Mar 16, 2026

You found a thyroid nodule. Maybe your doctor found it during a routine ultrasound. Maybe you felt a lump in your neck while shaving or clasping a necklace. Either way, you now have two facts competing for space in your head. Thyroid nodules are incredibly common, appearing in up to 68% of adults when detected by ultrasound. And tirzepatide, the GLP-1/GIP dual receptor agonist you were considering for weight loss or blood sugar management, carries an FDA boxed warning about thyroid tumors.
Those two facts create panic.
But panic is not the same thing as risk. And that boxed warning, the most serious label the FDA can apply, tells a more complicated story than most people realize. The warning exists because of what happened in rodents, not because of what has been observed in humans. Over 10,000 participants in clinical trials used tirzepatide for up to two years without any demonstrated increase in thyroid nodule formation or thyroid cancer incidence. A large retrospective cohort study involving 283,004 matched pairs found that tirzepatide users actually had a 65% lower incidence of malignant thyroid cancer compared to non-users, with a relative risk of 0.348. That is not a small difference.
None of this means the concern is imaginary. It means the concern is specific, limited to certain thyroid cancer types and genetic conditions that your doctor can screen for before you start treatment. This guide walks through every piece of evidence, every contraindication, every monitoring recommendation, and every practical question that researchers with thyroid nodules face when evaluating tirzepatide therapy. By the end, you will know exactly where you stand.

Understanding thyroid nodules before evaluating medication risk
Before diving into tirzepatide safety data, you need context about what thyroid nodules actually are. Without that context, the FDA warning reads like a death sentence. With it, the warning reads like a precaution based on incomplete evidence.
Thyroid nodules are growths that form within the thyroid gland, the butterfly-shaped organ at the base of your neck that regulates metabolism through hormone production. They range from tiny clusters of cells invisible to the naked eye all the way to visible lumps that change the shape of your neck. The thyroid gland contains two main cell types relevant to this discussion. Follicular cells produce thyroid hormones T3 and T4. Parafollicular cells, also called C-cells, produce calcitonin, a hormone involved in calcium regulation. This distinction matters enormously when evaluating tirzepatide risk because the FDA warning specifically concerns C-cell tumors, not the far more common follicular or papillary thyroid cancers.
How common are thyroid nodules?
Extremely common. Physical examination detects nodules in 5-7% of adults. Ultrasound examination finds them in 19-68% of adults. Autopsy studies reveal them in 8-65% of the general population. These numbers mean that if you are reading this article because a nodule showed up on a scan, you are in the majority, not the minority.
Women develop thyroid nodules at significantly higher rates than men. Prevalence increases with age. Iron deficiency, prior radiation exposure to the head or neck, and family history of thyroid disease all increase the likelihood of developing nodules. Most critically, the vast majority of these nodules are benign. Only about 5% of palpable thyroid nodules turn out to be malignant, and among those malignancies, approximately 95% are non-medullary thyroid cancers, specifically papillary and follicular types that carry excellent prognoses and are NOT the cancer type relevant to the tirzepatide warning.
Medullary thyroid carcinoma, the specific cancer type mentioned in the tirzepatide boxed warning, arises from C-cells and accounts for roughly 3-5% of all thyroid cancers. It is rare. Understanding this rarity is essential to putting the FDA warning in proper perspective.
Types of thyroid nodules and their clinical significance
Colloid nodules are the most common type, representing adenomatous benign neoplasms that do not pose an increased risk of malignancy. They are simply overgrowths of normal thyroid tissue. Thyroid cysts are fluid-filled nodules that are almost always benign. Inflammatory nodules can develop from conditions like Hashimoto thyroiditis or subacute thyroiditis. Follicular adenomas are benign tumors that sometimes require biopsy to distinguish from follicular carcinoma.
Then there are the malignant types. Papillary thyroid carcinoma accounts for about 87% of thyroid cancers and carries a five-year survival rate exceeding 98%. Follicular thyroid carcinoma makes up roughly 5-10% of thyroid cancers. Medullary thyroid carcinoma, the one connected to the GLP-1 receptor agonist warning, represents only 3-5% of thyroid cancers. And anaplastic thyroid carcinoma, the rarest and most aggressive type, accounts for less than 2%.
The takeaway here is direct. If you have a thyroid nodule, the overwhelming probability is that it is benign. If it happens to be malignant, the overwhelming probability is that it is papillary or follicular, neither of which is implicated in the tirzepatide safety concern. The specific cancer type that prompted the FDA warning represents a tiny fraction of an already uncommon outcome.
The FDA boxed warning explained
The FDA requires a boxed warning on all GLP-1 receptor agonists, including tirzepatide (marketed as Mounjaro for type 2 diabetes and Zepbound for weight management). This warning states that tirzepatide causes thyroid C-cell tumors at clinically relevant exposures in rodents, and that it is unknown whether tirzepatide causes thyroid C-cell tumors, including medullary thyroid carcinoma, in humans.
Read that again carefully. The warning does not say tirzepatide causes thyroid cancer in humans. It says it caused C-cell tumors in rodents, and the human risk is unknown. That distinction is everything.
What happened in the rodent studies
In two-year toxicology studies, rats receiving tirzepatide developed dose-dependent and treatment-duration-dependent increases in thyroid C-cell tumors, including both adenomas and carcinomas. The effect was observed in both male and female rats at clinically relevant plasma exposures. This finding triggered the boxed warning because the FDA applies precautionary standards, if an animal study shows a serious risk, the warning goes on the label until human data proves otherwise.
