Mar 23, 2026

No. Qsymia is not a GLP-1. Not even close. The two medications belong to entirely different drug classes, work through completely separate mechanisms, and produce different clinical outcomes. Yet the question keeps coming up, and for good reason. Both Qsymia and GLP-1 receptor agonists like semaglutide and tirzepatide are FDA-approved for chronic weight management. Both suppress appetite. Both require a prescription. And both show up in the same conversations when people start researching medical options for weight loss.
The confusion makes sense on the surface. But the differences between these two approaches run deep, from the molecular level all the way through to real-world results, side effect profiles, administration routes, and long-term safety data. Qsymia combines two older medications, phentermine and topiramate, into a single oral capsule. GLP-1 receptor agonists mimic a natural hormone your gut produces after eating, one that tells your brain you are full and slows the rate at which food leaves your stomach.
Understanding which category a medication falls into matters more than most people realize. The mechanism determines the side effects you will experience, how long you can safely take the medication, what monitoring your doctor needs to do, and ultimately how much weight you can expect to lose.
SeekPeptides has put together this comprehensive breakdown so you can understand exactly what Qsymia is, what GLP-1 medications are, and how they compare across every metric that matters.
This guide covers the pharmacology, the clinical trial data, the side effects, the costs, and the practical considerations that should inform your conversation with a healthcare provider. Whether you are considering qualifying for semaglutide or exploring alternatives to GLP-1 medications, what follows will give you the full picture.
What Qsymia actually is
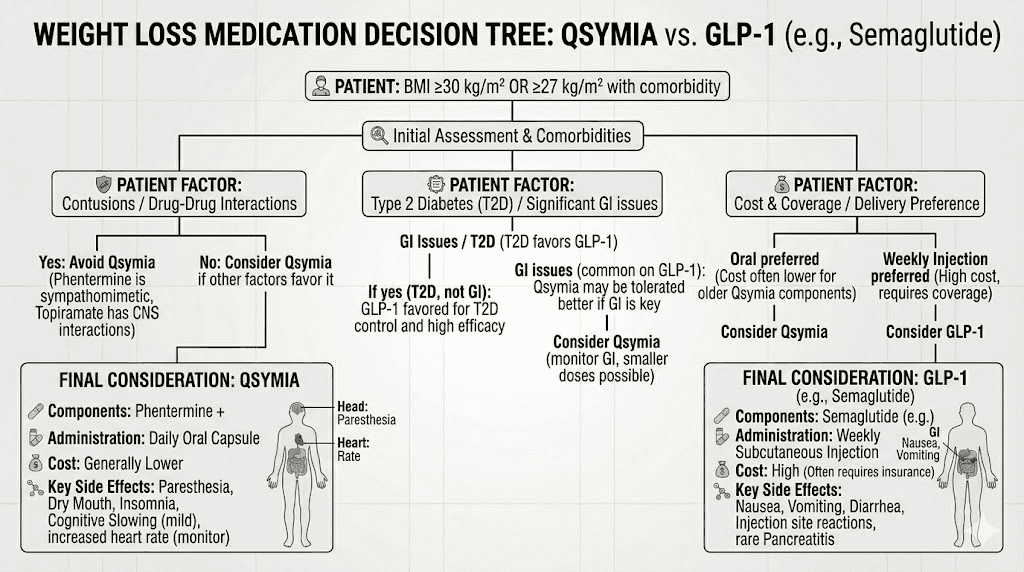
Qsymia is a combination prescription medication containing two active ingredients: phentermine and topiramate extended-release. The FDA approved it in 2012 for chronic weight management in adults with a BMI of 30 or greater, or a BMI of 27 or greater with at least one weight-related comorbidity such as high blood pressure, type 2 diabetes, or high cholesterol. In 2022, the approval expanded to include adolescents aged 12 and older.
Phentermine is a sympathomimetic amine. Think of it as a distant cousin of amphetamine, though with a much milder stimulant profile. It increases norepinephrine activity in the hypothalamus, the part of your brain that regulates hunger signals. The result is decreased appetite and reduced food consumption. Phentermine has been around since 1959. It is one of the oldest prescription weight loss medications still in use.
Topiramate is an anticonvulsant medication originally developed to treat epilepsy and prevent migraines. During clinical trials for those conditions, researchers noticed an interesting side effect: patients kept losing weight. The exact mechanism behind topiramate weight loss is not fully understood. Scientists believe it enhances satiety, the feeling of fullness after eating, through modulation of GABA receptors and voltage-gated sodium channels. It may also affect taste perception, making food less appealing.
The combination makes pharmacological sense. Phentermine reduces your desire to eat. Topiramate makes you feel full faster. Together, they attack appetite from two different angles. But neither of these mechanisms involves GLP-1 receptors, incretin hormones, or any of the pathways that GLP-1 receptor agonists target.
