Mar 3, 2026

Two drugs. Same manufacturer. Completely different approaches to the same problem. Retatrutide and Mounjaro both come from Eli Lilly, both target metabolic hormones, and both produce significant weight loss in clinical trials. But the similarities end there.
One activates two receptors. The other activates three. One sits in pharmacy shelves right now. The other remains locked behind clinical trial gates, with FDA approval still on the horizon. And the weight loss numbers, while impressive for both, tell very different stories when you look beneath the surface.
The gap between these two medications matters more than most comparison articles suggest. It is not simply about which produces bigger numbers on a scale. The retatrutide versus tirzepatide debate involves fundamentally different mechanisms, different risk profiles, different timelines for access, and different metabolic benefits that extend well beyond body weight. Understanding those differences is what separates informed decision-making from guesswork.
This guide breaks down everything researchers and patients need to know about retatrutide and Mounjaro (tirzepatide), from molecular mechanisms and clinical trial data to real-world availability and practical considerations. SeekPeptides has compiled the latest research, phase 3 results, and safety profiles so you can understand exactly what each drug offers and where each one falls short.
What is Mounjaro and how does it work?
Mounjaro is the brand name for tirzepatide, a dual-receptor agonist manufactured by Eli Lilly. It was FDA-approved for type 2 diabetes management, and its weight loss counterpart (marketed as Zepbound) received approval for chronic weight management. The drug is available by prescription from pharmacies right now, covered by many insurance plans, and widely prescribed across the United States.
The mechanism is straightforward but powerful.
Tirzepatide activates two hormone receptors simultaneously: GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide). Both receptors exist in brain regions that regulate appetite, and their combined activation produces stronger appetite suppression than targeting either receptor alone. This dual agonist approach was a breakthrough when it first hit the market, outperforming older single-receptor drugs like semaglutide in head-to-head trials.
How does it actually reduce body weight? Three primary pathways. First, it slows gastric emptying, meaning food stays in your stomach longer and you feel full faster. Second, it directly suppresses appetite through central nervous system signaling. Third, it improves insulin sensitivity and glucose metabolism, which reduces the metabolic dysfunction that drives fat storage in many patients.
The dosing protocol for Mounjaro follows a gradual titration. Patients start at 2.5 mg weekly and increase in 2.5 mg increments every four weeks, eventually reaching maintenance doses of 10 mg or 15 mg depending on tolerance and response. The maximum approved dose is 15 mg once weekly, typically reached by week 20 or 21 of treatment.
In the landmark SURMOUNT-1 trial, participants on the 15 mg dose achieved average weight loss of 22.5% of body weight over 72 weeks. The 10 mg group lost approximately 19.5%. Even the lowest maintenance dose of 5 mg produced meaningful results, averaging around 15% body weight reduction. These numbers reshaped expectations for what pharmaceutical weight management could achieve.
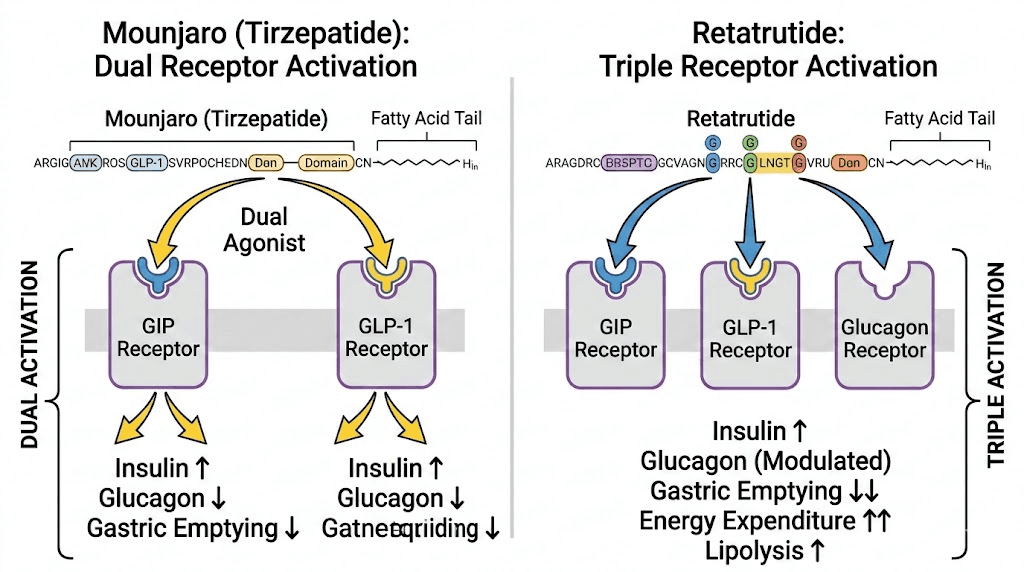
What is retatrutide and how does it differ?
Retatrutide is an investigational triple-receptor agonist, also developed by Eli Lilly. It targets the same two receptors as Mounjaro, GLP-1 and GIP, but adds a third: the glucagon receptor. That third receptor is what makes retatrutide fundamentally different from everything currently available.
Think of it this way. Mounjaro hits two targets. Retatrutide hits all three.
The glucagon receptor activation introduces metabolic effects that dual agonists simply cannot replicate. Glucagon increases energy expenditure by stimulating thermogenesis, meaning the body burns more calories at rest. It also promotes hepatic fat oxidation, which is a fancy way of saying it tells your liver to burn stored fat for fuel. And it influences amino acid metabolism in ways that may help preserve lean muscle mass during weight loss, though this specific benefit requires more long-term data to confirm.
