Feb 27, 2026

Before you increase your semaglutide dose, there is something your prescriber probably has not mentioned. Something that does not appear on the official drug label. Something that a growing number of researchers and patients are reporting with increasing urgency.
Your skin might start hurting. Not at the injection site. Everywhere.
Allodynia, a condition where normally painless sensations become painful, is emerging as a rare but significant side effect of semaglutide and other GLP-1 receptor agonists. The bedsheets against your legs at night become unbearable. A light breeze triggers burning. Your clothing feels like sandpaper against your skin. These are not exaggerations from anxious patients. These are documented clinical observations from peer-reviewed research published in the American Journal of Health-System Pharmacy and Cureus, describing a phenomenon that affects real people at real doses.
The connection between semaglutide and allodynia is complicated, and that complexity matters. Because while case reports link higher semaglutide doses to this painful skin sensitivity, other research shows GLP-1 receptor agonists actually reducing pain through anti-inflammatory mechanisms. This guide examines both sides of that paradox, covers what the research actually shows, and provides practical guidance for anyone experiencing unusual skin sensitivity while on semaglutide or considering dose adjustments. SeekPeptides has compiled the most current evidence available to help you understand this emerging topic.
What is allodynia and why does it matter for GLP-1 users
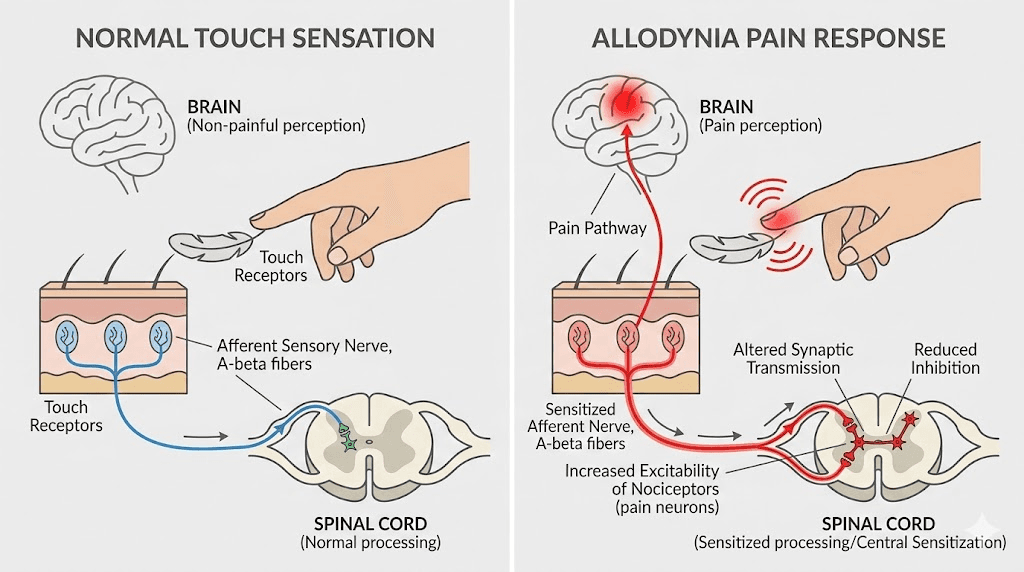
Allodynia is not ordinary pain. It is pain triggered by something that should not cause pain at all. A handshake. A cotton shirt. Cool air from an open window. The International Association for the Study of Pain defines it as pain resulting from a stimulus that does not normally provoke pain, and that definition matters because it distinguishes allodynia from hyperalgesia, where painful stimuli simply hurt more than expected.
The distinction is critical.
With hyperalgesia, stubbing your toe hurts worse than it should. With allodynia, someone gently touching your arm feels like a burn. The nervous system has fundamentally misinterpreted a harmless signal as dangerous, and the brain responds with genuine pain. This is not psychological. This is not imagined. This is a measurable neurological event involving nerve pathway dysfunction that researchers can observe and quantify.
Understanding allodynia requires knowing its three primary forms. Each type involves different nerve fibers, different triggers, and different implications for people using semaglutide or other GLP-1 medications.
Tactile allodynia (static)
Light touch or gentle pressure causes pain. This is the most commonly reported type in semaglutide case studies. Patients describe the sensation as feeling "sunburned" across large areas of skin, even without any sun exposure. Clothing rubbing against the body becomes painful. A light tap on the shoulder triggers sharp discomfort. The A-delta nociceptive fibers responsible for this type of allodynia have essentially lowered their activation threshold to the point where normal mechanical contact registers as a threat.
Mechanical allodynia (dynamic)
Movement across the skin surface triggers pain. Bedsheets being pulled across the legs at night, toweling off after a shower, or even wind blowing across exposed skin can all produce significant pain responses. This type involves myelinated A-beta fibers that normally carry non-painful touch information. When central sensitization occurs, these fibers begin triggering pain circuits they were never designed to activate. People dealing with GLP-1 side effects sometimes report this type without realizing it has a clinical name.
Thermal allodynia
Mild temperature changes, warmth that would normally feel pleasant or cool air that would normally feel refreshing, instead produce pain. A lukewarm shower feels scalding. A slightly cool room feels like ice against the skin. Both A-delta fibers and C-fibers mediate thermal allodynia, and the condition can make routine temperature exposure genuinely distressing.
Most people with allodynia experience more than one type simultaneously, creating a situation where nearly any skin contact becomes uncomfortable. For someone on a semaglutide dosing protocol, this can make even basic daily activities challenging.
How allodynia develops in the nervous system
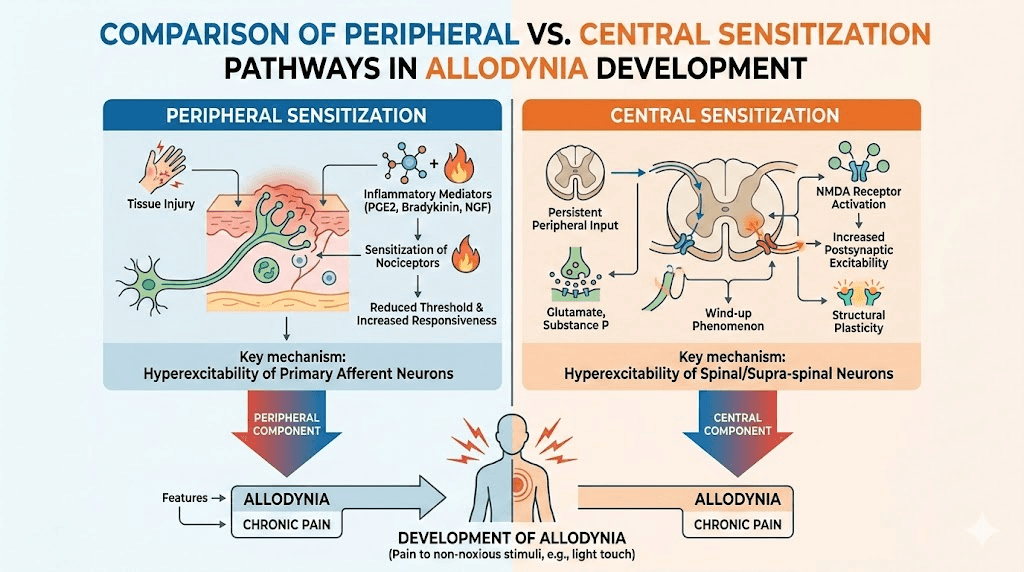
The mechanism behind allodynia involves two interconnected processes that researchers call peripheral sensitization and central sensitization. Both can contribute to allodynia in semaglutide users, and understanding them helps explain why the condition sometimes resolves on its own and sometimes requires intervention.
