Feb 26, 2026
You are wasting peptide. Not intentionally, of course. Nobody reconstitutes a 10 mg vial of retatrutide thinking they will lose a third of it to bad math, sloppy technique, or incorrect syringe readings. But that is exactly what happens when researchers work with a retatrutide dose without understanding the specific calculations that a 10 mg vial demands. The numbers matter here. Every unit on that insulin syringe corresponds to a precise milligram quantity, and getting it wrong means either underdosing (wasting time) or overdosing (wasting comfort).
Retatrutide sits in a category of its own. Unlike semaglutide or tirzepatide, this compound activates three receptor pathways simultaneously, targeting GLP-1, GIP, and glucagon receptors in a single molecule. That triple mechanism produced the most dramatic weight reduction data in modern clinical trials, with participants losing up to 24.2% of their body weight over 48 weeks. The 10 mg vial has become the standard research quantity for individual protocols, and understanding exactly how to work with it determines whether those results translate to real outcomes or expensive frustration.
This guide covers everything. Reconstitution ratios. Unit-to-milligram conversions. Week-by-week titration schedules built around the 10 mg supply. Storage protocols that preserve potency. Side effect management strategies backed by clinical data. If you are working with a 10 mg vial, or planning to, this is the reference you will return to repeatedly. SeekPeptides built this resource because the difference between wasted peptide and optimized protocols comes down to knowing these details cold.
What retatrutide 10 mg actually means
The "10 mg" on a retatrutide vial refers to the total amount of lyophilized (freeze-dried) peptide powder inside. That is it. Ten milligrams of active compound, sitting as a white or off-white powder at the bottom of a small glass vial, waiting to be reconstituted into an injectable solution.
This is not a dose. That distinction matters enormously.
A 10 mg vial contains enough retatrutide for multiple injections spread across several weeks. Nobody injects all 10 mg at once. The actual weekly doses in clinical trials ranged from 1 mg up to 12 mg, with most retatrutide dosage protocols starting at 2 mg per week and gradually escalating. So a single 10 mg vial provides anywhere from one to five weeks of supply, depending on where you are in your dose schedule.
The lyophilized form is intentional. Peptides degrade rapidly in liquid solution at room temperature. Freeze-drying removes all water, leaving a stable powder that can survive shipping and months of shelf storage without losing potency. Once you add bacteriostatic water back to the powder, the clock starts ticking, and proper peptide storage becomes critical.
Think of the vial as a reservoir. Your job is to reconstitute it correctly, calculate your doses accurately, draw them precisely, and store the remainder properly between injections. Every step in that chain affects potency. Every mistake compounds. The sections below walk through each step in detail, starting with what makes retatrutide fundamentally different from other weight loss peptides on the market.
How the triple receptor mechanism works
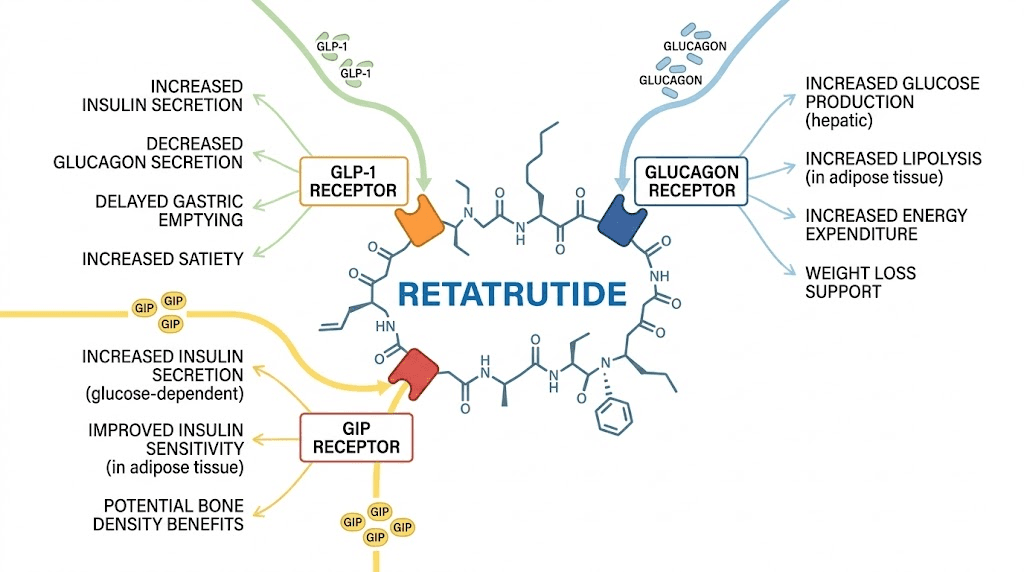
Retatrutide is not just another GLP-1 agonist. It is the first triple-hormone-receptor agonist to reach advanced clinical trials, activating three distinct metabolic pathways that individually would each justify their own drug development program. Understanding these pathways explains why the clinical data looks so different from semaglutide and tirzepatide.
GLP-1 receptor activation
The GLP-1 pathway is the one most people recognize. Semaglutide (Ozempic, Wegovy) works entirely through this single receptor. When retatrutide binds to GLP-1 receptors, it stimulates insulin release from the pancreas, reduces glucagon secretion, delays gastric emptying, and suppresses appetite through direct signaling to the brain. These effects are well-documented across dozens of clinical trials with GLP-1 medications.
The appetite suppression is powerful. People describe it as food noise disappearing. That constant background chatter about what to eat next, when to snack, whether there is something in the fridge, it quiets down. Dramatically. The onset of appetite suppression varies, but most researchers notice meaningful changes within the first two weeks of reaching therapeutic doses.
GIP receptor activation
Tirzepatide proved that adding a second receptor, GIP, produced meaningfully better outcomes than GLP-1 alone. Retatrutide takes this further. Its GIP receptor binding is actually the most potent of the three pathways, with an EC50 of 0.0643 nM compared to 0.775 nM for GLP-1. That means retatrutide activates GIP receptors at concentrations roughly twelve times lower than what it needs for GLP-1 activation.
GIP receptor activation contributes to insulin secretion, appetite modulation, and potentially beneficial effects on bone and cardiovascular health. The interplay between GIP and GLP-1 creates synergistic effects that neither pathway achieves alone, which is why tirzepatide results exceeded those of semaglutide in head-to-head comparisons.
