Apr 4, 2026

Tired of conflicting information about tirzepatide and PCOS? One article says it is a miracle. Another says the evidence is thin. Forums are full of personal stories that contradict each other, and your doctor may not even know this medication exists for polycystic ovary syndrome. The confusion is real. And it is costing women valuable time.
Here is the truth. Tirzepatide was not designed for PCOS. It was built for type 2 diabetes and weight management. But something unexpected happened during clinical use, something that caught researchers off guard and changed the conversation about hormonal health entirely. Women with PCOS started reporting dramatic improvements. Not just weight loss, but restored menstrual cycles, reduced cyst prevalence, lower testosterone levels, and genuine relief from symptoms they had been battling for years.
The numbers back this up. Over 90% of PCOS patients lost at least 10% of their body weight within 10 months on tirzepatide. Irregular menstrual cycles decreased from 85.7% to 32.1%. Ovarian cyst prevalence dropped from 89.3% to 41%. These are not marginal improvements. These are life-changing outcomes for women who had tried metformin, lifestyle changes, and birth control pills without meaningful results. The mechanism behind tirzepatide, its unique dual-receptor approach, targets the very metabolic dysfunction that drives PCOS in the first place. And that distinction matters more than most people realize.
This guide covers everything. The science behind why tirzepatide works for PCOS, the clinical data supporting its use, practical dosing protocols, side effect management, how it compares to semaglutide and other GLP-1 medications, fertility considerations, and what real-world outcomes actually look like. Whether you are just learning about this option or actively considering it with your healthcare provider, SeekPeptides has built this resource to be the most comprehensive reference available on tirzepatide and polycystic ovary syndrome.
Understanding PCOS and why traditional treatments fall short
Polycystic ovary syndrome affects roughly 8 to 13% of women of reproductive age worldwide. That makes it one of the most common endocrine disorders on the planet. Yet for something so prevalent, PCOS remains remarkably misunderstood, both by the general public and by many healthcare providers who treat it.
The name itself is misleading. Not every woman with PCOS has ovarian cysts. Not every woman with ovarian cysts has PCOS. The condition is actually a metabolic and hormonal disorder that manifests differently in every patient, which is precisely why it takes an average of two years and visits to three or more doctors before most women receive a correct diagnosis.
The insulin resistance connection most people miss
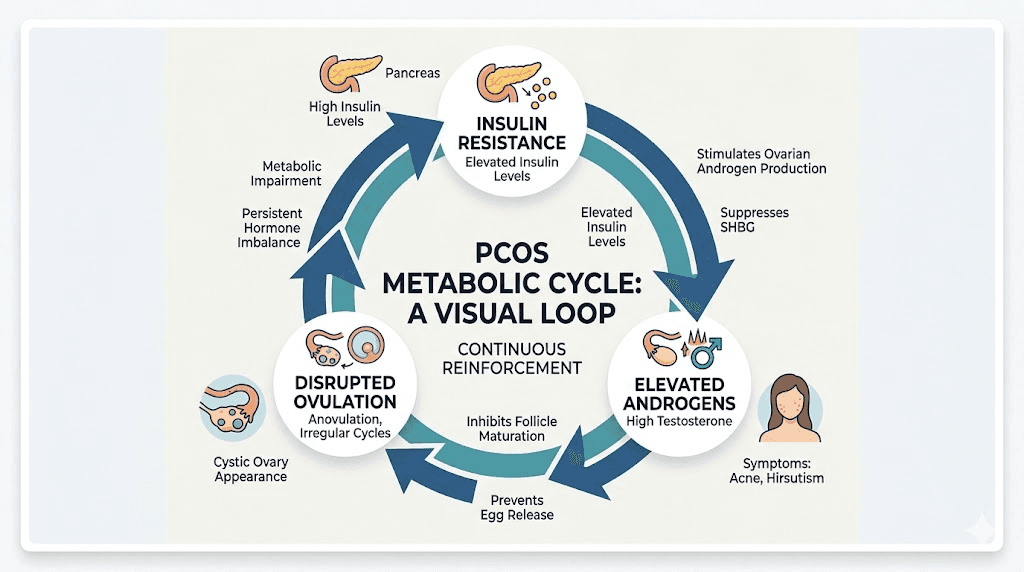
Here is what changes the entire conversation about PCOS treatment. Between 50% and 70% of women with PCOS have clinically significant insulin resistance, regardless of their body weight. That number rises even higher in women who also carry excess weight. Insulin resistance is not just a side effect of PCOS. It is increasingly recognized as the primary metabolic driver of the condition itself.
When cells become resistant to insulin, the pancreas compensates by producing more. These elevated insulin levels directly stimulate the ovaries to produce excess androgens, particularly testosterone. Higher testosterone disrupts normal follicle development, prevents regular ovulation, and produces the hallmark symptoms of PCOS, including acne, hirsutism, hair thinning, and irregular or absent periods.
The cascade goes deeper. Excess insulin also reduces production of sex hormone-binding globulin, or SHBG. This protein normally binds to testosterone and keeps it inactive. With less SHBG circulating, more free testosterone becomes available to cause symptoms. The cycle reinforces itself. Insulin resistance drives androgen excess, which promotes abdominal fat storage, which worsens insulin resistance further.
This is why weight loss of just 5 to 10% of body weight can restore regular menstrual cycles in up to 70% of women with PCOS. It is not the weight loss itself that matters most. It is the improvement in insulin sensitivity that accompanies it.
Why metformin and lifestyle changes are not enough for many women
Metformin has been the go-to pharmaceutical intervention for PCOS and insulin resistance for decades. It works. But it has limitations that leave many women frustrated and still symptomatic.
The average weight loss on metformin is modest, typically 2 to 5% of body weight over 6 months. For some women, that is enough to tip the metabolic balance. For many others, it falls short. Metformin also comes with gastrointestinal side effects that make compliance difficult, and it does not directly address the appetite dysregulation that accompanies insulin resistance. Women with PCOS often experience increased hunger signals and reduced satiety, a hormonal double bind that makes sustainable weight management exceptionally challenging even with the best intentions and dietary strategies.
Lifestyle modifications remain the first-line recommendation. And they should be. Regular exercise improves insulin sensitivity independently of weight loss. Dietary changes that reduce refined carbohydrates and processed foods lower insulin spikes. Stress management matters because cortisol worsens insulin resistance. But here is the problem: PCOS itself creates metabolic barriers that make these interventions less effective than they would be in someone without the condition. Women with PCOS burn fewer calories at rest, store fat more readily in the abdominal region, and have hormonal signaling that actively opposes weight loss efforts.
This is not a willpower issue. It is a metabolic one. And it is exactly where tirzepatide enters the picture with a fundamentally different approach.
How tirzepatide works as a dual GLP-1/GIP receptor agonist
Tirzepatide is different from every other medication in its class. That distinction is not marketing. It reflects a genuinely novel mechanism that explains why the clinical outcomes have been so striking, not just for diabetes and weight loss, but potentially for conditions like PCOS where metabolic dysfunction drives the disease.
The dual-receptor mechanism explained
Most GLP-1 receptor agonists, including semaglutide, work on a single target. They activate the GLP-1 receptor, which stimulates insulin release, slows gastric emptying, and reduces appetite through effects on the brain. These are powerful effects. But they represent only half of the incretin system.
