Feb 23, 2026

You just realized what you did. The injection is already in. And now you are staring at the vial, reading the date, doing the math in your head. That date passed. Maybe a week ago. Maybe a month. Your stomach drops, and not from the semaglutide.
Take a breath.
This is one of the most common medication concerns people face, and the answer is far less scary than you think. A single dose of expired semaglutide is extremely unlikely to cause you serious harm. The medication does not suddenly become toxic the day after its expiration date. It does not transform into something dangerous. What happens is much less dramatic, but still worth understanding fully so you know exactly where you stand.
Expired semaglutide primarily loses potency. The active peptide degrades gradually over time, which means you likely received a weaker dose than intended rather than a harmful one. That said, there are legitimate concerns worth addressing, from contamination risks to what this means for your weight loss progress and dosing schedule. Depending on how far past the expiration date the semaglutide has gone, and how it was stored before you used it, the risks range from negligible to worth monitoring closely.
This guide covers everything you need to know right now. What actually happens inside an expired vial at the molecular level. The specific symptoms to watch for in the hours and days ahead. A clear step-by-step action plan you can follow immediately. When to contact your healthcare provider versus when to simply move forward with your next scheduled dose. Whether you are using compounded semaglutide or a brand-name formulation like Ozempic or Wegovy, the information here applies to your situation. SeekPeptides has compiled the most current research and clinical guidance to help you navigate this with confidence.
What actually happens when you take expired semaglutide
The short answer is this: probably nothing alarming. But the complete answer requires understanding what expiration actually means for a peptide medication, and why it matters differently than it does for something like aspirin sitting in your medicine cabinet.
Semaglutide is a GLP-1 receptor agonist, a synthetic peptide that mimics a natural hormone your body produces to regulate blood sugar and appetite. Like all peptides, it has a specific molecular structure that must remain intact for the medication to work properly. When semaglutide expires, that structure begins to break down through chemical processes called deamidation and oxidation. The amino acid chains that make the drug effective slowly fragment and lose their three-dimensional shape.
Here is what that means in practical terms.
The peptide becomes less effective. Not more dangerous. Research on peptide degradation shows that semaglutide breaks down into inactive peptide fragments rather than harmful compounds. The tryptophan residue at position 25 is particularly vulnerable to oxidation, and this is one of the first degradation pathways that reduces potency. But these breakdown products are generally recognized as biologically inactive, meaning they neither help nor harm you in meaningful ways.
Think of it this way. A fully potent dose of semaglutide at 0.5mg delivers 0.5mg of active medication. An expired dose might deliver 0.3mg, or 0.2mg, or even less depending on how degraded the peptide has become. You are not getting a full therapeutic dose, but you are also not getting something that will hurt you. The gap between "less effective" and "dangerous" is enormous for this class of medication.
For people using semaglutide for weight management, a single weak dose means you might experience less appetite suppression than usual. You might feel hungrier that week. Your appetite signals might return to baseline faster. But these are temporary setbacks, not health emergencies.
For those using it primarily for blood sugar management, the concern is slightly different. A degraded dose may not control glucose levels as effectively, which could lead to temporary blood sugar elevation. This is worth monitoring, especially for people with type 2 diabetes who depend on consistent medication strength. We will cover exactly what blood glucose levels to watch for and when to seek help in the sections ahead.
One important distinction: expired semaglutide that was stored properly (refrigerated, protected from light, not frozen) degrades much more slowly than expired semaglutide that sat on a counter in direct sunlight for weeks. Storage history matters as much as the date on the label. If you accidentally left semaglutide out overnight before it expired, the effective potency may have already dropped below what the expiration date suggests. Conversely, a well-stored vial that expired last week likely retains most of its strength.
The bottom line: you almost certainly received a subtherapeutic dose rather than a harmful one. Your next step is understanding what to watch for and when to reach for a fresh vial.
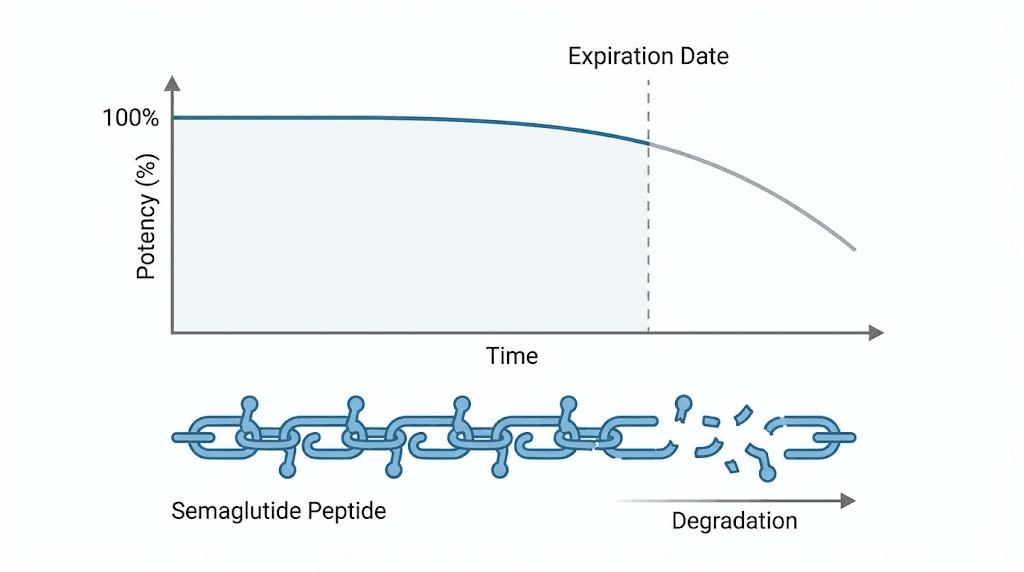
Why semaglutide expires in the first place
Every medication has a shelf life, but peptides like semaglutide are especially sensitive to the passage of time. Understanding why helps explain what you actually injected and what risks, if any, you face now.
Semaglutide is a 31-amino-acid peptide with a fatty acid chain attached at one end. That fatty acid chain (a C-18 fatty di-acid, specifically) is what gives semaglutide its long half-life in the body, allowing for once-weekly dosing. But this same complex molecular architecture also makes it vulnerable to environmental degradation.
The chemistry of degradation
Three main processes break down semaglutide over time.