Glucagon receptor activation
This is the pathway that makes retatrutide unique. Neither semaglutide nor tirzepatide touches the glucagon receptor. For years, researchers avoided glucagon activation because glucagon raises blood sugar, which seemed counterproductive for a metabolic medication. But the physiology is more nuanced than that simple framing suggests.
Glucagon does far more than regulate blood sugar. It reduces gastrointestinal motility, further slowing digestion. It decreases lipogenesis (fat creation) in the liver. It induces lipolysis (fat breakdown) in adipose tissue, increasing the production of free fatty acids and ketone bodies. And in animal models, glucagon activates brown fat thermogenesis, essentially turning up the body metabolic furnace to burn more calories as heat.
The combined effect of all three pathways creates a metabolic environment that attacks excess body fat from multiple angles simultaneously. Appetite goes down. Gastric emptying slows. Fat storage decreases. Fat burning increases. Energy expenditure rises. No single-receptor or even dual-receptor agonist can replicate this combination, which is why retatrutide produced weight loss numbers that surpassed both Ozempic and tirzepatide in clinical comparisons.
The compound has a half-life of approximately six days, which allows for convenient once-weekly dosing. Peak concentration occurs within 12 to 72 hours after injection. It does not interact with cytochrome P450 enzymes in the liver, which means fewer drug interaction concerns than many other medications. For researchers comparing options, our retatrutide vs tirzepatide dosage comparison breaks down the practical differences in detail.
Reconstituting a 10 mg vial step by step
Reconstitution is where most mistakes happen. Not because the process is complicated, but because small errors in technique degrade the peptide before you ever draw your first dose. The powder inside that vial is delicate. It is a chain of amino acids held together by precise chemical bonds, and rough handling breaks those bonds permanently.
Get this right and your vial delivers full potency for weeks. Get it wrong and you have expensive water.
What you need before starting
Gather everything first. Working with a partial setup leads to contamination and mistakes.
One 10 mg retatrutide vial (lyophilized powder)
One vial of bacteriostatic water (0.9% benzyl alcohol preservative)
One 3 mL mixing syringe with a longer needle (21-23 gauge)
Alcohol swabs (70% isopropyl)
A clean, flat, well-lit work surface
A vial label or marker for recording reconstitution date and concentration
Optional: U-100 insulin syringes (0.5 mL or 1 mL) for drawing doses later
Why bacteriostatic water specifically? Because the benzyl alcohol preservative prevents bacterial growth in the vial between doses. Sterile water lacks this preservative, making it suitable only for single-use reconstitution. Since you will be drawing multiple doses from your 10 mg vial over several weeks, bacteriostatic water is not optional. It is mandatory for safety. Our bac water guide for 10 mg retatrutide covers the rationale in greater depth.
Choosing your water volume
The amount of bacteriostatic water you add determines the concentration of your solution, which in turn determines how many units you draw for each dose. There is no single "correct" amount. Different volumes create different concentrations, each with trade-offs.
Here are the three most common options for a 10 mg vial:
1 mL of water = 10 mg/mL concentration. Very concentrated. Small injection volumes. Risk of drawing errors because tiny syringe movements represent large dose changes.
2 mL of water = 5 mg/mL concentration. The most popular choice. Balanced between precision and vial longevity. Most reconstitution charts default to this ratio.
3 mL of water = 3.33 mg/mL concentration. More dilute. Easier to measure small doses precisely. Uses slightly more syringe volume per injection.
For most researchers, 2 mL is the sweet spot. It creates a clean 5 mg/mL concentration where the math stays simple: every 10 units on a U-100 insulin syringe equals 0.5 mg of retatrutide. That simplicity prevents calculation errors during the weeks of titration ahead. The retatrutide dosage calculator can verify your numbers for any water volume you choose.
The reconstitution process
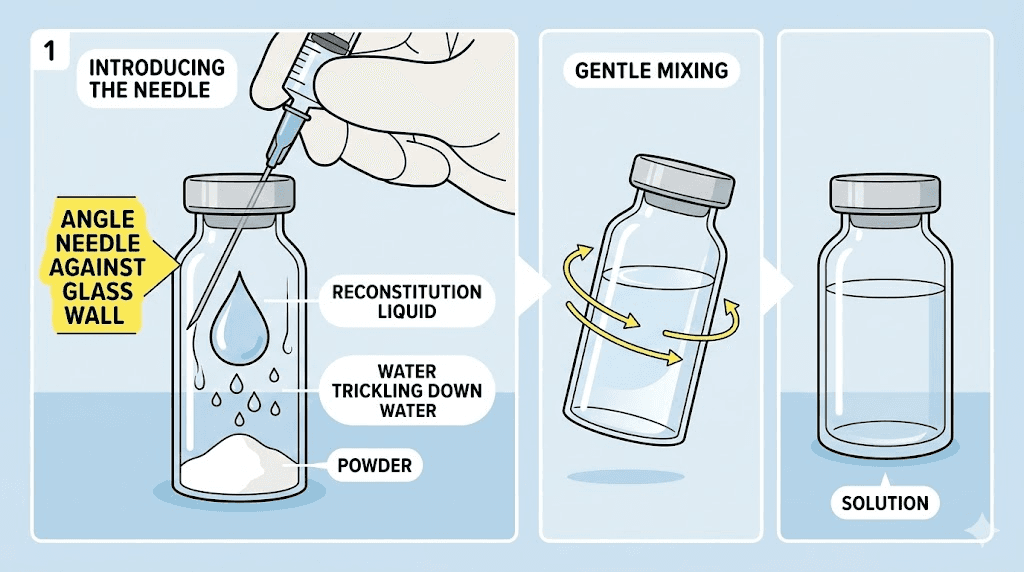
Step 1: Clean everything. Wash your hands thoroughly with soap for at least 20 seconds. Wipe the rubber stopper of both the retatrutide vial and the bacteriostatic water vial with alcohol swabs. Wait 10 to 15 seconds for the alcohol to evaporate completely before proceeding.
Step 2: Draw the water. Using your 3 mL mixing syringe, pull back the plunger to your target volume (2 mL for the recommended concentration). Insert the needle into the bacteriostatic water vial, inject the air, then invert the vial and withdraw 2 mL of water.