Tirzepatide activates both the GLP-1 receptor and the GIP receptor simultaneously. GIP, or glucose-dependent insulinotropic polypeptide, is the other major incretin hormone. For years, GIP was considered less important. Some researchers even thought blocking GIP might be beneficial for weight loss. Tirzepatide proved that theory wrong in spectacular fashion.
The dual activation creates effects that neither receptor can produce alone. GLP-1 activation reduces appetite and slows digestion. GIP activation enhances insulin sensitivity in adipose tissue, improves fat metabolism, and appears to reduce nausea, which is one of the most common complaints with pure GLP-1 agonists. Together, these pathways produce greater weight loss, better glucose control, and improved metabolic markers compared to targeting either receptor individually.
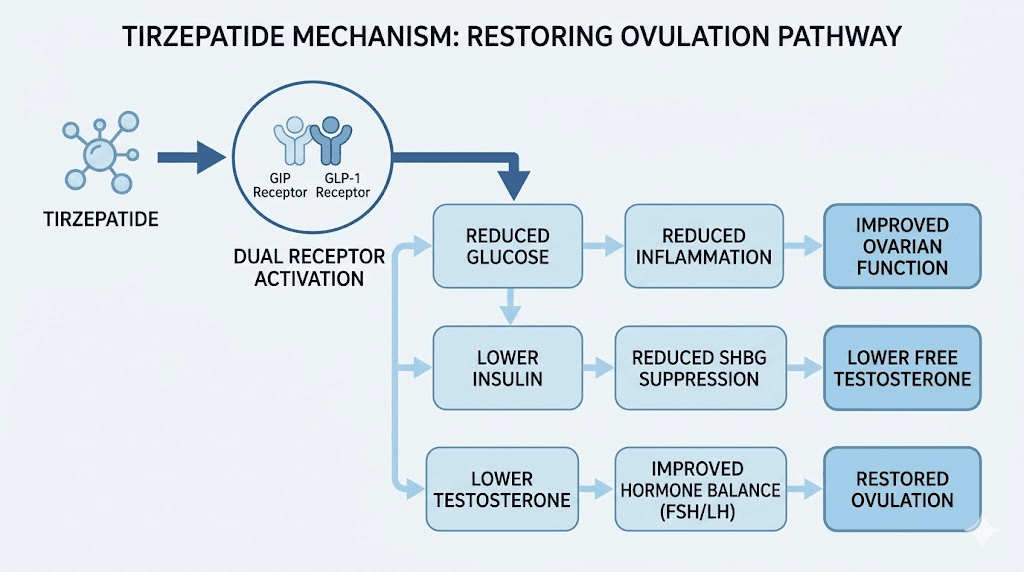
For women with PCOS, the dual mechanism of tirzepatide addresses the condition from multiple angles simultaneously. It improves insulin sensitivity both directly through GIP signaling and indirectly through weight reduction. It reduces the hyperinsulinemia that drives androgen excess. And it improves GLUT-4 expression, the glucose transporter responsible for getting sugar out of the bloodstream and into cells where it belongs.
Beyond weight loss: the metabolic cascade that benefits PCOS
Weight loss alone does not explain what happens to PCOS symptoms on tirzepatide. The metabolic improvements begin before significant weight reduction occurs, suggesting direct pharmacological effects on insulin signaling pathways.
Lower insulin means less ovarian stimulation. Less ovarian stimulation means lower testosterone production. Lower testosterone allows normal follicle development. Normal follicle development restores ovulation. Restored ovulation normalizes menstrual cycles. Each step builds on the last, creating a cascade of improvement that addresses PCOS at its root rather than merely treating symptoms.
Tirzepatide also increases SHBG levels. Remember, SHBG binds to free testosterone and renders it inactive. This is the same protein that insulin resistance suppresses. As insulin sensitivity improves, SHBG rises, and the amount of biologically active testosterone drops, even beyond what the reduction in total testosterone production would predict. Research shows GLP-1 receptor agonists can reduce testosterone levels by approximately 33%, a meaningful reduction that translates to visible improvements in androgen-driven symptoms.
There are also anti-inflammatory effects. PCOS is associated with chronic low-grade inflammation that further impairs insulin signaling and contributes to cardiovascular risk. Tirzepatide decreases markers of inflammation and reduces oxidative stress, addressing yet another dimension of the metabolic dysfunction that characterizes this condition.
Clinical evidence for tirzepatide in PCOS
The research on tirzepatide specifically for PCOS is growing rapidly. While the initial approvals were for type 2 diabetes and chronic weight management, the data emerging from both clinical studies and real-world observation is compelling enough that many endocrinologists and reproductive specialists are already incorporating it into their treatment approaches for women with polycystic ovary syndrome. Understanding how quickly tirzepatide produces results helps set appropriate expectations.
Weight loss outcomes that exceed previous treatments
The headline numbers are remarkable. In studies of women with PCOS treated with tirzepatide, 96.58% achieved at least 5% weight loss. Over 90% lost at least 10% of their body weight. And 75.96% reached the 15% threshold, a level of weight reduction previously achievable only through bariatric surgery for most patients.
The median weight loss reached 18.81% at 10 months. To put that in perspective, a woman weighing 200 pounds would lose approximately 37 to 38 pounds in that timeframe. For women with PCOS, where every percentage point of weight loss translates to measurable hormonal improvement, these numbers represent a genuine paradigm shift in what pharmaceutical treatment can accomplish.
Compare this to the alternatives. Metformin produces 2 to 5% weight loss on average. Tirzepatide demonstrated weight reduction of 17.60 kg compared to semaglutide at 11.85 kg in comparative analyses, a difference of nearly 6 kilograms. Lifestyle interventions alone typically yield 3 to 7% weight loss. The tirzepatide weight loss timeline shows that meaningful results begin within the first few weeks and continue progressively throughout treatment.
Menstrual cycle restoration
Perhaps the most meaningful outcome for women with PCOS is the restoration of regular menstrual cycles. Irregular periods are not just an inconvenience. They indicate anovulation, which affects fertility, bone density, endometrial health, and overall hormonal balance.
The data here is striking. Irregular menstrual cycles decreased from 85.7% of patients at baseline to just 32.1% during tirzepatide treatment. That means the majority of women who started with irregular cycles experienced normalization. This improvement reflects genuine restoration of the hypothalamic-pituitary-ovarian axis, not just breakthrough bleeding or artificially induced withdrawal bleeds like those produced by oral contraceptives.
Regular ovulation returning has implications far beyond fertility. It normalizes estrogen and progesterone cycling, which protects the endometrial lining from hyperplasia. It supports bone density maintenance. It improves mood stability and reduces the hormonal fluctuations that many women with PCOS describe as emotionally exhausting.
Reduction in ovarian cysts and androgen levels
Ovarian cyst prevalence dropped from 89.3% to 41% in patients receiving tirzepatide. These cysts, actually immature follicles that failed to ovulate, are the structural hallmark of PCOS on ultrasound. Their reduction indicates that follicle development is normalizing and ovulation is occurring more regularly.
Insulin resistance, the metabolic measurement most directly tied to PCOS pathology, improved from 80.4% to 50% of patients meeting criteria. That improvement occurred alongside the weight loss and hormonal changes, confirming the cascade mechanism. Better insulin sensitivity leads to lower insulin, which leads to lower androgen production, which leads to improved ovarian function.
