Feb 13, 2026

Cagrilintide sits at the center of the next wave in weight management research. A long-acting amylin analogue with an eight-day half-life, once-weekly dosing, and clinical trial data showing 11.8% body weight reduction as a standalone compound. The REDEFINE 1 trial published in the New England Journal of Medicine found that when paired with semaglutide, participants lost 22.7% of their body weight over 68 weeks. Sixty percent of those participants crossed the 20% threshold. Numbers like those have researchers paying attention.
But paying attention and actually sourcing high-quality cagrilintide for laboratory work are two very different things.
The research peptide market has exploded alongside public interest in GLP-1 agonists and weight loss peptides. That growth has brought legitimate suppliers to the table. It has also attracted companies cutting corners on purity, mislabeling vials, and shipping degraded product. When you are working with a compound as structurally complex as cagrilintide, a 39-amino-acid acylated peptide with specific mutations designed to prevent fibril formation, quality is not optional. One bad vial can invalidate weeks of work.
This guide covers everything a researcher needs before purchasing cagrilintide. We will walk through the science behind the compound, what separates a reliable vendor from a risky one, how to verify purity through third-party testing, proper storage and handling protocols, legal considerations, dosing frameworks from published literature, and how cagrilintide fits into the broader landscape alongside semaglutide, tirzepatide, and retatrutide. SeekPeptides has tracked the development of this compound since its earliest Phase 2 data, and the research community interest has only accelerated.
Whether you are evaluating cagrilintide for the first time or comparing vendors for your next order, this is the resource you will want bookmarked.
What is cagrilintide and why researchers want it
Before spending money on any research compound, understanding exactly what you are purchasing matters. Cagrilintide is not just another weight loss peptide. It represents an entirely different mechanism class from the GLP-1 receptor agonists that dominate current conversations.
The amylin pathway explained
Amylin is a 37-amino-acid peptide hormone co-secreted with insulin from pancreatic beta cells after meals. Think of it as insulin's quieter partner. While insulin handles glucose uptake, amylin tackles the appetite side of the equation. It slows gastric emptying, suppresses post-meal glucagon release, and signals satiety to brain regions involved in both homeostatic and hedonic appetite regulation.
The problem with natural amylin? It aggregates into fibrils within minutes. You cannot inject native human amylin and expect it to work therapeutically. The molecule self-destructs before it reaches meaningful circulating concentrations.
Pramlintide, the only FDA-approved amylin analogue until now, solved partial fibril formation issues but requires multiple daily injections. Its short half-life means constant dosing. Researchers using pramlintide know the frustration of three-times-daily administration schedules that make long-term protocol adherence difficult.
Cagrilintide changed the game completely.
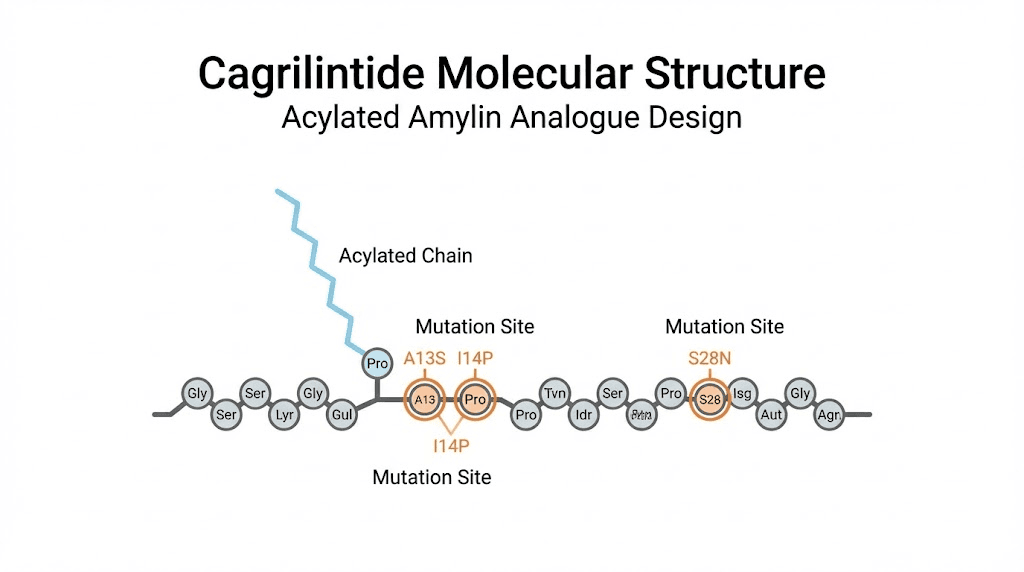
How Novo Nordisk engineered stability
The development team at Novo Nordisk published their approach in the Journal of Medicinal Chemistry, and the engineering is elegant. They started with the human amylin sequence and made strategic modifications. The 14E/17R mutations stabilize the central helix through a salt bridge. The 25P/28P/29P mutations, borrowed from rat amylin, reduce beta-sheet propensity and prevent the fibril formation that destroys native human amylin. A C-terminal proline improves potency specifically at the calcitonin receptor. And critically, an N-terminally linked C20 fatty acid chain enables reversible albumin binding.
That albumin binding is what gives cagrilintide its 159-195 hour half-life. Roughly eight days. Once-weekly dosing becomes not just possible but optimal. Compare that to pramlintide requiring injections before every meal, and you understand why the research community considers cagrilintide a generational leap forward in amylin receptor pharmacology.
The compound acts as a non-selective agonist at both amylin receptors (AMY1R and AMY3R) and calcitonin receptors (CTR). Recent research published in eBioMedicine confirmed that cagrilintide specifically requires AMY1R and AMY3R activation to produce its weight-lowering effects. This dual receptor engagement through the hypothalamus, hindbrain, and septum creates appetite suppression through pathways that GLP-1 agonists do not touch.
Clinical trial results worth knowing before you buy
The data supporting cagrilintide has progressed rapidly through clinical development, and researchers considering this compound should understand what the trials actually showed.
