Feb 28, 2026

A single statistic has been circulating through weight loss forums, Reddit threads, and health news outlets for months now. A 266% increased risk of deep vein thrombosis. That number comes from a meta-analysis of 21 semaglutide clinical trials involving over 26,000 participants, and it has understandably alarmed anyone currently injecting this GLP-1 receptor agonist for weight loss or blood sugar management.
But that statistic does not tell the whole story. Not even close.
The relationship between semaglutide and blood clots is far more complicated than a single relative risk number suggests. Newer research involving over 70,000 participants across 39 clinical trials paints a nuanced picture, one where certain types of clot risk may increase while overall cardiovascular outcomes actually improve. Real-world data from more than 540,000 patients tells yet another story, suggesting that semaglutide users may actually develop fewer blood clots than people on other diabetes medications. And at the molecular level, GLP-1 receptor agonists appear to directly inhibit platelet aggregation, reduce inflammatory markers, and improve endothelial function, all of which should theoretically protect against clots.
So what is actually happening? This guide examines every major study, case report, and biological mechanism connecting semaglutide to blood clot risk. Whether you are currently using semaglutide, considering starting it, or simply trying to separate fact from fear, the evidence may surprise you. SeekPeptides has assembled the most comprehensive breakdown of this topic available anywhere, pulling from peer-reviewed research, FDA regulatory filings, and clinical trial data that most articles never mention.
Understanding the semaglutide blood clot concern
Blood clots form when blood thickens and clumps together inside a vein or artery. Two types matter most in this discussion. Deep vein thrombosis, or DVT, occurs when a clot forms in a deep vein, usually in the legs. Pulmonary embolism, or PE, happens when a clot breaks free and travels to the lungs. Together, these are called venous thromboembolism, or VTE, and they can be life-threatening.
The concern about semaglutide specifically traces back to a 2021 meta-analysis published in the Endocrine Journal. Researchers pooled safety data from all available semaglutide randomized controlled trials, 10 PIONEER trials testing oral semaglutide and 11 SUSTAIN trials testing the injectable form. Across 26,436 total participants, they identified 252 different serious adverse events and calculated the relative risk for each one.
Deep vein thrombosis stood out.
The relative risk came in at 3.66, with a 95% confidence interval of 1.09 to 12.25 and a p-value of 0.036. Statistically significant. That translates to roughly a 266% increase compared to control groups. The I-squared value was zero, meaning there was no inconsistency between the trials, every study pointed in the same direction.
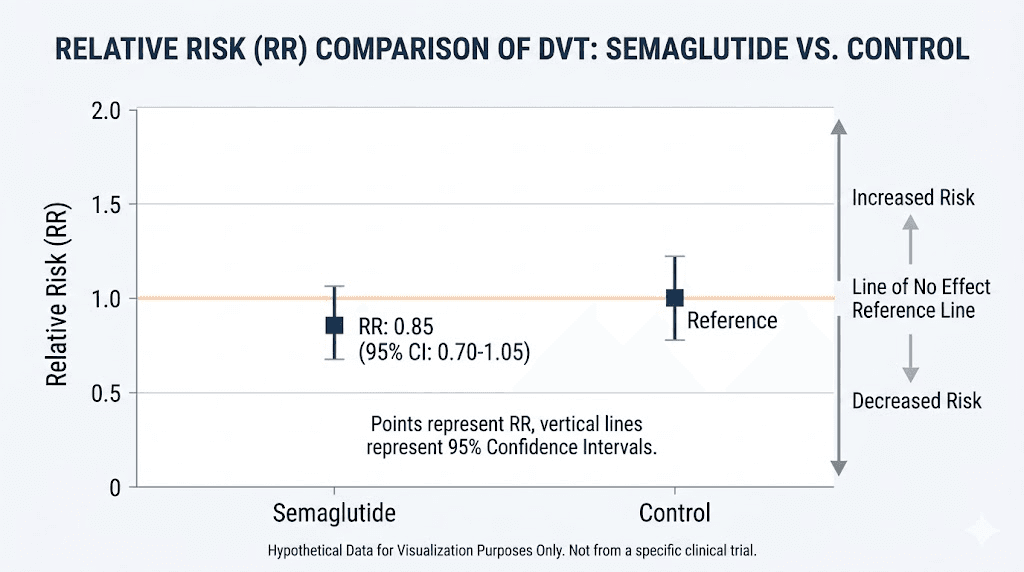
But context matters enormously here. The absolute number of DVT events across all 21 trials was small, which is exactly why the confidence interval stretches so wide, from 1.09 to 12.25. A true relative risk anywhere in that range would be considered statistically significant, but the practical implications differ dramatically between the low and high ends. When you are dealing with rare events in clinical trials, even a handful of additional cases can produce alarming-looking relative risk numbers.
The same meta-analysis found something equally important that rarely gets mentioned. Overall serious adverse events were actually reduced in semaglutide users compared to controls, with a relative risk of 0.92 (p = 0.005). Atrial fibrillation risk dropped by 31%. And cerebrovascular accidents, meaning strokes, trended toward a 48% reduction, though this did not quite reach statistical significance.
