Mar 20, 2026

Tired of conflicting answers about tirzepatide and blood work? One forum says it shows up on a standard panel. Another says it does not. Your healthcare provider might not even know what to look for, and if you are using compounded tirzepatide, the confusion only deepens. Here is the truth. Standard blood tests do not detect tirzepatide directly. Not a basic metabolic panel. Not a comprehensive metabolic panel. Not even a standard drug screen.
The molecule simply is not on the list of things those tests look for.
But that does not mean your blood work will look the same as it did before you started. Tirzepatide changes things. It shifts your glucose levels, alters your lipid profile, affects your liver enzymes, and can even influence your thyroid markers. A trained clinician looking at your labs might not see tirzepatide itself, but they will see its fingerprints everywhere.
This guide covers everything: which tests can and cannot detect tirzepatide, what blood markers change when you use it, how long it stays in your system, which labs you need before starting, the monitoring schedule that keeps you safe, and what to tell your doctor before any blood draw. Whether you are on branded Mounjaro or Zepbound or a compounded version, the science applies equally. SeekPeptides researchers have compiled the most comprehensive breakdown available, pulling from clinical trial data, pharmacokinetic studies, and real-world monitoring protocols.
The short answer: tirzepatide and standard blood tests
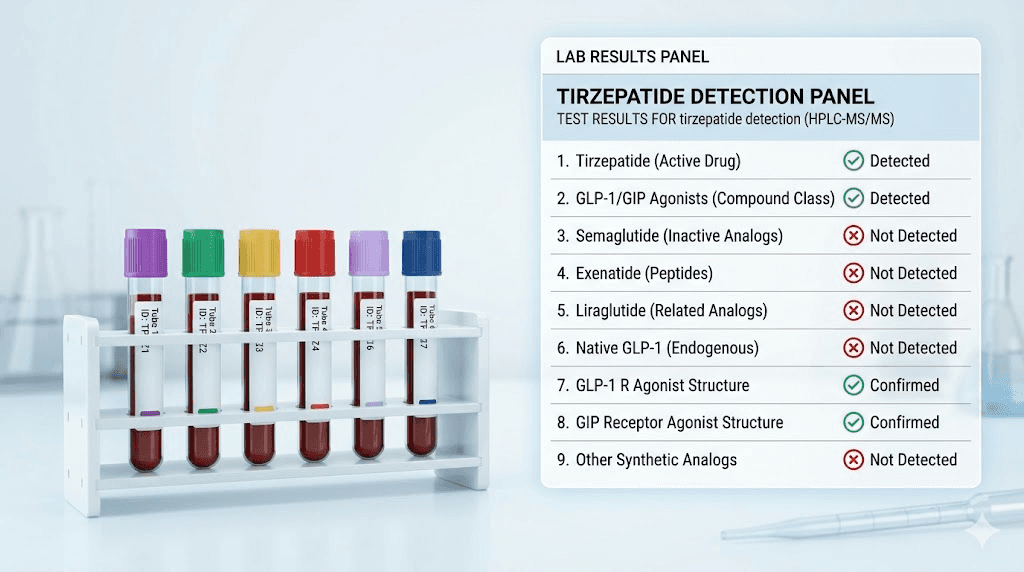
No. Tirzepatide does not show up on standard blood tests.
Standard blood panels, including the basic metabolic panel (BMP), comprehensive metabolic panel (CMP), complete blood count (CBC), and lipid panels, are not designed to detect peptide-based medications. They measure electrolytes, blood sugar, kidney markers, liver enzymes, cholesterol, and blood cell counts. Tirzepatide is not on that list. Neither is semaglutide, liraglutide, or any other GLP-1 receptor agonist.
Think of it this way. A standard blood panel is like a security checkpoint scanning for specific items on a known list. Tirzepatide is not on that list. The scanner does not recognize it, does not flag it, and does not report it. You could have therapeutic levels of tirzepatide circulating in your blood during the draw, and the results would say nothing about it.
This applies to every standard test your primary care doctor, endocrinologist, or clinic might order. The medication works quietly in the background, activating GIP and GLP-1 receptors, slowing gastric emptying, enhancing insulin secretion, and suppressing glucagon. All of those effects produce measurable changes in your blood chemistry. But the molecule itself remains invisible to routine testing.
