Mar 18, 2026

You took your semaglutide injection on Monday. Now it is Thursday, and you are wondering whether you can take tirzepatide before the week ends. Maybe your provider switched your prescription. Maybe you have both medications on hand and want faster results. Whatever the reason, this question comes up more often than most people realize.
The short answer is no. You should not take semaglutide and tirzepatide in the same week.
But the full answer involves understanding why these two medications overlap in ways that make same-week dosing genuinely dangerous, what happens inside your body when both are active simultaneously, and how to transition between them safely if switching is the goal. This guide covers everything researchers and patients need to know about the timing, the risks, and the proper protocol for moving from one GLP-1 receptor agonist to another without putting your health at risk.
SeekPeptides has compiled the most comprehensive breakdown available on this topic, pulling from prescribing information, clinical research, and real-world protocol guidance from experienced providers.
Why semaglutide and tirzepatide should never overlap in the same week
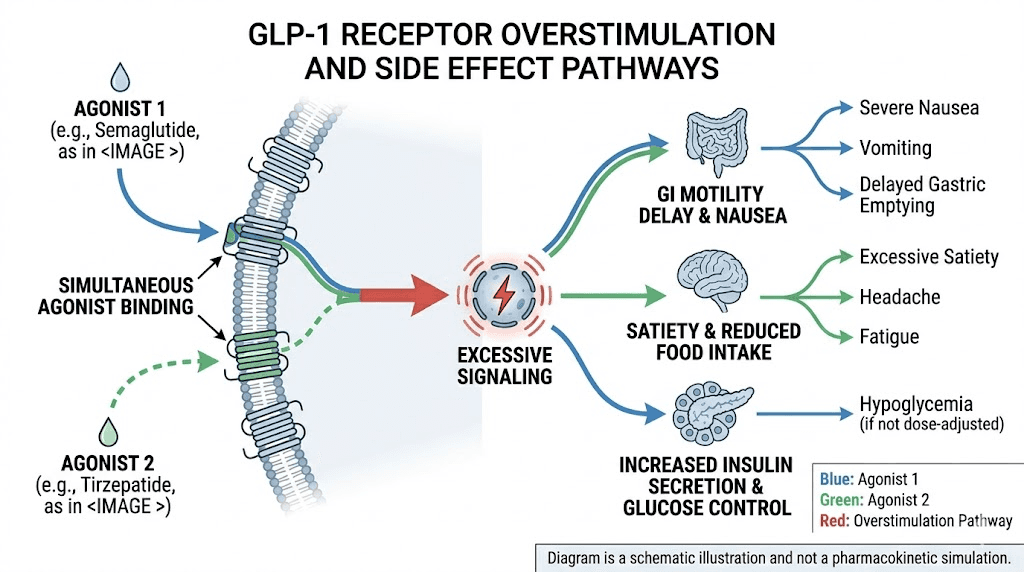
Both semaglutide and tirzepatide target the GLP-1 receptor pathway. Semaglutide is a pure GLP-1 receptor agonist. Tirzepatide is a dual GIP and GLP-1 receptor agonist, meaning it activates two incretin receptors instead of one. When both medications are present in your system during the same week, the GLP-1 pathway gets overstimulated. This does not produce better weight loss or improved blood sugar control. It produces stronger side effects.
Think of it this way. Your GLP-1 receptors have a capacity. One medication fills that capacity effectively. Adding a second medication targeting those same receptors does not double the benefit. It overwhelms the system.
The side effects of both medications are already significant for many users on their own. Nausea, vomiting, diarrhea, constipation, and abdominal pain are the most commonly reported issues during dose titration. When both medications are active simultaneously, every one of these side effects can intensify dramatically.
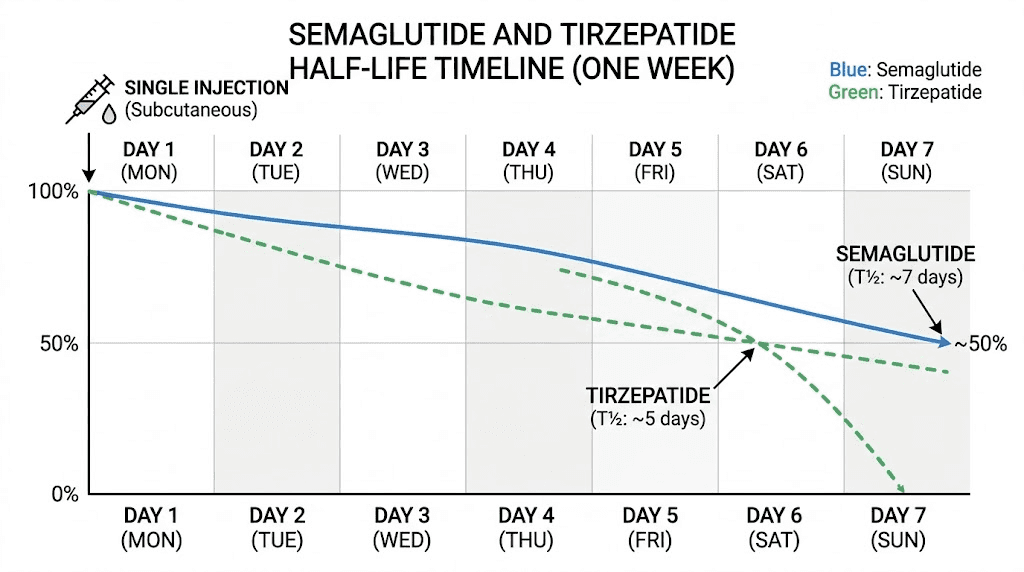
The half-life problem
This is where the "same week" question gets particularly important. Semaglutide has a half-life of approximately seven days. Tirzepatide has a half-life of roughly five days. A half-life means the time it takes for half the medication to clear your system.