Oxidation is the primary degradation pathway. The tryptophan amino acid at position 25 in the peptide chain is highly susceptible to oxidative stress. When oxygen interacts with this residue, it produces single and double oxidation products that reduce the peptide ability to bind to GLP-1 receptors. Without proper receptor binding, the medication cannot trigger the signaling cascade that suppresses appetite and regulates blood sugar. Research published in analytical chemistry journals confirms that oxidation at tryptophan dominates the degradation profile under stress conditions.
The second process is hydrolysis, where water molecules break peptide bonds between amino acids. This literally chops the semaglutide molecule into shorter fragments. Under thermal stress conditions, bonds between specific amino acids break, creating truncated versions of semaglutide that cannot function as the full-length peptide does. Think of it like cutting a key into pieces. Each piece is harmless metal, but none of them will open the lock anymore.
Aggregation is the third mechanism. Over time, individual semaglutide molecules can clump together, forming larger structures that the body cannot use effectively. Aggregated peptides may also trigger immune responses at the injection site, though this is more of a theoretical concern than a commonly reported clinical issue.
What accelerates these processes
Temperature is the biggest factor. Semaglutide stored at proper refrigeration temperatures (36 to 46 degrees Fahrenheit, or 2 to 8 degrees Celsius) degrades slowly and predictably. The same medication stored at room temperature degrades significantly faster. And semaglutide left at elevated temperatures can degrade in a fraction of the labeled time.
Light exposure accelerates oxidation. UV light directly attacks the tryptophan residue, speeding up the very degradation pathway that most reduces potency. This is why proper storage guidance says to keep semaglutide in its original packaging and away from direct light.
Freezing, counterintuitively, also damages semaglutide. Ice crystal formation can disrupt the peptide three-dimensional structure, causing irreversible damage that no amount of thawing will fix. If your semaglutide was accidentally frozen at any point before you used it, the effective potency may be lower than the label suggests regardless of the expiration date.
Repeated temperature cycling, such as moving a vial in and out of the refrigerator frequently, subjects the peptide to thermal stress that accelerates all three degradation mechanisms. Each cycle chips away at stability. When you are traveling with semaglutide, keeping temperature consistent matters enormously.
The role of preservatives
Both brand-name and compounded semaglutide contain preservatives to maintain sterility and slow degradation. Brand formulations like Ozempic contain phenol as a preservative, while compounded versions may use benzyl alcohol or other antimicrobial agents. These preservatives do two things: they prevent bacterial growth in the solution and they help buffer the pH to slow chemical degradation.
But preservatives have their own shelf life. Over time, they lose effectiveness. This is one of the key reasons expiration dates exist, not just because the active ingredient degrades, but because the protective systems that maintain the solution sterility and stability eventually fail as well. After the preservative loses enough potency, the solution becomes vulnerable to microbial contamination, a risk that compounds with each passing day beyond expiration.
The real risks of expired semaglutide
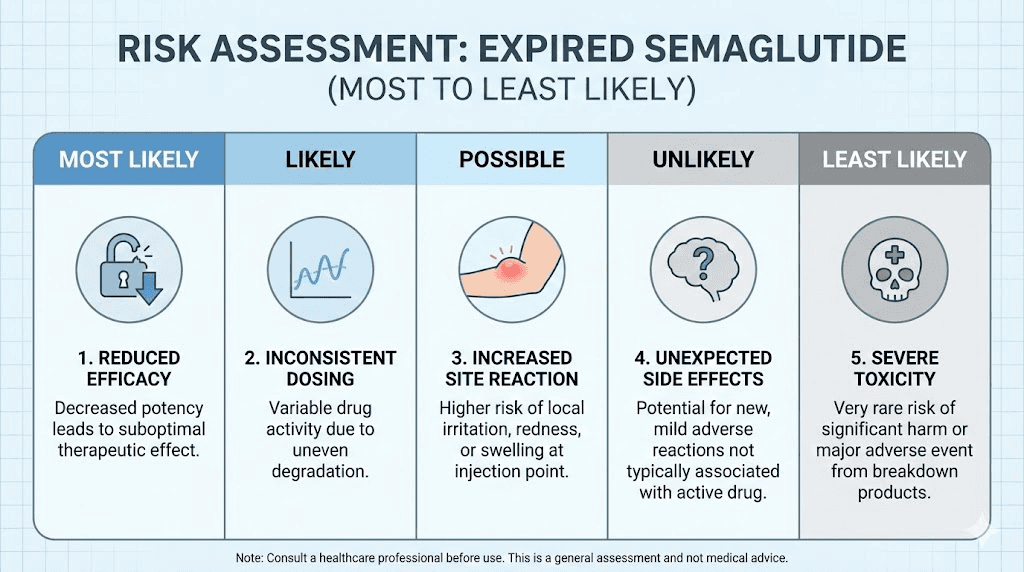
Now that you understand what happens chemically, here are the actual risks ranked from most likely to least likely. Not all expired semaglutide carries the same risk, and your specific situation depends on several factors.
Risk 1: Reduced therapeutic effect (very likely)
This is the primary and most common consequence. Your dose did not work as well as a fresh one would have. For weight loss, this means potentially less appetite suppression for that dosing period. You might feel hungrier than usual, experience stronger food cravings, or notice that the feeling of fullness after meals does not last as long.
A single subtherapeutic dose is not going to derail weeks or months of progress. Your body does not reset because of one weak injection. The medication residual effects from previous doses are still partially active in your system, thanks to semaglutide long 168-hour half-life. So even if this dose was significantly degraded, you still have active semaglutide circulating from your last proper dose.
That said, if you have been taking expired semaglutide for multiple doses without realizing it, the cumulative effect of reduced potency could explain a weight loss plateau or lack of expected results. Some people who report that semaglutide stopped working may actually be dealing with degraded medication rather than true treatment resistance.
Risk 2: Blood sugar fluctuations (moderate concern for diabetic patients)
For people using semaglutide to manage type 2 diabetes, a degraded dose may not control blood glucose as effectively. This could result in temporary hyperglycemia, elevated blood sugar that may cause increased thirst, more frequent urination, fatigue, and blurred vision.
Healthy individuals or those using semaglutide solely for weight management face a lower risk here because their bodies can typically regulate blood sugar through other mechanisms. But for anyone on a comprehensive diabetes management protocol, one ineffective dose can disrupt glucose control for days.
Monitor your blood glucose more frequently after taking an expired dose. If readings stay consistently above 200 mg/dL or spike above 300 mg/dL, contact your healthcare provider. These thresholds warrant professional guidance, though they are not typically emergency-level for a single occurrence in otherwise stable patients.
