Apr 4, 2026

At the cellular level, something remarkable happens when semaglutide reaches the brain. Receptors activate. Inflammatory cascades slow. Immune cells that were attacking myelin sheaths begin to quiet down. And for the millions of people living with multiple sclerosis, this cascade of events has sparked one of the most promising research avenues in years.
Multiple sclerosis destroys the protective coating around nerve fibers. Slowly. Relentlessly. The immune system turns on itself, stripping away myelin and leaving behind scars that disrupt every signal the brain tries to send. Walking becomes harder. Vision blurs. Fatigue sets in like a weight that never lifts. And for decades, the treatment options have focused almost exclusively on suppressing the immune system, not on protecting or repairing the nervous system itself.
Then came the GLP-1 receptor agonists. Drugs like semaglutide were developed for type 2 diabetes and weight management. But researchers began noticing something unexpected. Patients on these medications showed neuroprotective effects that extended far beyond blood sugar control. Animal studies revealed reduced neuroinflammation, preserved myelin, and even signs of remyelination, the holy grail of MS research. A massive cohort study of over 14,000 MS patients found that those taking GLP-1 agonists experienced significantly less disease progression across every functional category measured. This guide explores everything the current research tells us about semaglutide and multiple sclerosis, from the molecular mechanisms driving these effects to the practical considerations for people with MS who are already taking or considering semaglutide.
Understanding the connection between GLP-1 receptors and the nervous system
Before diving into the MS-specific research, it helps to understand why a diabetes drug would have any effect on the brain at all. The answer lies in where GLP-1 receptors actually exist in the body. Most people think of them as gut and pancreas receptors. They are. But they also populate the brain in significant numbers.
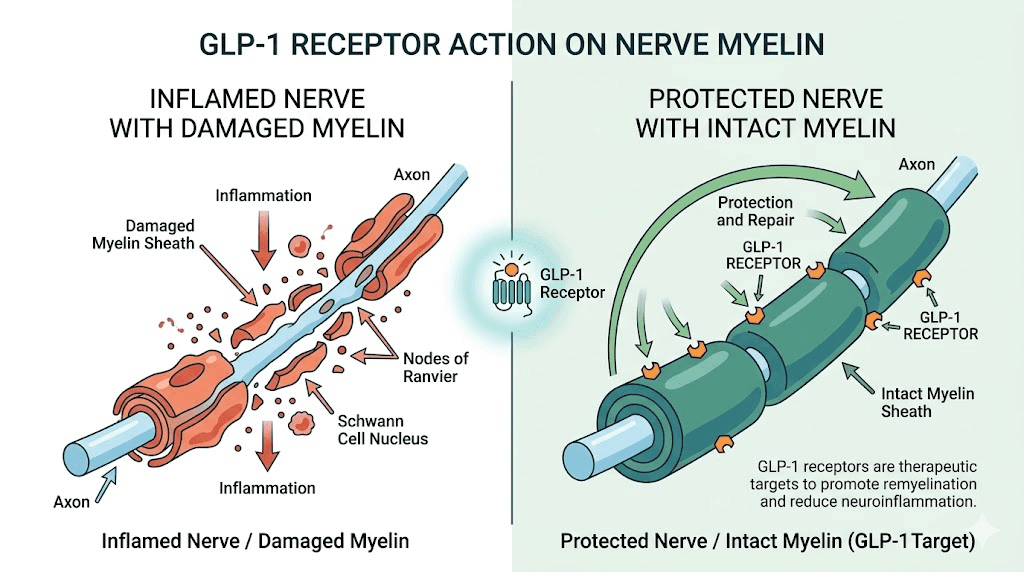
GLP-1 receptors appear throughout the central nervous system. They sit on neurons, on microglia (the immune cells of the brain), on astrocytes, and critically, on mature oligodendrocytes, the cells responsible for producing and maintaining myelin. When a GLP-1 receptor agonist binds to these receptors, it triggers a cascade of anti-inflammatory and protective signals that researchers are only beginning to fully map.
Here is what makes this especially relevant for multiple sclerosis. The disease attacks myelin. GLP-1 receptors exist on the very cells that make myelin. And activating those receptors appears to protect those cells, reduce the inflammation attacking them, and potentially even stimulate repair. That is not speculation. That is what the preclinical research consistently demonstrates. The timing and delivery of GLP-1 receptor activation matters, and understanding how these injection protocols work provides helpful context for the mechanism.
How semaglutide crosses the blood-brain barrier
One of the most important questions researchers had to answer was whether semaglutide could even reach the brain. The blood-brain barrier exists specifically to keep most substances out. Many drugs that show promise in lab dishes fail entirely because they cannot cross this barrier in a living organism.
GLP-1 receptor agonists like semaglutide can cross the blood-brain barrier. Research confirms this. The mechanism involves both passive diffusion and active transport, though the exact pathways are still being studied. What matters for MS patients is that semaglutide does reach the central nervous system in sufficient concentrations to activate GLP-1 receptors on brain cells, including oligodendrocytes and microglia. This is not a theoretical benefit. Neuroimaging and cerebrospinal fluid studies in humans confirm central nervous system activity of semaglutide.
The EVOKE and EVOKE+ trials for Alzheimer disease provide additional evidence. These large-scale phase 3 studies demonstrated that semaglutide significantly reduced cerebrospinal fluid levels of phosphorylated tau, total tau, and neurogranin, all biomarkers of neurodegeneration. If semaglutide can modify these markers, it is clearly reaching and affecting the brain.
GLP-1 receptors on immune cells
Beyond the brain itself, GLP-1 receptors also exist on peripheral immune cells. Macrophages, monocytes, and lymphocytes all express these receptors. When activated by semaglutide, these immune cells shift toward anti-inflammatory phenotypes. For a disease like MS, where peripheral immune cells cross into the brain and attack myelin, calming these cells before they even reach the central nervous system could be profoundly beneficial.
Research shows that GLP-1 receptor activation promotes regulatory T cell (Treg) expansion. Tregs are the immune system peacekeepers. They suppress autoimmune responses. In MS, Treg function is often impaired. The fact that semaglutide may enhance Treg activity adds another layer to its potential therapeutic value in autoimmune neurological conditions.
The landmark animal study: semaglutide in EAE mice
The most direct evidence for semaglutide in MS comes from a study published in International Immunopharmacology that tested semaglutide in the experimental autoimmune encephalomyelitis (EAE) mouse model. EAE is the gold standard animal model for multiple sclerosis. It mimics the autoimmune attack on myelin that defines the human disease.
The results were striking.
Mice received semaglutide at 25 nmol/kg/day via intraperitoneal injection. The researchers assessed cognitive function, motor ability, brain tissue integrity, and the molecular pathways involved. Every major outcome improved with semaglutide treatment.
Cognitive and motor improvements
EAE causes measurable cognitive and motor deficits in mice, just as MS does in humans. The semaglutide-treated mice showed significant improvements across multiple behavioral tests. Novel object recognition improved, indicating better memory and cognitive function. Open field testing showed improved exploratory behavior. Rotarod performance, which measures balance and coordination, improved substantially. And grip strength testing confirmed preserved motor neuron function.
These are not subtle improvements. The semaglutide-treated mice performed dramatically better than untreated EAE mice across every assessment. For people with MS who experience fatigue, cognitive fog, and progressive motor decline, these findings carry real significance. Understanding how semaglutide affects appetite and neurological side effects like dizziness helps contextualize the broader neurological activity of the drug.
Reduced demyelination and hippocampal damage
Histological examination of brain tissue revealed that semaglutide treatment attenuated hippocampal damage and corpus callosum demyelination. The corpus callosum is the thick band of nerve fibers connecting the two hemispheres of the brain, and it is one of the most commonly affected structures in MS. Preserving its myelin integrity would protect the communication between brain hemispheres that MS progressively disrupts.
The hippocampus, essential for memory and learning, also showed significantly less damage in semaglutide-treated animals. Hippocampal atrophy is a recognized feature of MS that correlates with cognitive decline. Protecting this structure could help preserve the cognitive abilities that many MS patients fear losing.