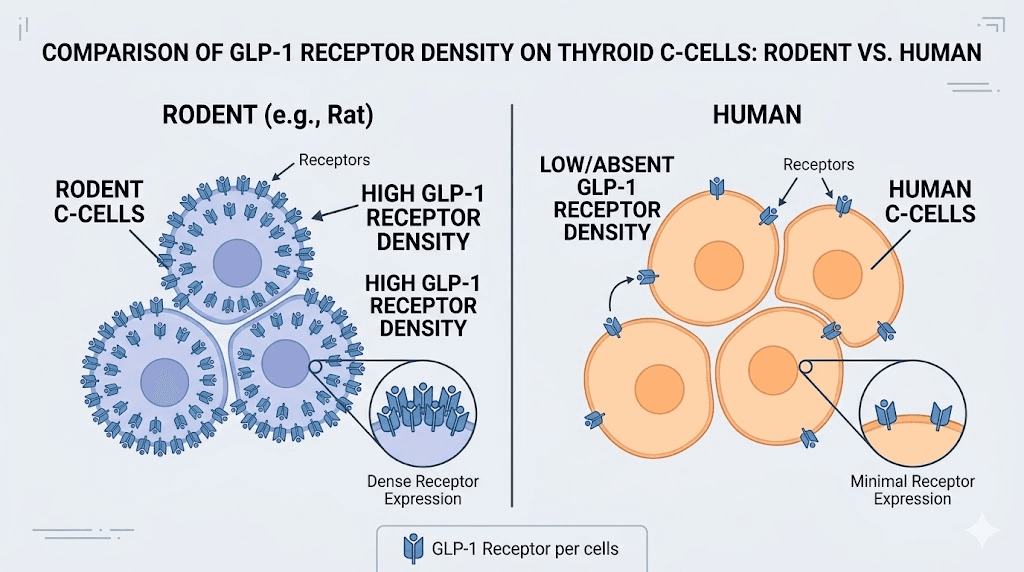
The mechanism behind these rodent tumors is well understood. Rodent thyroid C-cells express high levels of GLP-1 receptors. When GLP-1 receptor agonists chronically stimulate these receptors, the C-cells proliferate. Over time, this proliferation leads to hyperplasia, then adenomas, then potentially carcinomas. The pathway is clear, reproducible, and specific to the rodent thyroid.
Why rodent findings may not apply to humans
Here is where the story diverges sharply from the scary headline version. Human thyroid C-cells express substantially lower levels of GLP-1 receptors compared to rodent C-cells. The level of GLP-1 receptor expression in thyroid tissue is species-specific, with higher receptor expression reported in rodents compared with humans. This biological difference means that the mechanism driving C-cell tumors in rats, chronic GLP-1 receptor stimulation leading to C-cell proliferation, may simply not operate in human thyroid tissue, or at least not at anywhere near the same intensity.
Multiple lines of evidence support this interpretation. Primate studies, including those in cynomolgus monkeys, have not demonstrated the same C-cell effects seen in rodents. Human clinical trials involving tens of thousands of participants have not shown increased thyroid cancer incidence. And population-level observational studies have not identified a signal suggesting increased medullary thyroid carcinoma risk in GLP-1 receptor agonist users.
The FDA maintains the boxed warning because proving a negative, proving that something definitively does NOT cause cancer, requires decades of surveillance data. The warning represents appropriate regulatory caution, not confirmed human risk.
Clinical trial evidence on tirzepatide and thyroid safety
The most important evidence for anyone evaluating tirzepatide with thyroid nodules comes from large-scale clinical trials. These are not animal studies, not case reports, not theoretical models. These are controlled studies involving real humans taking real tirzepatide at therapeutic doses for extended periods.
The SURPASS and SURMOUNT trial programs
Tirzepatide was evaluated in over 10,000 participants across the SURPASS (type 2 diabetes) and SURMOUNT (obesity) trial programs. Treatment durations extended up to two years. Participants underwent regular monitoring including adverse event tracking. The result: clinical trials have not demonstrated increased thyroid nodule or cancer incidence in humans.
That sentence deserves emphasis because it directly contradicts the fear that the boxed warning creates. More than 10,000 people used tirzepatide in controlled clinical settings. Thyroid cancer did not increase. Thyroid nodule formation did not increase. The finding that drove the boxed warning, C-cell tumors in rodents, did not replicate in humans.
Calcitonin levels in clinical trials
Calcitonin is the hormone produced by thyroid C-cells, the same cells involved in medullary thyroid carcinoma. If tirzepatide were stimulating human C-cell proliferation the way it stimulates rodent C-cells, you would expect to see calcitonin levels rise. In clinical trials, serum calcitonin showed no clinically meaningful changes in the vast majority of participants. Some dose-dependent increases were observed at higher doses compared to placebo, but these remained within normal ranges and did not indicate pathological C-cell stimulation.
This calcitonin data is particularly reassuring. Elevated calcitonin, specifically levels exceeding 50 ng/L, serves as a tumor marker for medullary thyroid carcinoma. The fact that tirzepatide at therapeutic doses did not push calcitonin into concerning ranges provides direct biochemical evidence against the rodent-to-human translation of C-cell risk.
Post-marketing surveillance and real-world data
Beyond clinical trials, post-marketing surveillance through the FDA Adverse Event Reporting System (FAERS) has produced mixed but ultimately reassuring results. One analysis found a reporting odds ratio (ROR) of 2.09 for thyroid cancer with tirzepatide, meaning thyroid cancer was reported roughly twice as often as expected. However, this number requires careful interpretation.
Reporting odds ratios from FAERS data do not establish causation. They reflect how often a side effect is reported relative to other drugs, and they are heavily influenced by media attention, label warnings, and reporting bias. When a drug carries a boxed warning about thyroid cancer, doctors and patients are more likely to report any thyroid event, inflating the apparent signal. Notably, tirzepatide showed a lower ROR than other GLP-1 agonists, including liraglutide (ROR 15.59) and semaglutide (ROR 7.61).
