Mar 25, 2026

Before you take another dose, stop and think about something most guides never mention. Semaglutide and pregnancy planning do not mix the way most people assume they do. The relationship between this GLP-1 medication and fertility is complicated, contradictory, and surprisingly nuanced. Some women struggle for years to conceive, start semaglutide for weight loss, and find themselves pregnant within months. Others take it without realizing they need to stop well before trying to conceive. The stakes here are not abstract. They involve a developing life, your body, and decisions that require real information rather than guesswork.
The short answer is no, you should not take semaglutide while actively trying to get pregnant. The FDA and all major manufacturers recommend stopping the medication at least two months before planned conception. But the full picture involves far more than a simple yes or no. It involves the mechanism of how semaglutide works, its half-life in your body, what animal studies actually showed, what human data we have so far, and a phenomenon called "Ozempic babies" that has changed how fertility specialists think about GLP-1 weight loss medications entirely.
SeekPeptides created this guide to walk you through every aspect of semaglutide and pregnancy planning, from the science behind the washout period to practical steps for women who want to use semaglutide strategically before conception.
Why semaglutide and pregnancy do not mix
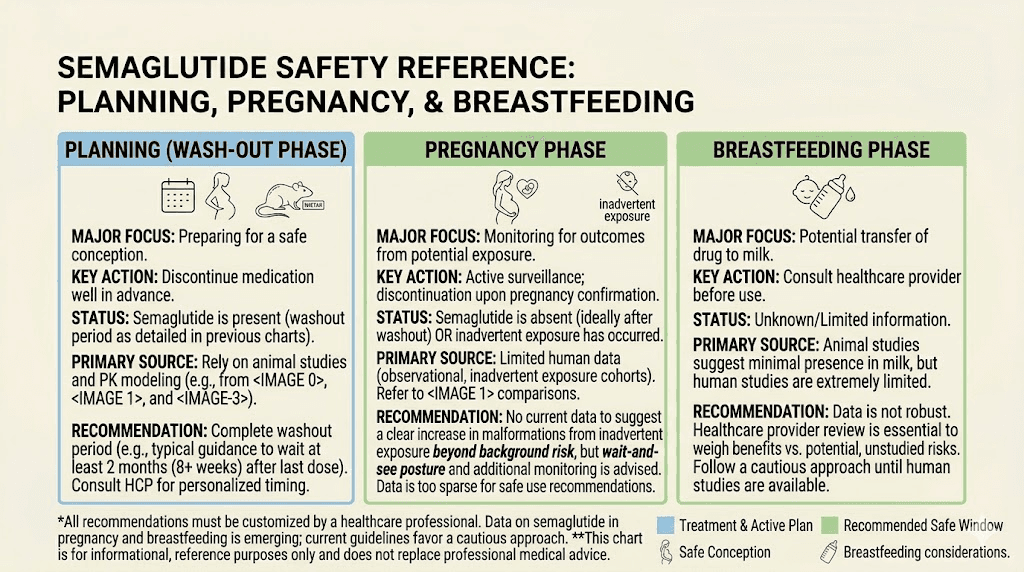
The core issue is straightforward. Semaglutide has not been proven safe for use during pregnancy in humans. That does not mean it has been proven dangerous. It means the data simply does not exist to confirm safety, and when it comes to a developing fetus, the medical standard is extreme caution. No responsible healthcare provider will tell you to continue semaglutide dosing while trying to conceive.
Here is why.
Semaglutide belongs to a class of medications called GLP-1 receptor agonists. These drugs mimic a hormone your body naturally produces to regulate blood sugar, appetite, and metabolism. The injectable forms, sold as Ozempic for type 2 diabetes and Wegovy for weight management, along with the oral form Rybelsus, all contain the same active compound. And that compound stays in your body for a long time. The half-life of semaglutide is approximately one week, meaning it takes roughly six weeks for most of the drug to clear your system after your last injection. This extended presence in the body is one of the primary reasons the timeline for semaglutide to take effect is gradual, and why it lingers long after you stop taking it.
The two-month washout period recommended by Novo Nordisk, the manufacturer, accounts for this extended clearance time plus an additional safety margin.
What animal studies revealed about semaglutide and fetal development
The reason for caution comes primarily from animal research. Studies conducted on pregnant rats, rabbits, and cynomolgus monkeys produced findings that warranted serious attention, even though the relevance to humans remains debated.
In rat studies, semaglutide administered during pregnancy caused increased fetal death and structural abnormalities. Researchers observed reduced growth, abnormal heart blood vessels, and changes in skeletal structures including cranial bones, vertebra, and ribs. These effects occurred at maternal exposures below the maximum recommended human dose.
Rabbit studies told a similar story. Early pregnancy losses, structural abnormalities, and reduced fetal weight were observed at doses equivalent to clinical human exposure. Again, these findings appeared at doses below what humans typically receive for standard semaglutide treatment.
In cynomolgus monkeys, early pregnancy losses and structural abnormalities appeared at exposures roughly two-fold the recommended human dose. Reduced fetal weight was also documented.
