Mar 3, 2026

Some researchers split their retatrutide dose into two or three smaller injections per week and report fewer side effects. Others follow the standard once-weekly protocol and lose 24% of their body weight in under a year. The difference between these two approaches is not just about convenience or preference. It is about pharmacokinetics, receptor activation thresholds, and whether the theoretical benefits of smoother drug levels actually translate into real-world results.
Retatrutide microdosing has become one of the most debated topics in the peptide community. The logic sounds reasonable on the surface. Take the same total weekly dose, split it into smaller amounts, inject more frequently, and reduce those brutal nausea spikes that hit a day or two after your full injection. The math works. The pharmacokinetic models support it. But the clinical data tells a more complicated story, one that involves retatrutide dosing fundamentals that most guides completely ignore.
This guide covers everything researchers need to know about microdosing retatrutide. From the science behind split dosing schedules to the glucagon receptor activation threshold that may render low doses less effective, from practical protocols and dosage charts to reconstitution math for multiple weekly injections. Whether microdosing makes sense for you depends on factors most online discussions never mention. And the answer is not as simple as either side wants it to be.
SeekPeptides has compiled the most thorough analysis of retatrutide microdosing available, drawing from Phase 2 and Phase 3 clinical trial data, pharmacokinetic modeling, community reports, and the underlying biology of triple receptor agonism. What follows is not opinion. It is evidence.
What retatrutide microdosing actually means
Microdosing retatrutide means splitting a standard weekly dose into two, three, or more smaller injections administered throughout the week. The total weekly amount stays the same. Only the frequency and individual injection volume change.
This is different from dose reduction.
A researcher on a 4mg weekly protocol who switches to microdosing might inject approximately 1.3mg three times per week, Monday, Wednesday, and Friday. The total still adds up to roughly 4mg. Someone reducing their dose would simply inject less total medication per week. These are fundamentally different strategies with different goals, and confusing them is where most problems start.
The standard retatrutide dose schedule follows a once-weekly injection pattern, consistent with how the molecule was studied in clinical trials. The Phase 2 trial published in the New England Journal of Medicine tested doses of 1mg, 4mg, 8mg, and 12mg, all administered as single weekly injections. Every piece of efficacy and safety data we have comes from this dosing frequency.
Microdosing emerged from the community, not from clinical research. Researchers who experienced significant gastrointestinal side effects, particularly nausea and vomiting in the first 24 to 48 hours after injection, began experimenting with split doses to smooth out the concentration curve. The reasoning borrowed heavily from similar approaches with tirzepatide microdosing and semaglutide split dosing that had gained traction in those communities.
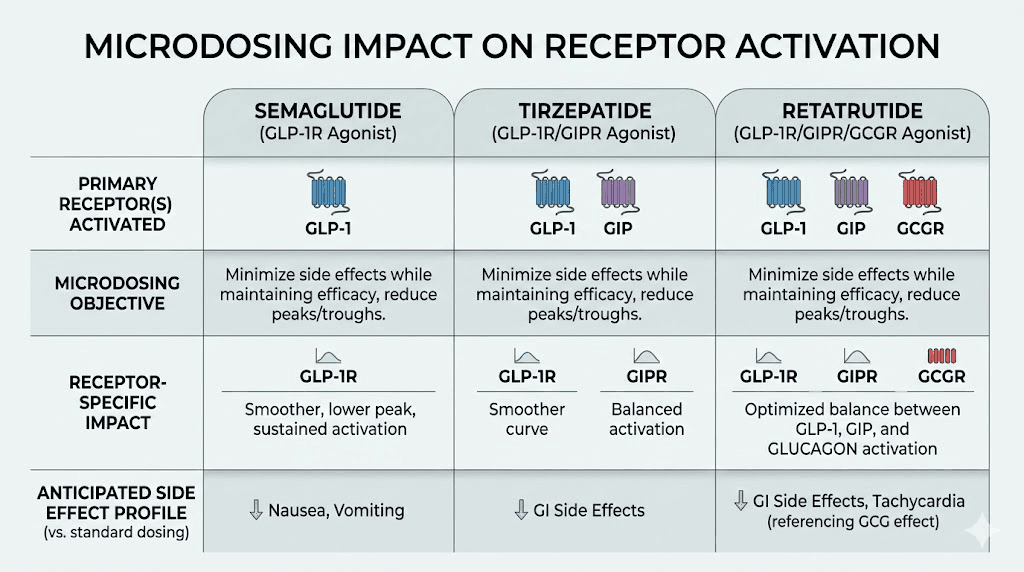
The triple agonist mechanism and why dose matters more than frequency
Retatrutide is not just another GLP-1 medication. It activates three separate receptor systems simultaneously, and this distinction matters enormously when discussing microdosing.
The three targets are the GLP-1 receptor, the GIP receptor, and the glucagon receptor. Each plays a distinct role in weight loss and metabolic health. Understanding what each receptor does explains why simply reducing peak concentrations through split dosing may not be a straightforward improvement.
GLP-1 receptor activation
The GLP-1 component works similarly to semaglutide and tirzepatide. It suppresses appetite through central nervous system signaling, slows gastric emptying to increase satiety, and improves insulin sensitivity. GLP-1 activation is primarily responsible for the appetite suppression that makes eating less feel effortless rather than torturous. This receptor responds well to sustained levels, meaning split dosing should theoretically maintain adequate appetite suppression.
GIP receptor activation
The GIP component enhances the metabolic effects of GLP-1 activation and contributes to improved glucose handling. GIP receptor agonism appears to amplify weight loss beyond what GLP-1 alone achieves, which is part of why dual agonists like tirzepatide outperform semaglutide in head-to-head comparisons. Like GLP-1, the GIP receptor likely responds to sustained exposure rather than requiring specific peak concentrations.
Glucagon receptor activation
Here is where things get complicated for microdosing advocates.
The glucagon receptor drives a fundamentally different mechanism. When activated, glucagon receptors increase energy expenditure through thermogenesis, promote lipolysis, which is the breakdown of stored fat, and enhance fatty acid oxidation in the liver. In clinical studies, researchers observed that levels of beta-hydroxybutyrate, a direct biomarker of fatty acid oxidation, increased two to threefold in a dose-related pattern at retatrutide doses of 4mg and higher.
