Mar 31, 2026
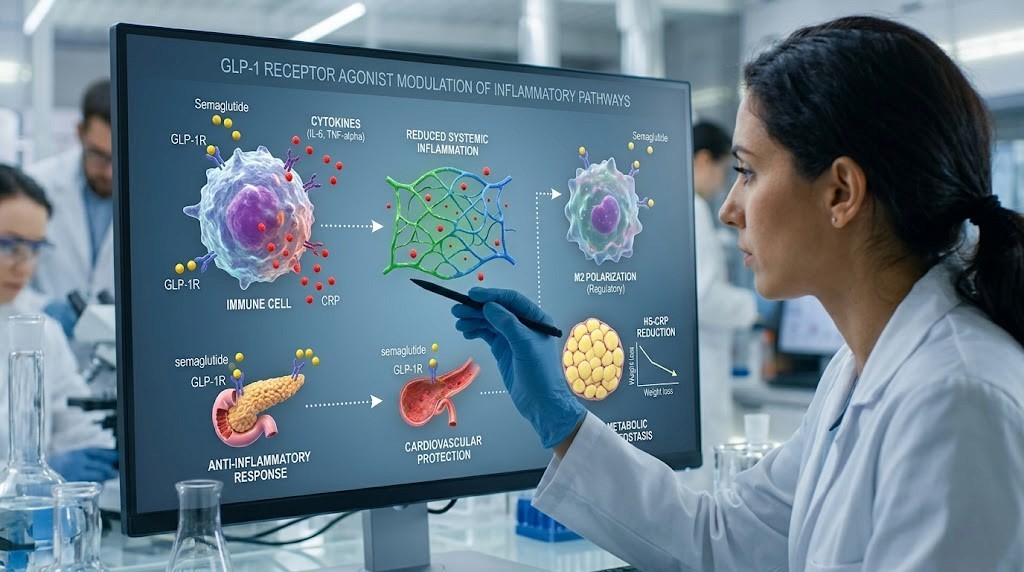
You have been on semaglutide for three weeks. Maybe four. And then you catch a cold. A bad one. The kind that knocks you flat for days when normally you would shake it off in 48 hours. Naturally, your first thought is the medication. Is semaglutide weakening my immune system? Am I more vulnerable now?
It is a fair question. And you are not the only one asking it.
Thousands of people starting semaglutide for weight loss or blood sugar management report feeling "run down" or wonder whether the medication is compromising their ability to fight infections. Forums are full of anecdotal reports. Reddit threads spiral into speculation. And the anxiety compounds when you realize just how little most prescribers explain about the immunological dimensions of GLP-1 receptor agonists.
Here is what the research actually shows. Semaglutide does not suppress your immune system in the way that immunosuppressive drugs like methotrexate or prednisone do. Clinical trial data from the SUSTAIN and STEP programs, involving tens of thousands of participants, found infection rates comparable to placebo groups. The FDA prescribing information for Ozempic, Wegovy, and Rybelsus does not list immune suppression among known risks or warnings. But the story is more nuanced than a simple yes or no, because semaglutide does interact with immune cells in ways researchers are only beginning to understand. GLP-1 receptors exist on macrophages, T cells, neutrophils, and eosinophils. The medication modulates inflammatory pathways, shifts macrophage polarization, and may even influence vaccine responses. This guide breaks down every mechanism, every study, and every practical consideration so you can make informed decisions about your health while using semaglutide.
How the immune system actually works (and what "lowering" it means)
Before we can answer whether semaglutide affects your immune system, we need to understand what the immune system actually does. It is not one thing. It is a layered, interconnected defense network with two major branches, and each branch handles threats differently.
Innate immunity: your first responders
The innate immune system is the frontline defense. It responds immediately to threats without needing to recognize specific pathogens. Think of it as the security guards at the door. They do not care who you are. If you look suspicious, you are getting stopped.
Key players include macrophages, which engulf and destroy invaders while signaling other immune cells to join the fight. Neutrophils arrive first at infection sites, releasing toxic substances to kill bacteria. Natural killer (NK) cells patrol for virus-infected or cancerous cells. And eosinophils handle parasitic infections and allergic responses. These cells communicate through chemical messengers called cytokines, including tumor necrosis factor alpha (TNF-alpha), interleukin-6 (IL-6), and interleukin-1 beta (IL-1beta). When these cytokines are elevated chronically, you get systemic inflammation, which drives conditions like Hashimoto's disease, lupus, and rheumatoid arthritis.
Adaptive immunity: the targeted response
The adaptive immune system is slower but more precise. It learns. It remembers. And it adapts to specific threats over time.
T cells are the commanders. Helper T cells coordinate immune responses. Cytotoxic T cells destroy infected cells directly. Regulatory T cells (Tregs) prevent your immune system from attacking your own tissues. B cells produce antibodies, proteins that lock onto specific pathogens and mark them for destruction. This is the branch that makes vaccines work. Your body encounters a weakened or inactive pathogen, learns to recognize it, and produces memory cells that respond rapidly if it ever encounters the real thing.
When people say a drug "lowers your immune system," they usually mean one of three things. Either it reduces the number of immune cells (like chemotherapy does). Or it suppresses the activity of immune cells (like prednisone does). Or it interferes with the communication between immune cells (like some biologics do). Semaglutide does none of these things in the traditional sense. But it does modulate immune pathways in ways that matter, particularly for people managing fatigue, liver function changes, or autoimmune conditions.
GLP-1 receptors on immune cells: the connection nobody talks about
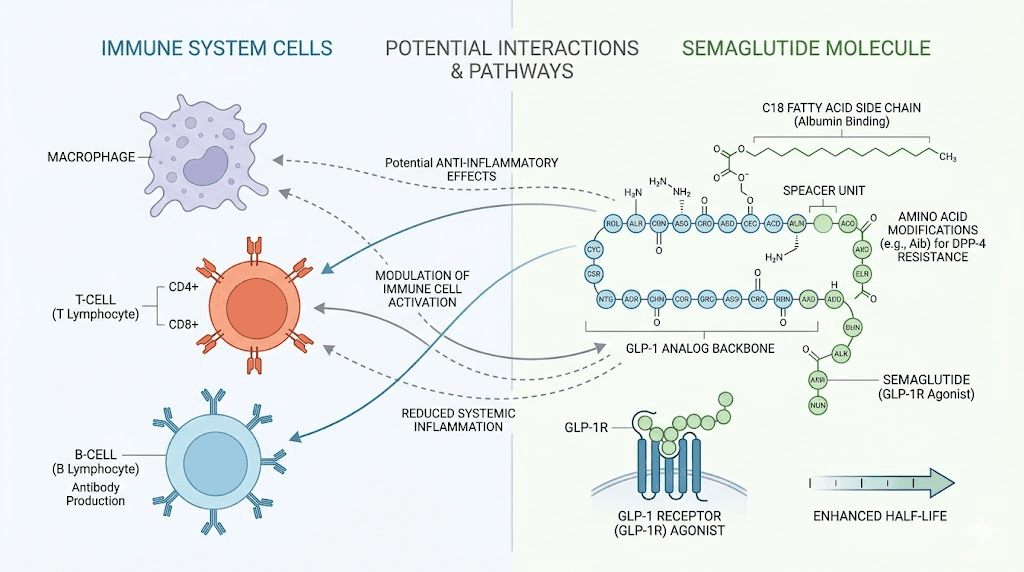
Here is where it gets interesting. GLP-1 receptors are not just in your pancreas and gut. They are expressed on multiple types of immune cells.