There is one important caveat. While standard blood tests do not detect tirzepatide, specialized laboratory techniques can. More on that in a later section. For now, the key takeaway is simple: your routine blood work will not reveal whether you are taking tirzepatide.
How tirzepatide works at the blood level
Understanding why tirzepatide affects blood work requires understanding what it does once injected. Tirzepatide is a dual GIP/GLP-1 receptor agonist. That dual mechanism is what separates it from semaglutide and other single-action GLP-1 medications. It activates two different incretin pathways simultaneously, and each pathway produces distinct, measurable changes in blood chemistry.
The GLP-1 pathway handles the familiar effects. Increased insulin secretion when blood sugar rises. Suppressed glucagon release. Delayed gastric emptying, which is why food choices matter on this medication. These actions directly lower fasting glucose and postprandial blood sugar spikes, both of which show up clearly on metabolic panels.
The GIP pathway adds something different. Glucose-dependent insulinotropic polypeptide enhances the insulin response beyond what GLP-1 alone achieves. It also influences lipid metabolism, which is why tirzepatide users often see dramatic improvements in their cholesterol and triglyceride numbers. Research from the SURPASS clinical trials demonstrated triglyceride reductions of 19-25% and improvements in HDL cholesterol that single-action GLP-1 drugs struggle to match.
Beyond glucose and lipids, tirzepatide affects appetite regulation through hypothalamic signaling. This reduces caloric intake, drives weight loss, and triggers a cascade of metabolic improvements. Your body starts burning stored fat more efficiently. Inflammatory markers decrease. Insulin sensitivity improves. All of these changes leave traces in blood work, even though the medication itself remains undetectable.
The SURPASS trials showed reductions in HbA1c of up to 2.5% and weight loss of approximately 15-21% of body weight. Those are massive changes that fundamentally alter blood chemistry. A healthcare provider looking at sequential lab results would see dramatic shifts in multiple markers, even without knowing what caused them.
Blood markers tirzepatide actually changes
While tirzepatide itself stays invisible on standard tests, its effects are anything but invisible. Here is exactly what changes and by how much, based on clinical trial data and real-world monitoring.
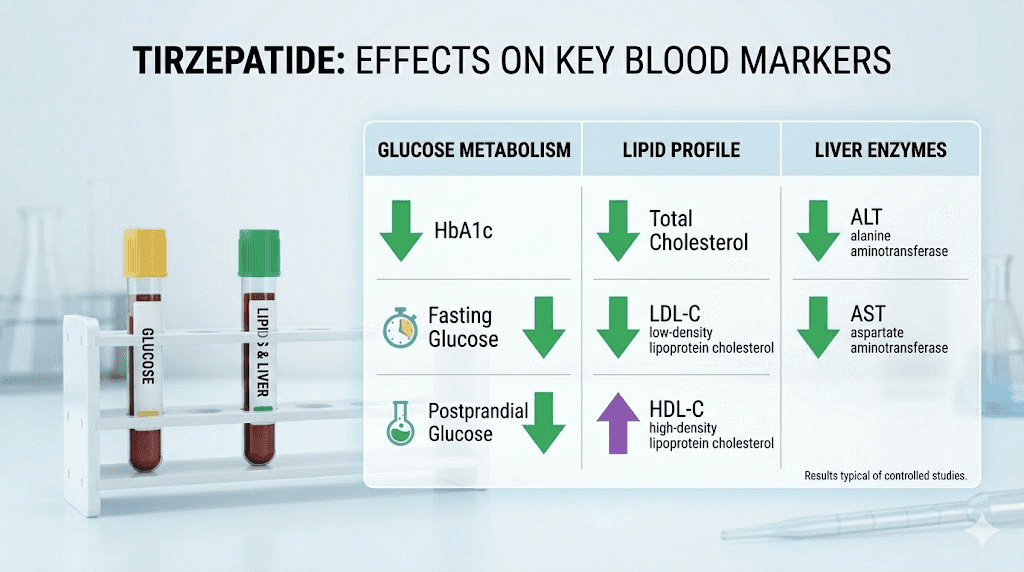
Blood glucose and HbA1c
This is the most dramatic shift. Fasting blood glucose drops significantly within the first few weeks of treatment. In the SURPASS-1 trial, patients on 15 mg tirzepatide saw fasting glucose reductions averaging 60-70 mg/dL. HbA1c, which reflects average blood sugar over 2-3 months, dropped by up to 2.5 percentage points. If your baseline A1c was 8.5%, it could drop to 6.0% or lower.