That phrase, "dose-related pattern," matters. It suggests that glucagon receptor activation may require reaching a specific concentration threshold to produce meaningful metabolic effects. Drop below that threshold by spreading the dose too thin, and you might maintain appetite suppression through the GLP-1 and GIP pathways while losing the unique fat-burning advantage that makes retatrutide different from semaglutide or tirzepatide.
This is the core tension of retatrutide microdosing. You may solve the side effect problem while undermining the mechanism that justifies choosing retatrutide over simpler alternatives.
Pharmacokinetics behind split dosing
The pharmacokinetic argument for microdosing is straightforward and mathematically sound. It just may not matter as much as advocates believe.
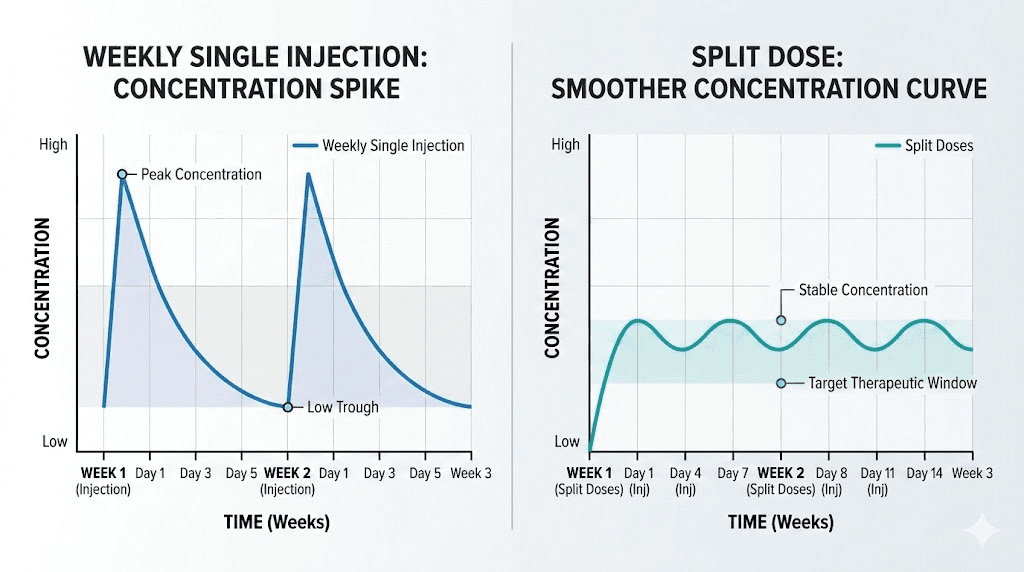
Retatrutide has an elimination half-life of approximately 6 days. Time to maximum concentration after injection ranges from 12 to 72 hours. The molecule demonstrates 99% albumin binding, creating a natural depot effect that already provides relatively stable blood levels compared to shorter-acting peptides.
With standard weekly dosing, the peak-to-trough ratio sits around 4.0. This means peak concentrations after injection are roughly four times higher than the lowest levels just before the next dose. That significant swing is what drives post-injection side effects for many researchers. The nausea, the fatigue, the gastrointestinal distress, these typically correlate with the concentration peak, not the average level.
How split dosing changes the concentration curve
Different microdosing frequencies produce measurably different pharmacokinetic profiles. The data from pharmacokinetic modeling shows clear patterns across common schedules.
Twice weekly dosing reduces peak concentrations by approximately 28% and brings the peak-to-trough ratio down to 2.1. For a researcher on 8mg weekly, this means injecting 4mg every 3.5 days. Theoretical efficacy retention sits around 95% based on area-under-the-curve calculations. This is the most popular split dosing schedule and likely the best compromise between side effect reduction and efficacy preservation.
Three times weekly dosing on a Monday, Wednesday, Friday schedule reduces peaks by 35% with a peak-to-trough ratio of 1.6. At 8mg weekly, each injection would be approximately 2.7mg. Theoretical efficacy drops slightly to around 90%, though this has never been validated in clinical trials.
Daily microdosing reduces peaks by 48% with a near-flat concentration profile and a peak-to-trough ratio of just 1.2. At 8mg weekly, daily injections would be approximately 1.14mg each. The theoretical efficacy concern grows here, dropping to roughly 75% of standard dosing based on modeling. Seven injections per week also dramatically increases practical burden and error risk.
Every-other-day dosing falls between twice weekly and three times weekly, with a 30% peak reduction and a peak-to-trough ratio of 1.8.
These numbers come from pharmacokinetic models, not clinical measurements. The actual concentration curves in living humans may differ. But the directional relationship is sound. More frequent dosing equals smoother levels equals lower peaks and higher troughs.
For detailed conversion calculations, the retatrutide dosage calculator can help determine exact split dosing amounts based on your total weekly target and chosen frequency.
Common microdosing schedules and protocols
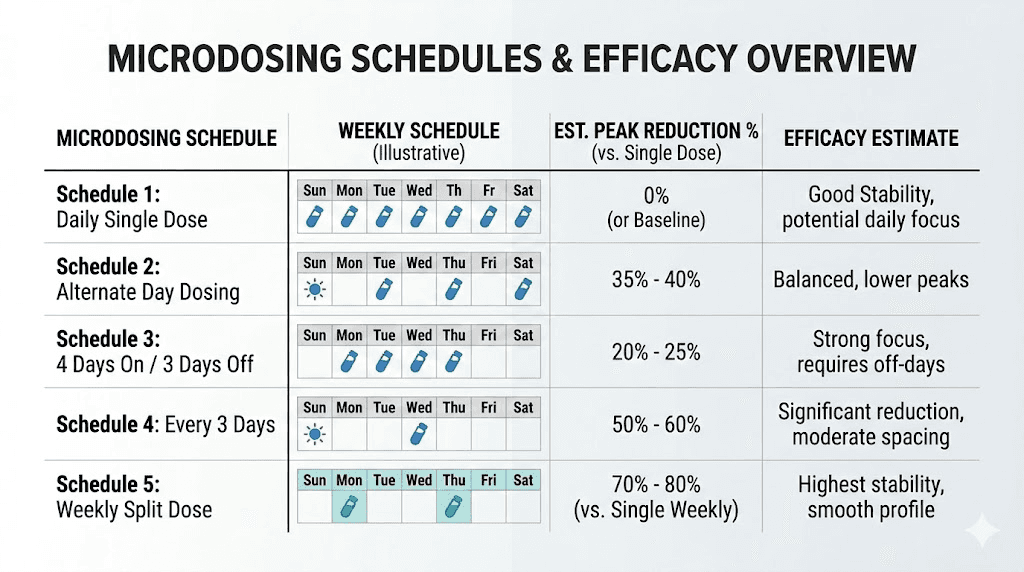
Researchers experimenting with retatrutide microdosing typically follow one of several established patterns. Each has distinct advantages and drawbacks that extend beyond simple pharmacokinetics.
