Feb 6, 2026

Some users report appetite suppression within days of starting Evolv GLP-1. Others take the tablets for weeks with minimal changes. Same product, same recommended dosage, completely different outcomes. The difference is not about the supplement itself but how it fits into the broader GLP-1 landscape, what realistic expectations look like, and whether biomimetic approaches can deliver what traditional peptides do without needles.
Evolv GLP-1 represents a fundamentally different approach to GLP-1 activation. Instead of injecting synthetic peptides that mimic glucagon-like peptide-1, this oral supplement uses engineered yeast to produce a molecule designed to engage GLP-1 and GIP receptors naturally. The concept is compelling. The execution involves cutting-edge biotechnology. But the clinical evidence remains incomplete, and understanding where this product fits requires examining what GLP-1 actually does, how traditional agonists work, and what biomimetic alternatives can realistically achieve.
The challenges with evaluating Evolv GLP-1 are significant. At the time of this writing, no peer-reviewed human studies demonstrate weight loss or metabolic benefits. The company has completed three studies and is conducting a fourth 90-day trial with 120 participants, but published data remains limited to company-run in-vitro assays, animal safety studies, and user testimonials. This is not inherently problematic for a new product, but it means we are working with preliminary evidence rather than established clinical proof.
This guide examines Evolv GLP-1 from every angle that matters. We will explore the science behind biomimetic GLP-1 activation, compare it with traditional semaglutide and tirzepatide protocols, analyze the available research data, review realistic dosing approaches, and identify who might benefit from this supplement versus who should stick with proven peptide therapies. For those navigating the expanding GLP-1 space, SeekPeptides provides comprehensive comparisons of traditional peptides, emerging alternatives, and evidence-based protocols.
What Evolv GLP-1 actually is
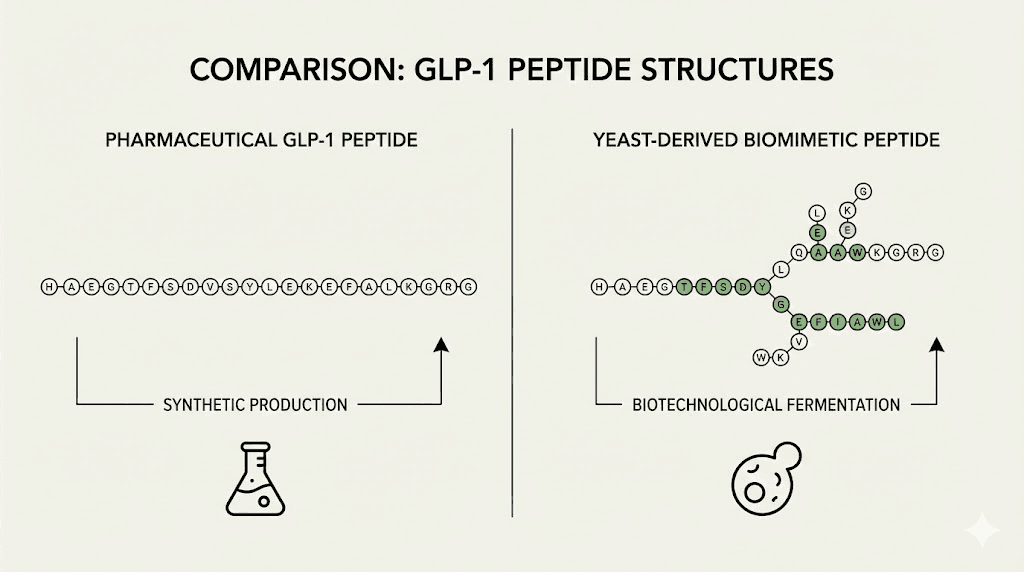
Evolv GLP-1 is not a peptide in the traditional sense. It is a biomimetic supplement, meaning it uses biological components engineered to mimic natural physiological processes. The active ingredient, EV1-Peptide, comes from a proprietary yeast strain modified through genetic engineering to produce a molecule that engages both GLP-1 receptors and glucose-dependent insulinotropic polypeptide (GIP) receptors.
This is fundamentally different from how semaglutide or tirzepatide work. Those peptides are synthetic molecules designed to resist degradation by the enzyme dipeptidyl peptidase-4 (DPP-4), which normally breaks down endogenous GLP-1 within minutes. By modifying the peptide structure, pharmaceutical companies extended the half-life to days or weeks, allowing once-weekly injections to maintain therapeutic levels.
Evolv takes a different route.
Instead of synthesizing a DPP-4-resistant peptide, Evolv uses yeast fermentation to create a molecule composed of canonical amino acids that the company claims has a half-life of 1-2 days. This molecule is not broken down as quickly by DPP-4, allowing oral administration to maintain receptor engagement longer than endogenous GLP-1 would naturally persist.
The yeast-derived approach is not entirely new. Yeast fermentation has been used for decades to produce recombinant proteins, including insulin and various therapeutic peptides. What makes Evolv distinct is the claim that their engineered yeast produces a peptide specifically optimized for oral bioavailability and receptor activation without requiring injection. Understanding oral versus injectable peptide delivery helps contextualize why this matters.
The engineering behind EV1-Peptide
The EV1-Peptide is produced through genetic modification of baker's yeast. The company describes this as creating a yeast hydrolysate, which is essentially a broken-down yeast product containing the active peptide fraction. The genetic modification instructs the yeast to produce a specific amino acid sequence that interacts with GLP-1 and GIP receptors.
The peptide is composed solely of canonical amino acids, the same building blocks found in dietary protein and endogenous peptides. This is important because non-canonical amino acids can trigger immune responses or create toxicity concerns. By sticking with standard amino acids, Evolv reduces the risk of adverse immunological reactions.
In-vitro receptor activation assays demonstrated that the molecule interacts with GLP-1 and GIP receptors. These assays involve isolating receptor proteins and measuring how effectively the test molecule binds and activates downstream signaling pathways. Positive results in these assays indicate the molecule has the structural characteristics needed for receptor engagement. However, in-vitro activity does not guarantee in-vivo efficacy. Many compounds that bind receptors in a dish fail to produce meaningful effects in living organisms due to poor bioavailability, rapid metabolism, or insufficient tissue distribution. For context, understanding peptide bioavailability is essential when evaluating oral delivery claims.
