Feb 24, 2026

Experienced researchers know something that beginners do not about retatrutide. The peptide is powerful on its own. Remarkably powerful. Phase 2 trials showed up to 24% body weight reduction at the highest dose, and Phase 3 data from the TRIUMPH program pushed that number even higher, reaching 28.7% in participants with obesity and knee osteoarthritis. But the researchers reporting the most dramatic metabolic changes are not running retatrutide alone.
They are adding cagrilintide.
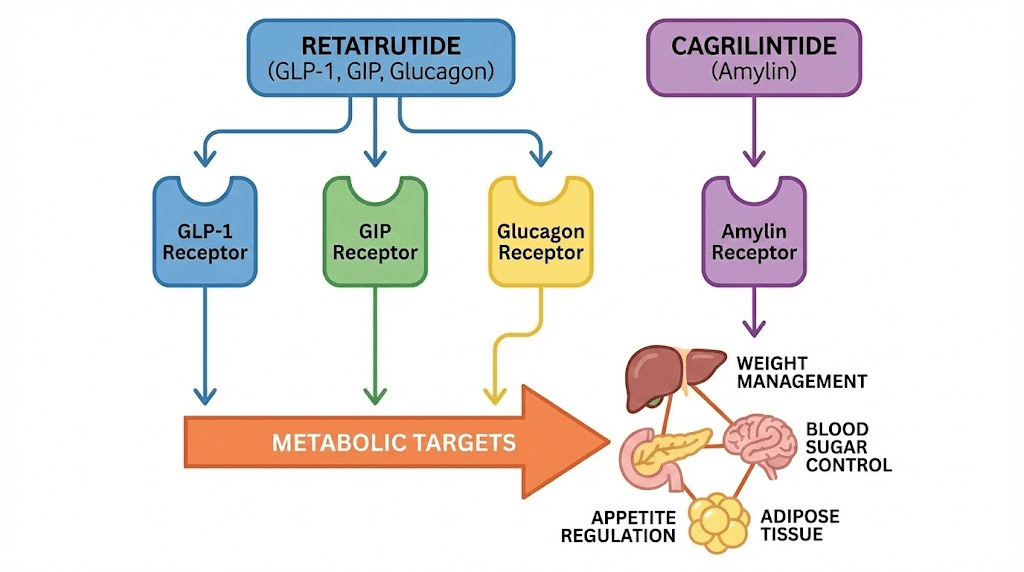
This combination targets four distinct metabolic pathways simultaneously, something no single compound currently achieves. Retatrutide activates GLP-1, GIP, and glucagon receptors through a single peptide. Cagrilintide works through an entirely separate system, the amylin receptor pathway, targeting different neuronal populations in the brainstem that regulate satiety and appetite. The result is a quadruple agonist approach to metabolic optimization that goes beyond what even the most aggressive single-agent protocols can deliver.
But here is where most people get it wrong.
They either dose the cagrilintide too aggressively alongside retatrutide, creating unnecessary gastrointestinal distress, or they under-dose it to the point of irrelevance. The margin between an effective stack and a wasted one comes down to understanding the specific dosage protocols, titration timing, and how these two compounds interact at the receptor level. This guide covers everything researchers need to know about combining cagrilintide with retatrutide, from the specific dosing schedules backed by clinical data, to reconstitution instructions, to the week-by-week titration protocol that minimizes side effects while maximizing metabolic coverage. Whether transitioning from tirzepatide or building a combination protocol from scratch, the information here provides the evidence-based framework for making informed decisions about this advanced peptide stack. SeekPeptides members already access detailed stacking protocols and dosing tools through our peptide stack calculator for combinations like this one.
Why researchers combine cagrilintide with retatrutide
The rationale behind this combination is not complicated, but it is deeply rooted in receptor pharmacology. Retatrutide already hits three major metabolic targets. Adding cagrilintide brings a fourth. And that fourth pathway, the amylin system, does not just add incremental benefit. It targets an entirely different set of neurons in the brain, creating what researchers call complementary satiety signaling.
Here is why that matters.
GLP-1 receptor agonists reduce appetite primarily through the hypothalamus and vagal afferent pathways. Amylin analogs like cagrilintide work through the area postrema and nucleus tractus solitarius in the brainstem, a region that processes satiety signals through a completely separate neuronal network. Research published in eBioMedicine confirmed that cagrilintide lowers body weight specifically through brain amylin receptors 1 and 3 (AMY1R and AMY3R), and these are distinct from the receptors that GLP-1 agonists activate. When both systems are engaged simultaneously, the appetite suppression is more robust and more sustained than either pathway alone can produce.
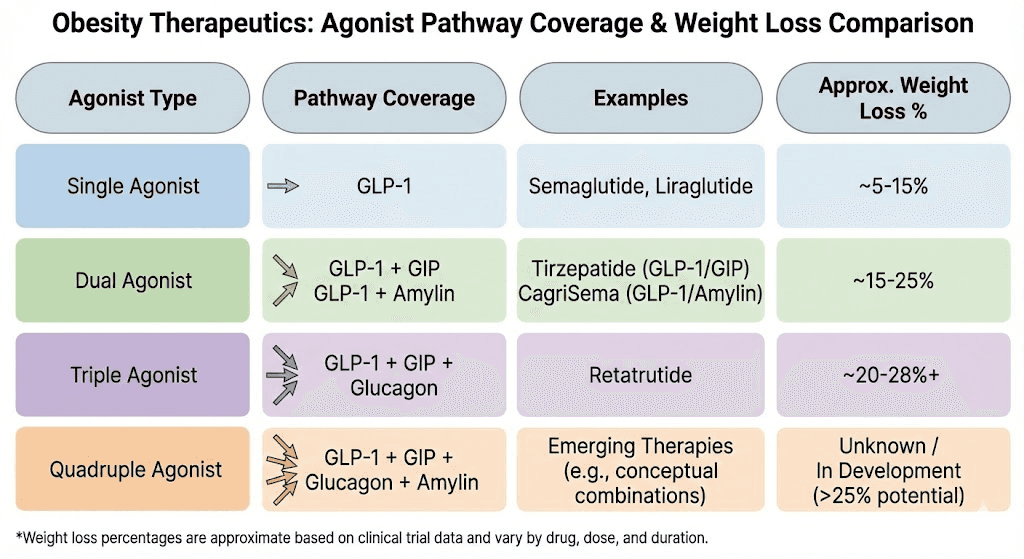
The clinical proof of concept comes from CagriSema, the combination of cagrilintide with semaglutide. In the REDEFINE 1 trial, published in the New England Journal of Medicine, participants receiving the combination achieved 20.4% mean body weight reduction at 68 weeks. Cagrilintide alone produced 11.5%. Semaglutide alone produced 14.9%. The combination outperformed both individual agents by a meaningful margin, and 60% of participants on the combination lost at least 20% of their body weight. The logic behind pairing cagrilintide with retatrutide follows the same principle, but with an even more powerful base compound.
