Mar 3, 2026

Three months into her tirzepatide protocol, Sarah noticed something new. Not the nausea she had expected. Not the appetite suppression everyone talks about. A sharp, stabbing pain under her right ribcage that came after dinner and refused to leave. Her doctor ordered an ultrasound the next morning. Gallstones. Two of them.
Sarah is not alone. And her experience raises a question that thousands of tirzepatide users need answered: does this medication actually cause gallbladder problems, or is the connection more complicated than the headlines suggest?
The answer, like most things in medicine, lives in the space between a simple yes and a simple no. Clinical trials show a measurable increase in gallbladder events among tirzepatide users. A systematic review of 9,871 participants found a relative risk of 1.97 for composite gallbladder and biliary disease. But that number needs context. It needs mechanisms. It needs practical guidance that most sources skip entirely.
This guide breaks down every piece of clinical evidence connecting tirzepatide to gallbladder disease. You will find the actual trial data, the biological mechanisms at play, the warning signs that demand immediate attention, and the prevention strategies that can dramatically reduce your risk. Whether you are considering tirzepatide, already using it, or managing gallbladder concerns alongside your protocol, every answer you need is here.
How tirzepatide affects your gallbladder
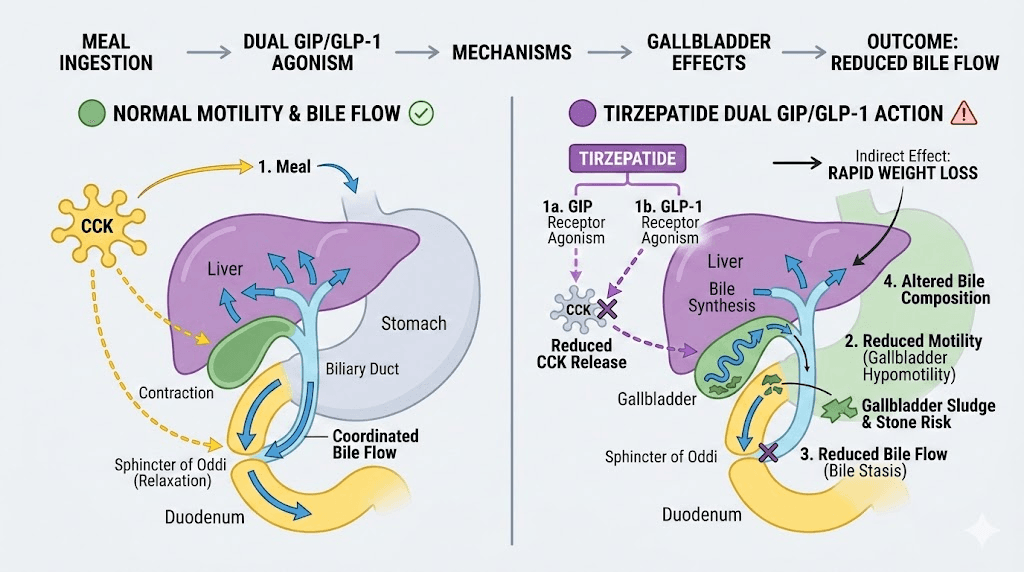
Understanding the connection between tirzepatide and gallbladder problems requires looking at three separate mechanisms. Each one contributes to increased risk. Together, they explain why some users develop issues while most do not.
The GLP-1 receptor pathway and gallbladder motility
Tirzepatide is a dual GIP/GLP-1 receptor agonist. That dual action is what makes it so effective for weight loss and blood sugar control. But GLP-1 receptor activation does something else entirely to your gallbladder.
GLP-1 has been shown to impair gallbladder motility and contractility by inhibiting cholecystokinin secretion. Cholecystokinin is the hormone that tells your gallbladder to contract and release bile after you eat. When GLP-1 receptors are activated, this signal gets dampened. Your gallbladder contracts less forcefully. Less frequently. Bile sits longer than it should.
Think of it like a pump that slows down. The fluid inside starts to stagnate. Particles that would normally get flushed out begin to settle and clump together. That is exactly what happens with bile when gallbladder motility decreases.
GIP, the other receptor tirzepatide activates, has also been reported to play a role in gallbladder relaxation. This means tirzepatide hits the gallbladder through two pathways simultaneously, something that pure GLP-1 agonists like semaglutide do not do to the same degree.
Rapid weight loss and bile composition changes
This is the bigger factor. And it has nothing to do with the medication itself.
When you lose weight rapidly, your body mobilizes cholesterol from adipose tissue. That cholesterol enters the bloodstream and eventually gets processed by the liver into bile. The result is bile that becomes supersaturated with cholesterol, meaning it contains more cholesterol than the bile salts can keep dissolved.
Cholesterol-supersaturated bile is the primary ingredient for gallstone formation. The excess cholesterol precipitates out of solution, forms crystals, and those crystals aggregate into stones over time. This process happens with any form of rapid weight loss, whether from semaglutide, bariatric surgery, very-low-calorie diets, or tirzepatide.
The National Institute of Diabetes and Digestive and Kidney Diseases states that losing more than 3 pounds per week significantly increases the risk of developing gallstones. Tirzepatide, at higher doses, can produce weight loss of 20.9% of body weight. For a 220-pound person, that could mean losing over 45 pounds. If that loss happens too quickly, the gallbladder pays the price.
Here is what makes this particularly relevant for tirzepatide dosing. The standard titration schedule increases the dose every four weeks. Each dose increase can accelerate weight loss. The fastest weight loss typically occurs between weeks 8 and 20 of treatment, precisely when the dose is climbing from 5mg to 10mg or higher. This is the window of highest gallbladder risk.