Protocol 1: twice weekly split (most common)
This is the most widely adopted microdosing approach. Inject half the weekly dose every 3 to 4 days. Most researchers choose Sunday and Wednesday or Monday and Thursday to maintain consistent spacing.
At a target of 4mg weekly during the titration schedule, this means 2mg per injection. At the 8mg maintenance level, it becomes 4mg per injection. At 12mg, it becomes 6mg per injection.
The advantages are meaningful. A 28% peak reduction is clinically significant for side effect management. Two injections per week is manageable without excessive burden. The 95% theoretical efficacy retention means you are unlikely to sacrifice meaningful results. And the injection spacing is flexible enough to accommodate travel or schedule changes.
The drawbacks are real but limited. Twice the injection frequency means twice the injection site management. You need to rotate between more locations to avoid tissue irritation. Reconstitution math becomes slightly more complex, and you need to ensure your reconstitution calculations produce concentrations that allow accurate measurement of smaller individual doses.
Protocol 2: three times weekly (Monday, Wednesday, Friday)
The Mon-Wed-Fri schedule provides smoother levels but introduces more complexity. At 4mg weekly, each injection is approximately 1.33mg. At 8mg weekly, approximately 2.67mg. At 12mg weekly, 4mg per injection.
This protocol may be worth considering for researchers who experience significant side effects even with twice-weekly dosing. The 35% peak reduction is substantial. But three injections per week starts to feel burdensome for many people, and the incremental benefit over twice-weekly dosing may not justify the additional injections.
Protocol 3: every other day
Every-other-day dosing creates an alternating schedule that some researchers find more natural than fixed days. At 4mg weekly, each injection is approximately 1.14mg, given that you inject 3.5 times per week on average.
The uneven weekly rhythm can cause confusion. Some weeks have three injections, others have four. This makes tracking and consistency more challenging. Most researchers who try every-other-day dosing eventually switch to a fixed twice or three-times-weekly schedule for simplicity.
Protocol 4: low-dose maintenance
After reaching goal weight, some researchers transition to a low-dose maintenance protocol. A common approach is 1mg twice weekly, totaling 2mg per week, which is significantly below the typical therapeutic range but may preserve appetite management and prevent rebound weight gain.
This approach preserves approximately 75% of the appetite suppression effect through sustained GLP-1 and GIP activation. Whether it maintains adequate glucagon receptor activation for metabolic benefits remains unknown. For researchers considering this approach, understanding how much retatrutide to take during maintenance is critical.
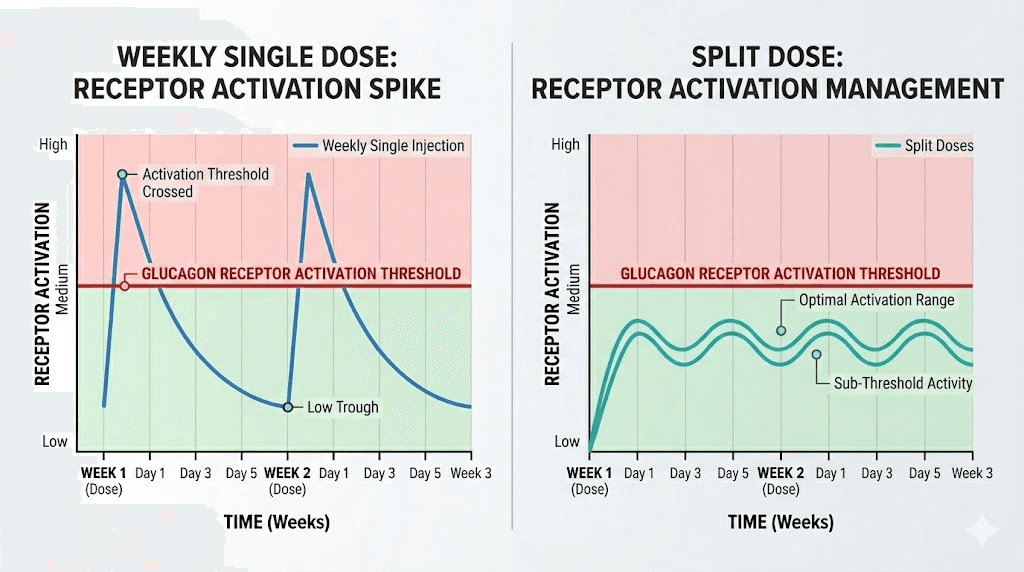
The glucagon receptor threshold problem
This is the most important section of this entire guide. Read it carefully.
Retatrutide works differently from semaglutide and tirzepatide because of its glucagon receptor component. That component is what produces the additional 5 to 10 percentage points of weight loss that separates retatrutide from dual agonists. But glucagon receptor activation appears to follow different rules than GLP-1 or GIP activation.
Glucagon acts on adipose tissue to decrease lipogenesis and induce lipolysis. It increases fatty acid oxidation in the liver. It promotes thermogenesis through effects on brown fat. These are energy-expensive processes that require significant receptor engagement to initiate.