After your last semaglutide injection, the medication remains pharmacologically active in your body for considerably longer than seven days. It takes approximately four to five half-lives for a drug to be effectively eliminated. For semaglutide, that means roughly 28 to 35 days of residual activity after your final dose. For tirzepatide, approximately 20 to 25 days.
If you take semaglutide on Monday and tirzepatide on Friday of the same week, both medications are at or near peak concentration simultaneously. This is the worst possible scenario for GLP-1 related side effects.
What the prescribing information says
The Wegovy (semaglutide) prescribing information explicitly states that coadministration with any other GLP-1 receptor agonist is not recommended. The Zepbound (tirzepatide) prescribing information contains the same warning. These are not suggestions. They are contraindications based on the mechanism of action and known side effect profiles of these drug classes.
No clinical trial has ever tested the combination of semaglutide and tirzepatide in the same patient during the same treatment week. There is no safety data supporting this practice. The FDA has not approved concurrent use for any indication.
What actually happens when both medications are active at once
Understanding the specific risks requires knowing what these medications do at the receptor level and how overlapping activity creates problems.
Gastrointestinal overstimulation
GLP-1 receptor activation slows gastric emptying. This is one of the primary mechanisms through which both semaglutide and tirzepatide reduce appetite and promote weight loss. Your stomach empties more slowly, you feel fuller longer, and caloric intake drops naturally.
When two GLP-1 agonists are active simultaneously, gastric emptying slows far beyond what either medication produces alone. The result is severe nausea that can last days. Vomiting becomes more likely. Bloating and abdominal distension worsen considerably.
Some patients report gastroparesis-like symptoms when overstimulation occurs, where the stomach essentially stops emptying normally for extended periods. This is not just uncomfortable. It can lead to dehydration, electrolyte imbalances, and in severe cases, hospitalization.
Blood sugar crashes
Both medications enhance insulin secretion in a glucose-dependent manner. One medication calibrates this response appropriately. Two medications stimulating the same pathway can push insulin release higher than intended, particularly after meals. For non-diabetic patients using these medications for weight loss, the risk of hypoglycemia increases substantially when both are on board.
Symptoms of hypoglycemia include shakiness, sweating, confusion, rapid heartbeat, and in extreme cases, loss of consciousness. People who take metformin alongside their GLP-1 medication face even higher risk when two agonists overlap.
Dehydration cascade
Severe nausea and vomiting from overlapping GLP-1 activity can trigger a dangerous dehydration cascade. Dehydration reduces kidney perfusion. Both semaglutide and tirzepatide have been associated with acute kidney injury in cases of severe gastrointestinal symptoms and dehydration. When both are active, the probability and severity of these gastrointestinal events increase, making kidney-related complications more likely.
This is not theoretical. Emergency departments have treated patients who combined GLP-1 agonists and experienced severe dehydration requiring IV fluid resuscitation.
The difference between taking them together and switching between them
This is the critical distinction that many people miss. Taking semaglutide and tirzepatide in the same week is different from switching from one to the other. Switching is common, medically accepted, and can be done safely with proper timing. Taking both in the same week as concurrent therapy is never recommended.
Why people consider same-week dosing
The question usually arises in a few specific scenarios.
Scenario 1: The impatient switch. Someone is transitioning from semaglutide to tirzepatide (or vice versa) and does not want to wait for the washout period. They take their last dose of one medication and start the other within days rather than waiting the recommended period.
Scenario 2: The "more is better" assumption. Someone has both medications available and reasons that if one works well, combining both should work even better. This assumption is incorrect and dangerous.
Scenario 3: The accidental overlap. Someone forgets they already took their weekly injection of one medication and takes the other. Or their provider switched their prescription mid-week without accounting for the residual activity of the first medication.
