Mar 25, 2026

Some people lose 20% of their body weight on tirzepatide. Others hit a wall at 8%. And a growing number cannot get it at all, whether because of supply constraints, insurance denials, side effects that make the drug unbearable, or a medical history that rules it out entirely. If you are reading this, you probably fall into one of those categories. You need something else. Something that works.
The good news is that the weight loss medication landscape has exploded. Tirzepatide was groundbreaking as the first dual GIP/GLP-1 receptor agonist, but it is no longer the only advanced option available. Researchers have developed triple agonists targeting three receptors simultaneously. Combination therapies pairing two proven compounds into a single injection now deliver results that rival or exceed what tirzepatide achieves alone. Oral pills that eliminate the need for needles entirely are completing Phase 3 trials with impressive data.
The alternatives are not just adequate anymore. Some of them may actually be better for your specific situation.
But choosing the right alternative requires more than scanning a list of drug names. You need to understand how each medication works at the receptor level, what the clinical trials actually show (not what marketing claims suggest), how side effect profiles compare, and which options are available right now versus still in development. This guide covers every viable alternative to tirzepatide, from the most established to the most cutting-edge, with the specific numbers and protocols that help you make an informed decision.
SeekPeptides has analyzed the clinical data on every major weight loss peptide and GLP-1 medication currently available or in late-stage development. What follows is the most comprehensive comparison you will find anywhere.
Why people look for tirzepatide alternatives
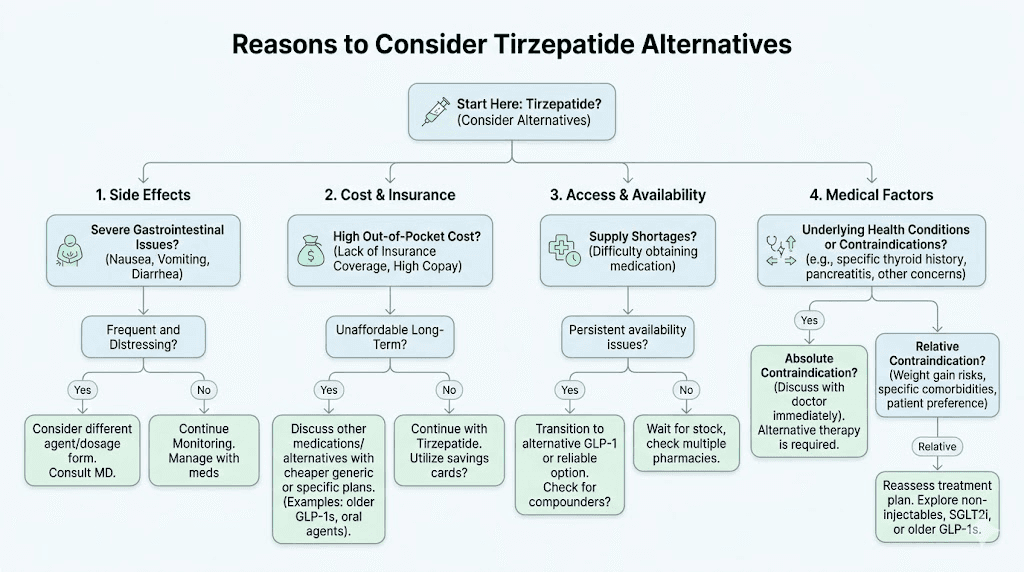
Before diving into the alternatives themselves, it helps to understand why someone would move away from tirzepatide in the first place. The reasons fall into several distinct categories, and your specific reason shapes which alternative makes the most sense.
Side effects that do not resolve
Tirzepatide works. Nobody disputes that. But for some people, the side effect profile makes it unsustainable. Persistent nausea that does not improve after the first few weeks. Constipation severe enough to require medical intervention. Body aches that interfere with daily life. Headaches that persist through dose adjustments. Insomnia that compounds the fatigue already caused by caloric restriction.
These are not rare complaints.
Clinical trials report gastrointestinal adverse events in 40-50% of tirzepatide users, with nausea alone affecting roughly one in three participants. Most people tolerate it. Some do not. And when side effects persist beyond the typical 4-6 week adjustment period, switching to an alternative with a different receptor binding profile or mechanism of action often resolves the issue entirely.
Inadequate response
Not everyone responds equally to tirzepatide. Genetic variations in GIP and GLP-1 receptor sensitivity, differences in gut microbiome composition, metabolic adaptation, and individual hormonal profiles all influence how much weight a person loses. If you have been on tirzepatide for several months without meaningful results, you are not alone. Our guide on tirzepatide not working anymore covers the troubleshooting steps, but sometimes the answer is simply that a different mechanism works better for your biology.
Some people plateau early. Others see initial results that stall completely. The reasons for not losing weight on tirzepatide are varied, but when optimization fails, exploring alternatives becomes the logical next step.
Access and cost barriers
Even with the shortage officially resolved, tirzepatide remains expensive without insurance coverage. Brand-name Mounjaro and Zepbound can cost $1,000 or more per month out of pocket. Insurance coverage remains inconsistent, with many plans excluding weight loss indications entirely. Affordable tirzepatide options exist through compounding pharmacies, but regulatory changes have tightened access to compounded tirzepatide significantly.
For many people, cost alone drives the search for alternatives.
Medical contraindications
Personal or family history of medullary thyroid carcinoma. Multiple endocrine neoplasia syndrome type 2. Severe gastroparesis. Pancreatitis history. Pregnancy or plans to become pregnant. These conditions may rule out tirzepatide entirely, making alternatives not just preferable but medically necessary.
How tirzepatide works and what makes an alternative "close"
Understanding tirzepatide at the receptor level is essential for evaluating alternatives. Without this foundation, comparing medications becomes guesswork.