Risk 3: Injection site reactions (low to moderate)
Degraded peptide fragments and aggregated proteins can sometimes trigger localized immune responses at the injection site. This might appear as increased redness, swelling, itching, or warmth around where you injected. These reactions are usually mild and resolve on their own within a few days.
If you notice unusual injection site reactions that go beyond what you normally experience, keep the area clean and monitor it. Signs of actual infection, such as spreading redness, increasing warmth, pus, or red streaks extending from the injection site, are different from a simple immune response and require medical attention. Choosing the proper injection site and maintaining clean injection technique reduces these risks significantly.
Risk 4: Microbial contamination (low but serious if present)
This is the risk that genuinely warrants caution, though it is statistically unlikely from a single dose. After expiration, the preservative systems in the solution may be less effective at preventing bacterial growth. Research on multi-dose medication vials found bacterial contamination rates of approximately 5.5% in opened vials, with organisms like Pseudomonas aeruginosa and Klebsiella pneumoniae identified as the most common contaminants.
The risk increases if the vial has been opened and used multiple times, stored improperly, handled with contaminated needles, or kept well past its expiration or beyond-use date. A sealed, refrigerated vial that expired one week ago carries minimal contamination risk. An opened, room-temperature vial that expired two months ago carries considerably more.
Contamination would manifest as injection site infection (redness, swelling, warmth, pain, pus), fever, chills, or in rare cases, systemic infection. If you notice any signs of infection developing in the 24 to 72 hours following your injection, seek medical care promptly. Early treatment of injection site infections is straightforward, but delayed treatment can lead to complications.
Risk 5: Allergic or immune response (rare)
Degradation products, particularly aggregated peptides, could theoretically trigger an allergic or immune response that the intact medication would not cause. This is more of a theoretical pharmacological concern than a commonly observed clinical phenomenon. If you notice any unusual allergic symptoms such as widespread rash, hives, difficulty breathing, or swelling of the face or throat, seek emergency medical care regardless of the cause.
Brand name vs compounded semaglutide expiration
Not all semaglutide is created equal when it comes to shelf life. The type of semaglutide you used, brand-name or compounded, significantly affects how quickly it degrades and what risks you face from an expired dose.
Brand-name semaglutide (Ozempic, Wegovy, Rybelsus)
Brand-name injectable semaglutide manufactured by Novo Nordisk comes in pre-filled pens with extensive stability testing behind the expiration dates. Ozempic and Wegovy pens have specific shelf lives:
Unopened and refrigerated: Up to 24 to 36 months from the manufacturing date, depending on the formulation. This is based on rigorous stability testing that demonstrates the peptide retains its specified potency throughout this period when stored at 2 to 8 degrees Celsius.
After first use: Ozempic pens can be stored at room temperature (up to 86 degrees Fahrenheit or 30 degrees Celsius) or refrigerated for 56 days. After 56 days from first use, the pen should be discarded regardless of the printed expiration date. This 56-day window accounts for the degradation that occurs once the pen seal is broken and the medication is exposed to repeated temperature changes from handling.
Rybelsus, the oral form of semaglutide, has different storage requirements because it is a tablet rather than an injectable solution. Oral semaglutide should be stored at room temperature (68 to 77 degrees Fahrenheit) and kept in its original blister packaging until use.
If you accidentally used an expired Ozempic or Wegovy pen, the good news is that these products have conservative expiration dates backed by extensive stability data. A pen that expired a few days or even a couple of weeks ago likely retains a significant portion of its potency, assuming proper storage.
Compounded semaglutide
This is where expiration dates become more complicated and more important to respect.
Compounded semaglutide is prepared by compounding pharmacies using semaglutide base powder, bacteriostatic water, and various excipients. Because each pharmacy uses different formulations, equipment, and processes, the stability of compounded products varies significantly.
Compounded semaglutide typically carries a beyond-use date (BUD) of 14 to 120 days, with most pharmacies assigning 28 to 90 days. The variation depends on whether the pharmacy has conducted its own stability testing or relies on default USP guidelines. Pharmacies like Empower Pharmacy and Olympia Pharmacy typically provide BUDs based on their internal stability data.
Once opened, compounded semaglutide vials are generally recommended for use within 28 days when refrigerated. This is notably shorter than the 56-day window for brand-name pens, largely because compounded products may have different preservative concentrations and are drawn from vials with rubber stoppers (which means a needle puncture creates a potential contamination entry point with each use).
If you accidentally used expired compounded semaglutide, the risk profile is slightly different from brand-name. The potency loss follows similar degradation pathways, but the contamination risk may be higher because compounding environments and preservative systems vary. A compounded vial that has been punctured multiple times and stored past its BUD carries more risk than a sealed, expired brand-name pen.
Understanding the reconstitution process for compounded semaglutide helps illustrate why these products are more sensitive. The mixing of bacteriostatic water with semaglutide powder creates a solution that starts its degradation clock from the moment of reconstitution, not from the date the powder was manufactured.
Key differences at a glance
Factor | Brand-name (Ozempic/Wegovy) | Compounded semaglutide |
|---|---|---|
Unopened shelf life | 24-36 months refrigerated | Varies by pharmacy (30-120 days typical) |
After opening | 56 days | 28 days (most pharmacies) |
Stability testing | Extensive FDA-required testing | Varies by pharmacy |
Preservative system | Standardized (phenol) | Varies (benzyl alcohol, others) |
Contamination risk post-expiry | Lower (sealed pen system) | Higher (vial with repeated punctures) |
Potency loss rate post-expiry | Gradual, well-characterized | Less predictable, depends on formulation |
Symptoms to monitor after taking expired semaglutide
You have taken the expired dose. Now what should you actually watch for? The symptoms fall into several categories, ranging from signs that the medication simply did not work as well (expected and manageable) to signs that warrant medical attention (uncommon but important to recognize).
Signs the dose was less potent (normal, expected)
These indicate the semaglutide was degraded and you received a subtherapeutic dose. They are not dangerous but tell you the medication did not fully do its job.
Increased appetite sooner than usual. If you typically experience strong appetite suppression for 5 to 6 days after your injection and it fades by day 3 or 4 this time, the dose was likely weaker than normal.
Less nausea than your typical dose. This sounds like a good thing, and in isolation it is. But if you usually experience mild nausea after injection (especially at higher doses) and felt nothing this time, it may indicate reduced potency rather than adaptation.
Stronger food cravings. The GLP-1 pathway that semaglutide activates plays a direct role in food reward signaling. A weaker dose means less suppression of these reward signals, which can manifest as stronger cravings for specific foods, particularly calorie-dense ones.