Peripheral sensitization happens at the nerve endings themselves. When a nerve is damaged or exposed to certain chemical signals, it can become hypersensitive. The threshold for activation drops. Signals that previously fell below the pain detection limit suddenly cross it. The nerve endings release higher quantities of neurotransmitters, creating inflammation around the nerve that further lowers the activation threshold. This creates a feedback loop where sensitivity breeds more sensitivity.
Central sensitization is more complex and more concerning.
In central sensitization, the spinal cord and brain amplify incoming signals regardless of their original intensity. Neurons in the dorsal horn of the spinal cord become hyperexcitable. They begin responding to inputs from A-beta touch fibers as if those inputs came from pain fibers. The technical term is long-term potentiation of sensory nerves, essentially an error in the way the nervous system processes and remembers pain signals. Once central sensitization develops, even removing the original trigger may not immediately resolve the pain, because the amplification occurs in the central nervous system rather than at the periphery.
This is why allodynia associated with semaglutide use can persist for weeks or even months after dose reduction. The central nervous system has essentially learned a new pain pattern, and unlearning it takes time. Research published in PMC confirms that the etiology of semaglutide-associated allodynia is "thought to be an error in the long-term potentiation of sensory nerves," directly implicating central sensitization as the primary mechanism.
The research connecting semaglutide to allodynia
The evidence linking semaglutide to allodynia comes from three distinct sources: clinical trial data, published case reports, and population-level epidemiological studies. Each provides a different piece of the puzzle, and together they paint a picture that is concerning enough to warrant attention but not alarming enough to discourage GLP-1 medication use entirely.
The Stark case series (2025)
The most detailed clinical evidence comes from a case series published in the American Journal of Health-System Pharmacy by Stark et al. This study documented four patients who developed allodynia while taking semaglutide 2.4 mg subcutaneously once weekly for weight management. Several important patterns emerged from these cases.
First, every patient developed symptoms specifically at the 2.4 mg dose. Not at lower doses. This dose-response relationship is significant because it suggests the effect is pharmacologically mediated rather than coincidental.
Second, the temporal relationship was clear. Symptoms appeared within weeks of reaching the highest dose, and in patients who discontinued the medication, symptoms resolved. Two patients stopped semaglutide entirely and both experienced complete resolution of allodynia. Two patients chose to continue the medication despite the side effect. One of those patients saw spontaneous resolution after approximately four months of continued use.
Third, no other explanation for the allodynia was identified in any of the four cases. The patients did not have conditions commonly associated with allodynia such as fibromyalgia, recent shingles, or diabetic neuropathy. The semaglutide was the only new variable.
The PMC review of clinical trial data
A broader analysis published in Cureus and indexed in PMC examined allodynia and dysesthesia across 22 clinical trials and case reports involving semaglutide and tirzepatide. The researchers found that among 334 patients receiving oral semaglutide at a 50 mg dose, three to nine patients experienced altered skin sensations including dysesthesias, hyperesthesia, neuralgia, pain of skin, paresthesia, sensitive skin, and skin burning sensation.
That translates to roughly 1-3% of patients in the oral formulation trials. For the injectable formulation at 2.4 mg, the reported rate was lower but still present: one of 407 patients reported dysesthesia.
These numbers warrant context. The gastrointestinal side effects of semaglutide, nausea, vomiting, diarrhea, affect 20-40% of users. Allodynia at 1-3% is far less common. But it is also far more disruptive to daily life than temporary nausea, and it is not something most patients or even prescribers are aware of as a possibility.
Epidemiological evidence from large cohort studies
The strongest population-level evidence comes from a study published on medRxiv that compared allodynia incidence between GLP-1 receptor agonist users and users of bupropion-naltrexone, an alternative weight management medication. The findings were striking.
New users of semaglutide or liraglutide had an allodynia incidence of 35 per 1,000 person-years. Bupropion-naltrexone users had an incidence of 15 per 1,000 person-years. That means GLP-1 receptor agonist users were more than twice as likely to develop allodynia compared to an active comparator group, not just compared to the general population.
This study is particularly valuable because using an active comparator controls for many confounding factors. Both groups were seeking weight loss treatment, so factors like obesity itself, which can independently affect nerve function, were present in both groups. The higher rate in the GLP-1 group suggests something specific about the medication class rather than the underlying condition being treated.
FDA adverse event data
The FDA Adverse Event Reporting System (FAERS) database provides additional context. Allodynia appears among the top 50 adverse reactions reported for tirzepatide in the FAERS system. For semaglutide, skin-related pain complaints have been documented in clinical trials but allodynia is not currently listed on the official drug label for any semaglutide product, whether Ozempic, Wegovy, or Rybelsus.
The absence from the drug label does not mean the connection is not real. It means the FDA has not yet determined that the evidence meets the threshold for a label change. Given that the first published case report appeared only recently, and that epidemiological studies are still emerging, a label update may follow as more data accumulates.
Documented case reports and what they reveal
Beyond the Stark case series, additional case reports provide granular detail about how semaglutide-associated allodynia presents in individual patients. These cases reveal important patterns about timing, severity, and resolution that can guide both patients and clinicians.
Case study: the sunburn sensation
A 56-year-old woman being treated for chronic weight management had previously tolerated liraglutide at doses ranging from 0.6 to 3 mg daily for six months without any skin-related complaints. When she transitioned to semaglutide and reached the 2.4 mg weekly dose, she developed dysesthesia described as a "sunburned" skin sensation. The pain was diffuse, not localized to the injection site, and affected multiple body areas simultaneously.
The resolution timeline is notable. Her symptoms resolved spontaneously after approximately six weeks of continued semaglutide use at the same dose. She did not require dose reduction or discontinuation. This case suggests that for some patients, the nervous system adapts to whatever change semaglutide produces, and the allodynia is self-limiting.
Case study: the burning body
A 75-year-old man with type 2 diabetes and pre-existing baseline neuropathy and chronic back pain was escalated from oral semaglutide 14 mg daily to tirzepatide. When he reached the tirzepatide 15 mg weekly dose, he developed allodynia described as a "burning sensation all over his body." This case is important for two reasons.
First, it involved a patient who already had neuropathy, raising the question of whether pre-existing nerve damage creates vulnerability to GLP-1-associated allodynia. His baseline neuropathy may have primed his nervous system for central sensitization in a way that would not occur in neurologically healthy patients.