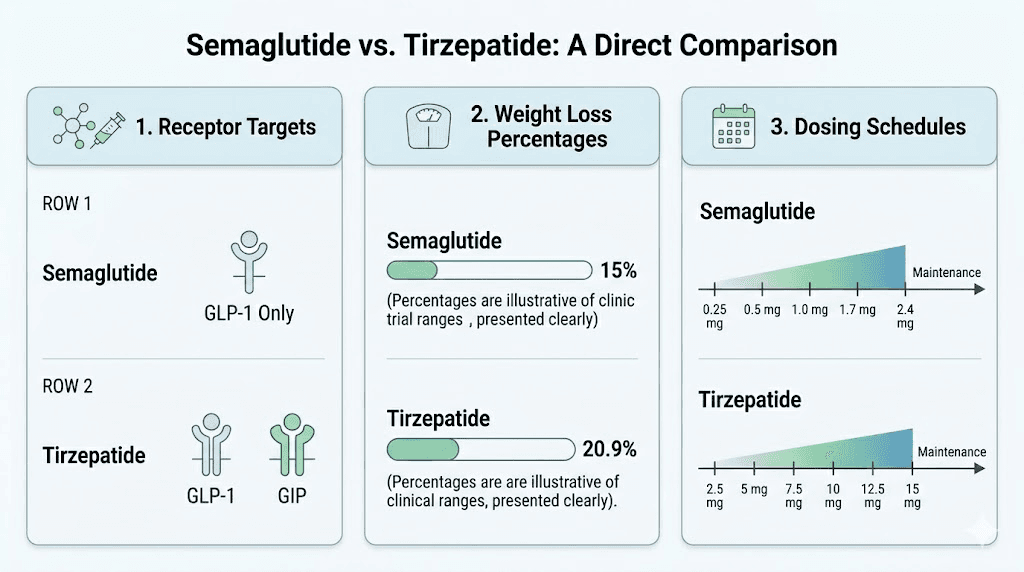
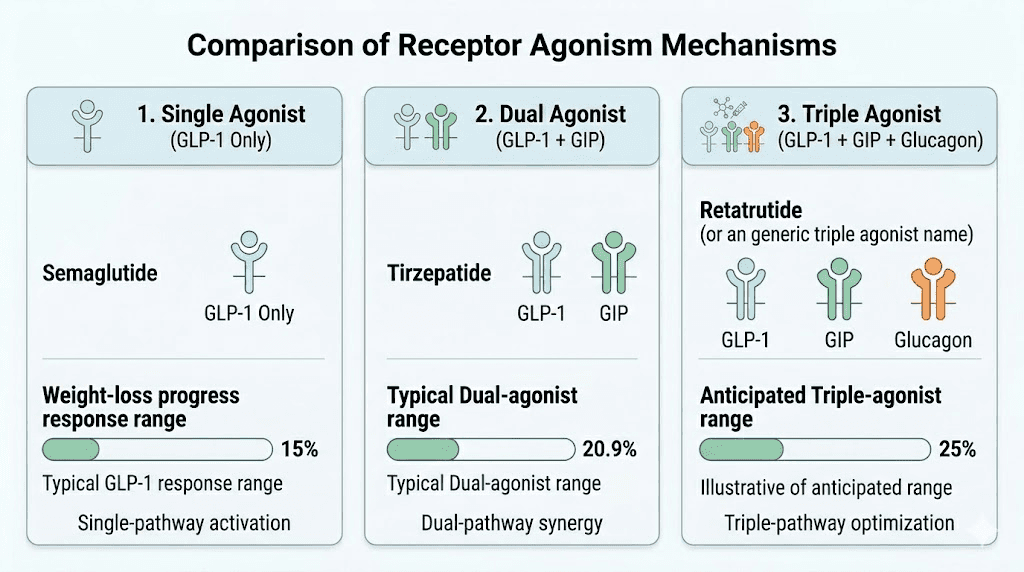
Tirzepatide is a dual GIP/GLP-1 receptor agonist. It simultaneously activates two incretin receptors that play central roles in glucose metabolism, appetite regulation, and energy balance. The GLP-1 receptor activation slows gastric emptying, reduces appetite through hypothalamic signaling, and enhances insulin secretion in a glucose-dependent manner. The GIP receptor activation adds a complementary mechanism, improving insulin sensitivity and potentially enhancing the metabolic effects beyond what GLP-1 alone achieves.
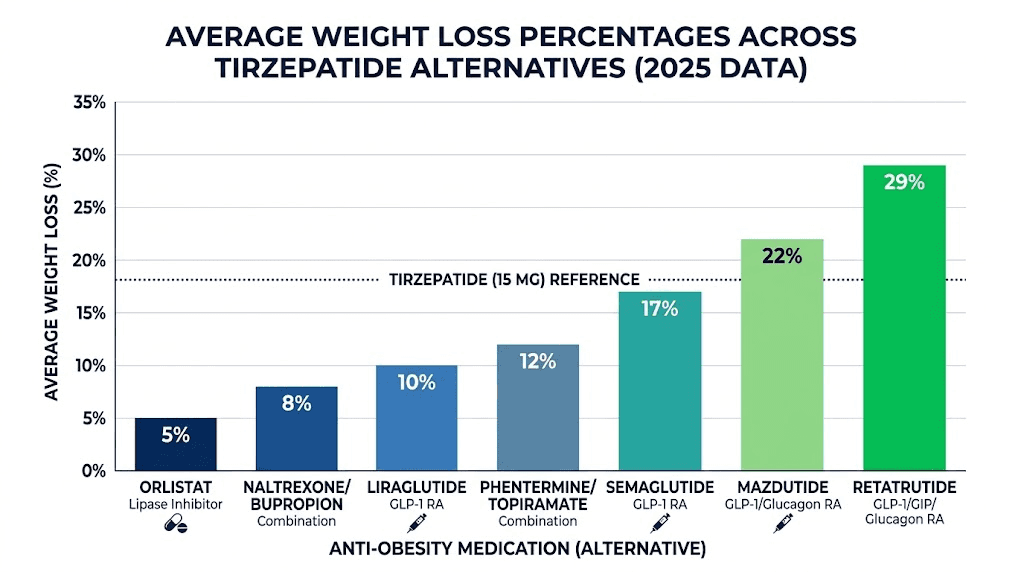
This dual mechanism is what separates tirzepatide from earlier GLP-1 receptor agonists and explains its superior weight loss results in clinical trials. The speed at which tirzepatide works and the magnitude of weight loss, averaging 20-22% of body weight in trials, set a high bar for alternatives.