The combined effect of all three receptor pathways creates something researchers call a synergistic metabolic cascade. The GLP-1 component suppresses appetite and improves insulin secretion. The GIP component enhances fat tissue function and further supports appetite control. And the glucagon component ramps up the metabolic engine itself, increasing basal energy expenditure while specifically targeting liver fat stores.
This triple mechanism has produced the most dramatic weight loss numbers ever recorded in clinical trials for an anti-obesity medication.
In the phase 2 trial published in the New England Journal of Medicine, participants on the 12 mg dose lost an average of 24.2% of their body weight in just 48 weeks. That is nearly a quarter of total body weight. And the weight loss curves had not yet plateaued at the study endpoint, suggesting even greater reductions with longer treatment duration.
The phase 3 TRIUMPH-4 trial confirmed and extended these findings. The 12 mg group achieved 23.7% average weight loss (27.2 kg or 60 pounds), while the 9 mg group lost 20.0%. In another phase 3 analysis, retatrutide at the highest dose reached up to 28.7% weight loss, making it the most potent weight loss medication ever tested in large-scale clinical trials.
The core mechanism difference: dual versus triple agonism
Understanding why retatrutide outperforms Mounjaro requires understanding what the glucagon receptor actually does when activated alongside GLP-1 and GIP.
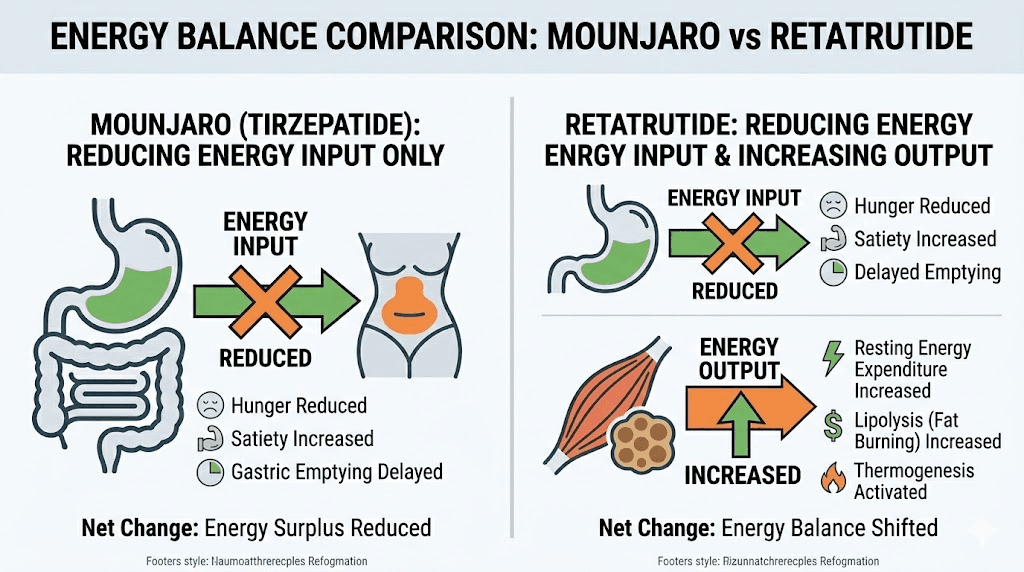
Mounjaro works primarily by reducing caloric intake. It makes people eat less. The dual GLP-1/GIP activation suppresses hunger signals, slows digestion, and creates powerful satiety. This is remarkably effective, as the trial results demonstrate. But the weight loss comes almost entirely from the input side of the energy balance equation: less food in.
Retatrutide works on both sides of the equation simultaneously.
Like Mounjaro, it reduces caloric intake through GLP-1 and GIP receptor activation. But the glucagon component adds something Mounjaro cannot: increased energy output. Glucagon receptor agonism stimulates hepatic glucose production, increases lipolysis (fat breakdown), promotes fatty acid oxidation in the liver, and elevates resting metabolic rate through thermogenesis.
The practical result? Retatrutide users lose weight both because they eat less AND because their bodies burn more energy at rest. It attacks obesity from two directions instead of one.
This dual attack explains why retatrutide consistently outperforms tirzepatide in head-to-head network meta-analyses. A comparative study found that retatrutide produced greater absolute weight reduction (16.34 kg versus 11.82 kg) and greater percentage weight loss (23.77% versus 16.79%) when data from equivalent trial durations were analyzed side by side.
But there is a cost to hitting three receptors instead of two. More receptor activation means more potential for side effects, more metabolic complexity, and more unknowns in the long-term safety profile. The glucagon receptor, in particular, has effects on heart rate that have drawn regulatory scrutiny and contributed to the extended timeline for FDA review.
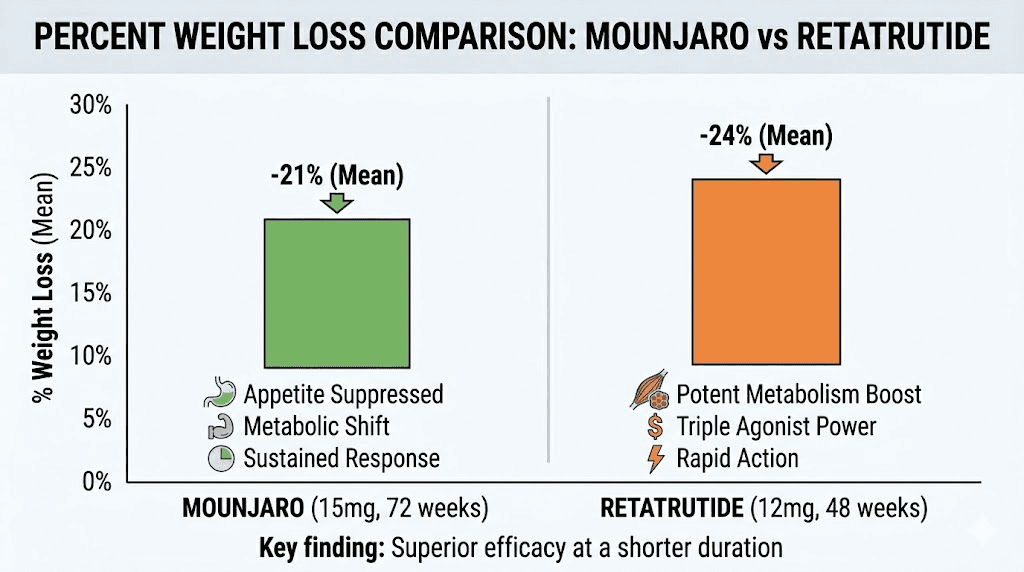
Weight loss results compared: the clinical trial data
Numbers tell the story more clearly than any marketing language could. Let me lay out the clinical trial data for both medications side by side.
