Feb 27, 2026

The prevailing narrative around semaglutide and female sexuality goes something like this: lose weight, feel confident, watch your sex drive skyrocket. It sounds logical. And for some women, that is exactly what happens. But for others, the experience is the complete opposite, a slow and confusing decline in desire that nobody warned them about and few providers seem to take seriously.
The reality is more complicated than any headline suggests.
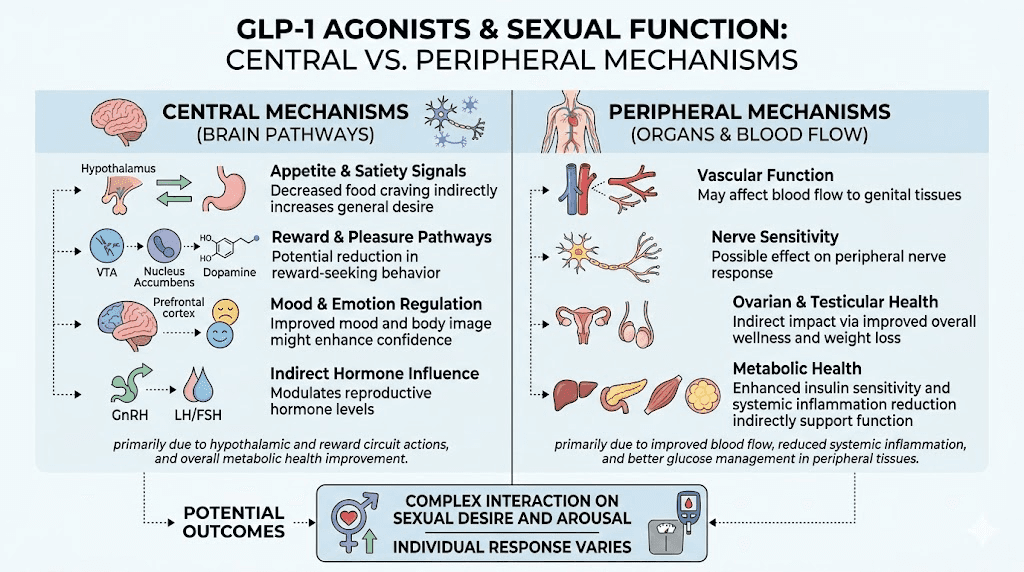
GLP-1 receptor agonists like semaglutide do not simply help women lose weight and call it a day. These medications interact with the brain reward system, energy metabolism, gastrointestinal function, and hormonal pathways that govern everything from appetite to arousal. When you change the signaling in areas of the brain responsible for pleasure, motivation, and reward, sexual desire can become collateral damage, or an unexpected beneficiary.
This guide examines every mechanism, every study, and every practical strategy relevant to semaglutide and sex drive in women. Whether you are experiencing a frustrating decline in libido, a surprising increase in desire, or just want to understand what might happen before starting treatment, the information here draws from clinical research, case reports, neurochemistry, and the lived experiences of thousands of women navigating this exact question. SeekPeptides has compiled this resource because the conversation around GLP-1 medications and female sexuality deserves more than vague reassurances. It deserves specifics.
How semaglutide works in the body and why sexual function gets involved
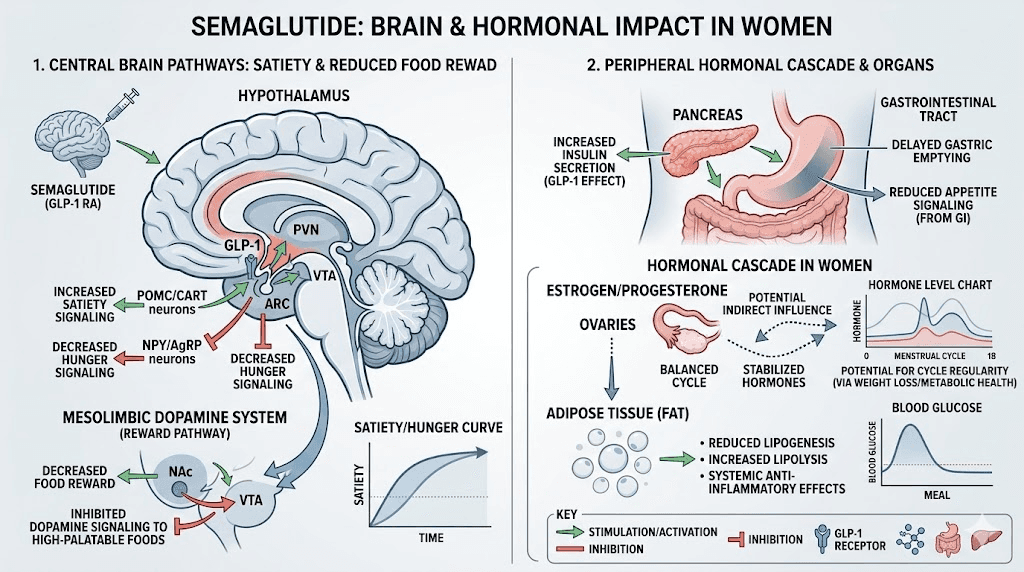
Understanding why semaglutide affects sex drive requires understanding what this medication actually does once it enters the body. Semaglutide is a GLP-1 receptor agonist, meaning it mimics a naturally occurring hormone called glucagon-like peptide-1. In a normally functioning body, GLP-1 gets released after eating. It tells the pancreas to produce insulin, slows gastric emptying, and signals to the brain that food has arrived.
Semaglutide amplifies these signals dramatically. Whether delivered as a weekly injection like brand-name or compounded formulations or as oral drops, the pharmacological mechanism is the same.
The medication binds to GLP-1 receptors throughout the body, and those receptors exist in far more places than the gut. Understanding how quickly semaglutide reaches these receptors helps explain the timeline of all side effects, including sexual ones. They sit in the pancreas, the cardiovascular system, the kidneys, and critically, the brain. The hypothalamus contains a dense concentration of GLP-1 receptors, and this is where things get relevant to sexual function. The hypothalamus regulates appetite, body temperature, sleep cycles, and, yes, sexual desire and arousal. When semaglutide activates receptors in this region, it does not selectively target only the appetite circuits. It influences the entire neighborhood, including pathways that govern appetite suppression, reward processing, and sexual motivation.
The timeline for appetite suppression typically begins within the first few weeks of treatment. For many women, changes in libido follow a similar, though less predictable, trajectory. Some notice shifts in desire within weeks of starting treatment. Others do not experience changes until months later, when cumulative weight loss begins altering their hormonal balance. And a significant portion of women notice no sexual side effects at all, which makes this topic particularly difficult to study in controlled settings.
The key insight is that semaglutide does not target one system in isolation. When you administer a medication that changes your dosage protocol to alter brain chemistry, hormonal balance, body composition, energy levels, and gastrointestinal function simultaneously, sexual desire becomes just one of many outputs that can shift in either direction. Whether you are using reconstituted semaglutide or a pre-mixed formulation, the mechanisms remain the same. The question is not whether semaglutide affects sex drive. The question is through which mechanism, in which direction, and for how long.
The libido paradox: why some women report higher desire and others report less
Ask ten women on semaglutide about their sex drive and you will likely hear ten different stories. This is not because semaglutide is unpredictable. It is because the medication triggers multiple competing forces that push libido in opposite directions simultaneously. Which force wins depends on the individual woman, her baseline hormonal status, her starting weight, her psychological relationship with her body, and her specific dosage.
On one side of the equation sits the confidence effect. Women who lose significant weight on compounded semaglutide or brand-name formulations often report feeling more attractive, more energetic, and more willing to initiate intimacy. The psychological burden of carrying excess weight, particularly in a culture that conflates thinness with desirability, can suppress sexual desire as effectively as any hormonal imbalance. Remove that burden and desire sometimes resurfaces with surprising intensity.