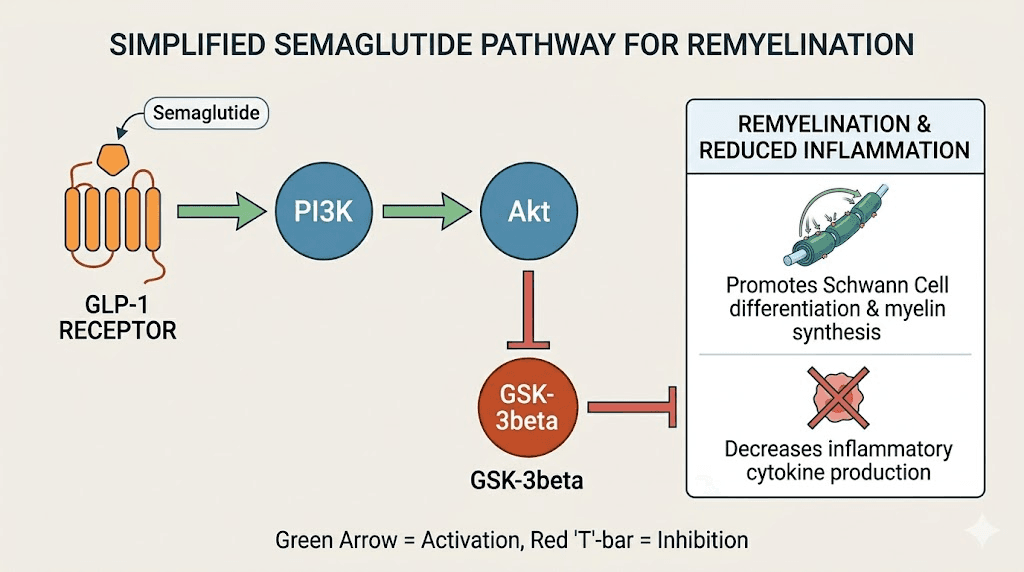
The PI3K/Akt/GSK-3beta pathway
The study identified the specific molecular pathway through which semaglutide exerts its protective effects. Semaglutide activates the PI3K/Akt signaling axis, which then inhibits GSK-3beta activity. This matters enormously because GSK-3beta inhibition triggers a cascade of protective effects.
First, it attenuates demyelination and triggers remyelination through CREB/BDNF signaling. BDNF (brain-derived neurotrophic factor) is a protein that supports neuronal survival and growth. Second, GSK-3beta inhibition boosts Nrf2 and SOD levels, protecting against oxidative stress, a major contributor to MS nerve damage. Third, it minimizes neuroinflammation by decreasing NF-kB and TNF-alpha levels, two of the primary inflammatory drivers in MS.
In simpler terms, semaglutide does not just reduce inflammation. It simultaneously promotes repair, protects against oxidative damage, and supports neuronal survival through a single integrated pathway. That kind of multi-target effect is exactly what MS researchers have been looking for. The glycine compound formulations and B12 glycine blends that are popular in compounded semaglutide may offer additional neuroprotective support through complementary pathways.
The TriNetX cohort study: 14,000 MS patients
Animal studies generate hypotheses. Human data tests them. And the largest human study to date on GLP-1 agonists and MS produced results that got the entire neurology community talking.
Presented at the American Academy of Neurology meeting, this retrospective cohort study used the TriNetX electronic health registry to compare outcomes in MS patients taking GLP-1 agonists versus those who were not. After propensity score matching to account for confounding variables, the researchers analyzed 7,046 MS patients receiving GLP-1 agonists and 7,046 matched controls over a five-year follow-up period.
Results across every functional category
The findings were consistent and statistically significant across the board. Patients not taking GLP-1 agonists demonstrated more MS disease progression in every functional category assessed using the Kurtzke Functional Systems Scores. These scores measure disability across pyramidal function (strength), cerebellar function (coordination), brainstem function, sensory function, bowel and bladder function, visual function, cerebral function, and ambulation.
The risk differences were clinically meaningful. Brainstem dysfunction showed a 4.36% risk difference (p < 0.0001). Cerebellar dysfunction showed a 4.54% risk difference (p < 0.0001). Bowel and bladder dysfunction showed a 4.98% risk difference (p < 0.0001). These are not small effect sizes for a disease where even modest slowing of progression represents a meaningful improvement in quality of life.
To put this in perspective, many approved MS treatments show similar or smaller effect sizes in clinical trials. And those drugs were specifically designed for MS. Semaglutide and other GLP-1 agonists are showing these benefits as what amounts to a side effect of their primary use for diabetes and weight management. Patients who combine semaglutide with metformin or other metabolic medications showed similar patterns, and the hepatic effects of semaglutide may provide additional metabolic benefits relevant to systemic inflammation.
Why this study matters despite its limitations
This is a retrospective observational study, not a randomized controlled trial. That distinction matters. Patients who received GLP-1 agonists likely had comorbid diabetes or obesity, which could introduce confounding factors. The propensity score matching helps address this, but it cannot eliminate all bias.
Still, the consistency of the results across every functional domain is noteworthy. If the benefit were driven by a confounding factor, you would expect it to appear in some domains but not others. The fact that GLP-1 agonist users showed less progression everywhere suggests a genuine protective effect, though only randomized trials can confirm this definitively.
The single-institution safety study
A separate retrospective cohort study from a single academic MS center examined the tolerability and safety of GLP-1 receptor agonists in MS patients specifically. This study, presented at ACTRIMS Forum, provided practical clinical data that complements the larger epidemiological findings.
Key safety findings
Among the MS patients taking GLP-1 medications, 85% were on semaglutide specifically. The average treatment duration was 15 months. The average weight loss was 12 pounds. And critically, no clinical relapses were identified during GLP-1 treatment.
Side effects were consistent with what the general population experiences on these drugs. About 24% of patients reported gastrointestinal side effects, including constipation, stomach pain, nausea, and vomiting. These are the same gastrointestinal effects seen in non-MS populations.
Two patients developed new lesions on magnetic resonance imaging during the study period. However, the researchers noted that new lesion development occurs in MS regardless of GLP-1 use, and this rate was not higher than expected for the MS population studied. The overall conclusion was that GLP-1 therapy is well tolerated in MS patients and does not appear to worsen disease activity. This safety profile is consistent with what we know about semaglutide side effect timelines in the general population. For people tracking their response to the drug, resources like the dosage charts and dosage calculator help maintain precise dosing.
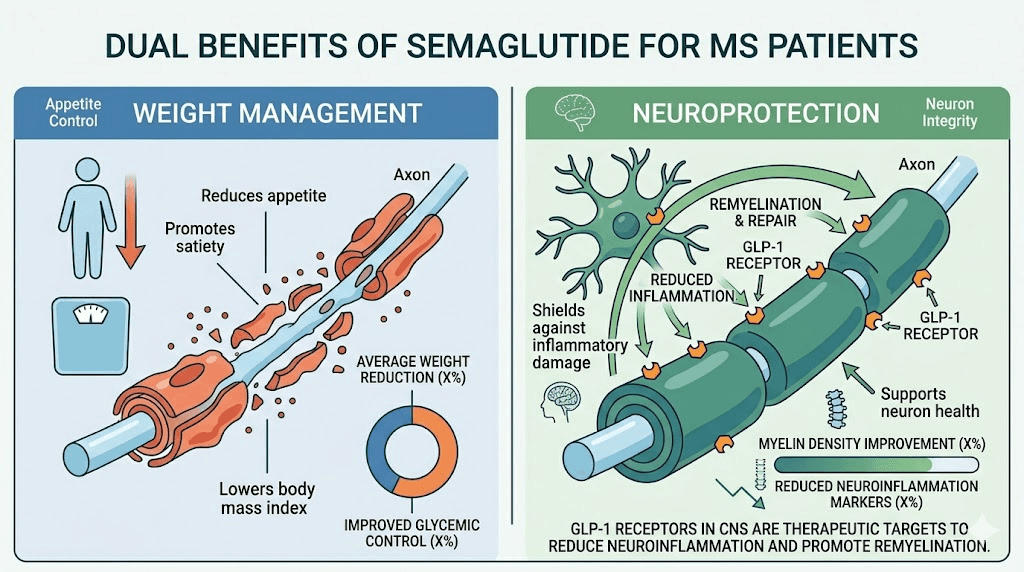
Weight management implications for MS
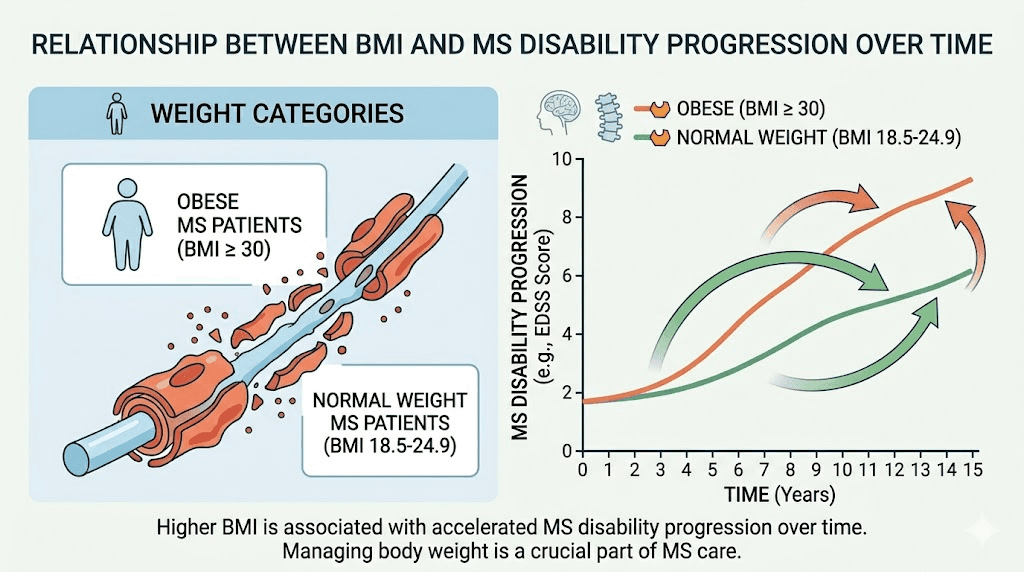
The weight loss component of this study deserves special attention because obesity significantly impacts MS outcomes. Research shows that people with a BMI of 30 or more at the time of MS diagnosis are twice as likely to reach significant disability within six years. Obesity accelerates disability progression, with obese MS patients showing a 0.022-point faster annual increase in Expanded Disability Status Scale scores compared to normal weight patients.