The picture is not as simple as "semaglutide causes blood clots." It is more accurate to say that semaglutide appears to shift certain cardiovascular risks in different directions simultaneously.
What the clinical research actually shows
Three categories of evidence exist regarding semaglutide and blood clots: randomized controlled trials, real-world observational studies, and individual case reports. Each tells a different part of the story, and understanding where they agree and disagree is critical for assessing actual risk.
Randomized controlled trial evidence
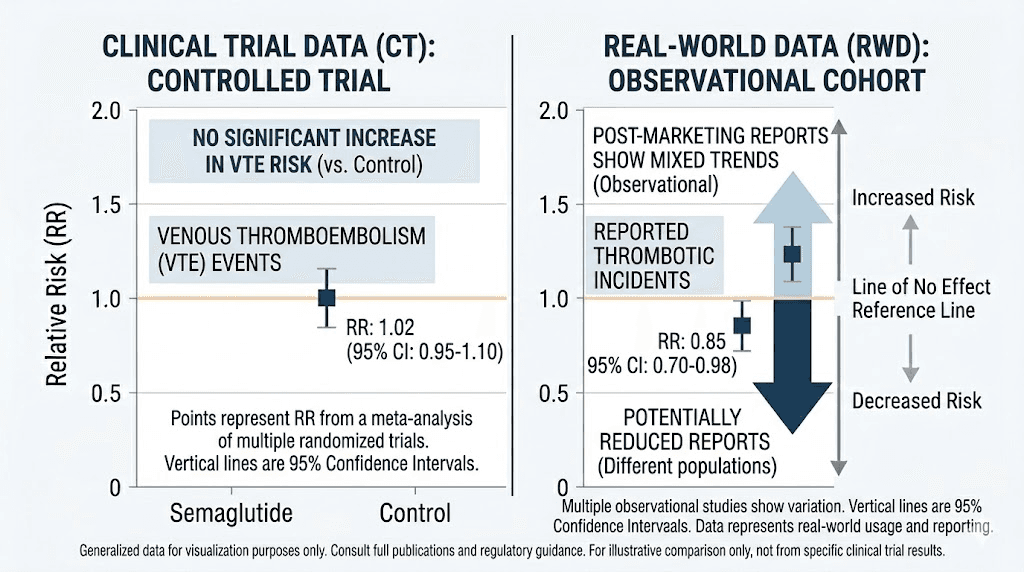
The most recent and comprehensive meta-analysis comes from the Journal of the American Heart Association, published in 2025. This study expanded far beyond just semaglutide, analyzing 39 randomized controlled trials covering all GLP-1 receptor agonists with 70,499 total participants.
The findings confirmed an association between GLP-1 medications and DVT, with an odds ratio of 1.64 (95% CI: 1.14-2.36). In absolute terms, this translates to approximately 25 additional DVT events per 10,000 person-years. For every 10,000 people taking GLP-1 medications for a year, roughly 25 more would experience DVT compared to those not taking the medication.
One finding from this analysis deserves special attention. Duration matters.
For treatment periods exceeding 1.5 years, the odds ratio jumped to 2.32 (95% CI: 1.49-3.60). The longer someone uses a GLP-1 receptor agonist, the higher the apparent DVT risk becomes. This duration-dependent relationship held up specifically in cardiovascular outcome trials, where the odds ratio reached 2.18 (95% CI: 1.36-3.49).
Critically, neither the original 2021 meta-analysis nor the 2025 JAHA study found a significant association between GLP-1 medications and pulmonary embolism. The increased risk appears specific to DVT, not to the more dangerous downstream complication of clots traveling to the lungs.
The major cardiovascular safety trials
The SUSTAIN-6 trial enrolled 3,297 patients with type 2 diabetes and high cardiovascular risk, following them for two years on semaglutide 0.5 mg or 1.0 mg weekly versus placebo. The primary finding was a 26% reduction in major adverse cardiovascular events (MACE), driven largely by a 39% reduction in nonfatal stroke. Heart attack rates trended lower but did not reach statistical significance.
DVT was not a pre-specified endpoint in SUSTAIN-6. This is important. The trial was designed to detect differences in heart attacks, strokes, and cardiovascular deaths, not blood clots. DVT events were captured as part of general safety monitoring, but the trial was not powered to detect meaningful differences in such a rare outcome.
The SELECT trial was even larger, enrolling 17,604 patients with obesity and pre-existing cardiovascular disease but without diabetes. At the higher weight loss dose of 2.4 mg weekly, semaglutide reduced MACE by 20% (HR 0.80, p less than 0.001) and all-cause mortality by 19%. Serious adverse events were fewer in the semaglutide group (33.4% vs 36.4%), including cardiac disorders specifically (11.5% vs 13.5%).
Again, DVT and PE data were not specifically broken out in the published SELECT results. The trial showed clear cardiovascular benefit but was not designed to answer the blood clot question directly.

Real-world observational evidence
Here is where the story takes a surprising turn.