The reduction in androgens is clinically significant. Women report improvements in acne within 2 to 3 months. Hirsutism, the unwanted facial and body hair growth that causes so much distress, begins to improve over 3 to 6 months as testosterone levels fall. Hair thinning at the scalp, another androgen-driven symptom, may take longer to show improvement because of the hair growth cycle, but the underlying hormonal driver is being addressed rather than masked.
Tirzepatide dosing protocol for PCOS
Getting the dose right matters enormously with tirzepatide. Too aggressive an approach leads to intolerable side effects that undermine compliance. Too conservative, and the metabolic benefits may not reach the threshold needed to improve PCOS symptoms. The standard tirzepatide dose chart provides a framework, but PCOS patients often benefit from a customized approach.
Standard titration schedule
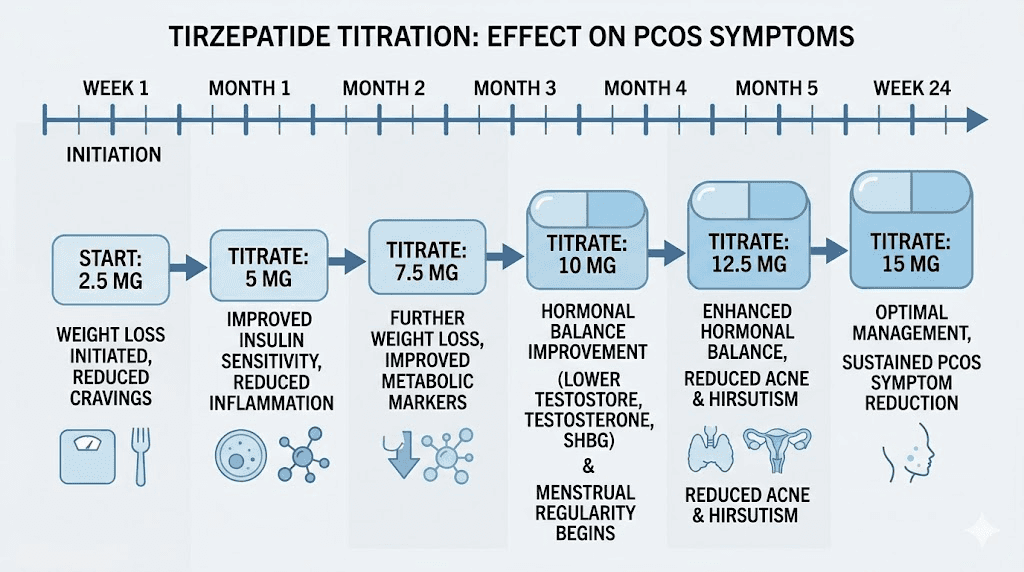
Tirzepatide uses a gradual dose escalation to minimize gastrointestinal side effects and allow the body to adapt. The starting dose is 2.5 mg administered once weekly via subcutaneous injection. This initial dose is considered a tolerability dose rather than a therapeutic dose for most patients.
The titration steps follow a defined progression. From the starting dose of 2.5 mg, the next step is 5 mg weekly after 4 weeks. Then 7.5 mg. Then 10 mg. Then 12.5 mg. And the maximum approved dose is 15 mg weekly. Each step typically lasts 4 weeks, though some clinicians extend this interval to 6 or 8 weeks if side effects are significant.
Not every patient needs the maximum dose. Some women with PCOS achieve meaningful metabolic improvement at 5 mg or 7.5 mg, particularly those who are primarily seeking hormonal normalization rather than substantial weight loss. The right dose is the one that produces the desired clinical outcome with acceptable side effects. You can use the SeekPeptides peptide calculator to help determine unit measurements based on your specific formulation concentration.
Understanding unit measurements
One of the most common sources of confusion with tirzepatide involves converting between milligram doses and syringe units, especially with compounded formulations. The relationship between milligrams and units depends entirely on the concentration of the solution.
For those using compounded tirzepatide, the compounded tirzepatide dosage calculator is essential. If your vial contains 10 mg/mL, then 2.5 mg equals 0.25 mL or 25 units on a standard insulin syringe. At the same concentration, 2.5 mg corresponds to 25 units, while 5 mg corresponds to 50 units. Understanding tirzepatide dosage in units prevents dangerous over or under-dosing.
The math changes with different concentrations. A 5 mg/mL solution requires twice the volume as a 10 mg/mL solution for the same dose. A 20 mg/mL solution requires half. Always verify the concentration on your specific vial before calculating. If you need help with the calculations, our guide on converting 20 units to milligrams, 30 units to milligrams, 40 units to milligrams, and 50 units to milligrams cover the most common conversion scenarios.
Reconstitution and storage for compounded tirzepatide
If you are working with lyophilized (freeze-dried) compounded tirzepatide, proper reconstitution is essential for both efficacy and safety. The process involves adding bacteriostatic water to the powder vial, swirling gently rather than shaking, and allowing the solution to fully dissolve before drawing a dose.
The amount of bacteriostatic water matters because it determines the concentration. For a 10 mg vial, adding 1 mL of bacteriostatic water creates a 10 mg/mL concentration. Adding 2 mL creates 5 mg/mL. Our guide on how much bacteriostatic water to mix with 10 mg tirzepatide walks through the process step by step. The peptide reconstitution calculator handles the math for any vial size and desired concentration.
Storage is straightforward but non-negotiable. Reconstituted tirzepatide must be kept refrigerated between 2 and 8 degrees Celsius. Our detailed guide on tirzepatide refrigeration requirements covers proper storage protocol. Once reconstituted, most compounded formulations remain stable for 28 to 30 days when properly stored. How long tirzepatide lasts in the fridge depends on the specific formulation and preservative used, but the general principle is simple: keep it cold, keep it clean, and use it within the recommended timeframe.
Injection technique and timing
Tirzepatide is administered as a once-weekly subcutaneous injection. The abdomen is the most common injection site, though the thigh and upper arm are also acceptable. Our guide on how to inject tirzepatide in the stomach area provides detailed instructions with visual references. Proper technique matters for consistent absorption and to minimize injection site reactions.
Pick the same day each week. Consistency helps maintain stable blood levels. If you miss your usual day, taking tirzepatide a day early is generally acceptable as long as at least 3 days have passed since the previous injection. The syringe dosage guide covers the practical aspects of drawing and administering the correct amount.
Microdosing tirzepatide for PCOS: a gentler approach
Not every woman needs the maximum dose to see improvements in PCOS symptoms. A growing number of clinicians and patients are exploring microdosing approaches to tirzepatide that prioritize metabolic improvement over aggressive weight loss.
Why microdosing may work for PCOS specifically
Remember that 5 to 10% weight loss can restore menstrual cycles in 70% of women with PCOS. That level of weight reduction does not require maximum-dose tirzepatide. Lower doses, in the range of 2.5 to 5 mg weekly, may produce sufficient insulin sensitization and modest weight loss to tip the hormonal balance back toward normal function.
The advantage of lower doses is dramatically reduced side effect burden. At 2.5 mg, most women tolerate tirzepatide without significant gastrointestinal discomfort. This improved tolerability translates to better long-term compliance, which matters enormously for a condition that requires sustained treatment rather than short-term intervention.
