Feb 20, 2026

Experienced researchers know something that newcomers to compounded semaglutide often miss. The pharmacy name on the vial matters more than the price on the invoice. And when it comes to BPI Labs, the story behind this Florida-based compounding facility is one that every informed researcher should understand before placing an order.
BPI Labs operates out of Largo, Florida as both an FDA-registered drug manufacturer and a 503B outsourcing facility. That dual registration is unusual. Most compounding pharmacies hold one designation or the other, not both. BPI claims to be the only FDA-registered drug manufacturer that also operates a 503B pharmacy producing GLP-1 medications, a claim that deserves scrutiny and context.
The company specializes in sterile injectable medications, servicing hospitals, clinics, and telehealth platforms across the United States. Their product lineup includes compounded versions of semaglutide and tirzepatide, the two GLP-1 receptor agonists that have dominated weight loss conversations. BPI Labs manufactures these compounds and distributes them through healthcare providers, telehealth companies, and pharmacy networks.
But here is where it gets complicated.
BPI Labs LLC is wholly owned by Belcher Pharmaceuticals LLC. That parent company carries a history that includes FDA scrutiny, executive fraud convictions, and regulatory violations spanning multiple years. Understanding BPI Labs means understanding that corporate lineage, because the manufacturing culture and compliance standards of a parent company inevitably influence its subsidiaries. This guide walks through every detail a researcher needs to make an informed decision about BPI Labs semaglutide, from product formulations and manufacturing processes to FDA history and the evolving legal landscape of GLP-1 compounding.
SeekPeptides has compiled this analysis by cross-referencing FDA inspection records, court documents, company filings, and verified user reports. The goal is not to make a purchasing recommendation. It is to give you the facts that most sources either do not have or choose not to share.
BPI Labs semaglutide products and formulations
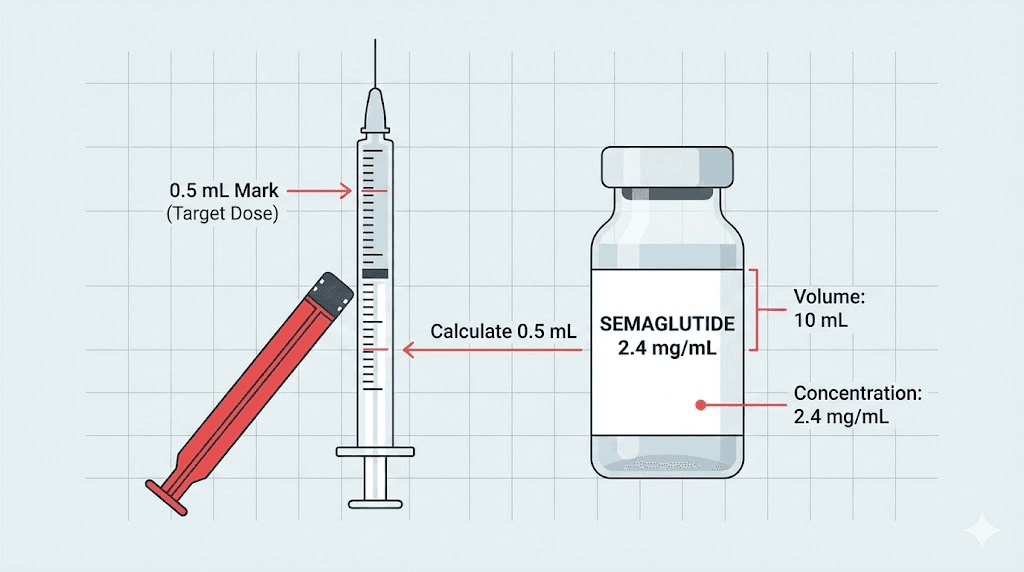
BPI Labs manufactures compounded semaglutide in injectable form. The most widely documented formulation is a 12.5mg/2.5ml vial, which works out to a concentration of 5mg/ml. This is a standard compounded semaglutide concentration that many providers use for dose titration across the typical escalation schedule.
What does that concentration mean in practice? At 5mg/ml, each 0.1ml drawn contains 0.5mg of semaglutide. A starting dose of 0.25mg would require drawing 0.05ml, or 5 units on a standard insulin syringe. These are small volumes that demand precise syringe measurement and an understanding of unit-to-milligram conversions.
The 12.5mg total per vial provides enough semaglutide for a multi-week supply depending on dose. At 0.25mg weekly, one vial lasts 50 weeks. At the higher 2.4mg weekly maintenance dose, one vial provides roughly five weeks of treatment. Use a semaglutide dosage calculator to determine exactly how long a vial will last at your specific dose.
Compounded semaglutide vs. FDA-approved versions
This distinction matters enormously. FDA-approved semaglutide (sold as Ozempic for diabetes and Wegovy for weight management) goes through rigorous clinical trials, manufacturing validation, and ongoing post-market surveillance. Compounded semaglutide, including what BPI Labs produces, is not FDA-approved. It has not been evaluated by the FDA for safety, effectiveness, or quality in the same way branded products have.
That does not automatically mean compounded semaglutide is unsafe. It means the regulatory oversight is different. A 503B outsourcing facility operates under different rules than a traditional pharmaceutical manufacturer, and those rules matter for quality assurance.
Compounded semaglutide from BPI Labs uses the same active pharmaceutical ingredient (API) as branded products. The company states that it sources semaglutide API and verifies its identity and purity through testing. However, the inactive ingredients, formulation process, and final product characteristics may differ from branded versions. BPI Labs has published FAQ pages addressing these questions, though the specific inactive ingredient lists require contacting the company directly for complete details.
Some compounded semaglutide products use semaglutide sodium salt rather than the base form used in FDA-approved products. The FDA has specifically warned about this distinction, noting that the salt form raises safety questions because it has not been studied in clinical trials. Whether BPI Labs uses the base form or salt form is a critical question that researchers should ask before accepting any compounded product.
BPI Labs tirzepatide products
Beyond semaglutide, BPI Labs also manufactures compounded tirzepatide with B6 (pyridoxine). This combination product reflects a broader trend among compounding pharmacies of adding vitamins or other compounds to GLP-1 formulations. The addition of B6 allows the product to be classified as a different formulation from the branded version, which has regulatory implications for compounding legality.
BPI Labs joined Empower Pharmacy in filing a petition with the U.S. Patent Trial and Appeal Board to challenge Eli Lilly patent claims on tirzepatide. This legal action demonstrates BPI interest in maintaining access to the compounded tirzepatide market and speaks to the broader industry battle over GLP-1 access and affordability.
For detailed comparisons between these two GLP-1 medications, including dosing equivalencies and side effect profiles, see our semaglutide vs. tirzepatide comparison and the complete dosage comparison chart.