The strongest real-world evidence comes from a retrospective cohort analysis published in the Journal of the Endocrine Society. This study used propensity score matching with 283,004 matched pairs from the TriNetX Global Collaborative Network. The finding was striking: patients treated with tirzepatide did not demonstrate a heightened risk of malignant thyroid cancer. In fact, the results showed a significantly lower incidence, with a relative risk of 0.348 (p < 0.001, 95% CI: 0.297-0.408). That translates to a 65% reduction in thyroid cancer incidence among tirzepatide users compared to matched controls.
The study authors noted that their findings align with similar research on other GLP-1 receptor agonists and suggested that the boxed warning may not be renewed as more evidence accumulates. While a single study cannot be considered definitive, a 65% risk reduction across nearly 300,000 matched pairs is difficult to dismiss.
Can you take tirzepatide if you have thyroid nodules?
This is the question driving most people to this article, and the answer depends on what type of thyroid nodule you have and your medical history. The short version: benign thyroid nodules are NOT a contraindication to tirzepatide use.
When tirzepatide is safe to use with thyroid nodules
Tirzepatide is not contraindicated for patients with benign thyroid nodules. Colloid nodules, thyroid cysts, inflammatory nodules, and follicular adenomas do not prevent you from using tirzepatide. Having a nodule discovered incidentally on imaging does not disqualify you from treatment.
Tirzepatide is also not contraindicated for patients with a history of common thyroid cancers, specifically papillary or follicular thyroid carcinoma. These cancer types arise from follicular cells, not C-cells, and are unrelated to the mechanism of concern described in the boxed warning. If you have had papillary thyroid cancer treated successfully, that history alone does not prevent starting tirzepatide.
SeekPeptides provides comprehensive guides to help researchers understand these distinctions and make informed decisions about their protocols. The nuance matters because blanket avoidance based on misunderstanding the boxed warning means missing out on a medication that could significantly benefit metabolic health.
When tirzepatide is absolutely contraindicated
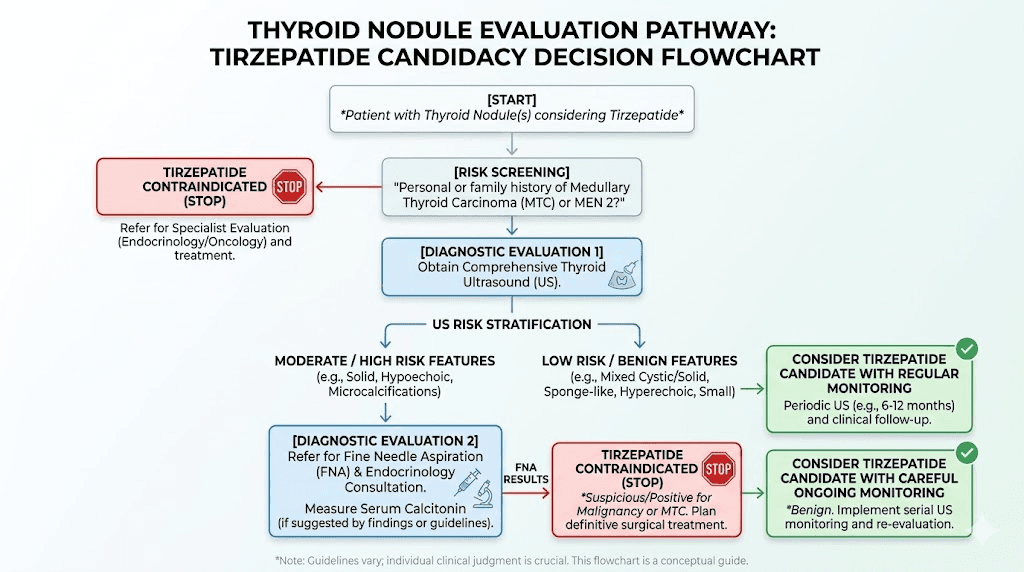
The FDA prescribing information lists two absolute contraindications related to thyroid conditions:
Personal or family history of medullary thyroid carcinoma (MTC). If you have ever been diagnosed with MTC, or if a first-degree relative (parent, sibling, child) has been diagnosed with MTC, tirzepatide should not be used. This contraindication exists because MTC arises from the same C-cells that showed tumor formation in rodent studies, and individuals with MTC history may have underlying genetic susceptibility that could theoretically interact with GLP-1 receptor stimulation.
Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). MEN 2 is a genetic condition that dramatically increases the risk of developing MTC. Individuals with MEN 2 carry mutations in the RET proto-oncogene that predispose C-cells to malignant transformation. Adding a medication that stimulates GLP-1 receptors on C-cells, even if human C-cells respond minimally compared to rodent C-cells, represents an unacceptable theoretical risk in this population.
These contraindications are specific and screenable. A simple family history discussion with your physician and, if needed, genetic testing can determine whether you fall into either category. The vast majority of people with thyroid nodules do not.
The gray area: nodules requiring further evaluation
The FDA prescribing information notes that patients with elevated serum calcitonin or thyroid nodules noted on physical examination or neck imaging should be further evaluated. This does not mean tirzepatide is contraindicated. It means your physician should assess the nodule before starting treatment.
Standard evaluation typically involves thyroid ultrasound with standardized risk stratification (ACR TI-RADS or similar system), fine needle aspiration biopsy for nodules meeting size and suspicion criteria, serum calcitonin measurement if medullary thyroid carcinoma is a concern, and review of family history for MTC and MEN 2.
If evaluation confirms a benign nodule, standard tirzepatide dosing protocols can proceed. If evaluation reveals a suspicious nodule requiring further workup, most endocrinologists will recommend completing the thyroid evaluation before initiating tirzepatide, not because tirzepatide would worsen the nodule, but because establishing a baseline before starting any new medication is sound medical practice.