A propensity-score matched study presented at the American Society of Hematology Annual Meeting in 2024 analyzed 656,588 patients with type 2 diabetes. After matching 168,428 patients in each group, GLP-1 receptor agonist users had a VTE rate of 11.0 per 1,000 patient-years compared to 12.9 per 1,000 patient-years in those taking DPP-4 inhibitors (a different class of diabetes medication). That is an 18% lower risk of blood clots in the GLP-1 group.
A separate target trial emulation study published in Blood Advances confirmed this finding in an even larger population. Among 540,258 patients (270,129 in each matched group), those on GLP-1 medications had fewer VTE events at every time point measured. At one year, pulmonary embolism occurred in 3.1 per 1,000 GLP-1 users versus 3.9 per 1,000 in the comparison group. DVT rates were 4.2 versus 5.0 per 1,000. Overall VTE rates were 6.5 versus 7.9 per 1,000, a 20% reduction.
These are not small studies. Combined, they represent over 1.2 million patients in real-world clinical practice, and they both show that people taking GLP-1 medications develop fewer blood clots than comparable patients on other treatments.
Individual case reports
Several case reports have documented blood clots in patients using semaglutide or related GLP-1 medications. While case reports cannot establish causation, they provide important clinical observations.
A 59-year-old woman with a BMI of 31.5 and type 2 diabetes developed intrahepatic portal vein thrombosis after four months on semaglutide 1 mg weekly. She had lost 5 kg and presented with lower back pain, nausea, and elevated inflammatory markers. Notably, her thrombophilia screening revealed a JAK2 V617F mutation, a known risk factor for unusual-site blood clots. She recovered fully with anticoagulation treatment.
A 57-year-old woman with a BMI of 45.5 and prior breast cancer developed an occlusive DVT of her left subclavian vein after eight months on tirzepatide 10 mg weekly (tirzepatide is a dual GLP-1/GIP agonist). Her complete hypercoagulability workup was negative, meaning no underlying clotting disorder could be identified. Using the Naranjo adverse drug reaction scale, researchers scored the association at 6 out of 13, indicating a "probable" relationship between the medication and the clot.
A third case involved a 20-year-old male who developed extensive lower extremity DVT while on tirzepatide.
These cases are concerning but must be interpreted carefully. Millions of people take semaglutide and tirzepatide worldwide. In any large population, blood clots will occur regardless of medication use, particularly in people with obesity and diabetes, two conditions that independently increase clot risk.
The scientific paradox: conflicting evidence explained
A genuine scientific paradox exists in the semaglutide blood clot data. Clinical trial meta-analyses show increased DVT risk. Real-world observational studies show decreased VTE risk. Both use large datasets. Both are published in respected journals. How can they both be right?
Several explanations may account for this discrepancy.
The small numbers problem
Clinical trials capture rare events in controlled settings. When the absolute number of DVT cases is small, even a few additional events can produce dramatic-looking relative risk numbers. The 2021 meta-analysis found a relative risk of 3.66, but the 95% confidence interval stretches from 1.09 to 12.25. This extreme width tells us the estimate is imprecise. The true risk could be barely above baseline or it could be substantially elevated, the data simply cannot pin it down with precision.
Different comparison groups
Clinical trials compared semaglutide to placebo or active comparators within the same trial. Real-world studies compared GLP-1 users to DPP-4 inhibitor users. The comparison group matters enormously. DPP-4 inhibitors may have their own effect on coagulation that makes GLP-1 medications look protective by comparison. Or placebo-treated patients in clinical trials may have different baseline risk profiles than real-world patients on alternative medications.
Population differences
Clinical trial participants are carefully selected. They tend to have more comorbidities and higher cardiovascular risk than the general population of semaglutide users. Real-world populations include a broader mix of patients, some healthier and some sicker than typical trial participants. These population differences can shift risk estimates in either direction.
Duration and monitoring effects
Most clinical trials lasted one year or less. The JAHA meta-analysis showed that DVT risk increases with duration beyond 1.5 years, suggesting a time-dependent relationship that shorter trials might underestimate. Conversely, real-world patients may receive more routine monitoring and earlier intervention, potentially catching and preventing clots before they become clinically significant events.
The dehydration hypothesis
Clinical trial protocols strictly define medication adherence and dosing. In real-world practice, patients who experience severe gastrointestinal side effects may adjust their behavior more freely, drinking more fluids, reducing doses, or taking breaks. If dehydration from GI side effects is the primary mechanism linking semaglutide to DVT, then real-world patients who self-manage these symptoms more aggressively might be better protected.
