Mar 23, 2026

No. Tresiba is not a GLP-1 receptor agonist. Not even close. The two medications belong to completely different drug classes, work through entirely different mechanisms, and produce dramatically different effects in the body. Yet this question comes up constantly, and the confusion makes sense when you consider that both Tresiba and GLP-1 medications are injectable treatments prescribed for blood sugar management and diabetes care. They look similar on the surface. Prefilled pens. Subcutaneous injections. Prescriptions from the same endocrinologist.
But the similarities end there.
Tresiba is insulin degludec, an ultra-long-acting basal insulin analog. It replaces the insulin your body cannot produce or does not produce enough of. GLP-1 receptor agonists like semaglutide and tirzepatide work differently. They mimic a gut hormone that tells your pancreas to release insulin when blood sugar rises, while simultaneously suppressing glucagon and slowing gastric emptying. One replaces a hormone directly. The other nudges your body to produce it more effectively. Understanding this distinction matters because choosing the wrong medication, or confusing what yours does, can lead to dangerous outcomes like hypoglycemia or inadequate blood sugar control.
This guide breaks down everything you need to know about Tresiba, GLP-1 medications, how they differ, and when doctors prescribe each one. Whether you are managing type 2 diabetes and wondering about your treatment options, or you have heard about semaglutide for weight loss and want to understand where Tresiba fits in, this article covers it all. SeekPeptides is committed to making these complex medication categories clear, evidence-based, and actionable for researchers and patients alike.
What Tresiba actually is
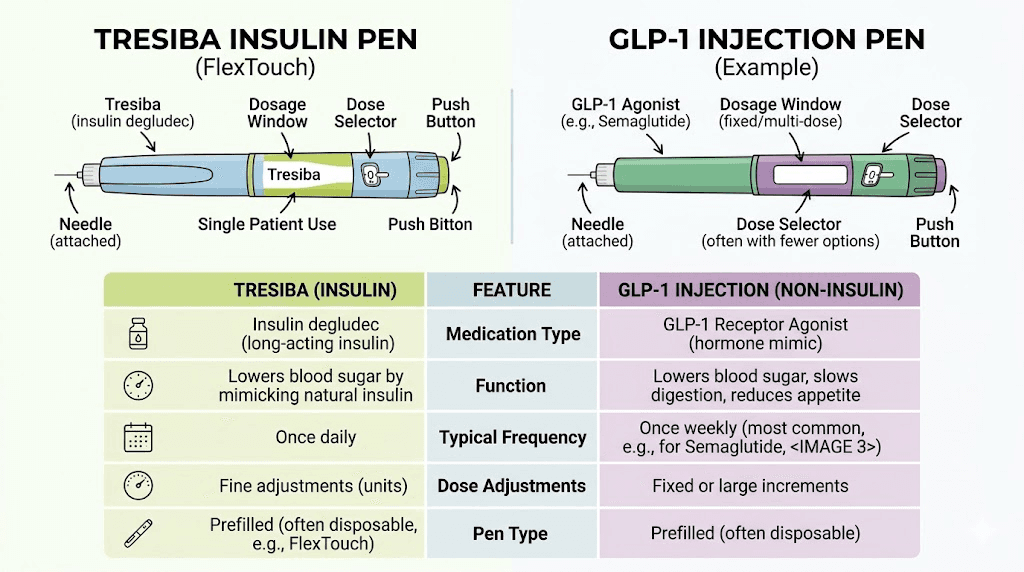
Tresiba is the brand name for insulin degludec. It is manufactured by Novo Nordisk, the same company behind Ozempic and Victoza. Approved by the FDA in 2015, Tresiba is classified as an ultra-long-acting basal insulin analog designed to provide consistent, background insulin coverage for up to 42 hours after a single injection.
That duration matters.
Most other long-acting insulins, like Lantus (insulin glargine) or Levemir (insulin detemir), last approximately 24 hours. Tresiba extends well beyond that window, which means small variations in injection timing do not cause the dramatic blood sugar swings that can occur with shorter-duration basal insulins. For people who travel frequently, work irregular schedules, or simply forget an injection occasionally, this extended duration provides a meaningful safety margin that other injectable medications do not offer.
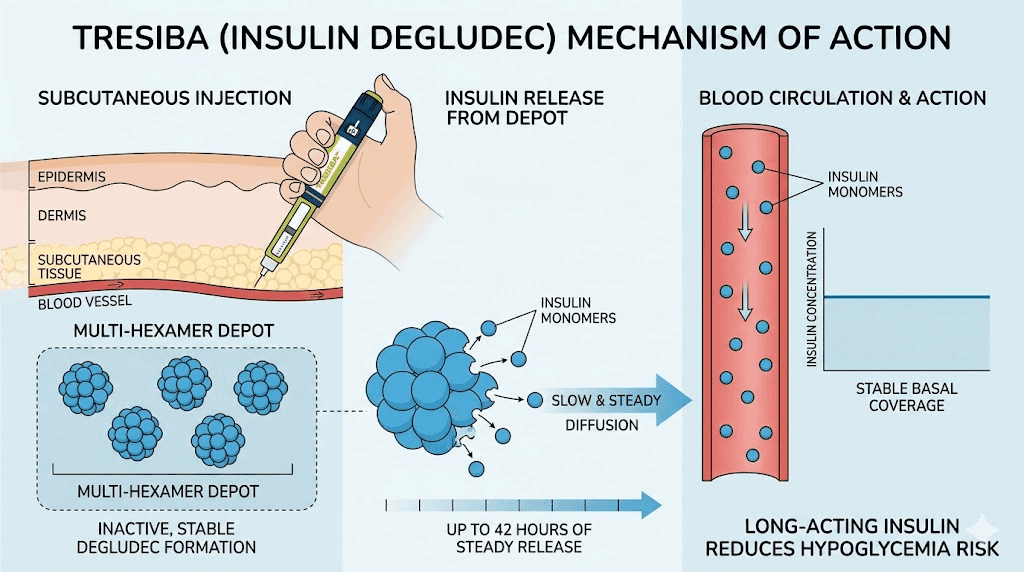
The mechanism is straightforward. After subcutaneous injection, insulin degludec forms multi-hexamer chains in the subcutaneous tissue. These chains create a depot, a reservoir of insulin that slowly releases individual monomers into the bloodstream at a steady, predictable rate. There are no peaks. No surges. Just a flat, stable background insulin level that mimics what a healthy pancreas provides between meals and overnight.
