Mar 14, 2026

At the cellular level, a single hormone triggers a cascade that controls hunger, blood sugar, gastric emptying, and even cardiovascular protection. That hormone is glucagon-like peptide-1. GLP-1. The body produces it naturally in the intestines every time you eat. But here is where things get interesting. Researchers discovered that synthetic versions of this peptide, pure GLP-1 receptor agonists, could replicate and amplify these effects far beyond what the body achieves on its own. And the results have reshaped metabolic medicine entirely.
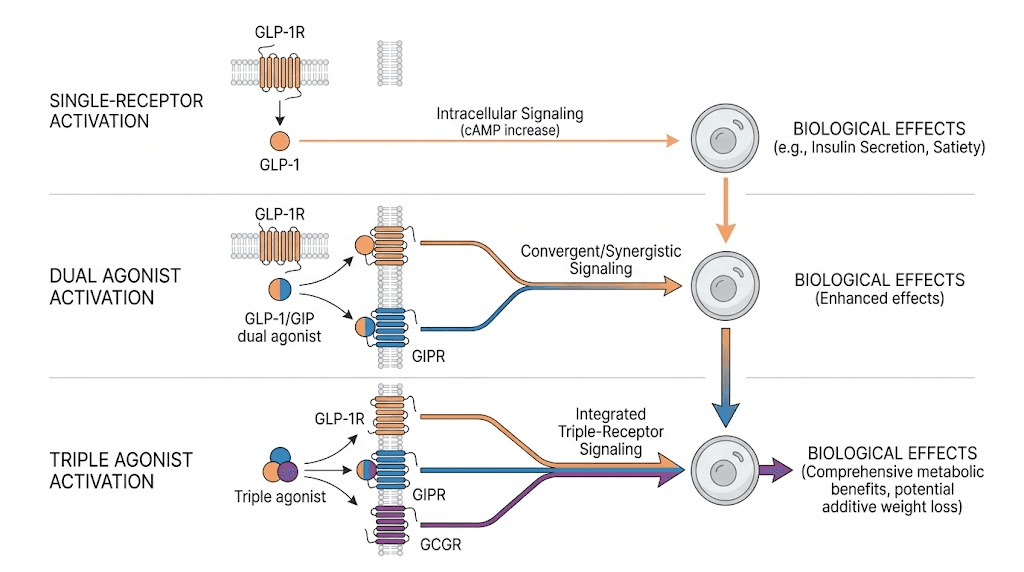
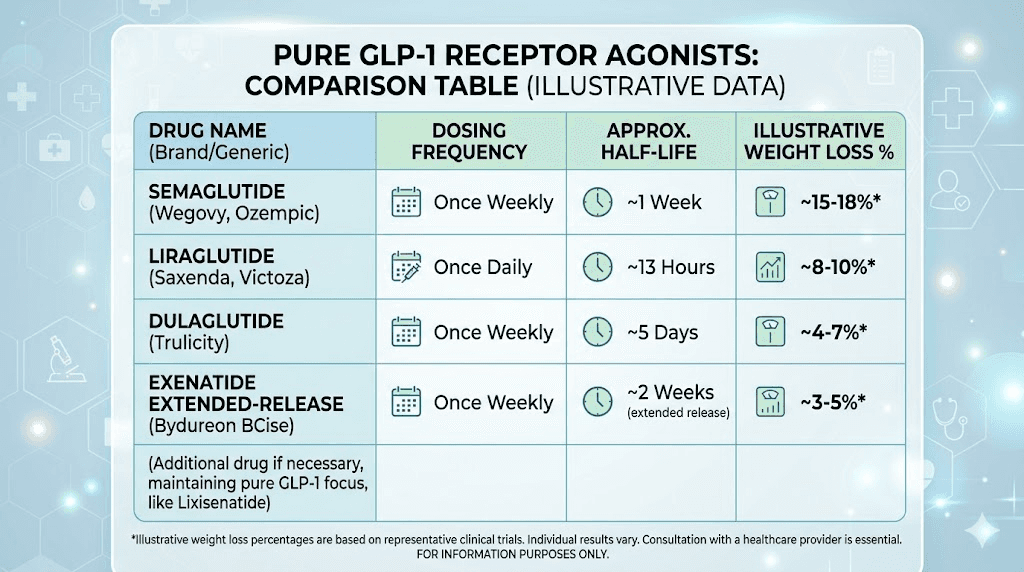
Pure GLP-1 agonists target one receptor. Just one. Unlike dual and triple agonists that activate multiple receptor pathways simultaneously, a pure GLP-1 agonist focuses all its pharmacological power on the GLP-1 receptor alone. This singular focus is not a limitation. It is a design choice that offers specific advantages for certain researchers and certain goals. Semaglutide, liraglutide, exenatide, dulaglutide, lixisenatide, these are the pure GLP-1 agonists that have transformed how the world approaches fat loss, glucose management, and metabolic health.
But the question everyone asks is simple. If dual agonists like tirzepatide and triple agonists like retatrutide hit more receptors and produce greater weight loss numbers, why would anyone choose a pure GLP-1? The answer involves nuance that most guides completely skip. Tolerability profiles, cardiovascular outcome data spanning decades, insurance coverage, oral formulations, established safety records, and situations where more aggressive multi-receptor activation is not actually better. This guide covers every dimension of pure GLP-1 agonists, from molecular mechanisms to practical protocols, so you can make an informed decision based on evidence rather than hype.
What pure GLP-1 actually means
The term pure GLP-1 refers to receptor agonists that exclusively target the GLP-1 receptor without activating GIP, glucagon, or other incretin receptors. To understand why this matters, you need to understand the hormone itself.
GLP-1 is a 30-amino-acid peptide hormone secreted by L-cells in the small intestine and colon. When food reaches these cells, they release GLP-1 into the bloodstream. The hormone then does several things simultaneously. It stimulates insulin secretion from pancreatic beta cells. It suppresses glucagon release from alpha cells. It slows gastric emptying, keeping food in the stomach longer. And it acts on the brain, specifically the hypothalamus and brainstem, to reduce appetite and increase satiety.
Natural GLP-1 has one critical problem. It gets destroyed almost immediately. The enzyme dipeptidyl peptidase-4 (DPP-4) breaks down endogenous GLP-1 within 2 to 3 minutes of release. This ultra-short half-life means the body never maintains high circulating levels of the hormone for extended periods. Synthetic pure GLP-1 agonists solve this problem through molecular modifications that resist DPP-4 degradation, extending the half-life from minutes to hours or even days.
That is the core concept. A pure GLP-1 agonist mimics endogenous GLP-1 but lasts dramatically longer in the body. Nothing more. Nothing less. It does not activate GIP receptors like tirzepatide does. It does not activate glucagon receptors like retatrutide does. It focuses entirely on one target.