Step 3: Add water to the peptide vial, slowly. This step is critical. Insert the needle into the retatrutide vial at an angle so the needle tip touches the glass wall. Inject the water very slowly, letting it trickle down the inside wall of the vial. Never spray water directly onto the powder. The force can break peptide bonds and reduce potency. This technique is identical to reconstituting any peptide with bac water, but patience matters especially with expensive compounds.
Step 4: Swirl gently. Never shake. Pick up the vial and roll it slowly between your palms. Tilt it gently in a circular motion. The powder should dissolve completely within one to five minutes. If it does not dissolve after ten minutes of gentle swirling, something may be wrong with the peptide. A cloudy solution after prolonged mixing indicates potential degradation. Do not use it.
The properly reconstituted solution should be clear and colorless. No particles. No cloudiness. No foam (a few small bubbles from the mixing process are normal and will dissipate).
Step 5: Label and store. Write the date of reconstitution, the concentration (5 mg/mL if you used 2 mL), and the discard date (28 days from reconstitution as a conservative guideline) on the vial or a label. Place it immediately in the refrigerator. Proper post-reconstitution storage is essential from this point forward.
The peptide reconstitution calculator on SeekPeptides can walk you through these calculations for any vial size and water volume combination. It eliminates the guesswork entirely.
Unit calculations and syringe markings for 10 mg vials
This is where people get lost. Units, milligrams, milliliters, IU markings, they all blur together when you are staring at a tiny syringe trying to figure out how far to pull the plunger. But the math is straightforward once you understand one core formula.
Here it is.
Insulin Units = (Desired Dose in mg / Concentration in mg per mL) x 100
That is the entire calculation. Everything else is just plugging in numbers. A standard U-100 insulin syringe has 100 units per milliliter. So dividing your desired dose by the concentration gives you the fraction of a milliliter you need, and multiplying by 100 converts that to syringe units.
Quick reference for 5 mg/mL concentration (2 mL water in 10 mg vial)
If you followed the recommended 2 mL reconstitution, every calculation becomes simple:
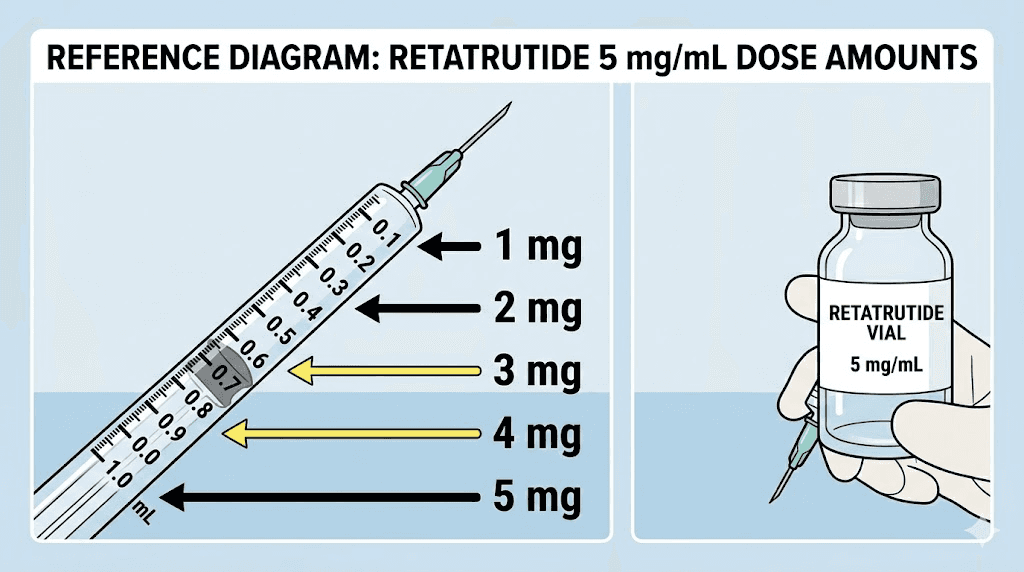
1 mg dose = 20 units (0.2 mL)
2 mg dose = 40 units (0.4 mL)
2.5 mg dose = 50 units (0.5 mL)
3 mg dose = 60 units (0.6 mL)
4 mg dose = 80 units (0.8 mL)
5 mg dose = 100 units (1.0 mL)
See the pattern? At 5 mg/mL, each milligram equals exactly 20 units. That clean ratio is why 2 mL of bac water is the most popular reconstitution volume. No decimals. No rounding errors. Just multiply your dose in milligrams by 20 to get your units. The peptide dosage calculation guide explains the underlying math for any concentration.
Quick reference for 3.33 mg/mL concentration (3 mL water in 10 mg vial)
Some researchers prefer more dilute solutions for easier measurement of smaller doses during the early titration phase:
1 mg dose = 30 units (0.3 mL)
2 mg dose = 60 units (0.6 mL)
3 mg dose = 90 units (0.9 mL)
4 mg dose = 120 units (1.2 mL, requires a larger syringe)
The trade-off is clear. More water means larger injection volumes and faster depletion of the vial, but greater precision for small doses. Less water means smaller injections and longer vial life, but higher risk of measurement errors. Choose based on where you are in your dosage protocol and which syringe size you are comfortable using.
Reading the syringe correctly
Insulin syringes come in two common sizes for peptide research. The 0.5 mL syringe has markings every single unit (1 IU increments) and tops out at 50 units. The 1 mL syringe has markings every two units and goes up to 100 units. For doses under 50 units, use the smaller syringe. Its finer gradations make accurate measurement much easier.
Always read the syringe at eye level with the plunger facing up. Look at where the flat bottom of the rubber plunger meets the unit markings, not the curved edge. This sounds trivial. It is not. A misread of even 2-3 units at a 5 mg/mL concentration changes your dose by 0.1-0.15 mg. Over weeks of daily or weekly injections, those errors compound significantly.
If math is not your strong suit, SeekPeptides offers a peptide calculator that handles all these conversions automatically. Enter your vial size, water volume, and desired dose, and it returns the exact number of syringe units to draw. No formulas required.
The complete titration schedule from a 10 mg vial
Nobody starts at the full dose. This is not optional caution. It is clinical protocol supported by hard data showing that slower dose escalation dramatically reduces side effects.
In the NEJM Phase 2 trial, participants assigned to an initial dose of 4 mg experienced significantly more adverse events than those who started at 2 mg, even when both groups ultimately reached the same target dose. Starting low is not being conservative. It is being smart.