Who needs Tresiba
Tresiba is prescribed for both type 1 and type 2 diabetes. In type 1 diabetes, the pancreas produces little to no insulin, so patients need external insulin to survive. Tresiba handles the basal (background) component, while rapid-acting insulins cover meals. In type 2 diabetes, Tresiba is typically added when oral medications and lifestyle changes cannot adequately control fasting blood sugar levels.
The starting dose for insulin-naive type 2 diabetes patients is typically 10 units once daily. For type 1 patients, Tresiba usually provides 30% to 50% of total daily insulin requirements. Doses are then adjusted based on fasting blood glucose readings, typically every 3 to 4 days.
This is fundamentally different from how GLP-1 medications are dosed. There is no titration based on blood sugar with GLP-1 drugs. Their dosing follows a fixed escalation schedule designed to minimize gastrointestinal side effects.
Tresiba side effects
The side effect profile of Tresiba is characteristic of insulin therapy in general. The most significant risk is hypoglycemia, where blood sugar drops too low. Symptoms include shakiness, sweating, confusion, rapid heartbeat, and in severe cases, loss of consciousness. Weight gain is another common side effect, which stands in stark contrast to the weight loss effects of GLP-1 medications.
Other reported side effects include injection site reactions (pain, redness, swelling), low potassium levels (hypokalemia), and potential heart failure when combined with thiazolidinediones. These side effects differ considerably from the gastrointestinal side effects that characterize GLP-1 therapy.
What GLP-1 receptor agonists actually are
GLP-1 receptor agonists are a completely different class of medication. GLP-1 stands for glucagon-like peptide-1, a naturally occurring incretin hormone that your gut produces after eating. This hormone signals your pancreas to release insulin, but only when blood sugar is elevated. That glucose-dependent mechanism is the key distinction. Unlike insulin, which lowers blood sugar regardless of whether it needs lowering, GLP-1 receptor agonists only stimulate insulin release when glucose levels are actually high.
This built-in safety mechanism dramatically reduces the risk of hypoglycemia compared to direct insulin therapy like Tresiba.
The most well-known GLP-1 receptor agonists include semaglutide (sold as Ozempic for diabetes and Wegovy for weight loss), liraglutide (Victoza, Saxenda), dulaglutide (Trulicity), and exenatide (Byetta, Bydureon). Tirzepatide (Mounjaro, Zepbound) is technically a dual GIP/GLP-1 receptor agonist, hitting two incretin receptors instead of one, which explains its superior weight loss outcomes in clinical trials.
How GLP-1 medications work
GLP-1 receptor agonists produce their effects through multiple pathways simultaneously. They stimulate glucose-dependent insulin secretion from pancreatic beta cells. They suppress inappropriate glucagon release from alpha cells, which reduces liver glucose output. They slow gastric emptying, keeping food in the stomach longer and creating prolonged satiety. And they act on the brain's appetite centers, reducing hunger and food cravings.
This multi-pronged approach is why GLP-1 medications have become so popular for both diabetes management and weight loss. They do not just lower blood sugar. They address the metabolic dysfunction that drives type 2 diabetes in ways that insulin alone cannot.
The weight loss component is particularly significant. In clinical trials, semaglutide produced average weight losses of 12% to 15% of body weight. Tirzepatide results were even more impressive, with some participants losing over 20% of their body weight. Compare that to Tresiba, which typically causes weight gain of 1 to 4 kg over the treatment period.
GLP-1 side effects
The side effect profile of GLP-1 medications is dominated by gastrointestinal symptoms. Constipation, diarrhea, sulfur burps, and acid reflux are common, especially during dose escalation. Most of these symptoms improve over time as the body adjusts. Fatigue and headaches can also occur in the early weeks of treatment.
More concerning but less common side effects include gallbladder problems, potential thyroid issues (a black box warning exists for medullary thyroid carcinoma risk in animal studies), and hair loss related to rapid weight loss rather than the medication itself. SeekPeptides provides comprehensive safety guides covering all documented side effects and management strategies for researchers exploring these treatment pathways.