On the other side sits the neurochemical dampening effect. Semaglutide modulates dopamine signaling in the mesolimbic pathway, the same circuit responsible for the anticipation of pleasure. This is actually how the medication reduces food cravings. It makes the reward associated with eating feel less compelling. But dopamine does not discriminate between food rewards and sexual rewards. When the brain reward system gets dialed down, all pleasurable anticipation can diminish, including sexual desire.
Then there is the hormonal component. Rapid weight loss changes estrogen production because adipose tissue is a significant source of estrogen in women. Lose fat quickly and estrogen levels can drop, sometimes causing hair thinning, mood changes, and decreased vaginal lubrication, all of which contribute to reduced sexual interest and comfort.
A third factor rarely gets discussed. The sheer physical misery of gastrointestinal side effects during the early weeks of treatment, nausea, bloating, digestive discomfort, can make the idea of intimacy genuinely unappealing. This is not a libido problem in the traditional sense. It is a comfort problem. And it usually resolves as the body adapts to the medication.
The paradox resolves when you stop looking for a single answer. Semaglutide does not increase sex drive. It does not decrease sex drive. It changes the landscape in which sex drive operates, and the outcome depends on which changes dominate for each individual woman.
Understanding the full timeline of how semaglutide works helps put these competing effects into perspective. The medication does not reach full therapeutic effect instantly. As blood levels build over weeks and months, different mechanisms come online at different times, which is why libido changes rarely follow a straight line in either direction.
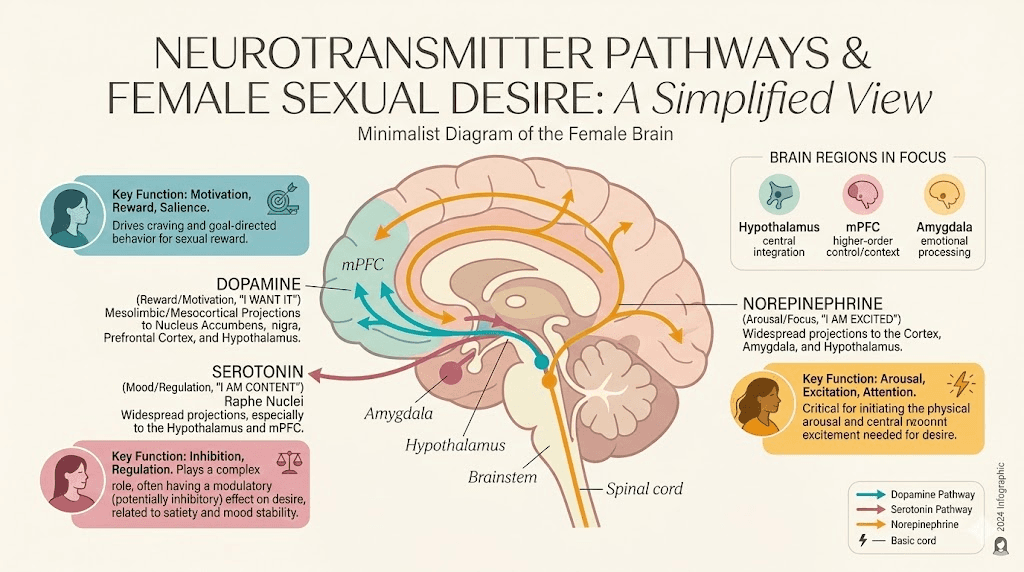
The neurochemistry behind semaglutide and sexual desire
Three neurotransmitter systems govern sexual desire in women: dopamine, serotonin, and norepinephrine. Semaglutide touches all three.
Dopamine and the reward pathway
Dopamine is the neurotransmitter of wanting. Not having, not enjoying, but wanting. It is what makes you think about sex before it happens, what creates the anticipatory excitement, the pull toward a partner. The mesolimbic dopamine pathway, running from the ventral tegmental area to the nucleus accumbens, fires when the brain anticipates something pleasurable.
Animal studies demonstrate that GLP-1 receptor agonists reduce dopamine signaling in this pathway. This is the mechanism behind reduced food cravings, reduced interest in alcohol, and potentially reduced interest in other reward-driven behaviors. The research published in preclinical models shows decreased rewarding effects across multiple pleasure categories when GLP-1 signaling increases.
For sexual desire, this means the anticipatory phase of arousal may feel blunted. Women describe it as not thinking about sex as often, not feeling the same pull toward intimacy, or finding that they can take or leave sexual encounters that previously felt compelling. The desire is not replaced by aversion. It simply fades into the background, like the volume on a radio slowly turned down.
This same reward pathway mechanism also explains the emerging reports of reduced interest in drinking alcohol on semaglutide, gambling, and compulsive shopping. The dopamine system does not differentiate between categories of reward. When GLP-1 receptor activation turns down the volume on wanting, everything pleasurable gets quieter, food, substances, and sex alike.
Serotonin and the 5-HT2C receptor
Here is where the mechanism gets specific. A clinical review published in 2025 proposed that GLP-1 agonist modulation via increased serotonergic activity at the 5-HT2C receptor may result in diminished sexual desire. This mirrors the mechanism behind anxiety-related effects reported with some GLP-1 medications. If that receptor name sounds familiar, it should. The 5-HT2C receptor is the same one implicated in the sexual side effects of SSRI antidepressants.
Women who have experienced decreased libido on medications like fluoxetine, sertraline, or escitalopram are likely familiar with this mechanism. SSRIs increase serotonin broadly, and the downstream effect on 5-HT2C receptors specifically suppresses sexual desire and delays or prevents orgasm. The emerging research suggests that semaglutide may activate similar serotonergic pathways, creating a parallel effect through a completely different medication class.
This connection matters because it suggests that women who are already sensitive to SSRI-induced sexual dysfunction may be at higher risk for experiencing similar effects on semaglutide. It also opens the door to potential treatment strategies borrowed from psychiatry, where providers have developed protocols for managing SSRI-related sexual side effects over decades.
Norepinephrine and arousal
Norepinephrine is the neurotransmitter of physiological arousal, the racing heart, the heightened sensation, the bodily readiness for sexual activity. It works alongside dopamine in the hypothalamus during sexual excitation. The case report literature on GLP-1 agonists and anorgasmia identifies decreased norepinephrine signaling as a potential contributor to orgasmic dysfunction, which represents a separate but related concern from decreased desire.
When semaglutide modulates hypothalamic activity, it may decrease both dopamine and norepinephrine in regions critical for sexual arousal and orgasm. The result is not just reduced desire but potentially reduced physical responsiveness, a slower arousal process, and difficulty reaching climax even when desire is present. This mechanism also overlaps with the pathways that cause headaches on GLP-1 medications and the broader musculoskeletal side effects that some women report. Understanding which neurotransmitter system is primarily affected can help guide treatment decisions, something we will explore in the strategies section below.
Hormonal shifts that change everything
The hormonal effects of semaglutide on female sexuality operate through two primary channels: the direct effects of weight loss on hormone production, and the indirect effects of metabolic changes on hormonal balance. Neither is simple.
Estrogen and adipose tissue
Fat cells produce estrogen through a process called aromatization, where the enzyme aromatase converts androgens into estrogens. Women with higher body fat percentages tend to have higher circulating estrogen levels. When semaglutide drives significant fat loss, sometimes 15 to 20 percent of body weight over a year, estrogen production from adipose tissue drops proportionally.
For premenopausal women, this shift is often manageable because the ovaries remain the primary estrogen source. But for perimenopausal and postmenopausal women, where adipose tissue becomes an increasingly important estrogen contributor, rapid fat loss can push estrogen levels low enough to cause symptoms. Those symptoms include vaginal dryness, decreased lubrication during arousal, hot flashes, mood changes, and reduced sexual desire. Women who are already navigating the hormonal transition of menopause while using semaglutide face a compound challenge that requires careful monitoring.