Two families of pure GLP-1 agonists
Not all pure GLP-1 agonists are created equal. They fall into two distinct families based on their molecular origin.
Human GLP-1 analogs are structurally similar to natural human GLP-1 with targeted modifications. Semaglutide, liraglutide, and dulaglutide belong to this family. They share the same amino acid backbone as endogenous GLP-1 but include fatty acid chains, amino acid substitutions, or Fc fragment fusions that extend their duration of action. Because they closely resemble the natural hormone, these analogs tend to have predictable pharmacological profiles.
Exendin-based agonists come from a completely different source. Exenatide was originally isolated from the saliva of the Gila monster lizard (Heloderma suspectum). It shares roughly 53% sequence homology with human GLP-1, enough to activate the same receptor but structurally distinct enough to resist DPP-4 degradation naturally. Lixisenatide is also exendin-based. These agents tend to have shorter durations of action compared to the human analogs, though extended-release formulations have narrowed this gap.
Understanding which family your pure GLP-1 agonist belongs to matters. It affects immunogenicity (the likelihood of developing antibodies against the medication), injection site reactions, and pharmacokinetic profiles.
Every pure GLP-1 agonist available right now
Six pure GLP-1 receptor agonists have received regulatory approval. Each has a distinct profile. Some are better for glucose control. Others excel at weight loss. Some require daily injection. Others need only weekly dosing. Knowing the differences helps you match the right molecule to the right goal.
Semaglutide
Semaglutide is the most potent pure GLP-1 agonist currently available. It is the active ingredient in Ozempic (for type 2 diabetes), Wegovy (for obesity), and Rybelsus (the oral formulation). Its molecular design includes a C-18 fatty diacid chain attached to a modified GLP-1 backbone, enabling strong albumin binding and a half-life of approximately 165 hours, roughly one week.
This weekly dosing convenience, combined with superior efficacy, has made semaglutide the dominant pure GLP-1 agonist in clinical practice. In the STEP trials, semaglutide 2.4 mg weekly produced average weight loss of approximately 15% of body weight over 68 weeks. No other pure GLP-1 agonist comes close to these numbers.
For researchers interested in practical details, understanding how to reconstitute semaglutide properly is essential for maintaining potency. The semaglutide dosage calculator helps determine exact unit conversions, and knowing the best time of day to take semaglutide can optimize results. Storage matters too, and researchers frequently ask how long semaglutide stays good once reconstituted.
Liraglutide
Liraglutide was the first pure GLP-1 agonist to gain FDA approval specifically for weight management (as Saxenda, at 3.0 mg daily). It also treats type 2 diabetes under the brand name Victoza at lower doses. Its half-life is approximately 13 hours, requiring daily injection.
While liraglutide produces less weight loss than semaglutide (roughly 8% versus 15% at optimal doses), it holds a critical advantage. Decades of cardiovascular outcome data. The LEADER trial demonstrated a 13% reduction in major adverse cardiovascular events (MACE) with liraglutide compared to placebo. This extensive safety track record makes liraglutide a preferred choice for researchers with significant cardiovascular risk factors who want a thoroughly studied pure GLP-1 option.
The daily dosing requirement is a drawback for some. But others actually prefer the finer control that daily administration provides. If withdrawal symptoms or rebound effects concern you, the shorter half-life of liraglutide means adjustments take effect faster.
Dulaglutide
Dulaglutide (Trulicity) combines a GLP-1 analog with an immunoglobulin G4 (IgG4) Fc fragment, creating a large molecule that clears slowly from the body. Its half-life is approximately 5 days, supporting weekly dosing. Dulaglutide produces moderate weight loss, typically 3-5% of body weight, positioning it more as a glucose management tool than a weight loss agent.
What distinguishes dulaglutide is the REWIND trial, which showed cardiovascular benefits even in patients without established cardiovascular disease. Most other GLP-1 cardiovascular outcome trials enrolled primarily high-risk patients. REWIND demonstrated that the protective effects extend to a broader population. For researchers primarily focused on metabolic health rather than aggressive weight loss, dulaglutide offers a well-tolerated weekly option.
Exenatide
Exenatide exists in two formulations. Byetta requires twice-daily injection. Bydureon is an extended-release formulation dosed weekly. As the first GLP-1 agonist ever approved (2005), exenatide pioneered the entire class. Its exendin-4 backbone provides natural resistance to DPP-4 degradation.
In terms of weight loss and glucose lowering, exenatide is the least potent of the commonly used pure GLP-1 agonists. Average weight loss with exenatide extended-release runs approximately 2-4% of body weight. However, it remains available and some researchers who started on exenatide years ago continue to use it successfully.
Lixisenatide
Lixisenatide (Adlyxin) is a once-daily exendin-based pure GLP-1 agonist primarily used for type 2 diabetes. It has a relatively short duration of action and produces modest weight loss. Its primary clinical niche involves combination with basal insulin (available as Soliqua, a fixed-ratio combination with insulin glargine).
For researchers focused on peptide-based approaches to metabolic management, lixisenatide represents a lighter-touch option that can complement other therapies without the potent appetite suppression of semaglutide.
Oral semaglutide
While technically the same molecule as injectable semaglutide, the oral formulation (Rybelsus) deserves separate mention. It uses an absorption enhancer called SNAC (sodium N-[8-(2-hydroxybenzoyl) amino] caprylate) to facilitate absorption through the gastric mucosa. This makes it the only oral pure GLP-1 agonist available.
Oral semaglutide produces somewhat less weight loss than the injectable version due to lower bioavailability (roughly 1% of the oral dose reaches systemic circulation). But for researchers who prefer oral administration over injections, it removes one of the biggest barriers to GLP-1 therapy. The emergence of GLP-1 drops and other oral formulations continues to expand delivery options.
How pure GLP-1 agonists work at every level
Understanding the mechanism of action is not just academic. It explains why pure GLP-1 agonists cause the side effects they do, why some researchers respond better than others, and why timing and dosing matter so much.
Pancreatic effects
Pure GLP-1 agonists are glucose-dependent insulin secretagogues. This is a critical distinction. They only stimulate insulin release when blood glucose is elevated. When blood sugar is normal or low, the insulin-stimulating effect diminishes dramatically. This glucose-dependent mechanism means pure GLP-1 agonists carry a much lower risk of hypoglycemia compared to sulfonylureas or exogenous insulin.
Simultaneously, these agonists suppress glucagon secretion from pancreatic alpha cells. Glucagon normally raises blood sugar by promoting hepatic glucose production. By suppressing glucagon in a glucose-dependent manner, pure GLP-1 agonists reduce the liver output of glucose after meals. This dual pancreatic effect, increasing insulin while decreasing glucagon, produces powerful glucose control without the dangerous lows that plague other diabetes medications.
