Feb 22, 2026

What if the reason your semaglutide is not working has nothing to do with dosing, timing, or your body? What if it is sitting in your fridge right now, slowly losing potency because you did not know when the clock actually started ticking?
It happens more than most people realize. A researcher pulls out a vial, draws the dose, injects as planned. The protocol looks perfect on paper. But the semaglutide inside that vial crossed its usable window three days ago, and nobody mentioned that the rules for vials are completely different from the rules for pre-filled pens.
The short answer is that most semaglutide vials remain good in the fridge for 28 to 90 days, depending on whether they are opened or unopened, brand-name or compounded, and how the compounding pharmacy determined the beyond-use date. But the real answer requires understanding why those numbers vary so dramatically, what actually happens to semaglutide molecules as they degrade, and how your specific storage habits either protect or destroy every dose remaining in that vial. This guide covers the complete picture, from compounded semaglutide vial protocols to brand-name pen comparisons, so you never waste another milligram of this peptide.
SeekPeptides members consistently report that understanding storage fundamentals transformed their results more than any dosing change ever did. That statement might sound dramatic until you realize that a 10-20% potency drop from poor storage compounds over weeks into results that look like the peptide simply stopped working.
Understanding semaglutide vial types
Before diving into shelf life numbers, you need to understand which type of semaglutide vial you actually have. This matters because the storage rules, expiration timelines, and handling protocols differ significantly between formats. Getting this wrong is one of the most common peptide mistakes beginners make, and it leads to wasted product and inconsistent results.
Pre-filled pens vs multi-dose vials
Brand-name semaglutide comes in two primary forms. Ozempic and Wegovy ship as pre-filled injection pens with fixed concentrations and built-in dose selectors. These pens contain enough semaglutide for multiple weekly injections, and the manufacturer has conducted extensive stability testing to determine exactly how long each pen remains viable.
Multi-dose vials are different. They contain a larger volume of semaglutide solution that you draw from using an insulin syringe. Each time you puncture the rubber stopper, you introduce a tiny amount of environmental exposure. This is why vials have different shelf life rules than sealed pens.
The concentration matters too. A 5mg semaglutide vial reconstituted in 2ml of bacteriostatic water gives you a different concentration than a 10mg vial in the same volume. Both concentrations follow the same general storage rules, but the number of doses you can draw before reaching the expiration window varies based on your dosage in units.
Compounded semaglutide vials explained
Most researchers using multi-dose vials are working with compounded semaglutide. These vials come from compounding pharmacies that prepare semaglutide solutions according to individual prescriptions or research needs. The compounded semaglutide landscape includes a range of formulations, each with its own stability profile.
Some compounding pharmacies use semaglutide base. Others use semaglutide sodium salt. The formulation type affects how long the solution remains stable because each form has slightly different molecular behavior in solution. Your pharmacy should specify which form they used and provide clear storage instructions specific to their formulation.
Common compounded vial sizes include 2.5mg, 5mg, and 10mg concentrations. Popular pharmacies like Empower Pharmacy, Olympia Pharmacy, and Direct Meds each have their own stability testing data and beyond-use date assignments. Do not assume that one pharmacy vial follows the same timeline as another.
How long an unopened semaglutide vial lasts in the fridge
An unopened vial is one that has never been punctured. The rubber stopper remains intact, no needle has entered, and the sterile environment inside remains undisturbed. This matters enormously for shelf life because the moment you pierce that stopper, a completely different set of rules applies.
Brand-name unopened storage
Unopened Ozempic pens stored in the refrigerator at 36-46 degrees Fahrenheit (2-8 degrees Celsius) remain good until the expiration date printed on the packaging. This expiration date typically falls 18-24 months from the manufacturing date because Novo Nordisk has conducted rigorous long-term stability testing on the formulation.
Wegovy follows similar rules for unopened storage. Keep it refrigerated at the same 36-46 degree range, and it stays viable until the printed expiration date. The key difference between these two products lies in what happens after you start using them, which we will cover in the next section.
For anyone wondering whether GLP-1 is the same as Ozempic, the answer involves understanding that semaglutide is the active ingredient in both brand names. The storage rules apply to the semaglutide molecule itself, regardless of which brand label sits on the box.
Compounded semaglutide unopened vials
Here is where things get complicated. Compounded semaglutide vials do not carry manufacturer expiration dates. Instead, they carry a beyond-use date, often abbreviated as BUD. This date is determined by the compounding pharmacy, not by the FDA, and it varies significantly from one pharmacy to the next.
Typical beyond-use dates for unopened compounded semaglutide vials stored in the refrigerator range from 30 to 90 days from the compounding date. Some pharmacies with extensive stability testing data assign BUDs of up to 120 days. Others, following more conservative USP guidelines, may assign BUDs as short as 14-28 days.
The variation comes down to stability testing rigor. Pharmacies that invest in comprehensive analytical testing of their specific formulations can demonstrate longer shelf life with confidence. Pharmacies that rely on default USP Chapter 797 guidelines without product-specific testing must use shorter, more conservative dates.
Always check the label on your vial. The BUD should be clearly printed. If it is not, contact your pharmacy immediately. Using a vial past its BUD means you are working with a product that has no guaranteed potency or sterility.
The beyond-use date explained
Think of the beyond-use date as a different concept from an expiration date. An expiration date on FDA-approved medications comes from years of accelerated and real-time stability studies conducted under strict regulatory oversight. The manufacturer tests thousands of units across multiple batches, tracking potency, purity, and sterility over extended periods.
A beyond-use date is more conservative by design. USP Chapter 797 sets the framework for how compounding pharmacies determine these dates. Without product-specific stability data, the default BUD for sterile compounded preparations stored in the refrigerator is relatively short. Pharmacies that conduct their own stability studies can extend these dates based on their findings.
This does not mean compounded semaglutide is inferior. It means the testing framework is different. Many compounding pharmacies produce high-quality semaglutide solutions that remain perfectly stable well past their BUD. But because you cannot independently verify potency at home, respecting the BUD protects your investment and your results.
For researchers who also work with other peptides, the concept of peptide expiration follows similar principles. All reconstituted peptide solutions have a finite window of reliable potency, and understanding that window is essential for peptide safety.
