Mar 15, 2026

Of the millions of people now taking semaglutide for weight loss or type 2 diabetes, a surprising number get bloodwork back and panic. ALT is up. Or down. Or different from last time. And suddenly the question hits: is this medication helping my liver or quietly destroying it?
The answer is more nuanced than most articles will tell you. For the vast majority of users, semaglutide actually improves liver enzyme levels. Clinical trials consistently show reductions in both ALT and AST, particularly in people who had elevated levels to begin with. The medication has become so promising for liver and metabolic health that the FDA approved it specifically for liver fibrosis caused by nonalcoholic fatty liver disease.
But there is another side to this story. Rare cases of drug-induced liver injury have been reported. At least ten cases sit in the FDA adverse event database. Some were severe. And for the person who happens to fall into that tiny percentage, the distinction between "rare" and "impossible" matters enormously. This guide breaks down everything the research shows about semaglutide and liver enzymes, from the mechanisms that make it protective for most people, to the warning signs that demand immediate attention, to the exact monitoring protocols that keep you safe throughout treatment. Whether your liver enzymes just came back elevated or you are starting semaglutide with existing liver concerns, the data here will help you make informed decisions with your healthcare provider.
Understanding liver enzymes and why they matter
Before diving into what semaglutide does to liver enzymes, you need to understand what these numbers on your bloodwork actually mean. Liver enzymes are proteins that your liver cells release into your bloodstream. When liver cells are damaged or inflamed, they leak more of these enzymes than usual. Higher levels on a blood test suggest something is stressing your liver. Lower levels generally mean things are calm.
Two enzymes dominate the conversation.
ALT (alanine aminotransferase) is the most liver-specific enzyme. When ALT goes up, it almost always points to liver cell damage. Normal ranges typically fall between 7 and 56 units per liter, though some labs use tighter ranges. ALT is the enzyme researchers watch most closely in semaglutide studies because it responds directly to changes in liver fat content and inflammation.
AST (aspartate aminotransferase) lives in the liver too, but also in heart muscle, skeletal muscle, and other tissues. An elevated AST does not automatically mean liver trouble. Normal ranges sit between 10 and 40 units per liter. When both ALT and AST rise together, that points more clearly toward the liver as the source.
There are other liver markers worth knowing. GGT (gamma-glutamyl transferase) helps identify bile duct problems and alcohol-related liver damage. ALP (alkaline phosphatase) points to bile duct obstruction or bone disease. Bilirubin, albumin, and INR round out the full liver function picture. But for the semaglutide conversation, ALT and AST are where the action is.

How semaglutide affects liver enzymes in most people
Here is the headline that matters most. In the overwhelming majority of cases, semaglutide lowers liver enzyme levels.
This is not a small effect. A pivotal study published in Alimentary Pharmacology and Therapeutics examined semaglutide treatment in subjects with type 2 diabetes and obesity who were at high risk for nonalcoholic fatty liver disease. The results were striking. Semaglutide significantly reduced elevated ALT levels alongside reductions in high-sensitivity C-reactive protein, a marker of systemic inflammation. These improvements were dose-dependent, meaning higher doses of semaglutide produced greater reductions in liver enzymes. And critically, the degree of ALT improvement correlated with the amount of weight loss achieved.
A 52-week real-life study tracking patients with type 2 diabetes found that once-weekly subcutaneous semaglutide improved fatty liver disease markers across the board. ALT dropped. AST dropped. Ultrasound-confirmed liver fat decreased. These were not marginal improvements seen only in ideal conditions. They happened in everyday clinical practice with real patients.
The STEP trials, which form the backbone of semaglutide approval data, consistently showed liver enzyme improvements as a secondary finding. Patients receiving semaglutide at therapeutic doses had liver enzyme changes that trended in the right direction compared to placebo groups. Serum enzyme elevations were no more common with semaglutide than with placebo or comparator agents.
Why does this happen? Several mechanisms work together.
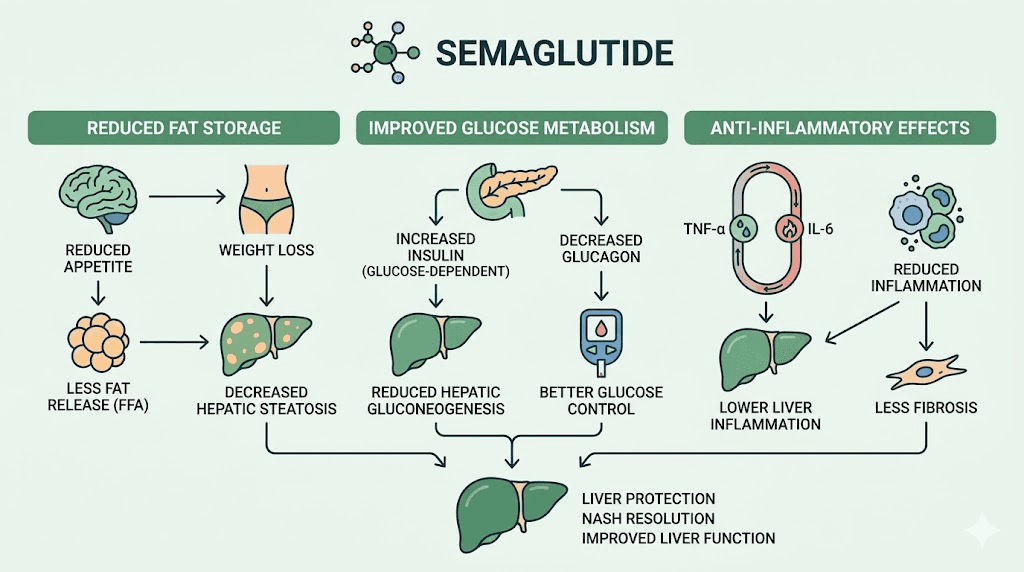
Weight loss reduces liver fat directly
The most obvious pathway is straightforward. Semaglutide causes significant weight loss. Losing weight, particularly visceral fat around organs, directly reduces the amount of fat stored in liver cells. Less fat means less inflammation. Less inflammation means fewer damaged liver cells. Fewer damaged liver cells means lower enzyme levels on your bloodwork. For someone who has been carrying excess liver fat for years, this reduction can be dramatic. Studies show that losing just 5-7% of body weight can meaningfully reduce liver fat content, and semaglutide users frequently lose 10-15% or more of their starting body weight.
