Mar 23, 2026

Before you order another vial from a website that promises 99% purity and ships without asking a single question, read this. The numbers do not lie. Independent laboratory testing of semaglutide products sold as research chemicals found actual purity levels between 7% and 14%. Not 99%. Not even close. And the problems go far deeper than a misleading label on a website.
The research chemical semaglutide market has exploded. Dozens of vendors sell lyophilized powder in vials marked "for research purposes only," complete with reconstitution instructions and dosing guides that make the intended use obvious. The FDA knows it. Researchers know it. And the people buying these products absolutely know it. But what most buyers do not know is exactly what they are getting, how it differs from pharmaceutical-grade semaglutide, and why the quality gap matters more than most people realize.
This guide breaks down everything the research reveals about research chemical semaglutide, from laboratory purity analysis and salt form complications to FDA enforcement actions and practical evaluation strategies. Whether you are trying to understand what separates a research chemical from a compounded semaglutide product, or you simply want to know what the risks actually look like, the data is here. All of it.
SeekPeptides exists to cut through exactly this kind of confusion, providing evidence-based guidance that helps researchers make informed decisions rather than gambling on products they do not fully understand.
What research chemical semaglutide actually is
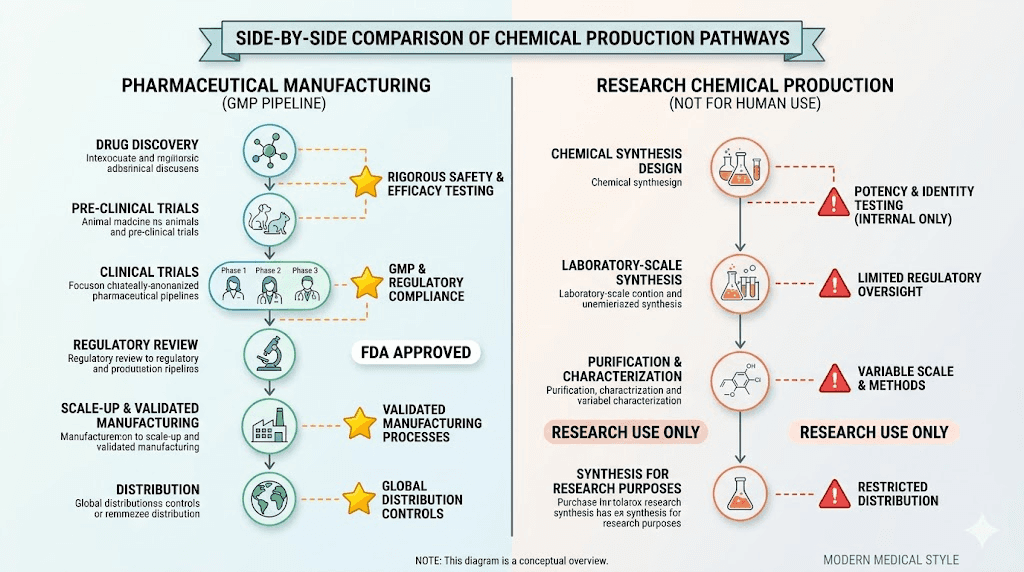
Semaglutide is a GLP-1 receptor agonist. A 31-amino-acid peptide analog of human glucagon-like peptide-1 with a C-18 fatty acid chain attached via a linker. That fatty acid chain is what gives it an extended half-life of roughly 7 days, making weekly dosing possible. The pharmaceutical versions, Ozempic and Wegovy, are manufactured by Novo Nordisk under strict FDA-regulated conditions using engineered yeast cells to produce the peptide backbone, followed by precise chemical modifications and multi-step purification.
Research chemical semaglutide is something else entirely.
These products are typically synthesized using solid-phase peptide synthesis (SPPS), a chemical manufacturing process that builds the peptide chain amino acid by amino acid. The end product arrives as a lyophilized (freeze-dried) powder in a vial, usually in 5mg or 10mg quantities. Vendors label these products "for research use only" or "not for human consumption," a legal disclaimer designed to circumvent pharmaceutical regulations.
The distinction matters. Pharmaceutical manufacturing involves current Good Manufacturing Practice (cGMP) standards, extensive quality control testing, validated processes, and regulatory oversight. Research chemical production has none of these requirements. The manufacturer decides their own quality standards, if they have any at all. Understanding these differences is fundamental before considering any semaglutide purchase decision.
Here is what that means practically. When you buy a vial of research chemical semaglutide, you are trusting that the manufacturer correctly synthesized a complex 31-amino-acid peptide, properly attached the C-18 fatty acid modification, purified the product to an acceptable standard, accurately measured the quantity, maintained sterile conditions during lyophilization, and stored the product correctly throughout the supply chain. Each of those steps represents a potential failure point.
The "research purposes only" label
This phrase appears on virtually every research chemical vendor website. It serves a specific legal purpose, creating a paper trail suggesting the product is not intended for human use. But the reality is transparent. A study published in the Journal of Medical Internet Research examined 38 websites selling GLP-1 drugs labeled as research chemicals. Only four asked buyers to check a box confirming they were researchers. None of the 38 verified that buyers were actual researchers.
Meanwhile, these same websites provide semaglutide dosing information, reconstitution instructions, injection guides, and dosage calculators. The disconnect between the "not for human use" disclaimer and the clearly human-focused content tells you everything about the intended market.
