Mar 20, 2026
Clinical trials say tirzepatide does not cause bone loss. Real-world data tells a different story. And somewhere between those two truths lies the answer that matters most to you, the person injecting this medication every week and wondering what it is doing to your skeleton.
The SURPASS and SURMOUNT trials, the foundational studies that earned tirzepatide its FDA approvals, did not identify osteoporosis or bone density loss as adverse events. Neither Mounjaro nor Zepbound lists bone loss or fractures in its official labeling. That sounds reassuring. But a large retrospective cohort study of nearly 460,000 patients found that tirzepatide users faced a 44% higher risk of osteoporosis or fragility fractures compared to people taking other GLP-1 receptor agonists. That sounds alarming. Both findings are real. Both require context.
This is not a simple yes-or-no question. The relationship between tirzepatide and bone health involves weight loss mechanics, hormonal shifts, gut microbiota changes, nutritional status, and individual risk factors that interact in ways most guides completely ignore. Understanding these mechanisms, and more importantly, knowing what you can do about them, separates informed users from those who discover problems too late. SeekPeptides members have access to detailed protocols for managing every aspect of tirzepatide therapy, including bone health strategies that most providers never discuss.
This guide covers every angle. The clinical evidence. The real-world signals. The biological mechanisms. The risk factors that make some people more vulnerable. And the concrete, evidence-based steps you can take to protect your bones while still getting the weight loss results you want.
What the clinical trials actually found
The pivotal clinical trial programs for tirzepatide span two major categories. The SURPASS trials evaluated tirzepatide for type 2 diabetes management. The SURMOUNT trials evaluated it for weight loss in people with obesity. Together, these programs enrolled thousands of participants across multiple dose levels and treatment durations.
None of them flagged bone loss as a significant adverse event.
That matters. But it also requires context. Most of these trials lasted 40 to 72 weeks, a timeframe that may not be long enough to detect gradual bone density changes. Bone remodeling happens slowly. A DEXA scan taken at the end of a 52-week trial might not capture the full trajectory of what is happening inside weight-bearing bones. More importantly, bone health was not a primary or secondary endpoint in any of these studies. The researchers were measuring HbA1c reduction, weight loss percentages, and cardiovascular markers, not running serial bone density scans.
The FDA labeling for both Mounjaro and Zepbound reflects this. Neither product lists osteoporosis, bone loss, bone mineral density reduction, or fractures among its known adverse effects. For many patients, this is where the conversation ends. Their doctor prescribes tirzepatide, they read the prescribing information, and they assume their bones are safe.
That assumption may be premature.
What the real-world evidence reveals
Outside the controlled environment of clinical trials, a different picture has started to emerge. And it is a picture worth paying attention to.
A large retrospective cohort study using the TriNetX database compared tirzepatide users to people taking other GLP-1 receptor agonists like semaglutide and liraglutide. The study included nearly 460,000 patients. The findings were significant. Tirzepatide users had a hazard ratio of 1.44 for new-onset osteoporosis or fragility fractures. They also had a hazard ratio of 1.61 for initiating osteoporosis therapy.
In plain terms, tirzepatide users were 44% more likely to develop osteoporosis or suffer a fragility fracture compared to people on other GLP-1 medications. And they were 61% more likely to need osteoporosis treatment.
Those numbers deserve attention.
A separate study presented at the Endocrine Society found that in patients at high risk for fractures, greater weight loss from tirzepatide correlated with greater declines in total hip bone mineral density. The specific numbers tell the story clearly. Significant declines appeared at the lumbar spine (1.6%), femoral neck (1.8%), and total hip (2.8%). These are not catastrophic losses in isolation. But for someone already at risk, particularly a postmenopausal woman or an older adult with existing low bone density, they could push someone from borderline to diagnosable osteoporosis.
An animal study published in the Journal of Orthopaedic Translation went further. Researchers found that tirzepatide administration promoted bone loss in obese mice through changes in gut microbial metabolites. Specifically, Lachnospiraceae, a bacterial family known to support bone health, was significantly reduced after tirzepatide treatment. When researchers transplanted Lachnospiraceae back into the treated mice, the bone loss improved.
Why the disconnect between trials and real-world data
Several factors explain why clinical trials missed what real-world data caught. Trial durations may have been too short. Bone density was not a measured endpoint. Trial populations were carefully selected and monitored, receiving regular nutritional guidance and follow-up that typical patients do not get. And the sheer magnitude of weight loss, with some patients losing 15 to 21% of their body weight, creates skeletal stress that takes time to manifest fully.
Real-world patients also face realities that trial participants do not. They skip meals because of nausea and appetite suppression. They do not always take calcium or vitamin D. They may not exercise. They may not get DEXA scans. They may not even think about their bones until something breaks.