Reduced food intake and gallbladder stimulation
Your gallbladder needs regular stimulation to function properly. Every time you eat, especially foods containing fat, your body releases cholecystokinin. This hormone triggers gallbladder contraction, which empties bile into the small intestine.
Tirzepatide profoundly suppresses appetite. Many users report eating significantly less, sometimes skipping meals entirely. Some struggle to consume enough calories to meet basic nutritional needs.
Less food means fewer gallbladder contractions. Fewer contractions mean bile stagnation. Stagnant bile means a higher likelihood of crystal formation and eventual stone development.
This creates a triple threat. The medication reduces gallbladder motility directly. Rapid weight loss changes bile composition. And reduced food intake removes the regular stimulation your gallbladder depends on to stay healthy.
What the clinical trial data actually shows
Numbers matter here. The difference between panic and appropriate caution lives in the actual data. Let us look at what the trials found.
SURPASS trials (type 2 diabetes)
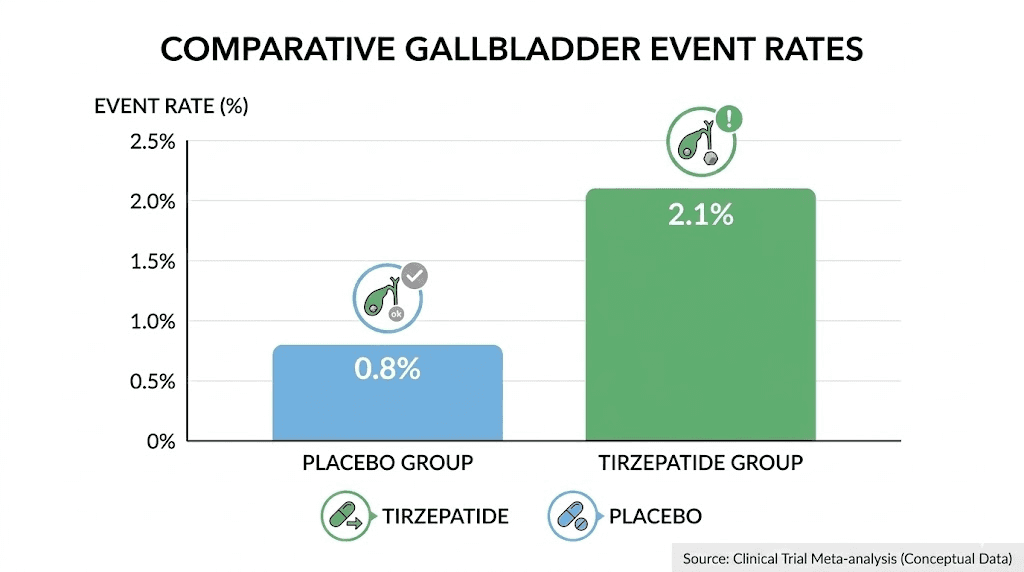
The SURPASS clinical trial program studied tirzepatide in patients with type 2 diabetes across multiple trials: SURPASS-1, SURPASS-3, SURPASS-4, SURPASS-5, and SURPASS J-mono. In the pooled analysis of placebo-controlled trials, acute gallbladder disease including cholelithiasis, biliary colic, and cholecystectomy was reported by 0.6% of tirzepatide-treated patients compared to 0% of placebo-treated patients.
That 0.6% number is important. It means that for every 167 patients taking tirzepatide in the diabetes trials, roughly one experienced an acute gallbladder event. Low. But not zero.
SURMOUNT trials (obesity)
The SURMOUNT trials studied tirzepatide specifically for weight management in patients without diabetes. SURMOUNT-1 and SURMOUNT-2 showed cholelithiasis in 1.1% of tirzepatide-treated patients versus 1.0% on placebo. The difference here is minimal.
However, cholecystitis, which is gallbladder inflammation and a more serious condition, was reported in 0.7% of tirzepatide-treated patients compared to 0.2% on placebo. That is a more meaningful difference, representing roughly a 3.5-fold increase in risk for the inflammatory condition specifically.
Meta-analysis findings
A systematic review and meta-analysis published in Frontiers in Endocrinology examined nine trials with 9,871 participants. The results showed a statistically significant association between tirzepatide and the composite outcome of gallbladder or biliary diseases, with a relative risk of 1.97 and a 95% confidence interval of 1.14 to 3.42.
A more recent meta-analysis of 12 studies involving 12,351 patients found tirzepatide was associated with gallbladder and biliary diseases at an overall relative risk of 1.52, with cholelithiasis specifically showing a relative risk of 1.67.
What does a relative risk of 1.52 to 1.97 mean in practical terms? If the baseline risk of gallbladder disease is roughly 1% per year in the general population, tirzepatide might increase that to approximately 1.5% to 2%. An additional 1 in 100 to 1 in 200 users might experience a gallbladder event that would not have happened otherwise.
Dose-response relationship
Here is where the data gets interesting. Subgroup analysis based on tirzepatide dose found no consistent dose-response relationship between different doses and the risk of gallbladder disease. The 10mg dose showed a relative risk of 1.91, while the 5mg and 15mg doses did not show statistically significant associations.
One analysis found that only the 5mg dose was significantly associated with cholelithiasis risk, with a relative risk of 2.19. This counterintuitive finding, where a lower dose shows higher risk than a higher dose, suggests that the relationship between tirzepatide and gallbladder problems may not be purely dose-dependent.
What likely matters more than the dose itself is the rate of weight loss. A person losing weight rapidly on 5mg might be at higher risk than someone losing weight gradually on 15mg. The pace of weight loss, not the medication dose, appears to be the critical variable.
How tirzepatide compares to other GLP-1 medications for gallbladder risk
Tirzepatide is not the only GLP-1 medication associated with gallbladder problems. In fact, this is a class-wide effect.