Retatrutide is not just a GLP-1 agonist. It is a triple agonist that also activates GIP and glucagon receptors, giving it metabolic advantages that semaglutide cannot match. The comparison between semaglutide, tirzepatide, and retatrutide shows that each successive generation of multi-receptor agonist delivers greater weight loss outcomes. If adding amylin agonism to a single GLP-1 agonist (CagriSema) produces 20.4% weight loss, the theoretical potential of adding it to a triple agonist is significant. This is why clinics offering peptides for fat loss are increasingly exploring this stack.
No clinical trial has studied the specific combination of retatrutide plus cagrilintide in humans. That is an important caveat. But the mechanistic rationale is strong, and early anecdotal reports from researchers and clinics using the combination suggest meaningful improvements in appetite control, energy expenditure, and overall metabolic response compared to retatrutide alone.
Understanding retatrutide: the triple receptor agonist
Before diving into combination dosing, understanding each compound individually is essential. Retatrutide is a 39-amino-acid peptide engineered from a GIP peptide backbone and modified to simultaneously activate three receptor systems. It has a half-life of approximately six days, which supports once-weekly subcutaneous injection. This long duration of action is one of the reasons it has generated so much attention in the weight loss research community.
Each receptor contributes distinct metabolic effects.
GLP-1 receptor activation reduces appetite, slows gastric emptying, promotes insulin secretion from pancreatic beta cells, and creates the characteristic feeling of fullness that GLP-1 agonist users report. This is the same pathway that semaglutide and the GLP-1 component of tirzepatide target. If you have used any GLP-1 based therapy before, you are already familiar with these effects. Our guide on how long GLP-1 takes to start working covers the typical onset timeline.
GIP receptor activation enhances insulin sensitivity and promotes fat metabolism. GIP (glucose-dependent insulinotropic polypeptide) was once considered primarily an incretin hormone, but research has revealed its broader metabolic role. GIP receptor activation in adipose tissue influences lipid storage and mobilization, and in the brain, it contributes to appetite regulation through mechanisms that complement GLP-1 signaling. This is the same additional pathway that tirzepatide uses beyond semaglutide.
Glucagon receptor activation is what truly sets retatrutide apart. Glucagon increases energy expenditure, promotes fat oxidation in the liver, reduces lipogenesis, and induces lipolysis in adipose tissue. While this might seem counterintuitive since glucagon raises blood glucose, the combined effect with GLP-1 and GIP agonism keeps glucose levels in check while dramatically increasing the metabolic rate. This glucagon component is the reason retatrutide produces greater weight loss than dual agonists like tirzepatide.
In the Phase 2 trial published in the New England Journal of Medicine, the retatrutide dosage chart showed clear dose-response results. At 1 mg weekly, participants lost modest amounts. At 4 mg, the losses became significant. At 8 mg and 12 mg, the results were dramatic, with the highest dose producing an average 24.2% body weight reduction over 48 weeks. The Phase 3 TRIUMPH-4 trial pushed these numbers even higher, with the 12 mg dose achieving 28.7% mean weight loss at 68 weeks. The standard retatrutide dose schedule involves titration from 1 mg up to 8-12 mg over approximately 16 weeks, increasing every four weeks.
Understanding the starting dose of retatrutide is critical because it affects how you layer in cagrilintide. Starting too high on both compounds simultaneously is the most common mistake researchers make, and it leads to avoidable gastrointestinal problems. Use the retatrutide dosage calculator to determine weight-based starting points before planning the combination.
Understanding cagrilintide: the amylin analog
Cagrilintide is a long-acting amylin analog developed by Novo Nordisk. Natural amylin is a 37-amino-acid peptide hormone co-secreted with insulin from pancreatic beta cells in response to food intake. It plays a crucial role in postprandial glucose regulation and satiety signaling that is entirely separate from the incretin system.
The problem with natural amylin is that it aggregates rapidly and has a very short half-life, making it impractical for therapeutic use. Cagrilintide was engineered from the human amylin backbone with modifications inspired by salmon calcitonin, and it includes a fatty acid chain that enables reversible albumin binding. This gives it a half-life long enough to support once-weekly dosing, a major practical advantage over earlier amylin-based therapies like pramlintide, which required multiple daily injections.
At the receptor level, cagrilintide is a non-selective agonist of the calcitonin receptor (CTR) and amylin receptors (AMYRs). Research published in Nature Communications detailed the structural and dynamic features of how cagrilintide binds to these receptors. The weight loss effect depends specifically on AMY1R and AMY3R activation, as confirmed by a study in eBioMedicine that showed cagrilintide lowers body weight through these specific receptor subtypes in the brainstem.
The side effect profile of cagrilintide is noteworthy. In clinical trials, the most common adverse effects were gastrointestinal, including nausea, vomiting, diarrhea, and constipation. However, these were predominantly mild to moderate and transient. A systematic review and meta-analysis published in PMC found that cagrilintide actually had the lowest odds ratio for nausea, vomiting, and diarrhea compared to other incretin-based therapies. This is significant for combination protocols because it suggests that adding cagrilintide to retatrutide may not dramatically increase the gastrointestinal burden, provided the titration is managed correctly.