How semaglutide interacts with your blood
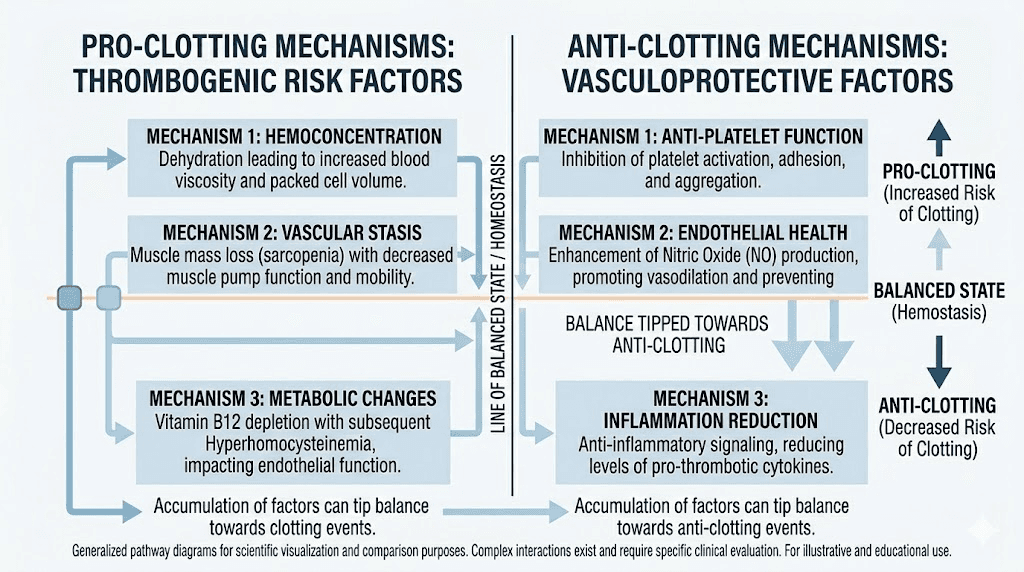
Understanding the biological mechanisms helps explain why the evidence points in two directions simultaneously. Semaglutide affects blood clotting through multiple pathways, some that could increase risk and others that clearly protect against it.
Mechanisms that could increase clot risk
Dehydration from gastrointestinal effects. This is the most commonly cited mechanism and arguably the most plausible. Semaglutide significantly increases the risk of diarrhea (relative risk 2.66 in the meta-analysis), and nausea affects up to 40% of users, particularly during the early weeks of treatment. Vomiting compounds the problem. When the body loses fluid through these routes without adequate replacement, blood becomes more concentrated. This hemoconcentration increases viscosity, making blood thicker and more prone to clotting. Dehydration itself trended toward a significant increase in the semaglutide trials (RR 2.16, p = 0.071).
Rapid weight loss and muscle wasting. Semaglutide produces substantial weight loss, often 15-20% of body weight over 68 weeks. Some of this weight comes from muscle mass, not just fat. The muscles in the legs serve as pumps that push blood back toward the heart. When these muscles shrink, the pumping action weakens, allowing blood to pool in the deep veins of the legs. Blood stasis is one of the three components of Virchow triad, the classic framework for understanding why blood clots form.
Vitamin B12 depletion. Semaglutide reduces stomach acid production, which impairs the absorption of vitamin B12. Low B12 levels lead to elevated homocysteine, an amino acid that damages blood vessel walls and promotes clot formation. This is not a theoretical concern. B12 deficiency is well-documented in long-term GLP-1 users, which is one reason why many compounded versions include B12 in the formulation.
Reduced physical activity. When nausea, fatigue, and dizziness from semaglutide side effects keep someone on the couch instead of moving, blood flow slows throughout the body. Immobility is one of the oldest and best-established risk factors for DVT. Even healthy people develop blood clots after long flights or periods of bed rest. Semaglutide side effects that reduce activity could contribute to the same mechanism.
Mechanisms that protect against clots
Direct platelet inhibition. This is perhaps the most compelling protective mechanism. A controlled study published in 2022 demonstrated that GLP-1 receptors are expressed directly on human platelets. When liraglutide (a closely related GLP-1 agonist) was administered for just two weeks, it reduced thromboxane-induced platelet aggregation by 54% (95% CI: 18%-90%, p = 0.003). This effect occurred before any weight loss happened, confirming it was a direct pharmacological action, not a secondary consequence of losing weight. Researchers confirmed it was GLP-1 receptor-dependent by showing that the effect reversed when they blocked the receptor with a specific antagonist.
Platelets are the blood cells primarily responsible for forming clots. A 54% reduction in their ability to clump together is substantial, comparable to the effect of low-dose aspirin.
Reduced platelet activation markers. A study of 30 patients with type 2 diabetes treated for 8 weeks showed measurable reductions in CD62p (a marker of platelet activation) and PAC-1 (a marker of glycoprotein IIb/IIIa complex activation, the final common pathway of platelet aggregation). Fibrinogen levels, which directly influence clot formation, also decreased. Standard coagulation tests like PT, APTT, and INR remained unchanged, suggesting the anti-clotting effect works through platelet function rather than the coagulation cascade.
Nitric oxide enhancement. GLP-1 receptor agonists activate endothelial nitric oxide synthase (eNOS) through the AMPK/Akt signaling pathway. Nitric oxide is a powerful vasodilator that also inhibits leukocyte adhesion and suppresses platelet aggregation. When blood vessels produce more nitric oxide, they actively resist clot formation. This mechanism contributes to the broader cardiovascular benefits observed in the SELECT and SUSTAIN-6 trials.
