Feb 28, 2026

Inside your thyroid gland, a quiet war is being fought. Your own immune system, the very machinery designed to protect you, has turned against the butterfly-shaped gland at the base of your neck. Hashimoto's thyroiditis affects roughly 14 million Americans, making it the most common autoimmune condition in the country. And if you have it, you already know the frustration. The fatigue that sleep does not fix. The weight that will not budge no matter how carefully you eat. The brain fog that makes simple tasks feel impossible.
Then you hear about GLP-1 receptor agonists. Semaglutide. Tirzepatide. The medications generating headlines for dramatic weight loss results. And you wonder: could these work for me? Or would they make my thyroid situation worse?
The answer is more nuanced than any headline suggests. GLP-1 medications interact with your body through pathways that overlap with thyroid function, immune regulation, gut health, and metabolic processing in ways that matter deeply when autoimmune thyroid disease is part of the equation. Understanding those interactions is not optional. It is essential. Because the difference between a successful protocol and a dangerous one often comes down to details that most guides completely skip.
This guide covers everything Hashimoto's patients need to understand about GLP-1 therapy, from the research on safety and thyroid function to practical monitoring schedules, supplement considerations, medication timing, and the specific scenarios where these medications could genuinely help versus situations where caution is warranted. SeekPeptides has compiled the research, the clinical data, and the practical guidance that thyroid patients need to make informed decisions.
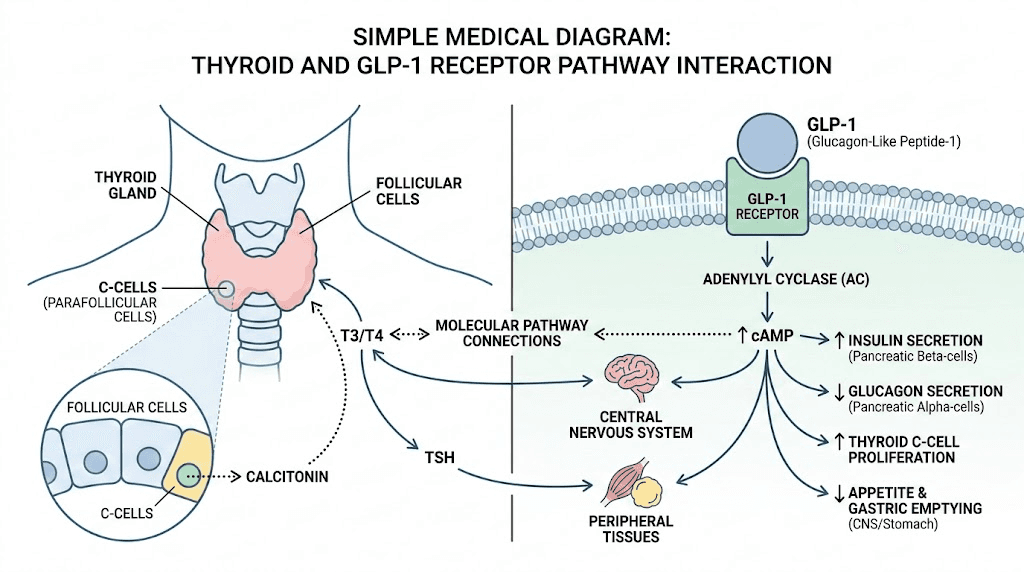
What Hashimoto's thyroiditis actually does to your body
Before diving into GLP-1 medications, understanding what Hashimoto's does at the cellular level makes everything else click into place.
Hashimoto's thyroiditis is an autoimmune condition where your immune system produces antibodies, specifically thyroid peroxidase antibodies (TPO) and thyroglobulin antibodies (TgAb), that attack your thyroid tissue. Over months and years, this sustained assault gradually destroys the gland's ability to produce thyroid hormones T3 and T4.
The consequences ripple outward through every system in your body.
Your metabolic rate drops. Basal metabolic rate, the calories your body burns simply existing, decreases significantly. Some research suggests reductions of 15-40% compared to individuals with healthy thyroid function. This is why weight gain happens even when caloric intake stays the same or decreases. Your body literally burns fewer calories doing the exact same activities.
Insulin resistance develops or worsens. Hashimoto's patients show higher rates of insulin resistance compared to the general population, even after thyroid hormone levels are optimized with medication. A study published in the Journal of Clinical Endocrinology found that patients with autoimmune thyroiditis had significantly higher HOMA-IR scores (a measure of insulin resistance) than matched controls, regardless of TSH levels. This creates a metabolic environment where weight loss becomes exceptionally difficult and fat storage, particularly visceral fat around the abdomen, becomes the default.
Inflammation becomes chronic. Hashimoto's is not just a thyroid problem. It is a systemic inflammatory condition. Elevated levels of C-reactive protein (CRP), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-alpha) are commonly found in Hashimoto's patients. This chronic inflammation drives fatigue, joint pain, muscle aches, brain fog, and mood disturbances that go far beyond simple thyroid hormone deficiency.
And then there is the gut connection.
The gut-thyroid axis and why it matters
Research has revealed a bidirectional relationship between intestinal health and thyroid function that researchers call the gut-thyroid axis. A landmark study published in Frontiers in Immunology found that patients with Hashimoto's thyroiditis had significantly altered gut microbiota composition and increased intestinal permeability compared to healthy controls.
Why does this matter for GLP-1 therapy? Because GLP-1 is produced primarily in the gut. L-cells in your intestinal lining produce and secrete GLP-1 in response to food intake. The health of your gut directly influences how much natural GLP-1 your body produces, how effectively it functions, and how you respond to exogenous GLP-1 medications.
Patients with Hashimoto's often have dysbiosis, an imbalance in gut bacteria that can reduce natural GLP-1 secretion, impair nutrient absorption (including thyroid medication absorption), and perpetuate the inflammatory cycle that drives autoimmune flares. Some specific bacterial strains like Lactobacillus acidophilus have been shown to promote GLP-1 secretion, and their depletion in Hashimoto's patients may partially explain why weight management becomes so challenging even before considering thyroid hormone levels.
How GLP-1 medications work and why Hashimoto's patients are interested
GLP-1 receptor agonists mimic the action of a natural hormone your body already produces. When you eat, your gut releases glucagon-like peptide-1, which signals your pancreas to release insulin, tells your brain you are full, and slows gastric emptying so nutrients absorb more gradually. The pharmaceutical versions amplify and extend these effects.