Qsymia is taken orally, once daily in the morning. It comes in four dosage strengths:
3.75 mg phentermine / 23 mg topiramate (starting dose, taken for 14 days)
7.5 mg phentermine / 46 mg topiramate (recommended dose)
11.25 mg phentermine / 69 mg topiramate (titration dose)
15 mg phentermine / 92 mg topiramate (top dose)
The dosing follows a gradual escalation. You start at the lowest dose, move to the recommended dose after two weeks, and only increase further if weight loss is insufficient after 12 weeks. This stepwise approach helps minimize side effects, particularly the cognitive and neurological effects that topiramate can cause.
What GLP-1 medications actually are
GLP-1 stands for glucagon-like peptide-1. It is a hormone your body produces naturally. When you eat, specialized cells in your small intestine release GLP-1 into your bloodstream. This hormone does several things simultaneously. It signals your pancreas to release insulin. It tells your liver to stop producing glucose. It slows gastric emptying, keeping food in your stomach longer. And it acts on appetite centers in your brain, reducing hunger and increasing feelings of satisfaction after meals.
Natural GLP-1 has a problem, though. Your body breaks it down within minutes. The enzyme dipeptidyl peptidase-4 (DPP-4) chews through GLP-1 so quickly that its effects are fleeting. GLP-1 receptor agonist medications solve this by using modified versions of the GLP-1 molecule that resist enzymatic breakdown. Some versions, like semaglutide, have a half-life of about a week, which is why they only require once-weekly dosing.
The GLP-1 receptor agonist class includes several medications:
Semaglutide (brand names Wegovy for weight loss, Ozempic for diabetes)
Liraglutide (brand names Saxenda for weight loss, Victoza for diabetes)
Tirzepatide (brand name Zepbound for weight loss, Mounjaro for diabetes), which is technically a dual GIP/GLP-1 receptor agonist
Dulaglutide (Trulicity, for diabetes)
Tirzepatide deserves special mention because it targets both GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptors. This dual mechanism appears to produce even greater weight loss than single GLP-1 agonists alone. Clinical trials showed average weight loss of 20-25% of body weight with tirzepatide at the highest doses, compared to 15-17% with semaglutide.
Most GLP-1 medications are administered via subcutaneous injection, typically in the abdomen, thigh, or upper arm. Newer formulations are emerging, including oral semaglutide drops and oral tirzepatide, but the injectable versions remain the standard for weight management.
The fundamental differences between Qsymia and GLP-1 medications
These two medication classes could hardly be more different. Understanding the distinctions helps explain why Qsymia is categorically not a GLP-1, and why that distinction has real clinical implications.
Mechanism of action
Qsymia works through central nervous system stimulation. Phentermine triggers norepinephrine release in the brain, creating a mild fight-or-flight response that suppresses hunger. Topiramate modulates GABA receptors and sodium channels, enhancing satiety through pathways that scientists still do not fully understand. Neither component interacts with GLP-1 receptors at all.
GLP-1 medications work by mimicking a natural gut hormone. They bind to GLP-1 receptors in the brain, pancreas, and digestive tract. The appetite suppression comes from hormonal signaling rather than central nervous system stimulation. This distinction matters because it affects the side effect profile, the drug interactions, and the long-term safety considerations.
Think of it this way. Qsymia turns up the volume on your sympathetic nervous system and adjusts neurotransmitter activity to make you less interested in food. GLP-1 medications speak the same language your body already uses after a meal, just louder and for longer. Both reduce appetite. But they do it through entirely different communication systems.
Administration
Qsymia is an oral capsule taken once daily in the morning. Simple. No needles, no reconstitution, no refrigeration. You take it with a glass of water before or after breakfast.
Most GLP-1 medications require weekly subcutaneous injections. This means using a syringe or auto-injector to deliver the medication under the skin, typically in the abdomen or thigh. The injections are relatively painless, using very thin needles, but the process requires some instruction and comfort with self-injection. Compounded versions of semaglutide and tirzepatide also need to be reconstituted before use, adding another step.
For people who strongly prefer oral medications, Qsymia has an obvious practical advantage. For people who do not mind a once-weekly injection, the frequency advantage goes to GLP-1 medications. Taking something once a week is easier to remember than daily dosing.
Drug class and regulatory status
Qsymia contains phentermine, which is a Schedule IV controlled substance due to its stimulant properties and potential for dependence. This classification adds prescribing restrictions and limits refill options in some states. Topiramate is not a controlled substance, but it carries its own regulatory requirements, including the REMS (Risk Evaluation and Mitigation Strategy) program that Qsymia must follow due to the risk of birth defects.
GLP-1 medications are not controlled substances. They carry no risk of physical dependence or abuse. This difference matters for long-term prescribing, as some providers are hesitant to prescribe Schedule IV medications for extended periods.
Weight loss effectiveness: what the clinical trials show
Numbers matter more than marketing. Here is what the clinical trial data actually shows for both medication classes.