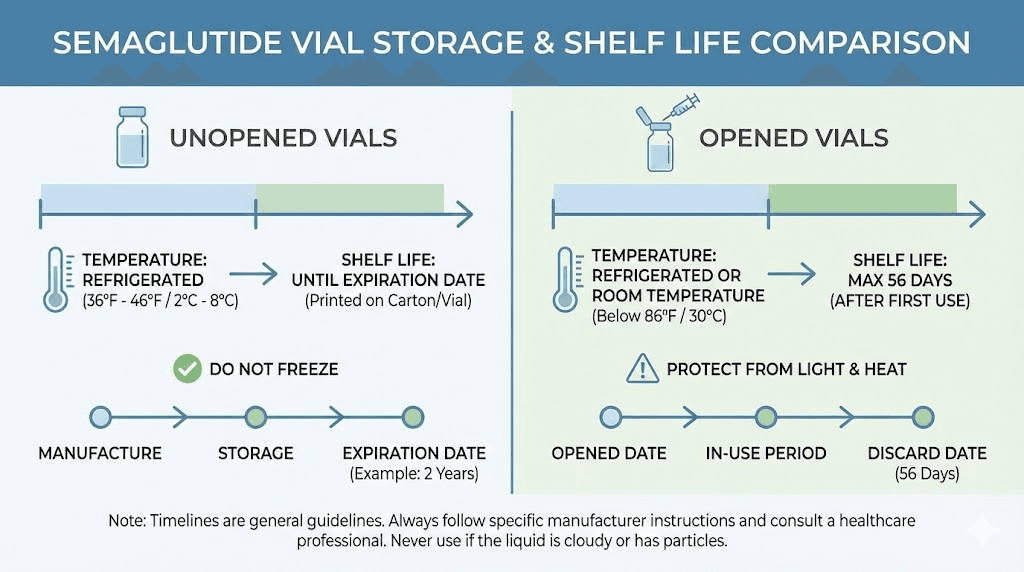
How long an opened semaglutide vial lasts in the fridge
Once you puncture that rubber stopper with a needle, the countdown accelerates. An opened vial is no longer a sealed system. Every subsequent needle insertion introduces microscopic amounts of air, potential contaminants, and small disruptions to the sterile barrier. The shelf life after opening is universally shorter than before opening, and this is where most confusion and most waste occurs.
Brand-name semaglutide after first use
Ozempic pens last 56 days after first use. That is eight weeks. You can store the pen in the refrigerator or at room temperature (59-86 degrees Fahrenheit) during this period. After 56 days, discard the pen even if solution remains inside.
Wegovy pens have a shorter post-opening window of 28 days. Same storage options, fridge or room temperature, but the usable period is exactly half of Ozempic. This difference exists because the two products have different concentrations, delivery mechanisms, and formulation details that affect post-opening stability.
If you are comparing these timelines to what you have read about semaglutide expiring after 28 days, the confusion likely stems from mixing up Ozempic and Wegovy guidelines. The 28-day figure applies specifically to Wegovy pens, not to all semaglutide products universally.
Compounded vials after first puncture
Most compounding pharmacies recommend using their semaglutide vials within 28-30 days after the first puncture when stored in the refrigerator. This is the most commonly cited number, and it applies regardless of how much solution remains in the vial.
Some pharmacies specify different post-puncture timelines based on their stability data. You might see windows as short as 14 days or as long as 45 days. The pharmacy label should specify the post-opening timeline separately from the overall BUD. If the label only shows a BUD with no post-opening instruction, a conservative approach is to use the vial within 28 days of first puncture or by the BUD, whichever comes first.
This 28-day post-puncture guideline is why dose planning matters. If you are on a low starting dose, say 0.25mg weekly, a 5mg vial contains roughly 20 weekly doses at that level. You cannot realistically use all 20 doses within 28 days of opening. This is where understanding your 5mg semaglutide dosage chart and planning your usage timeline becomes critical for avoiding waste.
Why the clock starts ticking once you open the vial
Three things change the moment a needle enters the vial.
First, sterility is compromised. Even with proper technique, using alcohol swabs on the stopper and sterile needles, the seal is no longer factory-intact. Microscopic organisms can potentially enter with each subsequent puncture.
Second, air exposure begins. The small amount of air that enters as you withdraw solution introduces oxygen, which can gradually oxidize the semaglutide peptide chains. This oxidation is slow at refrigerator temperatures but accelerates at room temperature.
Third, repeated punctures create small channels in the rubber stopper. After many uses, these channels can allow faster air exchange and potential contamination. This is why proper peptide storage protocols emphasize minimizing the number of times you puncture a single vial.
The combination of these three factors is why an opened vial in perfect fridge conditions still degrades faster than an unopened one. It is not about the fridge failing. It is about the seal being broken.
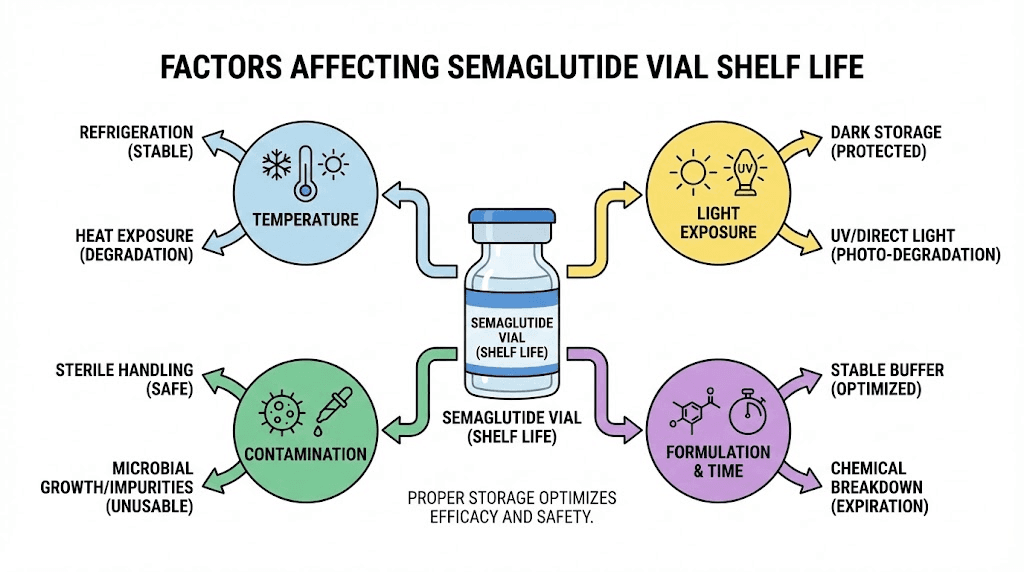
Factors that affect semaglutide vial shelf life
Shelf life is not a fixed number. It is a range determined by how well you control several variables. Two identical vials from the same pharmacy batch can have dramatically different effective shelf lives based entirely on how they are stored and handled. Understanding these factors gives you direct control over how much value you extract from every vial.
Temperature consistency
Semaglutide is a peptide, and peptides are proteins. Like all proteins, they have a three-dimensional structure that determines their biological activity. Temperature fluctuations cause these molecules to unfold slightly, then refold, and each cycle can result in minor structural changes that accumulate into potency loss.