Anti-inflammatory effects. Chronic inflammation damages blood vessel walls and creates a pro-coagulant environment. Semaglutide suppresses NLRP3 inflammasome activation, reduces proinflammatory cytokines and chemokines, and decreases adhesion molecules that attract immune cells to vessel walls. Lower inflammation means healthier blood vessels that are less likely to trigger clot formation.
Epicardial fat modification. A translational study of 21 patients with type 2 diabetes and obesity found that six months of semaglutide treatment changed what their epicardial fat tissue secreted. Specifically, semaglutide increased secretion of gelsolin, a protein with antithrombotic properties. Neutrophil adhesion to aortic endothelial cells decreased by approximately 15%, and neutrophil migration dropped by roughly 30%. These changes in the fat tissue surrounding the heart create a local anti-thrombotic environment.
PAI-1 modulation. Plasminogen activator inhibitor-1 (PAI-1) is the body primary brake on clot dissolution. When PAI-1 levels are high, the body cannot break down clots effectively. GLP-1 receptor agonists may reduce PAI-1 expression, enhancing the natural fibrinolytic system that dissolves clots before they become dangerous. Since PAI-1 exists in significant excess relative to tissue plasminogen activator (tPA) in plasma, even modest reductions could meaningfully improve the body ability to clear clots.
Risk factors that matter most
Not everyone on semaglutide faces the same blood clot risk. Understanding which factors elevate your baseline risk can help you and your healthcare provider make informed decisions about monitoring and prevention.
Pre-existing risk factors
Obesity. Ironically, the very condition semaglutide treats is itself a major risk factor for blood clots. People with a BMI over 30 have a significantly elevated baseline risk of VTE. Fat tissue produces inflammatory mediators and coagulation factors that create a pro-thrombotic state. As semaglutide reduces weight, this risk should theoretically decrease, but the transition period, where rapid weight loss causes fluid shifts and muscle wasting, may temporarily increase risk before the long-term protective effects kick in.
Type 2 diabetes. Diabetes itself creates a hypercoagulable state. Elevated blood sugar damages blood vessel walls, increases platelet reactivity, and impairs fibrinolysis. People with type 2 diabetes have roughly double the risk of VTE compared to non-diabetic individuals. Since many semaglutide users have diabetes, separating the medication risk from the disease risk becomes extremely difficult.
Personal or family history of blood clots. If you or a close family member has previously experienced DVT, PE, or other thrombotic events, your baseline risk is already elevated. Inherited thrombophilias like Factor V Leiden, prothrombin G20210A mutation, or antithrombin deficiency multiply the risk further. The portal vein thrombosis case report mentioned earlier occurred in a patient who was later found to carry a JAK2 V617F mutation.
Hormonal factors. Estrogen-containing oral contraceptives and hormone replacement therapy significantly increase blood clot risk. Women taking semaglutide who are also on hormonal birth control or HRT face compounded risk factors.
Cancer history. Cancer creates a hypercoagulable state through multiple mechanisms, including tumor-derived tissue factor, inflammatory cytokines, and cancer treatment effects. The tirzepatide DVT case report involved a patient with prior breast cancer. Anyone with active or recent cancer should discuss VTE risk specifically with their oncologist before starting GLP-1 therapy.
Semaglutide-specific risk factors
Severe GI side effects. Users who experience persistent diarrhea, vomiting, or severe nausea are at higher risk because of dehydration. The meta-analysis showed diarrhea risk increased by 166% with semaglutide. If you cannot keep fluids down or are having multiple loose stools daily, the resulting hemoconcentration could create clotting conditions.
Rapid weight loss without exercise. Losing weight quickly while remaining sedentary is a concerning combination. The muscle loss reduces venous pump function, and the lack of movement allows blood to pool. Maintaining physical activity during semaglutide treatment is important for multiple reasons, and DVT prevention is one of them.
Long-term use beyond 1.5 years. The JAHA meta-analysis specifically identified treatment duration as a modifier of DVT risk. The odds ratio more than doubled when treatment exceeded 1.5 years (OR 2.32 vs 1.64 overall). This does not necessarily mean you should stop after 18 months, but it does mean that long-term users should be more vigilant about monitoring and prevention strategies.
Inadequate hydration. This seems obvious but cannot be overstated. Many semaglutide users significantly reduce their food and fluid intake because of appetite suppression. The appetite reduction that makes semaglutide effective for weight loss can also lead to dangerously low fluid intake if users are not deliberately maintaining hydration. Your body needs adequate water to maintain proper blood viscosity regardless of how hungry you feel.
Smoking. Tobacco damages the endothelial lining of blood vessels, which is one of Virchow triad components. Combined with any of the semaglutide-related risk factors, smoking creates a multiplicative rather than additive increase in clot risk.
Recent surgery or immobilization. Surgery is a well-known DVT risk factor. If you are scheduled for a procedure, discuss with your surgeon whether to continue or temporarily pause semaglutide. The dehydration from fasting before surgery, combined with post-operative immobility, could compound semaglutide-related risks.
DVT warning signs every semaglutide user should know
Recognizing blood clot symptoms early can be the difference between a manageable medical event and a life-threatening emergency. Every person using semaglutide should know these signs.