Standard 10 mg vial titration protocol
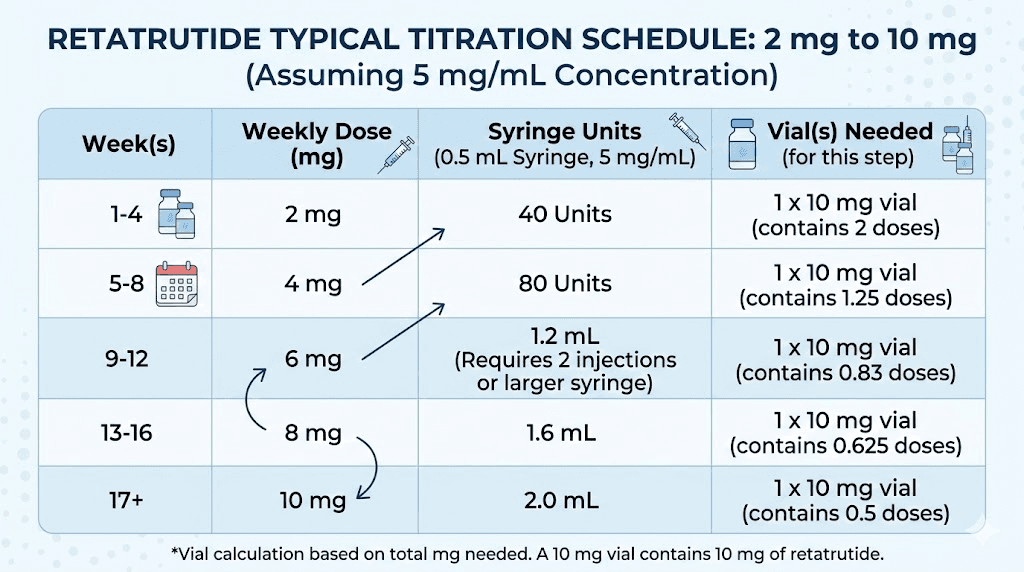
Based on clinical trial dose escalation schedules and adjusted for the 10 mg vial format, here is the most commonly referenced protocol. This assumes a 5 mg/mL concentration (2 mL bacteriostatic water).
Weeks 1-4: 2 mg per week (40 units)
This is the adaptation phase. The body is encountering triple-receptor activation for the first time. Gastrointestinal side effects peak during these early weeks as GLP-1 mediated gastric slowing takes effect. Most nausea occurs here. Most people who quit, quit here. Push through with the management strategies detailed later in this guide, and things improve markedly. At 2 mg per week, the 10 mg vial provides five full weeks of supply.
Weeks 5-8: 4 mg per week (80 units)
The first escalation doubles the dose. This is where the triple mechanism begins showing meaningful metabolic effects. Appetite suppression deepens. Weight loss accelerates. The clinical data showed the 4 mg group achieving 17.1% body weight reduction at 48 weeks, which already exceeds what most single-receptor GLP-1 agonists deliver at their maximum doses. At this dose, a 10 mg vial lasts 2.5 weeks. You will need a second vial partway through this phase.
Weeks 9-12: 8 mg per week (160 units, requires 2 draws from 1 mL syringe or a larger syringe)
The 8 mg dose is where retatrutide separates from the competition. At 48 weeks, the 8 mg group showed 22.8% mean body weight reduction, exceeding maximum tirzepatide dosing results. Note that 8 mg at 5 mg/mL concentration requires drawing 160 units (1.6 mL), which exceeds a standard 1 mL insulin syringe. You have two options: draw 80 units in two separate draws, or use a larger 3 mL syringe. Some researchers switch to a 3 mL reconstitution at this point for more manageable injection volumes. At 8 mg per week, a 10 mg vial lasts just 1.25 weeks.
Weeks 13+: 10-12 mg per week (maintenance)
The highest clinical doses. The 12 mg group achieved 24.2% weight loss at 48 weeks in Phase 2, and 28.7% at 68 weeks in the Phase 3 TRIUMPH-4 trial. At 10 mg per week (our vial size), one vial equals exactly one week. The starting dose guide provides additional context for researchers considering different escalation speeds.
How many vials you need for a full protocol
Let us do the math for a complete 24-week titration protocol:
Weeks 1-4 (2 mg/week): 8 mg total = 0.8 vials
Weeks 5-8 (4 mg/week): 16 mg total = 1.6 vials
Weeks 9-12 (8 mg/week): 32 mg total = 3.2 vials
Weeks 13-24 (10 mg/week): 120 mg total = 12 vials
Total for 24 weeks: approximately 17.6 vials (round to 18).
That is a significant investment. Understanding the retatrutide cost landscape helps with planning, and bulk purchasing options can reduce per-vial pricing substantially for longer protocols.
Adjusting the schedule based on tolerance
The schedule above is a template, not a rigid prescription. Some researchers tolerate dose increases faster. Others need more time at each tier. The guiding principle from clinical data is clear: escalate every four weeks, starting from the lowest effective dose, and never increase if side effects from the current dose have not stabilized.
If nausea, vomiting, or diarrhea remain significant at any dose, hold that dose for an additional two to four weeks before escalating. The dose selection guide outlines criteria for when to hold versus when to increase. Rushing the titration does not produce faster results. It produces worse side effects, more discontinuations, and often slower progress because severe GI symptoms prevent adequate nutrition and hydration.
How long does a 10 mg vial last
The answer depends entirely on your current dose. At the starting dose of 2 mg per week, one vial lasts five weeks. At the maintenance dose of 10 mg per week, one vial lasts exactly one week. Here is the complete breakdown.
2 mg/week: 5 weeks per vial
4 mg/week: 2.5 weeks per vial
5 mg/week: 2 weeks per vial
8 mg/week: 1.25 weeks per vial
10 mg/week: 1 week per vial
This creates a planning challenge. Early in the protocol, vials last long enough that storage becomes the limiting factor (reconstituted peptide should be used within 28 to 60 days). Later in the protocol, you burn through vials rapidly and need consistent supply. Understanding retatrutide shelf life at each stage helps prevent waste.
For the early titration phase, consider reconstituting with a smaller amount of water. If you are only using 2 mg per week, you will draw from the vial five times over five weeks. That is five needle punctures through the rubber stopper, each introducing a tiny risk of contamination. Bacteriostatic water mitigates this, but the fewer punctures, the better. Some researchers split the protocol across two vials instead, reconstituting each with 1 mL for a 10 mg/mL concentration and using one vial for the first 2.5 weeks before starting the second.