Gastric effects
Delayed gastric emptying is one of the most impactful effects of pure GLP-1 agonists, and also the source of their most common side effects. By slowing the rate at which food leaves the stomach, these agonists reduce postprandial glucose spikes, extend satiety, and decrease overall caloric intake.
This is why sulfur burps, constipation, and nausea are so common with GLP-1 therapy. The stomach is literally holding onto food longer than usual. For most researchers, these effects diminish over 4-8 weeks as the body adapts. Those experiencing persistent issues should review dietary strategies and foods to avoid while on GLP-1 therapy.
Central nervous system effects
GLP-1 receptors exist throughout the brain, particularly in the hypothalamus, area postrema, and nucleus tractus solitarius. Activation of these receptors reduces appetite, increases satiety, and may alter food preferences. Some research suggests GLP-1 agonism reduces the reward value of food, making hyperpalatable foods less appealing.
This central appetite suppression is arguably the most important mechanism for weight loss. It is why researchers on semaglutide often report simply not thinking about food. Not fighting cravings. Just genuine disinterest in eating beyond physiological need. Understanding how quickly appetite suppression begins helps set realistic expectations.
Recent research has also identified potential neuroprotective effects of GLP-1 receptor activation. Studies are exploring applications in Alzheimer disease, Parkinson disease, traumatic brain injury, and even substance use disorders. These emerging indications represent a frontier that pure GLP-1 agonists, with their extensive safety data, are uniquely positioned to explore.
Cardiovascular effects
Multiple cardiovascular outcome trials have demonstrated that pure GLP-1 agonists reduce major adverse cardiovascular events. The mechanisms include improved endothelial function, reduced inflammation (measured by C-reactive protein), improved lipid profiles, modest blood pressure reduction, and direct cardioprotective effects on myocardial cells.
The SELECT trial showed that semaglutide 2.4 mg weekly reduced MACE by 20% in individuals with overweight or obesity and established cardiovascular disease, even without diabetes. This was a landmark finding because it demonstrated cardiovascular benefit driven by weight loss and metabolic improvement rather than glucose control alone.
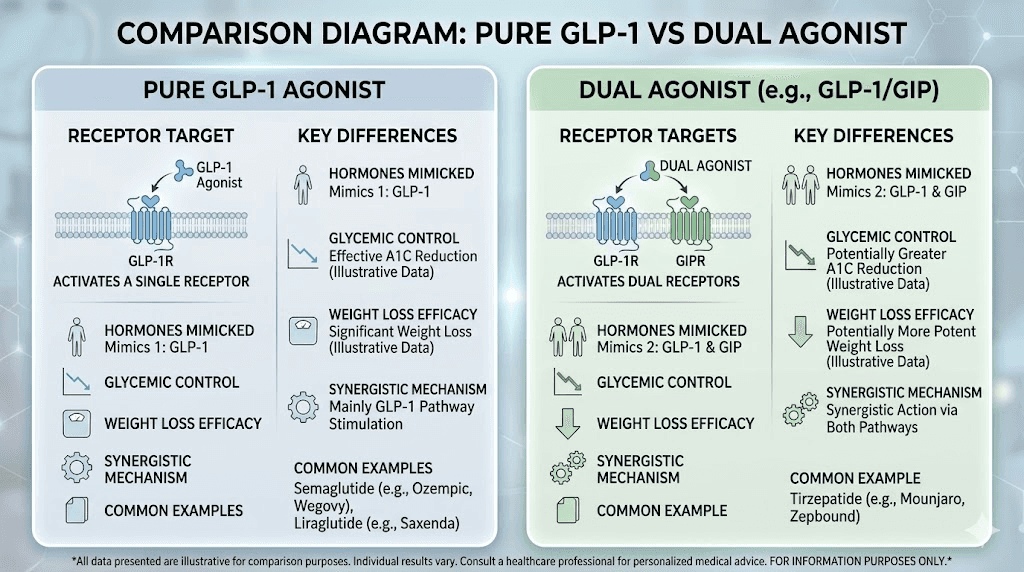
Pure GLP-1 versus dual agonists
This is the comparison everyone wants. Semaglutide versus tirzepatide. Pure GLP-1 versus GLP-1/GIP dual agonism. One receptor versus two.
The numbers tell one story. Tirzepatide versus semaglutide head-to-head trials (SURMOUNT and SURPASS programs) consistently show greater weight loss with tirzepatide. At maximum doses, tirzepatide produces approximately 20-26% body weight reduction compared to semaglutide at roughly 15-17%. The additional GIP receptor activation appears to enhance insulin sensitivity, improve lipid metabolism, and amplify weight loss through mechanisms that GLP-1 alone does not access.
But numbers do not tell the complete story.
Pure GLP-1 agonists have something tirzepatide does not. Time. Semaglutide and liraglutide have years of real-world safety data across millions of patients. Cardiovascular outcome trials with long follow-up periods. Extensive post-marketing surveillance. Tirzepatide, while promising, was approved more recently and its long-term safety profile is still being established.
There are practical differences too. Semaglutide has an oral formulation. Tirzepatide does not (though oral tirzepatide is in development). Insurance coverage is often better for established pure GLP-1 agonists. Compounded semaglutide has been more widely available through pharmacy compounding than tirzepatide formulations.
Some researchers experience different side effect profiles between the two classes. Tirzepatide tends to produce more diarrhea relative to semaglutide. Semaglutide tends to produce more nausea relative to tirzepatide. Individual tolerance varies enormously, and some researchers who do not respond well to one class respond excellently to the other. If semaglutide does not work, switching to a dual agonist is a reasonable next step, and vice versa.
When pure GLP-1 is the better choice
Choose a pure GLP-1 agonist when cardiovascular outcome data matters most to your decision-making. When you want the option of oral administration. When you prefer a molecule with the longest real-world safety track record. When insurance coverage is a factor. When you want access to compounded formulations. When your primary goal is glucose management rather than maximum weight loss. When you want the most conservative, well-studied approach to GLP-1 therapy.
Choose a dual agonist when maximum weight loss is the priority and you have discussed the newer safety profile with your physician. When you have plateaued on a pure GLP-1 and need additional receptor activation. When semaglutide plateau has stalled your progress despite optimization.
Pure GLP-1 versus triple agonists
Triple agonists like retatrutide activate three receptors simultaneously: GLP-1, GIP, and glucagon. The preliminary results are staggering. Phase 2 trials showed up to 24% body weight loss at 48 weeks, and the Phase 3 TRIUMPH-4 readout in late 2025 reported 28.7% weight loss. These are numbers that pure GLP-1 agonists simply cannot match.
