Mar 23, 2026

Everything you have read about Toujeo being a GLP-1 is wrong. And if you are making treatment decisions based on that misunderstanding, you could be heading down a path that does not match your goals, your body, or your medical needs. The confusion is understandable. Both Toujeo and GLP-1 medications are injectable. Both are prescribed for diabetes. Both involve subcutaneous needles, dosing schedules, and pharmacy conversations that sound remarkably similar. But the similarities end there.
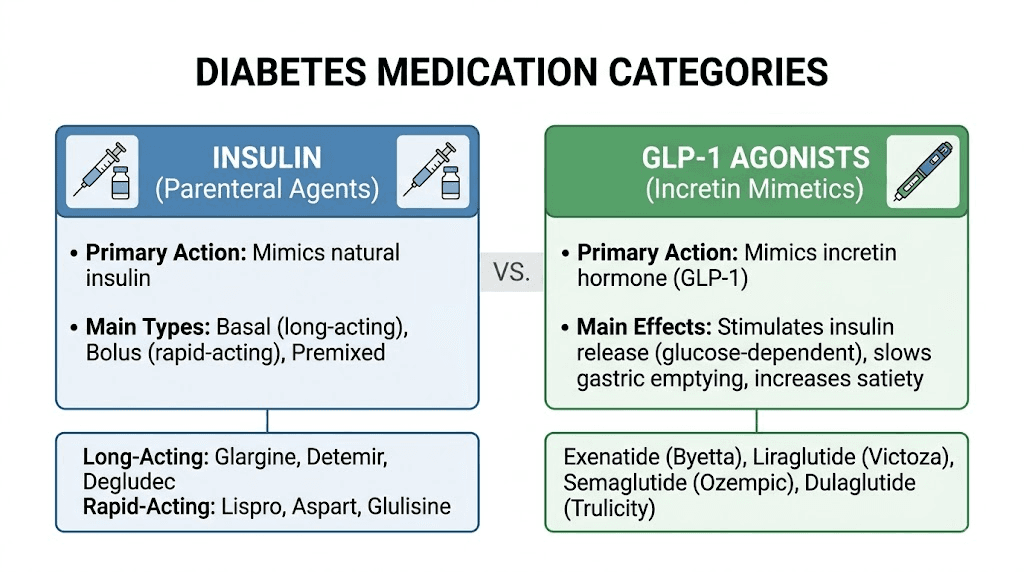
Toujeo is insulin. Specifically, it is insulin glargine at a concentration of 300 units per milliliter, manufactured by Sanofi. GLP-1 receptor agonists, like semaglutide and other GLP-1 medications, work through an entirely different mechanism. They mimic a gut hormone. Toujeo replaces a pancreatic hormone. These are fundamentally different approaches to blood sugar management, with different side effect profiles, different effects on body weight, and different implications for long-term health. Confusing the two is not just a semantic mistake. It is a clinical one.
This guide breaks down exactly what Toujeo is, what GLP-1 medications are, why the confusion happens so frequently, and what the differences mean for anyone navigating diabetes treatment or weight loss with peptide-based therapies.
Whether you are a patient trying to understand your prescription, a researcher comparing drug classes, or someone exploring GLP-1 options for fat loss, the distinction between insulin and incretin mimetics matters more than most people realize. SeekPeptides has put together this comprehensive resource to clear up the confusion once and for all.
What Toujeo actually is
Toujeo is a brand name for insulin glargine injection at 300 units per milliliter, commonly written as U-300. It belongs to the class of long-acting basal insulins. Not GLP-1 agonists. Not incretin mimetics. Not weight loss medications. Insulin.
The active ingredient in Toujeo is identical to what you find in Lantus, another well-known insulin product. Both contain insulin glargine. The difference is concentration. Lantus delivers 100 units per milliliter (U-100), while Toujeo packs three times that amount into the same volume. This higher concentration means a smaller injection volume for the same dose, and it also changes how the insulin absorbs into the body, creating a flatter, more prolonged activity profile that can last up to 36 hours.
Sanofi developed Toujeo as the next generation of their insulin glargine line. It received FDA approval in February 2015 for adults with both Type 1 and Type 2 diabetes. That approval for Type 1 diabetes is important because it is something no GLP-1 medication can claim. GLP-1 agonists require functioning beta cells to work. In Type 1 diabetes, those cells have been destroyed by autoimmune attack. Insulin replacement is not optional for these patients. It is survival.
How insulin glargine works in the body
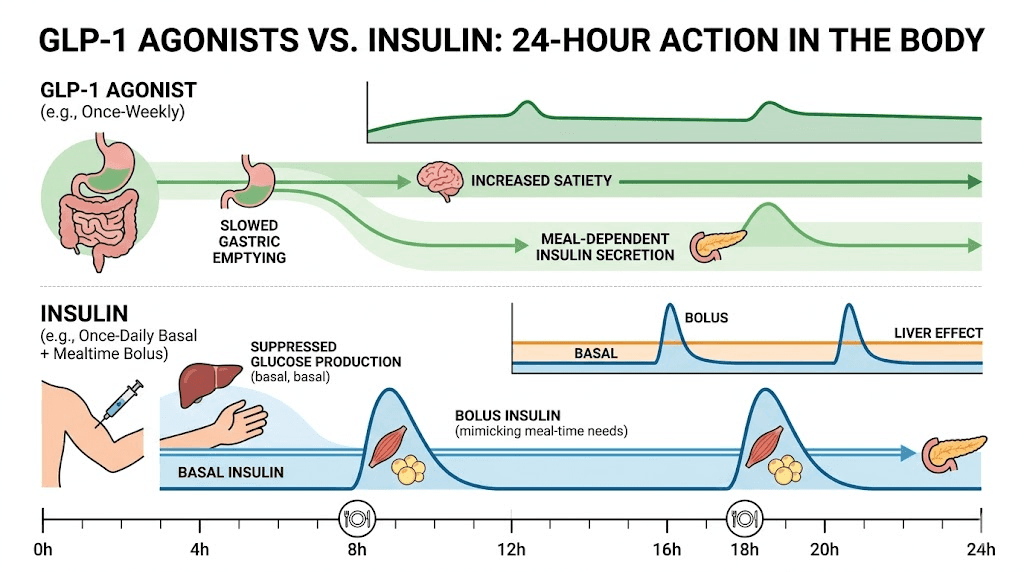
When you inject Toujeo, the insulin glargine forms microprecipitates under the skin. These tiny clusters slowly dissolve over the following hours, releasing insulin into the bloodstream at a steady, predictable rate. Think of it as a slow-release reservoir sitting just beneath the skin surface.
Once the insulin enters the bloodstream, it does what all insulin does. It binds to insulin receptors on cell surfaces throughout the body. This binding triggers cells to open glucose transporters, essentially unlocking the door that lets blood sugar enter cells for energy. Simultaneously, insulin tells the liver to stop producing new glucose. The combined effect is a gradual, sustained lowering of blood sugar levels.