The FDA has taken notice. In September 2025, the agency issued warning letters to more than 50 companies involved in the GLP-1 market, specifically targeting claims that were false or misleading. Products marketed as "for research purposes" while clearly intended for human injection fall squarely into this enforcement category.
Common forms available
Research chemical semaglutide typically comes in several forms. Lyophilized powder is the most common, requiring reconstitution with bacteriostatic water before use. You will also find pre-mixed solutions, though these raise additional stability concerns. Some vendors now offer sublingual formulations and oral drops, which add another layer of complexity to an already complicated quality picture.
The lyophilized powder format is generally considered more stable than pre-mixed solutions, which is why most research-focused vendors prefer it. But lyophilization quality varies wildly between manufacturers. Poor freeze-drying can damage the peptide structure, reducing potency even when the starting material was acceptable. Understanding proper peptide storage in powder form becomes critical when working with these products.
How research-grade differs from pharmaceutical-grade semaglutide
The differences are not subtle. They span every aspect of production, testing, and quality assurance.
Manufacturing environment. Pharmaceutical semaglutide is produced in cGMP-certified facilities with environmental controls, validated equipment, trained personnel, and documented procedures. Research chemicals may be produced in any laboratory setting, from sophisticated chemical facilities to minimally equipped operations. There are no regulatory inspections. No required certifications. No oversight.
Understanding the broader context of research versus pharmaceutical peptides helps frame why these distinctions exist and why they matter for every researcher evaluating their options.
Synthesis method. Novo Nordisk uses recombinant DNA technology with engineered Saccharomyces cerevisiae (yeast) to produce the peptide backbone, followed by chemical modification to attach the fatty acid sidechain. This biological production method tends to produce fewer impurities than fully synthetic approaches. Research chemical manufacturers typically use solid-phase peptide synthesis, which can introduce truncated sequences, deletion peptides, racemized amino acids, and residual coupling reagents as impurities.
Purification standards. Pharmaceutical-grade purification involves multiple chromatography steps with validated acceptance criteria. Research chemical purification might involve a single HPLC step, or in some cases, even less rigorous methods. The difference shows in the impurity profiles.
Testing requirements. Pharmaceutical semaglutide undergoes identity testing, potency assays, purity analysis (HPLC), residual solvent testing, endotoxin testing (LAL), sterility testing, particulate matter testing, and stability testing under controlled conditions. Research chemicals might include a certificate of analysis showing HPLC purity and mass spectrometry confirmation. Might.
When evaluating any peptide product, understanding the fundamentals of peptide quality assessment gives you a framework for making sense of what certificates of analysis actually tell you, and what they leave out.
The purity problem in real numbers
A landmark study published in the Journal of Medical Internet Research conducted LC-MS (liquid chromatography-mass spectrometry) analysis on semaglutide products purchased from online research chemical vendors. The findings were stark.
Claimed purity on labels: 99%.
Actual measured purity: 7.7% to 14.37%.
That is not a minor discrepancy. That is a product containing 85% to 93% of something that is not semaglutide. The study also found that the measured semaglutide content exceeded the labeled amount by 28% to 39% in each sample. So the products contained more total semaglutide than stated, but at dramatically lower purity. This combination means researchers are getting more active ingredient per vial than expected, but mixed with a substantial volume of unknown impurities.
For context, if you are following a semaglutide 5mg/mL dosage chart but your product is actually 7% pure, the math breaks down completely. Your calculations assume a known concentration. Unknown purity makes every dosing decision unreliable.
Endotoxin contamination
The same study detected endotoxins in all tested samples, with levels ranging from 2.16 EU/mg to 8.95 EU/mg. Endotoxins are bacterial cell wall components that can cause fever, inflammation, and in severe cases, septic shock when injected. Pharmaceutical products have strict endotoxin limits, typically below 5 EU/kg of body weight for injectable products.
This finding alone should give any researcher pause. Endotoxin contamination indicates either inadequate manufacturing conditions, poor raw material quality, or insufficient purification. It is a red flag that extends beyond purity concerns into direct safety territory. If you are researching how to properly administer semaglutide injections, endotoxin contamination adds a risk factor that no technique can mitigate.

The salt form problem that nobody talks about
Here is where things get technically complicated, and critically important.
FDA-approved semaglutide (Ozempic, Wegovy) uses semaglutide base, the unmodified active ingredient. Research chemical vendors and some compounding pharmacies sell semaglutide sodium or semaglutide acetate. These are different chemical entities.
Semaglutide sodium is formed by reacting semaglutide base with sodium hydroxide. Semaglutide acetate results from processing with acetic acid. While the core peptide structure remains the same, the salt form affects solubility, stability, pH behavior, and potentially bioavailability.
The FDA has been explicit about this distinction. The agency stated that semaglutide sodium and semaglutide acetate are "different active ingredients than are used in the approved drugs." They are not considered interchangeable with semaglutide base, and there is no evidence demonstrating that salt forms are safe or effective.
Why vendors use salt forms
The reason is practical. Semaglutide base has limited solubility in aqueous solutions at certain pH ranges. Salt forms dissolve more readily, making them easier to work with during manufacturing. Semaglutide sodium dissolves well at higher pH levels, while semaglutide acetate works better in slightly acidic conditions. For manufacturers optimizing for ease of production rather than regulatory compliance, salt forms simplify the process.