Research published in Frontiers in Immunology confirmed that human macrophages, neutrophils, eosinophils, and several types of T cells all express GLP-1 receptors. This means that when you take semaglutide, a long-acting GLP-1 receptor agonist, the medication does not just affect your blood sugar and appetite. It directly interacts with cells responsible for defending your body against infections, allergens, and abnormal cell growth.
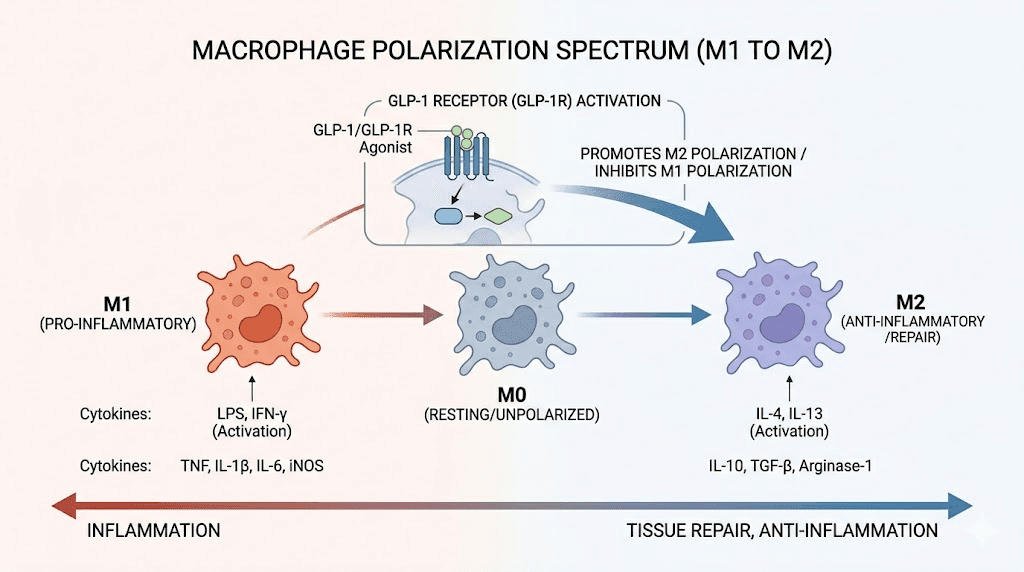
Macrophage polarization: M1 versus M2
This is arguably the most important immune effect of GLP-1 receptor agonists. And it is not what most people expect.
Macrophages exist on a spectrum between two states. M1 macrophages are pro-inflammatory. They are the attack dogs. They release TNF-alpha, IL-6, and other inflammatory cytokines that kill pathogens but also damage surrounding tissue. M2 macrophages are anti-inflammatory. They promote tissue repair, wound healing, and resolution of inflammation.
GLP-1 receptor agonists, including semaglutide, push macrophages toward the M2 phenotype.
Research demonstrates this through specific signaling pathways. When GLP-1 activates its receptor on macrophages, it triggers a cascade involving cyclic AMP (cAMP), protein kinase A (PKA), and STAT3 phosphorylation. The JNK inflammatory pathway gets attenuated. STAT3 activation increases. The result is macrophages that are less aggressive in their inflammatory response but more effective at tissue repair. Studies with exenatide, another GLP-1 receptor agonist, showed that it activates human monocyte-derived macrophages toward the M2 phenotype. Lixisenatide at 10 micrograms per kilogram per day decreased atheroma plaque size by reprogramming macrophages toward M2. This shift is why researchers are investigating GLP-1 agonists for conditions like autoimmune diseases and chronic inflammation.
But here is the nuance that matters. Shifting toward M2 does not mean your macrophages stop fighting infections. It means they fight less aggressively and clean up more efficiently. In most contexts, this is beneficial. In the context of an acute severe infection, it could theoretically slow the initial inflammatory response. No clinical data shows this happening with semaglutide at standard doses, but the mechanism exists.
Effects on neutrophils and eosinophils
Neutrophils and eosinophils also express GLP-1 receptors. The research shows that GLP-1 signaling significantly decreases eosinophil surface activation markers along with reduced production of IL-4, IL-8, and IL-13. For people with allergic conditions or inflammatory disorders, this could actually be helpful. Overactive eosinophils drive conditions like asthma, eosinophilic esophagitis, and certain skin conditions.
The pathway here involves PKA and NF-kB signaling. GLP-1 receptor activation on eosinophils dampens the NF-kB inflammatory cascade, reducing the production of inflammatory cytokines without eliminating the cells themselves. Your eosinophil count does not drop. They just become less reactive.
T cell effects: the vaccine question
This is the area that has generated the most scientific debate. And honestly, the most concern.
Activated T cells upregulate GLP-1 receptor expression starting about 24 hours after T cell receptor (TCR) activation, with peak expression at around 120 hours. In the context of vaccines, this means that vaccine-stimulated TCR activation can lead to increased expression of GLP-1 receptors on the very cells responsible for building immunity to the vaccine antigen.
A paper in ScienceDirect raised the possibility that long-acting GLP-1 receptor agonists like semaglutide may adversely affect vaccine response through GLP-1R signaling-induced T cell anergy. Anergy is a state where T cells become unresponsive. They are alive but functionally exhausted. Single-cell transcriptomic profiling confirmed that GLP-1R signaling mediates an anergic phenotype, with several characteristic markers of T cell exhaustion appearing after GLP-1 receptor activation.
A separate study published in Cell Metabolism described GLP-1R as a "T cell-negative costimulatory molecule." It showed that GLP-1R signaling prolonged allograft survival, mitigated alloimmune response, and reduced T lymphocyte graft infiltration. While that is beneficial in transplant medicine, it raises questions about whether the same mechanism could blunt your response to a flu shot or COVID booster.
Important context: no clinical study has yet demonstrated reduced vaccine effectiveness in patients taking semaglutide. The concerns are based on mechanistic data, not clinical outcomes. But the research calls for further investigation, and if you are concerned about semaglutide side effects, timing your vaccinations appropriately is worth discussing with your prescriber.
Regulatory T cells: the balancing act
GLP-1 receptor activation increases the frequency and function of regulatory T cells (Tregs). Research in diabetic NOD mice models showed enhanced IL-10 expression and increased inhibitory capacity following GLP-1 agonist treatment. Tregs are the peacekeepers of your immune system. They prevent autoimmune attacks by keeping other immune cells in check.
More Tregs means less autoimmune activity. That is why early research on GLP-1 agonists and conditions like rheumatoid arthritis, lupus, and endometriosis looks promising. But more Tregs also means slightly more immune tolerance, which in theory could mean slightly reduced vigilance against novel threats.
Again, clinical data has not shown this to be a problem in practice. But the mechanism deserves attention, particularly for people who are already immunocompromised for other reasons.
What the clinical trials actually found about infections
Let us move from mechanisms to outcomes. Because ultimately, what matters is whether people taking semaglutide actually get sick more often.