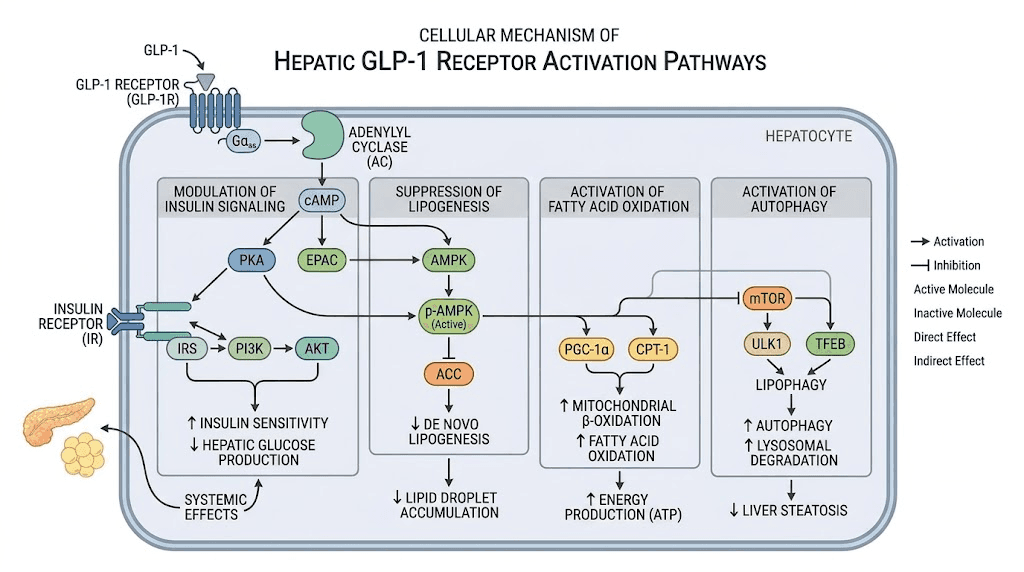
GLP-1 receptor activation provides direct hepatoprotection
The benefits go beyond weight loss. GLP-1 receptors exist on liver cells, and activating them triggers a cascade of protective effects that operate independently of how much weight you lose.
Research published in The Lancet Gastroenterology and Hepatology outlines the mechanisms. GLP-1 receptor agonists enhance hepatic glucose metabolism, meaning the liver handles sugar more efficiently. They reduce lipogenesis, the process by which the liver converts excess calories into stored fat. They promote fatty acid oxidation, actively burning liver fat for energy. They elevate adiponectin levels, a hormone that protects against insulin resistance and liver inflammation.
There is more. GLP-1 agonists interact with FXR and LXR pathways in the liver, reducing inflammation and fatty acid buildup at the cellular level. They enhance autophagy, the process by which liver cells clean up damaged components and recycle them. They reduce endoplasmic reticulum stress-related cell death. In plain language, semaglutide tells your liver cells to clean house, burn fat, and stop making more.
One particularly fascinating finding came from studies on dulaglutide, another GLP-1 receptor agonist. Researchers discovered that the hepatoprotective effects persisted even when controlling for weight loss, confirming that GLP-1 activation provides liver benefits through mechanisms that have nothing to do with dropping pounds on the scale. This suggests that semaglutide metabolic benefits extend deep into cellular function.
Reduced inflammation protects liver tissue
Semaglutide reduces systemic inflammation. High-sensitivity C-reactive protein drops. Inflammatory cytokines decrease. This matters for the liver because chronic low-grade inflammation is the engine that drives simple fatty liver into the more dangerous territory of steatohepatitis and fibrosis. By cooling that inflammatory fire, semaglutide slows or reverses the progression of liver disease in ways that show up clearly on enzyme panels.
Semaglutide and nonalcoholic fatty liver disease
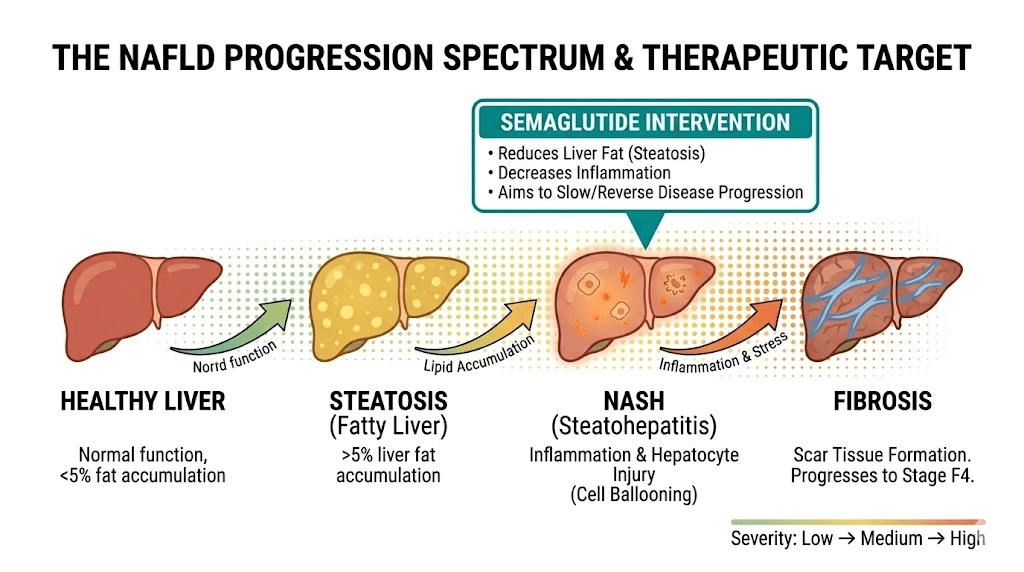
This is where the story gets genuinely exciting. Nonalcoholic fatty liver disease affects roughly 25% of the global population. It is the most common chronic liver condition in the world. And semaglutide may be one of the most effective treatments ever studied for it.