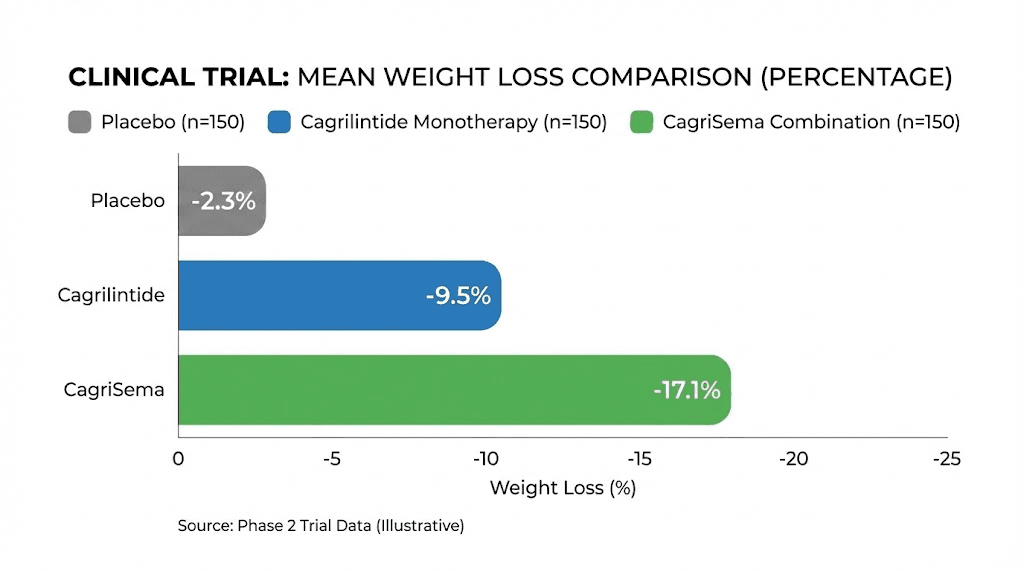
The Phase 2 dose-finding trial, published in The Lancet, tested weekly doses ranging from 0.3mg to 4.5mg in adults with overweight or obesity. At the 4.5mg dose, participants achieved approximately 10.8% body weight reduction over 26 weeks. Even at 2.4mg, the results showed dose-dependent weight loss that was statistically significant against placebo.
The REDEFINE 1 Phase 3 trial escalated things dramatically. Cagrilintide monotherapy at 2.4mg weekly produced 11.8% average weight loss over 68 weeks. That alone rivals semaglutide 2.4mg results. But the combination arm, CagriSema (cagrilintide 2.4mg plus semaglutide 2.4mg), achieved 22.7% mean body weight reduction. More than one in five participants lost 30% or more of their body weight.
The REDEFINE 2 trial focused on adults with type 2 diabetes and found CagriSema produced 15.7% weight loss versus 3.1% for placebo, with 88% of participants who had prediabetes returning to normoglycemia.
These are not marginal improvements. They represent a new ceiling for pharmaceutical weight management outcomes, and they explain why cagrilintide has become one of the most sought-after research peptides available.
Legal status and regulatory considerations
Understanding the legal framework around cagrilintide is essential before purchasing. The regulatory landscape differs significantly from approved compounds like semaglutide, and researchers need clarity on what they can and cannot do.
Current FDA status
Cagrilintide as a standalone compound is not FDA-approved for any indication. It has no approved medical use in the United States, Europe, or any other jurisdiction. The compound exists entirely within the clinical trial and research pipeline.
CagriSema, the fixed-dose combination of cagrilintide and semaglutide, had its New Drug Application submitted to the FDA by Novo Nordisk in December of the previous year. The FDA review is expected throughout this year, but approval is not guaranteed, and the timeline could extend.
This distinction matters for researchers. You are purchasing an investigational compound, not an approved therapeutic. The legal framework governing research peptides applies, not pharmaceutical distribution regulations.
Research use classification
In the United States, cagrilintide falls under the research chemical classification. It can be legally purchased, possessed, and used for legitimate laboratory research, in vitro studies, and educational purposes. Research peptide suppliers are required to label their products as "for research use only" and "not for human consumption."
This classification aligns with how other research peptides like retatrutide, mazdutide, and investigational compounds are distributed. The legal framework permits possession and research use while prohibiting marketing for human therapeutic application.
State laws can add complexity. Some states have additional regulations around peptide possession or distribution. Researchers should verify their specific state requirements before placing orders. Most established vendors will not ship to jurisdictions where local regulations create complications.
Import considerations
For researchers outside the United States, import regulations vary dramatically by country. The European Union, Australia, Canada, and the United Kingdom each have different frameworks for importing research chemicals. Customs declarations must accurately describe the product and its intended use. Misrepresenting research peptides on import documentation creates legal exposure that no researcher should accept.
Working with vendors who have experience shipping internationally reduces risk. Established companies understand customs requirements, proper documentation, and packaging that protects product integrity during transit. SeekPeptides provides guidance on navigating the regulatory landscape for various research compounds, helping researchers understand their obligations before purchasing.
How to evaluate cagrilintide vendors
The vendor you choose determines whether your research succeeds or fails. This is not an exaggeration. A peptide that tests at 95% purity when advertised at 99% can produce fundamentally different research outcomes. When cagrilintide costs significantly more than simpler peptides, wasting money on substandard product adds insult to scientific injury.
Third-party purity testing is non-negotiable
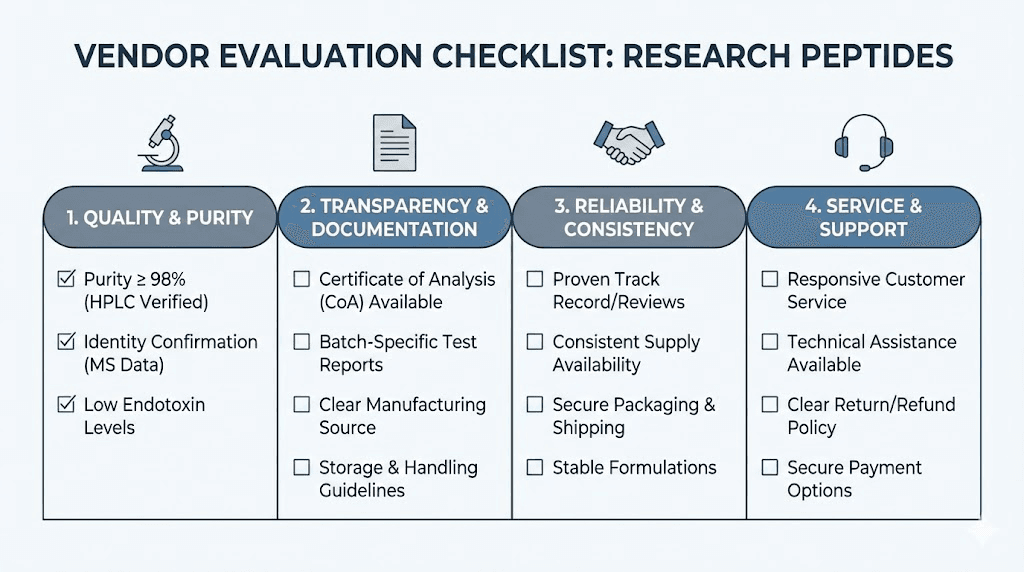
Every legitimate cagrilintide vendor provides certificates of analysis (CoAs) from independent, third-party laboratories. Not in-house testing. Third-party. The distinction matters because in-house testing creates obvious conflicts of interest.