Mounjaro (tirzepatide) weight loss data
The SURMOUNT program included multiple phase 3 trials evaluating tirzepatide for weight loss across diverse patient populations.
SURMOUNT-1 results (72 weeks, adults with obesity):
5 mg dose: approximately 15% body weight loss
10 mg dose: 19.5% body weight loss
15 mg dose: 22.5% body weight loss
Placebo: 3.1% body weight loss
At the 15 mg maximum dose, the average participant lost about 52 pounds over 72 weeks. Roughly 63% of participants on the highest dose achieved at least 20% weight loss, and about 40% achieved 25% or more. These were groundbreaking numbers that established tirzepatide as the most effective weight loss medication available at the time of approval.
The weight loss trajectory typically shows initial results within the first month, with patients losing about 4% of body weight. By three months, average reductions reach 4 to 7% depending on dose. The most significant changes accumulate between months four and twelve, with continued but gradually slowing progress through the full 72-week treatment period.
Retatrutide weight loss data
The retatrutide clinical program produced numbers that made the already-impressive Mounjaro data look modest by comparison.
Phase 2 trial results (48 weeks, adults with obesity):
1 mg dose: 7.2% body weight loss
4 mg dose: 12.9% body weight loss
8 mg dose: 17.3% body weight loss
12 mg dose: 24.2% body weight loss
Placebo: 1.6% body weight loss
Phase 3 TRIUMPH-4 results:
9 mg dose: 20.0% body weight loss (22.9 kg / 50.5 lbs)
12 mg dose: 23.7% body weight loss (27.2 kg / 60.0 lbs)
Placebo: 4.6% body weight loss
In a separate phase 3 analysis, retatrutide at the highest dose achieved up to 28.7% weight loss. That translates to an average loss of 71.2 pounds. And critically, the weight loss curves in both phase 2 and phase 3 trials were still trending downward at study completion, suggesting that maximum weight loss had not yet been reached.
Head-to-head comparison
Metric | Mounjaro (15 mg) | Retatrutide (12 mg) |
|---|---|---|
Average weight loss (%) | 22.5% at 72 weeks | 24.2% at 48 weeks |
Average weight loss (lbs) | ~52 lbs at 72 weeks | ~60 lbs at 48 weeks |
Time to results | Slower onset, steady progress | Faster onset, steeper curve |
Participants reaching 20%+ loss | ~63% | Data maturing from phase 3 |
Weight loss plateau reached? | Yes, around months 14-16 | Not yet at study endpoints |
The comparison reveals something important beyond raw percentages. Retatrutide achieved greater weight loss in a shorter timeframe. The 48-week phase 2 results (24.2%) already exceeded what Mounjaro accomplished at 72 weeks (22.5%). When phase 3 data pushed retatrutide to 28.7% at the highest dose, the gap widened further.
For anyone tracking before and after weight loss results, these numbers represent a meaningful clinical difference, not just a statistical one. Several additional percentage points of weight loss translate to significantly greater improvements in metabolic health markers, joint pain, mobility, and quality of life measures.
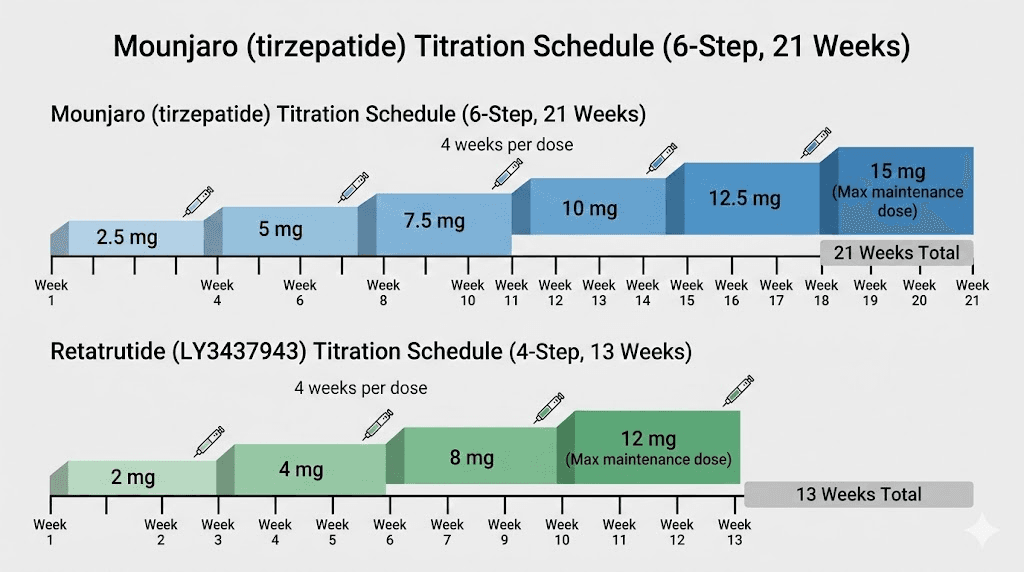
Dosage protocols: how each medication is administered
Both retatrutide and Mounjaro are once-weekly subcutaneous injections. Both use gradual dose titration to minimize gastrointestinal side effects. But the specific protocols differ in meaningful ways.