Compounded semaglutide formulation variants you should know about
BPI Labs semaglutide is just one formulation in a crowded market of compounded GLP-1 products. Understanding the differences between formulation variants helps you evaluate what you are actually receiving and how it compares to alternatives.
Semaglutide with B12 (cyanocobalamin)
Many compounding pharmacies combine semaglutide with vitamin B12. The rationale is twofold. First, B12 supplementation may help offset potential nutritional deficiencies caused by reduced food intake on GLP-1 therapy. Second, the addition of B12 creates a formulation that is technically different from branded semaglutide, which has implications for compounding legality under FDA regulations. Whether BPI Labs offers a semaglutide-B12 combination or a standalone semaglutide product depends on the specific product ordered. The compounded semaglutide with B12 dosage chart provides concentration and dosing details for these combination products.
Semaglutide with methylcobalamin
Some compounders use methylcobalamin rather than cyanocobalamin as the B12 component. Methylcobalamin is a bioactive form of B12 that does not require conversion in the body. Our semaglutide methylcobalamin guide explains the practical differences between these formulations and what they mean for absorption and effectiveness.
Semaglutide with glycine
Glycine is an amino acid sometimes added to compounded semaglutide formulations as a stabilizer. The addition of glycine may help maintain peptide stability in solution, potentially extending shelf life. For a deep dive into this combination, see our semaglutide with glycine analysis. Similar combinations exist for tirzepatide, including tirzepatide with glycine and the triple combination of tirzepatide, glycine, and B12.
Why formulation details matter
The inactive ingredients in a compounded product affect more than just the label. They influence stability (how long the product remains potent), compatibility (whether ingredients interact with each other or with the syringe material), and tolerability (some excipients can cause injection site reactions in sensitive individuals). When evaluating any compounded semaglutide, including BPI Labs products, ask about the complete formulation, not just the active ingredient. Understanding how peptides work at the molecular level helps you appreciate why these formulation differences can have real-world effects on your results.
Manufacturing and quality control claims
BPI Labs makes strong claims about its manufacturing capabilities. Understanding these claims requires separating marketing language from verifiable fact.
The company states that it uses robotic isolator systems for injectable production. Inside ISO-grade barriers, humanless robotic technology performs every fill, cap, and inspection in complete isolation from external touchpoints. This is a legitimate manufacturing advantage. Robotic aseptic filling eliminates the primary contamination vector in sterile manufacturing, which is human intervention. If accurate, this places BPI Labs ahead of many smaller compounding pharmacies that rely on manual filling processes in cleanroom environments.
ISO-grade barriers refer to the International Organization for Standardization classifications for cleanroom environments. ISO Class 5, the standard for aseptic processing, allows no more than 3,520 particles per cubic meter at 0.5 micrometers or larger. Meeting this standard with robotic systems rather than human operators significantly reduces contamination risk. This matters because sterile manufacturing is the single most critical quality factor for injectable medications.
BPI Labs also references adherence to current Good Manufacturing Practice (cGMP) regulations. These are the FDA standards that all drug manufacturers must follow. For a 503B outsourcing facility, cGMP compliance includes process validation, equipment qualification, environmental monitoring, stability testing, and documented procedures for every manufacturing step.
What "FDA-registered" actually means
Here is a distinction that many researchers miss. "FDA-registered" does not mean "FDA-approved." Any facility that manufactures, repackages, or distributes drugs in the United States must register with the FDA. Registration is a legal requirement, not an endorsement of quality. It simply means the facility is known to the FDA and subject to inspection.
The more meaningful question is: what has the FDA found during its inspections? And for BPI Labs, the inspection history tells a more nuanced story than the marketing materials suggest. We will examine those inspection results in detail in a subsequent section of this guide.
Understanding the difference between research-grade and pharmaceutical-grade products is essential for any researcher evaluating compounding pharmacies. FDA registration is necessary but not sufficient for quality assurance. The inspection record, enforcement actions, and transparency of a facility tell you far more than registration status alone.

The Belcher Pharmaceuticals connection
This is where the BPI Labs story becomes significantly more complex. Every researcher considering BPI Labs products needs to understand the corporate ownership structure and the legal history of the parent company.
BPI Labs LLC is wholly owned by Belcher Pharmaceuticals LLC, a Florida-based pharmaceutical company. Belcher Pharmaceuticals is itself connected to GeoPharma, a broader pharmaceutical enterprise. The individuals involved in the leadership of these connected entities have a documented legal history that directly relates to pharmaceutical manufacturing integrity.
Executive fraud convictions
Mihir Taneja, who served as CEO of GeoPharma (the parent entity), pleaded guilty on November 30, 2020, to "introduction of misbranded drugs into interstate commerce with the intent to defraud and mislead." This was not a minor regulatory infraction. The Department of Justice described it as part of a "Nationwide Telemedicine Pharmacy Health Care Fraud Conspiracy."
Taneja was sentenced to 10 months in federal prison plus one year of supervised release, and was ordered to pay nearly $21 million. He had co-founded Sterling-Knight, a pharmacy operation described in court documents as a sham pharmacy involved in the fraud scheme.
Arun Kapoor, identified as the Director of Business Development, was also convicted in connection with the same fraud scheme. Both individuals were involved in operations that the DOJ linked to fraudulent telemedicine practices and misbranded drug distribution.
What does this mean for BPI Labs today? The company has emphasized that these convictions involved individuals in their prior roles, not actions taken at BPI Labs. The company maintains that it operates independently with its own compliance framework. However, corporate culture and compliance standards flow from the top. When a parent company history includes fraud convictions related to pharmaceutical manufacturing and distribution, it raises legitimate questions about the culture of compliance that extends to subsidiaries.
What researchers should consider
Past conduct does not necessarily predict future behavior. Companies can and do reform. New leadership, new compliance programs, and stronger internal controls can transform an organization. The question is whether sufficient evidence exists to demonstrate that transformation.
For researchers evaluating BPI Labs products, the Belcher connection is one data point among many. It should not be the sole factor in a decision, but it should not be ignored either. Combined with the FDA inspection history (detailed below), it paints a picture that requires careful consideration. The evaluation of any peptide or pharmaceutical vendor should include corporate history as part of a comprehensive due diligence process.
FDA inspection history and compliance record
FDA inspections of compounding facilities produce documents called Form 483s. These are official notices issued when an investigator observes conditions that, in their judgment, may constitute violations of the Food, Drug, and Cosmetic Act. A Form 483 is not a final determination of violation, but it identifies areas of concern that the facility must address.