Calcitonin monitoring and thyroid screening guidelines
One of the most common questions researchers ask is whether they need ongoing thyroid monitoring while taking tirzepatide. The answer, based on current clinical guidelines, may surprise you.
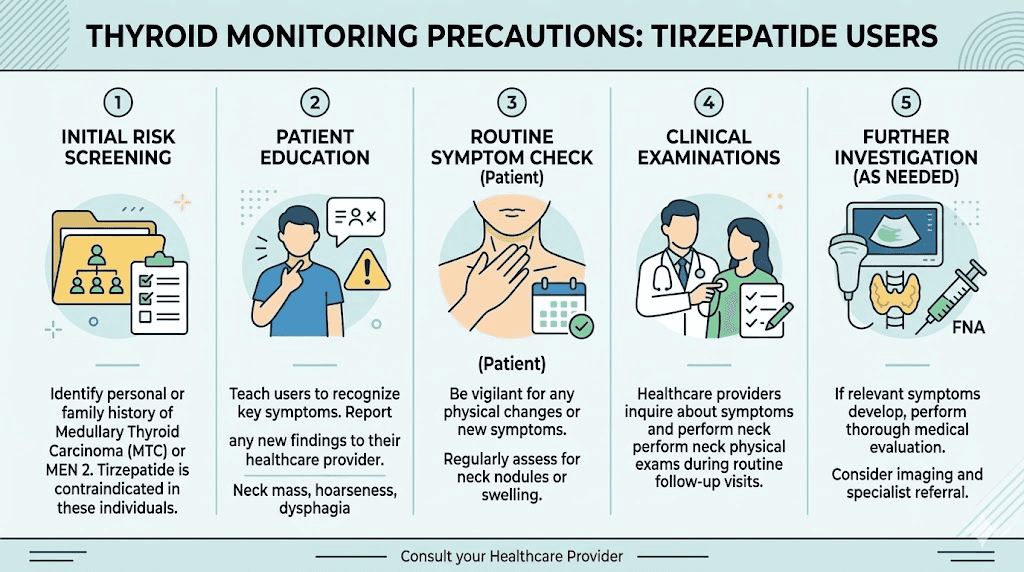
Routine calcitonin screening is not recommended
The American Diabetes Association and major endocrinology organizations do not recommend routine measurement of serum calcitonin levels before or during GLP-1 receptor agonist therapy in asymptomatic patients without specific risk factors. This recommendation may seem counterintuitive given the boxed warning, but it reflects several practical realities.
False-positive calcitonin results are common and lead to unnecessary anxiety, additional testing, and sometimes unnecessary surgery. The positive predictive value of calcitonin screening in the general population is low, meaning most elevated results do not indicate cancer. The clinical trial evidence does not support a meaningful risk of C-cell stimulation in humans taking tirzepatide. And universal screening would generate far more harm through false positives than benefit through early detection of an already rare cancer.
Calcitonin screening IS recommended in specific situations: when evaluating nodular thyroid disease (as part of the workup, not specifically because of tirzepatide), when a family history of MTC exists, and when MEN 2 is suspected. These are standard thyroid evaluation indications that apply regardless of whether someone is taking tirzepatide.
Thyroid ultrasound surveillance
Routine thyroid ultrasound surveillance is also not recommended for asymptomatic patients taking tirzepatide without specific risk factors. If you had a baseline thyroid ultrasound that showed benign-appearing nodules and you start tirzepatide, there is no guideline recommending surveillance ultrasounds specifically to monitor those nodules for tirzepatide-related changes. Your existing nodule surveillance schedule, based on standard American Thyroid Association guidelines for nodule management, should continue unchanged.
Symptoms that warrant evaluation
While routine screening is not recommended, certain symptoms should prompt medical evaluation regardless of whether you are taking tirzepatide. A new or growing lump in the neck could indicate nodule growth or a new nodule. Difficulty swallowing, called dysphagia, can suggest a nodule pressing on the esophagus. Persistent hoarseness not explained by a cold or allergy may indicate nerve involvement. Shortness of breath without obvious cause could suggest tracheal compression.
These symptoms warrant standard thyroid evaluation, including physical examination, ultrasound, and potentially biopsy. They are not specific to tirzepatide and would require the same workup in someone not taking any medication. The key point is that tirzepatide does not change the standard approach to thyroid symptom evaluation. You do not need special or accelerated monitoring simply because you are on a GLP-1 receptor agonist.
How tirzepatide interacts with thyroid function
Beyond the C-cell and cancer discussion, researchers often wonder whether tirzepatide affects overall thyroid function. The evidence here is nuanced and worth exploring in detail.
Thyroid hormone levels
Weight loss itself can affect thyroid hormone levels. When you lose significant weight, thyroid-stimulating hormone (TSH) and free T4 levels may shift as the body adjusts its metabolic setpoint. These changes are related to the weight loss rather than the medication causing it. Studies on tirzepatide efficacy show substantial weight reduction, up to 20% body weight in some trials, and changes of this magnitude naturally influence thyroid physiology.
Researchers taking tirzepatide who also have hypothyroidism or Hashimoto thyroiditis should be aware that thyroid hormone replacement doses may need adjustment as weight changes. This is not a safety concern specific to tirzepatide. It is a predictable metabolic consequence of significant weight loss that applies equally to weight lost through surgery, caloric restriction, or any other GLP-1 based approach.
Case reports of tirzepatide-associated thyroiditis
A published case report described a 32-year-old woman with no personal or family history of thyroid disease who developed painless biphasic thyroiditis after two months of tirzepatide therapy. This presentation involved initial thyrotoxicosis (excess thyroid hormone) followed by transient hypothyroidism (insufficient thyroid hormone), a pattern characteristic of destructive thyroiditis where inflammation damages thyroid tissue and releases stored hormones.