Mounjaro dosing schedule
The tirzepatide dosing protocol is well-established through FDA-approved labeling:
Week 1-4: 2.5 mg once weekly (starter dose, not a maintenance dose)
Week 5-8: 5 mg once weekly
Week 9-12: 7.5 mg once weekly
Week 13-16: 10 mg once weekly
Week 17-20: 12.5 mg once weekly
Week 21+: 15 mg once weekly (maximum dose)
Dose increases occur in 2.5 mg increments every four weeks. The starting dose of 2.5 mg is not considered therapeutic for weight loss; it exists purely to acclimate the gastrointestinal system. Patients who experience intolerable side effects at any dose can remain at their current level longer before titrating upward.
Some practitioners use alternative protocols. Microdosing tirzepatide has gained attention as an approach where lower doses are used long-term rather than titrating to maximum. The split dosing approach, where the weekly dose is divided into two smaller injections, may reduce peak-related side effects for some patients.
Administration is typically via subcutaneous injection in the abdomen, thigh, or upper arm. Injection sites should be rotated weekly. The medication can be stored in the refrigerator and should be brought to room temperature before injection. For those using compounded tirzepatide, reconstitution with bacteriostatic water follows standard protocols.
Retatrutide dosing schedule
The retatrutide dosing protocol from clinical trials follows a similar but slightly different titration pattern:
Week 1-4: 1-2 mg once weekly (starting dose varies by protocol)
Week 5-8: 4 mg once weekly
Week 9-12: 8 mg once weekly
Week 13+: 12 mg once weekly (maximum dose)
The dose increases are larger and less granular than Mounjaro. Each step roughly doubles the previous dose rather than adding a fixed increment. This means the jump from 4 mg to 8 mg is substantial, and so is the jump from 8 mg to 12 mg.
Clinical trial data showed that gradual titration is absolutely essential for retatrutide. In the phase 2 study, gastrointestinal symptom rates nearly doubled when participants were assigned directly to 8 mg without titrating from a lower starting dose. The lesson is clear: skipping steps or rushing the titration creates unnecessary discomfort and increases dropout risk.
The starting dose of retatrutide and optimal dose selection will likely be refined further when FDA labeling is finalized. Many participants in the trials achieved excellent results at 8 mg without needing to escalate to the 12 mg maximum, which suggests that the highest dose is not necessary or appropriate for everyone.
Dosing comparison table
Factor | Mounjaro | Retatrutide |
|---|---|---|
Starting dose | 2.5 mg weekly | 1-2 mg weekly |
Maximum dose | 15 mg weekly | 12 mg weekly |
Titration steps | 6 steps (2.5 mg increments) | 3-4 steps (variable increments) |
Time to max dose | ~20-21 weeks | ~12-13 weeks |
Administration | Subcutaneous injection | Subcutaneous injection |
Frequency | Once weekly | Once weekly |
Retatrutide reaches its maximum dose faster, in about 12 to 13 weeks versus 20 to 21 weeks for Mounjaro. This faster titration timeline partly explains why retatrutide achieves greater weight loss in shorter study durations. Patients spend more weeks at peak therapeutic doses.
Side effects and safety profiles compared
This is where the conversation gets complicated. And it should, because safety data often matters more than efficacy data for individual treatment decisions.
Mounjaro side effects
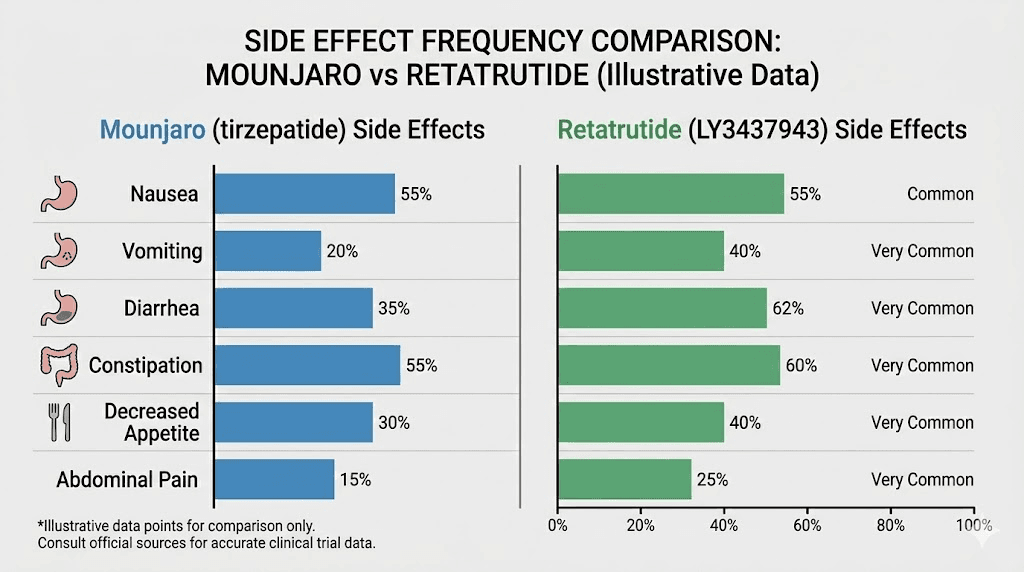
Tirzepatide has a well-documented safety profile thanks to years of post-market surveillance and extensive clinical trial data. The most common side effects of tirzepatide are gastrointestinal:
Nausea: Reported by 12-33% of participants depending on dose (most common early in treatment)
Diarrhea: 12-21% of participants
Constipation: 6-11% of participants
Vomiting: 5-13% of participants
Decreased appetite: Expected therapeutic effect but can be uncomfortable
These side effects are typically most intense during the first few weeks at each new dose level and tend to improve as the body adjusts. Most patients find that symptoms become manageable or resolve entirely by weeks 4 to 8 at a given dose.