This is fundamentally different from how semaglutide and other GLP-1 drugs work. Toujeo does not interact with GLP-1 receptors. It does not suppress appetite. It does not slow gastric emptying. It does not trigger the brain mechanisms that make you feel full. It simply provides the insulin your body either cannot make or cannot make enough of.
The onset of action for Toujeo is approximately six hours after injection. When you first start taking it, the medication needs up to five days to reach steady-state levels in your blood. After that, a single daily injection maintains relatively flat insulin coverage for 24 to 36 hours. Most physicians instruct patients to inject at the same time every day, typically in the morning or at bedtime, depending on the treatment protocol.
Who takes Toujeo
Toujeo serves a specific patient population. Adults with Type 1 diabetes who need basal insulin replacement. Adults with Type 2 diabetes who have not achieved adequate blood sugar control through diet, exercise, oral medications, or other non-insulin therapies. Patients who are already on basal insulin like Lantus and want a more concentrated option with potentially fewer injection-site reactions and slightly lower hypoglycemia risk.
It is not prescribed for weight loss. It is not prescribed for appetite suppression. In fact, one of the most common side effects of all insulin therapy, including Toujeo, is weight gain. This stands in stark contrast to the average weight loss seen with GLP-1 medications, which often ranges from 10 to 20 percent of body weight depending on the specific drug and dose.
What GLP-1 medications actually are
GLP-1 stands for glucagon-like peptide-1. It is a hormone your gut naturally produces when you eat food. This hormone does several important things simultaneously. It stimulates insulin release from the pancreas (but only when blood sugar is elevated). It suppresses glucagon, the hormone that tells your liver to dump glucose into the bloodstream. It slows gastric emptying, keeping food in your stomach longer. And it acts on appetite centers in the brain, reducing hunger and promoting satiety.
GLP-1 receptor agonists are medications that mimic this natural hormone. They bind to the same receptors that natural GLP-1 activates, but they last much longer in the body. Natural GLP-1 breaks down within minutes. Medications like semaglutide and tirzepatide can persist for days, providing sustained activation of these pathways.
The result is a multi-pronged approach to blood sugar control and weight management. Lower blood sugar through enhanced insulin secretion. Reduced liver glucose output through glucagon suppression. Slower nutrient absorption through delayed gastric emptying. Decreased food intake through central appetite suppression. All from a single medication class.
The GLP-1 medication landscape
The GLP-1 market has exploded in recent years. Multiple medications now exist, each with slightly different properties. Semaglutide is perhaps the most well-known, marketed as Ozempic for diabetes and Wegovy for weight management. It targets only GLP-1 receptors and is administered once weekly via injection, though oral formulations also exist.
Tirzepatide takes things a step further. Marketed as Mounjaro for diabetes and Zepbound for weight loss, tirzepatide activates both GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptors. This dual mechanism appears to produce even greater effects on blood sugar and body weight than GLP-1-only medications. The SURPASS-2 trial demonstrated that tirzepatide was superior to semaglutide in reducing both HbA1c and body weight.
Retatrutide represents the next frontier, targeting three receptors simultaneously: GLP-1, GIP, and glucagon. Still in clinical trials, this triple agonist has shown remarkable results in early studies. Then there are other established GLP-1 agonists like liraglutide (Victoza/Saxenda), dulaglutide (Trulicity), and exenatide (Byetta/Bydureon), each with their own dosing schedules and clinical profiles.
None of these medications are insulin. None of them directly provide the insulin hormone. They all work by leveraging the body's own incretin system to improve glucose regulation, and most of them produce significant weight loss as a beneficial side effect, or in some cases, as the primary intended effect.
Why people confuse Toujeo with GLP-1 medications
The confusion between Toujeo and GLP-1 drugs is not random. Several legitimate factors drive it, and understanding them helps clear up the misunderstanding.
Both are injectable. Both come in pen devices. Both are prescribed by endocrinologists. Both are used for diabetes management. Both involve subcutaneous injection into the abdomen, thigh, or upper arm. If you are standing in a pharmacy picking up your prescription, the physical similarity between a Toujeo SoloStar pen and an Ozempic pen is striking. Same general shape. Same injection technique. Same storage requirements.
The medical conversations overlap too. Your doctor discusses blood sugar targets with both. Insurance prior authorization processes look similar. Even the pharmaceutical advertising, with images of healthy people living active lives, uses nearly identical visual language for both drug classes.
Then there is the naming confusion. Drug names in the diabetes space are notoriously confusing. Toujeo sounds vaguely pharmaceutical. So does Ozempic. So does Mounjaro. Without medical training, there is no way to tell from the name alone which class a drug belongs to. Similar confusion exists around other diabetes medications like Qsymia, which is also not a GLP-1.
The combination product factor
Adding to the confusion, Sanofi (the same company that makes Toujeo) also manufactures Soliqua 100/33. This is a combination product that contains both insulin glargine and lixisenatide, which is a GLP-1 receptor agonist. So the same pharmaceutical company makes a pure insulin product (Toujeo), a combination insulin-plus-GLP-1 product (Soliqua), and markets them through similar channels to similar patient populations.
Similarly, Novo Nordisk makes both insulin degludec (Tresiba) and semaglutide (Ozempic), plus a combination product called Xultophy that pairs insulin degludec with liraglutide. When the same companies produce both insulins and GLP-1 drugs, and sometimes combine them into single products, it is easy to see why patients get confused about which is which.
The clinical evolution of diabetes treatment also plays a role. A decade ago, insulin was the standard next step when oral medications failed. Today, GLP-1 agonists have largely replaced insulin as the preferred injectable option for Type 2 diabetes. Many patients who expected to be prescribed insulin are now prescribed GLP-1 drugs instead, further blurring the line between the two categories in the public consciousness.
The fundamental differences between insulin and GLP-1 agonists
Understanding the differences between these drug classes is not academic. It has real implications for treatment outcomes, side effects, quality of life, and long-term health. Here is a detailed breakdown.
Mechanism of action
Insulin glargine (Toujeo) works by directly providing exogenous insulin to the body. It bypasses the pancreas entirely. The insulin you inject is the insulin your cells use. No intermediary steps. No dependence on remaining beta cell function. This is hormone replacement therapy in the most literal sense.
GLP-1 agonists work indirectly. They activate receptors that trigger a cascade of downstream effects, one of which is increased insulin release from your own pancreatic beta cells. But that insulin release is glucose-dependent, meaning it ramps up when blood sugar is high and backs off when blood sugar is normal. This built-in safety mechanism is why GLP-1 medications carry a much lower risk of hypoglycemia compared to insulin.
The distinction matters clinically. If you have Type 1 diabetes or advanced Type 2 diabetes with severely depleted beta cells, GLP-1 agonists alone will not adequately control your blood sugar. You need the direct insulin replacement that Toujeo provides. On the other hand, if you have earlier-stage Type 2 diabetes with preserved beta cell function, GLP-1 agonists can leverage your remaining insulin production while offering the bonus of appetite suppression and weight loss.