The ideal storage temperature is a steady 36-46 degrees Fahrenheit (2-8 degrees Celsius). Not 35 one day and 50 the next. Consistency matters more than hitting a specific number. A fridge that holds steady at 40 degrees is better than one that swings between 34 and 48 degrees, even if the average is the same.
This is the same principle that applies to all peptides stored in the fridge. The molecule does not care about the average temperature. It cares about the extremes.
Light exposure
UV and visible light accelerate peptide degradation through a process called photodegradation. Semaglutide vials should stay in their original packaging or in an opaque container inside the refrigerator. If your fridge has an internal light that turns on every time you open the door, consider placing the vial in a small box or bag to shield it.
This sounds minor. It adds up. A vial that gets hit with 30 seconds of light every time you open the fridge, multiplied by the 10-20 times per day most households open the fridge, accumulates meaningful light exposure over weeks. The effect is subtle but measurable, especially for researchers tracking precise outcomes.
Contamination risk from repeated punctures
Every needle insertion is a contamination event. Good technique reduces the risk dramatically, but it never eliminates it entirely. The more times you puncture a vial, the higher the cumulative contamination risk.
Best practices include using a fresh, sterile needle for every draw, swabbing the rubber stopper with an alcohol pad before each insertion, and never touching the needle tip to anything other than the stopper. These are fundamental peptide handling basics that protect both the vial contents and the person using them.
For researchers drawing from the same vial multiple times per week, consider whether post-reconstitution storage practices could be improved. Small changes in handling protocol often matter more than upgrading to a more expensive formulation.
Formulation type and additives
Compounded semaglutide vials are not all created equal. The base formulation, the solvent used, and any additives all influence stability.
Vials reconstituted with bacteriostatic water tend to have longer post-opening shelf lives than those made with sterile water alone. The benzyl alcohol preservative in bacteriostatic water actively inhibits microbial growth, extending the safe usage window. Most compounding pharmacies use bacteriostatic water for multi-dose vials specifically because of this preservative benefit.
Some formulations include additional stabilizers. B12 combinations like semaglutide with B12 or semaglutide with methylcobalamin may have different stability profiles than plain semaglutide solutions. The presence of additional compounds in the same vial can either stabilize or destabilize the semaglutide, depending on the specific interaction. Always follow the pharmacy-specific BUD for combination formulations.
Glycine-containing formulations like semaglutide with glycine may have enhanced stability because glycine can act as a cryoprotectant and stabilizer for peptide solutions. Again, the pharmacy BUD reflects their testing of the specific formulation they dispense.
Proper fridge storage for semaglutide vials
Knowing how long a vial lasts means nothing if your storage method is undermining the timeline. Most potency loss in home-stored semaglutide comes not from the peptide being inherently unstable but from storage conditions that accelerate degradation unnecessarily. Fix the storage, and you fix the potency problem.
Ideal temperature range
The target is 36-46 degrees Fahrenheit. That is 2-8 degrees Celsius. This range keeps the semaglutide peptide stable without freezing it. Freezing is catastrophic for semaglutide because ice crystals physically tear apart the molecular structure. A frozen vial should be discarded immediately, no exceptions.
Most home refrigerators run between 35-40 degrees Fahrenheit by default, which falls perfectly within the acceptable range. However, many people adjust their fridge temperature without checking, and internal temperatures can vary by 5-10 degrees depending on location within the fridge. A simple fridge thermometer costs a few dollars and eliminates all guessing.
For those who have experienced what happens when semaglutide gets warm, the lesson is clear. Even brief exposure to temperatures above 86 degrees Fahrenheit can cause irreversible damage. The degradation is invisible. The solution looks the same, but the molecules inside have changed.
Best placement inside the refrigerator
Not all spots in your fridge are equal. The back of the middle shelf offers the most stable temperature. It is far from the door, which experiences the largest temperature swings with every opening. It is not near the top, where warm air from the room tends to collect when the door opens. And it is not on the bottom shelf near the vegetable drawers, where some fridges run slightly colder and risk accidental freezing.
Never store semaglutide vials in the door. Door storage seems convenient because it is accessible, but the temperature in fridge doors can swing by 10-15 degrees every time you open and close it. Over weeks, those fluctuations compound into meaningful degradation.
If you use a dedicated mini-fridge for medications, even better. Medical-grade mini-fridges maintain tighter temperature control than kitchen refrigerators and eliminate the constant door openings from everyday food access.
What to avoid
The bathroom medicine cabinet is not a refrigerator. This seems obvious, but an alarming number of people store injectable medications at room temperature out of convenience or habit. Compounded semaglutide at room temperature degrades significantly faster than in the fridge, especially in humid environments like bathrooms.
Cars are another danger zone. Interior temperatures can exceed 130 degrees Fahrenheit in summer and drop below freezing in winter. Never leave semaglutide vials in a vehicle for any extended period. If you need to transport them, use an insulated cooler bag with a cold pack, making sure the vial does not contact the ice pack directly to avoid freezing.
For detailed travel protocols, check the complete semaglutide travel guide. Traveling with peptide vials requires planning, but it is entirely manageable with the right approach.
Traveling with semaglutide vials
Travel introduces unique storage challenges. A vial that sits perfectly in your home fridge now needs to survive airport security, hotel rooms, and potentially variable temperatures for days or weeks. The good news is that semaglutide can tolerate brief periods at room temperature without significant degradation.
Brand-name Ozempic tolerates room temperature (59-86 degrees Fahrenheit) for up to 56 days. Compounded semaglutide is generally safe at room temperature for 24-48 hours, though some formulations may tolerate longer. When traveling, use an insulated medication travel case and try to return the vial to refrigeration as soon as possible at your destination.
The room temperature tolerance of peptides varies by molecule, so do not assume all peptides in your travel kit follow the same rules. Semaglutide is relatively robust compared to some other research peptides, but it still benefits from refrigeration whenever available.
Signs your semaglutide vial has gone bad
Sometimes a vial expires before the date on the label. Improper storage, accidental temperature exposure, or contamination can render the solution unusable long before the BUD arrives. Knowing what to look for protects both your investment and your safety.
Visual inspection checklist
Before every single dose, inspect the vial. Hold it up to light and look for these warning signs.
Cloudiness. Normal semaglutide solution is clear and colorless. Any haziness, milkiness, or cloudiness indicates protein aggregation, which means the semaglutide molecules have clumped together and lost their proper structure. Cloudy semaglutide will not work as intended.
Particles. Look for floating specks, fibers, or sediment. These could indicate contamination or peptide precipitation. Even tiny particles that settle at the bottom of the vial are a reason to discard and replace.