Second, his allodynia initially resolved spontaneously after two weeks, then recurred six months later in a severe episode after antibiotic treatment. This recurrence pattern suggests that once the nervous system has been sensitized, other stressors can potentially reactivate the allodynia even without a dose change. Upon discontinuation of tirzepatide, his symptoms resolved completely. He was subsequently switched to oral semaglutide 14 mg and did not experience recurrence, suggesting the effect may be dose-dependent rather than strictly class-dependent.
What the case reports tell us collectively
Several patterns emerge across all published cases. The condition appears at higher doses. It can resolve spontaneously or upon discontinuation. It may recur if the nervous system has been sensitized. And it does not appear to occur at the same rate across all GLP-1 medications and doses. These patterns matter for anyone adjusting their semaglutide protocol and wondering whether unusual skin sensitivity might be medication-related.
The paradox: GLP-1 agonists as both pain cause and pain treatment
Here is where the semaglutide-allodynia story becomes genuinely interesting. While clinical reports document allodynia as an adverse effect, a parallel body of research shows that GLP-1 receptor agonists, including semaglutide, possess significant anti-pain and neuroprotective properties. This is not a contradiction so much as a reflection of how complex the nervous system truly is.
GLP-1 receptors and pain pathways
GLP-1 receptors are widely expressed throughout both the central and peripheral nervous systems. They appear in the spinal cord, the brainstem, the trigeminal nucleus, and in peripheral sensory neurons. When activated, these receptors can influence neuroinflammation, neuronal excitability, and nociceptive processing, the fundamental components of pain perception.
Research published in Experimental and Molecular Medicine demonstrated that GLP-1-derived peptides, including liraglutide, exendin-4, and exendin 9-39, effectively inhibited capsaicin-induced currents and calcium responses in cultured sensory neurons and TRPV1-expressing cell lines. TRPV1 is the receptor responsible for the burning sensation you feel when you eat hot peppers. By directly inhibiting this receptor, GLP-1 compounds reduced pain signaling at the most fundamental level.
In animal models, these compounds alleviated both acute pain and chronic pain induced by nerve injury. The implications are significant for anyone researching peptides for pain management.
Anti-inflammatory mechanisms
The pain-relieving properties of GLP-1 receptor agonists stem largely from their anti-inflammatory effects in neural tissue. Research shows these medications reduce the activation of the NLRP3 inflammasome in brain microglia, suppress pro-inflammatory cytokines including TNF-alpha, IL-1 beta, and IL-6 in the hippocampus and spinal cord, inhibit microglial activation in pain-processing centers, and reduce oxidative stress that contributes to nerve damage.
A study published in the Journal of Headache and Pain found that liraglutide attenuated trigeminal allodynia in a chronic migraine mouse model by stimulating IL-10 release in the trigeminal nucleus caudalis. IL-10 is an anti-inflammatory cytokine that suppresses pain signaling. The GLP-1 agonist was essentially treating allodynia rather than causing it.
Studies on inflammation and peptides consistently show this dual relationship between immune modulation and pain processing.
Diabetic neuropathy benefits
Perhaps most relevant to the semaglutide discussion, research demonstrates that GLP-1 receptor agonists can ameliorate diabetic neuropathic pain by inhibiting neuroinflammation in the spinal cord. A study published in PubMed showed that semaglutide itself reduced neuropathic pain markers in diabetic animal models through mechanisms involving reduced spinal cord inflammation.
This creates a genuine paradox. The same medication that can cause allodynia in some patients at higher doses appears to treat neuropathic pain in others through anti-inflammatory mechanisms. The resolution of this paradox likely involves dose, individual neurological vulnerability, and the difference between GLP-1 receptor activation in different nervous system compartments.
Resolving the paradox
Several hypotheses attempt to explain why the same medication class can both cause and relieve pain:
Dose-dependent effects. At therapeutic doses used for diabetes or moderate weight management, the anti-inflammatory effects may predominate. At the highest doses used for obesity treatment (2.4 mg semaglutide, 15 mg tirzepatide), the medication may overwhelm certain regulatory mechanisms and produce paradoxical sensitization.
Individual neurological vulnerability. Patients with pre-existing subclinical nerve issues, perhaps from metabolic stress, vitamin deficiencies, or genetic predisposition, may respond differently to GLP-1 receptor activation than neurologically healthy patients.
Treatment-induced neuropathy of diabetes. Rapid improvement in blood sugar from semaglutide can occasionally trigger acute painful neuropathy. This is a known phenomenon where neurons that had adapted to chronic hyperglycemia react adversely to rapid glycemic normalization. This mechanism applies primarily to diabetic patients rather than those using semaglutide solely for weight management.
Compartment-specific effects. GLP-1 receptor activation in the spinal cord and brainstem may be anti-inflammatory and pain-relieving, while effects on peripheral sensory neurons may, under certain conditions, produce sensitization. The net effect would depend on which compartment is most affected in a given individual.
Risk factors for developing allodynia on semaglutide
Not everyone who takes semaglutide develops allodynia. The vast majority do not. But identifying who might be at higher risk allows for more informed decision-making and earlier recognition if symptoms develop.
Dose level
The clearest risk factor is dose. Published cases overwhelmingly involve the highest available doses: 2.4 mg subcutaneous semaglutide weekly or 15 mg tirzepatide weekly. Lower doses appear to carry substantially less risk. This is consistent with a pharmacological dose-response relationship rather than an idiosyncratic reaction.
For people following a semaglutide dosing chart, this means the escalation from 1.7 mg to 2.4 mg is the critical transition point where allodynia risk appears to increase most significantly.
Rapid dose escalation
While not definitively established as an independent risk factor, the cases documented in clinical literature all involved standard dose escalation protocols. Theoretically, slower dose increases might allow the nervous system to adapt more gradually to GLP-1 receptor activation. Some clinicians who are aware of the allodynia risk recommend extended escalation timelines, particularly for patients approaching the 2.4 mg dose.
Research on starting semaglutide consistently emphasizes the importance of gradual dose increases for minimizing all side effects, not just allodynia.
Pre-existing neuropathy
The case of the 75-year-old man with baseline neuropathy who developed severe allodynia on tirzepatide suggests that pre-existing nerve damage may create vulnerability. If peripheral nerves are already compromised, the additional neurological changes produced by GLP-1 receptor agonists at high doses may be more likely to push the system past its sensitization threshold.
Conditions associated with neuropathy include diabetes, vitamin B12 deficiency, autoimmune disorders, and certain medications. People with any of these conditions should be particularly attentive to new skin sensitivity symptoms during semaglutide dose escalation.
Previous allodynia history
People who have experienced allodynia from other causes, such as migraines, fibromyalgia, complex regional pain syndrome, or shingles, may have a lower threshold for developing medication-associated allodynia. Their central pain processing systems have already demonstrated a capacity for sensitization, which could make them more susceptible to additional triggers.
Concurrent medications
The recurrence case in the literature involved allodynia returning after the patient received antibiotics while still on tirzepatide. This suggests that drug interactions or the physiological stress of infection treatment may contribute to allodynia development or recurrence. People taking multiple medications alongside semaglutide should discuss potential interactions with their prescriber, particularly if they notice new sensory symptoms after adding or changing other medications.
