Feb 24, 2026

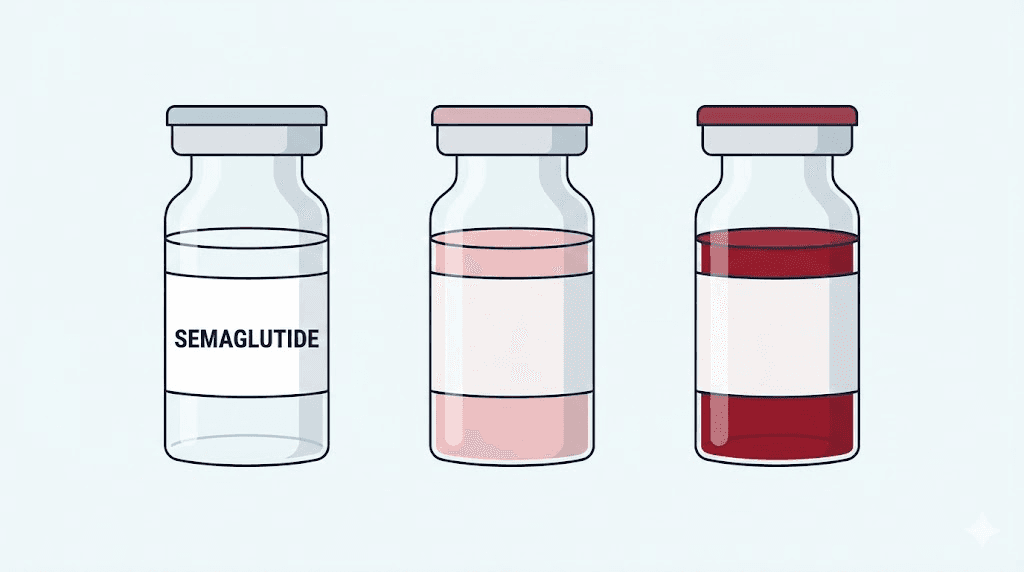
Before you draw up another dose, stop. Look at the vial. If the liquid inside is red, pink, or any shade other than perfectly clear, you need answers before that needle touches skin. This is not a minor cosmetic difference. The color of your semaglutide tells a story about what is inside it, how it was made, and whether it is safe to inject.
Here is the short answer. If your semaglutide contains vitamin B12 (cyanocobalamin or methylcobalamin) and the liquid is a clear pink or reddish hue, you are almost certainly fine. B12 is naturally red. Pharmacies add it deliberately. The color comes from the vitamin, not from contamination or degradation.
But that word "almost" matters. Because red semaglutide without B12 on the label is a completely different situation. And cloudy red semaglutide, regardless of what the label says, is a problem. The distinction between safe red and dangerous red comes down to three factors: what your label lists, how the liquid looks beyond just color, and how you have stored it. This guide walks through every scenario, every color variation, every warning sign, and every action step so you never have to guess. Whether you are using compounded semaglutide from a pharmacy or brand-name pens like Ozempic or Wegovy, the rules for color assessment are clear and specific. SeekPeptides has compiled the most comprehensive breakdown available so you can make an informed decision in the next five minutes.
What semaglutide should normally look like
Normal semaglutide looks like water. Clear. Colorless. Completely transparent.
That is the baseline for every FDA-approved semaglutide product. Ozempic pens contain a solution that should be entirely free of color, cloudiness, particles, and floating debris. Wegovy pens follow the same standard. Rybelsus tablets are a different format entirely, but the injectable forms share this universal rule: the liquid should look like you could mistake it for sterile water.
This applies whether you are looking at a 5mg vial, a 10mg vial, or a prefilled pen. The concentration does not change the color. A higher concentration of semaglutide does not make the solution yellow, pink, or cloudy. Pure semaglutide peptide in solution is colorless at every clinically relevant concentration.
When you hold the vial up to light, you should see through it completely. No haze. No tint. No specks. If someone handed you the vial without a label, you would have no visual way to distinguish it from bacteriostatic water or normal saline. That transparency is the gold standard, and any deviation from it requires investigation.
The same standard technically applies to compounded semaglutide when the formulation contains only semaglutide and a carrier solution. Pure compounded semaglutide without additives should also be clear and colorless. The moment color enters the equation, something else is in the vial. That something might be perfectly safe. Or it might not be.
How to properly inspect your vial
Hold the vial at eye level against a white background. Fluorescent lighting works best because it reveals subtle color shifts that incandescent bulbs can hide. Tilt the vial gently and watch the liquid move. Healthy semaglutide flows smoothly, like water. It does not leave streaks on the glass, form bubbles that linger, or show any visible texture.
Now hold it against a dark background. This step catches particles that white backgrounds miss. Look for any floating specks, fibers, crystals, or sediment settling at the bottom. Even one visible particle means something has gone wrong. Maybe a seal was compromised. Maybe the peptide has begun to aggregate. Either way, particles mean do not inject.
Finally, check the rubber stopper and the crimp seal. Any sign of moisture around the seal, discoloration of the stopper, or looseness suggests the vial integrity has been compromised. A compromised vial means potential contamination, and contaminated solutions can develop unexpected colors as bacteria or mold begin to grow.
The most common reason your semaglutide is red
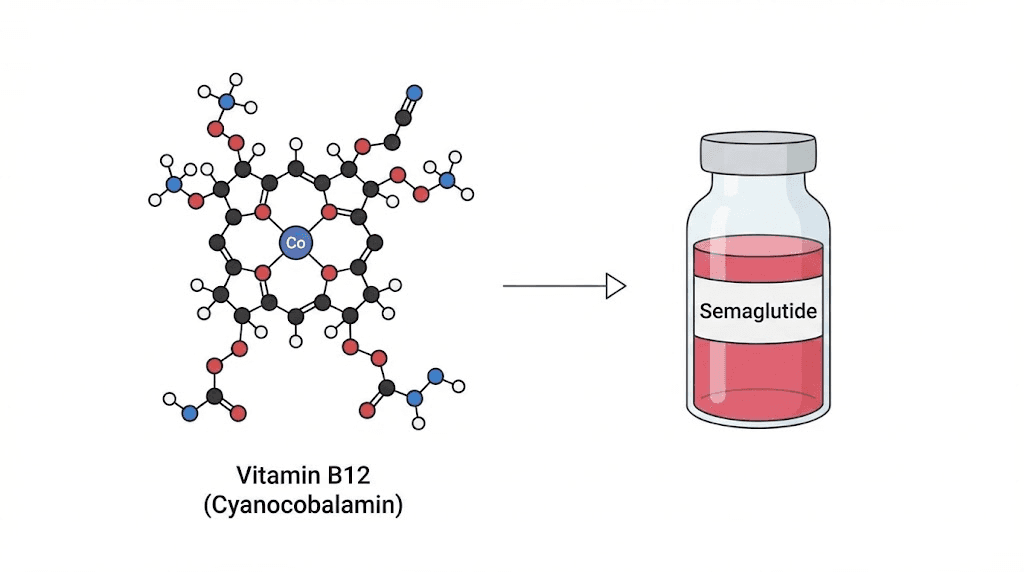
Vitamin B12. That is the answer for roughly 90 percent of people asking this question.