Color changes. Fresh semaglutide solution is colorless. Any yellowing, browning, or tinting suggests chemical degradation. Even slight color changes that develop gradually over time indicate the peptide is breaking down.
Unusual smell. While semaglutide solution is normally odorless or has a very faint smell, any strong or unusual odor after opening the vial indicates potential bacterial contamination. Do not use it.
The invisible degradation problem
Here is the frustrating truth. Most semaglutide degradation is invisible. The solution can look perfectly clear, colorless, and particle-free while having lost 20, 30, or even 50 percent of its potency. Peptide degradation at the molecular level does not always produce visible changes.
This is why BUD dates matter even when the vial looks fine. The pharmacy assigned that date based on testing that showed potency declining to unacceptable levels around that timeframe. Your eyes cannot detect what their analytical equipment measured.
If you have been using semaglutide from a vial and noticed results plateauing or weight loss stalling despite consistent dosing, expired or degraded semaglutide should be one of the first things you investigate. A fresh vial often resolves the mystery.
This same principle applies if you have been wondering about using expired semaglutide. The answer is not that it becomes dangerous overnight, but that potency loss makes dosing unpredictable. You do not know how much active semaglutide you are actually injecting, which makes protocol adjustments meaningless.
What happens if you use degraded semaglutide
Using mildly degraded semaglutide is unlikely to cause direct harm. The peptide fragments from degradation are not known to be toxic. However, the practical consequences are significant.
Reduced effectiveness means your dose is not what you think it is. If your vial has lost 30% potency, your 0.5mg dose is really delivering roughly 0.35mg of active semaglutide. This can lead to weeks of no weight loss despite doing everything else correctly.
Unpredictable dosing makes it impossible to properly titrate. When you think you are escalating from 0.5mg to 1.0mg, you might actually be going from 0.35mg to 0.7mg, a different change than planned. This complicates the entire dose escalation process outlined in most semaglutide dosing charts.
Contamination risk from vials used well past their intended timeline introduces potential for injection site reactions, unusual fatigue, or gastrointestinal issues beyond what semaglutide normally causes.

Compounded vs brand-name semaglutide vial longevity
The difference between compounded and brand-name semaglutide storage timelines confuses almost everyone. Both contain the same active molecule. Both require refrigeration. But the numbers on the label tell very different stories, and understanding why helps you make better decisions about which product fits your protocol.
Factor | Brand-name (Ozempic) | Brand-name (Wegovy) | Compounded vials |
|---|---|---|---|
Unopened shelf life | Until expiration (18-24 months) | Until expiration (18-24 months) | 30-90 days (BUD) |
Opened/in-use shelf life | 56 days | 28 days | 14-28 days typical |
Storage temperature | 36-46F fridge or 59-86F room temp | 36-46F fridge or 46-86F room temp | 36-46F fridge preferred |
Room temp tolerance | 56 days at 59-86F | 28 days at 46-86F | 24-48 hours generally |
Stability testing | FDA-mandated long-term studies | FDA-mandated long-term studies | Pharmacy-specific short-term studies |
Format | Pre-filled pen | Pre-filled pen | Multi-dose vial |
Preservative | Yes (manufacturer formulation) | Yes (manufacturer formulation) | Varies (bacteriostatic water common) |
Why compounded vials expire sooner
The shorter BUD on compounded semaglutide does not necessarily mean the product is lower quality. It reflects a different regulatory and testing framework.
Brand-name manufacturers like Novo Nordisk invest millions in stability testing programs that track product performance over years. They test across multiple temperature conditions, humidity levels, and storage scenarios. This extensive data allows them to confidently assign expiration dates that extend 18-24 months from manufacturing.
Compounding pharmacies operate under USP Chapter 797 guidelines, which are designed for smaller-batch production. The testing requirements are less extensive, and the default BUD assignments are deliberately conservative. A pharmacy that has not conducted product-specific stability testing might assign a 30-day BUD not because the product goes bad at 31 days, but because they cannot prove it does not.
This creates an interesting situation where the actual stability of compounded semaglutide often exceeds the labeled BUD. Some pharmacies that have invested in rigorous testing can demonstrate stability for 90-120 days. But without that testing, the conservative date protects patients from uncertainty.
USP standards and stability testing
USP Chapter 797 provides the framework that all compounding pharmacies must follow for sterile preparations. The chapter specifies default BUD assignments based on storage conditions and whether the pharmacy has conducted stability testing.
Without stability testing, the maximum BUD for a sterile compounded preparation stored in the refrigerator ranges from 14 to 45 days depending on the risk category. With stability-indicating testing that meets USP standards, pharmacies can extend BUDs based on their data, sometimes significantly.
When evaluating your compounded semaglutide dose chart and usage plan, the BUD becomes a critical factor. If your dose is low and the vial is large, you might not use all the product before the BUD. Planning your dose timeline around the BUD helps minimize waste and ensures every injection delivers full potency.
For comparison, tirzepatide vials in the fridge follow a similar pattern. Compounded tirzepatide also comes with pharmacy-assigned BUDs that are typically shorter than brand-name expiration dates. The storage principles are nearly identical across GLP-1 receptor agonist vials.
How to maximize your semaglutide vial shelf life
You cannot extend a vial beyond its BUD. But you can prevent it from degrading before the BUD arrives. The difference between a vial that maintains 95% potency at day 28 versus one that drops to 70% comes down to how well you control the variables within your power.
Storage best practices
Start with placement. Middle shelf, back of the fridge, away from the door. Use the original box or a small opaque container to block light. If you have a dedicated medication shelf in the fridge, even better.
Temperature monitoring is not optional if you take this seriously. A simple digital thermometer placed next to your vials shows you exactly what temperature your peptides experience. Many modern fridge thermometers log minimum and maximum temperatures over 24-hour periods, revealing fluctuations you would never notice otherwise.
Do not store vials on their side or upside down. Keep them upright so the rubber stopper stays on top and the needle puncture site remains above the solution level. This minimizes the chance of solution leaking or contamination entering through puncture channels.
Handling protocols
Clean hands every time. Wash with soap and water before handling vials and syringes. This is more important than any fancy storage container.
Swab the stopper with 70% isopropyl alcohol before every needle insertion. Let it dry for 10-15 seconds before puncturing. This simple step dramatically reduces contamination risk.
Use a fresh needle for every draw. Never reinsert a used needle into a vial. The needle that just went through your skin carries bacteria back into the vial. This rule applies even if you plan to draw again immediately, a fresh needle takes seconds and protects the entire remaining contents of the vial.
Minimize the number of times you puncture the vial. If your protocol allows, draw multiple doses at once into separate syringes and store the pre-filled syringes in the fridge. This reduces the total puncture count on the vial. Just make sure to label each pre-filled syringe with the date and dosage using proper peptide dosage calculation methods.