The Phase 2 clinical data supports a threshold model. Beta-hydroxybutyrate levels, the most direct biomarker of glucagon-driven fat oxidation, increased meaningfully only at doses of 4mg and above. At 1mg, the weight loss was 8.7% at 48 weeks. Respectable, but not dramatically different from what semaglutide achieves alone. The jump from 1mg to 4mg produced nearly double the weight loss, from 8.7% to 17.1%, suggesting that the glucagon component only becomes meaningfully active somewhere in that dose range.
Now consider what happens with microdosing. A researcher on a 4mg weekly protocol who splits into three injections is taking approximately 1.33mg per injection. That individual injection concentration may fall below the threshold needed for significant glucagon receptor activation. The GLP-1 and GIP components still work, maintaining appetite suppression. But the unique metabolic boost from glucagon, the reason you chose retatrutide over semaglutide or tirzepatide, may be partially or fully lost.
This is not proven. No clinical trial has directly compared split dosing to weekly dosing at the same total weekly amount. But the dose-response data strongly suggests that peak concentration, not just total exposure, matters for glucagon receptor activation.
Think about it differently. If you need 4mg to cross the glucagon activation threshold, and you split that into three 1.33mg doses, none of your individual injections reach the level needed. You get three sub-threshold glucagon exposures instead of one above-threshold exposure. The total amount is the same, but the biological effect may be dramatically different.
What the clinical trials actually show about dose response
Understanding the trial data is essential for making an informed decision about microdosing. The numbers tell a story that neither microdosing advocates nor opponents fully acknowledge.
Phase 2 results (48 weeks, published in NEJM)
The landmark Phase 2 trial randomized 338 participants across multiple dose groups. Weight loss at 48 weeks was clearly dose-dependent.
The placebo group lost 2.1%. The 1mg group lost 8.7%. The combined 4mg groups lost 17.1%. The combined 8mg groups lost 22.8%. And the 12mg group lost 24.2%, translating to approximately 58 pounds of absolute weight loss over 11 months.
The jump from placebo to 1mg represents GLP-1 and partial GIP activation. The jump from 1mg to 4mg, nearly doubling the weight loss percentage, likely represents the addition of meaningful glucagon receptor engagement. The more modest increases from 4mg to 8mg to 12mg suggest diminishing returns once the glucagon threshold is crossed.
Starting dose also mattered for tolerability. Groups that began at 2mg before escalating experienced fewer gastrointestinal side effects than groups that started at 4mg. This is the standard retatrutide titration approach and is relevant because proper titration may solve the same problems that microdosing attempts to address.
Phase 3 results (TRIUMPH-4, 68 weeks)
The Phase 3 TRIUMPH-4 trial evaluated 9mg and 12mg doses over 68 weeks in adults with obesity and knee osteoarthritis. Results exceeded even the impressive Phase 2 findings.
Participants taking 12mg lost an average of 28.7% body weight, approximately 71 pounds. Those on 9mg lost 26.4%. Among the 12mg group, 58.6% achieved 25% or greater weight loss, 39.4% achieved 30% or greater, and 23.7% achieved 35% or greater.
Beyond weight loss, retatrutide reduced non-HDL cholesterol, triglycerides, high-sensitivity C-reactive protein, and lowered systolic blood pressure by 14.0 mmHg at the highest dose. These cardiovascular benefits likely require sustained, adequate receptor activation across all three targets, raising further questions about whether microdosing preserves them.
For researchers comparing these results with other medications, our guides on retatrutide vs semaglutide and the retatrutide vs tirzepatide dosage comparison provide detailed head-to-head analyses.
Side effect profile by dose
Gastrointestinal side effects were the most common adverse events and were clearly dose-related. Nausea, diarrhea, vomiting, and constipation occurred more frequently at higher doses and were mostly mild to moderate in severity.
At 9mg, the discontinuation rate from adverse events was 12.2%. At 12mg, it was 18.2%. The placebo discontinuation rate was 4.0%. These numbers mean that approximately 82 to 88% of participants completed the trial without dropping out due to side effects, even at the highest doses.
A notable finding was dysesthesia, an abnormal sense of touch. This occurred in 8.8% of 9mg patients and 20.9% of 12mg patients compared to just 0.7% on placebo. This side effect is relatively unique to retatrutide and may relate to the glucagon receptor component.
Importantly, gradual dose escalation significantly reduced GI side effect rates. Starting at 2mg versus 4mg nearly halved the incidence of early nausea and vomiting. This suggests that proper titration scheduling may be more effective than microdosing for managing side effects.
When microdosing might actually make sense
Despite the concerns about glucagon receptor activation, there are specific scenarios where microdosing retatrutide could be a reasonable strategy. The key is being honest about the tradeoffs.
Severe gastrointestinal intolerance
Some researchers experience debilitating nausea and vomiting that does not improve with standard titration. If proper titration starting at the lowest dose with gradual 4-week escalation still produces intolerable GI effects, split dosing may allow continued use of the medication rather than abandoning it entirely.
The calculus is straightforward. Microdosing with potentially reduced glucagon activation is better than not taking retatrutide at all. A researcher who cannot tolerate weekly injections gains nothing from superior pharmacokinetics if they cannot adhere to the protocol.
Before concluding that microdosing is necessary, however, explore all tolerance strategies first. Proper dietary modifications, timing injections before bed, using antiemetics during the adjustment period, and ensuring adequate hydration can all reduce GI side effects without altering the dosing schedule.
Transitioning from tirzepatide
Researchers switching from tirzepatide to retatrutide sometimes experience increased side effects because of the added glucagon receptor component. A temporary split dosing approach during the transition period, returning to weekly dosing once adapted, can ease this adjustment.
The retatrutide dosage schedule after high-dose tirzepatide guide covers this transition in detail. Using split doses for the first 4 to 8 weeks while adapting to the glucagon component, then consolidating to weekly injections, is a pragmatic approach that some researchers report success with.
Cost-driven dose stretching
Some researchers use microdosing as a way to extend their supply. Taking a lower total weekly dose, say 2mg instead of the target 4mg, while splitting it into smaller frequent injections. This is dose reduction disguised as microdosing, and it carries more significant efficacy tradeoffs.
Understanding retatrutide cost considerations is important, but reducing the dose below therapeutic levels to save money may result in spending the same amount over a longer period with inferior results. The math often does not favor this approach.
