Feb 28, 2026

What if your doctor could tell you were taking semaglutide just by glancing at your blood work? Would your routine labs give you away? And more importantly, should you even care?
These questions keep popping up across forums, clinic waiting rooms, and late-night search sessions. Thousands of people on semaglutide want to know exactly what their blood work will show, what it will not show, and whether their healthcare provider can spot the medication without being told. The answer is more nuanced than a simple yes or no. Standard blood tests are not designed to detect semaglutide directly. But the medication leaves fingerprints everywhere, from your glucose markers to your liver enzymes to your lipid panel. Your labs will not name the drug, yet they will tell a story about what it is doing inside your body.
This guide breaks down every blood test relevant to semaglutide use. You will learn which panels detect the drug itself, which ones reveal its effects, what changes to expect at each stage of treatment, and exactly which markers your doctor monitors to keep you safe. Whether you are weeks into your first dose or months into a stable protocol, understanding your lab work transforms you from a passive patient into someone who can actually read the data and make informed decisions about your health.
For researchers and individuals tracking their progress with precision, SeekPeptides provides comprehensive guides to understanding peptide protocols and their measurable effects on your body.
The short answer: semaglutide does not show up in routine blood work
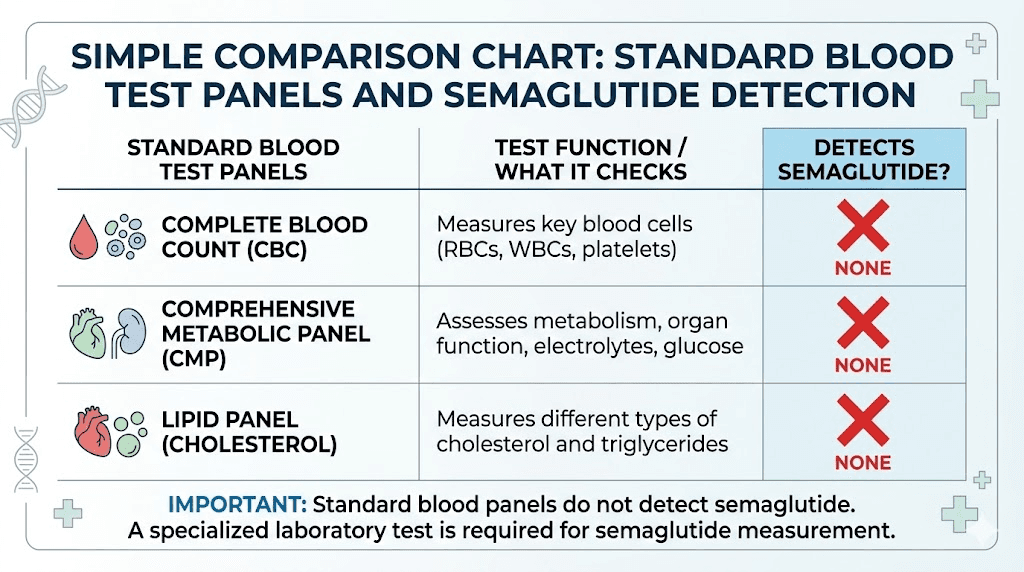
Here is the direct answer. No. Semaglutide does not appear in standard blood tests. Not in your complete blood count. Not in your basic metabolic panel. Not in your lipid profile. Not in your liver function tests. These panels measure specific biomarkers, things like red blood cell counts, electrolyte levels, cholesterol fractions, and enzyme activity. None of them are designed to detect GLP-1 receptor agonists circulating in your bloodstream.
Your doctor cannot look at a standard lab report and determine whether you are taking semaglutide, tirzepatide, or any other GLP-1 medication.
The reason is straightforward. Routine blood panels use immunoassay technology calibrated to detect specific substances, typically things like glucose, creatinine, thyroid hormones, and standard drug metabolites. Semaglutide is a modified peptide. Its molecular structure does not trigger any of the antibody-antigen reactions that standard immunoassays rely on. It is invisible to these tests by design, not by accident.
Think of it this way. Your routine blood work is like a security checkpoint looking for specific items on a list. Semaglutide is simply not on that list. The checkpoint does not recognize it, does not flag it, and does not report it. The medication passes through without a trace on the standard report.
What about drug tests and employment screening?
This deserves its own section because the anxiety around it is real. Semaglutide does not show up on standard drug screening panels. Period. Not on 5-panel tests. Not on 10-panel tests. Not on extended panels used for employment, legal proceedings, or probation monitoring.
Standard drug screenings test for substances of abuse: opioids, amphetamines, benzodiazepines, cannabinoids, cocaine, and sometimes barbiturates, methadone, or PCP. Semaglutide is a prescription peptide medication with zero abuse potential. The Drug Enforcement Administration does not classify it as a controlled substance. Its chemical structure, a modified GLP-1 peptide analog, bears absolutely no resemblance to any of the substances these panels screen for. You can learn more about how peptides interact with drug screening in our guide on whether peptides show up on drug tests.
Even confirmatory testing methods like gas chromatography-mass spectrometry (GC-MS) and standard liquid chromatography-mass spectrometry (LC-MS) will not detect semaglutide unless they are specifically programmed to look for it. And in routine drug testing contexts, they never are.
One exception exists. Competitive sports organizations may use specialized testing that can detect GLP-1 receptor agonists. The World Anti-Doping Agency (WADA) and certain athletic governing bodies have access to high-resolution, high-accuracy mass spectrometry (LC-HRMS) methods specifically designed to identify peptide hormones, including semaglutide. A study published in the journal Clinica Chimica Acta validated an LC-HRMS method capable of detecting semaglutide in whole blood samples. But this type of testing requires specialized equipment, trained personnel, and deliberate intent to screen for GLP-1 agonists. It is not something your employer, your doctor, or your insurance company will ever run.
Specialized tests that can detect semaglutide directly
While routine labs cannot find semaglutide, specialized assays can. If someone truly needed to confirm whether semaglutide was present in your blood, the technology exists.