BPI Labs has received Form 483 observations that reveal specific concerns about its manufacturing processes. Understanding these findings is critical for any researcher evaluating the quality of BPI Labs semaglutide.
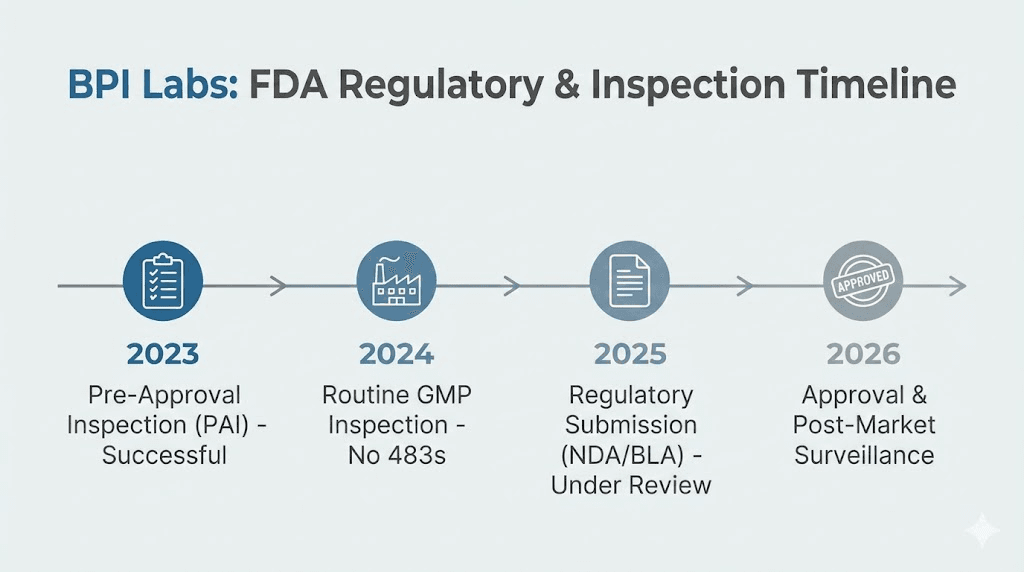
The 2021 FDA inspection
Following a 2021 inspection of BPI Labs, the FDA detailed numerous violations. The key findings included failure to use proper equipment, failure to adequately vet suppliers, and failure to thoroughly assess the risks associated with injectable drug products.
These are not minor paperwork issues. For a facility producing sterile injectables like semaglutide injections, equipment qualification and supplier vetting are foundational quality requirements. Using improper equipment can affect dose accuracy, sterility, and product stability. Failing to vet suppliers means the raw materials entering the manufacturing process may not meet the specifications necessary for safe injectable products.
BPI Labs has stated that this audit was closed with satisfactory compliance and that subsequent products received final FDA approval. The company also notes that no Warning Letters were issued. A Warning Letter is a more severe regulatory action than a Form 483, indicating that the FDA believes violations are significant enough to warrant formal enforcement consideration. The absence of a Warning Letter suggests the FDA accepted the corrective actions BPI Labs implemented.
February 2025 observations
More recently, in February 2025, FDA investigators observed additional violations at BPI Labs. These included improper labeling of compounded semaglutide and tirzepatide products. Labeling violations in pharmaceutical manufacturing can range from minor formatting issues to serious problems that affect patient safety, such as incorrect concentration statements or missing storage instructions.
The specific nature of the labeling violations observed at BPI Labs has not been fully detailed in public records. However, any labeling issue with injectable GLP-1 medications is concerning because dosing accuracy depends on correct concentration labeling. If a vial is labeled at 5mg/ml but the actual concentration differs, the dosage calculations a researcher performs will produce incorrect amounts.
Belcher Pharmaceuticals 2024 inspection
The parent company, Belcher Pharmaceuticals, also underwent FDA inspection in 2024. Investigators found numerous violations, including "failure to conduct testing for impurities" before releasing injectable testosterone packages. This finding is particularly relevant because impurity testing is one of the most fundamental quality control measures in pharmaceutical manufacturing.
When a company fails to test for impurities in one product line, it raises questions about whether similar gaps exist in other product lines. Researchers evaluating BPI Labs semaglutide should consider whether the quality control culture at the parent company influences practices at the subsidiary. The importance of third-party testing becomes even more apparent when a manufacturer inspection history reveals gaps in internal testing protocols.
Industry-wide context
It is worth noting that FDA inspections of 503B outsourcing facilities frequently identify violations. Of the 55 503B facilities that have been inspected, all but two (96%) received Form 483 observations. This does not excuse BPI Labs violations, but it provides context. The compounding industry as a whole faces significant quality challenges, and even well-regarded facilities often receive inspection findings.
The question is not whether a facility has ever received a Form 483, but what the findings reveal about systemic quality issues versus isolated incidents, and how the facility responds to findings. A facility that rapidly corrects issues, implements preventive measures, and demonstrates improvement in subsequent inspections shows a different compliance trajectory than one with recurring similar findings.
The evolving landscape of semaglutide compounding
The legal status of compounded semaglutide has shifted dramatically, and BPI Labs sits at the center of these changes. Understanding this regulatory landscape is essential for researchers who source semaglutide from any compounding pharmacy, not just BPI Labs.
The shortage that started it all
Compounding pharmacies were able to produce semaglutide because the FDA maintained semaglutide on its Drug Shortage List. Under federal law, 503B outsourcing facilities can compound drugs that are "currently in shortage" without individual patient prescriptions. This shortage designation opened the door for facilities like BPI Labs to manufacture compounded semaglutide at scale.
The shortage was real. Demand for semaglutide and other GLP-1 medications exploded as weight loss applications gained mainstream attention. Novo Nordisk, the manufacturer of Ozempic and Wegovy, could not produce enough to meet demand. Patients faced months-long waits, and compounding pharmacies stepped in to fill the gap.
The shortage resolution
On February 21, 2025, the FDA determined that the shortage of semaglutide injection products was resolved. This single decision had massive implications for the entire compounding industry. With the shortage resolved, the legal basis for 503B bulk compounding of semaglutide evaporated.
The FDA provided transition periods to avoid disrupting patient treatment. For 503A compounding pharmacies (which fill individual prescriptions), the deadline was April 22, 2025. For 503B outsourcing facilities like BPI Labs (which can produce without individual prescriptions), the deadline was May 22, 2025. After these dates, facilities could no longer rely on the shortage exception for compounding "essentially copies" of semaglutide products.
Legal challenges and ongoing uncertainty
The compounding industry did not accept the shortage resolution quietly. The Outsourcing Facilities Association filed a lawsuit against the FDA on February 24, 2025, challenging the determination that the semaglutide shortage had been resolved. The FDA indicated it would delay enforcement action until the District Court issued a decision on the preliminary injunction motion or until the stated deadlines, whichever came later.