But context matters enormously here. In all cases, these findings coincided with maternal body weight loss due to reduced food intake from semaglutide use. Mechanistic studies concluded that the effects on embryo-fetal development in rats were caused by a GLP-1 receptor-mediated impairment of the inverted yolk sac placenta. This is a structure that plays a critical role in rat embryonic nutrition but does not have the same function in human pregnancy. The GLP-1 receptor-related mechanism by which semaglutide causes embryotoxicity in rats is unlikely to be relevant to humans.
That last point is crucial. The mechanism that caused harm in rats does not apply to human physiology. But "unlikely to be relevant" is not the same as "definitely safe," and that distinction drives every recommendation you will find from medical authorities.
What human data actually shows
Human data on semaglutide exposure during pregnancy remains limited, but what exists is more reassuring than the animal studies might suggest.
One case report documented a woman who took semaglutide during the first three to four weeks of pregnancy before discovering she was pregnant. Her child was born without reported defects. A single case proves nothing, but it adds to the growing picture.
A larger study examined 168 pregnant women, including 51 who used semaglutide. No increased risk of birth defects was reported compared to the general population baseline of approximately 3% for all pregnancies.
A Danish study from recent research examined 32 pregnancies exposed to semaglutide and insulin, finding comparable risks of malformations, preterm birth, and neonatal hypoglycemia compared to pregnancies exposed solely to insulin. This matters because it suggests semaglutide exposure did not add measurable risk beyond what was already present from the underlying condition being treated.
A systematic review published in 2025 analyzed five studies involving 1,128 semaglutide-exposed pregnancies. The findings were mixed but notable. No consistent link was found between semaglutide use and major congenital malformations. Preterm birth, macrosomia, and neonatal hypoglycemia were observed in some cases, but researchers could not establish clear causal links after accounting for comorbidities like diabetes and obesity, which independently increase these risks. The review concluded that further large-scale studies are needed to clarify safety.
So the picture is this: animal studies show cause for caution through a mechanism that probably does not apply to humans. Limited human data has not found increased birth defect risk. But the data is not robust enough to declare semaglutide safe during pregnancy, and it may never be, since conducting controlled studies on pregnant women with unproven medications raises obvious ethical barriers.

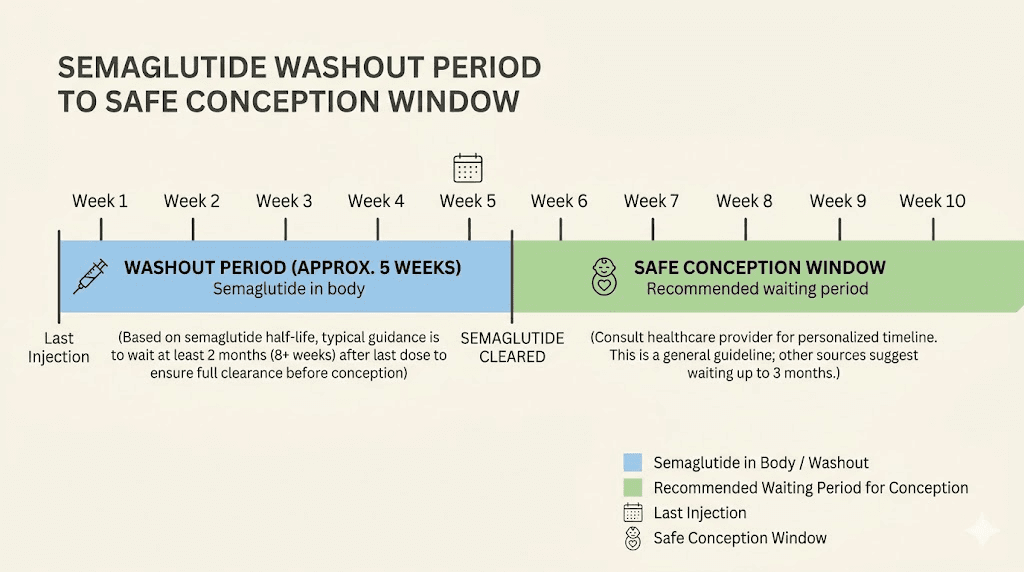
The two-month washout rule explained
Every major semaglutide product label, whether Ozempic, Wegovy, or Rybelsus, carries the same recommendation: women planning pregnancy should stop the medication at least two months before attempting to conceive.
Why two months specifically?
The math works like this. Semaglutide has a half-life of approximately seven days. After one week without a dose, half the drug remains in your system. After two weeks, roughly a quarter remains. By six weeks, the vast majority has been eliminated. The two-month recommendation adds approximately two additional weeks beyond the standard clearance time as a safety buffer.
This timeline applies to all forms of semaglutide. Whether you have been using compounded semaglutide, brand-name injectables, or oral semaglutide, the washout period remains the same because the active compound is identical regardless of delivery method.
Some practitioners recommend an even longer washout of three months for patients who have been on higher doses or who have been using the medication for extended periods. The reasoning is that higher semaglutide dosages may take slightly longer to fully clear, though the pharmacokinetic data suggests six weeks covers the vast majority of clearance regardless of dose.
What the washout period feels like
Stopping semaglutide is not without consequences. Many women experience what amounts to semaglutide withdrawal, though the term is somewhat misleading since semaglutide is not physically addictive. What happens is that appetite suppression fades, hunger returns, and the metabolic benefits gradually diminish.