The gold standard for direct semaglutide detection is liquid chromatography coupled with high-resolution mass spectrometry (LC-HRMS). This method isolates and identifies specific peptide molecules in blood samples with extraordinary precision. Research published in peer-reviewed journals has validated these methods for detecting semaglutide at therapeutic concentrations.
Here is what you need to know about these specialized tests.
They are expensive. A single LC-HRMS analysis can cost several hundred dollars or more, far exceeding the cost of a standard metabolic panel. They require specialized laboratory equipment that most clinical labs do not possess. They are not available through standard lab ordering portals like Quest Diagnostics or Labcorp for routine patient use. And they serve almost no clinical purpose for individual patient care.
In practical terms, these tests exist primarily for three purposes: anti-doping enforcement in competitive sports, forensic toxicology investigations, and pharmaceutical research. Your endocrinologist, your primary care doctor, and your weight management clinic have no reason to order them and no easy way to do so even if they wanted to. Understanding the difference between research-grade and pharmaceutical peptide testing helps put this in perspective.
The half-life of semaglutide is approximately 7 days, which means it remains detectable in the bloodstream for an extended period after each dose. For someone taking weekly injections, the drug would theoretically be detectable at any point during active treatment. After discontinuation, clearance takes approximately 5 weeks (roughly 5 half-lives) before levels drop below detectable thresholds.
How semaglutide changes your blood work: the markers that tell the story
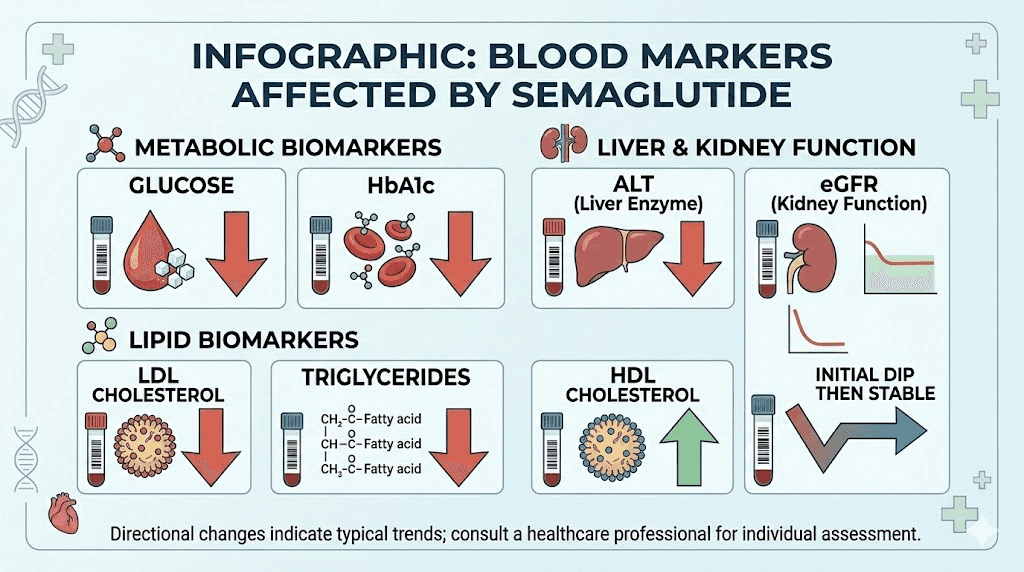
Here is where it gets interesting. While semaglutide itself is invisible to routine labs, its effects are not. The medication creates measurable, sometimes dramatic changes across multiple blood markers. An experienced clinician looking at your lab trends over time could reasonably suspect GLP-1 therapy based on the pattern of changes alone.
Think of it as footprints in the snow. You cannot see the person who walked through, but you can follow exactly where they went. Semaglutide leaves clear footprints across your metabolic panel, your lipid profile, your liver enzymes, and more.
HbA1c and blood glucose: the most dramatic shift
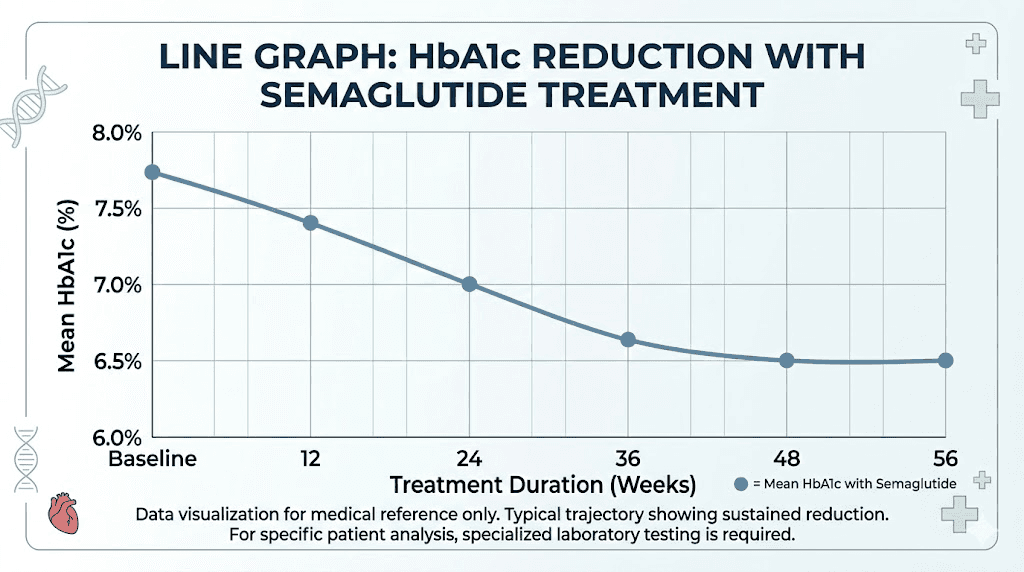
This is the most significant and well-documented change semaglutide produces in blood work. HbA1c, or glycated hemoglobin, measures your average blood sugar over the preceding 2-3 months. It is the single most important marker for glucose control.