For MS patients struggling with weight gain due to fatigue-related inactivity, medication side effects, or mobility limitations, semaglutide offers a potential dual benefit: managing weight while potentially providing neuroprotective effects. Approximately 42.4% of newly diagnosed MS patients are obese, making weight management a critical and often overlooked aspect of MS care. Understanding the timeline for semaglutide effects can help set realistic expectations. People wondering about the appetite suppression timeline should know that most people notice changes within the first one to two weeks, though full effects develop over several months. SeekPeptides offers comprehensive resources for understanding these timelines and optimizing protocol management.
The NLY01 clinical trial: from lab to patients
Perhaps the most exciting development in the GLP-1 and MS space is the clinical trial program led by Dr. Peter Calabresi at Johns Hopkins University. His team has been studying NLY01, a novel long-acting GLP-1 receptor agonist designed specifically to cross the blood-brain barrier more effectively than existing drugs.
Preclinical results with NLY01
The Johns Hopkins research tested NLY01 in two different mouse models of MS. In prevention trials using the EAE model, NLY01 significantly delayed disease onset and reduced severity. The drug achieved this by inhibiting immune cell activation in the spleen and preventing activated immune cells from trafficking into the central nervous system.
In treatment trials, where NLY01 was given to mice already showing EAE symptoms, the drug produced a significant reduction in clinical scores and secondary attacks. It also protected neurons in the retina, a finding that resonates with the visual problems so many MS patients experience. Optic neuritis, inflammation of the optic nerve, is often one of the first symptoms of MS, and retinal nerve fiber layer thinning is used as a biomarker for neurodegeneration in the disease.
The cuprizone demyelination model
The research team also tested NLY01 in the cuprizone model, which specifically examines demyelination and remyelination without the autoimmune component. Cuprizone is a toxin that selectively kills oligodendrocytes and strips myelin. When the toxin is removed, the brain attempts to remyelinate. This model helps researchers distinguish between anti-inflammatory effects and direct myelin repair effects.
The results from cuprizone studies were more nuanced. While behavioral improvements were observed, the direct remyelination effects of NLY01 in this model presented challenges that researchers are still working to understand. This complexity actually strengthens the overall picture, because it suggests that GLP-1 agonist benefits in MS likely come from both immunomodulation and direct neuroprotection, not just one mechanism.
FDA-approved phase 2 trial
Based on these preclinical findings, D&D Pharmatech received FDA approval to proceed with a phase 2 clinical trial of NLY01 for multiple sclerosis. The trial, led by researchers at Johns Hopkins, will enroll 240 patients and run for 96 weeks. Its primary focus is evaluating whether NLY01 can slow neurodegeneration in MS patients.
This represents a significant milestone. It means the evidence was compelling enough for the FDA to approve human testing specifically for MS. While the trial uses NLY01 rather than semaglutide, both drugs work through the same GLP-1 receptor, and positive results would validate the entire mechanism for MS treatment.
How semaglutide fights neuroinflammation in MS
The anti-inflammatory effects of semaglutide operate through multiple interconnected pathways. Understanding these mechanisms helps explain why the drug shows such consistent benefits in MS research and why it might complement existing disease-modifying therapies rather than replace them.
NF-kB suppression
Nuclear factor kappa B (NF-kB) is one of the master regulators of inflammation in the body. In MS, NF-kB activity is chronically elevated, driving the production of inflammatory cytokines, chemokines, and adhesion molecules that recruit immune cells to attack myelin. Semaglutide suppresses NF-kB activation, effectively turning down the volume on one of the primary inflammatory amplifiers in the disease.
Research shows that semaglutide suppresses LPS-induced expression of key inflammatory regulators in microglia, including Relb and Bcl3. These are transcription factors within the NF-kB family that drive inflammatory gene expression. By targeting these specific regulators, semaglutide reduces microglial activation without completely shutting down the immune surveillance that the brain needs for protection against infections and tumor cells. This selective suppression is exactly what you want in an MS therapy, enough reduction in autoimmune activity to slow disease progression, without leaving the brain defensively vulnerable.
TNF-alpha reduction
Tumor necrosis factor alpha (TNF-alpha) plays a complex role in MS. It is a major inflammatory cytokine that damages oligodendrocytes and promotes demyelination, but it also has some neuroprotective functions through specific receptor subtypes. Anti-TNF therapies have actually worsened MS in clinical trials, which highlights why the nuanced approach of GLP-1 agonists is important.
Semaglutide reduces TNF-alpha levels as part of its broader anti-inflammatory cascade, but it does not eliminate TNF-alpha signaling entirely. The reduction occurs primarily in the context of excessive inflammatory TNF-alpha production, preserving the baseline signaling that supports normal neural function. This is a fundamentally different approach from targeted anti-TNF biologics, and it may explain why semaglutide appears safe in MS while direct TNF inhibitors are not.
Microglial phenotype shifting
Microglia exist in two primary states. M1 microglia are pro-inflammatory, releasing cytokines and reactive oxygen species that damage tissue. M2 microglia are anti-inflammatory and reparative, clearing debris and promoting healing. In MS, the balance shifts heavily toward M1 activation, creating a toxic environment that destroys myelin and neurons.
GLP-1 receptor agonists promote the shift from M1 to M2 microglial phenotypes. This does not just reduce inflammation. It actively promotes repair. M2 microglia release growth factors, clear myelin debris (which is necessary before remyelination can occur), and support oligodendrocyte precursor cell maturation. The combination of reducing damage while promoting repair is what makes the GLP-1 mechanism so compelling for a disease like MS.
Oxidative stress protection
Oxidative stress is a major driver of tissue damage in MS. The inflammatory process generates reactive oxygen species that damage cell membranes, proteins, and DNA. Oligodendrocytes are particularly vulnerable to oxidative stress because of their high metabolic demands and large cell membranes.
Through GSK-3beta inhibition, semaglutide boosts Nrf2 and superoxide dismutase (SOD) levels. Nrf2 is the master regulator of the cellular antioxidant response, controlling the expression of over 200 cytoprotective genes. SOD directly neutralizes superoxide radicals, one of the most damaging reactive oxygen species. By enhancing both of these protective systems, semaglutide provides a robust defense against the oxidative damage that drives progressive neurodegeneration in MS.
Remyelination: the most exciting possibility
If semaglutide could only slow demyelination, that would be valuable. But the research suggests it may also promote remyelination, the regrowth of damaged myelin sheaths. This would represent a fundamentally different kind of therapy, one that repairs damage rather than merely preventing new damage.
How remyelination works
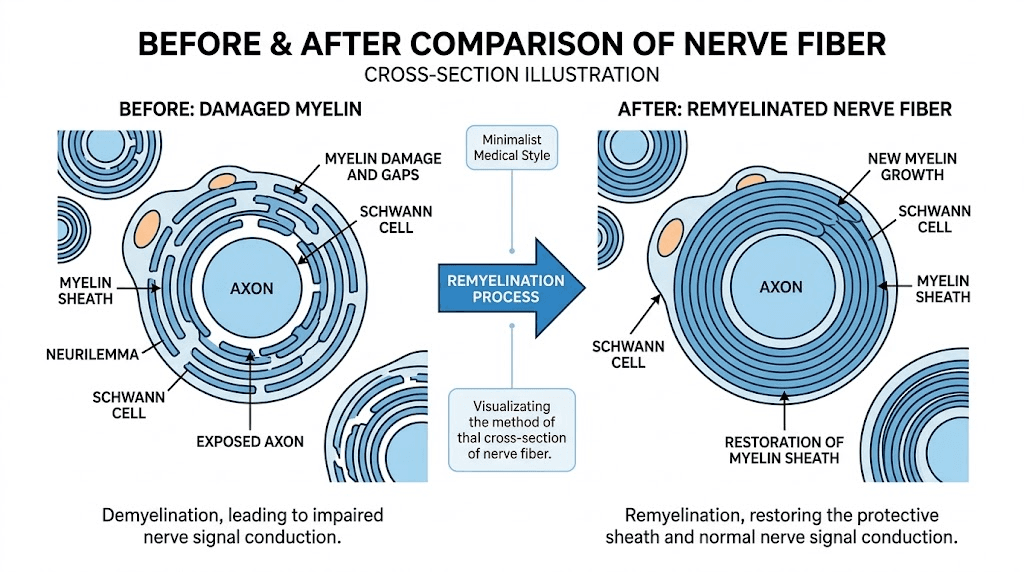
The brain contains oligodendrocyte precursor cells (OPCs) that can mature into myelin-producing oligodendrocytes. In healthy brains, these OPCs are recruited to areas of damage, where they differentiate into mature oligodendrocytes and rewrap nerve fibers with new myelin. In MS, this repair process fails for several reasons: chronic inflammation creates a hostile environment, inhibitory signals accumulate in damaged areas, and OPCs fail to mature properly.