Recognizing allodynia symptoms while on semaglutide
Early recognition matters. The sooner allodynia is identified, the more options are available for management, and the less likely central sensitization is to become deeply established. But recognizing allodynia requires distinguishing it from other common semaglutide side effects and from normal variations in skin sensitivity.
Key symptoms to watch for
The hallmark of allodynia is pain from stimuli that should not cause pain. Specifically, watch for burning or stinging sensations when clothing touches your skin, pain when water hits your skin during a shower at normal temperature, discomfort from bedsheets or blankets resting on your body, pain from light touch such as a pat on the back or a handshake, burning sensation from mild temperature changes that previously felt normal, and widespread skin tenderness that is not localized to the injection site.
The "not localized to the injection site" part is important. Injection site reactions are common with semaglutide and involve redness, swelling, or pain at the specific spot where the medication was administered. Allodynia is diffuse. It affects large areas of the body, often the torso, arms, legs, or entire body surface.
What allodynia feels like vs other side effects
Semaglutide produces numerous side effects that could be confused with allodynia. Understanding the differences helps with accurate identification.
Injection site reaction vs allodynia. Injection site reactions are localized to a small area around the injection point and typically resolve within days. Allodynia is widespread and persistent. If your skin hurts only near where you injected, that is almost certainly a local injection site reaction, not allodynia.
Muscle pain vs allodynia. Muscle pain from GLP-1 medications involves deep, aching sensations in muscle tissue. Allodynia involves surface-level skin sensitivity. If pressing into the muscle reproduces the pain, it is likely muscular. If light skin contact triggers the pain, it is more consistent with allodynia.
Body aches vs allodynia. General body aches from semaglutide tend to be dull, widespread, and similar to flu-like symptoms. Allodynia produces sharp, burning, or stinging sensations specifically triggered by touch or temperature. The quality of the pain is different even if the location is similar.
Headache-associated sensitivity vs allodynia. Some people experience headaches on GLP-1 medications accompanied by scalp tenderness. While scalp allodynia is indeed a form of allodynia (commonly seen with migraines), whole-body skin sensitivity is the pattern associated with semaglutide in the published literature.
When symptoms typically appear
Based on available case data, semaglutide-associated allodynia most commonly appears within the first few weeks of reaching the highest dose. For subcutaneous semaglutide, this is the 2.4 mg weekly dose. For oral semaglutide, higher doses may also carry risk.
The timing is important for causality assessment. If you developed skin sensitivity months before starting semaglutide, the medication is unlikely to be the cause. If the sensitivity appeared within weeks of a dose increase, the temporal relationship strongly suggests a medication effect.
What to do if you develop allodynia on semaglutide
If you suspect you are experiencing allodynia while taking semaglutide, the response should be proportional to the severity. Mild cases may warrant monitoring. Moderate cases deserve clinical attention. Severe cases may require immediate action.
Step 1: Document the symptoms
Before contacting your healthcare provider, document what you are experiencing. Record when the symptoms started, noting specifically how many days or weeks after your last dose increase. Describe the type of sensations, whether burning, stinging, aching, or electric shock-like. Map which body areas are affected. Note what triggers the pain, whether it is clothing, temperature, touch, or other stimuli. Rate the severity on a 1-10 scale. And document whether the symptoms are constant or intermittent.
This documentation serves two purposes. It helps your provider assess whether the pattern matches allodynia versus other conditions, and it creates a baseline for tracking whether symptoms improve, worsen, or remain stable over time.
Step 2: Assess severity and impact
Not all allodynia requires the same response. Consider how much the symptoms affect your daily functioning.
Mild allodynia: You notice increased skin sensitivity, but it does not significantly interfere with daily activities. You can still wear normal clothing, sleep without major disruption, and function normally. Monitoring may be appropriate.
Moderate allodynia: The sensitivity interferes with some daily activities. Certain clothing becomes uncomfortable. Sleep quality decreases. You avoid being touched. Clinical evaluation is warranted.
Severe allodynia: The pain significantly impairs daily functioning. Normal clothing is unbearable. Sleep is severely disrupted. Basic activities like bathing or dressing become painful. Immediate clinical attention is needed, and dose adjustment or discontinuation should be discussed urgently.
Step 3: Contact your healthcare provider
Bring your symptom documentation to your provider. Specifically mention that published research has linked semaglutide to allodynia, as many providers may not be aware of this association since it is not on the drug label. Reference the Stark et al. case series published in the American Journal of Health-System Pharmacy and the epidemiological data from medRxiv showing elevated allodynia rates in GLP-1 users.
This is not about self-diagnosing. It is about ensuring your provider has the most current information when evaluating your symptoms.
Step 4: Discuss management options
Based on the available evidence, several management strategies have been documented. The right approach depends on the severity of your allodynia, the benefit you are receiving from semaglutide, and your individual risk-benefit calculation.
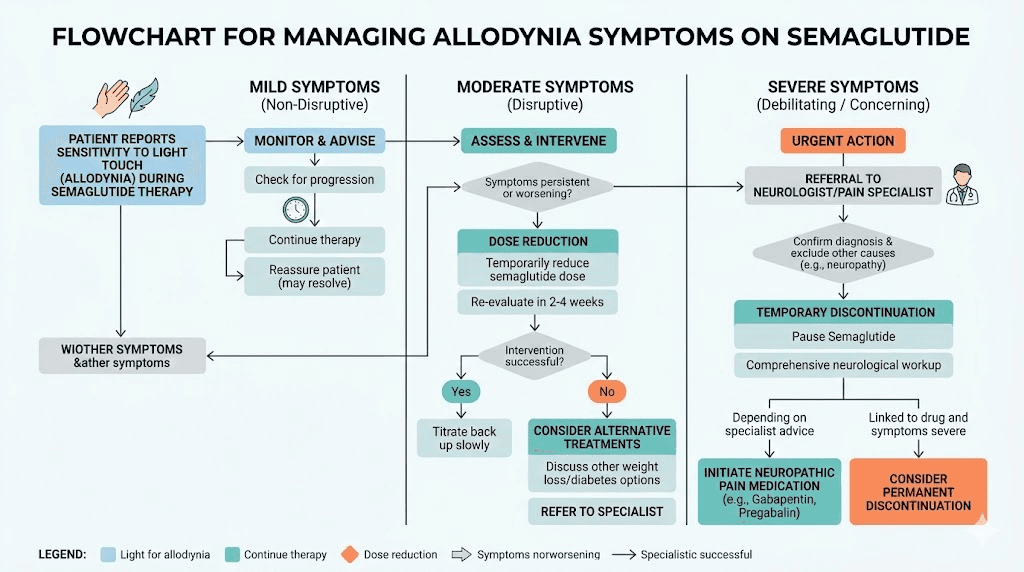
Treatment and management strategies for semaglutide-associated allodynia
The treatment approach for allodynia that develops during semaglutide use depends on what the published evidence shows has worked in documented cases. Because this is a recently recognized adverse effect, treatment strategies are drawn from case report outcomes, general allodynia management guidelines, and clinical experience with neuropathic pain.