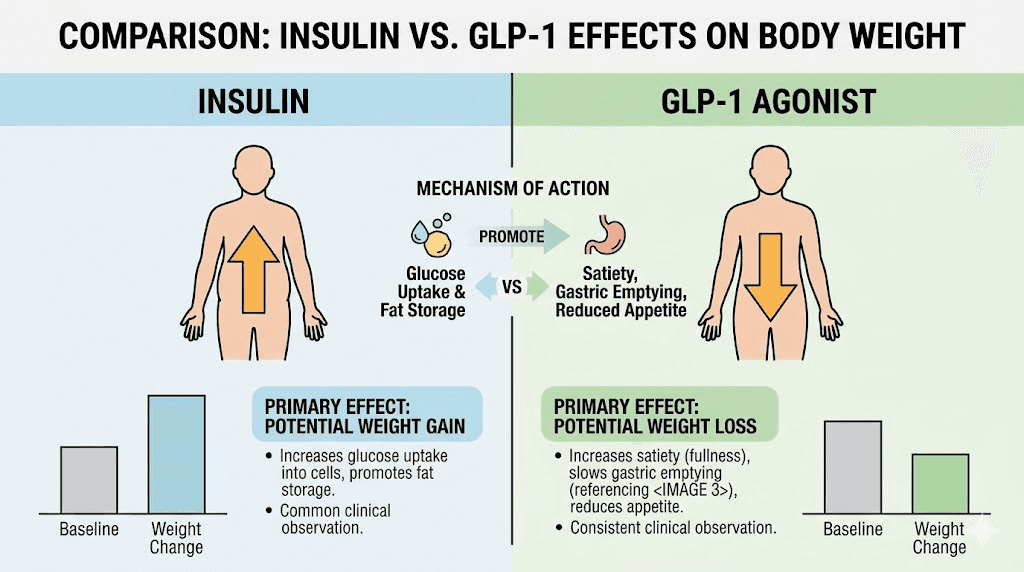
Effect on body weight
This is perhaps the most dramatic difference. Insulin therapy, including Toujeo, typically causes weight gain. The mechanism is straightforward. Insulin promotes glucose uptake into cells, including fat cells. It inhibits lipolysis (fat breakdown). And by lowering blood sugar more effectively, it reduces the amount of glucose lost in urine, meaning more calories are retained in the body. Most patients on basal insulin gain 2 to 4 kilograms in the first year of treatment.
GLP-1 medications produce the opposite effect. Through appetite suppression, delayed gastric emptying, and central nervous system effects on food reward pathways, these drugs cause significant weight loss. In clinical trials, semaglutide at the 2.4mg weekly dose (Wegovy) produced average weight loss of approximately 15 percent of body weight. Tirzepatide achieved even greater results, with some trial participants losing over 20 percent of their starting weight.
For someone asking whether Toujeo is a GLP-1, the weight implications alone should clarify the distinction. These drugs push body weight in opposite directions.
Hypoglycemia risk
Hypoglycemia, dangerously low blood sugar, is the most feared acute complication of insulin therapy. Because Toujeo provides a constant baseline of insulin regardless of whether you have eaten, skipping meals, exercising more than usual, or miscalculating your dose can all trigger hypoglycemia. Symptoms range from shakiness and sweating to confusion, seizures, and in severe cases, death.
Toujeo does carry a somewhat lower hypoglycemia risk compared to Lantus, thanks to its more gradual absorption profile. But the risk is still present and significant. It is an inherent consequence of providing exogenous insulin that works independently of blood sugar levels.
GLP-1 agonists have a fundamentally lower hypoglycemia risk. Because they stimulate insulin release in a glucose-dependent manner, the effect naturally diminishes as blood sugar drops toward normal levels. The insulin-stimulating effect of GLP-1 drugs essentially has a built-in off switch that insulin injections lack. Hypoglycemia with GLP-1 monotherapy is rare. The risk increases when GLP-1 drugs are combined with insulin or sulfonylureas, which is why dose adjustments are necessary in combination therapy.
Gastrointestinal effects
Toujeo has minimal gastrointestinal side effects. Injection site reactions, weight gain, and hypoglycemia dominate the side effect profile. Most patients tolerate insulin therapy without significant GI complaints.
GLP-1 agonists are a different story. By slowing gastric emptying and affecting gut motility, these medications commonly cause nausea, vomiting, diarrhea, and constipation. Up to 40 percent of patients experience nausea in the early weeks of treatment, particularly during dose escalation. Acid reflux, bloating, and sulfur burps are additional complaints. Most of these symptoms improve over time as the body adapts, but they can be significant enough to cause discontinuation in some patients.
The GI effects of GLP-1 drugs are actually part of their mechanism of action, not just unpleasant side effects. The slower gastric emptying contributes to the feeling of fullness and reduced food intake that drives weight loss. Managing these effects through dietary modifications and gradual dose titration is a standard part of GLP-1 therapy.
Detailed comparison table
Feature | Toujeo (insulin glargine U-300) | GLP-1 receptor agonists |
|---|---|---|
Drug class | Long-acting basal insulin | Incretin mimetic |
Active ingredient example | Insulin glargine | Semaglutide, tirzepatide, liraglutide |
Primary mechanism | Provides exogenous insulin directly | Activates GLP-1 receptors to enhance natural insulin release |
Type 1 diabetes | Yes (approved) | No (requires functioning beta cells) |
Type 2 diabetes | Yes (approved) | Yes (approved) |
Weight effect | Weight gain (2-4 kg typical) | Weight loss (10-20% body weight) |
Hypoglycemia risk | Moderate to high | Low (unless combined with insulin) |
GI side effects | Minimal | Common (nausea, vomiting, diarrhea) |
Appetite effect | May increase appetite | Significantly suppresses appetite |
Dosing frequency | Once daily | Once daily to once weekly (varies by drug) |
Cardiovascular benefit | Neutral | Demonstrated (especially semaglutide) |
Kidney protection | No direct evidence | Yes (semaglutide trials) |
Duration of action | Up to 36 hours | 24 hours to 7 days (varies by drug) |
Injection site | Abdomen, thigh, upper arm | Abdomen, thigh, upper arm |
Can be combined | Yes (with GLP-1 agonists) | Yes (with basal insulin) |
This table makes one thing clear. Toujeo and GLP-1 medications sit in completely different categories despite their surface-level similarities. The comparison is useful for understanding treatment options, but calling Toujeo a GLP-1 would be like calling aspirin an antibiotic because both come in pill form.