How biomimetic differs from pharmaceutical peptides
Pharmaceutical GLP-1 agonists like semaglutide and tirzepatide are precision-engineered molecules with extensive modifications to enhance stability and extend half-life. Semaglutide, for example, includes a fatty acid side chain that binds to albumin in the bloodstream, protecting it from enzymatic degradation and extending its half-life to approximately one week.
Tirzepatide goes further by engaging both GLP-1 and GIP receptors through a dual agonist mechanism, which appears to enhance weight loss and glycemic control beyond what GLP-1 activation alone achieves. The pharmaceutical development process for these peptides involves years of structural optimization, pharmacokinetic studies, dose-finding trials, and large-scale randomized controlled trials. By the time a peptide like semaglutide reaches market, thousands of patients have been studied, and efficacy is well-documented. Those interested in comparing these approaches should explore semaglutide versus tirzepatide mechanisms.
Biomimetic products do not follow the same development pathway. They are classified as dietary supplements rather than pharmaceuticals, which means they are not subject to the same regulatory scrutiny or clinical trial requirements. This allows faster time to market but also means the evidence base is thinner when products launch.
Evolv's yeast-derived peptide is not structurally identical to pharmaceutical GLP-1 agonists. It is a novel molecule designed to achieve similar receptor activation through a different mechanism. Whether this translates to comparable efficacy in humans remains an open question. For those exploring alternatives to injectable peptides, oral semaglutide represents another non-injection option with more established clinical data.
The science of GLP-1 and GIP receptor activation
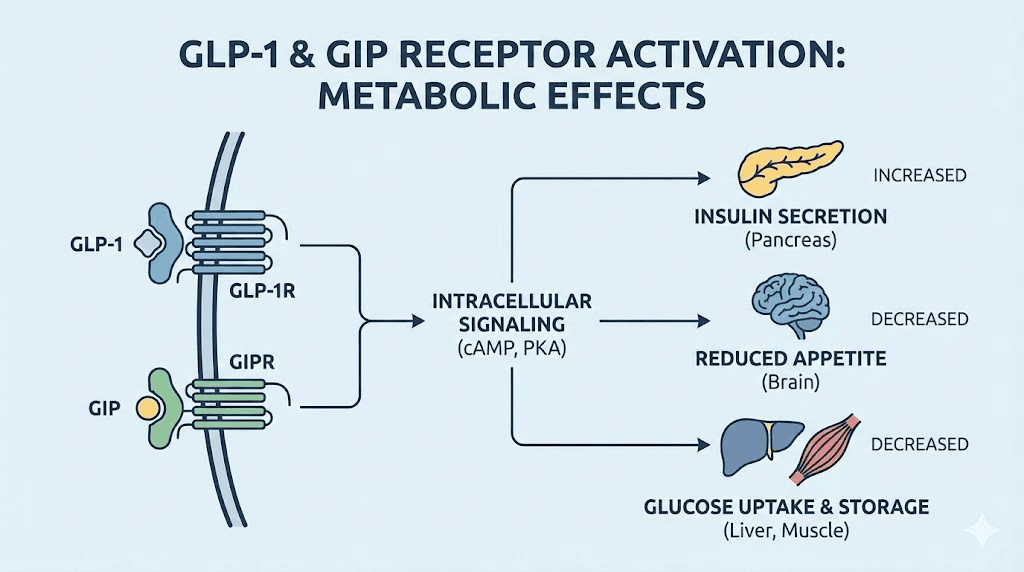
To understand what Evolv GLP-1 is attempting to do, you need to understand what GLP-1 and GIP actually do in the body. These are incretin hormones, meaning they are secreted by the gut in response to food intake and regulate glucose metabolism and appetite.
GLP-1 is produced by enteroendocrine L-cells in the distal ileum and colon. When you eat, especially foods containing carbohydrates or fats, these cells release GLP-1 into the bloodstream. GLP-1 then acts on multiple tissues. In the pancreas, it stimulates insulin secretion from beta cells in a glucose-dependent manner, meaning insulin release only occurs when blood glucose is elevated.
This reduces hypoglycemia risk compared to insulin or sulfonylureas.
GLP-1 also suppresses glucagon secretion from pancreatic alpha cells, reducing hepatic glucose production. In the brain, GLP-1 receptors in the hypothalamus and brainstem reduce appetite and food intake. In the stomach, GLP-1 slows gastric emptying, which prolongs satiety and reduces postprandial glucose spikes. These combined effects make GLP-1 a powerful regulator of metabolism and body weight. Understanding GLP-1 mechanisms in detail provides context for why activating these pathways matters.
The problem is that endogenous GLP-1 has a half-life of approximately 2 minutes. It is rapidly degraded by DPP-4, an enzyme expressed throughout the body. This short half-life is physiologically appropriate because it allows rapid on-off control of insulin secretion and appetite signaling. But it also means that simply taking oral GLP-1 would be ineffective, as it would be degraded before reaching systemic circulation.
Why GIP matters for dual agonism
Glucose-dependent insulinotropic polypeptide (GIP) is another incretin hormone, produced by enteroendocrine K-cells in the proximal small intestine. Like GLP-1, GIP stimulates insulin secretion in a glucose-dependent manner. It also promotes fat storage in adipose tissue and has complex effects on bone metabolism.
For years, GIP was considered less important for glucose control than GLP-1 because patients with type 2 diabetes often show reduced GIP responsiveness. However, the success of tirzepatide, which activates both GLP-1 and GIP receptors, demonstrated that dual agonism produces superior weight loss and glycemic control compared to GLP-1 agonism alone.
The mechanisms behind this are not fully understood. One hypothesis is that GIP activation enhances energy expenditure and lipolysis in adipose tissue, counteracting its lipogenic effects when combined with GLP-1 activation. Another possibility is that GIP improves beta-cell function and insulin sensitivity in ways that complement GLP-1's effects. Whatever the mechanism, the clinical data is clear. Dual agonists outperform single GLP-1 agonists for weight loss. Those comparing options should review tirzepatide versus semaglutide outcomes.
Evolv's claim to engage both GLP-1 and GIP receptors positions it as a biomimetic dual agonist, similar in concept to tirzepatide but delivered through an entirely different mechanism. Whether the yeast-derived peptide achieves sufficient receptor occupancy and downstream signaling to replicate pharmaceutical dual agonist effects is the central question.