During the washout period, you may notice increased appetite within the first two to three weeks. Fatigue can occur as your body readjusts. Some women report bloating or digestive changes as the GLP-1 effects wear off. Weight regain is possible, though not inevitable if you have established strong nutritional habits during treatment.
Planning for these changes matters. The washout period is not just about waiting for the drug to leave your system. It is about transitioning your body into a state that is ready for pregnancy, which means maintaining the nutritional and lifestyle improvements you made during treatment while your appetite regulation system returns to baseline.
The Ozempic babies phenomenon
Something unexpected happened when millions of women started taking GLP-1 medications for weight loss. Women who had struggled with infertility for years, some who had been told they might never conceive naturally, started getting pregnant. The media called them "Ozempic babies."
This is not a myth. It is not marketing. Fertility specialists across the country have documented the pattern, and the science behind it makes complete sense once you understand the connection between body weight, insulin resistance, and reproductive function.
The mechanism works through several interconnected pathways.
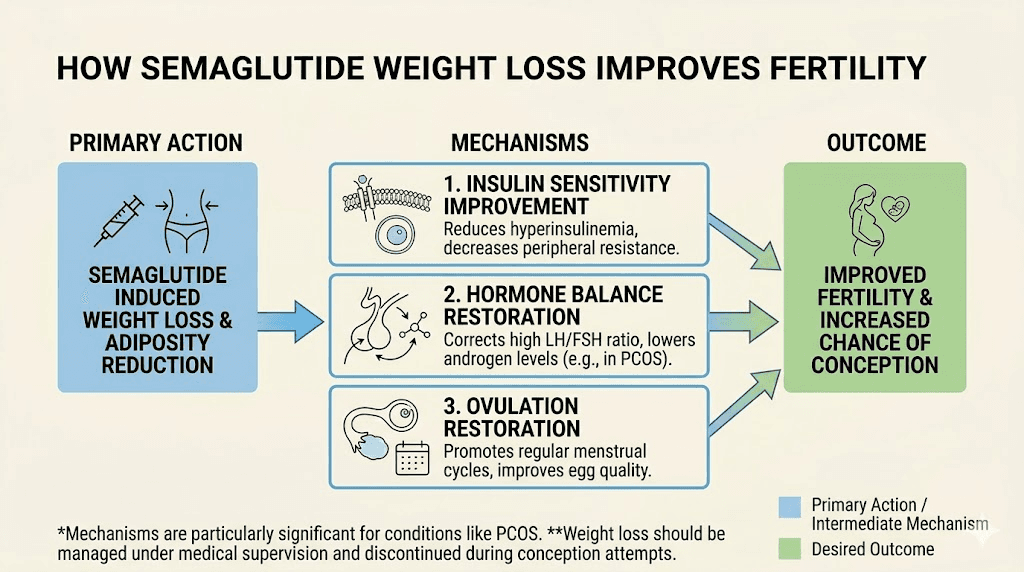
Weight loss restores ovulation
Excess body weight disrupts the hormonal balance required for regular ovulation. Fat tissue produces estrogen, and elevated estrogen levels can suppress the hypothalamic-pituitary-ovarian axis, the signaling pathway that controls your menstrual cycle. When women lose weight through semaglutide treatment, this hormonal balance often corrects itself. Research shows that losing just 5 to 10 percent of body weight can restore regular ovulation in women who were not ovulating.
This is not unique to semaglutide. Any method of significant weight loss can produce the same effect. But semaglutide makes substantial weight loss achievable for many women who could not achieve it through diet and exercise alone. The transformation that semaglutide enables extends far beyond the number on the scale.
Insulin resistance and fertility
Insulin resistance is one of the most significant but least discussed barriers to conception. When your body does not respond properly to insulin, your pancreas produces more of it to compensate. High insulin levels stimulate the ovaries to produce excess androgens, including testosterone, which interferes with normal follicle development and ovulation.
Semaglutide improves insulin sensitivity through multiple mechanisms. It enhances glucose-dependent insulin secretion, reduces glucagon release, and through weight loss, decreases the insulin resistance that comes with excess adipose tissue. Even in patients who do not achieve 5 percent weight loss, improvements in insulin resistance and blood glucose normalization have been documented, suggesting semaglutide has direct metabolic effects beyond what weight loss alone would produce.
PCOS and the fertility connection
The women most likely to experience the "Ozempic babies" effect are those with polycystic ovary syndrome. PCOS affects roughly 10 percent of women of reproductive age and is characterized by hormonal imbalance, insulin resistance, and irregular or absent ovulation. It is one of the leading causes of infertility.
The research on semaglutide and PCOS fertility is striking. In one prospective study, 60 percent of PCOS patients treated with semaglutide and metformin achieved pregnancy within the follow-up period. A total of 80 percent of patients who responded to treatment, defined as achieving at least 5 percent weight loss, normalized their menstrual cycles.
These numbers are remarkable. They do not mean semaglutide is a fertility treatment. It is not approved for that purpose, and no one should use it with the primary goal of getting pregnant. But they explain why so many women on semaglutide find themselves unexpectedly fertile, sometimes when they thought fertility was no longer possible.