But easier manufacturing does not equal equivalent products. The pharmacokinetic implications of salt form changes are not trivial. Absorption rates can differ. Stability profiles change. And without clinical data comparing salt forms to the base compound, nobody can say with certainty how these differences translate to real-world outcomes.
If you are comparing semaglutide products from different sources, understanding the difference between lyophilized and liquid peptide formulations provides additional context for evaluating what you are actually purchasing.
How to identify salt forms
Not every vendor discloses the salt form. Some simply label their product "semaglutide" without specifying whether it is base, sodium, or acetate. When evaluating a certificate of analysis, look for the molecular weight. Semaglutide base has a molecular weight of approximately 4,113.58 Da. Semaglutide sodium adds sodium ions (approximately 22.99 Da each, potentially multiple depending on the number of acidic groups neutralized). If the COA lists a molecular weight that does not match semaglutide base, you are likely looking at a salt form.
The mass spectrometry data on the COA should also clarify this. If it shows adduct ions consistent with sodium attachment, that confirms the salt form. This is one of many reasons why understanding how to read a COA matters for anyone evaluating peptide products and their true cost.
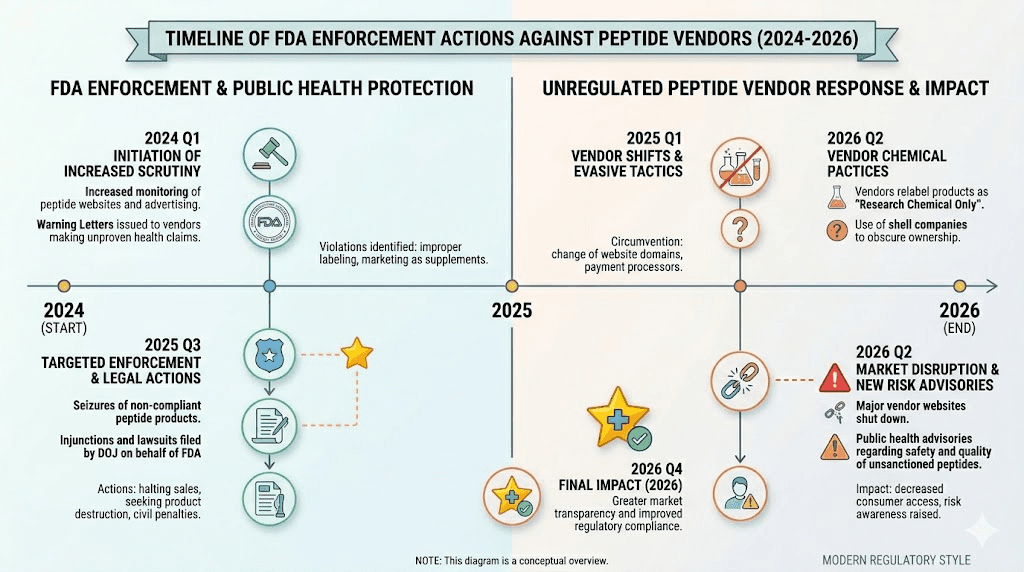
FDA enforcement actions and warning letters
The regulatory landscape around research chemical peptides has shifted dramatically.
In December 2024, the FDA issued a warning letter specifically to Summit Research Peptides for selling unapproved drugs, including semaglutide products marketed as research chemicals. The letter cited products that were "falsely labeled for research purposes or not for human consumption" while being sold directly to consumers with dosing instructions for injection.
Then in September 2025, the FDA escalated significantly, sending more than 50 warning letters to companies involved in the GLP-1 market. These letters targeted false or misleading claims, including assertions that compounded products were "generic versions" or contained the "same active ingredient" as FDA-approved drugs.
The enforcement pattern is clear. The FDA has moved from occasional actions to systematic targeting of the research chemical and compounding markets. For researchers who have been purchasing semaglutide through these channels, this regulatory pressure is likely to reshape availability and pricing in the near term.
The compounding pharmacy distinction
A critical regulatory nuance: compounded semaglutide from a licensed 503A or 503B compounding pharmacy is legally and practically different from research chemical semaglutide. Compounding pharmacies operate under FDA and state board of pharmacy oversight, require prescriptions, and must meet specific quality standards.
Research chemical vendors operate in a regulatory gray area. They are not pharmacies. They do not require prescriptions. And they are not subject to pharmacy board inspections. The "research use only" label is their primary legal shield, and as recent enforcement actions show, that shield is thinning.
In February 2025, the FDA announced that the nationwide shortage of semaglutide injections was resolved. This was significant because drug shortages had previously allowed compounding pharmacies to produce copies of FDA-approved drugs under certain conditions. With the shortage resolved, the legal basis for compounding semaglutide narrowed considerably. For the grey market in GLP-1 products, this represents a fundamental shift.
Red flags when evaluating research chemical vendors
Not all research chemical vendors are equal. Some operate with genuine quality standards. Others are essentially repackaging bulk powder from overseas suppliers with minimal or no quality testing. Learning to distinguish between them requires knowing what to look for, and what should send you running.