What to look for on a CoA:
HPLC purity above 98%. High-performance liquid chromatography remains the gold standard for peptide purity assessment. Results below 98% suggest manufacturing issues, improper purification, or degradation. Premium vendors consistently deliver 99%+ purity.
Mass spectrometry confirmation. The molecular weight should match cagrilintide precisely. Mass spec confirms you are receiving the correct compound, not a cheaper substitute or partial sequence.
Endotoxin testing. Bacterial endotoxin contamination poses real risks in research settings. LAL testing should show endotoxin levels below acceptable thresholds.
Amino acid analysis. This confirms the peptide sequence matches what was ordered. For a 39-amino-acid compound with specific mutations, sequence verification prevents costly mistakes.
Ask for batch-specific CoAs, not generic documents. Each manufacturing batch can vary, and a certificate from six months ago tells you nothing about the vial arriving at your door today.
Vendor reputation markers
Beyond documentation, several factors separate reliable vendors from problematic ones.
Longevity in the market matters. Companies that have operated for three or more years with consistent product availability have survived the period where most unreliable vendors disappear. Look for established track records, not flashy new websites with suspiciously low prices.
Customer reviews from verified purchasers provide real-world data. Check peptide research forums, Reddit communities, and independent review aggregators. Patterns matter more than individual reviews. One negative review could be an anomaly. Twenty complaints about purity testing revealing contamination is a pattern.
Responsive customer service indicates operational maturity. Before placing a large order, contact support with technical questions about their cagrilintide. Ask about the peptide content per vial (actual peptide weight versus total weight including counter-ions and water content), their lyophilization process, and shipping conditions. Legitimate companies answer these questions confidently. Companies selling repackaged product from unknown sources often cannot.
Payment methods reveal something too. Vendors accepting only cryptocurrency with no other options may be operating on the margins. Established companies typically offer credit card processing, which requires passing merchant verification processes that filter out many questionable operations.
Price expectations and red flags
Cagrilintide is a complex peptide to synthesize. The acylation step alone adds manufacturing difficulty and cost beyond standard peptide synthesis. Expect to pay more than you would for simpler compounds like BPC-157 or TB-500.
Current market pricing for research-grade cagrilintide typically falls in the range of $30-50 per 5mg vial, with bulk orders reducing per-unit cost. Prices significantly below this range should raise questions about purity, actual peptide content, or sourcing.
Red flags that should stop you from ordering:
No CoA available or only generic certificates
Prices dramatically below market average
Claims of pharmaceutical-grade product at research peptide prices
No clear contact information or physical address
Website launched within the last few months with no history
Marketing claims about therapeutic benefits or weight loss guarantees
Remember that the cheapest option costs the most when it ruins your research.
Cagrilintide specifications and what you receive
Understanding exactly what arrives when you order cagrilintide prevents confusion and ensures proper handling from the moment the package arrives at your laboratory.
Physical form and appearance
Research-grade cagrilintide arrives as a white to off-white lyophilized (freeze-dried) powder in sealed glass vials. Lyophilization removes water from the compound after freezing, creating a stable solid that maintains integrity during shipping and storage. The powder should appear fluffy and uniform. Discoloration, clumping, or liquid in the vial suggests degradation or improper lyophilization.
Standard vial sizes include 5mg and 10mg, with some vendors offering larger quantities for high-volume research programs. The vial will be sealed with a rubber stopper and aluminum crimp cap, maintaining sterility until reconstitution.
Molecular specifications
For researchers who need precise specifications:
Molecular formula: C187H291N49O57S2
Molecular weight: Approximately 4,003 Da
Sequence length: 39 amino acids (modified human amylin)
Modifications: N-terminal C20 fatty acid acylation, 14E/17R/25P/28P/29P mutations, C-terminal proline
CAS number: 2375388-75-5
Solubility: Soluble in bacteriostatic water, sterile water, or DMSO
These specifications help verify that what you receive matches the actual compound. Cross-reference the molecular weight from your mass spectrometry results against these values.
Peptide content versus total vial weight
A common source of confusion, and one that can throw off research dosing if misunderstood. When a vial says "5mg," this typically refers to total peptide weight, which includes the peptide itself plus counter-ions (usually acetate or TFA salts) and residual water content.
Actual peptide content, meaning pure cagrilintide only, is usually 70-85% of the labeled weight. A 5mg vial might contain 3.5-4.25mg of active cagrilintide. Premium vendors state the actual peptide content on their CoA. If this information is missing, ask for it before calculating your research dosing protocols.
This distinction becomes critical when trying to replicate published study protocols that specify exact microgram or milligram doses. Using total vial weight instead of actual peptide content introduces a systematic error that compounds across every administration.
Reconstitution and preparation protocols
Proper reconstitution determines whether your cagrilintide maintains potency or degrades before you can use it. The acylated fatty acid chain that gives cagrilintide its long half-life also makes it somewhat more sensitive to improper handling than simpler peptides.
What you need before starting
Gather all materials before beginning reconstitution. You will need:
Cagrilintide lyophilized vial
Bacteriostatic water (preferred) or sterile water
Alcohol swabs (70% isopropyl)
Insulin syringes (1mL with 29-31 gauge needles)
Clean workspace with minimal airflow
Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, extending the usable life of reconstituted peptide to approximately 30 days when refrigerated. Sterile water without preservative should be used within 48-72 hours of reconstitution. For most research protocols spanning weeks, bacteriostatic water is the practical choice.