The SUSTAIN trials
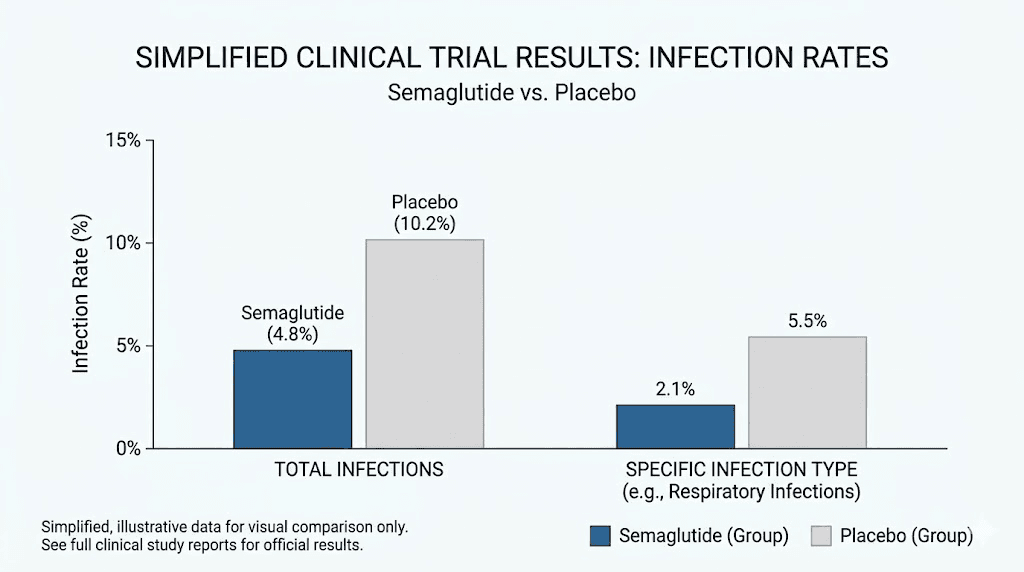
The SUSTAIN trial program evaluated semaglutide for type 2 diabetes management across multiple large-scale randomized controlled trials. Infection rates, including upper respiratory infections, urinary tract infections, and gastroenteritis, were tracked as adverse events. The results were clear: infection rates in the semaglutide group were comparable to those in the placebo and active comparator groups.
No signal. No trend. No statistically significant difference.
The STEP trials
The STEP program evaluated higher-dose semaglutide (2.4mg weekly) for weight management. These trials involved thousands of participants over extended periods. Once again, infection rates did not differ meaningfully between semaglutide and placebo groups. If semaglutide were suppressing immune function in a clinically relevant way, you would expect to see more infections, more severe infections, or longer recovery times. None of that appeared in the data.
This is consistent with what researchers studying semaglutide effectiveness have observed across multiple endpoints. The medication works primarily through metabolic pathways, and while it touches immune cells, the clinical impact on infection susceptibility appears minimal.
FDA safety data
The FDA prescribing information for Ozempic, Wegovy, and Rybelsus does not list immune suppression or increased infection risk among known risks, warnings, or precautions. The boxed warning relates to thyroid C-cell tumors observed in rodent studies. No immune-related warnings exist.
This matters because the FDA reviews not just trial data but also post-marketing surveillance. Millions of people now take semaglutide worldwide. If significant immune suppression were occurring, it would have shown up in the adverse event reporting system by now.
The anti-inflammatory properties of semaglutide (and why they matter)
Rather than suppressing your immune system, semaglutide appears to do something different. It reduces chronic inflammation.
There is an important distinction here. Immunosuppression means your immune system cannot fight threats effectively. Anti-inflammation means your immune system stops overreacting to things that are not actual threats. Chronic inflammation is not your immune system protecting you. It is your immune system damaging you. And semaglutide helps dial it back.
C-reactive protein reduction
C-reactive protein (CRP) is one of the most widely used markers of systemic inflammation. Multiple studies have shown that semaglutide reduces circulating CRP levels. This is significant because elevated CRP is associated with cardiovascular disease, type 2 diabetes complications, and chronic pain conditions.
For people managing liver inflammation or cardiovascular concerns, this anti-inflammatory effect is actually protective. Lower CRP means lower risk of inflammatory complications.
TNF-alpha and IL-6 suppression
Research shows that semaglutide and other GLP-1 receptor agonists reduce plasma levels of TNF-alpha and IL-6, two of the most potent pro-inflammatory cytokines. One study demonstrated that exenatide and semaglutide reduced TNF-alpha levels induced by agonists for TLR1, TLR2, TLR4, TLR5, and TLR9 receptors. These toll-like receptors (TLRs) are part of the innate immune pathogen recognition machinery. Reducing their inflammatory output does not prevent pathogen detection. It prevents the excessive inflammatory response that causes collateral tissue damage.
In the context of a study on cecal slurry-induced polymicrobial sepsis, a model of severe systemic infection, GLP-1 receptor agonists still reduced TNF-alpha while the immune system continued to fight the infection. The infection was controlled. The inflammation was modulated.
NF-kB pathway modulation
Nuclear factor kappa-B (NF-kB) is often called the master switch of inflammation. It controls the expression of hundreds of inflammatory genes. When NF-kB is chronically activated, you get persistent inflammation that drives tissue damage, metabolic dysfunction, and disease progression.
GLP-1 receptor agonists suppress NF-kB p65 activation through multiple mechanisms. Research on exendin-4, a GLP-1 agonist precursor, showed suppression of NF-kB and IL-1beta following surgical trauma. This is anti-inflammatory action, not immunosuppression. The cells remain functional. The pathways remain intact. The volume is just turned down from a chronic blare to a functional hum.
For people dealing with conditions driven by NF-kB overactivation, such as inflammatory conditions, gastrointestinal inflammation, and joint inflammation, this effect is genuinely therapeutic.
Why you might feel like you are getting sick more often on semaglutide
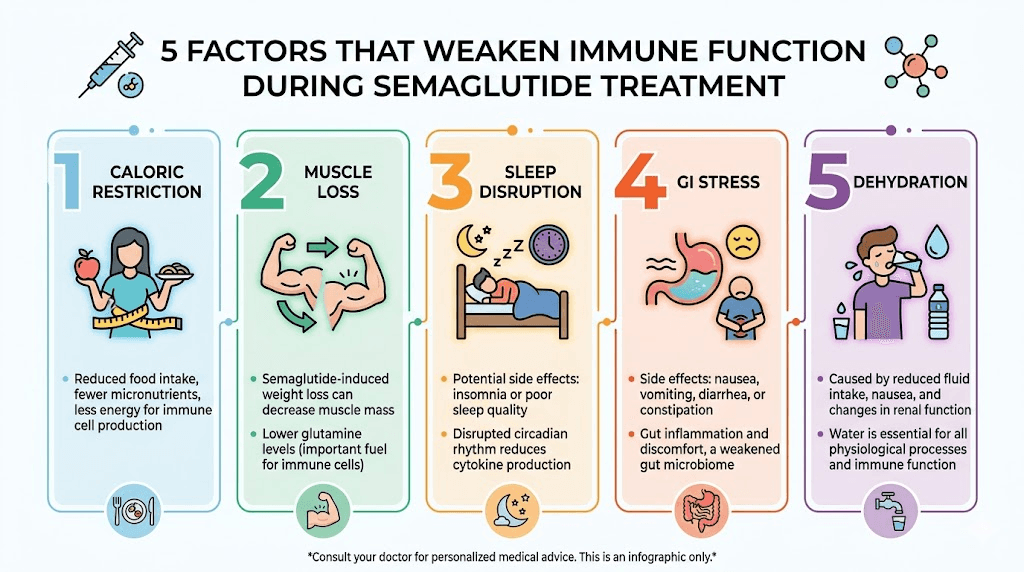
Here is the practical reality. Many people on semaglutide do report feeling more susceptible to illness. But the explanation is usually not immune suppression. It is one of several other factors that the medication creates or amplifies.