For researchers monitoring their progress, this is one of the clearest indicators that tirzepatide is working. A1c changes typically become apparent after 8-12 weeks on a stable dose. If you have been on tirzepatide for three months and your A1c has not moved, that is a signal something needs adjustment, whether it is your dose, timing, or dietary factors.
Lipid panel changes
Tirzepatide produces meaningful improvements in cholesterol. Total cholesterol decreases. LDL cholesterol drops modestly. But the real standout is triglycerides. The SURPASS trials showed triglyceride reductions of 19-25%, which is comparable to what some dedicated lipid-lowering medications achieve. HDL cholesterol, the protective kind, tends to remain stable or increase slightly.
These changes have implications for cardiovascular risk assessment. If your doctor evaluates your heart disease risk based on lipid panels, the improvements from tirzepatide could shift your risk category. This is one of the reasons proper dosing matters, as higher doses tend to produce more significant lipid improvements.
Insulin and C-peptide
Fasting insulin levels typically decrease on tirzepatide, reflecting improved insulin sensitivity. C-peptide, a marker of how much insulin your pancreas produces, may also shift. These changes indicate that your body is using insulin more efficiently rather than overproducing it to compensate for resistance. If your doctor orders an insulin panel, expect lower fasting insulin and improved HOMA-IR scores.
Inflammatory markers
C-reactive protein (CRP) and other inflammatory markers tend to decrease with tirzepatide use, largely driven by weight loss and improved metabolic function. This is relevant for researchers tracking systemic inflammation, particularly those dealing with conditions like autoimmune inflammation or lipedema.
Liver enzymes (ALT, AST)
This one surprises many people. Tirzepatide often improves liver enzyme levels. ALT and AST, markers of liver cell damage, frequently decrease during treatment. Research suggests GLP-1 receptor agonists reduce hepatic fat accumulation, and weight loss itself reduces the metabolic burden on the liver. Studies show maximum ALT improvement around 30 weeks of treatment.
However, in rare cases, liver enzymes can elevate. If your ALT or AST spike significantly after starting tirzepatide, that warrants immediate medical attention. It does not necessarily mean the medication caused it, but it needs investigation.
Kidney markers
Creatinine and estimated glomerular filtration rate (eGFR) require careful interpretation during tirzepatide treatment. Here is why: as you lose weight, you lose muscle mass along with fat. Creatinine is a byproduct of muscle metabolism, so less muscle means lower creatinine. Lower creatinine artificially inflates eGFR, making kidney function appear better than it actually is. Do not rely solely on creatinine-based eGFR during rapid weight loss. Cystatin C-based eGFR provides a more accurate picture because it is not affected by muscle mass changes.
Dehydration is another concern. Gastrointestinal side effects like nausea, vomiting, and diarrhea can cause fluid loss, which temporarily elevates creatinine and makes kidney function look worse than it is. If your kidney markers look off, consider whether hydration status is a factor before panicking.
Specialized detection: how tirzepatide can be found
Standard blood tests cannot find tirzepatide. But specialized laboratory techniques absolutely can.
The gold standard for tirzepatide detection is Liquid Chromatography-Mass Spectrometry/Mass Spectrometry, known as LC-MS/MS. This technique separates the components of a blood sample by their chemical properties and then identifies specific molecules by their mass-to-charge ratio. It can detect tirzepatide at extremely low concentrations, down to the nanogram-per-milliliter range.
Immunoassays represent another detection method. These tests use antibodies specifically designed to bind to tirzepatide. When the antibody-tirzepatide complex forms, it generates a measurable signal. Enzyme-linked immunosorbent assays (ELISA) and radioimmunoassays (RIA) fall into this category.
Who uses these tests? Almost exclusively researchers in clinical trial settings, pharmacokinetic studies, and specialized research environments. These tests are not available through Quest Diagnostics, LabCorp, or any standard commercial laboratory. They require specialized reagents, calibrated equipment, and trained technicians. The cost per test can range from hundreds to thousands of dollars.