This case is noteworthy but represents a single report. A meta-analysis of 45 randomized controlled trials observed a trend toward increased risk of thyroid dysfunction in patients treated with GLP-1 receptor agonists, although the findings did not reach statistical significance. Trends that fail to reach significance in meta-analyses of 45 trials are, by definition, small effects that may or may not be real.
For practical purposes, researchers starting tirzepatide should be aware that thyroid function changes are possible but uncommon. Symptoms like unexpected fatigue, feeling cold, rapid heart rate, or unexplained weight changes beyond expected medication effects should prompt thyroid function testing.
Thyroid volume changes
Some research has examined whether GLP-1 receptor agonists affect thyroid gland volume. The limited available data suggest that thyroid volume is not significantly affected by GLP-1 receptor agonist therapy in humans at therapeutic doses. This is consistent with the low GLP-1 receptor expression on human thyroid cells compared to rodent thyroid cells. The medication simply does not interact with human thyroid tissue intensely enough to cause volumetric changes.
Comparing thyroid risk across GLP-1 receptor agonists
Tirzepatide is not the only medication carrying a thyroid C-cell tumor boxed warning. All GLP-1 receptor agonists share this warning, and understanding how tirzepatide compares to other medications in this class provides useful context.
Tirzepatide vs semaglutide thyroid safety
Both tirzepatide and semaglutide carry identical boxed warnings about thyroid C-cell tumors based on rodent studies. In FAERS post-marketing data, tirzepatide showed a lower thyroid cancer reporting odds ratio (2.09) compared to semaglutide (7.61). This does not necessarily mean tirzepatide is safer for the thyroid than semaglutide, as reporting biases, time on market, and user populations differ between the two medications.
Tirzepatide is a dual GLP-1/GIP receptor agonist, while semaglutide is a pure GLP-1 receptor agonist. The additional GIP receptor activity in tirzepatide does not appear to add thyroid risk, as GIP receptors are not significantly expressed on human thyroid C-cells. The dual mechanism provides metabolic advantages, including potentially greater weight loss and metabolic improvement, without adding thyroid-specific concerns.
Liraglutide and the broader GLP-1 class
Liraglutide, one of the earliest GLP-1 receptor agonists, has the longest track record for evaluating thyroid safety in humans. The EXSCEL trial, which followed exenatide (another GLP-1 agonist) users for approximately three years across roughly 83,000 patient-years of follow-up, found no evidence that treatment increases serum calcitonin concentrations or risk of MTC in humans.
Interestingly, liraglutide showed the highest FAERS reporting odds ratio for thyroid cancer (15.59) among GLP-1 agonists, yet long-term population studies have not confirmed increased thyroid cancer risk with liraglutide either. This discrepancy between adverse event reporting data and actual incidence data reinforces the limitations of FAERS as a signal detection tool rather than a risk quantification tool.
The consistency across the entire GLP-1 class is reassuring. No GLP-1 receptor agonist has demonstrated confirmed increased thyroid cancer risk in humans through controlled studies or large observational cohorts, despite the class carrying boxed warnings for over a decade.
Retatrutide and newer triple agonists
Newer medications like retatrutide, a triple GLP-1/GIP/glucagon receptor agonist, will likely carry similar thyroid warnings based on the same rodent C-cell tumor class effect. The retatrutide clinical development program will need to demonstrate comparable thyroid safety in humans. Researchers considering switching from tirzepatide to retatrutide should know that the thyroid risk profile is expected to be similar based on the shared GLP-1 receptor agonist mechanism.
Practical protocols for researchers with thyroid nodules
Moving from evidence review to practical application, here is exactly what researchers with thyroid nodules should consider before, during, and while maintaining tirzepatide therapy.
Before starting tirzepatide
Complete a thorough personal medical history review covering any prior thyroid diagnoses, thyroid surgeries, or thyroid biopsies. Document your family history with specific attention to medullary thyroid carcinoma in any first or second-degree relative, MEN 2 or related endocrine neoplasia syndromes, and any family member with pheochromocytoma (a tumor of the adrenal gland associated with MEN 2).
If you have known thyroid nodules, ensure they have been evaluated per standard American Thyroid Association guidelines. This typically means ultrasound characterization with TI-RADS scoring, fine needle aspiration if the nodule meets size and suspicion criteria, and baseline thyroid function tests including TSH. If your physician orders a baseline calcitonin level, a result under 10 pg/mL is considered normal. Levels between 10-50 pg/mL are borderline and may warrant repeat testing. Levels above 50 pg/mL require further investigation including additional imaging and potentially referral to endocrinology.