Temporary weight fluctuation. A single subtherapeutic dose is unlikely to cause meaningful weight gain. But you might see a slight uptick on the scale from increased food intake or water retention. This is temporary and will correct once you resume with a properly potent dose. Understanding your expected month-to-month results helps put a single weak dose in perspective.
Signs of blood sugar disruption (monitor if diabetic)
These symptoms matter primarily for people managing type 2 diabetes. If you use semaglutide solely for weight management and do not have diabetes, these are less likely to be relevant, though they are still worth knowing.
Increased thirst. This is one of the earliest signs that blood glucose levels have risen above the normal range. If you find yourself unusually thirsty in the 24 to 48 hours after your injection, check your blood sugar if you have a glucose monitor available.
Frequent urination. Elevated blood glucose causes the kidneys to work harder to filter the excess sugar, pulling more water from the body in the process. This goes hand in hand with increased thirst.
Fatigue and brain fog. Fatigue on semaglutide is common even with proper doses, but unusually severe tiredness after an expired dose could indicate blood sugar levels are not being managed effectively.
Blurred vision. High blood sugar can temporarily affect the lens of the eye, causing vision changes. This symptom warrants prompt blood glucose testing and a call to your healthcare provider if it persists.
Signs of injection site infection (uncommon, watch for 72 hours)
Contamination from expired medication is uncommon with a single dose but worth monitoring. These symptoms typically develop 24 to 72 hours after injection.
Spreading redness around the injection site. Some redness immediately after injection is normal. Redness that spreads, intensifies, or develops a warm-to-touch quality over the following days is different.
Pus or discharge. Any fluid draining from the injection site is a clear sign of infection and requires medical evaluation.
Increasing pain or swelling. Mild tenderness at the injection site is common. Pain that worsens over days rather than improving is a warning sign.
Red streaks extending from the injection site. This indicates the infection may be spreading to surrounding tissue and warrants urgent medical attention.
Fever or chills. Systemic symptoms suggest the infection has moved beyond the local injection site. Seek medical care promptly if you develop fever following an injection with expired medication.
Signs of allergic reaction (rare, seek immediate help)
This is extremely uncommon from an expired dose, but these symptoms always require emergency care regardless of cause.
Widespread rash or hives. Swelling of the face, lips, tongue, or throat. Difficulty breathing or swallowing. Rapid heartbeat with dizziness. If you experience any of these symptoms, call emergency services immediately. Do not wait to see if they improve on their own.

What to do right now: your step-by-step action plan
You have the knowledge. Now here is the actionable plan. Follow these steps in order, and you will handle this situation exactly as a healthcare professional would advise.
Step 1: Check exactly how expired the semaglutide was
Find the vial or pen and read the expiration date or beyond-use date carefully. Calculate exactly how many days or weeks past expiration you are. This matters because a dose that expired 3 days ago is a fundamentally different situation from one that expired 3 months ago.
For brand-name pens (Ozempic, Wegovy): Look for the printed expiration date on the pen label. Also consider when you first used the pen, since the 56-day in-use window may have passed even if the printed date has not.
For compounded semaglutide: Look for the beyond-use date (BUD) assigned by your compounding pharmacy. Also check when the vial was first punctured if that date is noted. Many pharmacies from Empower to BPI Labs to Direct Meds label their products with clear BUD information.
Step 2: Assess storage history
Think back to how the medication was stored. Was it refrigerated consistently? Did it ever sit out at room temperature for an extended period? Was it ever left out overnight? Did it experience any temperature extremes (frozen, left in a hot car, exposed to direct sunlight)?
A well-stored vial that expired recently carries minimal risk. A poorly stored vial that expired months ago carries considerably more risk. Storage conditions determine the actual rate of degradation far more than the calendar date alone.
Step 3: Inspect the medication
Check the solution itself for visual signs of degradation.
Color changes: Semaglutide solution should be clear and colorless. Knowing what normal semaglutide looks like helps you spot problems. Any yellowing, cloudiness, or discoloration suggests significant degradation or contamination.
Particles: Hold the vial up to a light source and look for floating particles, fibers, or crystals. These indicate that the peptide has aggregated or that foreign matter has contaminated the solution.
Cloudiness: Clear solution is good. Hazy or turbid solution is a warning sign.
If the solution looks visually abnormal, the risk of having used it is higher than if it appeared perfectly clear. Note your observations in case your healthcare provider asks about them later.
Step 4: Monitor your symptoms
For the next 72 hours, pay attention to your body more carefully than usual. Keep track of:
Your appetite patterns compared to a normal dosing week. Any injection site changes (redness, warmth, swelling, pain). How you feel overall (energy, nausea, digestive function). Blood glucose levels if you have a monitor and use semaglutide for diabetes management.
You do not need to obsessively check for problems. Simply maintain a higher than usual awareness. Most people who accidentally take a single expired dose notice nothing at all, or notice only that their appetite suppression was weaker than expected.
Step 5: Dispose of the expired medication
Do not use the rest of the expired vial or pen. Mark it clearly as expired and dispose of it according to your pharmacy guidelines. Many pharmacies accept returned medications for safe disposal.
Step 6: Get a replacement
Contact your prescribing provider or pharmacy to obtain a fresh supply. Explain that your current medication has expired and you need a replacement. Most providers can send a new prescription quickly, especially for ongoing maintenance medications like semaglutide.
While waiting for your replacement, understand that missing one week of semaglutide is not catastrophic for your overall progress. The medication half-life of approximately 168 hours means that active semaglutide from your previous proper doses is still circulating in your system, providing some residual benefit. Semaglutide withdrawal symptoms typically do not begin with a single missed or reduced dose.
Step 7: Resume your normal schedule
Once you have a fresh supply, resume your regular dosing schedule at the same dose you were on before. Do not double the dose to "make up" for the expired injection. That is not how this medication works, and doubling up increases the risk of gastrointestinal side effects including nausea, vomiting, and dizziness.
When to call your doctor (and when to go to the ER)
Most people who accidentally take expired semaglutide will not need medical intervention. But there are specific scenarios where professional guidance is warranted, and a smaller set of situations that require urgent care.
Situations that do not require a doctor call
You took a dose that expired within the last 1 to 2 weeks and had been properly stored. The solution appeared clear and normal. You feel fine or just notice slightly less appetite suppression than usual. Your blood glucose levels (if monitored) remain in your normal range. The injection site looks normal or has only the mild redness you typically experience.
In these cases, simply dispose of the expired medication, obtain a replacement, and move on with your next dose at the scheduled time.