Qsymia clinical trial results
Two major phase 3 trials established the efficacy data for Qsymia:
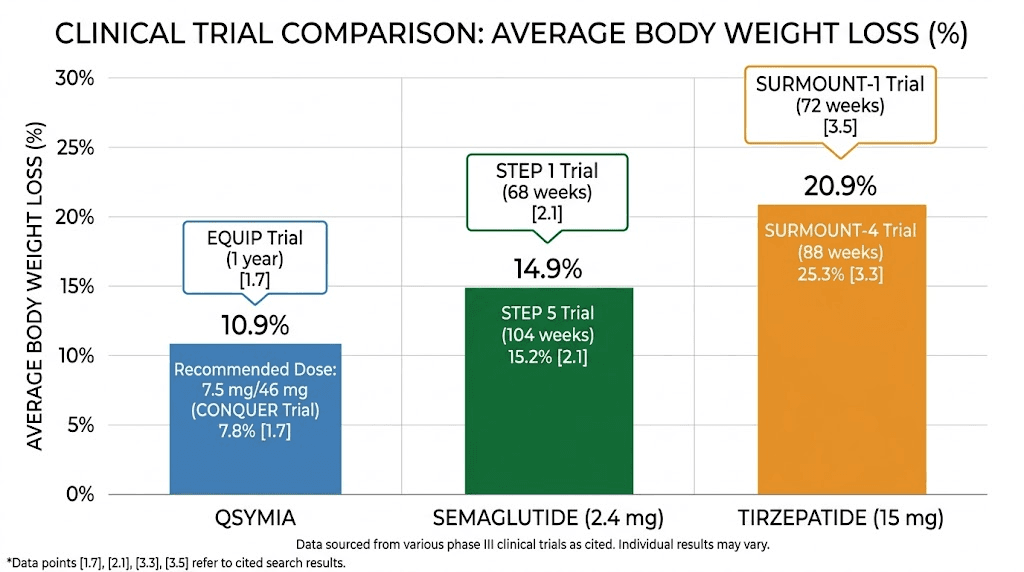
The CONQUER trial enrolled 2,487 overweight and obese adults. After 56 weeks:
Placebo group lost 1.2% of body weight
Recommended dose (7.5/46 mg) group lost 7.8% of body weight
Top dose (15/92 mg) group lost 9.8% of body weight
The EQUIP trial enrolled 1,267 adults with class II and III obesity (BMI 35+). After 56 weeks:
Placebo group lost 1.6% of body weight
Starting dose (3.75/23 mg) group lost 5.1% of body weight
Top dose (15/92 mg) group lost 10.9% of body weight
Beyond weight loss, the CONQUER trial showed significant improvements in blood pressure, waist circumference, lipid profiles, glycemic parameters, and inflammatory biomarkers compared to placebo. These cardiometabolic improvements matter because excess weight drives many of these risk factors.
GLP-1 clinical trial results
GLP-1 medications have consistently demonstrated greater weight loss in clinical trials.
The STEP trials for semaglutide 2.4 mg (Wegovy) showed:
Average weight loss of approximately 15% of body weight over 68 weeks
One-third of participants lost 20% or more of body weight
Significant reductions in waist circumference, blood pressure, and cardiovascular risk markers
The SURMOUNT trials for tirzepatide (Zepbound) showed even more impressive results:
Average weight loss of 20-25% at the highest dose (15 mg) over 72 weeks
More than half of participants lost 20% or more of body weight
Significant improvements in type 2 diabetes markers, blood pressure, and lipid profiles
The head-to-head comparison is clear. GLP-1 medications produce roughly 50-100% more weight loss than Qsymia at their respective top doses. A person taking the maximum Qsymia dose might expect to lose about 10% of their body weight. A person taking semaglutide at the weight loss dose might lose 15%. Someone on tirzepatide at the top dose might lose 20-25%.
Comparison table
Metric | Qsymia (top dose) | Semaglutide 2.4 mg | Tirzepatide 15 mg |
|---|---|---|---|
Average weight loss | 9.8-10.9% | ~15% | ~22% |
Participants losing 10%+ | ~48% | ~70% | ~85% |
Participants losing 20%+ | ~12% | ~32% | ~55% |
Trial duration | 56 weeks | 68 weeks | 72 weeks |
Route | Oral daily | Injection weekly | Injection weekly |
Drug class | Sympathomimetic + anticonvulsant | GLP-1 agonist | GIP/GLP-1 agonist |
Side effects and safety profiles
The side effect profiles of Qsymia and GLP-1 medications reflect their fundamentally different mechanisms. Knowing what to expect matters, especially if you are weighing one against the other.