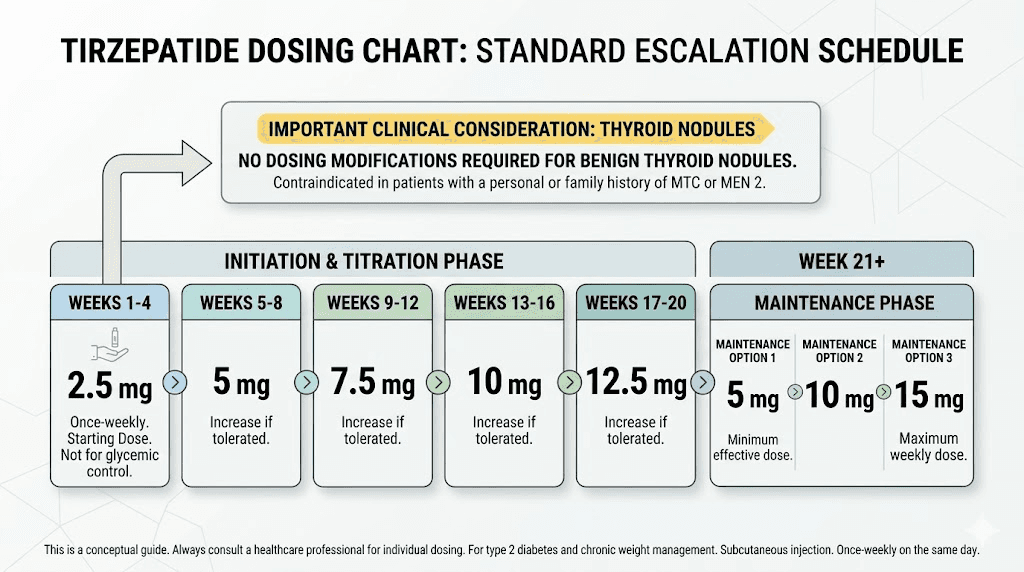
Once medullary thyroid carcinoma and MEN 2 have been excluded through history and appropriate testing, initiating tirzepatide follows standard protocols. The presence of benign thyroid nodules does not require dose modifications, delayed initiation, or special monitoring beyond what is already recommended for the nodules themselves.
During tirzepatide therapy
Continue any existing thyroid nodule surveillance schedule unchanged. If you were getting annual thyroid ultrasounds before starting tirzepatide, continue getting annual thyroid ultrasounds. If your nodules were deemed low-risk and follow-up was set for two years, maintain that schedule. Tirzepatide does not accelerate your surveillance timeline.
Monitor for new thyroid symptoms as described earlier: new neck lumps, swallowing difficulty, persistent hoarseness, or unexplained shortness of breath. Report these promptly to your healthcare provider. These symptoms warrant the same evaluation they would receive in someone not taking tirzepatide.
If you take thyroid hormone replacement medication (levothyroxine/Synthroid), expect potential dose adjustments as you lose weight on tirzepatide. Weight loss changes the body distribution of thyroid medication and may require dosing adjustments. Get thyroid function checked 6-8 weeks after significant weight changes and whenever symptoms suggest thyroid levels may be off.
Long-term maintenance considerations
For researchers maintaining tirzepatide therapy long-term, the question of cumulative thyroid risk naturally arises. Can years of GLP-1 receptor stimulation eventually cause the same C-cell problems seen in two-year rodent studies? The honest answer is that decades-long human data do not yet exist for tirzepatide specifically. However, liraglutide has been available since 2010, providing over 15 years of real-world use across millions of patients without confirmed increased MTC signal in humans.
The biological rationale for low human risk remains strong regardless of treatment duration. Human C-cells have low GLP-1 receptor expression. The proliferative pathway active in rodent C-cells does not appear to be meaningfully activated in human C-cells. And the population-level data consistently show no signal. Long-term tirzepatide users should maintain standard thyroid health practices: regular physical exams that include neck palpation, prompt evaluation of any new thyroid symptoms, and continued participation in any existing nodule surveillance programs.

Common concerns and misconceptions
Misinformation about tirzepatide and thyroid safety circulates widely. Addressing the most common misconceptions directly helps researchers make evidence-based decisions rather than fear-based ones.
Misconception: tirzepatide causes thyroid nodules
There is no evidence that tirzepatide causes thyroid nodules to form. Clinical trials did not show increased nodule incidence. The boxed warning concerns C-cell tumors (medullary thyroid carcinoma), not nodule formation. Thyroid nodules are overwhelmingly common in the general population regardless of medication use. Discovering a nodule while taking tirzepatide is almost certainly coincidental, not causal, particularly given that thyroid ultrasound has become more common in recent years, leading to more incidental nodule detection in all populations.
Misconception: the boxed warning means tirzepatide is dangerous for your thyroid
A boxed warning is the most serious warning the FDA can place on a medication. It does not mean the medication causes the warned condition in humans. It means that either human evidence confirms serious risk OR that animal data suggests potential serious risk warranting caution until human data accumulates. For GLP-1 receptor agonists and thyroid C-cell tumors, the warning falls into the second category. Rodent data triggered the warning. Human data has consistently failed to confirm the risk.
Many commonly used medications carry boxed warnings for conditions they rarely or never cause in practice. The presence of a boxed warning should prompt careful evaluation and discussion with your physician, not automatic avoidance of a medication that may provide substantial metabolic benefits.
Misconception: if you have any thyroid problem you cannot take tirzepatide
This is perhaps the most damaging misconception. Hypothyroidism, Hashimoto thyroiditis, Graves disease, benign thyroid nodules, and history of papillary or follicular thyroid cancer are NOT contraindications to tirzepatide. The only thyroid-related contraindications are personal or family history of medullary thyroid carcinoma and MEN 2. Everything else is manageable with standard thyroid care.
Researchers with hypothyroidism frequently use tirzepatide successfully. Their thyroid medication doses may need adjustment as weight changes, but the medications are compatible. SeekPeptides members frequently discuss managing thyroid conditions alongside tirzepatide protocols, and the community provides valuable real-world perspectives on navigating both conditions simultaneously.
Misconception: you need special thyroid tests before starting tirzepatide
No special thyroid tests are required beyond standard medical assessment. Your physician should take a thyroid-focused personal and family history. If that history is unremarkable and you have no palpable thyroid abnormalities, standard tirzepatide initiation can proceed without additional thyroid testing. Routine calcitonin screening is specifically NOT recommended by major endocrinology organizations for asymptomatic patients starting GLP-1 receptor agonists.
If you do have known thyroid nodules, they should be evaluated per standard guidelines, meaning the evaluation should follow thyroid nodule management recommendations, not special GLP-1-related protocols. The evaluation exists to characterize the nodule, not to clear you for tirzepatide specifically.