BPI Labs separately joined Empower Pharmacy in challenging Eli Lilly tirzepatide patent through the Patent Trial and Appeal Board. While this challenge targets tirzepatide rather than semaglutide, it demonstrates the broader strategy of compounding facilities fighting to maintain market access.
For researchers, this regulatory uncertainty creates practical questions. Can BPI Labs still legally manufacture compounded semaglutide? The answer depends on the specific legal mechanism being used. Some compounders have shifted to 503A operations (requiring individual prescriptions). Others argue that their formulations are "not essentially copies" of branded products because they contain different inactive ingredients or additional compounds like B12 or glycine. The legal landscape remains fluid, and staying informed about regulatory changes is critical for anyone relying on compounded products for their GLP-1 therapy.
The broader question extends beyond semaglutide. The FDA approach to GLP-1 compounding will set precedents that affect access to peptide therapy across many categories. If regulatory enforcement tightens significantly, researchers may need to explore alternative pathways for accessing these medications, including local peptide therapy clinics that operate under 503A pharmacy relationships with patients.
What "compounded" means in this new landscape
The distinction between different types of compounding is important. A 503B outsourcing facility can produce medications in bulk without individual prescriptions, similar to a drug manufacturer. A 503A compounding pharmacy fills prescriptions for individual patients, similar to a traditional pharmacy. Some facilities operate under both designations.
BPI Labs, which holds both FDA drug manufacturer registration and 503B designation, may have more regulatory flexibility than pure 503B facilities. However, the specific rules governing what BPI Labs can and cannot compound depend on the current shortage list status and any applicable court orders. Researchers should verify the current legal status before obtaining compounded semaglutide from any source, including understanding the difference between different types of pharmaceutical operations.
BPI Labs partnerships and distribution
BPI Labs does not sell directly to individual consumers. Instead, it manufactures compounded medications that are distributed through healthcare providers, telehealth platforms, and pharmacy networks. The most prominent partnership has been with Hims and Hers Health, one of the largest telehealth companies in the United States.
The Hims and Hers connection
Hims offered compounded semaglutide manufactured by BPI Labs for approximately $199 per month, significantly less than the branded versions of semaglutide. This pricing made GLP-1 therapy accessible to millions of people who could not afford or access branded Ozempic or Wegovy.
However, the consultation process raised concerns among healthcare professionals. Reports indicated that compounded semaglutide was offered via prescription as quickly as 20 minutes after completing an online survey. The consultation process reportedly involved automated messages about injection technique rather than comprehensive medical evaluation.
This matters because semaglutide is not appropriate for everyone. Contraindications include personal or family history of medullary thyroid carcinoma, multiple endocrine neoplasia syndrome type 2, and pancreatitis history. A thorough medical evaluation is essential before starting any GLP-1 therapy, whether branded or compounded. Rushing through that evaluation increases the risk of adverse outcomes.
Other distribution channels
Beyond Hims, BPI Labs products are distributed through various healthcare clinics, medical spas, and telehealth platforms across the country. Some of these providers offer more comprehensive medical oversight than others. The quality of medical supervision varies dramatically from one provider to another, which means the same BPI Labs vial can be part of either a well-managed treatment program or a minimal-oversight operation.
When evaluating any provider that uses BPI Labs products, consider the level of medical supervision, required lab work, follow-up protocols, and dose adjustment procedures. A provider that requires baseline labs, regular follow-ups, and individualized dosing demonstrates more commitment to patient safety than one that simply ships vials after a brief questionnaire. For guidance on proper GLP-1 injection technique and injection site selection, consult evidence-based resources rather than relying solely on provider communications.
How BPI Labs compares to other compounding pharmacies
BPI Labs is one of several major players in the compounded GLP-1 market. Understanding how it compares to alternatives helps researchers make informed decisions about product sourcing.
BPI Labs vs. Empower Pharmacy
Empower Pharmacy, headquartered in Houston, Texas, is another major 503B outsourcing facility and 503A compounding pharmacy. Empower has been one of the most prominent compounders of semaglutide and tirzepatide, and the two companies share a notable connection: they have collaborated on legal challenges to tirzepatide patents.
Key differences include scale, regulatory approach, and transparency. Empower operates what it describes as a cGMP manufacturing facility and has been more publicly transparent about its formulations and quality control processes. BPI Labs emphasizes its robotic isolator technology and dual FDA manufacturer/503B designation.
Both companies have faced FDA scrutiny. The question is not which company has a perfect record, because neither does. The question is which company demonstrates a stronger trajectory of compliance improvement and transparency.
BPI Labs vs. Olympia Pharmacy
Olympia Pharmacy is another frequently mentioned compounder in the GLP-1 space. Olympia has taken a different approach to formulation, often combining semaglutide with B12 (cyanocobalamin) as a standard offering. The addition of B12 serves a dual purpose: providing a potential nutritional benefit for patients on calorie-restricted diets and creating a formulation that is technically different from the branded product.
Comparison overview
Factor | BPI Labs | Empower Pharmacy | Olympia Pharmacy |
|---|---|---|---|
Facility type | FDA manufacturer + 503B | 503B + 503A | 503B + 503A |
Location | Largo, FL | Houston, TX | Orlando, FL |
Robotic filling | Yes (ISO-grade isolators) | Automated systems | Varies |
FDA 483 history | 2021 + 2025 findings | Documented findings | Documented findings |
Parent company concerns | Belcher fraud convictions | None publicly documented | None publicly documented |
Semaglutide formulations | Semaglutide injectable | Semaglutide + B12 | Semaglutide + B12 |
Tirzepatide | Tirzepatide + B6 | Tirzepatide + B6 | Tirzepatide formulations |
Transparency | Limited public detail | More publicly available | Moderate |
This comparison is not exhaustive, and individual experiences may vary. The compounded GLP-1 market changes rapidly as regulatory pressures and legal challenges reshape the industry. For the most current vendor evaluations, check our complete vendor guide and the grey market peptides guide for understanding sourcing nuances.
How to evaluate any compounding pharmacy for GLP-1 medications
Whether you are considering BPI Labs or any other compounding pharmacy, a systematic evaluation framework protects you from quality and safety risks. Too many researchers focus exclusively on price when choosing a compounding source. Price matters. But it should never be the primary factor for an injectable medication that goes directly into your body.
Check FDA registration and inspection records
Every legitimate compounding facility should be registered with the FDA. You can verify registration through the FDA online database. But registration alone tells you very little. The inspection record tells you much more.