At higher doses, the math simplifies. One vial per week at 10 mg is clean and straightforward. The reconstituted peptide longevity guide provides detailed timelines for maintaining potency at different storage conditions.
Clinical trial data behind the 10 mg dose range
The most compelling argument for retatrutide comes from the numbers. Not marketing numbers. Not anecdotal reports. Published, peer-reviewed, placebo-controlled clinical trial data from some of the most rigorous obesity research ever conducted.
Phase 2 trial (NEJM, 2023)
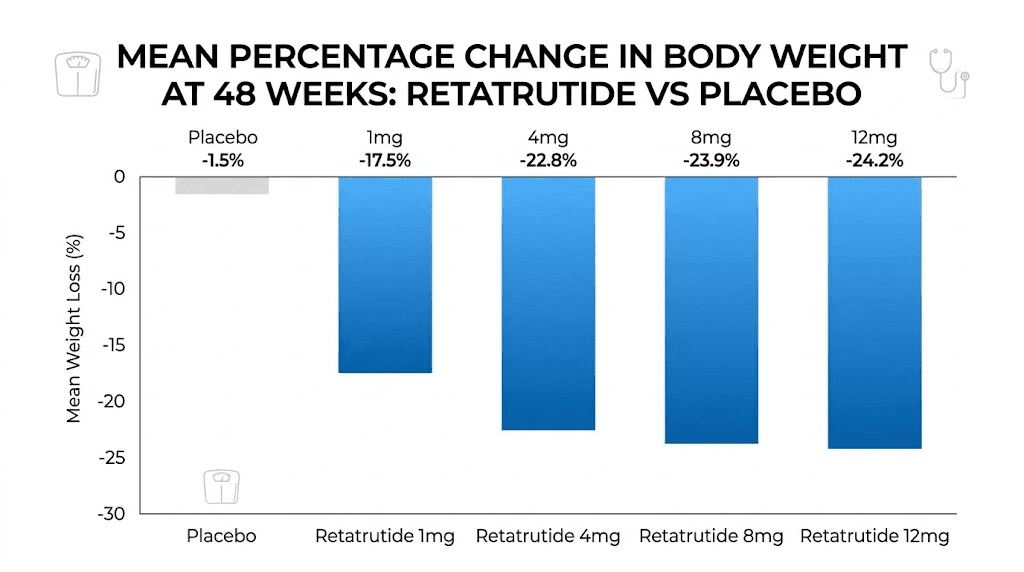
The landmark Phase 2 trial, published in the New England Journal of Medicine, randomized 338 adults with obesity to receive weekly subcutaneous retatrutide at doses of 1 mg, 4 mg, 8 mg, or 12 mg, or placebo, for 48 weeks. The dose escalation design tested different starting doses (2 mg vs 4 mg) to evaluate tolerability.
Results at 48 weeks were remarkable. The placebo group lost 2.1% of body weight. The 1 mg group lost 8.7%. The 4 mg group lost 17.1%. The 8 mg group lost 22.8%. And the 12 mg group lost 24.2%, averaging 27.2 kg (approximately 60 pounds) of weight reduction.
These numbers require context. Semaglutide 2.4 mg (Wegovy) produces approximately 15-17% weight loss in similar trials. Tirzepatide 15 mg (the highest approved dose) produces approximately 20-22%. Retatrutide at 12 mg exceeded both, and the 8 mg dose roughly matched the maximum tirzepatide result. For researchers weighing their options, the weight loss dosage chart compares these outcomes side by side.
The 10 mg dose was not studied as a specific tier in Phase 2. The tested doses jumped from 8 mg to 12 mg. However, researchers working with 10 mg vials often target 8-10 mg as their maintenance dose, falling within the range that produced 22-24% weight loss in the trial. The dose-response curve between 8 and 12 mg shows diminishing returns, meaning 10 mg captures most of the benefit while potentially reducing some of the dose-related side effects seen at 12 mg.
Phase 3 TRIUMPH trials
The Phase 3 program, called TRIUMPH, expanded to much larger populations across multiple indications. The TRIUMPH-4 trial, focused on adults with obesity and knee osteoarthritis, reported results showing 28.7% mean weight loss at 68 weeks with the 12 mg dose. That is nearly one-third of body weight, among the highest ever recorded for any anti-obesity medication.
Beyond weight loss, TRIUMPH-4 demonstrated significant improvements in osteoarthritis symptoms. Pain scores (measured by WOMAC) improved by up to 75.8%, with more than one in eight participants becoming completely pain-free by the end of the trial. These results highlight retatrutide potential beyond weight management alone.
For liver health, a Phase 2a trial in patients with metabolic dysfunction-associated steatotic liver disease (MASLD, formerly known as NAFLD) showed extraordinary results. At 48 weeks, 93% of participants on the 12 mg dose achieved normal liver fat levels (below 5%), with a mean relative reduction of 86% in hepatic fat content. These numbers suggest retatrutide could fundamentally change the treatment landscape for fatty liver disease.
The broader comparison between retatrutide and its competitors is detailed in the mazdutide vs retatrutide analysis and the survodutide comparison guide. The field is evolving rapidly, but retatrutide currently holds the weight loss efficacy record among investigational compounds.
Type 2 diabetes data
In a separate Phase 2 trial, retatrutide was tested in adults with type 2 diabetes. The 12 mg dose escalation group achieved a 2.02% reduction in HbA1c at 24 weeks, along with 16.94% body weight loss at 36 weeks. These results exceeded those of 1.5 mg dulaglutide (Trulicity) at the higher dose tiers. Researchers exploring retatrutide for both weight management and glycemic control can find additional protocol details in the timeline to results guide.
Storing your reconstituted 10 mg vial properly
Reconstituted retatrutide is a fragile solution. It is not like a bottle of medication sitting in a medicine cabinet. It is a dissolved peptide in water, held together by chemical bonds that degrade with heat, light, agitation, and time. Proper storage is not a suggestion. It is the difference between a vial that delivers consistent potency across all your doses and one that becomes progressively weaker with each passing day.
Temperature: 2-8 degrees Celsius (35.6-46.4 degrees Fahrenheit). That is standard refrigerator temperature. Not the door shelf where temperatures fluctuate every time you open the fridge. The back of the middle shelf, where temperatures remain most stable. This mirrors the storage requirements for other reconstituted peptides.