In the SUSTAIN clinical trial program, semaglutide at the 1.0 mg weekly dose reduced HbA1c by 1.5% to 1.8% over 30 to 56 weeks of treatment. To put that in perspective, dropping from an HbA1c of 8.0% to 6.2% represents a transformation from poorly controlled diabetes to near-normal glucose regulation. The SUSTAIN 7 trial showed average reductions of 1.8% at the 1.0 mg dose compared to 1.4% with dulaglutide at its highest dose. The SUSTAIN 10 trial demonstrated a 1.7% reduction at week 30.
Fasting glucose drops as well, typically within the first few weeks of treatment. If you were tracking how long semaglutide takes to produce effects, glucose changes often appear before appetite suppression or weight loss become obvious.
For individuals without diabetes using semaglutide for weight management, the glucose changes are less dramatic but still measurable. Fasting glucose typically drops into the lower end of the normal range. HbA1c may decrease by 0.3% to 0.5%, moving from high-normal toward optimal levels. These shifts are clinically meaningful even when starting from a normal baseline.
Your doctor will see these changes clearly. A sudden, sustained improvement in glucose markers without corresponding changes in diet or exercise habits is one of the clearest laboratory signatures of GLP-1 therapy. If your first week on semaglutide or subsequent weeks are producing results, your blood work will confirm it.
Lipid panel: cholesterol and triglyceride improvements
Semaglutide produces consistent, favorable changes in lipid profiles. A systematic review and meta-analysis of randomized controlled trials in non-diabetic adults with overweight or obesity found the following changes.
Total cholesterol decreased by an average of 6.39 mg/dL. That number might not sound enormous on its own, but combined with other lipid improvements, the cardiovascular impact is meaningful.
LDL cholesterol, the so-called bad cholesterol, dropped by an average of 6.01 mg/dL. For someone with borderline-high LDL, this shift could move them from a risk category that warrants medication consideration to one that does not.
Triglycerides showed the most dramatic improvement, declining by an average of 14.75 mg/dL. Some studies showed even more impressive reductions. Fasting triglyceride concentrations were 12% lower with semaglutide compared to placebo in controlled settings, with VLDL concentrations dropping by 21%. High triglycerides represent one of the most common lipid abnormalities in people with obesity and insulin resistance, and this reduction addresses a genuine cardiovascular risk factor.
HDL cholesterol, the protective form, increased by an average of 1.82 mg/dL. While modest, this improvement was sustained at both 26-week and 52-week assessments. Understanding these changes helps explain why semaglutide demonstrated cardiovascular benefits in the SELECT trial. People tracking their progress with tools like the semaglutide dosage calculator should know that proper dosing correlates with these metabolic improvements.
What makes these lipid changes interesting from a detection standpoint is that they appear partially independent of weight loss. Semaglutide has direct effects on lipid metabolism beyond what you would expect from weight reduction alone. This means even individuals who have not lost significant weight may show improved lipid panels, a pattern that could suggest pharmacological intervention to an observant clinician.
Liver enzymes: ALT, AST, and GGT changes
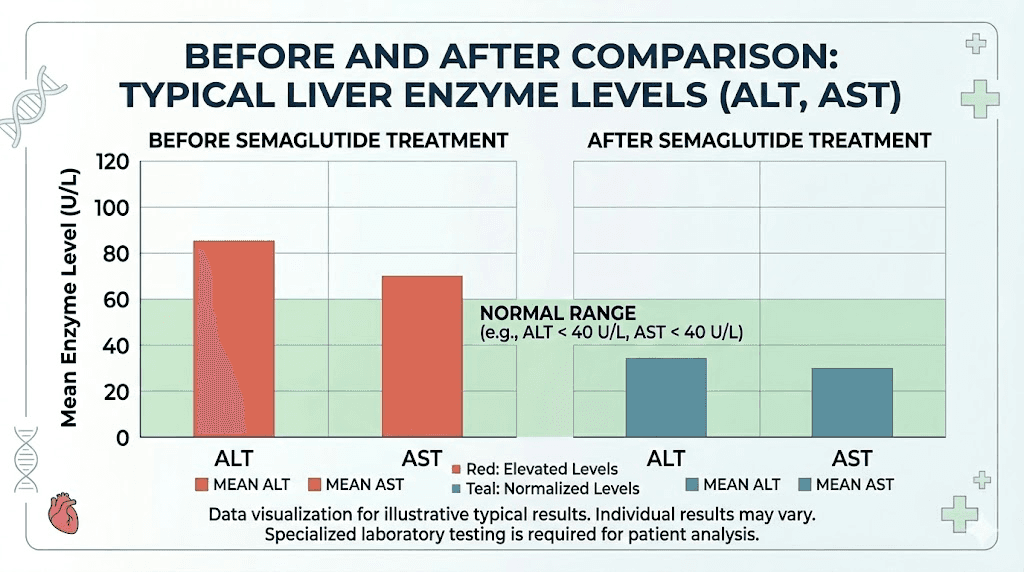
Liver enzyme changes on semaglutide are clinically significant and worth understanding in detail. The two primary markers, alanine aminotransferase (ALT) and aspartate aminotransferase (AST), both tend to improve during treatment.
In weight management trials, semaglutide reduced ALT levels by 6% to 21% compared to placebo in participants who had elevated baseline values. Even more impressively, a pooled analysis showed that 25% to 46% of subjects with elevated baseline ALT achieved normalization by week 52, compared to only 18% of those receiving placebo. This is a substantial difference.
The improvement in liver enzymes reflects semaglutide reducing hepatic fat content. Fatty liver disease, technically called metabolic dysfunction-associated steatotic liver disease (MASLD), is extremely common in people with obesity. Semaglutide addresses it through multiple mechanisms: reduced caloric intake, improved insulin sensitivity, decreased de novo lipogenesis, and direct hepatoprotective effects.
In fact, the evidence became so compelling that in August of last year, the FDA approved semaglutide (Wegovy) for the treatment of non-cirrhotic metabolic dysfunction-associated steatohepatitis (MASH) with moderate to advanced liver fibrosis, based on the phase 3 ESSENCE trial. This trial demonstrated superior improvement in liver fibrosis with no worsening of steatohepatitis, along with improvements in ALT, AST, and GGT.