Cyanocobalamin, the most common supplemental form of vitamin B12, is a deep cherry-red compound. When a compounding pharmacy adds B12 to semaglutide, the resulting solution takes on a pink to red hue depending on the concentration of B12 in the formulation. Higher B12 concentrations produce deeper red. Lower concentrations produce a gentle pink, sometimes so faint it looks almost salmon-colored.
This is not contamination. This is not degradation. This is chemistry working exactly as expected. The combination of semaglutide with B12 has become one of the most popular compounded formulations available, and the red color is simply what B12 looks like in solution.
Methylcobalamin, another form of B12, produces a slightly different shade. It tends toward a pinkish-red rather than the deep cherry of cyanocobalamin. Semaglutide with methylcobalamin formulations may appear lighter overall, but the principle is the same. The vitamin creates the color.
Why pharmacies add B12 to semaglutide
There are three main reasons compounding pharmacies combine B12 with semaglutide, and none of them are arbitrary.
First, nausea reduction. Semaglutide slows gastric emptying, which is part of how it suppresses appetite and promotes weight loss. But that same mechanism causes nausea in a significant percentage of users, especially during the early weeks of treatment. B12 appears to help some patients tolerate the medication better, though the research on this specific combination remains limited.
Second, energy support. Fatigue is a common side effect of semaglutide, particularly during caloric restriction. B12 plays a central role in energy metabolism, red blood cell formation, and neurological function. Adding it to the injection provides a direct supplementation route that bypasses the digestive system, which is especially relevant since semaglutide itself reduces nutrient absorption by slowing digestion.
Third, weight loss can deplete B12 stores over time. Rapid weight loss, reduced food intake, and changes in gut motility all contribute to potential micronutrient deficiencies. Including B12 in the semaglutide formulation addresses this proactively rather than reactively.
The dosage charts for semaglutide with B12 typically specify both the semaglutide concentration and the B12 concentration per milliliter, so you can verify exactly what your pharmacy included. If your label shows B12 or cyanocobalamin or methylcobalamin as an ingredient, and the liquid is a consistent pink or red without cloudiness, your medication is behaving exactly as formulated.
Brand-name vs compounded semaglutide color differences
This distinction is critical, and getting it wrong could mean injecting something unsafe or throwing away perfectly good medication.
Brand-name semaglutide (Ozempic, Wegovy)
Brand-name semaglutide pens should always, without exception, contain clear and colorless liquid. Novo Nordisk manufactures these products under strict FDA oversight. The formulation contains semaglutide and excipients like disodium phosphate dihydrate, propylene glycol, phenol, and water for injection. None of these ingredients produce color.
If your Ozempic or Wegovy pen contains pink, red, yellow, brown, or any colored liquid, do not use it. This is not normal. This suggests degradation, contamination, or a manufacturing defect. Contact your pharmacy immediately. Do not inject. The manufacturer prescribing information is explicit: the solution should be clear and colorless to almost colorless.
There is no scenario where a brand-name semaglutide pen should be red. Period. No additive, no formulation variation, no acceptable exception. If it is red, something is wrong.
Compounded semaglutide
Compounded semaglutide is a different story entirely. These formulations come from compounding pharmacies that create custom preparations, and the ingredient list varies by pharmacy and by prescription.
A compounded semaglutide vial might contain:
Semaglutide only (should be clear and colorless)
Semaglutide plus B12/cyanocobalamin (will be pink to red)
Semaglutide plus methylcobalamin (will be pinkish-red)
Semaglutide plus glycine (should be clear, glycine is colorless)
Semaglutide plus B12 plus glycine (will be pink to red)
Semaglutide plus B6/pyridoxine (may have slight yellowish tint)
The key action here is simple. Read your label. Every compounding pharmacy is required to list all active ingredients and their concentrations on the vial label. If B12 appears on that label and the color matches what B12 produces, you have your answer. If B12 does not appear on the label and the liquid is red, you have a different kind of answer, and it means you should not inject that medication until you have spoken with your pharmacy.
Complete semaglutide color guide
Different colors tell different stories. Here is what each one means, what causes it, and what you should do.
Clear and colorless
This is the standard for pure semaglutide. Brand-name and compounded formulations without B12 should look like this. If your semaglutide is clear and you have stored it properly, proceed with confidence. No action needed beyond normal inspection for particles.
Light pink to salmon
Almost always indicates a low concentration of B12 in the formulation. Check your label. If cyanocobalamin or methylcobalamin is listed, this color is expected and safe. The lighter shade simply means less B12 per milliliter compared to deeper red formulations. Many pharmacies like Olympia Pharmacy and Empower Pharmacy use varying B12 concentrations.
Medium pink to rose
Standard appearance for semaglutide-B12 combinations at typical concentrations. Nothing unusual here if your label confirms B12 content. The solution should still be completely clear, meaning you can see through it without any haze, despite the color.
Deep red to cherry
Higher B12 concentrations produce this deeper color. Still normal if label-confirmed, but worth noting because deeper red can make it harder to spot cloudiness or particles. When inspecting a deeply colored vial, use bright lighting and check from multiple angles. Look for any areas where the color appears uneven, which could indicate layering or precipitation.
Yellow or amber
This is where things get concerning. A yellow tint in semaglutide can indicate oxidation of the peptide, which means the molecular structure has begun to break down. Oxidized semaglutide may have reduced potency or may be entirely inactive. The amino acid residues in semaglutide, particularly methionine and tryptophan, are susceptible to oxidation, and this process can produce yellow-brown discoloration.
One exception: some formulations containing vitamin B6 (pyridoxine) may have a very faint yellowish tint. But B6-containing semaglutide formulations are far less common than B12 combinations. If your label does not show B6 and the liquid is yellow, do not use it.