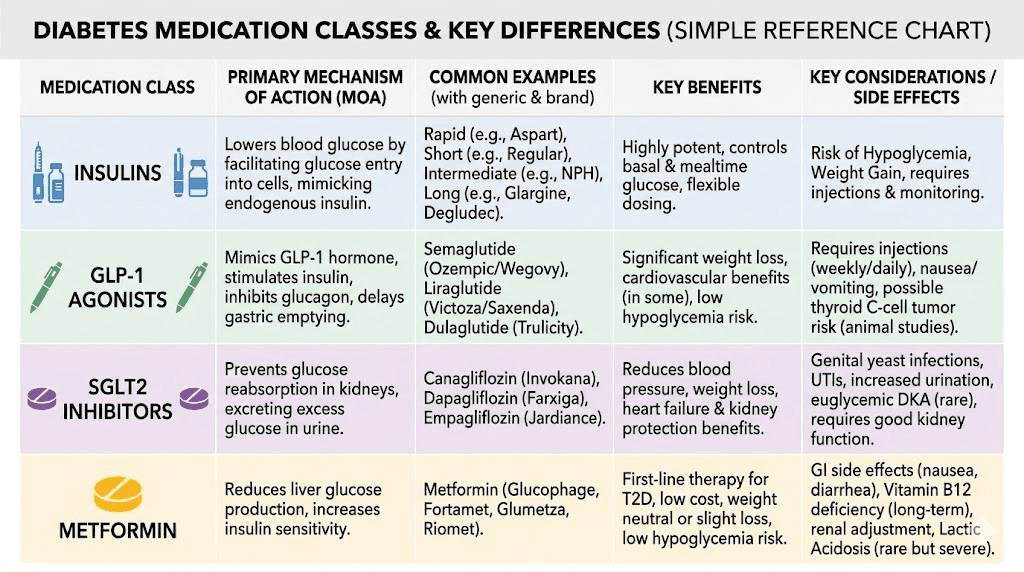
The 7 key differences between Tresiba and GLP-1 medications
The confusion between Tresiba and GLP-1 medications usually stems from surface-level similarities. Both are injectable. Both treat diabetes. Both come from the same manufacturer (Novo Nordisk makes both Tresiba and Ozempic). But the differences are fundamental, and understanding them could literally save your life if you are making treatment decisions.
1. Drug class and mechanism
Tresiba is a basal insulin analog. It directly replaces the insulin hormone. When you inject Tresiba, you are adding exogenous insulin to your bloodstream, period. It does not stimulate your pancreas or interact with gut hormones. It IS the hormone.
GLP-1 receptor agonists work indirectly. They mimic GLP-1, a gut hormone that triggers your pancreas to produce its own insulin. The distinction matters because GLP-1 medications require functioning beta cells in the pancreas to work. If your pancreas cannot produce insulin at all (as in type 1 diabetes), GLP-1 medications will not help.
2. Approved indications
Tresiba is approved for type 1 diabetes and type 2 diabetes. It serves a critical, life-sustaining role for type 1 patients who produce no insulin.
GLP-1 receptor agonists are approved primarily for type 2 diabetes and, in some formulations, for weight management. They are not approved for type 1 diabetes because they depend on residual beta cell function. Semaglutide (as Wegovy) and tirzepatide (as Zepbound) carry additional approvals specifically for chronic weight management in adults with obesity or overweight with at least one weight-related comorbidity.
3. Dosing frequency
Tresiba is injected once daily. Its 42-hour duration provides flexibility, but daily injection is the standard protocol.
Most modern GLP-1 medications are injected once weekly. Semaglutide and tirzepatide both follow weekly dosing schedules. Older GLP-1 medications like exenatide (Byetta) required twice-daily dosing, but this is no longer the standard. The weekly dosing convenience is one reason GLP-1 medications have gained such widespread adoption, with many patients preferring one injection per week to the daily injections that insulin protocols require.
4. Effect on body weight
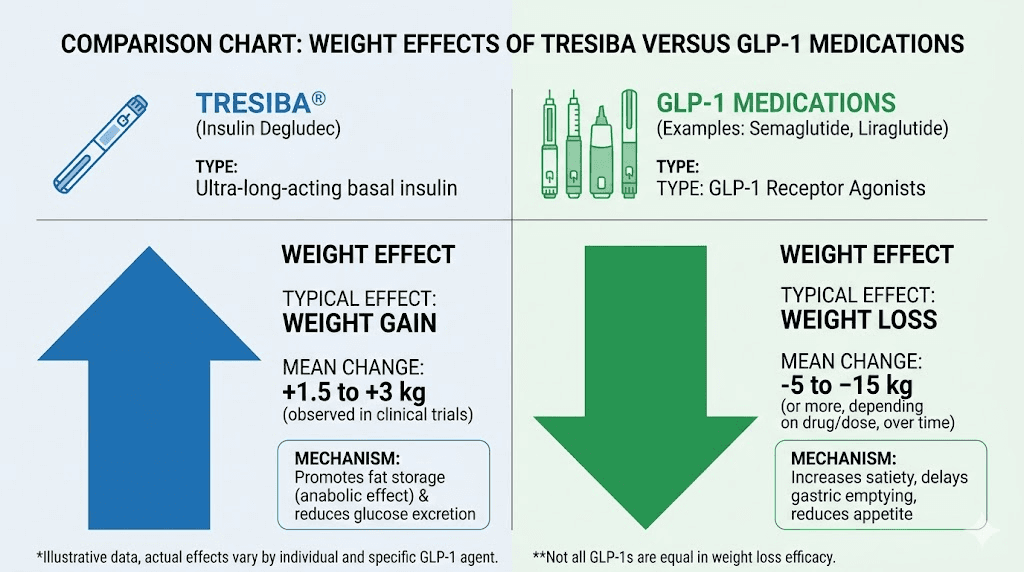
This is perhaps the most dramatic difference. Tresiba commonly causes weight gain. Insulin promotes glucose uptake into cells, including fat cells, and the improved metabolic control often leads to increased caloric storage. Clinical trials showed average weight gains of 1 to 4 kg with Tresiba use.