When to discard and replace
Discard a vial when any of these conditions apply:
The BUD has passed. Period. Even if the solution looks perfect. Even if there is product remaining. The date exists for a reason.
The 28-day post-opening window has closed (or whatever your pharmacy specifies for post-puncture use). Track this by writing the date of first use directly on the vial with a permanent marker.
Any visual changes have appeared. Cloudiness, particles, color change, or unusual smell means immediate disposal.
The vial was accidentally frozen. Freezing damages semaglutide irreversibly. A vial that froze even briefly should be discarded.
The vial was exposed to temperatures above 86 degrees Fahrenheit for more than a few hours. Brief exposure might be tolerable, but prolonged heat exposure degrades the peptide significantly.
For SeekPeptides members, the protocol database includes specific storage checklists and discard criteria for every common peptide formulation. Having a clear decision framework eliminates the guesswork that leads to using compromised product.
Common mistakes that shorten vial shelf life
Knowing the right way to store semaglutide is only half the battle. Knowing the specific mistakes that destroy shelf life, often without any visible sign, protects you from the invisible potency loss that ruins protocols.
Storing vials in the refrigerator door
This is the number one storage mistake. Fridge doors experience temperature swings of 10-15 degrees with every opening. A busy household that opens the fridge 15-20 times daily subjects door-stored vials to hundreds of temperature fluctuations per month. Each fluctuation stresses the peptide structure.
Move your vials to the back of the middle shelf. It takes five seconds and can add meaningful shelf life.
Forgetting to note the opening date
You punctured the vial on... when exactly? If you cannot answer this question instantly, you have already lost track of the post-opening window. Memory is unreliable for tracking dates across weeks.
Write the date of first puncture directly on the vial label with a permanent marker. Some researchers use a small piece of tape with the date written on it. However you do it, make the opening date impossible to forget.
Temperature swings from unnecessary handling
Taking the vial out of the fridge, drawing a dose, getting distracted, leaving the vial on the counter for 30 minutes, then returning it to the fridge. This sequence sounds harmless. Repeated twice weekly over a month, it subjects the semaglutide to 8-10 room temperature excursions that accelerate degradation.
Get everything ready before removing the vial from the fridge. Prepare your syringe, swab your injection site, have the alcohol pad ready. Then take the vial out, draw the dose, and return it immediately. Minimize the time the vial spends outside the fridge.
Reusing needles on vials
Some people reinsert the same needle they just used for injection back into the vial to save needles. This introduces skin bacteria directly into the sterile vial environment. It is one of the fastest ways to contaminate a multi-dose vial and potentially cause an infection at the injection site.
Needles are inexpensive. Vials of semaglutide are not. Use a fresh needle for every vial interaction without exception.
Ignoring pharmacy-specific instructions
Generic advice says 28 days after opening. Your pharmacy might say 14 days. Or 45 days. The pharmacy-specific instruction reflects their formulation and their testing. Ignoring it in favor of what you read online is gambling with potency.
When in doubt, call your compounding pharmacy. They have the stability data. They know their product. Their guidance supersedes anything you find in a general blog post, including this one.

Semaglutide vial storage by concentration
Concentration affects how many doses you can draw from a single vial, which directly impacts whether you can use the entire vial within the post-opening shelf life window. Matching your dose to the right vial size prevents waste and ensures you are always working with fresh, potent semaglutide.
5mg semaglutide vials
A 5mg vial is the most common starting point for researchers new to semaglutide. At a starting dose of 0.25mg weekly, this vial theoretically contains 20 weekly doses. At a maintenance dose of 1.0mg weekly, it contains 5 weekly doses.
The math problem becomes obvious. At 0.25mg weekly, 20 doses would take 20 weeks to use. Your post-opening shelf life is 28 days. You will waste the majority of the vial unless you increase your dose or share with another researcher in your household.
The practical solution is matching your vial size to your current dose. At lower doses, consider whether a smaller vial or a pre-filled syringe format might be more economical. Use the semaglutide dosage calculator to determine exactly how many doses your specific concentration provides, then compare that to your 28-day window.
For detailed 5mg/2ml dosage charts and 5mg/ml dosage charts, the reconstitution volume directly affects concentration and the number of usable draws per vial.
10mg semaglutide vials
A 10mg vial makes more sense for researchers on higher doses. At 2.0mg weekly, you get 5 weekly doses from a single vial, fitting neatly within the 28-day post-opening window. At 1.0mg weekly, you get about 10 doses, still within the 28-day limit if you dose frequently enough or if you are titrating up during that period.
The 10mg semaglutide dosage chart shows the relationship between reconstitution volume, concentration, and doses per vial. Higher concentrations mean smaller injection volumes per dose, which many researchers prefer for comfort at the injection site.
Reconstitution plays a critical role here. The amount of bacteriostatic water you mix with 10mg of semaglutide determines the final concentration, which determines how many units you draw per dose. Getting this right from the start, using a proper 10mg reconstitution chart, prevents dosing errors that compound over the life of the vial.
Different concentrations and shelf life
Higher concentrations do not inherently last longer or shorter in the fridge than lower concentrations. The shelf life is determined by the formulation stability, not the amount of semaglutide per milliliter. A 2.5mg/ml solution and a 5mg/ml solution from the same pharmacy, using the same base and preservative, should have similar BUDs.
What changes is the practical usage timeline. Higher concentrations mean smaller draw volumes per dose, which means less air enters the vial with each use (assuming the same needle gauge). This could theoretically reduce oxidation exposure slightly, though the difference is marginal compared to the other factors discussed above.
The semaglutide mixing chart and reconstitution chart resources can help you determine the optimal concentration for your dose level and vial size, balancing ease of measurement against the post-opening shelf life constraint.
What to do if your semaglutide vial was left out
It happens. You get distracted after drawing a dose. The vial sits on the counter. Maybe overnight. Maybe for a few hours. The question is whether the vial is still usable or whether you need a replacement.
Brief room temperature exposure (under 4 hours)
A few hours at room temperature is generally not catastrophic for semaglutide. The peptide is reasonably stable at temperatures up to 86 degrees Fahrenheit (30 degrees Celsius). Brand-name Ozempic is explicitly rated for room temperature storage for up to 56 days, which tells you the molecule itself can handle it.
However, compounded semaglutide may not have the same room temperature stability as brand-name formulations. The preservative system, pH, and formulation details all affect room temperature tolerance. If your compounded vial sat out for under 4 hours at normal room temperature (68-75 degrees Fahrenheit), it is most likely fine. Return it to the fridge promptly and continue using it.