A landmark JAMA Internal Medicine meta-analysis of 76 randomized clinical trials found that GLP-1 receptor agonists as a class were associated with increased risks of cholelithiasis (relative risk 1.27), cholecystitis (relative risk 1.36), and biliary disease broadly (relative risk 1.55). The absolute risk difference was an additional 27 events per 10,000 patients per year.
Here is how tirzepatide compares to semaglutide specifically:
Factor | Tirzepatide | Semaglutide |
|---|---|---|
Cholelithiasis rate (obesity trials) | 1.1-2.5% | 1.6% |
Cholecystitis rate | 0.7% | 0.4% |
Maximum weight loss | Up to 20.9% | Up to 15-17% |
Receptor targets | GIP + GLP-1 (dual) | GLP-1 only |
Gallbladder motility impact | Dual pathway inhibition | Single pathway inhibition |
The higher cholecystitis rate with tirzepatide compared to semaglutide may relate to its greater weight loss efficacy. More weight lost means more cholesterol mobilized, more bile composition changes, and more gallbladder stress. The dual GIP/GLP-1 mechanism may also contribute by affecting gallbladder motility through two pathways rather than one.
Long-acting GLP-1 receptor agonists like semaglutide and tirzepatide appear to exert a more pronounced inhibitory effect on gallbladder contraction compared to short-acting agents like exenatide. The extended half-life means the gallbladder experiences continuous motility suppression rather than intermittent effects.
For context, newer agents like mazdutide and survodutide are being studied for similar gallbladder effects, though data remains limited.
Who is most at risk for tirzepatide-related gallbladder problems
Not everyone on tirzepatide faces the same gallbladder risk. Several factors significantly increase vulnerability.
Established risk factors for gallstone disease
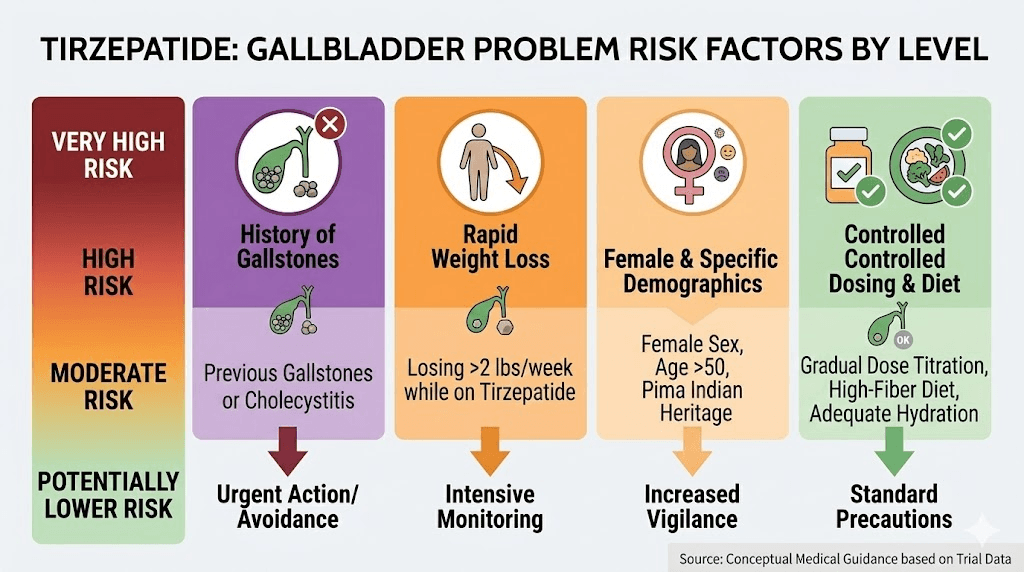
The traditional risk factors for gallstones apply even more strongly when tirzepatide enters the picture:
Female sex. Women develop gallstones two to three times more frequently than men. Estrogen increases cholesterol secretion into bile, and progesterone slows gallbladder emptying. These hormonal effects compound the motility changes caused by tirzepatide.
Age over 40. Gallstone incidence rises steadily with age. The gallbladder becomes less efficient at contracting and emptying. Adding tirzepatide to an already sluggish gallbladder creates more stagnation.
Obesity and metabolic syndrome. Paradoxically, the very condition tirzepatide treats is itself a major risk factor for gallstones. Obesity increases biliary cholesterol secretion and creates a larger pool of cholesterol available for mobilization during weight loss. This is why people with higher starting BMIs face greater gallbladder risk during GLP-1 treatment.
Family history. Genetics account for roughly 25-30% of gallstone risk. If your parents or siblings have had gallstones, your risk is significantly elevated regardless of medication use.
Ethnicity. Hispanic and Native American populations have higher baseline gallstone prevalence due to genetic factors affecting bile composition.
Treatment-specific risk factors
Beyond baseline gallstone risk, certain treatment patterns create additional vulnerability:
Rapid dose escalation. Advancing through the tirzepatide starting dose schedule faster than recommended can accelerate weight loss beyond safe rates. Each dose increase should occur at minimum four-week intervals, allowing the body to adjust.
Very low caloric intake. Users who eat very little, whether from severe appetite suppression or intentional restriction, deprive their gallbladder of the stimulation it needs. If you are eating fewer than 1000 calories per day for extended periods, your gallbladder risk increases substantially.
High-carbohydrate, low-fat diets. Fat is the primary trigger for gallbladder contraction. Diets extremely low in fat reduce gallbladder emptying frequency. While dietary choices on tirzepatide matter for many reasons, including adequate fat intake specifically protects gallbladder function.