GLP-1 medications cause significant weight loss. The appetite suppression, slowed gastric emptying, and central nervous system effects combine to reduce caloric intake by 20% to 40% in many patients. Semaglutide results show progressive weight loss over 68 weeks, with most patients losing 10% to 15% of their starting weight. Tirzepatide weight loss timelines show even greater reductions.
5. Hypoglycemia risk
Tresiba carries a meaningful risk of hypoglycemia. Because it provides a constant level of insulin regardless of blood sugar status, blood glucose can drop dangerously low if a patient skips meals, exercises vigorously, or takes too much insulin. Though Tresiba has a lower hypoglycemia rate than some other insulins due to its flat action profile, the risk is always present.
GLP-1 medications have a very low risk of hypoglycemia when used alone. Their glucose-dependent mechanism means they only stimulate insulin release when blood sugar is elevated. When blood sugar drops to normal levels, the insulin-stimulating effect turns off. This built-in safeguard makes GLP-1 medications significantly safer from a hypoglycemia standpoint, though the risk increases when combined with insulin or sulfonylureas.
6. Cardiovascular effects
GLP-1 receptor agonists have demonstrated cardiovascular benefits in large outcome trials. Semaglutide reduced major adverse cardiovascular events by 26% in the SUSTAIN-6 trial. Liraglutide showed similar benefits in the LEADER trial. These cardiovascular advantages have made GLP-1 medications a preferred choice for type 2 diabetes patients with established cardiovascular disease.
Tresiba has been shown to be cardiovascular-safe (non-inferior to placebo) in the DEVOTE trial, but it has not demonstrated the cardiovascular benefit that GLP-1 medications provide. It does not make heart disease worse, but it does not actively protect against it either.
7. Effect on appetite and eating behavior
Tresiba has no effect on appetite or eating behavior. It is purely a glucose-lowering hormone replacement.
GLP-1 medications profoundly alter appetite and food preferences. Patients commonly report reduced hunger, earlier satiety, decreased cravings for high-calorie foods, and sometimes complete food indifference. This appetite modulation occurs through both peripheral mechanisms (slowed gastric emptying) and central mechanisms (direct effects on hypothalamic appetite centers). Understanding why some people still feel hungry on GLP-1 medications requires examining individual factors like dosing, timing, and metabolic status.
Why people confuse Tresiba with GLP-1 medications
The confusion is understandable. Several factors contribute to this common misconception.
Both are injectable. For decades, injectable diabetes medications meant insulin. Period. Now that GLP-1 receptor agonists have entered the market as injectable treatments, patients naturally assume all injectable diabetes medications work the same way. They do not.
Same manufacturer. Novo Nordisk makes both Tresiba and Ozempic (semaglutide). The company also makes Victoza (liraglutide), another GLP-1. When patients see the same pharmaceutical company name on different medications, they may assume the drugs are related or work similarly.
Similar pen devices. Both Tresiba and several GLP-1 medications come in prefilled pen devices that look nearly identical. The injection technique is the same. Subcutaneous injection into the abdomen, thigh, or upper arm using a small needle. Without understanding the pharmacology, the physical similarity reinforces the misconception.
Media coverage overlap. The explosion of GLP-1 media coverage has created confusion about what falls under the GLP-1 umbrella. People hear about GLP-1 options and injectable diabetes treatments in the same conversations, leading to category conflation.
Combination products exist. Xultophy, another Novo Nordisk product, combines insulin degludec (the active ingredient in Tresiba) with liraglutide (a GLP-1 receptor agonist) in a single injection. This combination product further blurs the line between the two drug classes. If your doctor mentions Tresiba and GLP-1 in the same sentence when discussing Xultophy, it is easy to conclude they are the same thing.
Tresiba and GLP-1 combination therapy
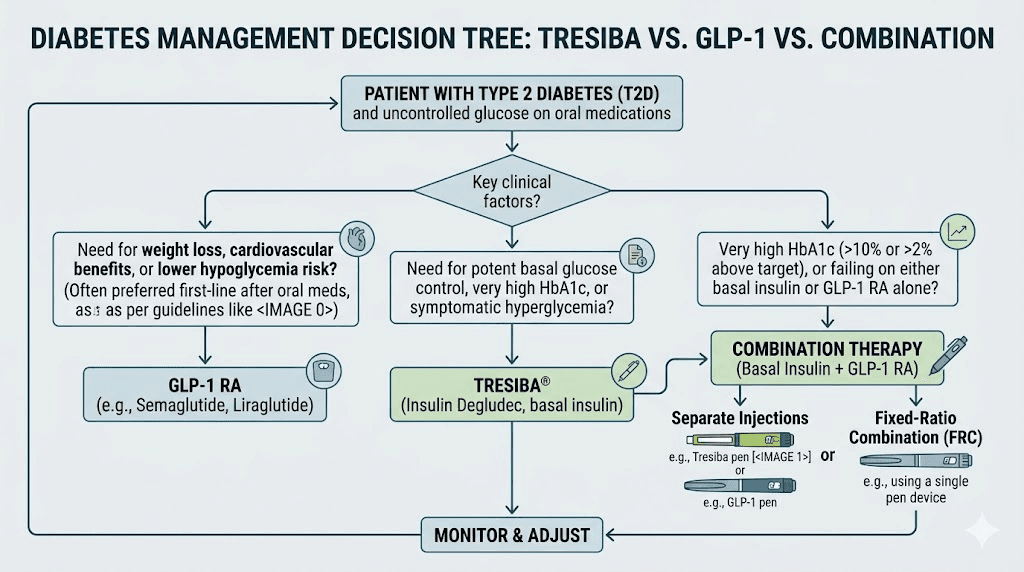
While Tresiba is not a GLP-1, the two drug classes are frequently prescribed together. This combination leverages the strengths of both medications while minimizing their individual weaknesses.