Qsymia side effects
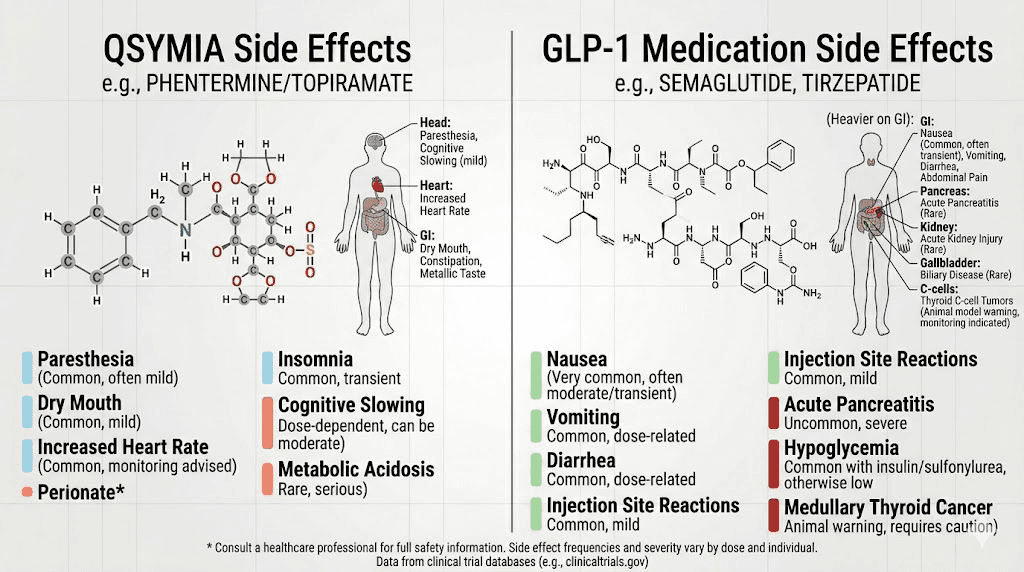
The most common side effects reported in clinical trials include:
Paresthesia (tingling or numbness, especially in hands and feet), reported by up to 20% of users at the top dose
Dizziness
Dysgeusia (altered taste, things taste metallic or strange)
Insomnia (due to the stimulant properties of phentermine)
Constipation
Dry mouth
But Qsymia carries several serious warnings that set it apart from GLP-1 medications:
Birth defects. Topiramate causes cleft lip and cleft palate in developing fetuses. This risk is so significant that Qsymia is available only through a restricted REMS program. Women must have a pregnancy test before starting and monthly while taking it. Effective contraception is mandatory.
Suicidal ideation. Topiramate is an antiepileptic drug, and this class carries an FDA warning about increased risk of suicidal thoughts and behavior. Patients with depression history need careful monitoring.
Cognitive dysfunction. Many users report difficulty with concentration, memory, and word-finding. This is a well-known topiramate effect, sometimes called "dopamax" in medical circles because of how foggy it makes people feel. The cognitive effects can be significant enough to interfere with work performance.
Metabolic acidosis. Topiramate can increase blood acid levels, potentially leading to kidney stones, weakened bones, and harm to unborn children. Regular blood work is recommended to monitor bicarbonate levels.
Elevated heart rate. Phentermine is a stimulant. It can increase resting heart rate, and patients with cardiovascular disease need to approach it cautiously. Qsymia is not recommended for people with uncontrolled hypertension or a history of heart disease.
GLP-1 side effects
GLP-1 medications have a different side effect profile, heavily weighted toward gastrointestinal symptoms:
Nausea (the most common, affecting 40-50% initially, but typically improving over weeks)
Bloating and abdominal discomfort
The key difference is trajectory. GLP-1 side effects tend to be worst during the first few weeks of treatment or after dose increases, then gradually improve as the body adjusts. Most people find the nausea manageable after the first month. The timeline for side effects typically follows a predictable pattern, peaking early and fading.
GLP-1 medications also carry some serious (but rare) warnings:
Potential risk of thyroid C-cell tumors (observed in rodent studies, unclear relevance in humans)
Risk of gallbladder problems
Pancreatitis (rare but documented)
Hair loss (temporary, related to rapid weight loss rather than the drug itself)
One notable advantage of GLP-1 medications: they do not cause cognitive dysfunction, do not carry teratogenicity warnings as severe as topiramate, and are not controlled substances.
Cost and insurance coverage
Cost is often the deciding factor between these medications, especially since insurance coverage varies dramatically.
Qsymia costs
Without insurance, Qsymia typically costs around $200-310 per month at retail pharmacies. However, several savings options exist. GoodRx coupons can bring the price down to approximately $89 for a 30-day supply. The manufacturer also offers Qsymia Engage, a cash-pay program that fixes pricing at $98 per month, or $70 per month with a 90-day supply.
Generic phentermine-topiramate ER became available in 2025, which has further reduced costs. The generic version may be available for even less than the brand-name savings programs.
Private insurance plans may cover Qsymia for patients who meet BMI criteria, though coverage varies by plan. Medicare prescription drug plans generally do not cover Qsymia or any weight loss medications.