Testosterone and female desire
Testosterone is not just a male hormone. Women produce testosterone in the ovaries and adrenal glands, and it plays a critical role in sexual desire, arousal, and overall energy levels. The relationship between semaglutide and female testosterone is complex. In women with polycystic ovary syndrome, weight loss from GLP-1 treatment reduces elevated testosterone levels by approximately 33 percent, which is therapeutically beneficial because excess testosterone drives unwanted symptoms like acne and hirsutism in PCOS.
But in women without PCOS, any reduction in already-normal testosterone levels could theoretically decrease sexual desire. The research on semaglutide and testosterone in non-PCOS women is extremely limited, and most of what we know comes from general weight loss studies rather than semaglutide-specific trials. What we do know is that caloric restriction and rapid weight loss can temporarily lower free testosterone in women, and semaglutide-driven caloric restriction is no exception. Women interested in peptide approaches to fat loss should understand these hormonal trade-offs before selecting a protocol.
The cortisol connection
Chronic stress elevates cortisol, and elevated cortisol suppresses both testosterone production and sexual desire. Here is where semaglutide can actually help. Many women report reduced anxiety and improved mood after starting treatment, partly from the weight loss itself and partly from the direct neurological effects of GLP-1 receptor activation on mood-regulating brain circuits. Women who previously struggled with weight loss plateaus often carry additional cortisol burden from the frustration of failed diets. Lower cortisol means better conditions for sexual desire. This is another reason why the libido response to semaglutide varies so dramatically between individuals, the net hormonal effect depends on which imbalances existed before treatment started.
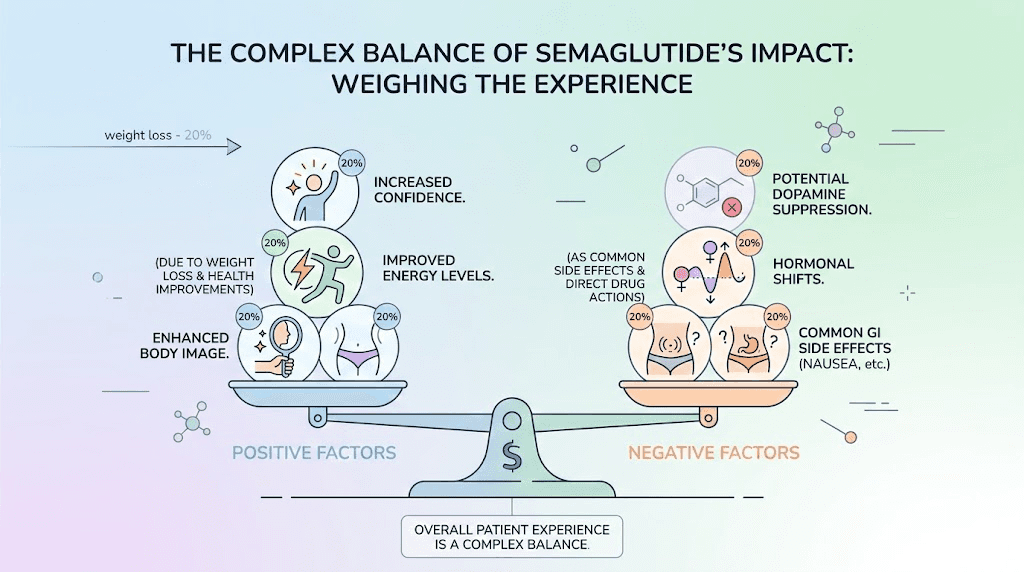
Weight loss, body image, and the confidence factor
The psychological dimension of semaglutide and female sex drive may be the most powerful variable of all, and it is the one that medical research captures least effectively.
Body image distress is one of the most common suppressors of female sexual desire. Women who feel self-conscious about their bodies during intimacy experience what researchers call spectatoring, mentally observing and evaluating themselves rather than being present in the sexual experience. This psychological distance from the physical experience of sex effectively kills desire over time. Why would the brain generate desire for an experience that consistently produces self-criticism and shame?
Semaglutide can break this cycle dramatically. Women who lose 30, 50, 80 or more pounds often describe feeling like they inhabit a different body. Clothes fit differently. Movement feels easier. The mirror becomes less adversarial. And the bedroom becomes a space of possibility rather than vulnerability. For these women, semaglutide does not directly increase sex drive. It removes the psychological barrier that was suppressing it.
The confidence effect extends beyond body image alone. Improved energy levels mean more stamina for physical intimacy. Better cardiovascular function from weight loss improves blood flow, including blood flow to the genitals. Women who have seen dramatic before-and-after transformations on GLP-1 medications often describe a complete shift in how they experience their own bodies during intimacy. Reduced joint pain makes previously uncomfortable positions accessible again. These physical improvements create a foundation on which desire can rebuild naturally.
But the confidence effect has limits. If the primary cause of decreased libido is neurochemical, meaning dopamine suppression or serotonin receptor activation, no amount of improved body image will overcome it. A woman can love how she looks and still feel zero sexual desire if her neurotransmitter profile has been altered sufficiently. Recognizing which factor dominates is essential for choosing the right management strategy.
PCOS, semaglutide, and the hormone rebalancing effect
Polycystic ovary syndrome affects an estimated 8 to 13 percent of reproductive-age women, and the intersection of PCOS, semaglutide, and sexual function deserves special attention because the dynamics are fundamentally different from those in women without PCOS.
Women with PCOS typically have elevated androgens, including testosterone, which drives symptoms like acne, excess body hair, and irregular menstrual cycles. Many also have insulin resistance, which further disrupts hormonal balance. The relationship between PCOS and sexual function is complicated. While testosterone is associated with desire, the elevated testosterone in PCOS is often accompanied by such significant psychological distress, from symptoms like hirsutism and weight gain, that sexual desire suffers despite adequate hormonal levels.
Semaglutide addresses multiple PCOS-related issues simultaneously. A meta-analysis of randomized controlled trials found that GLP-1 agonists in women with PCOS reduced body mass index, serum triglycerides, total testosterone, and waist circumference. Treatment normalized menstrual cycles in approximately 80 percent of responsive patients. A prospective study combining semaglutide with metformin showed reduced inflammatory markers, improved insulin resistance, alleviated menstrual irregularities, and increased natural pregnancy rates.
For sexual function specifically, the testosterone reduction in PCOS women is actually a net positive because it brings abnormally high levels back toward normal range. Combined with weight loss, improved body image, and normalized cycles, many women with PCOS report improved sexual satisfaction on semaglutide, even when the general population shows mixed results.
However, there is a nuance. If semaglutide brings testosterone levels too low, even PCOS women can experience decreased desire. The ideal outcome is a reduction that resolves PCOS symptoms without overcorrecting into deficiency. This requires monitoring hormone levels during treatment, something that many providers do not routinely do when prescribing semaglutide for weight management. If you have PCOS and notice decreased libido on semaglutide, requesting a testosterone level check is a reasonable first step that your dosage protocol should account for. Using the semaglutide dosage calculator can help ensure your dose matches your current weight and treatment goals while minimizing hormonal overcorrection.
Vaginal health changes during GLP-1 treatment
The term "Ozempic vulva" appeared in popular media and quickly captured attention, but the clinical reality behind it is more nuanced than the catchy name suggests. Vaginal health changes during semaglutide treatment are real, but they are not caused by the medication directly. They are downstream effects of the rapid body composition changes that semaglutide facilitates.
Vaginal dryness and decreased lubrication
When estrogen levels drop due to fat loss, the vaginal tissue can become thinner, less elastic, and less capable of producing adequate lubrication during arousal. This is the same mechanism that causes vaginal atrophy during menopause, but in the context of semaglutide, it happens faster because the weight loss occurs over months rather than years. The result is discomfort during intercourse, which naturally decreases interest in sexual activity regardless of desire levels.
This effect is more pronounced in perimenopausal and postmenopausal women, where baseline estrogen levels are already declining. Premenopausal women with functioning ovaries typically have enough estrogen production to compensate for the loss from adipose tissue, though exceptions exist, especially with very rapid or very significant weight loss.