When standard weekly dosing is the better choice
For most researchers, standard weekly dosing remains the superior option. Here is why.
Every piece of clinical evidence supports it. The 24.2% weight loss at 48 weeks and 28.7% at 68 weeks came from weekly injections. These are the largest weight loss numbers ever reported for any obesity medication in clinical trials. There is no evidence that split dosing produces equivalent results.
The side effects are manageable for most people. Approximately 82 to 88% of trial participants completed the study without discontinuing due to side effects. Proper titration, starting low and escalating slowly, dramatically reduces early GI issues. If you have not tried standard titration with a 2mg starting dose and 4-week escalation intervals, you have not given weekly dosing a fair chance.
Glucagon receptor activation likely requires threshold concentrations. The unique advantage of retatrutide, its ability to increase energy expenditure and fat oxidation beyond what GLP-1/GIP alone achieve, may depend on reaching peak concentrations that only weekly dosing reliably provides.
Fewer injections means fewer opportunities for error. Each injection is a moment where dosing mistakes, contamination, and tissue damage can occur. One injection per week minimizes these risks. For researchers learning proper retatrutide injection technique, simplicity matters.
Adherence is better with simpler protocols. Research consistently shows that medication adherence decreases as dosing frequency increases. One injection per week is easier to maintain over months and years than two, three, or seven injections per week.
How to properly titrate retatrutide to reduce side effects
Before considering microdosing, optimize your titration. Many researchers who think they need split dosing actually need better titration.
The standard titration approach
Based on clinical trial protocols, the evidence-based titration follows a consistent pattern. Start at the lowest effective dose and escalate every 4 weeks only if the current dose is well tolerated.
Weeks 1 through 4 begin at 1mg once weekly. This is the adaptation phase where the body adjusts to GLP-1 and GIP receptor activation. Side effects at this level are typically minimal. Some researchers start even lower at 0.5mg for the first week or two.
Weeks 5 through 8 increase to 2mg once weekly. The first meaningful escalation introduces more significant receptor engagement. Mild nausea may appear in the first 1 to 2 days after the new dose.
Weeks 9 through 12 move to 4mg once weekly if tolerated. This is where most researchers begin to experience the glucagon receptor component. The jump from 2mg to 4mg is where many people report the most noticeable GI effects.
Weeks 13 through 16 escalate to 8mg once weekly. A significant dose increase that may require an extra week or two at 4mg if side effects are pronounced. Using the retatrutide dosage calculator helps ensure precise measurements at these higher concentrations.
Weeks 17 and beyond reach the 12mg maintenance dose if tolerated. Not all researchers need to reach 12mg. If weight loss is progressing well at 8mg with manageable side effects, there may be no need to escalate further.
Side effect management during titration
Several strategies can reduce GI side effects without altering the dosing schedule.
Timing matters. Many researchers find that injecting in the evening, before bed, allows them to sleep through the initial peak. By morning, the worst of the nausea has often passed.
Hydration is critical. Dehydration amplifies nausea and can trigger headaches that compound the discomfort. Aiming for at least 64 ounces of water daily, more during the first few days after injection, helps significantly. Researchers experiencing GLP-1 headaches should especially prioritize hydration.
Dietary modifications reduce GI burden. Smaller, more frequent meals are easier to tolerate than large ones. Avoiding high-fat and greasy foods during the first 48 hours post-injection minimizes nausea triggers. Our guide on foods to eat while on GLP-1 medications provides detailed dietary guidance.
Ginger, whether as tea, capsules, or chews, has evidence supporting its antiemetic properties. Many researchers keep ginger products available for the first few days after dose escalation.
If standard approaches do not work, extending the escalation timeline is preferable to microdosing. Spending 6 to 8 weeks at each dose instead of 4 gives the body more time to adapt while maintaining the pharmacokinetic profile that the clinical trials validated.
Microdosing retatrutide vs microdosing tirzepatide vs semaglutide
The microdosing discussion differs significantly between these three medications because of their different receptor profiles.
Semaglutide microdosing
Semaglutide is a pure GLP-1 receptor agonist with a half-life of approximately 7 days. Because it targets only one receptor, microdosing is relatively straightforward. The dose-response curve for GLP-1 activation is more linear, meaning split dosing is less likely to sacrifice significant efficacy. Researchers who microdose semaglutide generally maintain good appetite suppression because sustained, moderate GLP-1 levels are sufficient for that effect.
For researchers exploring semaglutide specifically, our guides on semaglutide dosage in units and semaglutide reconstitution cover the practical details.
Tirzepatide microdosing
Tirzepatide targets both GLP-1 and GIP receptors. Microdosing tirzepatide is more complex than semaglutide because of the dual receptor engagement, but both GLP-1 and GIP respond well to sustained lower levels. The microdosing tirzepatide chart shows that split dosing preserves much of the efficacy because neither receptor requires high peak concentrations for activation.
The tirzepatide microdose chart provides specific protocols that have become popular in the community. Many researchers who successfully microdose tirzepatide assume the same approach will work for retatrutide. That assumption may be flawed because of the glucagon receptor difference.
Retatrutide microdosing: the unique challenge
Retatrutide is the most complex to microdose because of the glucagon receptor component. While GLP-1 and GIP respond to sustained moderate levels, glucagon receptor activation appears more threshold-dependent. This means retatrutide microdosing carries risks that do not apply to the other two medications.
A direct comparison illustrates the point. If you microdose semaglutide and lose some efficacy, you might go from 15% weight loss to 13% weight loss. The GLP-1 mechanism still works. If you microdose retatrutide and lose the glucagon component, you might go from 24% weight loss to 15% weight loss, essentially converting your triple agonist into a less efficient version of what you could get from semaglutide alone.
This does not mean retatrutide microdosing never works. It means the stakes are higher and the decision requires more careful consideration. The retatrutide vs tirzepatide comparison helps researchers evaluate whether the added complexity of retatrutide is worth it given their individual tolerance profile.
Side effects at different dose levels
Understanding which side effects occur at which doses helps researchers make informed decisions about microdosing versus tolerance strategies.