Semaglutide and tirzepatide achieve approximately 15-20% body weight reduction in clinical trials. That is not a marginal improvement. For someone carrying 200 pounds, that translates to 30-40 pounds of weight loss. For Hashimoto's patients who have spent years fighting metabolic dysfunction, those numbers are genuinely life-changing.
But weight loss is only part of the picture.
Anti-inflammatory effects that matter for autoimmune conditions
A growing body of research suggests that GLP-1 receptor agonists have significant anti-inflammatory properties independent of weight loss. A narrative review published in Cureus searched PubMed, Embase, and Scopus through August 2025, examining the intersection of GLP-1 therapy and autoimmune thyroid disease. The findings were notable.
GLP-1 receptor agonists suppress pro-inflammatory cytokines. Specifically, they reduce levels of IL-6, TNF-alpha, and CRP, the exact inflammatory markers that run high in Hashimoto's patients. They enhance regulatory T-cell function, which is significant because defective T-cell regulation is a core driver of autoimmune thyroid destruction. And they improve immune signaling from adipose tissue and the gut, two sites intimately connected to autoimmune thyroid disease progression.
This does not mean GLP-1 medications treat Hashimoto's directly. They do not reduce thyroid antibodies. They do not reverse thyroid tissue damage. They do not replace levothyroxine. But they address several of the metabolic and inflammatory factors that make living with Hashimoto's so difficult.
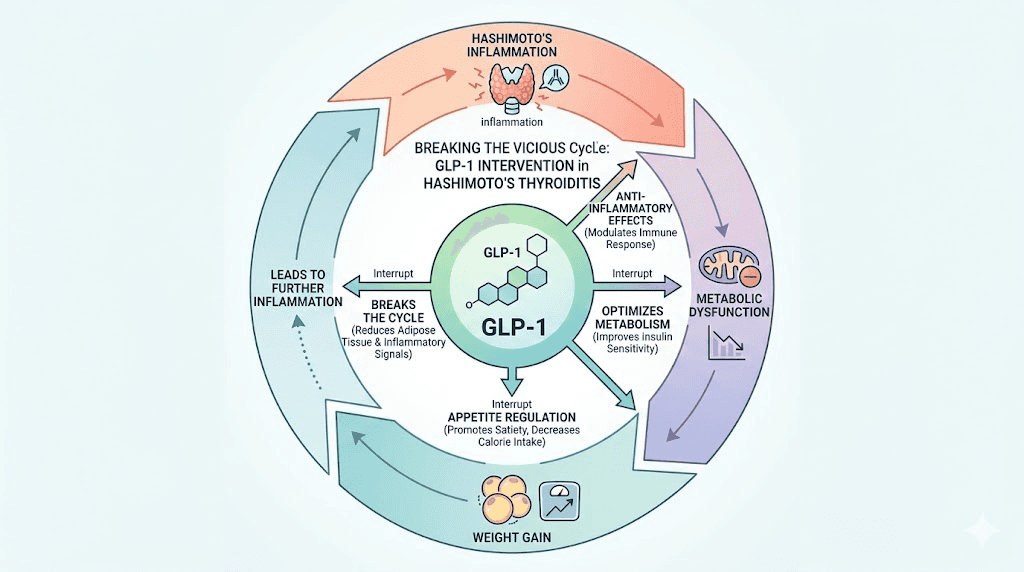
The metabolic reset that thyroid patients need
Here is what makes GLP-1 therapy particularly relevant for Hashimoto's patients. Autoimmune thyroid disease frequently coexists with metabolic syndrome, a cluster of conditions including obesity, insulin resistance, dyslipidemia, and fatty liver disease. This is not coincidental. The bidirectional relationship between immune dysregulation and metabolic stress means each condition amplifies the other.
GLP-1 medications can interrupt this cycle. By reducing body weight, improving insulin sensitivity, lowering inflammatory markers, and addressing metabolic dysfunction, they target the very factors that make Hashimoto's management so challenging. Even when thyroid hormone levels are perfectly optimized with medication, many patients still struggle with weight that will not budge, energy levels that remain low, and inflammatory symptoms that persist. GLP-1 therapy offers a potential pathway to address those residual symptoms.
Is it safe to take GLP-1 medications with Hashimoto's?
The short answer: yes, for most people. The longer answer requires understanding specific safety considerations that thyroid patients cannot afford to ignore.
No established medical contraindication exists for using GLP-1 receptor agonists in patients with Hashimoto's thyroiditis. The American Thyroid Association, the Endocrine Society, and the FDA have not identified autoimmune thyroid disease as a reason to avoid these medications. Hashimoto's is not listed as a contraindication for semaglutide, tirzepatide, or any other approved GLP-1 receptor agonist.
However. There is one absolute contraindication that every thyroid patient must know about.
The medullary thyroid carcinoma warning
All GLP-1 receptor agonists carry a black-box warning, the most serious warning the FDA issues, regarding thyroid C-cell tumors. In rodent studies, GLP-1 medications caused thyroid C-cell tumors, including medullary thyroid carcinoma (MTC). This is why every GLP-1 medication carries a warning against use in patients with a personal or family history of MTC or Multiple Endocrine Neoplasia syndrome type 2 (MEN2).
Now, context matters here. Human thyroid tissue expresses GLP-1 receptors at much lower levels than rodent tissue, with expression not exceeding 30% of human thyrocytes and parafollicular C-cells. A meta-analysis of 45 clinical trials including 52,600 patients found no statistically significant increased risk of thyroid cancer in GLP-1 receptor agonist users. However, a retrospective cohort study published in Diabetes Care did find that use of GLP-1 medications for 1-3 years was associated with a slightly increased risk of all thyroid cancer (adjusted hazard ratio 1.58) and medullary thyroid cancer specifically (adjusted hazard ratio 1.78).
The evidence is mixed. The risk is likely very small for most people. But for Hashimoto's patients, who already have compromised thyroid tissue and may have a higher baseline risk of thyroid nodules, this is not something to dismiss casually.
What to do: Before starting any GLP-1 medication, ensure you have had a thorough thyroid evaluation including thyroid ultrasound. If you have thyroid nodules, discuss the findings with your endocrinologist before proceeding. If you have any family history of medullary thyroid cancer or MEN2, GLP-1 medications are absolutely contraindicated, no exceptions.
