Feb 22, 2026

That vial in your refrigerator door is losing potency right now. Not because semaglutide is fragile, not because your fridge is broken, but because the door shelf is the worst possible spot for a temperature-sensitive peptide. And that is just one of dozens of storage mistakes that quietly degrade the medication people spend hundreds of dollars on every month.
Here is the frustrating part. The answer to "how long does semaglutide last in the fridge" changes completely depending on which form you are using. Ozempic gets 56 days after first use. Wegovy gets 28. Compounded semaglutide from a pharmacy might last anywhere from 30 to 90 days, depending on who made it and how they tested it. And lyophilized powder, the kind researchers reconstitute themselves, follows an entirely different timeline.
Most storage guides lump all these together. They give you one number. One temperature range. One set of rules. That is not good enough. Because if you are following Ozempic guidelines for your compounded semaglutide, you could be injecting degraded peptide without knowing it. And if you are treating compounded vials like they expire in a week, you might be throwing away perfectly good medication.
This guide breaks down the exact refrigeration timelines for every form of semaglutide, the science behind why temperature matters so much for this particular peptide, and the storage mistakes that silently destroy potency. Whether you are using brand-name pens, oral formulations, or compounded vials, you will find your specific numbers here. SeekPeptides created this comprehensive reference because the stakes are real. Every degree matters. Every day counts. And the wrong storage decision can turn a $300 monthly investment into expensive water.
The quick answer for every form of semaglutide
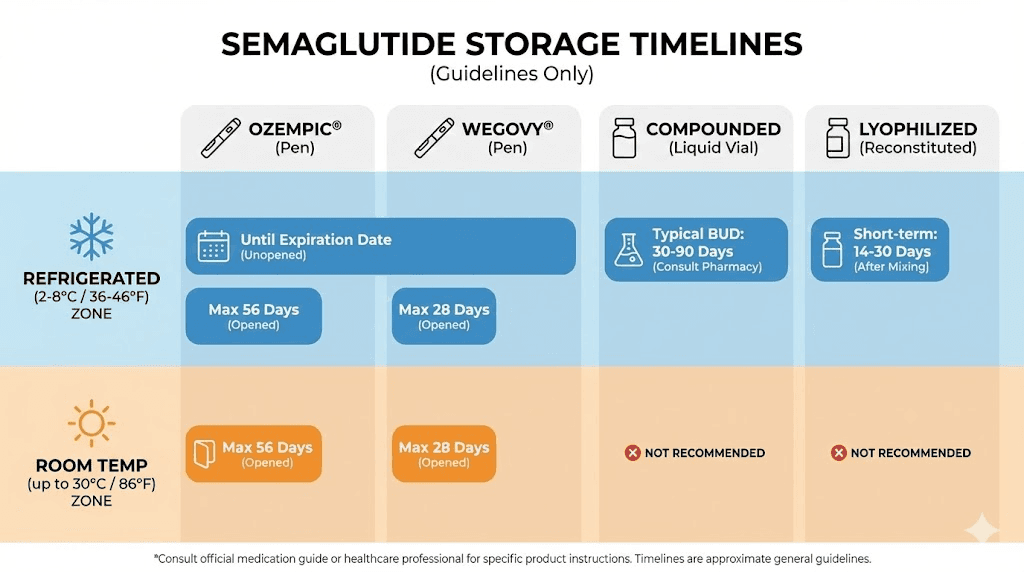
Before we get into the details, here is the summary table. Find your specific product. Get your number. Then read the sections below for the context that makes these numbers actually useful.
Product | Unopened (refrigerated) | After first use (refrigerated) | Room temperature |
|---|---|---|---|
Ozempic pen | Until expiration date | 56 days | 56 days (59-86F) |
Wegovy pen | Until expiration date | 28 days | 28 days (46-86F) |
Rybelsus tablets | No refrigeration needed | N/A | Until expiration (68-77F) |
Compounded injectable | 30-90 days (BUD) | 28 days typical | Not recommended |
Lyophilized powder | Months to years (sealed) | 28-30 days after reconstitution | Varies by formulation |
Those numbers look simple. They are not. The difference between 28 and 56 days has caught thousands of people off guard, and the compounded semaglutide timeline depends entirely on your specific pharmacy. Keep reading for the details that actually matter.
Ozempic in the fridge: the 56-day rule
Ozempic is the most commonly prescribed brand of injectable semaglutide for type 2 diabetes. Its storage rules are more generous than most people expect.
Before first use: Keep Ozempic refrigerated between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). In this state, it remains good until the manufacturer expiration date printed on the box, which is typically about two years from the date of manufacture.
That two-year shelf life is significant. It means you can stock up during periods of availability without worrying about waste, as long as your refrigerator maintains proper temperature. Novo Nordisk conducts extensive stability testing to establish these timelines, and the data supports the full expiration window when storage conditions are met.
After first use: Once you remove the cap and inject your first dose, the clock starts. You now have exactly 56 days. That is 8 weeks. The pen can stay refrigerated or sit at room temperature during this period, as long as room temperature stays between 59 and 86 degrees Fahrenheit (15 to 30 degrees Celsius).
At day 57, throw it away. Even if there is medication left in the pen. Even if it looks perfectly clear. Even if you stored it flawlessly. The 56-day limit exists because the rubber seal and pen mechanism introduce contamination risks over time, not because the semaglutide molecule itself suddenly degrades at the 57-day mark.
Why Ozempic gets more time than Wegovy
This confuses people. Same active ingredient. Same molecule. Same manufacturer. Different storage windows. The reason comes down to formulation and concentration. Ozempic contains semaglutide at concentrations of 1.34 mg/mL, with specific preservatives and stabilizers optimized for multi-dose pen delivery over 8 weeks. The pen design, the preservative system, and the dosing schedule all work together to support the longer 56-day window.
If you are calculating your semaglutide dosage and wondering whether to switch between brands, understand that these storage differences might affect your protocol planning. A pen that lasts 56 days gives you more flexibility than one that expires in 28. Use the peptide calculator to determine exact amounts for your concentration. For a deeper comparison of semaglutide versus other GLP-1 options, understanding the practical differences goes beyond just the active ingredient. Whether semaglutide truly qualifies as Ozempic or stands alone as a class depends on the formulation, and each formulation carries its own storage profile.
Common Ozempic storage mistakes
The refrigerator door. That is where most people put it. Convenient, visible, easy to remember. And the absolute worst location in your fridge. Every time you open the door, the temperature swings by 5 to 10 degrees. Over the course of a day, items on the door shelf experience more temperature fluctuation than anything else in the refrigerator. These repeated thermal cycles accelerate peptide degradation far more than a single temperature excursion.
The better location is the middle shelf, toward the back. Temperatures there stay the most consistent. If you keep your semaglutide injection schedule regular, you will develop a habit of reaching for the same spot every time.
Another common mistake is freezing. The freezer compartment in most home refrigerators sits directly above or behind the main cooling element. Place your Ozempic pen too close to the back wall on certain shelves and the temperature can drop below freezing, especially in older refrigerators with less precise temperature control. Frozen semaglutide must be discarded immediately. Even if it thaws and looks normal, the freeze-thaw cycle damages the peptide structure in ways that are not visible to the naked eye.
How semaglutide storage compares to tirzepatide
If you use both semaglutide and tirzepatide, or if you are considering switching between the two, understanding the storage differences matters. Tirzepatide (Mounjaro, Zepbound) also requires refrigeration, but the details differ in ways that catch people off guard.