Animal research has illuminated some of the molecular pathways involved. Studies show that GLP-1 receptor agonists regulate the ovarian PI3K/AKT pathway, affecting FoxO1 phosphorylation and follicular development. In PCOS animal models, both liraglutide and semaglutide were associated with reductions in insulin and androgen levels, decreased inflammation, lower testosterone and LH levels, and improvements in ovarian morphology. These changes create an environment where ovulation becomes possible again.
The birth control question
If semaglutide can improve fertility, the obvious question becomes: does it interfere with birth control?
The pharmacological answer is reassuring. A clinical study published in the Journal of Clinical Pharmacology found that semaglutide does not reduce the bioavailability of ethinylestradiol or levonorgestrel, the two most common components in combined oral contraceptive pills. Coadministration of semaglutide is not expected to affect the efficacy of oral contraceptive medications based on this data.
But there is a practical caveat that matters enormously.
Semaglutide causes nausea and vomiting, especially during the first weeks of treatment and after dose increases. If you vomit within a couple of hours after taking your birth control pill, your body may not have absorbed it. This creates a window of vulnerability that has nothing to do with drug interactions and everything to do with the gastrointestinal side effects of GLP-1 medications.
The practical recommendation for women on semaglutide who do not want to get pregnant: consider backup contraception methods that do not depend on oral absorption. IUDs, implants, injections, and barrier methods are not affected by nausea or vomiting. If you rely solely on the pill, follow your missed pill guidance any time you vomit shortly after taking it.
It is worth noting that tirzepatide has a different interaction profile. Unlike semaglutide, tirzepatide has been shown to reduce how much oral contraceptive the body absorbs by around 20 percent. The tirzepatide manufacturer specifically advises using backup birth control for four weeks after starting or increasing the dose, or switching to a non-oral contraceptive method entirely.
Strategic use of semaglutide before pregnancy
Here is where the conversation gets interesting and practical. While you cannot take semaglutide during pregnancy or while actively trying to conceive, using it strategically in the months before your conception window can significantly improve both fertility and pregnancy outcomes.
The pre-conception optimization approach
Think of semaglutide as a preparation tool rather than something you take through the process. The goal is to use the medication to achieve metabolic improvements and weight loss that persist after you stop taking it, creating the optimal conditions for conception and a healthy pregnancy.
The evidence supports this approach. Women who enter pregnancy at a healthier weight have lower risks of gestational diabetes, preeclampsia, cesarean delivery, and complications for the baby including macrosomia and birth defects associated with obesity. The weight loss achievable with semaglutide, often 15 to 20 percent of starting body weight, can transform pregnancy risk profiles.
Timeline for pre-conception semaglutide use
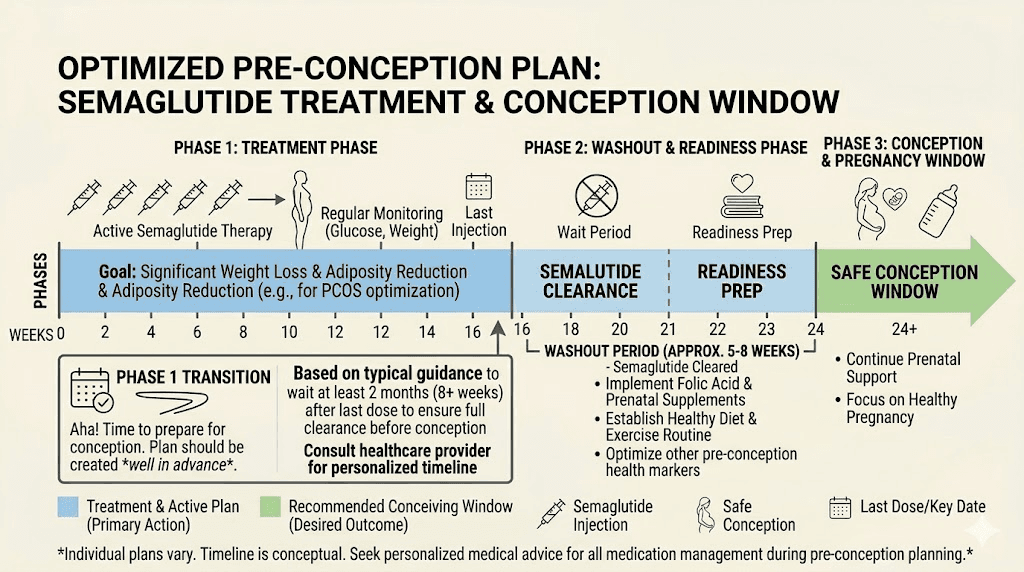
A practical timeline looks like this.
Months 1 through 6 (or longer): Use semaglutide for weight loss and metabolic optimization. Follow proper nutrition guidelines to build sustainable eating habits. Focus on nutrient-dense foods that support both weight loss and future pregnancy. Take prenatal vitamins during this phase to build nutrient stores. Monitor your menstrual cycle for signs of improved regularity.
Month 7 (two months before planned conception): Take your final semaglutide dose. Begin the washout period. Continue the eating patterns and exercise habits you established during treatment. Expect appetite to gradually return over two to three weeks. This is normal and expected.