FDA inspection reports (Form 483s) are available through Freedom of Information Act requests. These documents reveal exactly what FDA investigators observed during facility inspections. Look for patterns: are the same types of violations appearing across multiple inspections, or do findings change, suggesting the facility addresses problems? Recurring findings in the same areas (sterility, equipment, supplier vetting) suggest systemic issues rather than isolated incidents.
Understand the 503A vs. 503B distinction
A 503A pharmacy compounds medications based on individual patient prescriptions. A 503B outsourcing facility can compound in bulk without individual prescriptions. Both are legal, but they operate under different regulatory frameworks. 503B facilities face more stringent manufacturing requirements because they produce at larger scale, including cGMP compliance, adverse event reporting, and regular FDA inspections.
Ask your provider which type of facility compounds their medications. Then verify that information independently. The distinction between different pharmaceutical regulatory frameworks directly affects the quality controls applied to the products you receive.
Evaluate transparency
Quality compounding pharmacies are transparent about their operations. They willingly share information about API sources, testing protocols, certificates of analysis, stability testing results, and inspection history. A pharmacy that deflects questions about these topics or provides vague answers is a red flag.
Specific questions to ask include:
Where do you source your semaglutide API?
Do you use semaglutide base or semaglutide sodium salt?
Can you provide a certificate of analysis for the specific lot?
What stability testing have you conducted?
What is the beyond-use date for your compounded semaglutide?
How do you verify potency and sterility of finished products?
These are not unreasonable requests. Any reputable pharmacy should be able to provide this information. For understanding the true cost of quality compounded medications, recognize that extensive testing and quality control add cost. If a price seems impossibly low, the quality controls may be insufficient.
Look for third-party testing
The gold standard for product verification is independent third-party testing. Some compounding pharmacies submit their products to independent laboratories for identity, potency, sterility, and endotoxin testing. This provides an external check on internal quality control processes.
Ask whether the compounder uses third-party testing and whether they will share results. Facilities like independent peptide testing laboratories can verify the identity and purity of compounded medications. Some researchers choose to independently test compounded products they receive, particularly from new or unfamiliar sources.
Red flags to watch for
Certain indicators suggest a compounding pharmacy may not meet quality standards:
No FDA registration or refusal to provide registration number
No certificates of analysis available
Prices dramatically lower than competitors without explanation
No physician oversight required for obtaining prescriptions
Claims that compounded products are "the same as" FDA-approved versions
Inability or unwillingness to describe quality control procedures
History of FDA Warning Letters (more serious than Form 483s)
No stability testing data for their specific formulations
Avoiding common mistakes when selecting pharmaceutical products starts with understanding that the cheapest option is rarely the safest option, and the most expensive option is not automatically the best.
Safety considerations for compounded semaglutide
The FDA has issued specific warnings about compounded GLP-1 medications that every researcher must understand. These warnings apply to compounded semaglutide from any source, including BPI Labs.
Adverse event reports
Between February 2019 and December 2023, the FDA received 352 adverse event reports related to compounded GLP-1 drugs. These reports included five deaths. While adverse event reports do not establish causation (the events may have been caused by factors unrelated to the compounded medication), the volume of reports signals a need for heightened caution.
Common adverse events with semaglutide, whether branded or compounded, include nausea, vomiting, diarrhea, constipation, and injection site reactions. These are expected GLP-1 side effects related to the mechanism of action. More serious adverse events can include pancreatitis, gallbladder problems, and severe gastrointestinal reactions. For managing common side effects, see our guides on semaglutide constipation treatment and semaglutide fatigue.
The concern with compounded products specifically is that manufacturing quality issues could introduce additional risks beyond those inherent to the medication itself. Contamination, incorrect potency, improper formulation, or inadequate sterility could all contribute to adverse events that would not occur with properly manufactured products.
The salt form question
One of the most important safety considerations for compounded semaglutide is the form of the active ingredient. FDA-approved semaglutide products use semaglutide in its base form. Some compounding pharmacies have used semaglutide sodium salt, which is a different chemical entity.
The FDA has warned that semaglutide sodium salt has not been studied in clinical trials and may have different properties than the base form. Differences could include absorption rate, bioavailability, stability, and side effect profile. When evaluating any compounded semaglutide product, including those from BPI Labs, determining which form is used is a critical safety question.
Storage and handling
Compounded semaglutide typically requires refrigeration, similar to branded products. However, the stability profile of compounded formulations may differ from FDA-approved versions because the excipients and formulation process are different. Follow the specific storage instructions provided with your compounded product, and consult resources on compounded semaglutide refrigeration requirements and shelf life after opening.
Proper storage is not optional. Temperature excursions can degrade the peptide, reducing potency or creating breakdown products that may cause adverse reactions. If you receive a compounded semaglutide vial and it appears cloudy, discolored, or contains particles, do not use it. Clear, colorless solution is the expected appearance for properly compounded semaglutide. Understanding peptide storage fundamentals protects both product integrity and your safety.
Dose verification
Because compounded semaglutide concentrations may differ from branded products, verifying your dose is essential. Use a semaglutide dosage calculator to confirm that the volume you are drawing corresponds to the intended milligram dose based on the concentration stated on your vial.
For example, a BPI Labs vial at 5mg/ml concentration requires different syringe volumes than a branded Ozempic pen. Drawing 10 units (0.1ml) from a 5mg/ml vial delivers 0.5mg of semaglutide. Understanding semaglutide dosing in units and unit-to-milligram conversions prevents dosing errors that could lead to either subtherapeutic dosing or overdose.
Understanding semaglutide dosing from compounded vials
Dosing semaglutide from a compounded multi-dose vial is fundamentally different from using a branded pre-filled pen. The branded Ozempic pen has pre-set dose clicks. A compounded vial requires manual calculation and syringe measurement. Getting this right is non-negotiable.
Standard dose escalation
The typical semaglutide dose escalation schedule follows this pattern:
Weeks 1-4: 0.25mg once weekly
Weeks 5-8: 0.5mg once weekly
Weeks 9-12: 1.0mg once weekly
Weeks 13-16: 1.7mg once weekly
Week 17 onward: 2.4mg once weekly (maintenance dose for weight management)
This escalation exists for a reason. Starting at a lower dose allows your body to adapt to GLP-1 receptor activation, reducing the severity of gastrointestinal side effects. Rushing the escalation increases the likelihood of severe nausea, vomiting, and other adverse effects. For detailed information on how quickly semaglutide produces results, understand that therapeutic effects begin even at lower starting doses.