Immediate red flags
No certificate of analysis available. Any vendor that does not provide a COA for each batch is not worth considering. Period. The COA should include HPLC purity data, mass spectrometry confirmation, and ideally endotoxin testing results. If they cannot produce this documentation, assume the worst about their product quality.
COA from the manufacturer, not a third party. A COA generated by the company selling you the product has obvious conflict-of-interest problems. Independent third-party testing provides far more reliable data. Look for COAs from recognized analytical laboratories, not in-house results printed on the vendor letterhead.
Prices significantly below market. Semaglutide synthesis is complex and expensive. If a vendor offers dramatically lower prices than competitors, they are likely cutting corners somewhere. Maybe the purity is lower. Maybe the quantity is less than labeled. Maybe the product is a different compound entirely. When evaluating cheaper semaglutide options, the price-quality relationship matters enormously.
No batch numbers or lot tracking. Legitimate quality-focused vendors assign batch numbers to each production run, allowing traceability. If the vial you receive has no batch number, or if the batch number on the vial does not match the COA, something is wrong.
Dosing instructions on the website. This one is counterintuitive, but hear the logic. A vendor that provides detailed human dosing instructions while labeling their product "for research use only" is telling you their legal disclaimer is performative. While this does not necessarily mean the product quality is poor, it does mean the vendor is comfortable with contradictory claims, which should make you question other statements they make.
Positive indicators
Third-party COAs from recognized labs. Batch-specific testing rather than a single COA applied to all products. Transparent information about their synthesis methods and suppliers. Active quality programs with documented rejection rates. Customer service that can answer technical questions about their products.
Vendors who invest in quality typically charge more. That price premium reflects actual analytical testing costs, better raw materials, and more rigorous manufacturing processes. When comparing vendors, understanding the broader cost structure of peptide products helps calibrate expectations.
Why people turn to research chemicals in the first place
Understanding the demand side helps explain why this market exists and continues to grow despite well-documented quality concerns.
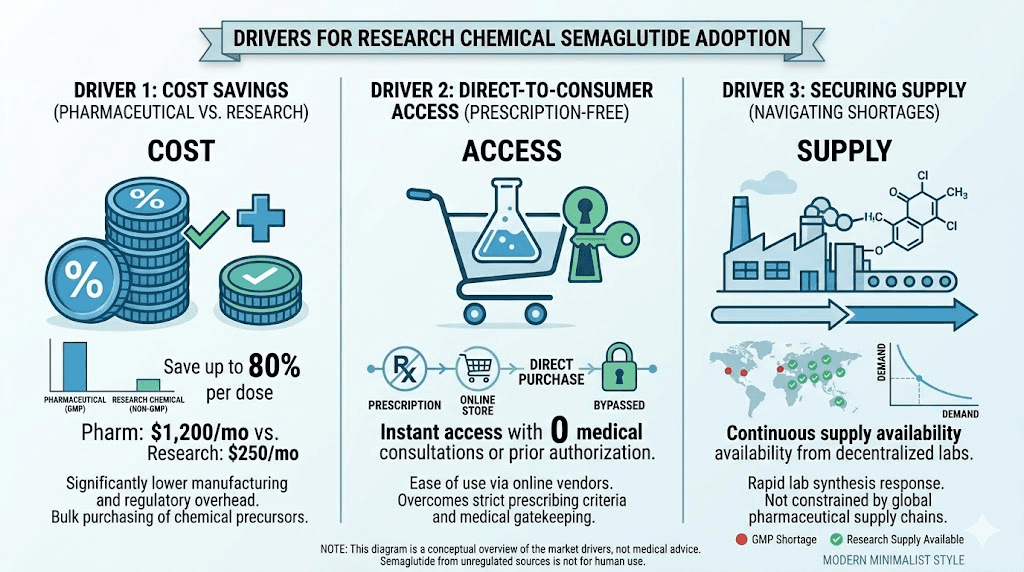
Cost
This is the primary driver. Pharmaceutical semaglutide without insurance can cost $1,000 or more per month. Compounded versions, while less expensive, still run several hundred dollars monthly. Research chemical semaglutide can cost a fraction of these prices, sometimes $50-100 for a multi-month supply at common dosing levels.
The math is simple. The cost difference is enormous. For many people, the choice between an unaffordable pharmaceutical product and a cheap research chemical is not really a choice at all. Financial pressure pushes people toward the lowest-cost option, even when they understand the risks.
Access barriers
Getting a prescription for semaglutide requires a healthcare provider visit, meeting specific diagnostic criteria, insurance authorization (often denied), and ongoing medical supervision. Each of these steps represents a barrier. Research chemicals bypass all of them. No prescription. No doctor visit. No insurance required. Order online, receive in days.
For researchers who have struggled with the qualification process for semaglutide, the appeal of unrestricted access is obvious. But convenience and safety do not always align.
Supply shortages
The global semaglutide shortage that began in 2022 and persisted through much of 2024 pushed many people toward alternative sources. When pharmacies could not fill prescriptions, research chemical vendors could ship immediately. Even though the FDA declared the shortage resolved in February 2025, the supply chain disruption established purchasing patterns that persist.
The researcher identity
Some buyers genuinely purchase semaglutide for in vitro research, cell culture studies, or animal research protocols. Legitimate research applications exist. But the volume of research chemical sales compared to the number of actual research laboratories purchasing these products tells a clear story about who is really buying and why.