But retatrutide is not yet FDA-approved. It is still in clinical trials with an expected FDA submission in late 2026 and potential approval by mid-2027. This means researchers cannot access it through traditional pharmaceutical channels. Those obtaining it do so through research peptide suppliers, which introduces questions about purity, potency, and manufacturing standards that do not exist with approved pure GLP-1 medications.
The glucagon receptor activation in triple agonists is a double-edged sword. Glucagon increases energy expenditure and promotes hepatic fat oxidation, which enhances weight loss. But glucagon also raises blood sugar. The balance between GLP-1 mediated glucose lowering and glucagon mediated glucose raising requires careful dose titration. For researchers with significant insulin resistance or prediabetes, this dynamic adds complexity that a pure GLP-1 agonist avoids entirely.
Understanding retatrutide dose schedules and dosage charts is essential for those considering triple agonism. The retatrutide versus semaglutide comparison highlights the key tradeoffs. And for those weighing all three tiers, the three-way comparison guide provides the most complete analysis available.
Weight loss results with pure GLP-1 agonists
Let us look at the actual clinical data. Numbers matter. Ranges matter. Timelines matter.
Semaglutide weight loss data
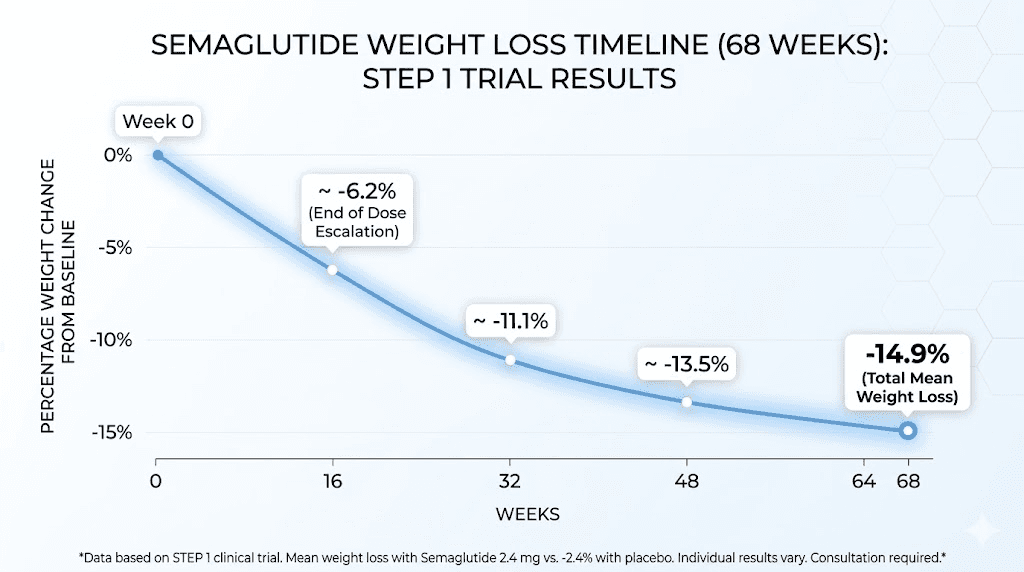
The STEP clinical trial program provides the most comprehensive data on semaglutide for weight loss. STEP 1 enrolled adults with obesity (BMI 30 or higher) or overweight (BMI 27 or higher) with at least one weight-related comorbidity. At 68 weeks, the 2.4 mg weekly dose produced mean weight loss of 14.9% versus 2.4% with placebo. Approximately one-third of participants lost 20% or more of their body weight.
STEP 2 focused on adults with type 2 diabetes and obesity. Weight loss was somewhat less, approximately 9.6% at 68 weeks, reflecting the metabolic complexity of established diabetes. STEP 3 combined semaglutide with intensive behavioral therapy and showed 16% mean weight loss. STEP 5 extended the observation period to 104 weeks and demonstrated that weight loss was maintained with continued use.
What happens week by week on semaglutide? Most researchers notice appetite suppression within the first 1-2 weeks. Measurable weight loss typically begins by weeks 4-6. The rate of loss is fastest during months 2-5, then gradually slows as a new equilibrium is reached. How fast semaglutide works depends on individual factors including starting weight, diet quality, activity level, and metabolic status.
Some researchers experience disappointing early results. If you are 4 weeks on semaglutide with no weight loss, that does not necessarily indicate failure. It often means the titration schedule needs more time to reach therapeutic doses, or dietary patterns need adjustment. The complete guide on why weight loss stalls on semaglutide covers every common cause and solution.
Liraglutide weight loss data
The SCALE trials evaluated liraglutide 3.0 mg daily for weight management. SCALE Obesity and Prediabetes showed mean weight loss of 8.0% versus 2.6% with placebo at 56 weeks. About 33% of participants lost 10% or more. These numbers are respectable but clearly below semaglutide performance.
However, liraglutide has one advantage in the weight loss timeline. Because it is dosed daily, some researchers achieve steady-state faster and experience less of the pronounced peak-trough cycle that can occur with weekly semaglutide injections. The consistent daily exposure can provide more stable appetite suppression for certain individuals.
Comparative weight loss table
Pure GLP-1 agonist | Dose | Frequency | Mean weight loss | Trial duration | Top performers |
|---|---|---|---|---|---|
Semaglutide | 2.4 mg | Weekly | 14.9% | 68 weeks | 33% lost 20%+ |
Liraglutide | 3.0 mg | Daily | 8.0% | 56 weeks | 33% lost 10%+ |
Dulaglutide | 4.5 mg | Weekly | 5.0% | 36 weeks | Modest |
Exenatide ER | 2 mg | Weekly | 2.3% | 28 weeks | Variable |
The hierarchy is clear. Among pure GLP-1 agonists, semaglutide stands alone at the top for weight loss. Liraglutide occupies a solid middle ground. Dulaglutide and exenatide are primarily glucose management tools where weight loss is a secondary benefit.
For context, tirzepatide before and after results show 20-26% weight loss at maximum doses, and retatrutide trials have reported up to 28.7%. But these are multi-receptor agonists, not pure GLP-1.
Side effects of pure GLP-1 agonists
Every medication has side effects. Pure GLP-1 agonists are no exception. But their side effect profile is well-characterized after years of clinical use and post-marketing surveillance. Knowing what to expect, and what to do about it, separates successful protocols from abandoned ones.
Gastrointestinal effects
Nausea is the most common side effect. It affects 20-44% of researchers depending on the specific agonist and dose. Nausea is typically worst during the titration phase, when doses are being increased, and improves significantly within 4-8 weeks at a stable dose. Eating smaller, more frequent meals helps. Avoiding high-fat and heavily processed foods helps more.
Vomiting occurs in roughly 10-25% of cases. Like nausea, it tends to be transient and dose-related. Diarrhea affects 10-20% of researchers and is usually mild. Constipation is paradoxically also common (10-15%), reflecting the slowed gastric motility. Both tend to improve with dietary adjustments and adequate hydration.