As monotherapy, cagrilintide produced 11.8% weight loss in the Phase 3 REDEFINE 1 trial at 68 weeks with the 2.4 mg dose. While this is less than semaglutide or tirzepatide, the value of cagrilintide is not as a standalone compound. Its value is in what it adds when combined with GLP-1 agonism. The cagrilintide and semaglutide combination (CagriSema) demonstrated this clearly, with the combination exceeding the sum of individual effects. Novo Nordisk has already filed for FDA approval of CagriSema based on these results.
For researchers interested in sourcing cagrilintide, understanding the standard cagrilintide dosing protocols is the first step. The clinical titration starts at 0.25 mg weekly and increases every four weeks up to a maintenance dose of 2.4 mg weekly, though some research protocols extend up to 4.5 mg. The compound is typically supplied in lyophilized form requiring reconstitution, and men may have specific considerations covered in our cagrilintide for men guide.
How four metabolic pathways create synergy
The real power of combining cagrilintide with retatrutide is not simply that you activate more receptors. It is that you activate receptors on different neuronal populations that independently regulate appetite, metabolism, and energy balance. Understanding this distinction is essential for appreciating why this combination may produce results beyond what any triple agonist alone can achieve.
Let us start with the brainstem.
The dorsal vagal complex (DVC) in the brainstem is where much of the appetite regulation occurs at a fundamental level. This complex includes the nucleus tractus solitarius (NTS), the area postrema (AP), and the dorsal motor nucleus of the vagus nerve (DMV). Both amylin and GLP-1 receptor agonists act within this region. However, and this is the critical finding, they activate different populations of neurons within the DVC. Research on brainstem mechanisms of amylin-induced anorexia confirmed that amylin-responsive and GLP-1-responsive neurons are largely separate populations. This means combining the two does not create redundancy. It creates additive and potentially synergistic suppression of appetite through parallel circuits.
Now add the GIP and glucagon components from retatrutide.
GIP receptor activation in the brain and adipose tissue influences energy partitioning and fat metabolism through mechanisms distinct from both GLP-1 and amylin. Glucagon receptor activation increases thermogenesis and energy expenditure primarily through hepatic and adipose tissue effects. Neither of these pathways overlap significantly with amylin signaling. The four-pathway model therefore targets appetite (GLP-1 and amylin through separate neuronal circuits), fat metabolism (GIP and glucagon), energy expenditure (glucagon primarily), and glucose homeostasis (GLP-1, GIP, and amylin all contribute through different mechanisms).
Think of it this way. Semaglutide suppresses appetite through one channel. Tirzepatide suppresses appetite through two channels. Retatrutide works through three channels, and the glucagon component adds an energy expenditure effect that is absent from the other two. Cagrilintide adds a fourth channel that specifically enhances satiety signaling through brainstem amylin receptors. Each additional pathway fills a gap that the others cannot reach.
For researchers tracking their metabolic response, metabolic rate changes and energy level shifts become important markers. The combination of all four pathways should theoretically produce earlier onset of appetite suppression, more sustained fullness between meals, increased resting energy expenditure from the glucagon component, and a reduced tendency toward the metabolic adaptation that commonly causes weight loss plateaus on single-agent therapy. The fat loss peptide landscape is rapidly moving toward multi-receptor combinations for exactly these reasons.
Cagrilintide dosage protocols for the retatrutide stack
Now for the practical details that researchers actually need. The cagrilintide dosing in a combination with retatrutide requires a more conservative approach than monotherapy dosing, at least during the initial titration period. This is because both compounds affect gastrointestinal motility and appetite through their respective mechanisms, and layering them too aggressively creates compounding GI effects that can derail compliance.
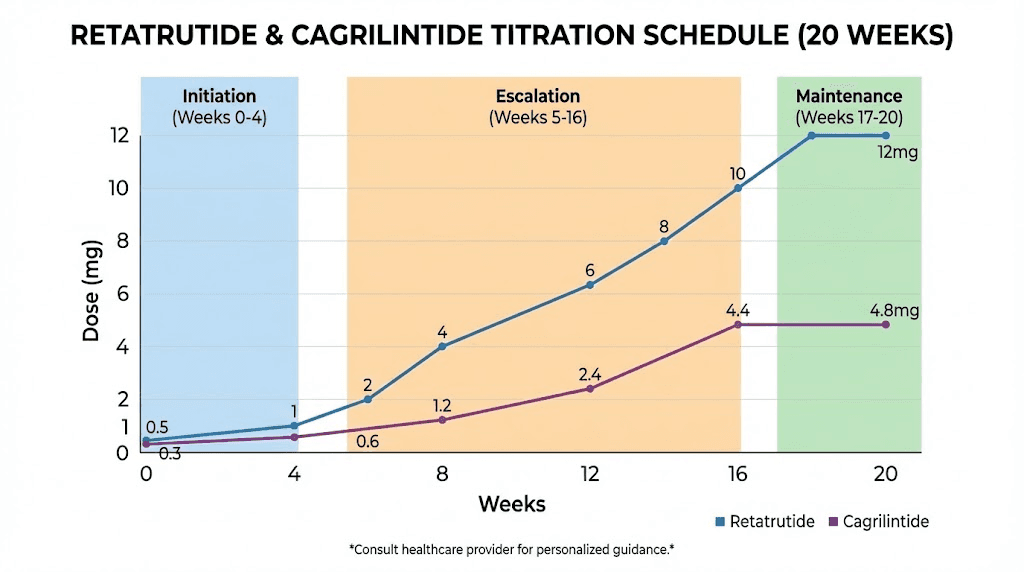
The standard cagrilintide monotherapy titration follows this schedule: 0.25 mg weekly for weeks 1-4, then 0.5 mg for weeks 5-8, then 1.0 mg for weeks 9-12, then 1.7 mg for weeks 13-16, and finally 2.4 mg for week 17 onward as the maintenance dose. In a combination protocol, the same titration applies, but the timing of when you introduce cagrilintide relative to your retatrutide titration becomes the critical variable.
Starting cagrilintide when already on retatrutide
If you are already established on retatrutide at a stable dose (meaning you have been at your current retatrutide dose for at least four weeks without significant GI issues), you can introduce cagrilintide at its lowest starting dose of 0.25 mg weekly. This is the safest approach and the one most researchers use. The key is that you do not escalate both compounds simultaneously. Hold your retatrutide dose steady while titrating cagrilintide up. Only after reaching your target cagrilintide dose should you consider any further increases to retatrutide.
