yet at a therapeutic level for weight loss. Give it time.
Check storage and handling. Peptide potency depends on proper storage and handling. If your retatrutide was exposed to high temperatures, was not refrigerated properly, or was reconstituted incorrectly, the active ingredient may have degraded. The peptide storage guide covers proper handling in detail.
If you have been on an adequate dose for 8-12 weeks with confirmed proper technique, storage, and supportive lifestyle factors, and you are still seeing no response whatsoever, you may be a non-responder to this particular medication. This is uncommon but not impossible. The mazdutide comparison and survodutide comparison explore alternative options in the next-generation obesity pharmacotherapy space.
The clinical trial data in detail
For those who want the precise numbers from the published research, here is a comprehensive summary of the key data points that inform the retatrutide timeline.
Phase 1b trial (12 weeks, type 2 diabetes population)
This early-stage trial established proof of concept. In participants with type 2 diabetes, treatment with retatrutide resulted in a placebo-adjusted least-squares mean weight reduction of 8.96 kg (approximately 10%) in the highest-dose 12 mg group after just 12 weeks. This was the first signal that retatrutide could produce exceptionally rapid weight loss.
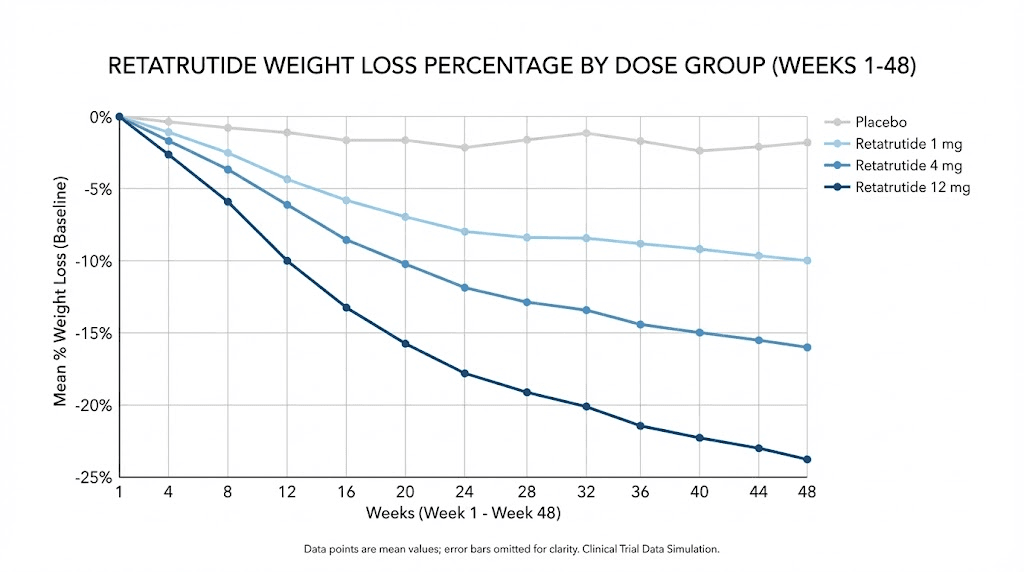
Phase 2 trial (48 weeks, obesity population)
Published in the New England Journal of Medicine (Jastreboff et al.), this trial enrolled 338 adults with a body mass index of 30 or higher, or 27-30 with at least one weight-related condition. Participants were 51.8% men, which is notable because many obesity trials skew heavily female.
Participants were randomized 2:1:1:1:1:2:2 to receive placebo or retatrutide at 1 mg, 4 mg (starting at 2 mg), 4 mg (starting at 4 mg), 8 mg (starting at 2 mg), 8 mg (starting at 4 mg), or 12 mg (starting at 2 mg) administered subcutaneously once weekly for 48 weeks.
Key weight loss results at 48 weeks by group:
Dose group | 24-week change | 48-week change |
|---|---|---|
Placebo | -1.6% | -2.1% |
1 mg | -7.2% | -8.7% |
4 mg (combined) | -12.9% to -14.5% | -17.1% |
8 mg (combined) | -14.5% to -17.5% | -22.8% |
12 mg | -17.5% | -24.2% |
Response rates at 48 weeks (12 mg dose):
100% achieved greater than or equal to 5% weight loss
93% achieved greater than or equal to 10% weight loss
83% achieved greater than or equal to 15% weight loss
The cardiometabolic improvements were also significant, with reductions in blood pressure, triglycerides, LDL cholesterol, HbA1c, fasting glucose, and fasting insulin across all dose groups.