What the research shows about GLP-1 and thyroid function
A comprehensive literature review published in the journal Medicines examined the effects of GLP-1 receptor agonists on thyroid volume, cancer risk, functionality, and TSH levels. The findings provide reassurance on several fronts.
GLP-1 medications do not appear to worsen autoimmune thyroid inflammation. They do not accelerate thyroid tissue destruction. They do not increase thyroid antibody levels. In fact, the immunomodulatory effects mentioned earlier, suppression of inflammatory cytokines and enhancement of regulatory T-cell function, could theoretically have mild protective effects on thyroid tissue, though no clinical study has confirmed this.
There is one notable case report worth knowing about. A 32-year-old woman with no prior thyroid history developed painless biphasic thyroiditis (initial hyperthyroidism followed by temporary hypothyroidism) after two months of tirzepatide therapy. Her condition resolved after discontinuation. While this is a single case report and not necessarily generalizable, it underscores why thyroid monitoring during GLP-1 therapy is not optional, it is essential.
How GLP-1 medications affect your levothyroxine
This is where practical concerns get very real for Hashimoto's patients. If you take levothyroxine (or liothyronine, natural desiccated thyroid, or any thyroid hormone replacement), GLP-1 medications can affect how your body processes that medication in several important ways.
Weight loss changes your levothyroxine dose requirements
Levothyroxine dosing is weight-based. Standard dosing protocols typically start around 1.6 mcg per kilogram of body weight. When you lose significant weight on a GLP-1 medication, your levothyroxine requirements change. This is not speculation. It is pharmacological certainty.
Research from Epic Research found that patients who lost weight after starting GLP-1 medications were more likely to see thyroid level improvements. But "improvement" in this context means TSH levels changed, and that can go in either direction depending on your current dose.
If you lose 10% or more of your body weight while taking levothyroxine at a dose calibrated for your higher weight, you may become relatively over-medicated. Symptoms of overmedication include heart palpitations, anxiety, tremor, excessive sweating, difficulty sleeping, and unintentional additional weight loss. These symptoms can easily be mistaken for side effects of the GLP-1 medication itself, leading to mismanagement.
Conversely, GLP-1 medications slow gastric emptying significantly. This altered gastrointestinal transit time can affect levothyroxine absorption in some patients, potentially reducing the amount of medication that reaches your bloodstream, which could worsen hypothyroid symptoms.
Medication timing becomes critical
Levothyroxine absorption is famously sensitive to timing and stomach contents. It should be taken on an empty stomach, typically 30-60 minutes before eating, separated from other medications and supplements by at least 4 hours (for calcium, iron, and similar minerals).
GLP-1 medications complicate this. If you take oral semaglutide (Rybelsus), it must also be taken on an empty stomach with no more than 4 ounces of plain water, at least 30 minutes before other oral medications, food, or drink. This creates a direct timing conflict with levothyroxine.
Practical solution: Take levothyroxine first thing in the morning. Wait at least 30 minutes. If taking oral semaglutide, take it at a completely different time (some clinicians recommend evening dosing for one or the other). Injectable GLP-1 formulations (subcutaneous semaglutide, tirzepatide) do not have this timing issue since they bypass the gastrointestinal tract for absorption. This is one reason many endocrinologists prefer injectable GLP-1 medications for patients on thyroid hormone replacement.
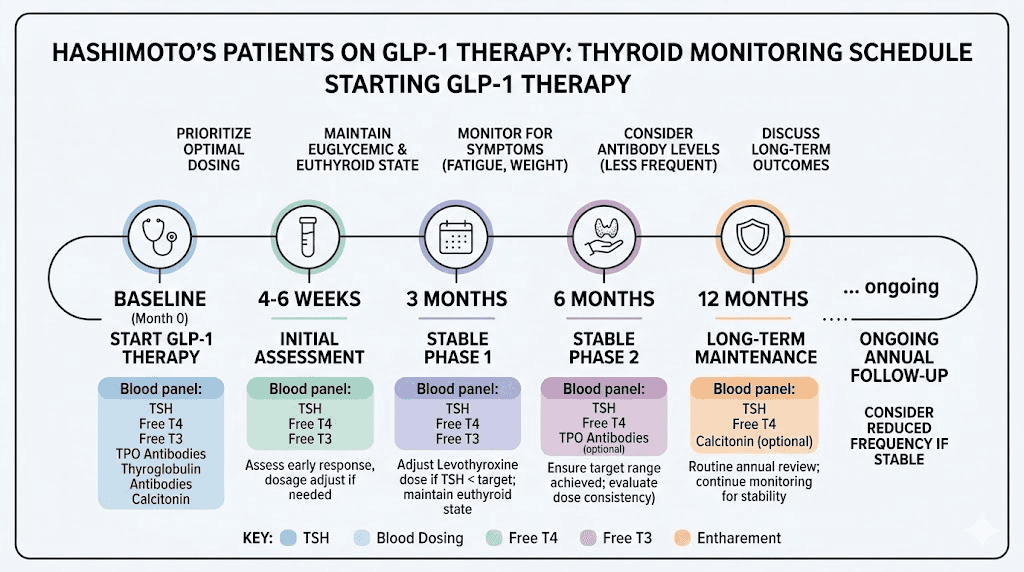
The monitoring schedule you need to follow
Standard thyroid monitoring for Hashimoto's patients on stable levothyroxine doses involves TSH checks every 6-12 months. When you add a GLP-1 medication, that schedule needs to tighten considerably.
Recommended monitoring protocol:
Baseline: Full thyroid panel (TSH, free T4, free T3, TPO antibodies, thyroglobulin antibodies) plus thyroid ultrasound before starting GLP-1 therapy
6-8 weeks after starting GLP-1: Recheck TSH and free T4. This catches early absorption changes
After each significant weight milestone (every 10-15 pounds lost): Recheck TSH and free T4
Every 3-4 months during active weight loss phase: Full thyroid panel including TSH, free T4, free T3
After weight stabilization: Return to every 6 months for the first year, then annually if stable
This is more frequent than most GLP-1 prescribers recommend for non-thyroid patients. But for Hashimoto's patients, catching dose changes early prevents weeks or months of unnecessary symptoms.
Choosing between semaglutide and tirzepatide with Hashimoto's
Both semaglutide and tirzepatide are options for Hashimoto's patients, but they work differently and have distinct considerations worth understanding.