There is one important caveat. Rare cases of semaglutide-associated liver injury have been reported. One documented case involved a 44-year-old woman who developed elevated liver enzymes after starting semaglutide, with levels normalizing after discontinuation and worsening upon rechallenge. These cases are exceedingly rare, but they underscore why liver enzyme monitoring during treatment is standard practice. If you are experiencing semaglutide fatigue, elevated liver enzymes could be one contributing factor worth investigating.
Kidney function markers: eGFR and creatinine
Kidney function markers deserve careful attention because semaglutide produces a pattern that can initially look alarming but is actually expected and, in the long run, beneficial.
During the first 12 to 16 weeks of treatment, many people experience a small, transient decline in estimated glomerular filtration rate (eGFR), which corresponds to a slight increase in serum creatinine. In the SELECT trial, this initial eGFR dip reached its lowest point (nadir) at approximately 8 weeks after starting semaglutide. By week 20, eGFR levels were similar between the semaglutide and placebo groups.
This early dip is believed to reflect hemodynamic changes rather than actual kidney damage. Semaglutide reduces blood pressure and alters renal blood flow dynamics during the adjustment period. It is analogous to the initial eGFR dip seen with ACE inhibitors or ARBs, medications widely recognized as kidney-protective despite causing a transient decline in filtration rate.
The long-term kidney data is reassuring. The landmark FLOW trial demonstrated that semaglutide reduced the risk of significant kidney events by 24% compared to placebo in patients with type 2 diabetes and chronic kidney disease. The mean annual eGFR slope, which measures how quickly kidney function declines over time, was 1.16 mL/min/1.73m2 per year less steep in the semaglutide group. This means semaglutide actually slowed the progression of kidney disease.
If your blood work shows a slight creatinine increase or eGFR decrease in the first few months of semaglutide, do not panic. But do make sure your doctor is aware you are taking the medication so they can interpret the results in context. Dehydration from gastrointestinal side effects like nausea or vomiting can exacerbate kidney stress, so staying hydrated is especially important during the dose-escalation phase. Knowing how long semaglutide takes to work helps set expectations for when these initial adjustments resolve.
Thyroid markers: TSH and calcitonin
Semaglutide carries a boxed warning about thyroid C-cell tumors based on rodent studies. This has created significant anxiety about thyroid blood work, so let us separate fact from fear.
In animal studies, rats exposed to GLP-1 receptor agonists at high doses over their lifetimes developed C-cell tumors, including medullary thyroid carcinoma (MTC). However, human thyroid tissue has significantly fewer GLP-1 receptors on C-cells compared to rodent thyroid tissue, which likely explains why this risk has not materialized in human clinical data.
In clinical trials involving semaglutide, the incidence of thyroid cancer in treated patients was less than 1%, and no statistically significant increase compared to placebo was established. A consistent, clinically meaningful elevation in calcitonin (the marker for C-cell activity) has not been demonstrated with GLP-1 receptor agonist use in humans.
Current FDA guidance does not recommend routine calcitonin monitoring in patients using GLP-1 medications unless they have specific risk factors. Those risk factors include a personal or family history of medullary thyroid cancer and a diagnosis of multiple endocrine neoplasia syndrome type 2 (MEN2). Routine neck ultrasounds or calcitonin testing are not recommended for the general semaglutide-using population.
TSH (thyroid-stimulating hormone) testing, which is part of many standard wellness panels, is generally unaffected by semaglutide. The medication does not alter thyroid hormone production or regulation in a clinically significant way. If your TSH was normal before starting semaglutide, it should remain normal during treatment. Any changes in TSH would more likely reflect an underlying thyroid condition unrelated to the medication.
Pancreatic enzymes: amylase and lipase
Pancreatic enzyme levels are relevant because of the known (though rare) association between GLP-1 receptor agonists and acute pancreatitis. Understanding how semaglutide affects amylase and lipase helps you interpret these results without unnecessary alarm.
Studies on GLP-1 receptor agonists show modest increases in both amylase and lipase during treatment. Data from liraglutide trials (a closely related GLP-1 agonist) demonstrated approximately a 7% increase in amylase and a 28% to 31% increase in lipase compared to placebo. These increases are believed to reflect adaptive growth of pancreatic tissue in response to GLP-1 receptor activation rather than inflammatory damage.
Crucially, the positive predictive value of elevated amylase and lipase for actual pancreatitis was less than 1%. In other words, having slightly elevated pancreatic enzymes while on a GLP-1 agonist almost never means you are developing pancreatitis. The enzymes returned to baseline after medication discontinuation, confirming the elevations were reversible and non-pathological in the vast majority of cases.
A meta-analysis of randomized clinical trials specifically evaluating semaglutide did not find an increased risk of acute pancreatitis compared to placebo. However, individual case reports do exist documenting semaglutide-associated pancreatitis, sometimes with dramatically elevated lipase levels (above 3,000 U/L in some cases). These events are rare and more common in patients with pre-existing risk factors: prior history of pancreatitis, high triglycerides, heavy alcohol use, or genetic predisposition.
Current guidelines do not recommend routine amylase and lipase monitoring for all semaglutide users. However, if you develop severe abdominal pain, especially pain radiating to the back accompanied by nausea and vomiting, these enzymes should be checked immediately. Knowing the difference between common GI side effects like bloating and symptoms that warrant urgent evaluation is important for every semaglutide user.