Situations that warrant a call to your doctor
The medication was significantly past its expiration date (more than 30 days). The solution looked abnormal (discolored, cloudy, particles). You have type 2 diabetes and your blood glucose is running higher than usual. You are experiencing gastrointestinal symptoms that are unusual for you or more severe than your typical post-injection response. The injection site is showing increasing redness, warmth, or swelling 24 or more hours after injection. You are pregnant or think you may be pregnant. You have compromised immunity or are on immunosuppressive medications.
Your healthcare provider can assess whether any monitoring, testing, or treatment adjustments are needed. In most cases, the advice will be to discard the expired medication and resume with a fresh supply, but professional reassurance is valuable when concern is warranted.
Situations that require emergency care
Blood glucose readings consistently above 300 mg/dL despite management efforts. Signs of diabetic ketoacidosis: fruity-smelling breath, deep or rapid breathing, nausea with vomiting, abdominal pain, confusion. Signs of severe allergic reaction: throat swelling, difficulty breathing, widespread hives, rapid heartbeat. Signs of spreading infection: fever above 101 degrees Fahrenheit, red streaks extending from the injection site, pus, rapidly expanding redness, severe pain at the site. Severe and persistent vomiting that prevents keeping any fluids down.
These emergency situations are extremely rare from a single expired dose, but they are included here so you can recognize them if they occur. When in doubt, calling your provider or a nurse hotline is always appropriate.
How expired is too expired: the degradation timeline
The degree of risk scales with time. Not all expired semaglutide is equally degraded, and understanding the timeline helps you assess your specific situation more accurately.
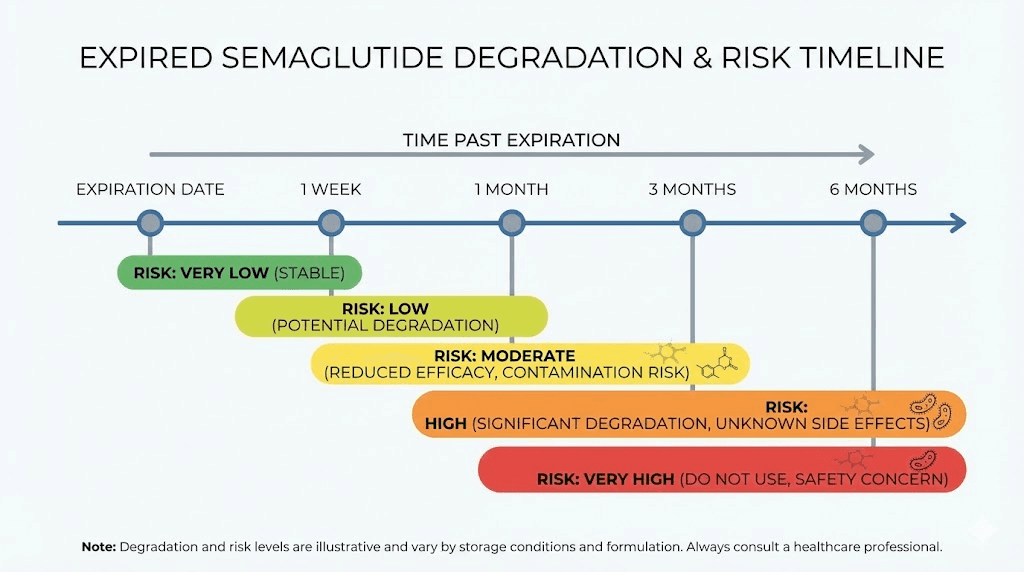
1 to 7 days past expiration
This is the lowest risk window. Expiration dates include built-in safety margins, meaning the medication was likely still effective on the day it "expired" and retains most of its potency for a short period afterward. FDA stability testing ensures potency through the labeled date, but degradation does not suddenly accelerate the next day.
Brand-name semaglutide that expired within the past week and was properly refrigerated likely retains 90% or more of its labeled potency. Compounded semaglutide in this window is also likely to be close to its intended strength, assuming proper storage.
Your practical risk: Minimal. You probably received close to a full therapeutic dose.
1 to 4 weeks past expiration
Degradation becomes more noticeable in this range. The medication is still unlikely to be harmful, but potency has dropped measurably. Depending on storage conditions, you may have received 60 to 85% of the intended dose.
The FDA Shelf Life Extension Program (SLEP), which tests stockpiled medications for the Department of Defense, has found that 88% of 3,005 drug lots tested retained their potency past expiration, with average extensions of 5.5 years. However, peptide medications like semaglutide were not specifically included in these broad findings, and peptides are generally more sensitive to degradation than small-molecule drugs.
Your practical risk: Low. Reduced effectiveness is likely, but safety concerns remain minimal if storage was proper.
1 to 3 months past expiration
Now we enter territory where potency loss becomes significant and other risks begin to compound. The peptide structure has had enough time for meaningful oxidation, hydrolysis, and potential aggregation. The preservative system has further weakened, especially if the vial has been opened and punctured multiple times.
You may have received 30 to 60% of the intended dose, or less if storage was suboptimal. The contamination risk is moderately elevated. Understanding how long semaglutide remains good and what the 28-day window means helps frame why this timeframe matters.
Your practical risk: Moderate. Contact your healthcare provider if you have diabetes or notice any unusual symptoms. Monitor your injection site carefully for 72 hours.
3 to 6 months past expiration
This is the range where using the medication is generally inadvisable and where accidental use warrants more careful monitoring. Potency may have dropped to 20% or less of the labeled amount. The preservative system may be largely ineffective. The risk of contamination is meaningfully elevated, especially for opened, multi-use vials.
Your practical risk: Moderate to high. Monitor carefully. Contact your healthcare provider to discuss the situation and get guidance specific to your health profile.
Beyond 6 months past expiration
At this point, the semaglutide is effectively a different product from what was originally dispensed. Significant degradation has occurred, preservative protection is essentially gone, and the risk-benefit calculus has shifted entirely to the risk side. There is very little chance of therapeutic benefit and a meaningful chance of contamination.
Your practical risk: High. Contact your healthcare provider. Monitor closely for any signs of infection or adverse reaction. Consider whether any visual abnormalities in the solution were present when you used it.
Storage mistakes that accelerate expiration
Understanding what speeds up degradation helps you assess the actual condition of the expired semaglutide you used. A well-stored medication that is slightly past its date is vastly different from a poorly stored medication that is slightly past its date.