Extended room temperature exposure (4-24 hours)
Longer exposure increases the risk of degradation. If your vial was out for 4-24 hours at moderate room temperature, the risk depends heavily on the actual temperature. A climate-controlled room at 72 degrees presents less risk than a warm kitchen in summer at 85 degrees.
For brand-name semaglutide, this duration at room temperature falls well within the manufacturer-tested window and should not cause significant potency loss. For compounded semaglutide, use your judgment. If the room was cool and the vial was not in direct sunlight, the product is probably acceptable. If conditions were warm, consider how much product remains and whether starting a fresh vial provides better peace of mind.
Freezing exposure
There is no debate on this one. If a semaglutide vial froze, discard it. Freezing forms ice crystals that physically disrupt the three-dimensional structure of the semaglutide molecule. Once those structures are disrupted, they do not reform correctly when the solution thaws. The result is a clear liquid that contains broken, inactive peptide fragments instead of functional semaglutide.
This rule applies even if the vial was only partially frozen or frozen briefly. The damage occurs during the freezing process itself, not from the duration of frozen storage. A vial that froze for 10 minutes sustains the same type of damage as one frozen for 10 hours.
If you are unsure whether your vial froze, check for ice crystals or look for changes in solution clarity after thawing. But honestly, if you suspect freezing occurred, the safest approach is replacement. The cost of a new vial is always less than the cost of weeks on a degraded product that delivers inconsistent results.
Planning your vial usage to minimize waste
Smart vial planning is where storage knowledge meets practical protocol design. The goal is to use every usable dose in the vial before the shelf life expires, maximizing the value of every milligram you purchased.
Matching vial size to your dose
If you are on a stable maintenance dose, calculate how many weeks of doses your vial provides and compare that to the post-opening shelf life. Here are some common scenarios:
A 5mg vial at 0.25mg per week provides 20 weeks of doses. Post-opening shelf life is 28 days (4 weeks). You will use only 1mg of the 5mg before the vial expires. That is 80% waste.
A 5mg vial at 1.0mg per week provides 5 weeks of doses. Post-opening shelf life is 28 days (4 weeks). You will use 4mg of 5mg. Minimal waste.
A 10mg vial at 2.0mg per week provides 5 weeks of doses. Post-opening shelf life is 28 days. You use 8mg of 10mg. Roughly 20% waste, which is reasonable.
For anyone on the dose escalation schedule where doses increase weekly or biweekly, a single vial might see 0.25mg the first week, 0.5mg the second, 0.75mg the third, and 1.0mg the fourth. That totals 2.5mg in 28 days, making a 2.5mg or 5mg vial ideal.
Use the peptide calculator or peptide cost calculator to determine the most cost-effective vial size for your specific dose and duration.
Dose escalation and vial timing
Most semaglutide protocols involve a dose escalation schedule where you start low and gradually increase over weeks or months. The standard escalation pattern for most protocols is:
Weeks 1-4 at 0.25mg weekly. Weeks 5-8 at 0.5mg weekly. Weeks 9-12 at 1.0mg weekly. Then potentially higher depending on response and protocol goals.
Understanding how long semaglutide takes to work helps frame why the escalation exists. Starting at a full dose often causes significant gastrointestinal side effects. The gradual increase allows your body to adjust.
Each vial should align with your current escalation phase. Opening a 10mg vial during the 0.25mg phase means most of that vial will expire before you use it. Better to use a smaller vial during the low-dose phase and switch to larger vials as your dose increases.
Pre-filling syringes for the week
Some researchers draw multiple doses at once, storing pre-filled syringes in the refrigerator. This approach reduces the number of times you puncture the vial and can be convenient for weekly dosing preparation.
Pre-filled syringes should be used within 7-14 days. Label each syringe with the date it was filled and the dose. Store them capped, needle-up, in a clean container in the fridge. This approach works well for researchers who dose more than once per week or who want to minimize their interaction with the vial.
The semaglutide syringe dosage chart and conversion chart for syringe dosage can help you draw precise amounts with confidence.
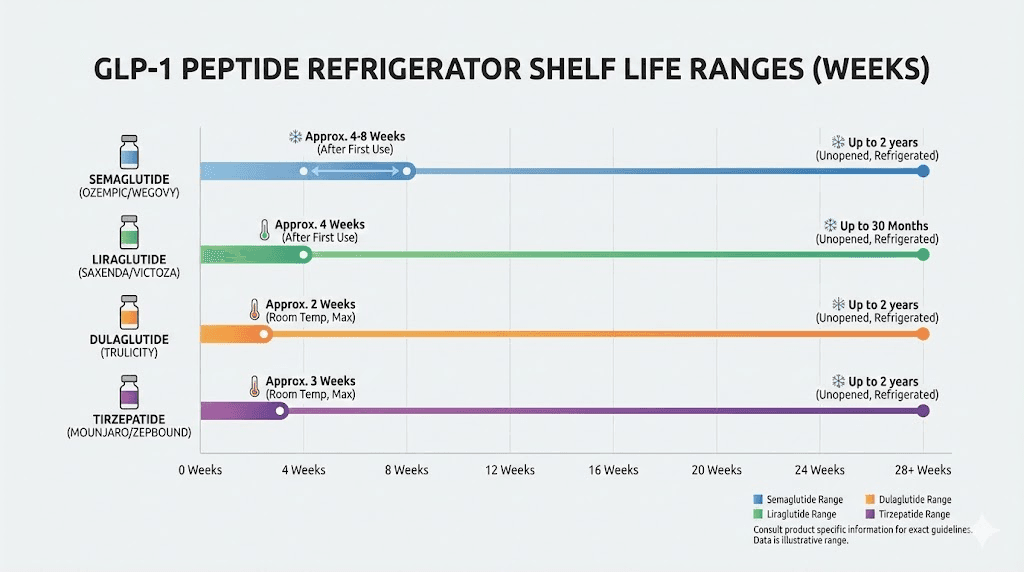
Semaglutide vial storage compared to other GLP-1 peptides
If you work with multiple GLP-1 receptor agonists, understanding how semaglutide vial storage compares to other peptides in this class helps you organize a consistent storage protocol across your entire research setup.
Tirzepatide vial storage
Compounded tirzepatide vials in the fridge follow very similar storage rules to semaglutide. Same temperature range (36-46 degrees Fahrenheit), similar post-opening windows (28-30 days typically), and the same sensitivity to freezing and light exposure.
The main difference is that tirzepatide, being a dual GIP/GLP-1 agonist, has a slightly different molecular structure that some pharmacies say gives it marginally different stability characteristics. In practice, the storage protocols are nearly interchangeable.
Researchers who use both semaglutide and tirzepatide should clearly label each vial and track opening dates separately. Mixing up which vial was opened when leads to uncertainty about whether either product is still within its usable window. The semaglutide vs tirzepatide comparison page covers the pharmacological differences between these two peptides.