Tirzepatide in the fridge follows its own timeline. Mounjaro allows 21 days at room temperature after first use, compared to 56 days for Ozempic. If you are managing storage for both medications simultaneously, keeping the timelines straight prevents costly mix-ups. The dosage comparison between the two medications shows they are quite different in potency, and their storage requirements reflect those formulation differences.
For compounded versions, compounded tirzepatide has its own room temperature rules that differ from compounded semaglutide. The preservative systems, concentrations, and stability profiles are not interchangeable. If you are researching both peptides through our semaglutide versus tirzepatide comparison, storage logistics should factor into your decision alongside efficacy and side effect profiles.
Wegovy in the fridge: the 28-day rule
Wegovy is semaglutide prescribed specifically for weight management, at higher doses than Ozempic. Despite containing the same active molecule, its storage rules are stricter.
Before first use: Refrigerate between 36 and 46 degrees Fahrenheit. Good until the expiration date on the packaging. Same as Ozempic here.
After first use: 28 days. That is it. Whether you keep it refrigerated or at room temperature (46 to 86 degrees Fahrenheit), you have four weeks to use or discard. Note that Wegovy allows a slightly wider room temperature range on the lower end, starting at 46 degrees instead of 59, but the overall window is half of what Ozempic provides.
This shorter timeline creates real practical challenges. If your semaglutide results have been progressing well and you need to pause for surgery or travel, the 28-day window means you cannot simply set the pen aside and come back to it next month. Understanding when to resume semaglutide after surgery becomes even more important when storage timelines constrain your options.
The Wegovy dose escalation storage challenge
Wegovy uses a five-step dose escalation schedule. You start at 0.25 mg for four weeks, then move to 0.5 mg, then 1.0 mg, then 1.7 mg, and finally 2.4 mg. Each dose comes in its own single-use pen. This actually simplifies the storage question in one way: since each pen is used once, the "after first use" timeline is less relevant for most patients.
But here is where it gets complicated. Some patients experience side effects that require managing gastrointestinal symptoms by staying at a lower dose longer than the standard four weeks. If your prescriber tells you to extend time at 0.5 mg, you might have leftover pens at the 0.25 mg dose. Those unused pens stay good until their expiration date as long as they remain sealed and refrigerated. Do not throw them away prematurely.
The unit conversion for semaglutide doses matters significantly during the escalation phase. Understanding exactly how much medication you are receiving at each step helps you plan your supply and minimize waste. SeekPeptides members access detailed dose escalation protocols that account for these practical storage considerations.

Rybelsus: the semaglutide that does not need a fridge
Rybelsus is oral semaglutide in tablet form. It is the exception to everything discussed so far.
No refrigeration required. Store at room temperature between 68 and 77 degrees Fahrenheit (20 to 25 degrees Celsius). Keep in the original packaging. Protect from moisture. That is the complete set of storage instructions.
The reason Rybelsus breaks from the refrigeration requirement is its formulation. The tablet uses a technology called SNAC (sodium N-[8-(2-hydroxybenzoyl)amino] caprylate) to enhance oral absorption. The semaglutide in tablet form is protected by the solid matrix of the tablet itself, making it far less susceptible to the temperature-induced degradation that affects liquid injectable formulations.
However, moisture is the enemy of Rybelsus. The SNAC technology is hygroscopic, meaning it attracts water from the environment. Humidity breaks down the absorption enhancer before the semaglutide even reaches your stomach, reducing the amount of peptide that actually makes it into your bloodstream. This is why you should never remove tablets from the blister pack until you are ready to take them.
If you are weighing the differences between injectable versus oral peptides, the storage simplicity of Rybelsus is a genuine advantage. No cold chain management. No worrying about leaving it out overnight. No counting days since first use. Just a dry, room-temperature cabinet. For people who travel frequently with semaglutide, the oral form eliminates virtually all storage complications. Understanding the broader landscape of research versus pharmaceutical grade peptides also helps contextualize why storage requirements vary so dramatically between different semaglutide products.
Compounded semaglutide: the complicated one
This is where storage rules get genuinely confusing. Compounded semaglutide does not come with the standardized storage data that brand-name products provide. Different pharmacies use different formulations, different concentrations, different preservatives, and different stability testing protocols. The result is a wide range of shelf life claims.
Beyond-use date versus expiration date
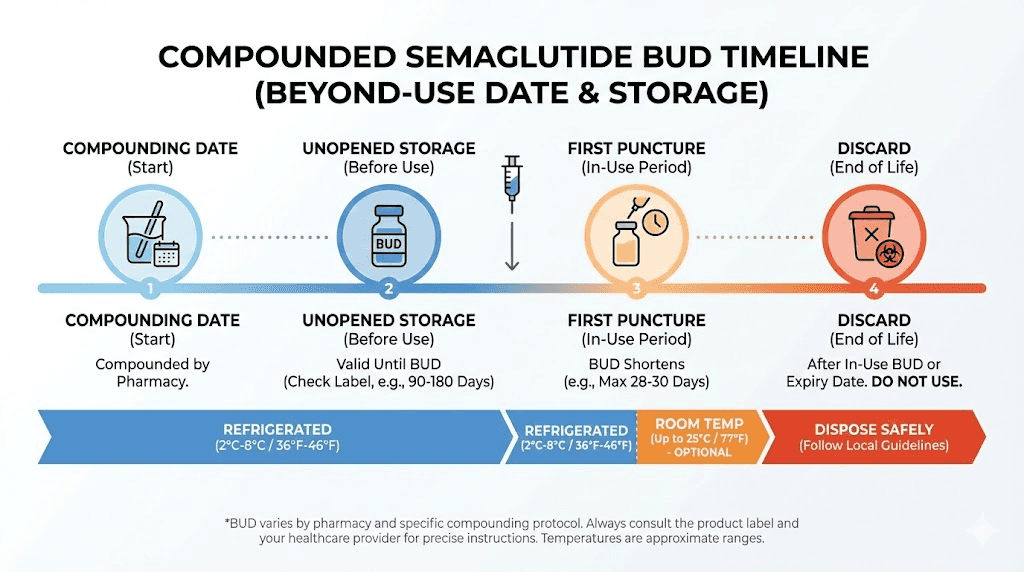
Brand-name drugs have expiration dates established through years of rigorous stability testing submitted to the FDA. Compounded medications have something different: a beyond-use date, or BUD. The BUD represents the date after which the pharmacy cannot guarantee the medication meets its labeled potency and sterility standards.
For compounded semaglutide, the BUD typically ranges from 30 to 90 days when stored refrigerated at 36 to 46 degrees Fahrenheit. Some pharmacies with advanced stability testing programs have demonstrated BUDs of up to 120 days. Others, particularly those relying on default USP guidelines rather than product-specific testing, assign a conservative 14 to 28 day BUD.
This means two vials of compounded semaglutide from different pharmacies can have wildly different shelf lives, even if they contain the same concentration. The difference comes down to testing. Pharmacies that invest in third-party stability studies, potency testing at intervals, and sterility validation can justify longer BUDs. Those that follow minimum regulatory requirements typically assign shorter ones.
For a deeper dive into the specific storage requirements of the compounded form, our complete guide to compounded semaglutide refrigeration covers the nuances in detail. And if you are unsure whether compounded semaglutide requires refrigeration at all, the short answer is yes, almost always.
Concentration matters for shelf life
Compounded semaglutide comes in various concentrations. The most common are 2.5 mg/mL, 5 mg/mL, and 10 mg/mL. Higher concentrations generally have slightly better stability profiles because the ratio of active peptide to degradation-promoting solvent is more favorable. But this advantage is modest and should not override the BUD assigned by your pharmacy.