Tirzepatide dosing considerations for people with thyroid conditions
The standard tirzepatide dosing schedule does not change based on thyroid nodule status. However, certain thyroid conditions may warrant practical adjustments to optimize outcomes.
Standard dosing protocol
Tirzepatide is initiated at 2.5 mg weekly for four weeks, then increased to 5 mg weekly. Further dose escalation occurs in 2.5 mg increments at minimum four-week intervals up to a maximum of 15 mg weekly. This titration schedule is the same regardless of thyroid nodule status. There is no evidence suggesting that slower titration, lower maximum doses, or modified schedules are needed for patients with benign thyroid nodules.
For researchers using compounded tirzepatide, the same principles apply. Dosing is based on metabolic goals and tolerance, not thyroid nodule status. Calculate your exact dose using the appropriate conversion resources and follow standard injection techniques.
Managing thyroid medication alongside tirzepatide
If you take levothyroxine for hypothyroidism, take it on an empty stomach at least 30-60 minutes before other medications or food. Tirzepatide is administered weekly, so timing conflicts are minimal, but be aware that tirzepatide slows gastric emptying, which could theoretically affect absorption of oral medications taken around the same time. Separating levothyroxine administration from tirzepatide injection by several hours eliminates any theoretical absorption concern.
As you lose weight on tirzepatide, your levothyroxine dose may need reduction. Weight loss decreases the body mass that determines thyroid hormone requirements. Monitor for symptoms of over-replacement, such as anxiety, rapid heart rate, insomnia, or unintentional weight loss beyond expected tirzepatide effects. Get TSH checked after significant weight milestones, typically every 10-15 pounds lost.
Microdosing approaches
Some researchers with multiple medical conditions, including thyroid disorders, prefer starting with microdosing tirzepatide to minimize side effects and assess tolerance before standard dosing. A microdose schedule might start at 1 mg or even lower, gradually building to the standard starting dose. This approach is based on individual tolerance preference, not thyroid safety data, as there is no evidence that lower doses carry different thyroid risk profiles than standard doses.
Microdosing for inflammation has also gained attention among researchers interested in GLP-1 agonist effects beyond weight loss, including potential anti-inflammatory benefits for autoimmune conditions that may include autoimmune thyroiditis.
Side effects to watch that may overlap with thyroid symptoms
One practical challenge for tirzepatide users with thyroid conditions is distinguishing between tirzepatide side effects and thyroid-related symptoms. Several symptoms overlap, and knowing which is which prevents unnecessary alarm or, conversely, missing an important thyroid change.
Fatigue
Fatigue is common during tirzepatide therapy, particularly during dose escalation and periods of rapid weight loss. It is also a cardinal symptom of hypothyroidism. If you develop new or worsening fatigue on tirzepatide and you have a thyroid condition, get your TSH checked before attributing it solely to the medication. If TSH is normal, the fatigue is likely tirzepatide-related and typically improves as your body adjusts to the current dose.
Feeling cold
Feeling cold on tirzepatide is widely reported and relates to reduced caloric intake, lower body fat insulation, and metabolic changes from weight loss. Cold intolerance is also a classic hypothyroidism symptom. Again, thyroid function testing can distinguish between the two causes, though in practice, many tirzepatide users with normal thyroid function still report feeling cold as they lose weight.
Hair thinning
Rapid weight loss from any cause can trigger telogen effluvium, a form of temporary hair shedding. GLP-1 related hair changes follow this pattern and typically resolve within 6-12 months. Thyroid disorders can also cause hair changes, making it important to verify thyroid function is optimal if hair thinning occurs. Adequate protein intake, which can be challenging on tirzepatide due to appetite suppression, is essential for preventing nutrition-related hair loss.
Gastrointestinal symptoms
Nausea, constipation, and diarrhea are the most common tirzepatide side effects and do not indicate thyroid problems. However, severe gastrointestinal symptoms can impair absorption of thyroid medications, making it important to ensure levothyroxine is taken at a time when nausea is minimal and absorption is not compromised by vomiting.
Neck discomfort
Tirzepatide does not cause neck pain or neck lumps. Any new neck lump, pain, or swelling while taking tirzepatide should be evaluated by your physician. Body aches and joint pain have been reported with tirzepatide use, but these affect muscles and joints, not the thyroid gland. True thyroid-related neck symptoms feel different, typically a painless lump, difficulty swallowing, or a sensation of pressure at the base of the throat.
What the experts say about moving forward
The clinical consensus on tirzepatide and thyroid nodules has evolved as evidence accumulates. Leading endocrinology organizations and thyroid cancer experts have weighed in with increasingly clear guidance.
A perspective published in the Journal of Clinical Endocrinology and Metabolism titled "Approach to the Patient With Thyroid Nodules: Considering GLP-1 Receptor Agonists" concluded that GLP-1 receptor agonist therapy should not be withheld from patients with benign thyroid nodules. The article emphasized the importance of standard thyroid nodule risk stratification and surveillance according to established American Thyroid Association guidelines, independent of GLP-1 receptor agonist therapy decisions.
The Endocrine Society has noted that the rodent C-cell tumor findings, while appropriately triggering regulatory caution, have not translated into demonstrated human risk across the now extensive clinical trial and post-marketing experience with GLP-1 receptor agonists as a class.
A research presentation titled "Much Ado About Nothing: Tirzepatide and Medullary Thyroid Cancer Debunked," presented at the Endocrine Society annual meeting, directly addressed the disconnect between the boxed warning and clinical evidence, arguing that the accumulating data strongly suggest the warning will eventually be revised as human evidence continues to show no signal.
SeekPeptides members access comprehensive protocol guidance that incorporates the latest evidence on tirzepatide safety, including thyroid considerations.