Temperature abuse
This is the number one factor. Semaglutide belongs in the refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Every hour spent at room temperature accelerates degradation. Extended exposure to temperatures above 77 degrees Fahrenheit dramatically speeds up oxidation and hydrolysis.
If your semaglutide spent significant time at elevated temperatures before you used it past its expiration, the actual potency was likely lower than what the expiration date alone would suggest. A vial that spent its life properly refrigerated may actually be more potent past its expiration than a vial stored at room temperature that technically has not expired yet.
Light exposure
UV light directly targets the tryptophan residue in the semaglutide molecule, accelerating the primary oxidative degradation pathway. Semaglutide stored on a counter near a window, in a bathroom with fluorescent lighting, or anywhere with direct light exposure degrades faster than protected medication.
The original packaging exists for a reason. It blocks light. If your semaglutide was stored outside its packaging in a well-lit area, factor that into your assessment of how degraded it might have been.
Freezing
Freezing semaglutide causes ice crystal formation that physically disrupts the peptide three-dimensional structure. Unlike temperature-driven chemical degradation, freeze damage is immediate and irreversible. A vial that froze once, even briefly, may have lost significant potency regardless of what the expiration date says.
This is particularly relevant for people who store semaglutide in the back of a refrigerator where temperatures can drop below freezing, or for those who travel with semaglutide and encounter freezing conditions during transport. Proper refrigerator storage means middle shelf, not the back wall where ice can form.
Repeated handling
Every time you remove a vial from the refrigerator, draw medication, and return it, the temperature cycling introduces stress. For compounded semaglutide drawn from multi-dose vials, each needle puncture also introduces a microscopic pathway for potential contamination. The more times a vial has been punctured, the higher the cumulative contamination risk, especially past the beyond-use date.
This is one reason why compounded semaglutide vials have shorter use windows than sealed brand-name pens. The delivery mechanism itself affects how quickly the product degrades and how much contamination risk accumulates over time.
Contamination during use
Using unclean needles, touching the rubber stopper of a vial with ungloved hands, drawing medication in a dusty or unclean environment, or reusing syringes all introduce contaminants that can grow once the preservative weakens. Good injection practice matters not just for the current dose, but for maintaining the integrity of the remaining medication in the vial.
Preventing this from happening again
Once is an accident. Twice is a pattern. Here is how to make sure this does not happen again.
Create a labeling system
When you receive a new vial or pen of semaglutide, immediately write two dates on it with a permanent marker: the expiration or beyond-use date (already printed, but make it more visible), and the date you first opened or used it. For Ozempic pens, add 56 days to the open date. For compounded semaglutide, add 28 days (or whatever your pharmacy specifies).
Keep a small piece of tape on the vial or pen with the "discard by" date clearly visible. This eliminates the need to read small print or do mental math when you are half-awake on injection day.
Set calendar reminders
When you start a new vial or pen, set a reminder on your phone for one week before the discard date. This gives you time to order a replacement without the stress of running out. Many people using semaglutide on a regular schedule find that syncing their medication calendar with their injection day reminders creates a reliable system.
Store medications correctly from day one
Designate a specific spot in your refrigerator for your semaglutide. Not the back wall (freezing risk), not the door (temperature fluctuation from opening), and not next to anything that could freeze or leak onto it. A small dedicated container on a middle shelf works well.
Keep the medication in its original packaging to protect it from light. Never leave it out at room temperature longer than necessary (brief periods for injection preparation are fine). Consistent refrigeration is the single most important factor in maximizing the useful life of your medication.
Maintain a medication log
Track your injections in a simple log that includes the date, time, dose, and remaining quantity. This makes it immediately obvious when you are running low and need to reorder. It also helps you catch situations where a vial should have been used up by now but still has medication remaining, which might indicate you have been underdosing. The semaglutide dosage calculator on SeekPeptides can help you understand exactly how many doses remain in your vial based on your current protocol.
Order refills proactively
Do not wait until you are on your last dose to order more. Build a buffer of 1 to 2 weeks into your ordering schedule. This accounts for shipping delays, pharmacy processing time, insurance issues, and the inevitable moments where life gets in the way. Running out and then using expired medication because the fresh supply has not arrived yet is one of the most common ways people end up in this situation.
What the FDA and research say about expired medications
The broader context of medication expiration dates adds useful perspective to your situation, even though peptide medications have specific considerations that set them apart from many other drugs.
The FDA position
The FDA officially recommends against using medications past their labeled expiration dates. Their position is clear: potency and safety cannot be guaranteed once the expiration date has passed. This is a legally and medically conservative stance designed to protect consumers in all possible scenarios.
However, the FDA also operates the Shelf Life Extension Program (SLEP) in partnership with the Department of Defense. This program tests stockpiled medications for stability well beyond their labeled expiration dates. The results are instructive.
The SLEP findings
Testing of 3,005 lots across 122 different drug products found that 88% of tested medications retained their potency past expiration. The average extension granted was 5.5 years beyond the original expiration date. Some medications demonstrated stability for over 20 years past their printed dates.
These findings suggest that expiration dates for many medications are conservative. Drug manufacturers set dates based on the period for which they have stability data (typically 2 to 3 years of testing), not based on the point where medications actually become unsafe. The result is that many medications are discarded while still fully effective.
But peptides are different
Here is the important caveat. The SLEP findings apply primarily to small-molecule drugs (tablets, capsules) stored in ideal conditions. Peptide medications like semaglutide are fundamentally different for several reasons.
Peptides are large, complex molecules with three-dimensional structures that are essential for function. Unlike a simple chemical compound like acetaminophen that either has the molecule or does not, a peptide can be partially intact but functionally impaired.
Peptides in solution degrade faster than dry tablets. The aqueous environment promotes hydrolysis and facilitates oxidation in ways that solid-state medications avoid.
Peptides require specific storage conditions (refrigeration, light protection) that are harder to maintain perfectly over extended periods. A bottle of ibuprofen in a medicine cabinet is in a relatively stable environment. A vial of semaglutide in a refrigerator is subject to temperature fluctuations every time the door opens.
So while the SLEP data is reassuring for medication stability in general, it should not be directly extrapolated to injectable peptide medications. Your expired semaglutide may retain some potency past its date, but the rate and degree of degradation are less predictable than for a standard oral medication.
What medical professionals actually advise
Most healthcare providers take a pragmatic approach. A single dose of recently expired, properly stored semaglutide is generally not considered a medical concern. They will advise you to obtain a fresh supply and resume your normal schedule. Multiple doses of significantly expired medication warrant closer discussion and potential monitoring, especially for diabetic patients.