Dose reduction
Reducing the semaglutide dose is often the first strategy considered, and for good reason. The dose-response relationship in published cases strongly suggests that lower doses carry lower allodynia risk. Stepping down from 2.4 mg to 1.7 mg, or even further to 1 mg, may resolve symptoms while maintaining some of the metabolic benefits of the medication.
The semaglutide dosage chart can help visualize the available dose steps. The key question is whether the lower dose still provides adequate therapeutic benefit. For many patients, the 1.7 mg dose delivers meaningful appetite suppression and weight management effects, making dose reduction a viable compromise.
Temporary discontinuation
In the Stark case series, both patients who discontinued semaglutide experienced complete resolution of allodynia symptoms. This is the most definitive treatment approach and the one with the strongest evidence base. If the allodynia is severe enough to significantly impair quality of life, temporary discontinuation allows the nervous system to reset.
The question then becomes whether and when to restart. Some patients may tolerate re-escalation if done more slowly. Others may find that the allodynia returns at the same dose threshold. Research on restarting semaglutide after a break provides guidance on re-initiation protocols, though specific allodynia-focused re-escalation data is limited.
Continued use with monitoring
One of the four patients in the Stark case series continued semaglutide and experienced spontaneous resolution of allodynia after approximately four months. The 56-year-old woman in the PMC case report saw resolution after six weeks of continued use. These cases suggest that for some patients, allodynia is self-limiting even without dose changes.
This approach is reasonable for mild-to-moderate allodynia where the patient is receiving significant benefit from semaglutide and is willing to tolerate temporary discomfort. Close monitoring is essential, with clear parameters established for escalating to dose reduction or discontinuation if symptoms worsen.
Switching medications
The case of the patient who developed allodynia on tirzepatide 15 mg but tolerated oral semaglutide 14 mg without recurrence raises an important possibility: switching between GLP-1 medications may help. The allodynia may be specific to a particular formulation, dose, or receptor activation profile rather than a universal class effect.
Options for switching include moving from injectable to oral or sublingual semaglutide, transitioning between tirzepatide and semaglutide, or considering a dose conversion to a different GLP-1 agonist at a lower relative dose.
Pharmacological treatment of allodynia itself
When allodynia persists despite dose adjustment, or when a patient needs to remain on the current semaglutide dose for clinical reasons, treating the allodynia directly becomes necessary. First-line medications for neuropathic pain and allodynia include the following options.
Pregabalin (Lyrica). An anticonvulsant that reduces nerve excitability by blocking calcium channels. It is FDA-approved for neuropathic pain and is considered a first-line treatment for allodynia. Typical starting doses are 75 mg twice daily, titrated as needed.
Gabapentin (Neurontin). Similar mechanism to pregabalin with a longer track record. Starting doses are usually 300 mg daily, gradually increased. Gabapentin has the advantage of more flexible dosing and lower cost. Research on neuropathy treatment frequently cites gabapentin as a foundational therapy.
Duloxetine (Cymbalta). A serotonin-noradrenaline reuptake inhibitor with established efficacy in neuropathic pain. It works by increasing the concentration of neurotransmitters that naturally suppress pain signaling in the spinal cord.
Topical options. Capsaicin 8% patches desensitize TRPV1 receptors on nerve endings, reducing pain signaling over time. Lidocaine 5% patches provide local numbing for specific areas of severe sensitivity. These topical approaches can be used alongside systemic medications or as standalone treatments for localized allodynia.
Non-pharmacological approaches
Physical therapy techniques, particularly desensitization therapy, can help retrain the nervous system to correctly interpret non-painful stimuli. This involves gradually exposing affected areas to increasing levels of gentle stimulation, essentially teaching the nervous system that these sensations are safe.
Cognitive behavioral therapy has evidence supporting its use in chronic pain conditions including allodynia. The psychological impact of persistent pain should not be underestimated, and addressing the cognitive and emotional components of the pain experience can meaningfully improve outcomes.
Lifestyle modifications that may help include wearing loose, soft-textured clothing made from natural fibers, using silk or satin bedsheets that produce less friction, maintaining a consistent room temperature to avoid thermal triggers, practicing stress reduction techniques since psychological stress can amplify central sensitization, and optimizing sleep hygiene since poor sleep worsens pain perception.
Allodynia in the context of common semaglutide side effects
Understanding where allodynia fits within the broader landscape of semaglutide side effects helps put the risk in perspective. The medication produces a range of effects on the body, and knowing which are common versus rare helps calibrate concern appropriately.
Common side effects (affecting 10% or more of users)
Nausea affects 20-40% of semaglutide users, particularly during dose escalation. Constipation, diarrhea, and bloating round out the gastrointestinal effects that most users experience to some degree. Headache (8-14%) and dizziness (4-8%) are also relatively common. Fatigue affects a significant percentage of users, particularly in the early weeks.
Uncommon side effects (affecting 1-10% of users)
Hair thinning, excessive burping, changes in sex drive, insomnia, and menstrual changes fall into this category. Skin-related sensory changes, including dysesthesia and altered skin sensation, also fall here based on clinical trial data showing 1-3% incidence.
Rare side effects (affecting less than 1% of users)
Full-blown allodynia, where non-painful stimuli produce genuine pain across large body areas, appears to be rare based on current evidence. The epidemiological rate of 35 per 1,000 person-years (3.5%) includes a broad definition that may encompass milder sensory changes as well as true allodynia.
Putting these numbers together, allodynia is less common than nausea by a large margin but more common than many other side effects that receive more attention. Its impact on quality of life, however, can be disproportionately severe compared to its frequency.
Semaglutide dose escalation and allodynia prevention
While no proven prevention strategy exists specifically for semaglutide-associated allodynia, the dose-dependent nature of the condition suggests that thoughtful dose management may reduce risk.
Standard vs extended escalation
The standard semaglutide escalation protocol increases the dose every four weeks: 0.25 mg for weeks 1-4, 0.5 mg for weeks 5-8, 1 mg for weeks 9-12, 1.7 mg for weeks 13-16, and 2.4 mg from week 17 onward. Each step roughly doubles the dose, placing significant new demands on the body systems affected by GLP-1 receptor activation.
An extended escalation approach, spending six to eight weeks at each dose rather than four, may allow the nervous system to fully adapt before the next increase. This is not specific guidance for allodynia prevention, as no study has tested this hypothesis directly. But it aligns with the general principle that slower escalation produces fewer side effects across all categories.
Using a semaglutide dosage calculator can help you and your provider plan an appropriate escalation timeline based on your individual response.
Evaluating the need for maximum dose
Not every patient needs the 2.4 mg dose. If you are achieving satisfactory weight loss results at 1 mg or 1.7 mg, escalating further introduces additional side effect risk without guaranteed additional benefit. The optimal semaglutide dose is the lowest dose that produces adequate therapeutic effect, not automatically the highest available dose.
This principle applies broadly across all semaglutide side effects but has particular relevance for allodynia given its strong dose-response relationship.
Monitoring during escalation
Active monitoring during dose escalation can catch allodynia symptoms early, before central sensitization has time to become deeply established. Pay attention to any new skin sensitivity in the first two to four weeks after each dose increase. Ask yourself: does clothing feel different? Does water temperature seem off? Does touch feel different than it did before the dose change?