Phase 2a MASLD trial (48 weeks, liver disease population)
In participants with metabolic dysfunction-associated steatotic liver disease (formerly NAFLD/NASH), retatrutide demonstrated remarkable liver fat reduction. The 12 mg group showed 86% mean relative reduction in liver fat, with 93% of participants achieving normal liver fat levels below 5% at 48 weeks.
Phase 3 TRIUMPH trials
The most recent data comes from the phase 3 program. The treatment-regimen estimand results at 68 weeks showed -20.0% body weight change (approximately 50.5 pounds) in the 9 mg group and -23.7% (approximately 60 pounds) in the 12 mg group. In the osteoarthritis trial, participants lost up to an average of 71.2 pounds with substantial relief from osteoarthritis pain.
These phase 3 results confirm and extend the phase 2 findings, establishing retatrutide as potentially the most effective obesity pharmacotherapy ever studied. For researchers following the development pipeline, the retatrutide availability and cost guide track the latest access information.
Long-term considerations and weight maintenance
A question that naturally follows "how long does retatrutide take to work" is "how long do you need to take it?" The honest answer, based on all available data from GLP-1 class medications, is that discontinuation typically leads to weight regain.
The withdrawal experience from semaglutide provides instructive context. Studies show that within 12-18 months of stopping semaglutide, participants regained approximately two-thirds of the weight they had lost. Similar patterns are expected with retatrutide, because the underlying metabolic and appetite-regulating mechanisms revert when the medication is removed.
This does not mean retatrutide is a failure if you cannot take it indefinitely. Even temporary weight loss produces lasting health benefits including improved cardiovascular risk markers, reduced joint stress, improved sleep apnea, and better metabolic function. And for many people, a period of substantial weight loss creates a window to establish exercise habits and dietary patterns that are easier to maintain at a lower body weight.
The key insight is managing expectations. Retatrutide is a treatment, not a cure. It works for as long as you take it, and its effects are dose-dependent and duration-dependent. Planning for long-term use, including the cost implications, is a practical necessity.
For researchers exploring their options, SeekPeptides provides comprehensive protocol guidance, dosing calculators, and community support to help navigate the complexities of long-term peptide therapy. Understanding not just when the medication starts working but how to sustain those results requires exactly the kind of evidence-based, personalized approach that SeekPeptides was built to provide.
Combining retatrutide with other peptides
Some researchers explore combinations of retatrutide with other peptides to enhance specific outcomes. While clinical trial data on combinations is limited, the theoretical rationale and community experience provide some guidance.
Cagrilintide with retatrutide is one combination that has generated interest. Cagrilintide targets the amylin receptor, adding another appetite-suppressing pathway that does not overlap with GLP-1, GIP, or glucagon signaling. The combination could theoretically produce additive appetite suppression, though formal studies are needed.
Tesamorelin with retatrutide targets visceral fat specifically through growth hormone-releasing hormone pathways. For individuals whose primary concern is abdominal fat rather than total body weight, this combination addresses both the metabolic environment (retatrutide) and the growth hormone axis (tesamorelin).
For muscle preservation during rapid weight loss, some researchers incorporate peptides like BPC-157 for tissue repair and recovery, or TB-500 for its regenerative properties. These do not directly affect weight loss but support the body during a period of significant metabolic change.
The peptide stack calculator helps researchers plan combinations, and the stacking guide covers general principles. Always research potential interactions thoroughly before combining any peptides, and consult with a knowledgeable practitioner.
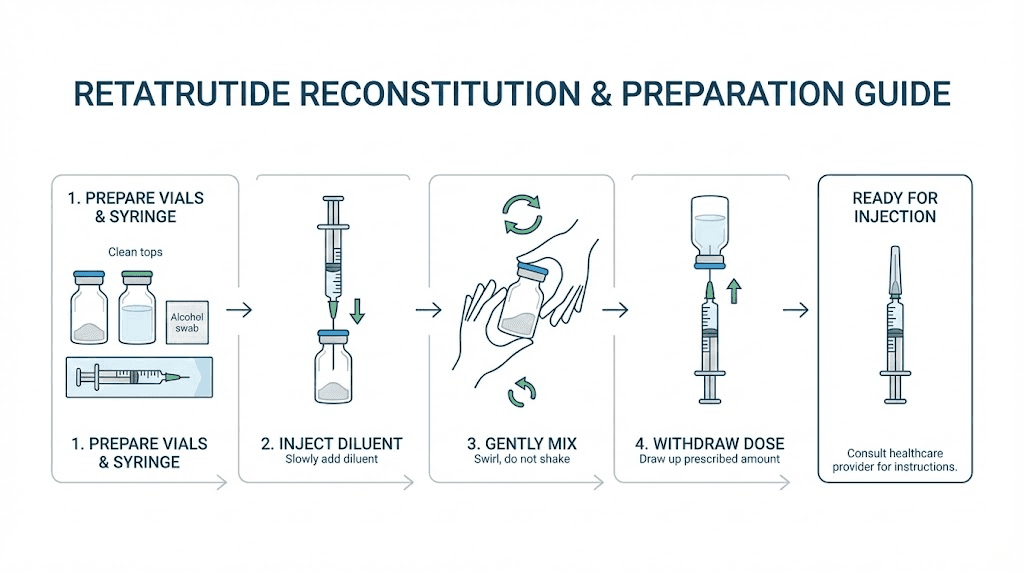
Proper preparation and administration
The timeline of retatrutide results assumes proper preparation and administration. Errors at any stage can reduce potency and delay onset.