Starting both compounds together from baseline
If beginning both compounds from scratch, start retatrutide first. Run the initial retatrutide starting dose of 1 mg weekly for the first four weeks. Assess tolerance. If GI side effects are manageable, add cagrilintide at 0.25 mg weekly starting in week 5, while simultaneously increasing retatrutide to 2 mg as per its standard titration. From this point forward, escalate only one compound at a time. If increasing retatrutide from 2 mg to 4 mg, hold cagrilintide at its current dose. If increasing cagrilintide from 0.25 mg to 0.5 mg, hold retatrutide steady.
Cagrilintide target doses in the combination
The optimal cagrilintide dose in the combination context appears to be between 1.0 mg and 2.4 mg weekly based on available data. The CagriSema trials used 2.4 mg as the target, and the Phase 3 monotherapy data also used 2.4 mg. However, some clinics offering the retatrutide-cagrilintide combination use a ratio of approximately 5:1 (retatrutide to cagrilintide), which at a retatrutide dose of 10-12 mg would put cagrilintide at 2.0-2.5 mg. Use our peptide calculator to determine exact volumes based on your reconstitution concentration.
The best time to administer both injections is the same day, but at different injection sites. Many researchers choose to inject both on the same morning to maintain a consistent weekly schedule. Injection sites should be rotated, and our guides on how to inject GLP-1 compounds and optimal injection locations cover the specific techniques. Both compounds are administered subcutaneously, typically in the abdomen, thigh, or upper arm.
Retatrutide titration schedule in the combination
The retatrutide titration in a combination protocol follows the same fundamental escalation pattern as monotherapy, but with two important modifications. First, the escalation timeline may be extended if GI tolerance is a concern. Second, the final maintenance dose may be lower than what you would use with retatrutide alone, because the cagrilintide component provides additional appetite suppression and metabolic benefit that reduces the need for maximum retatrutide dosing.
The standard retatrutide dose schedule is:
Weeks 1-4: 1 mg once weekly
Weeks 5-8: 2 mg once weekly
Weeks 9-12: 4 mg once weekly
Weeks 13-16: 8 mg once weekly
Week 17 onward: 8-12 mg once weekly (maintenance)
In the combination context, many researchers find that 8 mg is sufficient for their maintenance dose rather than pushing to 12 mg, because the cagrilintide provides enough additional appetite suppression and satiety to make the extra 4 mg of retatrutide unnecessary. This lower retatrutide dose also means fewer GI side effects from the retatrutide itself, which is a meaningful quality-of-life improvement over the long term. Check our guide on how much retatrutide to take for individualized decision-making frameworks.
The retatrutide peptide dosage chart shows that GI side effects nearly doubled in Phase 2 trials when participants were assigned directly to higher doses without titration. The same principle applies with even more urgency in a combination protocol. GI symptom rates at the 12 mg dose included nausea (38-43%), diarrhea (33-35%), constipation (21-25%), and vomiting (20-21%). Adding cagrilintide, which has its own GI effects, to an aggressively-dosed retatrutide protocol without careful titration is a recipe for poor tolerance and treatment discontinuation.
For researchers transitioning from tirzepatide, the retatrutide dosage schedule after high-dose tirzepatide provides specific guidance on bridging between compounds. The retatrutide versus tirzepatide dosage chart can help translate your current tirzepatide dose into an approximate retatrutide equivalent. However, because retatrutide has the additional glucagon receptor component, starting at a lower-than-expected dose and titrating up remains the safest approach.
Use the retatrutide dosage calculator to determine your specific volume per injection based on the concentration of your reconstituted peptide.
Complete week-by-week combined protocol
The following protocol represents a conservative approach to introducing both compounds, designed to minimize GI side effects while building toward full therapeutic doses. This is not the only valid approach, but it reflects the general principles that experienced researchers and clinics follow when combining these peptides.
Phase 1: Retatrutide foundation (weeks 1-8)
During this initial phase, the goal is to establish retatrutide tolerance before adding cagrilintide.
Weeks 1-4: Retatrutide 1 mg weekly. No cagrilintide. Focus on assessing GI tolerance, injection site reactions, and baseline appetite changes. Track weight, waist circumference, and any symptoms daily.
Weeks 5-8: Retatrutide 2 mg weekly. Still no cagrilintide. The retatrutide dose increase may cause temporary GI effects. Wait until these stabilize before proceeding. The retatrutide dose chart shows expected responses at each dose level.
Phase 2: Cagrilintide introduction (weeks 9-16)
Once retatrutide at 2 mg is well-tolerated, introduce cagrilintide at the lowest dose.
Weeks 9-12: Retatrutide 4 mg weekly + Cagrilintide 0.25 mg weekly. Both injected on the same day at different sites. This is the critical transition period. Monitor closely for compounding GI effects. Use separate syringes for each compound unless using a pre-mixed blend. Our retatrutide injection guide covers proper technique.
Weeks 13-16: Retatrutide 4 mg weekly (hold steady) + Cagrilintide 0.5 mg weekly. Remember: only escalate one compound at a time. The retatrutide stays at 4 mg while cagrilintide increases.
Phase 3: Retatrutide escalation (weeks 17-24)
Now that cagrilintide is established at a stable low dose, resume retatrutide escalation.
Weeks 17-20: Retatrutide 8 mg weekly + Cagrilintide 0.5 mg weekly (hold steady). The jump from 4 mg to 8 mg retatrutide is the biggest escalation step and commonly produces the most significant GI effects. Having cagrilintide at a stable low dose during this transition is manageable for most researchers.
Weeks 21-24: Retatrutide 8 mg weekly (hold steady) + Cagrilintide 1.0 mg weekly. Increase cagrilintide while holding retatrutide. At this point, you should be experiencing significant appetite suppression from both compounds. Track food intake to ensure adequate protein consumption. Our protein shake guide for GLP-1 users provides practical nutrition strategies.
Phase 4: Maintenance optimization (weeks 25 onward)
This is where individual adjustment matters most.