GLP-1 receptor agonists address several of these barriers simultaneously. By reducing the inflammatory environment, they create conditions more conducive to OPC maturation. By activating PI3K/Akt signaling, they directly promote OPC differentiation. And by boosting BDNF through CREB signaling, they provide the neurotrophic support that OPCs need to survive and mature.
Evidence for GLP-1 agonist effects on oligodendrocytes
Research has confirmed that mature oligodendrocytes express GLP-1 receptors, though interestingly, OPCs do not. This means GLP-1 agonists may protect existing myelin-producing cells rather than directly stimulating precursor cell maturation. However, the indirect effects on the cellular environment, reducing inflammation, clearing debris, boosting growth factors, create conditions that strongly favor OPC differentiation and new myelin production.
Studies with liraglutide, another GLP-1 agonist, demonstrated improved remyelination in the cuprizone model through stimulating oligodendrocyte progenitor cell differentiation. While these results were achieved with a different GLP-1 agonist, the shared receptor mechanism suggests semaglutide would produce similar effects. The remyelination observed in the semaglutide EAE study supports this expectation, as CREB/BDNF-mediated remyelination was specifically identified as part of the therapeutic mechanism.
What remyelination means for MS symptoms
Remyelination could potentially reverse some of the disability that MS causes. Nerve fibers that have been stripped of myelin conduct signals slowly and inefficiently, leading to the weakness, numbness, vision problems, and cognitive difficulties that characterize the disease. If new myelin could be laid down, signal conduction would improve, and some lost function could return.
This is not guaranteed, and the extent of remyelination achievable with GLP-1 agonists in humans remains unknown. Animal studies show promising results, but the human brain is more complex and the damage in long-standing MS may be more difficult to repair. Still, the possibility of even partial remyelination distinguishes this approach from current MS therapies, which focus almost exclusively on preventing new damage.
Practical considerations for MS patients considering semaglutide
The research is promising, but it is important to understand what we know, what we do not know, and how to approach this practically. Semaglutide is not approved for MS treatment. Anyone considering it for MS-related reasons needs to make informed decisions in consultation with their neurologist.
Current FDA-approved uses
Semaglutide is currently approved for type 2 diabetes (as Ozempic and Rybelsus) and for chronic weight management (as Wegovy). Many MS patients receive semaglutide legitimately for one of these approved conditions. The research we have discussed applies to these patients, suggesting they may be receiving an unintended neurological benefit alongside the metabolic one. Various forms are available, including oral drops, troches, and standard injectable formulations.
For MS patients without diabetes or obesity, the path to semaglutide is more complicated. Some physicians may prescribe it off-label based on the emerging evidence, but this remains a personal medical decision that depends on individual circumstances, risk tolerance, and the availability of alternatives.
Dosing considerations
The animal studies used specific doses that do not translate directly to human dosing. The EAE study used 25 nmol/kg/day intraperitoneally, which is a different route and dose than the standard human subcutaneous injection. Standard semaglutide dosing for weight management starts at 0.25 mg weekly and titrates up to 2.4 mg weekly over 16-20 weeks.
Whether the doses used for diabetes and weight management provide sufficient brain concentrations for neuroprotection in humans is not yet established. It is possible that the neurological benefits require higher doses, lower doses, or the same doses currently used. The ongoing clinical trials should help answer this question. For now, calculating appropriate doses should always be done in consultation with a prescribing physician. Tools like the peptide calculator and reconstitution calculator can help with precise preparation, and understanding reconstitution procedures is essential for compounded formulations.
Interactions with MS disease-modifying therapies
Most MS patients take one or more disease-modifying therapies (DMTs) such as ocrelizumab, natalizumab, fingolimod, dimethyl fumarate, or others. The safety of combining semaglutide with these medications has not been extensively studied, but the available data is reassuring.
In the single-institution safety study, MS patients taking GLP-1 agonists were also on various DMTs, and no concerning interactions were identified. There are 376 drugs known to interact with semaglutide according to drug interaction databases, but the major interactions involve medications that affect blood sugar levels, not immunological drugs. No specific warnings exist for combining semaglutide with any approved MS therapy.
That said, both GLP-1 agonists and some MS therapies affect immune function. Understanding how semaglutide affects the immune system is important for anyone on immunomodulatory MS treatments. The immune effects of semaglutide appear to be modulatory rather than suppressive, meaning it rebalances immune function rather than broadly suppressing it. This is different from many MS drugs and suggests a favorable interaction profile, but individual monitoring is still essential.
Gastrointestinal side effects and MS
The most common side effects of semaglutide are gastrointestinal: nausea, vomiting, constipation, diarrhea, and acid reflux. For MS patients, these side effects deserve extra consideration because MS itself can cause bowel dysfunction. The bowel and bladder problems are among the most common and distressing MS symptoms, affecting up to 70% of patients.
Adding GI side effects from semaglutide on top of existing MS-related bowel issues could be problematic for some individuals. The 24% GI side effect rate observed in the MS safety study is actually lower than the rates seen in the general population on these drugs, which may suggest that MS patients tolerate semaglutide reasonably well. However, starting at the lowest dose and titrating slowly is especially important for MS patients with pre-existing bowel symptoms. Many people find that dietary adjustments and practical tips can minimize these effects.
Fatigue and energy considerations
MS fatigue is one of the most debilitating and poorly understood symptoms of the disease. It affects up to 80% of MS patients and is often described as completely different from normal tiredness. Some semaglutide users report fatigue as a side effect, particularly during the titration phase. This could temporarily worsen MS-related fatigue.
However, the opposite could also be true. Weight loss reduces the physical burden on the body, which can improve energy levels. And the anti-inflammatory effects of semaglutide might address some of the inflammatory drivers of MS fatigue. The net effect on fatigue is likely to vary from person to person, and monitoring this symptom closely during the first few months is important. Understanding whether semaglutide affects energy levels positively or negatively requires individual assessment.
The obesity and MS connection
Even setting aside the neuroprotective effects, semaglutide has a strong rationale for MS patients purely through weight management. The relationship between obesity and MS is bidirectional and deeply consequential.
How obesity accelerates MS progression
Research published in Neurology: Neuroimmunology and Neuroinflammation demonstrated that obesity affects MS disease activity, progression, cognitive functioning, and quality of life. Obese MS patients showed faster disability accumulation, more brain atrophy, and worse cognitive performance compared to normal-weight patients.
The numbers are stark. People with a BMI of 30 or more at diagnosis are twice as likely to reach significant disability within six years. Obesity was associated with a 0.022-point faster annual increase in EDSS scores. Over a decade, that adds up to an additional 0.22 points of disability, which can mean the difference between walking independently and needing a cane. Understanding BMI requirements for GLP-1 therapy helps determine eligibility, and the average monthly weight loss data sets realistic expectations for MS patients starting treatment.
Adipose tissue is not inert. It is an active endocrine organ that produces inflammatory cytokines, including TNF-alpha, IL-6, and leptin. These cytokines contribute to the systemic inflammation that drives MS. Reducing adipose tissue through effective weight loss could lower this inflammatory burden and slow disease progression independently of any direct neuroprotective effects.
Why weight management is so difficult with MS
MS creates a vicious cycle with weight gain. Fatigue limits physical activity. Reduced mobility limits exercise options. Some MS medications, particularly corticosteroids used for relapse treatment, promote weight gain. Depression, which affects up to 50% of MS patients, can lead to emotional eating. And the disease itself may alter metabolic rate through hypothalamic inflammation.