Microbiome changes
Rapid weight loss and metabolic changes can alter the vaginal microbiome, potentially increasing susceptibility to yeast infections and bacterial vaginosis. While semaglutide does not cause these infections directly, the hormonal shifts it triggers can create a less hospitable environment for the lactobacilli that protect vaginal health. Women who experience recurrent infections during treatment should discuss this pattern with their provider rather than attributing it to unrelated causes.
Management strategies for vaginal health
Over-the-counter vaginal moisturizers, used regularly rather than just during sexual activity, can maintain tissue hydration. Water-based lubricants during intercourse address the immediate comfort issue. For more significant symptoms, low-dose vaginal estrogen creams or rings, which act locally rather than systemically, can restore vaginal tissue health without significantly affecting blood estrogen levels. These are prescription options that require a provider conversation but are highly effective and safe for most women. Maintaining overall tissue health through gut health optimization also supports the mucosal membranes throughout the body, including vaginal tissue, because the same inflammatory and microbiome factors affect both systems.
How GI side effects quietly sabotage intimacy
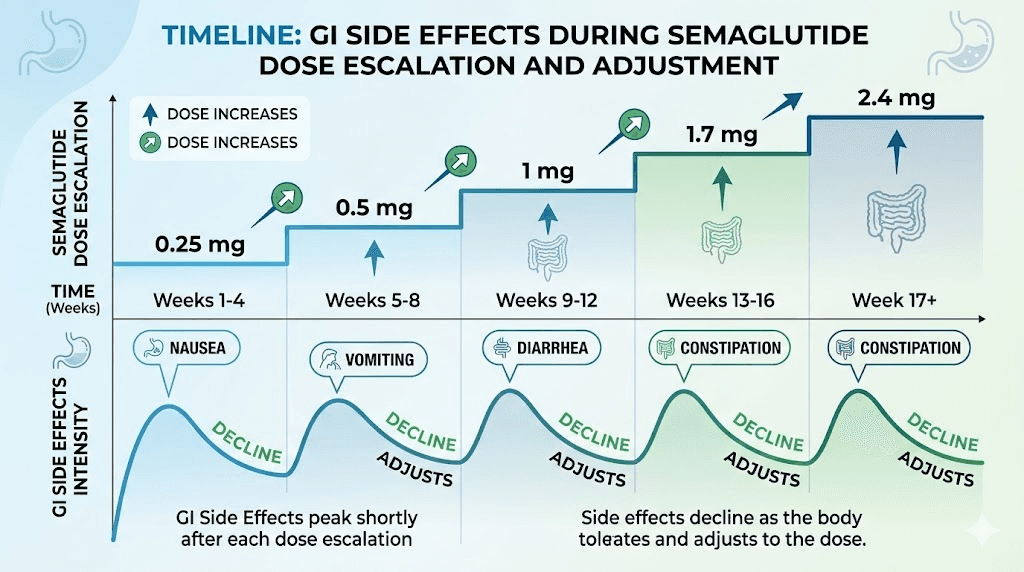
This section addresses the elephant in the room that nobody wants to discuss. Nausea is reported in 16 to 44 percent of semaglutide users, particularly during dose escalation phases. Add in constipation, bloating, acid reflux, and occasional diarrhea, and you have a recipe for intimacy avoidance that has nothing to do with hormones or neurotransmitters.
When your stomach feels like it is staging a revolt, sex is the last thing on most people agenda. This is not pathological. It is common sense. The body prioritizes discomfort signals over pleasure signals, and chronic nausea is a powerful deterrent to any activity that involves movement, physical contact, or vulnerability. The same digestive challenges that affect tirzepatide users apply to semaglutide, though the specific pattern and severity differ between the two medications.
The good news is that GI side effects are typically worst during the first few weeks of treatment and during each dose escalation. Most women find that these symptoms diminish significantly once they stabilize at their target dose. The timing of semaglutide administration can also make a difference. Some women find that injecting at a time that places the peak GI side effects during sleep rather than during evening hours when intimacy typically occurs helps preserve their interest in sex.
Dietary strategies also play a role. Eating smaller, more frequent meals, avoiding fatty or greasy foods during peak medication activity, and staying hydrated can all reduce GI symptoms. Our semaglutide diet plan guide covers nutrition strategies that minimize digestive discomfort while maintaining adequate nutrition. The list of foods to eat while on semaglutide provides specific meal ideas that are gentle on the digestive system.
If GI side effects persist beyond the adjustment period or are severe enough to significantly impact quality of life, dose adjustment may be warranted. Some women find that stepping back to a lower dose and titrating up more slowly resolves the issue without sacrificing weight loss efficacy. Others find that oral semaglutide formulations produce a different side effect profile than injections, though the overall efficacy data differs between delivery methods.
Menstrual cycle changes and what they mean for sexual health
Semaglutide does not directly regulate menstrual cycles, but the weight loss it produces absolutely does. And menstrual cycle regularity is intimately connected to sexual desire, arousal, and overall reproductive health in premenopausal women.
When women lose a significant amount of weight, the hormonal cascade that governs menstruation can shift dramatically. Estrogen and progesterone, the two hormones responsible for ovulation and menstruation, are both affected by body composition changes. Some women who had irregular periods before treatment find that their cycles normalize as they approach a healthier weight. Others, particularly those who lose weight very rapidly, may experience temporary irregularity, missed periods, or changes in flow.
The connection to sexual health is direct. Estrogen levels fluctuate throughout the menstrual cycle, and these fluctuations directly influence desire. Most women experience peak sexual desire around ovulation, when estrogen is highest. If semaglutide-induced weight loss disrupts ovulatory patterns, these natural desire peaks can flatten or disappear temporarily, leading to a subjective sense of decreased libido even when other systems are functioning normally. Tracking these patterns with the GLP-1 progress tracking tools can help women identify whether their sexual changes correlate with cycle disruptions.
For women using hormonal contraception, the interaction between semaglutide and contraceptive efficacy is worth discussing with a provider. While semaglutide does not directly interact with oral contraceptives, the delayed gastric emptying it causes could theoretically affect absorption of oral medications. The same gastric emptying delay that makes food choices matter during GLP-1 treatment can also affect how the body absorbs other oral medications. Most providers recommend using backup contraception or switching to non-oral methods during the initial months of semaglutide treatment. Our guide on GLP-1 medications during breastfeeding covers related reproductive health considerations.
Importantly, the improved menstrual regularity seen in women with PCOS on semaglutide, where 80 percent of responsive patients normalized their cycles, suggests that the medication can be profoundly positive for reproductive and sexual health in the right population. The key variable is whether the weight loss brings hormones into better balance or pushes them past the optimal range.
Anorgasmia and GLP-1 medications: what the case reports reveal
Perhaps the most concerning sexual side effect associated with GLP-1 agonists is anorgasmia, the inability to achieve orgasm despite adequate stimulation and desire. While decreased libido is frustrating, anorgasmia is a different experience entirely, and the emerging case report literature deserves attention.
The most detailed published case involves a 71-year-old postmenopausal woman who presented to a sexual medicine center reporting sudden loss of sexual arousal and inability to achieve orgasm. She described her condition as the "clitoris went to sleep all of a sudden." The timeline was telling. Anorgasmia developed within approximately two weeks of starting liraglutide, a daily GLP-1 agonist. When she was switched to semaglutide, a weekly GLP-1 agonist, the symptoms persisted for two additional months.
The resolution is equally informative. When the patient was switched to tirzepatide, a dual GIP/GLP-1 receptor agonist, orgasmic function returned within two weeks. This rapid reversal upon changing medications strongly suggests that the mechanism was pharmacological rather than psychological, and that different GLP-1 agonists may have different effects on sexual function.