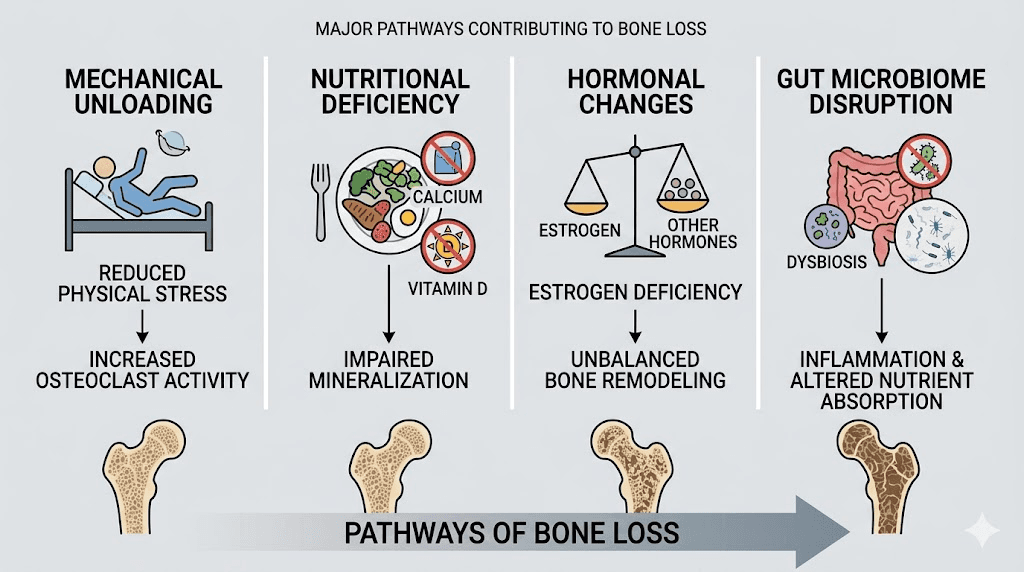
How tirzepatide affects your bones: the mechanisms
Understanding why tirzepatide might impact bone health requires looking at multiple interconnected pathways. This is not a single-mechanism problem. It is a cascade.
Mechanical unloading from weight loss
Your bones respond to the forces placed on them. This is a fundamental principle of bone biology known as mechanotransduction. When you carry more weight, your weight-bearing bones adapt by maintaining higher density. When that weight drops rapidly, the mechanical stimulus decreases.
Think of it like a building. If you remove several floors from a skyscraper, the foundation no longer needs to support as much load. Over time, the foundation, your skeleton, remodels to match the reduced demand. This is not unique to tirzepatide. Bariatric surgery studies consistently show bone density decreases of 2 to 10% in the first year after the procedure, with the hip and spine most affected.
Tirzepatide produces weight loss comparable to some surgical interventions. In the SURMOUNT trials, participants on the highest dose lost an average of 22.5% of their body weight. That level of rapid weight reduction creates the same mechanical unloading effect that drives post-surgical bone loss.
Nutritional deficiency pathways
Rapid weight loss on tirzepatide often involves dramatically reduced food intake. The appetite suppression that makes the medication effective for weight loss also reduces total nutrient consumption. Three nutrients matter most for bone health.
Calcium. The building block of bone mineral. Adults need 1,000 to 1,200 mg daily. When caloric intake drops by 40 to 60%, calcium consumption typically falls with it, especially in people who are not consciously supplementing.
Vitamin D. Essential for calcium absorption. Without adequate vitamin D, your body cannot use the calcium you consume. Serum 25-hydroxyvitamin D levels need to stay above 20 to 30 ng/mL for optimal bone health. Many people on tirzepatide eating plans are not getting enough from food or sunlight alone.
Protein. The structural framework of bone. Roughly 50% of bone volume is protein, primarily collagen. During rapid weight loss, protein intake often falls below the 1.0 to 1.2 g/kg body weight daily minimum needed to maintain both muscle mass and bone integrity.
The gastrointestinal side effects of tirzepatide compound this problem. Nausea affects 12 to 33% of users. Vomiting hits 5 to 12%. Diarrhea affects 9 to 23%. These symptoms make it harder to eat enough, harder to absorb nutrients, and harder to maintain the nutritional foundation bones need.
Hormonal disruption
Fat tissue is not just storage. It is an endocrine organ that produces estrogen through a process called aromatization. When you lose substantial body fat on tirzepatide, estrogen production from adipose tissue decreases. For women, particularly those who are perimenopausal or postmenopausal, this reduction compounds the estrogen decline already happening naturally.
Estrogen is one of the most important hormones for bone preservation. It inhibits osteoclasts, the cells that break down bone tissue. When estrogen drops, osteoclast activity increases, and bone resorption accelerates. This is why postmenopausal women are the highest-risk group for osteoporosis, and why adding significant fat loss on top of age-related estrogen decline creates a particularly dangerous combination.
Leptin also plays a role. This hormone, produced by fat cells, influences bone formation through complex signaling pathways. Rapid fat loss reduces circulating leptin levels, which may further impair bone building processes.
Gut microbiome disruption
This is the newest and perhaps most surprising mechanism. Research published in the Journal of Orthopaedic Translation demonstrated that tirzepatide alters the gut microbiome in ways that directly affect bone metabolism.
Lachnospiraceae, a bacterial family that produces short-chain fatty acids supportive of bone health, was significantly reduced in tirzepatide-treated mice. When researchers transplanted Lachnospiraceae bacteria back into the treated animals, bone loss improved. This suggests that targeting gut microbiota composition could be a viable strategy for protecting bone health during tirzepatide treatment.
The implications are significant. If gut bacteria play a mediating role in tirzepatide-related bone loss, then probiotic supplementation might offer real protective value, not just for digestive comfort, but for skeletal preservation.
The diabetes paradox and why it matters for tirzepatide users
Here is something most tirzepatide guides completely miss. The relationship between diabetes, bone density, and fracture risk is paradoxical, and understanding this paradox is critical for anyone using tirzepatide.
People with type 2 diabetes often have normal or even elevated bone mineral density on DEXA scans. By standard diagnostic criteria, their bones look fine. Sometimes better than fine. Yet they experience 40 to 70% higher fracture risk compared to people without diabetes.
How is that possible?
Chronic hyperglycemia creates advanced glycation end products, or AGEs, within bone collagen. These AGEs alter the structural properties of bone, making it more brittle even when mineral density appears adequate. It is like having a concrete wall that looks solid but has internal micro-fractures throughout its structure. The density measurement says strong. The structural reality says fragile.