The platform provides evidence-based guides, detailed protocols, and a community of experienced researchers who help navigate exactly these kinds of nuanced safety questions.
The role of genetic testing
For researchers with thyroid nodules who want maximum reassurance before starting tirzepatide, genetic testing offers a definitive answer about MEN 2 risk, the primary genetic condition that contraindicates GLP-1 receptor agonist use.
RET proto-oncogene testing
MEN 2 is caused by germline mutations in the RET proto-oncogene. Genetic testing for RET mutations is highly accurate and widely available. A negative RET test essentially eliminates the MEN 2 contraindication, providing definitive clearance from the genetic risk perspective. Testing is particularly valuable for individuals with family histories that are incomplete or uncertain, those with thyroid nodules that have any features suggestive of MTC on biopsy, and anyone with personal or family history of pheochromocytoma or hyperparathyroidism (the other components of MEN 2).
When genetic testing is unnecessary
Most people do not need RET genetic testing before starting tirzepatide. If you have no family history of MTC, no family history of MEN 2 features, and your thyroid nodule has been evaluated as benign through standard workup, genetic testing adds cost without meaningful clinical value. The decision to test should be made in consultation with your physician based on your specific family history and nodule characteristics.
Tirzepatide benefits that may support thyroid health
While this article focuses primarily on thyroid safety concerns, it is worth noting that some tirzepatide effects may indirectly benefit thyroid health.
Weight loss and thyroid function
Obesity is associated with mild TSH elevation and alterations in thyroid hormone metabolism. Significant weight loss with tirzepatide can normalize these obesity-related thyroid changes. Some researchers with borderline hypothyroidism find that substantial weight loss reduces or eliminates the need for thyroid medication, though this should always be monitored with laboratory testing rather than self-adjusted.
Anti-inflammatory effects
GLP-1 receptor agonists demonstrate anti-inflammatory properties that extend beyond metabolic effects. For researchers with Hashimoto thyroiditis, the autoimmune condition causing most cases of hypothyroidism, the anti-inflammatory effects of GLP-1 agonists could theoretically reduce thyroid autoimmune activity. Preliminary research in this area is encouraging, though not yet sufficient to recommend GLP-1 agonists specifically for thyroid autoimmunity management.
Metabolic improvement
Better metabolic control through improved insulin sensitivity, reduced inflammation, and weight normalization creates a healthier physiological environment for all organ systems, including the thyroid. Improved metabolism supports optimal thyroid function rather than threatening it.
Frequently asked questions
Can I take tirzepatide if I have a benign thyroid nodule?
Yes. Benign thyroid nodules are not a contraindication to tirzepatide use. The FDA boxed warning concerns medullary thyroid carcinoma, a specific rare cancer type, not benign nodules. If your nodule has been evaluated and determined to be benign through standard workup, you can proceed with tirzepatide therapy following normal protocols.
Does tirzepatide cause thyroid nodules to grow?
There is no clinical evidence that tirzepatide causes thyroid nodules to form or grow. Clinical trials involving over 10,000 participants did not show increased nodule incidence. Continue your existing nodule surveillance schedule as recommended by your endocrinologist, regardless of tirzepatide use.
Should I get a thyroid ultrasound before starting tirzepatide?
A thyroid ultrasound is not routinely required before starting tirzepatide unless your physician identifies a palpable thyroid abnormality or your medical history suggests specific risk factors. If you already have known nodules, ensure they have been appropriately evaluated per standard thyroid guidelines, but this evaluation is for nodule management, not tirzepatide clearance specifically.
What thyroid conditions absolutely prevent tirzepatide use?
Only two thyroid-related conditions are absolute contraindications: personal or family history of medullary thyroid carcinoma (MTC) and Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). All other thyroid conditions, including hypothyroidism, Hashimoto thyroiditis, Graves disease, benign nodules, and history of papillary or follicular thyroid cancer, are NOT contraindications.
Will tirzepatide interfere with my thyroid medication?
Tirzepatide slows gastric emptying, which could theoretically affect absorption of oral medications like levothyroxine. Separating levothyroxine administration from tirzepatide by several hours minimizes any potential interaction. As you lose weight on tirzepatide, your levothyroxine dose may need reduction, so monitor thyroid function regularly during significant weight loss.
Is tirzepatide safer for the thyroid than semaglutide?
Both medications carry identical FDA boxed warnings based on rodent C-cell tumor data. Post-marketing reporting data shows a lower thyroid cancer reporting odds ratio for tirzepatide (2.09) compared to semaglutide (7.61), but reporting differences and time on market make direct comparison unreliable. Neither medication has demonstrated confirmed thyroid cancer risk in controlled human studies.
How often should I check my thyroid while on tirzepatide?
Routine thyroid monitoring specifically for tirzepatide is not recommended for patients without thyroid conditions. If you have hypothyroidism and take thyroid medication, check TSH every 6-8 weeks during periods of significant weight loss and adjust medication accordingly. If you have known thyroid nodules, continue your existing surveillance schedule unchanged.
Can tirzepatide affect TSH levels?
Weight loss itself can affect TSH levels. Significant weight reduction may lower TSH in patients with obesity-related TSH elevation. For patients on thyroid hormone replacement, weight loss may cause TSH to drop below target, indicating over-replacement that requires dose reduction. These are weight loss effects, not direct tirzepatide effects on thyroid function.
External resources
Approach to the Patient With Thyroid Nodules: Considering GLP-1 Receptor Agonists (JCEM)
Tirzepatide and Cancer Risk: Systematic Review and Meta-Analysis
In case I do not see you, good afternoon, good evening, and good night. May your thyroid stay healthy, your nodules stay benign, and your protocols stay evidence-based.