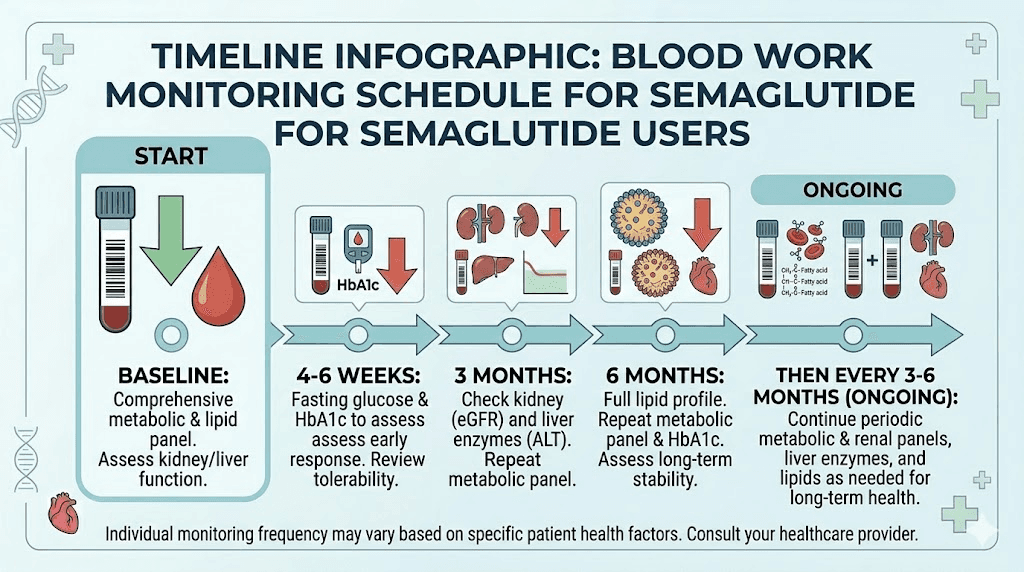
Complete blood work monitoring schedule for semaglutide users
Knowing which tests your doctor should order, and when, gives you the ability to advocate for your own care. Here is the evidence-based monitoring schedule recommended for semaglutide therapy.
Before starting semaglutide (baseline labs)
A comprehensive baseline blood panel establishes your starting point. Without these numbers, neither you nor your doctor can measure progress or identify problems. Most clinics require blood work completed within 90 days of starting treatment.
The essential baseline panel should include:
Complete metabolic panel (CMP) covering glucose, electrolytes (sodium, potassium, chloride, bicarbonate), BUN, creatinine, eGFR, calcium, albumin, total protein, and liver enzymes (ALT, AST, alkaline phosphatase, bilirubin). This panel establishes your kidney function baseline, liver health, electrolyte balance, and fasting glucose level.
HbA1c provides your 2-3 month glucose average. Even if you do not have diabetes, this establishes a critical baseline for tracking metabolic improvement. People exploring whether semaglutide suppresses appetite immediately should know that glucose improvements often precede noticeable appetite changes.
Lipid panel including total cholesterol, LDL, HDL, triglycerides, and ideally VLDL. This captures your cardiovascular risk baseline. Given that semaglutide consistently improves lipid markers, having accurate starting numbers lets you quantify the benefit.
Complete blood count (CBC) measures red blood cells, white blood cells, hemoglobin, hematocrit, and platelets. While semaglutide does not directly alter these values significantly, a baseline CBC can identify anemia or other blood disorders that might complicate treatment.
TSH checks thyroid function. While semaglutide does not typically affect thyroid hormones, baseline thyroid assessment is prudent given the boxed warning. Undiagnosed hypothyroidism can also affect weight and metabolism, potentially confounding your response to treatment.
Urinalysis and urine albumin-to-creatinine ratio (UACR) assess kidney health more comprehensively than blood tests alone. The UACR detects early kidney damage (microalbuminuria) that might not yet affect serum creatinine or eGFR. This is especially important for people with diabetes or hypertension. If you are following a specific semaglutide diet plan, adequate hydration is critical for kidney health.
First 3-6 months: close monitoring phase
The dose-escalation period is when most side effects emerge and when your body adjusts to the medication. Blood work during this phase should be more frequent than during maintenance.
At the 4-6 week mark, a focused panel makes sense. Check a basic metabolic panel (BMP) to assess kidney function, since the initial eGFR dip typically occurs around week 8. Checking at 4-6 weeks provides an early data point. If creatinine is climbing significantly or you are experiencing dizziness or signs of dehydration, intervention can happen before the dip reaches its nadir.
At the 3-month mark, a more comprehensive panel is warranted. This is your first HbA1c recheck. You should see a meaningful decline. Include the full CMP, lipid panel, and HbA1c. This visit is also a good time to assess whether your early results are tracking with expectations.
At the 6-month mark, repeat the comprehensive panel. By now, you should be at or near your maintenance dose. HbA1c improvements should be approaching their maximum effect. Lipid changes should be well-established. Liver enzymes should be stable or improved. Kidney function should have recovered from any initial dip. This set of labs establishes your new metabolic baseline on treatment. People wondering how long they will stay on semaglutide should use these 6-month results to inform that discussion with their provider.
Ongoing maintenance: every 3-6 months
Once you are established on a stable dose and your labs are trending favorably, routine monitoring can space out. Most clinicians recommend comprehensive blood work every 3 to 6 months during ongoing treatment.
The maintenance panel should include HbA1c (every 3-6 months), CMP with liver enzymes (every 6 months), lipid panel (every 6-12 months), and TSH (annually). Additional testing may be indicated based on individual circumstances, comorbidities, or symptoms that emerge during treatment.
If you are using compounded semaglutide, monitoring may need to be slightly more frequent since compounded products have less standardized quality control than branded pharmaceuticals. Understanding compounded semaglutide considerations helps you work with your provider to set appropriate monitoring intervals.
What your doctor sees when comparing blood work before and after semaglutide
Let us put real numbers to this. Here is what a typical set of lab results might look like for someone who has been on semaglutide 1.0 mg weekly for 6 months, assuming they started with common metabolic abnormalities associated with obesity.
Marker | Before semaglutide | After 6 months | Change |
|---|---|---|---|
HbA1c | 7.2% | 5.8% | -1.4% |
Fasting glucose | 132 mg/dL | 96 mg/dL | -36 mg/dL |
Total cholesterol | 228 mg/dL | 209 mg/dL | -19 mg/dL |
LDL cholesterol | 148 mg/dL | 131 mg/dL | -17 mg/dL |
HDL cholesterol | 38 mg/dL | 43 mg/dL | +5 mg/dL |
Triglycerides | 196 mg/dL | 142 mg/dL | -54 mg/dL |
ALT | 52 U/L | 34 U/L | -18 U/L |
AST | 38 U/L | 28 U/L | -10 U/L |
Creatinine | 0.9 mg/dL | 0.9 mg/dL | No change |
eGFR | 92 mL/min | 94 mL/min | +2 mL/min |
TSH | 2.1 mIU/L | 2.3 mIU/L | No meaningful change |
Look at that table carefully. An experienced clinician seeing these changes in a patient who also lost 15-20% of their body weight would strongly suspect pharmacological intervention. The combination of dramatic glucose improvement, lipid optimization, and liver enzyme normalization in a 6-month window tells a clear story, even though semaglutide itself never appears by name on the lab report.