Low dose (1-2mg weekly)
At the lowest effective doses, side effects are typically mild. Occasional nausea affects some researchers, particularly in the first 1 to 2 weeks. Mild appetite suppression is noticeable but not overwhelming. Energy levels may shift slightly. Most researchers tolerate these doses with minimal complaint, and the side effect profile closely resembles low-dose semaglutide.
Medium dose (4mg weekly)
This is where the glucagon component becomes active and side effects become more noticeable. Nausea is more common, particularly in the first 24 to 48 hours after injection. Diarrhea and constipation may alternate, a confusing pattern that reflects the complex receptor interactions. Decreased appetite becomes more pronounced. Some researchers report fatigue during the adjustment period, similar to GLP-1-related fatigue seen with other medications.
The 4mg dose is often where researchers consider microdosing because the side effect jump from 2mg is significant. However, this is also where the glucagon activation threshold likely sits, making it the worst dose at which to reduce peak concentrations through splitting.
High dose (8-12mg weekly)
Higher doses produce the most dramatic weight loss but also the most significant side effects. Nausea and GI effects are most pronounced during the first 2 to 3 weeks at each new dose level, then typically moderate. The dysesthesia reported in trials, that unusual tingling or altered touch sensation, appears primarily at these higher doses.
Interestingly, hair loss concerns are reported anecdotally at higher doses, likely related to the rapid weight loss rather than a direct drug effect. Researchers should ensure adequate protein intake and consider monitoring for nutritional deficiencies during aggressive weight loss phases.
For managing specific side effects, the following guides provide targeted strategies: constipation treatment, tirzepatide constipation management, and managing fatigue on GLP-1 medications.
Practical reconstitution for split dosing
If you decide to proceed with microdosing, proper reconstitution becomes even more critical. Smaller individual doses require more precise measurement, and drawing from the same vial multiple times per week increases contamination risk.
Reconstitution math for split dosing
The goal is to create a concentration that allows you to accurately measure your split dose with a standard insulin syringe. Too concentrated and small volume differences create large dose variations. Too dilute and you are injecting unnecessarily large volumes.
For a 10mg vial reconstituted with 2mL of bacteriostatic water, the concentration is 5mg/mL. On a twice-weekly schedule targeting 8mg total, each injection is 4mg, which equals 0.8mL or 80 units on a standard insulin syringe. That is an easy, accurate measurement.
For three times weekly at the same 8mg target, each injection is approximately 2.67mg, which equals about 0.53mL or 53 units. Still measurable but less forgiving of error.
For daily microdosing at 8mg weekly, each injection is 1.14mg, which equals about 0.23mL or 23 units. At this volume, even a 2-unit measurement error represents nearly 10% dose variation. This is one reason daily microdosing is problematic from a practical standpoint.
The bacteriostatic water guide for retatrutide provides detailed reconstitution ratios for various scenarios. For general reconstitution principles, the peptide reconstitution calculator handles the math automatically.
Storage considerations with multiple draws
Drawing from the same vial multiple times per week accelerates degradation and increases contamination risk. Each needle insertion introduces potential contaminants, even with proper sterile technique. Use bacteriostatic water rather than sterile water for reconstitution, as the benzyl alcohol preservative provides ongoing antimicrobial protection between draws.
Store reconstituted retatrutide in the refrigerator between 2 and 8 degrees Celsius. Do not freeze. The peptide remains stable for approximately 4 to 6 weeks when properly stored, but multiple weekly draws may shorten this window. Our guide on storing peptides after reconstitution covers best practices in detail.
Consider pre-loading syringes if your protocol calls for three or more injections per week. Draw all doses from the vial at once during a single sterile preparation, cap the pre-loaded syringes, and store them in the refrigerator. This minimizes vial punctures and ensures consistent dosing throughout the week.
Injection site rotation for frequent dosing
More injections per week means you need more injection sites to rotate through. Standard sites include the abdomen, avoiding a 2-inch radius around the navel, the front of the thighs, and the back of the upper arms.
With twice-weekly dosing, alternating between left and right sides of the abdomen is usually sufficient. With three or more injections per week, incorporate the thighs and upper arms into your rotation to prevent tissue irritation and lipodystrophy at overused sites. The retatrutide injection site guide provides detailed anatomical guidance, and our broader GLP-1 injection technique guide covers proper subcutaneous injection form.
Tracking progress on a microdosing protocol
Whether you choose standard dosing or microdosing, tracking your progress systematically helps you evaluate whether your protocol is working and make data-driven adjustments.
What to track
Weight is the most obvious metric, but it is also the most misleading when measured in isolation. Daily weight fluctuates by 2 to 5 pounds based on water retention, food volume, and hydration status. Weekly morning weigh-ins, taken on the same day at the same time before eating or drinking, provide more reliable trend data.
Appetite scores, rated on a 1 to 10 scale at consistent times of day, reveal whether your GLP-1 and GIP receptor activation is adequate. If microdosing maintains appetite suppression comparable to weekly dosing, that is a positive indicator. If hunger returns between doses, you may be dropping below effective levels.
Energy levels and exercise performance reflect metabolic activity. The glucagon receptor component should increase resting energy expenditure. If you feel sluggish or notice reduced thermogenesis compared to weekly dosing, the glucagon activation may be compromised.
Side effect logging, tracking nausea severity, GI symptoms, and any other effects after each injection, helps quantify whether microdosing is actually reducing your side effect burden. Some researchers discover that their side effects improve equally with time on standard dosing, making microdosing unnecessary.
Body composition measurements provide the most meaningful long-term data. The GLP-1 plotter tool can help visualize your progress over time, and periodic body fat measurements, whether by DEXA scan, body fat calipers, or even waist circumference, tell you more than the scale alone.
When to adjust your protocol
If after 4 to 6 weeks of microdosing you see significantly slower progress than expected, consider returning to weekly dosing. The adjustment period should be long enough to establish a meaningful trend, but short enough that you do not waste months on a suboptimal protocol.
Specifically, be concerned if weight loss stalls completely for more than 3 weeks despite caloric deficit, if appetite suppression is noticeably weaker than during initial weeks, if you do not notice any thermogenic effect, or if your weekly weight loss rate drops below half of what clinical trials show for your dose level. These signs may indicate insufficient glucagon receptor activation.