Sulfur burps deserve special mention. While not dangerous, they are unpleasant and underreported in clinical trials. They result from prolonged gastric retention of food, allowing bacterial fermentation to produce hydrogen sulfide gas. Reducing portion sizes and avoiding sulfur-rich foods (eggs, cruciferous vegetables) typically resolves the issue.
Fatigue and energy
Fatigue on GLP-1 therapy is common, particularly in the early weeks. It results from reduced caloric intake, metabolic adaptation, and sometimes dehydration. Some researchers report that GLP-1 fatigue improves dramatically once they optimize their protein intake and hydration. The question of whether semaglutide gives you energy long-term depends largely on these nutritional factors.
Headaches
GLP-1 headaches occur in approximately 10-15% of researchers, most commonly during the first 2-4 weeks. They are often related to dehydration, blood sugar fluctuations, or caffeine intake changes as appetite decreases. Maintaining fluid intake of at least 2-3 liters daily usually prevents or resolves this issue. For persistent headaches, the headache treatment guide covers evidence-based remedies.
Hair loss
Hair loss on GLP-1 therapy affects a minority of researchers but causes significant concern when it occurs. The mechanism is telogen effluvium, a stress response triggered by rapid weight loss rather than the medication itself. It typically begins 2-4 months after significant weight loss starts and resolves within 6-12 months. Adequate protein intake (1.2-1.6 g/kg body weight daily) and nutritional supplementation can minimize this effect. The GLP-1 vitamin guide covers essential nutritional support.
Hormonal effects
Questions about menstrual changes on semaglutide are increasingly common. Rapid weight loss and metabolic shifts can alter estrogen levels, potentially affecting cycle regularity. Understanding whether semaglutide increases estrogen levels helps female researchers prepare for possible changes. These effects are generally related to body fat changes rather than direct hormonal actions of the medication.
Serious but rare side effects
Pancreatitis occurs in approximately 0.1-0.3% of GLP-1 agonist users. While rare, it requires immediate medical attention. Symptoms include severe, persistent abdominal pain radiating to the back, nausea, and vomiting that differs from the typical GI side effects.
Gallbladder problems are more common with rapid weight loss and affect approximately 1-3% of researchers on GLP-1 therapy. This is a class effect related to weight loss velocity rather than the specific medication.
Medullary thyroid carcinoma (MTC) risk has been identified in rodent studies with all GLP-1 agonists. The clinical significance in humans remains uncertain, but GLP-1 agonists are contraindicated in individuals with a personal or family history of MTC or Multiple Endocrine Neoplasia syndrome type 2 (MEN2).
Dosing protocols for pure GLP-1 agonists
Proper dosing makes or breaks a GLP-1 protocol. Too fast and side effects become intolerable. Too slow and results lag expectations. The titration schedule exists for a reason, and skipping steps rarely ends well.
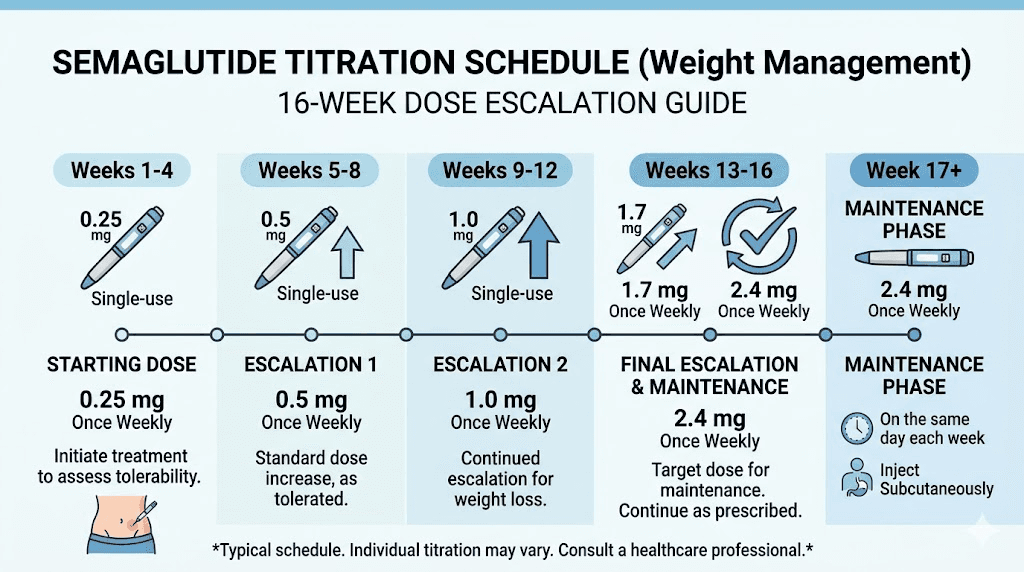
Semaglutide titration protocol
The standard semaglutide titration for weight management follows a 16-week escalation:
Weeks 1-4: 0.25 mg weekly
Weeks 5-8: 0.5 mg weekly
Weeks 9-12: 1.0 mg weekly
Weeks 13-16: 1.7 mg weekly
Week 17 onward: 2.4 mg weekly (maintenance)
Each step increase can temporarily worsen GI side effects. If side effects are severe at any step, extending that step by an additional 2-4 weeks before escalating is perfectly acceptable and often recommended. The semaglutide dosage calculator helps with precise unit conversions for compounded formulations. Understanding semaglutide dosage in units is essential for researchers working with insulin syringes.
For researchers using compounded semaglutide vials, knowing how much bacteriostatic water to mix determines concentration accuracy. The 5mg vial dosage chart provides visual reference for common reconstitution ratios. Proper injection site selection and technique affect absorption and comfort.
Liraglutide titration protocol
Liraglutide titrates over 5 weeks for weight management:
Week 1: 0.6 mg daily
Week 2: 1.2 mg daily
Week 3: 1.8 mg daily
Week 4: 2.4 mg daily
Week 5 onward: 3.0 mg daily (maintenance)
The faster titration compared to semaglutide means researchers reach therapeutic doses sooner. But daily injection compliance becomes the key variable. Missing doses with liraglutide has a more immediate impact on appetite suppression compared to missing a weekly semaglutide dose.
Microdosing approaches
Some researchers use sub-therapeutic doses of pure GLP-1 agonists, particularly for applications beyond weight loss. Microdosing GLP-1 for autoimmune conditions is an emerging area of research based on the anti-inflammatory properties of GLP-1 receptor activation. These protocols typically use 10-25% of the standard therapeutic dose.
The rationale is that lower doses may provide anti-inflammatory and neuroprotective benefits without the profound appetite suppression and GI effects of full-dose therapy. While the evidence base for microdosing is still developing, the approach is gaining traction in the research community.