Microvascular complications from diabetes also impair blood supply to bone tissue. Peripheral neuropathy increases fall risk. These factors mean that a diabetic patient on tirzepatide who sees stable DEXA numbers might still be accumulating fracture risk through mechanisms that density scans cannot detect.
Interestingly, research suggests that GLP-1 receptor agonists affect bone differently depending on diabetes status. One study found that semaglutide and tirzepatide use was associated with greater annualized total hip bone loss in patients without diabetes, while bone loss was comparable between GLP-1 RA users and controls in patients with diabetes. This suggests that in non-diabetic patients, the weight loss itself is the primary driver of bone loss, while in diabetic patients, other metabolic factors may be buffering the effect.
What FRAX scores miss in this population
The Fracture Risk Assessment Tool, known as FRAX, is the standard clinical tool for estimating 10-year fracture probability. But FRAX tends to underestimate fracture risk in patients with type 2 diabetes because it relies partly on BMD values, which may look falsely reassuring in this population.
If you have diabetes and are using tirzepatide, a normal FRAX score does not necessarily mean your bones are safe. The combination of diabetes-related bone quality changes and tirzepatide-induced weight loss may create risk that standard assessment tools cannot fully capture.
Who faces the highest risk
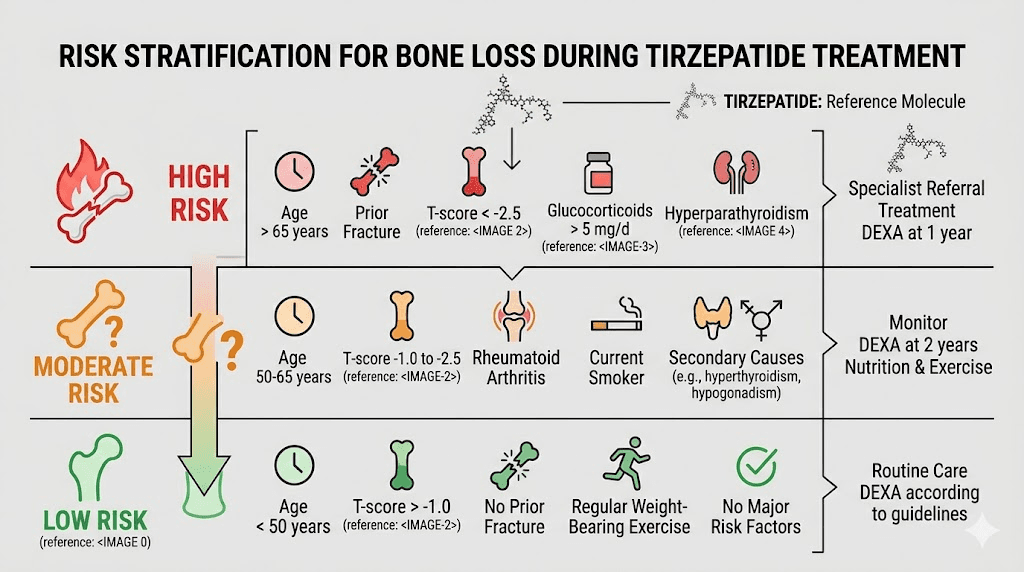
Not everyone on tirzepatide faces the same level of bone risk. Several factors determine where you fall on the spectrum.
High-risk categories
Postmenopausal women. Already experiencing estrogen-driven bone loss. Adding substantial weight loss compounds an existing vulnerability. If you are postmenopausal and losing 15%+ of your body weight on tirzepatide, your skeleton is under significant stress from multiple directions simultaneously.
Adults over 65. Age-related bone loss is already underway. Reduced mechanical loading from tirzepatide weight loss accelerates a process that was already happening. Fall risk also increases with age, making even modest bone density reductions more dangerous.
People with prior fractures. A previous fragility fracture is the single strongest predictor of future fractures. If you have broken a bone from a minor fall or impact, your baseline risk is already elevated. Tirzepatide-related fatigue and potential dizziness can further increase fall risk.
Those with BMI approaching or below 20 after weight loss. Extremely lean individuals have less mechanical stimulus on their bones and less adipose tissue producing protective estrogen. If tirzepatide brings your BMI from 35 down to 22, the net change in skeletal loading is enormous.
People with chronic kidney disease. Impaired kidney function affects vitamin D activation and calcium metabolism. Combined with tirzepatide GI side effects that reduce nutrient absorption, this creates a particularly unfavorable environment for bone health.
Moderate-risk categories
Premenopausal women. Estrogen levels are still protective, but rapid weight loss can still impact bone density. Worth monitoring but less urgent than postmenopausal status.
Men under 70 without other risk factors. Testosterone provides some bone protection, and the mechanical unloading effect is partially offset by typically higher baseline bone density. Still worth tracking if weight loss exceeds 15%.
People on corticosteroids. Long-term glucocorticoid use (more than 3 months at 5 mg prednisone equivalent or higher daily) is an independent risk factor for bone loss. Adding tirzepatide creates additive risk.
Lower-risk categories
Younger adults with adequate nutrition and exercise habits. If you are under 50, maintain good calcium and vitamin D intake, perform regular resistance training, and do not have other risk factors, your bones are better positioned to tolerate the mechanical unloading from weight loss.
People losing moderate amounts of weight. The bone impact appears dose-dependent with weight loss magnitude. Someone losing 8% of body weight faces substantially less skeletal stress than someone losing 22%.
How tirzepatide compares to other GLP-1 medications for bone safety
The TriNetX study that flagged tirzepatide specifically compared it to other GLP-1 receptor agonists. This comparison is important because it suggests the bone risk is not uniform across the medication class.