Semaglutide: the pure GLP-1 approach
Semaglutide is a pure GLP-1 receptor agonist. It mimics only GLP-1, activating receptors in the pancreas, brain, and gut to reduce appetite, improve insulin sensitivity, and slow gastric emptying. Clinical trials demonstrate approximately 15% body weight reduction with semaglutide 2.4 mg weekly.
For Hashimoto's patients, semaglutide has the longest track record. It has been available longer than tirzepatide, which means more post-market surveillance data exists for patients with thyroid conditions. The side effect profile is well-characterized: nausea, constipation, and gastrointestinal discomfort are the most common issues, typically resolving within the first 4-8 weeks.
Available forms include injectable (Ozempic for diabetes, Wegovy for weight management) and oral (Rybelsus). As discussed above, the oral form creates timing complications with levothyroxine that injectable semaglutide avoids entirely.
Tirzepatide: the dual-action option
Tirzepatide is a dual GLP-1/GIP receptor agonist. It activates both the GLP-1 receptor and the glucose-dependent insulinotropic polypeptide (GIP) receptor. This dual mechanism produces greater weight loss in head-to-head comparisons, with clinical trials showing approximately 20-22% body weight reduction.
The additional GIP activation provides stronger effects on insulin sensitivity and glucose metabolism, which may be particularly relevant for Hashimoto's patients dealing with insulin resistance. GIP receptor activation also appears to have independent effects on fat metabolism and energy expenditure.
However, tirzepatide is the medication associated with the case report of induced thyroiditis mentioned earlier. While one case report does not establish causation, it is worth noting when making a choice between the two options.
Head-to-head comparison for thyroid patients
Factor | Semaglutide | Tirzepatide |
|---|---|---|
Mechanism | GLP-1 only | GLP-1 + GIP dual agonist |
Average weight loss | ~15% | ~20-22% |
Insulin sensitivity improvement | Significant | Greater than semaglutide |
Track record with thyroid patients | Longer (more safety data) | Shorter (newer medication) |
Thyroiditis case reports | Subclinical hypothyroidism reported | Biphasic thyroiditis case reported |
Oral option available | Yes (Rybelsus) | Not yet (trials ongoing) |
Levothyroxine interaction risk | Higher with oral form | Lower (injectable only) |
Anti-inflammatory effect | Documented | Documented (potentially stronger) |
Neither option is clearly superior for all Hashimoto's patients. Semaglutide offers more safety data and an oral option. Tirzepatide offers greater weight loss and potentially stronger metabolic benefits. The choice depends on your individual situation, insurance coverage, and your endocrinologist's assessment of your specific needs.
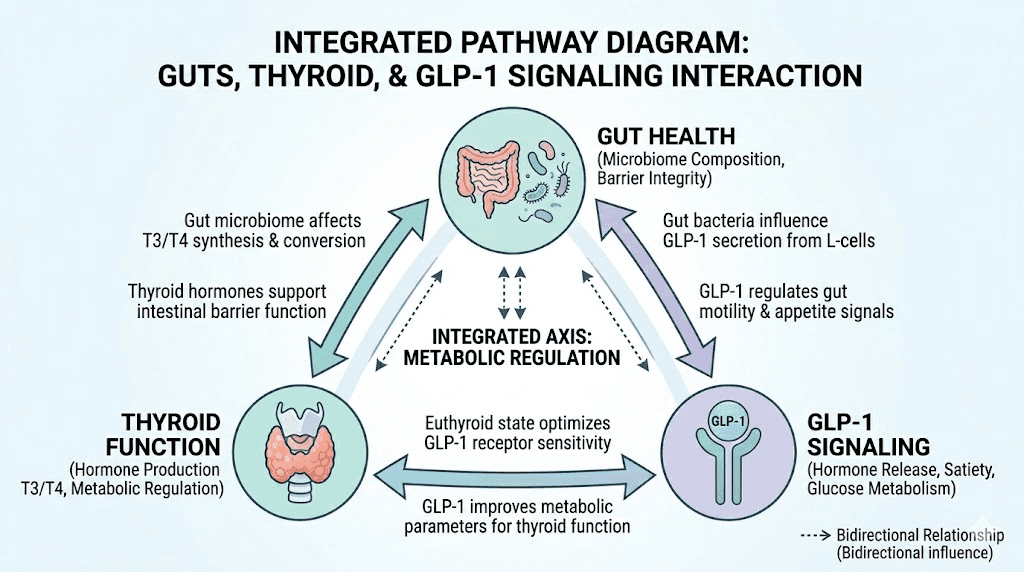
The gut-thyroid-GLP-1 triangle
One of the most fascinating aspects of GLP-1 therapy in Hashimoto's patients is the three-way relationship between gut health, thyroid function, and GLP-1 signaling. Understanding this triangle provides insights that most guides on this topic completely miss.
How Hashimoto's damages your gut
Hashimoto's patients have documented alterations in gut microbiota composition. Research published in Frontiers in Immunology demonstrated that these patients show reduced microbial diversity, decreased populations of beneficial bacteria (including Lactobacillus and Bifidobacterium species), and increased intestinal permeability, commonly known as leaky gut.
This intestinal permeability allows partially digested food particles, bacterial endotoxins, and other inflammatory triggers to cross the intestinal barrier and enter the bloodstream. The immune system responds to these foreign invaders, generating more inflammation, which further damages the gut lining, which allows more particles through. It is a self-reinforcing cycle that perpetuates both the gut damage and the autoimmune response against the thyroid.
How your gut produces GLP-1
Here is the connection most people miss. Your body's natural GLP-1 production depends heavily on gut health. L-cells in your intestinal lining produce GLP-1 in response to nutrients reaching the lower intestine. But these L-cells need a healthy intestinal environment to function properly.
Specific gut bacteria play direct roles in GLP-1 secretion. Lactobacillus acidophilus can regulate intestinal endocrine cell function and promote GLP-1 secretion. Short-chain fatty acids (SCFAs), produced by beneficial gut bacteria during fiber fermentation, are potent stimulators of GLP-1 release. When Hashimoto's disrupts the gut microbiome, it potentially reduces the body's natural GLP-1 production, contributing to the metabolic dysfunction that makes weight management so difficult.