Brown or dark amber
Advanced degradation. This color suggests significant chemical breakdown of either the peptide or an additive in the solution. Never inject brown semaglutide. The protein structure has likely denatured beyond the point where any therapeutic benefit remains, and injecting degraded peptide material carries risks that include injection site reactions, immune responses, and zero therapeutic effect.
Cloudy or milky (any color)
Cloudiness indicates particulate matter suspended in the solution. This could be aggregated peptide, bacterial contamination, or precipitated ingredients. Regardless of the base color, clear pink with cloudiness is a no. Clear and colorless with cloudiness is a no. Any cloudiness at all means discard the vial and contact your pharmacy.
Understanding proper peptide storage prevents most color-change issues before they start.

Deep dive into vitamin B12 and semaglutide formulations
Since B12 is the most common reason for red semaglutide, it deserves a thorough explanation.
The chemistry behind the color
Cyanocobalamin is a cobalt-containing compound with a corrin ring structure. The cobalt ion at the center of this ring absorbs light in the yellow-green spectrum, which means the wavelengths it reflects back to your eyes fall in the red range. This is not a dye or an artificial coloring. It is the natural optical property of the B12 molecule itself, the same reason that a B12 supplement capsule or a B12 injection from any pharmacy appears red.
When dissolved in the same solution as semaglutide, the B12 distributes evenly throughout the liquid. This is why properly prepared semaglutide-B12 formulations have a uniform color from top to bottom. If you see color that is darker at the bottom or lighter at the top, that suggests incomplete mixing or separation, and the vial should be gently swirled (never shaken) before inspection.
Typical B12 concentrations in compounded semaglutide
Most compounding pharmacies add B12 at concentrations between 1,000 mcg/mL and 2,500 mcg/mL. Some formulations go as high as 5,000 mcg/mL. The higher the B12 concentration, the deeper the red color. Here is a general guide:
1,000 mcg/mL B12: Light pink, almost translucent
2,000 mcg/mL B12: Medium pink to rose
2,500 mcg/mL B12: Rose to light red
5,000 mcg/mL B12: Deep red, cherry-colored
These are approximate ranges, and the exact shade depends on the semaglutide concentration, the total volume of solution, and the specific form of B12 used. But they give you a reasonable reference for what to expect when you open a new vial of compounded semaglutide with B12.
Cyanocobalamin vs methylcobalamin: color differences
Cyanocobalamin produces the classic deep cherry-red that most people associate with B12 injections. It is the most common form used in compounded semaglutide because it is stable, inexpensive, and well-studied.
Methylcobalamin is the bioactive form of B12, meaning the body can use it directly without conversion. It produces a somewhat different shade, often described as pinkish-red or salmon rather than deep cherry. Methylcobalamin is also less stable than cyanocobalamin and more sensitive to light, which means formulations containing it may need extra protection from light exposure.
Some patients prefer methylcobalamin because it does not require the enzymatic conversion step that cyanocobalamin needs. However, both forms are effective for B12 supplementation, and the choice between them is a clinical decision that your prescribing provider and pharmacy make based on your specific situation.
Does the B12 affect semaglutide potency?
Current evidence does not suggest that B12 degrades semaglutide when properly formulated and stored. Reputable compounding pharmacies conduct stability testing on their formulations to verify that both the semaglutide and the B12 remain at labeled potency throughout the assigned beyond-use date.
However, the FDA has not approved any semaglutide-B12 combination product. This means the combination has not undergone the rigorous multi-phase clinical trial process that Ozempic and Wegovy completed. The clinical data supporting the combination is limited to pharmacy-level stability testing and theoretical rationale rather than large-scale efficacy studies.
This does not make the combination dangerous. It means the evidence base is different from what exists for the brand-name products. If you are using compounded semaglutide, understanding this distinction helps you make informed decisions about your care.
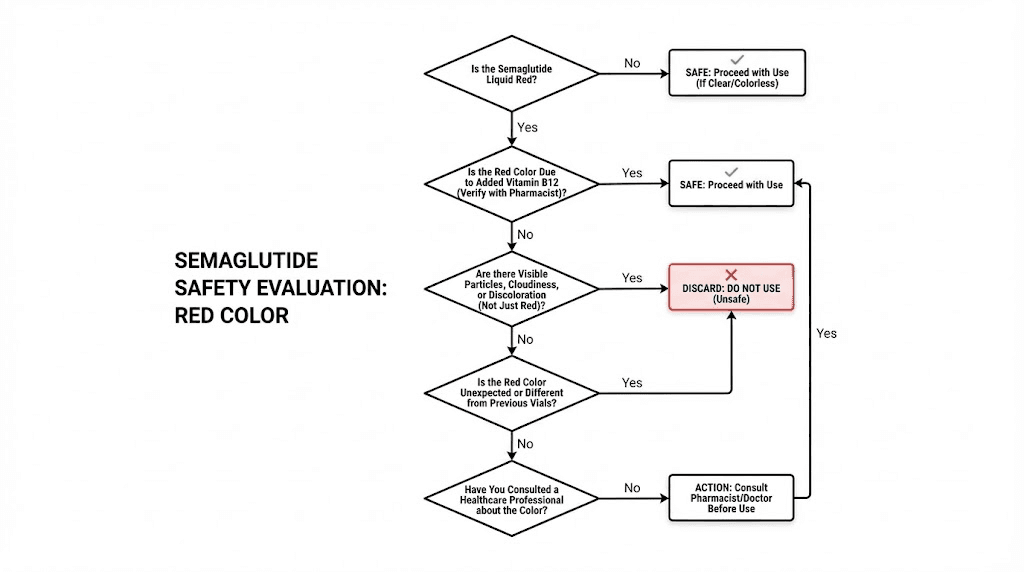
When red semaglutide is safe to use
Red semaglutide is safe to use when all four of these conditions are met simultaneously. Not three. Not two. All four.
Condition 1: The label lists B12. Check the vial label for cyanocobalamin, methylcobalamin, vitamin B12, or any B12 variant. If B12 is listed as an ingredient, the red color has a known, expected source.
Condition 2: The liquid is clear. Despite being colored, the solution must be transparent. Hold it up to light and look through it. You should be able to read text through the vial. Color is acceptable. Cloudiness is not. A solution can be red and clear at the same time, and that combination is what you want to see.