The microdose tirzepatide schedule typically involves staying at 2.5 mg for 8 to 12 weeks rather than the standard 4 weeks, then advancing to 5 mg only if clinical response is insufficient. Some patients remain at 2.5 or 5 mg indefinitely, achieving the metabolic improvements they need without escalating to higher doses. This approach is particularly relevant for women who are primarily seeking hormonal normalization and fertility improvement rather than major weight reduction.
Managing side effects of tirzepatide in PCOS patients
Side effects are the primary reason women discontinue tirzepatide before achieving full benefit. Understanding what to expect, when to be concerned, and how to manage common issues makes the difference between successful treatment and premature abandonment of a medication that might have transformed your health.
Gastrointestinal effects: the most common challenge
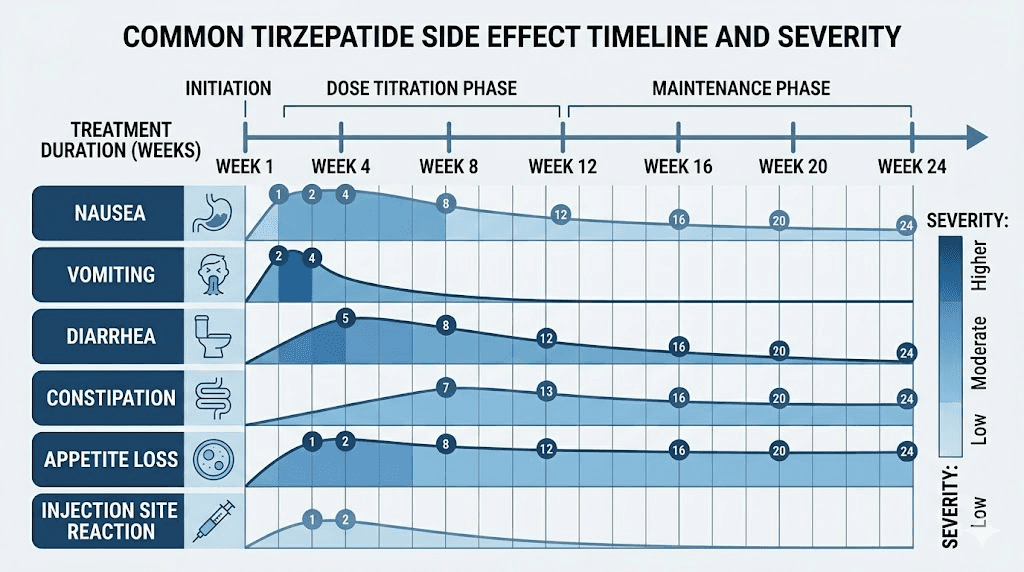
The most frequently reported side effects are gastrointestinal. In studies of women with PCOS on tirzepatide, heartburn affected 42.86% of patients. Nausea and vomiting occurred in 39.29%. General weakness was reported by 33.93%. These numbers sound high, but context matters. Most gastrointestinal side effects are dose-dependent, meaning they occur primarily during dose escalation and often resolve or significantly improve within 2 to 4 weeks at each new dose level.
Nausea is the side effect that drives the most discontinuation. It tends to peak in the first 2 to 3 days after each injection, particularly when moving to a higher dose. Eating smaller, more frequent meals helps. Avoiding high-fat and greasy foods during the first few days after injection reduces the severity. Staying hydrated is essential, especially if vomiting occurs. The complete guide to foods to avoid on tirzepatide provides specific dietary recommendations, and the tirzepatide diet plan offers meal-by-meal strategies.
Constipation is another common complaint. Slowed gastric emptying is a feature of GLP-1 receptor agonists, not a bug, as it contributes to satiety and blood sugar control. But it can lead to uncomfortable constipation that requires active management. Our tirzepatide constipation treatment guide covers evidence-based approaches from fiber supplementation to osmotic laxatives. On the other end of the spectrum, some women experience diarrhea that typically resolves within the first few weeks of treatment.
Fatigue and energy changes
Many women wonder whether tirzepatide causes fatigue. The answer is nuanced. During the initial weeks of treatment, reduced caloric intake can produce a temporary energy dip, particularly in women who were previously consuming significantly more calories than their new, medication-influenced appetite allows. This is not a direct drug side effect so much as a caloric adjustment period.
Ironically, many women with PCOS report improved energy over time on tirzepatide. The question of whether tirzepatide gives you energy is answered differently by different patients, but the metabolic improvements, better blood sugar stability, reduced inflammation, and weight loss all contribute to higher baseline energy in many cases. Body aches and sleep disturbances can occur but are typically transient.
Other side effects relevant to PCOS patients
Headaches occur in a subset of patients, particularly during early treatment. The complete guide to tirzepatide and headaches covers causes and management strategies. Anxiety symptoms have been reported by some users, though it remains unclear whether this represents a direct pharmacological effect or a response to rapid metabolic changes. Joint pain and muscle pain occasionally occur, particularly during weight loss, and may relate to changes in mechanical loading or inflammatory markers.
For women specifically concerned about PCOS-related hair issues, the relationship between GLP-1 medications and hair loss is important to understand. Rapid weight loss from any cause can trigger telogen effluvium, a temporary shedding phase. This typically resolves once weight stabilizes, and the long-term hormonal improvements from reduced androgens generally benefit hair health overall.
Tirzepatide and menstrual cycles: what PCOS patients need to know
Menstrual irregularity is often the symptom that initially brings women with PCOS to medical attention. It is also one of the symptoms most responsive to tirzepatide treatment. But the path from irregular to regular is not always straightforward, and understanding what to expect prevents unnecessary worry during the transition.
How tirzepatide restores ovulation
The mechanism is elegantly simple once you understand the underlying physiology. In PCOS, elevated insulin drives the ovaries to produce excess androgens. These androgens, primarily testosterone, disrupt the delicate hormonal signaling that governs follicle development and ovulation. Multiple follicles begin to develop each cycle, but none reaches the maturity needed for ovulation. These stalled follicles become the "cysts" visible on ultrasound.
Tirzepatide breaks this cycle by reducing insulin levels. Lower insulin means the ovaries receive less stimulation to produce androgens. With testosterone falling, the hypothalamic-pituitary-ovarian axis begins to function more normally. Follicle-stimulating hormone and luteinizing hormone regain their normal pulsatile pattern. A dominant follicle can emerge, mature, and actually ovulate.
This process takes time. Most women begin to notice cycle changes within 2 to 4 months of starting tirzepatide, coinciding with the period needed for meaningful metabolic improvement. Some women experience irregular bleeding patterns initially as their hormonal milieu shifts. This is usually temporary and resolves as cycles normalize.
Period changes during tirzepatide treatment
The question of whether tirzepatide affects your period comes up constantly, and the answer is yes, often dramatically and for the better. Women who had been menstruating only 2 to 4 times per year may find themselves moving toward monthly cycles. Women who had completely absent periods may see menstruation return for the first time in years.
But the transition is not always smooth. Some women experience heavier than expected periods as cycles resume, particularly if the endometrial lining has built up during prolonged anovulation. Others experience spotting between periods during the first few months. These changes, while sometimes concerning, typically represent the hormonal system recalibrating rather than a problem requiring intervention.