Regardless of the scenario, the biological reality is the same. Both medications will be active in your system simultaneously, creating the risks described above.
The proper way to switch medications
If your goal is to move from semaglutide to tirzepatide (the most common direction), the established protocol is straightforward.
Step 1: Take your last dose of semaglutide on your normal injection day.
Step 2: Wait at least seven days. Many providers recommend a 7 to 14 day washout period, depending on your dose level and how well you tolerated semaglutide. If you experienced significant gastrointestinal side effects on semaglutide, a longer washout is advisable.
Step 3: Begin tirzepatide at the lowest starting dose of 2.5 mg, regardless of what semaglutide dose you were on previously. This is not optional. Starting at a higher tirzepatide dose because you were on a high semaglutide dose dramatically increases side effect risk, especially with residual semaglutide still in your system.
Step 4: Follow the standard tirzepatide titration schedule, increasing dose every four weeks or longer based on tolerability.
For the reverse switch, from tirzepatide to semaglutide, the protocol is similar. Wait at least seven days after your last tirzepatide dose before starting semaglutide at 0.25 mg weekly. The semaglutide to tirzepatide conversion page on our site provides detailed dose equivalency guidance.
How long semaglutide stays in your system after your last dose
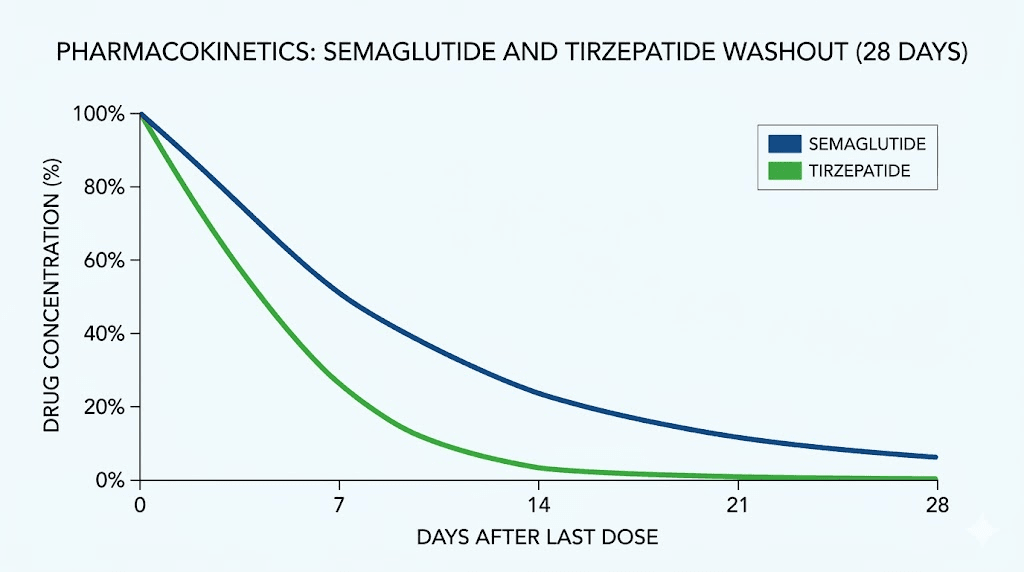
Understanding the pharmacokinetics helps explain why even a one-week gap between medications does not eliminate overlap entirely.
Semaglutide reaches peak plasma concentration approximately one to three days after injection. Its half-life of approximately seven days means that one week after your last injection, roughly 50% of the drug remains active. Two weeks out, about 25% remains. Three weeks, approximately 12.5%. Four weeks, around 6%.
Practically, this means that if you take your last semaglutide dose and start tirzepatide exactly one week later, there is still significant semaglutide activity in your body. The overlap is real, but at the seven-day mark, your provider may judge the residual level acceptable for starting a new medication at its lowest dose. This is why medical supervision matters for the transition.
For people who were on higher doses of semaglutide (1.7 mg or 2.4 mg weekly), the residual drug levels are proportionally higher. A longer washout of 10 to 14 days is often recommended in these cases. Your semaglutide dosage calculator can help you understand your current dosing level relative to maximum dose.
How long tirzepatide stays in your system
Tirzepatide has a slightly shorter half-life of approximately five days. Peak concentration occurs about 8 to 72 hours post-injection. The same pharmacokinetic principles apply.
One week after your last tirzepatide injection, approximately 38% of the drug remains active (slightly less than semaglutide because of the shorter half-life). Two weeks out, about 14%. The full duration of tirzepatide activity extends roughly three to four weeks from your final injection.
If switching from tirzepatide to semaglutide, the shorter half-life of tirzepatide means slightly less overlap risk at the seven-day mark compared to the reverse direction. But overlap still exists, and starting at the lowest semaglutide dose remains critical.