Condition 3: No particles are visible. Tilt the vial slowly and watch. Look against both light and dark backgrounds. No specks, no fibers, no crystals, no sediment. Particles indicate either contamination or peptide aggregation, and neither is acceptable regardless of the color.
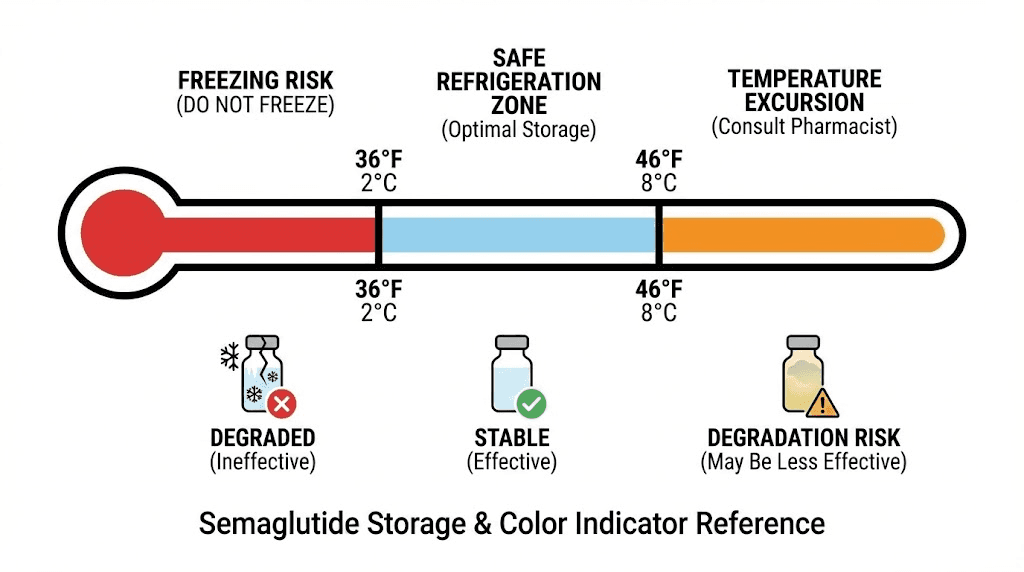
Condition 4: The vial has been properly stored. This means refrigeration between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius) unless your pharmacy specifies different storage conditions. If the vial sat in a hot car, on a sunny countertop, or anywhere above recommended temperatures for extended periods, the visual appearance alone cannot guarantee safety. Temperature damage to peptides is often invisible, meaning the semaglutide can look fine while being biochemically compromised.
When all four conditions check out, your red semaglutide is functioning exactly as your pharmacy intended it to function. Proceed with your normal dosing protocol.
When red semaglutide is NOT safe to use
Any single one of these scenarios is reason enough to stop and call your pharmacy before injecting.
Your label does not list B12
If the label shows semaglutide only (or semaglutide with glycine, or any other combination that does not include B12), the liquid should be colorless. Red or pink color in a non-B12 formulation suggests contamination, a compounding error, or degradation. Do not inject. Call the pharmacy immediately and describe what you see. Most reputable pharmacies will replace the vial at no charge because they take color discrepancies seriously.
The color changed from your last vial
Consistency matters. If your previous three vials from the same pharmacy were light pink and the new one is deep red, something changed. Maybe the pharmacy adjusted their B12 concentration. Maybe a different batch of B12 was used. Maybe something went wrong. Contact the pharmacy and ask. This does not necessarily mean the medication is unsafe, but an unexplained color change deserves an explanation before you proceed.
The color changed within the same vial
This is more concerning than vial-to-vial variation. If your semaglutide started as light pink last week and now looks darker, or if it was clear and has developed any color, something is changing inside the vial. Chemical reactions, bacterial growth, or light-induced degradation could all cause progressive color changes. Beyond-use dates exist for a reason, and color changes within a vial often signal that the formulation is breaking down faster than expected.
Cloudiness accompanies the color
Say it again for emphasis. Red plus clear equals potentially fine. Red plus cloudy equals never inject. Cloudiness means particles too small to see individually have formed a visible haze in the solution. These particles could be aggregated peptide, precipitated ingredients, or microbial contamination. None of those outcomes are compatible with safe injection.
You see particles, crystals, or sediment
Any visible solid material in the vial is a disqualifier. It does not matter whether the liquid is red, pink, clear, or any other color. Particles mean the solution is compromised. Some people mistake air bubbles for particles, so tilt the vial gently and wait thirty seconds. Bubbles rise and disappear. Particles remain suspended or settle to the bottom.
The vial seal looks compromised
If the flip-top cap was loose, the rubber stopper looks pushed in or damaged, or there is any evidence of moisture around the seal, the sterility of the vial may have been compromised. Vial integrity is foundational to medication safety, and a compromised seal means potential bacterial or fungal contamination.
Degradation signs every user should recognize
Semaglutide is a peptide. Peptides are proteins. And proteins are fragile. They maintain their therapeutic function only when their three-dimensional structure remains intact, and a surprising number of everyday conditions can damage that structure beyond repair.
How semaglutide degrades
Peptide degradation happens through several chemical pathways, and understanding them helps you recognize the signs.
Oxidation is the most common pathway. The methionine and tryptophan amino acid residues in the semaglutide molecule are particularly vulnerable to oxidative damage. When these residues oxidize, the peptide structure changes. The molecule may still look like semaglutide under basic analysis, but its ability to bind to GLP-1 receptors is compromised. Oxidation can be caused by exposure to air, light, heat, or certain chemicals. Visual signs include yellowing, though early-stage oxidation often produces no visible change at all.
Hydrolysis is the breaking of peptide bonds by water. This sounds counterintuitive since semaglutide is dissolved in water, but the pH and temperature of the solution are carefully controlled to minimize hydrolysis. When storage conditions change, particularly when temperature rises, the rate of hydrolysis increases. The peptide chain breaks into fragments that may or may not be visible as changes in the solution.
Aggregation occurs when damaged peptide molecules clump together. These aggregates can be invisible at first, then appear as cloudiness or visible particles as they grow larger. Aggregated semaglutide is not only ineffective but potentially immunogenic, meaning it could trigger an immune response at the injection site.
Denaturation is the unfolding of the peptide three-dimensional structure. Think of it like straightening a paper clip. The metal is still there, but it no longer functions as a paper clip. Denatured semaglutide may still be present in the solution at full concentration by weight, but it cannot bind to its target receptors effectively.