Receptor binding versus physiological effect
Demonstrating that a molecule binds to GLP-1 and GIP receptors in vitro is a necessary but insufficient condition for therapeutic efficacy. Receptor binding affinity, intrinsic activity, and duration of receptor occupancy all influence downstream signaling.
Pharmaceutical peptides are optimized for high-affinity binding and prolonged receptor engagement. Semaglutide, for instance, binds the GLP-1 receptor with similar affinity to native GLP-1 but remains bound much longer due to its albumin-binding properties. This sustained receptor occupancy produces continuous signaling, which drives the therapeutic effects.
A molecule that binds weakly or transiently may not produce sufficient signaling to impact appetite, glucose metabolism, or body weight. Additionally, oral bioavailability is a major hurdle. Peptides are notoriously difficult to deliver orally because they are degraded by gastric acid and proteolytic enzymes in the GI tract. Even if a peptide survives digestion, it must cross the intestinal epithelium to reach systemic circulation, which is challenging for molecules composed of amino acids.
Evolv claims their yeast-derived peptide overcomes these challenges, but the mechanisms by which this occurs are not publicly detailed. Without published pharmacokinetic data showing plasma levels after oral administration, it is difficult to assess whether the peptide reaches concentrations sufficient for receptor activation. This contrasts with oral tirzepatide formulations, which use absorption enhancers to improve bioavailability.
Available research and clinical evidence
As of this writing, Evolv has completed three studies on their formulation and is midway through a fourth. The completed studies measured safety and efficacy across key biomarkers related to appetite regulation, metabolic function, and nutrient absorption. The ongoing study is a 90-day trial with 120 participants examining weight loss, appetite suppression, and HbA1C changes.
None of these studies have been published in peer-reviewed journals. The available data comes from company-run in-vitro assays, animal safety and toxicity studies, and anecdotal user reports. This is not unusual for a newly launched supplement, but it means we are evaluating a product based on preliminary evidence rather than established clinical proof.
In-vitro receptor activation assays
The company conducted in-vitro studies demonstrating that EV1-Peptide binds and activates GLP-1 and GIP receptors. These assays typically involve expressing the receptor in cell lines and measuring downstream signaling events, such as cyclic AMP production or calcium flux, when the test molecule is added.
Positive results indicate the molecule has the structural features needed to engage the receptor. However, in-vitro activity does not predict in-vivo efficacy. The concentrations used in cell assays may be far higher than what is achievable in human plasma after oral dosing. Additionally, in-vitro assays do not account for metabolism, distribution, or the complex physiological environment in which the peptide must function.
For context, many peptides show strong receptor activation in vitro but fail to produce clinical effects because they cannot be delivered at effective concentrations in living organisms. Understanding peptide receptor pharmacology clarifies why in-vitro results are just the first step.
Animal safety and appetite studies
Evolv conducted animal studies to evaluate safety and toxicity. These studies are essential for identifying potential adverse effects before human trials. The company reports that no significant safety concerns were identified, and qualitative appetite signals suggested reduced food intake in treated animals.
Animal data provides useful directional information but often does not translate directly to humans. Rodents, for example, metabolize drugs differently than humans, and their behavioral responses to appetite-suppressing compounds can differ. Additionally, qualitative observations of appetite are less rigorous than quantitative measurements of food intake, body weight, or fat mass.
Without published details on the animal study design, sample sizes, dosing protocols, and statistical analyses, it is difficult to assess the robustness of these findings. Comparing this with the extensive preclinical work behind semaglutide's development illustrates the difference in evidentiary standards between pharmaceutical peptides and dietary supplements.
Ongoing human clinical trial
The most informative data will come from the ongoing 90-day randomized controlled trial with 120 participants. This study is examining weight loss, appetite suppression, and HbA1C changes, which are the primary outcomes of interest for a GLP-1 agonist.
Key questions this trial should answer include:
1. What is the magnitude of weight loss? Pharmaceutical GLP-1 agonists produce 10-20% body weight reduction over 6-12 months in clinical trials. If Evolv produces 2-3% weight loss over 90 days, it suggests modest efficacy. If it produces 7-10%, it would be competitive with pharmaceutical options.
2. Is the effect statistically significant compared to placebo? Randomized, placebo-controlled trials are the gold standard for demonstrating efficacy. If Evolv participants lose significantly more weight than placebo participants, it strengthens the case for genuine GLP-1 activation.
3. What is the dropout rate and adverse event profile? High dropout rates or significant side effects would limit practical utility even if efficacy is demonstrated.
4. Are there biomarker changes consistent with GLP-1 activation? Reductions in HbA1C, fasting glucose, or insulin levels would support the mechanism of action.
Until this trial is completed and published, we are evaluating Evolv based on incomplete information. Users considering this supplement should understand they are trying a product without established clinical proof. Those seeking evidence-based approaches should explore GLP-1 clinical trial evidence for pharmaceutical options.
User testimonials and anecdotal reports
The company and early users report appetite suppression, reduced food noise, and weight loss. These reports are valuable for generating hypotheses but cannot substitute for controlled trials. Anecdotal evidence is subject to placebo effects, reporting bias, and confounding variables like concurrent diet or exercise changes.
Placebo responses in weight loss trials are substantial. Studies show that participants receiving placebo often lose 2-5% of body weight through increased attention to diet, regular weigh-ins, and behavioral support provided by trial participation. Without a placebo control group, it is impossible to determine how much of the reported benefit is due to the supplement versus these non-specific factors.
This does not mean user reports are worthless. They provide real-world perspectives on tolerability, subjective effects, and practical considerations. But they cannot establish efficacy. For comparison, semaglutide user experiences are supported by randomized controlled trial data showing consistent, reproducible effects.
Dosage protocols and practical use
Evolv recommends starting with two tablets daily and adjusting up to three tablets daily as desired. Each bottle contains 30 tablets, costing approximately $148, which translates to $4.93 per day at two tablets or $7.40 per day at three tablets.
The lack of specific dosing guidelines reflects the product's supplement status rather than pharmaceutical development. Pharmaceutical peptides undergo dose-finding studies to determine the minimum effective dose, maximum tolerated dose, and optimal dose for balancing efficacy and side effects. Evolv's recommendation to start at two tablets and adjust as needed places the burden of optimization on the user.
Starting protocol for new users
For those trying Evolv GLP-1, a conservative approach minimizes side effects while allowing assessment of individual response. Start with one tablet daily for the first 3-5 days. This is lower than the recommended starting dose but allows observation of initial effects without overwhelming the system.