Traditional weight loss approaches, eat less and exercise more, are not always feasible for someone dealing with severe fatigue, mobility limitations, heat sensitivity (exercise intolerance is common in MS), and the cognitive load of managing a complex chronic disease. Semaglutide combined with appropriate exercise offers a pharmacological approach to appetite suppression and metabolic improvement that does not depend on the patient ability to maintain intensive exercise programs. Some MS patients also wonder about losing weight on semaglutide without exercise, which is feasible though combining movement when possible improves outcomes. The early weeks without weight loss are normal and should not discourage continuation, as the drug requires time to reach therapeutic levels.
Weight loss results in MS patients
The single-institution study reported an average weight loss of 12 pounds over 15 months in MS patients on GLP-1 therapy. This is modest compared to the results seen in the general population, where semaglutide typically produces 15-20% body weight reduction. The difference could be due to MS-related factors that limit weight loss, lower doses used, shorter treatment duration, or the complicating effects of MS medications.
Even 12 pounds of weight loss, however, can be clinically meaningful for an MS patient. Reduced body weight means less mechanical stress on joints affected by spasticity, less energy expenditure for mobility, and reduced inflammatory cytokine production from adipose tissue. For someone navigating MS, every improvement in physical function counts. Setting realistic expectations by understanding the weight loss timeline is important. Reviewing before and after results from other patients provides visual context for what to expect, and understanding how to handle weight loss plateaus helps maintain progress over time.
How semaglutide compares to current MS treatments
To understand where semaglutide might fit in the MS treatment landscape, it helps to compare its mechanism with existing disease-modifying therapies.
Current MS treatment approaches
Existing MS treatments fall into several categories. Interferon-based therapies (like interferon beta-1a) were among the first DMTs and work by broadly modulating immune function. B-cell depleting therapies (like ocrelizumab) target specific immune cells involved in MS attacks. S1P receptor modulators (like fingolimod) trap lymphocytes in lymph nodes, preventing them from reaching the brain. And newer therapies like cladribine deplete specific lymphocyte populations.
All of these approaches share a common strategy: suppress or redirect the immune attack on myelin. None of them directly promote remyelination or provide neuroprotection independent of their immunomodulatory effects. This is the critical gap that GLP-1 agonists might fill.
What semaglutide adds to the picture
Semaglutide works through fundamentally different mechanisms than any approved MS therapy. Its benefits include immunomodulation (not immunosuppression), direct neuroprotection via PI3K/Akt signaling, antioxidant enhancement through Nrf2 activation, potential remyelination support through BDNF upregulation, weight management benefits, and metabolic improvements that reduce systemic inflammation.
This complementary mechanism of action suggests that semaglutide could potentially be used alongside existing MS therapies rather than replacing them. A patient might take ocrelizumab to prevent immune attacks while semaglutide provides neuroprotective support and metabolic benefits. This combination approach is exactly what the current clinical trials are designed to evaluate. Understanding the differences between semaglutide and tirzepatide may also inform which GLP-1 agonist might best complement a given MS treatment regimen. The broader landscape of peptides for weight loss provides additional context for metabolic management options.
The neuroprotection gap in MS treatment
One of the biggest unmet needs in MS treatment is neuroprotection. Current therapies reduce relapse frequency and slow disability progression by dampening the immune attack. But they do relatively little to protect neurons and oligodendrocytes from the damage that still occurs. Progressive MS, where disability accumulates independently of relapses, is particularly poorly served by current treatments.
Semaglutide potential to provide neuroprotection, not just immunomodulation, makes it especially interesting for progressive forms of MS. The PI3K/Akt/GSK-3beta pathway activation, Nrf2-mediated antioxidant enhancement, and BDNF-driven neurotrophic support could protect nerve cells from the slow-burning neurodegeneration that drives progressive disability. The cardiovascular safety profile of semaglutide is also relevant here, as MS patients face elevated cardiovascular risk from reduced mobility and chronic inflammation. Some patients explore alternatives like phentermine for weight management, but these lack the neuroprotective mechanisms that make semaglutide uniquely interesting for MS.
The epidemiological evidence: GLP-1 and reduced MS risk
Beyond treating existing MS, there is intriguing evidence that GLP-1 agonists might reduce the risk of developing MS in the first place. While this is less directly relevant for people already diagnosed, it reinforces the biological plausibility of the protective mechanism.
Reduced MS incidence in GLP-1 users
Analysis of the FDA Adverse Event Reporting System database revealed striking associations. Semaglutide use was associated with a 76.2% lower likelihood of developing MS. Dulaglutide showed an 83.5% reduction. Liraglutide showed an 83.9% reduction. These numbers come from pharmacovigilance data rather than controlled trials, so they should be interpreted with caution. But the consistency across multiple GLP-1 agonists and the magnitude of the effect are notable.
A separate real-world study found that obesity medications, including GLP-1 agonists, were tied to a reduced chance of MS diagnosis. This could be partly explained by weight loss reducing the inflammatory state that increases MS risk, but the neuroprotective mechanisms of GLP-1 agonists likely contribute independently.
What this means for MS prevention research
These epidemiological findings do not prove that semaglutide prevents MS. They show an association that is consistent with the biological mechanisms we have discussed. Confounding factors could explain part of the effect. People taking GLP-1 agonists may differ from the general population in ways that independently affect MS risk.
However, the convergence of epidemiological data, animal studies, and human cohort studies all pointing in the same direction makes a compelling case for further research. If even a fraction of the protective effect seen in these observational studies holds up in controlled trials, GLP-1 agonists could become an important tool not just for treating MS but for preventing it in high-risk individuals.
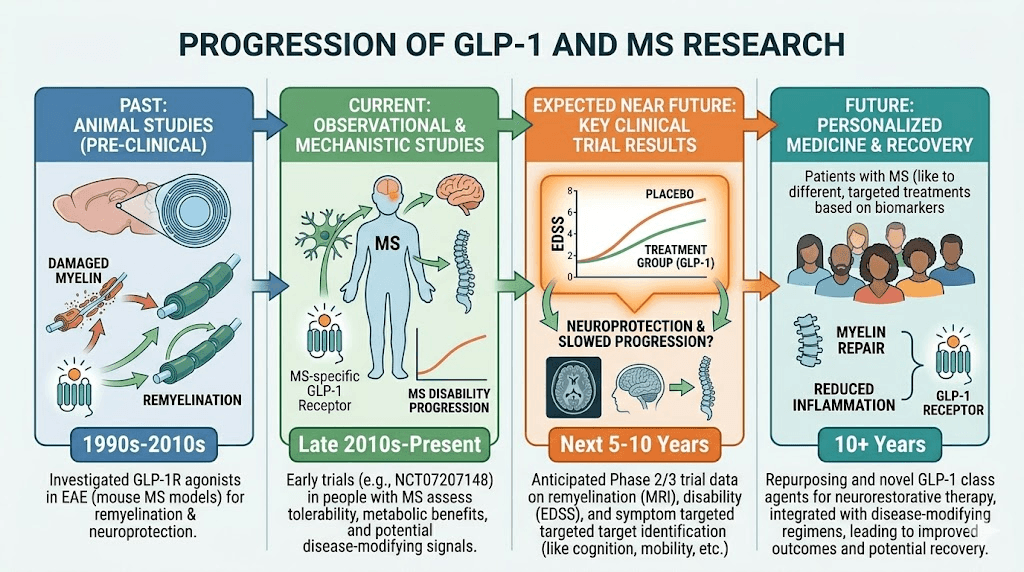
Ongoing clinical trials and future research
The field is moving rapidly. Several clinical trials are underway or planned that will provide much stronger evidence about the role of GLP-1 agonists in MS.
The NLY01 phase 2 trial
The Johns Hopkins-led phase 2 trial of NLY01 in MS will enroll 240 patients and run for 96 weeks. This trial is specifically designed to evaluate whether a GLP-1 receptor agonist can slow neurodegeneration in MS patients. Primary outcomes will likely include brain volume measurements (atrophy is a marker of neurodegeneration), disability progression scores, and possibly biomarkers of myelin integrity.
This trial is particularly important because NLY01 was designed to cross the blood-brain barrier more effectively than existing GLP-1 agonists. If it shows clear benefits, it would strongly support developing brain-penetrant GLP-1 agonists specifically for neurological conditions.
The semaglutide/dulaglutide MS trial
Another clinical trial is studying the effects of semaglutide and dulaglutide in adults with MS aged 18-60 with high disease activity. This trial is expected to conclude by December 2026, with data collected to determine the effects on chronic axonal damage and neurodegeneration. This trial uses commercially available drugs at standard doses, making its results more immediately applicable to clinical practice.
What we still need to learn
Despite the promising evidence, significant questions remain. What is the optimal dose for neuroprotection in MS? Do benefits differ between relapsing and progressive forms of MS? How long do neuroprotective effects persist after stopping the drug? Do benefits increase with longer treatment duration? Are certain GLP-1 agonists more effective than others for MS? Can GLP-1 agonists produce measurable remyelination in human brains? How do GLP-1 agonists interact with specific MS disease-modifying therapies?