If you are working with a 5 mg/mL semaglutide formulation, the reconstitution and storage parameters will differ from a 10 mg/mL formulation. Always check the documentation that came with your specific vial. The concentration affects not only how many units you draw per dose but also how long the vial remains stable.
What about multi-use vials?
Most compounded semaglutide comes in multi-use vials, meaning you draw multiple doses from the same container. Each time you pierce the rubber stopper with a needle, you introduce a small amount of contamination risk. This is why the BUD for a compounded vial after first puncture is typically shorter than the overall BUD of an unopened vial.
A common pattern: the pharmacy labels the vial with a 90-day BUD from compounding date. But once you puncture the stopper, the effective shelf life drops to 28 days regardless of the original BUD. This two-tiered system catches people off guard. They see 90 days on the label, start using the vial, and assume they have 90 days from first use. They do not.
Proper injection technique matters here too. If you are using a syringe to draw from a vial, alcohol swabbing the stopper before each draw and using a fresh needle every time reduces contamination risk and supports the full BUD window. Reusing needles or skipping the alcohol wipe accelerates bacterial introduction and can shorten the safe window even further.
Lyophilized semaglutide: storage before and after reconstitution
Lyophilized (freeze-dried) semaglutide is the form most commonly used in research settings. It arrives as a dry powder in a sealed vial and must be reconstituted with bacteriostatic water or sterile water before use.
Before reconstitution
Dry lyophilized semaglutide is remarkably stable. In sealed vials, stored at refrigerator temperatures (36 to 46 degrees Fahrenheit), the powder can remain viable for months to years depending on the manufacturer and specific formulation. The absence of water is the key. Without a liquid medium, the degradation pathways that break down the semaglutide molecule are essentially frozen in place.
Some suppliers even allow short-term storage of sealed lyophilized vials at room temperature without significant degradation. However, for maximum shelf life, refrigeration is still recommended. The general principle with lyophilized versus liquid peptides applies here: the dry form always wins on storage stability.
After reconstitution
Once you add bacteriostatic water to the lyophilized powder, the storage rules change dramatically. The reconstituted solution must be refrigerated at 36 to 46 degrees Fahrenheit, and most protocols recommend using it within 28 to 30 days.
The type of water you use for reconstitution matters significantly. Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, which helps prevent bacterial growth in the reconstituted solution. This preservative is what allows the 28-30 day window. If you use plain sterile water instead, which contains no preservative, the reconstituted solution should ideally be used within a few days and stored with extreme care.
For detailed reconstitution instructions, including exactly how much bacteriostatic water to mix with 10 mg of semaglutide, the ratios for mixing 5 mg vials, or the specific process for reconstituting semaglutide properly, getting the ratio right directly affects both dosing accuracy and storage stability. Our complete semaglutide reconstitution chart provides visual reference for common concentrations. The peptide reconstitution calculator on SeekPeptides can help you determine the exact amounts for your specific vial size.
Reconstitution concentration and stability
There is an interesting relationship between reconstitution concentration and refrigerated shelf life. Higher concentrations (more peptide per mL of water) tend to be slightly more stable because there are fewer water molecules available per peptide molecule to drive hydrolysis reactions. This is one reason why protocols for reconstituting 5 mg semaglutide might suggest different water volumes than those for mixing 5 mg vials depending on whether the priority is accuracy or stability.
The tradeoff is precision. More concentrated solutions mean smaller injection volumes, which can be harder to measure accurately with standard insulin syringes. Understanding unit-to-mg conversions for semaglutide becomes critical when working with concentrated reconstitutions. A one-unit error at 10 mg/mL is very different from a one-unit error at 2.5 mg/mL. Whether you are measuring 10 units, 20 units, or 40 units of semaglutide, the concentration of your specific vial determines the actual milligram dose and the conversion chart for your formulation should always be your reference.
The science behind semaglutide temperature sensitivity
Understanding why temperature matters helps you make better storage decisions when the guidelines are not perfectly clear. Semaglutide is a modified GLP-1 receptor agonist, a 31-amino acid peptide with specific structural features that make it both more durable and more vulnerable than you might expect.
What makes semaglutide more stable than most peptides
Novo Nordisk engineered semaglutide with a C-18 fatty acid chain attached to the peptide backbone through a linker. This modification gives semaglutide its long half-life in the body (approximately 7 days, which is why you only inject weekly). It also provides some structural protection against degradation. The fatty acid chain acts partly as a shield, reducing the exposure of vulnerable amino acid residues to degradation-promoting factors in solution.
Additionally, the amino acid sequence includes specific substitutions, notably at positions 8 and 34 compared to native GLP-1, that make the molecule more resistant to enzymatic breakdown. These same substitutions provide modest improvements in chemical stability during storage.
This engineered durability is why semaglutide can tolerate room temperature for days or weeks, unlike many research peptides that degrade rapidly outside refrigeration. If you are comparing storage requirements across your full supplement protocol, semaglutide is actually one of the more forgiving peptides when it comes to brief temperature excursions.
What still degrades semaglutide
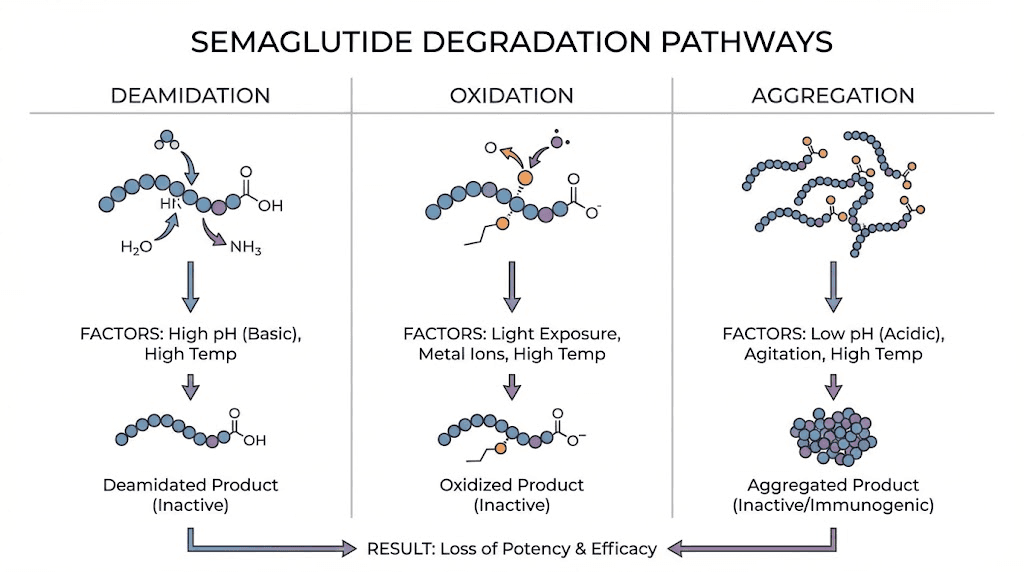
Despite its engineered stability, semaglutide is still vulnerable to several degradation pathways.
Deamidation. This is the primary degradation mechanism. Asparagine residues in the peptide chain spontaneously convert to aspartic acid, changing the molecule structure. The rate of deamidation increases with temperature and is influenced by pH. Recent preformulation studies published in the Journal of Peptide Science found that semaglutide is most vulnerable to degradation between pH 4.5 and 5.5, near its isoelectric point of pH 5.4. This is why commercial formulations are buffered above pH 7.0.