Deep vein thrombosis symptoms
DVT most commonly occurs in the legs, though it can develop in the arms, pelvis, or abdomen. The classic presentation includes swelling in one leg (not both, asymmetry is a key indicator), pain or tenderness that often starts in the calf and feels like cramping or soreness, redness or discoloration of the skin over the affected area, and warmth in the swollen area.
Some people notice their affected leg feels heavier than usual. Others describe a persistent aching that worsens with standing or walking. The pain typically increases when you flex your foot upward (dorsiflexion), a clinical sign called Homans sign, though this test is neither sensitive nor specific enough for diagnosis.
Not every DVT causes symptoms. Some are discovered incidentally during imaging for other reasons. But when symptoms appear, they demand immediate medical evaluation.
Pulmonary embolism symptoms
If a DVT breaks free and travels to the lungs, the symptoms are different and more urgent. Sudden shortness of breath, especially with exertion, is the most common. Sharp chest pain that worsens with deep breathing or coughing. Rapid heart rate. Coughing up blood, even small amounts. Feeling lightheaded or faint. Excessive sweating.
A massive PE can cause sudden cardiovascular collapse. This is a medical emergency requiring immediate 911 activation.
When to seek care
Any new, unexplained leg swelling or pain while on semaglutide warrants a same-day medical evaluation. Do not wait to see if it improves. DVT diagnosis typically involves a D-dimer blood test (which measures clot breakdown products) followed by duplex ultrasound if the D-dimer is elevated. Treatment with anticoagulation is highly effective when started promptly.
For any symptoms suggesting PE, do not drive yourself. Call emergency services.
How to reduce blood clot risk while using semaglutide
Regardless of whether semaglutide directly increases DVT risk or the apparent association reflects confounding factors, minimizing blood clot risk while on this medication is straightforward and backed by strong evidence.
Hydration is everything
This is the single most important protective measure. Aim for at least 64 ounces (eight cups) of water daily, and increase this amount if you are experiencing GI side effects like diarrhea or vomiting. Some semaglutide users find that their reduced appetite extends to water as well, meaning they simply forget to drink. Set reminders. Keep a water bottle visible at all times. Track your intake if needed.
Signs you are not drinking enough include dark urine, dry mouth, headaches, and dizziness when standing. If your urine is consistently light yellow to clear, you are probably adequately hydrated.
Electrolyte balance matters too. Prolonged diarrhea depletes sodium, potassium, and other electrolytes that affect blood viscosity and cardiovascular function. Consider adding electrolyte supplements or drinking electrolyte-enhanced water if GI symptoms persist.
Stay physically active
Movement is the most effective DVT preventive measure, and it costs nothing. Walking for 30 minutes daily activates the calf muscle pump that pushes blood through the deep veins of the legs. Even on days when fatigue or nausea makes vigorous exercise unappealing, light walking, calf raises, ankle circles, and leg stretches maintain venous blood flow.
Avoid prolonged sitting or standing. If your job requires long periods in one position, take breaks every hour to walk around. During long car rides or flights, flex your calves and ankles regularly. Compression stockings can help maintain venous return during extended periods of immobility.
Resistance training deserves special mention. Because semaglutide can cause muscle loss alongside fat loss, strength training serves double duty: it preserves the muscle mass that drives venous return AND it provides the movement needed to keep blood flowing. The dietary strategy should also prioritize adequate protein to support muscle maintenance.
Monitor B12 levels
Ask your doctor to check serum B12 and homocysteine levels periodically while on semaglutide. If B12 is low or homocysteine is elevated, supplementation is simple and inexpensive. Many compounding pharmacies now include B12 directly in their semaglutide formulations, which addresses this concern proactively.
Methylcobalamin (the active form of B12) is generally preferred over cyanocobalamin because it does not require conversion and is better utilized by the body. Typical supplementation ranges from 1,000 to 2,500 mcg daily when deficiency is present.
Manage GI side effects aggressively
Do not simply power through severe nausea, vomiting, and diarrhea. These side effects cause the dehydration that most plausibly links semaglutide to blood clot risk. If standard measures, such as eating small, bland meals, avoiding high-fat foods, and eating slowly, are not sufficient, talk to your prescriber about anti-nausea medications or dose adjustments.
Slow titration helps. Starting at the lowest dose and increasing gradually gives the body time to adapt, reducing the severity of GI side effects. The standard dosing schedule builds up slowly for this reason, and some providers use even more gradual approaches for patients who are particularly sensitive.
Know your baseline risk
Before starting semaglutide, discuss your personal blood clot risk with your healthcare provider. If you have a history of VTE, inherited thrombophilia, active cancer, or are on estrogen-containing medications, additional monitoring or preventive anticoagulation may be warranted. A simple risk assessment can identify whether you need extra precautions.

Semaglutide cardiovascular benefits vs. clot concerns
It would be irresponsible to discuss semaglutide blood clot risk without placing it in the context of the medication overall cardiovascular impact. The data overwhelmingly shows net cardiovascular benefit.