Converting concentration to dose volume
With a BPI Labs 5mg/ml vial, the math works like this:
Target dose | Volume to draw | Insulin syringe units |
|---|---|---|
0.25mg | 0.05ml | 5 units |
0.5mg | 0.10ml | 10 units |
1.0mg | 0.20ml | 20 units |
1.7mg | 0.34ml | 34 units |
2.4mg | 0.48ml | 48 units |
These calculations assume exactly 5mg/ml concentration. If the concentration differs, every number in this table changes. Always verify the concentration printed on your specific vial before calculating doses. For more detailed conversion tables, see our semaglutide 5mg/ml dosage chart and the complete conversion chart.
Syringe selection and technique
Use a U-100 insulin syringe for drawing compounded semaglutide. The 0.3ml (30-unit) syringe provides the most accurate measurement for lower doses. The 0.5ml (50-unit) syringe works better for higher doses. The 1.0ml (100-unit) syringe is generally unnecessary for semaglutide dosing and offers less precision at the small volumes required for starting doses.
Proper injection technique matters as much as dose accuracy. Subcutaneous injection into the abdomen, thigh, or upper arm is standard for semaglutide. Rotate injection sites to prevent lipodystrophy (hardening of tissue at injection sites). Our guide on how to inject GLP-1 medications covers technique in detail.
Reconstitution vs. ready-to-use
BPI Labs semaglutide vials are typically supplied as ready-to-use liquid formulations rather than lyophilized (freeze-dried) powder requiring reconstitution. This is an important distinction. Ready-to-use formulations eliminate the reconstitution step and the associated risks of using incorrect diluent volumes or contaminating the product during mixing.
If you ever encounter a semaglutide product that requires reconstitution, use only bacteriostatic water as specified by the compounder. The volume of diluent determines the final concentration, so precise measurement using the peptide reconstitution calculator is essential. For specific guidance, see our articles on mixing bacteriostatic water with 10mg semaglutide and the 5mg mixing guide.
What to expect on compounded semaglutide
Regardless of whether your semaglutide comes from BPI Labs, another compounder, or a branded manufacturer, the medication works through the same mechanism. It activates GLP-1 receptors in the brain and gut, reducing appetite, slowing gastric emptying, and improving insulin sensitivity. The therapeutic effects should be similar if the compounded product contains the correct amount of properly formulated semaglutide.
Timeline of effects
Most people notice appetite reduction within the first one to two weeks of treatment, even at the starting dose of 0.25mg. This is among the earliest signs that the medication is working. For a detailed breakdown, see our guide on how long semaglutide takes to suppress appetite.
Weight loss typically becomes measurable by weeks four through eight. The rate varies significantly between individuals, influenced by starting weight, diet, activity level, and metabolic factors. If you are not seeing results after four weeks, our guide on four weeks on semaglutide with no weight loss covers troubleshooting steps. For broader troubleshooting, see why you might not be losing weight on semaglutide.
Maximum weight loss effects are typically seen between months three and six, with continued but slower progress afterward. The question of how long to stay on semaglutide depends on individual goals, response, and medical guidance.
Common side effects and management
The most frequently reported side effects of semaglutide include nausea (especially in the first weeks and after dose increases), constipation, diarrhea, abdominal pain, and fatigue. These effects are dose-dependent and tend to improve with time as the body adjusts.
Managing these side effects involves several strategies:
Eat smaller, more frequent meals rather than large portions
Avoid high-fat and overly processed foods that worsen nausea
Stay hydrated, as GLP-1 medications can contribute to dehydration
Follow the dose escalation schedule without skipping steps
Consider timing of injection relative to meals and activities
For dietary guidance, our articles on the semaglutide diet plan and foods to eat while on semaglutide provide specific meal planning strategies. The best time of day to take semaglutide can also influence side effect severity.
For questions about combining semaglutide with other substances, we have detailed guides covering alcohol and semaglutide, phentermine and semaglutide, and semaglutide with B12.
When to be concerned
Certain symptoms require immediate medical attention regardless of the semaglutide source. These include severe abdominal pain (possible pancreatitis), changes in vision, signs of thyroid tumors (lump in the neck, difficulty swallowing, hoarseness), severe allergic reactions, and signs of rebound effects after stopping.
The difference between expected side effects and warning signs is important to understand. Expected nausea after a dose increase is normal. Persistent severe vomiting that prevents adequate fluid intake is not. Mild fatigue during dose escalation is common. Extreme exhaustion that impairs daily function warrants evaluation. If you experience persistent burping or hair thinning, these may be related to your GLP-1 therapy and deserve investigation.
Proper storage and handling of compounded semaglutide from BPI Labs
Storage matters more than most researchers realize. A perfectly compounded semaglutide product can become ineffective or even harmful through improper handling. And compounded products may have different stability profiles than branded versions because of differences in formulation and preservatives.
Refrigeration requirements
Compounded semaglutide from BPI Labs should be stored refrigerated at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). This is consistent with the standard storage requirements for compounded semaglutide products. Do not freeze compounded semaglutide, as freezing can damage the peptide structure and alter potency.
Once removed from refrigeration for an injection, allow the vial to reach room temperature briefly before drawing your dose. Cold injections can be more uncomfortable. However, do not leave the vial at room temperature for extended periods. Return it to the refrigerator promptly after use. For detailed guidance on how long compounded semaglutide lasts in the fridge, consult our complete storage guide.
Expiration and beyond-use dating
Compounded medications have "beyond-use dates" rather than traditional expiration dates. The beyond-use date is determined by the compounder based on their stability testing data. It may be significantly shorter than the expiration date on branded products. Ask your provider about the beyond-use date for BPI Labs semaglutide and do not use the product beyond that date. For guidance on whether expired semaglutide retains any potency, understand that the answer depends on many factors including storage conditions and formulation type. The general recommendation is to discard expired products. If you are managing multiple peptide vials, understanding overall peptide shelf life in refrigeration helps you organize your supply effectively.
Travel considerations
If you need to travel with semaglutide, maintaining the cold chain is essential. Use an insulated travel case with cold packs. Do not pack semaglutide in checked luggage where temperature extremes in the cargo hold can damage the product. Keep it in your carry-on with appropriate documentation from your healthcare provider. Understanding how long peptides last at room temperature helps you plan for situations where refrigeration is temporarily unavailable.
Verifying product integrity
Before each injection, visually inspect your BPI Labs semaglutide vial. The solution should be clear, colorless, and free of particles. Cloudiness, discoloration, visible particles, or an unusual odor are all signs of potential degradation or contamination. Do not use a product that fails visual inspection. This basic check is one of the most important safety practices for anyone using compounded injectable medications, and it applies equally to all peptide products regardless of source.
Practical tips for researchers using compounded semaglutide
Beyond choosing a source and understanding dosing, several practical considerations affect the quality of your semaglutide experience. These apply whether you use BPI Labs or any other compounder.