How GLP-1 medications may improve gut health
GLP-1 receptor agonists have been demonstrated in preclinical and clinical trials to alter gut microbiota composition. They change gastric emptying rate and transit time, which modifies the intestinal environment including local pH levels and nutrient availability. Some research suggests these changes could actually improve microbial diversity over time.
Additionally, GLP-1 and its sister peptide GLP-2 both play roles in maintaining intestinal barrier integrity. By activating GLP-1 receptors in the gut, these medications may help support the intestinal barrier function that is compromised in Hashimoto's patients.
This creates an intriguing possibility: GLP-1 therapy might address not just the weight management challenges of Hashimoto's, but also contribute to improving the gut dysfunction that drives the autoimmune process itself. This is theoretical and not proven in clinical trials, but the mechanistic pathways are well-documented.
Practical protocols for Hashimoto's patients starting GLP-1 therapy
Theory is useful. Practical guidance is essential. Here is what a well-designed GLP-1 protocol looks like when you have Hashimoto's thyroiditis.
Pre-start checklist
Before your first injection, these items need to be completed:
Thyroid function optimized: Your TSH should be within your optimal range (not just "normal," but where you feel best, often in the lower half of the reference range for Hashimoto's patients). Uncontrolled hypothyroidism should be addressed before starting GLP-1 therapy
Full thyroid panel completed: TSH, free T4, free T3, TPO antibodies, thyroglobulin antibodies. This establishes your baseline
Thyroid ultrasound: Document any existing nodules. This is essential before starting medication that carries a thyroid cancer warning
Rule out MTC/MEN2: Confirm no personal or family history of medullary thyroid carcinoma or MEN2. If either exists, GLP-1 medications are absolutely contraindicated
Medications reviewed: Ensure your medication timing plan accounts for levothyroxine separation
Supplement review: Identify any supplements that might interact with either medication
Starting dose strategy for thyroid patients
Both semaglutide and tirzepatide are started at low doses and titrated upward. For Hashimoto's patients, there is a strong argument for slower titration than standard protocols.
Standard semaglutide titration:
Weeks 1-4: 0.25 mg weekly
Weeks 5-8: 0.5 mg weekly
Weeks 9-12: 1.0 mg weekly
Week 13+: 1.7 mg or 2.4 mg weekly (target dose)
Hashimoto's-adjusted approach:
Weeks 1-6: 0.25 mg weekly (extended to allow thyroid monitoring at week 6)
Weeks 7-12: 0.5 mg weekly (TSH check before increasing)
Weeks 13-16: 1.0 mg weekly
Week 17+: Assess whether higher doses are needed
The extended timeline allows for thyroid function checks at each step, ensuring levothyroxine doses are adjusted proactively rather than reactively. Many Hashimoto's patients find that moderate doses (0.5-1.0 mg semaglutide, 5-10 mg tirzepatide) provide meaningful benefits without the gastrointestinal intensity of maximum doses.
Some clinicians are exploring microdosing GLP-1 for autoimmune conditions specifically, using lower-than-standard doses to capture anti-inflammatory benefits while minimizing side effects and metabolic disruption. This approach is still emerging but represents an interesting frontier for thyroid patients who want immune-modulating effects without aggressive weight loss.
Managing side effects with thyroid disease
GLP-1 side effects can overlap with and mimic Hashimoto's symptoms, creating diagnostic confusion. Understanding which symptom comes from where is critical.
Symptom | GLP-1 side effect? | Hashimoto's symptom? | How to tell the difference |
|---|---|---|---|
Fatigue | Yes (from reduced caloric intake) | Yes (from low thyroid hormones) | Check TSH. If normal, likely GLP-1 related caloric deficit |
Nausea | Yes (common, especially early) | Less common | Usually GLP-1. Should improve within 4-8 weeks |
Constipation | Yes (from slowed gastric emptying) | Yes (from hypothyroidism) | If new or worsened after GLP-1 start, likely medication-related |
Hair loss | Yes (from rapid weight loss/nutrient deficiency) | Yes (classic Hashimoto's symptom) | Check ferritin, zinc, thyroid levels. Timing relative to GLP-1 start |
Brain fog | Possible (from caloric restriction) | Yes (common) | If worsened after GLP-1, check thyroid levels. May need dose adjustment |
Anxiety/palpitations | Rare | If hyperthyroid/overmedicated | Check TSH. Weight loss may have made levothyroxine dose too high |
Cold intolerance | Possible (less insulating fat) | Yes (classic hypothyroid) | Check TSH. Both can contribute |
Muscle pain | Yes (reported) | Yes (myalgia is common) | If new after GLP-1, may be medication. If chronic, likely Hashimoto's |
The key takeaway: when symptoms appear or worsen after starting GLP-1 therapy, the first step is always to check thyroid levels. Weight loss can shift levothyroxine requirements rapidly, and what looks like a GLP-1 side effect may actually be thyroid medication overmedication or undermedication.
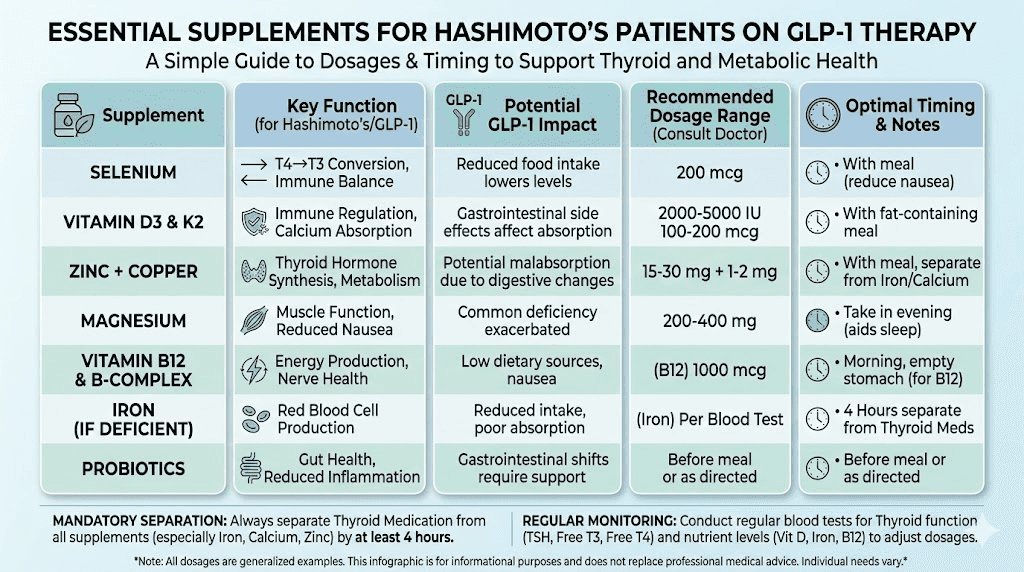
Nutrients and supplements that matter for both conditions
Hashimoto's patients on GLP-1 medications face a double challenge: the autoimmune condition depletes certain nutrients, and the GLP-1 medication's appetite suppression and altered gastric emptying can reduce nutrient intake and absorption further. Proactive supplementation is not optional, it is necessary.