NAFLD exists on a spectrum. Simple steatosis means fat accumulation without significant inflammation. Nonalcoholic steatohepatitis, or NASH, adds inflammation and cell damage to the picture. Left unchecked, NASH can progress to fibrosis, cirrhosis, and eventually liver failure or hepatocellular carcinoma. The progression is slow, often taking decades, but it is relentless in the absence of intervention.
The landmark NASH trial
The study that changed everything was published in the New England Journal of Medicine. Researchers enrolled 320 patients with biopsy-confirmed NASH and liver fibrosis at stages F1 through F3. They randomized patients to receive semaglutide at doses of 0.1mg, 0.2mg, or 0.4mg daily, or placebo, for 72 weeks.
The results were remarkable.
NASH resolution with no worsening of fibrosis occurred in 40% of the 0.1mg group, 36% of the 0.2mg group, 59% of the 0.4mg group, and just 17% of the placebo group. Improvement in the NAFLD activity score was observed in 71% of patients at 0.1mg, 80% at 0.2mg, and 83% at 0.4mg, compared to 44% with placebo. These numbers represent a genuine breakthrough. No other medication had demonstrated this level of NASH resolution in a randomized controlled trial.
There was one important caveat. Despite improvements in inflammation and steatosis, the fibrosis endpoint did not reach statistical significance. Semaglutide resolved the disease activity but did not convincingly reverse existing scar tissue. Subsequent studies and meta-analyses have confirmed this pattern, showing that while semaglutide dramatically improves metabolic liver parameters, its effect on established fibrosis remains less clear.
NASH-related cirrhosis trial
A phase 2 trial published in The Lancet Gastroenterology and Hepatology went further, testing semaglutide 2.4mg once weekly in patients who already had NASH-related cirrhosis. This is the most advanced and dangerous stage of fatty liver disease. Even in this population, semaglutide showed improvements in liver inflammation markers and metabolic parameters, though the trial was designed primarily to assess safety rather than efficacy.
FDA approval for liver fibrosis
The accumulated evidence was strong enough that the FDA approved semaglutide (as Wegovy) for the treatment of liver fibrosis due to NAFLD, also now called metabolic dysfunction-associated steatotic liver disease or MASLD. This makes semaglutide one of the first medications specifically approved for this indication, a recognition of just how profound the therapeutic effects are.
For people with concurrent obesity and fatty liver disease, semaglutide now addresses both conditions simultaneously. The weight loss improves the liver. The direct hepatoprotective effects improve the liver further. The result is a compound benefit that neither weight loss alone nor any previous liver medication could match.
When liver enzymes go up on semaglutide
Not everyone sees their liver enzymes improve. A small number of people experience the opposite. And while rare, these cases deserve serious attention.
Transient elevations during dose escalation
Some people see a temporary bump in liver enzymes during the first few weeks of treatment or when increasing their dose. This is usually mild, self-limiting, and resolves without intervention. The proposed mechanism involves rapid metabolic changes as the body adjusts to the medication. Fat mobilization from the liver can temporarily increase enzyme release before the overall trend becomes favorable.
If your ALT or AST goes up slightly during dose escalation but stays below 3 times the upper limit of normal, and you have no symptoms, most clinicians will monitor and continue. The elevation typically resolves within 4-8 weeks as your body adapts.
Rapid weight loss effects
Rapid weight loss itself can temporarily elevate liver enzymes. This is not unique to semaglutide. It happens with bariatric surgery, very low calorie diets, and any intervention that causes fast fat mobilization. When stored fat leaves liver cells quickly, the cells undergo stress that can temporarily raise enzyme levels. This phenomenon has been well documented in weight management research for decades.
The key distinction is trajectory. If enzymes bump up but then trend downward over subsequent weeks, that pattern suggests beneficial remodeling. If they continue climbing, something else may be happening.
Drug-induced liver injury: rare but real
The most concerning scenario is drug-induced liver injury, or DILI. This is when the medication itself causes liver damage through an idiosyncratic reaction, meaning it is unpredictable, unrelated to dose, and affects only a tiny subset of individuals who happen to be susceptible.
The NCBI LiverTox database documents this clearly. In large clinical trials, no instances of clinically apparent liver injury were attributed to semaglutide. The database notes that treatment with semaglutide and other GLP-1 analogues was usually associated with improvements in serum aminotransferase levels. However, since the medication entered widespread use, rare cases have emerged.
At least ten cases of hepatic injury attributable to semaglutide have been reported to the FDA Adverse Event Reporting System. Published case reports describe patients developing significantly elevated ALT and AST levels, sometimes reaching 10 to 20 times the upper limit of normal. In these cases, liver enzyme levels normalized after semaglutide was discontinued, confirming the medication as the cause.
A case report published in the ACG Case Reports Journal described semaglutide-induced liver injury in detail.
The patient developed markedly elevated transaminases without other identifiable causes. Discontinuation led to normalization. Rechallenge was not attempted, which is typical in suspected DILI cases.
Another case published in Clinical Case Reports described semaglutide-induced hepatic injury as idiosyncratic, emphasizing that it affected a susceptible individual in a way that could not have been predicted from the clinical trial data or the patient medical history.