Specific scenarios and what to do in each one
Different situations call for different responses. Here is exactly what to do in the most common scenarios people face.
You accidentally took both in the same week
Do not panic, but do take this seriously. Contact your healthcare provider immediately. Monitor yourself for severe nausea, vomiting, and signs of dizziness or dehydration. Stay extremely well hydrated. Eat small, bland meals. Avoid fatty or heavy foods that would further challenge your already-compromised gastric motility.
Skip your next scheduled dose of whichever medication you plan to continue. Do not take either medication the following week. Allow the overlap to clear before resuming single-agent therapy.
Watch for warning signs that require emergency care: inability to keep fluids down for more than 24 hours, severe abdominal pain, confusion, fainting, or dark urine indicating dehydration.
Your provider switched your prescription mid-cycle
This happens more often than it should. A provider writes a new prescription for tirzepatide while you still have semaglutide injections remaining, or vice versa. The provider may not explicitly discuss the transition timing.
Do not start the new medication the same week you took your last dose of the old one. Ask your provider specifically about the washout period. A good response from your provider should include how long to wait and what starting dose to use for the new medication.
If your provider does not address washout timing proactively, bring it up yourself. This is important patient advocacy.
You want to switch because your current medication is not working
Plateaus happen with both medications. If semaglutide has plateaued and you want to try tirzepatide, or if tirzepatide has stopped working and you want to try semaglutide, a switch can be worthwhile. But rushing the transition by overlapping them in the same week will not jumpstart your progress. It will make you sick.
Research from the SURMOUNT and STEP trials shows that tirzepatide may be more effective for patients who plateau on semaglutide. The dual GIP/GLP-1 mechanism provides additional pathways for appetite suppression and metabolic improvement. But those benefits only materialize with proper dosing, not by stacking medications.
You have leftover medication and want to use it up
Some people have remaining doses of their previous medication and feel they should use them up rather than waste them. Do not alternate between medications week by week. This creates a constant state of receptor overlap where you never achieve steady-state concentration of either drug. Both alternating between semaglutide and tirzepatide and taking them in the same week carry the same fundamental risk of overstimulation.
If you have leftover medication, store it properly. Semaglutide is stable in the fridge for the duration of its shelf life. Tirzepatide similarly maintains potency when properly refrigerated. You may need it in the future if you switch back.
Why "more is not better" with GLP-1 medications
The desire to combine medications for faster results is understandable but fundamentally misguided when it comes to GLP-1 receptor agonists. Here is why.
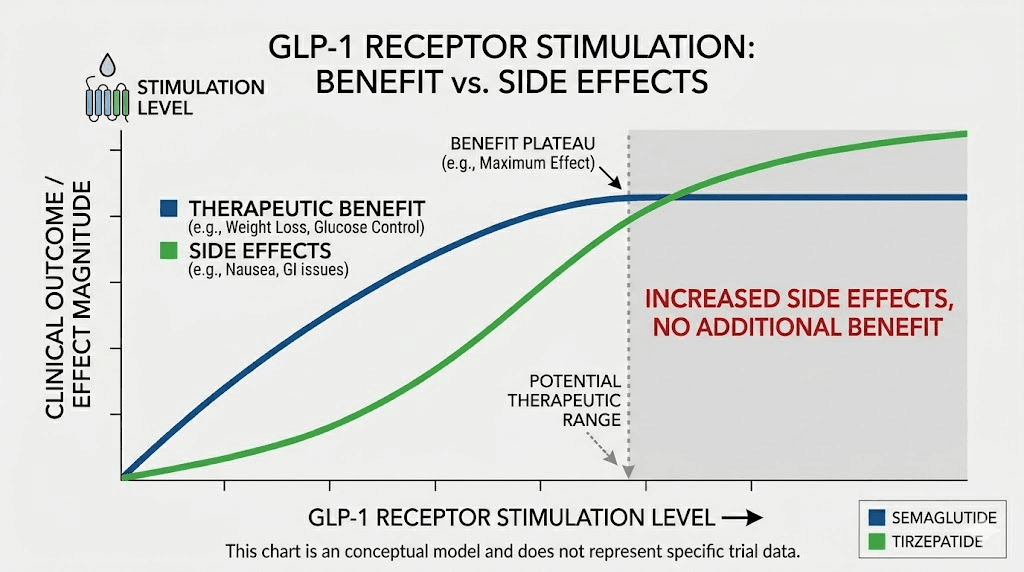
Receptor saturation is real
GLP-1 receptors can only be activated to a certain degree. Once receptor occupancy reaches a threshold, adding more agonist does not increase the biological response. It is like trying to pour water into a glass that is already full. The extra water just spills over. In biological terms, the "spillover" manifests as side effects without additional therapeutic benefit.