If no side effects occur and appetite suppression is minimal, increase to two tablets daily (the recommended starting dose). Take one tablet in the morning and one in the early afternoon. Splitting the dose may provide more consistent receptor engagement throughout the day compared to taking both tablets at once.
Maintain two tablets daily for at least 2-3 weeks before considering further increases. GLP-1 effects can take time to manifest fully, and premature dose escalation increases side effect risk without necessarily improving outcomes. Monitor appetite, food intake, body weight, and any gastrointestinal symptoms during this period.
If appetite suppression is inadequate after 2-3 weeks at two tablets, increase to three tablets daily. Distribute these as one tablet in the morning, one at midday, and one in the early afternoon. Avoid taking tablets in the evening, as appetite suppression may interfere with dinner or cause nocturnal GI symptoms. For comparison, understanding semaglutide dosing schedules illustrates how pharmaceutical peptides approach titration.
Timing and administration considerations
Evolv does not specify whether tablets should be taken with or without food. For oral peptides, food can influence absorption, though the direction of the effect varies by formulation. Some oral peptides are taken fasting to maximize absorption, while others are taken with food to reduce GI irritation.
In the absence of specific guidance, a reasonable approach is to take tablets 30-60 minutes before meals. This timing leverages the appetite-suppressing effects to reduce meal size and may improve absorption if gastric emptying influences bioavailability. If GI side effects occur, taking tablets with a small amount of food may improve tolerance.
Consistency is critical. Take tablets at the same times each day to maintain steady plasma levels and predictable effects. Erratic dosing makes it difficult to assess whether the supplement is working and complicates side effect management. Those familiar with peptide timing strategies will recognize these principles.
Adjusting based on response
Response to GLP-1 activation varies widely. Some individuals are highly sensitive and experience strong appetite suppression at low doses. Others require higher doses to achieve meaningful effects. This variability is seen with pharmaceutical GLP-1 agonists and likely applies to biomimetic alternatives.
If appetite suppression is strong at two tablets daily and you are losing weight at a desirable rate (0.5-1% body weight per week), there is no reason to increase the dose. Higher doses do not necessarily accelerate weight loss and may increase side effects.
If appetite suppression is minimal after 3-4 weeks at three tablets daily, the product may not be effective for you. Some individuals may not absorb the yeast-derived peptide effectively, or their receptors may be less responsive to this particular molecule. In this case, continuing the supplement is unlikely to produce better results, and switching to a proven pharmaceutical option may be more effective. Exploring GLP-1 non-responder phenotypes can provide insight into individual variability.
Cost considerations and comparisons
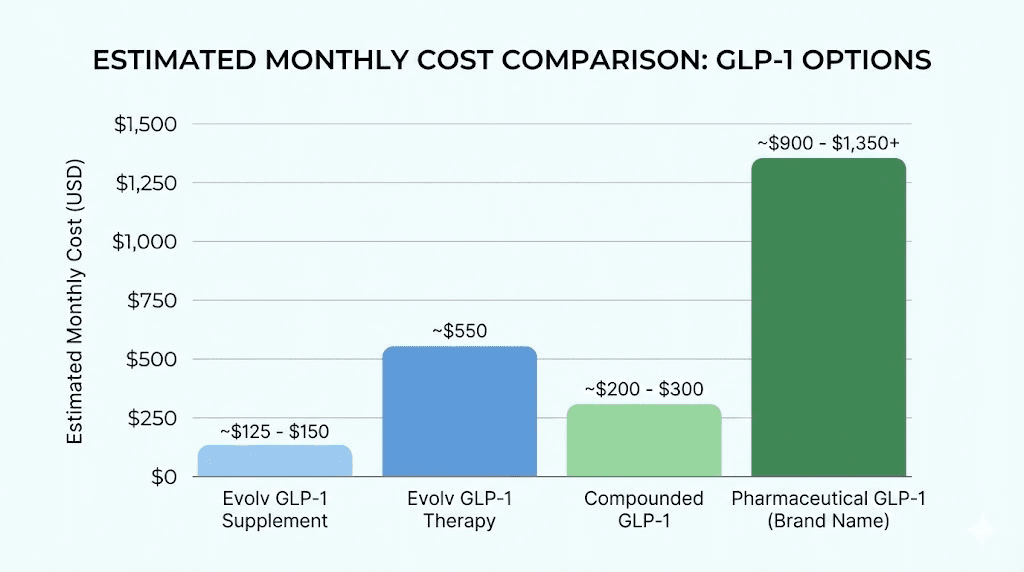
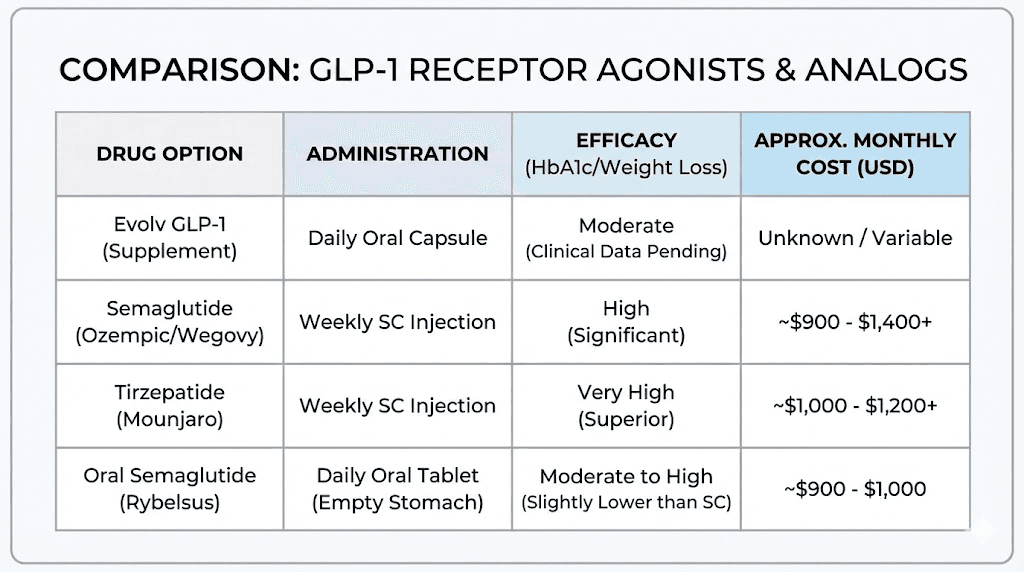
At two tablets daily, a 30-tablet bottle lasts 15 days, costing approximately $296 per month. At three tablets daily, the cost rises to $444 per month. This is comparable to or higher than compounded semaglutide, which costs $200-400 per month depending on the source and dosage.