Fatigue is another commonly reported experience, particularly during the first weeks of treatment or after dose increases. Some patients also report headaches, gastrointestinal discomfort, anxiety, and sleep disturbances.
Serious adverse events with tirzepatide are rare but include pancreatitis, gallbladder problems, and potential thyroid concerns (based on animal studies). The cardiovascular safety profile appears favorable, with no increased risk of major adverse cardiac events in available data.
Retatrutide side effects
Retatrutide shares the same GI-related side effect profile but at higher frequencies. The glucagon receptor activation adds additional metabolic effects that create unique safety considerations.
From the phase 3 trials at the 9 mg and 12 mg doses:
Nausea: 38.1% (9 mg) to 43.2% (12 mg) versus 10.7% with placebo
Diarrhea: 34.7% (9 mg) to 33.1% (12 mg) versus 13.4% with placebo
Constipation: 21.8% (9 mg) to 25.0% (12 mg) versus 8.7% with placebo
Vomiting: 20.4% (9 mg) to 20.9% (12 mg)
Compare those numbers to Mounjaro and the difference is clear. Retatrutide produces GI side effects at roughly 1.5 to 2 times the rate of tirzepatide. A network meta-analysis confirmed that adverse events were significantly more frequent with retatrutide (relative risk 4.10) compared to tirzepatide (relative risk 2.78) when both were measured against placebo.
But the bigger safety concern is cardiovascular.
Retatrutide was associated with a greater increase in resting heart rate than tirzepatide in clinical trials. This cardiac effect, attributed primarily to the glucagon receptor activation, is one of the main reasons the drug remains in testing. An elevated resting heart rate can increase long-term cardiovascular risk, and regulators need robust data confirming that the metabolic benefits outweigh this potential downside.
Safety comparison summary
Safety Factor | Mounjaro | Retatrutide |
|---|---|---|
GI side effects frequency | Moderate (12-33%) | Higher (33-43%) |
Heart rate impact | Minimal | Greater increase observed |
Long-term safety data | Years of real-world data | Limited to clinical trials |
Serious adverse events | Rare, well-characterized | Rare in trials, monitoring ongoing |
Regulatory confidence | FDA approved | Still in review |
For anyone weighing these options, the safety difference is not trivial. Mounjaro comes with years of post-marketing surveillance data confirming its safety in millions of real-world patients. Retatrutide has trial data that looks promising but lacks the long-term, large-scale, real-world confirmation that only comes after FDA approval and widespread clinical use.
Managing constipation on tirzepatide, body aches, and other common side effects has become well-established through clinical experience. Supportive supplements and dietary adjustments can help manage many of these symptoms effectively.
Availability and FDA approval status
This is perhaps the most practically important section of the entire comparison. It does not matter how impressive a drug is on paper if you cannot access it.
Mounjaro: available now
Mounjaro (tirzepatide) is FDA-approved and currently available by prescription. For weight loss specifically, tirzepatide is marketed under the brand name Zepbound. Both formulations are manufactured by Eli Lilly and can be obtained through:
Primary care physicians
Endocrinologists
Weight management specialists
Telehealth weight loss clinics
Compounding pharmacies (compounded tirzepatide versions)
Insurance coverage varies widely. Many plans cover Mounjaro for type 2 diabetes but may not cover Zepbound for weight management alone. Affordable tirzepatide options include compounded versions from pharmacies like Empower Pharmacy, Southend Pharmacy, and various telehealth platforms.
The bottom line: you can get a prescription, fill it, and start treatment today.
Retatrutide: still in clinical trials
Retatrutide is not yet FDA-approved. It is not available by prescription. You cannot legally obtain it from a pharmacy.
The retatrutide availability timeline depends on ongoing clinical trials and regulatory review:
Current status: Multiple phase 3 trials (TRIUMPH program) ongoing
Phase 3 primary completion: Expected through mid-year
NDA submission: Eli Lilly expected to submit to FDA in late future
FDA review period: Approximately 10 months after submission
Estimated approval: Potentially late next year at earliest
Commercial launch: Could follow shortly after approval
It is important to note that Eli Lilly has not announced an official filing timeline. The estimates above are based on trial completion dates and standard regulatory timelines. Any delays in phase 3 results, safety signals, or manufacturing readiness could push the timeline further back.
Some individuals have sought access to research-grade retatrutide through peptide vendors, but this approach carries significant risks. Research-grade peptides are not manufactured under pharmaceutical standards, have no guaranteed purity or potency, and are not intended for human use. SeekPeptides strongly emphasizes the importance of understanding these distinctions before making any decisions about grey market peptides.
Metabolic benefits beyond weight loss
Weight loss is the headline, but both medications produce metabolic improvements that extend far beyond the number on the scale. And this is where retatrutide starts to separate itself even further from Mounjaro.
Mounjaro metabolic benefits
Tirzepatide consistently demonstrates improvements across multiple metabolic markers:
Blood sugar control: HbA1c reductions of 1.87% to 2.07% in type 2 diabetes trials
Blood pressure: Modest systolic blood pressure reductions of 4-7 mmHg
Lipids: Improvements in triglycerides and cholesterol profiles
Waist circumference: Significant reductions correlating with visceral fat loss
Inflammatory markers: Reductions in high-sensitivity C-reactive protein
These benefits make tirzepatide more than just a weight loss drug. It addresses the constellation of metabolic risk factors collectively known as metabolic syndrome, which is the primary driver of cardiovascular disease, type 2 diabetes, and premature death in patients with obesity. Metabolic improvements from tirzepatide persist as long as treatment continues.