Concurrent fasting or caloric restriction. Combining tirzepatide with intermittent fasting or very-low-calorie diets multiplies the risk. The medication already reduces appetite significantly. Adding intentional restriction on top creates prolonged periods without gallbladder stimulation.
Previous gallstone episodes. If you have had gallstones before, even if they resolved, your risk of recurrence is substantially higher. The gallbladder has already demonstrated a tendency toward stone formation, and tirzepatide can reactivate that tendency.
Recognizing gallbladder symptoms while on tirzepatide
One of the biggest challenges with gallbladder problems during tirzepatide use is distinguishing them from the medication normal side effects. Nausea, abdominal discomfort, and digestive upset are common with tirzepatide regardless of gallbladder involvement. Knowing the differences could save your gallbladder, or your life.
Common tirzepatide side effects versus gallbladder warning signs
Symptom | Normal tirzepatide side effect | Possible gallbladder problem |
|---|---|---|
Nausea | Generalized, mild to moderate, improves with dose adjustment | Intense, associated with right-sided pain, worse after fatty meals |
Abdominal pain | General stomach discomfort, cramping, usually central or diffuse | Sharp or stabbing pain specifically under right ribcage, radiating to right shoulder or between shoulder blades |
Vomiting | Occasional, related to overeating or rapid eating | Persistent, cannot keep food down, associated with severe pain |
Bloating | General bloating, improves with smaller meals | Localized right upper abdomen, tender to touch |
Duration | Comes and goes, typically improves over weeks | Episodes lasting 30 minutes to several hours, may worsen over time |
The five warning signs that demand medical attention
1. Right upper quadrant pain. This is the hallmark. Pain concentrated under your right ribcage, often described as sharp, stabbing, or squeezing. It may radiate to your right shoulder blade or between your shoulder blades. This pattern is distinctly different from the generalized discomfort tirzepatide typically causes.
2. Pain triggered by fatty meals. Gallbladder attacks classically follow meals high in fat. The fat triggers cholecystokinin release, which causes the gallbladder to contract. If stones are present, that contraction pushes them against the cystic duct opening, causing sudden intense pain. If your abdominal pain consistently appears 30-60 minutes after eating fatty foods, suspect gallbladder involvement.
3. Fever with abdominal pain. This combination suggests cholecystitis, which is inflammation of the gallbladder. Tirzepatide side effects do not cause fever. If you develop a temperature above 100.4 degrees F alongside right-sided abdominal pain, this is a medical emergency. Do not wait.
4. Jaundice. Yellowing of the skin or whites of the eyes indicates bile duct obstruction. A gallstone has moved from the gallbladder into the common bile duct, blocking bile flow. This is serious and requires immediate emergency care.
5. Clay-colored stools or dark urine. These indicate bile is not reaching the intestine properly. Normal stool gets its brown color from bile pigments. If bile flow is blocked, stools become pale or clay-colored while urine becomes very dark. This pattern points to biliary obstruction.
The Murphy sign
Healthcare providers use a specific physical examination technique called the Murphy sign to assess for gallbladder inflammation. During a deep breath, the examiner presses on the right upper quadrant. If pain causes you to stop breathing in mid-inspiration, the Murphy sign is positive. This test is 65% sensitive and 87% specific for cholecystitis, making it a useful but imperfect screening tool.
You can approximate this at home. Press firmly under your right ribcage and take a deep breath. If the pain intensifies sharply and stops your breath, contact your healthcare provider promptly.
Prevention strategies that actually work
The good news is that most tirzepatide-related gallbladder problems are preventable. These strategies target each of the three mechanisms driving risk.
Control the pace of weight loss
This is the single most impactful thing you can do.
The NIDDK recommends keeping weight loss below 3 pounds per week to minimize gallstone risk. For most people on tirzepatide, this means being thoughtful about dose escalation rather than rushing to the highest tolerated dose.
Practical approaches include staying at a given dose for longer than the minimum four weeks if weight is dropping rapidly. If you are losing weight faster than 3 pounds per week consistently, discuss delaying your next dose increase with your prescriber. Some clinicians recommend extending the interval between dose increases to 6-8 weeks during periods of rapid loss.
Track your weekly weight loss. A simple dosage tracking approach helps you spot when loss accelerates beyond the safe zone. The fastest weight loss typically happens between weeks 8 and 20 of treatment, so pay extra attention during this window.
Maintain adequate fat intake
This sounds counterintuitive when you are trying to lose weight. But your gallbladder needs fat to function.
Dietary fat triggers cholecystokinin release, which causes gallbladder contraction. Regular contractions keep bile flowing and prevent stagnation. Aim for at least 20-30 grams of healthy fats daily, distributed across meals.
Good sources include avocados, olive oil, nuts, fatty fish, and eggs. These foods also tend to be well tolerated on tirzepatide and provide essential nutrients that support overall health during weight loss.
If you are following a tirzepatide diet plan, ensure fat is not eliminated. Some users, dealing with nausea, gravitate toward bland, low-fat foods exclusively. This is understandable but increases gallbladder risk. Even small amounts of fat at each meal help.
Eat regular meals
Skipping meals is one of the most common behaviors on tirzepatide, and one of the most damaging to gallbladder health.
Each meal triggers a gallbladder contraction cycle. Going 12 or more hours without eating creates extended bile stagnation. Try to eat at least three small meals daily, even if appetite is minimal.
This is especially important for users who combine tirzepatide with intermittent fasting. While intermittent fasting can have benefits, the combination with tirzepatide creates prolonged fasting windows that stress the gallbladder. If you practice intermittent fasting, consider shortening your fasting window while on the medication.
Breakfast is particularly important. After an overnight fast, your morning meal is the first gallbladder stimulation of the day. Include some fat in that meal to get bile flowing.