Why combining makes sense
Insulin and GLP-1 receptor agonists are complementary. Insulin provides reliable, consistent basal glucose control. GLP-1 medications add postprandial (after-meal) glucose control through glucose-dependent insulin secretion, glucagon suppression, and delayed gastric emptying. The GLP-1 component also counteracts the weight gain that insulin causes, creating a more balanced metabolic profile.
Clinical data supports this approach. The DUAL studies showed that patients receiving the combination of insulin degludec and liraglutide achieved better A1C reduction with less hypoglycemia and less weight gain compared to either drug alone. The odds of hitting an A1C below 7% without hypoglycemia or weight gain were significantly improved with combination therapy.
How the combination is prescribed
Two approaches exist for combining basal insulin with GLP-1 therapy.
The first is using separate injections. A patient takes Tresiba daily and a GLP-1 medication like tirzepatide or semaglutide weekly. This allows independent dose adjustments for each medication.
The second is using Xultophy, the fixed-ratio combination product containing insulin degludec and liraglutide in a single pen. Each unit of Xultophy delivers 1 unit of insulin degludec and 0.036 mg of liraglutide. This approach simplifies the regimen to a single daily injection but limits dosing flexibility.
When adding Tresiba to an existing GLP-1 regimen, the typical starting dose is 10 units daily. When adding a GLP-1 to an existing Tresiba regimen, most guidelines recommend reducing the Tresiba dose by approximately 20% to avoid hypoglycemia as the GLP-1 begins its own glucose-lowering effects.
Comparing Tresiba to specific GLP-1 medications
Understanding how Tresiba compares to each major GLP-1 medication clarifies why these are fundamentally different treatments serving different purposes.
Tresiba vs semaglutide (Ozempic)
Semaglutide is the most prescribed GLP-1 receptor agonist worldwide. It is injected once weekly at doses ranging from 0.25 mg (starting dose) to 2.0 mg (maximum dose for diabetes). Semaglutide typically starts working within the first week, with full effects developing over 4 to 5 weeks as the dose escalates.
Tresiba is injected daily, with doses measured in units rather than milligrams. A type 2 diabetes patient might take anywhere from 10 to 80+ units daily, adjusted based on fasting glucose readings. The dose-response relationship is entirely different from semaglutide, where dosing follows a fixed titration schedule.
The clinical outcomes differ dramatically. Semaglutide reduces A1C by approximately 1.5% to 1.8% while producing significant weight loss. Tresiba reduces A1C by approximately 1.0% to 1.3% but causes modest weight gain. For patients whose primary concern is weight management alongside blood sugar control, semaglutide offers clear advantages in that regard.
Tresiba vs tirzepatide (Mounjaro)
Tirzepatide is a dual GIP/GLP-1 receptor agonist that has produced the most impressive glycemic and weight loss results of any diabetes medication to date. In the SURPASS trials, tirzepatide at its highest dose (15 mg weekly) reduced A1C by approximately 2.4%, with average weight losses exceeding 12 kg.
Comparing that to Tresiba highlights the fundamental difference in what these medications do. Tresiba replaces insulin. Tirzepatide enhances the body's own metabolic responses through dual incretin receptor activation. For type 2 diabetes patients with significant insulin resistance and obesity, tirzepatide may address the root metabolic dysfunction in ways that exogenous insulin cannot.
However, Tresiba remains essential for patients who need direct insulin replacement, particularly those with type 1 diabetes or advanced type 2 diabetes with significant beta cell failure.
Tresiba vs liraglutide (Victoza)
Liraglutide holds particular relevance to the Tresiba discussion because the two are combined in Xultophy. As a standalone GLP-1, liraglutide differs from semaglutide in several ways. It is injected daily rather than weekly, has a shorter half-life (approximately 13 hours versus 7 days for semaglutide), and produces somewhat less weight loss.
The daily dosing of both Tresiba and liraglutide probably contributes to the confusion. If a patient takes both medications daily via pen injections, distinguishing between them becomes harder without understanding the underlying pharmacology. But the mechanisms remain completely distinct. Tresiba provides exogenous insulin. Liraglutide stimulates endogenous insulin production while offering appetite suppression and cardiovascular protection.
Other medications commonly confused with GLP-1s
Tresiba is not the only medication that people mistake for a GLP-1 receptor agonist. The diabetes medication landscape has become increasingly complex, and several drug classes share surface-level similarities that lead to confusion.
Other insulins
All insulin medications, including Lantus, Levemir, Humalog, NovoLog, and Tresiba, are distinct from GLP-1 receptor agonists. They provide the insulin hormone directly rather than stimulating its production. Some patients on insulin therapy hear about GLP-1 medications and wonder if their insulin injections are GLP-1s. They are not.
Phentermine
Phentermine is also not a GLP-1. It is a sympathomimetic amine that suppresses appetite through central nervous system stimulation. While it shares the weight loss indication with some GLP-1 medications, the mechanism and side effect profile are entirely different. Phentermine vs GLP-1 comparisons reveal significant differences in safety, efficacy, and duration of use.