For practical purposes, if you are wondering whether your doctor, employer, or insurance company can detect tirzepatide in your blood, the answer is no. Not through any test they would typically order. The technology exists, but it is not deployed in routine clinical or occupational settings.
One emerging exception: the GLP-1 Test Panel from specialized labs like Request A Test. This panel specifically measures GLP-1 levels and can indicate whether exogenous GLP-1 receptor agonist activity is present. It does not identify tirzepatide specifically, but elevated GLP-1 activity in someone not prescribed these medications could raise questions. This test remains uncommon and is not part of standard screening protocols.
How long tirzepatide stays in your system
Understanding tirzepatide pharmacokinetics matters for blood work timing. The medication does not clear your system overnight, and its lingering presence continues to influence blood markers long after your last injection.
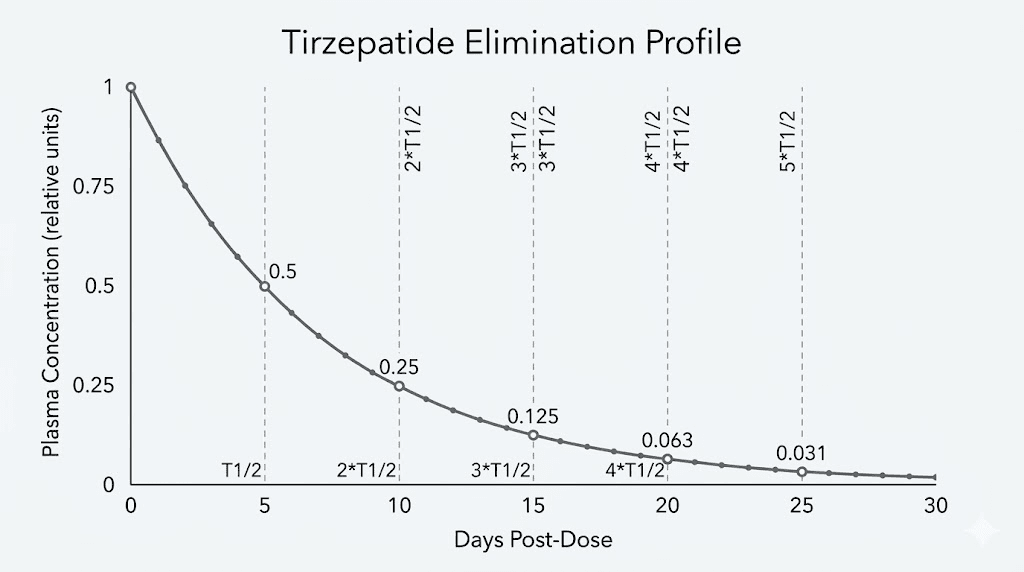
Tirzepatide has an elimination half-life of approximately 5 days. That means every 5 days, the concentration in your bloodstream drops by half. After one half-life, 50% remains. After two half-lives (10 days), 25% remains. After three half-lives (15 days), 12.5% remains. After four half-lives (20 days), roughly 6% remains. After five half-lives (25 days), less than 3% of the original dose circulates.
Complete elimination, defined as less than 3% remaining, takes approximately 25-30 days after the last injection. However, metabolic effects persist beyond drug clearance. Your improved insulin sensitivity, altered lipid metabolism, and reduced appetite do not snap back to baseline the moment the molecule clears.
Peak plasma concentration
After a subcutaneous injection, tirzepatide reaches peak blood levels (Tmax) somewhere between 8 and 72 hours. Individual variation is significant here. Injection site, body composition, and dose all influence absorption speed. Whether you inject in your stomach, thigh, or arm affects how quickly the peptide enters circulation.
Steady-state concentration, the point where weekly doses produce consistent blood levels, takes approximately 4 weeks of regular dosing. This is why the standard titration protocol starts at 2.5 mg and increases every 4 weeks. Each dose level needs about a month to reach equilibrium.
Elimination pathways
Your body eliminates tirzepatide through two primary routes. Approximately 66% is excreted via urine as metabolites. The remaining 33% exits through feces. The medication undergoes proteolytic degradation, meaning enzymes break it down into smaller peptide fragments and amino acids before elimination.
The extended half-life is not natural for a peptide this size. It results from deliberate structural modifications, specifically a C20 fatty diacid moiety that promotes albumin binding. When tirzepatide binds to albumin in your blood, it is shielded from enzymatic breakdown and renal filtration. This albumin binding is what allows once-weekly dosing instead of the multiple daily injections that unmodified peptides would require.