Tirzepatide is unique. It is a dual GLP-1 and GIP receptor agonist. Other medications like semaglutide and liraglutide target only the GLP-1 receptor. This dual mechanism produces greater weight loss, which likely drives greater mechanical unloading and, consequently, more bone impact.
The GIP receptor component adds another layer of complexity. GIP receptors are found on bone cells, and the interaction between GIP signaling and bone metabolism is not fully understood. Some early research suggests GIP may actually support bone formation in certain contexts, which makes the net effect of tirzepatide on bone biology more nuanced than simple weight-loss-drives-bone-loss models would predict.
An ongoing clinical trial (NCT07094568) is directly comparing tirzepatide to liraglutide for bone outcomes. This head-to-head comparison should provide clearer data on whether the dual receptor mechanism itself contributes to bone risk or whether the difference is purely a function of greater weight loss.
Semaglutide and bone: a reference point
Semaglutide data on bone health is also mixed. GLP-1 receptors are expressed on osteoblasts, the cells that build bone. Some animal studies suggest GLP-1 signaling may actually support bone formation. But in human studies, the weight loss produced by semaglutide still correlates with bone density declines, particularly at the hip. A JAMA Network Open study found that exercise combined with GLP-1 RA treatment helped preserve bone compared to medication alone, highlighting the importance of physical activity as a countermeasure.
For people considering switching from tirzepatide to semaglutide or vice versa for bone health reasons, the evidence does not support one being dramatically safer than the other. The primary driver of bone risk is weight loss magnitude, and both medications can produce substantial weight loss. The 44% higher risk finding for tirzepatide likely reflects its tendency to produce greater weight loss rather than a fundamentally different mechanism of bone damage.
Protecting your bones during tirzepatide treatment
The good news. Bone loss associated with tirzepatide is not inevitable. Multiple evidence-based strategies can mitigate the risk. Some are nutritional. Some are physical. Some involve monitoring. All of them work better when implemented proactively rather than reactively.
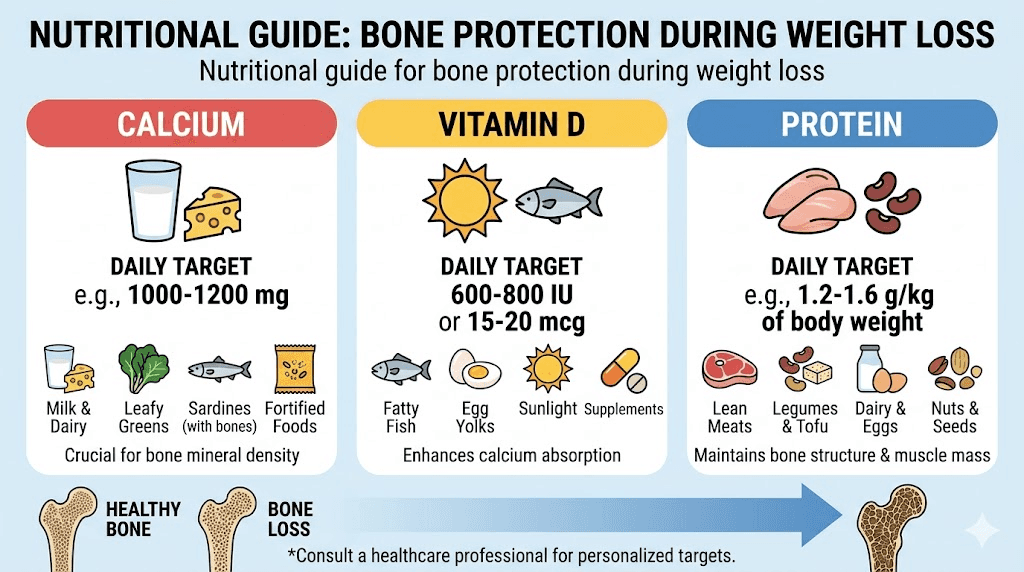
Calcium: the foundation
Your bones need 1,000 to 1,200 mg of calcium daily. Period. This does not change because you are on tirzepatide. If anything, it becomes more important.
Food sources first. Dairy products remain the most bioavailable source. One cup of milk provides roughly 300 mg. One cup of yogurt, about 350 mg. One ounce of cheese, roughly 200 mg. If dairy is not an option, fortified plant milks, sardines with bones, leafy greens like kale and collard greens, and calcium-set tofu are alternatives.
But here is the problem. Tirzepatide dramatically reduces appetite. Many users struggle to eat enough calories, let alone enough calcium-rich foods specifically. If you are eating 1,000 to 1,200 calories per day because your appetite is essentially absent, getting 1,200 mg of calcium from food alone is extremely difficult.
Supplementation fills the gap. Calcium carbonate is best taken with food because it requires stomach acid for absorption. Calcium citrate can be taken with or without food. Split doses of 500 to 600 mg are absorbed better than a single large dose. Take calcium separately from iron supplements, as they compete for absorption.
Vitamin D: the enabler
Without adequate vitamin D, calcium supplementation is largely pointless. Your body needs vitamin D to absorb and utilize calcium from the gut. Target serum 25-hydroxyvitamin D levels of at least 30 ng/mL, though many experts suggest 40 to 60 ng/mL for optimal bone health.
Most people need 1,000 to 2,000 IU of vitamin D3 daily. Some need more, particularly those with darker skin, limited sun exposure, obesity (vitamin D gets sequestered in fat tissue), or malabsorption issues. Testing serum levels is the only way to know if your dose is adequate.
Vitamin D3 (cholecalciferol) is preferred over D2 (ergocalciferol) because D3 raises serum levels more effectively and maintains them longer. Take it with a meal containing fat for best absorption.