The same metabolic improvements that affect menstrual cycles in women with PCOS also influence reproductive hormones in other ways. Similarly, semaglutide has been shown to affect periods through the same insulin-mediated mechanism, though tirzepatide dual-receptor action may produce faster or more pronounced changes.
Tirzepatide and fertility in PCOS
For many women with PCOS, the relationship between this medication and fertility is the most important consideration. PCOS is the leading cause of anovulatory infertility, and any treatment that restores ovulation has direct implications for reproductive potential.
The fertility surprise: unplanned pregnancies on GLP-1 medications
Here is something pharmaceutical companies did not emphasize in their marketing but real-world experience has made clear: women who were previously unable to conceive are becoming pregnant on GLP-1 receptor agonists, sometimes unexpectedly. The phenomenon has become common enough that some fertility specialists now joke about these medications being the most effective fertility treatment available.
The stories of women getting pregnant on tirzepatide are both heartwarming and cautionary. For women actively trying to conceive, restored ovulation is exactly the desired outcome. But for women who believed they could not become pregnant due to PCOS and were not using contraception, the surprise is significant. If you are taking tirzepatide and are sexually active but not trying to conceive, use reliable contraception. Your fertility may be substantially better than you believe.
It is critical to note that tirzepatide should be discontinued before attempting conception and during pregnancy. The medication has not been studied for safety during pregnancy, and the reduced caloric intake it produces is not appropriate for fetal development. Most guidelines recommend stopping tirzepatide at least 2 months before planned conception attempts. For women who discover pregnancy while on the medication, the recommendation is to stop immediately and consult with an obstetrician.
Using tirzepatide as a bridge to fertility treatment
Some reproductive endocrinologists are now using tirzepatide as a pre-treatment before formal fertility interventions. The logic is sound. A woman who enters IVF or ovulation induction with better insulin sensitivity, lower androgens, and healthier body composition has significantly better outcomes than one who begins fertility treatment with unaddressed metabolic dysfunction.
A typical approach involves 3 to 6 months of tirzepatide treatment to optimize metabolic parameters, followed by discontinuation and a washout period before beginning fertility medications. During this pre-treatment phase, some women conceive spontaneously, eliminating the need for more intensive interventions entirely.
Breastfeeding considerations
For women who become pregnant and deliver while their PCOS is being managed, the question of tirzepatide during breastfeeding inevitably arises. Current guidelines recommend against tirzepatide while nursing, as the medication has not been studied in lactating women and could theoretically affect milk production or composition. Similarly, GLP-1 medications in general are not recommended during breastfeeding. Women should work with their healthcare providers to develop an alternative PCOS management strategy during the breastfeeding period.
Tirzepatide versus semaglutide for PCOS
The comparison between tirzepatide and semaglutide is perhaps the most common question among women with PCOS considering GLP-1 therapy. Both medications have demonstrated benefits for the metabolic and hormonal aspects of the condition. But they are not identical, and the differences may matter depending on individual circumstances and treatment goals. The comprehensive semaglutide versus tirzepatide comparison at SeekPeptides covers the broader differences in detail.
Head-to-head comparison on key outcomes
On weight loss, tirzepatide has consistently outperformed semaglutide. The data shows tirzepatide producing weight reduction of 17.60 kg compared to semaglutide at 11.85 kg, a difference of nearly 6 kilograms. For women with PCOS where weight loss directly correlates with hormonal improvement, this difference is clinically meaningful.
The difference likely stems from tirzepatide dual-receptor mechanism. GIP receptor activation provides additional metabolic benefits that GLP-1 alone does not, including improved fat tissue insulin sensitivity and enhanced nutrient-dependent insulin secretion. The result is greater total weight loss, more fat mass reduction specifically, and potentially better preservation of lean muscle mass.
On insulin sensitization, both medications improve insulin resistance significantly. However, some evidence suggests tirzepatide produces faster and more pronounced improvement in insulin sensitivity markers, which would translate to earlier and more robust hormonal improvement for PCOS patients. This theoretical advantage needs more direct comparison data to confirm, but the mechanistic logic is strong.
Side effect profiles compared
The side effect comparison between semaglutide and tirzepatide matters for treatment selection. Both medications cause gastrointestinal side effects, primarily nausea, vomiting, and diarrhea. However, some data suggests that tirzepatide dual mechanism may actually reduce GI side effects compared to pure GLP-1 agonists, because the GIP component appears to have a moderating effect on nausea.
Both medications can cause fatigue, particularly early in treatment. Both can cause headaches. Both affect appetite profoundly, which is the intended effect. The question of whether tirzepatide suppresses appetite faster depends on individual response, but many patients report noticeable appetite reduction within the first week.
For women considering switching between these medications or wanting to understand how doses compare, the semaglutide to tirzepatide conversion chart provides guidance on equivalent dosing. The semaglutide dosage calculator helps with precise measurements for those currently on semaglutide who are considering the switch.
Factor | Tirzepatide | Semaglutide |
|---|---|---|
Mechanism | Dual GLP-1/GIP agonist | GLP-1 agonist only |
Average weight loss | 17.60 kg | 11.85 kg |
Insulin sensitization | Direct plus indirect | Primarily indirect |
GI side effects | Potentially lower (GIP moderation) | Standard GLP-1 profile |
Dosing range | 2.5 to 15 mg weekly | 0.25 to 2.4 mg weekly |
PCOS-specific data | Growing rapidly | More established |
Cost | Higher | Lower generic options emerging |
When to choose tirzepatide over semaglutide for PCOS
Tirzepatide may be the better choice when insulin resistance is severe and central to the PCOS presentation, when significant weight loss is needed to achieve hormonal improvement, when a patient has not responded adequately to semaglutide alone, or when the dual mechanism offers potential for fewer GI side effects. The speed at which tirzepatide works may also be a factor for women eager to see improvements in their symptoms and cycles.
Semaglutide may be preferred when cost is a primary consideration, when a patient has already responded well to GLP-1 therapy and simply needs to maintain, or when the more extensive safety data for semaglutide provides additional reassurance. Both the semaglutide onset timeline and the appetite suppression onset are well-documented.
Tirzepatide versus metformin for PCOS
Metformin has been the standard pharmacological treatment for PCOS and insulin resistance for decades. Comparing it to tirzepatide illuminates how dramatically the treatment landscape is shifting.
Mechanism comparison
Both medications address insulin resistance, but through fundamentally different pathways. Metformin primarily works by reducing hepatic glucose production and modestly improving peripheral insulin sensitivity. It does not significantly affect appetite, body weight, or the incretin system. Tirzepatide works through incretin receptor activation, producing profound effects on appetite, body weight, and insulin sensitivity simultaneously.
The weight loss difference is the most striking distinction. Metformin produces 2 to 5% weight loss on average, while tirzepatide produces median weight loss of 18.81% at 10 months. For PCOS patients whose symptoms are strongly linked to metabolic status and body composition, this difference in weight loss potential translates directly to greater symptom improvement.
Practical considerations
Metformin is taken orally, usually twice daily. Tirzepatide is a once-weekly injection, though oral tirzepatide formulations are in development. The comparison between tablet and injection formats is relevant for women who strongly prefer oral medications. For those comfortable with injections, the once-weekly schedule is actually more convenient than twice-daily metformin.