Duration: 28 to 60 days maximum after reconstitution. Conservative guidelines recommend 28 days. Some sources extend this to 60 days for vials stored properly with bacteriostatic water. The reality is that potency degrades gradually, not all at once. A vial at day 30 is not dramatically different from day 28, but a vial at day 60 has meaningfully less active peptide than one at day 14. Use the freshest solution possible for consistent results.
Never freeze reconstituted solution. Freezing creates ice crystals that can physically tear apart peptide chains. This damage is invisible, you cannot see it, but your body will notice reduced efficacy. If you need long-term storage, keep the peptide in its original lyophilized (powder) form. Unreconstituted retatrutide stored in a freezer or refrigerator can last months to years. The powder form storage guide covers this in detail.
Avoid light exposure. Keep the vial in its original box or wrap it in aluminum foil. UV and visible light accelerate peptide degradation through photooxidation. Minimize the time the vial spends out of the refrigerator during each dose drawing session. Pull it out, draw your dose with clean technique, and return it within a few minutes.
One more detail that researchers often overlook: every time you puncture the rubber stopper with a needle, you create a tiny pathway for contamination. Use a fresh needle for every draw. Never reuse needles between sessions. And always wipe the stopper with an alcohol swab before each puncture. These hygiene practices seem obvious until the fourth week when routine has dulled your attention. Stay disciplined. Your peptide storage protocols determine whether you get full value from every milligram.
Side effects and how to manage them at higher doses
Side effects are real. They are well-documented. And they are manageable. The clinical trial data gives us precise numbers for what to expect, which is far more useful than vague warnings.
Gastrointestinal effects
The most common side effects are gastrointestinal, driven primarily by the GLP-1 mediated slowing of gastric emptying. At the 12 mg dose level, here is what clinical trials reported:
Nausea: 43.2% of participants (versus 10.7% on placebo)
Diarrhea: 33.1% of participants (versus 13.4% on placebo)
Constipation: 25.0% of participants (versus 8.7% on placebo)
Vomiting: 20.9% of participants
These percentages tell an important story. Yes, GI effects are common. But they are also dose-dependent and temporary. Most participants who experienced nausea reported it during the initial weeks and dose escalation periods, with symptoms improving as the body adapted. This pattern mirrors what researchers see with semaglutide GI side effects and tirzepatide GI side effects.
The starting dose matters enormously for tolerability. Participants who began at 2 mg experienced fewer adverse events than those who started at 4 mg, even when both groups reached the same final dose. This validates the slower titration approach described in the protocol section above.
Practical nausea management
Eat smaller, more frequent meals instead of three large ones. This is the single most effective dietary adjustment. When GLP-1 agonists slow gastric emptying, large meals sit in the stomach longer, creating that heavy, nauseated feeling. Five smaller meals spread throughout the day prevent the stomach from ever becoming uncomfortably full.
Avoid fatty, greasy, and spicy foods during the initial titration weeks. These foods take longer to digest under normal circumstances. Combined with peptide-induced gastric slowing, they can trigger pronounced nausea. Bland foods like rice, toast, bananas, and lean proteins digest more easily and cause less distress.
Stay hydrated. Sip water throughout the day rather than drinking large amounts at once. Dehydration worsens nausea, and the vomiting and diarrhea that some researchers experience can create a dehydration cycle that amplifies symptoms. Electrolyte solutions help replace what is lost. For comprehensive nutrition strategies, the GLP-1 diet plan and best foods to eat on GLP-1 agonists guides apply equally to retatrutide.
Timing your injection matters. Some researchers find that injecting in the evening before sleep allows them to sleep through the worst of any initial nausea. Others prefer morning injections so they can manage symptoms during waking hours. Experiment to find what works for you. The best time to take your GLP-1 shot guide explores this in detail.
Constipation and diarrhea management
These two side effects can alternate, which confuses people. You might experience diarrhea during the first week of a new dose level, followed by constipation as your GI tract adjusts to the slower motility. Both are manageable.
For constipation: increase fiber intake gradually, stay hydrated, and maintain physical activity. Gentle movement helps keep the bowels moving even when gastric emptying is slowed. If dietary measures are not sufficient, over-the-counter osmotic laxatives can provide relief without interfering with the peptide mechanism.
For diarrhea: the BRAT diet (bananas, rice, applesauce, toast) provides easily digestible nutrition while managing symptoms. Electrolyte replacement is essential because diarrhea depletes sodium, potassium, and other minerals rapidly. If diarrhea persists beyond a few days at any dose level, it may indicate that the dose escalation happened too quickly. The GI side effect timeline provides benchmarks for normal duration versus concerning persistence.
Other reported effects
Beyond GI symptoms, clinical trials noted several other effects worth monitoring:
Increased heart rate: A small, dose-dependent increase in resting heart rate was observed, consistent with what occurs with other GLP-1 agonists. This is typically modest (3-5 beats per minute) and does not require intervention unless accompanied by symptoms like palpitations or chest discomfort.
Fatigue: Some participants reported fatigue, particularly during the early titration period. This often correlates with reduced caloric intake as appetite suppression takes effect. Ensuring adequate nutrition, even when appetite is reduced, helps mitigate this. The GLP-1 fatigue management guide covers strategies in depth.
Headaches: Reported at rates slightly above placebo, headaches can often be attributed to dehydration or rapid dietary changes rather than direct pharmacological effects. Staying hydrated resolves most cases. The GLP-1 headache guide provides targeted solutions.
Hair thinning: Rapid weight loss from any cause can trigger telogen effluvium, a temporary increase in hair shedding. This is not specific to retatrutide but occurs with any significant caloric deficit. It typically resolves 3-6 months after weight stabilizes. The retatrutide and hair loss article explores whether the compound itself contributes beyond simple caloric restriction, and the broader GLP-1 hair loss guide covers prevention strategies.
Skin hyperesthesia: An unusual side effect reported in some trial participants, this involves increased skin sensitivity or unusual sensations. It is uncommon and typically mild, resolving without intervention.
The discontinuation rate in clinical trials ranged between 20% and 50% within the first year, with GI intolerance being the primary reason. Slower titration and proactive side effect management significantly improve retention rates. Do not push through severe symptoms. Hold your dose. Manage the side effects. Escalate only when comfortable.