Retatrutide and newer GLP-1 peptides
Newer triple-agonist peptides like retatrutide are primarily available as research-grade lyophilized powder that requires reconstitution. The storage rules for reconstituted peptides in the fridge apply here, with most reconstituted research peptides lasting 14-30 days refrigerated depending on the specific molecule and the solvent used.
For anyone exploring the broader peptide landscape, understanding how long peptides last in powder form versus reconstituted solution provides important context. Lyophilized (freeze-dried) peptide powder is dramatically more stable than reconstituted solution, lasting months or even years when stored properly. This is why many researchers prefer to reconstitute only what they will use within 2-4 weeks rather than reconstituting an entire supply at once.
General peptide storage principles that apply to all vials
Whether you are storing semaglutide, tirzepatide, BPC-157, or any other research peptide, certain principles apply universally:
Refrigeration extends shelf life for all reconstituted peptides. Room temperature accelerates degradation for all of them. Freezing destroys virtually all reconstituted peptide solutions. Light exposure harms all peptides through photodegradation. And contamination from repeated punctures threatens any multi-dose vial.
The comprehensive peptide storage guide covers these principles in detail for researchers working with multiple peptides simultaneously. Building a consistent storage routine that applies to all your research materials is more effective than trying to remember different rules for each individual product.
The reconstitution factor in vial shelf life
If you are working with lyophilized (powdered) semaglutide rather than pre-mixed liquid vials, the reconstitution process itself plays a crucial role in determining how long your vial will last in the fridge. The solvent you choose, the technique you use, and the cleanliness of the process all affect the starting point of your shelf life clock.
Bacteriostatic water vs sterile water
This choice matters more than most people realize. Bacteriostatic water contains 0.9% benzyl alcohol as a preservative. This preservative actively inhibits bacterial growth in the reconstituted solution, extending the safe multi-use window to 28-30 days.
Sterile water for injection contains no preservative. Once you reconstitute a peptide with sterile water and puncture the vial, there is nothing stopping microbial growth except refrigeration temperature. Vials reconstituted with sterile water should be used within 24-48 hours for maximum safety, or up to 7 days refrigerated in very conservative protocols.
For multi-dose semaglutide vials that you plan to draw from multiple times over weeks, bacteriostatic water is the clear choice. The guide on how to mix peptides with bacteriostatic water walks through the technique step by step.
Proper reconstitution technique
How you reconstitute the vial sets the trajectory for everything that follows. Rough handling during reconstitution can damage peptide molecules before you even store the vial.
The critical rules: add bacteriostatic water slowly along the inside wall of the vial, not directly into the powder. Never shake the vial. Shaking creates foam and subjects the peptide to mechanical stress that damages molecular structure. Instead, gently swirl the vial until the powder dissolves completely. This should take 1-3 minutes for most semaglutide formulations.
The complete semaglutide reconstitution guide covers the technique in full detail, including specific instructions for 5mg vials and the proper 5mg mixing chart for different reconstitution volumes. Getting the reconstitution right protects every subsequent dose from that vial.
The amount of bacteriostatic water matters
Using too little water creates a highly concentrated solution that can be difficult to draw accurately with standard insulin syringes. Using too much creates a dilute solution that requires larger injection volumes.
For a 5mg vial, common reconstitution volumes are 1ml or 2ml of bacteriostatic water. For a 10mg vial, 2ml is the most common choice. The peptide reconstitution calculator can help you determine the exact volume needed for your target concentration.
The reconstitution volume does not directly affect shelf life. Whether you use 1ml or 3ml of bacteriostatic water, the BUD remains the same. What changes is the per-dose volume you need to draw and how precisely you can measure each dose using the markings on your syringe.
Special considerations for semaglutide combination vials
Many compounding pharmacies now offer semaglutide combined with other active ingredients in a single vial. These combination formulations can have different shelf life profiles than plain semaglutide, and understanding the specific storage requirements for each combination protects your investment.
Semaglutide and B12 combination vials
The semaglutide with B12 combination is one of the most popular compounded formulations. Cyanocobalamin (B12) is a relatively stable molecule that does not typically interfere with semaglutide stability. However, combination formulations undergo separate stability testing because the presence of multiple active ingredients can sometimes affect the solution pH or create unexpected interactions.
The compounded semaglutide with B12 dosage chart accounts for both components in the dose calculations. Storage rules for the combination vial should come from your specific pharmacy, as they have tested their particular formulation for stability.
Semaglutide and glycine vials
Glycine is an amino acid sometimes added to semaglutide formulations as a stabilizer. It can act as a buffer and may help maintain the proper pH of the solution over time. Some pharmacies report that glycine-containing formulations have longer stability profiles, though the BUD will still reflect the pharmacy-specific testing data.
Semaglutide with methylcobalamin
Semaglutide with methylcobalamin is another popular combination. Methylcobalamin is the active form of B12 and is slightly less stable than cyanocobalamin. This means methylcobalamin-containing combination vials may have slightly shorter BUDs than cyanocobalamin-containing ones. Again, defer to the pharmacy label for your specific product.
No matter which combination formulation you use, the fundamental storage rules remain the same. Refrigerate at 36-46 degrees Fahrenheit, protect from light, minimize puncture events, track your opening date, and discard at the BUD or post-opening limit, whichever comes first.
How semaglutide vial storage affects your results
Storage is not just about keeping the peptide alive. It directly determines the quality of every injection you take from that vial. A vial stored at the edge of acceptable conditions delivers subtly different results than one stored optimally, even if both pass a visual inspection.
The potency degradation curve
Semaglutide does not suddenly become inactive at the BUD. Degradation follows a curve. A freshly compounded vial might contain 100% of labeled potency. At day 14, it might be at 97%. At day 28, perhaps 92%. At day 45, maybe 80%. The exact numbers depend on formulation and storage conditions, but the pattern holds. Potency declines gradually, then accelerates as degradation products accumulate and begin catalyzing further breakdown.
This curve explains something frustrating that many researchers experience. The first two weeks on a new vial feel great. Appetite suppression is strong. Energy is stable. The appetite-suppressing effects kick in reliably. Then around week 3-4, things feel slightly different. Not dramatically worse, just a little off. The dose seems less effective. The temptation is to increase the dose, but the real problem might be a 10-15% potency drop from accumulated storage stress.
Understanding this curve changes how you think about vial management. The goal is not just to use the vial before it goes bad, but to use it while it is still near peak potency. For researchers chasing precise protocols and consistent results, this distinction matters.
Why consistent storage produces consistent results
Protocol consistency requires dosing consistency, and dosing consistency requires potency consistency. If your vial delivers 95% potency on week 1 and 80% on week 4, you are effectively running two different dose levels without realizing it.