GLP-1 costs
Brand-name GLP-1 medications are significantly more expensive. Wegovy lists at approximately $1,300-1,400 per month. Zepbound (tirzepatide) lists at around $1,000 per month. Without insurance, these prices put GLP-1 medications out of reach for many people.
However, compounded semaglutide and compounded tirzepatide offer more affordable alternatives, typically ranging from $100-400 per month depending on the pharmacy and dosage. Insurance coverage for GLP-1 weight loss medications has been expanding, with some states now requiring coverage and new Medicare provisions taking effect.
The cost comparison depends heavily on your insurance situation. With good insurance, GLP-1 medications might cost similar copays to Qsymia. Without insurance, Qsymia (especially generic) is substantially cheaper than brand-name GLP-1 medications, though compounded versions narrow the gap.
For people exploring the most affordable tirzepatide or affordable semaglutide options, compounding pharmacies have become a popular route. But understanding that Qsymia is not a GLP-1 is important when comparing costs, because you are comparing medications with different effectiveness levels.
Who is Qsymia better suited for?
Despite GLP-1 medications showing superior weight loss in clinical trials, Qsymia still has a place in the treatment landscape. Several scenarios make it a reasonable or even preferred choice.
People who will not do injections. Some people have genuine needle phobia or simply refuse to inject themselves weekly. Qsymia is taken orally. Period. No needles, no syringes, no injection technique to learn. Until oral GLP-1 formulations become widely available and proven for weight loss, Qsymia remains the strongest oral option.
People who need migraine prevention. Topiramate is FDA-approved for migraine prevention. If you have both obesity and chronic migraines, Qsymia addresses both conditions simultaneously. This dual benefit is genuinely unique and can simplify a medication regimen.
People who experience severe GI side effects on GLP-1 medications. Some individuals cannot tolerate the nausea, vomiting, and digestive disruption that GLP-1 medications cause. If someone has tried semaglutide or tirzepatide and the gastrointestinal effects were intolerable even after proper titration, Qsymia offers a completely different side effect profile. You trade GI problems for potential tingling, cognitive effects, and insomnia.
Cost-sensitive patients without insurance coverage. At $70-98 per month through savings programs or even less for the generic, Qsymia costs a fraction of brand-name GLP-1 medications. For someone paying entirely out of pocket, the math may favor Qsymia despite its lower efficacy.
People with epilepsy. If you take topiramate for seizure control, Qsymia packages that treatment with weight loss benefits. The overlapping indication creates efficiency.
Who should choose GLP-1 medications instead?
For most people seeking maximum weight loss and a favorable long-term safety profile, GLP-1 medications are the stronger choice. But specific situations make the case even clearer.
People with type 2 diabetes. GLP-1 medications do double duty here. They reduce blood sugar through insulin regulation while simultaneously producing weight loss. Qsymia has no direct glucose-lowering mechanism. If you have diabetes and obesity, GLP-1 medications address both conditions through a single medication class.
People with cardiovascular disease. Semaglutide (Wegovy) has demonstrated cardiovascular risk reduction in clinical trials, reducing the risk of major cardiovascular events by 20%. Phentermine, the stimulant component of Qsymia, can increase heart rate and is generally avoided in patients with significant cardiovascular disease.
People who need maximum weight loss. If your BMI is 40+ and you need substantial weight reduction for health reasons, the 20-25% body weight loss achievable with tirzepatide vastly exceeds the 10% typical of Qsymia. For severe obesity, every percentage point matters.
Women of childbearing age. While pregnancy should be avoided on any weight loss medication, the teratogenicity risk with Qsymia (specifically topiramate causing cleft palate) is uniquely severe. GLP-1 medications require a washout period before pregnancy but do not carry the same level of birth defect risk.
People who value cognitive clarity. Topiramate cognitive effects are not subtle for many users. Difficulty finding words mid-sentence, trouble concentrating during meetings, feeling mentally foggy. If your work requires sharp cognitive function, GLP-1 medications do not carry this risk.
People concerned about dependence. Phentermine is a Schedule IV controlled substance. While physical dependence at prescribed doses is uncommon, the classification carries stigma and prescribing restrictions. GLP-1 medications have no abuse potential and no controlled substance designation.
Can you take Qsymia and GLP-1 medications together?
This question comes up frequently, and the answer is nuanced. No major pharmacokinetic interactions exist between Qsymia and GLP-1 receptor agonists. They work through different mechanisms and do not compete for the same receptors or metabolic pathways.
However, combination therapy with two weight loss medications is generally not recommended without careful medical supervision. The concern is not about drug interactions but about amplified side effects. Combining appetite suppressants that work through different mechanisms could theoretically enhance weight loss, but it could also increase the risk and severity of side effects from both medications simultaneously.