Metformin
Metformin is an oral diabetes medication that reduces hepatic glucose production. It is not an injection, not a peptide, and not a GLP-1 receptor agonist. However, patients often ask whether metformin can be taken with GLP-1 medications or combined with tirzepatide. The answer is yes, as they work through different pathways and complement each other well.
SGLT2 inhibitors
Drugs like empagliflozin (Jardiance) and dapagliflozin (Farxiga) are sodium-glucose cotransporter 2 inhibitors. They work by blocking glucose reabsorption in the kidneys, causing excess glucose to be excreted in urine. Like GLP-1 medications, they can promote modest weight loss and provide cardiovascular benefits, but they work through an entirely different mechanism.
When doctors prescribe Tresiba instead of GLP-1 medications
Understanding when each medication is appropriate helps clarify why they are not interchangeable.
Type 1 diabetes
Tresiba (or another basal insulin) is essential for type 1 diabetes management. GLP-1 medications are not approved and generally not effective for type 1 diabetes because the autoimmune destruction of beta cells means there are no functioning insulin-producing cells for GLP-1 to stimulate. Tresiba provides the background insulin these patients need to survive.
Advanced type 2 diabetes with beta cell failure
As type 2 diabetes progresses, the pancreatic beta cells can become exhausted or destroyed. When this happens, GLP-1 medications lose their effectiveness because there are insufficient beta cells to respond to the GLP-1 signal. At this stage, exogenous insulin like Tresiba becomes necessary regardless of other medications being used.
When GLP-1 medications are contraindicated
Some patients cannot tolerate GLP-1 medications due to severe gastrointestinal side effects, a personal or family history of medullary thyroid carcinoma, or active gallbladder disease. For these patients, insulin therapy with Tresiba may be the preferred option for glucose control.
Pregnancy and breastfeeding
Insulin is the preferred diabetes treatment during pregnancy because it does not cross the placenta and has decades of safety data. GLP-1 medications are contraindicated in pregnancy. Researchers exploring GLP-1 use during breastfeeding face similar restrictions, with insulin remaining the safer choice.
When doctors prescribe GLP-1 medications instead of Tresiba
The trend in type 2 diabetes management has shifted toward earlier GLP-1 use, often before insulin is considered.
Early to moderate type 2 diabetes
For patients with type 2 diabetes who have not achieved adequate control with metformin alone, GLP-1 receptor agonists are increasingly the second-line choice. Their ability to lower blood sugar, promote weight loss, and reduce cardiovascular risk makes them attractive as early interventions. Many endocrinologists now prescribe semaglutide or tirzepatide before considering insulin therapy.
Weight management in type 2 diabetes
For type 2 diabetes patients who are overweight or obese (which describes the majority), GLP-1 medications address both the glycemic and the weight components of their disease. Since excess weight worsens insulin resistance, the weight loss produced by GLP-1 medications actually improves the underlying metabolic dysfunction. Tresiba addresses the symptom (high blood sugar) without addressing the root cause (insulin resistance driven by excess adiposity).
Understanding dietary approaches during GLP-1 therapy and knowing caloric targets while on semaglutide can maximize the metabolic benefits of GLP-1 treatment.
Cardiovascular risk reduction
For type 2 diabetes patients with established cardiovascular disease, GLP-1 receptor agonists are preferred over insulin due to their proven cardiovascular benefits. Guidelines from the American Diabetes Association and the European Association for the Study of Diabetes recommend GLP-1 medications as preferred agents for patients with atherosclerotic cardiovascular disease, heart failure, or chronic kidney disease.
Weight loss without diabetes
GLP-1 medications have expanded beyond diabetes treatment entirely. Higher-dose formulations of semaglutide (Wegovy, 2.4 mg weekly) and tirzepatide (Zepbound, up to 15 mg weekly) are approved specifically for chronic weight management in non-diabetic patients. Tresiba has no role in weight management. It would be harmful to give insulin to a non-diabetic person for weight loss, as it would cause dangerous hypoglycemia.