Community data on retatrutide microdosing
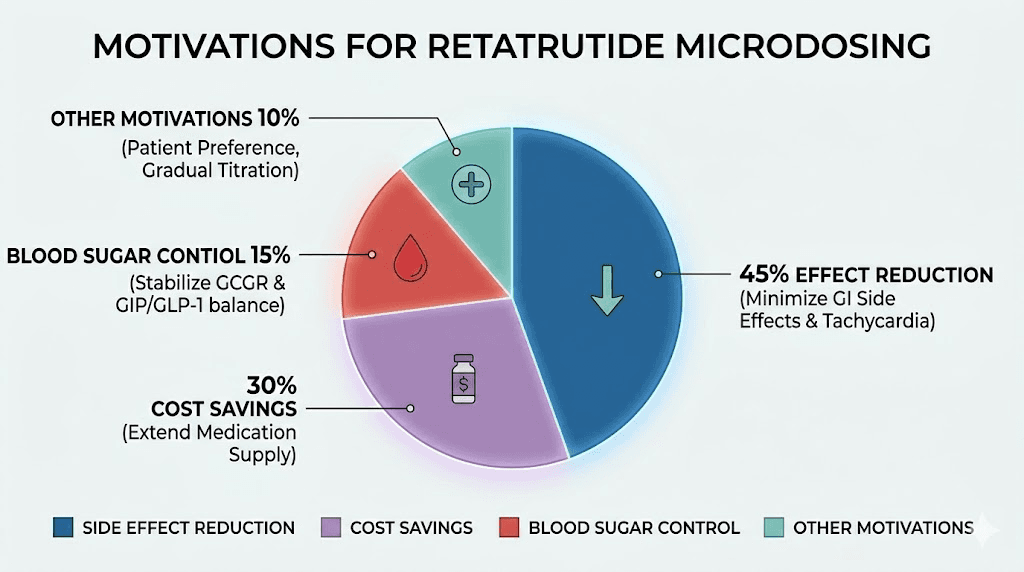
While no clinical trials exist on retatrutide microdosing specifically, community surveys provide some insight into real-world experiences.
Approximately 66% of researchers who try microdosing cite side effect reduction as their primary motivation. About 38% are motivated by cost savings through dose stretching. Only 28% are specifically seeking better blood sugar control or metabolic improvements.
The regret rate among microdosers sits at approximately 22%, meaning roughly one in five researchers who try split dosing eventually return to weekly injections. The most common reason for reverting is slower-than-expected progress, suggesting that the glucagon threshold concern is not merely theoretical.
Perhaps most concerning, 55% of researchers who microdose make the decision without consulting a physician. This is not a judgment, it is a risk factor. Complex dosing modifications for a triple-receptor agonist ideally involve medical guidance, particularly given the absence of clinical trial data on alternative schedules.
For those exploring retatrutide through various sourcing options, understanding the retatrutide availability landscape and reviewing vendor options helps ensure product quality, which becomes even more critical when precise microdosing depends on accurate peptide content.
Safety considerations and risk assessment
Microdosing retatrutide introduces specific safety concerns that standard weekly dosing does not.
Increased injection frequency risks
Each subcutaneous injection carries a small risk of infection, bruising, and tissue damage. At one injection per week, these risks are minimal. At three to seven injections per week, they compound. Proper sterile technique is not optional, it is essential. Use alcohol swabs on injection sites, never re-use needles, and inspect vials for particulates or cloudiness before each draw.
Lipohypertrophy, the buildup of fatty tissue at frequently injected sites, becomes a concern with increased injection frequency. This is not just cosmetic. Injecting into lipohypertrophic tissue reduces drug absorption, creating unpredictable pharmacokinetics that undermine the very stability microdosing aims to achieve.
Dosing accuracy concerns
Smaller individual doses are harder to measure accurately. A 5-unit error on a 100-unit injection represents 5% variation. The same 5-unit error on a 20-unit injection represents 25% variation. Inconsistent dosing can produce erratic blood levels that are worse than the peaks and troughs of standard weekly dosing.
Using insulin syringes with half-unit markings improves accuracy for small volumes. The retatrutide reconstitution chart can help you calculate concentrations that produce measurable volumes for your chosen protocol.
Unknown long-term effects of altered pharmacokinetics
No one knows the long-term safety profile of microdosed retatrutide because no one has studied it. The clinical trial safety data applies exclusively to weekly dosing. Altering the pharmacokinetic profile, even while maintaining the same total weekly dose, could theoretically affect receptor desensitization patterns, immune response to the peptide, or metabolic adaptation in ways that have not been characterized.
This does not mean microdosing is dangerous. It means the safety profile is unknown rather than established. For a medication with documented cardiovascular benefits and a specific metabolic profile, that unknown should factor into your decision.
Interaction considerations
Researchers using retatrutide alongside other supplements or medications should be especially cautious with microdosing. Altered peak and trough levels may change interaction profiles. For example, the effect of retatrutide on gastric emptying varies with concentration, which could affect the absorption of oral medications taken at different times relative to injection.
For researchers combining retatrutide with other compounds, understanding potential supplement interactions and medication combination risks is essential regardless of dosing schedule.