The absolute risk is extremely small. With millions of prescriptions written, ten reported cases puts the incidence in the range of 1 in several hundred thousand. But if you are the one experiencing it, the rarity provides cold comfort. This is why monitoring matters.
Complete liver enzyme monitoring protocol
Smart monitoring catches problems early and confirms benefits. Here is what the evidence supports.
Before starting semaglutide
Get baseline liver function tests. This is non-negotiable. You need to know where you stand before the medication changes anything. The panel should include ALT, AST, GGT, ALP, total bilirubin, and albumin. If any values are already elevated, your prescriber needs to investigate the cause before starting treatment. Pre-existing liver disease does not necessarily preclude using semaglutide, but it does change the monitoring intensity.
If you already have diagnosed NAFLD or NASH, the baseline becomes even more important. Document your starting enzymes precisely. These are the numbers you will measure improvement against. Consider asking for a comprehensive metabolic panel rather than just a basic one.
During dose escalation (months 1-4)
Check liver enzymes at each dose increase. Standard semaglutide dose escalation happens every 4 weeks. At each step up, repeat the panel. This is the window where both transient benign elevations and early DILI would become apparent.
What to watch for:
ALT or AST rising above 3x the upper limit of normal (ULN) without symptoms: increase monitoring frequency to every 2 weeks. Continue medication with caution.
ALT or AST rising above 5x ULN: temporarily discontinue semaglutide. Investigate other causes. Do not restart until enzymes normalize and cause is identified.
Any elevation accompanied by symptoms (jaundice, dark urine, right upper quadrant pain, unexplained fatigue): stop semaglutide immediately and seek urgent evaluation.
During maintenance therapy (month 5+)
Once you are on a stable dose and liver enzymes are stable or improving, monitoring can decrease to every 3-6 months. Most clinicians fold this into routine metabolic panels done as part of standard long-term semaglutide management.
For patients with pre-existing liver disease, more frequent monitoring (every 2-3 months) remains appropriate even during maintenance.
What normal improvement looks like
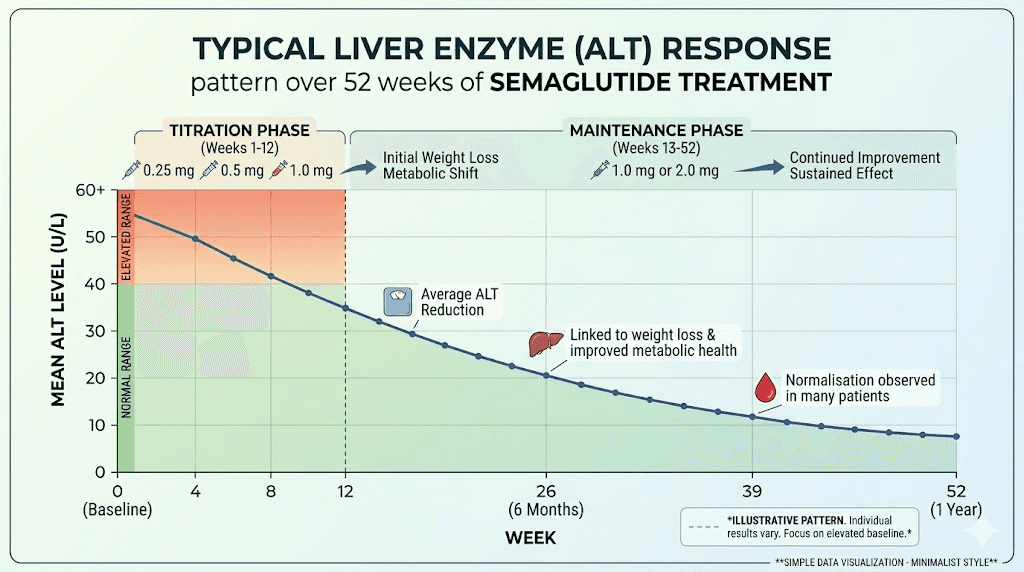
If you started with elevated liver enzymes, here is the typical pattern on semaglutide:
Weeks 1-4: Enzymes may stay flat or bump slightly upward during initial dose.
Weeks 4-12: Gradual downward trend begins. ALT and AST start moving toward normal ranges.
Weeks 12-24: Most significant improvement occurs. ALT often drops by 20-40% from elevated baselines.
Weeks 24-52: Continued improvement, stabilizing near or within normal ranges. The degree of improvement correlates with amount of weight lost.
Specific liver enzyme scenarios on semaglutide
Different starting points create different experiences. Here is what to expect based on where your liver enzymes begin.
Scenario 1: normal baseline liver enzymes
If your ALT and AST were normal before starting semaglutide, they will almost certainly stay normal throughout treatment. The clinical trial data is reassuring on this point. Serum enzyme elevations were no more common with semaglutide than with placebo in subjects with normal baseline values. You may even see your enzymes improve slightly as metabolic health improves overall.
Monitoring recommendation: standard schedule. Baseline, then every 3-6 months as part of routine care.
Scenario 2: mildly elevated baseline (1-3x ULN)
This is the group that often sees the most dramatic improvement. Many people with mildly elevated liver enzymes have undiagnosed NAFLD. The excess liver fat causes chronic low-level inflammation that keeps ALT and AST slightly above normal range. Semaglutide addresses this directly through both weight loss and direct hepatoprotective mechanisms.
Expect to see improvement beginning around week 8-12, with enzymes potentially normalizing by week 24-52. The landmark studies showed the greatest absolute reductions in people who started with the highest baseline values.
Monitoring recommendation: check at each dose escalation, then every 3 months during the first year.