The ongoing trials should begin answering some of these questions within the next few years. In the meantime, the preclinical and observational evidence provides a strong rationale for cautious optimism. For researchers and clinicians, SeekPeptides provides comprehensive resources to stay current on the latest peptide research developments.
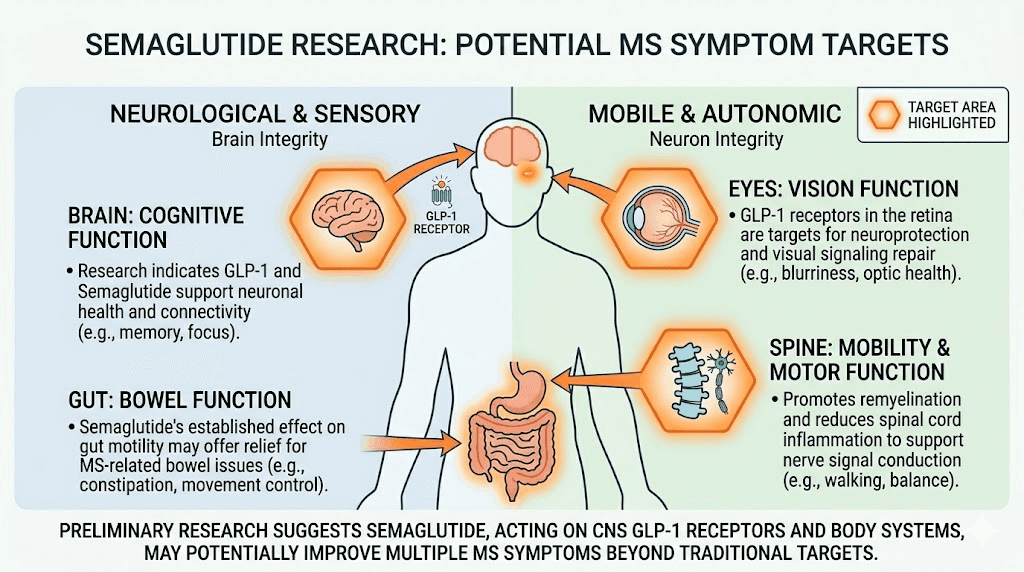
Semaglutide and MS-related symptoms: what the research suggests
Beyond the disease-modifying potential, semaglutide may affect several specific MS symptoms through its metabolic, anti-inflammatory, and neuroprotective actions.
Cognitive function
Cognitive impairment affects 40-70% of MS patients. Memory problems, slowed processing speed, and difficulty with attention and executive function are among the most common complaints. The EAE study showed that semaglutide improved cognitive function in mice, specifically memory and recognition tasks assessed through novel object recognition testing.
The mechanism is plausible. Hippocampal damage, which semaglutide reduced in the animal model, directly correlates with memory impairment in MS. BDNF upregulation through CREB signaling supports synaptic plasticity, the cellular basis of learning and memory. And reducing neuroinflammation in the hippocampus and cortex could improve the cognitive environment broadly.
Human evidence is indirect but supportive. The EVOKE trials for Alzheimer disease showed semaglutide effects on neurodegeneration biomarkers in humans. Cognitive outcomes in MS patients on GLP-1 agonists have not been formally studied yet, but the biological rationale is strong. People experiencing cognitive changes should track them carefully, potentially using tools like those available through GLP-1 monitoring resources.
Fatigue
MS fatigue has multiple drivers: neuroinflammation, demyelination, sleep disruption, depression, and the sheer energy cost of a body fighting to compensate for neurological damage. Semaglutide could address several of these. Reduced neuroinflammation might lower the inflammatory component of fatigue. Weight loss reduces the physical effort of daily activities. And improved sleep, which some GLP-1 agonist users report, could help with the restorative rest that MS patients desperately need.
The flip side is that semaglutide can cause fatigue, especially during dose titration. For MS patients, this temporary increase in fatigue needs to be weighed against potential long-term benefits. Gradual titration starting at the lowest dose is critical for managing this transition period. People who notice persistent low energy should review whether semaglutide eventually improves energy as the body adapts, and whether sleep disruption could be contributing to daytime fatigue.
Pain and sensory symptoms
Neuropathic pain affects approximately 50% of MS patients. It results from damaged nerve fibers sending inappropriate pain signals. The anti-inflammatory and neuroprotective effects of semaglutide could theoretically reduce some of this pain by protecting nerve fibers and reducing the inflammatory environment that sensitizes pain pathways.
The allodynia research is particularly interesting here. Studies examining semaglutide and allodynia suggest effects on pain processing pathways that could be relevant for MS-related neuropathic pain. However, no studies have specifically examined semaglutide effects on MS pain, so this remains theoretical.
Vision
Optic neuritis is one of the most recognizable MS symptoms, and visual pathway damage is common even in patients who have never experienced a clinical episode of optic neuritis. The NLY01 studies at Johns Hopkins specifically showed retinal neuroprotection in EAE mice, which is directly relevant to visual function in MS.
Retinal nerve fiber layer (RNFL) thickness is used as a biomarker for neurodegeneration in MS clinical trials. If GLP-1 agonists can preserve RNFL thickness, this would provide objective evidence of neuroprotection that is directly linked to visual function preservation.
Nutrition and lifestyle strategies for MS patients on semaglutide
For MS patients considering or already taking semaglutide, optimizing nutrition and lifestyle factors is especially important. The combination of MS and semaglutide creates unique nutritional needs and challenges. Resources on GLP-1 friendly recipes, breakfast ideas, and dinner planning can help address the practical challenges of eating well while managing appetite changes.
Protein intake for muscle preservation
Both MS and semaglutide-induced weight loss can lead to muscle loss. MS causes muscle wasting through reduced physical activity, neurogenic atrophy, and inflammation-driven catabolism. Semaglutide weight loss involves both fat and lean tissue reduction. Together, this creates a significant risk of sarcopenia that must be actively managed.
MS patients on semaglutide should aim for 1.2-1.6 grams of protein per kilogram of body weight daily, at the higher end of general recommendations. Understanding protein requirements on semaglutide becomes even more critical when MS-related muscle preservation is a concern. High-quality protein sources include lean meats, fish, eggs, dairy, and legumes. Spreading protein intake across meals is more effective than consuming it all at once.
Anti-inflammatory nutrition
Diet can either fuel or fight inflammation. For MS patients on semaglutide, an anti-inflammatory diet amplifies the drug anti-inflammatory effects. The semaglutide diet plan should emphasize omega-3 fatty acids from fatty fish, walnuts, and flaxseeds, which reduce pro-inflammatory cytokines. Colorful fruits and vegetables provide antioxidants that complement semaglutide Nrf2-mediated oxidative stress protection. Fiber from whole grains and vegetables supports gut health, which is increasingly recognized as important in autoimmune conditions.
Foods to emphasize include salmon, sardines, leafy greens, berries, turmeric, extra virgin olive oil, and fermented foods. Understanding what foods work best with semaglutide helps maximize both weight management and anti-inflammatory benefits. Foods to avoid include processed meats, refined sugars, excessive saturated fats, and ultra-processed foods that promote inflammation.
Vitamin D considerations
Vitamin D deficiency is exceptionally common in MS and may contribute to disease progression. MS patients should have their vitamin D levels checked regularly and supplement as needed, typically aiming for blood levels of 40-60 ng/mL. Semaglutide does not directly affect vitamin D metabolism, but reduced food intake during appetite suppression could limit dietary vitamin D sources. Taking a vitamin D supplement is generally recommended for MS patients regardless of semaglutide use.
Supplements that complement both conditions
Several supplements have evidence for benefit in both MS and semaglutide use. B12 is important because MS and semaglutide can both affect absorption, and B12 deficiency causes neurological symptoms that mimic MS. Many semaglutide formulations include B12 for this reason. Omega-3 supplements support the anti-inflammatory effects of both the drug and the dietary approach. Magnesium can help with muscle cramps and spasticity, both common in MS and sometimes exacerbated by reduced food intake. L-carnitine supplementation may support both energy metabolism and neuroprotection. Some patients also explore niacinamide combinations and pyridoxine (B6) formulations. A comprehensive guide to supplements with GLP-1 medications can help identify the most beneficial additions. Choosing the right electrolyte supplement is especially important for MS patients who may already be at risk for electrolyte imbalances.
Exercise strategies
Exercise is one of the most evidence-based interventions for MS, improving strength, balance, fatigue, and quality of life. Semaglutide-induced weight loss can make exercise easier by reducing the physical burden. However, MS patients face unique challenges including heat sensitivity (Uhthoff phenomenon), fatigue, balance issues, and spasticity.