Step-by-step reconstitution
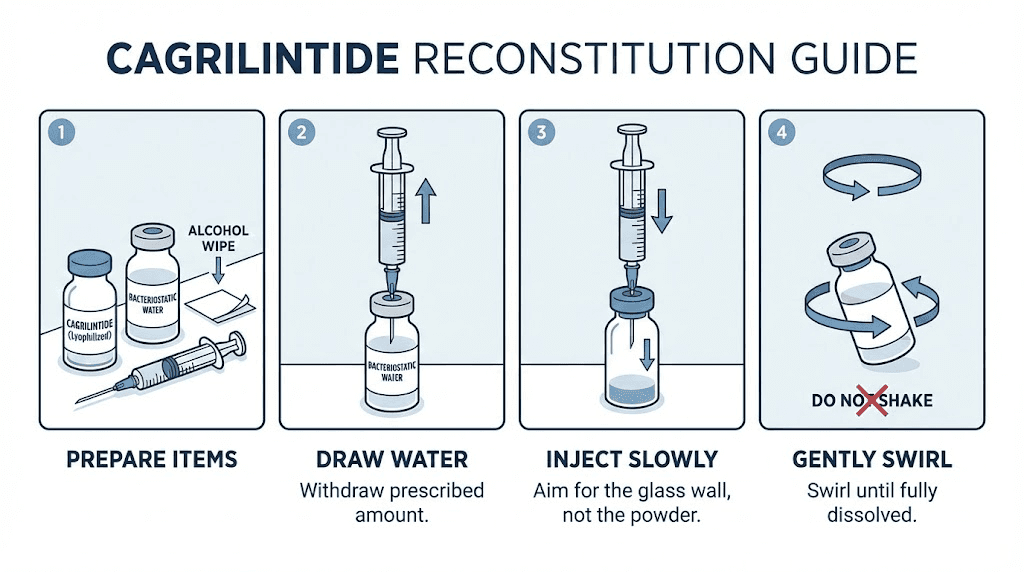
Clean your workspace thoroughly. Wash hands and consider wearing nitrile gloves to prevent contamination.
Swab the rubber stopper on the cagrilintide vial with an alcohol pad. Do the same for the bacteriostatic water vial. Allow both to dry for 30 seconds. Alcohol kills surface bacteria but needs to evaporate completely before you draw or inject any liquid.
Draw your chosen volume of bacteriostatic water into the syringe. For a 5mg vial, adding 2.0mL creates a concentration of 2.5mg/mL. For a 10mg vial, adding 2.0mL creates 5.0mg/mL. Your specific concentration choice depends on the dose volumes your protocol requires. Smaller volumes mean more concentrated solution but require more precise measurement. Larger volumes provide easier measurement but may exceed practical injection volumes for some protocols.
Insert the needle through the rubber stopper at an angle, with the bevel facing the glass wall of the vial. Inject the water slowly, letting it run down the inside wall of the glass. Never spray water directly onto the lyophilized powder. Direct impact can damage peptide structure through mechanical force.
Once all water is in the vial, remove the syringe. Gently swirl the vial between your fingers. Do not shake it. Do not vortex it. Aggressive agitation creates foam and can cause peptide denaturation at air-liquid interfaces, particularly for acylated peptides like cagrilintide where the fatty acid chain makes the molecule somewhat amphiphilic.
The powder should dissolve within 2-5 minutes of gentle swirling. If particulates remain after 10 minutes of gentle agitation, place the vial in the refrigerator for 30 minutes and try again. Persistent cloudiness or visible particles after this process may indicate degraded product.
Concentration calculations
Researchers using the peptide reconstitution calculator can simplify these calculations, but understanding the math ensures accuracy.
For a 5mg vial reconstituted with 2.0mL of bacteriostatic water:
Concentration: 5mg / 2.0mL = 2.5mg/mL = 2,500mcg/mL
Each 0.1mL (10 units on an insulin syringe) = 250mcg
Each 0.01mL (1 unit on an insulin syringe) = 25mcg
For a 10mg vial reconstituted with 2.0mL:
Concentration: 10mg / 2.0mL = 5.0mg/mL = 5,000mcg/mL
Each 0.1mL = 500mcg
Each 0.01mL = 50mcg
Choose your reconstitution volume based on the doses your protocol requires. If you need 250mcg doses and want to measure 10 units on the syringe each time, 2.0mL in a 5mg vial gives you exactly that. Mathematical precision at this stage prevents cumulative errors throughout the protocol.
Storage and stability guidelines
Cagrilintide stability depends on storage conditions at every stage, from the moment the package arrives through the final administered dose. Mishandling at any point degrades the compound and compromises research integrity.
Unreconstituted (lyophilized) storage
Lyophilized cagrilintide maintains stability for two or more years when stored at -20 degrees Celsius in a freezer, protected from light and moisture. This is your long-term storage solution for vials you do not plan to use immediately.
For shorter-term storage of up to six months, refrigeration at 2-8 degrees Celsius is acceptable. This temperature range applies to vials awaiting reconstitution within a reasonable timeframe.
Room temperature storage should be limited to the briefest possible duration, primarily during shipping and initial receipt. Extended room temperature exposure degrades peptide integrity, particularly for acylated compounds where the fatty acid chain can undergo oxidation.
Keep vials in their original packaging or wrapped in foil to prevent light exposure. UV and visible light both catalyze degradation reactions. A freezer-safe container with desiccant packets provides optimal conditions.
Reconstituted storage
Once reconstituted with bacteriostatic water, cagrilintide should be stored at 2-8 degrees Celsius and used within 30 days. This timeline assumes the vial remains properly sealed and refrigerated between uses.
Never freeze reconstituted peptide solution. The freeze-thaw cycle damages protein structure through ice crystal formation and concentration effects at the liquid-solid interface. This applies universally to reconstituted peptides, not just cagrilintide, and the same principle governs tirzepatide storage and semaglutide refrigeration protocols.
If reconstituted with sterile water (no preservative), use within 48-72 hours. Bacteriostatic water is strongly preferred for any protocol lasting longer than a few days.