Caloric restriction and immune function
Semaglutide works partly by suppressing appetite. Dramatically. Many users report eating 30 to 50 percent less than their pre-medication intake. While that drives weight loss, severe caloric restriction has well-documented effects on immune function.
When you eat significantly less, your body gets fewer of the micronutrients that fuel immune responses. Zinc, vitamin C, vitamin D, iron, and B vitamins all play critical roles in immune cell production and function. If your reduced caloric intake means you are getting inadequate amounts of these nutrients, your immune function will suffer. Not because of semaglutide directly, but because of the dietary changes it induces.
This is why tracking caloric intake on semaglutide, maintaining adequate protein intake, and eating nutrient-dense foods is so important. It is not optional. It is essential for maintaining immune function during weight loss.
Muscle loss and immune vulnerability
Lean mass reductions ranging from nearly 0 percent to 40 percent of total weight reduction have been observed across semaglutide studies. Muscle is not just for strength. It is an endocrine organ that produces myokines, signaling molecules that support immune function. Significant muscle loss can impair your immune response independently of any direct drug effect.
Maintaining muscle mass through adequate protein intake (1.2 to 1.6 grams per kilogram of body weight per day) and resistance training is critical. Not just for body composition, but for keeping your immune system functional. People who lose weight on semaglutide while maintaining muscle through exercise and proper nutrition report better overall health outcomes than those who lose weight passively. You can explore whether it is possible to build muscle while on GLP-1 medications and how to approach it strategically.
Sleep disruption
Some semaglutide users report insomnia or disrupted sleep patterns. Sleep is foundational to immune function. During deep sleep, your body produces cytokines needed to fight infection and inflammation. Chronic sleep deprivation reduces the production of protective cytokines and antibodies while increasing inflammatory markers. If semaglutide is disrupting your sleep, either directly or through gastrointestinal side effects that wake you up at night, your immune function will decline. This is a sleep problem, not an immunosuppression problem.
Gastrointestinal stress
Nausea, constipation, bloating, and other GI side effects are common with semaglutide, especially in the early weeks. Your gut houses approximately 70 percent of your immune system. The gut-associated lymphoid tissue (GALT) is the largest collection of immune cells in your body. When your gut is stressed, your immune surveillance capabilities can be compromised.
GI distress can also reduce nutrient absorption, compounding the caloric restriction problem described above. If you are nauseous and barely eating, and then what you do eat is not being properly absorbed because of altered gastric motility, your immune-supporting nutrient levels drop further.
Managing GI side effects is not just about comfort. It is about maintaining the gut environment that your immune system depends on. Proper dietary approaches on semaglutide, including fiber supplementation and probiotics, can make a significant difference.
Dehydration
Reduced appetite often means reduced fluid intake too. Many people on semaglutide simply forget to drink enough water because they are not eating as much and do not feel thirsty. Dehydration impairs every aspect of immune function, from the production of lymph fluid that carries immune cells through your body to the maintenance of mucous membranes that serve as your first physical barrier against pathogens.
Semaglutide and autoimmune conditions: what the research shows
For the millions of people living with autoimmune conditions who are also interested in semaglutide for weight management, this section is critical.
The anti-inflammatory benefit for autoimmune patients
Preliminary research suggests that GLP-1 receptor agonists may actually benefit autoimmune conditions rather than worsen them. The mechanism makes sense given what we know about macrophage polarization and Treg enhancement. By shifting the immune response away from aggressive M1 inflammation and toward regulatory tolerance, semaglutide could theoretically reduce autoimmune flares.
Research has shown that GLP-1 reduces cytokines such as TNF-alpha, IL-6, and IL-1beta, which are major drivers of autoimmune diseases and arthritis. Preclinical research found that semaglutide ameliorated experimental autoimmune encephalomyelitis-induced multiple sclerosis in mice. These are early findings, but the direction is clear and consistent.
For people managing autoimmune conditions alongside GLP-1 therapy, the existing evidence is more reassuring than concerning.
Rheumatoid arthritis: encouraging signals
Studies examining GLP-1 medications and rheumatoid arthritis have found that treatment with GLP-1 agonists helped relieve RA symptoms. Patients experienced fewer flares, less joint pain and stiffness, better movement, and more energy. This aligns with the anti-inflammatory mechanisms described earlier, particularly the reduction in TNF-alpha and IL-6, both of which are therapeutic targets in RA treatment.
No study has shown that semaglutide worsens rheumatoid arthritis. If anything, the weight loss itself provides additional benefit by reducing mechanical stress on inflamed joints.
Lupus: a more complex picture
Lupus (systemic lupus erythematosus) presents a more nuanced situation. Post-marketing surveillance data from Ozempic showed that SLE-like symptoms were reported by 7 out of 12,332 individuals, a rate of 0.06 percent. This adverse effect was predominantly reported in females over the age of 60 who had been using the medication for less than one month.
There was one documented case report of semaglutide-induced lupus erythematosus with multiorgan involvement, published in PMC. This is concerning but extremely rare, and the causal relationship is not established. Drug-induced lupus is a recognized phenomenon with many medications, and it typically resolves when the medication is discontinued.
For people with existing lupus, the situation is unclear. Some researchers believe the anti-inflammatory properties could help. Others urge caution given the rare but documented reports. If you have lupus or another connective tissue disease, discuss semaglutide specifically with your rheumatologist before starting. You can also explore peptide approaches for lupus management.
Hashimoto's thyroiditis
Hashimoto's is the most common autoimmune condition in the United States, and many people with Hashimoto's are interested in semaglutide for the weight management challenges that hypothyroidism creates. The anti-inflammatory properties of GLP-1 agonists could theoretically reduce thyroid inflammation. No study has shown that semaglutide worsens Hashimoto's.
However, weight loss itself can alter thyroid hormone requirements. People on levothyroxine who lose significant weight on semaglutide may need dose adjustments. This is a medication management issue, not an immune issue, but it is important to monitor. Our guides on Hashimoto's and GLP-1 and GLP-1 for Hashimoto's disease cover this in detail.
Transplant patients: an important exception
One population where the immune-modulating effects of GLP-1 agonists have been studied carefully is organ transplant recipients. These patients already take immunosuppressive medications to prevent organ rejection. Research with liraglutide and dulaglutide in solid organ transplant patients demonstrated significant improvement in metabolic control and, critically, no interference with immunosuppressive drugs. This suggests that GLP-1 agonists do not significantly alter immune function in ways that compete with or undermine traditional immunosuppression.
The gut microbiome connection
Your gut microbiome is not separate from your immune system. It is deeply intertwined with it. And semaglutide changes it.
How semaglutide alters gut bacteria
Animal model studies have consistently demonstrated that semaglutide significantly alters gut microbiota composition. The medication slows gastric emptying, changes the pH of different sections of the digestive tract, and reduces food volume, all of which shift the balance of bacterial populations in your gut.
Some researchers believe that part of semaglutide metabolic benefit may derive from its capacity to modulate inflammation via gut-immune-microbiota interactions. The gut microbiome influences immune function through multiple pathways. Beneficial bacteria produce short-chain fatty acids that fuel intestinal immune cells. They maintain the integrity of the gut barrier, preventing bacterial translocation that triggers systemic inflammation. And they directly interact with immune cells in the GALT.
Implications for immune function
Changes in gut microbiome composition can affect immune function in both positive and negative ways. A shift toward more anti-inflammatory bacterial species could enhance immune regulation. A loss of microbial diversity could reduce immune resilience.