Month 8: Continue washout. Most semaglutide should be cleared from your system by the end of this month. Focus on maintaining protein intake and nutritional quality. Monitor weight and adjust calorie intake if needed to prevent rapid regain.
Month 9 and beyond: Begin actively trying to conceive. Your body should be free of semaglutide, metabolically healthier, and at a weight that supports optimal fertility and pregnancy outcomes.
Maintaining weight loss after stopping
The biggest concern most women have about stopping semaglutide before trying to conceive is weight regain. This is a legitimate concern. Research shows that without intervention, many people regain a significant portion of lost weight after stopping GLP-1 medications.
But weight regain is not inevitable. The key is using the semaglutide treatment period to fundamentally change your relationship with food and activity. Knowing how many calories to consume and establishing those patterns while appetite suppression makes it easier is the strategy. When semaglutide wears off, those habits should be ingrained enough to maintain most of your progress.
Some practical approaches include maintaining a structured meal plan, avoiding foods that trigger overeating, and keeping protein intake high to preserve lean mass and promote satiety.
What happens if you get pregnant while on semaglutide
Despite best planning, unintended pregnancies happen. The Ozempic babies phenomenon proves this point. So what should you do if you discover you are pregnant while still taking semaglutide?
First, do not panic. The human data available, while limited, has not shown increased birth defect rates from early pregnancy semaglutide exposure. The animal studies that raised concerns involved mechanisms that are unlikely to apply to human pregnancy.
Second, stop taking semaglutide immediately. Do not take your next scheduled dose.
Third, contact your healthcare provider right away. They will want to know when your last dose was, how long you were taking the medication, what dose you were on, and your estimated date of conception. This information helps them assess potential exposure and plan appropriate monitoring.
The fact that semaglutide has a long half-life means some of the drug will remain in your system for weeks after your last dose, even if you stop immediately upon learning you are pregnant. This is unavoidable and is one of the reasons the two-month pre-conception washout exists. But the declining levels after cessation are very different from continued active dosing throughout early pregnancy.
Your healthcare provider may recommend additional monitoring, such as detailed ultrasounds to check fetal development, more frequent prenatal visits during the first trimester, blood glucose monitoring if you were using semaglutide for diabetes, and nutritional counseling to ensure adequate calorie and nutrient intake now that appetite suppression is wearing off.
The emotional component
Discovering an unplanned pregnancy while on any medication creates anxiety. This is normal and valid. But it helps to know that the available human evidence, while limited, does not support the catastrophic outcomes that internet searches might suggest. The 168-woman study, the Danish cohort data, and the systematic review of over 1,100 exposed pregnancies all point toward reassuring, if not definitive, safety signals.
What you should not do is spend your pregnancy in fear based on rat studies that investigated a mechanism irrelevant to human biology.
Work with your healthcare provider, follow their monitoring recommendations, and focus on the things you can control: nutrition, prenatal care, and stress management.
Semaglutide and male fertility
The pregnancy planning conversation usually focuses on women, but what about men? If the male partner is taking semaglutide, does he need to stop before trying to conceive?
Current evidence says no. Animal studies examining the effects of semaglutide on male reproductive function found no changes in male fertility at doses equivalent to human therapeutic levels. There is no recommendation for men to discontinue semaglutide before attempting to conceive with their partner.
That said, there are indirect ways semaglutide might affect male fertility positively. Obesity in men is associated with lower testosterone levels, reduced sperm quality, and decreased fertility. Weight loss through semaglutide treatment can improve all of these parameters. Men who have been struggling with low energy and reduced vitality due to obesity-related hormonal changes may find that the metabolic improvements from treatment indirectly support fertility.
Semaglutide and breastfeeding
After pregnancy, many women wonder when they can resume semaglutide, particularly if they are breastfeeding.
The research here is more encouraging than you might expect. A study of eight nursing mothers using subcutaneous semaglutide at doses ranging from 0.25 to 1 mg weekly found that semaglutide was undetectable in breast milk. The maximum relative infant dose projected was 1.26 percent, far below the standard 10 percent safety threshold used in lactation medicine.
The infants in the study ranged from 4 to 23 months of age and were mixed-fed, receiving breastmilk for 3 to 9 weeks while their mothers were taking semaglutide. All infants were reported to have normal growth and development.
Based on this data, injectable semaglutide is generally considered acceptable during breastfeeding. However, the oral form of semaglutide is not recommended during lactation because it contains an absorption enhancer called salcaprozate sodium, which may enter milk and accumulate in infants.
The primary concern with semaglutide during breastfeeding is not direct drug transfer to the infant. It is the effect on the mother. Semaglutide suppresses appetite and promotes weight loss. Breastfeeding already increases caloric demands by approximately 500 calories per day. Combining aggressive appetite suppression with the metabolic demands of milk production could lead to insufficient calorie intake, inadequate nutrient reserves, and reduced milk supply. If you are breastfeeding and taking semaglutide, monitoring calorie intake and milk production closely is essential. For more detailed information, see our guide on whether you can take GLP-1 while breastfeeding.
How semaglutide compares to tirzepatide for pregnancy planning
Women considering GLP-1 medications for weight loss before pregnancy often wonder how semaglutide compares to tirzepatide in the context of family planning.