Proposed mechanisms for GLP-1 anorgasmia
The researchers proposed two potential mechanisms. The peripheral mechanism involves GLP-1 agonist vasoconstriction of smooth muscle, which reduces blood flow and oxygen delivery to the genitals. Adequate genital blood flow is essential for clitoral engorgement and the rhythmic smooth muscle contractions that produce orgasm. If semaglutide constricts the blood vessels supplying the clitoral and vulvar tissue, the physical infrastructure for orgasm becomes compromised.
The central mechanism involves decreased dopamine and norepinephrine signaling in the hypothalamus, where these neurotransmitters are produced and released during sexual excitation. Without adequate dopamine and norepinephrine, the neurological signals required for orgasm may not reach sufficient intensity, regardless of physical stimulation.
The treatment implications differ based on which mechanism is responsible. If reduced genital blood flow is the primary issue, PDE-5 inhibitors (the same class of medications that includes sildenafil) could theoretically help by increasing blood flow to genital tissue. If central dopamine suppression is responsible, pro-dopaminergic agents like flibanserin or bremelanotide, both FDA-approved for hypoactive sexual desire disorder in premenopausal women, represent potential solutions. Understanding how fatigue differs between GLP-1 medications can also inform whether the issue is medication-specific or class-wide.
One case report does not establish prevalence, and it is entirely possible that anorgasmia on GLP-1 agonists is rare. But the mechanism is plausible, the timeline is consistent with a pharmacological cause, and the resolution upon medication change strongly supports the association. Women who develop new-onset orgasmic difficulty after starting semaglutide should not dismiss it as coincidence. The storage conditions of your medication and whether your semaglutide has expired can also affect medication potency and potentially contribute to unpredictable side effect profiles.
When sexual side effects typically start and when they usually resolve
Timing matters enormously when interpreting sexual side effects on semaglutide. Different mechanisms produce changes at different points in the treatment timeline, and understanding this can help distinguish temporary adjustments from persistent problems.
Weeks 1 to 4: the GI phase
During the initial weeks, particularly during the 0.25 mg starting dose and the first escalation to 0.5 mg, GI side effects dominate. Any decrease in sexual interest during this period is most likely attributable to nausea, fatigue, and general physical discomfort rather than neurochemical or hormonal changes. The early weeks of semaglutide treatment focus on adjustment, and sexual function typically recovers as GI symptoms improve.
Months 1 to 3: the neurochemical phase
As semaglutide reaches therapeutic levels and the brain adapts to altered GLP-1 signaling, changes in reward processing become more apparent. This is when dopamine-mediated effects on desire typically emerge. Women may notice they think about sex less frequently, feel less responsive to sexual cues, or experience a general flattening of desire. The neurochemical adaptation period varies significantly between individuals.
Months 3 to 6: the hormonal phase
As cumulative weight loss becomes significant, hormonal changes come into play. Estrogen shifts, testosterone fluctuations, and the metabolic consequences of sustained caloric deficit all influence sexual function during this period. This is also when the positive effects of weight loss on body image and confidence begin to counterbalance negative neurochemical effects, creating the paradoxical experience many women describe.
Months 6 and beyond: the new equilibrium
For most women, sexual function stabilizes after six months on a stable dose. The body has adapted to the medication, GI side effects have resolved, hormonal levels have reached a new baseline, and the brain reward system has partially recalibrated. Many women who experienced decreased desire during the first few months report that their libido returns to baseline or even improves by the six-month mark, particularly if they have experienced significant weight loss and improved body image.
Women who are planning long-term semaglutide use should be aware of this timeline and resist making permanent conclusions about their sexual function based on the first few months of treatment. Using tools like the GLP-1 progress plotter to track multiple variables over time, including sexual function, helps distinguish temporary disruptions from persistent concerns. Patience is warranted, though persistent symptoms beyond six months deserve medical evaluation.
10 practical strategies to support libido during semaglutide treatment
Theory is useful, but most women reading this article want to know what they can actually do. Here are ten evidence-informed strategies organized from simplest to most involved.
Strategy 1: optimize your dose timing
The best time to take semaglutide varies by individual, but choosing a day and time that minimizes GI side effects during times when intimacy is most likely can make a meaningful difference. Some women find that injecting on Monday morning allows side effects to peak during the workweek and diminish by the weekend. Experiment with timing to find what works for your schedule and relationship patterns.
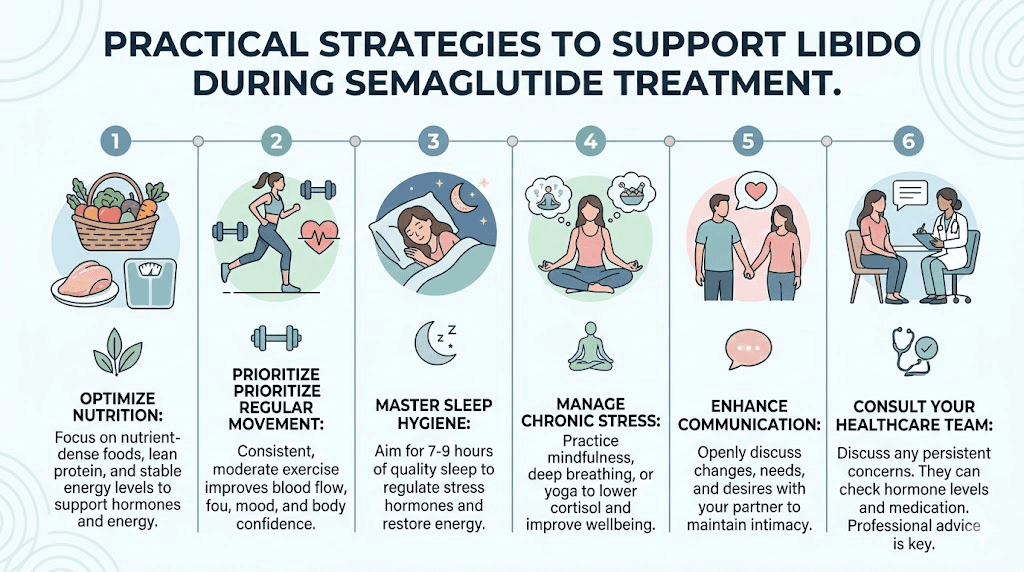
Strategy 2: prioritize protein and micronutrients
Caloric restriction on semaglutide can create nutritional deficiencies that compound sexual dysfunction. Zinc, iron, B vitamins, and vitamin D all play roles in sexual desire and arousal. Our guide to eating on semaglutide emphasizes nutrient density, which matters not just for body composition but for sexual health. B12 supplementation is particularly relevant because B12 supports nerve function, energy production, and mood regulation, all of which influence desire. Some providers recommend semaglutide with methylcobalamin specifically to address the energy and neurological support needs during treatment.
Strategy 3: address GI symptoms aggressively
Do not accept persistent nausea as an inevitable cost of treatment. Work with your provider to optimize your semaglutide dose and escalation timeline. Slower titration schedules often reduce GI side effects significantly without compromising long-term weight loss. Our dietary planning guide includes specific meal strategies that minimize nausea. If constipation is an issue, addressing it proactively removes another barrier to comfort during intimacy.
Strategy 4: consider supplementation with glycine
Glycine is an amino acid that supports collagen production, sleep quality, and neurotransmitter balance. Some women on GLP-1 agonists find that glycine supplementation helps with sleep quality and overall mood stability, both of which support healthy sexual function. Glycine also supports the production of glutathione, a powerful antioxidant that protects cellular health during the metabolic stress of rapid weight loss.
Strategy 5: exercise strategically
Regular exercise, particularly resistance training and cardiovascular exercise, naturally boosts dopamine, norepinephrine, and endorphin levels. If semaglutide is dampening these neurotransmitters, exercise can partially compensate. Aim for 150 minutes of moderate activity per week, with at least two sessions of resistance training. Exercise also preserves lean muscle mass during weight loss, which is critical for maintaining healthy hormone production and overall body composition.