Visual signs of degradation
Here is the frustrating truth. Most semaglutide degradation is invisible. A vial that has lost 50 percent of its potency to oxidation might look identical to a fresh vial. This is why proper storage matters far more than visual inspection alone.
That said, when degradation becomes visible, these are the signs:
Color change: Clear becoming yellow, amber, or brown (in non-B12 formulations)
Cloudiness: Loss of transparency, haziness, or milky appearance
Particles: Visible specks, fibers, crystals, or sediment
Foaming: Excessive bubbles that do not resolve after gentle swirling
Viscosity change: Solution becomes thicker or stickier than water-like consistency
If your semaglutide shows none of these visual signs but you suspect it has been exposed to improper conditions, the safest approach is still to discard it and request a replacement. Using expired or compromised semaglutide might save money in the short term but costs you in wasted doses that produce no therapeutic effect.
The invisible degradation problem
This deserves special attention because it catches so many people off guard. You pull your semaglutide out of the fridge. It looks clear. It looks colorless. It passes every visual inspection test. But you have noticed that your appetite suppression is weaker than it was two weeks ago. You are not losing weight as expected. Something feels off.
This is what invisible degradation looks like from the patient side. The semaglutide plateau that some people attribute to receptor adaptation or metabolic adjustment might actually be a potency problem. If your vial was exposed to temperature excursions, even brief ones, during shipping or storage, the peptide may have partially degraded without any visible evidence.
This is one reason SeekPeptides emphasizes proper storage protocols so heavily. Visual inspection is a necessary step, but it is not sufficient on its own. Consistent cold chain management from pharmacy to injection is the only reliable way to ensure your medication retains full potency.
How storage mistakes change semaglutide color
Temperature, light, and time are the three enemies of peptide stability. Each one can independently cause color changes, and when they combine, degradation accelerates dramatically.
Temperature exposure
Semaglutide should be refrigerated at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius) before first use. After first use, brand-name Ozempic pens can be kept at room temperature up to 86 degrees Fahrenheit (30 degrees Celsius) for up to 56 days. Wegovy pens can stay at room temperature up to 86 degrees for up to 28 days.
What happens outside these ranges? Heat accelerates every degradation pathway simultaneously. Oxidation speeds up. Hydrolysis increases. Aggregation becomes more likely. The rate of degradation roughly doubles for every 18-degree Fahrenheit (10-degree Celsius) increase in temperature. So semaglutide stored at 100 degrees Fahrenheit degrades approximately four times faster than semaglutide stored at 64 degrees.
Freezing is equally problematic. When the solution freezes, ice crystals form that can physically damage the peptide structure. The expansion and contraction of freezing and thawing can also crack the peptide three-dimensional folding. Never freeze semaglutide. If it has frozen, discard it even if it looks normal after thawing.
Understanding what happens when semaglutide gets warm is essential for anyone managing their own medication storage.
Light exposure
Ultraviolet light from sunlight and certain artificial sources drives photodegradation of peptides. UV radiation provides the energy needed to trigger oxidation reactions in susceptible amino acid residues. This is why semaglutide vials should be stored in their original packaging or in a dark location within the refrigerator.
B12-containing formulations are particularly sensitive to light. Methylcobalamin is especially photosensitive and can degrade significantly with prolonged light exposure, which would change the color of the solution as the B12 breaks down. If your methylcobalamin-containing semaglutide was exposed to direct sunlight or bright artificial light for extended periods, the B12 may have degraded even if the semaglutide itself is unaffected.
Time beyond expiration
Every semaglutide product and every compounded formulation has an assigned expiration or beyond-use date. These dates are not arbitrary. They are based on stability testing that demonstrates the medication maintains acceptable potency and purity through that date when stored properly.
Using semaglutide past its expiration date increases the risk of reduced potency and degradation byproducts. Color changes are more likely in expired medication because the degradation processes have had more time to produce visible effects. If your semaglutide is past its date and shows any color change, the two factors compound each other in terms of risk.
For detailed timelines on how long your vial remains viable, check the complete semaglutide shelf life guide and the refrigerator storage timeline.
Injection site redness vs medication color
Some people searching "why is my semaglutide red" are actually asking about redness at the injection site, not the color of the medication itself. These are completely different issues with completely different causes and implications.
Why injection sites turn red
When a needle penetrates skin, it triggers a localized inflammatory response. Your immune system detects the tissue disruption and sends white blood cells and inflammatory mediators to the area. Blood vessels dilate to increase blood flow. The result is a small area of redness, warmth, and sometimes mild swelling that appears within minutes of the injection.
This is normal. It happens with virtually every subcutaneous injection, not just semaglutide. According to FDA prescribing information, injection site reactions occur in approximately 1.4 percent of Ozempic users and 2.4 percent of Wegovy users. The vast majority are mild and resolve on their own within hours to a few days.
If you experience injection site reactions regularly, there are several techniques to minimize them. Rotating injection sites prevents repeated trauma to the same tissue. Allowing refrigerated medication to warm slightly (not above room temperature) before injecting reduces the thermal shock to tissues. Injecting slowly rather than quickly gives the tissue time to accommodate the fluid volume.
When injection site redness is a concern
Normal injection site redness is a small area, perhaps the size of a quarter, that fades within a few hours. Concerning redness looks different.
Watch for redness that expands over time rather than shrinking. If the red area is getting larger hours after injection, that suggests a more significant inflammatory or allergic response. Redness accompanied by increasing pain, warmth, hardness, or pus could indicate infection, especially if these symptoms develop 24 to 72 hours after injection rather than immediately.
Bright red, blistered, or hot skin at the injection site warrants medical evaluation. While true infection from subcutaneous peptide injections is rare when proper sterile technique is used, it does happen, and early treatment prevents complications.
To minimize injection discomfort and complications, follow evidence-based GLP-1 injection techniques and rotate between recommended injection sites consistently.
What to do right now if your semaglutide is red
Here is the step-by-step action plan. Follow it in order.
Step 1: Read the label completely
Look at every ingredient listed on your vial. Search for any form of B12: cyanocobalamin, methylcobalamin, hydroxocobalamin, or simply "vitamin B12." Also check for other color-producing additives like B6 (pyridoxine). Write down or photograph the ingredient list for reference.
Step 2: Determine your formulation type
Is this a brand-name pen (Ozempic, Wegovy) or a compounded formulation? Brand-name pens should never be red. If you have a brand-name pen with red liquid, skip directly to Step 6. If you have a compounded formulation, continue to Step 3.