Both medications carry similar pregnancy warnings. Neither should be taken during pregnancy or while actively trying to conceive. Both require a washout period before conception.
But there are meaningful differences.
Washout period: Tirzepatide has a half-life of approximately five days, compared to seven days for semaglutide. This means tirzepatide clears the body somewhat faster, though the recommended pre-conception cessation period is similar for both medications.
Birth control interaction: This is where the two drugs diverge significantly. Semaglutide does not reduce oral contraceptive absorption. Tirzepatide does, by approximately 20 percent. Women using tirzepatide need backup contraception, while women on semaglutide generally do not, unless vomiting is an issue. Understanding the full side effect differences between semaglutide and tirzepatide helps inform this decision.
Weight loss magnitude: Clinical trials suggest tirzepatide may produce somewhat greater weight loss than semaglutide, which could be advantageous for women who need to lose more weight before pregnancy. However, both medications produce clinically meaningful results for most users.
Fertility effects: Anecdotal reports of the Ozempic babies phenomenon exist for both medications. Women have reported unexpected pregnancies on tirzepatide as well. The underlying mechanism, improved fertility through weight loss and metabolic optimization, applies to both GLP-1 agonists.
Period effects: Both medications can affect menstrual cycles. Semaglutide has been documented to affect periods in some women, and similar reports exist for tirzepatide and menstrual changes. These changes are generally related to weight loss and hormonal shifts rather than direct effects of the medications on reproductive organs.
Nutritional considerations during pre-conception semaglutide use
Using semaglutide before pregnancy carries a specific nutritional challenge. The appetite suppression that makes weight loss possible can also make it difficult to consume adequate nutrients, and nutrient status before conception directly affects early fetal development.
Critical nutrients for pre-conception
Folate is the most important pre-conception nutrient. Neural tube defects, which occur in the first 28 days after conception, are strongly linked to folate deficiency. Women should start taking 400 to 800 micrograms of folic acid daily at least one month before trying to conceive, but ideally three months before. Since semaglutide treatment often reduces food intake, supplementation becomes even more critical.
Iron stores take months to build. Semaglutide-induced appetite suppression can reduce iron intake from food, and iron deficiency before pregnancy increases the risk of anemia during pregnancy, preterm birth, and low birth weight. Have your ferritin levels checked during the pre-conception phase.
Vitamin D plays a role in implantation and early placental development. Many women are deficient, and reduced food intake during semaglutide treatment can worsen this. Supplementation with 1,000 to 2,000 IU daily is commonly recommended.
Omega-3 fatty acids, particularly DHA, are critical for fetal brain development. Since semaglutide often reduces fat intake as part of overall calorie reduction, targeted supplementation or emphasis on fatty fish consumption matters. The supplements recommended with GLP-1 medications should include a quality prenatal vitamin with DHA if pregnancy is on the horizon.
Protein intake becomes doubly important. You need adequate protein to preserve lean mass during weight loss and to build nutrient stores for pregnancy. Aim for at least 1.2 grams per kilogram of body weight during the pre-conception phase. Our guide on protein intake while on semaglutide provides detailed recommendations.
Foods to prioritize
Even when appetite is suppressed, making every calorie count matters more during pre-conception than at any other time. Leafy greens provide folate and iron. Eggs offer choline, which is critical for fetal brain development. Fatty fish delivers omega-3s and vitamin D. Legumes provide both folate and plant-based iron. Dairy or fortified alternatives supply calcium and vitamin D. For a comprehensive food list, see our guide on the best foods to eat on semaglutide and modify it with a pre-conception lens.

Managing side effects during the pre-conception phase
Side effects from semaglutide can complicate pre-conception nutrition in ways that deserve attention.
Constipation is one of the most common complaints. During the pre-conception phase, adequate fiber and hydration are critical not just for comfort but for nutrient absorption and hormonal balance. A quality fiber supplement can help without adding significant calories.
Dizziness and lightheadedness can indicate dehydration or inadequate calorie intake, both of which need to be addressed during pre-conception. Staying well hydrated with quality electrolytes supports both side effect management and fertility.
Acid reflux and bloating can reduce food intake further. If these side effects are severe during the pre-conception phase, discuss dose reduction with your healthcare provider. The goal at this stage is metabolic improvement, not necessarily maximum weight loss, and a lower dose that allows adequate nutrition may serve your pregnancy goals better than the highest tolerable dose.
Insomnia affects overall health and hormonal balance. Sleep disruption can impair fertility through its effects on cortisol, melatonin, and reproductive hormones. If semaglutide is affecting your sleep, addressing this before conception is important.
Leg cramps may indicate magnesium or potassium deficiency, both of which matter for pregnancy. Correcting these mineral deficiencies serves double duty.
Monitoring your cycle during and after semaglutide
If you are using semaglutide with an eye toward future pregnancy, tracking your menstrual cycle provides valuable information about your fertility status.
Many women on semaglutide notice their cycles becoming more regular as they lose weight. This is particularly true for women with PCOS or obesity-related amenorrhea. The return of regular periods is often one of the first signs that semaglutide is working on a metabolic level beyond just weight loss.
Track the following during your semaglutide treatment and washout period:
Cycle length. Normal cycles range from 21 to 35 days. If your cycles were previously irregular and are becoming more predictable, that is a positive fertility sign.