Strategy 6: use vaginal moisturizers proactively
Do not wait for discomfort during sex to address vaginal dryness. Use a hyaluronic acid-based vaginal moisturizer several times per week as maintenance, regardless of sexual activity. This keeps tissue hydrated and comfortable, removing one barrier to desire. For women experiencing significant vaginal atrophy symptoms, discuss low-dose vaginal estrogen with a provider.
Strategy 7: get hormone levels tested
Request baseline hormone levels before starting semaglutide if possible, and follow-up testing at the three-month and six-month marks. Key markers include total and free testosterone, estradiol, DHEA-S, and thyroid function. These numbers provide concrete data for decision-making rather than guessing about the cause of sexual changes. The peptide calculator tools at SeekPeptides can help you track and optimize your protocols with precise measurements.
Strategy 8: slow your dose escalation
The standard semaglutide titration schedule moves from 0.25 mg to 0.5 mg to 1 mg to 1.7 mg to 2.4 mg at four-week intervals. Nothing prevents extending these intervals to six or even eight weeks if side effects are problematic. Many women find that slower escalation allows their bodies to adapt more gradually, reducing the severity of both GI and sexual side effects. Understanding semaglutide dosing in units helps when working with your provider to customize your titration schedule. For those on compounded formulations, the reconstitution process and mixing ratios allow for very precise dose adjustments.
Strategy 9: explore pro-dopaminergic supports
Certain supplements and lifestyle modifications can support dopamine production naturally. L-tyrosine is a precursor to dopamine synthesis. Adequate sleep supports dopamine receptor sensitivity. Novelty and new experiences stimulate dopamine release. If the primary mechanism of your decreased desire appears to be reward system dampening, these approaches can help restore some of the neurochemical balance. Some women also explore peptides for broader wellness as part of their overall optimization approach, while others find that compound formulations with glycine and B12 provide neurochemical support alongside their primary GLP-1 treatment. The peptide reconstitution calculator helps ensure accurate preparation of any supplementary protocols.
Strategy 10: consider medication switching if symptoms persist
The case report showing that anorgasmia resolved when a patient switched from semaglutide to tirzepatide raises an important possibility. Not all GLP-1 medications affect sexual function identically. Women considering affordable tirzepatide options may find that the switch addresses both cost and sexual side effect concerns. If sexual side effects are persistent and significantly impacting quality of life after exhausting other strategies, discussing a switch to a different medication class or a different GLP-1 agonist with your provider is reasonable. The side effect profiles differ between semaglutide and tirzepatide, and what does not work for one woman may work perfectly well for another.
When to talk to your provider and what to say
Many women hesitate to bring up sexual concerns with their prescribing provider, especially when the provider is focused on weight loss outcomes. But sexual health is health. A medication that achieves weight loss goals while destroying quality of life in the bedroom is not a complete success, and providers cannot address problems they do not know about.
When to schedule the conversation
Schedule a specific appointment to discuss sexual side effects if any of the following apply. You have experienced decreased desire or arousal that has persisted for more than eight weeks. You have developed new-onset difficulty reaching orgasm. You are experiencing vaginal dryness or pain during intercourse that is not responding to over-the-counter solutions. Your sexual changes are causing significant relationship distress. You have a history of SSRI-induced sexual dysfunction and are noticing similar patterns on semaglutide.
What to ask for
Come prepared with specific requests. Ask for hormone level testing, including testosterone, estradiol, and DHEA-S. Ask whether a slower dose escalation or a lower maintenance dose might reduce sexual side effects while preserving weight loss benefits. Use the semaglutide dosage calculator to explore what different dose levels look like in practical terms. Ask about the possibility of switching to a different GLP-1 medication if symptoms persist, and review the conversion protocols if a switch is recommended. Ask whether any medications approved for hypoactive sexual desire disorder might be appropriate as an adjunct to your current treatment.
If your provider dismisses your concerns, consider consulting a sexual medicine specialist. These providers have specific training in medication-induced sexual dysfunction and can offer targeted solutions that general practitioners may not be familiar with.
Semaglutide versus tirzepatide: does one affect sex drive less?
This is one of the most common questions women ask, and the honest answer is that we do not have enough data for a definitive comparison. But the available evidence provides some clues.
Semaglutide is a pure GLP-1 receptor agonist. Tirzepatide is a dual GIP/GLP-1 receptor agonist, meaning it activates both the GLP-1 receptor and the glucose-dependent insulinotropic polypeptide receptor. This dual mechanism produces different downstream effects on brain chemistry, appetite regulation, and potentially sexual function.
The case report demonstrating that anorgasmia on liraglutide and semaglutide (both pure GLP-1 agonists) resolved completely when the patient switched to tirzepatide is suggestive but not conclusive. It is a single case. However, the mechanism is plausible. GIP receptor activation may modulate the dopamine-suppressing effects of GLP-1 receptor activation, effectively buffering the sexual side effects.
The side effect profiles of semaglutide and tirzepatide differ in several respects. Tirzepatide may produce less nausea at equivalent weight loss levels, which removes one indirect contributor to decreased libido. The dosage comparison between the two medications shows different titration schedules and potency levels. For women considering a switch, understanding the conversion between semaglutide and tirzepatide is important for maintaining treatment continuity.
Tirzepatide onset of action and the timeline for results may also influence the overall experience. Some women find that the tirzepatide dosing approach allows for a more gradual adjustment that produces fewer acute side effects, including sexual ones. The tirzepatide dose chart and microdosing options provide flexibility that pure semaglutide formulations may not offer.
Future GLP-1 medications may offer even more options. Retatrutide, a triple receptor agonist targeting GLP-1, GIP, and glucagon receptors simultaneously, is in clinical development. The retatrutide dosing protocols are still being refined through clinical trials. Whether its broader receptor profile translates to different sexual side effects remains to be seen. The next generation of weight loss peptides continues to expand the options available to women who need effective weight management without sacrificing sexual health. Understanding the side effect profiles of emerging GLP-1 compounds will help women make informed choices as more options reach the market.
Nutritional deficiencies that quietly destroy libido during treatment
Semaglutide causes significant appetite reduction. That is the whole point. But when caloric intake drops substantially, nutritional deficiencies develop quickly, and several of those deficiencies directly impact sexual function in ways most women never connect to their diet.
Zinc deficiency
Zinc is essential for testosterone production in both men and women, and it plays a direct role in sexual arousal and lubrication. Women on calorie-restricted diets frequently develop subclinical zinc deficiency within months. The symptoms mimic medication side effects, reduced desire, fatigue, and mood changes, which means the nutritional component gets overlooked entirely. Our guide to semaglutide-friendly foods includes zinc-rich options. Red meat, oysters, pumpkin seeds, and chickpeas are among the best dietary sources. Supplementing with 15 to 30 mg daily during active weight loss is a reasonable strategy, though testing zinc levels through a simple blood test provides more precise guidance.
Iron deficiency
Women of reproductive age already face higher iron demands than men, and reduced food intake on semaglutide compounds the risk. Iron deficiency, even without full-blown anemia, causes fatigue that permeates every aspect of life, including sexual energy. The exhaustion from iron deficiency feels different from regular tiredness. It is a bone-deep weariness that makes the idea of physical activity, including sex, feel overwhelming rather than appealing. Getting adequate energy support during semaglutide treatment requires attention to iron status, especially for women with heavy menstrual periods.
Vitamin D and omega-3 fatty acids
Vitamin D deficiency is epidemic in the general population, and reduced dietary intake on semaglutide exacerbates it. Women already managing the fatigue that accompanies GLP-1 treatment may find that vitamin D deficiency compounds the problem substantially. Low vitamin D has been associated with decreased sexual satisfaction in women across multiple studies. Omega-3 fatty acids support hormone production, reduce inflammation, and maintain the health of mucous membranes throughout the body, including vaginal tissue. Women following a structured semaglutide diet plan that prioritizes fatty fish, eggs, and fortified foods will naturally address both deficiencies. For those who cannot eat enough, supplementation fills the gap.