Reconstitution. Retatrutide typically arrives as a lyophilized powder that requires reconstitution with bacteriostatic water. The mixing ratio for 10 mg retatrutide and the reconstitution chart provide exact volumes for accurate dosing. Use the peptide reconstitution calculator to determine the precise amount of bacteriostatic water needed based on your vial concentration and desired dose per injection.
Storage. Once reconstituted, retatrutide must be refrigerated at 36-46 degrees Fahrenheit (2-8 degrees Celsius). The shelf life guide covers duration, and the storage guide addresses specific handling questions. Exposure to heat, direct sunlight, or repeated freeze-thaw cycles degrades the peptide and reduces effectiveness.
Injection technique. Subcutaneous injection into the abdomen, thigh, or upper arm is standard. Rotate injection sites to avoid lipodystrophy. The injection site guide covers anatomical landmarks, and the broader injection guide covers technique for all subcutaneous peptides. Proper technique ensures consistent absorption and predictable pharmacokinetics, which is essential for staying on schedule with the dosing protocol.
Sourcing. The potency, purity, and sterility of the retatrutide you use directly impacts results. Impure or degraded product will not produce clinical trial-level results regardless of dose or protocol. Research sourcing options carefully, verify third-party testing, and review the purchasing guide for due diligence criteria.
Frequently asked questions
How long until I feel appetite suppression on retatrutide?
Most people notice some degree of reduced appetite within the first one to three weeks, with 86% of clinical trial participants reporting eating behavior changes within the first eight weeks. The effect becomes more pronounced as the dose increases through the titration schedule.
Is retatrutide faster than semaglutide for weight loss?
Yes. Retatrutide achieved approximately 17.5% weight loss at 24 weeks compared to semaglutide approximately 10-11% at the same time point. The triple-receptor mechanism, particularly the added glucagon receptor activation, accelerates results. See the full retatrutide vs semaglutide comparison.
What if I do not lose weight in the first month?
The first month is primarily an adjustment period, especially if you are on a starting dose of 1-2 mg. Clinically meaningful weight loss typically begins as the dose reaches 4 mg or higher. Do not evaluate the medication effectiveness until you have been on a therapeutic dose for at least four to six weeks.
How much weight can I expect to lose on retatrutide?
Phase 2 trial data shows average weight loss of 24.2% at 48 weeks on the 12 mg dose, which translates to roughly 58 pounds for the average participant. Phase 3 data shows up to 71.2 pounds at 68 weeks. Individual results vary based on starting weight, dose, diet, and activity level. The weight loss dosage chart helps estimate results by dose.
Do the side effects go away?
For most people, yes. Gastrointestinal side effects are most pronounced during the first two weeks and after each dose increase, then diminish as the body adapts. By week eight to twelve, most people report minimal ongoing side effects. Slower titration reduces both the severity and duration of side effects.
Can I switch from tirzepatide to retatrutide?
Yes, many people transition between GLP-1 class medications. The dosage schedule after high-dose tirzepatide provides specific guidance for this transition, including starting dose recommendations and what to expect during the switch.
Is retatrutide FDA approved?
As of early 2026, retatrutide is not yet FDA approved. Phase 3 TRIUMPH trials are expected to complete by mid-2026, with potential FDA approval in 2027. Current access is through clinical trials and research channels. The availability guide tracks the latest access information.
How long do I need to take retatrutide?
Based on data from other GLP-1 class medications, weight regain typically occurs after discontinuation. Most researchers anticipate that long-term treatment will be necessary to maintain results, similar to how blood pressure medication must be continued to maintain blood pressure control.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, dosing calculators, and a community of thousands who have navigated these exact questions. Whether you are just starting retatrutide or looking to optimize your results, SeekPeptides members access detailed protocol guidance that goes far beyond what any single article can provide.
In case I do not see you, good afternoon, good evening, and good night. May your titration stay steady, your appetite stay managed, and your results stay consistent.