Shipping and transit considerations
Quality vendors ship lyophilized cagrilintide with cold packs during warm months and insulated packaging year-round. Transit time matters because extended exposure to temperature extremes during shipping degrades product before it ever reaches your freezer.
When your order arrives, inspect the packaging immediately. The cold pack should still be cool (though not necessarily frozen). The vial should contain intact lyophilized powder. If the powder appears wet, dissolved, or discolored, contact the vendor before using. Reputable companies replace compromised shipments.
For researchers ordering multiple vials, consider having them shipped in separate batches if transit times are long. Losing one vial to a shipping delay is preferable to losing your entire supply.
Dosing protocols from published research
Understanding the dosing frameworks that produced results in clinical trials gives researchers the foundation for designing their own protocols. These doses were established through rigorous Phase 1, 2, and 3 clinical trials with safety monitoring.
Dose escalation approach
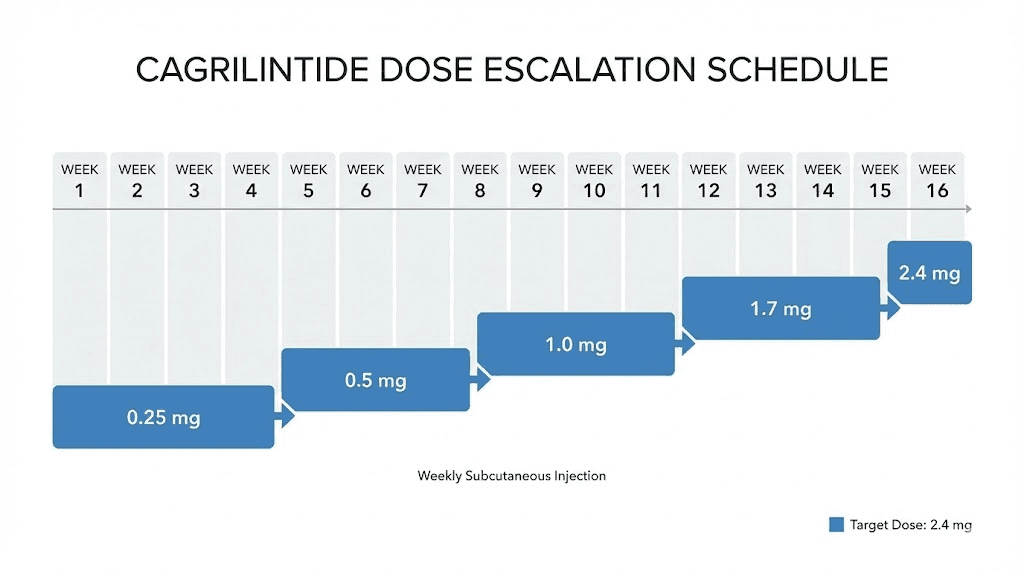
Every published cagrilintide protocol uses gradual dose escalation. Starting at the full target dose produces unnecessary gastrointestinal side effects that slow research and reduce protocol adherence. The escalation schedule from clinical trials follows a clear pattern:
Weeks 1-4: 0.25mg once weekly. This initial phase establishes baseline tolerance. Gastrointestinal effects at this dose are minimal in most subjects.
Weeks 5-8: 0.5mg once weekly. A modest increase that allows adaptation while beginning to produce measurable pharmacological effects.
Weeks 9-12: 1.0mg once weekly. At this dose, appetite suppression becomes consistently measurable in research settings.
Weeks 13-16: 1.7mg once weekly. Approaching the therapeutic range used in major clinical trials.
Week 17 onward: 2.4mg once weekly (maintenance dose for most protocols). This is the dose used in both the REDEFINE 1 and REDEFINE 2 trials.
Some Phase 2 protocols explored doses up to 4.5mg weekly for monotherapy research. The higher doses produced greater weight reduction but also increased gastrointestinal adverse events proportionally.
Monotherapy versus combination protocols
Cagrilintide produces meaningful results as a monotherapy, but the data overwhelmingly shows that combining it with a GLP-1 receptor agonist amplifies outcomes dramatically.
The rationale is mechanistic. Cagrilintide activates amylin receptors in brain regions that regulate appetite through pathways distinct from GLP-1 signaling. Semaglutide activates GLP-1 receptors in overlapping but different neural circuits. Together, they create complementary appetite suppression that neither achieves alone.
Monotherapy results at 2.4mg weekly: 11.8% mean body weight reduction over 68 weeks.
CagriSema combination at 2.4mg each: 22.7% mean body weight reduction over the same period.
The combination nearly doubles the weight loss outcome. This is not additive, it is synergistic, with the combination producing more than the sum of individual effects.
For researchers considering combination protocols, the published literature provides the framework. Semaglutide follows its own dose escalation schedule simultaneously with cagrilintide escalation. Both compounds reach their target doses by approximately week 16-20 of the protocol.
Administration details
Clinical trials administered cagrilintide via subcutaneous injection once weekly. The preferred injection sites include the abdomen (at least 2 inches from the navel), the front of the thigh, and the upper arm. Rotating injection sites weekly reduces the injection site reactions that approximately 17% of trial participants reported.
The once-weekly schedule reflects the compound pharmacokinetics. With a half-life of 159-195 hours, steady-state concentrations are achieved after approximately 4-5 weekly doses. Peak plasma concentration occurs 24-72 hours after injection, with dose-proportional exposure across the studied range.
Timing of the weekly dose does not appear to significantly impact outcomes based on available data. Consistency matters more than the specific day or time chosen. Pick a day and stick with it throughout the protocol.
How cagrilintide compares to other weight management peptides
Researchers rarely evaluate a compound in isolation. Understanding where cagrilintide fits relative to established and emerging alternatives helps inform purchasing decisions, protocol design, and research priorities.
Cagrilintide versus semaglutide
Semaglutide is the current benchmark. As a GLP-1 receptor agonist, it targets a completely different receptor system than cagrilintide. The STEP trials established semaglutide 2.4mg as capable of producing approximately 15-17% body weight reduction in adults with obesity.
Cagrilintide monotherapy at the same 2.4mg dose produced 11.8% weight loss in the REDEFINE trials. On a head-to-head basis, semaglutide monotherapy appears to produce somewhat greater weight loss than cagrilintide monotherapy.