B vitamin complex
The B vitamins, particularly B6, B12, and folate, support neurotransmitter synthesis, nerve function, and energy production. Semaglutide may impair B12 absorption through its effects on gastric acid production and delayed gastric emptying. Women who combine semaglutide with B12 supplementation or use methylcobalamin-enhanced formulations report better energy levels and mood stability, both of which support healthy sexual function. B6 specifically supports the conversion of tryptophan to serotonin and the production of dopamine, making it directly relevant to the neurochemical mechanisms discussed earlier.
The role of body composition and lean mass preservation
One factor that rarely enters the semaglutide and sex drive conversation is the impact of lean muscle mass loss on sexual function. Semaglutide produces weight loss, but not all of that weight loss comes from fat. Studies indicate that 25 to 40 percent of weight lost on GLP-1 agonists can be lean mass, including muscle tissue.
Muscle tissue is metabolically active. It contributes to overall energy production, hormone synthesis, and physical capacity. Women who lose significant muscle mass alongside fat may experience disproportionate fatigue, hormonal disruption, and reduced physical stamina, all of which affect sexual function. The early weeks of treatment are critical for establishing habits that preserve lean mass.
Resistance training, adequate protein intake (1.2 to 1.6 grams per kilogram of body weight daily), and strategic supplementation can significantly reduce lean mass loss during semaglutide treatment. Supplements that support muscle preservation during GLP-1 treatment include creatine, essential amino acids, and vitamin D. The nutritional guidelines for GLP-1 treatment apply equally to semaglutide users and emphasize protein-forward eating. Using the peptide stack calculator can help those exploring complementary protocols to support their overall wellness goals alongside semaglutide. Maintaining muscle mass preserves the metabolic foundation on which healthy hormone production and sexual function depend.
Women who combine semaglutide with a structured exercise program and adequate nutrition report better outcomes across virtually every metric, including sexual function. Tracking progress with tools like the peptide cost calculator helps manage the financial aspect of treatment while the performance optimization resources and muscle growth guides at SeekPeptides can help structure an approach that maximizes the benefits of treatment while minimizing unwanted side effects. Understanding real weight loss results from GLP-1 treatment gives realistic expectations for what body composition changes are achievable with proper lean mass preservation.
Sleep, stress, and the foundation of female desire
Sexual desire in women does not exist in a vacuum. It emerges from a complex interplay of physical health, hormonal status, psychological wellbeing, and relationship dynamics. Two foundational factors, sleep and stress, deserve special attention in the context of semaglutide treatment.
Sleep quality and semaglutide
Many women report improved sleep quality on semaglutide, partly because weight loss reduces sleep apnea risk and partly because reduced appetite means less late-night eating that disrupts sleep architecture. Choosing the optimal injection timing also plays a role in sleep quality. Better sleep directly supports sexual function through several mechanisms. Sleep is when the body produces the majority of sex hormones. Sleep deprivation reduces testosterone production in both men and women. And adequate sleep supports dopamine receptor sensitivity, partially counteracting the dopamine-dampening effects of semaglutide.
However, some women experience disrupted sleep during dose escalation phases, particularly if nausea or digestive discomfort interferes with nighttime rest. Addressing sleep disruption during these periods is important not just for general wellbeing but specifically for sexual health. The fatigue management guide includes strategies for maintaining energy levels during treatment that apply to sexual energy as well. Understanding the broader GLP-1 and fatigue connection helps contextualize why tiredness during treatment can feel so pervasive.
Stress management
Cortisol, the stress hormone, is one of the most powerful suppressors of female sexual desire. When cortisol is chronically elevated, the body prioritizes survival over reproduction, and sexual interest declines accordingly. Semaglutide treatment occurs within the context of real life, meaning work stress, relationship dynamics, parenting demands, and financial pressures all continue to operate alongside medication effects.
Women who attribute decreased desire entirely to semaglutide may be overlooking concurrent stressors that would suppress libido regardless of medication. Women dealing with hair loss concerns during GLP-1 treatment, for instance, often experience additional body image stress that compounds sexual side effects. Before concluding that semaglutide is the sole cause of sexual changes, honestly assess whether stress levels, relationship quality, and life circumstances have also shifted during the treatment period.
The relationship dimension: communication during body transformation
Semaglutide produces physical changes that affect both partners in a relationship, even though only one person is taking the medication. Rapid body transformation can shift relationship dynamics in unexpected ways, and these shifts can profoundly affect intimacy.
Some partners become more attracted as the semaglutide user loses weight. Others feel threatened by the change, consciously or unconsciously. Some women feel more sexually confident as they lose weight, especially those who have seen significant body transformation results on GLP-1 treatment. Others feel vulnerable and exposed in a smaller body, particularly if they used their weight as emotional armor. These psychological dynamics affect sexual desire in ways that no medication can address alone.
Open communication about sexual changes during semaglutide treatment is essential. Saying "I have noticed my desire has changed since starting this medication, and I want to work through it with you" is infinitely more productive than silently withdrawing from intimacy and leaving a partner to wonder why. Couples navigating these changes together might also explore whether anti-aging peptide protocols or other wellness approaches can complement the weight loss journey while supporting overall vitality and intimacy.
For women experiencing decreased desire while their partner experience is unchanged, scheduled intimacy, while it sounds unromantic, can actually help. The research on responsive desire versus spontaneous desire shows that many women experience desire in response to sexual activity rather than before it. If spontaneous desire has decreased on semaglutide, creating intentional opportunities for physical connection can activate the responsive desire system. The frustration of medication effects plateauing applies to sexual side effects too, where the initial disruption typically moderates over time as both partners adapt.
Long-term outlook: does libido recover after stopping semaglutide?
For women who ultimately decide to discontinue semaglutide, whether because of sexual side effects or other reasons, understanding the withdrawal and discontinuation timeline is important. Semaglutide has a half-life of approximately one week, meaning it takes about five weeks for the medication to fully clear the system after the last dose.
Sexual function changes that are primarily neurochemical, meaning dopamine or serotonin mediated, typically resolve within one to two months of discontinuation as the brain reward system recalibrates. Hormonal changes related to weight loss take longer to stabilize and may be permanent if the weight loss is maintained. Body image improvements tend to persist as long as the weight remains off.
The challenge, of course, is that discontinuing semaglutide often leads to weight regain, which brings its own set of sexual function implications. The challenges of weight management on semaglutide and the strategies for maintaining results are important considerations when weighing the decision to continue or stop treatment.
Women who experienced improved sexual function from the weight loss and body image benefits of semaglutide face a difficult trade-off. Stopping the medication may resolve neurochemical sexual side effects but reintroduce the psychological barriers to desire that existed before treatment. Working with both an obesity medicine specialist and a sexual health provider can help navigate this decision with full awareness of the trade-offs involved. Understanding the complete duration of semaglutide treatment helps frame these decisions within a realistic timeline rather than reacting to temporary side effects.
What the research still needs to answer
The current evidence base on semaglutide and female sexual function has significant gaps. No large-scale randomized controlled trial has specifically studied sexual side effects as a primary endpoint. Most of what we know comes from post-marketing surveillance, case reports, social media analysis, and clinical observation.
The questions that remain unanswered include the actual prevalence of sexual side effects (we do not know if it affects 5 percent or 30 percent of women), whether certain doses are more likely to cause sexual dysfunction than others, whether compounded semaglutide formulations produce different sexual side effect profiles than brand-name versions, whether specific pharmacy sources or compounding pharmacies produce formulations with different tolerability profiles, and whether pre-treatment screening can identify women at higher risk for sexual dysfunction on GLP-1 agonists.
Until these questions are answered, women and their providers must make decisions based on incomplete information, the principles of individualized medicine, and careful monitoring of symptoms over time. SeekPeptides continues to track the evolving research on GLP-1 medications and sexual health, providing evidence-based guidance as new studies emerge.
Comparing GLP-1 delivery methods and sexual side effects
Semaglutide is available in multiple formulations, and the delivery method may influence sexual side effects through differences in pharmacokinetics, side effect profiles, and patient experience.