If the answer to any of these questions is yes, inform your provider before the next scheduled dose increase. Early intervention, even if it is just a temporary pause at the current dose, may prevent progression from mild sensory changes to full allodynia.
Allodynia and tirzepatide: is it a class effect
The question of whether allodynia is specific to semaglutide or a broader GLP-1 receptor agonist class effect has significant implications for treatment decisions. If it is semaglutide-specific, switching to tirzepatide might resolve the issue. If it is a class effect, switching medications within the same class would not help.
Evidence for a class effect
Several pieces of evidence suggest allodynia may be a GLP-1 receptor agonist class effect rather than specific to semaglutide. The PMC case report documents allodynia with tirzepatide, which is a dual GIP/GLP-1 receptor agonist rather than a pure GLP-1 agonist. The FAERS database lists allodynia among the top 50 adverse reactions for tirzepatide specifically. And the epidemiological study used a combined semaglutide-and-liraglutide cohort, with elevated allodynia rates for the class as a whole.
These data points suggest that GLP-1 receptor activation itself, rather than any semaglutide-specific property, may be responsible for the allodynia risk. The side effect profiles of semaglutide and tirzepatide share many commonalities, and allodynia may be another shared characteristic.
Evidence against a universal class effect
However, one case provides a compelling counterexample. The patient who developed allodynia on tirzepatide 15 mg was successfully switched to oral semaglutide 14 mg without recurrence. If allodynia were a pure GLP-1 class effect, one would expect it to appear with any GLP-1 agonist at sufficient doses. The fact that this patient tolerated oral semaglutide suggests that formulation, route of administration, or specific receptor binding profile may matter.
Tirzepatide activates both GIP and GLP-1 receptors, which may produce different neurological effects than pure GLP-1 agonism. Newer agents like retatrutide, which activates three receptors (GLP-1, GIP, and glucagon), and cagrilintide-semaglutide combinations may have different allodynia risk profiles that are not yet characterized.
Practical implications
For patients who develop allodynia on one GLP-1 medication, switching to a different medication within the class (or to a different formulation of the same medication) is a reasonable strategy to try. The available evidence suggests this works for some patients. But it is not guaranteed, and close monitoring during the switch is essential.
Understanding the dosage conversion between medications is important for any transition. Starting at the lowest dose of the new medication and escalating slowly provides the best opportunity to identify any recurring allodynia early.
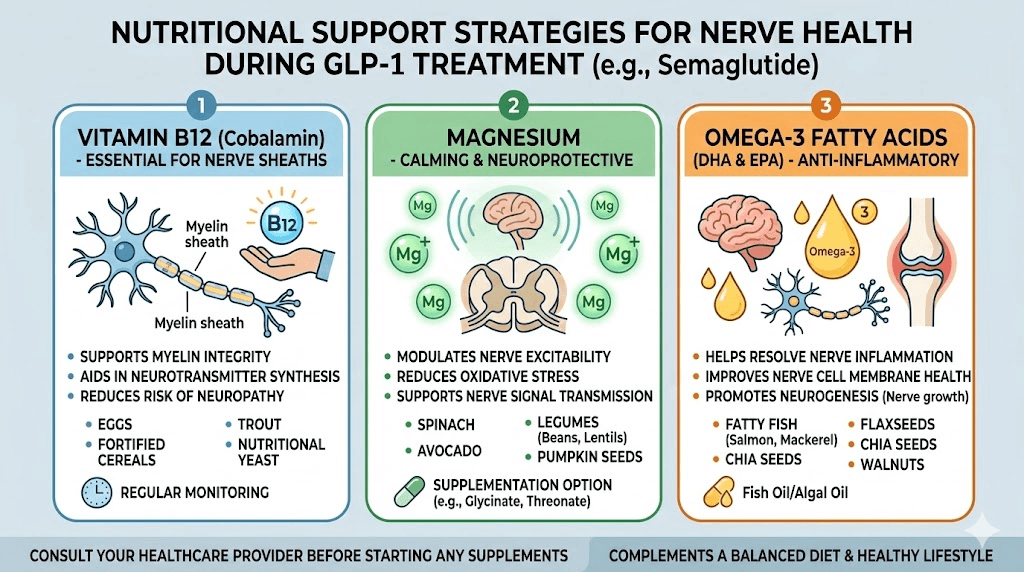
Nutritional and lifestyle factors that may influence allodynia risk
While no direct evidence links specific nutrients to semaglutide-associated allodynia, general neuropathic pain research identifies several nutritional and lifestyle factors that influence nerve health and pain sensitivity. Optimizing these factors during semaglutide use may theoretically reduce allodynia risk.
B vitamin status
Vitamin B12 deficiency is a well-established cause of peripheral neuropathy, and several GLP-1 formulations now include B12 specifically because of concerns about nutritional status during weight loss. Semaglutide with B12 and tirzepatide with B12 formulations exist partly to address this concern.
Reduced food intake on semaglutide can lead to decreased B12 absorption from dietary sources. Methylcobalamin, the active form of B12, supports nerve function and may help maintain the resilience of the nervous system against sensitization. If you are on semaglutide, monitoring B12 levels and supplementing if needed is a reasonable precaution for nerve health generally, not just for allodynia prevention specifically.
Magnesium
Magnesium plays a critical role in nerve function, and deficiency is associated with increased pain sensitivity. NMDA receptor activation, a key mechanism in central sensitization, is modulated by magnesium. Adequate magnesium levels help keep NMDA receptors in check, potentially raising the threshold for central sensitization.
Reduced food intake on GLP-1 medications can compromise magnesium intake. Supplementation with magnesium glycinate or magnesium threonate may support nerve health during semaglutide use.
Dietary considerations
The reduced appetite produced by semaglutide makes dietary quality even more important than usual. Every calorie consumed needs to deliver maximum nutritional value. Following a structured diet plan that prioritizes nutrient density, particularly foods rich in B vitamins, omega-3 fatty acids, and antioxidants, supports the nervous system during a period of metabolic change.
The best foods to eat on semaglutide include fatty fish (omega-3s for nerve membrane health), leafy greens (B vitamins and magnesium), nuts and seeds (vitamin E and minerals), and colorful vegetables (antioxidants that reduce neuroinflammation). Complete food lists can help guide meal planning during GLP-1 treatment.
Sleep and stress management
Poor sleep amplifies pain perception through multiple mechanisms, including increased cortisol, reduced endogenous opioid activity, and impaired glymphatic clearance of inflammatory mediators. Sleep disruption from semaglutide is reported by some users, and if present, addressing it may help reduce vulnerability to allodynia.
Chronic stress maintains the hypothalamic-pituitary-adrenal axis in an activated state, which promotes neuroinflammation and lowers pain thresholds. Stress management is not a substitute for medical treatment of allodynia, but it is a modifiable factor that influences the nervous system environment in which allodynia develops.
Long-term outlook for semaglutide-associated allodynia
The prognosis for allodynia that develops during semaglutide use appears to be generally favorable based on available evidence, though the data is limited and more research is needed.