Selenium
Selenium is the single most studied nutrient for Hashimoto's thyroiditis. Multiple randomized controlled trials show that selenium supplementation (200 mcg daily as selenomethionine) can reduce TPO antibody levels and may slow autoimmune thyroid destruction. It also plays a critical role in T4-to-T3 conversion through selenoprotein deiodinase enzymes.
GLP-1 medications can reduce selenium intake through decreased food consumption. Since Brazil nuts (the richest food source) may not be appealing on a suppressed appetite, supplementation becomes important.
Vitamin D
Vitamin D deficiency is significantly more prevalent in Hashimoto's patients compared to the general population. Low vitamin D is associated with higher thyroid antibody levels and increased autoimmune activity. GLP-1 medications can reduce dietary vitamin D intake, and significant fat loss can temporarily release stored vitamin D from fat cells before new equilibrium is established.
Most endocrinologists recommend maintaining vitamin D levels above 40-60 ng/mL for autoimmune thyroid patients, often requiring 2,000-5,000 IU daily depending on baseline levels.
B12 and iron
Both vitamin B12 and iron deserve special attention. Hashimoto's patients have higher rates of pernicious anemia (B12 deficiency) due to autoimmune gastritis co-occurrence. GLP-1 medications can further impair B12 absorption through altered gastric acid production and slowed transit time.
Iron deficiency is common in Hashimoto's, particularly in menstruating women. Both iron and B12 are critical for thyroid hormone production and should be monitored regularly.
Zinc and magnesium
Zinc supports thyroid hormone synthesis, immune function, and T4-to-T3 conversion. Magnesium supports over 300 enzymatic reactions and is commonly depleted in both autoimmune conditions and during caloric restriction. Both can be supplemented safely alongside GLP-1 medications, but should be taken at least 4 hours away from levothyroxine to avoid absorption interference.
Probiotics
Given the gut-thyroid connection discussed earlier, probiotic supplementation makes physiological sense for Hashimoto's patients on GLP-1 therapy. Strains like Lactobacillus acidophilus and Bifidobacterium lactis have been shown to support intestinal barrier function, promote natural GLP-1 secretion, and may modulate autoimmune responses. A probiotic specifically designed for GLP-1 users addresses the gut microbiome changes that both Hashimoto's and GLP-1 medications create.
Diet and lifestyle considerations specific to this combination
Hashimoto's patients on GLP-1 medications need a more strategic approach to nutrition than the average GLP-1 user. The reduced appetite makes every calorie count more, and the autoimmune condition places specific demands on dietary composition.
Protein becomes non-negotiable
GLP-1 medications cause weight loss. Not all weight loss is equal. Without adequate protein intake, a significant portion of weight lost comes from muscle tissue rather than fat. This is problematic for everyone. For Hashimoto's patients, it is particularly concerning because muscle mass directly influences basal metabolic rate, which is already compromised by thyroid dysfunction.
Target a minimum of 0.7-1.0 grams of protein per pound of lean body mass daily. This may require protein supplementation on days when appetite suppression is strongest. Prioritize protein-rich foods at every meal: eggs, fish, poultry, lean meats, and if tolerated, dairy products.
The gluten question
The relationship between gluten and Hashimoto's remains debated but is worth discussing. Some research suggests that molecular mimicry between gluten proteins (gliadin) and thyroid tissue may trigger or worsen autoimmune thyroid attacks. Not all Hashimoto's patients are sensitive to gluten, but those who are often report significant symptom improvement on a gluten-free diet.
GLP-1 medications do not change this calculus. If you have identified gluten sensitivity alongside your Hashimoto's, maintaining gluten avoidance on GLP-1 therapy is still important. If you have not explored this, the dietary changes that GLP-1 medications necessitate might be a good time to trial a gluten-free period and assess symptom response.
Anti-inflammatory eating patterns
Given that both Hashimoto's and metabolic dysfunction involve chronic inflammation, an anti-inflammatory dietary approach makes sense. This does not mean a specific named diet. It means prioritizing foods that reduce inflammation and avoiding foods that promote it.
Prioritize: Fatty fish (omega-3s), colorful vegetables, berries, turmeric, ginger, olive oil, nuts, seeds
Limit: Refined sugars, processed oils (soybean, corn, canola), ultra-processed foods, excessive alcohol
The dietary strategy for GLP-1 users needs to account for the reduced appetite window. When you are eating less overall, the quality of what you eat matters exponentially more. An anti-inflammatory approach ensures that every meal serves double duty: providing essential nutrients while supporting both thyroid and metabolic health.
Exercise with thyroid disease on GLP-1
Exercise is more important, and more complicated, for Hashimoto's patients on GLP-1 therapy than for other populations. Resistance training is essential to preserve muscle mass during weight loss. But Hashimoto's patients often deal with fatigue, joint pain, and exercise intolerance that make intense training difficult.
Start with what you can do consistently. Walking, swimming, and light resistance training are excellent starting points. As weight decreases and energy improves (both from better thyroid management and GLP-1 therapy), gradually increase intensity. Aim for at least 150 minutes of moderate activity weekly plus 2-3 resistance training sessions.
One important note: intense exercise can temporarily increase inflammation in Hashimoto's patients. During autoimmune flares, reduce intensity and focus on gentle movement. Listen to your body. The goal is consistent, sustainable activity, not exhaustive workouts that trigger flare responses.
Special populations and scenarios
Women with Hashimoto's and PCOS
A significant number of women have both Hashimoto's thyroiditis and polycystic ovary syndrome. Both conditions involve insulin resistance, hormonal imbalance, and difficulty with weight management. GLP-1 medications are particularly beneficial in this overlap population because they address the insulin resistance that drives both conditions.