For researchers tracking their own data, the GLP-1 plotter tool can help visualize trends over time. And using the peptide calculator ensures your dosing stays accurate, which directly affects how consistently these improvements appear in your lab work.
Blood work changes that signal potential problems
Most blood work changes on semaglutide are positive. But some patterns warrant attention and potential intervention. Knowing what to watch for is just as important as understanding the expected improvements.
Significant creatinine elevation
As discussed, a small early increase in creatinine and corresponding eGFR dip is expected. But a creatinine increase exceeding 30% from baseline, or one that continues rising beyond the first 12-16 weeks, needs investigation. This could indicate dehydration (common with GI side effects), pre-existing kidney disease being unmasked, or genuine acute kidney injury.
Case reports of acute kidney injury associated with semaglutide exist, though they are rare and typically linked to severe dehydration from persistent nausea and vomiting during dose escalation. The fix is usually aggressive hydration and slower dose titration, not discontinuation. But the blood work signal cannot be ignored. Understanding what happens when you stop or adjust semaglutide is relevant if dose modification becomes necessary.
Dramatically elevated liver enzymes
Semaglutide typically improves liver enzymes. If your ALT or AST rises significantly during treatment, especially if it exceeds 3 times the upper limit of normal, this is abnormal and requires evaluation. While extremely rare, idiosyncratic drug-induced liver injury has been documented with semaglutide. Other causes (alcohol use, new medications, viral hepatitis, autoimmune conditions) should be investigated first, but the timing correlation with semaglutide initiation matters.
Severely elevated lipase with abdominal symptoms
As noted earlier, mild lipase elevation is common and benign on GLP-1 therapy. However, lipase levels exceeding 3 times the upper limit of normal, accompanied by severe epigastric pain radiating to the back, represent a potential pancreatitis emergency. This requires urgent evaluation regardless of whether the cause is semaglutide, gallstones, or another trigger.
People taking semaglutide who also consume alcohol should be particularly vigilant about this, as alcohol is an independent risk factor for pancreatitis that can compound any GLP-1-related risk.
Hypoglycemia markers
Semaglutide used alone rarely causes hypoglycemia because its insulin-stimulating effect is glucose-dependent. But when combined with sulfonylureas or insulin, the risk increases substantially. Fasting glucose levels consistently below 70 mg/dL, or symptoms like shakiness, confusion, and sweating, should prompt a blood glucose check and medication adjustment discussion.
If you are exploring combining semaglutide with other medications, understanding how each affects glucose metabolism helps prevent hypoglycemic episodes.
How semaglutide affects less common blood markers
Beyond the standard panels, semaglutide influences several markers that may appear on specialized or expanded blood work.
Inflammatory markers: CRP and ESR
Semaglutide demonstrates anti-inflammatory effects that show up in blood work. C-reactive protein (CRP), a general marker of systemic inflammation, tends to decrease during treatment. This reduction is partially attributable to weight loss (adipose tissue is metabolically active and produces inflammatory cytokines) and partially to direct anti-inflammatory effects of GLP-1 receptor activation.
If your doctor orders a high-sensitivity CRP (hs-CRP) as part of cardiovascular risk assessment, expect to see improvement on semaglutide. This contributes to the overall cardiovascular benefit demonstrated in the SELECT trial, where semaglutide reduced major adverse cardiovascular events by 20% compared to placebo. Understanding how different GLP-1 medications compare in their effects can help inform treatment decisions.
Insulin and C-peptide levels
Fasting insulin levels typically decrease on semaglutide, reflecting improved insulin sensitivity. As cells become more responsive to insulin, the pancreas does not need to produce as much to maintain glucose control. C-peptide, which is co-secreted with insulin and serves as a marker of endogenous insulin production, follows a similar pattern.
These changes are particularly relevant for people with insulin resistance or early type 2 diabetes. Decreasing fasting insulin levels, combined with improving glucose markers, suggests genuine metabolic improvement rather than simply masking the problem with more insulin production.
Uric acid levels
Semaglutide has been associated with modest decreases in serum uric acid levels. This is relevant because hyperuricemia is common in people with obesity and metabolic syndrome, and elevated uric acid is associated with gout and cardiovascular risk. The reduction is thought to result from improved renal uric acid excretion and decreased production related to metabolic improvements. People who experience joint pain on GLP-1 medications should have uric acid levels checked if gout is a possibility.
Vitamin and mineral levels
One area where semaglutide blood work effects are less discussed but clinically relevant is micronutrient status. Reduced food intake over months of treatment can lead to deficiencies. Markers to watch include:
Vitamin B12 levels may decrease over time, especially in people with reduced dietary intake or those combining semaglutide with metformin (which independently reduces B12 absorption). Some compounded formulations include B12 specifically to address this concern, which is why options like semaglutide with B12 and semaglutide with methylcobalamin have become popular.
Iron studies (ferritin, serum iron, TIBC) may shift if dietary iron intake drops significantly. Pre-menopausal women on semaglutide are at higher risk for iron deficiency due to combined effects of reduced intake and menstrual blood loss.
Vitamin D levels, already commonly low in people with obesity (vitamin D is sequestered in adipose tissue), may paradoxically improve as body fat decreases and more vitamin D becomes bioavailable. Or they may worsen if dietary intake drops. Monitoring makes sense.
Following a well-planned diet while on semaglutide helps prevent deficiencies. Guides on what foods to eat on semaglutide and foods to avoid can help maintain nutritional adequacy despite reduced appetite.