How Toujeo fits into the diabetes treatment algorithm
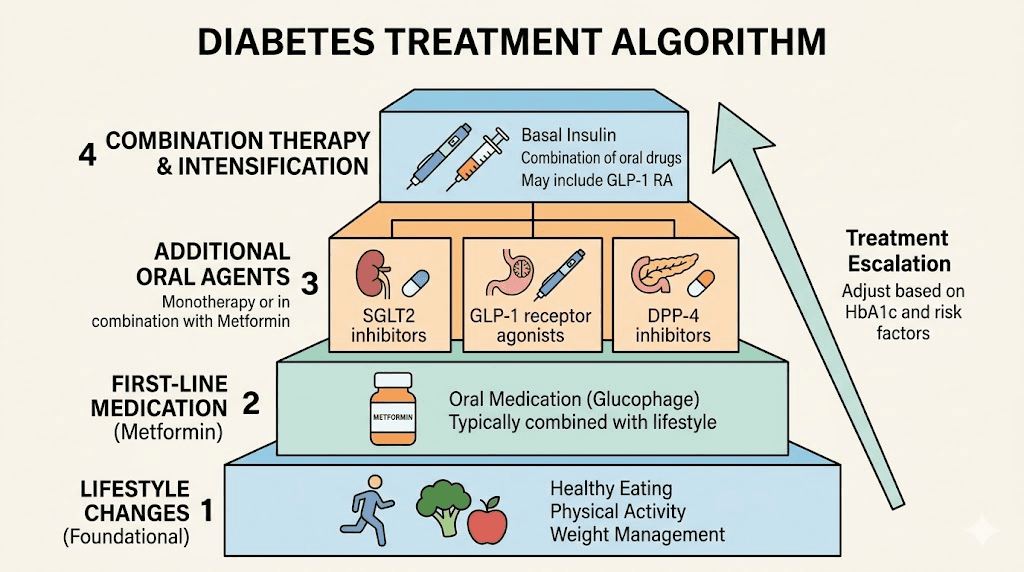
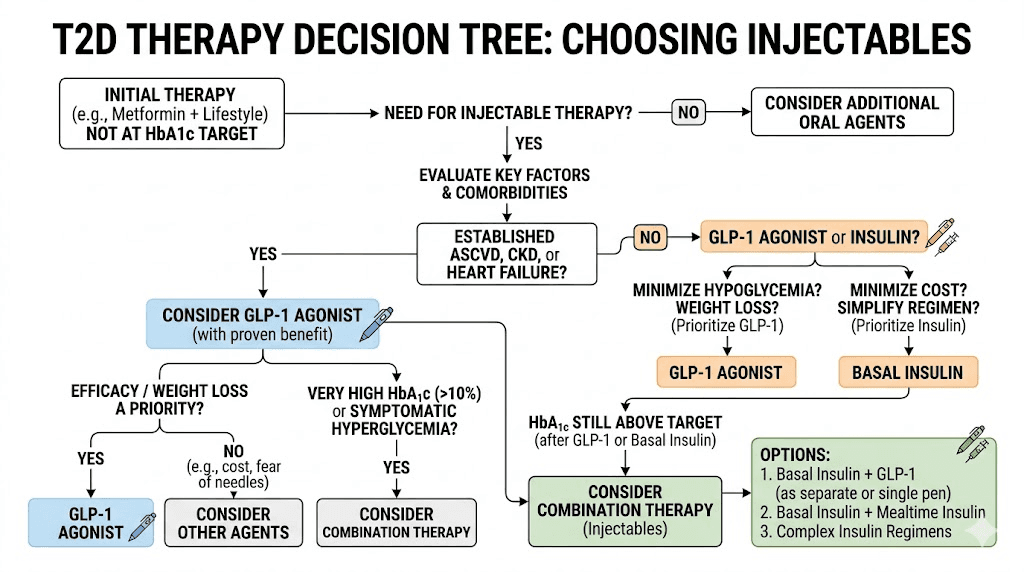
Modern diabetes treatment follows a stepwise approach. Understanding where Toujeo and GLP-1 drugs fit in this progression clarifies why both exist and why they serve different purposes.
Step one: lifestyle and metformin
For most patients with newly diagnosed Type 2 diabetes, the first-line treatment is lifestyle modification (diet, exercise, weight management) combined with metformin, an oral medication that reduces liver glucose production and improves insulin sensitivity. Metformin has been the backbone of Type 2 diabetes treatment for decades, and it remains the recommended starting point in most guidelines.
At this stage, neither Toujeo nor GLP-1 medications are typically involved. The focus is on dietary changes, physical activity, and oral medication. However, for patients with very high blood sugar at diagnosis (HbA1c above 10 percent) or symptoms of hyperglycemia, insulin may be started right away, bypassing the usual stepwise approach.
Step two: adding injectable therapy
When metformin alone (or metformin plus other oral agents) fails to achieve blood sugar targets, the next decision is whether to add a GLP-1 agonist or insulin. This is where the landscape has shifted dramatically in recent years.
Current guidelines from the American Diabetes Association now favor GLP-1 agonists over insulin as the first injectable therapy for most patients with Type 2 diabetes. The reasoning is compelling. GLP-1 drugs produce comparable blood sugar reduction with the added benefits of weight loss, lower hypoglycemia risk, and cardiovascular protection. Insulin remains the preferred choice only when GLP-1 drugs are contraindicated, unaffordable, or insufficient alone, or when the patient has Type 1 diabetes.
Toujeo specifically enters the picture when a patient needs basal insulin and either has already tried and failed GLP-1 therapy, has a contraindication to GLP-1 drugs, has Type 1 diabetes, or has very advanced Type 2 diabetes with negligible beta cell function. It is not the first-choice injectable for uncomplicated Type 2 diabetes anymore, and understanding this shift helps explain why the GLP-1 question comes up so often.
Step three: combination and intensification
For patients who do not reach their targets on either medication alone, combining basal insulin with a GLP-1 agonist is an increasingly popular strategy. The combination makes pharmacological sense. Basal insulin covers the fasting glucose component. The GLP-1 agonist handles post-meal glucose spikes and provides appetite suppression that counteracts insulin-related weight gain.
Pre-mixed combination products like Soliqua (insulin glargine plus lixisenatide) and Xultophy (insulin degludec plus liraglutide) simplify this approach into a single injection. Some researchers have explored concurrent use of different GLP-1 agents, though this is less established in clinical practice.
Side effects of Toujeo versus GLP-1 medications
The side effect profiles of these two drug classes are so different that comparing them further reinforces how distinct they are. Understanding what to expect from each helps patients and researchers make informed decisions.
Toujeo side effects
The most significant side effect of Toujeo is hypoglycemia. Low blood sugar can occur at any time, particularly if doses are not properly adjusted for food intake, exercise, or changes in other medications. Severe hypoglycemia can cause confusion, loss of consciousness, seizures, and can be life-threatening. Patients on Toujeo must monitor blood sugar regularly and know how to recognize and treat low glucose episodes.
Weight gain is the second major side effect. As discussed earlier, insulin promotes fat storage and calorie retention. Most patients on Toujeo can expect to gain some weight, which can be frustrating for those already struggling with obesity-related Type 2 diabetes. This weight gain effect is the opposite of what patients experience on tirzepatide or semaglutide.