Scenario 3: moderately elevated baseline (3-5x ULN)
Moderately elevated enzymes require investigation before starting semaglutide. Your prescriber should rule out viral hepatitis, autoimmune liver disease, hemochromatosis, Wilson disease, and medication-related causes before attributing elevations to fatty liver alone. If the cause is confirmed as NAFLD or NASH, semaglutide may be particularly beneficial, but monitoring needs to be tighter.
Monitoring recommendation: baseline comprehensive panel, check at every dose change, then monthly for the first 3 months of maintenance, then every 2-3 months ongoing.
Scenario 4: liver enzymes rise during treatment
If liver enzymes that were normal or improving suddenly start climbing during treatment, do not panic immediately. First, consider common explanations. Did you start any new medications? Are you consuming more alcohol than usual? Have you been taking acetaminophen regularly? Did you recently increase your semaglutide dose?
If the elevation is mild (under 3x ULN), recheck in 2-4 weeks. Transient bumps often resolve spontaneously. If it persists or worsens, broader investigation is warranted. The differential diagnosis for rising liver enzymes on semaglutide includes not just the medication itself but every other cause of liver enzyme elevation.
Drug-induced liver injury from semaglutide typically presents within the first few months of treatment, often during dose escalation. Late-onset DILI (appearing after months of stable dosing) is possible but even rarer.
Scenario 5: diagnosed NAFLD or NASH starting semaglutide
You represent the population with the most to gain. The clinical evidence specifically supports semaglutide for your situation. The FDA approval for liver fibrosis due to NAFLD validates what the trials have shown.
Expect improvement, but be patient. NASH resolution takes time. The landmark trial used a 72-week treatment period to demonstrate NASH resolution rates of 59% at the highest dose. Liver enzyme improvements typically precede histological improvements, meaning your bloodwork will get better before biopsy changes become apparent.
Work with a hepatologist if possible, not just a primary care provider. Managing semaglutide in the context of established liver disease benefits from specialist oversight. Serial imaging (ultrasound or FibroScan) can track improvements that blood tests alone may miss.
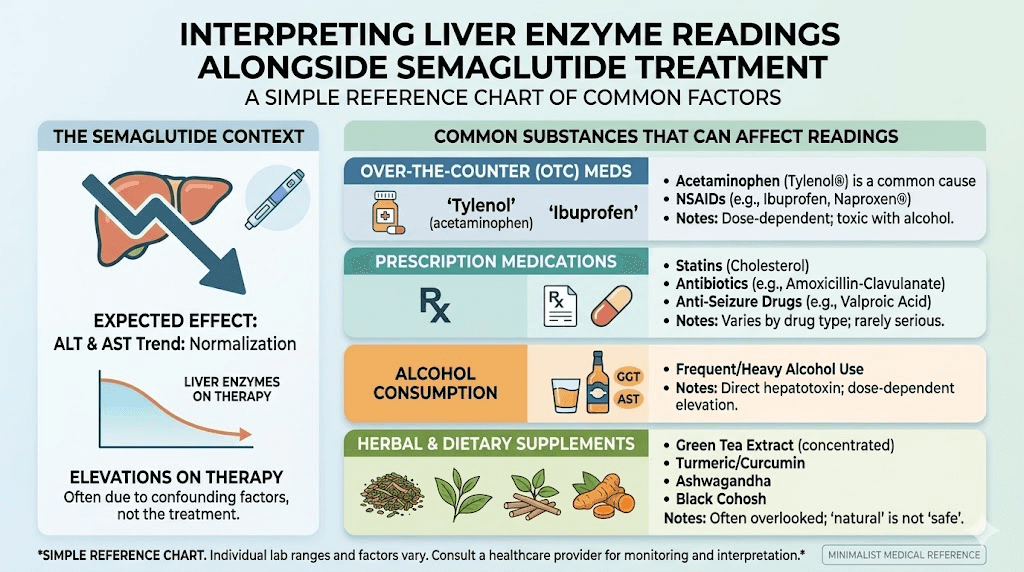
Medications and supplements that affect liver enzymes alongside semaglutide
You are probably not taking semaglutide in isolation. Many people on semaglutide also take other medications or supplements that can independently affect liver enzymes. Understanding these interactions prevents false alarms and missed diagnoses.
Common medications that elevate liver enzymes
Statins are the most frequent culprit. Atorvastatin, rosuvastatin, and other statins can cause mild ALT elevations in 1-3% of users. If you start a statin around the same time as semaglutide, an enzyme bump may be blamed on the wrong medication. Many people on semaglutide for metabolic health are also managing cholesterol with multiple medications.
Acetaminophen (Tylenol) is hepatotoxic at high doses and can cause mild enzyme elevations even at therapeutic doses in some people. If you are taking acetaminophen regularly for pain, your liver enzymes may reflect this rather than semaglutide effects.
NSAIDs like ibuprofen and naproxen can cause liver enzyme elevations, particularly with prolonged use. Antibiotics, particularly amoxicillin-clavulanate and fluoroquinolones, are common causes of drug-induced liver injury that could be confused with semaglutide effects if taken concurrently.
Metformin, often prescribed alongside semaglutide for type 2 diabetes, can occasionally cause liver enzyme elevations, though this is uncommon.
The combination of metformin and semaglutide is generally considered safe for the liver, and the two may even have synergistic hepatoprotective effects.
Supplements that affect liver enzymes
Herbal supplements are a frequently overlooked cause of liver enzyme abnormalities. Green tea extract in concentrated supplement form has been linked to hepatotoxicity. Kava, comfrey, and chaparral are known hepatotoxins. Even seemingly benign supplements like turmeric or ashwagandha can cause liver enzyme elevations in susceptible individuals.
If your liver enzymes are elevated on semaglutide, make a complete list of everything you are taking, including supplements, vitamins, and herbal products. Your prescriber needs the full picture.