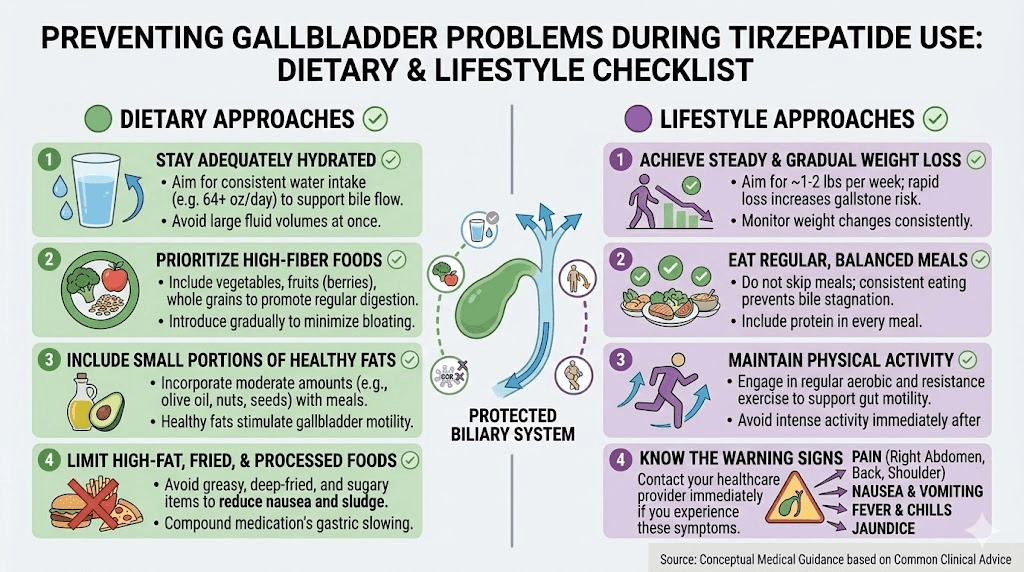
Stay hydrated
Adequate hydration supports bile flow and reduces bile concentration. Dehydration makes bile thicker and more prone to crystal formation. Aim for at least 64 ounces of water daily, more if you are active or in a warm climate.
Some users find that tirzepatide-related nausea makes drinking water difficult. Try small, frequent sips rather than large volumes. Adding a slice of lemon or ginger can help with palatability.
The ursodiol question
Ursodeoxycholic acid, sold as ursodiol, is an FDA-approved medication for preventing gallstone formation during rapid weight loss. It works by reducing bile cholesterol saturation and preventing cholesterol crystal nucleation.
The data supporting ursodiol is strong. In bariatric surgery patients losing weight rapidly, gallstones developed in 28% of placebo patients compared to only 3% receiving 600mg daily of ursodiol. A meta-analysis found ursodiol reduced the risk of ultrasound-verified gallstones by 67% compared to control groups.
Currently, there are no specific guidelines recommending ursodiol prophylaxis for GLP-1 medication users. However, given the established gallbladder risk, some clinicians prescribe it for patients with multiple risk factors, particularly those losing weight rapidly on higher doses.
If you have a history of gallstones, a strong family history, or are experiencing rapid weight loss on tirzepatide, ask your healthcare provider about ursodiol prophylaxis. The typical preventive dose is 300-600mg daily.
Consider moderate exercise
Regular physical activity independently reduces gallstone risk by improving gallbladder motility and reducing biliary cholesterol secretion. The evidence suggests that 30 minutes of moderate exercise most days of the week provides meaningful protection.
Exercise also helps moderate the pace of weight loss, converting some of the deficit into lean mass preservation rather than pure fat loss. This is beneficial for gallbladder health and overall body composition during GLP-1 treatment.
What to do if you develop gallbladder symptoms
Despite best prevention efforts, some users will develop gallbladder symptoms. The response strategy depends on severity.
Mild symptoms: monitoring approach
If you experience occasional right-sided discomfort after fatty meals that resolves within 30 minutes and does not involve fever or jaundice, this may represent biliary colic from small, asymptomatic stones. Schedule an appointment with your healthcare provider for an ultrasound evaluation. Continue tirzepatide but keep a symptom diary noting what you ate, when pain occurred, how long it lasted, and its severity.
Moderate symptoms: medical evaluation needed
Pain lasting more than an hour, recurring episodes, or pain that wakes you from sleep warrants prompt medical evaluation. Your provider will likely order an abdominal ultrasound, which is 95% sensitive for detecting gallstones.
At this stage, your prescriber may recommend temporarily holding tirzepatide until the gallbladder situation is clarified. This is not necessarily a permanent stop. It is a pause to assess the situation safely.
If asymptomatic gallstones are found on ultrasound, many clinicians will recommend continuing tirzepatide with closer monitoring. Asymptomatic stones often remain silent and may not require intervention.
Severe symptoms: emergency care
Seek emergency medical care immediately for any of these:
Severe, unrelenting right upper abdominal pain lasting more than 4-6 hours
Fever above 100.4 degrees F with abdominal pain
Jaundice (yellowing of skin or eyes)
Clay-colored stools with dark urine
Severe vomiting with inability to keep fluids down
Pain so intense you cannot find a comfortable position
These symptoms may indicate acute cholecystitis, choledocholithiasis (stone in the common bile duct), or cholangitis (bile duct infection). All three require urgent evaluation and potentially surgical intervention.
Cholecystectomy: when surgery becomes necessary
If gallstones cause recurrent symptoms or complications, cholecystectomy (gallbladder removal) may be recommended. This is one of the most common surgeries performed, with over 700,000 procedures annually in the United States alone.
Laparoscopic cholecystectomy is minimally invasive, typically involves same-day discharge, and has a recovery period of 1-2 weeks. Most patients return to normal activity within 7-10 days.