Injection site reactions, including pain, redness, itching, and swelling at the injection point, occur in some patients.
Over time, repeated injections in the same area can cause lipodystrophy, a thickening or thinning of fat tissue that affects insulin absorption. Rotating injection sites helps prevent this.
Less common but serious side effects include severe allergic reactions, hypokalemia (low potassium), fluid retention, and heart failure in patients with pre-existing cardiac conditions. Toujeo can also interact with numerous other medications, making careful pharmacological management essential.
GLP-1 agonist side effects
The GLP-1 side effect profile looks completely different. Gastrointestinal symptoms dominate. Nausea is the most common complaint, affecting 20 to 40 percent of patients during initial dosing. Vomiting, diarrhea, and constipation are also frequent. Burping and indigestion round out the GI picture. These symptoms are usually worst during the first few weeks and during each dose increase, then gradually improve as the body adapts.
Fatigue is another commonly reported side effect of GLP-1 medications, particularly in the early stages of treatment. This may be related to reduced caloric intake, changes in blood sugar patterns, or direct effects on the central nervous system. Headaches and dizziness also appear on the side effect list, though they tend to be mild and transient.
Hair loss has been reported by some patients on GLP-1 therapy, likely related to rapid weight loss and nutritional changes rather than a direct drug effect. This is similar to telogen effluvium seen with any significant weight loss, regardless of the method used.
More serious concerns include a possible increased risk of pancreatitis (inflammation of the pancreas) and, in animal studies, thyroid C-cell tumors. The thyroid tumor risk has not been confirmed in humans, but GLP-1 drugs carry a boxed warning about medullary thyroid carcinoma risk and are contraindicated in patients with a personal or family history of this cancer type. Gallbladder problems, including gallstones, have also been associated with GLP-1 therapy, likely related to rapid weight loss and changes in bile composition.
Cardiovascular and organ protection
One of the most significant recent developments in diabetes pharmacology has been the recognition that GLP-1 agonists provide benefits beyond blood sugar control. This is another major differentiator from insulin.
GLP-1 cardiovascular outcomes
Multiple large-scale cardiovascular outcomes trials have demonstrated that GLP-1 agonists reduce the risk of major adverse cardiovascular events, including heart attack, stroke, and cardiovascular death. Semaglutide, in the SUSTAIN-6 and SELECT trials, showed particularly impressive cardiovascular and metabolic benefits. Liraglutide, in the LEADER trial, demonstrated a 13 percent reduction in cardiovascular death.
These are not just blood-sugar effects. GLP-1 drugs appear to have direct protective effects on the cardiovascular system, reducing inflammation, improving endothelial function, and potentially stabilizing atherosclerotic plaques. For patients with Type 2 diabetes who also have or are at high risk for cardiovascular disease, GLP-1 agonists offer a dual benefit that insulin cannot match.
Kidney protection
The FLOW trial with semaglutide demonstrated significant kidney protective effects in patients with Type 2 diabetes and chronic kidney disease. GLP-1 agonists reduce albuminuria (protein in the urine, a marker of kidney damage) and appear to slow the progression of diabetic kidney disease. This is an actively evolving area of research, but the early results are promising enough to change clinical practice.
Insulin cardiovascular profile
Insulin, including Toujeo, has a cardiovascular profile best described as neutral. The ORIGIN trial showed that insulin glargine neither increased nor decreased cardiovascular events compared to standard care. It does not provide the active cardioprotection seen with GLP-1 drugs. In patients with heart failure, insulin can actually worsen fluid retention and edema, making careful monitoring essential.
This cardiovascular distinction is clinically significant. For a patient with Type 2 diabetes and established heart disease, choosing a GLP-1 agonist over insulin could provide meaningful protection against future cardiovascular events, above and beyond blood sugar control.
The complete list of GLP-1 and related medications
To further clarify what is and is not a GLP-1, here is a comprehensive rundown of the major players in the incretin-based therapy space.
Pure GLP-1 receptor agonists
Semaglutide is the most prescribed GLP-1 medication worldwide. Available as Ozempic (diabetes, injectable), Wegovy (weight management, injectable), and Rybelsus (diabetes, oral). It binds exclusively to GLP-1 receptors and has the broadest evidence base for cardiovascular and kidney protection. Dosing starts low and escalates gradually.
Liraglutide, marketed as Victoza for diabetes and Saxenda for weight management, was one of the first successful GLP-1 drugs. It requires daily injection, unlike the weekly dosing of semaglutide. Despite both being GLP-1 agonists, liraglutide and semaglutide differ significantly in potency, dosing frequency, and clinical outcomes.
Dulaglutide (Trulicity) is a once-weekly GLP-1 agonist commonly prescribed for Type 2 diabetes. Exenatide comes in two forms: Byetta (twice daily) and Bydureon (once weekly). These older GLP-1 drugs remain in use but have been largely overtaken by semaglutide and tirzepatide in new prescriptions.
Dual and triple agonists
Tirzepatide is the first dual GIP/GLP-1 receptor agonist. It activates both GLP-1 and GIP receptors, producing effects that exceed those of pure GLP-1 drugs in head-to-head trials. Marketed as Mounjaro for diabetes and Zepbound for weight management, tirzepatide has rapidly become one of the most prescribed medications in the diabetes and obesity space.
Retatrutide is a triple agonist targeting GLP-1, GIP, and glucagon receptors simultaneously. Still in Phase 3 clinical trials, early results show unprecedented weight loss, with some participants losing over 24 percent of body weight. Survodutide is another dual agonist in development, targeting GLP-1 and glucagon receptors. CagriSema combines semaglutide with cagrilintide, an amylin analog, for potentially enhanced effects.
Orforglipron is a next-generation oral GLP-1 agonist that could eventually make daily pills as effective as weekly injections. Compared to tirzepatide, orforglipron offers the convenience of oral administration but may not achieve the same degree of efficacy.
What is NOT a GLP-1
Toujeo is not a GLP-1. But it is not alone in being commonly confused with this drug class. Other medications that are frequently mistaken for GLP-1 agonists include:
Phentermine is a sympathomimetic amine appetite suppressant, not a GLP-1. It works through norepinephrine release, not incretin pathways. Qsymia is a combination of phentermine and topiramate, also not a GLP-1. Metformin is a biguanide that works on the liver, not a GLP-1. Contrave combines bupropion and naltrexone, working on brain reward pathways rather than GLP-1 receptors. All insulins, including Toujeo, Lantus, Tresiba, Levemir, Humalog, and NovoLog, are insulin products, not GLP-1 medications.
Can you use Toujeo and GLP-1 medications together?
Yes. Not only can you use them together, but the combination is becoming one of the most effective approaches in modern diabetes management. Understanding how and why they complement each other underscores the fact that they are distinct drug classes.