Oxidation. Certain amino acids in the semaglutide chain, particularly methionine residues, are susceptible to oxidation. Exposure to light, elevated temperatures, and dissolved oxygen in solution all accelerate this process. This is why storage instructions consistently recommend keeping semaglutide in its original packaging, away from direct light.
Aggregation. At elevated temperatures, semaglutide molecules can unfold and stick together, forming aggregates. Aggregated peptide is not only inactive, it can potentially trigger immune responses. Temperature cycling, where the vial repeatedly warms up and cools down, promotes aggregation more aggressively than sustained exposure at a constant higher temperature.
This last point is crucial. It is not just about average temperature. It is about consistency. A vial stored at a steady 40 degrees Fahrenheit will outperform a vial that swings between 35 and 50 degrees multiple times per day, even if the average of both is the same. This is the scientific basis for keeping your semaglutide away from the refrigerator door.
Signs your semaglutide has gone bad
Visual inspection cannot detect most degradation. A vial can lose 30% of its potency and still look perfectly clear. But when degradation has progressed to the point of visible changes, you are well past the point of reduced effectiveness. You are in territory where using the product could be genuinely problematic.
Visual warning signs
Normal semaglutide solution is clear and colorless. Any deviation from this should trigger immediate concern.
Cloudiness or turbidity. If the solution looks hazy, milky, or anything other than crystal clear, do not use it. Cloudiness indicates aggregation or bacterial contamination, both of which mean the product is no longer safe. This is different from tiny bubbles, which can form during drawing and are normal.
Color change. Any yellowing, browning, or other discoloration signals chemical degradation. The byproducts of deamidation and oxidation can produce colored compounds at advanced stages of breakdown. If your semaglutide has color, it is telling you something went very wrong during storage.
Particles or precipitate. Floating specks, crystals, flakes, or sediment at the bottom of the vial are all signs of degradation or contamination. Sometimes gentle swirling can distinguish between actual particles and air bubbles. Bubbles rise to the top and disappear. Particles do not.
If you are wondering about the appearance of fresh semaglutide for comparison, our guide on what color semaglutide should be provides detailed visual reference points.
Non-visual warning signs
Unusual odor. Semaglutide solution should have essentially no smell. A chemical, sour, or otherwise unusual odor indicates breakdown or contamination.
Reduced effectiveness. This is the most insidious sign because it develops gradually. If your appetite suppression from semaglutide has been declining despite consistent dosing, and you have not recently changed anything else in your protocol, degraded medication is a possibility worth investigating. Many people blame tolerance or their own body when the real culprit is a vial that has been slowly losing potency. If you are experiencing a semaglutide plateau, check your storage conditions before assuming the medication has stopped working.
Increased side effects. Paradoxically, degraded semaglutide can sometimes cause more side effects, not fewer. Breakdown products and aggregates can trigger injection site reactions, dizziness, or unusual gastrointestinal symptoms that differ from the typical fatigue or nausea associated with properly stored medication.
When in doubt
If you are unsure whether your semaglutide is still good, the safest approach is to discard it. The cost of a wasted partial vial is always less than the cost of injecting degraded medication that either does not work or causes adverse reactions. Understanding whether expired semaglutide is safe to use helps you make informed decisions, but the conservative approach is almost always the right one.
What happens when semaglutide gets warm
Life happens. You forget the pen on the counter. The pharmacy delivers it and it sits on the porch for three hours. The power goes out and your fridge warms up. These situations are more common than people admit, and the consequences depend entirely on how warm and for how long.
Brief temperature excursions
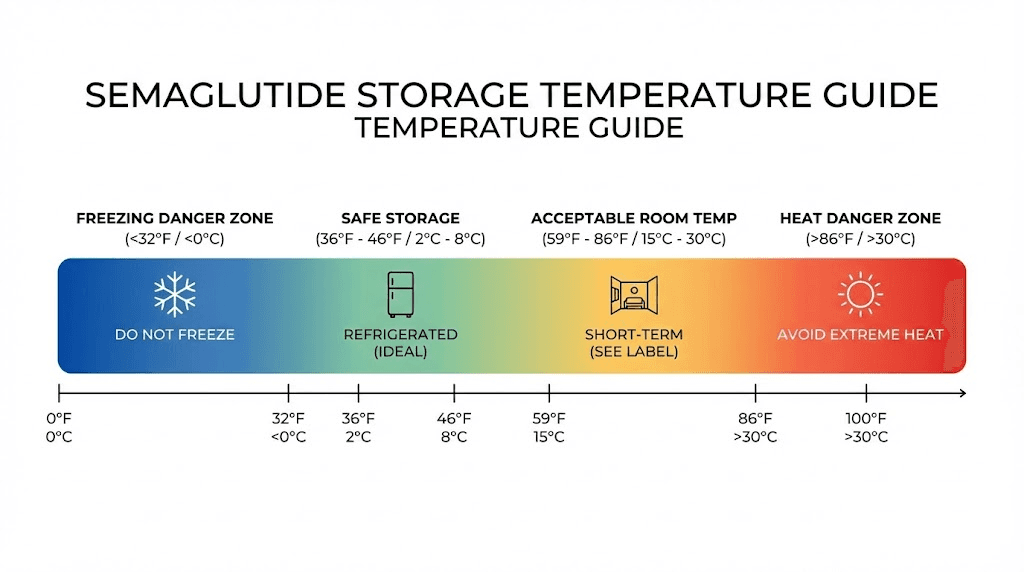
Semaglutide is more resilient than many people think. A pen left on the counter for 30 minutes while you prepare your injection is not damaged. Even a few hours at room temperature is unlikely to cause measurable degradation, as long as room temperature means below 86 degrees Fahrenheit.
Stability studies show that semaglutide remains remarkably stable at 25 degrees Celsius (77 degrees Fahrenheit) for extended periods. The brand-name products would not have room temperature allowances of 28 to 56 days if brief excursions were problematic. The molecule can handle it.
Extended heat exposure
The rules change above 86 degrees Fahrenheit (30 degrees Celsius). At these temperatures, degradation rates increase significantly. A vial left in a hot car on a summer day (where interior temperatures can exceed 140 degrees Fahrenheit) should be discarded. Period. No exceptions.
Research published in the Journal of Peptide Science examined semaglutide stability at various temperatures. At 40 degrees Celsius (104 degrees Fahrenheit), measurable degradation occurred within 24 hours. At 60 degrees Celsius (140 degrees Fahrenheit), significant breakdown occurred within hours. At 80 degrees Celsius (176 degrees Fahrenheit), the peptide remained stable for approximately 3 hours before substantial degradation.
For a comprehensive breakdown of heat-related concerns, including what to do in specific scenarios, our guide on what happens when semaglutide gets warm covers every situation you might encounter.
The freezing problem
Freezing is worse than moderate warming. When semaglutide solution freezes, ice crystals form within the liquid. These crystals physically disrupt the peptide structure through a process called freeze-induced denaturation. The ice crystals act like tiny blades, cutting through the three-dimensional structure that the peptide needs to function.
Unlike moderate warming, which causes gradual chemical degradation that may leave some peptide intact, freezing can cause sudden structural damage to a large percentage of the molecules simultaneously. And here is the real problem: frozen-then-thawed semaglutide often looks perfectly normal. Clear. Colorless. No visible signs of damage. But the peptide has been compromised.
This is why every storage guide for every form of semaglutide says the same thing: if it has been frozen, discard it. No exceptions. Even if it looks fine. Even if it was only frozen briefly. The damage is done. The same principle applies to other injectable peptides, including tirzepatide. If you also store tirzepatide in your fridge, the freezing rule is equally absolute for both medications.