The important question many users ask: can you continue tirzepatide after gallbladder removal?
Taking tirzepatide after gallbladder removal
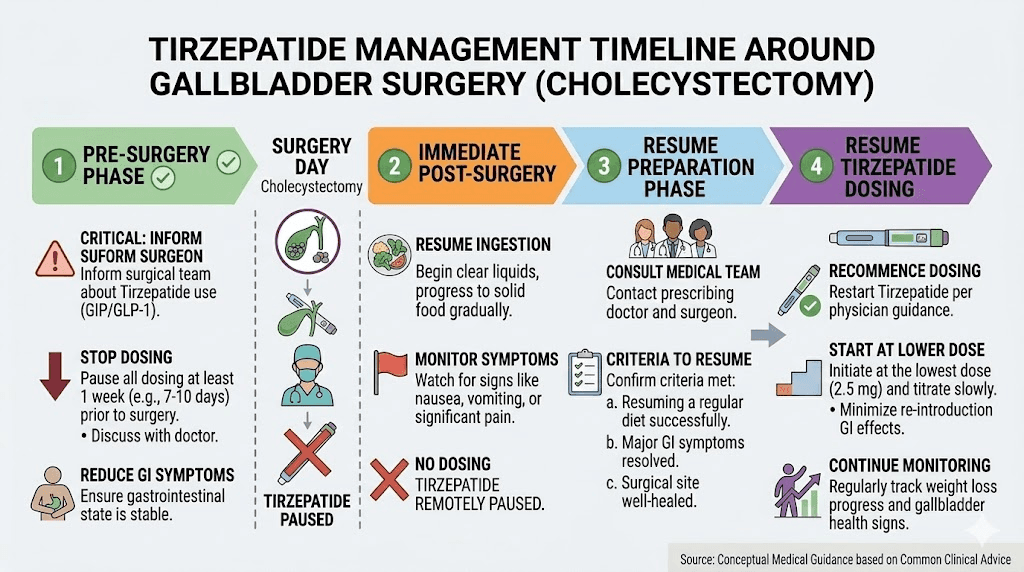
Yes. Tirzepatide can be safely used after cholecystectomy.
The FDA prescribing information does not list gallbladder removal as a contraindication. Eli Lilly, the manufacturer of Mounjaro and Zepbound, confirms that patients with gallbladder disease or prior gallbladder removal can take tirzepatide. The medication is administered subcutaneously and undergoes proteolytic degradation, meaning it does not require biliary processing for absorption or metabolism.
Once the gallbladder is removed, the risk of gallstones and gallbladder inflammation is eliminated entirely. You cannot form gallstones in an organ you no longer have. However, there are some practical considerations:
Overlapping digestive symptoms. Both tirzepatide and life without a gallbladder can cause diarrhea, bloating, and nausea. After cholecystectomy, bile flows continuously from the liver into the intestine rather than being stored and released in concentrated bursts. This can cause loose stools, particularly after fatty meals. Adding tirzepatide on top may intensify these symptoms initially.
Fat absorption changes. Without a gallbladder, fat digestion efficiency decreases somewhat. You may need to adjust your fat intake distribution, eating smaller amounts of fat spread across more meals rather than large amounts at once.
No dose adjustment needed. Tirzepatide dosing does not require modification based on cholecystectomy status. The standard starting protocol of 2.5mg weekly with gradual escalation applies regardless of gallbladder status.
Timing considerations. If you undergo cholecystectomy while on tirzepatide, most clinicians recommend pausing the medication for 1-2 weeks around surgery to allow recovery. The medication can then be resumed at the same dose or stepped down briefly if GI symptoms are significant.
Tirzepatide and existing gallbladder disease
What if you already have gallstones or gallbladder issues before starting tirzepatide? This is a nuanced clinical decision.
Known asymptomatic gallstones
Many people have gallstones that cause no symptoms, often discovered incidentally on imaging performed for other reasons. Current evidence does not contraindicate tirzepatide in patients with asymptomatic gallstones, but the risk of those stones becoming symptomatic may increase.
If you have known asymptomatic gallstones and are considering GLP-1 treatment, discuss prophylactic strategies with your provider. Ursodiol prophylaxis, careful dose titration, and regular symptom monitoring are reasonable precautions.
History of symptomatic gallstones
If you have had previous gallstone episodes that resolved, your risk of recurrence during tirzepatide treatment is elevated. Your provider may recommend a baseline ultrasound before starting treatment and periodic imaging during the first year.
Active gallbladder disease
Starting tirzepatide during active gallbladder symptoms is generally not recommended. Resolve the acute gallbladder issue first. If cholecystectomy is needed, have the surgery and recover before beginning the medication. This eliminates the organ at risk entirely and allows you to use tirzepatide without gallbladder concerns.
The broader gallbladder picture: GLP-1 medications as a class
It would be unfair to single out tirzepatide without acknowledging that gallbladder risk is a feature of the entire GLP-1 medication class.
A FAERS database pharmacovigilance analysis of FDA adverse event reports found significant signals for both cholecystitis and cholelithiasis across multiple GLP-1 receptor agonists. The risk appears highest with long-acting agents that produce greater weight loss.
The JAMA Internal Medicine meta-analysis of 76 randomized trials quantified the class-wide risk:
Cholelithiasis: relative risk 1.27 across all GLP-1 RAs
Cholecystitis: relative risk 1.36
Biliary disease composite: relative risk 1.55
Higher doses carried higher risk than lower doses
Longer treatment duration carried higher risk than shorter duration
For high-risk populations, particularly older women, close monitoring for cholecystitis and cholelithiasis during GLP-1 therapy is recommended. If patients present with symptoms such as upper abdominal pain, nausea, or vomiting, prompt imaging evaluation should be conducted.