Weeks 25-28: Retatrutide 8-12 mg weekly + Cagrilintide 1.7 mg weekly. Some researchers will be satisfied with 8 mg retatrutide and never need to go higher. Others may push to 12 mg. The decision depends on body weight response, tolerance, and goals.
Week 29 onward: Retatrutide 8-12 mg weekly + Cagrilintide 2.0-2.4 mg weekly. Full maintenance doses of both compounds. Monitor body composition, metabolic markers, and side effects on a regular basis.
The peptide cost calculator can help estimate the ongoing expense of maintaining this dual-peptide protocol. For researchers who have been tracking their retatrutide injection sites, adding a second compound means you need to be even more disciplined about site rotation to prevent lipodystrophy or injection site reactions. The optimal injection timing for the combination is consistent from week to week, ideally the same day and approximate time each week.
Reconstitution and administration for both peptides
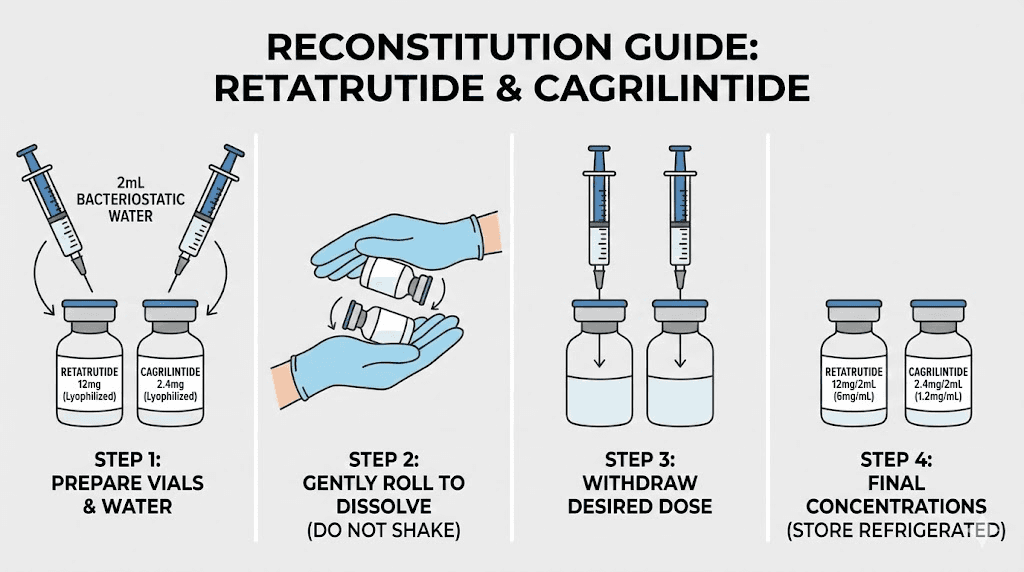
Both cagrilintide and retatrutide are typically supplied as lyophilized (freeze-dried) powders that require reconstitution with bacteriostatic water before use. Getting the reconstitution right is non-negotiable. Incorrect dilution means incorrect dosing, and incorrect dosing means either subtherapeutic effects or unnecessary side effects.
Retatrutide reconstitution
The retatrutide reconstitution chart provides specific water volumes based on vial concentration. Most research-grade retatrutide comes in 10 mg or 20 mg vials. For a 10 mg vial, adding 2 mL of bacteriostatic water creates a concentration of 5 mg/mL. Each 0.1 mL (10 units on an insulin syringe) would then deliver 0.5 mg. For detailed calculations, see our guide on how much bacteriostatic water for 10 mg retatrutide.
For researchers using the 20 mg retatrutide vial, a common approach is to add 2 mL of bacteriostatic water for a concentration of 10 mg/mL, or 4 mL for 5 mg/mL. The higher concentration means smaller injection volumes, which some researchers prefer. The lower concentration allows for more precise dosing at lower doses during the titration phase.
Cagrilintide reconstitution
Cagrilintide vials commonly come in 5 mg or 10 mg sizes. For a 5 mg vial, adding 2 mL of bacteriostatic water creates a concentration of 2.5 mg/mL. Each 0.1 mL (10 units) delivers 0.25 mg, which is the standard starting dose. This makes the lowest dose conveniently equal to the smallest easily-measured volume on an insulin syringe. The peptide reconstitution calculator handles these calculations automatically for any vial size and desired concentration.
Pre-mixed blends
Some research suppliers offer pre-mixed retatrutide/cagrilintide blends, typically in ratios like 12.5 mg retatrutide / 2.5 mg cagrilintide per vial. These blends simplify administration to a single injection but limit dosing flexibility. During the titration phase when you need to adjust each compound independently, separate vials are strongly recommended. Pre-mixed blends are best reserved for the maintenance phase once both compounds are at stable target doses.
Administration best practices
Both compounds are injected subcutaneously using insulin syringes (typically 29-31 gauge, 0.5 inch needles). Injection technique follows the same principles covered in our retatrutide injection guide and GLP-1 injection guide.
Key administration points for the combination:
Separate injection sites: Never inject both compounds at the same anatomical location. If injecting retatrutide in the right abdomen, inject cagrilintide in the left abdomen or thigh. Maintain at least 2 inches (5 cm) between injection sites.
Same-day administration: Both compounds can be injected on the same day. Many researchers prefer this for scheduling simplicity. There is no known pharmacological interaction from same-day administration.
Storage: Both reconstituted peptides should be refrigerated at 2-8 degrees Celsius and are generally stable for 4-6 weeks after reconstitution. Understanding how injectable peptides compare to oral alternatives and the difference between lyophilized and liquid peptide formats helps in making informed sourcing decisions.
Reconstitution technique: When adding bacteriostatic water to the lyophilized powder, direct the stream against the side of the vial and allow it to flow gently onto the powder. Do not shake or vortex. Gently swirl the vial until the powder is fully dissolved. If you are unfamiliar with the process, our peptide reconstitution guide walks through each step with visual instructions.