This matters most during the dose escalation phase. When you increase from 0.25mg to 0.5mg weekly, you expect to feel a specific change in appetite suppression and appetite response timing. If the vial is at 85% potency, your 0.5mg dose is really about 0.425mg. The expected jump in effect does not happen, and you might conclude that 0.5mg is not enough for you, when the real problem is storage-related potency loss.
Researchers who maintain optimal storage conditions report more predictable responses at each dose level. They can more accurately identify their ideal maintenance dose because each injection delivers what the label says it should. This is the hidden benefit of paying attention to storage. It does not just save money on wasted product. It makes your entire protocol more reliable.
The cost of poor storage
Consider the math. A 10mg vial costs a certain amount. If poor storage degrades 20% of the product before you use it, that is 2mg of semaglutide wasted. Multiply that across months of use, and the cost adds up quickly. The peptide cost calculator can help quantify these losses based on your specific dose and vial size.
Beyond the direct product waste, there is the indirect cost of suboptimal results. Weeks spent on degraded semaglutide that does not work as expected means delayed progress, unnecessary dose escalation, and frustration that could have been avoided with better storage practices.
The researchers who get the best results from semaglutide, the ones who report steady progress from month one onward, almost universally cite good storage habits as a foundational practice. It is not glamorous. It does not make for exciting conversation. But it separates consistent results from inconsistent ones more reliably than almost any other variable.
Understanding your semaglutide vial label
The label on your semaglutide vial contains critical information that many researchers overlook. Reading and understanding every element of that label prevents the most common storage and usage mistakes.
Key information on the label
Your vial label should include the beyond-use date (BUD), the concentration (usually in mg/ml), the total volume, the lot number, and storage instructions. Some labels also include the compounding date, which helps you calculate how much of the BUD window has already elapsed before you received the vial.
Pay close attention to the difference between the compounding date and the BUD. If a pharmacy assigns a 60-day BUD and the vial was compounded 15 days before it arrived at your door, you have 45 days remaining. Not 60.
Shipping time matters. If your vial traveled for 3 days in a shipping container with an ice pack, those 3 days count. The clock started at compounding, not at delivery.
What to do if the label is unclear
Call the pharmacy. If there is no BUD on the label, if the date is smudged, or if you are unsure about any detail, contact the compounding pharmacy directly. They maintain batch records and can tell you exactly when the vial was compounded and what BUD it carries.
Never guess on BUD dates. The cost of a phone call is zero. The cost of injecting degraded semaglutide for weeks is significant in terms of both wasted product and wasted time.
Tracking your vial freshness
A simple tracking system prevents the most common vial management mistake, which is losing track of when the vial was opened and when it expires. You do not need a complicated spreadsheet. A few basic habits cover it.
The minimum tracking system
Write two dates on every vial with a permanent marker. The date you first punctured it, and the discard-by date (either BUD or 28 days post-opening, whichever is sooner). Check both dates before every use. That is the entire system.
Some researchers add a tally mark each time they draw from the vial. This gives a quick visual count of how many punctures the stopper has received, which is useful for estimating contamination risk and remaining volume.
Using a fridge thermometer log
A min/max fridge thermometer costs under 10 dollars and provides invaluable data. Check it weekly and note the minimum and maximum temperatures. If you ever see a reading below 33 degrees (potential freezing risk) or above 48 degrees (accelerated degradation), investigate and fix the issue.
This simple monitoring practice catches problems like a fridge thermostat malfunction, a door seal that is not closing properly, or a power outage that went unnoticed. Any of these events could compromise your vial without any visible signs.
For researchers committed to optimizing their semaglutide protocols and storage practices, SeekPeptides offers comprehensive storage tracking tools and protocol builders that integrate vial management into the broader dosing and timing workflow. Members find that systematizing these details removes the mental load of tracking multiple variables simultaneously.

Frequently asked questions
Can I use a semaglutide vial past its beyond-use date if it looks fine?
It is not recommended. The BUD reflects the last date that potency and sterility are reliably guaranteed based on testing. Visual inspection cannot detect molecular degradation. The solution can look perfectly clear and colorless while having lost significant potency. For consistent results and safety, respect the BUD and replace the vial. Learn more about the risks of using expired semaglutide.
Does the brand of compounding pharmacy affect how long the vial lasts?
Yes. Each pharmacy uses different formulations, preservatives, and stability testing protocols. A vial from Empower Pharmacy may have a different BUD than one from Olympia or another provider, even if the semaglutide concentration is identical. Always follow the specific pharmacy instructions on your vial label.
What happens if I freeze my semaglutide vial by accident?
Discard it immediately. Freezing creates ice crystals that permanently damage the peptide molecular structure. Even brief freezing causes irreversible harm. The vial may look normal after thawing, but the semaglutide molecules inside have been structurally compromised and will not work as intended.
Is it better to buy smaller vials more frequently or larger vials less often?
Match the vial size to your dose. If you are on a low dose (0.25-0.5mg weekly), smaller vials minimize waste because you can use the entire contents within the post-opening window. If you are on higher doses (1.0-2.4mg weekly), larger 10mg vials are more cost-effective because you will use most of the product before expiration. The peptide cost calculator can help you compare the economics.
Can I store semaglutide in a mini-fridge?
Yes, as long as the mini-fridge maintains a consistent 36-46 degrees Fahrenheit (2-8 degrees Celsius). Dedicated medication mini-fridges are actually better than kitchen refrigerators because they are opened less frequently, resulting in fewer temperature fluctuations. Just verify the temperature with a thermometer, as some mini-fridges have less precise temperature control.
How does the time of day I inject affect vial shelf life?
The time of injection does not directly affect vial shelf life. However, establishing a consistent injection time means you remove the vial from the fridge at the same time each week, creating a predictable temperature excursion pattern. Consistency in handling supports consistency in storage conditions.
Should I let the vial warm to room temperature before drawing a dose?
Some researchers prefer to let the vial sit at room temperature for 5-10 minutes before injecting because cold injections can be slightly more uncomfortable. This brief warming period will not affect shelf life. Just return the vial to the fridge immediately after drawing your dose. Do not leave it out for extended periods.
What if my semaglutide is a slightly different color than my last vial?
Minor batch-to-batch variations in appearance are normal for compounded medications. However, if the solution is noticeably yellow, brown, or any color other than clear/colorless, do not use it. Contact your pharmacy. True semaglutide solution should be clear and colorless. Learn more about what color semaglutide should be.
External resources
For researchers serious about optimizing their semaglutide protocols and storage practices, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, proper dosing charts, storage protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your vials stay potent, your fridge stay steady, and your protocols stay effective.