This class-wide effect means that switching from tirzepatide to semaglutide or another GLP-1 agonist will not eliminate gallbladder risk. It may reduce it marginally since tirzepatide produces greater weight loss, but the fundamental mechanisms remain.
The weight loss surgery comparison
Putting tirzepatide gallbladder risk in perspective helps. After bariatric surgery, which also produces rapid significant weight loss, gallstone incidence ranges from 22% to 71% depending on the study and procedure type. That is dramatically higher than the 1-3% seen with tirzepatide in clinical trials.
The bariatric surgery gallstone rate is 10 to 70 times higher than what tirzepatide produces. This context matters because it shows that tirzepatide-related gallbladder risk, while real, is relatively modest compared to other weight loss interventions that produce comparable results.
Ursodiol prophylaxis is standard of care after bariatric surgery precisely because the gallstone risk is so high. The fact that it is not yet standard for GLP-1 users reflects the lower, though still elevated, risk level.
Monitoring protocols for gallbladder health
Proactive monitoring can catch gallbladder problems early, when they are easiest to manage.
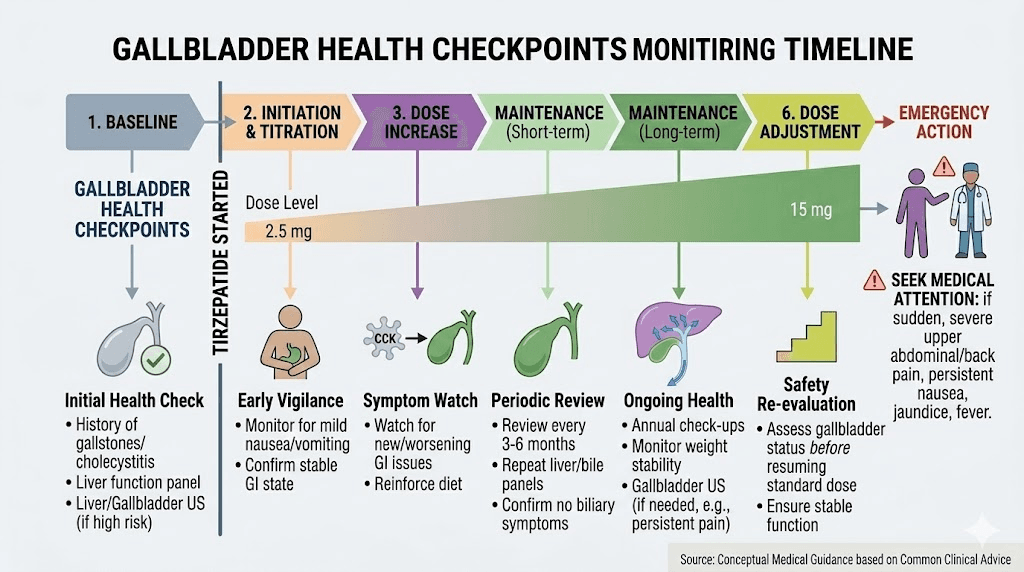
Baseline assessment
Before starting tirzepatide, consider a baseline abdominal ultrasound if you have any of these risk factors:
Female sex plus age over 40
BMI over 40
Family history of gallstones
History of rapid weight cycling
Hispanic or Native American ethnicity
History of biliary symptoms
A baseline ultrasound establishes whether stones are already present before treatment begins. This information helps guide monitoring intensity and prophylactic decisions.
During treatment monitoring
For the first six months of tirzepatide treatment, especially during dose titration when weight loss is typically fastest:
Monthly weight tracking. Monitor weekly weight loss rate. Flag any sustained period of more than 3 pounds per week for discussion with your prescriber. Use a tracking tool to stay organized.
Symptom awareness. Know the five warning signs described earlier. Pay particular attention to right upper quadrant pain, especially after meals.
Dietary logging. Track fat intake to ensure you are getting at least 20-30 grams daily. Dietary planning during tirzepatide treatment serves multiple purposes, but gallbladder protection is an underappreciated one.
Periodic ultrasound. For high-risk patients, some clinicians recommend ultrasound at 3-6 months of treatment to check for new stone formation. This is not standard practice but may be warranted for patients with multiple risk factors.
Long-term monitoring
After the first year, gallbladder risk typically stabilizes as weight loss slows and bile composition adjusts. Continue standard monitoring but recognize that the highest-risk period has passed. Maintenance-phase tirzepatide use at stable weight carries substantially lower gallbladder risk than the active weight loss phase.
Practical protocols for gallbladder protection
Based on all available evidence, here are three practical protocols based on your risk level.
Protocol 1: Standard risk (no additional risk factors)
Who this applies to: Males under 40, no family history, no prior gallstones, BMI under 35
Approach:
Follow standard dose titration schedule
Eat at least 3 meals daily with moderate fat content
Stay hydrated (64+ ounces daily)
Monitor weight loss rate weekly
Know the warning signs
No baseline ultrasound needed unless desired
Protocol 2: Moderate risk (1-2 additional risk factors)
Who this applies to: Females, age 40+, family history, BMI 35-45, or history of weight cycling
Approach:
Consider baseline abdominal ultrasound
Follow standard dose titration but extend intervals if weight loss exceeds 3 lbs/week
Include 25-30g healthy fats daily across meals
Avoid prolonged fasting (do not skip meals)
Stay well hydrated
Discuss ursodiol prophylaxis with your provider
Monitor closely during weeks 8-20
Schedule follow-up ultrasound at 6 months if high concern
Protocol 3: High risk (3+ risk factors or prior gallstones)
Who this applies to: Females over 40 with family history, prior gallstones, BMI over 40, or Native American/Hispanic ethnicity with obesity
Approach:
Baseline abdominal ultrasound before starting tirzepatide
Strongly consider ursodiol 300-600mg daily during active weight loss phase
Extended dose titration (6-8 week intervals rather than 4)
Include 30g+ healthy fats daily
No intermittent fasting during treatment
Regular meals every 4-5 hours during waking hours
Follow-up ultrasound at 3 months and 6 months
Lower threshold for medical evaluation of any abdominal symptoms
Discuss prophylactic cholecystectomy if large asymptomatic stones are found
Supplements and natural approaches for gallbladder support
Several natural approaches may complement the prevention strategies above. While none replace medical management, they can serve as additional protective layers.