Compounded semaglutide versus research chemicals: the critical distinctions
These terms are not interchangeable, though they are frequently confused.
Compounded semaglutide is prepared by a licensed compounding pharmacy, typically a 503A (patient-specific) or 503B (outsourcing facility) pharmacy. These pharmacies are regulated by state boards of pharmacy and the FDA. They require a valid prescription. They must use pharmaceutical-grade ingredients from FDA-registered suppliers. They must follow specific compounding standards (USP 795 or 797 depending on the dosage form). And they are subject to inspection.
Many compounded formulations include additional ingredients like vitamin B12, glycine, or niacinamide as stabilizers or complementary compounds. These additions are documented, with known concentrations and quality controls.
Research chemical semaglutide is sold by a vendor with no pharmacy license. No prescription required. No regulatory oversight of the manufacturing process. No requirement to use pharmaceutical-grade starting materials. No mandatory testing standards. The only real requirement is the "for research use only" label.
The practical differences show up in predictable ways.
Factor | Compounded semaglutide | Research chemical semaglutide |
|---|---|---|
Prescription required | Yes | No |
Regulatory oversight | FDA and state pharmacy boards | None |
Ingredient sourcing | FDA-registered suppliers | Unregulated, often overseas |
Testing requirements | USP standards, beyond-use dating | Vendor discretion |
Endotoxin testing | Required for injectables | Rarely performed |
Cost | $150-400/month typical | $30-100/month typical |
Traceability | Full chain of custody | Limited or none |
If you are exploring compounded options, understanding the specific offerings from pharmacies like Empower Pharmacy, Olympia Pharmacy, or Belmar Pharmacy provides concrete comparison points. Each operates under regulatory frameworks that research chemical vendors do not.
The blended formulation angle
Some compounding pharmacies offer semaglutide combined with other ingredients in specific ratios. Semaglutide with glycine and B12 is a common combination. Semaglutide with L-carnitine targets additional metabolic support. Semaglutide with methylcobalamin provides a bioactive form of B12.
Research chemical vendors sometimes mimic these combinations, but without the regulatory requirements that ensure accurate concentrations and ingredient quality. The result is a product that looks similar on the label but may have wildly different actual composition.
How to evaluate a certificate of analysis
If you are going to assess any semaglutide product, whether research chemical or compounded, understanding the COA is essential.
What a legitimate COA includes
Identity confirmation. Mass spectrometry data (typically ESI-MS or MALDI-TOF) showing the molecular weight matches semaglutide. The expected molecular weight for semaglutide base is approximately 4,113.58 Da. Look for the [M+H]+ or multiply charged ion peaks consistent with this mass.
Purity by HPLC. High-performance liquid chromatography separates the target compound from impurities. A legitimate COA shows the chromatogram (the graph) with the main peak identified as semaglutide and any impurity peaks quantified. Pharmaceutical standards typically require greater than 95% purity. Research chemical COAs claiming 99% should be viewed skeptically given independent testing results.
Endotoxin testing (LAL). The Limulus Amebocyte Lysate test detects bacterial endotoxins. For injectable products, this is critical. USP limits for injectable products are typically 5 EU/kg body weight. A COA that does not include endotoxin data for an injectable product is incomplete.
Residual solvent analysis. Peptide synthesis uses organic solvents (DMF, NMP, DCM, TFA) that can persist in the final product. GC-MS testing should confirm these solvents are below acceptable limits per ICH Q3C guidelines. Most research chemical COAs omit this entirely.
Water content. Karl Fischer titration measures residual moisture in lyophilized products. Excessive water content reduces stability and shelf life. Understanding semaglutide shelf life starts with knowing the water content of the product when it arrives.
Red flags in COAs
Generic COAs not tied to a specific batch number. COAs showing round numbers (exactly 99.0% purity) rather than precise measurements (98.73%). Missing chromatogram images. No contact information for the testing laboratory. COAs dated significantly before the product was shipped. These all suggest the documentation may not reflect the actual product in your hands.
When you receive a product and want to verify its quality, the visual appearance provides initial clues. Understanding what semaglutide should look like helps identify obviously degraded or contaminated products. The lyophilized powder should be white to off-white. Significant discoloration, unusual texture, or unexpected color changes warrant concern.
Safety risks of unregulated semaglutide products
The risks fall into several categories, each with different severity and likelihood profiles.
Impurity-related risks
When a product is 85-93% impurities (as the published testing data suggests for some research chemicals), those impurities matter. They could include truncated peptide sequences that are biologically active in unpredictable ways. Deleted sequences missing one or more amino acids. Racemized amino acids that alter the peptide three-dimensional structure. Residual coupling reagents from synthesis. Deprotection byproducts. Aggregated peptide forms.
None of these impurities have been studied for safety in human tissues. Their effects are unknown. That is the fundamental problem with low-purity products. You are not just getting less semaglutide. You are getting an unknown mixture of compounds with unknown biological activity.
If a product causes unexpected dizziness, fatigue, bloating, or acid reflux beyond what is typical for pharmaceutical semaglutide, impurities could be contributing factors that are impossible to diagnose or manage without knowing what they are.