So what makes an alternative "close" to tirzepatide? Three factors matter most.
Receptor overlap. Medications targeting similar or additional incretin pathways come closest to replicating tirzepatide effects. A pure GLP-1 agonist covers half the mechanism. A triple agonist covers the same two receptors plus glucagon. The more receptor overlap, the more similar the metabolic effects.
Weight loss magnitude. Clinical trial data showing comparable percentage body weight reduction is the most direct measure of how close an alternative comes to tirzepatide performance.
Side effect profile. An alternative that achieves similar weight loss but with fewer gastrointestinal side effects or different tolerability characteristics may actually be preferable, even if the mechanism differs.
Tier 1: the closest alternatives available now
These medications are FDA-approved and available through prescribers today. They represent the most practical alternatives for anyone who needs to switch from tirzepatide immediately.
Semaglutide (Wegovy, Ozempic)
Semaglutide is the most established alternative and the one most people consider first. As a pure GLP-1 receptor agonist, it covers one of the two receptor pathways that tirzepatide targets. The clinical data is extensive, the prescribing infrastructure is well-established, and both injectable and oral formulations are available.
How it compares to tirzepatide. The SURMOUNT and SURPASS trials put this head to head. Tirzepatide consistently outperformed semaglutide for weight loss, with the highest tirzepatide dose achieving approximately 22% body weight reduction compared to semaglutide 2.4 mg at roughly 15%. That is a meaningful gap. But 15% weight loss is still clinically significant, and for many people, semaglutide delivers excellent results with potentially better tolerability.
The switching process from tirzepatide to semaglutide requires careful dose management. The conversion chart helps align equivalent doses, but most prescribers recommend starting semaglutide at a lower dose and titrating up regardless of previous tirzepatide dose to minimize gastrointestinal side effects during the transition.
Dosing protocol. Semaglutide for weight loss (Wegovy) follows a standardized titration schedule. You start at 0.25 mg weekly for 4 weeks, increase to 0.5 mg for 4 weeks, then 1.0 mg for 4 weeks, followed by 1.7 mg for 4 weeks, reaching the target dose of 2.4 mg weekly. The full semaglutide dosage breakdown and dosage calculator provide exact unit-level calculations for compounded versions.
Side effect differences. The gastrointestinal side effect profile is similar but not identical. Semaglutide constipation, bloating, and fatigue are common complaints. Some people who cannot tolerate tirzepatide find semaglutide more manageable, likely because the absence of GIP receptor activation changes the overall gastrointestinal response. Others experience the opposite. The only way to know is to try.
Formulation options. This is where semaglutide has a clear advantage over tirzepatide. Injectable Wegovy and Ozempic are the standard, but sublingual semaglutide, compounded versions, and the newly approved oral Wegovy pill offer multiple routes of administration. If needle aversion is part of why you want an alternative, semaglutide provides options that tirzepatide currently does not match.
Cost. Brand-name Wegovy runs $1,300-1,500 per month without insurance. However, qualifying for semaglutide through insurance is often easier than qualifying for tirzepatide, as semaglutide has broader coverage. Compounded semaglutide, where still available, runs $150-300 per month.
Best for: People who want a proven, widely available alternative with multiple formulation options and established long-term safety data.
Liraglutide (Saxenda)
Liraglutide is the older sibling in the GLP-1 family. Approved for weight management as Saxenda, it requires daily injections rather than weekly, which is a significant practical difference. The injection process is identical, but doing it seven times per week instead of once changes the experience considerably.
Weight loss data. Clinical trials show average weight loss of 5-10% of body weight over 56 weeks with liraglutide 3.0 mg daily, compared to tirzepatide at 20-22%. The gap is substantial. Liraglutide is a weaker alternative by the numbers, but it remains FDA-approved for weight management and has the longest real-world safety track record of any GLP-1 for obesity.
When it makes sense. Liraglutide may be appropriate when both tirzepatide and semaglutide have been tried and failed, when insurance covers Saxenda but not other options, or when a patient needs a medication with the most extensive long-term safety data. The fatigue profile tends to be milder with liraglutide, and some patients prefer the daily dosing because it provides more flexible dose adjustment opportunities.
Best for: People who need an established option with extensive safety data, or who have insurance that specifically covers Saxenda.
Compounded tirzepatide
Technically, this is not an alternative to tirzepatide. It is tirzepatide, just from a compounding pharmacy rather than the brand manufacturer. But for many people searching for alternatives, the real issue is cost or access to the brand-name product, making compounded tirzepatide the closest possible "alternative."
Regulatory changes have complicated this landscape significantly. The FDA ended the official tirzepatide shortage, which restricts the legal basis for 503A pharmacies to compound it. However, 503B outsourcing facilities and certain state-specific regulations create pathways that still allow compounded tirzepatide access in some situations. The grey market adds another layer of complexity and risk.
If you go this route, understanding proper reconstitution, correct syringe dosing, and storage requirements is essential for both safety and efficacy. Use the compounded tirzepatide dosage calculator to verify your measurements.
Best for: People who respond well to tirzepatide but need a more affordable access pathway.
Tier 2: advanced alternatives in late-stage development
These medications are not yet FDA-approved but have completed or are completing Phase 3 clinical trials. They represent the next generation of obesity therapeutics and several may become available within the next 12-24 months.
Retatrutide: the triple agonist
If tirzepatide was revolutionary as a dual agonist, retatrutide aims to be transformative as a triple agonist. It targets GLP-1, GIP, and glucagon receptors simultaneously, adding a third mechanism that directly increases energy expenditure and fat oxidation through the liver.
Weight loss data. The Phase 2 trial data is remarkable. Participants on the highest dose lost up to 24.2% of body weight over 48 weeks. The Phase 3 TRIUMPH-4 trial has reported even more impressive results, with participants achieving up to 28.7% body weight reduction. That puts retatrutide potentially ahead of every currently available medication, including tirzepatide.