Step 3: Match the color to your ingredients
B12 on the label and pink-to-red liquid with no cloudiness? That checks out. B12 on the label but the color is different from your previous vials? Note the discrepancy and continue to Step 4 before using. No B12 on the label and red liquid? Skip to Step 6.
Step 4: Perform a thorough visual inspection
Check clarity (can you see through the liquid?), check for particles (against white and dark backgrounds), check the seal integrity, and verify the expiration or beyond-use date. If anything fails this inspection, proceed to Step 6. If everything passes, your semaglutide is likely safe to use.
Step 5: Verify storage history
Has this vial been properly refrigerated since you received it? Was it shipped with cold packs? Did it sit at room temperature for extended periods during transit? If you are unsure about the cold chain history, err on the side of caution. Traveling with semaglutide introduces particular storage challenges that deserve careful attention.
Step 6: Contact your pharmacy
If anything about the color, clarity, particles, seal, or storage history raises concern, call the pharmacy that dispensed the medication. Describe what you see. Most pharmacies will ask you to bring the vial in for inspection or will issue a replacement. Document the vial with photographs before contacting them.
Step 7: Report to the FDA if necessary
If you suspect contamination or a compounding error, report it to the FDA through MedWatch at 1-800-FDA-1088 or online. These reports help protect other patients and hold pharmacies accountable. You can also report to your state board of pharmacy.
How to verify your compounding pharmacy
Not all compounding pharmacies are created equal. The difference between a reputable compounder and a questionable one can be the difference between safe medication and a vial of unknowns.
FDA registration
Legitimate compounding pharmacies register with the FDA as either 503A (traditional compounding) or 503B (outsourcing facility) entities. 503B facilities are subject to FDA inspection and must follow current good manufacturing practices (cGMP). 503A pharmacies operate under state pharmacy board oversight with less federal regulation. Both can produce quality products, but 503B facilities have an additional layer of accountability.
You can verify a pharmacy FDA registration through the FDA website. If the pharmacy is not registered, that is a significant red flag.
State licensing
Every compounding pharmacy must hold a valid license in the state where it operates and in any state where it ships medication. You can verify licensing through your state board of pharmacy website. Legitimate pharmacies display their license numbers prominently.
Third-party testing
The best compounding pharmacies voluntarily submit their products to independent third-party testing for potency, purity, sterility, and endotoxin levels. Ask your pharmacy whether they conduct third-party testing and whether they can share certificates of analysis for their semaglutide formulations.
Third-party testing is particularly important for peptide medications because potency testing confirms that the labeled amount of semaglutide is actually present in the vial, and sterility testing confirms that no microbial contamination is present.
The semaglutide salt form issue
The FDA has warned that some compounders use semaglutide sodium or semaglutide acetate rather than semaglutide base. These salt forms are chemically different from the active ingredient in Ozempic and Wegovy. While some compounding pharmacies argue that the salt forms are therapeutically equivalent, the FDA has not evaluated these claims, and the salt forms may have different bioavailability, stability, and safety profiles.
Ask your pharmacy what form of semaglutide they use. A transparent pharmacy will answer this question directly. If they are evasive or unclear, consider finding a different provider. The grey market for peptides introduces additional risks that informed users should understand.
Comparing semaglutide colors across popular pharmacies
Different pharmacies produce visibly different products, and knowing what to expect from your specific pharmacy reduces unnecessary worry.
Empower Pharmacy formulations
Empower Pharmacy offers semaglutide combined with cyanocobalamin. Their formulations typically appear as a medium pink to light red, consistent with the B12 concentrations they use. Empower is a 503B outsourcing facility, which means it operates under FDA inspection and cGMP requirements. Their products come with certificates of analysis, and the color should be consistent from vial to vial within the same formulation.
Olympia Pharmacy formulations
Olympia Pharmacy also produces semaglutide-B12 combinations, and their color profile is similar, ranging from light pink to medium rose depending on the specific formulation ordered. Some of their semaglutide-glycine combinations without B12 should be clear and colorless.
Strive Pharmacy formulations
Strive Pharmacy offers multiple semaglutide formulations with varying additive combinations. Check the Strive Pharmacy semaglutide dosage chart for specifics on their current formulations and expected appearances.
Other pharmacies
For any compounding pharmacy not listed here, the same principles apply. Read the label. Match the color to the ingredients. Verify the pharmacy credentials. And when in doubt, call and ask. Pharmacists are trained professionals who expect these questions and prefer that patients ask rather than guess.
Whether you are comparing options through Direct Meds or evaluating other providers, the label is always your starting point for color assessment.
Related color questions for other GLP-1 medications
If you are using or considering other GLP-1 receptor agonists, you might wonder whether color concerns apply to them as well.
Tirzepatide color
Brand-name tirzepatide (Mounjaro, Zepbound) should be clear and colorless to slightly yellow. A very faint yellow tint is acceptable for brand-name tirzepatide, which differs from semaglutide where any yellow is concerning. Compounded tirzepatide formulations follow the same rules as compounded semaglutide. If tirzepatide contains B12, it will be pink to red. If it does not, it should be clear.
For detailed information on tirzepatide formulations, the tirzepatide glycine B12 compound guide and the tirzepatide methylcobalamin guide cover what to expect from different formulations. Storage requirements for tirzepatide refrigeration and shelf life mirror those for semaglutide.
Retatrutide color
Retatrutide is still in clinical trials and not yet commercially available. Research-grade retatrutide should be clear and colorless after proper reconstitution. Any color in a reconstituted retatrutide solution suggests either an additive or contamination, and the same inspection principles apply.
Converting between GLP-1 medications
If you are switching between GLP-1 medications, the color difference between your old medication and your new one can be jarring if you are moving from a B12-containing formulation to a plain one, or vice versa. This is purely cosmetic. The therapeutic effect depends on the active ingredient and dosage, not the color. Use the conversion chart for accurate dose translation when transitioning.
Reconstitution and mixing: color considerations
If you are working with lyophilized (freeze-dried) semaglutide that requires reconstitution, the mixing process introduces its own set of color-related considerations.