Managing side effects of the cagrilintide retatrutide stack
Side effects are the primary concern with any multi-peptide protocol, and combining cagrilintide with retatrutide creates the potential for compounding gastrointestinal effects. The good news is that these effects are manageable with proper titration and awareness. The bad news is that rushing the titration will almost certainly result in more severe symptoms than either compound would cause alone.
Gastrointestinal effects
Nausea is the most commonly reported side effect with both compounds individually. Retatrutide at the 12 mg dose produced nausea in 38-43% of Phase 3 trial participants. Cagrilintide at 2.4 mg produced nausea in approximately 55% of REDEFINE trial participants, though this was predominantly mild and transient. When combining the two, expect nausea to be most pronounced during dose escalation periods, particularly when both compounds are being increased in close succession.
Management strategies include smaller, more frequent meals rather than large ones, avoiding high-fat meals that slow gastric emptying further, staying upright for at least 30 minutes after eating, and using ginger or peppermint tea as natural anti-nausea agents. The detailed approaches in our cagrilintide side effects guide apply directly to the combination protocol as well.
Constipation is another common issue, reported in 21-25% of retatrutide users and a significant percentage of cagrilintide users. Both compounds slow gastric motility, so the effect can be additive. Our constipation treatment guide and semaglutide constipation strategies provide practical solutions that apply equally to this stack. Adequate hydration (at least 2-3 liters daily), fiber supplementation, and magnesium citrate at bedtime are the first-line approaches.
Diarrhea, vomiting, and decreased appetite round out the common GI effects. The appetite decrease is of course part of the intended mechanism, but it can become excessive during aggressive titration, leading to inadequate caloric and protein intake. Monitor protein consumption closely. Muscle preservation during rapid weight loss requires at least 1.2-1.6 grams of protein per kilogram of body weight daily.
Fatigue and energy changes
GLP-1-related fatigue is a recognized phenomenon that can occur with any peptide in this class. Adding cagrilintide to retatrutide may temporarily exacerbate fatigue during the titration phase, particularly if caloric intake drops significantly. Ensuring adequate nutrition, staying hydrated, and maintaining gentle exercise can mitigate this effect. Most researchers report that fatigue resolves within 2-3 weeks of reaching a stable dose.
Headaches
GLP-1-related headaches occur in a subset of users, typically during the first few weeks or after dose escalation. The combination protocol increases the number of dose escalation events (since you are titrating two compounds), which may mean more frequent but generally mild headaches. Standard analgesics and adequate hydration usually resolve these within days.
Hair loss considerations
Rapid weight loss from any cause can trigger telogen effluvium, a temporary form of hair shedding. With a combination protocol that may produce more rapid weight loss than monotherapy, the risk is theoretically higher. Our guides on retatrutide and hair loss and GLP-1-related hair loss cover prevention strategies including adequate protein intake, biotin supplementation, and iron monitoring.
Dysesthesia
The TRIUMPH-4 trial data revealed a notable safety signal: dysesthesia (abnormal sensations like tingling, burning, or numbness) was reported in 8.8% of participants on the 9 mg dose and 20.9% on the 12 mg dose of retatrutide. This is specific to retatrutide and not typically seen with cagrilintide. Researchers using the combination should be aware of this potential effect, particularly at higher retatrutide doses. If dysesthesia occurs, reducing the retatrutide dose is the primary intervention.
Expected results and realistic timelines
Setting realistic expectations is important for maintaining protocol adherence. The combination of cagrilintide and retatrutide is not a shortcut. It is a more comprehensive approach that may ultimately produce greater total weight loss, but the timeline still requires patience and consistency.
Weeks 1-4: Foundation period
During the initial retatrutide-only phase at 1 mg, expect mild appetite changes and possibly slight weight loss (1-3 lbs). Some researchers report no noticeable effects at this dose. That is normal. The 1 mg dose is primarily about establishing tolerance, not producing dramatic results. Our guide on how long GLP-1 takes to start working covers the typical onset timeline in detail.
Weeks 5-12: Building momentum
As retatrutide increases to 2 mg and then 4 mg, and cagrilintide is introduced at 0.25 mg, appetite suppression becomes noticeable. Weight loss typically accelerates during this period, with most researchers reporting 4-8% body weight reduction by week 12. The retatrutide onset timeline varies between individuals but most report clear effects by the 2-4 mg dose range.
Weeks 13-24: Primary weight loss phase
This is where the combination begins to show its potential. With retatrutide at 8 mg and cagrilintide building toward 1.0-1.7 mg, the four-pathway metabolic suppression is fully engaged. Weight loss during this phase is typically the most rapid, with many researchers reporting 1-3% body weight loss per month. By week 24, total weight loss from baseline commonly falls in the 12-18% range, though individual results vary significantly based on starting weight, diet, exercise, and metabolic factors.
Weeks 25 and beyond: Maintenance and optimization
At full maintenance doses, the rate of weight loss gradually slows as the body approaches a new equilibrium. This is not a plateau in the traditional sense, but rather the expected deceleration that occurs as body weight decreases and metabolic rate adjusts. Total weight loss at 48-68 weeks on the combination is difficult to predict without clinical trial data, but based on individual compound data (retatrutide at 24-28.7% and the additive benefit seen with CagriSema), a reasonable estimate for the combination is 25-35% total body weight reduction in high responders. These numbers are theoretical and should be treated as directional, not guaranteed.
For researchers who have previously used semaglutide or tracked their tirzepatide weight loss timeline, the combination protocol should produce noticeably greater results, but the exact magnitude depends on individual response and adherence.