This is why tirzepatide was designed as a dual GIP/GLP-1 agonist rather than simply being a stronger GLP-1 agonist. By activating a second receptor type (GIP), tirzepatide achieves additional benefits through a different pathway rather than hitting the same pathway harder. Adding semaglutide on top does not provide a third pathway. It just hammers the GLP-1 pathway more aggressively.
The dose-response curve plateaus
Clinical data shows clear dose-response relationships for both medications, but the curves flatten at higher doses. The SURMOUNT-1 trial demonstrated that tirzepatide at 15 mg weekly produced about 22.5% body weight reduction. Higher doses did not proportionally increase weight loss because the biological pathways were already maximally activated.
Similarly, semaglutide at 2.4 mg weekly (the maximum approved dose for weight management) produced roughly 15% body weight reduction in the STEP trials. The appetite suppression effects plateau at a certain receptor activation level.
Combining both medications does not move you beyond these plateaus. The GLP-1 component is already saturated by either medication alone at therapeutic doses.
Side effects scale differently than benefits
Here is the cruel irony. While therapeutic benefits plateau with increased receptor stimulation, side effects continue to increase. Gastrointestinal side effects are dose-dependent and continue to worsen with increased GLP-1 activity well beyond the point where weight loss benefits stop improving.
A 2025 meta-analysis published in Annals of Saudi Medicine found that tirzepatide produced gastrointestinal events in 79.8% of participants versus 25% in the control group. Adding semaglutide on top of tirzepatide would push that number even higher while providing zero additional weight loss benefit.
What the research says about combining incretin therapies
While no study has specifically tested semaglutide plus tirzepatide in the same patient, research on incretin therapy combinations provides clear guidance.
GLP-1 agonists and DPP-4 inhibitors
DPP-4 inhibitors (like sitagliptin) work by preventing the breakdown of natural GLP-1 in the body. When patients are already on a GLP-1 receptor agonist like semaglutide, adding a DPP-4 inhibitor provides no additional benefit because the synthetic agonist already provides GLP-1 activity far exceeding what natural GLP-1 levels could achieve even without degradation.
This principle extends directly to combining two GLP-1 agonists. If one synthetic agonist already maximally activates the receptor, a second one cannot provide additional activation.
The SURPASS trials comparison data
The SURPASS-2 trial compared tirzepatide directly against semaglutide 1 mg weekly in type 2 diabetes patients. Tirzepatide at all tested doses (5 mg, 10 mg, and 15 mg) outperformed semaglutide on both HbA1c reduction and weight loss. This is important because it shows that tirzepatide alone already exceeds what semaglutide provides, making the addition of semaglutide redundant even in theory.
The comparison between semaglutide, tirzepatide, and retatrutide shows how each generation of incretin therapy improves upon the last by targeting additional receptors, not by doubling down on GLP-1 alone.
Emerging multi-agonist approaches
The pharmaceutical industry is pursuing triple agonists like retatrutide (GLP-1/GIP/glucagon receptor agonist) rather than combining existing drugs. This approach activates multiple receptor types through a single, carefully calibrated molecule. The safety profile can be studied. The dose-response relationship is predictable.
Combining two separate medications creates unpredictable pharmacokinetic interactions that no safety study has evaluated. The dose relationship between two drugs with different half-lives, different receptor binding affinities, and different distribution profiles cannot be reliably predicted from individual drug data. For comparison, mazdutide and survodutide are other next-generation multi-agonists being developed as single molecules rather than combinations.
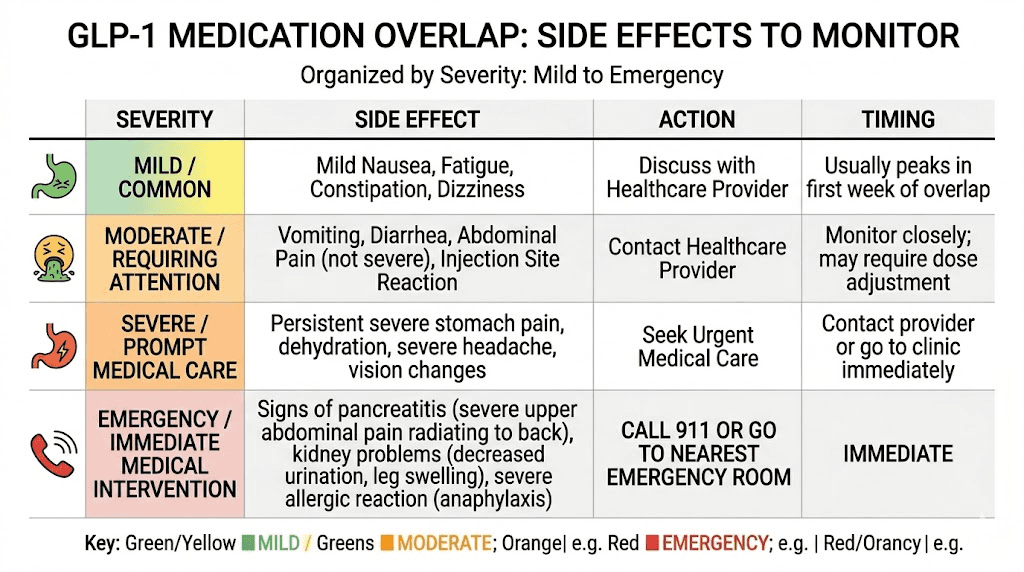
Side effects to watch for if overlap occurs
Whether accidental or intentional, if you do end up with both medications active in your system during the same week, here are the specific symptoms to monitor.