Protein: the structural support
Bone is roughly 50% protein by volume. Collagen provides the framework upon which minerals deposit. Without adequate protein, even perfect calcium and vitamin D intake cannot maintain bone integrity.
During weight loss, protein needs actually increase. The minimum target is 1.0 to 1.2 g/kg body weight daily, but many experts recommend 1.2 to 1.6 g/kg during active weight loss to preserve both muscle and bone. For a 180-pound person, that means 82 to 131 grams of protein daily.
This is one of the biggest challenges for tirzepatide users. Appetite suppression makes hitting protein targets feel impossible. Strategies that help include front-loading protein at meals, using protein shakes designed for GLP-1 users, prioritizing protein-dense foods first before anything else on the plate, and using protein bars or collagen powder when solid food feels like too much.
Resistance training: the strongest countermeasure
If there is one single intervention that provides the most bone protection during tirzepatide treatment, it is resistance training. Nothing else comes close.
Resistance exercise directly stimulates osteoblasts through mechanical loading. When you lift weights, the forces transmitted through your skeleton trigger bone remodeling signals that say: strengthen here, add density here, reinforce this structure. This mechanical signal partially replaces the stimulus lost from reduced body weight.
The evidence is clear. The JAMA Network Open study on GLP-1 RA treatment found that exercise combined with medication preserved bone better than medication alone. Resistance training specifically targets the sites most vulnerable to loss during weight loss, the hip and spine.
Minimum effective protocol:
2 to 3 resistance training sessions per week
Focus on compound movements that load the spine and hips: squats, deadlifts, lunges, overhead presses, rows
Progressive overload: gradually increase weight over time
Include weight-bearing cardio: walking, jogging, stair climbing, dancing
What does not count: Swimming and cycling, while excellent for cardiovascular health, do not provide the gravitational and impact forces that stimulate bone remodeling. They are supplements to resistance training, not substitutes.
For people who have not lifted weights before, starting with bodyweight exercises and progressing to light weights is perfectly appropriate. The key is consistency over time, not intensity on any single day. Long-term maintenance of exercise habits matters more than short-term heroics.
Balance and fall prevention
Bone density is only half the equation. The other half is whether you fall. A person with borderline bone density who never falls will never fracture. A person with adequate density who falls regularly is still at risk.
Tirzepatide-related fatigue, dizziness, and dehydration can all increase fall risk. So can rapid weight loss itself, which can affect proprioception as your body adjusts to a different center of gravity.
Balance exercises reduce fall risk by 23 to 30% in older adults. Simple practices like single-leg stands, heel-to-toe walking, yoga, and tai chi can make a meaningful difference. For people on tirzepatide, particularly older adults, incorporating 10 to 15 minutes of balance work 3 to 4 times per week is a low-effort, high-impact investment in fracture prevention.
DEXA scans and bone monitoring
DEXA (dual-energy X-ray absorptiometry) is the gold standard for measuring bone mineral density. It is painless, takes about 10 to 15 minutes, and uses minimal radiation. The results produce a T-score that tells you where your bone density falls relative to a healthy young adult.
T-score interpretation:
0 to -1.0: Normal bone density
-1.0 to -2.5: Osteopenia (low bone density, increased risk)
-2.5 or lower: Osteoporosis
When to get a baseline DEXA
Consider a baseline DEXA scan before starting or early in tirzepatide treatment if you meet any of the following criteria:
Woman over 65 or man over 70
Postmenopausal woman with one or more risk factors
History of fragility fracture at any age
Family history of hip fracture
BMI below 20 or expected to reach below 25 after weight loss
Chronic corticosteroid use (more than 3 months)
Current smoker
Excessive alcohol consumption (more than 3 drinks daily)
Chronic kidney disease
Vitamin D deficiency history
A baseline scan establishes your starting point. Without it, you have no way to know whether your density changes over time are normal age-related shifts or accelerated loss from treatment.
Follow-up frequency
Standard recommendations for DEXA monitoring vary by risk level:
High risk: Every 1 to 2 years
Moderate risk: Every 3 to 5 years
Low risk: Every 10 to 15 years
For tirzepatide users losing significant weight (more than 10% of body weight), many bone health experts suggest more frequent monitoring than standard guidelines recommend. A follow-up scan 12 to 18 months after starting treatment provides the first meaningful comparison point.
Beyond DEXA: bone turnover markers
Blood tests can measure markers of bone turnover that change faster than density measurements. CTX (C-terminal telopeptide) measures bone resorption. P1NP (procollagen type 1 N-terminal propeptide) measures bone formation. Together, they give a dynamic picture of what is happening in your skeleton right now, not what accumulated over the past year.
These markers can detect accelerated bone loss months before it shows up on a DEXA scan. For tirzepatide users concerned about bone health, asking your provider about CTX and P1NP testing, alongside standard vitamin D levels and calcium, can provide early warning signals that allow for intervention before density drops significantly.
The role of medications that affect bone
Tirzepatide does not exist in a vacuum. Many medications that people commonly use alongside it have their own effects on bone metabolism, for better or worse.
Medications that can worsen bone loss
Proton pump inhibitors (PPIs). Commonly used for the acid reflux and GI symptoms that tirzepatide can cause. Long-term PPI use reduces calcium absorption and has been associated with increased fracture risk. If you are taking a PPI alongside tirzepatide, the combined impact on calcium absorption deserves attention.
Corticosteroids. Prednisone, dexamethasone, and other glucocorticoids are potent drivers of bone loss. Even at low doses, long-term use (3+ months) significantly increases fracture risk. If you are on both corticosteroids and tirzepatide, bone monitoring should be aggressive.