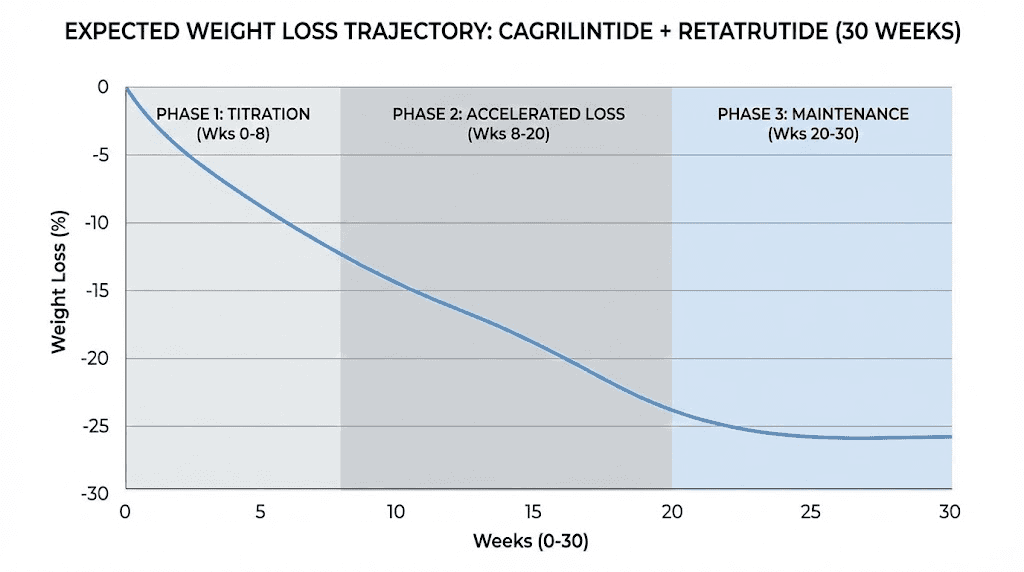
Who should consider this combination
This is an advanced protocol. It is not appropriate for everyone, and understanding who benefits most helps researchers make informed decisions about whether the added complexity and cost are justified.
Strong candidates
Researchers who have plateaued on tirzepatide or hit a wall with semaglutide and are looking for additional metabolic leverage often find the retatrutide-cagrilintide combination provides the breakthrough they need. The four-pathway approach activates mechanisms that single or dual agonists cannot reach, potentially overcoming resistance that develops with prolonged use of less comprehensive compounds.
Individuals with significant body weight to lose, typically those with a BMI that qualifies for GLP-1 therapy and who have 30% or more body weight to lose, may benefit from the more aggressive metabolic intervention this combination provides. The theoretical 25-35% total body weight reduction potential makes this stack particularly relevant for individuals with a higher starting weight who need more than what single-agent therapy can deliver.
Researchers already experienced with GLP-1 compounds who understand titration, injection technique, and side effect management are better equipped to handle the complexity of a dual-peptide protocol. This is not a beginner stack.
Who should avoid this combination
Individuals who have never used any GLP-1 agonist or metabolic peptide should not start with this combination. Begin with a single compound, learn how your body responds, and only consider combination therapy after establishing that foundation. Those who experienced severe GI side effects on tirzepatide or semaglutide that required treatment discontinuation should proceed with extreme caution, as the combination will likely produce similar or potentially more pronounced effects. Pregnant or breastfeeding individuals should avoid both compounds entirely, as detailed in our GLP-1 and breastfeeding guide.
Researchers with a history of pancreatitis, medullary thyroid carcinoma, or MEN2 syndrome should avoid both compounds due to the GLP-1 receptor activation component. These contraindications apply to all GLP-1 agonists and extend to combination protocols.
Comparing this stack to other combinations
The cagrilintide plus retatrutide combination is one of several multi-peptide approaches researchers explore for metabolic optimization. Understanding how it compares to alternatives helps in selecting the right protocol for individual goals.
Cagrilintide + retatrutide vs. CagriSema (cagrilintide + semaglutide)
CagriSema is the most directly comparable combination because it also pairs cagrilintide with a GLP-1 agonist. The key difference is that retatrutide provides triple agonism (GLP-1 + GIP + glucagon) while semaglutide provides only GLP-1 agonism. CagriSema achieved 20.4% weight loss at 68 weeks in REDEFINE 1. The theoretical advantage of cagrilintide plus retatrutide is the additional GIP and glucagon receptor activation, which should translate to greater energy expenditure and fat metabolism. Our cagrilintide and semaglutide guide provides a detailed breakdown of the CagriSema data.
Cagrilintide + retatrutide vs. cagrilintide + tirzepatide
The cagrilintide dosage with tirzepatide combination targets three pathways (GLP-1, GIP, and amylin) versus four with the retatrutide combination (GLP-1, GIP, glucagon, and amylin). The missing glucagon component means tirzepatide-based stacks lack the thermogenic and lipolytic advantages that glucagon agonism provides. However, the tirzepatide combination has the advantage of using an FDA-approved base compound with extensive safety data.
Retatrutide alone vs. retatrutide + cagrilintide
Retatrutide alone at 12 mg produced 24-28.7% weight loss in clinical trials. Adding cagrilintide theoretically provides additional satiety signaling through the amylin pathway, potentially pushing total weight loss beyond 30%. The trade-off is increased complexity, cost, two separate injections, and additional side effect management. For researchers who respond well to retatrutide alone, the incremental benefit of adding cagrilintide must be weighed against these practical considerations. The three-way comparison of semaglutide, tirzepatide, and retatrutide provides context for how single-agent retatrutide stacks up.
Other relevant comparisons
For researchers evaluating the broader landscape, our comparison guides cover retatrutide versus semaglutide, Ozempic versus retatrutide, mazdutide versus retatrutide, CagriSema versus tirzepatide, and orforglipron versus tirzepatide. Each comparison addresses different decision points depending on individual goals, budget, and available options.
Common mistakes and troubleshooting
Even experienced researchers make errors when combining peptides. Recognizing these mistakes early prevents wasted product, unnecessary side effects, and suboptimal results.
Mistake 1: Escalating both compounds simultaneously
This is the most common error and the most consequential. Increasing both retatrutide and cagrilintide in the same week makes it impossible to determine which compound is causing any side effects that develop. If nausea spikes after a dual escalation, do you reduce the retatrutide, the cagrilintide, or both? Without knowing which compound caused the problem, you end up either reducing both unnecessarily or guessing wrong and suffering through avoidable symptoms. Always escalate one compound at a time with at least a two-week stabilization period between escalations.
Mistake 2: Insufficient protein intake
The aggressive appetite suppression from four metabolic pathways can make eating feel like a chore. But inadequate protein intake during rapid weight loss leads to muscle loss, metabolic rate reduction, and worse long-term outcomes. Aim for at least 1.2-1.6 grams of protein per kilogram of body weight daily. Protein shakes designed for GLP-1 users and supplements for peptide therapy can help hit these targets even when appetite is severely suppressed.