Tracking your response
Keep a log of your dose, injection date, injection site, and any side effects. This information is invaluable for troubleshooting and for conversations with your healthcare provider. If you switch between compounders or formulations, detailed tracking helps you identify whether changes in response are related to the product switch or other factors. The first month on semaglutide is especially important for establishing your baseline response.
Recognizing a plateau
Weight loss on semaglutide does not proceed in a straight line. Plateaus are normal and expected, occurring when the body adapts to a given dose level. A plateau does not necessarily mean your compounded product is defective. It often means a dose increase is appropriate, or that dietary and activity adjustments are needed. Our guide on semaglutide plateaus covers specific strategies for breaking through stalls.
Nutrition on GLP-1 therapy
Reduced appetite means every calorie counts more. With lower food intake, nutritional density becomes critical. Focus on high-protein foods and supplements to prevent muscle loss during weight reduction. Protein intake should target at least 0.7 to 1.0 grams per pound of lean body mass. The semaglutide diet plan provides meal frameworks designed specifically for GLP-1 therapy, while our guide on foods to eat while on semaglutide offers specific food recommendations.
When to consider switching
If you are not responding to compounded semaglutide after adequate titration (reaching at least 1.0mg weekly for eight or more weeks), consider whether the issue is the product, the dose, or other factors. Possible reasons for poor response include suboptimal product quality, insufficient dose, medication interactions, underlying metabolic conditions, or simply being a non-responder to semaglutide.
Options for switching include trying a different compounder, switching to branded semaglutide if accessible, or exploring alternative GLP-1 medications like tirzepatide. The semaglutide to tirzepatide conversion chart provides dosing equivalencies for switching between these two medications. Some researchers also explore combining GLP-1 therapy with other weight loss peptide stacks to enhance results, though this should only be done under medical supervision.
Alternatives to BPI Labs compounded semaglutide
If the information in this guide raises concerns about BPI Labs, several alternatives exist for accessing semaglutide therapy. Each comes with its own set of advantages and limitations.
Branded semaglutide (Ozempic/Wegovy)
The FDA-approved versions offer the highest quality assurance, backed by extensive clinical trials and ongoing post-market surveillance. The primary barrier is cost. Without insurance coverage, branded semaglutide can exceed $1,000 per month. With insurance, copays vary widely. This remains the gold standard for safety and efficacy verification, and any researcher who can access branded products through insurance or manufacturer programs should strongly consider that option.
Other compounding pharmacies
Numerous 503A and 503B pharmacies compound semaglutide with varying degrees of quality and transparency. Empower Pharmacy, Olympia Pharmacy, and Direct Meds are among the more established names. Apply the evaluation framework discussed earlier in this guide to any compounding pharmacy you consider.
Oral semaglutide options
For those who prefer to avoid injections entirely, oral semaglutide formulations exist, both branded (Rybelsus) and compounded (sublingual drops). The bioavailability of oral semaglutide is lower than injectable, meaning higher oral doses are needed to achieve similar effects. Our guide on sublingual semaglutide covers this option in detail.
Alternative GLP-1 medications
Semaglutide is not the only GLP-1 option. Tirzepatide is a dual GIP/GLP-1 receptor agonist that has shown even greater weight loss in clinical trials. Retatrutide, a triple agonist targeting GIP, GLP-1, and glucagon receptors, is in clinical development and has shown remarkable results in early trials. These alternatives each have their own availability challenges and compounding considerations, but understanding the full landscape helps researchers make informed decisions about which GLP-1 approach best fits their needs.
For those exploring peptide-based weight loss strategies more broadly, the options extend well beyond GLP-1 medications. Our comprehensive guide to the best peptides for weight loss covers the full spectrum of available compounds and their evidence base.
The future of BPI Labs and compounded GLP-1 medications
The compounded GLP-1 market is at a crossroads, and BPI Labs position within it depends on several unresolved regulatory and legal questions.
Regulatory trajectory
With semaglutide off the shortage list, the legal basis for 503B bulk compounding has narrowed significantly. BPI Labs dual status as both an FDA manufacturer and 503B facility may provide some regulatory flexibility that pure compounders do not have. However, the specifics of what BPI Labs can legally produce going forward remain subject to ongoing litigation and FDA enforcement decisions.
The Outsourcing Facilities Association lawsuit challenging the shortage resolution could reinstate compounding authority if successful. Alternatively, the court could affirm the FDA position, further restricting compounding operations. Either outcome will directly impact BPI Labs ability to continue producing compounded semaglutide at scale.
Market evolution
Several factors are reshaping the compounded GLP-1 landscape. Novo Nordisk is expanding manufacturing capacity to meet demand for branded semaglutide. New GLP-1 and multi-agonist medications are entering the market, potentially reducing supply pressure on existing drugs. Patent challenges to tirzepatide could open new compounding opportunities even as semaglutide compounding faces restrictions.
For researchers, the practical implication is uncertainty. A supply source that exists today may not exist in six months. Building knowledge about multiple sourcing options, understanding the legal landscape for peptides and GLP-1 medications, and maintaining relationships with multiple providers creates resilience against supply disruptions.
Quality improvement potential
FDA scrutiny of the compounding industry has intensified dramatically since GLP-1 medications became a mass-market product. This increased oversight, while disruptive to compounders, ultimately benefits consumers by raising quality standards across the industry. BPI Labs, like all compounders, faces pressure to demonstrate consistent quality and compliance.
The question is whether that pressure produces genuine improvement or simply better documentation of existing practices. Researchers should monitor FDA inspection results, enforcement actions, and company communications over time to assess whether BPI Labs is on an upward quality trajectory. SeekPeptides continues to track these developments and will update this guide as new information becomes available.
Making an informed decision about BPI Labs semaglutide
After examining BPI Labs from every angle, including products, manufacturing, corporate history, FDA record, and the regulatory landscape, what should an informed researcher conclude?
The picture is genuinely mixed.
On the positive side, BPI Labs operates robotic isolator technology that represents a legitimate manufacturing advantage for sterile products. The dual FDA manufacturer and 503B registration provides a regulatory framework that many competitors lack. The company has not received Warning Letters, which suggests the FDA considers their corrective actions to previous findings adequate.
On the concerning side, the Belcher Pharmaceuticals ownership brings a corporate history that includes fraud convictions related to pharmaceutical distribution. FDA inspections have identified violations including improper equipment use, inadequate supplier vetting, and labeling issues. The parent company separately faced findings of inadequate impurity testing. These are not trivial concerns for a company producing injectable medications.