Aromatase inhibitors. Used in breast cancer treatment, these dramatically reduce estrogen levels. Combined with estrogen loss from tirzepatide-driven fat reduction, the skeletal impact can be severe.
Certain antidepressants. SSRIs have been associated with modest increases in fracture risk. Given that tirzepatide can affect mood, some users may be prescribed these alongside their weight loss medication.
Medications that may help
Metformin. Generally considered bone-neutral or mildly protective. If you are taking metformin with tirzepatide, it likely has a neutral or slightly positive effect on your skeleton.
Bisphosphonates (alendronate, risedronate, zoledronic acid). The first-line treatment for osteoporosis. If monitoring reveals significant bone loss during tirzepatide treatment, your provider may recommend one of these. They work by inhibiting osteoclasts, slowing bone breakdown.
Denosumab. A biologic medication that also inhibits bone resorption. Used for osteoporosis when bisphosphonates are not tolerated or not effective enough.
Practical protocols for bone protection
Theory is useful. Protocols are actionable. Here are concrete approaches based on risk level.
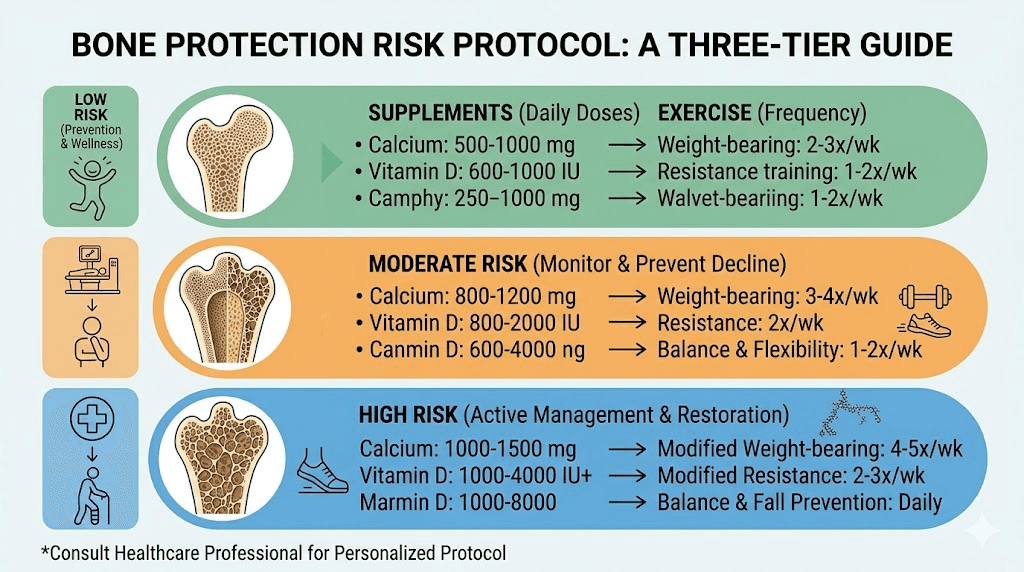
Protocol 1: Low-risk baseline (younger, no risk factors)
Goal: Maintain bone density during tirzepatide treatment
Calcium: 1,000 mg daily (food first, supplement to fill gaps)
Vitamin D: 1,000 to 2,000 IU daily, test levels at 6 months
Protein: 1.2 g/kg body weight daily
Exercise: Resistance training 2x/week, weight-bearing cardio 3x/week
Monitoring: Baseline vitamin D level, consider DEXA if weight loss exceeds 15%
Protocol 2: Moderate-risk (perimenopausal, family history, corticosteroid use)
Goal: Prevent accelerated bone loss, detect early changes
Calcium: 1,200 mg daily (split doses)
Vitamin D: 2,000 to 4,000 IU daily, test levels at baseline and 6 months
Protein: 1.2 to 1.5 g/kg body weight daily
Exercise: Resistance training 3x/week with compound movements, daily walking minimum 30 minutes, balance exercises 3x/week
Monitoring: Baseline DEXA, repeat at 12 to 18 months, bone turnover markers (CTX, P1NP) at baseline and 6 months
Consider: Probiotic supplementation targeting Lachnospiraceae
Protocol 3: High-risk (postmenopausal, prior fracture, T-score below -1.5)
Goal: Aggressive bone preservation, early intervention if deterioration detected
Calcium: 1,200 mg daily (split doses, calcium citrate preferred)
Vitamin D: 2,000 to 5,000 IU daily (physician-guided, based on serum levels)
Protein: 1.5 g/kg body weight daily
Exercise: Resistance training 3x/week (supervised if new to lifting), weight-bearing cardio 5x/week, balance exercises daily
Monitoring: Baseline DEXA and bone turnover markers, repeat DEXA at 12 months, bone markers every 6 months
Discussion with provider: Consider whether tirzepatide dose optimization (lower doses producing moderate weight loss) is appropriate, evaluate need for bisphosphonate or denosumab if rapid bone loss detected
Additional: Ensure adequate magnesium, vitamin K2, and boron intake, which all support bone metabolism
SeekPeptides provides members with detailed bone health monitoring protocols, supplement dosing guides, and exercise templates designed specifically for people using GLP-1 and GIP-based medications. These resources go beyond generic recommendations to provide weight-based, risk-adjusted strategies that account for the unique challenges of maintaining bone health during aggressive weight loss.
Gut health and bone: the emerging connection
The discovery that tirzepatide alters gut microbiota in ways that affect bone metabolism opens an entirely new avenue for prevention. This is cutting-edge research, but the early findings are compelling enough to inform practical action.