Severe gastrointestinal symptoms
Watch for nausea that does not resolve with standard remedies. The constipation or acid reflux that might be manageable with one medication can become severe with two. Vomiting that persists beyond 24 hours is a red flag. Bloating and abdominal distension may become painful rather than merely uncomfortable.
The sulfur burps that some patients experience on GLP-1 medications can worsen dramatically with overlapping doses, indicating severely delayed gastric emptying.
Neurological symptoms
Headaches are common with both medications individually. Overlapping doses can trigger more severe headaches and dizziness. This is often related to dehydration from increased gastrointestinal losses combined with reduced fluid intake from severe nausea.
Confusion or disorientation warrants immediate medical attention, as this may indicate hypoglycemia or severe dehydration.
Cardiovascular symptoms
Rapid heartbeat, especially upon standing, can indicate dehydration-related cardiovascular stress. Both medications can affect heart rate independently. Overlapping doses may cause more pronounced heart rate increases.
Fatigue and energy depletion
Both semaglutide and tirzepatide can cause fatigue on their own. The combination tends to produce profound fatigue that goes well beyond what either medication causes individually. If you are experiencing GLP-1 related fatigue that is significantly worse than your baseline, overlapping medication activity may be the cause.
Pancreatitis warning signs
While analysis has not shown a significant increase in pancreatitis risk with either medication alone in obese patients without diabetes, the risk profile of concurrent use is unknown. Severe, persistent abdominal pain that radiates to the back, especially accompanied by vomiting, should be treated as a potential pancreatitis emergency.
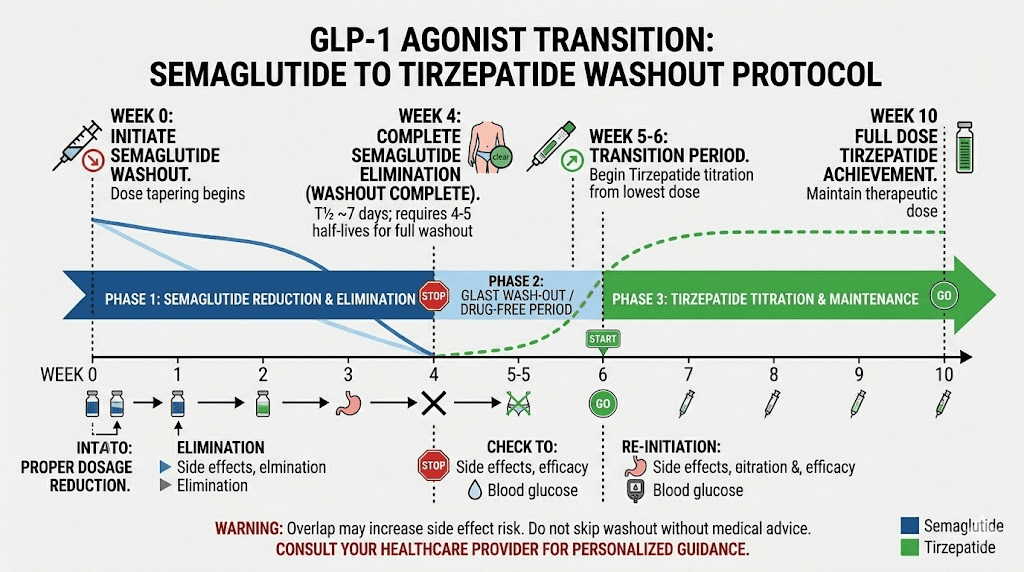
The proper washout timeline for switching between medications
If you are planning a switch between these medications, timing matters enormously. Here is the detailed protocol based on current clinical guidance.
Switching from semaglutide to tirzepatide
Minimum wait: 7 days after last semaglutide dose
Recommended wait for patients on high-dose semaglutide (1.7 mg or 2.4 mg): 10 to 14 days
Recommended wait for patients with significant GI side effects on semaglutide: 14 days
Starting dose: Always 2.5 mg tirzepatide weekly, regardless of previous semaglutide dose. The semaglutide to tirzepatide conversion chart provides equivalent dosing guidance, but initial dosing should still start at the bottom of the tirzepatide scale.