Resolution rates
In published cases, every patient who discontinued the medication experienced complete resolution of allodynia symptoms. Among patients who continued the medication, some experienced spontaneous resolution after weeks to months. No published case reports document permanent allodynia from semaglutide or tirzepatide use.
This is encouraging, but caution is warranted. The published case literature is small, only documenting a handful of patients in detail. It is possible that more severe or persistent cases exist but have not yet been published.
Timeline for resolution
Based on available data, the timeline for resolution varies by management strategy. With medication discontinuation, symptoms typically resolve within days to weeks. With continued medication use, spontaneous resolution has been documented at four to six weeks and up to four months. With dose reduction, the timeline is not well documented but would be expected to fall somewhere between discontinuation and continued use.
Risk of recurrence
The case of the patient whose allodynia recurred six months after initial resolution raises the possibility that semaglutide-associated allodynia can recur. Once the nervous system has undergone central sensitization, it may be more vulnerable to re-sensitization from the same or different triggers. This is consistent with central sensitization research more broadly, where initial sensitization episodes lower the threshold for future episodes.
For patients who have experienced allodynia on semaglutide and then resume the medication or escalate to higher doses, closer monitoring for recurrence is advisable. The nervous system has demonstrated a capacity for this particular malfunction, and vigilance helps ensure early detection if it returns.
Ongoing research
The semaglutide-allodynia connection is an active area of investigation. The first case reports were only recently published, and epidemiological studies are still emerging. As GLP-1 medications continue to be prescribed to millions of people worldwide, more data will accumulate on the incidence, risk factors, mechanisms, and optimal management of this side effect.
For the most current evidence, SeekPeptides monitors emerging research on GLP-1 medications and their effects, providing members with updated information as new studies are published.
When to seek immediate medical attention
While semaglutide-associated allodynia is generally not a medical emergency, certain presentations warrant urgent evaluation.
Seek immediate medical attention if:
You experience sudden, severe pain with numbness or weakness in any extremity. This could indicate acute nerve compression or vascular event rather than allodynia. You develop allodynia accompanied by vision changes, balance problems, or difficulty speaking. These symptoms could suggest central nervous system involvement beyond peripheral sensitization. Your allodynia is accompanied by a rash, blisters, or visible skin changes. This pattern is more consistent with shingles, allergic reaction, or autoimmune process than medication-associated allodynia. The pain is severe enough to prevent you from performing basic activities of daily living. Severe allodynia requires aggressive management, and delaying treatment allows central sensitization to deepen.
Schedule a non-urgent but prompt appointment if:
You notice new skin sensitivity that correlates temporally with a semaglutide dose increase. The sensitivity is present most of the time rather than intermittently. The sensation is spreading to new body areas over time. Or you are unable to identify any other cause for the symptoms.
The key principle is that any new neurological symptom during medication use deserves clinical evaluation. Even if the most likely explanation is a known medication effect, ruling out other causes is important.
Understanding related sensory phenomena on GLP-1 medications
Allodynia exists on a spectrum of altered sensory phenomena that can occur with GLP-1 receptor agonists. Understanding the full spectrum helps patients and clinicians identify and describe their symptoms more precisely.
Dysesthesia
Dysesthesia refers to unpleasant, abnormal sensations that can occur spontaneously or be triggered by stimulation. Unlike allodynia, which specifically involves pain from non-painful stimuli, dysesthesia encompasses a broader range of abnormal sensations including tingling, prickling, crawling, and numbness. The Cureus/PMC study documented both allodynia and dysesthesia in semaglutide and tirzepatide users, suggesting they may represent different points on the same mechanistic spectrum.
Paresthesia
Paresthesia, the "pins and needles" sensation familiar to anyone who has sat on their leg too long, is among the altered skin sensations documented in semaglutide clinical trials. Three to nine out of 334 patients in one oral semaglutide trial experienced paresthesia. While less painful than allodynia, paresthesia can be a precursor to or coexist with allodynia.
Hyperesthesia
Hyperesthesia involves increased sensitivity to sensory stimuli without necessarily reaching the level of pain. Touch feels more intense than usual. Temperature changes feel more extreme. This heightened sensitivity may represent an early or mild form of the same process that produces full allodynia at higher intensities. Monitoring hyperesthesia during semaglutide dose escalation may help catch developing allodynia before it becomes severe.
Neuralgia
Neuralgia, or nerve pain, involves sharp, shooting, or burning pain along the course of a nerve. This was also documented in the oral semaglutide clinical trial data. Neuralgia differs from allodynia in that the pain occurs spontaneously rather than being triggered by normally non-painful stimuli, though the two conditions can coexist.
Comparing allodynia risk across GLP-1 medications
As the GLP-1 medication landscape continues to expand, understanding the relative allodynia risk across different agents becomes increasingly relevant. While comprehensive comparative data is not yet available, existing evidence provides some initial guidance.
Semaglutide (Ozempic/Wegovy). The most documented agent for allodynia, with published case series and epidemiological data. Risk appears highest at the 2.4 mg dose. Available in both injectable and oral formulations. The compounded semaglutide market has grown significantly, and whether compounded formulations carry the same allodynia risk as brand-name products is unknown.
Tirzepatide (Mounjaro/Zepbound). At least one published case of allodynia. Listed in FAERS top 50 adverse reactions. Dual GIP/GLP-1 mechanism may produce different neurological effects than pure GLP-1 agonism. Risk may be dose-dependent similar to semaglutide, with 15 mg being the documented trigger in the published case.
Liraglutide (Saxenda/Victoza). Included in the epidemiological cohort showing elevated allodynia rates. Shorter half-life than semaglutide may theoretically allow faster recovery if allodynia develops. Less commonly prescribed for weight management compared to semaglutide and tirzepatide.
Retatrutide. A triple-agonist (GLP-1/GIP/glucagon) currently in clinical trials. No allodynia data is available. The additional glucagon receptor activation introduces another variable that could influence neurological effects in either direction. Retatrutide dosage protocols and side effect profiles are still being characterized.
Oral formulations. The case where a patient tolerated oral semaglutide after developing allodynia on injectable tirzepatide raises the possibility that oral GLP-1 formulations may carry lower allodynia risk. This could relate to different pharmacokinetic profiles, lower peak drug levels, or differences in how the oral versus injectable forms interact with the nervous system. Oral versus injectable GLP-1 comparisons are an active area of research.
Practical guidance for current semaglutide users
For the millions of people currently taking semaglutide, allodynia awareness does not need to create anxiety. It needs to create informed vigilance.
If you are starting semaglutide
Follow the prescribed escalation schedule. Do not rush to higher doses. Pay attention to any changes in skin sensitivity during each dose increase. Document any new sensations, even minor ones, as a baseline reference. Ensure adequate nutritional support throughout treatment. And discuss allodynia awareness with your prescriber at your initial appointment.
If you are currently on a stable dose
Continue your medication as prescribed. The fact that you have been on a stable dose without allodynia symptoms is reassuring. Be aware that dose changes, including increases, are the most common trigger for this side effect. If you experience any new skin sensitivity, document it and report it to your provider.