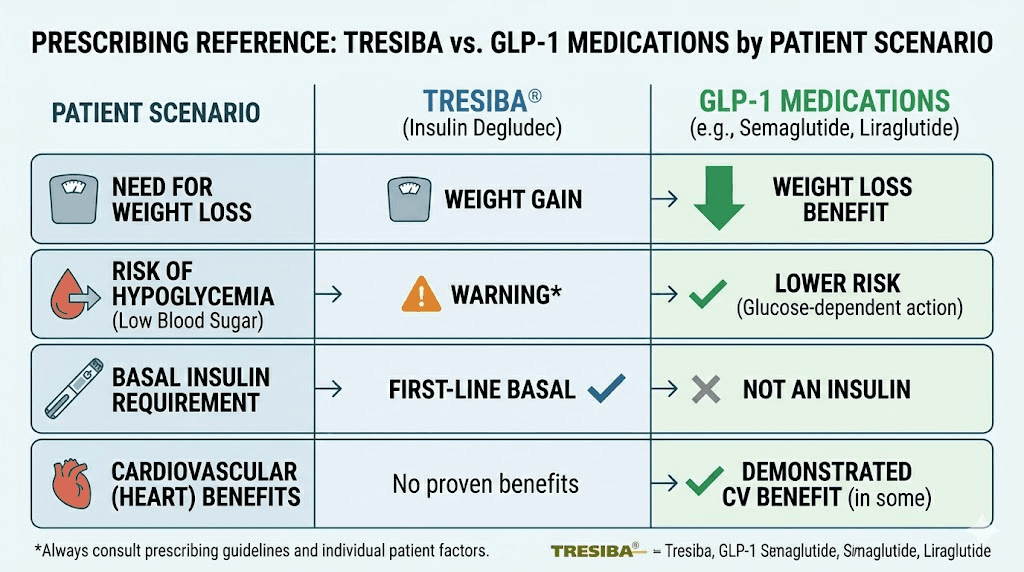
The complete comparison table
Feature | Tresiba (Insulin Degludec) | GLP-1 Receptor Agonists |
|---|---|---|
Drug class | Ultra-long-acting basal insulin | Incretin mimetic |
Mechanism | Directly replaces insulin hormone | Stimulates endogenous insulin release, suppresses glucagon, slows gastric emptying |
Dosing frequency | Once daily | Once weekly (most modern formulations) |
Weight effect | Weight gain (1-4 kg) | Weight loss (5-20% body weight) |
Hypoglycemia risk | Moderate (lower than older insulins) | Very low (when used alone) |
Cardiovascular benefit | Neutral (safe, but no benefit) | Proven benefit (reduced MACE events) |
Type 1 diabetes | Approved and essential | Not approved |
Type 2 diabetes | Approved (often later-line) | Approved (increasingly first-line after metformin) |
Weight management | Not indicated | Approved (Wegovy, Zepbound) |
Appetite effect | None | Significant appetite suppression |
Duration of action | Up to 42 hours | Varies (13 hours to 7+ days) |
Common side effects | Hypoglycemia, weight gain, injection site reactions | Nausea, diarrhea, constipation, injection site reactions |
Manufacturer | Novo Nordisk | Novo Nordisk (semaglutide), Eli Lilly (tirzepatide), others |
This table makes the distinction clear. These are not variations of the same drug class. They are fundamentally different medications with different mechanisms, different outcomes, and different patient populations.
Understanding insulin versus incretin-based therapies
To fully grasp why Tresiba is not a GLP-1, it helps to understand the broader categories of diabetes medications and how they relate to each other.
The insulin pathway
Insulin is a peptide hormone produced by the beta cells of the pancreatic islets of Langerhans. It is the primary anabolic hormone in the body, facilitating glucose uptake into muscle, fat, and liver cells while inhibiting glucose production by the liver. Without insulin, cells cannot access glucose for energy, and blood sugar rises to dangerous levels.
Tresiba and all other insulin medications simply provide this hormone exogenously. Whether rapid-acting (Humalog, NovoLog), intermediate-acting (NPH), or long-acting (Lantus, Levemir, Tresiba), the fundamental job is the same. Put insulin into the bloodstream so cells can absorb glucose.
The incretin pathway
Incretins are gut hormones released after eating. The two major incretins are GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide). These hormones account for approximately 50% to 70% of the insulin response after a meal, a phenomenon called the incretin effect.
In type 2 diabetes, the incretin effect is impaired. The body produces less GLP-1, and the remaining GLP-1 is rapidly degraded by the enzyme DPP-4. GLP-1 receptor agonists bypass this problem by providing synthetic GLP-1 analogs that are resistant to DPP-4 degradation, restoring the incretin response.
This is why researchers at SeekPeptides emphasize understanding the mechanistic differences between drug classes. The incretin pathway and the direct insulin pathway are both essential for glucose homeostasis, but they represent fundamentally different therapeutic approaches.
Can you switch from Tresiba to a GLP-1 medication?
This is a common question, and the answer depends on the individual clinical situation.
When switching is possible
Some type 2 diabetes patients who were started on insulin like Tresiba may be candidates for transitioning to or adding a GLP-1 medication. This is particularly true for patients who were started on insulin early in their disease course when their beta cells still had significant function, gained weight on insulin therapy, have A1C levels that might respond to GLP-1-mediated insulin secretion, or want the cardiovascular and weight loss benefits that GLP-1 medications offer.
The transition must be medically supervised. Abruptly stopping insulin can cause dangerous hyperglycemia. Typically, the GLP-1 medication is started first and titrated up, while the insulin dose is gradually reduced under close blood glucose monitoring.
When switching is not possible
Patients with type 1 diabetes cannot switch from insulin to a GLP-1 medication. They need exogenous insulin to survive. Similarly, type 2 patients with significant beta cell failure will still require insulin regardless of GLP-1 therapy. For these patients, the combination approach discussed earlier is more appropriate than a complete switch.
Resources like the guide on switching between GLP-1 medications and understanding whether GLP-1 medications can be combined provide additional context for patients navigating treatment transitions.
The future of combined insulin and GLP-1 therapy
The line between insulin and GLP-1 therapy is becoming increasingly blurred, not because they are the same thing, but because they are increasingly used together.
Fixed-ratio combinations
Xultophy (insulin degludec + liraglutide) represents the current state of combined therapy. Future products may combine basal insulin with newer, more potent GLP-1 or dual GIP/GLP-1 receptor agonists. These combination products simplify treatment by reducing injection burden while providing the benefits of both drug classes.
Co-formulations under development
Pharmaceutical companies are exploring co-formulations that combine ultra-long-acting insulin with once-weekly GLP-1 receptor agonists. These products would allow weekly dosing for both components, dramatically simplifying diabetes management for patients who need both basal insulin and incretin-based therapy.