Injectable semaglutide (Ozempic, Wegovy) is administered weekly via subcutaneous injection. The steady-state blood levels achieved with weekly dosing mean consistent GLP-1 receptor activation, which may produce more persistent effects on brain chemistry, including the dopamine and serotonin changes that affect sexual function. Understanding proper injection technique and GLP-1 injection guidelines ensures optimal absorption and consistent blood levels. For those using compounded injectable formulations, proper reconstitution of 5mg semaglutide or correct mixing ratios are essential for achieving consistent dosing that minimizes side effect variability.
Oral semaglutide formulations produce different pharmacokinetic profiles, with more variable absorption and potentially different peaks and troughs in blood levels. Whether this variability translates to different sexual side effects is unknown, but some clinicians theorize that less consistent receptor activation could mean fewer persistent neurochemical effects. The comparison between injectable and oral delivery methods extends beyond semaglutide to the broader peptide landscape.
Emerging delivery methods like GLP-1 patches represent another option that could change the side effect profile through different absorption characteristics. The tirzepatide oral drops and tablet formulations are also expanding the delivery method options beyond injections. Our comprehensive comparison of research and pharmaceutical peptides covers the broader landscape of formulation differences. As these options become more widely available, women will have more flexibility to choose delivery methods that optimize their treatment experience across all quality-of-life measures, including sexual function.
Frequently asked questions
Does semaglutide directly decrease sex drive in women?
Semaglutide does not have a single, direct effect on female sex drive. It influences multiple systems simultaneously, some of which can decrease desire (dopamine suppression, serotonin changes, hormonal shifts) and some of which can increase desire (weight loss, improved body image, better cardiovascular health). The net effect varies by individual. Clinical studies have not established decreased libido as an official side effect, but the emerging case report literature and proposed neurochemical mechanisms suggest it occurs in a subset of women.
How long do semaglutide sexual side effects last?
Most women who experience sexual side effects report that they are worst during the first three to four months of treatment, particularly during dose escalation. GI-related effects on intimacy typically resolve within the first six to eight weeks. Neurochemical effects on desire may persist longer but often improve as the body adapts. Most women reach a new equilibrium by six months. Women who are still experiencing significant sexual dysfunction after six months on a stable dose should consult their provider, as this may warrant intervention or medication adjustment. Understanding the overall semaglutide timeline helps set realistic expectations.
Can I take anything to boost my libido while on semaglutide?
Several approaches can support libido during treatment. B12 supplementation supports energy and nerve function. Glycine supports sleep quality and neurotransmitter balance. Regular exercise naturally boosts dopamine and endorphins. Adequate protein and micronutrient intake support hormone production. For more persistent issues, prescription options like flibanserin (for premenopausal women) or hormone replacement therapy may be appropriate, and your provider can guide these decisions.
Is tirzepatide better for sex drive than semaglutide?
Limited evidence suggests it may be. One published case report showed that anorgasmia present on both liraglutide and semaglutide resolved within two weeks of switching to tirzepatide. The dual receptor mechanism of tirzepatide (GIP + GLP-1) may buffer some of the dopamine-suppressing effects of pure GLP-1 agonism. However, this is based on a single case report, not large-scale comparative trials. The complete side effect comparison between the two medications covers additional differences worth considering.
Will my sex drive return to normal if I stop semaglutide?
Neurochemically mediated sexual side effects typically resolve within one to two months after discontinuation, as semaglutide clears the system and brain chemistry recalibrates. Hormonal changes related to weight loss may persist as long as the weight stays off. Body image improvements tend to maintain their positive effect on desire as long as the weight remains stable. The withdrawal timeline provides context for what to expect during and after discontinuation.
Does semaglutide dose affect sex drive more at higher amounts?
This has not been studied systematically, but the pharmacological principle of dose-response suggests that higher doses may produce more pronounced neurochemical effects, including effects on the reward system. Some women find that maintaining a lower dose (for example, 1.0 mg rather than escalating to 2.4 mg) achieves adequate weight loss with fewer side effects, including sexual ones. The semaglutide dosage calculator can help you and your provider explore dosing options.
Can semaglutide cause vaginal dryness?
Semaglutide itself does not directly cause vaginal dryness, but the rapid fat loss it produces can reduce estrogen levels because adipose tissue produces estrogen through aromatization. This estrogen decline can cause vaginal dryness, particularly in perimenopausal and postmenopausal women. Over-the-counter vaginal moisturizers and lubricants are first-line treatments, and low-dose vaginal estrogen is available by prescription for more significant symptoms.
Should I talk to my doctor about sex drive changes on semaglutide?
Yes. Sexual health is health. If you are experiencing changes in desire, arousal, or orgasmic function that are persisting beyond the initial adjustment period and affecting your quality of life or relationships, discuss this with your prescribing provider or a sexual medicine specialist. Specific requests to make include hormone level testing, dose optimization, and referral to a sexual health specialist if needed.
Does the semaglutide formulation matter for sex drive?
Compounded semaglutide and brand-name formulations contain the same active molecule, so the core neurochemical effects should be similar. However, compounded versions often include different additives or combination compounds that may affect overall tolerability and side effect profiles. The storage and handling of compounded formulations can also affect potency and consistency, which may influence how the medication affects various body systems.
Can exercise help maintain sex drive on semaglutide?
Yes. Regular exercise is one of the most effective strategies for supporting sexual desire during GLP-1 treatment. Resistance training and cardiovascular exercise naturally boost dopamine, norepinephrine, and endorphins, partially counteracting the neurotransmitter changes semaglutide produces. Exercise also preserves lean muscle mass, supports hormone production, improves cardiovascular function and blood flow to genital tissue, and enhances body image. Aim for at least 150 minutes of moderate activity weekly, incorporating both performance-oriented training and flexibility work.
Does alcohol make semaglutide sex drive effects worse?
Alcohol and semaglutide interact in multiple ways that can compound sexual side effects. Many women on semaglutide report reduced alcohol tolerance, and alcohol itself is a central nervous system depressant that further suppresses the dopamine signaling already dampened by semaglutide. Additionally, alcohol dehydrates mucous membranes, potentially worsening vaginal dryness. Limiting alcohol consumption during semaglutide treatment supports both weight loss goals and sexual health.
What if semaglutide is not working for weight loss but is affecting my sex drive?
If you are experiencing sexual side effects without corresponding weight loss benefits, this is an important conversation to have with your provider. The reasons semaglutide may not produce weight loss vary from dosage issues to metabolic factors. If the medication is causing sexual dysfunction without delivering its primary benefit, a dose adjustment, medication switch, or treatment reassessment may be warranted. Some women find that combining semaglutide with other approaches achieves better weight loss results at lower semaglutide doses, which may reduce sexual side effects.
How does semaglutide compare to phentermine for sex drive effects?
Phentermine and semaglutide work through completely different mechanisms. Phentermine is a stimulant that increases norepinephrine and dopamine, which can actually increase libido in some women, at least temporarily. Semaglutide modulates the reward system in the opposite direction. However, phentermine carries its own risks and is typically only approved for short-term use, making direct comparison complicated. Understanding whether phentermine is a GLP-1 medication (it is not) helps clarify these different mechanisms.
External resources
Anorgasmia following initiation of GLP-1 agonist, PMC Case Report
International Society for Sexual Medicine, GLP-1 Agonists and Sexuality
GLP-1 Receptor Agonists, Compulsive Behavior, and Libido, PMC Research
GLP-1 Agonist Weight Loss Medications and Sexual Desire, Journal of Sexual Medicine
For researchers committed to understanding the full picture of how GLP-1 medications affect female health, SeekPeptides provides evidence-based protocols, comprehensive guides, and a community of knowledgeable researchers navigating these exact questions with the depth and nuance they deserve.
In case I do not see you, good afternoon, good evening, and good night. May your hormones stay balanced, your desire stay present, and your research stay thorough.