The SELECT trial demonstrated a 20% reduction in heart attacks, strokes, and cardiovascular death. A 19% reduction in all-cause mortality. A significant reduction in heart failure events. These are outcomes that kill far more people than DVT does.
The SUSTAIN-6 trial showed a 39% reduction in nonfatal stroke, arguably the most devastating non-fatal cardiovascular event a person can experience. The semaglutide meta-analysis found a 31% reduction in atrial fibrillation, a condition that itself causes blood clots in the heart that can travel to the brain and cause stroke.
Consider the absolute numbers. The JAHA meta-analysis estimated 25 additional DVT events per 10,000 person-years with GLP-1 agonist use. The SELECT trial showed approximately 150 fewer MACE events per 10,000 person-years. Even taking the DVT increase at face value, the cardiovascular benefit outweighs the clot risk by roughly 6:1.
And DVT, while serious, is highly treatable. Most DVTs resolve completely with 3-6 months of anticoagulation therapy. Heart attacks and strokes, by contrast, often cause permanent damage or death. The risk-benefit calculation strongly favors semaglutide for most patients, particularly those with existing cardiovascular risk factors.
This does not mean the DVT signal should be ignored. It means that for the vast majority of semaglutide candidates, the benefits substantially outweigh this particular risk, especially when preventive measures are implemented.
What the FDA says (and does not say)
As of early 2026, the FDA-approved prescribing information for Ozempic (semaglutide for diabetes) and Wegovy (semaglutide for weight loss) does not list blood clots, DVT, or VTE as known side effects or warnings. The labels mention cardiovascular risk reduction based on the SELECT trial data but do not address thrombotic risk.
In January 2024, the FDA announced it was investigating semaglutide for several potential safety signals, including blood clots, alongside hair loss, aspiration risk, and suicidal ideation. This investigation was based on adverse event reports submitted to the FDA Adverse Event Reporting System (FAERS). Through the end of 2023, the FAERS database contained 22,287 total semaglutide adverse reaction records (16,346 for subcutaneous semaglutide and 2,496 for oral semaglutide).
An FDA investigation does not mean a safety problem has been confirmed. It means the agency has identified enough reports to warrant closer examination. The investigation could result in a label change, additional warnings, or a determination that no label change is needed.
In September 2023, the FDA did update the semaglutide label to add ileus (a severe intestinal blockage) as a warning, demonstrating that the agency does act on emerging safety signals when the evidence supports it.
Multiple lawsuits have been filed against Novo Nordisk alleging that semaglutide caused blood clots. These legal proceedings are ongoing and do not constitute scientific evidence. However, they may accelerate regulatory review and encourage more systematic tracking of VTE events in semaglutide users.
SeekPeptides monitors FDA regulatory developments closely and updates its resources as new information becomes available. Understanding the current regulatory landscape helps users have informed conversations with their healthcare providers about risk management.
Comparison with other GLP-1 medications
The blood clot question extends beyond semaglutide to the entire GLP-1 receptor agonist class. Here is how the available evidence compares across medications.
Medication | Class | DVT/VTE signal | Cardiovascular outcomes | Platelet data |
|---|---|---|---|---|
Semaglutide (Ozempic/Wegovy) | GLP-1 RA | RR 3.66 for DVT in meta-analysis | 20% MACE reduction (SELECT) | Anti-inflammatory, anti-platelet mechanisms |
Liraglutide (Victoza/Saxenda) | GLP-1 RA | No specific DVT signal | 13% MACE reduction (LEADER) | 54% reduction in platelet aggregation |
Tirzepatide (Mounjaro/Zepbound) | Dual GLP-1/GIP | Two DVT case reports | SURPASS/SURMOUNT showed no VTE signal | Limited platelet-specific data |
Dulaglutide (Trulicity) | GLP-1 RA | No specific DVT signal | 12% MACE reduction (REWIND) | Limited data |
Triple agonist | Insufficient data | Phase 3 ongoing | No published platelet data |
The 2025 JAHA meta-analysis examined the GLP-1 class as a whole and did not find statistically significant differences between individual medications for DVT risk. However, semaglutide has been the most extensively studied, with the largest number of clinical trials analyzed. This means the DVT signal is most visible for semaglutide simply because there is more data available, not necessarily because it carries a uniquely higher risk.
Liraglutide stands out for having the strongest direct evidence of platelet inhibition, the controlled study showing 54% reduction in thromboxane-induced aggregation. Whether semaglutide has the same platelet effect at equivalent receptor activation levels remains unconfirmed, though the shared mechanism of action makes it plausible.
For users considering switching between GLP-1 medications, current evidence does not support choosing one over another specifically to reduce blood clot risk. The class-wide DVT signal and the class-wide cardiovascular benefits suggest these are properties of the GLP-1 receptor activation pathway itself, not of any individual drug.
The bottom line on semaglutide and blood clots
After reviewing every available study, case report, and biological mechanism, here is what we can say with reasonable confidence.
Clinical trial meta-analyses show a statistically significant association between semaglutide (and GLP-1 agonists broadly) and increased DVT risk. The absolute risk increase is approximately 25 additional events per 10,000 person-years. This risk appears to increase with treatment duration beyond 1.5 years.