The rationale for combination therapy
Basal insulin like Toujeo addresses fasting blood sugar by providing a steady background level of insulin. But it does little for the blood sugar spikes that occur after meals. GLP-1 agonists, through their effects on insulin secretion, glucagon suppression, and gastric emptying, are particularly effective at controlling post-meal glucose excursions. Combining the two creates comprehensive 24-hour glucose coverage.
The weight benefit is also complementary. GLP-1-induced weight loss can offset insulin-related weight gain. Some studies show that adding a GLP-1 agonist to basal insulin results in better blood sugar control with neutral weight effects, or even mild weight loss, compared to intensifying insulin alone.
Hypoglycemia risk with the combination is lower than with insulin intensification strategies like adding mealtime insulin. The glucose-dependent insulin secretion of GLP-1 drugs provides a natural safety buffer. However, when starting a GLP-1 agonist in someone already on basal insulin, the insulin dose often needs to be reduced by 10 to 20 percent to prevent hypoglycemia.
Available combination products
Soliqua 100/33 combines insulin glargine 100 units/mL with lixisenatide 33 mcg/mL in a single pen. It is administered once daily. The insulin glargine in Soliqua is the same active ingredient as in both Lantus and Toujeo, though at the U-100 concentration rather than Toujeo U-300 concentration.
Xultophy 100/3.6 combines insulin degludec 100 units/mL with liraglutide 3.6 mg/mL. This product uses a different basal insulin (degludec rather than glargine) paired with a GLP-1 agonist.
For patients who need both Toujeo specifically (the U-300 concentration) and a GLP-1 agonist, separate injections are currently the only option, as there is no combination product using the U-300 concentration. Many patients take their GLP-1 medication on one day and manage their insulin separately.
Cost and insurance considerations
The financial landscape for these medications is complex and worth discussing, as it often influences treatment decisions as much as clinical factors.
Toujeo pricing
Toujeo, as a mature insulin product, has relatively established pricing, though insulin costs in the United States remain controversial. List prices for Toujeo pens run several hundred dollars per month without insurance. However, most insurance plans cover basal insulin, and Sanofi offers patient assistance programs. Generic biosimilar insulin glargine products have also begun to enter the market, potentially reducing costs for patients who need this medication class.
GLP-1 pricing
GLP-1 medications are significantly more expensive than insulin. Monthly list prices for brand-name GLP-1 drugs like Ozempic and Mounjaro exceed $1,000 in many markets. Insurance coverage varies widely, with some plans requiring prior authorization, step therapy (trying cheaper medications first), or denying coverage altogether.
Compounded versions of semaglutide and tirzepatide have emerged as lower-cost alternatives through compounding pharmacies, though their regulatory status has been evolving. FSA and HSA eligibility for these medications depends on the specific indication and insurance plan.
For patients trying to navigate insurance coverage for GLP-1 medications or understanding what their plan covers, the process can be frustrating. Knowing how to talk to your doctor about GLP-1 options can make a significant difference in accessing these treatments.
Toujeo versus other insulins
While this article focuses on the Toujeo versus GLP-1 distinction, it helps to understand where Toujeo sits within the insulin landscape itself.
Toujeo versus Lantus
Both contain insulin glargine. Toujeo is U-300 (300 units/mL) while Lantus is U-100 (100 units/mL). The higher concentration in Toujeo means smaller injection volumes and a more gradual, extended absorption profile. Clinical trials have shown that Toujeo produces slightly fewer hypoglycemic events, particularly nocturnal hypoglycemia, compared to Lantus at equivalent doses. However, patients switching from Lantus to Toujeo may need a slightly higher total daily dose to achieve the same level of glucose control.
Toujeo versus Tresiba
Tresiba (insulin degludec) is another ultra-long-acting basal insulin, produced by Novo Nordisk. It has an even longer duration of action than Toujeo, lasting up to 42 hours, which allows for more flexible injection timing. Head-to-head trials (CONCLUDE and BRIGHT) showed comparable glucose control between the two, with some differences in hypoglycemia patterns depending on the trial design. Both are considered advanced basal insulin options for patients who need long-acting glucose coverage.
Toujeo versus rapid-acting insulins
Toujeo should not be confused with rapid-acting insulins like Humalog (insulin lispro), NovoLog (insulin aspart), or Apidra (insulin glulisine). These fast-acting insulins are taken at mealtimes to cover post-meal glucose spikes, while Toujeo provides background coverage throughout the day and night. Some patients with Type 1 diabetes or advanced Type 2 diabetes use both a basal insulin like Toujeo and a rapid-acting insulin at meals, a regimen known as basal-bolus therapy.
Making the right treatment choice
Whether someone ends up on Toujeo, a GLP-1 medication, or both depends on multiple individual factors. Here is a practical framework for understanding which direction a treatment plan might go.
Factors favoring GLP-1 therapy
Type 2 diabetes with preserved beta cell function. Obesity or overweight with diabetes. Established cardiovascular disease or high cardiovascular risk. Desire for weight loss alongside glucose control. Preference for less frequent injections (weekly versus daily). Concern about hypoglycemia risk. Earlier in the diabetes treatment progression.
BMI requirements may apply for insurance coverage of GLP-1 drugs when prescribed specifically for weight management rather than diabetes. The clinical benefits of GLP-1-based weight loss therapy extend beyond the scale, improving blood pressure, lipid profiles, sleep apnea, and conditions like lipedema.
Factors favoring insulin therapy (Toujeo)
Type 1 diabetes (absolute indication). Very advanced Type 2 diabetes with severe beta cell depletion. Very high HbA1c at diagnosis (above 10 percent) with hyperglycemic symptoms. GLP-1 intolerance or contraindication. Personal or family history of medullary thyroid carcinoma. History of pancreatitis. Pregnancy (insulin is the preferred treatment for gestational and pre-existing diabetes). Cost barriers to GLP-1 therapy with adequate insulin coverage.
Factors favoring combination therapy
Type 2 diabetes not controlled on basal insulin alone. Desire to avoid mealtime insulin injections. Insulin-related weight gain causing concern. High post-meal glucose spikes despite adequate fasting glucose control. Healthcare provider recommending treatment intensification beyond basal insulin.
The role of emerging therapies
The diabetes and obesity treatment landscape continues to evolve rapidly. Understanding where things are heading provides additional context for the Toujeo versus GLP-1 discussion.
Next-generation GLP-1 drugs
Retatrutide, the triple agonist, could become the most potent weight loss and glucose-lowering medication ever developed. Phase 2 trial results showed average weight loss exceeding 24 percent, surpassing even tirzepatide. If Phase 3 trials confirm these results, retatrutide could significantly shift the treatment paradigm.
Mazdutide, a dual GLP-1/glucagon agonist, is in development and shows promising results in early trials, particularly in Asian populations. Survodutide is another dual agonist in the pipeline with encouraging phase 2 data.