Some obesity medicine specialists do prescribe combination approaches in carefully selected patients, particularly those who have plateaued on a single medication. But this is an off-label use that requires close monitoring. If your weight loss has plateaued on semaglutide or tirzepatide has stopped working, adding Qsymia should only happen under direct physician supervision.
Importantly, there are no large clinical trials studying the combination of Qsymia with GLP-1 medications. The evidence base for this approach is limited to case reports and clinical experience. Until proper studies are conducted, it remains an area of clinical judgment rather than evidence-based medicine.
If you are considering phentermine with semaglutide or phentermine with tirzepatide, understand that these are separate clinical decisions that require professional guidance.
Other non-GLP-1 weight loss medications worth knowing
If you are exploring alternatives to GLP-1 medications, Qsymia is not the only option. Understanding the full landscape helps you have better conversations with your healthcare provider.
Contrave (naltrexone/bupropion) combines an opioid antagonist with an antidepressant. It produces modest weight loss (5-8% of body weight) and may be particularly helpful for people whose overeating has a compulsive or emotional component. Like Qsymia, it is oral. Unlike Qsymia, it does not contain a controlled substance.
Phentermine alone is available as a standalone prescription. It is approved only for short-term use (up to 12 weeks), which limits its utility for chronic weight management. But it is inexpensive and widely prescribed. Our guide on whether phentermine is a GLP-1 covers this in detail, and the answer is the same: no, phentermine is not a GLP-1 either.
Orlistat (Xenical/Alli) blocks fat absorption in the gut. It produces modest weight loss (3-5%) but carries notorious gastrointestinal side effects including oily stools and fecal urgency. It works through yet another mechanism entirely unrelated to GLP-1.
The comparison between phentermine and semaglutide, phentermine and tirzepatide, and phentermine and GLP-1 medications broadly consistently shows the GLP-1 class producing greater weight loss with a more favorable long-term safety profile.
The future of weight loss medications
The weight loss medication landscape is evolving rapidly. Several next-generation treatments are in clinical trials that may further shift the balance.
Retatrutide is a triple agonist that targets GLP-1, GIP, and glucagon receptors simultaneously. Phase 2 trial data showed weight loss of up to 24% at the highest dose, with phase 3 trials underway. If approved, it would represent yet another step forward in the GLP-1 receptor agonist family.
Survodutide is a dual GLP-1/glucagon receptor agonist showing promising results in clinical trials. Orforglipron is an oral small-molecule GLP-1 agonist that could eventually provide GLP-1 level weight loss without injections, potentially making the oral advantage of Qsymia less relevant.
CagriSema combines semaglutide with cagrilintide (an amylin receptor agonist), targeting yet another hormone pathway involved in appetite regulation. Early data suggests even greater weight loss than semaglutide alone.
As these newer medications reach the market, Qsymia will likely move further down the treatment algorithm. But for now, it remains a viable option for specific patient populations, particularly those who cannot use injectable medications.
How to decide which medication is right for you
The choice between Qsymia and a GLP-1 medication is not one you should make alone. This decision requires a healthcare provider who understands your complete medical history, current medications, risk factors, and treatment goals. But you can come to that conversation prepared.
Start by understanding your priorities. If maximum weight loss is the goal and you have no contraindications, GLP-1 medications are almost always the first-line recommendation in current obesity medicine guidelines. Qsymia has become a second-line option, recommended when GLP-1 medications are not tolerated, not accessible, or contraindicated.
Consider your practical constraints. Cannot afford $1,000+ per month for brand-name GLP-1 medications? Check if compounded versions are available in your area. Still too expensive? Qsymia or generic phentermine-topiramate may be the pragmatic choice. Will not do injections? Qsymia or Contrave are your oral options until oral GLP-1 formulations for weight loss become widely available.
Think about your medical history. Migraines? Qsymia might pull double duty. Diabetes? GLP-1 medications address both conditions. Heart disease? GLP-1 medications have the cardiovascular benefit data. Planning pregnancy? Both require stopping, but Qsymia carries more severe teratogenicity risks.
Document your questions before your appointment. Ask about:
Which medication class your provider recommends given your specific situation
What your insurance covers and what the out-of-pocket costs will be
What monitoring will be required (blood work, pregnancy tests, heart rate checks)
What the plan is if the first medication does not work
How long treatment should continue
SeekPeptides members access comprehensive medication comparison tools, detailed protocol guides, and community discussions that help inform these conversations with healthcare providers.