Cost is a significant factor. Metformin is available as a generic medication and costs very little. Tirzepatide remains expensive, though affordable tirzepatide options are becoming more accessible through compounding pharmacies. The peptide cost calculator can help estimate and compare expenses over treatment periods.
Some clinicians use both medications together. Metformin addresses hepatic glucose production while tirzepatide handles incretin-mediated insulin sensitization, appetite regulation, and weight management. This combination may be particularly effective for women with severe insulin resistance who need maximum metabolic support.
Diet and nutrition on tirzepatide for PCOS
What you eat while taking tirzepatide influences both how well you tolerate the medication and how effectively it addresses PCOS. The appetite reduction is powerful, but it makes every calorie you consume more important because you are eating less overall. Nutritional quality becomes paramount.
Optimizing nutrition for PCOS and tirzepatide together
Women with PCOS already benefit from a lower-glycemic, anti-inflammatory dietary pattern. Tirzepatide amplifies the effectiveness of these dietary choices by improving the metabolic environment in which food is processed. The comprehensive tirzepatide diet plan provides detailed meal frameworks, and the downloadable meal plan offers practical daily templates.
Protein intake deserves special attention. During weight loss, adequate protein intake preserves lean muscle mass and supports satiety between meals. Women on tirzepatide should aim for at least 1.2 to 1.6 grams of protein per kilogram of body weight daily. When appetite is reduced, meeting this target requires deliberate planning, as protein-rich foods are often the first things that become unappealing. Understanding what to eat on tirzepatide helps prioritize the most nutrient-dense options when total food intake is naturally lower.
Certain foods should be minimized or avoided while on tirzepatide, both for PCOS management and medication tolerability. The foods to avoid on tirzepatide include high-fat fried foods that worsen nausea, rapidly digested refined carbohydrates that spike insulin, and carbonated beverages that exacerbate GI discomfort. For PCOS specifically, dairy and high-glycemic foods may worsen insulin resistance and inflammatory markers.
Supplements that support tirzepatide and PCOS treatment
Several supplements complement tirzepatide treatment for PCOS. Inositol, particularly myo-inositol and D-chiro-inositol in a 40:1 ratio, has its own evidence base for improving insulin sensitivity and ovarian function in PCOS. Vitamin D deficiency is common in PCOS and associated with worse metabolic parameters. Omega-3 fatty acids reduce the inflammation that characterizes the condition.
Some compounded tirzepatide formulations include additional compounds. Tirzepatide with B12 is increasingly common, as B12 supports energy metabolism and neurological function. The compounded tirzepatide with B12 formulation is particularly relevant for women also taking metformin, which can deplete B12 over time. Other combination formulations include tirzepatide with glycine, tirzepatide with niacinamide, and the triple compound of tirzepatide with glycine and B12, each designed to address specific nutritional needs during treatment. The methylcobalamin formulation provides a bioactive form of B12 that may be better absorbed than standard cyanocobalamin.
Alcohol and tirzepatide for PCOS
The question of whether you can drink alcohol on tirzepatide requires thoughtful consideration, especially for women with PCOS. Alcohol worsens insulin resistance, increases inflammatory markers, disrupts sleep quality, and provides empty calories that offer no nutritional value during a period when caloric intake is naturally reduced. It also intensifies the nausea and GI side effects of tirzepatide.
For PCOS management specifically, alcohol impairs liver function that is essential for metabolizing hormones and processing excess androgens. It can also disrupt the menstrual cycle improvements that tirzepatide is helping to produce. The general recommendation is to minimize alcohol consumption during treatment, and many women find their tolerance is significantly lower while on the medication.
The rapid rise of GLP-1 prescribing for PCOS
The medical community is catching up to what the data has been showing. GLP-1 prescribing for PCOS has increased from 2.4% to 17.6%, a seven-fold surge that reflects growing recognition of these medications as legitimate PCOS treatments rather than off-label curiosities. This shift is happening faster than many anticipated.
Why physicians are embracing GLP-1 therapy for PCOS
For decades, PCOS treatment followed a predictable script. Lifestyle modifications first. Metformin if insulin resistance was prominent. Oral contraceptives for cycle regulation. Spironolactone for androgen symptoms. Fertility medications when conception was desired. This approach addressed individual symptoms but rarely resolved the underlying metabolic dysfunction driving the condition.
GLP-1 receptor agonists, and tirzepatide in particular, represent a fundamentally different philosophy. By addressing insulin resistance more effectively than any previous pharmaceutical option, and producing weight loss that was previously achievable only through surgery, these medications treat the root cause. The downstream improvements in hormones, cycles, and symptoms follow naturally. Clinicians who have witnessed the transformation in their PCOS patients are becoming advocates for earlier and more aggressive metabolic intervention.
The shift also reflects updated understanding of PCOS as a metabolic disorder first and a reproductive disorder second. When the metabolic environment is corrected, reproductive function tends to normalize on its own, a principle that tirzepatide demonstrates more convincingly than any medication before it. Understanding how long GLP-1 medications take to start working helps both patients and providers set realistic expectations for this process.
Current prescribing landscape
Despite the rapid growth in GLP-1 prescribing for PCOS, access remains uneven. Not all insurance plans cover tirzepatide for this indication, as the FDA approvals are specifically for diabetes and weight management. Women whose BMI qualifies them for the weight management indication often have an easier time obtaining coverage, while normal-weight women with PCOS and insulin resistance may face more barriers.
Compounded formulations have filled some of this access gap. These compounded versions are available at lower cost through specialty pharmacies, though the regulatory landscape continues to evolve. The Empower pharmacy tirzepatide guide and grey market tirzepatide overview discuss the various access pathways, including their respective advantages and risks. International options are also explored by some patients seeking affordability.
Long-term management and what happens when you stop
One of the most important questions about tirzepatide for PCOS is whether the benefits persist after treatment ends. This question does not have a simple answer, because the response depends heavily on what changes occurred during treatment and whether those changes were maintained through other means.
Sustaining improvements after tirzepatide
If tirzepatide treatment produces 15 to 20% weight loss and that weight loss is maintained through dietary habits and exercise after discontinuation, the insulin sensitization, hormonal improvement, and cycle regularity often persist. The medication created a metabolic reset. Sustaining that reset depends on maintaining the conditions, primarily body weight and composition, that support improved insulin sensitivity.
If weight regains after stopping, PCOS symptoms typically return. This is not a failure of the medication. It reflects the chronic nature of PCOS as a metabolic condition that requires ongoing management. Some women may need extended or indefinite treatment, similar to how blood pressure medication is used continuously for hypertension. Others achieve sufficient metabolic improvement and behavioral change during treatment that they can maintain benefits without ongoing medication.
The phenomenon of tirzepatide seeming to stop working sometimes represents a weight loss plateau rather than true treatment failure. Weight loss naturally slows as body weight decreases and metabolic rate adjusts. The strategies for managing GLP-1 plateaus apply equally to tirzepatide. Understanding what happens when getting off GLP-1 medications helps with planning the transition.
Combining tirzepatide with other PCOS treatments
Tirzepatide does not have to be the only treatment. In fact, a multimodal approach often produces the best outcomes for PCOS. The medication addresses metabolic dysfunction and weight management. Other interventions can complement it by targeting specific symptoms.