Retatrutide metabolic benefits
Retatrutide produces all of the metabolic benefits seen with Mounjaro plus additional improvements that appear unique to triple agonism:
Blood sugar control: HbA1c reductions of 1.3% to 2.0% at doses of 4-12 mg in type 2 diabetes
Blood pressure: Systolic blood pressure reduction of up to 14.0 mmHg at the highest dose (roughly double what Mounjaro achieves)
Lipids: Non-HDL cholesterol and triglyceride reductions
Prediabetes reversal: 72% of participants with prediabetes at baseline reverted to normoglycemia
Inflammatory markers: Significant reductions in high-sensitivity C-reactive protein
But the standout metabolic benefit, the one that has researchers most excited, is the effect on liver health.
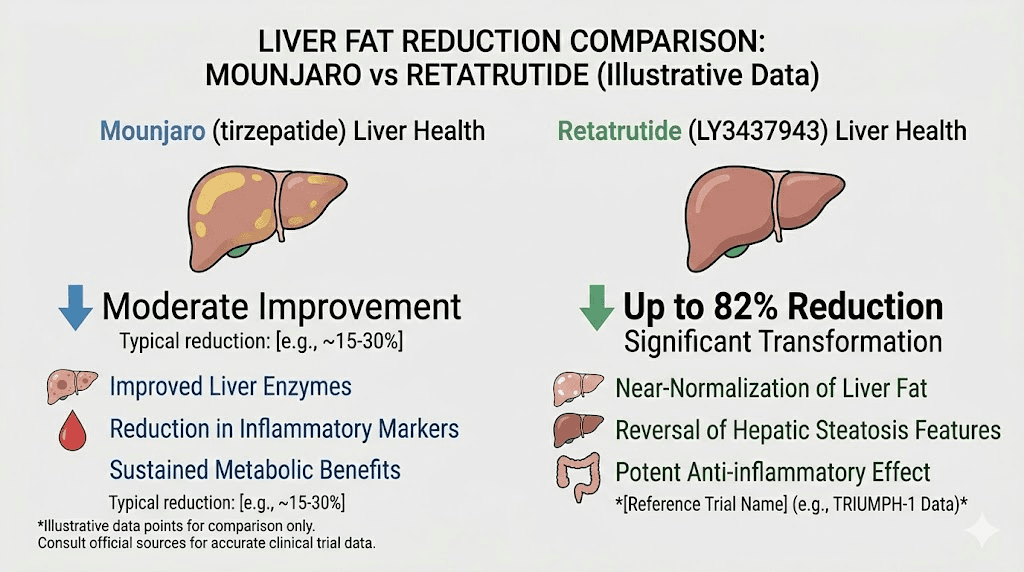
The liver health advantage
Retatrutide produced remarkable reductions in liver fat in clinical trials. The phase 2a liver-specific trial showed:
1 mg dose: 42.9% reduction in liver fat
4 mg dose: 57.0% reduction in liver fat
8 mg dose: 81.4% reduction in liver fat
12 mg dose: 82.4% reduction in liver fat
An 82% reduction in liver fat is extraordinary. Metabolic dysfunction-associated steatotic liver disease (formerly called NAFLD) affects roughly 30% of adults globally and has no approved pharmacotherapy. Retatrutide may become the first effective pharmaceutical treatment for this condition.
The glucagon receptor activation is the key driver of this liver benefit. Glucagon promotes hepatic fat oxidation, essentially commanding the liver to burn its stored fat. This mechanism operates independently of, and in addition to, the weight loss itself. In other words, the liver benefits are not simply a downstream consequence of losing weight. They are a direct pharmacological effect of the glucagon component.
This has implications for people with fatty liver disease who may not have significant obesity. It also suggests that retatrutide could eventually find clinical applications beyond weight management.
For those exploring the broader landscape of metabolic peptides, SeekPeptides members access detailed breakdowns of emerging treatments, mechanism comparisons, and evidence-based analysis of how different GLP-1 approaches to fat loss compare in real-world settings.
Comparing both to semaglutide (Ozempic/Wegovy)
Any conversation about retatrutide versus Mounjaro would be incomplete without placing both in the context of semaglutide, the drug that started the GLP-1 weight loss revolution.
Semaglutide (Ozempic for diabetes, Wegovy for weight loss) is a single GLP-1 receptor agonist. It activates one receptor. Mounjaro activates two. Retatrutide activates three.
The progression in weight loss results follows that receptor count almost perfectly:
Medication | Receptors | Max weight loss | Trial duration |
|---|---|---|---|
Semaglutide (2.4 mg) | GLP-1 only | ~15-17% | 68 weeks |
Mounjaro/Zepbound (15 mg) | GLP-1 + GIP | ~20-22.5% | 72 weeks |
Retatrutide (12 mg) | GLP-1 + GIP + Glucagon | ~24-28.7% | 48-72 weeks |
Each additional receptor target adds roughly 5 to 8 percentage points of weight loss. That pattern has prompted researchers to explore whether adding even more targets, or fine-tuning the balance between existing ones, could push results further still.
For people currently on semaglutide who have plateaued or are not seeing adequate results, switching between GLP-1 medications is an increasingly common clinical practice. Conversion charts between semaglutide and tirzepatide exist to help guide these transitions. When retatrutide becomes available, switching protocols from Mounjaro to retatrutide will follow similar principles, and data on switching from tirzepatide to retatrutide is already being generated in clinical trials.