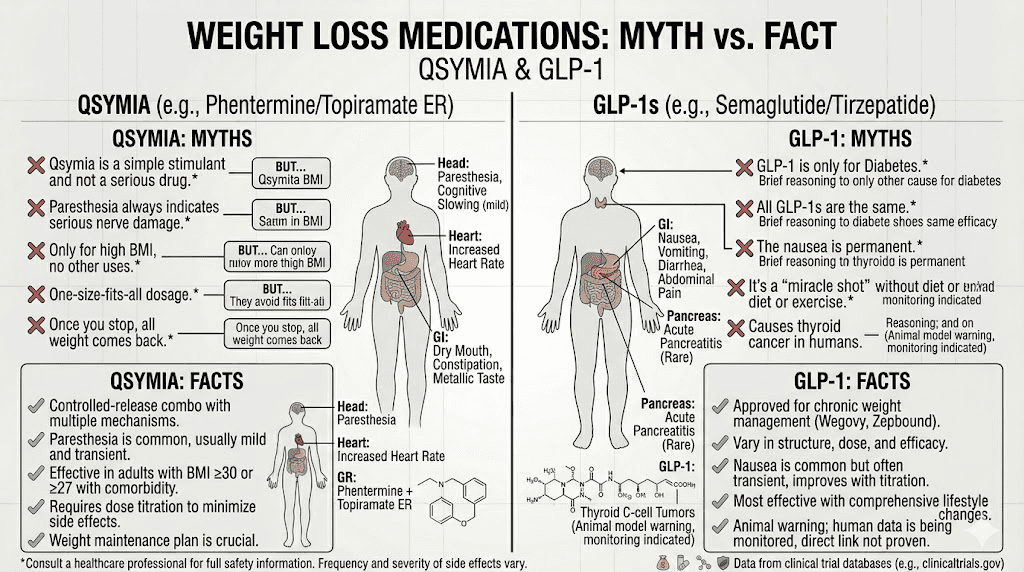
Common misconceptions about Qsymia and GLP-1 medications
Several myths circulate online that deserve correction.
Misconception: all weight loss medications work the same way
False. Qsymia works through CNS stimulation and neurotransmitter modulation. GLP-1 medications work through incretin hormone mimicry. Orlistat blocks fat absorption. Contrave combines opioid antagonism with norepinephrine/dopamine reuptake inhibition. Each class has a unique mechanism, which is exactly why they have different effectiveness levels, side effects, and appropriate patient populations.
Misconception: Qsymia is just as effective as GLP-1 medications
The clinical trial data does not support this. Qsymia produces 7-11% weight loss on average. Semaglutide produces approximately 15%. Tirzepatide produces 20-25%. These are meaningful differences that compound over time. However, any medication that helps a patient achieve clinically significant weight loss (typically defined as 5% or more) provides health benefits.
Misconception: GLP-1 medications are just fancy appetite suppressants
While appetite suppression is a major component, GLP-1 medications do much more. They slow gastric emptying, improve insulin sensitivity, reduce hepatic glucose production, and appear to have direct cardiovascular benefits independent of weight loss. The benefits beyond weight loss are substantial and growing as more research emerges.
Misconception: you can switch between Qsymia and GLP-1 medications without any transition
While switching is possible, it requires medical guidance. Stopping Qsymia requires a taper, not abrupt discontinuation, because topiramate withdrawal can trigger seizures in susceptible individuals. Starting a GLP-1 medication requires slow dose titration to minimize gastrointestinal side effects. The transition period needs planning.
Misconception: natural supplements can replace either medication class
No supplement has demonstrated weight loss results comparable to either Qsymia or GLP-1 medications in rigorous clinical trials. Products marketed as GLP-1 vitamins or GLP-1 boosters typically contain ingredients that may marginally increase natural GLP-1 production, but the magnitude of effect does not approach what prescription medications achieve. GLP-1 probiotics and similar products fall into the same category, potentially beneficial as supplements but not replacements for prescription medications.
Living with either medication: practical considerations
Beyond the pharmacology and clinical trials, daily life on these medications differs in practical ways that matter.
Diet and lifestyle on Qsymia
Qsymia is approved as an adjunct to diet and exercise. The appetite suppression helps, but the medication works best when combined with a structured eating plan and regular physical activity. Many users report that food becomes less interesting rather than unappetizing, a subtle but important distinction from the nausea-driven appetite suppression of GLP-1 medications.
Stay well-hydrated. Topiramate increases kidney stone risk, and adequate fluid intake is the simplest protective measure. Avoid alcohol, as both phentermine and topiramate can amplify its effects and increase the risk of side effects. Take the medication in the morning to minimize insomnia from phentermine stimulant effects.
Diet and lifestyle on GLP-1 medications
GLP-1 medications fundamentally change your relationship with food. The slowed gastric emptying means meals sit longer, and portion sizes naturally decrease. Many users find they need to focus on food choices that provide adequate nutrition in smaller volumes, especially protein to preserve muscle mass during weight loss.
A structured diet plan while on semaglutide or tirzepatide typically emphasizes high-protein foods, adequate fiber, and hydration. The foods to avoid tend to be high-fat, greasy, or overly processed items that exacerbate the GI side effects.
Exercise remains important on both medications. Weight loss from any source, medication or otherwise, carries the risk of muscle loss. Resistance training and adequate protein intake help preserve lean mass regardless of which medication you use.