Pharmaceutical semaglutide (Wegovy) costs approximately $1,300 per month without insurance, making Evolv appear more affordable. However, compounded semaglutide and tirzepatide are widely available at lower costs and have established efficacy. The cost-benefit analysis depends on whether Evolv produces meaningful weight loss. If it achieves 50% of pharmaceutical efficacy at 50% of the cost, it may be a reasonable option. If it produces minimal effects, any cost is wasted. Understanding peptide cost comparisons helps evaluate value.
Comparing Evolv GLP-1 with traditional peptides
The most relevant comparisons are with semaglutide, tirzepatide, and oral semaglutide formulations. Each has different mechanisms, administration methods, efficacy profiles, and side effect patterns.
Evolv GLP-1 versus injectable semaglutide
Injectable semaglutide is administered subcutaneously once weekly. It produces dose-dependent weight loss, with most patients losing 10-15% of body weight over 6-12 months at therapeutic doses. The efficacy is well-established through multiple randomized controlled trials involving thousands of participants.
Semaglutide's primary advantages are proven efficacy, predictable dosing, and extensive safety data. The main disadvantages are the need for weekly injections, potential GI side effects (nausea, vomiting, diarrhea), and cost. Understanding semaglutide injection technique reduces discomfort for those using this option.
Evolv's potential advantages are oral administration, no needles, and lower cost than brand-name pharmaceutical peptides. The disadvantages are unproven efficacy, lack of peer-reviewed data, and unclear long-term safety. If Evolv produces even 50-70% of semaglutide's weight loss without injections, it would be compelling for needle-averse individuals. If efficacy is substantially lower, the convenience of oral administration may not justify the cost.
Evolv GLP-1 versus tirzepatide
Tirzepatide is a dual GLP-1 and GIP agonist, administered subcutaneously once weekly. It produces greater weight loss than semaglutide, with average reductions of 15-20% body weight in clinical trials. The dual agonist mechanism appears to enhance both appetite suppression and metabolic effects.
Evolv claims to engage both GLP-1 and GIP receptors, positioning it as a biomimetic dual agonist. If this claim is accurate and the yeast-derived peptide achieves sufficient receptor occupancy, Evolv could theoretically produce effects similar to tirzepatide. However, without clinical data demonstrating dual agonist efficacy, this remains speculative. For detailed comparisons, see tirzepatide dosing protocols.
Tirzepatide's main disadvantages are the need for weekly injections, higher incidence of GI side effects compared to semaglutide, and cost. If Evolv produces comparable weight loss orally, it would be a significant advancement. If efficacy is substantially lower, tirzepatide's superior weight loss may justify the inconvenience of injections.
Evolv GLP-1 versus oral semaglutide
Oral semaglutide (Rybelsus) is a pharmaceutical formulation combining semaglutide with an absorption enhancer (SNAC) to improve bioavailability. It is taken daily on an empty stomach with a small amount of water, and patients must wait 30 minutes before eating or drinking.
Oral semaglutide produces weight loss comparable to low-dose injectable semaglutide but less than high-dose injections. Clinical trials show approximately 5-10% body weight reduction, which is meaningful but lower than what weekly injections achieve. The advantage is avoiding needles. The disadvantages are strict administration requirements, daily dosing, and lower efficacy than injections. Detailed protocols are covered in oral semaglutide guides.
Evolv GLP-1 is simpler to take than oral semaglutide (no fasting requirement, no waiting period) but lacks the clinical validation that Rybelsus has. If Evolv produces weight loss in the 5-10% range, it would be directly comparable to oral semaglutide with more convenient dosing. If efficacy is lower, oral semaglutide's established track record makes it the safer choice.
Side effect profile comparison
The most common side effects of pharmaceutical GLP-1 agonists are gastrointestinal: nausea, vomiting, diarrhea, constipation, and abdominal discomfort. These typically occur during dose escalation and diminish over time as the body adapts. Severe GI side effects can lead to discontinuation in 5-10% of users. Managing these effects is covered in semaglutide side effect guides.
Evolv has not published detailed adverse event data. User reports suggest some individuals experience mild GI discomfort, but the incidence and severity are unclear. If Evolv produces meaningful GLP-1 activation, it should cause similar side effects to pharmaceutical agonists. If side effects are minimal, it may indicate weak receptor activation and limited efficacy. The absence of significant side effects is not necessarily reassuring if it reflects insufficient pharmacological activity.
Other potential concerns with long-term GLP-1 agonist use include gallbladder disease (cholelithiasis, cholecystitis), pancreatitis, and potential effects on thyroid C-cells. Pharmaceutical peptides have extensive safety monitoring and post-market surveillance. Evolv, as a dietary supplement, is not subject to the same reporting requirements, making long-term safety harder to assess.
Who should consider Evolv GLP-1
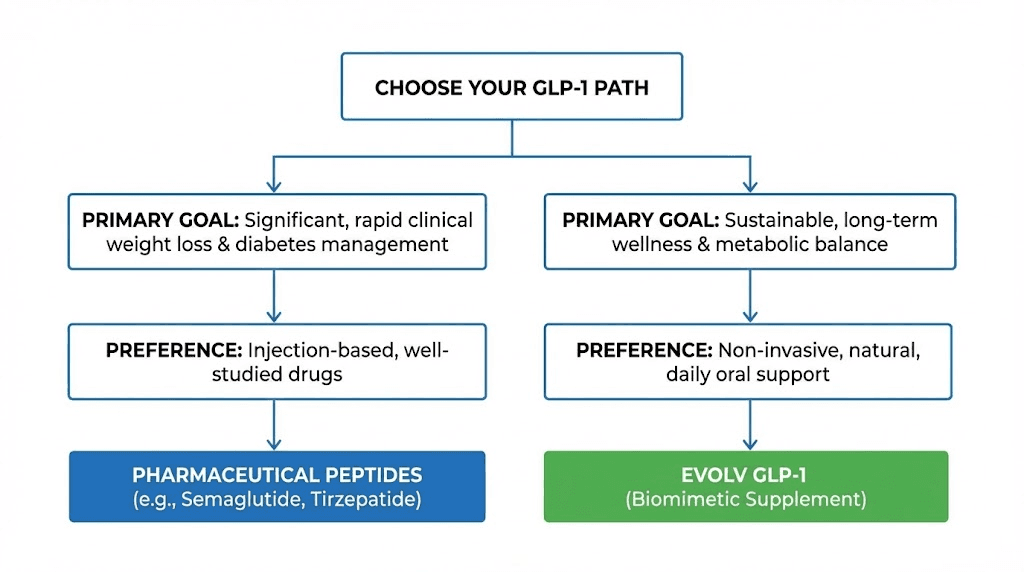
The decision to try Evolv GLP-1 depends on individual circumstances, preferences, risk tolerance, and weight loss goals. Different user profiles will find different risk-benefit ratios.