Endotoxin-related risks
Endotoxins trigger immune responses. Low-level endotoxin exposure from injectable products can cause injection site inflammation, fever, malaise, and flu-like symptoms. Higher levels can cause more serious systemic reactions. The injection site reactions that some users report may be partially attributable to endotoxin contamination rather than the peptide itself.
Dosing uncertainty
When purity is unknown, dosing becomes guesswork. If you calculate a dose based on a semaglutide dosage chart assuming 99% purity but the actual purity is 10%, your effective dose is dramatically different from what you intended. This uncertainty compounds with every injection. Some batches might be closer to the claimed purity. Others might be far below. Without testing each batch independently, consistency is impossible.
The semaglutide dosage calculator on our site assumes a known concentration. Research chemical products with uncertain purity make these calculations unreliable. This is not a minor inconvenience. It is a fundamental barrier to achieving predictable, safe outcomes.
Storage and stability concerns
Even if a research chemical product starts with acceptable quality, storage conditions throughout the supply chain can degrade it. Semaglutide is sensitive to temperature. Exposure to heat accelerates degradation. If your product was exposed to warm temperatures during shipping or storage, potency may be compromised regardless of what the COA says about the original batch.
Understanding how long semaglutide lasts in the fridge after reconstitution is critical, but that guidance assumes you started with a product of known quality. An already degraded product will deteriorate faster than expected. If you have ever wondered about semaglutide refrigeration requirements, the answer becomes even more critical when dealing with products of uncertain quality.
Products that arrive at room temperature, or worse, arrive hot during summer shipping, represent a specific degradation risk. And leaving semaglutide out overnight after reconstitution compounds whatever quality issues existed from the start.
What experienced researchers actually look for
People who have worked with research peptides for years develop evaluation strategies that go beyond reading a COA.
Vendor consistency testing
Experienced researchers often purchase from the same vendor multiple times and compare results across batches. If the same vendor consistently delivers product that reconstitutes clearly, appears correctly as white to off-white colored powder, and produces predictable results, that track record has value. But it does not replace independent testing, and past performance does not guarantee future quality.
Community intelligence
Online forums, subreddits, and peptide communities aggregate user experiences across vendors. While individual reports are unreliable (placebo effect, poor technique, batch variation), patterns across many reports can identify consistently good or bad vendors. A vendor with dozens of negative reports about product quality, inconsistent results, or contamination should be avoided regardless of what their website claims.
Reconstitution behavior
The way a lyophilized peptide dissolves provides crude quality information. Semaglutide should dissolve relatively quickly in bacteriostatic water with gentle swirling. Excessive foaming, particulate matter that will not dissolve, unusual color after reconstitution, or slow dissolution can indicate problems. Proper reconstitution technique matters, but even perfect technique cannot overcome a degraded or contaminated starting product.
If you are reconstituting a 5mg vial of semaglutide, the amount of bacteriostatic water to use determines your concentration. For a 10mg vial, the calculation changes accordingly. But all of these calculations assume you actually have the stated amount of semaglutide in the vial, an assumption that published testing data calls into question.
Independent testing
Some researchers send samples to independent analytical laboratories for testing. This costs money, typically $100-300 per sample for basic identity and purity analysis. But it provides actual data about what you have rather than what a vendor claims you have. For anyone spending significant amounts on research chemicals over time, periodic independent testing is a rational investment in knowing what you are working with.
The supply chain behind research chemicals
Understanding where research chemical semaglutide comes from helps explain the quality variation in the market.
Chinese bulk manufacturing
The majority of research-grade semaglutide peptide powder originates from Chinese chemical manufacturers. Some of these manufacturers operate sophisticated facilities with genuine quality control. Others prioritize volume and low cost over product quality. The range is enormous.
A study published in the journal SSM - Qualitative Research in Health examined the international supply chain for semaglutide and other weight-loss drugs sold online. The research found that Chinese manufacturers openly advertise bulk semaglutide powder on platforms like Alibaba, with prices as low as a few dollars per gram at bulk quantities. These products are then repackaged by vendors worldwide, often with minimal additional quality testing.
This pattern mirrors what happens in other peptide markets. Bulk retatrutide and bulk retatrutide from China follow similar supply chain patterns, with the same quality variability concerns.
Vendor repackaging
Many US-based research chemical vendors are not manufacturers. They are resellers. They purchase bulk powder from overseas suppliers, aliquot it into individual vials, lyophilize it (or sell it as-is), and apply their own labels. Some perform quality testing on the bulk powder before repackaging. Others rely entirely on the certificate of analysis provided by their upstream supplier, which may itself be unreliable.
The gap between a vendor website showing a professional laboratory setup and the reality of their operations can be significant. Some vendors do maintain clean rooms and analytical equipment. Others operate out of far less controlled environments. Without visiting the facility or seeing independent verification, it is impossible to know which category a given vendor falls into.
The legal landscape and what it means for buyers
The legal situation for research chemical peptides exists in a gray area that is rapidly narrowing.
Current legal status
Semaglutide is a prescription drug. Selling it without a prescription for human use is illegal under federal law. The "research use only" label creates a legal fig leaf, but the FDA has demonstrated its willingness to look past that label when the evidence shows products are being sold for human consumption.