The retatrutide versus tirzepatide comparison is compelling. The retatrutide versus semaglutide data shows an even wider gap. And the three-way comparison puts the hierarchy into clear perspective.
Dosing. The retatrutide dosage chart follows a titration protocol starting at lower doses. The dose schedule typically begins at 1 mg weekly, titrating up to 8-12 mg over several weeks. The starting dose recommendations emphasize slow titration to manage gastrointestinal side effects, which are more frequent with the triple agonist mechanism.
Side effects. Higher rates of gastrointestinal adverse events compared to tirzepatide. Nausea, diarrhea, and vomiting occur more frequently, particularly during dose escalation. Hair loss concerns have been raised, similar to other aggressive weight loss interventions. The trade-off between greater efficacy and increased side effect burden is real.
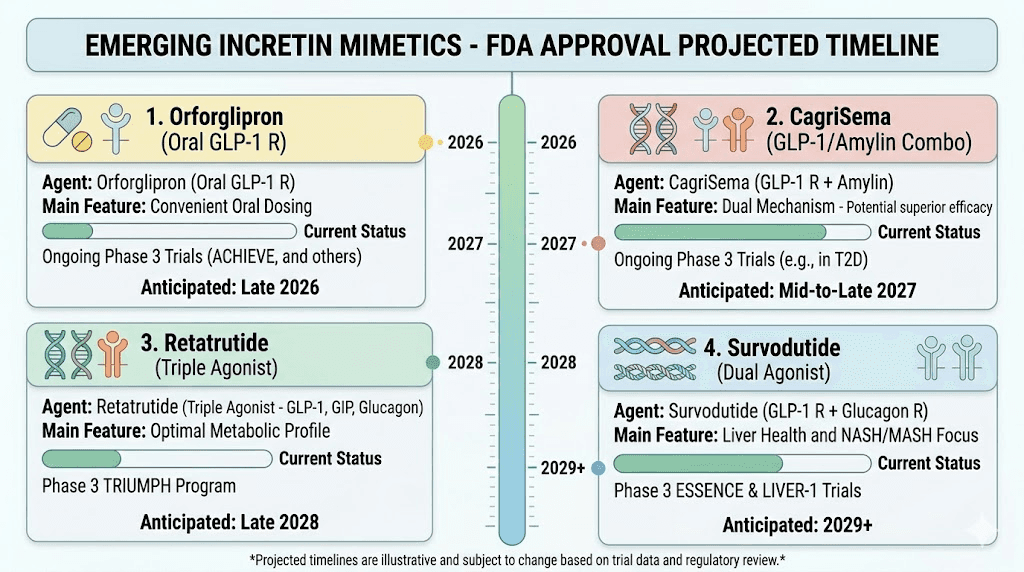
Availability. Retatrutide availability remains limited. It is not FDA-approved. Research-grade retatrutide can be found through certain channels, and our guide on where to buy retatrutide covers the options. The cost for research-grade product varies widely. FDA approval could come as early as mid-2026 or early 2027 if Phase 3 trials continue their positive trajectory.
For those switching from tirzepatide to retatrutide, the transition protocol involves careful consideration of the timeline for retatrutide to take effect after discontinuing tirzepatide.
Best for: People who want the most aggressive weight loss option and are willing to accept higher side effect rates and the current lack of FDA approval.
CagriSema (cagrilintide plus semaglutide)
CagriSema takes a different approach to exceeding tirzepatide results. Rather than targeting more receptors with a single molecule, it combines two separate compounds, cagrilintide (an amylin analogue) and semaglutide (a GLP-1 agonist), into a single weekly injection.
Weight loss data. The REDEFINE 1 trial published results showing 20.4% mean body weight reduction at 68 weeks. That matches or slightly exceeds tirzepatide performance at the same timepoint. Remarkably, 60% of participants achieved at least 20% weight loss, and 23% lost 30% or more of their body weight.
For context, semaglutide alone achieved 14.9% weight loss in the same trial. Cagrilintide alone achieved 11.5%. The combination delivers substantially more than either component individually, suggesting genuine synergy between the amylin and GLP-1 pathways.
How amylin adds to GLP-1. Amylin is a hormone co-secreted with insulin from pancreatic beta cells. It slows gastric emptying, suppresses glucagon secretion, and promotes satiety through direct brain signaling. These mechanisms complement GLP-1 effects through different neural and hormonal pathways, creating a more comprehensive appetite suppression signal than GLP-1 alone.
The CagriSema versus tirzepatide comparison is particularly interesting because both achieve similar weight loss magnitudes through completely different receptor combinations. This means that if you do not respond well to GIP receptor activation (which tirzepatide relies on), CagriSema amylin pathway may work where tirzepatide did not.
Side effects. Gastrointestinal adverse events affected 79.6% of the CagriSema group in REDEFINE 1, compared to 39.9% with placebo. Nausea, vomiting, diarrhea, constipation, and abdominal pain were mainly transient and mild to moderate in severity. The side effect profile is broadly similar to tirzepatide, though the specific character of symptoms may differ due to the amylin component.
Availability. CagriSema has completed Phase 3 trials and Novo Nordisk is expected to file for FDA approval. It could become available within 12-18 months. Currently, there is no way to access it outside of clinical trial settings.
Best for: People who want tirzepatide-level results through a completely different mechanism, especially those who may not respond well to GIP receptor activation.
Survodutide
Survodutide is a dual glucagon/GLP-1 receptor agonist developed by Boehringer Ingelheim. Like tirzepatide, it targets two receptors simultaneously. But instead of pairing GLP-1 with GIP, survodutide pairs GLP-1 with the glucagon receptor. This creates a fundamentally different metabolic profile.
The survodutide versus tirzepatide comparison highlights how different receptor pairings produce different metabolic effects despite comparable weight loss magnitudes.