Room temperature storage: the rules for each form
Sometimes refrigeration is not available. Travel, power outages, or transitions between locations create situations where semaglutide must spend time at room temperature. Knowing the exact rules for your specific product prevents unnecessary waste and unnecessary risk.
Ozempic at room temperature
After first use, Ozempic can be stored at room temperature (59 to 86 degrees Fahrenheit) for up to 56 days. This is the same 56-day window whether you refrigerate the in-use pen or not. The timer starts at first use, not when you remove it from the fridge.
One important clarification: an unopened Ozempic pen that has been at room temperature should be used or discarded within 56 days from when it left refrigeration. You cannot leave an unopened pen at room temperature for 55 days, put it back in the fridge, and reset the clock. Once it leaves the cold chain, the room temperature timer is running.
Wegovy at room temperature
Wegovy permits room temperature storage (46 to 86 degrees Fahrenheit) for up to 28 days. Note the lower bound: 46 degrees Fahrenheit, not 59. This wider acceptable range on the cool side gives you more flexibility with cooler room environments.
The same clock applies. Once Wegovy leaves the refrigerator, you have 28 days. No resets. No do-overs. If you need to travel with your semaglutide, plan your trip within the 28-day window or bring a cooling case.
Compounded semaglutide at room temperature
Most compounding pharmacies do not validate their semaglutide for room temperature storage. The standard recommendation is to keep compounded formulations refrigerated at all times. If your compounded semaglutide has been at room temperature for more than a few hours, contact your pharmacy for guidance specific to their formulation.
This is one of the significant practical differences between brand-name and compounded semaglutide. The brand-name products come with room temperature data backed by extensive stability studies. Compounded products typically do not. If room temperature flexibility matters to your lifestyle, this is worth factoring into your decision.
Rybelsus at room temperature
Already covered, but worth restating: Rybelsus lives at room temperature. Store between 68 and 77 degrees Fahrenheit. No refrigeration needed. Just keep it dry.
Traveling with semaglutide: keeping it cold on the go
Travel creates the biggest storage challenge for semaglutide users. Airports, hotels, road trips, and international destinations all introduce temperature management problems that do not exist at home.
Air travel
Injectable semaglutide is permitted in carry-on luggage. The TSA allows medically necessary liquids and syringes. Declare them at the security checkpoint. Having the original pharmacy label on the pen or vial helps, though it is not strictly required by TSA regulations.
Never check your semaglutide in luggage. Cargo holds on aircraft can drop below freezing, and your medication will be destroyed. Always carry it on. Use a small insulated case with a gel pack to maintain cool temperatures during the flight. Medical cooling cases designed for insulin work perfectly for semaglutide.
For multi-day trips, hotel room refrigerators are generally acceptable for storage, though they tend to be slightly warmer and less consistent than home refrigerators. Place the vial toward the back of the mini-fridge, away from the door. If the room refrigerator has a freezer compartment, keep the semaglutide as far from it as possible to avoid accidental freezing.
Road trips
Car interiors can exceed 140 degrees Fahrenheit on hot days. Never leave semaglutide in a parked car, even for short periods. A cooler with ice packs works well for road trips, but create a barrier between the ice packs and the vial. Direct contact with frozen ice packs can freeze the medication, which is just as damaging as heat exposure.
The ideal setup is a small insulated bag with a gel pack, wrapped in a cloth or towel, placed in the passenger cabin where air conditioning keeps temperatures moderate. This maintains the 36 to 46 degree Fahrenheit range without risk of freezing.
International travel
If you are traveling internationally, carry a letter from your healthcare provider documenting your prescription. Some countries have restrictions on importing injectable medications without documentation. Also consider the climate at your destination. If you are traveling to a tropical location, plan your cold chain carefully.
For people using the oral formulation, international travel is significantly simpler. Oral semaglutide needs no refrigeration, no cooling case, and minimal documentation beyond a standard prescription. Even the newer sublingual semaglutide formulations offer travel-friendly storage advantages over injectable versions.
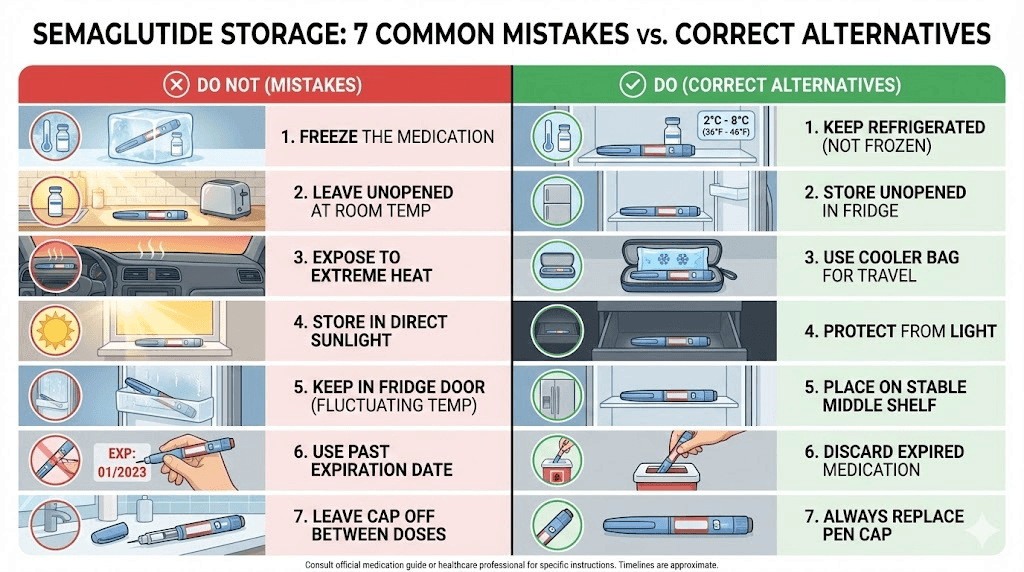
Storage mistakes that destroy semaglutide potency
After analyzing thousands of community reports and pharmacist consultations, certain storage mistakes come up again and again. Avoid these and your semaglutide will maintain its potency for the full duration of its labeled shelf life.
Mistake 1: the refrigerator door
Already mentioned, but it bears repeating because it is the single most common mistake. The door shelf experiences temperature swings of 5 to 15 degrees with every opening. These cycles promote aggregation. Move your semaglutide to the middle shelf, toward the back.
Mistake 2: storing near the back wall of the freezer side
In side-by-side refrigerators, cold air from the freezer compartment often leaks into the refrigerator section along the shared wall. Items placed directly against this wall can experience temperatures below freezing. Keep your semaglutide at least two inches from any wall that borders the freezer.
Mistake 3: assuming all forms have the same timeline
The difference between Ozempic at 56 days and Wegovy at 28 days is not trivial. People who switch between brands or formulations often apply the wrong storage rules to the wrong product. If you have recently started switching between GLP-1 medications, double-check the storage requirements for your new product. They are almost certainly different from what you were using before.
Mistake 4: using a faulty fridge thermometer
Most people have never checked their refrigerator temperature with an independent thermometer. The built-in thermostat dial with settings from 1 to 5 gives you no actual temperature data. A digital refrigerator thermometer costs less than $10 and tells you exactly what temperature your medication is experiencing. If the reading falls outside 36 to 46 degrees Fahrenheit, adjust immediately.
Mistake 5: leaving the cap off
Ozempic and Wegovy pens should be stored with the cap on to protect the needle and the medication from light exposure. Light drives photodegradation reactions that break down the peptide. The cap is not just for convenience. It is a storage requirement.