Alcohol and semaglutide
Alcohol is the most common cause of elevated liver enzymes worldwide. Interestingly, many semaglutide users report decreased interest in alcohol during treatment, which may contribute to liver enzyme improvements beyond what weight loss alone would explain. However, if you continue drinking while on semaglutide, alcohol can keep your enzymes elevated despite the medication beneficial effects.
There is no absolute prohibition on alcohol with semaglutide, but if your goal includes liver health improvement, reducing or eliminating alcohol will dramatically enhance the results you see on bloodwork.
Semaglutide compared to other GLP-1 medications for liver health
Semaglutide is not the only GLP-1 receptor agonist with liver benefits. How does it compare?
Semaglutide vs tirzepatide for liver enzymes
Tirzepatide is a dual GIP/GLP-1 receptor agonist that has also shown liver enzyme improvements in clinical trials. The SURPASS trial program demonstrated reductions in ALT comparable to or potentially exceeding those seen with semaglutide, likely because tirzepatide produces greater average weight loss. For someone choosing between the two medications specifically for liver health, both are excellent options.
The dual receptor mechanism of tirzepatide may provide additional hepatoprotective effects through GIP receptor activation, though this hypothesis needs more research to confirm. Head-to-head trials comparing liver outcomes specifically have not been completed.
Semaglutide vs liraglutide for liver enzymes
Liraglutide, the older daily GLP-1 agonist, was actually the first in its class to demonstrate NASH resolution in a clinical trial (the LEAN study). However, semaglutide appears to produce superior liver outcomes, consistent with its greater efficacy for weight loss and metabolic improvement overall. The once-weekly dosing of semaglutide also provides more consistent receptor activation compared to the daily peaks and troughs of liraglutide administration.
Semaglutide vs retatrutide for liver enzymes
Retatrutide, the triple receptor agonist (GLP-1/GIP/glucagon), has shown promising early data for liver fat reduction that may exceed even semaglutide effects. The glucagon receptor component specifically targets liver fat mobilization, theoretically making it the most liver-directed of the incretin-based medications. Phase 2 data showed liver fat reductions of up to 80% at the highest doses. However, retatrutide is not yet approved, and long-term liver safety data is still accumulating.
For now, semaglutide remains the GLP-1 medication with the strongest evidence base for liver health, the broadest clinical experience, and the only FDA approval specifically for liver fibrosis.
Comparison table: GLP-1 medications and liver effects
Medication | Liver enzyme improvement | NASH resolution data | Liver-specific approval | Evidence strength |
|---|---|---|---|---|
Semaglutide | Strong (20-40% ALT reduction) | 59% resolution at 0.4mg | Yes (FDA approved) | 9/10 |
Strong (comparable to semaglutide) | Trials ongoing | Not yet | 7/10 | |
Liraglutide | Moderate | 26% resolution (LEAN trial) | No | 6/10 |
Very strong (up to 80% fat reduction) | Phase 2 data promising | Not yet approved | 5/10 | |
Preliminary positive data | Limited data | No | 3/10 |
The science behind GLP-1 liver protection
Understanding the mechanisms helps you appreciate why semaglutide works for the liver, not just that it works. This section goes deeper into the cellular biology.
Insulin signaling improvement
Insulin resistance is the root cause of most NAFLD. When liver cells become resistant to insulin, they lose the ability to properly regulate fat storage and glucose production. Fat accumulates because the signals to burn it are blocked. GLP-1 receptor activation promotes phosphorylation of AKT and other molecules downstream of IRS-2, restoring the insulin signaling pathway. When insulin signaling works properly again, liver cells reduce triglyceride storage and the cascade toward fatty liver slows or reverses.
De novo lipogenesis reduction
Your liver constantly converts excess carbohydrates and calories into new fat through a process called de novo lipogenesis. In people with insulin resistance and obesity, this process runs in overdrive, flooding liver cells with fat they cannot export fast enough. Semaglutide and other GLP-1 agonists dial down de novo lipogenesis, reducing the rate at which new fat is manufactured. Less new fat production combined with increased fat oxidation creates a net negative fat balance in the liver, draining the excess that drives inflammation and enzyme elevation.
Enhanced fatty acid oxidation
While reducing fat production, GLP-1 agonists simultaneously increase the rate at which existing liver fat is burned for energy. This dual mechanism, less production plus more burning, is why the effects on liver fat can be so dramatic. Some imaging studies show liver fat reductions of 30-50% within the first six months of treatment.
Autophagy enhancement
Autophagy is the cellular self-cleaning process. Damaged organelles, misfolded proteins, and accumulated lipid droplets get tagged for destruction and recycling. In NAFLD, autophagy is impaired, allowing damaged components to accumulate and drive further inflammation. Research shows that GLP-1 receptor activation enhances autophagy in liver cells, helping them clear the cellular debris that contributes to enzyme elevation and disease progression.
Endoplasmic reticulum stress reduction
The endoplasmic reticulum is a cellular factory where proteins are folded and processed. In fatty liver disease, the ER becomes overwhelmed and stressed, triggering cell death pathways that release liver enzymes into the bloodstream. Treatment with GLP-1 agonists reduces ER stress-related apoptosis in human hepatocytes, keeping liver cells alive longer and reducing enzyme leakage.
Anti-inflammatory pathway modulation
GLP-1 agonists interact with multiple inflammatory pathways relevant to liver disease. They reduce NF-kB activation, a master switch for inflammatory gene expression. They decrease TNF-alpha and IL-6 production. They modulate the FXR pathway, which controls bile acid metabolism and has direct anti-inflammatory effects in the liver. These combined anti-inflammatory actions explain why semaglutide improves not just fat content but also the inflammatory component of liver disease.
Oral vs injectable semaglutide: does the delivery method matter for liver effects?
Semaglutide comes in two forms: oral tablets (Rybelsus) and subcutaneous injections (Ozempic, Wegovy). Does the route of administration affect liver outcomes?