This is an area where practical steps matter. Supporting your gut microbiome during semaglutide treatment through probiotic supplementation, prebiotic fiber, and diverse food choices is not just about digestive comfort. It is about maintaining the immune foundation that lives in your gut.
The gut response support protocols developed for GLP-1 users address this specifically, and SeekPeptides members have access to detailed guides on optimizing gut health during peptide therapy.
Semaglutide and vaccine effectiveness: should you be concerned?
This deserves its own section because the research, while preliminary, raises questions that matter.
The mechanism of concern
As described earlier, activated T cells upregulate GLP-1 receptors starting about 24 hours after activation, peaking at 120 hours. When you receive a vaccine, your T cells activate in response to the vaccine antigen. If semaglutide is circulating at the same time and binding to those newly expressed GLP-1 receptors on your activated T cells, it could theoretically induce a degree of T cell anergy, reducing the vigor of the immune response to the vaccine.
The transcriptomic data supports this concern. Characteristic markers of T cell exhaustion and anergy-related genes are upregulated following GLP-1R signaling in T cells. The paper describing GLP-1R as a "T cell-negative costimulatory molecule" reinforces this mechanism.
What we do not know
No clinical study has measured vaccine antibody titers in patients taking semaglutide compared to controls. Most research has focused on acute metabolic outcomes, with limited studies exploring long-term immune adaptations. The concern is based on mechanistic data and in vitro findings, not real-world vaccine failure rates.
Millions of people on semaglutide have received flu shots, COVID boosters, and other vaccines. There are no widespread reports of vaccine failure in this population. That does not mean the effect does not exist, as it could be subtle enough to reduce vaccine effectiveness without causing complete failure. But it does mean we should approach this with proportionate concern, not panic.
Practical vaccine recommendations
Until more data is available, reasonable precautions include discussing vaccine timing with your prescriber. Some researchers suggest scheduling vaccinations during the trough of semaglutide activity cycle (the days before your next weekly injection) when drug levels are lowest. Others recommend simply proceeding with normal vaccination schedules since the theoretical risk is small and the risk of missing vaccinations is larger.
If you are immunocompromised for other reasons (HIV, post-transplant, autoimmune medications) and also taking semaglutide, this conversation becomes more important. Stacking multiple factors that could reduce vaccine effectiveness warrants more careful planning.
The HIV and immunocompromised research
One of the more interesting lines of research involves semaglutide in people with HIV-associated lipohypertrophy. A study published in Open Forum Infectious Diseases examined the effects of semaglutide on inflammation and immune activation in this population.
This is particularly relevant because HIV-positive individuals on antiretroviral therapy often have chronic low-grade inflammation that drives metabolic complications, even when their viral load is undetectable. The study found that semaglutide reduced inflammatory markers in this population without compromising immune function or antiretroviral efficacy.
This is powerful evidence against the "semaglutide lowers your immune system" narrative. In a population where immune function is already compromised and closely monitored, the medication did not cause further immune decline. It reduced inflammation, which is a different and generally beneficial effect.
Specific immune markers to watch
If you are concerned about immune function while taking semaglutide, here are the specific markers worth monitoring through regular blood work. Talk to your healthcare provider about including these in your routine labs.
Complete blood count (CBC) with differential
This is the most basic immune function test. It measures your white blood cell count and breaks it down into individual cell types: neutrophils, lymphocytes, monocytes, eosinophils, and basophils. If semaglutide were suppressing your immune system, you would see decreased counts in one or more of these cell types.
Most people on semaglutide have completely normal CBCs. If yours shows abnormalities, they are more likely related to nutritional deficiencies (iron deficiency from reduced food intake, for example) than to direct immune suppression. Regular blood work is important for anyone on semaglutide, as our guide on whether semaglutide shows up in blood work explains.
C-reactive protein (CRP)
As mentioned, semaglutide typically lowers CRP, which is beneficial. But tracking it gives you a baseline and helps identify if inflammation is increasing for other reasons (new infection, autoimmune flare, or unrelated condition).
Immunoglobulin levels
IgG, IgA, and IgM levels reflect the ability to produce antibodies. If semaglutide were truly immunosuppressive, you might see declining immunoglobulin levels over time. This has not been reported in clinical trials, but individual monitoring provides reassurance.
Vitamin and mineral levels
Given that reduced food intake is the most likely cause of perceived immune decline on semaglutide, checking vitamin D, zinc, iron, B12, and folate levels is practical. Deficiencies in any of these can impair immune function and are easily correctable with supplementation. Exploring supplements to take with GLP-1 medications and vitamin protocols for GLP-1 users can help you fill these gaps proactively.
How to protect your immune system while on semaglutide
Whether or not semaglutide directly affects your immune system, you can take concrete steps to ensure your immune function remains robust during treatment. These recommendations are evidence-based and practical.
Prioritize protein
Protein is the building block of immune cells, antibodies, and cytokines. When you cut calories on semaglutide, protein is the last macronutrient you should sacrifice. Aim for 1.2 to 1.6 grams per kilogram of body weight per day. For a 180-pound person, that is roughly 98 to 131 grams daily.
Each meal should include a high-quality protein source. Eggs, chicken, fish, Greek yogurt, cottage cheese, lean beef. If nausea makes solid food difficult, protein shakes become essential. Our guide on protein shakes for GLP-1 users covers the best options.
Supplement strategically
At minimum, consider these supplements during semaglutide treatment:
Vitamin D (2,000 to 5,000 IU daily) supports T cell function and innate immune responses. Deficiency is extremely common and is associated with increased susceptibility to respiratory infections. Zinc (15 to 30mg daily) is essential for the development and function of neutrophils, NK cells, and T cells. Vitamin C (500 to 1,000mg daily) supports epithelial barrier function and accumulates in phagocytic cells like neutrophils. B12 is particularly important if you are eating less red meat, as it plays a role in immune cell production. Interestingly, many compounded semaglutide formulations include B12 for this reason. Our guide on semaglutide with B12 explores this combination in depth.
Maintain gut health
A high-quality probiotic containing Lactobacillus and Bifidobacterium strains supports the gut immune barrier. Prebiotic fiber from foods like garlic, onions, leeks, and asparagus feeds beneficial bacteria. Fermented foods like kimchi, sauerkraut, and kefir provide additional microbial diversity.
This is especially important during the first few weeks of semaglutide when GI side effects are most pronounced. Your gut needs support while it adapts to the effects on gastric motility from the medication and microbiome composition. The constipation relief strategies and GLP-1 friendly recipes can help you maintain a diet that supports both weight loss and immune function.
Prioritize sleep
Seven to nine hours nightly. Not negotiable for immune function. If semaglutide-related GI symptoms are disrupting your sleep, address them aggressively. Take your injection earlier in the day if evening injections cause nighttime nausea. Elevate the head of your bed if acid reflux is an issue. Our guides on semaglutide and acid reflux and best injection timing can help optimize your schedule.
Exercise regularly
Moderate exercise enhances immune function through multiple mechanisms. It improves immune cell circulation, reduces chronic inflammation, and supports gut microbiome diversity. Resistance training is particularly important on semaglutide to preserve muscle mass and the immune-supporting myokines that muscles produce.
Aim for 150 minutes of moderate cardio plus 2 to 3 resistance training sessions weekly. This combination supports both weight loss goals and immune resilience. If you are wondering about the interplay between exercise and GLP-1 therapy, our guide on whether you can lose weight on semaglutide without exercise discusses why exercise remains important even with the appetite suppression from the medication.