Weight loss data. Phase 2 trial results demonstrated up to 19% body weight reduction after 46 weeks at the highest dose (4.8 mg). That is approaching tirzepatide territory, particularly considering the shorter trial duration. Phase 3 trials (SYNCHRONIZE-1 and SYNCHRONIZE-2) are currently underway and will provide the definitive comparison data.
The glucagon advantage. Unlike GIP receptor activation, glucagon receptor activation directly stimulates hepatic fat oxidation and thermogenesis. This means survodutide may provide additional benefits for fat loss specifically, particularly visceral and liver fat, beyond what GIP/GLP-1 dual agonism achieves. Early data shows significant improvements in liver fat content, which has implications for metabolic health beyond weight loss alone.
Side effects. The gastrointestinal side effect profile is consistent with other GLP-1 based therapies. Nausea, diarrhea, and vomiting are the most common adverse events, particularly during dose escalation. The glucagon receptor component may also contribute to mild increases in heart rate in some individuals.
Availability. Not yet FDA-approved. Phase 3 trial results expected in the coming months. If positive, approval could follow within 18-24 months.
Best for: People interested in a dual agonist with a different receptor profile than tirzepatide, particularly those concerned about liver fat or metabolic health.
Mazdutide
Mazdutide is another GLP-1/glucagon dual receptor agonist, developed by Innovent Biologics. It shares the same receptor targets as survodutide but has a different molecular structure and pharmacokinetic profile. The mazdutide versus tirzepatide comparison and the mazdutide versus retatrutide analysis provide detailed breakdowns.
Weight loss data. The GLORY-2 study showed mazdutide 9 mg achieving up to 20.1% weight loss in Chinese adults with obesity over one year. A meta-analysis across randomized controlled trials found significant reductions in body weight averaging 12.4%, BMI by 4.0 kg/m2, and waist circumference by 8.0 cm. These results are notable because mazdutide achieves over 20% weight loss with just a 2-step dose titration, compared to the more complex titration schedules required by other medications.
Additional benefits. Mazdutide demonstrates improvements in blood lipids, blood pressure, uric acid levels, liver enzymes, and liver fat content. The glucagon receptor activation enhances hepatic fat metabolism, similar to survodutide, providing metabolic benefits that extend well beyond the number on the scale.
Availability. Mazdutide has received approval in China for type 2 diabetes. Global Phase 3 trials for obesity are progressing. Approval outside of China likely remains 2-3 years away.
Best for: People looking at the global pipeline of alternatives and considering future options with strong metabolic profiles.
Orforglipron: the oral revolution
Orforglipron represents something entirely different. It is not a peptide at all. It is a small-molecule oral GLP-1 receptor agonist developed by Eli Lilly, the same company behind tirzepatide. And it could fundamentally change how weight loss medications are delivered.
The distinction between oral versus injectable medications matters enormously for patient adherence. Many people who would benefit from GLP-1 therapy never start or discontinue early because of needle aversion. Tablet formulations remove that barrier entirely.
Weight loss data. The Phase 3 ATTAIN-1 trial showed the 36 mg dose achieved 11.2% mean body weight reduction over 72 weeks. Among those on the highest dose, 54.6% achieved at least 10% weight loss, 36.0% achieved 15% or more, and 18.4% achieved 20% or more. These numbers are lower than tirzepatide injectable results, but the convenience of a daily pill with no food or water restrictions changes the risk-benefit calculation considerably.
Orforglipron also demonstrated superiority over oral semaglutide (Rybelsus) in a head-to-head trial for blood sugar control and weight loss in type 2 diabetes, delivering better results with fewer food and water intake restrictions.
The convenience factor. Unlike oral semaglutide (which requires taking the pill on an empty stomach with limited water and waiting 30 minutes before eating), orforglipron has no food or water restrictions. Take it any time. With or without food. This is a massive practical advantage that improves real-world adherence.
It also opens the door for people who have tried tirzepatide drops or orally disintegrating tablets and want a fully oral option backed by Phase 3 data.
Side effects. Consistent with other GLP-1 receptor agonists. Constipation, nausea, diarrhea, and vomiting are the most common adverse events. The oral route does not eliminate gastrointestinal side effects because they stem from the systemic GLP-1 receptor activation, not the route of administration.
Availability. Not yet FDA-approved. Eli Lilly has completed multiple Phase 3 trials and is expected to file for approval. Market availability could come within 12-18 months.
Best for: People who want to avoid injections entirely, or those looking for a convenient daily oral alternative with competitive weight loss results.
Complete comparison table: all tirzepatide alternatives
Numbers tell the story faster than paragraphs. This table compares every significant alternative across the metrics that matter most when choosing a replacement for tirzepatide.
Medication | Receptor targets | Weight loss | Dosing | FDA approved | Monthly cost |
|---|---|---|---|---|---|
Tirzepatide (reference) | GLP-1 + GIP | 20-22% | Weekly injection | Yes | $1,000+ |
Semaglutide injectable | GLP-1 | 15% | Weekly injection | Yes | $1,300+ |
Semaglutide oral | GLP-1 | 15% | Daily pill | Yes | $1,300+ |
Liraglutide | GLP-1 | 5-10% | Daily injection | Yes | $1,200+ |
Retatrutide | GLP-1 + GIP + Glucagon | 24-29% | Weekly injection | No (Phase 3) | Research only |
CagriSema | GLP-1 + Amylin | 20% | Weekly injection | No (Phase 3 complete) | Not available |
Survodutide | GLP-1 + Glucagon | 19% | Weekly injection | No (Phase 3) | Not available |
Mazdutide | GLP-1 + Glucagon | 20% | Weekly injection | China only | Not available (US) |
Orforglipron | GLP-1 | 11% | Daily pill | No (Phase 3 complete) | Not available |
Several patterns emerge from this data. First, the next generation of medications, retatrutide, CagriSema, survodutide, and mazdutide, all approach or exceed tirzepatide weight loss results. Second, the gap between currently available alternatives (semaglutide at 15%) and tirzepatide (20-22%) may close significantly within the next 1-2 years as these newer options reach the market. Third, oral options like orforglipron trade some efficacy for dramatically improved convenience.