Mistake 6: returning to the fridge after extended room temp
Some people think they can extend the life of their semaglutide by alternating between room temperature and refrigeration. They leave the pen out during the day, put it back in the fridge at night, and assume the cold restores whatever was lost during the warm period. This is wrong. Temperature cycling promotes aggregation more than stable storage at either temperature. Once the room temperature clock starts, it does not pause when you return to cold storage.
Mistake 7: ignoring the reconstitution date
For lyophilized semaglutide, the storage clock starts at reconstitution, not at first injection. If you reconstitute a vial but do not use it for a week, you have already used one-quarter of your 28-day window. Write the reconstitution date on the vial with a permanent marker. Do not trust your memory.
Understanding whether semaglutide expires after 28 days depends entirely on which form you are using and when the clock started. Get the timing wrong and you risk either wasting good medication or using bad medication.
How to check if your fridge temperature is right
The ideal storage temperature for all injectable semaglutide forms is between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). Here is how to verify your refrigerator hits this range and how to fix it if it does not.
Testing your refrigerator
Buy a digital refrigerator thermometer. Place it on the shelf where you store your semaglutide. Leave it for 24 hours. Check the reading.
If it reads between 36 and 46 degrees Fahrenheit, you are good. If it reads below 36, your semaglutide might be at risk of freezing, especially on cold days when your fridge runs more aggressively. If it reads above 46, your medication is degrading faster than it should.
Better thermometers record minimum and maximum temperatures over a period. These are especially useful because they catch the temperature swings that a single reading misses. Your fridge might read 40 degrees when you check at 2 PM, but drop to 33 degrees at 3 AM when the compressor cycles aggressively in a quiet house.
Adjusting your refrigerator
Most home refrigerators run between 35 and 38 degrees Fahrenheit at their default setting. This is fine for semaglutide, though it is on the cold end of the acceptable range. If your thermometer shows temperatures dropping below 36 degrees regularly, consider raising the temperature setting by one increment and monitoring for another 24 hours.
The crisper drawers at the bottom of most refrigerators tend to maintain the most consistent temperature. If you have space, these can be excellent locations for medication storage, as long as they are not set to high-humidity mode (which some models offer for produce storage).
Storage timelines at a glance: your complete reference
Let us put everything together in one comprehensive reference. This section is designed to be bookmarked and returned to whenever you have a storage question.
Ozempic complete storage reference
Unopened, refrigerated (36-46F): Until manufacturer expiration date (typically 2 years from manufacture).
Unopened, room temperature (59-86F): 56 days from leaving refrigeration.
After first use, refrigerated: 56 days from first use.
After first use, room temperature (59-86F): 56 days from first use.
Above 86F: Discard.
Frozen: Discard immediately.
Wegovy complete storage reference
Unopened, refrigerated (36-46F): Until manufacturer expiration date.
Unopened, room temperature (46-86F): 28 days from leaving refrigeration.
After first use, refrigerated: Single-use pens, use immediately.
After first use, room temperature (46-86F): 28 days from leaving refrigeration.
Above 86F: Discard.
Frozen: Discard immediately.
Rybelsus complete storage reference
Room temperature (68-77F): Until manufacturer expiration date.
Humidity: Keep in original blister pack until use.
Refrigeration: Not required, not harmful.
Freezing: Not applicable (solid tablet).
Compounded semaglutide complete storage reference
Unopened, refrigerated (36-46F): Until beyond-use date (BUD), typically 30-90 days from compounding.
After first puncture, refrigerated: 28 days or as specified by pharmacy (whichever comes first).
Room temperature: Not recommended unless pharmacy specifically validates.
Frozen: Discard immediately.
Lyophilized semaglutide complete storage reference
Unreconstituted, refrigerated: Months to years (check manufacturer).
Unreconstituted, room temperature: Weeks to months (check manufacturer).
Reconstituted with bacteriostatic water, refrigerated: 28-30 days.
Reconstituted with sterile water, refrigerated: 3-7 days.
Reconstituted, room temperature: Not recommended.
Frozen (reconstituted): Discard immediately.
Power outages and emergency situations
Power outages happen. They might last hours or days. Knowing what to do with your semaglutide during and after an outage can save you hundreds of dollars and keep your treatment on track.
Short outages (under 4 hours)
Keep the refrigerator door closed. A closed refrigerator maintains safe temperatures for approximately 4 hours without power. Your semaglutide will be fine. Do not open the door to check on it. Every opening lets cold air escape and shortens the safe window.
Extended outages (4-24 hours)
After 4 hours, refrigerator temperatures start to climb above the safe zone. If you have a cooler and ice packs, transfer your semaglutide to the cooler. Place a barrier between the medication and the ice to prevent freezing. A digital thermometer in the cooler helps you monitor conditions.
If you do not have a cooler, keep the refrigerator door closed as long as possible and check the temperature once power returns. If the thermometer shows the fridge stayed below 46 degrees Fahrenheit throughout, the medication is fine. If it climbed above 46 but stayed below 86, the room temperature clock has started for your specific product (56 days for Ozempic, 28 for Wegovy).
Prolonged outages (24+ hours)
In multi-day outages, maintaining cold chain for injectable medication becomes genuinely difficult. Contact your pharmacy or healthcare provider for guidance. Some insurance plans and pharmacies will replace medication that was damaged due to documented power outages. Keep records of the outage duration and any temperature readings you captured.
If you also use other GLP-1 medications or peptides, the storage challenge compounds. Products like tirzepatide have their own refrigeration requirements, and managing multiple temperature-sensitive medications during an outage requires careful triage.
Compounded versus brand-name: the storage showdown
The practical storage differences between compounded and brand-name semaglutide are significant enough to influence which form works best for your situation.
Brand-name advantages
Extensive stability data. FDA-reviewed storage guidelines. Consistent preservative systems across batches. Room temperature tolerance validated through rigorous testing. Longer overall shelf life. Pre-filled pen packaging that minimizes contamination risk.
The reliability of brand-name storage data cannot be overstated. When Novo Nordisk says Ozempic is good for 56 days at room temperature, that claim is backed by accelerated stability studies, real-time stability data, and post-market surveillance. You can trust the number.
Compounded advantages
Customizable concentrations. Potentially lower cost. Availability during brand-name shortages. Access to formulations that brand-name products do not offer, such as combination products containing semaglutide with B12 or semaglutide with glycine for enhanced tolerability.
The tradeoff is storage uncertainty. Without the extensive testing that brand-name products undergo, you are relying on your pharmacy BUD assignment and general compounding guidelines. Some pharmacies are excellent. Others assign conservative BUDs because they have not invested in product-specific stability testing. Understanding the reputation and testing standards of your specific compounding pharmacy matters more for storage than it does for almost any other aspect of compounded medication use.
Which matters more: storage convenience or cost
For people who travel frequently, live in hot climates, or have unreliable home refrigeration, the generous room temperature allowance of brand-name products provides real peace of mind. For people with stable home environments and reliable cold storage, the cost savings of compounded semaglutide may outweigh the slightly more demanding storage requirements.
Neither choice is wrong. But making the decision without understanding the storage implications is a mistake. Factor storage into your total cost of ownership. A cheaper vial that loses 20% of its potency due to suboptimal storage is not actually cheaper. For detailed pharmacy reviews and comparisons, resources on providers like Olympia pharmacy semaglutide, Direct Meds semaglutide, and BPI Labs semaglutide can help you evaluate vendors based on their storage and shipping practices as well as product quality.