Needle-averse individuals seeking modest weight loss
If you are strongly opposed to injections and need to lose 10-20 pounds, Evolv may be worth trying. The oral administration eliminates needle anxiety, and if efficacy is even modest (3-5% body weight loss), it could help you reach your goal without pharmaceutical peptides.
The downside is cost and uncertainty. At $300-450 per month, you are paying a premium for unproven efficacy. If the product does not work for you, you have spent considerable money with no benefit. A trial period of 2-3 months is reasonable to assess response. If appetite suppression and weight loss are minimal, discontinue and consider alternatives. Exploring non-injection weight loss options provides additional context.
Early adopters willing to experiment
If you are comfortable trying novel compounds before extensive clinical data is available, Evolv represents an opportunity to test biomimetic GLP-1 activation. You may be among the first to experience a new approach to metabolic health, and your self-experimentation can inform future decisions.
The risks are financial (potentially wasting money on an ineffective product) and opportunity cost (time spent trying Evolv could be spent on proven alternatives). The potential reward is finding an effective oral GLP-1 solution before it becomes mainstream. Document your experience carefully, track weight, appetite, and side effects, and share your findings with communities interested in metabolic optimization.
Individuals seeking to avoid pharmaceutical side effects
Some people experience severe GI side effects with semaglutide or tirzepatide that limit tolerability. If you have tried pharmaceutical peptides and discontinued due to side effects, Evolv might be better tolerated.
However, if Evolv genuinely activates GLP-1 receptors, it should produce similar side effects. The mechanisms causing nausea (delayed gastric emptying, central appetite suppression) are intrinsic to GLP-1 activation, not specific to pharmaceutical formulations. If Evolv lacks GI side effects, it may indicate weak receptor activation and limited efficacy rather than better tolerability. Managing side effects is discussed in GLP-1 side effect management guides.
Who should avoid Evolv GLP-1
If you need significant weight loss (more than 20% body weight), proven pharmaceutical peptides are the better choice. Semaglutide and tirzepatide have demonstrated efficacy in this range. Evolv's efficacy is unproven, and gambling on an unvalidated product when you need substantial weight loss is not advisable.
If you have type 2 diabetes requiring glycemic control, pharmaceutical GLP-1 agonists are FDA-approved for this indication and have extensive evidence supporting their use. Evolv is marketed as a supplement for healthy individuals seeking weight management, not a diabetes treatment. Using an unproven supplement for diabetes management is risky. Pharmaceutical options are covered in semaglutide for diabetes guides.
If you have a history of pancreatitis, gallbladder disease, or medullary thyroid carcinoma, GLP-1 agonists (including biomimetic alternatives) carry potential risks. Consult a physician before using any GLP-1-activating compound.
If cost is a major concern and you need proven efficacy, compounded semaglutide or tirzepatide may provide better value. While Evolv is cheaper than brand-name pharmaceuticals, it is comparable to compounded options that have established efficacy.
Combining Evolv GLP-1 with other interventions
GLP-1 activation is not a standalone solution for weight loss or metabolic health. It works best when combined with dietary modifications, exercise, sleep optimization, and other lifestyle interventions. Evolv should be viewed as one component of a comprehensive approach.
Dietary strategies to enhance GLP-1 effects
GLP-1 agonists reduce appetite, but food choices still matter. High-protein diets enhance satiety and preserve lean mass during weight loss. Aim for 1.2-2.0 grams of protein per kilogram of body weight daily. Distribute protein across meals rather than concentrating it in one sitting. Understanding protein intake strategies with GLP-1 optimizes results.
High-fiber foods slow gastric emptying and promote satiety, complementing GLP-1's effects. Include vegetables, legumes, whole grains, and fruits in most meals. Fiber also supports gut microbiome health, which influences metabolic function and inflammation.
Minimize ultra-processed foods high in refined carbohydrates and added sugars. These foods are engineered to override satiety signals, counteracting appetite suppression. Even with GLP-1 activation, consuming hyperpalatable processed foods can undermine weight loss.
Hydration is critical. GLP-1 agonists can reduce thirst perception, and dehydration worsens side effects like constipation and fatigue. Drink at least 2-3 liters of water daily, more if exercising or in hot environments.
Exercise and movement optimization
Exercise enhances weight loss, preserves lean mass, and improves metabolic health independently of GLP-1 effects. Resistance training is particularly important during weight loss to maintain muscle mass. Losing muscle along with fat worsens metabolic rate and functional capacity. Aim for 3-4 resistance training sessions per week, focusing on compound movements like squats, deadlifts, presses, and rows. Strategies are detailed in resistance training with GLP-1 guides.
Cardiovascular exercise supports calorie expenditure and cardiovascular health. Include 150-300 minutes of moderate-intensity activity per week, such as brisk walking, cycling, or swimming. High-intensity interval training (HIIT) can be effective but may be harder to sustain if GLP-1 side effects reduce energy levels.
Non-exercise activity thermogenesis (NEAT) contributes substantially to daily calorie expenditure. Walking more, taking stairs, and staying active throughout the day can add several hundred calories of expenditure without formal exercise sessions.
Sleep and stress management
Poor sleep undermines weight loss by increasing hunger hormones (ghrelin), reducing satiety hormones (leptin), and impairing insulin sensitivity. Aim for 7-9 hours of quality sleep per night. Prioritize sleep hygiene: consistent bed and wake times, dark and cool bedroom, limited screen time before bed. Sleep optimization is covered in sleep and metabolic health guides.