Spironolactone continues to be valuable for androgen-mediated symptoms like hirsutism and acne, even while tirzepatide reduces testosterone production. The two work through different mechanisms and can be used together safely. For women not seeking pregnancy, oral contraceptives provide additional cycle regulation and androgen suppression. The combination of tirzepatide with these traditional PCOS medications creates a comprehensive treatment approach that addresses the condition from multiple angles simultaneously.
Some women also explore combination approaches with other medications. Whether phentermine can be combined with tirzepatide is a question that arises occasionally, though this combination lacks strong evidence and introduces additional cardiovascular considerations. The phentermine versus GLP-1 comparison helps clarify when each option might be more appropriate.
Choosing the right tirzepatide formulation for PCOS
The tirzepatide market has expanded beyond the original branded injectable pen to include compounded formulations, emerging oral options, and combination products. Understanding the differences helps women make informed choices about which formulation best suits their needs and budget.
Injectable versus oral tirzepatide
The standard and most established form of tirzepatide is the subcutaneous injection. Learning where to inject GLP-1 medications and proper injection technique takes minimal practice and becomes routine quickly. The oral versus injection comparison reveals that injectable forms currently provide the most reliable absorption and dose accuracy.
Oral formulations are emerging as an alternative. The orally dissolving tablet formulation and tirzepatide drops offer needle-free administration options, though bioavailability differs from injectable forms. Women who cannot tolerate injections or prefer oral medication may find these alternatives worth discussing with their healthcare provider. The injectable versus oral peptides comparison page at SeekPeptides explains the pharmacokinetic differences in detail.
Compounded formulations
Compounded tirzepatide is available from specialty pharmacies and typically comes as lyophilized powder that requires reconstitution. The compound dosage chart helps navigate the concentration and dosing specifics that vary between compounding pharmacies. These formulations often cost significantly less than branded products, making long-term treatment more accessible for women managing a chronic condition like PCOS.
Quality varies between compounding pharmacies, so selecting a reputable source matters. The peptide sciences tirzepatide guide covers one established source, but women should verify that any compounding pharmacy they use follows current good manufacturing practices and provides certificates of analysis. The peptide stack calculator can help those using compounded formulations manage multiple compounds simultaneously.
Emerging alternatives and next-generation treatments
The GLP-1 space is evolving rapidly. Mazdutide is another dual agonist in development. Retatrutide is a triple agonist targeting GLP-1, GIP, and glucagon receptors simultaneously. GLP-1 patches are being developed for needle-free transdermal delivery. Even probiotic approaches that naturally boost GLP-1 production are under investigation.
For women with PCOS, these developments are encouraging because they suggest an expanding toolkit for addressing the metabolic dysfunction at the core of their condition. However, tirzepatide remains the most studied and clinically established dual-agonist option currently available.
Real-world outcomes: what women with PCOS are experiencing
Clinical trial data provides the framework, but real-world outcomes tell the human story. Women with PCOS who have used tirzepatide report experiences that range from dramatic transformation to more modest but still meaningful improvement. Understanding the spectrum of outcomes helps set appropriate expectations.
Weight loss trajectories
The tirzepatide before and after results from real patients reveal a pattern that clinical trials confirmed. The first 4 to 8 weeks produce modest weight loss, primarily because the starting doses are sub-therapeutic for weight management. The most rapid weight loss typically occurs between months 2 and 6, as doses escalate into the therapeutic range and the medication full metabolic effects take hold.
The detailed before and after results show that individual variation is significant. Some women lose steadily and consistently throughout treatment. Others experience periods of rapid loss alternating with plateaus. The results differ somewhat between men and women, with hormonal factors playing a role in response patterns. For women with PCOS specifically, the hormonal improvements sometimes precede significant weight loss, suggesting direct metabolic effects beyond caloric deficit.
Hormonal and symptom improvements
Beyond the scale, women with PCOS report improvements that matter more to their daily lives. Acne clearing within 2 to 3 months is common. Hirsutism gradually improves over 4 to 6 months as testosterone levels fall. Energy levels improve as blood sugar stability increases. Mood stabilizes as hormonal cycling normalizes. These quality-of-life improvements are often what women describe as most valuable, even more than the weight loss itself.
Sleep quality frequently improves as well, both because of reduced sleep apnea risk with weight loss and because of improved hormonal balance. Some women experience insomnia initially, but this typically resolves within the first few weeks. The timing of the GLP-1 injection can influence sleep quality, with many women finding that morning or early afternoon administration is better tolerated than evening dosing.
Understanding the BMI and eligibility question
Access to tirzepatide often depends on meeting specific BMI criteria, which creates a particular challenge for women with PCOS. The BMI requirements for GLP-1 medications vary by indication and insurance provider, and understanding these thresholds helps with the practical aspects of obtaining treatment.
For the weight management indication, the general requirement is a BMI of 30 or above, or 27 or above with at least one weight-related comorbidity. PCOS itself, along with insulin resistance, dyslipidemia, and hypertension (all common in PCOS), may qualify as weight-related comorbidities that lower the BMI threshold. This is important because some women with PCOS have BMIs in the 27 to 30 range and might not qualify under the higher threshold alone.
Normal-weight PCOS is a real and recognized phenotype affecting up to 30% of women with the condition. These women have insulin resistance and hormonal dysfunction without significant obesity. Access to tirzepatide for this population is more challenging, as most prescribing criteria are weight-based. Some clinicians prescribe based on the insulin resistance and metabolic dysfunction itself, but insurance coverage may not follow. This is an area where advocacy and evolving clinical guidelines are slowly expanding access.
Combining tirzepatide with exercise for PCOS
Exercise is a cornerstone of PCOS management that becomes even more effective when combined with tirzepatide treatment. The two work synergistically, with the medication improving the metabolic environment in which exercise produces its benefits.
Resistance training and muscle preservation
During any weight loss, preserving lean muscle mass is essential. This is doubly important for PCOS because muscle tissue is a primary site of glucose uptake and insulin-mediated metabolism. More muscle means better insulin sensitivity, which means better hormonal balance. Resistance training 2 to 3 times weekly protects muscle mass during tirzepatide-induced weight loss and may actually improve body composition beyond what the medication achieves alone.
The concern about muscle pain on tirzepatide sometimes discourages women from maintaining exercise routines. In most cases, mild muscle discomfort during weight loss is normal and not a reason to stop training. Distinguishing between typical exercise-related soreness and medication side effects helps women stay consistent with their training programs.
Cardiovascular exercise and insulin sensitivity
Aerobic exercise independently improves insulin sensitivity through mechanisms that complement tirzepatide effects. A single session of moderate-intensity cardio can enhance insulin-mediated glucose uptake for 24 to 72 hours. Regular cardiovascular exercise, combined with tirzepatide insulin-sensitizing effects, creates a cumulative improvement that exceeds what either intervention achieves alone.
For women with PCOS, the combination of reduced caloric intake (from tirzepatide appetite suppression), improved insulin sensitivity (from both medication and exercise), and progressive weight loss creates the metabolic conditions most favorable for hormonal normalization and symptom resolution. The peptides for fat loss resource at SeekPeptides explores how various compounds support body composition goals.
Monitoring progress on tirzepatide for PCOS
Tracking the right metrics ensures you and your healthcare provider can assess whether tirzepatide is producing the desired effects and adjust treatment accordingly. PCOS requires monitoring beyond simple weight checks.