How storage affects your semaglutide results
You started semaglutide expecting results. Weight loss. Better blood sugar control. Reduced appetite. But what if your storage habits are quietly undermining those results?
The gradual potency loss problem
Semaglutide does not go from 100% potency to zero overnight. It degrades gradually. A vial stored at slightly too warm a temperature might lose 1 to 2% of potency per week. Over the course of a 30-day vial, that adds up to 5 to 8% loss. You probably would not notice a 5% reduction in a single dose. But over weeks of using suboptimally stored medication, the cumulative effect can look like the medication "stopped working."
This is exactly why some people experience a stall in weight loss at four weeks on semaglutide or wonder why they are not losing weight on semaglutide. Before assuming the medication is not working or that you need a higher dose, verify your storage conditions. A simple refrigerator thermometer check can rule out or confirm whether suboptimal storage is contributing to lackluster results.
The dose escalation timing factor
Storage quality intersects with dose escalation in a subtle way. If you are using degraded medication at a lower dose and not seeing results, you might move up to the next dose sooner than necessary. But the problem was never the dose. It was the potency. Moving up means more side effects for the same effective dose you should have been getting at the lower level.
Understanding how long semaglutide takes to work is only useful if the medication you are taking is actually at full strength. Storage is the hidden variable that most timeline discussions completely ignore. For a detailed look at realistic expectations with properly stored medication, our semaglutide one-month results guide provides benchmarks you can compare against. People tracking their semaglutide before-and-after progress sometimes attribute inconsistent results to tolerance when degraded medication is the actual explanation.
The cost of waste
Bad storage does not just reduce potency. It can force you to discard medication entirely. A brand-name Ozempic pen that costs $900+ per month, thrown away because it froze in the back of the fridge. A compounded vial at $200+ discarded because the power went out and nobody noticed for two days. These losses add up fast.
The peptide cost calculator can help you understand the true cost of your protocol. But that calculation only matters if every vial you buy maintains its potency throughout its use period. Storage is not a minor detail. It is a core part of the financial equation.
Storage and your semaglutide diet protocol
Here is something most storage guides never mention: your diet and nutrition protocol while on semaglutide directly interacts with how much the storage timeline matters to you.
If you follow a structured semaglutide diet plan, you are likely using each dose as part of a carefully calibrated approach. Degraded medication disrupts that calibration. The appetite suppression you are counting on to maintain your planned food intake becomes unreliable when potency drops. You end up hungry between meals, break the plan, and blame yourself when the real issue was storage.
People who avoid certain foods on semaglutide to manage gastrointestinal side effects might notice those side effects shifting if they switch between fresh and degraded vials. The breakdown products can affect GI tolerance differently than intact semaglutide. If your side effect pattern suddenly changes without a dose change, check your vial dates before adjusting your diet.
For those pursuing weight loss on semaglutide without exercise, the medication carries even more of the weight loss burden, which makes consistent potency even more critical. And people supplementing with protein shakes alongside GLP-1 therapy are investing in their nutrition. Wasting that investment on degraded medication that cannot properly suppress appetite defeats the purpose.
Other GLP-1 storage considerations
Semaglutide is not the only GLP-1 receptor agonist with specific storage requirements. If you use or are considering other options in this class, understanding the storage landscape helps you compare across the board.
The timeline for GLP-1 medications to start working varies by compound, and so do the storage requirements. Tirzepatide refrigeration follows a different protocol than semaglutide. Oral options like GLP-1 patches and oral GLP-1 liquids have their own storage profiles entirely.
For tracking your overall progress with any GLP-1 medication, tools like the GLP-1 plotter and the GLP-1 companion app can help you correlate changes in results with potential storage issues. If you notice your semaglutide before-and-after progress stalling, cross-referencing with your storage timeline can identify whether degraded medication is a contributing factor.
Side effects like GLP-1 fatigue and GLP-1 headaches may present differently with degraded medication. Some users report that switching to a fresh vial after unknowingly using a degraded one results in a sudden increase in both efficacy and side effects, essentially catching up to the dose they should have been experiencing all along.
Knowing the best time to take your GLP-1 shot and where to inject it are important parts of your protocol. But none of those details matter if the medication you are injecting has lost potency due to poor storage. Get the storage right first. Everything else builds on that foundation.
Setting up the perfect semaglutide storage system
Stop thinking about storage as something you do after you buy the medication. Set up a proper system once and never worry about it again.
Step 1: get a dedicated thermometer
A digital min/max thermometer placed on the shelf where you store your semaglutide. Under $10. Check it weekly. Adjust your fridge if readings fall outside 36 to 46 degrees Fahrenheit.
Step 2: choose the right shelf
Middle shelf, center to rear of the refrigerator. Away from the door. Away from walls bordering the freezer. Away from the crisper drawer air vents (which blow extra-cold air). The goal is the most temperature-stable location in the fridge.
Step 3: create a barrier
Place your semaglutide in a small sealed container or bag inside the refrigerator. This adds one more layer of temperature buffering and protects the medication from accidental contact with frozen items, spills, or food contaminants.
Step 4: label everything
Write the date of first use (for brand-name pens) or the reconstitution date (for lyophilized vials) directly on the product with a permanent marker. Do the same for the BUD on compounded vials if it is not already clearly labeled. Do not rely on memory or apps. Write it on the vial.
Step 5: set disposal reminders
Create a phone reminder for the disposal date. 56 days after first use for Ozempic. 28 days for Wegovy. 28 days after reconstitution for lyophilized. Whatever your product specific timeline is, set the reminder on day one and forget about it until the alert fires.
Step 6: keep a backup plan
A small insulated cooler bag and two gel packs in your freezer give you an emergency cold chain for power outages, travel, or any other disruption. This $15 investment can save hundreds of dollars in medication.
Semaglutide storage and injection timing
Your storage routine and your injection routine should work together. The way you handle semaglutide during the injection process affects both the comfort of the injection and the long-term stability of the remaining medication.
Should you inject cold semaglutide
Cold injections are more uncomfortable than room temperature ones. The viscosity of the solution increases at lower temperatures, making the injection feel thicker and more painful. Many users find that removing the pen or vial from the refrigerator 15 to 30 minutes before injection allows it to warm slightly, reducing discomfort.
This brief warm-up period is perfectly safe. Fifteen to thirty minutes at room temperature causes no measurable degradation. Just remember to return the medication to the fridge promptly after your dose.
Injection site and storage
This might seem unrelated, but the best injection site for semaglutide can be influenced by medication temperature. Cold solution injected into fatty tissue in the abdomen may cause more stinging than the same solution injected at room temperature into the thigh. If you are experiencing injection site discomfort, try the brief warm-up technique before switching injection sites.
For people using the standard GLP-1 injection technique, consistency in both storage and injection routine leads to the most predictable absorption and the fewest side effects.
How long does semaglutide really last: what the research says
Manufacturer guidelines are conservative by design. They are based on guaranteed minimum potency at the end of the stated period under specified conditions. The actual stability of semaglutide often exceeds these published numbers. But using medication beyond its stated shelf life is a personal risk assessment, not a recommendation.
What stability studies show
Pharmaceutical stability studies involve storing the product at controlled temperatures and testing potency at regular intervals. Products must maintain at least 90% of labeled potency throughout the stated shelf life. This means that on the last day of the shelf life, your medication still contains at least 90% of the active ingredient listed on the label.
In practice, many products retain 95% or more of potency well beyond their labeled expiration or beyond-use dates. The manufacturer knows this but cannot extend the label claim without additional regulatory submissions and testing.