But the comparison misses the point. Cagrilintide was not designed to replace semaglutide. It was designed to complement it. The combination outperforms either compound alone by a significant margin. Researchers interested in either compound should understand both, since the most promising research direction involves using them together.
From a side effect perspective, cagrilintide monotherapy shows lower rates of nausea, vomiting, and diarrhea compared to semaglutide. For researchers whose protocols are complicated by GI side effects affecting data quality, cagrilintide monotherapy may offer a cleaner signal.
Semaglutide is also far more established in terms of sourcing. Compounded semaglutide is available through numerous channels, and researchers can reference extensive literature on its pharmacology. Cagrilintide sourcing requires more due diligence given fewer established suppliers and a shorter market history for the research compound.
Cagrilintide versus tirzepatide
Tirzepatide is a dual GIP/GLP-1 receptor agonist that produced approximately 20-22% weight loss in the SURMOUNT trials at its highest dose. This puts tirzepatide results in the same ballpark as CagriSema, though the mechanisms differ fundamentally.
Tirzepatide hits two incretin pathways. CagriSema hits an incretin pathway (GLP-1 via semaglutide) plus the amylin pathway (via cagrilintide). The receptor targets are different, the downstream signaling differs, and the potential for combination with other compounds differs.
An interesting research question, one that published data has not yet fully addressed, is whether cagrilintide could be combined with tirzepatide. Since tirzepatide does not activate amylin receptors and cagrilintide does not activate GIP receptors, the mechanistic rationale for combination exists. No clinical trial has tested this combination, making it a theoretical consideration rather than an evidence-based protocol.
Practically, tirzepatide is more widely available through compounding pharmacies and research suppliers. Pricing for tirzepatide research peptide has also decreased as competition among suppliers has increased. Cagrilintide remains somewhat more expensive per milligram due to synthesis complexity and lower production volumes.
Cagrilintide versus retatrutide
Retatrutide targets three receptors simultaneously: GLP-1, GIP, and glucagon. Phase 2 data showed approximately 24% weight loss at the highest dose, making it one of the most potent single agents in development. The retatrutide dosing protocols share similar escalation approaches with cagrilintide.
Cagrilintide and retatrutide occupy different mechanistic spaces. Retatrutide covers the incretin pathways more broadly than any single agent. Cagrilintide covers the amylin pathway that retatrutide does not touch. From a research perspective, both compounds offer unique pharmacological profiles worth investigating.
Availability of retatrutide for research purchase has expanded, though both compounds remain less accessible than semaglutide or tirzepatide from a vendor standpoint.
Comparison summary
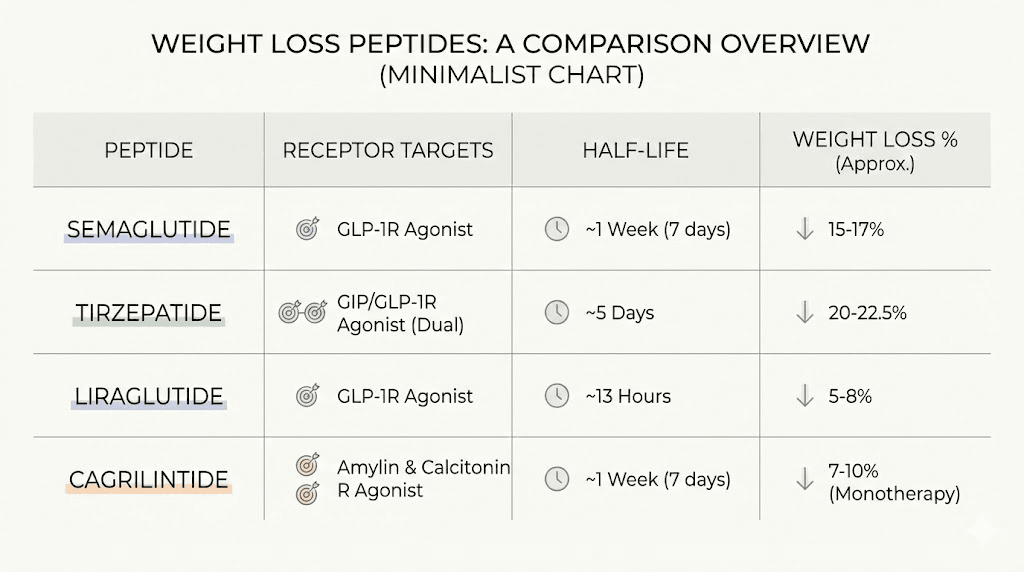
Feature | Cagrilintide | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|---|
Primary targets | AMY1R, AMY3R, CTR | GLP-1R | GLP-1R, GIPR | GLP-1R, GIPR, GCGR |
Dosing frequency | Once weekly | Once weekly | Once weekly | Once weekly |
Half-life | ~8 days | ~7 days | ~5 days | ~6 days |
Monotherapy weight loss | 11.8% | 15-17% | 20-22% | ~24% |
Combination potential | CagriSema: 22.7% | Part of CagriSema | Not yet studied | Not yet studied |
GI side effect profile | Lowest among class | Moderate | Moderate-high | Moderate-high |
Research availability | Moderate | Widely available | Widely available | Moderate |
Relative cost | Higher | Lower | Moderate | Higher |
Side effects and safety considerations
No compound worth researching comes without risk considerations. Cagrilintide has an established safety profile from multiple clinical trials, and researchers should understand both common and less common adverse events before incorporating it into protocols.
Gastrointestinal effects
Nausea is the most frequently reported side effect. It tends to be mild to moderate, dose-dependent, and transient, typically resolving within the first few weeks at each dose level. The dose escalation protocol exists specifically to minimize this effect, and researchers who skip the escalation phase to reach target doses faster consistently report higher rates of GI complaints.
Other GI effects include constipation, diarrhea, and occasional abdominal discomfort. Meta-analysis data shows cagrilintide produces lower rates of nausea, vomiting, and diarrhea compared to semaglutide and tirzepatide. This favorable GI profile is one reason researchers exploring the amylin pathway find cagrilintide attractive.
When combined with semaglutide in the CagriSema formulation, GI adverse events increase, with approximately 79.6% of participants reporting at least one GI event versus 39.9% in the placebo group. Most were mild to moderate and transient, but the combination clearly produces more GI effects than either compound alone.