Titration: Increase by 2.5 mg every four weeks as tolerated. The standard tirzepatide titration schedule applies. Do not accelerate the titration just because you were previously on a high semaglutide dose. Your body needs to adjust to the dual GIP/GLP-1 mechanism, which affects the gut differently than GLP-1 alone.
During the washout period, you may notice some return of appetite and slight weight fluctuation. This is normal and temporary. The dietary strategies you developed during semaglutide treatment will help maintain your progress during the gap.
Switching from tirzepatide to semaglutide
Minimum wait: 7 days after last tirzepatide dose
Recommended wait for patients on high-dose tirzepatide (10 mg or above): 10 to 14 days
Starting dose: 0.25 mg semaglutide weekly. Again, this is non-negotiable regardless of your previous tirzepatide dose. Use the semaglutide dosage calculator to plan your titration schedule.
Titration: Follow the standard semaglutide titration schedule, increasing every four weeks. The semaglutide 5 mg/mL dosage chart provides detailed unit breakdowns for compounded formulations.
Many patients who switch from tirzepatide to semaglutide do so because of insurance changes, availability issues, or cost considerations. Understanding the compounded semaglutide landscape can help with this transition.
Same-day switch protocol (clinical settings only)
Some clinicians may consider a same-day switch for stable, well-tolerating patients where the risk of heightened side effects is judged by the clinician to be low. This is only appropriate when done under direct medical supervision with clear follow-up protocols in place. Same-day switching is not the same as combining both medications. It means stopping one and starting the other on the same day, at the lowest dose of the new medication.
This approach is controversial and most conservative clinical guidelines recommend against it. The one to two week washout remains the preferred approach for most patients.
Special considerations for compounded formulations
Many patients using these medications through compounding pharmacies face unique challenges when switching.
Concentration differences
Compounded semaglutide and tirzepatide come in various concentrations. Semaglutide 10 mg vials, 5 mg vials, and other formulations all deliver the same active ingredient but at different concentrations per unit drawn. When switching between medications, make sure you understand your actual milligram dosing, not just unit counts.
The same applies to tirzepatide. A compounded tirzepatide dosage chart will differ from brand-name Mounjaro auto-injector dosing. Calculate your actual weekly milligram dose before planning a switch.
Multi-ingredient compounded formulations
Some compounding pharmacies produce semaglutide or tirzepatide combined with other ingredients like B12, glycine, niacinamide, or L-carnitine. When switching between medications, you are also switching between these additional ingredients. Some of these combinations may have their own transition considerations.
For instance, if your semaglutide formulation includes B12 and your tirzepatide formulation does not, you may want to supplement B12 separately during the transition. The supplements to take with GLP-1 medications guide covers these considerations.
Storage during transitions
If you have remaining vials of your previous medication during a switch, proper storage matters. Compounded semaglutide has a specific shelf life and storage requirements. Tirzepatide similarly has a defined shelf life when stored properly. Do not waste medication by leaving it out, as you may need it if you decide to switch back.
Keep both medications properly refrigerated during and after the transition period.
Managing appetite return during the washout period
One of the biggest concerns people have about waiting between medications is the return of appetite during the gap. This is a legitimate concern, but it is manageable.
What to expect during washout
During the first few days after your last dose, you will likely still feel significant appetite suppression. Semaglutide and tirzepatide levels remain high during this period. Around days 5 to 7, depending on which medication you are coming off, appetite begins to return.
This return is not instant. It is gradual. Most people describe it as food becoming more interesting again rather than experiencing sudden, overwhelming hunger. The strategies for managing hunger during active treatment also apply during washout periods.
Dietary strategies for the gap
High-protein meals remain your best tool for appetite management during the transition. Protein provides the strongest satiety signal independent of GLP-1 activity. The protein shakes optimized for GLP-1 users work just as well during washout periods.
Fiber intake should also increase during the gap. Soluble fiber slows gastric emptying naturally, partially mimicking the GLP-1 effect. Vegetables, legumes, and whole grains become even more important during this period. The foods to avoid list for GLP-1 medications also applies during washout, as your gut is still adjusting.
Stay hydrated. Your body is transitioning out of the altered gastric motility state. Adequate hydration supports this process and prevents the headaches and cold sensations that can accompany GLP-1 medication changes.
Exercise during transition
Maintain your exercise routine during the washout. Physical activity independently supports appetite regulation, metabolic health, and weight maintenance. The relationship between exercise and GLP-1 medication shows that physical activity provides additive benefits regardless of medication status.
If anything, the washout period is a good time to slightly increase activity levels to compensate for reduced pharmaceutical appetite suppression.