The ideal exercise approach for MS patients on semaglutide combines resistance training (for muscle preservation during weight loss), aquatic exercise (which provides cooling and buoyancy), and gentle cardiovascular activity. Combining semaglutide with exercise produces better outcomes than either intervention alone, and this is especially true for MS patients where exercise has independent disease-modifying effects.
Questions to ask your neurologist about semaglutide
If you have MS and are interested in semaglutide, whether for weight management, diabetes, or potential neuroprotective benefits, here are informed questions for your care team.
About safety with your specific MS treatment
Ask whether semaglutide has any known interactions with your specific disease-modifying therapy. Ask about monitoring parameters, what should be tracked more closely if you start semaglutide while on MS treatment. Ask whether any adjustments to your current medications might be needed.
About managing overlapping symptoms
If you already have MS-related bowel dysfunction, ask how to manage potential GI side effects from semaglutide. If fatigue is a major issue, discuss the expected timeline for semaglutide-related fatigue versus potential long-term energy improvements from weight loss. If you have bladder issues, discuss how appetite suppression and reduced fluid intake might affect bladder function.
About monitoring disease activity
Ask whether any additional MRI monitoring should be done while on semaglutide. Discuss how to distinguish between MS-related symptoms and semaglutide side effects. Ask about tracking cognitive function to see if semaglutide provides any benefit in this area.
About the emerging research
Ask whether your neurologist has seen the TriNetX cohort data and what they think of it. Ask whether they are aware of any clinical trials you might be eligible for. Discuss whether the potential neuroprotective benefits factor into the risk-benefit analysis for your specific case.
Understanding the different types of MS and how semaglutide might apply
MS is not one disease. It exists on a spectrum, and the relevance of semaglutide may differ across types.
Relapsing-remitting MS (RRMS)
RRMS is characterized by discrete attacks (relapses) followed by partial or complete recovery. This is the most common form at diagnosis. For RRMS patients, semaglutide potential benefits include reduced relapse severity through immunomodulation, neuroprotection during and between relapses, weight management to reduce disability risk, and potential support for remyelination during recovery periods.
The TriNetX cohort study showed benefits across functional domains in a population that likely included many RRMS patients. The absence of clinical relapses during GLP-1 treatment in the safety study is encouraging, though the study was not powered to detect relapse rate changes.
Secondary progressive MS (SPMS)
SPMS develops when RRMS transitions to a phase of gradual disability accumulation, with or without continued relapses. This is where current treatments are least effective, because the damage becomes driven more by neurodegeneration than by acute immune attacks.
Semaglutide potential for neuroprotection makes it particularly interesting for SPMS. The PI3K/Akt/GSK-3beta pathway activation, antioxidant enhancement, and BDNF-driven neurotrophic support could address the neurodegenerative component that current therapies largely miss. The NLY01 phase 2 trial specifically focuses on slowing neurodegeneration, which is the central problem in SPMS.
Primary progressive MS (PPMS)
PPMS involves gradual disability accumulation from onset without clear relapses. Only one drug, ocrelizumab, has shown modest benefit in PPMS. The neuroprotective mechanisms of GLP-1 agonists could be particularly relevant here, as the disease is driven primarily by chronic neurodegeneration and progressive demyelination.
However, PPMS patients have not been specifically studied in the context of GLP-1 agonists. The TriNetX study likely included some PPMS patients, but the results were not stratified by MS type. Future research specifically examining GLP-1 agonist effects in progressive MS is critically needed.
Semaglutide, the gut-brain axis, and MS
An emerging area of research connects the gut microbiome to MS through the gut-brain axis. Semaglutide effects on gut function and the microbiome may have additional relevance for MS patients.
The gut-brain connection in MS
Research increasingly shows that MS patients have altered gut microbiomes compared to healthy controls. Specific bacterial populations are over- or under-represented, and these changes correlate with disease activity. The gut microbiome influences immune function through multiple pathways, including regulatory T cell development, cytokine production, and intestinal barrier integrity.
Semaglutide slows gastric emptying and alters gut transit time, which can change the gut environment in ways that affect bacterial populations. While the specific effects of semaglutide on the MS-relevant microbiome have not been studied, the connection is biologically plausible and represents another potential pathway through which the drug could influence MS outcomes.
Gut permeability and autoimmunity
Increased intestinal permeability, sometimes called leaky gut, has been observed in MS patients. This allows bacterial products and food antigens to enter the bloodstream, potentially triggering or amplifying autoimmune responses. GLP-1 receptor agonists have been shown to improve intestinal barrier function in some contexts, which could reduce the autoimmune trigger load in susceptible individuals. Understanding the relationship between gut health and peptide research provides additional context for this connection.
Probiotics and semaglutide for MS
Given the microbiome connection, combining semaglutide with targeted probiotic supplementation could theoretically enhance both metabolic and immunological outcomes for MS patients. Specific probiotic strains have shown benefit in MS mouse models, and supporting gut health during semaglutide treatment makes physiological sense. However, this combination has not been clinically tested in MS patients specifically.
Comparing semaglutide with other GLP-1 agonists for MS
Semaglutide is not the only GLP-1 agonist being studied in the context of MS. Understanding how different drugs in this class compare helps contextualize the research.
Semaglutide versus liraglutide
Liraglutide was one of the first GLP-1 agonists studied for neurological conditions. It has a shorter half-life than semaglutide and requires daily injection rather than weekly. In the cuprizone demyelination model, liraglutide improved behavioral profiles and promoted remyelination through oligodendrocyte progenitor cell differentiation. However, semaglutide generally achieves greater weight loss and has stronger metabolic effects, and the weekly dosing improves adherence. Understanding the differences between these drugs helps inform treatment decisions. The storage and shelf life considerations also differ between formulations, which matters for patients managing complex medication regimens.
Semaglutide versus exenatide
Exenatide has the most extensive neurological research history among GLP-1 agonists, particularly for Parkinson disease. A phase 2 trial showed sustained improvement in motor function in Parkinson patients. In MS-related research, exenatide has shown delay in disease onset and reduced severity in EAE models. The Parkinson data provides a proof of concept that GLP-1 agonists can have meaningful clinical neurological benefits in humans.
NLY01 versus semaglutide
NLY01 was specifically engineered for enhanced blood-brain barrier penetration and sustained GLP-1 receptor activation in the central nervous system. If it proves more effective than standard GLP-1 agonists for MS in clinical trials, it could lead to the development of brain-targeted GLP-1 agonists specifically for neurological conditions. For now, semaglutide is the only commercially available option with significant MS-related evidence.
Tirzepatide considerations
Tirzepatide is a dual GIP/GLP-1 receptor agonist that has shown superior weight loss compared to semaglutide. Whether the additional GIP receptor activation provides additional neurological benefits for MS is unknown. Some MS patients already use tirzepatide for autoimmune considerations, and the comparison between semaglutide and tirzepatide side effects may influence the choice for MS patients prioritizing specific outcomes. The question of using both agents together has also been explored, though this is not recommended without medical supervision. For those considering tirzepatide specifically, understanding its relationship with autoimmune conditions like lupus provides additional context.
What the skeptics say and why caution is warranted
It is important to present a balanced view. Not everyone in the neurology community is convinced that GLP-1 agonists will prove beneficial for MS, and there are legitimate reasons for caution.
The evidence hierarchy
The strongest evidence, randomized controlled trials in humans, does not yet exist for GLP-1 agonists in MS. What we have is animal studies (strong but not always translatable), retrospective cohort studies (suggestive but confounded), and small safety studies (reassuring but underpowered for efficacy). Each step up the evidence hierarchy could reveal that the benefits are smaller than expected, or that risks emerge that were not apparent in earlier studies.
The translation gap
Many drugs that show remarkable results in EAE mouse models fail in human MS trials. The EAE model is imperfect. It does not fully replicate the complexity of human MS, particularly the progressive forms. The doses used in animal studies often do not translate directly to human dosing. And the mouse immune system, while similar to the human system, has important differences.
Potential risks
While the safety data is reassuring so far, long-term effects of GLP-1 agonists on the immune system are not fully characterized. The immunomodulatory effects that benefit MS could theoretically interfere with immune surveillance in ways that take years to manifest. The pancreatic safety concerns that apply to all GLP-1 agonist users (rare but potential pancreatitis risk, uncertain long-term thyroid effects) apply equally to MS patients.
For MS patients specifically, the GI side effects could be problematic given existing bowel dysfunction. The weight loss, while generally beneficial, needs to be managed carefully to prevent muscle loss that could worsen mobility. And the interaction between semaglutide immunomodulatory effects and existing immunosuppressive MS therapies needs more study.
The commercial factor
It would be naive not to mention that GLP-1 agonists are among the most profitable drugs in pharmaceutical history. The enormous commercial success of these drugs creates incentives for research that could inflate expectations. Independent, publicly funded research is essential to provide unbiased evidence about GLP-1 agonists in MS. The NLY01 trial, funded partly through the Department of Defense Multiple Sclerosis Research Program, represents exactly this kind of rigorous, independent investigation.