The conservative buffer
Both Novo Nordisk (for Ozempic and Wegovy) and compounding pharmacies build conservative buffers into their storage timelines. An Ozempic pen that the manufacturer says is good for 56 days after first use has likely been tested and shown stability beyond that point. The 56-day claim is a guaranteed minimum, not the point at which the medication suddenly becomes dangerous.
This does not mean you should use semaglutide beyond its stated shelf life. It means that if you accidentally used your Ozempic on day 58 instead of discarding it on day 56, you almost certainly did not inject degraded medication. But making a habit of pushing past the stated limits erodes the safety margin that manufacturers intentionally built in.
Accelerated stability data
Pharmaceutical companies also conduct accelerated stability studies, where they store the product at higher temperatures (typically 40 degrees Celsius and 75% relative humidity) to predict long-term stability in a shorter timeframe. For semaglutide, these studies generally show that the molecule is more robust than many other peptide drugs, thanks to its fatty acid modification and engineered amino acid substitutions.
This is why semaglutide has more generous room temperature allowances compared to other peptides that researchers might be familiar with. If you work with peptides like BPC-157 or tirzepatide, you will notice that semaglutide is generally more forgiving in terms of storage flexibility. Understanding how long semaglutide remains good in various conditions gives you practical flexibility without compromising safety.
Semaglutide storage for specific populations
Certain groups face unique storage challenges that generic guidelines do not fully address.
Women and menstrual cycle considerations
Some women report that semaglutide affects their menstrual cycle, particularly during the first few months. If you are tracking cycle changes alongside your medication use, inconsistent potency from storage issues can create confusing data. A vial that has lost 10% of its potency might produce different hormonal effects than a fresh vial, making it harder to determine whether the medication itself or the degradation is affecting your cycle.
Post-surgical patients
People who need to resume semaglutide after surgery face a specific storage challenge. If you paused your medication for a surgical procedure, the pen or vial sitting in your fridge has been aging during your recovery. Check the dates before resuming. A pen that has been sitting for six weeks post-first-use has exceeded the 56-day Ozempic window or the 28-day Wegovy window.
Combination therapy users
Researchers using semaglutide alongside other medications face compound storage challenges. If you are combining with phentermine and semaglutide together or comparing phentermine versus semaglutide options, each medication has its own storage requirements that need to be managed independently. Keeping multiple temperature-sensitive medications organized requires labeling discipline and fridge space planning.
For those on compounded combination products like semaglutide with methylcobalamin, the additional ingredients may alter the stability profile. The B12 component, for instance, is itself light-sensitive, which reinforces the importance of keeping these formulations in opaque containers or their original packaging within the refrigerator.
People considering breastfeeding
Women who are evaluating GLP-1 use while breastfeeding should be especially cautious about medication integrity. If the decision involves weighing risk versus benefit, that calculation only works if the medication is at full potency. Using degraded semaglutide introduces risk without the corresponding benefit, making the risk-benefit analysis meaningless.
Special situations and edge cases
What if I accidentally left my semaglutide out overnight
Do not panic. One night at room temperature is well within the tolerance of all brand-name semaglutide products. Check the temperature where it was sitting. If it was below 86 degrees Fahrenheit, the medication is fine. Put it back in the fridge and continue using it normally. The room temperature clock has started, but one night does not meaningfully reduce the overall shelf life.
For compounded semaglutide, contact your pharmacy. They may advise continued use if your room temperature was moderate, or they may recommend discarding depending on their specific formulation stability data.
What if my semaglutide arrived warm
Mail-order pharmacies ship semaglutide with cold packs. Sometimes those cold packs are depleted by the time the package arrives, especially during summer months. If the medication arrived warm to the touch but the cold packs still have some coolness to them, the product is very likely fine. Refrigerate immediately.
If the package arrived with cold packs completely warm and the medication feels warm to the touch, contact the pharmacy for a replacement. Most reputable pharmacies will replace medication that arrived outside temperature specifications.
What if I have two partially used vials
Never combine medication from two different vials, even if they are the same product at the same concentration. Each vial has its own contamination timeline and beyond-use date. Combining them mixes potentially degraded or contaminated medication with fresher product.
Use the older vial first. When it is empty or expired, move to the newer one. This first-in, first-out approach minimizes waste while maintaining safety.
What about semaglutide compound formulations
Compounded semaglutide is increasingly available in combination formulations. Products containing semaglutide with methylcobalamin or other additives may have different storage requirements than plain semaglutide. The additional ingredients can either improve or reduce overall stability depending on their chemical properties. Always follow the specific BUD and storage instructions for your exact formulation.
Frequently asked questions
Can I put semaglutide back in the fridge after it has been at room temperature?
Yes, but it does not reset the clock. If your Ozempic was out for 3 days and you return it to the fridge, you still have 53 days remaining from the original removal date. Refrigeration slows degradation but does not undo it. For brand-name products, the manufacturer room temperature timeline applies regardless of whether you alternate between fridge and counter.
Does semaglutide go bad faster once the pen has been used?
The first use introduces potential contamination through the needle seal, which is why the post-use storage window (56 days for Ozempic, 28 for Wegovy) is shorter than the overall unopened shelf life. The semaglutide molecule itself does not degrade faster after first use, but the contamination risk changes the safety calculation. For proper dosage measurement, using within the stated window ensures both potency and sterility.
Is it safe to use semaglutide that has been stored at 50 degrees Fahrenheit?
Yes. Fifty degrees Fahrenheit (10 degrees Celsius) falls within the acceptable range for Wegovy (46-86F) and is just slightly above the refrigeration range for Ozempic (36-46F). At this temperature, degradation is minimal and the medication remains effective. However, 50 degrees is warmer than ideal for long-term storage, so do not make it your permanent storage temperature.
How can I tell if my compounded semaglutide has expired?
Check the beyond-use date on the label. If no BUD is written, call your pharmacy. Visual inspection, specifically checking for cloudiness, color changes, or particles, can catch advanced degradation. But potency loss can occur without any visible signs. When in doubt, ask your pharmacy or discard the vial. Understanding the risks of using expired semaglutide helps inform this decision.
Does the semaglutide dosage affect how long it lasts in the fridge?
The dosage you draw does not affect the remaining medication shelf life. A vial at any dosage level follows the same storage timeline whether you take 10 units or 50 units per dose. What matters is the date of first puncture (for vials) or first use (for pens), the storage temperature, and the product specific shelf life. Use our semaglutide dosage calculator to ensure you are drawing the right amount from each vial.
Can I freeze semaglutide to make it last longer?
No. Never freeze injectable semaglutide in any form. Freezing destroys the peptide structure through ice crystal formation. This applies to Ozempic, Wegovy, compounded formulations, and reconstituted lyophilized solutions. Frozen semaglutide must be discarded even if it looks normal after thawing.
What happens if I use semaglutide that was stored incorrectly?
Degraded semaglutide may simply be less effective, meaning reduced appetite suppression and slower results. In more advanced degradation, breakdown products could cause injection site reactions, increased nausea, or other unexpected side effects. If you suspect you have been using improperly stored medication, switch to a fresh supply and monitor your response.
How long can semaglutide sit out during an injection?
The 15 to 30 minutes most people take from removing semaglutide from the fridge, preparing the injection site, injecting, and returning the medication is completely fine. Even an hour is insignificant. The room temperature timelines (28-56 days) are measured in weeks, not minutes. Focus on returning it to the fridge within a reasonable time and do not stress about brief handling periods.
External resources
For researchers serious about optimizing their semaglutide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based storage guides, proven protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your refrigerator stay calibrated, your peptides stay potent, and your storage habits stay impeccable.