However, women managing both conditions need more frequent monitoring and may need coordinated care between endocrinology and gynecology. The hormonal changes from weight loss can affect menstrual cycles, PCOS symptoms, and thyroid function simultaneously, creating a complex picture that requires experienced medical oversight.
Hashimoto's patients in perimenopause
Perimenopause adds another layer of complexity. Fluctuating estrogen levels affect thyroid binding globulin, which can alter how much thyroid hormone is bioavailable even when total levels appear normal. Weight redistribution during perimenopause tends toward visceral fat, which increases inflammation and insulin resistance.
GLP-1 medications can be genuinely helpful during this transition, but the perimenopause hormonal fluctuations mean thyroid levels may need even more frequent monitoring. Some women find their levothyroxine needs shift not just from weight loss but from changing estrogen levels simultaneously.
Pregnancy planning
GLP-1 medications must be discontinued well before conception. The current recommendation is to stop semaglutide at least 2 months before planned pregnancy and tirzepatide at least 1 month before. For Hashimoto's patients, this discontinuation requires careful planning because:
Thyroid hormone requirements increase significantly during pregnancy (typically 25-50% higher)
Weight regain after GLP-1 discontinuation will change levothyroxine requirements again
Optimal thyroid function before conception is critical for fetal brain development
If you discover you are pregnant while on a GLP-1 medication, stop the medication immediately and contact your healthcare provider for urgent thyroid function testing and levothyroxine dose adjustment.
What GLP-1 medications cannot do for Hashimoto's
Managing expectations is important. GLP-1 medications are powerful tools, but they are not thyroid treatments. Understanding their limitations prevents disappointment and dangerous self-management decisions.
They do not treat the autoimmune process
No clinical evidence demonstrates that GLP-1 receptor agonists reduce thyroid antibody levels or modify the autoimmune destruction of thyroid tissue. The immunomodulatory effects documented in research are systemic anti-inflammatory effects, not targeted autoimmune therapy. Your Hashimoto's will continue to require the same immune management regardless of GLP-1 use.
They do not replace thyroid hormone
GLP-1 medications do not improve thyroid hormone production. If your thyroid gland has been damaged by Hashimoto's to the point where it cannot produce adequate T4 and T3, no amount of GLP-1 therapy will change that. Levothyroxine (or other thyroid hormone replacement) remains necessary.
Some patients notice improved energy and reduced fatigue on GLP-1 therapy and wonder if they can reduce their thyroid medication. Do not do this without laboratory confirmation. The improved energy may come from weight loss, reduced inflammation, or better metabolic function, not from improved thyroid hormone production. Reducing thyroid medication without testing can lead to dangerous hypothyroidism.
They do not guarantee weight loss in all thyroid patients
While GLP-1 medications are remarkably effective for weight loss in clinical trials, individual responses vary. Some Hashimoto's patients, particularly those with significantly compromised thyroid function, persistent untreated insulin resistance, or cortisol dysregulation, may experience less dramatic results. If you are not losing weight on GLP-1 therapy, thyroid optimization should be the first thing investigated. Many patients are "treated" with levothyroxine but not optimized, meaning their TSH is within the reference range but not in the lower portion where many Hashimoto's patients feel and function best.
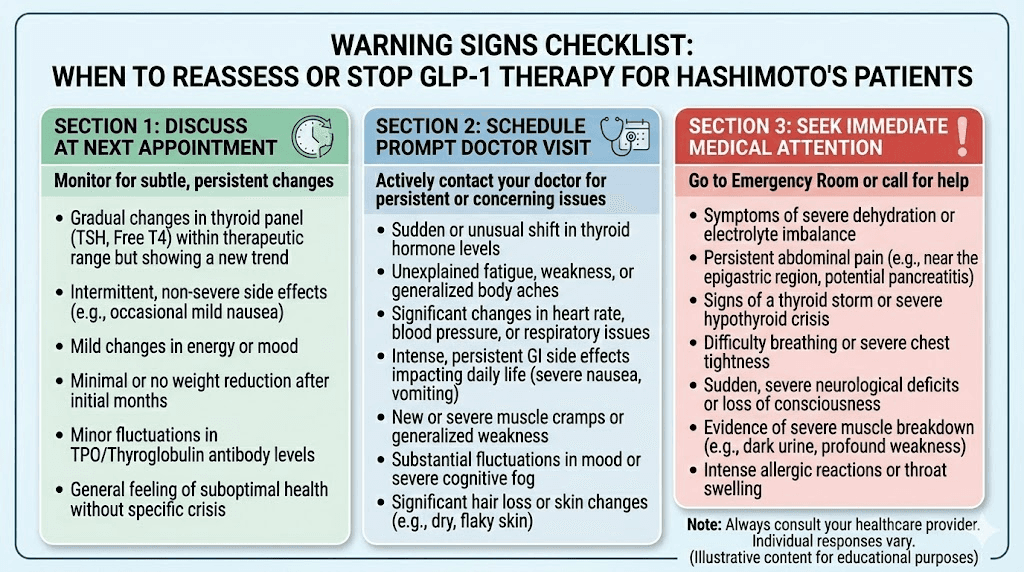
When to stop or reconsider GLP-1 therapy
Not every Hashimoto's patient should stay on GLP-1 medications indefinitely. There are clear signals that something needs to change.
Stop immediately and contact your doctor if:
Severe neck swelling or a palpable thyroid mass develops
Symptoms of thyrotoxicosis appear: rapid heartbeat, severe anxiety, excessive sweating, tremor, sudden unexplained weight loss
Severe pancreatitis symptoms: intense upper abdominal pain radiating to the back with nausea and vomiting
You discover you are pregnant
Discuss with your endocrinologist if:
TSH levels become unstable despite levothyroxine adjustments
New thyroid nodules appear on follow-up ultrasound
Hair loss is severe and persistent despite adequate nutrient levels
Fatigue worsens despite thyroid levels appearing normal
Autoimmune flare frequency appears to increase
You reach your target weight and want to explore discontinuation
For patients who achieve their weight loss goals and want to stop GLP-1 therapy, tapering is recommended over abrupt discontinuation. Gradual dose reduction over 4-8 weeks allows the body to readjust, and thyroid levels should be checked both during and after the taper, since weight stabilization changes levothyroxine requirements once again.