The consensus among endocrinologists and obesity medicine specialists is that the biggest risk from expired semaglutide is therapeutic failure (the medication not working), not toxicity (the medication causing harm). This distinction is important and should provide considerable reassurance if you are worried right now.
Common myths about expired semaglutide
Misinformation about expired medications circulates widely online. Here is what is actually true and what is not.
Myth: Expired semaglutide becomes toxic
Reality: Semaglutide does not become toxic after its expiration date. The peptide breaks down into inactive fragments through oxidation, hydrolysis, and aggregation. These fragments are not harmful, they are simply not therapeutically active. This is different from a very small number of medications (most notably tetracycline antibiotics in older formulations) that can theoretically produce harmful degradation products. Semaglutide is not in that category.
Myth: One expired dose will ruin your progress
Reality: Semaglutide has a half-life of approximately 168 hours (7 days). This means that at any given time, you have active medication in your system from multiple previous doses. One subtherapeutic dose does not erase the drug circulating in your bloodstream from prior injections. Your overall results depend on consistent use over weeks and months, not on any single injection.
Myth: If it looks fine, it is fine
Reality: Visual inspection is a useful but incomplete assessment tool. Semaglutide can lose significant potency while still appearing perfectly clear and colorless. Chemical degradation at the molecular level is invisible to the naked eye until it reaches an advanced stage. Cloudiness, discoloration, or visible particles indicate severe degradation, but the absence of these signs does not guarantee full potency.
Think of it like checking if milk has gone bad by looking at it. If it looks green and chunky, it is definitely bad. But if it looks normal, it might still be slightly turned. Visual inspection catches the worst cases, not the borderline ones.
Myth: Refrigeration prevents all degradation
Reality: Refrigeration dramatically slows degradation, but it does not stop it entirely. The chemical processes that break down semaglutide (oxidation, hydrolysis) are temperature-dependent but not temperature-eliminated. Even at perfect storage temperatures, semaglutide gradually loses potency over time, which is precisely why it has an expiration date in the first place.
Proper storage extends the usable life of the medication and ensures it remains potent through the labeled date. It does not make the medication immortal.
Myth: All expired medications are equally risky
Reality: The risk profile varies enormously by medication type, formulation, and storage history. An expired aspirin tablet is a very different situation from an expired injectable peptide. Among injectable peptides, a sealed brand-name pen that expired last week is a very different situation from an opened compounded vial that expired two months ago. Generalizations about "expired medications" are not particularly useful for evaluating your specific situation.
Myth: You should take a double dose to compensate
Reality: Never double your semaglutide dose without medical guidance. Taking too much semaglutide significantly increases the risk of severe nausea, vomiting, diarrhea, and other gastrointestinal side effects. The correct approach is to simply resume your normal dose with a fresh supply at your next scheduled injection.

Getting back on track after an expired dose
The practical question remains: how do you get back to your normal semaglutide routine as quickly and effectively as possible?
When to take your next dose
Resume your normal dosing schedule with a fresh, non-expired supply. If your regular injection day is Wednesday and you accidentally took an expired dose on Wednesday, your next dose should be the following Wednesday with proper medication. Do not try to "make up" for the expired dose by injecting sooner or at a higher amount.
If your next scheduled dose is several days away and you are concerned about the gap in effective treatment, contact your prescribing provider. They may advise you to take your next fresh dose slightly early or to maintain your regular schedule. This decision depends on your specific clinical situation and the dose you are currently on.
Will this affect your long-term progress?
One expired dose will not meaningfully impact your long-term results. If you have been consistently using semaglutide for weeks or months, the progress you have made is built on a foundation of many effective doses. One weak injection does not undo that work.
You may notice a temporary dip in appetite suppression or a slight increase on the scale over the next few days. This is temporary. Once you resume with a properly potent dose, your body will respond as it normally does. Many people who experience a plateau on semaglutide find that the issue resolves once they ensure their medication is properly potent and stored.
If you were unknowingly using expired medication for multiple weeks
This is a different scenario that deserves specific attention. If you have been using the same expired vial for several weeks, you have been receiving progressively weaker doses over that entire period. This could explain:
Gradual return of appetite and food cravings. Stalled weight loss despite maintaining your protocol. Feeling like the medication "stopped working." A general sense that semaglutide is less effective than it used to be.
The solution is straightforward. Obtain a fresh, properly dated supply and resume your protocol. You do not need to restart at a lower dose or re-titrate. Simply continue at your current prescribed dose with the new medication. Most people notice a return to normal effectiveness within 1 to 2 weeks of switching to fresh medication.
Supporting your results during the gap
While waiting for your replacement medication or during the period when the expired dose is less effective than usual, you can support your progress by focusing on the lifestyle factors that complement semaglutide treatment.
Prioritize protein intake to support satiety. Stick to your semaglutide diet plan even if appetite suppression is reduced. Focus on avoiding trigger foods that tend to drive overeating. Stay hydrated and maintain regular physical activity. Lean on the food strategies that work well with GLP-1 medications to bridge the gap.
These strategies are not a replacement for properly potent medication, but they help minimize any setback from a single expired dose while you wait for your fresh supply.
Understanding expiration across different GLP-1 medications
If you use or have considered other GLP-1 medications alongside or instead of semaglutide, understanding their expiration profiles provides useful context.
Tirzepatide, the dual GIP/GLP-1 agonist sold as Mounjaro and Zepbound, has similar storage requirements and expiration concerns. Like semaglutide, tirzepatide expires through peptide degradation, and proper refrigeration is essential. Compounded tirzepatide follows the same beyond-use date principles as compounded semaglutide, with refrigerated shelf life typically limited to 28 days after first use.
The comparison between semaglutide and tirzepatide extends to their degradation profiles. Both are peptide-based medications vulnerable to the same types of chemical breakdown. If you are considering switching between them, the conversion chart can help you understand equivalent dosing, while articles on switching between these medications cover the practical considerations.
Oral GLP-1 formulations like oral semaglutide drops and GLP-1 oral liquids have their own stability considerations. Oral formulations face additional challenges from gastric acid and digestive enzymes, but their storage and expiration profiles differ from injectable forms because the delivery mechanism is fundamentally different.
Newer medications in the pipeline like retatrutide, the triple-agonist GLP-1/GIP/glucagon receptor agonist, will face the same peptide stability challenges. The principles you learn about semaglutide expiration apply broadly to this entire class of medications.
Companion supplements and expired semaglutide
Some people use companion supplements alongside their semaglutide to address common side effects or enhance results. If you took an expired dose, certain supplements may help bridge the gap in effectiveness.