Use the peptide cost calculator to compare your actual costs across different options based on your specific dosing needs.
Non-GLP-1 alternatives
Not every alternative needs to target incretin pathways. Several FDA-approved weight loss medications work through completely different mechanisms. These become relevant when GLP-1 class medications are contraindicated, when you want to combine approaches, or when the GLP-1 pathway simply does not produce adequate results for your biology.
Phentermine-topiramate (Qsymia)
This combination medication pairs an appetite suppressant (phentermine) with an anticonvulsant (topiramate) that has weight loss properties. Clinical trials show average weight loss of 7-10% of body weight over one year at the highest dose.
The phentermine versus semaglutide comparison shows that while phentermine-based medications produce less weight loss than GLP-1 agonists, they work through completely different pathways. The mechanism is distinct from GLP-1 class drugs. Phentermine acts as a sympathomimetic amine, releasing norepinephrine and dopamine to suppress appetite centrally. Topiramate modulates GABA receptors and voltage-gated ion channels.
Some prescribers combine phentermine with GLP-1 agonists for enhanced effect. The phentermine and tirzepatide combination and phentermine and semaglutide combination are increasingly common in clinical practice, though this represents off-label use.
Best for: People who cannot use any GLP-1 class medication, or as an add-on to boost results.
Naltrexone-bupropion (Contrave)
Contrave combines an opioid antagonist (naltrexone) with an antidepressant (bupropion). The combination targets the brain reward pathways involved in food cravings, particularly emotional and hedonic eating. Average weight loss in clinical trials is approximately 5-6% of body weight.
This medication is particularly useful for people whose weight management challenges are primarily driven by cravings, emotional eating, or food addiction rather than hunger. The mechanism is complementary to GLP-1 effects, and some practitioners use it alongside peptide-based weight loss approaches.
Best for: People whose primary challenge is food cravings or emotional eating rather than physical hunger.
Metformin
Metformin is not FDA-approved for weight loss, but it produces modest weight reduction (2-3% of body weight) as a side effect of its primary mechanism, improving insulin sensitivity and reducing hepatic glucose production. It is inexpensive, widely available, extremely well-studied, and has a favorable safety profile spanning decades of use.
Metformin is often used as a supportive medication alongside GLP-1 agonists rather than as a standalone alternative. Its role in the alternatives landscape is more about metabolic optimization than aggressive weight loss.
Best for: People with insulin resistance or pre-diabetes who want a well-established, affordable metabolic support medication.
Orlistat (Xenical, Alli)
Orlistat works by blocking fat absorption in the digestive tract, preventing roughly 30% of dietary fat from being absorbed. Average weight loss is 5-7% of body weight. The gastrointestinal side effects (oily stools, flatulence, fecal urgency) are well-documented and directly tied to dietary fat intake.
This is the only weight loss medication that works entirely in the gut without systemic effects. It requires no prescription at lower doses (Alli 60 mg), making it the most accessible option for people who cannot or do not want to see a prescriber.
Best for: People who want an over-the-counter option or cannot use systemic medications.
Lifestyle and supplement approaches
Medication is not the only path. Some people seek tirzepatide alternatives specifically because they want to move away from pharmaceutical approaches. Others want to combine lifestyle interventions with lower-dose medications for a more balanced strategy.
Berberine
Berberine activates AMPK (AMP-activated protein kinase), a metabolic master switch that influences glucose uptake, fat metabolism, and insulin sensitivity. Clinical studies show modest weight loss of 2-5 pounds over 12 weeks, with more significant improvements in metabolic markers like fasting glucose and HbA1c. The berberine and semaglutide combination has gained attention as a way to enhance GLP-1 effects naturally.
GLP-1 supporting supplements
Several supplements claim to support natural GLP-1 production. Pendulum GLP-1 Probiotic Pro contains bacterial strains shown to increase endogenous GLP-1 secretion. The evidence is early but promising, and these products represent the mildest possible alternative to pharmaceutical GLP-1 agonists.
Other supplements that support metabolic function during weight loss include B12, which is commonly compounded with GLP-1 medications. The semaglutide with B12 and tirzepatide with B12 combinations are popular for addressing the energy and neurological support needs during aggressive caloric restriction. Supplements to take alongside GLP-1 medications can make any alternative more effective.
High-protein, calorie-controlled diet
The most fundamental alternative to any weight loss medication is dietary modification. The tirzepatide diet plan principles apply broadly: high protein intake (0.7-1.0 g per pound of body weight), adequate fiber, strategic meal timing, and controlled caloric deficit. The foods to avoid and optimal foods guidance translates directly to any weight management approach, with or without medication.
Weight loss without exercise is possible through diet alone, but combining dietary changes with regular physical activity improves outcomes across every metric, from fat loss to muscle preservation to metabolic health.
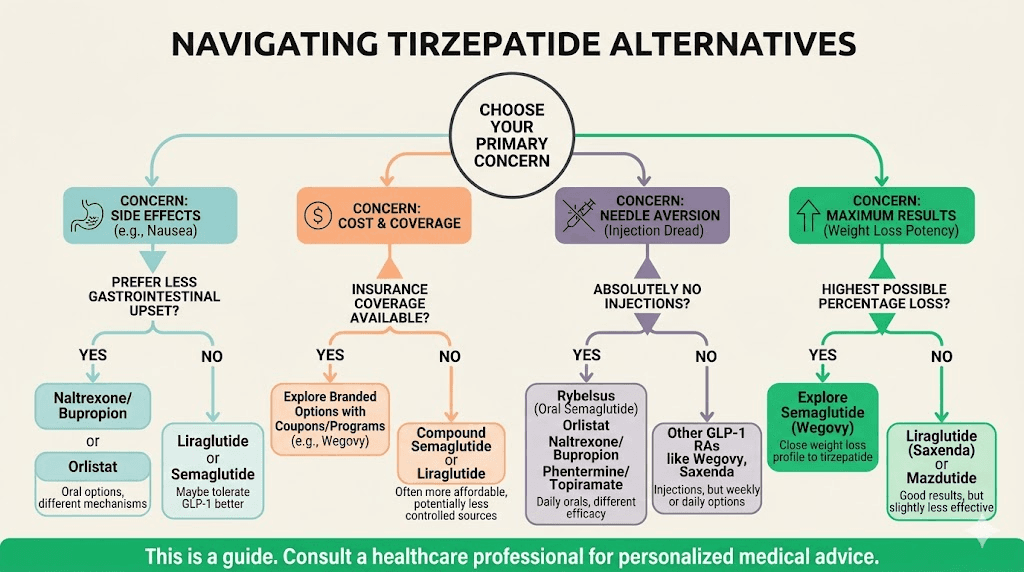
How to choose the right alternative for your situation
The sheer number of options can feel overwhelming. Here is a decision framework based on your specific circumstances.