Chronic stress elevates cortisol, which promotes fat storage, increases appetite, and impairs glucose metabolism. Stress management practices like meditation, deep breathing, yoga, and time in nature can mitigate these effects. Even 10-15 minutes of daily stress reduction practice can improve outcomes.
Stacking with other peptides or supplements
Some individuals combine GLP-1 agonists with other peptides or supplements to enhance weight loss or address specific goals. BPC-157 is popular for gut healing and may help manage GI side effects from GLP-1 activation. CJC-1295 with ipamorelin is sometimes stacked to preserve lean mass during weight loss by increasing growth hormone secretion.
Metformin is occasionally combined with GLP-1 agonists for additional metabolic benefits, particularly in individuals with insulin resistance or prediabetes. Metformin improves insulin sensitivity, reduces hepatic glucose production, and may have longevity benefits. However, combining multiple interventions increases complexity and makes it harder to attribute effects to specific compounds.
Berberine is a plant-derived compound with effects on glucose metabolism and gut microbiome composition. Some users combine it with GLP-1 agonists for additional glycemic control. Omega-3 fatty acids support cardiovascular health and may reduce inflammation during weight loss. Vitamin D, magnesium, and other micronutrients should be maintained at adequate levels to support metabolic function. Stacking strategies are explored in peptide stacking guides.
Troubleshooting common issues
Even if Evolv GLP-1 works as intended, users may encounter challenges that require adjustment. Recognizing these issues early and responding appropriately improves outcomes.
Minimal appetite suppression
If you experience little to no appetite suppression after 2-3 weeks at the recommended dose, several factors could be responsible. First, you may be a non-responder. Individual variability in GLP-1 receptor sensitivity, peptide absorption, and metabolic pathways means some people respond strongly while others see minimal effects. If you are a non-responder to Evolv, pharmaceutical peptides with proven bioavailability may work better. Non-responder patterns are discussed in GLP-1 non-responder guides.
Second, the product may not be delivering sufficient receptor activation. If the yeast-derived peptide has poor oral bioavailability or weak receptor binding, it may not produce meaningful effects even in individuals who would respond to pharmaceutical peptides.
Third, your baseline eating patterns may be masking subtle effects. If you consume ultra-processed foods or eat in response to cues other than hunger (stress, boredom, social situations), reduced physiological appetite may not translate to reduced food intake. Focus on eating only when physically hungry and prioritize whole, minimally processed foods to test whether appetite suppression is present but being overridden by environmental factors.
GI side effects: nausea, vomiting, diarrhea
GI side effects are the most common issue with GLP-1 activation. If nausea occurs, reduce the dose temporarily. Drop from three tablets to two, or from two to one, and maintain the lower dose for an additional week before attempting to increase again. Slower dose escalation allows the GI system to adapt.
Eat smaller, more frequent meals rather than large meals. GLP-1 slows gastric emptying, so large meals can cause uncomfortable fullness, nausea, or bloating. Focus on easily digestible foods like lean proteins, cooked vegetables, and simple carbohydrates during the adaptation period.
Avoid high-fat and high-fiber foods if nausea is severe. These slow gastric emptying further, exacerbating discomfort. Once tolerance improves, gradually reintroduce these foods. Ginger tea, peppermint, and small amounts of carbohydrate can help settle the stomach. GI side effect management is detailed in managing GLP-1 nausea guides.
If vomiting occurs, discontinue the supplement temporarily and consult a physician. Persistent vomiting can lead to dehydration and electrolyte imbalances. Do not resume dosing until symptoms resolve completely, and restart at a lower dose.
Constipation
GLP-1 activation slows GI motility, which can cause constipation. Increase fluid intake to at least 3 liters per day. Add fiber gradually through vegetables, fruits, legumes, and whole grains. Sudden large increases in fiber can worsen constipation if fluid intake is insufficient, so increase both together.
Magnesium citrate (200-400 mg daily) promotes bowel movements and is generally well-tolerated. Prunes or prune juice provide both fiber and sorbitol, which has a mild laxative effect. Regular physical activity also supports GI motility. If constipation persists despite these measures, over-the-counter laxatives like polyethylene glycol (Miralax) can be used short-term. Chronic constipation solutions are covered in peptide-induced constipation guides.
Fatigue or low energy
Some individuals experience fatigue when starting GLP-1 agonists. This can result from reduced calorie intake, changes in macronutrient balance, or direct effects of GLP-1 signaling. Ensure you are consuming adequate calories to support basic metabolic needs. Aggressive calorie restriction combined with GLP-1 activation can produce excessive energy deficits and fatigue.
Check electrolyte status, particularly sodium, potassium, and magnesium. Reduced food intake and increased fluid consumption can dilute electrolyte levels. Add salt to meals if you are not hypertensive, and consider an electrolyte supplement if deficiency is suspected.
Monitor sleep quality. GLP-1 agonists should not directly impair sleep, but if you are taking doses late in the day, residual GI discomfort may disrupt rest. Take tablets earlier in the day if evening dosing coincides with fatigue or sleep issues. Energy optimization strategies are explored in maintaining energy during weight loss guides.
Weight loss plateau
Weight loss plateaus are common during any weight loss intervention. After initial rapid loss (often 5-10 pounds in the first month, much of which is water weight), the rate of loss slows as metabolic adaptation occurs. This is normal and does not indicate the product has stopped working.
Reassess calorie intake. As you lose weight, your energy expenditure decreases, so maintaining a deficit requires eating less than before. Use a food diary or tracking app to ensure you are still in a calorie deficit. Increase physical activity if possible to maintain energy expenditure. Add an extra walking session, increase training volume, or incorporate more movement throughout the day. Plateau management strategies are covered in breaking weight loss plateaus guides.
Consider whether the plateau reflects body composition changes rather than fat loss. If you are resistance training, you may be gaining muscle while losing fat, which can mask progress on the scale. Use body composition measurements (waist circumference, body fat percentage, progress photos) to assess changes beyond weight alone.
Long-term considerations and sustainability
Weight loss is only part of the challenge. Maintaining lost weight long-term is harder than losing it initially. Most people who lose significant weight regain it within 2-5 years. GLP-1 agonists can support maintenance, but they are not a permanent solution if underlying behaviors and environments do not change.
Duration of use
How long should you use Evolv GLP-1? Pharmaceutical GLP-1 agonists are often continued long-term, sometimes indefinitely, because discontinuation frequently leads to weight regain. The same principle likely applies to Evolv. If you stop taking it after reaching your goal weight, appetite and food intake may return to baseline, leading to regain.