For blood work purposes, this matters because tirzepatide is actively influencing your blood chemistry for the entire week between injections. There is no "window" where the medication has worn off and your blood work reflects your unmedicated baseline. If you want truly unmedicated lab values, you would need to stop tirzepatide for at least 4-5 weeks, and even then, some metabolic effects might persist.
Tirzepatide and drug testing
Will tirzepatide show up on a drug test? No.
Standard employment drug screens test for specific categories of controlled substances: amphetamines, barbiturates, benzodiazepines, cannabinoids, cocaine, opioids, phencyclidine, and sometimes expanded panels including synthetic opioids and designer drugs. Tirzepatide is not a controlled substance. It is not a drug of abuse. It is not on any standard drug screening panel.
A 5-panel, 10-panel, or even 12-panel drug test will not detect tirzepatide. Neither will specialized panels used by the Department of Transportation, military, or federal agencies. The immunoassay and gas chromatography-mass spectrometry methods used in workplace drug testing are calibrated for entirely different molecular targets.
Athletic drug testing is a different conversation. The World Anti-Doping Agency (WADA) does not currently list GLP-1 receptor agonists as prohibited substances. However, anti-doping regulations evolve, and metabolic modulators are an area of ongoing regulatory attention. Athletes subject to anti-doping testing should check current prohibited substance lists before using tirzepatide or any similar medication.
Insurance-related blood work is where things get nuanced. Insurance companies do not test for tirzepatide. But they do look at metabolic markers. If you apply for life insurance and your blood work shows dramatically improved glucose, A1c, and lipid levels compared to previous records, an underwriter might ask questions. This is not detection of the drug. It is observation of suspicious improvements that could indicate undisclosed medication use.
Essential blood work before starting tirzepatide
Getting baseline labs before your first injection is not optional. It is essential. Without baseline values, you have no reference point to assess whether tirzepatide is helping, hurting, or doing nothing. Every reputable tirzepatide protocol should begin with comprehensive lab work.
The baseline panel
Here is the complete list of tests to request before starting tirzepatide:
Metabolic markers:
Comprehensive metabolic panel (CMP), which includes glucose, electrolytes, BUN, creatinine, calcium, and liver enzymes
Hemoglobin A1c (HbA1c), your 2-3 month blood sugar average
Fasting insulin and fasting glucose
HOMA-IR calculation (derived from fasting insulin and glucose)
Lipid panel:
Total cholesterol
LDL cholesterol (direct measurement preferred)
HDL cholesterol
Triglycerides
VLDL cholesterol
Liver function:
ALT (alanine aminotransferase)
AST (aspartate aminotransferase)
Alkaline phosphatase (ALP)
Total bilirubin
GGT (gamma-glutamyl transferase), optional but useful
Kidney function:
Serum creatinine
Blood urea nitrogen (BUN)
Estimated glomerular filtration rate (eGFR)
Cystatin C (recommended for more accurate kidney assessment)
Urine albumin-to-creatinine ratio (UACR), especially if diabetic
Thyroid function:
TSH (thyroid stimulating hormone)
Free T4
Free T3 (optional but informative)
Thyroid antibodies if history of thyroid disease
Complete blood count (CBC):
White blood cells, red blood cells, hemoglobin, hematocrit, platelets
Differential (neutrophils, lymphocytes, monocytes, eosinophils, basophils)
Additional recommended tests:
Vitamin B12 (especially if taking tirzepatide with B12)
Vitamin D
Iron panel (ferritin, TIBC, serum iron)
Magnesium
Pancreatic enzymes (lipase, amylase), baseline reference
This might seem like an extensive list. It is. But each test serves a purpose. Tirzepatide affects multiple organ systems, and having baseline values for all of them ensures you can identify positive changes and catch potential problems early. The cost of comprehensive baseline labs is minimal compared to the cost of missing an early warning sign.
The complete monitoring schedule
Once you have baseline labs, you need a monitoring schedule. How often depends on your health status, dose escalation timeline, and individual risk factors. Here is the evidence-based approach.