Oral semaglutide passes through the gastrointestinal tract and undergoes first-pass metabolism in the liver before reaching systemic circulation. This means the liver sees higher local concentrations of the drug compared to the injectable form. Theoretically, this could provide enhanced direct hepatoprotective effects. A pilot study published in JGH Open examined oral semaglutide in patients with NAFLD and type 2 diabetes, finding improvements in hepatic steatosis and liver enzymes consistent with what the injectable trials demonstrated.
In practice, the liver outcomes appear similar between oral and injectable forms at equivalent effective doses. The major studies demonstrating NASH resolution and liver enzyme improvement used injectable semaglutide, so the evidence base is stronger on that side. However, nothing in the pharmacology suggests the oral form would be inferior for liver effects.
For patients specifically targeting liver health improvement, injectable semaglutide at higher doses (2.4mg weekly for Wegovy) currently has the most robust evidence and the FDA approval for liver fibrosis. But oral semaglutide remains a reasonable option for those who prefer pills over injections, particularly at the higher 14mg daily dose.
Lifestyle factors that maximize liver enzyme improvement on semaglutide
Semaglutide is powerful, but it works best when combined with lifestyle modifications that support liver health. The medication does heavy lifting, but you can amplify the results significantly.
Diet modifications for liver health
The Mediterranean diet has the strongest evidence base for NAFLD improvement, and it pairs perfectly with semaglutide. Emphasize olive oil, fatty fish, nuts, vegetables, and whole grains. Minimize added sugars and refined carbohydrates, which drive de novo lipogenesis in the liver.
Fructose deserves special attention. High-fructose corn syrup and excessive fruit juice consumption directly promote liver fat accumulation through metabolic pathways that bypass normal caloric regulation. Reducing fructose intake while on semaglutide can accelerate liver fat reduction beyond what either intervention achieves alone.
Coffee, interestingly, has consistent evidence for hepatoprotective effects. Multiple large studies show that regular coffee consumption reduces liver enzyme levels, decreases fibrosis progression, and lowers hepatocellular carcinoma risk. Drinking 2-3 cups of coffee daily while on semaglutide may provide additive liver benefits.
Exercise for liver fat reduction
Both aerobic exercise and resistance training reduce liver fat independently of weight loss. Aerobic exercise at moderate intensity for 150 minutes per week has the most evidence. Resistance training 2-3 times weekly adds further benefit. The combination of exercise plus semaglutide plus dietary modification creates a triple threat against liver fat that no single intervention can match.
Even modest physical activity helps. Walking 30 minutes daily reduces liver fat content by measurable amounts within weeks. You do not need extreme exercise protocols. Consistency matters more than intensity for metabolic health improvement.
Alcohol reduction or elimination
If liver health is a primary goal, reducing alcohol is one of the most impactful changes you can make. Even moderate alcohol consumption adds stress to liver cells that are already dealing with metabolic overload. Many semaglutide users report naturally reduced desire for alcohol, making this an easier transition than expected.
For someone with diagnosed NAFLD or NASH, complete alcohol elimination is the recommendation from most hepatology guidelines, regardless of whether the liver disease is classified as "nonalcoholic." Any alcohol adds fuel to the inflammatory fire that drives disease progression.
Sleep and stress management
Poor sleep and chronic stress both elevate cortisol, which promotes liver fat accumulation and insulin resistance. Optimizing sleep quality (7-9 hours nightly, consistent schedule) and managing stress through evidence-based approaches supports the liver improvements semaglutide provides. These factors are frequently overlooked in liver health discussions but meaningfully impact enzyme levels.
When to worry: red flags that demand immediate attention
Most liver enzyme changes on semaglutide are positive. But certain scenarios require urgent medical evaluation. Know these red flags.
Stop semaglutide and seek immediate care if you experience:
Jaundice (yellowing of skin or eyes): this indicates bilirubin is accumulating because the liver cannot process it properly. Always urgent.
Dark urine (cola or tea-colored): suggests bilirubin is being excreted through the kidneys because the liver is failing to handle it. Combined with other symptoms, this is an emergency.
Persistent right upper quadrant pain: the liver sits under your right ribs. Pain here that does not resolve with simple measures suggests liver inflammation or swelling severe enough to stretch the liver capsule.
Unexplained severe fatigue (beyond normal semaglutide fatigue): liver dysfunction causes a distinctive exhaustion that differs from normal tiredness. If you are sleeping adequately but can barely function, liver involvement should be investigated.
ALT or AST above 5x the upper limit of normal: this level of elevation warrants medication discontinuation and workup regardless of symptoms.
Any elevation plus symptoms: even a moderate enzyme bump (2-3x ULN) combined with nausea, loss of appetite, or malaise should prompt evaluation.
What your doctor should investigate
If liver enzymes rise significantly on semaglutide, the workup should include:
Complete hepatitis panel: hepatitis B surface antigen, hepatitis C antibody, hepatitis A IgM
Autoimmune markers: ANA, anti-smooth muscle antibody, immunoglobulin levels
Metabolic workup: ferritin (for hemochromatosis), ceruloplasmin (for Wilson disease), alpha-1 antitrypsin
Imaging: right upper quadrant ultrasound at minimum, with consideration for MRI or FibroScan
Medication review: every prescription, over-the-counter, and supplement should be scrutinized
Alcohol history: honest assessment of consumption patterns
Semaglutide-related DILI is a diagnosis of exclusion. It should only be attributed to the medication after other causes have been thoroughly ruled out.
Compounded semaglutide and liver safety considerations
The rise of compounded semaglutide adds another dimension to the liver enzyme conversation. Compounded versions are not identical to brand-name products. They may contain different salts (semaglutide sodium vs. semaglutide base), different concentrations, and different excipients.