The reality is that no compounding pharmacy operates without some level of regulatory finding. The industry is under unprecedented scrutiny, and even well-regarded facilities face challenges. What distinguishes a trustworthy compounder from a risky one is transparency, responsiveness to findings, and a demonstrated commitment to continuous improvement.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. The membership includes access to vendor evaluations, dosing calculators, cost analysis tools, and expert guidance for making informed decisions about every aspect of peptide research.
BPI Labs semaglutide and the broader peptide research landscape
Compounded semaglutide from BPI Labs exists within a much larger ecosystem of peptide therapeutics. Researchers who start with GLP-1 medications often discover an entire world of peptide compounds that serve different purposes, from healing and recovery to cognitive enhancement and longevity.
Understanding how GLP-1 medications relate to the broader peptide landscape helps contextualize your research. Semaglutide and tirzepatide belong to the incretin mimetic category, but peptides also include healing peptides like BPC-157, recovery peptides like TB-500, skin and anti-aging compounds like GHK-Cu, and longevity peptides. Each category has different sourcing challenges, quality standards, and regulatory considerations.
The same principles that apply to evaluating BPI Labs, specifically checking FDA registration, reviewing inspection history, verifying product quality through testing, and understanding the regulatory framework, apply equally to sourcing any peptide product. Researchers who develop strong vendor evaluation skills for GLP-1 medications carry those skills into every other peptide category they explore.
For those just beginning their peptide research journey, our getting started with peptides guide provides a comprehensive foundation. For experienced researchers considering expanding beyond GLP-1 medications, the peptide stacking guide covers how to safely combine multiple compounds for synergistic effects. And for understanding the legal framework that governs peptide access in the United States, our peptide legality guide breaks down federal and state regulations.
SeekPeptides members gain access to detailed vendor evaluations, protocol libraries, and a community of experienced researchers who share firsthand knowledge about product quality across dozens of peptide categories. When you encounter questions that this guide does not answer, the membership community often provides the most current and practical insights available anywhere.
Frequently asked questions
Is BPI Labs semaglutide FDA-approved?
No. BPI Labs semaglutide is a compounded medication, not an FDA-approved product. While BPI Labs is an FDA-registered facility, the compounded semaglutide itself has not been evaluated by the FDA for safety, effectiveness, or quality. FDA-approved semaglutide is sold under the brand names Ozempic (for diabetes) and Wegovy (for weight management). Understanding this distinction is essential for making informed decisions about your peptide research.
How much does BPI Labs semaglutide cost?
BPI Labs does not sell directly to consumers. Pricing depends on the healthcare provider or telehealth platform distributing the product. Through the Hims partnership, compounded semaglutide from BPI Labs has been available for approximately $199 per month. Other providers may charge different amounts. Compare this against the full cost landscape for peptide therapy before making a decision, and use the peptide cost calculator to evaluate long-term expenses.
Can I still get compounded semaglutide from BPI Labs?
The availability of compounded semaglutide from BPI Labs depends on the current regulatory environment. Since the FDA removed semaglutide from the shortage list in February 2025, the legal basis for 503B compounding has been restricted. However, ongoing litigation and different legal mechanisms may still allow some compounding operations. Check with your provider for the most current availability and consult peptide regulation updates for the latest developments.
Is BPI Labs semaglutide safe?
No compounded medication carries the same safety assurance as an FDA-approved product. BPI Labs uses robotic filling systems that reduce contamination risk, which is a positive factor. However, FDA inspections have identified quality control concerns, and the parent company has a documented history of regulatory issues. The FDA has reported 352 adverse events related to compounded GLP-1 medications overall. For comprehensive safety information, review our peptide safety guide.
What concentration is BPI Labs semaglutide?
The most documented BPI Labs semaglutide formulation is 12.5mg/2.5ml, providing a concentration of 5mg/ml. This is a standard concentration for compounded semaglutide. At this concentration, 0.05ml (5 units) delivers 0.25mg, and 0.1ml (10 units) delivers 0.5mg. Always verify the concentration on your specific vial and use a semaglutide dosage calculator to confirm your dose calculations.
How does BPI Labs compare to Empower Pharmacy for semaglutide?
Both are significant players in the compounded GLP-1 market. Empower Pharmacy has been more publicly transparent about formulations and operates from Houston, Texas. BPI Labs emphasizes its robotic manufacturing technology and dual FDA registration. The key differentiator is that BPI Labs parent company (Belcher Pharmaceuticals) carries a history of fraud convictions and FDA violations that Empower does not. Both have received FDA inspection findings. Read our full Empower Pharmacy semaglutide guide for detailed comparison.
Should I switch from BPI Labs to branded semaglutide?
If you can access and afford branded semaglutide (Ozempic or Wegovy), it offers the highest quality assurance backed by FDA approval and clinical trials. Switching between GLP-1 formulations requires careful dose adjustment. Consult with your healthcare provider about the transition, as abrupt changes in formulation or dosing can affect tolerability and efficacy. The semaglutide dosage chart in ml can help you understand equivalent dosing across different formulations.
Does BPI Labs test for semaglutide purity?
BPI Labs states that it verifies the identity and form of its semaglutide API through testing. However, the specific testing protocols, frequency of testing, and willingness to share certificates of analysis with end users vary. For the highest confidence in product quality, request a certificate of analysis for your specific lot and consider independent third-party testing to verify potency and purity.
How do I store BPI Labs semaglutide properly?
Store BPI Labs semaglutide vials refrigerated between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). Do not freeze. Protect from light. Return the vial to the refrigerator promptly after drawing your dose. If a vial has been left at room temperature for an extended period, check with your provider about whether it remains safe to use. Our comprehensive peptide storage guide covers best practices for maintaining potency across all injectable peptide products.
Can I use BPI Labs semaglutide with other peptides?
Combining semaglutide with other peptides is a decision that should involve medical guidance. Some researchers use GLP-1 medications alongside healing peptides like BPC-157 and TB-500 for concurrent weight management and recovery goals. Others explore peptides specifically recommended alongside GLP-1 therapy. However, combining multiple injectable compounds increases complexity and requires careful attention to timing, injection sites, and potential interactions. Consult our peptide stack calculator for compatibility information.
What is the difference between a 503A and 503B pharmacy?
A 503A pharmacy compounds medications based on individual patient prescriptions, similar to a traditional pharmacy. A 503B outsourcing facility can produce medications in bulk without individual prescriptions, operating more like a small manufacturer. BPI Labs operates as a 503B facility and also holds FDA drug manufacturer registration. 503B facilities face more stringent requirements including cGMP compliance, adverse event reporting, and regular FDA inspections. Understanding these distinctions helps researchers evaluate the options available for obtaining peptide therapy.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your vendors stay transparent, your formulations stay pure, and your research stay informed.