Injection site reactions
Approximately 17% of participants receiving cagrilintide in clinical trials reported injection site reactions, primarily mild redness, itching, or irritation. These reactions are more common with cagrilintide than with semaglutide, likely due to the compound structural properties.
Rotating injection sites between the abdomen, thigh, and upper arm reduces the frequency and severity of local reactions. Allowing the reconstituted solution to reach room temperature before injection (10-15 minutes out of the refrigerator) also helps reduce local irritation.
Metabolic considerations
Cagrilintide does not appear to increase hypoglycemia risk in individuals without diabetes, which aligns with its mechanism as an amylin analogue rather than an insulin secretagogue. In clinical trials involving participants with type 2 diabetes and concurrent sulfonylurea use, hypoglycemia rates were higher but remained within ranges seen with other anti-obesity agents.
Researchers should monitor for changes in appetite that could affect nutritional status in extended protocols. The appetite-suppressing effects of cagrilintide are its primary mechanism for producing weight loss, and in some subjects, this suppression can be more pronounced than expected, particularly during the first weeks at a new dose level.
Discontinuation rates
The overall discontinuation rate due to adverse events was approximately 10% in the REDEFINE trials. This is comparable to rates seen with other anti-obesity medications in their clinical programs. Serious adverse events were rare and occurred at similar rates in treatment and placebo groups.
For perspective, the cagrilintide side effects profile is generally considered favorable compared to most compounds in the metabolic research space. Researchers who have worked with tirzepatide or high-dose semaglutide often note that cagrilintide produces less GI disruption at equivalent efficacy levels.
Ordering process and what to expect
For researchers ready to purchase, understanding the typical ordering process reduces friction and prevents common mistakes that delay research timelines.
Standard ordering workflow
Most research peptide vendors follow a similar process. You create an account, browse their catalog for cagrilintide, select your desired quantity and vial size, and proceed to checkout. Some vendors require a brief verification step to confirm research purposes, which may involve providing institutional affiliation or a statement of intended use.
Payment processing typically takes 1-2 business days. Shipping times vary by vendor location and shipping method selected. Domestic U.S. orders usually arrive within 3-7 business days. International shipping adds time and may require customs processing.
Upon placing your order, request the batch-specific certificate of analysis if it is not automatically included. Some vendors email CoAs separately from shipping confirmations. Do not begin using any peptide without reviewing its documentation first.
Quantity planning
Calculate your total protocol needs before ordering. A standard 16-week escalation protocol to 2.4mg weekly requires approximately:
Weeks 1-4: 0.25mg x 4 = 1.0mg
Weeks 5-8: 0.5mg x 4 = 2.0mg
Weeks 9-12: 1.0mg x 4 = 4.0mg
Weeks 13-16: 1.7mg x 4 = 6.8mg
Maintenance at 2.4mg x number of additional weeks
Total for 16-week escalation alone: approximately 13.8mg. Add an additional 2.4mg per week for each maintenance week. A 24-week protocol (16 escalation + 8 maintenance) requires roughly 33mg total. Factor in 10-15% overage for syringe dead space and measurement precision, bringing the practical need to approximately 36-38mg.
Ordering sufficient quantity upfront, from the same manufacturing batch when possible, ensures consistency across your entire protocol. Mid-protocol vendor changes or batch variations introduce variables that complicate data interpretation.
Handling upon arrival
When your shipment arrives, inspect immediately. Verify that the correct number of vials are present and that the lyophilized powder appears intact. Store unreconstituted vials according to the guidelines above. Only reconstitute vials as needed for immediate use, keeping the remainder in long-term storage to maximize shelf life.
Document the batch number, receipt date, and storage conditions as part of your research records. Good laboratory practice requires traceability from product receipt through final use.
Using cagrilintide with other research compounds
The combination potential of cagrilintide extends beyond the CagriSema paradigm. Researchers exploring metabolic pathways often want to understand how cagrilintide interacts with other compounds in their research toolkit.
Cagrilintide plus semaglutide (the CagriSema protocol)
This is the most extensively studied combination and the one with the strongest clinical evidence. The rationale is straightforward: amylin receptor activation (cagrilintide) plus GLP-1 receptor activation (semaglutide) produces complementary appetite suppression through distinct neural pathways.
Both compounds follow their own dose escalation schedules simultaneously. The clinical protocol reaches target doses of 2.4mg each by approximately week 16-20. Since both are administered once weekly via subcutaneous injection, they can be given on the same day at different injection sites.
Researchers interested in this combination should review the REDEFINE trial protocols for detailed methodology. The results, 22.7% mean weight loss over 68 weeks, represent the current high-water mark for pharmaceutical weight management interventions.
Cagrilintide with B12 or glycine
Some research protocols incorporate vitamin B12 or glycine alongside weight management peptides. Semaglutide with B12 combinations have become common in compounded formulations, and the rationale may extend to cagrilintide protocols.
B12 supplementation addresses the potential for nutrient absorption changes that caloric restriction and gastric emptying modulation can produce. Glycine serves as an amino acid with roles in collagen synthesis, neurotransmitter function, and metabolic regulation. Neither compound is expected to interfere with cagrilintide receptor activity, but formal interaction studies are limited.
Compounds to approach with caution
Combining cagrilintide with other agents that affect gastric emptying warrants careful consideration. The additive effect on gastric motility could amplify GI side effects beyond what either compound produces alone. This consideration applies to combinations with pramlintide, high-dose GLP-1 agonists beyond those studied in the CagriSema trials, or any compound with significant effects on gut motility.
Insulin-sensitizing compounds should be monitored carefully in combination with cagrilintide, particularly in protocols involving subjects with type 2 diabetes. While cagrilintide does not directly cause hypoglycemia, its effects on appetite and food intake can change glucose dynamics in ways that interact with insulin or sulfonylurea protocols.
The CagriSema FDA timeline and what it means for researchers
Novo Nordisk submitted the CagriSema New Drug Application to the FDA in December of last year, setting up a review process that will likely conclude later this year. Understanding this timeline helps researchers plan their work and anticipate market changes.