From a liver safety perspective, several considerations apply to compounded formulations:
Purity and potency variability. Compounding pharmacies operate under different regulatory standards than pharmaceutical manufacturers. Variability in actual semaglutide content could mean patients receive inconsistent doses, complicating the interpretation of liver enzyme trends. If enzymes fluctuate unexpectedly on compounded semaglutide, dose inconsistency is a possible explanation.
Additional ingredients. Some compounded formulations include vitamin B12, glycine, niacinamide, L-carnitine, or other additives. Each of these has its own (generally benign) liver profile, but the combination could theoretically produce different enzyme patterns than pure semaglutide alone.
Lack of specific liver data. The clinical trials that demonstrated semaglutide liver benefits used brand-name pharmaceutical-grade product. These results cannot be automatically extrapolated to compounded versions, though the active ingredient mechanism would be expected to produce similar effects.
If you are using compounded semaglutide and concerned about liver health, the monitoring protocol should be the same as or more vigilant than what you would follow with brand-name product. Consider choosing established compounding pharmacies with strong quality control records.
Special populations and liver enzyme considerations
People with type 2 diabetes
Type 2 diabetes and NAFLD share a bidirectional relationship. Insulin resistance drives both conditions, and each worsens the other. Semaglutide addresses both simultaneously, making it a particularly valuable medication for this population. The liver enzyme improvements seen in diabetic patients on semaglutide tend to be among the most pronounced, reflecting improvements in both glycemic control and hepatic fat content.
Diabetes medications like pioglitazone and metformin can also affect liver enzymes. When interpreting bloodwork in diabetic patients on semaglutide, consider the full medication picture.
People with obesity
Obesity is the strongest risk factor for NAFLD. Weight loss through any mechanism improves liver enzymes. Semaglutide provides both the weight loss stimulus and direct hepatoprotection, creating a compound benefit. For people with BMI above 35 and known fatty liver, the liver argument for semaglutide treatment is compelling even independently of the weight loss benefits.
People over 65
Aging affects liver metabolism and drug clearance. Older adults may have a higher baseline level of liver enzyme variability. While semaglutide has been studied in older populations and shown acceptable safety profiles, liver enzyme monitoring should be somewhat more vigilant in patients over 65, particularly those on multiple medications.
People with alcohol use history
A history of heavy alcohol use, even if currently abstinent, means the liver may have pre-existing damage that complicates interpretation of enzyme changes on semaglutide. Baseline imaging to assess for fibrosis or cirrhosis is particularly important in this population. Semaglutide can still provide liver benefits, but the starting point and monitoring expectations differ from someone with a clean hepatic slate.
Frequently asked questions
Does semaglutide cause liver damage?
In the vast majority of cases, semaglutide improves liver health rather than harming it. Clinical trials consistently show reduced ALT and AST levels with treatment. However, rare cases of drug-induced liver injury have been reported, affecting roughly 1 in several hundred thousand users. Proper monitoring with baseline and periodic liver function tests catches any problems early.
Should I get liver function tests before starting semaglutide?
Yes. Baseline liver function tests are strongly recommended before starting semaglutide. These provide a reference point against which future values can be compared. If baseline values are already significantly elevated, your prescriber should investigate the cause before beginning treatment.
My ALT went up slightly after starting semaglutide. Should I stop?
Not necessarily. Mild, transient ALT elevations during dose escalation are common and usually resolve within weeks. If the elevation stays below 3 times the upper limit of normal and you have no symptoms (jaundice, dark urine, abdominal pain), continued monitoring with repeat testing in 2-4 weeks is typically appropriate. Discuss the specific numbers with your prescriber for personalized guidance.
Can semaglutide help reverse fatty liver disease?
Strong evidence supports semaglutide for reversing NAFLD and NASH. A landmark NEJM trial showed 59% NASH resolution at the 0.4mg daily dose versus 17% with placebo. The FDA has approved semaglutide for liver fibrosis caused by NAFLD, making it one of the first medications with this specific indication. Benefits include reduced liver fat, improved inflammation markers, and normalized liver enzymes over time.
How often should liver enzymes be monitored on semaglutide?
Check baseline before starting, at each dose escalation (typically monthly for the first 4 months), then every 3-6 months during maintenance. People with pre-existing liver disease or elevated baseline enzymes should be monitored more frequently, typically every 2-3 months. Any new symptoms should prompt immediate retesting.
Does compounded semaglutide affect the liver differently than brand name?
The active ingredient is the same, so the mechanism of liver protection should be similar. However, compounded formulations may contain additional ingredients like methylcobalamin or glycine, and purity can vary between pharmacies. No specific clinical trials have evaluated compounded semaglutide liver effects. Following the same or more vigilant monitoring protocol as brand-name product is prudent.
Is semaglutide safe for people with existing liver disease?
For NAFLD and NASH specifically, semaglutide is not just safe but potentially therapeutic. For other types of liver disease (autoimmune hepatitis, viral hepatitis, cirrhosis from alcohol), the data is more limited. Patients with advanced cirrhosis (Child-Pugh B or C) were generally excluded from clinical trials. Consult a hepatologist before starting semaglutide if you have significant liver disease beyond simple metabolic fatty liver.
Will my liver enzymes normalize completely on semaglutide?
Many patients with mildly elevated liver enzymes at baseline see complete normalization within 6-12 months of treatment, particularly those achieving significant weight loss. Patients with more advanced NAFLD or NASH may see substantial improvement without complete normalization. The degree of improvement correlates with weight loss achieved and duration of treatment.
External resources
Effect of semaglutide on liver enzymes and markers of inflammation (PMC)
Subcutaneous semaglutide in nonalcoholic steatohepatitis (NEJM)
GLP-1 receptor agonists in NAFLD: evidence and perspectives (PMC)
For researchers serious about understanding how their protocols affect liver health and metabolic function, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, dosing calculators, proven protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your liver enzymes stay normal, your metabolic health stay improving, and your treatment decisions stay informed.