Vitamin C. Studies suggest that vitamin C deficiency correlates with increased gallstone risk. Adequate vitamin C intake supports bile acid synthesis and may help prevent cholesterol supersaturation. This is one of several supplements worth considering during tirzepatide treatment.
Coffee. Regular coffee consumption is associated with reduced gallstone risk in epidemiological studies. Caffeine stimulates gallbladder contraction and may improve bile flow. If you tolerate coffee, 2-3 cups daily may provide some protection.
Fiber. Adequate dietary fiber, particularly soluble fiber, supports bile acid metabolism and may reduce biliary cholesterol saturation. Aim for 25-30 grams daily from vegetables, fruits, legumes, and whole grains.
Magnesium. Low magnesium levels are associated with increased gallstone risk in several studies. Magnesium supports smooth muscle function, including gallbladder contractility. Consider a magnesium supplement of 200-400mg daily if dietary intake is insufficient.
Omega-3 fatty acids. Fish oil provides both the fat needed to stimulate gallbladder contraction and omega-3 fatty acids that may favorably influence bile composition. This does double duty for gallbladder protection.
Frequently asked questions
Does tirzepatide directly cause gallstones?
Not directly. Tirzepatide does not place cholesterol into bile or create stones. It increases gallstone risk through three indirect mechanisms: reducing gallbladder motility via GLP-1 and GIP receptor activation, promoting rapid weight loss that changes bile composition, and suppressing appetite in ways that reduce gallbladder stimulation. The gallstones that form are cholesterol stones, the same type that form during any rapid weight loss scenario. Prevention strategies targeting these mechanisms substantially reduce risk.
How common are gallbladder problems with tirzepatide?
In clinical trials, gallstone formation occurred in 1.1-2.5% of tirzepatide users compared to 0-1% on placebo, depending on the study population. Cholecystitis (gallbladder inflammation) occurred in about 0.7% of users versus 0.2% on placebo. For perspective, that means roughly 97-99% of tirzepatide users complete treatment without any gallbladder event.
Should I get my gallbladder checked before starting tirzepatide?
A baseline ultrasound is not required for all patients but is strongly recommended for those with existing risk factors: women over 40, family history of gallstones, BMI over 40, Hispanic or Native American ethnicity, or history of gallbladder symptoms. If you have no risk factors, standard monitoring during treatment is typically sufficient.
Can I take tirzepatide if I have already had my gallbladder removed?
Yes. Cholecystectomy eliminates the risk of gallstones and gallbladder inflammation entirely. Tirzepatide dosing does not need modification after gallbladder removal. You may experience overlapping digestive symptoms (diarrhea, bloating) from both the medication and the absence of a gallbladder, but these typically improve with dietary adjustments and time.
Is tirzepatide worse for the gallbladder than semaglutide?
The data is mixed. Tirzepatide produces greater weight loss than semaglutide, which may translate to higher gallbladder risk through increased cholesterol mobilization. Tirzepatide also affects the gallbladder through two receptor pathways (GIP and GLP-1) compared to semaglutide one (GLP-1 only). However, when comparing similar rates of weight loss, the difference in gallbladder risk between the two medications narrows considerably.
What should I eat to protect my gallbladder on tirzepatide?
Focus on regular meals containing moderate healthy fats. Include avocados, olive oil, nuts, fatty fish, and eggs to stimulate gallbladder contraction. Avoid very low-fat diets and prolonged fasting. Eat at least three meals daily, even small ones. Adequate fiber from vegetables and whole grains supports bile acid metabolism. For detailed guidance, see our foods to avoid on tirzepatide guide, which also covers foods to emphasize.
When during treatment is gallbladder risk highest?
Weeks 8 through 20 of treatment typically carry the highest risk. This is the period when dose titration is active and weight loss is usually most rapid. Once weight loss stabilizes at maintenance levels, gallbladder risk decreases substantially. Extra vigilance during this window, including closer weight monitoring and dietary attention, provides the most protection.
Does microdosing tirzepatide reduce gallbladder risk?
Microdosing tirzepatide produces slower weight loss, which theoretically reduces gallbladder risk by minimizing rapid bile composition changes. However, no studies have specifically examined gallbladder outcomes with microdosing protocols. The principle of slower weight loss equaling lower gallbladder risk is well established across all weight loss interventions, making the logic sound even without direct trial data.
External resources
GLP-1 receptor agonist use and risk of gallbladder and biliary diseases - JAMA Internal Medicine
Gallstones - National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK)
Risk of biliary diseases in patients treated with tirzepatide - Journal of Diabetes Investigation
For researchers serious about understanding and managing their protocols, SeekPeptides offers comprehensive guides, evidence-based protocols, and a community of thousands who have navigated these exact questions. From dosage calculators to dosing charts and supplement guidance, every tool you need to optimize your protocol safely is available in one place.
In case I do not see you, good afternoon, good evening, and good night. May your gallbladder stay quiet, your bile stay flowing, and your protocols stay safe.