If you stopped tirzepatide because of side effects
Try semaglutide first. It covers one of the same receptor pathways but eliminates the GIP component, which may be responsible for your specific side effects. The switching protocol is well-established. Start at a low dose regardless of your previous tirzepatide dose. If semaglutide produces similar side effects, the issue is likely GLP-1 related rather than GIP related, and a non-GLP-1 alternative like Qsymia or Contrave becomes the better path.
If tirzepatide stopped working
Distinguish between true pharmacological tolerance and a weight loss plateau caused by metabolic adaptation. Most "plateaus" on tirzepatide are actually expected metabolic slowdowns at lower body weights, not medication failure. The full troubleshooting guide covers dose optimization, diet adjustments, and exercise modifications before switching.
If you have genuinely exhausted optimization on tirzepatide, retatrutide (when available) offers additional receptor activation that may overcome the plateau. CagriSema approaches the problem through an entirely different receptor combination. Adding metformin or phentermine to your existing tirzepatide may also restart progress without switching entirely.
If cost is the primary driver
Compounded semaglutide (where available) at $150-300 per month. Generic liraglutide if accessible. Phentermine-topiramate (Qsymia) which has generic availability for some components. Qualifying for insurance coverage of brand-name medications through type 2 diabetes or cardiovascular indications rather than weight loss alone can also dramatically reduce costs.
If you want to avoid injections
Oral semaglutide (Rybelsus or oral Wegovy) is available now. Orforglipron will likely be available within 12-18 months with no food or water restrictions. Qsymia and Contrave are both oral. Sublingual semaglutide offers another non-injection route.
If you want the most aggressive weight loss possible
Retatrutide leads the field at 24-29% weight loss in trials. CagriSema at 20% matches tirzepatide. If you need something available right now, high-dose semaglutide (2.4 mg) combined with phentermine-topiramate represents the most aggressive currently available combination, though this requires a prescriber willing to manage a multi-drug regimen.
Side effect comparison across all alternatives
Side effects often determine which alternative you actually stick with long-term, regardless of efficacy numbers. This section compares the most clinically relevant adverse events across every major option.
Gastrointestinal effects
Every GLP-1 based medication shares a core set of gastrointestinal side effects because they stem from the mechanism of action itself, slowing gastric emptying and altering gut motility. The frequency and severity vary by compound.
Tirzepatide: nausea (40-50%), diarrhea (15-25%), constipation (10-20%), vomiting (10-15%). Most resolve within 4-6 weeks. Diarrhea duration and constipation management are well-documented.
Semaglutide: similar rates overall. Bloating may be more prominent with semaglutide specifically. Constipation treatment approaches are similar across the class.
Retatrutide: higher overall rates due to triple receptor activation. The glucagon component adds its own gastrointestinal effects on top of the GLP-1 and GIP effects.
CagriSema: 79.6% gastrointestinal adverse event rate in REDEFINE 1. The amylin component contributes additional nausea beyond what GLP-1 alone produces.
Non-GLP-1 alternatives have completely different side effect profiles. Qsymia can cause tingling, taste changes, and cognitive effects. Contrave may cause headache, constipation, and insomnia. Orlistat causes gastrointestinal effects (oily stools, flatulence) but only when dietary fat is consumed.
Fatigue and energy
Tiredness on tirzepatide is common, primarily due to reduced caloric intake rather than direct medication effects. Semaglutide fatigue and GLP-1 fatigue generally follow the same pattern. Switching between GLP-1 medications rarely resolves fatigue because the cause is caloric deficit, not a specific drug. Energy levels on semaglutide and energy levels on tirzepatide tend to improve once body weight stabilizes.
Hair loss
GLP-1 related hair loss occurs with any medication that causes rapid weight loss. It is telogen effluvium, a temporary shedding triggered by physiological stress, not permanent hair loss. The rate correlates with the speed and magnitude of weight loss rather than the specific medication used. Retatrutide, with its more aggressive weight loss, may produce more noticeable hair thinning than milder alternatives.
Reproductive effects
Period changes on tirzepatide and semaglutide are well-documented. Increased fertility is a real concern. Breastfeeding considerations and GLP-1 use during lactation require careful medical guidance regardless of which alternative you choose.
Switching protocols: how to transition safely
Switching from tirzepatide to any alternative requires a thoughtful approach. Abrupt discontinuation followed by immediate full-dose initiation of a new medication increases side effect risk and may cause a temporary rebound in appetite and weight.
General principles
Wait for the last tirzepatide dose to clear (approximately 5 half-lives, or about 25 days for tirzepatide) before expecting full effects from the new medication. However, most prescribers start the new medication 7 days after the last tirzepatide injection to prevent a gap in appetite suppression.
Start any new GLP-1 medication at the lowest available dose regardless of your previous tirzepatide dose. Your gastrointestinal system needs time to adjust to the new compound even if the mechanism is similar.
The weaning protocol for tirzepatide may be appropriate if you are discontinuing GLP-1 therapy entirely rather than switching. Gradual dose reduction over 4-8 weeks helps minimize withdrawal-like effects including appetite rebound and rapid weight regain.
Tirzepatide to semaglutide
The most common switch. Use the conversion chart as a reference, but always start semaglutide at 0.25 mg weekly for the first month. Even if you were on high-dose tirzepatide, the semaglutide-specific side effect profile requires independent titration. Most people reach their target semaglutide dose within 16-20 weeks following the standard titration schedule.