Oral formulations are advancing rapidly. Oral tirzepatide is in clinical trials, and if successful, could eliminate the injection barrier entirely for this class. Sublingual semaglutide, tirzepatide drops, and GLP-1 patches represent additional non-injection delivery methods under development.
Smart insulin and closed-loop systems
On the insulin side, research is advancing toward glucose-responsive insulin formulations that would automatically adjust their activity based on blood sugar levels, essentially building in the safety feature that GLP-1 drugs inherently have. Closed-loop insulin delivery systems (artificial pancreas technology) combine continuous glucose monitoring with automated insulin pumps to create more physiological insulin delivery patterns.
These advances do not change the fundamental classification. Insulin remains insulin. GLP-1 drugs remain GLP-1 drugs. But the gap in clinical outcomes between the two classes may narrow as insulin technology catches up with the incretin-based revolution.
Practical tips for patients navigating both drug classes
If you are a patient who has been prescribed either Toujeo or a GLP-1 medication, or if you are trying to understand your options, these practical considerations can help.
Questions to ask your healthcare provider
Ask why a specific drug class was chosen for you. If prescribed Toujeo, ask whether a GLP-1 agonist was considered and why it was not selected. If prescribed a GLP-1 drug, ask whether insulin might be needed in the future. Understand the goal of your treatment. Is it purely glucose control? Weight management? Cardiovascular protection? The answers to these questions should align with the drug class prescribed.
Monitoring differences
Patients on Toujeo need regular blood glucose monitoring, including fasting glucose checks and possibly continuous glucose monitoring. Hypoglycemia awareness is critical. Patients on GLP-1 medications also benefit from glucose monitoring but with less emphasis on hypoglycemia and more attention to GI symptoms, nutritional intake, and weight tracking.
Home monitoring tools for GLP-1 therapy can help track progress and identify issues early. Whether you are on insulin, a GLP-1, or both, regular HbA1c testing (typically every three months) remains the standard for assessing long-term glucose control.
Storage and handling
Both Toujeo and GLP-1 medications require refrigeration before first use. Once opened, both can typically be stored at room temperature for a limited period (28 days for most products). Traveling with injectable diabetes medications requires planning for temperature control. A travel case designed for GLP-1 or insulin pens can help maintain proper temperatures during transport.
Injection technique
Proper injection technique matters for both drug classes. GLP-1 injection site selection and injection method follow similar principles to insulin injection, which may contribute to the confusion between the classes. Both are given subcutaneously. Both benefit from site rotation. But the medications themselves work through completely different mechanisms once absorbed.
What researchers should know
For peptide researchers exploring the GLP-1 landscape, the distinction between insulin and incretin mimetics is foundational knowledge. The research community around SeekPeptides has been actively following developments in both the insulin and GLP-1 spaces, and understanding the pharmacological differences is essential for informed research.
Comparing different GLP-1-based therapies requires understanding what makes each one unique. Semaglutide activates GLP-1 receptors only. Tirzepatide activates both GLP-1 and GIP receptors. Retatrutide targets GLP-1, GIP, and glucagon receptors. Each additional receptor target appears to enhance efficacy but also changes the side effect profile and mechanism of action.
Microdosing protocols for GLP-1 medications represent an emerging area of research interest, particularly for applications beyond weight loss and diabetes, including autoimmune conditions. Microdosing tirzepatide and similar approaches reflect the growing recognition that these peptides have effects throughout the body, not just on blood sugar and appetite.
The semaglutide dosage calculator and peptide calculator on SeekPeptides can help researchers determine appropriate dosing for their specific protocols. Understanding the difference between insulin and GLP-1 agonists is step one. Getting the dosing right for whichever medication class is being studied is step two.
Lifestyle factors that complement both treatments
Regardless of whether someone is on Toujeo, a GLP-1 medication, or both, lifestyle factors play a critical role in treatment success. The specific dietary and exercise recommendations differ somewhat between the two drug classes.
Diet on insulin versus GLP-1 therapy
Patients on Toujeo need consistent carbohydrate intake to match their insulin dose. Skipping meals or drastically reducing carbs without adjusting insulin can trigger hypoglycemia. Carbohydrate counting is often a central part of insulin management.
Patients on GLP-1 medications face different dietary challenges. Choosing the right foods on semaglutide involves prioritizing protein to prevent muscle loss during weight reduction, eating smaller meals to accommodate reduced appetite and slowed gastric emptying, and avoiding foods that exacerbate GI side effects. Certain foods are best avoided on tirzepatide due to their tendency to worsen nausea or bloating.
Protein intake becomes especially important on GLP-1 therapy because significant weight loss can include lean muscle mass if protein intake is inadequate. Protein shakes designed for GLP-1 users and specific recipes can help patients meet their nutritional needs while navigating reduced appetite.
Supplementation is another consideration. Patients on GLP-1 drugs may benefit from fiber supplements for digestive regularity, probiotics for gut health, and B12 supplementation to prevent deficiency that can occur with reduced food intake. Some GLP-1 formulations now include B12, glycine, or niacinamide in their compounded versions.
Exercise considerations
Exercise benefits both drug classes but requires different precautions. Insulin users must plan exercise carefully, often reducing their insulin dose before planned physical activity to prevent exercise-induced hypoglycemia. Carrying fast-acting glucose during workouts is standard practice.
GLP-1 users face less hypoglycemia risk during exercise but may struggle with nausea if exercising too soon after injections. While weight loss can occur on GLP-1 medications without exercise, adding resistance training helps preserve lean muscle mass and improves metabolic health outcomes beyond what medication alone achieves.
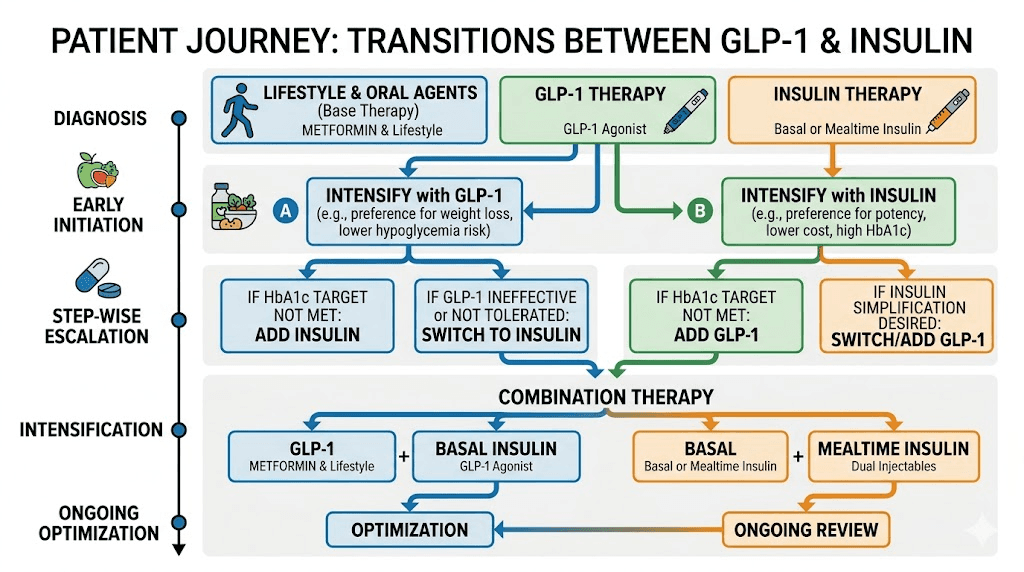
Switching between drug classes
Some patients transition from one drug class to another during their treatment journey. Understanding what these transitions look like reinforces the distinction between insulin and GLP-1 medications.