Injection technique and timing
Knowing where to inject GLP-1 medications affects both absorption and comfort. The abdomen, thigh, and upper arm are standard sites. Rotating between sites prevents lipodystrophy. Proper injection technique includes using the right syringe type, the correct syringe selection, pinching the skin appropriately, and injecting at the proper angle.
Timing matters too. Choosing the best time for your GLP-1 shot can minimize side effects and optimize efficacy. Many researchers prefer evening injection to sleep through the initial nausea peak. Others prefer morning injection to maximize daytime appetite suppression.
Optimizing results on pure GLP-1 therapy
Taking a pure GLP-1 agonist is not a passive process. The medication provides a foundation. What you build on that foundation determines your results.
Nutrition strategy
Protein intake is the single most important dietary factor during GLP-1 therapy. Reduced appetite means reduced food intake, and without deliberate protein prioritization, muscle mass loss can account for 20-40% of total weight lost. Aim for 1.2-1.6 grams of protein per kilogram of body weight daily. Protein shakes designed for GLP-1 users can help hit targets when appetite makes solid food unappealing.
Meal composition matters beyond protein. Foods that work well with semaglutide tend to be lean proteins, complex carbohydrates, and fiber-rich vegetables. Foods to avoid include high-fat items, heavily processed foods, and sugary drinks that exacerbate GI side effects without providing nutritional value.
Meal planning can dramatically improve the GLP-1 experience. A structured semaglutide diet plan ensures adequate nutrition despite reduced appetite. High-protein breakfast ideas and GLP-1 dinner recipes provide practical guidance for daily meal preparation.
Exercise considerations
Can you lose weight on semaglutide without exercise? Yes. The clinical trials demonstrated significant weight loss with semaglutide alone. But exercise, particularly resistance training, dramatically improves body composition outcomes. Researchers who combine GLP-1 therapy with resistance training 2-4 times weekly preserve significantly more muscle mass and achieve a more favorable fat-to-lean tissue ratio.
Cardiovascular exercise remains beneficial for heart health and metabolic parameters. But the priority during GLP-1 mediated weight loss should be resistance training to protect lean body mass.
Supplement support
The essential vitamins and supplements for GLP-1 users address nutritional gaps created by reduced food intake. Key supplements include a high-quality multivitamin, vitamin D (often deficient in individuals with obesity), magnesium, omega-3 fatty acids, and a good probiotic to support gut health during the microbiome shifts caused by altered eating patterns.
Compound formulations that combine semaglutide with specific vitamins and cofactors have gained popularity. Semaglutide with B12 is among the most common combinations, designed to prevent the B12 deficiency that can occur with reduced dietary intake. Semaglutide with glycine aims to provide additional metabolic support. Niacinamide semaglutide compounds offer another combination approach.
Monitoring and adjustments
Regular monitoring during pure GLP-1 therapy should include body weight and composition measurements, fasting glucose and HbA1c (if applicable), lipid panels, liver function tests, kidney function, and basic metabolic panels. Many researchers use a GLP-1 plotter tool to track their progress visually over time.
If progress stalls, the GLP-1 reset protocol offers evidence-based strategies for restoring receptor sensitivity. Understanding why weight loss stalls on semaglutide and addressing the underlying cause, whether it is dose plateau, dietary drift, or metabolic adaptation, is critical for long-term success.
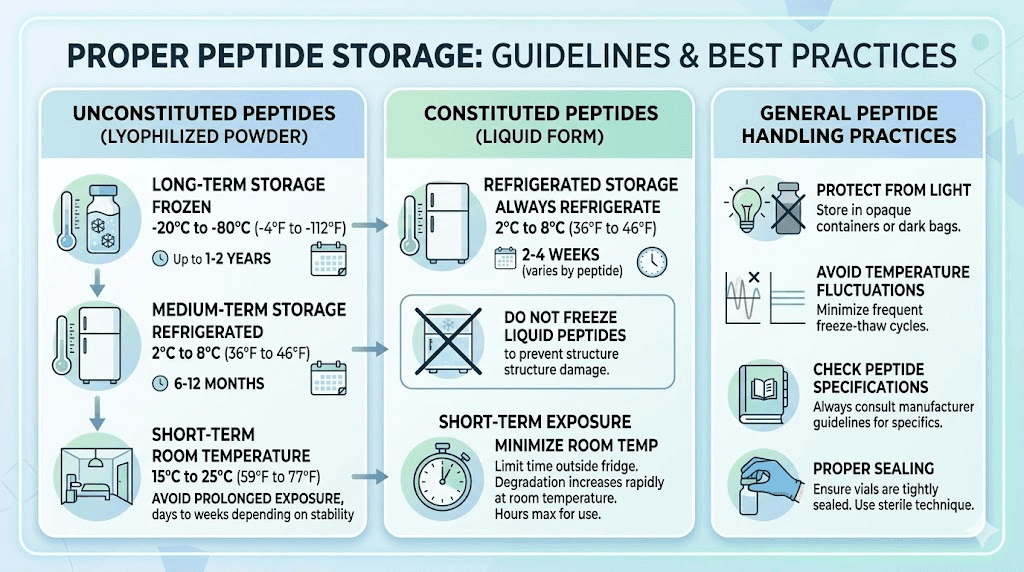
Practical storage and handling
Pure GLP-1 agonists are peptides. They are sensitive to temperature, light, and mechanical stress. Proper storage directly affects potency and safety.
Unopened vials and pens should be refrigerated at 2-8 degrees Celsius (36-46 degrees Fahrenheit). Once in use, most formulations can remain at room temperature (up to 30 degrees C / 86 degrees F) for a defined period, typically 28-56 days depending on the product. Knowing how long semaglutide remains potent and how long it lasts in the fridge prevents waste and ensures efficacy.
Temperature excursions happen. If your semaglutide arrives hot during shipping, assessment of potency is needed. Brief temperature excursions (a few hours) may not significantly degrade the peptide, but extended exposure to heat or freezing temperatures will. If you accidentally leave semaglutide out overnight, the viability depends on ambient temperature and duration of exposure.
Travel with GLP-1 medications requires planning. The complete travel with semaglutide guide covers TSA regulations, cooling cases, international considerations, and how to maintain the cold chain during transit.
Questions about expiration are common. Whether expired semaglutide retains any potency depends on storage conditions, but using expired peptides is generally not recommended. Understanding the 28-day expiration guideline for opened products helps plan dosing schedules appropriately.
Compounded versus pharmaceutical pure GLP-1
The distinction between FDA-approved pharmaceutical GLP-1 products and compounded versions is important for researchers to understand.