Semaglutide and specialized monitoring panels
Several commercial labs now offer semaglutide-specific monitoring panels that bundle relevant tests into a single order. Understanding what these include helps you evaluate whether they provide value.
A typical semaglutide monitoring panel includes HbA1c, comprehensive metabolic panel (glucose, electrolytes, kidney function, liver enzymes), lipid panel, thyroid function (TSH), and sometimes insulin and C-reactive protein. Some panels add vitamin B12, vitamin D, and iron studies.
These bundled panels are convenient but not strictly necessary if your doctor already orders the component tests individually. The advantage is completeness, ensuring nothing gets missed. The disadvantage is cost, as these specialty panels often carry a premium compared to ordering the same tests a la carte through a standard lab requisition.
For people self-managing their semaglutide protocol, particularly those using compounded semaglutide or following their own dosing schedule, these comprehensive panels provide accountability. You can order them through direct-to-consumer lab services without a doctor requisition in most states, though discussing results with a healthcare provider is always advisable.
The SeekPeptides platform offers resources to help members understand their lab results in the context of peptide protocols, providing detailed guides on what each marker means and how it relates to treatment efficacy and safety.
How to talk to your doctor about semaglutide and blood work
Whether you want your doctor to know about your semaglutide use or not, honest communication produces better outcomes. Here is a practical framework for navigating these conversations.
If you want your doctor to know
Disclosing semaglutide use to your healthcare provider allows them to interpret your lab results accurately. A sudden improvement in glucose markers, liver enzymes, and lipid profiles has a differential diagnosis. Your doctor could attribute these changes to diet modifications, exercise, a new supplement, or pharmacological intervention. Knowing you are on semaglutide simplifies interpretation and guides monitoring.
When disclosing, provide the specifics. What dose are you taking? How often? What brand or formulation? When did you start? Are you using a branded product (Ozempic, Wegovy) or a compounded version? This information helps your doctor assess quality, dosing appropriateness, and potential interactions with your other medications. Knowing how to qualify for semaglutide through proper medical channels can simplify this conversation.
If you prefer privacy
Some people take semaglutide without their primary care provider knowing, particularly if they obtained it through telehealth, a weight loss clinic, or an online pharmacy. While disclosure is medically ideal, it is your right to maintain privacy about your medications.
Be aware, however, that your doctor may notice the laboratory changes described throughout this article. A dramatic improvement in metabolic markers without a corresponding explanation (new diet, exercise program, other medication changes) may prompt questions. Having a response prepared, even if it is simply "I have made some changes to my health routine that I am not ready to discuss in detail," preserves your privacy while being honest.
What you should never do is lie about medications when asked directly, especially before procedures that require anesthesia, during pregnancy evaluations, or when other medications are being prescribed that could interact with semaglutide. The risks of non-disclosure in these contexts can be genuinely dangerous. Our guide on resuming semaglutide after surgery explains why surgical teams need to know about GLP-1 medications specifically.
Blood work differences between semaglutide formulations
Not all semaglutide formulations produce identical blood work changes. Understanding the differences helps you interpret your labs more accurately.
Branded injectable (Ozempic, Wegovy) versus compounded injectable
Branded products undergo rigorous quality control with precise dosing. Each injection delivers exactly the amount of semaglutide specified. This consistency means blood work changes should closely mirror what clinical trial data predicts.
Compounded semaglutide can vary in actual potency despite the labeled dose. Some compounded products have been found to contain more or less active ingredient than specified. This variability means your blood work changes might be less predictable. You might see stronger or weaker metabolic improvements than expected for your stated dose. Understanding how to reconstitute semaglutide properly and using accurate tools like the semaglutide dosage calculator helps minimize variability when working with compounded formulations.
Compounded products also sometimes include additional ingredients, such as B12, glycine, L-carnitine, or niacinamide. These additives can independently affect certain blood markers. B12 will obviously affect B12 levels. L-carnitine may affect lipid metabolism markers. Being aware of what is in your specific formulation helps explain any unexpected lab findings.
Oral versus injectable semaglutide
Oral semaglutide (Rybelsus) has lower bioavailability than injectable forms, meaning more of the drug is lost to first-pass metabolism in the gut and liver. Blood work changes on oral semaglutide tend to be less pronounced than on injectable forms at equivalent labeled doses. HbA1c reductions are typically in the 1.0% to 1.4% range with oral semaglutide versus 1.5% to 1.8% with injectable. Lipid improvements follow a similar pattern.
If you are using oral semaglutide drops or sublingual semaglutide, set your blood work expectations accordingly. Smaller changes do not necessarily mean the medication is not working. They reflect the pharmacokinetic reality of oral peptide delivery.
Semaglutide versus tirzepatide: blood work comparison
Since many people consider switching between these medications, understanding how their blood work effects differ is practical. Our detailed semaglutide versus tirzepatide comparison covers this comprehensively, but in terms of blood work specifically:
Tirzepatide tends to produce larger HbA1c reductions (up to 2.0% to 2.3% in the SURPASS trials) compared to semaglutide. Triglyceride improvements are also typically more pronounced with tirzepatide. Liver enzyme improvements appear similar between the two. Kidney function effects follow comparable patterns. If you are switching between these medications, expect some adjustment in your lab values during the transition. The conversion chart helps ensure dosing equivalence during switches.
Special situations: blood work considerations
Blood work during pregnancy
Semaglutide is contraindicated in pregnancy and should be discontinued at least 2 months before a planned pregnancy (given its 7-day half-life, approximately 5 weeks of clearance time is recommended). Blood work during this washout period should show a gradual return of glucose and lipid markers toward pre-treatment values.
Importantly, pregnancy blood work (gestational diabetes screening, prenatal panels) is not affected by prior semaglutide use once the drug is fully cleared. The metabolic improvements from weight loss may persist independent of the medication, which is actually beneficial for pregnancy outcomes. Resources on how semaglutide affects menstrual cycles are relevant for women of reproductive age.
Blood work during dose changes
When titrating up (increasing dose) or down (decreasing or discontinuing), blood work markers shift. During dose escalation, expect progressive improvement in metabolic markers at each dose level. The most dramatic changes typically occur during the first few dose increases.