What approval would change
If approved, CagriSema would become the first combination amylin-GLP-1 therapeutic, creating a new drug category. Pharmaceutical-grade CagriSema would become available through prescription channels, potentially changing the research landscape in several ways.
First, increased public awareness of cagrilintide would drive more research interest, potentially expanding the supplier base and improving quality competition. Second, pharmaceutical-grade reference standards would become more accessible for quality comparison. Third, the published FDA review documents would provide additional safety and efficacy data that enriches the research literature.
For researchers currently using research-grade cagrilintide, approval would not change the legal framework around research peptide use. Research-grade compounds would continue to be available through existing channels. The two markets, pharmaceutical and research, operate in parallel.
What researchers should do now
The period before potential approval is actually an optimal time for researchers to establish protocols and generate preliminary data. Once CagriSema potentially receives approval, the research community interest will spike, likely affecting both pricing and availability of research-grade cagrilintide.
Building familiarity with cagrilintide handling, reconstitution, and administration now positions researchers to work more efficiently when broader scientific attention arrives. Having established vendor relationships and validated quality protocols gives you a head start over researchers entering the field after approval.
Common mistakes when buying cagrilintide
Learning from other researchers mistakes saves time, money, and frustration. These are the errors that come up most frequently in research communities.
Mistake 1: Buying based on price alone
The cheapest cagrilintide is rarely the best value. A $20 vial that tests at 93% purity contains less active compound than a $40 vial at 99% purity, and the impurities in that 7% gap could include synthesis byproducts that confound your research. Factor in the cost of wasted protocol time and repeat experiments, and the cheap vial becomes the expensive choice.
Mistake 2: Not verifying the CoA
Accepting a vendor certificate of analysis at face value without scrutiny is common but risky. Check that the CoA is batch-specific, from a third-party lab, recent (within 6 months of your purchase), and includes HPLC purity and mass spec data at minimum. Some researchers go a step further and send a sample to an independent lab for verification, particularly on their first order from a new vendor.
Mistake 3: Improper storage on arrival
Leaving a delivery box on the porch for hours during summer, or storing vials at room temperature because the freezer is full, degrades product before research even begins. Have your storage solution ready before ordering. Make sure someone is available to receive and properly store the shipment on arrival day.
Mistake 4: Skipping dose escalation
Researchers in a hurry sometimes jump directly to maintenance doses. The clinical trial dose escalation protocol exists for good reason. Skipping it increases adverse events, particularly nausea, that can disrupt data collection and reduce protocol adherence. The four extra weeks of escalation time saves weeks of dealing with unnecessary side effects.
Mistake 5: Mixing up peptide content and total vial weight
As discussed earlier, the difference between total vial weight and actual peptide content introduces systematic dosing errors. A researcher who assumes all 5mg in a vial is active cagrilintide might be administering 15-30% less than intended. Over a multi-week protocol, this underdosing accumulates and can mean the difference between meaningful results and inconclusive data.
Mistake 6: Not planning total quantity needed
Ordering one or two vials to "try it out" then needing more mid-protocol creates problems. Different batches may have slightly different purities or peptide content. Shipping delays can create gaps in weekly dosing. Plan the full protocol supply and order it together when feasible.
Frequently asked questions
Is cagrilintide the same as semaglutide?
No. Cagrilintide is a long-acting amylin analogue that activates amylin and calcitonin receptors. Semaglutide is a GLP-1 receptor agonist that targets a completely different receptor system. They affect appetite through distinct neural pathways, which is why combining them (as in CagriSema) produces significantly greater weight loss than either alone.
How much does cagrilintide cost for research purposes?
Research-grade cagrilintide typically costs between $30-50 per 5mg vial from reputable vendors, with 10mg vials proportionally priced. Bulk purchases reduce per-unit cost. Prices vary by vendor, purity level, and market conditions. Always prioritize quality and verified purity over the lowest price available.
Can I take cagrilintide orally?
Cagrilintide is a peptide that would be degraded by digestive enzymes if taken orally. All published clinical research administers cagrilintide via subcutaneous injection. Unlike some compounds that have oral formulations, no oral cagrilintide delivery system has been developed or studied.
How long does cagrilintide last once reconstituted?
When reconstituted with bacteriostatic water and stored at 2-8 degrees Celsius, cagrilintide maintains stability for approximately 30 days. Using sterile water without preservative reduces this to 48-72 hours. Proper refrigeration is essential for maintaining potency throughout the usable period.
What is the difference between cagrilintide and CagriSema?
Cagrilintide is the standalone amylin analogue compound. CagriSema is Novo Nordisk branded combination product containing both cagrilintide 2.4mg and semaglutide 2.4mg in a single injection. CagriSema is currently under FDA review. Research-grade cagrilintide can be used alone or combined with separately sourced semaglutide to create a similar combination protocol.
Does cagrilintide need to be refrigerated?
Lyophilized (unreconstituted) cagrilintide should be stored frozen at -20 degrees Celsius for long-term storage or refrigerated at 2-8 degrees for shorter periods up to 6 months. Once reconstituted, it must be refrigerated and used within 30 days. Never freeze reconstituted solution, as the freeze-thaw cycle destroys peptide structure.
What are the most common side effects of cagrilintide?
Mild to moderate nausea (transient, dose-dependent), injection site reactions (reported by approximately 17% of subjects), occasional constipation or diarrhea, and reduced appetite (which is the intended mechanism of action). Cagrilintide side effects are generally milder than those reported with GLP-1 receptor agonists like semaglutide.
Can cagrilintide be combined with tirzepatide?
No published clinical trial has tested cagrilintide with tirzepatide. The mechanistic rationale exists since they target different receptor systems, but safety and efficacy data for this specific combination is not available. Researchers considering novel combinations should exercise appropriate caution and consult relevant literature on each compound individual safety profile.
External resources
Cagrilintide: A Long-Acting Amylin Analog for Treatment of Obesity - PubMed
Development of Cagrilintide, a Long-Acting Amylin Analogue - Journal of Medicinal Chemistry
For researchers serious about optimizing their metabolic research protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Whether you are evaluating cagrilintide dosing for the first time or building complex multi-compound protocols, the depth of resources and community expertise makes the difference between guessing and knowing.
In case I do not see you, good afternoon, good evening, and good night. May your peptides stay potent, your vendors stay honest, and your research stay rigorous.