Flow characteristics. Changes in menstrual flow can indicate hormonal shifts. Lighter or heavier periods during semaglutide treatment are common and usually normalize after the medication clears your system.
Ovulation signs. Cervical mucus changes, basal body temperature shifts, and ovulation predictor kit results can confirm whether you are ovulating regularly. Documenting these during the washout period helps you identify your fertile window when you begin trying to conceive.
Any spotting or breakthrough bleeding. Report this to your healthcare provider, as it can sometimes indicate issues that should be addressed before pregnancy.
Common questions about the washout period
The two-month washout generates a lot of anxiety. Here are the questions women most commonly ask.
Can I use a lower dose instead of stopping completely?
No. The recommendation is complete cessation, not dose reduction. Even small amounts of semaglutide still carry the theoretical risk that prompted the pregnancy warning. The half-life means even low doses take weeks to fully clear.
What if I only used semaglutide for a short time?
The duration of use does not change the washout recommendation. Whether you used semaglutide for two weeks or two years, the two-month cessation period applies. The drug needs to clear regardless of how long you took it.
Can I switch to a different weight loss medication during washout?
This depends entirely on the medication. Some weight loss drugs also carry pregnancy warnings. Phentermine, for example, is also contraindicated during pregnancy. Metformin is actually one of the few medications in this space that is sometimes continued during pregnancy for women with PCOS or diabetes, though this decision must be made with your healthcare provider.
Will I regain all the weight during the washout?
Not necessarily. The two-month washout is relatively short. Most people do not experience dramatic weight regain in just eight weeks, especially if they maintain the habits they built during treatment. Some appetite increase is normal, but the structural changes to your eating patterns should help maintain most of your progress. Understanding weight loss plateaus and how your body adapts helps set realistic expectations.
What if I have been on semaglutide for a long time?
Women who have been on semaglutide for extended periods, particularly those who have been on long-term treatment, may have more anxiety about stopping. The pharmacokinetics do not change with duration of use. The drug still clears at the same rate. What may differ is the psychological adjustment to losing the appetite suppression you have come to rely on. Working with a therapist or nutritionist during the washout can provide support.
When to talk to your doctor
Certain situations require immediate medical guidance rather than general information.
Talk to your healthcare provider if you are currently taking semaglutide and have a positive pregnancy test. Do not wait for your next scheduled appointment. Contact them the same day.
Speak with your doctor before starting semaglutide if pregnancy is something you are considering within the next year. They can help you develop a timeline that maximizes the metabolic benefits while ensuring adequate washout time.
Consult a reproductive endocrinologist if you have PCOS and are considering semaglutide specifically to improve fertility. While the data is promising, a specialist can determine whether semaglutide is the right approach for your specific situation or whether other interventions might be more appropriate.
See your provider if your periods have not returned to normal within three months of stopping semaglutide. While some cycle irregularity during washout is expected, prolonged amenorrhea after stopping the medication should be evaluated.
Reach out if you experience significant symptoms after stopping semaglutide that affect your quality of life or ability to maintain adequate nutrition during the pre-conception phase.
The bigger picture: metabolic health and pregnancy
Stepping back from the specific question of semaglutide timing, the broader principle matters more. Metabolic health before conception directly influences pregnancy outcomes, fetal development, and even the long-term health of the child.
Women who enter pregnancy with well-controlled blood sugar, healthy insulin sensitivity, adequate nutrient stores, and a body weight within a healthier range have measurably better outcomes across nearly every metric. Lower rates of gestational diabetes. Fewer cases of preeclampsia. Reduced risk of cesarean delivery. Better birth weights. Even potential benefits for the child metabolic health decades later, as emerging research on epigenetic programming suggests.
Semaglutide, used strategically before pregnancy and stopped with appropriate lead time, can be a powerful tool for achieving these metabolic goals. The key is treating it as a preparation phase, not an ongoing treatment that overlaps with conception.
For women who have struggled with weight, insulin resistance, or PCOS-related infertility, the emergence of GLP-1 medications represents a genuinely new option. Not a fertility drug, but a metabolic optimization tool that happens to remove some of the biggest barriers to conception. The weight loss journey with semaglutide can set the stage for one of the most important transitions of your life.
Understanding how to qualify for semaglutide and approaching treatment with pregnancy planning in mind from the start gives you the best chance of using the medication effectively while keeping your future family safe.
Returning to semaglutide after pregnancy
Many women want to know when they can resume semaglutide after having a baby, especially if they gained weight during pregnancy.
If you are not breastfeeding, you can discuss resuming semaglutide with your healthcare provider once you have recovered from delivery. There is no specific waiting period beyond normal postpartum recovery, though most providers recommend waiting at least six weeks postpartum before starting any new medication.
If you are breastfeeding, as discussed earlier, injectable semaglutide appears safe based on the available data showing undetectable levels in breast milk. However, the appetite suppression effects need to be managed carefully to ensure adequate calorie intake for milk production. Starting at the lowest dose and increasing slowly is typically recommended.
The decision about restarting semaglutide after pregnancy involves weighing the benefits of postpartum weight management against the demands of breastfeeding and early parenthood. There is no universal right answer. It depends on your individual health situation, breastfeeding plans, and priorities.