Pharmaceutical products (Ozempic, Wegovy, Rybelsus, Victoza, Trulicity) undergo rigorous manufacturing controls, batch testing, and regulatory oversight. Their potency, sterility, and purity are guaranteed by the manufacturer and monitored by the FDA.
Compounded semaglutide is prepared by compounding pharmacies and has been available under FDA shortage allowances. These formulations can be significantly less expensive. However, they are not FDA-approved products and quality varies between pharmacies. Researchers should verify that their compounding pharmacy is licensed, accredited (preferably PCAB accredited), and performs appropriate potency testing.
Sourcing research-grade semaglutide peptide introduces additional considerations. Purity certificates (HPLC and mass spectrometry data), supplier reputation, and proper reconstitution technique all affect the quality of the research product. Proper reconstitution using bacteriostatic water at the correct ratio is non-negotiable for maintaining peptide integrity.
The peptide reconstitution calculator eliminates guesswork from the mixing process. And the peptide cost calculator helps researchers compare the true cost per dose across different sources and formulations.
Emerging applications of pure GLP-1 agonists
Weight loss and glucose control were the starting point. But the therapeutic potential of pure GLP-1 agonists extends far beyond metabolism.
Cardiovascular protection
The cardiovascular benefits of pure GLP-1 agonists represent perhaps their most significant advantage over newer multi-receptor agonists. Multiple large-scale cardiovascular outcome trials (LEADER, SUSTAIN-6, SELECT, PIONEER-6) have demonstrated reduced rates of heart attack, stroke, and cardiovascular death with GLP-1 agonist therapy. The SELECT trial was particularly groundbreaking because it showed cardiovascular benefit in individuals without diabetes, purely on the basis of overweight/obesity with established cardiovascular disease.
No dual or triple agonist has yet completed a cardiovascular outcome trial of comparable size and duration. This gives pure GLP-1 agonists a unique evidence advantage for researchers where cardiovascular risk reduction is a primary consideration.
Neurodegenerative disease
GLP-1 receptors in the brain make pure GLP-1 agonists candidates for neurodegenerative conditions. Liraglutide and semaglutide are being studied in clinical trials for Alzheimer disease and Parkinson disease. Preclinical data suggests GLP-1 receptor activation reduces neuroinflammation, promotes neuronal survival, and improves synaptic plasticity.
The implications are enormous. If GLP-1 agonists demonstrate neuroprotective effects in ongoing trials, the clinical applications of these pure compounds will expand dramatically beyond metabolic medicine.
Liver disease
Non-alcoholic steatohepatitis (NASH), now called metabolic dysfunction-associated steatohepatitis (MASH), is a growing epidemic closely linked to obesity and insulin resistance. Semaglutide has shown significant improvements in liver histology in clinical trials, with resolution of steatohepatitis in a meaningful proportion of patients. The combined effects of weight loss, improved insulin sensitivity, and potentially direct hepatoprotective mechanisms make pure GLP-1 agonists a promising treatment for liver disease.
Substance use disorders
Perhaps the most unexpected emerging application involves addiction. Observational data and early clinical trials suggest GLP-1 agonists may reduce alcohol consumption, tobacco use, and potentially other substance use behaviors. The mechanism likely involves modulation of reward pathways in the brain, the same pathways that reduce food reward. While these applications remain investigational, they highlight the broad biological impact of GLP-1 receptor activation.
Autoimmune and inflammatory conditions
The anti-inflammatory properties of GLP-1 receptor activation have generated interest in autoimmune conditions. Research into microdosing GLP-1 for autoimmune disease is exploring whether sub-therapeutic doses can provide anti-inflammatory benefits without the metabolic effects of full-dose therapy. Early results are intriguing but preliminary.
Conditions like lipedema, endometriosis, and Hashimoto thyroiditis are areas where GLP-1 therapy is being explored for both metabolic and inflammatory benefits.
Who should consider pure GLP-1 agonists
Not everyone needs the most aggressive metabolic intervention available. Pure GLP-1 agonists occupy a specific and valuable position in the therapeutic landscape.
Ideal candidates
Researchers with a BMI qualifying for GLP-1 therapy who want the most evidence-supported approach to weight management. Those with cardiovascular risk factors who value the extensive outcome trial data. Individuals with type 2 diabetes who need glucose management as a primary goal with weight loss as a secondary benefit. Researchers who prefer oral administration options. Those who have been told they need GLP-1 therapy and want the most conservative, well-studied first step.
When to consider alternatives
If you have tried pure GLP-1 agonists at maximum tolerated doses and achieved insufficient weight loss, stepping up to a dual agonist is reasonable. If your semaglutide has plateaued despite optimization, the conversion to tirzepatide may provide additional receptor activation. Some researchers explore whether alternating between semaglutide and tirzepatide offers benefits, though evidence for this approach remains limited.
Combination approaches are another consideration. Combining phentermine with semaglutide provides complementary mechanisms, with phentermine offering norepinephrine-mediated appetite suppression alongside GLP-1 agonism. Understanding how phentermine compares to semaglutide and the broader phentermine versus GLP-1 comparison helps inform these decisions.
Lifestyle interactions with pure GLP-1 therapy
Real life does not pause during GLP-1 therapy. Researchers have questions about alcohol, surgery, pregnancy, and daily activities. The answers matter.
Alcohol
Drinking on semaglutide is technically possible but comes with important caveats. GLP-1 agonists slow gastric emptying, which can alter alcohol absorption patterns. Some researchers report increased alcohol sensitivity, feeling the effects of smaller amounts. Others report decreased interest in alcohol entirely, consistent with the reward pathway modulation discussed earlier. Moderation is the standard recommendation, particularly during the titration phase when GI sensitivity is highest.
Surgery
Pure GLP-1 agonists delay gastric emptying, which creates aspiration risk during anesthesia. Current guidelines generally recommend discontinuing GLP-1 agonists 1-3 weeks before elective surgery involving general anesthesia. Understanding when to resume semaglutide after surgery is equally important for maintaining progress.
Pregnancy and breastfeeding
Pure GLP-1 agonists are contraindicated during pregnancy. They should be discontinued at least 2 months before a planned pregnancy (longer for semaglutide given its extended half-life). The question of GLP-1 use while breastfeeding is less clear cut, but current guidance generally advises against it due to insufficient safety data in nursing mothers.
Sleep
Some researchers report sleep disruption during GLP-1 therapy. Whether semaglutide causes insomnia is a question of active investigation. Low blood sugar during the night, altered eating patterns, and GI discomfort may all contribute to sleep disturbance. Adjusting injection timing and ensuring adequate evening nutrition can help.
The future of pure GLP-1 agonists
Pure GLP-1 agonists are not going away. Despite the emergence of dual and triple agonists, the pure GLP-1 class continues to evolve and expand.