Implications for treatment selection
As combination products become more common, the distinction between insulin and GLP-1 therapy becomes more important, not less. Patients need to understand what each component does, why it was prescribed, and how to manage the different side effect profiles. A combination product does not make insulin into a GLP-1 or vice versa. It simply delivers both medications simultaneously.
Managing diabetes medications safely
Whether you take Tresiba, a GLP-1 medication, or both, safe management requires understanding what each medication does and how to use it properly.
For Tresiba users
Monitor fasting blood glucose regularly. Adjust doses gradually (every 3-4 days). Never skip doses without medical guidance. Carry glucose tablets or another fast-acting carbohydrate source for hypoglycemia. Learn the signs of low blood sugar (shakiness, sweating, confusion, rapid heartbeat). Store Tresiba properly, as it is a temperature-sensitive biological product that requires refrigeration before first use.
For GLP-1 users
Follow the dose escalation schedule. Do not increase doses faster than prescribed, as this worsens gastrointestinal side effects. Take on the same day each week for weekly formulations. Learn about optimal nutrition during GLP-1 therapy. Stay hydrated, especially if experiencing nausea or diarrhea. Report any severe or persistent side effects to your healthcare provider promptly. Understand injection site reactions and proper injection site rotation.
For combination therapy users
Understand which medication does what. Know that the insulin component creates hypoglycemia risk while the GLP-1 component causes gastrointestinal effects. Track both fasting and postprandial blood sugars. Never adjust one medication without considering the impact on the other. Home monitoring tools can help track glucose trends and identify patterns that guide dosing decisions.
What to tell your doctor
If you arrived at this article because you were unsure whether your Tresiba prescription is a GLP-1 medication, here is what to discuss with your healthcare provider.
Ask specifically what drug class each of your medications belongs to. Ask why insulin was chosen over a GLP-1 medication, or vice versa. If you are on Tresiba and interested in the weight loss benefits of GLP-1 medications, ask whether adding a GLP-1 is appropriate for your clinical situation. If you are experiencing side effects, make sure you are attributing them to the correct medication. Constipation is a GLP-1 side effect, not an insulin side effect. Hypoglycemia is an insulin risk, not typically a GLP-1 risk.
Understanding these distinctions empowers you to have more productive conversations with your medical team and make more informed decisions about your treatment. SeekPeptides members access detailed medication guides, protocol comparisons, and expert-reviewed resources that help navigate these complex treatment landscapes with confidence.

Frequently asked questions
Is Tresiba an Ozempic alternative?
No. Tresiba and Ozempic are completely different medications. Tresiba is a basal insulin that replaces the insulin hormone, while Ozempic is a GLP-1 receptor agonist that stimulates your body to produce its own insulin. They serve different clinical purposes and cannot substitute for each other.
Does Tresiba cause weight loss like GLP-1 medications?
No. Tresiba typically causes weight gain of 1 to 4 kg, which is the opposite of GLP-1 medications that produce significant weight loss. If weight management is a priority alongside blood sugar control, GLP-1 medications offer a clear advantage in that area.
Can I take Tresiba and a GLP-1 at the same time?
Yes. Combining basal insulin with a GLP-1 receptor agonist is a well-established treatment strategy for type 2 diabetes. The combination product Xultophy contains both insulin degludec (Tresiba) and liraglutide (a GLP-1) in a single injection. Alternatively, Tresiba and a GLP-1 can be taken as separate injections.
Is Tresiba better than a GLP-1 for type 2 diabetes?
It depends on the individual situation. For early to moderate type 2 diabetes, GLP-1 medications are increasingly preferred due to weight loss benefits, lower hypoglycemia risk, and cardiovascular protection. For advanced type 2 diabetes with significant beta cell failure, or for type 1 diabetes, Tresiba (or another insulin) is essential and often superior to or required in addition to GLP-1 therapy.
Why did my doctor prescribe Tresiba instead of Ozempic?
Your doctor may have prescribed Tresiba because you have type 1 diabetes, your beta cells are no longer producing enough insulin for GLP-1 medications to be effective, you have contraindications to GLP-1 therapy, or your blood sugar requires the reliable, direct glucose-lowering that only exogenous insulin provides. Discuss your specific situation with your provider if you have questions about your treatment plan.
Is insulin degludec the same as semaglutide?
No. Insulin degludec (Tresiba) is a synthetic insulin analog. Semaglutide (Ozempic, Wegovy) is a GLP-1 receptor agonist. They are different molecules, different drug classes, and work through completely different mechanisms. Both are made by Novo Nordisk, which may contribute to the confusion.
Does Tresiba suppress appetite?
No. Tresiba has no effect on appetite or eating behavior. Appetite suppression is a hallmark of GLP-1 receptor agonists, which act on brain appetite centers and slow gastric emptying. If you are looking for appetite suppression, that is a GLP-1 effect, not an insulin effect.
Can Tresiba be used for weight loss?
No. Tresiba should never be used for weight loss. It is an insulin medication that typically causes weight gain. Using insulin in a non-diabetic person for weight loss purposes would cause dangerous hypoglycemia and is medically inappropriate. For weight management, approved GLP-1 medications like semaglutide (Wegovy) and tirzepatide (Zepbound) are the evidence-based options.
External resources
For researchers serious about understanding the full landscape of peptide-based therapies, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, detailed medication comparisons, and a community of thousands who navigate these complex treatment decisions every day.
In case I do not see you, good afternoon, good evening, and good night. May your blood sugar stay stable, your medications stay clear, and your treatment decisions stay informed.