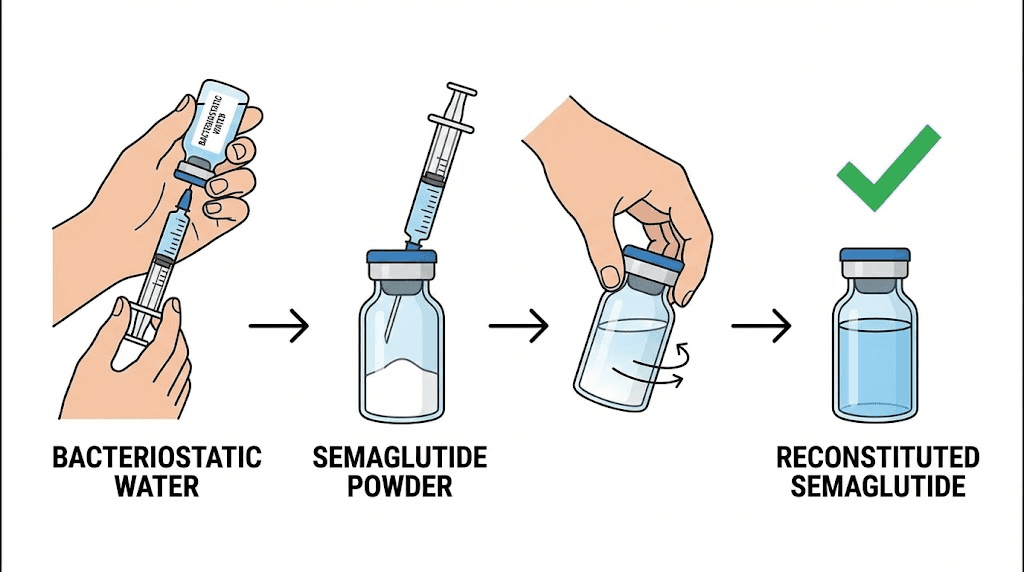
What reconstituted semaglutide should look like
When you add bacteriostatic water to semaglutide powder, the result should be a clear, colorless solution. The powder itself is typically white to off-white. As it dissolves, the solution transitions from cloudy (undissolved powder) to clear. This process should take only gentle swirling over one to two minutes. If the powder does not dissolve fully or the solution remains cloudy after several minutes, the peptide may have degraded during storage in powder form.
Never shake peptide vials during reconstitution. Aggressive mixing creates foam and can physically damage the peptide through shear forces. The complete semaglutide reconstitution guide covers proper technique in detail, and the reconstitution chart provides exact water-to-powder ratios for different concentrations.
Using the right water
Bacteriostatic water is the standard diluent for peptide reconstitution. It contains 0.9 percent benzyl alcohol as a preservative, which helps maintain sterility over multiple uses. Bacteriostatic water itself is clear and colorless. If your bacteriostatic water has any color, do not use it.
Sterile water for injection is an alternative for single-use vials but does not contain a preservative. Using sterile water for multi-dose vials increases contamination risk with each needle entry. For detailed guidance on diluent selection, see the peptide mixing guide and the bacteriostatic water measurement guide.
Common reconstitution mistakes that affect appearance
Several errors during mixing can cause unexpected color or clarity issues:
Using too little water: Over-concentrated solutions may appear slightly hazy because the peptide is at the edge of its solubility limit
Contaminated water source: Non-pharmaceutical-grade water can introduce ions or organisms that cause discoloration
Dirty needles or syringes: Reusing needles introduces contaminants that can cloud the solution
Powder was degraded before mixing: If the lyophilized peptide was stored improperly, it may not dissolve cleanly even with proper technique
The most common mistakes beginners make with peptides often involve the reconstitution step, and color anomalies are one of the first visible signs that something went wrong.
Use the peptide reconstitution calculator on SeekPeptides to determine exact volumes for your specific vial concentration and desired dosage.
Semaglutide storage best practices to prevent color changes
Prevention beats troubleshooting every time. These storage practices keep your semaglutide in optimal condition throughout its entire use period.
Refrigerator placement
Store semaglutide in the main body of the refrigerator, not in the door. Door shelves experience the widest temperature fluctuations because they are exposed to room temperature air every time the door opens. The back of the middle shelf typically maintains the most consistent temperature.
Do not store semaglutide near the freezer compartment or against the back wall of the refrigerator where ice formation is more likely. A small thermometer placed near your medication can verify that the temperature stays within the 36 to 46 degree Fahrenheit range.
Light protection
Keep the vial in its original box or wrap it in aluminum foil if the packaging has been discarded. This prevents photodegradation from the refrigerator light, which turns on every time you open the door. Every exposure to light is a small but cumulative hit to stability.
If you keep your medication in a pill organizer or a designated drawer, make sure it remains dark. The comprehensive peptide storage guide covers additional protection strategies for all types of peptide medications.
Travel considerations
Maintaining the cold chain during travel with semaglutide requires planning. Use an insulated cooler bag with ice packs, but ensure the vial does not come into direct contact with the ice pack (wrap the vial in a cloth or paper towel). Direct contact with a frozen ice pack can freeze the medication, which is just as damaging as heat exposure.
For air travel, keep semaglutide in your carry-on luggage, never in checked bags. Cargo holds experience temperature extremes that can reach well below freezing at altitude. Most airlines allow medications in carry-on bags regardless of liquid restrictions when accompanied by a prescription label.
Handling your vial
Minimize the number of times you remove the vial from refrigeration. Take it out, draw your dose, and return it promptly. Each minute at room temperature is cumulative over the life of the vial. If you are using a multi-dose vial, this means potentially dozens of temperature excursions over its use period, and each one chips away at stability.
Never touch the rubber stopper with bare fingers before inserting a needle. Clean it with an alcohol swab every time. This simple step prevents introducing skin bacteria or oils into the sterile solution, which could eventually cause discoloration or contamination.
Understanding dosage and potency concerns with discolored semaglutide
When semaglutide changes color due to degradation rather than additives, the potency question becomes critical. How much active medication remains in a partially degraded vial?
The honest answer is that you cannot know without laboratory testing. Visual inspection cannot quantify potency loss. A vial that looks slightly yellow might have lost 10 percent potency or 50 percent potency. There is no visual correlation between the degree of color change and the percentage of active ingredient remaining.
This matters because semaglutide dosing is carefully calibrated. A 5mg dosage protocol assumes you are receiving the full 5mg per the concentration on the label. If degradation has reduced the actual potency, you are effectively underdosing without knowing it. The semaglutide dosage calculator can help you verify your intended dose, but no calculator can account for degradation in the vial.