Patients currently on semaglutide who have hit a weight loss plateau often see renewed progress when transitioning to tirzepatide, likely due to the additional GIP receptor activation. The question many researchers are asking is whether a similar jump in results will occur when transitioning from tirzepatide to retatrutide, thanks to that glucagon component.
Who should consider each option?
Making the right choice between these medications, when retatrutide eventually becomes available, will depend on individual circumstances, risk tolerance, and clinical needs.
Mounjaro may be the better choice if:
You need treatment now (retatrutide is not yet available)
You prefer a medication with extensive real-world safety data
Your BMI qualifies you for current GLP-1 prescriptions
You have type 2 diabetes (Mounjaro has robust diabetes data)
You are sensitive to GI side effects and want a well-established titration protocol
Your weight loss goals are achievable with 15 to 22% body weight reduction
You want insurance coverage options (available for some plans)
Mounjaro is the proven, accessible, right-now option. It works. It is safe. And for the vast majority of people with obesity, a 15 to 22% weight loss represents life-changing improvement in health markers, physical function, and quality of life.
Retatrutide may be worth waiting for if:
You have not responded adequately to existing GLP-1 medications
Your obesity is severe enough to benefit from the additional weight loss potential
You have metabolic dysfunction-associated steatotic liver disease (MASLD/NAFLD)
You are comfortable with a medication that has less real-world safety data
You can wait for FDA approval without significant health deterioration
Your cardiovascular status has been evaluated and heart rate increases are not contraindicated
Retatrutide represents the next frontier. Greater efficacy. More metabolic benefits. But also more unknowns, more potential side effects, and no current legal pathway to access.
The practical timeline: when could you switch?
For patients currently on Mounjaro who are interested in eventually transitioning to retatrutide, here is a realistic timeline based on available information:
Phase 3 trials complete and data is analyzed. Eli Lilly submits an NDA to the FDA. The FDA conducts a standard review process lasting approximately 10 months. If approved, manufacturing ramp-up and distribution follow. Commercial availability could come at any point after approval.
During the waiting period, optimizing your current Mounjaro protocol makes the most sense. Ensure you are at the right dose, following appropriate dietary guidelines, managing food choices, and building sustainable habits that will support long-term success regardless of which medication you use.
When switching from Mounjaro to retatrutide does become possible, the transition will likely follow established principles for transitioning between GLP-1 medications. Clinical trials comparing the two drugs directly are already underway at Eli Lilly, and that data will eventually inform switching protocols.
Cost considerations
Nobody should pick a medication based solely on cost. But pretending cost does not factor into the decision would be equally wrong.
Mounjaro currently carries a significant monthly expense. Brand-name Mounjaro without insurance can cost over $1,000 per month, though manufacturer coupons, savings programs, and insurance coverage can reduce this substantially. Compounded tirzepatide from 503B pharmacies offers a more affordable alternative for many patients.
Retatrutide pricing will not be finalized until FDA approval and commercial launch. However, Eli Lilly will likely price it competitively with existing GLP-1 medications. Whether it will cost more, less, or roughly the same as Mounjaro/Zepbound remains unknown.
The cost-effectiveness calculation also depends on treatment duration. If retatrutide achieves greater weight loss faster, patients may spend fewer total months on the medication to reach their goals, potentially offsetting a higher per-month cost. This is speculative, but it is how health economists will likely evaluate the drug once real-world pricing data becomes available.
Storage and handling considerations
Both medications require proper storage to maintain potency and safety.
Mounjaro is available in pre-filled injection pens (brand name) or as reconstituted vials (compounded). The pre-filled pens should be stored in the refrigerator at 36 to 46 degrees Fahrenheit and can be kept at room temperature (up to 86 degrees Fahrenheit) for up to 30 days if needed. Compounded versions have their own stability profiles, and understanding how long tirzepatide lasts in the fridge is essential for anyone using vials rather than pre-filled pens.
For those navigating storage questions, our guides on tirzepatide refrigeration requirements, what happens if tirzepatide gets warm, and traveling with tirzepatide cover the practical details comprehensively.
Retatrutide storage protocols will be established with commercial labeling. In clinical trials, the medication was stored under standard refrigeration conditions similar to other injectable peptide medications. Our guide on how long retatrutide lasts covers what we know from trial protocols.
Other emerging competitors in the pipeline
Retatrutide and Mounjaro are not the only players in this space. The GLP-1 landscape is expanding rapidly, and understanding the competitive context helps frame the retatrutide versus Mounjaro comparison.
Survodutide (from Boehringer Ingelheim) is another dual agonist targeting GLP-1 and glucagon, but without the GIP component. CagriSema (from Novo Nordisk) combines cagrilintide and semaglutide in a single injection. Orforglipron is an oral GLP-1 agonist that could eliminate the need for injections entirely. Mazdutide and mazdutide compared to retatrutide represents another angle of competition in the global market.
The cagrilintide pathway adds amylin receptor agonism to the mix, creating yet another multi-receptor approach. And GLP-1 patches are being developed that could offer transdermal delivery as an alternative to weekly injections.
Within a few years, patients and clinicians will have a menu of options ranging from single-receptor oral pills to triple-receptor injectable agonists, each with distinct efficacy, safety, and convenience profiles. Retatrutide currently sits at the top of that menu in terms of raw weight loss efficacy. Whether it remains there as the field evolves is an open question.
Injection technique and administration tips
Both medications use subcutaneous injection, but practical technique matters for both comfort and absorption.
The recommended injection sites for GLP-1 medications include the abdomen (at least 2 inches from the navel), the front of the thigh, and the back of the upper arm. Rotating injection sites weekly helps prevent lipohypertrophy (fatty lumps at the injection site) and maintains consistent absorption.