For buyers, the legal risk is generally lower than for sellers. Purchasing a research chemical is not typically a criminal offense. But the products themselves exist outside any consumer protection framework. If a product causes harm, there is no regulatory recourse, no product liability in most cases, and no guarantee that the vendor will still be in business when problems emerge.
Regulatory trends
The direction is unmistakable. More enforcement. More warning letters. More seizures of products at customs. More pressure on payment processors to stop servicing research chemical vendors. More coordination between the FDA, DEA, and state pharmacy boards.
The February 2025 resolution of the semaglutide shortage removed the primary legal justification for many compounding operations. The September 2025 wave of 50+ warning letters signaled escalating enforcement. For anyone building a long-term research program around access to affordable semaglutide, the regulatory trajectory suggests decreasing availability and increasing risk over time.
This broader shift affects the entire peptide landscape, not just semaglutide. Understanding the online peptide therapy landscape helps contextualize where research chemicals fit within the larger ecosystem of options available to researchers.
Making informed decisions about semaglutide research
For researchers who have evaluated the risks and still choose to work with research chemical semaglutide, several practices can reduce (though never eliminate) the uncertainty.
Start with proper dosing knowledge
Before working with any semaglutide product, understanding the full dosing landscape provides essential context. The semaglutide dosage in units guide covers how insulin syringe units translate to milligram doses. Conversion charts help with the math. And knowing how many milligrams common unit measurements represent prevents basic calculation errors.
For specific measurements: 10 units, 20 units, 25 units, 50 units, and 100 units each have specific milligram equivalents that depend on your reconstitution concentration. Getting these calculations right starts with the semaglutide reconstitution chart and the mixing chart for your specific vial size.
Storage protocols matter even more
With products of uncertain baseline quality, proper storage becomes even more critical. Understanding semaglutide shelf life helps set realistic expectations. Keep lyophilized powder refrigerated. After reconstitution, maintain cold chain storage continuously. The fridge storage duration for reconstituted semaglutide assumes optimal starting conditions, so err on the shorter end when working with research chemicals.
If you need to travel with semaglutide, maintaining the cold chain becomes an additional challenge. Research chemical products without the stabilizing formulation components found in pharmaceutical versions may be more sensitive to temperature excursions.
Monitor for unexpected effects
When using any semaglutide product, knowing what to expect helps identify problems early. The first week on semaglutide typically involves appetite suppression and mild gastrointestinal effects. If you experience effects that fall outside the normal range, product quality could be a factor.
Common expected effects include some degree of constipation, burping, and appetite changes. But unusual severity, unexpected symptoms, or reactions that do not match published semaglutide side effect profiles may indicate impurity-related problems. Understanding the typical timeline for appetite suppression provides a baseline for comparison.
Know when semaglutide is not working
If you are four weeks in with no results, the product quality should be among the first things you investigate. While many factors can explain poor response to semaglutide, a product that contains 7-14% actual semaglutide (as independent testing has found for some research chemicals) simply cannot produce the results that a full-purity product would.
The phenomenon of semaglutide plateaus is well documented even with pharmaceutical products. But if results plateau immediately or never begin, product quality deserves scrutiny before adjusting dose or switching protocols.
Alternative approaches worth considering
For researchers who have weighed the risks of research chemicals and want to explore other options, several alternatives exist with different risk-benefit profiles.
Compounded semaglutide with added compounds
Licensed compounding pharmacies can prepare semaglutide with complementary ingredients. Semaglutide with B12 is popular for energy support. Semaglutide with glycine adds a stabilizing amino acid. These formulations come from regulated sources with quality testing, at a higher price point than research chemicals but with substantially more quality assurance.
Oral and sublingual options
The semaglutide delivery landscape has expanded beyond injection. Sublingual semaglutide and oral semaglutide drops offer needle-free alternatives, though bioavailability differs from injectable forms. Semaglutide troches represent another oral delivery method. Each has different absorption characteristics that affect dosing and efficacy.
Other GLP-1 peptides
Semaglutide is not the only GLP-1 receptor agonist available. Tirzepatide targets both GLP-1 and GIP receptors, with some research suggesting superior weight loss outcomes. Understanding the side effect differences between semaglutide and tirzepatide helps inform the comparison. Adding retatrutide to the comparison introduces a triple agonist (GLP-1, GIP, and glucagon receptors) that represents the newest generation of these compounds.
For those exploring the full landscape of weight management options, understanding how semaglutide compares to older options like phentermine and whether combining them is feasible provides additional decision-making context. Other combinations like berberine with semaglutide and metformin with semaglutide also have research behind them.
Lifestyle optimization alongside any peptide protocol
Regardless of semaglutide source, lifestyle factors dramatically influence outcomes. Understanding the best foods to eat on semaglutide, knowing which foods to avoid, and planning caloric intake appropriately all enhance results regardless of product source. A comprehensive semaglutide diet plan combined with practical tips helps maximize the value of whichever product a researcher chooses.
The timing of semaglutide administration also matters. Research suggests that injection timing relative to meals can influence tolerability and effectiveness. And injection site selection affects absorption consistency.
Vendor-specific considerations
Without endorsing any specific research chemical vendor (quality varies by batch, and past performance does not guarantee future results), understanding what established vendors in the compounding space offer provides useful benchmarks.