If you are experiencing mild skin sensitivity
Do not panic. Mild, transient skin sensitivity is different from clinical allodynia. Document what you are experiencing. Consider whether it correlates with a recent dose change. Discuss it with your provider at your next scheduled appointment unless symptoms are worsening rapidly. Try supportive measures like loose clothing, consistent room temperature, and adequate B vitamin intake.
If you are experiencing significant skin pain
Contact your healthcare provider promptly. Bring your symptom documentation. Discuss the published research on semaglutide and allodynia. Work with your provider to determine the best management strategy based on the severity of your symptoms and the benefit you are receiving from the medication.
For those looking for comprehensive guidance on navigating GLP-1 side effects, SeekPeptides provides detailed protocols, side effect management guides, and evidence-based resources that help members make informed decisions about their treatment.
The broader context: semaglutide safety profile
Allodynia should be understood within the broader safety context of semaglutide. The medication has been prescribed to tens of millions of people worldwide and has a well-characterized safety profile overall. The safety considerations for any peptide or medication involve weighing benefits against risks for each individual patient.
The most common side effects, gastrointestinal symptoms, affect a large percentage of users but are generally manageable and often improve with time. Dietary modifications, proper timing of doses, and gradual escalation all help minimize gastrointestinal distress.
Less common side effects like hair thinning, fatigue, and now allodynia deserve awareness without causing unnecessary alarm. The risk-benefit calculation for semaglutide remains favorable for most patients with obesity or type 2 diabetes, conditions that carry their own serious long-term health risks.
The key is informed consent. Patients should know about allodynia as a possibility, understand what to watch for, and have a plan for what to do if symptoms develop. This is not fundamentally different from being aware of any medication side effect. It is simply newer information that has not yet made it into standard patient education materials.
What researchers do not yet know
Honesty about the limitations of current knowledge is important. Several critical questions remain unanswered.
No one yet knows the precise pharmacological mechanism by which semaglutide produces allodynia. Is it direct GLP-1 receptor activation on sensory neurons? Is it an indirect effect mediated through metabolic changes? Is it related to rapid weight loss and its effects on nerve function? The mechanism remains unknown, and until it is identified, targeted prevention strategies cannot be developed.
The true incidence rate is uncertain. The epidemiological estimate of 35 per 1,000 person-years may overcount mild sensory changes as allodynia or undercount cases that are never reported. A prospective study specifically designed to measure allodynia incidence would provide more reliable numbers.
Whether certain populations are at higher risk remains speculative. The case reports involved patients aged 56 and 75, but whether age is a risk factor or simply reflects the demographics of semaglutide users is unclear.
The optimal management algorithm has not been established through controlled trials. Current management is based on case report outcomes and extrapolation from general neuropathic pain treatment. A randomized trial comparing dose reduction, temporary discontinuation, and symptomatic treatment would be valuable but has not been conducted.
Whether allodynia risk differs between brand-name and compounded semaglutide is unknown. The case reports all involved brand-name products, but the growing compounded market means millions of people are using formulations that have not been specifically studied for this side effect.
Frequently asked questions
Can semaglutide cause nerve damage?
Current evidence does not suggest that semaglutide causes permanent nerve damage. The allodynia associated with semaglutide appears to resolve with dose reduction or discontinuation, indicating functional change rather than structural damage. However, the research is still early, and long-term studies specifically examining nerve health in semaglutide users have not been completed. Paradoxically, some research suggests GLP-1 agonists may actually protect nerves through anti-inflammatory mechanisms.
Is allodynia from semaglutide permanent?
Based on published case reports, no. Every documented case of semaglutide-associated allodynia has resolved either with medication discontinuation, dose reduction, or spontaneously over weeks to months while continuing the medication. No permanent allodynia from semaglutide has been reported in the medical literature.
At what dose does semaglutide allodynia typically occur?
Published cases consistently involve the 2.4 mg subcutaneous weekly dose, which is the highest approved dose for weight management. Lower doses (0.25 mg through 1.7 mg) have not been prominently associated with allodynia in the literature, supporting a dose-dependent relationship. For tirzepatide, the documented case occurred at the 15 mg weekly dose.
Should I stop semaglutide if I develop skin sensitivity?
Not necessarily. The decision to continue, reduce, or stop semaglutide should be made with your healthcare provider based on symptom severity, the benefit you are receiving from the medication, and alternative management options. Mild skin sensitivity may warrant monitoring rather than immediate discontinuation. Severe allodynia that significantly impairs daily function may warrant dose reduction or temporary discontinuation.
Is allodynia the same as injection site pain?
No. Injection site reactions are localized to the specific area where semaglutide was injected and typically involve redness, swelling, and tenderness at that site. Allodynia is a widespread condition affecting large areas of the body, where normally non-painful stimuli like light touch or clothing contact produce pain. The two are distinct phenomena with different mechanisms and different management approaches.
Does allodynia from semaglutide affect everyone?
No. Allodynia affects a small minority of semaglutide users. The vast majority of people taking semaglutide do not develop allodynia. Even at the highest dose of 2.4 mg, where the risk appears greatest, most patients tolerate the medication without developing this side effect.
Can I prevent allodynia while taking semaglutide?
No proven prevention strategy exists specifically for semaglutide-associated allodynia. However, following a gradual dose escalation schedule, maintaining adequate B vitamin and mineral intake, optimizing sleep quality, managing stress, and monitoring for early sensory changes during dose increases may help reduce risk or enable early detection.
Will my doctor know about semaglutide and allodynia?
Many healthcare providers may not yet be aware of this association because allodynia is not listed on the semaglutide drug label and the research is recent. You may need to share the published literature with your provider. Key references include the Stark et al. case series in the American Journal of Health-System Pharmacy and the allodynia incidence study published on medRxiv.
Can other GLP-1 medications also cause allodynia?
Yes. Tirzepatide has at least one published case report of allodynia, and liraglutide was included in the epidemiological study showing elevated allodynia rates in GLP-1 users as a class. The evidence suggests this may be a class effect related to GLP-1 receptor activation rather than specific to semaglutide alone. However, individual responses vary, and some patients tolerate one GLP-1 medication without allodynia while experiencing it on another.
What is the difference between allodynia and hyperalgesia?
Allodynia is pain caused by stimuli that do not normally cause pain, such as light touch or normal clothing contact. Hyperalgesia is an increased pain response to stimuli that do normally cause pain, such as a pinch hurting far more than expected. Both involve altered pain processing, but allodynia involves a fundamentally inappropriate pain response to non-painful input, while hyperalgesia involves an exaggerated response to actually painful input.
External resources
PubMed: Allodynia (skin tenderness) associated with semaglutide case series
PMC: Allodynia and dysesthesia associated with semaglutide and tirzepatide
For researchers seeking comprehensive, evidence-based guidance on GLP-1 medications, side effect management, and peptide safety, SeekPeptides provides the most thorough resource available, with regularly updated protocols, detailed guides, and a community of experienced researchers who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your nerves stay calm, your skin stay comfortable, and your protocols stay well-informed.