Stay hydrated
Aim for at least 64 ounces of water daily, more if you are exercising or in warm climates. Set reminders if you need to, because reduced appetite can also reduce your awareness of thirst. Hydration supports lymph fluid production, mucous membrane integrity, and every other aspect of immune function.
Manage stress
Chronic psychological stress suppresses immune function through cortisol elevation. If weight loss, medication side effects, or other life stressors are overwhelming you, your immune system will pay the price regardless of what semaglutide is or is not doing at the cellular level.
Semaglutide dose and immune effects: does higher dose mean more risk?
An important question for people managing semaglutide dosing is whether the immune-modulating effects increase with dose.
Dose-dependent anti-inflammatory effects
The anti-inflammatory properties of GLP-1 agonists do appear to be dose-dependent in preclinical studies. Higher concentrations produce greater reductions in inflammatory cytokines and more pronounced macrophage M2 polarization. In the context of reducing chronic inflammation, this is generally positive. Higher doses of semaglutide (like the 2.4mg used in the STEP weight loss trials compared to the 1.0mg maximum used for diabetes) may provide greater anti-inflammatory benefit.
Clinical infection rates across doses
However, clinical trial data does not show increased infection rates at higher semaglutide doses. The STEP trials used the higher 2.4mg dose and still found infection rates comparable to placebo. If dose-dependent immunosuppression were occurring, you would expect to see it most clearly at the highest doses. It does not appear.
This suggests that while the immune-modulating effects may intensify with dose, they do not cross the threshold into clinically meaningful immunosuppression at any approved dose level. For people working through the semaglutide dose chart and titrating upward, this should be reassuring.
The compounded semaglutide consideration
People using compounded semaglutide sometimes take doses or formulations that differ from commercially available versions. The immune-modulating effects described in this article apply to the semaglutide molecule itself, regardless of the formulation. The key variable is the total weekly dose, not whether it comes from a pen or a compounded vial.
If your compounded formulation includes additional ingredients like B12, glycine, or L-carnitine, those additional compounds may independently support immune function. B12 in particular is important for immune cell production, which is one reason it is commonly added to compounded GLP-1 formulations. Understanding the semaglutide glycine B12 blend rationale can help you choose the right formulation.
Comparing semaglutide to other GLP-1 agonists on immune effects
Semaglutide is not the only GLP-1 receptor agonist available. How do others compare in terms of immune modulation?
Liraglutide (Saxenda, Victoza)
Liraglutide has been studied more extensively than semaglutide for immune effects due to its longer time on the market. Research shows similar anti-inflammatory properties: macrophage M2 polarization via the cAMP-PKA-STAT3 pathway, NF-kB suppression, and reduction in inflammatory cytokines. At 30 micrograms per kilogram per day, liraglutide ameliorated macrophage accumulation in periodontitis models by decreasing M1 markers while preserving M2 phenotypes.
Liraglutide has also been used safely in transplant patients taking immunosuppressive medications, demonstrating that it does not meaningfully alter immune function in clinically relevant ways. If you are curious about the differences between these medications, our comparison guide on liraglutide versus semaglutide covers the pharmacological distinctions.
Tirzepatide (Mounjaro, Zepbound)
Tirzepatide is a dual GIP/GLP-1 receptor agonist. It activates both the GLP-1 receptor and the glucose-dependent insulinotropic polypeptide (GIP) receptor. The GLP-1 receptor-mediated immune effects described in this article apply to tirzepatide as well. The GIP receptor component adds another layer of complexity, as GIP receptors are also expressed on some immune cells.
Clinical trials for tirzepatide have not shown increased infection rates compared to placebo, consistent with the semaglutide data. For people choosing between these medications, immune effects should not be a differentiating factor. Our detailed comparison of semaglutide versus tirzepatide side effects covers the full spectrum of differences.
Retatrutide
Retatrutide is a triple agonist targeting GLP-1, GIP, and glucagon receptors. As a newer compound with less clinical data, its immune effects are less well characterized. However, the GLP-1 receptor component would be expected to produce similar anti-inflammatory and immune-modulating effects as semaglutide. The glucagon receptor component could add additional metabolic effects that indirectly influence immune function through changes in energy metabolism.
For a broader comparison of these newer compounds, our guide on semaglutide versus tirzepatide versus retatrutide provides a comprehensive head-to-head analysis.
Specific situations where immune function matters most
Before surgery
If you are scheduled for surgery while on semaglutide, the anti-inflammatory effects could theoretically be beneficial for post-operative healing (M2 macrophage polarization supports tissue repair). However, the reduced caloric intake and potential nutritional deficiencies could impair wound healing and infection resistance. Most surgeons recommend stopping semaglutide before surgery primarily due to aspiration risk from delayed gastric emptying, not immune concerns. Our guide on resuming semaglutide after surgery covers the timing considerations.
During chemotherapy
There is insufficient data on combining semaglutide with chemotherapy. Chemotherapy is truly immunosuppressive, destroying rapidly dividing cells including immune cells. Adding the immune-modulating effects of semaglutide on top of chemotherapy-induced immunosuppression has not been studied. If you are undergoing cancer treatment, semaglutide decisions should involve your oncologist.
During pregnancy
Semaglutide is contraindicated during pregnancy and should be stopped at least two months before conception. The immune system undergoes dramatic changes during pregnancy (shifting toward Th2 dominance to tolerate the fetus), and the effects of semaglutide on this process are unknown. If you are concerned about fertility while on semaglutide, our guide on semaglutide while trying to get pregnant and semaglutide and fertility cover the evidence.
During cold and flu season
There is no reason to stop semaglutide during cold and flu season. The clinical data shows no increased susceptibility to respiratory infections. Focus instead on the supportive measures outlined above: nutrition, sleep, hydration, and supplementation. These are far more impactful for your seasonal immune resilience than any direct effect of semaglutide on your immune cells.
If you are already immunocompromised
For people with existing immune compromise (HIV, primary immunodeficiency, post-transplant, on immunosuppressive medications for autoimmune conditions), starting semaglutide deserves careful consideration with your treating specialist. The research in HIV patients is actually reassuring, showing anti-inflammatory benefit without immune compromise. But each situation is unique, and stacking multiple factors that touch the immune system warrants individualized guidance.
SeekPeptides provides detailed resources for navigating these complex situations, with evidence-based protocols and community support from researchers who have managed similar challenges.
What does "immune modulation" actually mean for your daily life?
Let us bring this back to practical reality. After reviewing all the mechanisms, pathways, and clinical data, what does this mean for you, the person taking semaglutide?
You are not immunosuppressed
Semaglutide does not suppress your immune system in any clinically meaningful way. You do not need to avoid crowds, wear masks beyond normal recommendations, or take special infection precautions solely because of semaglutide.
Your inflammation is probably lower
The anti-inflammatory effects of semaglutide are real and generally beneficial. Lower chronic inflammation means reduced cardiovascular risk, less tissue damage from overactive immune responses, and potentially improved outcomes in inflammatory conditions. This is a feature, not a bug.
Your indirect risk factors matter more
If you are getting sick more often on semaglutide, the cause is almost certainly related to reduced nutrition, sleep disruption, dehydration, GI stress, or muscle loss rather than direct immune suppression. Address these factors and your immune function will improve regardless of what the medication is doing at the receptor level.
Vaccines still work
Until evidence shows otherwise, proceed with your normal vaccination schedule. The theoretical concern about T cell anergy is real at the mechanistic level but has not translated into clinical vaccine failure. Missing a flu shot because of unproven concerns about semaglutide interaction is riskier than getting the shot.