Laboratory markers to track
Fasting insulin and glucose should be checked at baseline and every 3 months during treatment. These are the most direct measures of the insulin resistance improvement that drives all other PCOS benefits. HOMA-IR, a calculated measure of insulin resistance derived from fasting glucose and insulin, provides a single number to track over time.
Testosterone levels, both total and free, should be measured at baseline and at 3 to 6 month intervals. A reduction in free testosterone is often the first laboratory sign that hormonal balance is improving. SHBG levels rise as insulin resistance improves, and tracking this protein provides another confirmation of metabolic improvement.
Lipid panels, liver function tests, and inflammatory markers like C-reactive protein round out the monitoring picture. Women on tirzepatide typically see improvements across all these parameters, confirming the broad metabolic benefit of the medication.
Clinical markers to monitor
Menstrual cycle tracking is perhaps the most practical indicator of PCOS improvement for women not using hormonal contraception. A simple calendar notation of cycle start dates, duration, and flow quality provides invaluable longitudinal data. The transition from irregular or absent cycles to regular monthly periods is one of the clearest signs that treatment is working at the hormonal level.
Body composition changes matter more than scale weight alone. Women who are simultaneously losing fat and building muscle through exercise may see modest changes on the scale while experiencing significant improvements in how their clothes fit, how they feel, and how their metabolic markers look. Waist circumference is a particularly useful measurement, as abdominal fat is the most metabolically active and most directly tied to insulin resistance.
Addressing common concerns about tirzepatide for PCOS
Women considering tirzepatide for PCOS often have specific concerns that go beyond general medication questions. Addressing these directly helps with informed decision-making.
Is tirzepatide safe long-term?
The long-term safety data for tirzepatide continues to accumulate. The medication class, GLP-1 receptor agonists, has been in use since the mid-2000s, and no unexpected long-term safety signals have emerged with extended use. Tirzepatide specifically has been studied in trials lasting up to several years, with a safety profile that remained consistent over time.
For PCOS specifically, the long-term considerations are somewhat different from the general population. Women with PCOS already have elevated cardiovascular risk due to their metabolic profile. Tirzepatide addresses multiple cardiovascular risk factors, including weight, insulin resistance, inflammation, and dyslipidemia, potentially providing a net cardiovascular benefit rather than introducing new risk.
Will my insurance cover tirzepatide for PCOS?
Insurance coverage for tirzepatide specifically for PCOS diagnosis varies widely. Most coverage pathways currently require either a type 2 diabetes diagnosis or meeting BMI criteria for the weight management indication. Women with PCOS who also meet these criteria typically have better luck with coverage. Prior authorization with documentation of insulin resistance, failed metformin therapy, and metabolic comorbidities improves approval rates.
For women without insurance coverage, the affordable tirzepatide guide explores alternatives including compounding pharmacy options, manufacturer savings programs, and other cost-reduction strategies. The cost calculator helps project long-term expenses for different formulation choices.
Can I take tirzepatide while on birth control?
Yes, tirzepatide can be taken concurrently with hormonal birth control. However, there is an important caveat. The slowed gastric emptying caused by tirzepatide may reduce the absorption of oral medications, including oral contraceptive pills. This concern is most relevant during the first few months of treatment and during dose escalation. Women relying on oral contraceptives should discuss backup methods or alternative contraceptive forms, such as IUDs, implants, or rings, with their healthcare provider.
The importance of reliable contraception while on tirzepatide cannot be overstated for PCOS patients. As we discussed, the medication can restore ovulation in women who were previously anovulatory. A woman who has been told she cannot get pregnant because of PCOS may suddenly become fertile. If pregnancy is not desired, this is a scenario that requires proactive planning.
Frequently asked questions
How quickly does tirzepatide improve PCOS symptoms?
Most women notice changes within the first 2 to 4 months of treatment. Appetite reduction and early weight loss begin within weeks, but meaningful hormonal improvements, including cycle restoration and androgen reduction, typically require 3 to 6 months. The tirzepatide weight loss timeline provides detailed expectations for each phase of treatment.
Is tirzepatide FDA-approved specifically for PCOS?
No. Tirzepatide is FDA-approved for type 2 diabetes and chronic weight management. Its use for PCOS is considered off-label, though growing clinical evidence supports its effectiveness. Many endocrinologists and reproductive specialists prescribe it for PCOS based on the strong data linking metabolic improvement to hormonal normalization. The seven-fold increase in GLP-1 prescribing for PCOS reflects this clinical confidence.
Can lean women with PCOS benefit from tirzepatide?
Potentially yes. Even normal-weight women with PCOS often have insulin resistance that drives their hormonal dysfunction. Tirzepatide insulin-sensitizing effects may benefit these patients independently of significant weight loss. However, access and insurance coverage may be more challenging, and lower doses are typically used. The microdosing approach may be particularly appropriate for lean PCOS patients.
What happens to PCOS symptoms if I stop taking tirzepatide?
If weight regains after stopping, PCOS symptoms typically return because the underlying metabolic condition has not changed. However, women who maintain their weight loss through lifestyle modifications often sustain their hormonal improvements. Some clinicians recommend ongoing low-dose treatment for PCOS patients, similar to how long-term GLP-1 treatment is used for weight maintenance. Understanding potential withdrawal effects helps with planning discontinuation.
Can I take tirzepatide while trying to get pregnant?
Tirzepatide should be discontinued before actively trying to conceive, typically at least 2 months prior. However, some fertility specialists use tirzepatide as a pre-treatment to optimize metabolic parameters before conception attempts, discontinuing it before the actual trying-to-conceive window. The experiences of women who became pregnant on tirzepatide highlight the importance of contraception planning during treatment if pregnancy is not desired.
Does tirzepatide work better than semaglutide for PCOS?
Tirzepatide produces greater average weight loss (17.60 kg versus 11.85 kg for semaglutide), and its dual-receptor mechanism provides theoretical advantages for insulin sensitization. However, both medications significantly improve PCOS symptoms. The detailed comparison helps determine which may be more appropriate for individual circumstances. Cost, availability, and personal response all influence which medication is the better choice for a specific patient.
What is the best starting dose of tirzepatide for PCOS?
The standard starting dose is 2.5 mg once weekly, maintained for at least 4 weeks before considering escalation. Some clinicians keep PCOS patients at this dose or at 5 mg longer than the standard titration schedule, particularly when the primary goal is hormonal normalization rather than maximum weight loss. The starting dose guide and full dose chart provide comprehensive dosing information.
Are there any interactions between tirzepatide and common PCOS medications?
Tirzepatide can be used with metformin, spironolactone, and most other common PCOS medications without significant drug interactions. The main pharmacokinetic concern is that slowed gastric emptying may affect absorption of oral medications. This is most relevant for oral contraceptives and medications that require consistent blood levels. Taking oral medications at least 1 hour before the tirzepatide injection can help mitigate this concern.
External resources
National Library of Medicine - Tirzepatide and GLP-1 agonists in PCOS research
American College of Obstetricians and Gynecologists - PCOS FAQ
For researchers serious about understanding the full scope of GLP-1 therapy for hormonal and metabolic conditions, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, detailed protocols, and a community of thousands who navigate these exact questions every day.
In case I do not see you, good afternoon, good evening, and good night. May your cycles stay regular, your insulin stay sensitive, and your hormones stay balanced.