Some individuals use GLP-1 agonists to lose weight, then transition to a maintenance approach combining lower doses, intermittent use, or reliance on lifestyle modifications alone. This requires careful monitoring and willingness to resume the supplement if regain begins. Long-term use strategies are discussed in long-term GLP-1 use guides.
The financial sustainability of long-term use is a consideration. At $300-450 per month, Evolv represents a significant ongoing expense. Can you sustain this cost for years? If not, you may need to develop an exit strategy that includes dietary discipline, exercise habits, and environmental modifications to maintain weight loss without pharmacological support.
Building sustainable habits
Use the appetite suppression from GLP-1 activation as an opportunity to build lasting habits. The reduced hunger makes it easier to practice portion control, eat more slowly, and choose nutrient-dense foods. These behaviors become ingrained over time, making maintenance easier even if you discontinue the supplement.
Develop non-food coping strategies for stress, boredom, and emotional discomfort. Many people eat for reasons unrelated to hunger. If GLP-1 removes physiological hunger but you continue eating for emotional reasons, weight loss will be limited. Address the psychological and environmental drivers of eating through therapy, mindfulness practices, or behavioral coaching. Habit formation is explored in building sustainable eating habits guides.
Create an environment that supports healthy choices. Stock your kitchen with nutritious foods, remove tempting ultra-processed items, and organize your schedule to prioritize meal preparation and physical activity. Environmental design is one of the most powerful tools for sustaining behavior change, yet it is often overlooked in favor of willpower-based approaches.
Monitoring health markers
If using Evolv GLP-1 long-term, monitor health markers to assess effects and detect potential issues early. Track body weight weekly, but do not obsess over daily fluctuations. Weekly or biweekly averages provide a clearer trend than daily measurements.
Measure waist circumference monthly. This correlates with visceral fat, which has stronger metabolic implications than subcutaneous fat. A reducing waist circumference indicates beneficial fat loss even if scale weight is stable.
Check fasting glucose and HbA1C every 3-6 months if you have prediabetes or are using Evolv for metabolic health. Improvements in these markers suggest beneficial effects on glucose metabolism. Lipid panels (cholesterol, triglycerides) every 6-12 months assess cardiovascular risk. Weight loss typically improves lipid profiles, but monitoring confirms this.
Liver and kidney function tests annually ensure no adverse effects on these organs. While GLP-1 agonists are generally safe, long-term supplement use carries unknown risks, and periodic lab work provides reassurance. Health monitoring protocols are detailed in metabolic health monitoring guides.
Frequently asked questions
Is Evolv GLP-1 a real peptide or just a supplement?
Evolv GLP-1 is a dietary supplement containing a yeast-derived peptide. It is not a pharmaceutical peptide like semaglutide or tirzepatide, which are synthetic molecules approved by regulatory agencies. The distinction matters because supplements are not subject to the same rigorous testing and approval processes as drugs. Understanding research versus pharmaceutical peptides clarifies these differences.
Can I combine Evolv GLP-1 with semaglutide or tirzepatide?
Combining Evolv with pharmaceutical GLP-1 agonists is not recommended. Both activate the same receptors, so stacking them increases the risk of side effects without necessarily improving efficacy. If you are using semaglutide or tirzepatide and achieving good results, there is no reason to add Evolv. If you are not responding to pharmaceutical peptides, Evolv is unlikely to provide additional benefit. Combination strategies are explored in combining GLP-1 peptides guides.
How long does it take to see results with Evolv GLP-1?
If the product is effective, appetite suppression should be noticeable within 3-7 days. Weight loss typically follows within 1-2 weeks. If you see no changes after 3-4 weeks at the recommended dose, it may not be effective for you. Timeline expectations are discussed in GLP-1 results timeline guides.
Do I need to refrigerate Evolv GLP-1 tablets?
Check the product label for storage instructions. Tablets are generally more stable than liquid formulations and may not require refrigeration. However, storing supplements in a cool, dry place away from direct sunlight preserves potency. Proper storage is covered in peptide storage guides.
Will I regain weight if I stop taking Evolv GLP-1?
Most people regain weight after discontinuing GLP-1 agonists if they do not maintain the dietary and lifestyle changes that supported weight loss. Appetite typically returns to baseline, and food intake increases. To prevent regain, transition to a maintenance phase with careful attention to eating habits, physical activity, and environmental factors. Weight maintenance strategies are explored in maintaining weight loss guides.
Is Evolv GLP-1 safe for people with diabetes?
Evolv is marketed as a supplement for healthy individuals, not a diabetes treatment. If you have diabetes, use pharmaceutical GLP-1 agonists that are FDA-approved for glycemic control and have extensive safety data. Do not rely on unproven supplements for managing a chronic disease. Diabetes-specific protocols are detailed in GLP-1 for diabetes guides.
Can I use Evolv GLP-1 if I am breastfeeding or pregnant?
No. GLP-1 agonists are contraindicated during pregnancy and breastfeeding. Weight loss is not appropriate during these periods, and the effects of GLP-1 activation on fetal development or infant nutrition are unknown. Avoid all GLP-1 agonists, including supplements, if you are pregnant, planning to become pregnant, or breastfeeding. Safety considerations are covered in GLP-1 safety guides.
What is the refund policy if Evolv GLP-1 does not work for me?
Check the company website for return and refund policies. Many supplement companies offer money-back guarantees within a certain timeframe (30-60 days). If the product does not produce appetite suppression or weight loss after a reasonable trial period, contact customer service to request a refund. Document your experience to support the refund request if needed.
External resources
Glucagon-like peptide-1 receptor agonists for type 2 diabetes - National Institutes of Health
Medications containing semaglutide - U.S. Food and Drug Administration
How GLP-1 drugs work - National Institutes of Health
For those serious about optimizing metabolic health, SeekPeptides members access comprehensive protocols comparing traditional GLP-1 peptides with emerging alternatives like Evolv, evidence-based dosing guides for both pharmaceutical and biomimetic options, side effect management strategies refined through thousands of user experiences, and real-time updates as new clinical data becomes available. The platform provides the depth of research and practical guidance needed to make informed decisions in the rapidly evolving GLP-1 landscape.
In case I do not see you, good afternoon, good evening, and good night. May your appetite stay balanced, your metabolism stay optimized, and your weight loss stay sustainable.