Monitor but do not worry
Regular blood work including a CBC with differential will catch any significant changes in immune cell counts. Track your CRP to monitor inflammation. Check vitamin levels to ensure nutritional adequacy. These routine measures will give you and your prescriber the data needed to make informed decisions.
The neuroinflammation angle
One emerging area of research that deserves mention is the effects of semaglutide on neuroinflammation. GLP-1 receptors exist in the brain, and some researchers believe semaglutide may reduce inflammation within the central nervous system.
Studies have shown inhibition of microglia-mediated neuroinflammation from semaglutide use. Microglia are the brain's resident immune cells, analogous to macrophages in the body. When chronically activated, they drive conditions like Alzheimer's disease, Parkinson's disease, and chronic brain fog.
Novo Nordisk initiated a study examining how semaglutide affects the immune system in people with Alzheimer's disease, with results expected in the near future. The primary endpoint was changes in gene expression in immune cells in blood and cerebrospinal fluid, assessed by single-cell RNA sequencing.
For semaglutide users experiencing brain fog or cognitive changes, this research may eventually provide answers about whether the medication helps or hinders neural immune function. The best peptides for brain function guide covers compounds that work through complementary neurological pathways.
Signs your immune system is actually compromised (versus normal adjustment)
It helps to distinguish between normal adaptation symptoms and genuine immune compromise. Not every sniffle means your immune system is failing.
Normal adjustment symptoms (not immune compromise)
Mild fatigue in the first two to four weeks as your body adapts to lower caloric intake. Occasional headaches related to GLP-1 side effects rather than infection. Feeling cold more often, which is a common side effect related to reduced caloric thermogenesis, not immune dysfunction. Our guide on feeling cold on semaglutide explains the mechanism. Mild dizziness or lightheadedness from reduced food intake. These are metabolic adjustment symptoms, not immune problems, and they typically resolve within four to six weeks as your body acclimates to the new metabolic state.
Potential warning signs (worth investigating)
Wounds that take significantly longer to heal than your normal baseline. Recurring oral thrush or yeast infections, which could indicate immune surveillance decline, and our guide on semaglutide and yeast infections covers this. Shingles reactivation, which can occur when cellular immunity declines. Persistent low-grade fevers without obvious cause. Infections that seem disproportionately severe relative to the pathogen. These warrant blood work and a conversation with your provider. They are not expected effects of semaglutide, but they could indicate nutritional deficiencies or other factors that need addressing.
Common misconceptions about semaglutide and immunity
Misconception: semaglutide is an immunosuppressant
False. Immunosuppressants like cyclosporine, tacrolimus, and methotrexate directly inhibit specific immune cell populations or signaling pathways critical for immune defense. Semaglutide modulates inflammatory responses without compromising immune system capacity to fight infections. The clinical trial data is unambiguous on this point.
Misconception: getting sick on semaglutide proves it weakens your immune system
Correlation is not causation. People start semaglutide at various times of year, often during periods of stress (making a major health change is inherently stressful). They eat less, potentially sleep worse, and their gut microbiome shifts. All of these factors can increase susceptibility to illness independently of any direct immune effect. Attributing every cold to the medication ignores these confounding factors.
Misconception: semaglutide cancels out vaccines
No clinical evidence supports this. The T cell anergy mechanism is a theoretical concern that warrants further study, not a reason to skip vaccinations. Continue following recommended vaccination schedules. If concerned, discuss timing with your provider.
Misconception: higher doses are more dangerous for immunity
Clinical data does not show dose-dependent increases in infection risk. The 2.4mg weight loss dose was as safe from an immune perspective as the lower diabetes doses. Higher doses do produce more anti-inflammatory effect, but anti-inflammation is not the same as immunosuppression.
Misconception: you should stop semaglutide if you get sick
This is a decision to make with your healthcare provider, not based on internet speculation. In most cases, continuing semaglutide during minor illness is fine. For serious illness requiring hospitalization, your medical team will make medication decisions based on your specific situation. The more relevant concern during illness is maintaining hydration and nutrition, which can be challenging when semaglutide is already suppressing appetite.
Misconception: natural GLP-1 does not affect immunity, only the drug does
Your body naturally produces GLP-1 after meals. Natural GLP-1 has the same receptor interactions on immune cells as the medication. The difference is duration and intensity, as semaglutide keeps GLP-1 receptor activation elevated continuously rather than in brief post-meal bursts. The same immune modulation occurs naturally. Semaglutide simply amplifies and extends it.
The big picture: inflammation, obesity, and immune function
Here is the perspective that most articles on this topic miss entirely.
Obesity itself is an immunocompromising condition. Excess adipose tissue produces pro-inflammatory cytokines that chronically activate the immune system, leading to a state of constant low-grade inflammation. This chronic activation impairs immune system capacity to respond to acute threats. Obese individuals have documented higher rates of infections, worse outcomes from respiratory illnesses (including COVID-19), impaired vaccine responses, and higher rates of cancer.
By helping people lose weight, semaglutide may actually improve immune function in the long run, even if the mechanisms described in this article produce modest short-term modulation. The reduction in chronic inflammation, the improvement in metabolic health, the decrease in adipose-derived inflammatory signaling, all of these contribute to a healthier, more responsive immune system.
The question is not just "does semaglutide lower your immune system" but "does losing weight on semaglutide improve your immune system over time?" The answer, based on what we know about obesity and immunity, is very likely yes.
For people working through plateaus, managing stalls in weight loss, or considering long-term semaglutide use, this bigger picture matters. The short-term immune modulation from the medication is likely far outweighed by the long-term immune benefit of achieving and maintaining a healthier weight.
When to talk to your doctor about immune concerns
While semaglutide does not suppress your immune system, certain symptoms warrant medical attention regardless of the cause. Contact your healthcare provider if you experience:
Recurrent infections (three or more in a three-month period), especially if they are infections you do not normally get or ones that are unusually severe. Infections that do not resolve with standard treatment or that last significantly longer than expected. Unexplained fevers lasting more than a few days. Swollen lymph nodes that persist for weeks without apparent cause, as our guide on semaglutide and swollen lymph nodes explains. Significant fatigue that does not improve with adequate sleep, nutrition, and hydration, which may warrant investigation beyond what our semaglutide fatigue guide covers. Any new autoimmune symptoms such as joint pain, skin rashes, or unusual hair loss.
These symptoms could indicate issues unrelated to semaglutide, or they could signal nutritional deficiencies, thyroid changes from weight loss, or other conditions that need attention. The point is not to blame or exonerate semaglutide but to ensure any immune changes are properly evaluated.
The future of GLP-1 and immune research
The intersection of GLP-1 biology and immunology is an active and expanding field of research. Several directions are worth watching.
Autoimmune therapeutics. Multiple research groups are investigating whether GLP-1 agonists could be repurposed as treatments for autoimmune conditions. The Treg-enhancing, M2-polarizing, NF-kB-suppressing properties make them interesting candidates for conditions like rheumatoid arthritis, multiple sclerosis, and inflammatory bowel disease. If these studies succeed, the narrative around GLP-1 and immunity will shift from "does it suppress" to "how can we harness."
Vaccine optimization studies. Researchers have called for clinical trials specifically measuring vaccine antibody titers in patients on long-acting GLP-1 agonists. These studies will either confirm or refute the T cell anergy concern. Until they are completed, the question remains theoretical.