Injection technique for retatrutide
Proper injection technique ensures consistent absorption and minimizes injection site reactions. Retatrutide is administered subcutaneously, meaning the needle goes into the fat layer beneath the skin, not into muscle. This is the same approach used for all GLP-1 injections.
Approved injection sites
Three areas are standard for subcutaneous peptide injections:
Abdomen: The most common site. Inject at least two inches from the belly button, rotating positions across the entire abdominal area. Avoid the belt line where clothing friction can irritate the site.
Thigh: The front and outer areas of the upper thigh provide ample subcutaneous tissue. Avoid the inner thigh where skin is thinner.
Upper arm: The back of the upper arm, in the fatty area between the shoulder and elbow. This site is harder to self-inject and often requires assistance.
Rotate between sites weekly. Never inject in the same spot twice in a row. Repeated injections in one location can cause lipohypertrophy (thickened fatty tissue) or lipodystrophy, which affects peptide absorption and creates visible bumps. Maintaining a rotation pattern, such as left abdomen one week, right abdomen the next, then left thigh, right thigh, keeps each site fresh. The retatrutide injection site guide includes visual diagrams showing optimal rotation patterns.
The injection process
Clean the injection site with an alcohol swab and let it dry completely. Pull back on the insulin syringe plunger to your calculated dose. Hold the syringe like a pencil or dart at a 45 to 90 degree angle. With your non-dominant hand, gently pinch a fold of skin at the chosen site. Insert the needle smoothly in one motion. Inject the solution slowly and steadily. Wait five seconds before withdrawing the needle to allow the solution to disperse. Apply gentle pressure with a clean cotton ball or gauze. Do not rub.
For detailed visual instructions, the complete retatrutide injection guide walks through each step with troubleshooting tips. If you are new to subcutaneous injections entirely, the beginner peptide injection guide covers fundamentals like needle selection, air bubble removal, and anxiety management.
Comparing retatrutide 10 mg to other options
The peptide landscape for weight management has expanded dramatically. Understanding where retatrutide fits helps researchers make informed decisions about their protocols.
Retatrutide vs semaglutide
Semaglutide works through a single receptor (GLP-1). Retatrutide works through three. This fundamental difference drives the efficacy gap. At maximum doses, semaglutide produces approximately 15-17% body weight reduction. Retatrutide produces 22-24% at comparable timeframes. That is roughly 40-50% more weight loss from the triple receptor approach.
Side effect profiles are similar in type (GI effects dominate both) but different in severity. Retatrutide GI side effects tend to be more pronounced at equivalent efficacy levels, likely due to the additional glucagon-mediated GI motility effects layered on top of the GLP-1 gastric slowing. The detailed retatrutide vs semaglutide comparison breaks down every dimension of this matchup.
Availability is another factor. Semaglutide is FDA-approved and widely available as branded medications. Retatrutide remains investigational, available only through research channels or clinical trials. This affects sourcing, cost, and quality assurance considerations.
Retatrutide vs tirzepatide
Tirzepatide is the closest competitor. It activates two of the same three receptors (GLP-1 and GIP), missing only the glucagon pathway. In clinical comparisons, tirzepatide maximum dose (15 mg) produces approximately 20-22% weight loss. Retatrutide 8 mg roughly matches this, and the 12 mg dose exceeds it.
The glucagon component appears to be the differentiator. Animal studies showed retatrutide producing lower expression levels of inflammatory and fibrotic mediators compared to tirzepatide, suggesting the triple mechanism may offer benefits beyond additional weight loss. The retatrutide vs tirzepatide analysis evaluates whether the additional efficacy justifies the investigational status and potentially higher cost.
For researchers currently on tirzepatide considering a switch, the retatrutide dosing after tirzepatide guide covers the transition protocol, and the retatrutide onset after tirzepatide article addresses what to expect during the switch.
Where other compounds fit
The broader competitive landscape includes several emerging triple agonists and next-generation compounds. Mazdutide is another GLP-1/glucagon dual agonist showing promising results. Cagrilintide works through the amylin pathway, offering a different mechanism entirely. Orforglipron represents the oral GLP-1 frontier, potentially eliminating the need for injections altogether.
For a comprehensive overview of all available options, the best peptide for weight loss guide ranks current and investigational compounds across efficacy, side effects, cost, and convenience dimensions. The three-way comparison of semaglutide, tirzepatide, and retatrutide provides the most detailed head-to-head analysis available.
Sourcing considerations for 10 mg vials
Retatrutide is not FDA-approved. It does not come from a pharmacy with a prescription. This reality creates both challenges and risks that researchers must navigate carefully.
Quality varies dramatically between suppliers. The peptide research market includes reputable manufacturers who produce pharmaceutical-grade compounds alongside low-quality operators who sell degraded or mislabeled products. A 10 mg vial that actually contains 7 mg of active peptide (with the rest being filler or degraded material) will produce inconsistent results regardless of how perfectly you execute your reconstitution and dosing protocol.
Third-party testing is the minimum standard. Reputable suppliers provide certificates of analysis (COA) showing purity, identity, and potency for each batch. Look for HPLC purity above 98% and mass spectrometry confirmation of the correct molecular weight. The best place to buy retatrutide guide evaluates suppliers based on these quality metrics, and the complete purchasing guide covers the due diligence process in detail.
Pricing for 10 mg vials varies substantially. Domestic suppliers typically charge more but offer faster shipping and easier returns. International suppliers, particularly from established peptide manufacturing regions, can offer significant cost savings with trade-offs in shipping time and customs risk. The retatrutide pricing guide tracks current market rates across different supplier tiers. For larger orders, the online purchasing guide and bulk sourcing analysis cover volume discount structures and quality verification for international orders.
Storage during shipping is another quality factor. Lyophilized peptide tolerates a wider temperature range than reconstituted solution, but extreme heat during summer shipping can still cause degradation. Reputable suppliers use insulated packaging and cold packs for warm-weather shipments. When your vial arrives, inspect the powder. It should be a compact cake or loose powder at the bottom of the vial. If it looks wet, discolored, or has moved to the sides of the vial (indicating exposure to moisture or excessive heat), contact the supplier before reconstituting.
Nutrition and lifestyle optimization during retatrutide protocols
The peptide creates the metabolic environment. Your nutrition and lifestyle choices determine how effectively that environment translates to results. Retatrutide does not replace the need for adequate nutrition. It dramatically reduces appetite, which means you must be intentional about what you eat when you do eat.