Travel considerations
Qsymia wins on travel convenience. Oral capsules do not require refrigeration, do not need syringes, and do not raise questions at airport security. You can toss the bottle in your bag and forget about it.
GLP-1 injections require more planning. You need to consider temperature control during travel, carry syringes and alcohol swabs, and potentially deal with TSA questions. A travel case helps, but it adds logistical complexity that Qsymia simply does not have.
Monitoring requirements
Qsymia requires more routine monitoring than GLP-1 medications:
Monthly pregnancy tests for women of childbearing age
Regular heart rate monitoring
Periodic blood work for bicarbonate levels (metabolic acidosis screening)
Kidney function tests
Mood and cognition assessment
GLP-1 medications generally require less intensive monitoring, though periodic blood work to check pancreatic enzymes and kidney function is still recommended. Both medication classes benefit from regular follow-up visits to track weight loss progress and adjust dosing.
What happens when you stop each medication
Weight regain after stopping weight loss medications is a reality for both classes, but the dynamics differ.
Stopping Qsymia requires a gradual taper. Abruptly discontinuing topiramate can cause seizures, even in people who have never had seizures before. The typical taper takes one to two weeks, stepping down through the lower doses before stopping completely. Once off the medication, appetite typically returns to baseline within days to weeks, and weight regain commonly follows.
Stopping GLP-1 medications does not require a taper, but withdrawal effects can occur. Many people experience a rapid return of appetite, sometimes feeling hungrier than they did before starting the medication. Studies show that approximately two-thirds of weight lost on GLP-1 medications is regained within a year of stopping. This is one reason many obesity medicine experts now view these medications as long-term treatments rather than temporary interventions.
The strategies for maintaining weight loss after tirzepatide typically involve either continuing the medication at a lower maintenance dose, transitioning to a less expensive option, or implementing intensive lifestyle changes to offset the lost pharmacological support.
For researchers interested in understanding the weaning process, gradual dose reduction may help minimize rebound hunger and weight regain, though evidence for this approach is still emerging.
Frequently asked questions
Is Qsymia an injectable medication?
No. Qsymia is an oral capsule taken once daily in the morning. It does not require injection. This is one of its main advantages over GLP-1 injectable medications.
Can I switch from Qsymia to a GLP-1 medication?
Yes, but the transition requires medical supervision. Qsymia must be tapered gradually to avoid seizure risk from topiramate withdrawal. Your provider will design a transition plan that may include an overlap period or a gap between medications. The switching process between weight loss medications should always involve your prescribing physician.
Does Qsymia work on the same hormones as Ozempic or Wegovy?
No. Qsymia works through central nervous system stimulation (phentermine) and neurotransmitter modulation (topiramate). Ozempic and Wegovy (both semaglutide) work by mimicking the GLP-1 hormone. These are completely different biological pathways. Our guide on GLP-1 versus Ozempic explains the hormone mechanism in detail.
Which is safer for long-term use: Qsymia or GLP-1 medications?
Both are FDA-approved for long-term use in chronic weight management. GLP-1 medications generally have a more favorable long-term safety profile, with cardiovascular benefits demonstrated in clinical trials. Qsymia carries ongoing risks of cognitive effects, metabolic acidosis, and requires more intensive monitoring. Your healthcare provider can assess which profile better fits your individual risk factors.
Is phentermine the same as a GLP-1?
No. Phentermine is a sympathomimetic amine (a stimulant that suppresses appetite through norepinephrine release). It is one of the two active ingredients in Qsymia but is also prescribed as a standalone medication. Our guide on whether phentermine is a GLP-1 covers this topic comprehensively.
Why is Qsymia cheaper than GLP-1 medications?
Qsymia combines two older, well-established medications (phentermine from 1959, topiramate from 1996). The development costs were lower, generic competition is now available, and the manufacturing process for oral medications is simpler than producing injectable biologics. GLP-1 medications are complex peptide-based drugs that are expensive to manufacture, relatively new, and still under patent protection for brand-name versions.
Can Qsymia cause the same nausea as GLP-1 medications?
Qsymia can cause nausea, but it is less common and typically less severe than with GLP-1 medications. The dominant side effects of Qsymia are neurological (tingling, cognitive fog, insomnia) rather than gastrointestinal. If nausea is your primary concern with weight loss medications, Qsymia may be better tolerated in that specific regard.
Do I need a BMI of 30 to get Qsymia?
You need either a BMI of 30 or greater, or a BMI of 27 or greater with at least one weight-related condition such as hypertension, type 2 diabetes, or high cholesterol. This is the same basic threshold used for GLP-1 weight loss medications. Our guide on BMI requirements for GLP-1 medications covers the qualification criteria in detail.
External resources
For researchers serious about understanding the full landscape of weight loss medications and making informed decisions about peptide-based treatments, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, detailed comparison tools, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your medications stay effective, your decisions stay informed, and your progress stay consistent.