For Mounjaro specifically, injecting in the abdomen and understanding proper injection technique can reduce discomfort and injection site reactions. Some patients experience redness and itching at the injection site, which typically resolves within a few hours to days.
For retatrutide, injection protocols and recommended injection sites follow similar principles to other subcutaneous peptide medications. The general guidance for GLP-1 injections applies to both medications.
Diet and lifestyle considerations during treatment
Medication is one piece of the puzzle. Diet and lifestyle modifications during treatment can dramatically influence outcomes with either Mounjaro or retatrutide.
Both medications reduce appetite significantly, which creates both an opportunity and a challenge. The opportunity: eating less becomes much easier. The challenge: ensuring that the food you do eat is nutritionally dense enough to prevent deficiencies and muscle loss.
Diet plans for tirzepatide users emphasize high-protein meals to preserve lean mass, adequate hydration (both medications can increase fluid loss), and avoiding certain foods that worsen GI side effects. Optimal food choices on tirzepatide include lean proteins, vegetables, and whole grains while minimizing high-fat and high-sugar foods that can exacerbate nausea.
Protein shakes designed for GLP-1 users have become increasingly popular as a practical way to meet protein targets when appetite is suppressed. Many patients find that liquid nutrition is easier to tolerate than solid food, especially during the initial weeks of treatment.
For those on semaglutide, similar dietary principles apply. Our guides on semaglutide diet plans, foods to avoid on semaglutide, and best foods to eat on semaglutide contain transferable principles that apply across the entire GLP-1 medication class.
Common questions about both medications
Patients considering either medication tend to ask similar practical questions. Addressing those here.
Can you drink alcohol on either medication? Alcohol and tirzepatide is a common concern. Both medications affect gastric motility and liver metabolism, which can alter alcohol tolerance. Most clinicians advise caution and reduced alcohol consumption during treatment. Some alcoholic beverages are better tolerated than others, but moderation is the consistent recommendation.
What about effects on menstrual cycles? Tirzepatide can affect menstrual cycles, primarily through rapid weight loss altering hormone levels. Similar effects are expected with retatrutide. Patients should discuss reproductive planning with their healthcare provider before starting either medication. There are specific concerns about tirzepatide during breastfeeding and pregnancy that warrant careful medical supervision.
What happens when you stop treatment? Weight regain after discontinuing GLP-1 medications is a documented concern for both drugs. Studies show that patients tend to regain weight when treatment stops, which is why maintaining weight loss after tirzepatide requires sustained lifestyle changes. Some patients explore gradual weaning protocols rather than abrupt discontinuation to minimize rebound.
Frequently asked questions
Is retatrutide the same as Mounjaro?
No. While both are manufactured by Eli Lilly and both activate the GLP-1 and GIP receptors, retatrutide is a triple agonist that also activates the glucagon receptor. Mounjaro (tirzepatide) is a dual agonist. They are different molecules with different mechanisms, different efficacy profiles, and different regulatory statuses.
Can I get retatrutide prescribed today?
No. Retatrutide has not received FDA approval and is currently only available through clinical trials. The earliest possible commercial availability is likely late next year, though timelines may shift. Check our guide on retatrutide availability for the latest status updates.
Is retatrutide more effective than Mounjaro for weight loss?
Clinical trial data suggests yes. Retatrutide achieved approximately 24 to 28.7% weight loss at its highest dose over 48 to 72 weeks, compared to Mounjaro achieving approximately 20 to 22.5% at its highest dose over 72 weeks. The additional glucagon receptor activation contributes to faster and more extensive weight reduction. However, individual results vary, and the best medication for any person depends on their unique medical profile.
Will retatrutide replace Mounjaro?
Not likely. These medications will probably serve different segments of the patient population. Mounjaro remains an excellent choice with proven safety and availability. Retatrutide may become the preferred option for patients who need more aggressive weight management or who have specific conditions like fatty liver disease that benefit from glucagon receptor activation.
Does retatrutide have worse side effects than Mounjaro?
The frequency of gastrointestinal side effects is higher with retatrutide (nausea affects about 43% vs 12-33% for Mounjaro). Retatrutide also produces a greater increase in resting heart rate. However, the types of side effects are similar, and most are manageable with proper dose titration.
What about combining both medications?
There are no clinical trials combining retatrutide and tirzepatide, and this would not be medically advisable. Both drugs activate the same GLP-1 and GIP receptors, so combining them would create overlapping and potentially dangerous receptor overstimulation. However, some research has examined transitioning from high-dose tirzepatide to retatrutide as a sequential approach.
How do the reconstitution and preparation processes compare?
Brand-name Mounjaro comes in pre-filled injection pens requiring no preparation. Compounded tirzepatide requires reconstitution with bacteriostatic water. Retatrutide commercial formulation details have not been finalized, but it will likely be available in similar delivery formats upon approval. For research purposes, retatrutide reconstitution follows standard peptide preparation protocols.
External resources
New England Journal of Medicine: Retatrutide Phase 2 Trial for Obesity
PMC: Comparative Efficacy of Tirzepatide vs Retatrutide Network Meta-Analysis
Nature Medicine: Retatrutide for Metabolic Dysfunction-Associated Steatotic Liver Disease
U.S. Food and Drug Administration: Drug Approvals and Databases
For researchers serious about understanding the full landscape of GLP-1 based weight management, SeekPeptides provides the most comprehensive resource available. Members access detailed protocol comparisons, evidence-based dosing guides, and a community of thousands navigating these exact decisions.
In case I do not see you, good afternoon, good evening, and good night. May your receptors stay responsive, your titrations stay smooth, and your metabolic health stay on the right trajectory.