Empower Pharmacy operates as a 503B outsourcing facility with published testing standards. Olympia Pharmacy has established protocols for compounded semaglutide with various formulations. Direct Meds, BPI Labs, and Belmar Pharmacy each have their own approach to semaglutide compounding. Reviewing multiple providers like Amino Asylum, Amino USA, Brello, Elevate Health, WeightCare, and Levity helps build a comprehensive understanding of the available options.
Each vendor has different dosage charts, pricing structures, and formulation options. Some offer 10mg dosage configurations, while others focus on 5mg vial options. The compounded semaglutide dose chart varies by provider, so understanding each vendor specific protocols is essential before starting.
Long-term considerations for semaglutide researchers
Whether using research chemicals or compounded products, several long-term factors deserve attention.
Duration of use
Research on how long to stay on semaglutide suggests that weight management benefits require ongoing use. Discontinuation typically leads to weight regain, which raises the question of withdrawal symptoms and what happens when stopping abruptly. Restarting after a break involves its own protocol considerations.
For long-term users, cumulative exposure to impurities from research chemical products represents an unknown risk. A few weeks of use exposes you to relatively limited amounts of unknown compounds. Years of use multiply that exposure significantly.
Monitoring and safety
Regular monitoring helps catch problems early regardless of product source. Awareness of how semaglutide affects liver enzymes, menstrual cycles, blood clotting, and sleep quality helps researchers identify when something is not right. Blood work provides objective data that subjective assessment cannot.
Energy levels and libido changes may also signal how the body is responding to the product. Tracking these markers over time against known semaglutide effect profiles helps distinguish normal response from product quality issues.
Results tracking
Documenting results systematically provides data that subjective impression cannot. Week-by-week results tracking establishes a baseline and reveals trends. One-month checkpoints help evaluate whether a product is performing as expected based on published clinical trial data.
If results from a research chemical product fall significantly below what clinical trials show for pharmaceutical semaglutide at equivalent doses, product quality is the most likely explanation. This comparison is imperfect since research chemicals may not contain the actual dose you calculated, but the pattern provides useful information.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Understanding the difference between research-grade and pharmaceutical-grade products is just the starting point. SeekPeptides members access detailed protocol builders, vendor comparison tools, and direct guidance from experienced researchers who can help translate these concepts into practical decisions.
Frequently asked questions
Is research chemical semaglutide the same as Ozempic?
No. Research chemical semaglutide targets the same active ingredient but is produced without FDA oversight, cGMP manufacturing standards, or mandatory quality testing. Independent analysis has shown purity levels as low as 7-14% in tested research chemical products, compared to the rigorous standards applied to FDA-approved Ozempic. The salt form may also differ, with research chemicals often using sodium or acetate salts rather than semaglutide base.
How can I test research chemical semaglutide purity?
Independent analytical laboratories can perform HPLC purity analysis and mass spectrometry confirmation for approximately $100-300 per sample. This provides actual purity data rather than relying on vendor-supplied certificates of analysis. Look for labs that specialize in peptide analysis and can provide chromatograms along with numerical results.
Are research chemicals legal to buy?
The legal status exists in a gray area. Purchasing research chemicals is not typically a criminal offense for buyers. However, selling prescription drug analogs labeled "for research use only" while clearly marketing to consumers is increasingly subject to FDA enforcement, as demonstrated by the 50+ warning letters issued in September 2025.
What is the difference between semaglutide base and semaglutide sodium?
Semaglutide base is the active ingredient used in FDA-approved products. Semaglutide sodium is a salt form created by reacting semaglutide with sodium hydroxide. The FDA considers these different active ingredients and has stated there is no evidence showing salt forms are safe or effective. Salt forms have different solubility, stability, and potentially different pharmacokinetic properties.
Can I trust the certificate of analysis from a research chemical vendor?
Vendor-supplied COAs have inherent conflict-of-interest limitations. Third-party COAs from independent laboratories are more reliable. Look for batch-specific testing, chromatogram images, and contact information for the testing laboratory. COAs showing exactly 99.0% purity or lacking batch numbers should be viewed skeptically.
Why is research chemical semaglutide so much cheaper?
Lower costs reflect the absence of cGMP manufacturing requirements, regulatory compliance expenses, clinical testing, prescription infrastructure, and comprehensive quality assurance programs. The true cost of quality peptide products includes substantial testing and manufacturing overhead that research chemical vendors skip.
Should I switch from research chemicals to compounded semaglutide?
Compounded semaglutide from a licensed pharmacy offers significantly more quality assurance than research chemicals. The tradeoff is higher cost and the requirement for a prescription. For researchers concerned about product quality and safety, the additional cost of a compounded product may be justified by the reduced risk and greater consistency.
How do I store research chemical semaglutide properly?
Store lyophilized powder refrigerated (2-8 degrees Celsius) in its original sealed vial. After reconstitution, keep refrigerated and use within 28-30 days. Protect from light. Never freeze reconstituted solutions. Monitor the color and clarity of reconstituted product before each use, as changes may indicate degradation.
External resources
FDA: Concerns with unapproved GLP-1 drugs used for weight loss
PMC: Multifactor quality and safety analysis of semaglutide products sold online
NABP: RogueRx activity report on injectable weight loss drugs
In case I do not see you, good afternoon, good evening, and good night. May your peptides stay pure, your protocols stay evidence-based, and your decisions stay informed.