When switching actually makes sense versus sticking with one medication
Not everyone who considers switching needs to switch. Before planning a transition between medications, consider whether the switch is truly warranted.
Good reasons to switch
Persistent side effects that do not resolve. If headaches on tirzepatide or insomnia on semaglutide continue despite dose optimization, the other medication may be better tolerated due to subtle receptor profile differences.
Weight loss plateau despite maximum tolerated dose. If you have been on the maximum dose of semaglutide for 12 or more weeks without continued progress, switching to tirzepatide adds the GIP receptor pathway. Conversely, some patients respond better to the pure GLP-1 approach. The troubleshooting guide for semaglutide plateaus can help determine if a switch is warranted or if other factors are at play.
Insurance or availability changes. Supply chain disruptions, formulary changes, or cost shifts may necessitate a switch regardless of clinical preference.
Provider recommendation based on lab results. Changes in HbA1c, liver enzymes, or other metabolic markers may prompt your provider to recommend a switch to the medication with a better profile for your specific situation.
Poor reasons to switch
Faster results. Switching between medications does not accelerate weight loss. Each switch requires restarting at the lowest dose and titrating up over weeks to months. You lose momentum every time you switch. The tirzepatide weight loss timeline and semaglutide week-by-week results show that consistent use of one medication produces better outcomes than switching.
Temporary side effects during titration. Nausea and gastrointestinal symptoms during dose increases are expected and usually resolve within two to three weeks at each new dose level. Switching to avoid these temporary effects just restarts the same process with a different medication.
Someone else had better results with the other medication. Individual response varies dramatically. The fact that your friend lost more weight on tirzepatide does not mean you will too. Genetic factors, metabolic differences, and lifestyle variables all play significant roles.
Frequently asked questions
Can I take a half dose of each medication in the same week to reduce the overlap risk?
No. Reducing the dose of each medication does not make same-week dosing safe. Both half-doses still activate the GLP-1 receptor pathway simultaneously, creating overstimulation. The combined effect of two half-doses is not equivalent to one full dose of either medication because the pharmacokinetic profiles, receptor binding characteristics, and metabolic pathways differ between the drugs.
My provider prescribed both medications. Is that safe?
If your provider prescribed both for concurrent use, seek a second opinion immediately. No current medical guideline supports using semaglutide and tirzepatide simultaneously. The prescribing information for both medications explicitly advises against coadministration with other GLP-1 receptor agonists. If your provider intended for you to switch from one to the other, clarify the transition protocol including washout timing.
What if I took my tirzepatide dose early and my semaglutide is due the same week?
Skip the semaglutide dose entirely. Do not take both in the same week under any circumstances. Contact your provider to determine which medication you should continue and establish a single-medication schedule going forward. The guidelines for taking tirzepatide early address timing flexibility but never support overlapping with a second GLP-1 agonist.
How long should I wait between my last semaglutide dose and first tirzepatide dose?
At minimum, seven days. For patients on higher semaglutide doses (1.7 mg or 2.4 mg weekly), 10 to 14 days is recommended. Patients with a history of significant gastrointestinal side effects should wait the full 14 days. Always start tirzepatide at the lowest dose of 2.5 mg regardless of your previous semaglutide dose level.
Will I gain weight during the washout period between medications?
Most patients experience minimal weight change during a 7 to 14 day washout period. The weight maintenance strategies used after medication discontinuation apply here. Focus on protein intake, maintaining your established eating patterns, and regular physical activity. A few days without medication will not undo months of progress.
Can I take oral semaglutide and injectable tirzepatide in the same week since they are different formulations?
No. The route of administration does not change the fundamental pharmacology. Oral semaglutide activates the same GLP-1 receptors as injectable semaglutide. Taking oral semaglutide with injectable tirzepatide creates the same overstimulation risks as combining two injectable formulations.
Are there any situations where both medications can be safely used in the same week?
No clinically validated scenario supports using both medications in the same week. This applies regardless of dose, formulation (oral dissolving tablets, injections, sublingual drops), or brand (Wegovy, Ozempic, Mounjaro, Zepbound, compounded versions). The biological mechanism of risk is the same in every case.
If tirzepatide already activates GLP-1 receptors, why would anyone add semaglutide?
They should not. Tirzepatide provides GLP-1 receptor activation as part of its dual-agonist mechanism. Adding semaglutide provides no additional receptor type activation. It simply adds redundant GLP-1 stimulation. For patients seeking enhanced results beyond what tirzepatide alone provides, the clinical path forward involves next-generation triple agonists like retatrutide, not medication stacking.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. SeekPeptides members access detailed switching protocols, dose conversion tools, and personalized guidance for safe medication transitions.
In case I do not see you, good afternoon, good evening, and good night. May your transitions stay smooth, your receptors stay balanced, and your progress stay consistent.