If you do resume semaglutide while breastfeeding, monitoring is essential. Track your milk supply, your infant weight gain, and your own nutritional intake. The combination of postpartum recovery, breastfeeding demands, and appetite suppression can create nutritional deficits that affect both you and your baby if not actively managed.
Practical checklist for semaglutide and pregnancy planning
Whether you are currently on semaglutide and thinking about pregnancy, or considering starting semaglutide with future pregnancy plans, this checklist covers the essential steps.
Before starting semaglutide: Tell your provider about your pregnancy timeline. Start prenatal vitamins now, even if pregnancy is months away. Choose a contraception method that does not depend solely on oral absorption. Set weight loss goals that align with your conception timeline. Understand the dosing schedule and plan your treatment duration accordingly.
During semaglutide treatment: Track your menstrual cycles monthly. Maintain adequate protein and micronutrient intake despite reduced appetite. Continue prenatal vitamins throughout treatment. Monitor your progress with appropriate tools. Know the signs of unintended pregnancy, since improved fertility from weight loss is real. Use reliable contraception consistently.
Two months before planned conception: Take your final semaglutide dose. Inform your healthcare provider you are entering the washout phase. Continue prenatal vitamins. Prepare for appetite return by having structured meal plans ready. Begin ovulation tracking if you have not already.
During washout (weeks 1 through 8): Do not take any additional semaglutide doses. Focus on maintaining nutritional quality. Manage returning appetite with high-protein, high-fiber meals. Track any menstrual cycle changes. Stay in contact with your healthcare provider. Avoid alcohol as you approach your conception window.
After conception: Confirm pregnancy with your provider. Report any semaglutide exposure timing. Follow standard prenatal care recommendations. Do not restart semaglutide until after delivery and any breastfeeding period, or until cleared by your provider.
Frequently asked questions
How long does semaglutide stay in your system after the last dose?
Semaglutide has a half-life of approximately seven days, meaning it takes roughly six weeks for the vast majority of the drug to be eliminated from your body. The two-month washout recommendation adds extra safety margin beyond this clearance period. Understanding how semaglutide duration works helps explain why the timeline is what it is.
Can semaglutide cause a miscarriage?
Human studies have not demonstrated that semaglutide increases miscarriage risk. Animal studies showed increased pregnancy losses, but these occurred through a mechanism specific to rodent placental biology that does not apply to humans. The current evidence, while limited, does not support a causal link between semaglutide exposure and miscarriage in humans.
Does semaglutide cause birth defects?
In human studies including over 1,100 exposed pregnancies, no consistent link has been found between semaglutide use and major congenital malformations. The baseline birth defect rate in the general population is approximately 3 percent, and semaglutide-exposed pregnancies have not shown rates significantly above this baseline. However, the data is not yet robust enough to make definitive safety claims.
Why do people call them Ozempic babies?
The term refers to women who became pregnant unexpectedly after starting semaglutide (brand name Ozempic) or other GLP-1 medications. Many of these women had previously struggled with infertility, often related to PCOS or obesity. The weight loss and metabolic improvements from semaglutide restored their fertility, sometimes catching them by surprise. It is not that semaglutide is a fertility drug. Rather, it removes metabolic barriers that were preventing conception.
Can my husband keep taking semaglutide while we try to conceive?
Yes. Animal studies found no effects on male fertility at therapeutic doses of semaglutide. There is no recommendation for men to stop semaglutide before their partner attempts to conceive. In fact, if the male partner has obesity, continued semaglutide use may indirectly improve fertility through weight loss and hormonal optimization.
Should I use semaglutide or tirzepatide if I am planning pregnancy later?
Both medications require a washout period before conception and carry similar pregnancy warnings. The main practical difference is that tirzepatide reduces oral contraceptive absorption by about 20 percent, while semaglutide does not. If you rely on birth control pills and want to avoid accidental pregnancy during treatment, semaglutide may be the more convenient choice. Discuss your specific situation with your provider.
Will I gain back all the weight during the two-month washout?
Most women do not experience significant weight regain in just two months. Appetite typically returns gradually over the first two to three weeks after the last dose, but eight weeks is a short enough window that maintained eating habits can prevent major regain. Focus on structured meals, adequate protein, and the nutrition strategies you established during treatment.
Is semaglutide safe while breastfeeding?
Injectable semaglutide has been found undetectable in breast milk in a study of eight nursing mothers, with a projected relative infant dose of 1.26 percent, well below safety thresholds. The oral form is not recommended during breastfeeding due to its absorption enhancer. The bigger concern is ensuring adequate calorie intake for milk production while appetite is suppressed. Our guide on GLP-1 medications and breastfeeding covers this topic in detail.
External resources
For researchers serious about understanding how semaglutide works and navigating its use around major life events like pregnancy, SeekPeptides provides the most comprehensive resource available. Members access evidence-based guides, dosage calculators, and a community of thousands who have navigated these exact questions. Whether you are planning pregnancy months from now or just starting to think about it, having reliable, research-backed information makes all the difference.
In case I do not see you, good afternoon, good evening, and good night. May your fertility stay strong, your planning stay smart, and your family stay healthy.