Vitamin B12 is one of the most commonly paired supplements with semaglutide. GLP-1 medications can affect B12 absorption over time, and some compounded formulations include methylcobalamin directly in the solution. If you have been relying on B12 included in your compounded semaglutide and that medication is expired, the B12 component may also be degraded.
Glycine is another supplement frequently combined with semaglutide in compounded formulations. Glycine serves as a stabilizer in some formulations and may provide additional metabolic benefits. Like the active peptide, glycine in solution can degrade over time, though it is generally more stable than the semaglutide molecule itself.
If you normally take supplements alongside your GLP-1 medication, continue taking them as scheduled regardless of whether your most recent semaglutide dose was expired. These supplements support your overall health independently of the semaglutide potency.
Special considerations for specific populations
Certain groups face unique considerations when dealing with expired semaglutide exposure.
People with type 2 diabetes
If you rely on semaglutide as part of your blood glucose management strategy, an expired dose that fails to control blood sugar effectively is more clinically significant than for someone using it solely for weight management. Monitor your blood glucose more frequently for the next 48 to 72 hours. Have a plan for managing any spikes with your existing diabetes management tools (other medications, dietary adjustments, physical activity).
Contact your endocrinologist or primary care provider if blood glucose readings are consistently elevated above your target range following the expired dose. They may temporarily adjust your other medications to compensate until you resume treatment with a fresh semaglutide supply.
People on higher doses
If you are at the maximum semaglutide dose, you have less margin for reduced potency. A degraded dose at 2.4mg that delivers only 60% effective medication still provides roughly 1.44mg of active semaglutide, which is above many starting and maintenance doses. But if you are on a higher dose because lower doses were insufficient for your goals, that reduction in effective dose may be more noticeable.
People who are pregnant or could be pregnant
Semaglutide should not be used during pregnancy. If you accidentally used expired semaglutide and are pregnant or suspect you might be pregnant, contact your healthcare provider immediately. The concern is not about the medication being expired specifically, but about any semaglutide exposure during pregnancy.
People with compromised immune systems
If you have a compromised immune system due to medication or medical conditions, the contamination risk from expired medication is more significant for you than for someone with normal immune function. Your body may be less able to handle any bacterial contamination that might be present in an expired solution. Monitor for signs of infection more carefully, and have a lower threshold for contacting your healthcare provider.
People who combine semaglutide with other medications
If you take semaglutide alongside other medications like phentermine or insulin, a subtherapeutic semaglutide dose can disrupt the balance of your overall medication regimen. The other medications remain at full strength while the semaglutide is weakened, which could produce unexpected interactions or effects. Discuss with your provider if you are on a complex multi-medication protocol.
The bigger picture: medication management and GLP-1 therapy
This incident is an opportunity to strengthen your overall approach to medication management. People who use semaglutide and other GLP-1 medications for weight management or diabetes treatment are typically on long-term therapy, which means medication management becomes a recurring part of life.
Establishing reliable systems now prevents future problems. The few minutes it takes to label your medication, set a calendar reminder, and organize your storage pays dividends over months and years of treatment. It also removes the anxiety of wondering whether your medication is still good every time you reach for it.
SeekPeptides members access comprehensive medication management guides, including storage protocols, reconstitution calculators, and dosage tools that help take the guesswork out of peptide management. Having reliable resources for calculating doses, understanding unit-to-milligram conversions, and tracking your protocol reduces the chance of medication errors of all kinds.
For researchers serious about optimizing their GLP-1 protocols safely and effectively, SeekPeptides provides the most thorough resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
Frequently asked questions
Can expired semaglutide make me sick?
A single dose of recently expired, properly stored semaglutide is extremely unlikely to make you sick. The primary risk is reduced effectiveness rather than toxicity. The peptide degrades into inactive fragments, not harmful compounds. If the medication was significantly expired or improperly stored, there is a small risk of infection from potential contamination, which would present as injection site redness, warmth, or fever in the 24 to 72 hours after injection.
How do I know if my semaglutide has expired?
Check the expiration date (brand-name) or beyond-use date (compounded) printed on the label. For opened brand-name pens, count 56 days from the date you first used the pen. For opened compounded semaglutide vials, count 28 days from the first puncture (or whatever your pharmacy specifies). Also inspect the solution visually for any color changes, cloudiness, or particles.
Should I take an extra dose to make up for the expired one?
No. Never double your semaglutide dose. Taking too much semaglutide dramatically increases the risk of severe nausea, vomiting, and other gastrointestinal side effects. Simply resume your normal dose with fresh medication at your next scheduled time.
I have been using the same vial for months. Could all those doses have been expired?
Yes, if you exceeded the beyond-use date or expiration date of your vial. Compounded semaglutide is typically good for 28 days after opening. If you have been drawing from the same vial for 2 to 3 months, you have likely been receiving increasingly degraded doses. This could explain any perceived plateau in your results. Check your dates, discard the old vial, and start fresh.
Does expired semaglutide lose all of its potency at once?
No. Degradation is gradual and continuous. Semaglutide does not go from 100% potent to 0% potent on the expiration date. Potency declines slowly over time, with the rate depending on storage conditions. A well-stored vial that expired a week ago may still retain 90% of its potency, while a poorly stored vial that expired two months ago may have lost most of its effectiveness.
Can I test if my semaglutide is still good?
There is no at-home test for semaglutide potency. You can visually inspect for discoloration, cloudiness, or particles, which indicate severe degradation. But the absence of visible problems does not guarantee full potency. The only reliable way to know is to use medication within its labeled expiration or beyond-use date.
Is expired compounded semaglutide riskier than expired brand-name?
Generally, yes. Compounded semaglutide may have different preservative concentrations, shorter stability windows, and variable quality depending on the compounding pharmacy. Brand-name products undergo rigorous FDA-mandated stability testing. Additionally, compounded vials drawn with needles have more contamination risk than sealed pre-filled pens.
What if I used expired semaglutide and now feel fine?
That is the most common outcome. Most people who accidentally use a single expired dose experience nothing adverse. If you feel fine, your blood sugar is normal (if you monitor it), and your injection site looks normal, you can simply discard the expired medication and resume your protocol with a fresh supply.
External resources
PubMed: Stability Profiles of Drug Products Extended Beyond Labeled Expiration Dates
Biosynth: Pharmacological Implications of Semaglutide Impurities
In case I do not see you, good afternoon, good evening, and good night. May your medications stay potent, your storage stay cold, and your next dose stay on schedule.