Oral semaglutide formulations are improving. Higher-dose oral semaglutide tablets (25 mg and 50 mg) have shown weight loss approaching injectable levels in clinical trials. This could eliminate the injection barrier entirely for pure GLP-1 therapy.
New delivery systems are emerging. GLP-1 patches offer transdermal delivery. Sublingual semaglutide provides another needle-free option. These innovations make pure GLP-1 therapy more accessible and comfortable.
AI-driven peptide design is creating next-generation GLP-1 molecules with optimized receptor binding, longer duration of action, and reduced side effect profiles. Research groups have generated thousands of de novo GLP-1 receptor agonist candidates using computational approaches, with dozens showing promise in preclinical screening.
The expanding landscape of clinical applications, from neurodegeneration to addiction to autoimmune disease, ensures that pure GLP-1 agonists will remain a cornerstone of peptide research for years to come. SeekPeptides continues to track these developments and provides members with updated protocols, research summaries, and practical guidance as the field evolves.
Common mistakes with pure GLP-1 protocols
Years of community experience have identified recurring mistakes that undermine results. Avoiding these accelerates progress and reduces frustration.
Titrating too fast. Jumping doses to reach therapeutic levels quickly increases side effects and dropout rates. The titration schedule exists because GLP-1 receptors need time to adapt. Respect the schedule.
Neglecting protein. This cannot be overstated. Inadequate protein during GLP-1 mediated weight loss leads to excessive muscle loss, metabolic slowdown, and the "skinny fat" outcome nobody wants. Track protein intake daily.
Ignoring hydration. Reduced food intake means reduced water intake from food sources. Many side effects attributed to the medication, headaches, fatigue, constipation, dizziness, are actually dehydration. Aim for 2.5-3.5 liters of water daily.
Stopping at the first plateau. Weight loss on pure GLP-1 agonists is not linear. Plateaus are normal and expected, typically occurring every 4-8 weeks. They do not mean the medication stopped working. They mean the body is recalibrating. The comprehensive semaglutide plateau guide explains why this happens and what to do about it.
Not addressing sleep and stress. Cortisol from chronic stress and sleep deprivation directly counteracts the metabolic benefits of GLP-1 agonism. Researchers who optimize sleep hygiene and stress management consistently achieve better outcomes.
Expecting pure GLP-1 to match dual agonist numbers. Comparing your semaglutide results to tirzepatide results is comparing different pharmacological classes. A pure GLP-1 agonist producing 12-15% weight loss is performing exactly as designed. That is not a failure. That is a different tool for a different situation.
Choosing your pure GLP-1 agonist
With six approved options, the choice can feel overwhelming. Here is a framework for decision-making.
If your priority is... | Consider... | Why |
|---|---|---|
Maximum weight loss | Semaglutide 2.4 mg weekly | Highest efficacy data among pure GLP-1 agonists |
Cardiovascular protection | Liraglutide or Semaglutide | Strongest MACE outcome trial data |
No needles | Oral semaglutide | Only oral pure GLP-1 option |
Weekly convenience | Semaglutide or Dulaglutide | Once-weekly dosing |
Fine dose control | Liraglutide | Daily dosing allows precise adjustments |
Primary glucose control | Dulaglutide | REWIND trial, good tolerability |
Cost sensitivity | Compounded semaglutide | Lower cost than brand-name options |
Combination with insulin | Lixisenatide (as Soliqua) | Designed for basal insulin combination |
For most researchers starting GLP-1 therapy today, semaglutide is the default recommendation. Its combination of efficacy, convenience (weekly dosing), oral option availability, cardiovascular outcome data, and extensive real-world experience makes it the most versatile pure GLP-1 agonist available.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact decisions. The peptide calculator tools, stacking calculator, and detailed protocol guides provide the practical support that transforms knowledge into results.
Frequently asked questions
What is the difference between pure GLP-1 and a dual agonist?
A pure GLP-1 agonist activates only the GLP-1 receptor. A dual agonist like tirzepatide activates both GLP-1 and GIP receptors. Pure GLP-1 agonists include semaglutide, liraglutide, dulaglutide, and exenatide. The semaglutide versus tirzepatide comparison covers the key differences in efficacy, side effects, and clinical evidence.
Is semaglutide a pure GLP-1 agonist?
Yes. Semaglutide is the most potent pure GLP-1 receptor agonist currently available. It exclusively targets the GLP-1 receptor without activating GIP or glucagon receptors. It is available as injectable semaglutide (Ozempic, Wegovy) and oral semaglutide (Rybelsus).
Which pure GLP-1 agonist causes the most weight loss?
Semaglutide 2.4 mg weekly produces the highest average weight loss among pure GLP-1 agonists, approximately 15% of body weight over 68 weeks. Liraglutide is second at roughly 8%. Check the week-by-week results timeline for detailed expectations.
Can I switch from a pure GLP-1 to a dual agonist?
Yes. Switching from semaglutide to tirzepatide is a common transition for researchers seeking additional weight loss. The conversion chart provides dose equivalency guidance. Most physicians recommend a brief washout or direct dose conversion based on the current pure GLP-1 dose.
How long do pure GLP-1 side effects last?
Most GI side effects (nausea, vomiting, diarrhea) improve within 4-8 weeks at a stable dose. They tend to recur briefly with each dose escalation during titration. When side effects appear and how long they persist varies by individual, but most researchers find them manageable by the maintenance dose phase.
Do pure GLP-1 agonists work for people without diabetes?
Absolutely. The STEP trials and SELECT trial enrolled participants without diabetes and demonstrated significant weight loss and cardiovascular benefits. Semaglutide (as Wegovy) and liraglutide (as Saxenda) are FDA-approved specifically for weight management regardless of diabetes status. The BMI requirements for GLP-1 therapy apply to non-diabetic individuals as well.
Are there natural ways to boost GLP-1?
The body produces GLP-1 naturally in response to food, particularly protein, fiber, and healthy fats. Certain probiotics may enhance GLP-1 secretion, which is why GLP-1 postbiotic supplements and gut response supplements have gained popularity. However, these natural approaches produce dramatically lower GLP-1 levels compared to pharmaceutical agonists.
Can I take other medications with a pure GLP-1 agonist?
Most medications can be used alongside pure GLP-1 agonists, but timing may need adjustment due to delayed gastric emptying. Oral medications taken concurrently may have altered absorption. Phentermine and semaglutide combinations are increasingly common. Researchers using other peptide stacks for weight loss should evaluate interactions carefully.
External resources
For researchers serious about mastering pure GLP-1 protocols, SeekPeptides members access detailed dosing guides, safety databases, nutritional companion guides, and a community of experienced researchers who share real-world insights that clinical trials never capture.
In case I do not see you, good afternoon, good evening, and good night. May your receptors stay sensitive, your peptides stay potent, and your protocols stay evidence-based.