Tirzepatide to retatrutide
The tirzepatide to retatrutide transition requires extra caution because retatrutide adds glucagon receptor activation. Start at 1 mg weekly and titrate slowly. The onset timeline varies, but most people notice appetite suppression effects within the first 1-2 weeks.
Maintaining weight during the transition
The transition period between medications is the highest risk window for weight regain. Maintain strict dietary adherence during this time. Track everything. Keep protein high. The optimal food choices and foods to avoid apply to any GLP-1 transition period.
SeekPeptides members access detailed transition protocols, dose conversion guides, and personalized support for navigating medication switches safely and effectively.
The future of tirzepatide alternatives
The weight loss medication pipeline is more active than any other therapeutic area in pharmaceutical development. Several additional compounds deserve mention for anyone planning their long-term strategy.
Pemvidutide
A balanced 1:1 GLP-1/glucagon dual agonist showing particular promise for liver fat reduction. The Phase 2b IMPACT trial demonstrated MASH resolution without worsening fibrosis, with weight losses of 4-6% over 24 weeks at current doses. The FDA granted both Fast Track and Breakthrough Therapy designations, indicating regulatory priority. While the weight loss numbers are currently modest compared to tirzepatide, the liver-specific benefits could make pemvidutide particularly valuable for people with fatty liver disease.
Ecnoglutide
A long-acting GLP-1 receptor agonist designed for twice-monthly or potentially monthly dosing. Less frequent injections could significantly improve adherence for people who find weekly injections burdensome.
Combination approaches
The trend toward combining compounds with different mechanisms is accelerating. Beyond CagriSema, researchers are exploring GLP-1 combined with FGF21 analogues, GLP-1 combined with leptin sensitizers, and even triple or quadruple receptor agonists. The next decade will likely see weight loss medications that routinely achieve 30%+ body weight reduction with acceptable tolerability.
Staying informed about these developments helps you plan your long-term weight management strategy. What is cutting-edge research today becomes clinical practice within a few years.
Practical considerations when switching
Beyond the pharmacology, several practical factors influence which alternative works best in your daily life.
Storage and handling
All injectable GLP-1 medications require refrigeration. Temperature excursions compromise potency. If you travel frequently, cold chain maintenance is a practical consideration regardless of which medication you use. Oral alternatives like orforglipron (when available) or Qsymia eliminate storage concerns entirely.
Injection technique
If you are switching between injectable options, the best injection sites, injection location guidance, and stomach injection technique remain largely the same. The injection site reactions that occurred with tirzepatide may or may not recur with a new medication.
Monitoring during transition
Weigh yourself under consistent conditions (same time, same clothing) throughout the transition. Track appetite changes subjectively on a 1-10 scale. Monitor blood glucose if you have diabetes or pre-diabetes. Report any unusual symptoms to your prescriber promptly. The first week on a new GLP-1 establishes your baseline response.
Alcohol and lifestyle interactions
Alcohol tolerance changes on GLP-1 medications are well-documented. Alcohol on semaglutide produces similar effects. Switching medications does not reset your altered alcohol sensitivity. Continue to exercise caution with alcohol regardless of which alternative you use.
For researchers serious about optimizing their weight management protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, detailed dosing charts, calculators, and a community of thousands who have navigated these exact transitions.
Frequently asked questions
What is the closest drug to tirzepatide?
Semaglutide (Wegovy/Ozempic) is the closest FDA-approved alternative, targeting the GLP-1 receptor that tirzepatide also activates. However, CagriSema and retatrutide (both in late-stage development) more closely match tirzepatide weight loss results, with CagriSema achieving 20% and retatrutide achieving up to 29% body weight reduction in trials.
Can I switch from tirzepatide to semaglutide without gaining weight?
Most people maintain their weight during the transition if they follow proper switching protocols, maintain dietary adherence, and titrate semaglutide appropriately. Some temporary weight fluctuation (1-3 pounds) is normal during the transition period as your body adjusts to the new medication.
Is retatrutide better than tirzepatide?
Clinical trial data suggests retatrutide produces greater weight loss than tirzepatide (24-29% vs 20-22%), but with higher rates of side effects. Retatrutide is not yet FDA-approved, so direct comparison in real-world use is not yet possible. "Better" depends on your tolerance for side effects and your weight loss goals.
What is the best oral alternative to tirzepatide?
Currently, oral semaglutide (Rybelsus or oral Wegovy) is the only FDA-approved oral GLP-1 option. Orforglipron, expected to reach the market within 12-18 months, showed competitive results in Phase 3 trials with the added convenience of no food or water restrictions. For non-GLP-1 oral options, Qsymia provides the strongest weight loss results among oral alternatives.
How long should I wait between stopping tirzepatide and starting a new medication?
Most prescribers recommend starting the new medication 7 days after your last tirzepatide injection. Tirzepatide has a half-life of approximately 5 days, so substantial levels remain in your system for 2-3 weeks after the last dose. Starting the new medication within a week prevents a significant gap in appetite suppression.
Will I need to change my diet when switching medications?
The core dietary principles remain the same regardless of medication: high protein, adequate fiber, controlled calories. You may need to adjust meal sizes during the transition if appetite changes differ between medications. The optimal food choices for GLP-1 therapy apply universally across the class.
Are compounded versions of these alternatives safe?
Compounded semaglutide from FDA-registered 503B pharmacies following USP-797 standards can be safe, though compounded medications are not FDA-approved. Verify that any compounding pharmacy is properly licensed, uses third-party testing, and follows current good manufacturing practices. The grey market carries significant risks and should be approached with extreme caution.
Can I combine tirzepatide alternatives for better results?
Some combinations are used in clinical practice. Phentermine with semaglutide is increasingly common. Using multiple GLP-1 medications simultaneously is generally not recommended due to overlapping mechanisms and compounded side effect risk. Always discuss combination approaches with your prescriber.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your alternatives stay effective, your transitions stay smooth, and your results stay lasting.