Moving from insulin to GLP-1
A patient on basal insulin whose Type 2 diabetes is relatively well controlled might transition to a GLP-1 agonist if they are gaining excessive weight on insulin, want to simplify their regimen, have developed cardiovascular risk factors that would benefit from GLP-1 protection, or have adequate remaining beta cell function. This transition requires careful medical supervision, as stopping insulin while starting a GLP-1 drug involves monitoring for hyperglycemia during the switch period.
Moving from GLP-1 to insulin
Conversely, some patients on GLP-1 therapy may eventually need insulin if their Type 2 diabetes progresses and beta cell function declines further. Signs that insulin may be needed include persistently elevated blood sugar despite maximum GLP-1 doses, declining endogenous insulin production, and treatment plateaus that suggest the incretin-based approach has reached its limits.
Switching within drug classes
Switching from one GLP-1 to another, such as moving from semaglutide to tirzepatide, is a different kind of transition entirely. This is not a class change but a medication change within the same therapeutic category. Conversion charts exist to help guide dosing during these within-class switches.
Similarly, switching from Lantus to Toujeo is a within-class insulin change, not a transition between drug categories. These within-class switches are generally simpler than cross-class transitions because the underlying mechanism of action remains the same.
Special populations and considerations
Different patient populations have different considerations when it comes to choosing between insulin and GLP-1 therapy.
Pregnancy and breastfeeding
Insulin is the preferred treatment for diabetes during pregnancy. No GLP-1 medication is currently approved for use in pregnant women. Animal studies have shown potential risks with GLP-1 drugs during pregnancy, and these medications should be discontinued before conception if possible. Accidental pregnancy while on GLP-1 therapy is a recognized concern, particularly given the increased fertility that can accompany weight loss. Breastfeeding considerations also differ between the drug classes, with insulin generally considered compatible with breastfeeding while GLP-1 drugs are not recommended.
Elderly patients
Older adults are at higher risk for hypoglycemia with insulin therapy, including Toujeo. Reduced kidney function, irregular meal patterns, and polypharmacy all increase the danger. GLP-1 agonists may be preferable in some elderly patients due to lower hypoglycemia risk, though GI side effects and potential for weight loss (which can be undesirable in frail elderly patients) require careful consideration.
Thyroid conditions
Patients with Hashimoto thyroiditis or other thyroid conditions may have special considerations when choosing between drug classes. The boxed warning about thyroid C-cell tumors applies to all GLP-1 drugs and prohibits their use in patients with personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2. Thyroid nodules in patients on tirzepatide warrant careful monitoring. Insulin has no thyroid-related contraindications.
Autoimmune conditions
The relationship between GLP-1 therapy and autoimmune conditions is an area of active research. Microdosing GLP-1 for autoimmune conditions has emerged as a research interest based on the anti-inflammatory properties these peptides demonstrate in preclinical studies. GLP-1 and endometriosis is another area being explored. Insulin, as a hormone replacement, does not have these potential anti-inflammatory applications.
Frequently asked questions
Is Toujeo the same as Ozempic?
No. Toujeo is insulin glargine, a long-acting basal insulin. Ozempic is semaglutide, a GLP-1 receptor agonist. They belong to completely different drug classes, work through different mechanisms, and have different effects on weight, appetite, and cardiovascular health.
Can Toujeo help with weight loss?
No. Toujeo typically causes weight gain because insulin promotes glucose uptake into cells, including fat cells, and reduces glucose loss through urine. If weight loss is a treatment goal, GLP-1 medications are a more appropriate choice.
Is Toujeo safer than GLP-1 medications?
Each has a different safety profile. Toujeo carries higher hypoglycemia risk and causes weight gain but has minimal GI side effects. GLP-1 drugs have lower hypoglycemia risk and promote weight loss but commonly cause nausea, vomiting, and other GI symptoms. The "safer" option depends on the individual patient profile and treatment priorities.
Why did my doctor prescribe Toujeo instead of a GLP-1?
Possible reasons include Type 1 diabetes diagnosis, advanced Type 2 diabetes with severely depleted beta cells, contraindication to GLP-1 drugs (thyroid cancer history, pancreatitis history), insurance or cost barriers, prior GLP-1 intolerance, or very high blood sugar requiring immediate insulin coverage. Ask your doctor specifically why Toujeo was chosen over a GLP-1 agonist.
Can I switch from Toujeo to a GLP-1 medication?
Potentially, but only under medical supervision. The switch depends on your remaining beta cell function, current glucose control, overall health status, and treatment goals. Some patients successfully transition from insulin to GLP-1 therapy, particularly if their Type 2 diabetes was caught relatively early.
Is Tresiba a GLP-1?
No. Tresiba is insulin degludec, another long-acting basal insulin similar to Toujeo. Like Toujeo, it provides exogenous insulin rather than activating GLP-1 receptors. The confusion between basal insulins and GLP-1 drugs extends across the entire insulin category, not just Toujeo.
Do GLP-1 medications replace insulin?
For Type 2 diabetes patients with adequate beta cell function, GLP-1 medications can sometimes replace or delay the need for insulin therapy. For Type 1 diabetes patients, GLP-1 drugs cannot replace insulin because these patients lack the beta cells needed for GLP-1-stimulated insulin secretion. GLP-1 drugs take time to reach full effectiveness, so any transition from insulin must be carefully managed.
What happens if I confuse my Toujeo pen with a GLP-1 pen?
Accidentally injecting the wrong medication could have serious consequences. Injecting Toujeo when you meant to inject a GLP-1 drug could cause severe hypoglycemia, especially at insulin doses. Injecting a GLP-1 drug when you meant to inject Toujeo could result in hyperglycemia from missing your insulin dose. Always clearly label and separately store your medications. If a mix-up occurs, contact your healthcare provider immediately.
External resources
For researchers serious about understanding the full landscape of GLP-1-based therapies, peptide protocols, and evidence-based dosing guidance, SeekPeptides offers the most comprehensive resource available. Members access detailed comparison tools, protocol databases, and a community of thousands who have navigated these exact questions about medication classification and treatment selection.
In case I do not see you, good afternoon, good evening, and good night. May your medications stay correctly identified, your blood sugar stay well controlled, and your treatment decisions stay fully informed.