During dose reduction or discontinuation, markers tend to drift back toward pre-treatment values over weeks to months. HbA1c rises, lipids may worsen, and liver enzymes may increase. This is not necessarily a "rebound" effect, it is simply the removal of the pharmacological intervention that was driving improvement. Understanding what happens at a semaglutide plateau helps differentiate between dose-related stalls and true treatment resistance.
Blood work and fasting requirements
Semaglutide does not change the standard fasting requirements for blood work. A 10-12 hour fast is still recommended for accurate fasting glucose, triglycerides, and certain other markers. Semaglutide can be taken as scheduled regardless of when your blood work is drawn, as the medication itself does not interfere with test accuracy. The timing of your weekly injection relative to your blood draw matters only for the minor point that glucose may be slightly lower in the 24-48 hours immediately following an injection versus 5-6 days after. For the most representative results, draw blood at the same point in your dosing cycle each time (for example, always on day 4 after injection).
Knowing the best time to take semaglutide in relation to your lab schedule can help standardize your results.
What your blood work cannot tell you about semaglutide
Blood work is powerful but limited. Here is what your lab results will not reveal about your semaglutide experience.
Appetite suppression. There is no blood marker that quantifies how much semaglutide is reducing your hunger. This is purely subjective. You cannot measure GLP-1 receptor occupancy in the brain through a blood test. Tracking how long it takes for appetite suppression to develop requires self-reporting, not lab work.
Body composition changes. Blood work tells you about metabolic health but not about how much of your weight loss is fat versus muscle. You could have excellent metabolic improvements (great blood work) while losing disproportionate muscle mass, or vice versa. Body composition assessment requires different tools: DEXA scans, bioelectrical impedance, or at minimum, waist circumference measurements. Before and after semaglutide results are best tracked with a combination of lab work and physical measurements.
Medication quality. Your blood work cannot distinguish between pharmaceutical-grade semaglutide and a degraded or counterfeit product. Improved metabolic markers suggest the medication is working, but unchanged markers could mean the product is inactive OR that your body is not responding, and blood work alone cannot differentiate between these possibilities.
Optimal dosing. Lab results show you the effects of your current dose but cannot tell you whether a higher or lower dose would serve you better. Dose optimization requires integrating blood work data with clinical response (weight trajectory, appetite, side effects, energy levels). Proper dosing calculations using the semaglutide dosage calculator and syringe dosage charts ensure you are administering what you intend.
Frequently asked questions
Will my insurance company find out I am taking semaglutide through blood work?
No. Insurance companies do not receive your actual lab values in a format that would reveal semaglutide use. They see billing codes for the tests ordered, not the test results themselves. Your improved metabolic markers would not trigger any flags. The only way an insurer would know about semaglutide use is through pharmacy claims if you fill a prescription through your insurance.
Can my employer detect semaglutide through a pre-employment drug screening?
No. Pre-employment drug screenings test for substances of abuse (opioids, amphetamines, benzodiazepines, cannabinoids, cocaine). Semaglutide is not a controlled substance and bears no chemical resemblance to any screened drug class. It will not produce a positive result on any standard employment drug test. For more details, see our guide on peptides and drug tests.
How soon after starting semaglutide will my blood work change?
Fasting glucose may improve within the first 1-2 weeks. HbA1c, which reflects a 2-3 month average, shows meaningful change at the 3-month mark. Lipid improvements typically become statistically significant by 12-26 weeks. Liver enzyme improvements develop gradually over 12-52 weeks. Tracking your first week on semaglutide can reveal early glucose changes.
Should I fast before blood work if I take semaglutide?
Standard fasting requirements (10-12 hours, water only) apply regardless of semaglutide use. The medication does not change fasting protocols. Take your semaglutide on its normal schedule, as it does not interfere with test accuracy. For the most consistent results over time, try to have blood drawn at the same point in your weekly dosing cycle.
Can semaglutide cause false positive results on any blood tests?
Semaglutide does not cause false positives on standard blood tests or drug screenings. It does not cross-react with immunoassays for common drugs of abuse. The metabolic changes it produces (lower glucose, improved lipids) are real physiological improvements, not artifacts or false readings.
What blood work should I get if I am experiencing side effects on semaglutide?
For persistent nausea and vomiting, check kidney function (BMP) to rule out dehydration-related kidney stress. For severe abdominal pain, check lipase and amylase to evaluate for pancreatitis. For unusual fatigue, check thyroid function (TSH, free T4), iron studies, and vitamin B12. For dizziness, check blood pressure and a basic metabolic panel for electrolyte imbalances. For hair loss, check iron, ferritin, zinc, biotin, and thyroid function.
Do I need blood work before starting compounded semaglutide?
Yes. The same baseline labs recommended for branded semaglutide apply to compounded versions. In fact, monitoring may be even more important with compounded products because potency can vary between batches. Baseline labs establish your starting point so you and your provider can assess whether the compounded product is producing expected effects.
Will stopping semaglutide change my blood work back to pre-treatment levels?
Partially, over time. Glucose markers (HbA1c, fasting glucose) typically begin rising within 4-8 weeks of discontinuation. Lipid changes may persist longer if weight loss is maintained. Liver enzyme improvements often persist as long as reduced body weight is maintained. The metabolic benefits that depended on the direct pharmacological effect of semaglutide will fade. The benefits attributable to sustained weight loss may persist. Understanding semaglutide withdrawal symptoms helps you prepare for changes after stopping.
External resources
New England Journal of Medicine: FLOW Trial (semaglutide kidney outcomes)
PMC: Effect of semaglutide on liver enzymes and markers of inflammation
Ulta Lab Tests: Semaglutide blood biomarker monitoring guide
Diabetes Care: GLP-1 impact on amylase, lipase, and pancreatitis
For researchers serious about understanding every marker and optimizing their protocols with precision, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, detailed protocol databases, and a community of thousands who track their lab work alongside their peptide research.
In case I do not see you, good afternoon, good evening, and good night. May your lab results stay reassuring, your markers stay improving, and your protocols stay informed.