Protein is non-negotiable
GLP-1 agonists cause weight loss through multiple mechanisms, but not all weight loss is equal. Losing muscle along with fat produces a worse metabolic outcome long-term. The only reliable way to preserve muscle mass during rapid weight loss is adequate protein intake combined with resistance training.
Target 0.7 to 1.0 grams of protein per pound of body weight daily. Yes, even with reduced appetite. This often requires conscious effort and protein-first meal planning. Lean meats, fish, eggs, Greek yogurt, and protein supplements become essential dietary anchors. The best foods to eat on GLP-1 agonists guide prioritizes protein-rich options, and the foods to avoid guide explains which choices undermine your protocol.
Hydration requires attention
Reduced appetite often means reduced fluid intake. People forget to drink when they are not eating. Add in the potential for GI side effects that deplete fluids, and dehydration becomes a real risk during retatrutide protocols.
Aim for a minimum of 64 ounces of water daily, more if you are experiencing diarrhea or vomiting during dose adjustments. Set reminders if needed. Carry a water bottle. Make hydration a conscious practice rather than relying on thirst signals, which may be suppressed along with appetite.
Supplements to consider
Rapid weight loss and reduced food intake can create nutritional gaps. A quality multivitamin covers the basics. Beyond that, the supplements guide for GLP-1 users recommends considering:
Electrolytes: Especially during the first few weeks when GI side effects are most prevalent
B12: Often combined with GLP-1 agonists in compounded formulations, as semaglutide with B12 and tirzepatide with B12 combinations demonstrate
Fiber: To support digestive regularity, which can be disrupted by altered gastric motility
Biotin: Particularly if hair thinning occurs, as a preventive measure alongside adequate protein
Exercise during retatrutide use
Resistance training is more important than cardio during a GLP-1 protocol. The primary risk of rapid weight loss is muscle catabolism. Strength training sends a signal to the body that muscle tissue is actively needed, redirecting catabolism toward fat stores instead. Three to four resistance sessions per week covering major muscle groups provides adequate stimulus.
Cardio has its place, but it should complement resistance training, not replace it. Walking 30 to 60 minutes daily provides cardiovascular benefits and supports general metabolism without the recovery demands of intense exercise. During the early titration phase when GI symptoms may limit exercise tolerance, walking is often the only practical option. That is perfectly fine. Consistency matters more than intensity. You can explore whether weight loss without exercise is viable, but combining peptide protocols with physical activity consistently produces superior body composition outcomes.
Frequently asked questions
How many doses can I get from one 10 mg retatrutide vial?
It depends on your current weekly dose. At 2 mg per week (the starting dose), one vial provides five doses across five weeks. At 5 mg per week, you get two doses. At 10 mg per week, one vial equals one dose. The dose chart maps every common dose to the number of vials needed per month.
Can I split a 10 mg vial across multiple smaller doses?
Yes, and that is exactly how most protocols work. You reconstitute the entire vial once, then draw individual doses from the same vial over multiple weeks during the early titration phase. The bacteriostatic water preservative keeps the solution safe for multi-dose use when stored properly between 2-8 degrees Celsius. For reconstitution specifics, see the reconstitution chart.
Is 10 mg a safe weekly dose of retatrutide?
Clinical trials tested weekly doses up to 12 mg for 48 weeks with manageable side effect profiles. The 10 mg dose falls within the studied range. However, no dose should be the starting dose. The titration protocol (starting at 2 mg and increasing every four weeks) is essential for tolerability. Jumping directly to 10 mg would likely cause severe gastrointestinal side effects and is not recommended under any circumstances. Review the dosing selection guide for personalized recommendations.
What happens if my reconstituted vial gets warm during a power outage?
Brief temperature excursions (a few hours at room temperature) typically cause minimal potency loss. Extended exposure (12+ hours above 8 degrees Celsius) degrades the peptide progressively. If you know the vial was at room temperature for more than a few hours, the safest approach is to start fresh with a new vial. The financial loss is real but less costly than running an entire protocol on degraded compound. Our room temperature peptide stability guide provides specific degradation timelines.
Can I use a 10 mg vial if I am switching from tirzepatide?
Yes, many researchers transition from tirzepatide to retatrutide. The transition protocol depends on your current tirzepatide dose. Researchers on high-dose tirzepatide (10-15 mg) typically begin retatrutide at 4-8 mg rather than the standard 2 mg starting dose, since their GI tract is already adapted to incretin receptor activation. The transition protocol from tirzepatide provides specific guidance for this switch.
How do I know if my 10 mg vial is still potent?
Visual inspection is your first check. The reconstituted solution should remain clear and colorless. Any cloudiness, particulate matter, or discoloration suggests degradation or contamination. Beyond visual checks, the most reliable indicator is efficacy, if appetite suppression diminishes or weight loss stalls unexpectedly mid-protocol, a degraded vial is a possible cause. Always check your retatrutide shelf life against your reconstitution date.
Is a 10 mg vial better than a 20 mg vial?
Neither is inherently better. A 10 mg vial is ideal for the titration phase when weekly doses are small (2-4 mg), because the vial gets used within its optimal potency window. A 20 mg vial becomes more practical at higher maintenance doses (8-12 mg/week) where a 10 mg vial would be consumed in one to two sessions. Many researchers start with 10 mg vials and switch to larger vials once they reach their target maintenance dose.
Do I need a prescription for retatrutide?
Retatrutide is not FDA-approved and does not have a standard prescription pathway. It is available through research peptide suppliers for investigational use. The availability guide covers the current regulatory landscape and sourcing options in detail.
External resources
NEJM Phase 2 Trial: Triple Hormone Receptor Agonist Retatrutide for Obesity
PMC Review: Retatrutide - A Game Changer in Obesity Pharmacotherapy
ClinicalTrials.gov: Retatrutide TRIUMPH Clinical Trial Registry
Nature Medicine: Retatrutide for Metabolic Dysfunction-Associated Steatotic Liver Disease
For researchers serious about optimizing their retatrutide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, personalized protocol builders, dosing calculators, and a community of thousands who have navigated these exact questions. SeekPeptides members access detailed retatrutide protocols, weight-based dosing tools, and expert-reviewed titration schedules that account for individual factors most resources ignore.
In case I do not see you, good afternoon, good evening, and good night. May your reconstitutions stay precise, your titrations stay smooth, and your protocols stay effective.