Mistake 3: Not tracking intake and symptoms
A dual-peptide protocol requires more careful monitoring than monotherapy. Track daily caloric intake, protein consumption, water intake, body weight (weekly), waist circumference (biweekly), side effects (type, severity, duration), and energy levels. Without this data, making informed dose adjustments becomes guesswork.
Mistake 4: Ignoring food quality
When appetite is suppressed, every calorie matters more. Choosing nutrient-dense foods over empty calories ensures the body receives the micronutrients it needs during rapid weight loss. Our guides on foods to avoid on peptide therapy and best foods to eat on GLP-1 compounds provide specific dietary recommendations.
Mistake 5: Poor reconstitution practices
Using the wrong amount of bacteriostatic water, shaking the vial vigorously (which denatures the peptide), or using expired diluent all compromise the integrity of the compound. Follow the reconstitution guides precisely. Use our peptide reconstitution calculator to verify your calculations before mixing.
Troubleshooting persistent nausea
If nausea persists beyond the first 1-2 weeks at a given dose combination, consider reducing the most recently escalated compound by one dose level. Ensure you are eating slowly and in small amounts. Avoid lying down immediately after meals. If nausea is severe enough to prevent adequate nutrition, reduce both compounds to the last tolerated dose and extend the stabilization period before attempting re-escalation. This is not a failure. It is appropriate protocol management.
Troubleshooting insufficient weight loss
If weight loss stalls despite being at target doses, evaluate compliance first. Are injections being administered on schedule? Is the peptide stored correctly and within shelf life? Is dietary compliance consistent? Review our guide on weight loss stalls for systematic troubleshooting. Sometimes the answer is patience. Weight loss on peptide therapy is not linear, and temporary stalls of 1-3 weeks are normal even on optimal protocols.
Frequently asked questions
Can I inject cagrilintide and retatrutide in the same syringe?
No. Unless using a commercially prepared pre-mixed blend from a reputable source, do not mix these compounds in the same syringe. Compatibility data for combining these specific peptides in a single solution is limited, and mixing could potentially affect stability or bioactivity. Use separate syringes and separate injection sites for each compound. Follow our GLP-1 injection technique guide for proper administration.
How long should I wait between injecting retatrutide and cagrilintide?
There is no required waiting period between the two injections when given on the same day. Most researchers inject both within a few minutes of each other, simply switching to a new syringe and a different injection site. The compounds are absorbed independently from separate subcutaneous depots and do not interact at the injection site level.
What if I cannot tolerate the combination at all?
Some individuals are more GI-sensitive than others. If even the lowest combination doses (retatrutide 2 mg + cagrilintide 0.25 mg) produce intolerable side effects, the combination may not be appropriate for you. Consider running retatrutide as monotherapy at its full dose range first, as the triple agonist alone produces excellent results. Not everyone needs four-pathway coverage to achieve their goals. Our complete retatrutide dosing guide covers monotherapy protocols in detail.
Is this combination legal?
Neither retatrutide nor cagrilintide is currently FDA-approved. Retatrutide is in Phase 3 clinical trials through Eli Lilly, and cagrilintide is in Phase 3 trials through Novo Nordisk (both as monotherapy and as part of CagriSema). Both compounds are available through research supply channels. Legal status varies by jurisdiction, and researchers should understand the regulatory framework in their location before acquiring either compound. See our research versus pharmaceutical peptides comparison for context.
How does this compare to just using a higher dose of retatrutide?
Higher retatrutide doses (10-12 mg) produce more GI side effects and carry a higher risk of dysesthesia (tingling/numbness) without engaging the amylin pathway. Adding cagrilintide at a moderate dose (1.0-2.4 mg) provides amylin-mediated appetite suppression through a completely different neuronal circuit, which may allow you to use a lower retatrutide dose (8 mg instead of 12 mg) while achieving comparable or superior total metabolic effect. The guide on retatrutide dosing decisions helps evaluate this trade-off.
Can I switch from CagriSema to cagrilintide plus retatrutide?
Yes, though the transition requires careful planning. If currently on cagrilintide 2.4 mg plus semaglutide 2.4 mg, you would maintain the cagrilintide dose and replace semaglutide with retatrutide at a starting dose of 1-2 mg, then titrate up. Because retatrutide includes GLP-1 agonism (which semaglutide was providing) plus GIP and glucagon agonism, the transition should not result in loss of appetite suppression during the switch. However, starting retatrutide at a low dose is still recommended to assess tolerance of the glucagon receptor component specifically.
What about adding other peptides to this stack?
Researchers sometimes ask about adding BPC-157, AOD-9604, or other peptides. While AOD-9604 with GLP-1 agonists is an area of interest, adding more compounds to an already complex dual-peptide protocol increases variables and makes troubleshooting more difficult. The recommendation is to establish and optimize the cagrilintide-retatrutide combination first before considering any additional peptides.
How much does this combination cost per month?
Cost depends on the source, vial sizes, and maintenance doses. At target doses of retatrutide 8-12 mg weekly and cagrilintide 2.0-2.4 mg weekly, monthly costs typically range from moderate to significant depending on the supplier. Use the peptide cost calculator with your specific source pricing to get an accurate estimate. Our retatrutide cost guide and cagrilintide buying guide cover sourcing options.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, detailed stacking protocols, dosing calculators, and a community of thousands who have navigated these exact questions. SeekPeptides members access personalized protocol guidance, interaction databases, and expert-reviewed combination strategies that go beyond what any single article can cover.
External resources
New England Journal of Medicine: CagriSema REDEFINE 1 Trial Results
New England Journal of Medicine: Retatrutide Phase 2 Trial for Obesity
PMC: Efficacy and Safety of Cagrilintide Systematic Review and Meta-Analysis
PMC: Cagrilintide Lowers Bodyweight Through Brain Amylin Receptors
In case I do not see you, good afternoon, good evening, and good night. May your titrations stay gradual, your pathways stay synergistic, and your protocols stay evidence-based.