The research landscape and what is coming
The intersection of GLP-1 therapy and autoimmune thyroid disease is an active area of research. Understanding where the science is heading helps set realistic expectations about what we know now versus what we might learn soon.
Immunometabolism: the emerging framework
Researchers are increasingly studying "immunometabolism," the bidirectional relationship between metabolic processes and immune function. GLP-1 receptor agonists sit right at this intersection. Their ability to improve metabolic health while exerting anti-inflammatory and immunomodulatory effects makes them uniquely interesting for autoimmune conditions.
Current research is examining whether GLP-1 therapy can modify the trajectory of autoimmune diseases by addressing the metabolic dysfunction that amplifies immune dysregulation. For Hashimoto's specifically, the question is whether improving insulin sensitivity, reducing visceral fat, and lowering inflammatory markers could slow the autoimmune destruction of thyroid tissue. No clinical trial has answered this question definitively yet, but the mechanistic rationale is compelling.
Next-generation GLP-1 medications
The GLP-1 medication landscape is expanding rapidly. Retatrutide, a triple agonist targeting GLP-1, GIP, and glucagon receptors simultaneously, has shown even greater weight loss in early trials (up to 24% body weight reduction). CagriSema, combining semaglutide with cagrilintide (an amylin analog), represents another approach.
For Hashimoto's patients, these newer medications will need the same careful thyroid monitoring and management. The greater the weight loss potential, the more significant the levothyroxine dose adjustments will need to be. But these medications may also bring stronger anti-inflammatory and metabolic benefits, potentially making them even more useful for autoimmune populations.
Personalized medicine approaches
The future of GLP-1 therapy in Hashimoto's likely involves more personalized approaches. Genetic testing may help predict which patients will respond best to specific GLP-1 medications. Gut microbiome analysis could guide probiotic and prebiotic interventions that optimize both natural GLP-1 production and autoimmune management. And more sophisticated thyroid monitoring, including real-time tracking of free T3 and T4 alongside TSH, could enable proactive medication adjustments rather than reactive ones.
Finding the right medical team
Managing Hashimoto's on GLP-1 therapy requires coordinated medical care. This is not a condition where a single general practitioner can manage everything optimally.
The ideal medical team includes:
Endocrinologist: Manages thyroid function, monitors TSH and antibodies, adjusts levothyroxine, screens for thyroid nodules and cancer risk
Obesity medicine specialist or prescribing clinician: Manages GLP-1 dosing, titration, and side effects
Registered dietitian: Develops nutrition plans that address both autoimmune support and adequate intake on reduced appetite
Communication between these providers is essential. Your endocrinologist needs to know about GLP-1 dose changes and weight milestones. Your GLP-1 prescriber needs to know about thyroid level changes and levothyroxine adjustments. If these providers are not communicating, you become the bridge, and that means being informed and proactive.
SeekPeptides members access comprehensive guides on finding qualified providers, understanding lab results, and navigating the complexities of peptide therapy alongside existing medical conditions. For Hashimoto's patients exploring GLP-1 options, having reliable, evidence-based resources is the difference between informed decision-making and risky guesswork.
Frequently asked questions
Can GLP-1 medications cure Hashimoto's thyroiditis?
No. GLP-1 medications do not treat or cure Hashimoto's thyroiditis. They do not reduce thyroid antibodies, reverse thyroid tissue damage, or modify the autoimmune process. They can help manage metabolic complications associated with Hashimoto's, particularly weight management and insulin resistance, but thyroid hormone replacement therapy remains necessary.
Do I need to adjust my levothyroxine dose when starting a GLP-1 medication?
Likely yes, but not immediately. As you lose weight, your levothyroxine requirements will decrease. Weight loss of 10% or more typically necessitates a dose reduction. Check TSH levels 6-8 weeks after starting GLP-1 therapy and after each significant weight milestone. Never adjust thyroid medication without laboratory confirmation.
Is semaglutide or tirzepatide better for Hashimoto's patients?
Neither is definitively better. Semaglutide has a longer safety track record. Tirzepatide produces greater weight loss and may have stronger effects on insulin sensitivity. The choice depends on individual factors including thyroid status, metabolic profile, insurance coverage, and your endocrinologist's assessment.
Can I take oral semaglutide with levothyroxine?
Yes, but timing is critical. Both medications require empty-stomach administration. Take levothyroxine first thing in the morning, wait at least 30 minutes, then take oral semaglutide. Alternatively, consider injectable semaglutide to avoid the absorption conflict entirely.
Will GLP-1 medications worsen my thyroid antibodies?
Current evidence says no. GLP-1 medications have not been shown to increase TPO or thyroglobulin antibody levels. Their anti-inflammatory effects may theoretically provide mild benefit, though this has not been confirmed in clinical trials focused on Hashimoto's specifically.
How often should I check my thyroid levels while on GLP-1 therapy?
More frequently than standard Hashimoto's monitoring. Check at baseline, 6-8 weeks after starting, after each 10-15 pounds of weight loss, every 3-4 months during active weight loss, and every 6 months after weight stabilization. Full panels including TSH, free T4, and free T3 are recommended.
Can GLP-1 medications cause thyroid cancer?
The risk is debated. Animal studies showed C-cell tumors in rodents. Human evidence is mixed, with some studies showing no increased risk and others showing a small increase. The FDA black-box warning applies to all GLP-1 medications. Patients with personal or family history of medullary thyroid carcinoma or MEN2 must not use these medications.
Should I get a thyroid ultrasound before starting GLP-1 therapy?
Yes. A baseline thyroid ultrasound is recommended for all Hashimoto's patients starting GLP-1 medications. This documents any existing nodules and provides a comparison point for future monitoring. Discuss follow-up ultrasound timing with your endocrinologist.
External resources
The Thyroid Twist: How GLP-1 agonists are influencing autoimmune thyroid care (PMC)
Implications of GLP-1 receptor agonist on thyroid function: literature review (PMC)
Alterations in gut microbiota and intestinal permeability in Hashimoto thyroiditis (Frontiers)
Risk of thyroid tumors with GLP-1 receptor agonists (Diabetes Care)
For researchers serious about understanding the full picture of peptide therapy alongside autoimmune conditions, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, detailed protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your thyroid levels stay stable, your inflammation stay low, and your protocols stay informed.