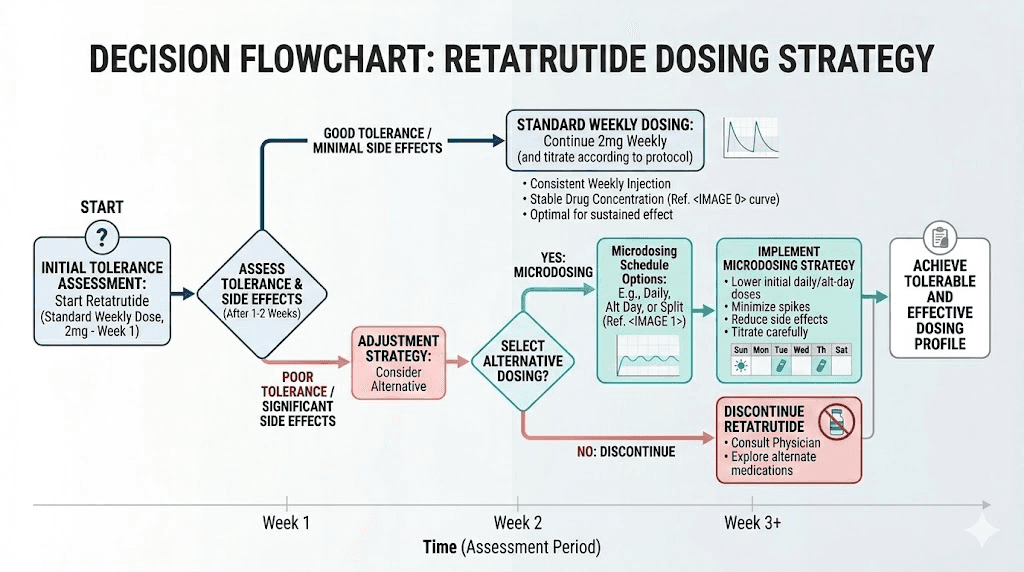
Making the decision: a framework for researchers
SeekPeptides recommends a systematic approach to deciding whether microdosing retatrutide is appropriate for your situation.
First, exhaust standard titration options. Start at the lowest dose, escalate every 4 to 6 weeks, use all available side effect management strategies. If you can tolerate weekly dosing, the evidence strongly supports sticking with it.
Second, if standard titration fails, try twice-weekly dosing first. The 28% peak reduction with 95% theoretical efficacy retention makes this the best compromise. Give it at least 4 to 6 weeks before evaluating results.
Third, track everything. Compare your microdosing results to the expected trajectory from clinical trial data. If weight loss is within 80% of the expected rate for your dose, microdosing may be working adequately. If it is significantly below expected, consider returning to weekly dosing or accepting a slower but tolerable path.
Fourth, do not microdose at the dose level where glucagon activation begins. If you are escalating from 2mg to 4mg and want to try split dosing, recognize that you may be spreading the dose below the glucagon threshold. At higher total doses like 8mg or 12mg, split dosing is more likely to maintain adequate peak concentrations for glucagon activation because each individual injection is still a substantial amount.
Fifth, consult a healthcare provider. This guidance applies to all retatrutide use, but it is especially important for alternative dosing schedules. A provider experienced with GLP-1 medications can help monitor your response and adjust your protocol based on clinical measurements rather than subjective assessment alone.
For researchers who want comprehensive support navigating these decisions, SeekPeptides members access detailed protocol guides, dosing calculators, and a community of experienced researchers who have worked through these exact questions.
Frequently asked questions
Is microdosing retatrutide the same as taking a lower dose?
No. Microdosing maintains the same total weekly dose but splits it across multiple injections. Dose reduction means taking less total medication per week. The goals are different. Microdosing aims to smooth blood levels while maintaining total exposure. Dose reduction aims to decrease total drug exposure, usually to manage side effects or stretch supply.
How long should I try microdosing before deciding if it works?
Give any protocol change at least 4 to 6 weeks before evaluating. This allows your body to reach new steady-state levels, which takes approximately 4 to 5 half-lives (roughly 24 to 30 days for retatrutide). Compare your progress to the expected weight loss trajectory for your dose level to determine if microdosing is producing adequate results.
Can I microdose during the titration phase?
Starting with microdosed retatrutide during titration is not recommended. The clinical trial titration protocols used weekly dosing, and the body needs time to adapt to each new dose level. Microdosing during titration adds complexity and makes it harder to assess tolerance. Complete your titration with weekly dosing, then consider split dosing if side effects remain problematic at your target maintenance dose.
Does microdosing retatrutide affect appetite suppression?
Appetite suppression from the GLP-1 and GIP components appears well-maintained with microdosing, particularly at twice-weekly frequency. Some researchers actually report more consistent appetite control because the smoother concentration curve prevents the hunger breakthroughs that occur just before the next weekly dose. However, this is anecdotal and not validated by clinical research.
What is the minimum effective dose of retatrutide?
The Phase 2 trial showed meaningful weight loss even at 1mg weekly (8.7% at 48 weeks), but the dramatic results associated with retatrutide begin at 4mg and above. For the full triple-agonist effect including glucagon-driven fat oxidation, doses of 4mg weekly or higher appear necessary. Consult the complete retatrutide dosing guide for personalized recommendations.
Should I microdose if I had good results microdosing tirzepatide?
Not necessarily. Tirzepatide microdosing success does not predict retatrutide microdosing success because the glucagon receptor component changes the equation. Tirzepatide lacks glucagon receptor activation, so split dosing preserves more of its efficacy. With retatrutide, you risk losing the additional metabolic benefit that makes it worth the added complexity and cost.
Can I switch between microdosing and weekly dosing?
Yes. There is no physiological reason you cannot alternate between protocols. Some researchers use weekly dosing most of the time and switch to split dosing during periods when side effects are particularly bothersome, such as during dose escalation. Allow 2 to 3 weeks for your body to adjust after any protocol change before evaluating results.
How does retatrutide microdosing compare to microdosing semaglutide?
Semaglutide is a simpler molecule targeting only the GLP-1 receptor, making microdosing more straightforward. The efficacy tradeoffs with semaglutide split dosing are smaller because there is no glucagon threshold concern. If your primary goal is appetite suppression with minimal side effects, microdosing semaglutide is a more conservative and better-understood approach than microdosing retatrutide.
External resources
NEJM: Triple Hormone-Receptor Agonist Retatrutide for Obesity Phase 2 Trial
PMC: Retatrutide - A Game Changer in Obesity Pharmacotherapy
PMC: Efficacy and Safety of Retatrutide - Systematic Review and Meta-Analysis
American Diabetes Association: Retatrutide Clinical Highlights
Nature Medicine: Retatrutide for Metabolic Dysfunction-Associated Steatotic Liver Disease
For researchers serious about optimizing their retatrutide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, personalized dosing tools, and a community of thousands who have navigated these exact decisions.
In case I do not see you, good afternoon, good evening, and good night. May your dosing stay precise, your side effects stay manageable, and your results stay consistent.