Autoimmune conditions beyond MS: what the broader picture tells us
The relationship between semaglutide and MS does not exist in isolation. Research is exploring GLP-1 agonists across multiple autoimmune conditions, and the broader pattern strengthens the case for MS-specific benefits.
Rheumatoid arthritis
GLP-1 agonists have shown anti-inflammatory effects in rheumatoid arthritis models, reducing joint inflammation and cartilage destruction. The shared inflammatory pathways between RA and MS suggest common mechanisms of benefit.
Inflammatory bowel disease
GLP-1 receptor activation reduces intestinal inflammation in preclinical IBD models. Given the gut-brain axis connection to MS, benefits in IBD could have secondary relevance for MS through reduced intestinal permeability and altered immune cell trafficking.
Hashimoto thyroiditis
The relationship between Hashimoto disease and GLP-1 is being explored, with potential immunomodulatory benefits for autoimmune thyroid conditions. MS patients have higher rates of other autoimmune conditions, including Hashimoto thyroiditis, making this research doubly relevant. Research into GLP-1 specifically for Hashimoto provides additional context.
The autoimmune pattern
Across autoimmune conditions, GLP-1 agonists consistently show reduced inflammatory cytokine production, enhanced regulatory T cell function, M2 macrophage polarization, and NF-kB suppression. This pattern suggests a genuine immunomodulatory effect rather than condition-specific findings. For MS patients who may also manage other autoimmune conditions, semaglutide potential benefits could extend across multiple disease processes. Understanding GLP-1 approaches for autoimmune conditions provides a broader perspective on this emerging field.
Hormonal considerations for MS patients on semaglutide
MS affects women at roughly three times the rate of men, and hormonal factors play a significant role in both MS and semaglutide response. Understanding these interactions is important for the majority of MS patients who are female.
Estrogen, MS, and semaglutide
Estrogen has known neuroprotective effects, which may partly explain why MS is less aggressive during pregnancy (when estrogen levels are high) and why it worsens postpartum. Weight loss affects estrogen levels because adipose tissue produces estrogen. Significant weight loss on semaglutide could theoretically reduce estrogen production, which might have implications for MS disease activity. Research on whether semaglutide affects estrogen is still evolving, but MS patients should be aware of this potential interaction.
Menstrual effects
Some women report menstrual changes on semaglutide, and MS itself can affect menstrual regularity. Changes in menstrual patterns could affect MS symptom fluctuations that many women experience in relation to their cycle. Tracking these changes can help distinguish between drug effects and disease-related patterns. Understanding the broader hormonal picture, including the estrogen effects and libido changes some women experience, provides a more complete view.
Fertility considerations
MS is commonly diagnosed in women of reproductive age. Semaglutide is not recommended during pregnancy, and the drug manufacturer recommends stopping it at least two months before attempting to conceive. MS patients planning pregnancy should discuss this with both their neurologist and reproductive specialist. Understanding the considerations around semaglutide and pregnancy planning is essential for this population.
HRT and GLP-1 interactions
Some MS patients, particularly those in perimenopause or menopause, take hormone replacement therapy. The interaction between HRT and GLP-1 medications is an area of active research. For MS patients, the combined neuroprotective effects of estrogen replacement and GLP-1 receptor activation could theoretically be synergistic, but this has not been studied.
Building a comprehensive MS management plan with semaglutide
For MS patients who are already taking or seriously considering semaglutide, integrating it into a comprehensive management plan maximizes potential benefits. SeekPeptides provides comprehensive protocol resources and community support for people navigating peptide-based treatments alongside complex medical conditions.
Monitoring framework
MS patients on semaglutide should have baseline measurements before starting the drug, including brain MRI with volumetric analysis, EDSS score, cognitive testing (at minimum, the Symbol Digit Modalities Test), weight, BMI, and body composition, HbA1c and fasting glucose, lipid panel, vitamin D level, and B12 level.
Regular monitoring should include MRI per standard MS protocols (typically annually), clinical assessment for MS symptoms and semaglutide side effects, weight and body composition tracking, and nutritional status assessment. Using a systematic approach to tracking GLP-1 treatment can help identify both benefits and concerns early. The blood work considerations for semaglutide users are straightforward, and storage and expiration awareness ensures medication potency throughout treatment. For MS patients who travel with semaglutide, maintaining proper cold chain storage is especially important.
Titration strategy for MS patients
Given the unique fatigue and GI challenges of MS, an even slower titration than standard may be appropriate. Starting at 0.25 mg weekly for a full four weeks (rather than the standard two to four) before increasing allows MS patients to separate drug side effects from disease fluctuations. Each dose increase should be followed by a monitoring period to assess impact on MS symptoms before proceeding further.
When to stop or pause
MS patients should pause semaglutide during acute relapses being treated with high-dose corticosteroids, as the metabolic effects of steroids could interact unpredictably with semaglutide. If new or worsening neurological symptoms develop that cannot be attributed to either MS disease activity or known semaglutide side effects, the drug should be held pending medical evaluation. Understanding the considerations for stopping semaglutide and restarting after a break helps plan for these situations. Patients should also be aware of potential withdrawal effects when discontinuing, and the troubleshooting guide for non-responders provides helpful strategies if the drug does not seem to be working as expected. SeekPeptides members access detailed protocol adjustment guides for exactly these scenarios.
Frequently asked questions
Is semaglutide approved for treating multiple sclerosis?
No. Semaglutide is currently approved only for type 2 diabetes and chronic weight management. The research on semaglutide and MS is promising but preliminary. Clinical trials are underway, and it may take several years before regulatory approval for MS could be considered. MS patients who take semaglutide currently do so for its approved indications, with any neurological benefits being an added potential advantage.
Can semaglutide replace my current MS medication?
No. Semaglutide should not be used as a replacement for approved disease-modifying therapies for MS. The evidence suggests it could potentially complement existing treatments, not replace them. Never stop your MS medication to start semaglutide without discussing this with your neurologist.
Is semaglutide safe to take with ocrelizumab or other MS drugs?
The limited evidence available suggests that GLP-1 agonists are well tolerated alongside MS disease-modifying therapies, with no concerning interactions identified in the studies conducted so far. However, comprehensive safety data for specific drug combinations is lacking. Your neurologist should be aware of all medications you are taking.
How long would I need to take semaglutide to see neurological benefits?
This question cannot be answered definitively yet. The TriNetX cohort study showed benefits over a five-year follow-up period. The standard treatment duration for semaglutide varies, but neurological benefits likely require sustained treatment. The ongoing clinical trials will help establish optimal treatment duration for neurological outcomes.
Will my insurance cover semaglutide if I have MS but not diabetes or obesity?
Currently, insurance coverage for semaglutide requires an approved indication (type 2 diabetes or obesity with BMI of 30 or above, or 27 with comorbidities). MS alone would not qualify for coverage. If you meet criteria for an approved indication, coverage depends on your specific plan. Some people explore compounded semaglutide options as an alternative.
Does the type of MS I have matter for semaglutide potential benefits?
Potentially, yes. The neuroprotective mechanisms may be most relevant for progressive forms of MS (SPMS and PPMS), where neurodegeneration drives disability progression. The immunomodulatory effects may be more relevant for relapsing forms. However, the research has not yet stratified results by MS type, so this remains speculative.
Can semaglutide promote remyelination in humans?
Animal studies show evidence of remyelination support through multiple mechanisms, including BDNF upregulation and oligodendrocyte protection. Whether this translates to clinically meaningful remyelination in humans with MS is not yet known. This is one of the key questions the ongoing clinical trials should help answer.
What about oral semaglutide for MS?
Oral semaglutide (Rybelsus) achieves lower blood levels than the injectable form, which could mean less brain penetration and weaker neuroprotective effects. For MS-related potential benefits, injectable semaglutide would theoretically be preferred due to higher and more consistent blood levels. However, no studies have directly compared oral versus injectable semaglutide for neurological outcomes.
External resources
TriNetX cohort study: GLP-1 agonists and MS progression (Neurology)
Brain Plasticity and GLP-1 Receptor Agonist Treatment clinical trial
Department of Defense MS Research Program: GLP-1R Agonist Highlight
For researchers serious about staying ahead of the rapidly evolving GLP-1 and neurological research landscape, SeekPeptides provides evidence-based guides, comprehensive protocol databases, and a community of thousands navigating these exact questions. Members access detailed resources on peptide mechanisms, safety profiles, and the latest research developments, making it the most trusted platform for peptide education and informed decision-making.
In case I do not see you, good afternoon, good evening, and good night. May your myelin stay intact, your inflammation stay quiet, and your research stay thorough.