The gut-bone axis, as researchers call it, works through several mechanisms. Short-chain fatty acids produced by gut bacteria influence calcium absorption, modulate immune cells that regulate bone remodeling, and affect hormone signaling that controls osteoblast and osteoclast activity.
Tirzepatide reduces Lachnospiraceae abundance. This bacterial family produces butyrate, a short-chain fatty acid with documented bone-protective properties. The reduction in Lachnospiraceae after tirzepatide treatment may partially explain why bone loss occurs even when nutritional intake appears adequate.
What you can do about it
Dietary fiber. Lachnospiraceae and other beneficial gut bacteria thrive on dietary fiber. Increasing fiber intake through vegetables, legumes, whole grains, and fermented foods supports a microbial environment favorable to bone health. For GLP-1 users struggling with appetite, fiber supplements like psyllium husk or partially hydrolyzed guar gum can help, though whole food sources are preferable.
Probiotic supplementation. While specific Lachnospiraceae-targeted probiotics are not yet commercially available, broad-spectrum probiotics containing Lactobacillus and Bifidobacterium species have shown modest bone benefits in clinical studies. Look for products with multiple strains and at least 10 billion CFU.
Fermented foods. Yogurt, kefir, sauerkraut, kimchi, and other fermented foods provide both live bacteria and the substrates they need to flourish. Regular consumption supports microbial diversity. Kefir specifically has shown bone-protective effects in some studies, making it a particularly strategic choice for tirzepatide meal planning.
Prebiotic foods. Garlic, onions, leeks, asparagus, and bananas contain prebiotic compounds that feed beneficial gut bacteria. Including these in your diet, even in small quantities, supports the microbial ecosystem that appears to protect bone health.
Tirzepatide dose and bone risk: is lower better for bones?
The bone risk from tirzepatide appears to correlate primarily with weight loss magnitude rather than medication dose directly. But since higher doses produce greater weight loss, there is an indirect relationship worth examining.
Tirzepatide is available in three primary dose levels: 5 mg, 10 mg, and 15 mg. The standard titration starts at 2.5 mg and increases gradually. Weight loss increases with dose. In the SURMOUNT-1 trial, average weight loss was approximately 15% at 5 mg, 19.5% at 10 mg, and 20.9% at 15 mg.
For bone health specifically, the question becomes: is moderate weight loss at a lower dose substantially safer for bones than aggressive weight loss at a higher dose?
The evidence suggests yes, though the relationship is not perfectly linear. A 10% weight loss produces less mechanical unloading than a 20% loss. Less weight lost means less hormonal disruption, less nutritional stress, and potentially less gut microbiome disruption. For people with bone risk factors, discussing with their provider whether a moderate dose achieving 10 to 15% weight loss might be preferable to maximum dose achieving 20%+ could be a bone-protective strategy.
The concept of microdosing tirzepatide takes this even further. Some practitioners advocate for lower doses that produce more gradual weight loss, giving the skeleton time to adapt to changing loads. While the evidence base for this specific approach is limited, the physiological logic is sound. Bones adapt better to gradual change than sudden shifts.
Signs your bones may be struggling
Some warning signs suggest that bone health may be deteriorating during tirzepatide treatment. None of these are definitive on their own, but any of them should prompt a conversation with your healthcare provider about bone assessment.
Unexplained bone pain. Persistent aching in the spine, hips, or long bones that is not explained by muscle pain from exercise or other causes.
Height loss. Losing more than half an inch in height can indicate vertebral compression fractures, which sometimes occur silently (without acute pain).
Fractures from minimal trauma. Breaking a bone from a fall from standing height or less, or from an impact that would not normally cause a fracture.
Nail brittleness and dental issues. While not specific to bone loss, these can indicate broader calcium and mineral metabolism problems that also affect bone health.
Severe or persistent GI symptoms. Chronic nausea, vomiting, or diarrhea that limits food intake for extended periods increases the risk of nutritional deficiencies that affect bone. If your GI symptoms are severe enough that you are eating very little for weeks at a time, your bones are not getting what they need.
Muscle weakness or wasting. Bone and muscle health are closely linked. If you are losing significant muscle mass alongside weight, bone loss may be occurring simultaneously. Muscle preservation strategies also support bone preservation.
What the ongoing research tells us
The field is evolving rapidly. Several ongoing studies will provide better answers in the coming years.
NCT07094568 is a clinical trial directly comparing tirzepatide to liraglutide for bone outcomes. This will be the first head-to-head comparison between a dual agonist and a pure GLP-1 RA specifically looking at bone health as a primary endpoint.
Researchers are also investigating whether the GIP receptor component of tirzepatide has independent effects on bone, separate from weight loss. GIP receptors on bone cells could theoretically provide some protective signaling, but the net effect appears to be overwhelmed by the bone-depleting mechanisms of rapid weight loss.
The gut microbiome research is particularly promising. If Lachnospiraceae transplantation or targeted probiotics can mitigate tirzepatide-related bone loss in humans (as they did in mice), this would represent a relatively simple, low-risk intervention that could be widely implemented.
Long-term follow-up studies of tirzepatide users, extending beyond the 72-week trials, will also be critical. Bone loss from weight loss medications may plateau after initial weight stabilization, or it may continue. The trajectory matters enormously for long-term fracture risk prediction.
What your doctor may not tell you
Most prescribing providers are focused on weight loss outcomes and metabolic parameters. They check your HbA1c, your weight, your blood pressure, maybe your liver enzymes. Very few proactively discuss bone health before starting tirzepatide.
This is not negligence. The FDA labeling does not flag bone risk. The clinical guidelines do not currently mandate bone monitoring for tirzepatide users. And in a 15-minute appointment, there are only so many topics a physician can cover.