Underdosing semaglutide produces predictable consequences. Appetite suppression weakens. Appetite control takes longer to establish. Weight loss slows or stalls. Blood glucose management becomes less consistent. These symptoms are often attributed to tolerance or metabolic adaptation when the real culprit is simply receiving less medication than intended.
If you notice a sudden decrease in effectiveness that coincides with a color change in your vial, the connection is more than coincidental. Replace the vial and see if effectiveness returns.
FDA safety considerations for compounded semaglutide
The broader safety landscape for compounded semaglutide deserves mention because color concerns often intersect with quality concerns.
FDA warnings and alerts
The FDA has issued multiple warnings about compounded semaglutide products. In August 2024, they received complaints about black particulates in semaglutide vials from certain compounders. In October 2024, FDA inspections revealed compounding operations using non-sterile ingredients without adequate sterilization steps. In July 2024, the FDA warned that dosing errors with compounded injectable semaglutide had led to patients receiving five to twenty times the intended dose.
These warnings do not mean all compounded semaglutide is dangerous. They mean that the quality of compounded products varies significantly by pharmacy, and patients need to exercise due diligence in selecting their provider. The peptide regulation landscape continues to evolve, and staying informed protects your safety.
What the FDA recommends
The FDA recommends obtaining semaglutide from FDA-approved sources (Ozempic, Wegovy) when possible. When compounded semaglutide is used, they recommend verifying that the compounding pharmacy is FDA-registered, state-licensed, and follows appropriate quality standards. They also recommend reporting any adverse events or quality concerns through the MedWatch system.
For researchers and users navigating the compounded semaglutide landscape, understanding these regulatory dynamics helps inform both pharmacy selection and quality expectations.
Optimizing your semaglutide experience beyond color concerns
Once you have confirmed that your semaglutide is safe to use, the next question is how to get the most out of every dose. Color assessment is just one piece of the medication management puzzle.
Timing and administration
The best time to take semaglutide varies by individual, but consistency matters more than specific timing. Pick a day of the week and a general time window, then stick with it. Semaglutide has a half-life of approximately one week, so small timing variations of a few hours do not significantly impact blood levels.
For injection technique, proper syringe preparation and site rotation prevent both injection site reactions and uneven absorption. Injecting into the abdomen typically provides the most consistent absorption rates, followed by the thigh and upper arm.
Diet and lifestyle support
Semaglutide works best alongside appropriate dietary choices. The semaglutide diet plan and recommended food list provide evidence-based nutritional guidance tailored to how semaglutide affects digestion and appetite. Knowing which foods to avoid can reduce common side effects like nausea and constipation.
Managing side effects
Side effects are most common during dose escalation and typically improve with time. Fatigue, dizziness, burping, and gastrointestinal discomfort are the most frequently reported. Sleep disruption affects some users. Most of these resolve as the body adjusts, but persistent side effects should be discussed with your prescribing provider.
If side effects become unmanageable, your provider may adjust your dosage, slow your titration schedule, or consider switching to a different GLP-1 medication with a different side effect profile.
Tracking results
Understanding how long semaglutide takes to work sets realistic expectations. Most users notice appetite changes within the first week or two, with meaningful weight loss beginning around weeks four to eight. The one-month results typically show 3 to 5 percent body weight reduction, with continued loss over subsequent months as the dose is titrated upward.
If you are four weeks in with no weight loss, review your medication storage (color and potency), dosage accuracy, dietary intake, and whether you are actually taking the dose consistently. Many apparent non-responders turn out to have one or more correctable factors affecting their results.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
Frequently asked questions
Is red semaglutide safe to inject?
Red semaglutide is safe to inject if and only if the label lists vitamin B12 as an ingredient, the liquid is clear despite being colored, no particles are visible, and the vial has been properly stored. If any of these conditions are not met, do not inject and contact your pharmacy. Brand-name Ozempic and Wegovy should never be red.
What color should my compounded semaglutide be?
It depends on the formulation. Compounded semaglutide without additives should be clear and colorless. Formulations with vitamin B12 will range from light pink to deep red. Formulations with glycine only should be clear and colorless. Always check your label to know what color to expect from your specific formulation.
Can semaglutide turn red on its own without B12?
Semaglutide itself does not turn red. Without B12 or another colored additive, a red or pink color in semaglutide indicates contamination, bacterial growth, or an unidentified substance in the solution. This is not safe. Do not use it and contact your pharmacy immediately for a replacement and investigation.
My semaglutide was clear but now looks slightly yellow. Is it safe?
A yellow tint in previously clear semaglutide suggests oxidation or degradation. This means the peptide structure may be compromised and the medication may have reduced or zero potency. Discard the vial and contact your pharmacy for a replacement. Review your storage practices to prevent this from happening again.
How can I tell if my compounding pharmacy is reputable?
Verify their FDA registration, state licensing, and whether they use third-party testing. Ask what form of semaglutide they use (base vs salt forms). Check for any FDA warning letters or state board actions. Reputable pharmacies are transparent about their processes, testing, and ingredient sourcing. The peptide testing guide explains what quality testing entails.
Does the B12 in my semaglutide actually help with weight loss?
There is no clinical trial evidence that adding B12 to semaglutide enhances weight loss beyond what semaglutide alone achieves. The theoretical benefits include reduced nausea, improved energy levels, and prevention of B12 deficiency during weight loss. These are supportive benefits rather than direct weight loss enhancement. The complete semaglutide with B12 guide covers the evidence for and against the combination.
Should I switch to a non-B12 formulation if the red color concerns me?
You can request a semaglutide-only or semaglutide-glycine formulation without B12 if the color causes anxiety. Discuss this with your prescribing provider. Some patients supplement B12 separately through oral tablets or standalone injections rather than having it mixed into their semaglutide. The therapeutic effect of semaglutide itself is identical regardless of whether B12 is present in the formulation.
What happens if I inject degraded semaglutide?
Degraded semaglutide that has changed color likely has reduced or zero potency, meaning you would receive little to no therapeutic benefit from the injection. In addition to wasted medication, degradation products could potentially cause injection site reactions or immune responses. The primary risk is not acute toxicity but rather loss of therapeutic effect and wasted time in your treatment plan.
External resources
FDA: Concerns with unapproved GLP-1 drugs used for weight loss
PubMed: Effect of pH, buffers, and temperature on semaglutide degradation
In case I do not see you, good afternoon, good evening, and good night. May your vials stay clear, your storage stay cold, and your results stay consistent.