Neuroinflammation trials. The Novo Nordisk Alzheimer's study and others examining the effects of semaglutide on brain inflammation could open entirely new therapeutic applications. If semaglutide reduces pathological neuroinflammation without compromising peripheral immune function, it could become a dual-purpose medication for metabolic and neurodegenerative conditions.
Gut-immune axis research. The relationship between semaglutide-induced microbiome changes and immune function is largely unexplored in clinical settings. Future studies mapping specific bacterial changes to specific immune outcomes will help clinicians optimize gut support during GLP-1 therapy.
SeekPeptides tracks these developments closely, providing members with up-to-date research summaries and evidence-based guidance as new data emerges.
Semaglutide, weight loss medications, and the broader immune landscape
Understanding the immune effects of semaglutide becomes clearer when you compare it to the broader landscape of weight loss interventions and their documented impacts on immune function.
Semaglutide versus surgical weight loss
Bariatric surgery produces far more dramatic immune changes than semaglutide. Rapid weight loss after gastric bypass or sleeve gastrectomy causes significant nutritional deficiencies, microbiome disruption, and metabolic shifts that can transiently suppress immune function. Patients commonly experience increased infection susceptibility in the first three to six months post-surgery. The gradual weight loss approach with semaglutide, typically 1 to 2 pounds per week, is far gentler on the immune system. The slower pace allows the body to adapt, maintain nutritional status more easily, and preserve more muscle mass. If you are comparing weight loss approaches and wondering about average weight loss per month on GLP-1 medications, the rate is considerably more moderate than surgical interventions.
Semaglutide versus crash dieting
Crash diets, those extreme caloric restrictions below 800 calories per day, have well-documented immunosuppressive effects. They reduce lymphocyte counts, impair phagocytic cell function, and decrease secretory IgA production. Semaglutide, even at its most appetite-suppressing, rarely drives caloric intake that low. Most users settle at 1,200 to 1,800 calories per day, well above the threshold for diet-induced immunosuppression.
The key difference is sustainability. Crash diets produce immune deficits because they are extreme and unsustainable. Semaglutide produces gradual, sustained caloric reduction that the body can adapt to if nutritional quality is maintained. Tracking your intake using a semaglutide diet plan ensures you stay above the threshold where immune function declines.
Semaglutide versus phentermine
People sometimes ask whether phentermine or semaglutide is "harder" on the immune system. Phentermine is a sympathomimetic amine that stimulates the central nervous system. Chronic sympathetic activation can suppress immune function through cortisol elevation and reduced vagal tone. Semaglutide does not stimulate the sympathetic nervous system and does not elevate cortisol. From a pure immune perspective, semaglutide is the gentler option.
For those considering combination approaches, our guides on combining phentermine and semaglutide and whether phentermine is a GLP-1 clarify the pharmacological differences.
The role of other medications taken alongside semaglutide
Many semaglutide users take other medications that have their own immune effects. Metformin has documented anti-inflammatory properties and may complement the immune modulation from semaglutide. Berberine, a popular supplement often taken alongside GLP-1 medications, also has immunomodulatory properties. Even creatine, commonly used to preserve muscle mass during weight loss, has been shown to support immune cell energy metabolism.
The point is that your immune function on semaglutide is not determined by a single medication. It is the sum of everything, your nutrition, sleep, exercise, stress, hydration, supplementation, and the entirety of your medication regimen. Isolating semaglutide as the sole variable misses the bigger picture.
Long-term versus short-term immune considerations
Most concerns about semaglutide and immunity focus on the short term, the first few weeks or months when the body is adjusting. GI side effects are strongest, appetite suppression is most dramatic, and nutritional changes are most abrupt. This is when immune vulnerability peaks, not because of the medication itself but because of the lifestyle disruption it creates.
Long term, the picture reverses. People who have been on semaglutide for a year or more have typically lost significant weight, reduced their chronic inflammation, improved their metabolic health markers, and established sustainable dietary patterns. Their immune function is almost certainly better than it was at their pre-treatment weight. The initial adjustment period is a temporary cost that pays dividends over time.
Understanding the week-by-week results timeline and what to expect in the first week helps set appropriate expectations for this adjustment period. And for those thinking about stopping semaglutide, understanding the withdrawal process and maintaining results is equally important for long-term immune health.
For researchers serious about optimizing their peptide protocols and understanding the full scope of how these medications interact with every system in the body, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
Frequently asked questions
Does semaglutide suppress the immune system like steroids do?
No. Steroids like prednisone broadly suppress immune cell function and reduce white blood cell counts. Semaglutide modulates inflammatory responses without reducing immune cell numbers or compromising the ability to fight infections. Clinical trial infection rates are comparable to placebo, and the FDA does not list immune suppression as a risk. The two medications work through completely different mechanisms on the immune system.
Should I stop semaglutide before getting a vaccine?
Current evidence does not support stopping semaglutide for vaccinations. While mechanistic research suggests possible effects on T cell responses, no clinical study has shown reduced vaccine effectiveness in semaglutide users. Continue your normal vaccination schedule and discuss any concerns with your prescribing provider.
Why do I keep getting colds since starting semaglutide?
This is almost certainly due to reduced nutritional intake, not direct immune suppression. When you eat 30 to 50 percent less food, you get fewer immune-supporting nutrients like zinc, vitamin D, vitamin C, and B vitamins. Addressing nutritional supplementation, protein intake, and hydration typically resolves the issue.
Is semaglutide safe for people with autoimmune conditions?
The evidence suggests semaglutide may actually benefit some autoimmune conditions through its anti-inflammatory properties. Research shows reduced flares in rheumatoid arthritis and potential benefits in multiple sclerosis models. However, rare cases of drug-induced lupus-like symptoms have been reported. Discuss your specific autoimmune condition with your specialist before starting semaglutide. Our guides on microdosing GLP-1 for autoimmune conditions and peptides for autoimmune diseases provide additional context.
Does the dose of semaglutide affect immune function?
Anti-inflammatory effects may increase with dose in laboratory settings, but clinical trial data shows no increased infection rates at higher doses (2.4mg versus 1.0mg). The semaglutide dosing chart and dosage calculator can help you navigate appropriate dosing levels.
Can semaglutide help with chronic inflammation?
Yes. Semaglutide reduces CRP, TNF-alpha, and IL-6, all key markers of chronic inflammation. It also shifts macrophages toward anti-inflammatory M2 phenotypes and enhances regulatory T cell function. For people with conditions driven by chronic inflammation, these effects are genuinely therapeutic. Exploring inflammation-targeting peptides can provide complementary approaches.
Should I get my immune system tested while on semaglutide?
Routine blood work including a CBC with differential is sufficient for most people. Add CRP to monitor inflammation trends and check vitamin D, zinc, B12, and iron levels to catch nutritional deficiencies early. There is no need for specialized immunological testing unless you are experiencing recurrent infections or have a pre-existing immune condition.
Does natural GLP-1 from food affect immunity too?
Yes. Your body produces GLP-1 naturally after eating, and it interacts with the same receptors on immune cells. The difference with semaglutide is duration and consistency, as the medication maintains elevated GLP-1 receptor activation continuously rather than in brief post-meal pulses. The immune modulation from natural GLP-1 is part of normal physiology.
External resources
GLP-1 receptor agonist as a modulator of innate immunity (Frontiers in Immunology)
GLP-1R agonist therapy and vaccine response: Neglected implications (ScienceDirect)
Anti-inflammatory benefits of semaglutide: State of the art (PMC)
In case I do not see you, good afternoon, good evening, and good night. May your immune cells stay vigilant, your inflammation stay controlled, and your health stay on the upward trajectory you deserve.