But the real-world evidence is now strong enough that an informed patient should raise the topic themselves. Asking about baseline DEXA screening, vitamin D testing, and calcium supplementation before starting tirzepatide is reasonable and appropriate. Asking about bone turnover markers after 6 months of treatment is proactive but not excessive.
For researchers serious about optimizing every aspect of their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, monitoring frameworks, and a community of thousands who have navigated these exact questions. Understanding how to protect your bone health while achieving weight loss goals is exactly the kind of nuanced, multi-factor challenge that SeekPeptides helps members navigate.
Putting it all together: a decision framework
Should you be worried about bone loss on tirzepatide? That depends on your individual risk profile. Here is a framework for thinking about it.
If you have no bone risk factors, are under 50, exercise regularly, and eat well: The bone risk is real but manageable. Ensure adequate calcium, vitamin D, and protein. Maintain resistance training. Consider a baseline DEXA if weight loss exceeds 15%. Do not panic, but do not ignore it either.
If you have one or two moderate risk factors (perimenopausal, family history, sedentary): Proactive monitoring is warranted. Get a baseline DEXA. Supplement calcium and vitamin D. Commit to resistance training at least twice per week. Ask your provider about bone turnover markers at 6 months. These steps significantly reduce your risk.
If you are high risk (postmenopausal, prior fracture, corticosteroid use, low BMD): Bone health should be a primary consideration in your treatment plan, not an afterthought. Baseline DEXA is essential. Supplement aggressively. Resistance train consistently. Discuss dose optimization with your provider. Consider bone-protective medications if monitoring shows deterioration. Have your provider evaluate whether the benefits of aggressive weight loss outweigh the bone risks in your specific case.
The answer to "does tirzepatide cause bone loss" is not a clean yes or no. The more accurate answer is: tirzepatide produces weight loss that can cause bone loss, and tirzepatide may also affect bone through gut microbiome changes. But these effects are modifiable. With proper nutrition, exercise, monitoring, and medical guidance, most people can use tirzepatide effectively while keeping their bones strong.
Frequently asked questions
Does tirzepatide directly damage bone tissue?
No direct bone toxicity has been demonstrated. The primary pathway of bone impact is indirect, through the weight loss tirzepatide produces and the downstream effects of reduced mechanical loading, nutritional changes, and hormonal shifts. Animal research also implicates gut microbiome changes as an additional mechanism. But tirzepatide does not appear to be directly toxic to bone cells.
Should I get a DEXA scan before starting tirzepatide?
If you have any risk factors for osteoporosis, yes. A baseline DEXA establishes your starting bone density and gives you a reference point for monitoring changes over time. For women over 65, men over 70, anyone with prior fractures, or those with family history of osteoporosis, a baseline scan is strongly recommended. For younger, lower-risk individuals, a baseline scan is optional but still informative, especially if significant weight loss is expected.
Can resistance training completely prevent bone loss from tirzepatide?
Resistance training is the most effective countermeasure, but it may not completely prevent all bone density changes, particularly in high-risk individuals losing substantial weight. However, it significantly reduces the magnitude of loss and can help maintain bone density at critical fracture sites like the hip and spine. Combined with adequate nutrition and supplementation, resistance training makes bone loss manageable rather than inevitable.
How much calcium do I need while on tirzepatide?
1,000 to 1,200 mg daily, ideally from a combination of dietary sources and supplements. Split supplement doses into 500 to 600 mg servings for better absorption. Pair with adequate vitamin D (1,000 to 2,000 IU daily minimum) because calcium without vitamin D is poorly absorbed.
Does the bone loss from tirzepatide reverse when I stop the medication?
Some bone density recovery can occur after weight stabilization, as the skeleton readjusts to a new steady-state mechanical load. However, the recovery is typically partial, not complete, and depends on age, hormonal status, and ongoing nutrition and exercise habits. Younger individuals with good nutrition and active lifestyles tend to recover more density than older individuals with multiple risk factors. For guidance on stopping tirzepatide safely, consult your healthcare provider.
Is semaglutide safer for bones than tirzepatide?
The evidence does not conclusively show that semaglutide is dramatically safer for bones. The TriNetX study showed tirzepatide had higher bone risk than other GLP-1 RAs, but this likely reflects greater weight loss rather than a fundamentally different mechanism. Semaglutide also produces weight-loss-related bone density changes. The key factor is magnitude of weight loss, not the specific medication used.
Should I take a probiotic for bone health while on tirzepatide?
Emerging research suggests it may help. Tirzepatide reduces Lachnospiraceae bacteria that produce bone-protective short-chain fatty acids. While Lachnospiraceae-specific probiotics are not yet available commercially, broad-spectrum probiotics with Lactobacillus and Bifidobacterium strains, combined with adequate dietary fiber and fermented foods, can support overall gut health and may provide some bone protection.
Do I need to worry about bone loss if I am only losing a small amount of weight?
The bone risk appears to scale with weight loss magnitude. Losing 5 to 8% of body weight produces significantly less skeletal stress than losing 20%+. For people on lower tirzepatide doses achieving moderate weight loss, the bone risk is lower, though not zero. Basic bone health practices (calcium, vitamin D, exercise) are still recommended regardless of weight loss amount.
External resources
NIH Osteoporosis and Related Bone Diseases National Resource Center
ClinicalTrials.gov: Tirzepatide vs Liraglutide Bone Study (NCT07094568)
PMC: Tirzepatide Promotes Bone Loss via Gut Microbial Metabolites
In case I do not see you, good afternoon, good evening, and good night. May your bones stay dense, your protocols stay informed, and your results stay lasting.