Real-world data from over a million patients shows the opposite: GLP-1 users have 18-20% fewer blood clots than comparable patients on other diabetes medications. This paradox has not been fully explained.
At the molecular level, semaglutide has both pro-thrombotic effects (primarily through dehydration, muscle loss, and B12 depletion) and anti-thrombotic effects (through direct platelet inhibition, nitric oxide enhancement, anti-inflammatory pathways, and PAI-1 modulation). The net effect likely depends on individual patient factors and how well the pro-thrombotic risks are managed.
The overall cardiovascular benefit of semaglutide massively outweighs the potential DVT risk. A 20% reduction in heart attacks, strokes, and cardiovascular death prevents far more serious harm than the potential increase in treatable DVT events.
The FDA has not added blood clots to the semaglutide label but is investigating the signal.
Practical risk reduction is straightforward: stay hydrated, stay active, monitor B12, manage GI side effects, and know the warning signs of DVT.
For researchers and users seeking the most thorough understanding of semaglutide safety, SeekPeptides offers comprehensive, evidence-based guides that cover not just the headlines but the complete scientific picture. Members access detailed safety databases, compound-specific guides, and a community of experienced researchers who have navigated these exact concerns.
Frequently asked questions
Does semaglutide directly cause blood clots?
The relationship is not definitively established as causal. Meta-analyses of clinical trials show a statistical association between semaglutide use and increased DVT risk (relative risk 3.66), but real-world studies involving over a million patients show the opposite trend. The most likely explanation is that semaglutide side effects, particularly dehydration from GI symptoms, indirectly increase clot risk in some users, while the drug direct anti-platelet and anti-inflammatory effects provide protection in others.
Should I stop taking semaglutide because of blood clot risk?
For most users, no. The cardiovascular benefits of semaglutide (20% reduction in heart attacks, strokes, and cardiovascular death) substantially outweigh the potential DVT risk. However, if you have a personal history of blood clots, known thrombophilia, or multiple additional risk factors, discuss your specific situation with your healthcare provider. Stopping semaglutide should always be a decision made with medical guidance.
How common are blood clots in semaglutide users?
Based on the JAHA meta-analysis, there are approximately 25 additional DVT events per 10,000 person-years of GLP-1 agonist use. This means that for every 10,000 people taking semaglutide for one year, roughly 25 more would experience DVT compared to non-users. While statistically significant, this absolute risk increase is small compared to the baseline DVT rate in the general population (approximately 1-2 per 1,000 per year in adults over 40).
Does the blood clot risk increase the longer I take semaglutide?
The JAHA meta-analysis found that DVT risk was significantly higher in studies with treatment duration exceeding 1.5 years (OR 2.32 vs 1.64 overall). This suggests a duration-dependent relationship, though it has not been confirmed in prospective studies designed specifically to test this question. Long-term semaglutide users should maintain extra vigilance about hydration, activity, and symptom awareness.
Does tirzepatide have the same blood clot risk as semaglutide?
The evidence is less extensive for tirzepatide specifically, but two case reports of DVT have been published. The JAHA meta-analysis examined GLP-1 agonists as a class and found the DVT association applied broadly, not just to semaglutide. Since tirzepatide activates the GLP-1 receptor (along with GIP), similar mechanisms likely apply. However, the SURPASS and SURMOUNT trials did not report a significant VTE signal.
Can I take blood thinners with semaglutide?
Yes, many patients take anticoagulants alongside semaglutide. There are no known direct drug interactions between semaglutide and common blood thinners like warfarin, apixaban, or rivaroxaban. However, semaglutide slows gastric emptying, which could theoretically affect the absorption timing of oral anticoagulants. If you are on both medications, your healthcare provider should monitor your anticoagulation levels more closely, especially when starting or adjusting semaglutide doses.
Will drinking more water prevent blood clots while on semaglutide?
Adequate hydration is one of the most effective strategies for reducing clot risk, but it is not a guarantee. Dehydration increases blood viscosity and is the most plausible mechanism linking semaglutide to DVT. Maintaining proper fluid intake, at least 64 ounces daily and more if experiencing GI side effects, removes this risk factor. However, other factors like immobility, genetic predisposition, and hormonal influences also affect clot risk independently of hydration status.
Are blood clots from semaglutide treatable?
DVTs are highly treatable when diagnosed promptly. Standard treatment involves anticoagulation therapy (blood thinners) for 3-6 months, and most clots resolve completely. The case reports of DVT in GLP-1 users all described successful treatment with anticoagulants. The key is early recognition of symptoms, specifically new leg swelling, pain, or redness, and immediate medical evaluation.
External resources
Journal of the American Heart Association: GLP-1 RAs and risk of VTE meta-analysis
Endocrine Journal: Comprehensive safety analysis of semaglutide (SUSTAIN/PIONEER meta-analysis)
American Society of Hematology: GLP-1 RA and VTE propensity-matched study
In case I do not see you, good afternoon, good evening, and good night. May your blood flow freely, your veins stay clear, and your health decisions stay informed.