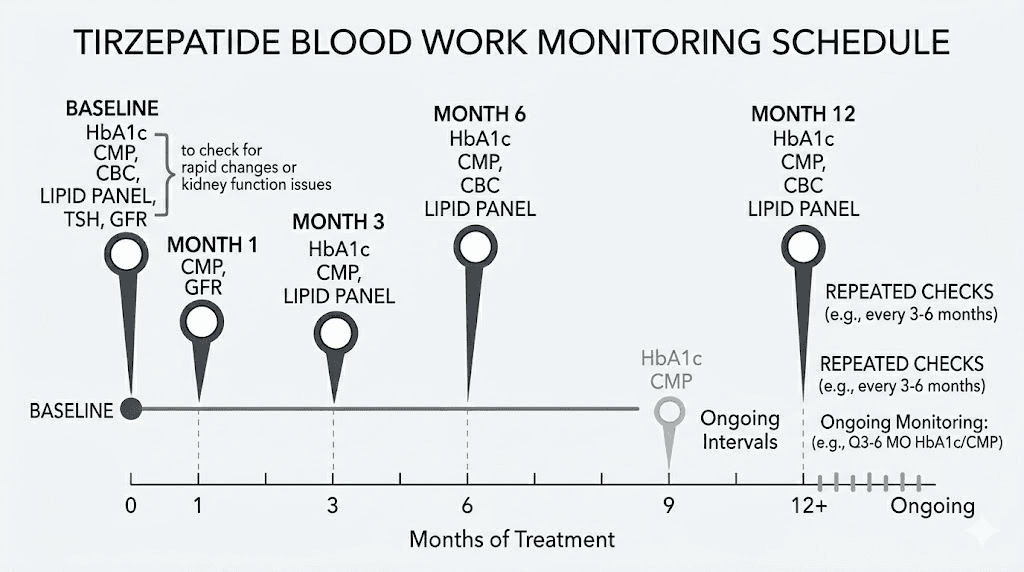
Month 1: initial assessment
Run a basic metabolic panel and fasting glucose at 4 weeks, just before your first dose escalation from 2.5 mg to 5 mg. This catches any acute metabolic changes and confirms you are tolerating the medication. Check kidney function markers if you experienced significant gastrointestinal side effects that could cause dehydration.
Month 3: comprehensive check
At 12 weeks, order the full panel again. CMP, A1c, lipids, liver function, kidney function, CBC. This is your first opportunity to assess meaningful trends. A1c reflects the previous 2-3 months, so this draw captures your early treatment period. Compare everything to baseline. You should see glucose and A1c improving, lipids trending better, and liver enzymes stable or decreasing.
Month 6: progress evaluation
Repeat the comprehensive panel. By now, you are likely on a stable maintenance dose, whether that is a microdose, the standard 10-15 mg, or somewhere in between. This draw tells you whether the metabolic improvements are sustaining. Add thyroid function (TSH, Free T4) if not checked since baseline. Include vitamin levels (B12, D, iron) to catch any nutritional deficiencies from reduced food intake.
Ongoing: every 3-6 months
Once stable on a maintenance dose with no concerning trends, comprehensive labs every 6 months is reasonable for most people. Those with pre-existing kidney disease, liver conditions, thyroid disorders, or diabetes should maintain the 3-month schedule. Anyone experiencing new side effects should get relevant labs checked promptly rather than waiting for the next scheduled draw.
Timepoint | Tests | Purpose |
|---|---|---|
Baseline (before starting) | Full panel: CMP, A1c, lipids, liver, kidney, thyroid, CBC, vitamins | Establish reference values |
4 weeks | BMP, fasting glucose, kidney markers if GI issues | Catch acute changes before dose increase |
12 weeks | Full panel: CMP, A1c, lipids, liver, kidney, CBC | First comprehensive trend assessment |
24 weeks | Full panel plus thyroid, vitamins, nutritional markers | Progress evaluation and nutritional status |
Every 6 months after | CMP, A1c, lipids, liver, kidney, thyroid annually | Ongoing monitoring |
Liver function and tirzepatide: what to watch
The liver deserves its own section because tirzepatide has a complex relationship with liver health. For most people, tirzepatide improves liver function. For a small number, it can cause problems.
The good news first. GLP-1 receptor agonists, including tirzepatide, reduce hepatic fat accumulation. This is significant for the millions of people with non-alcoholic fatty liver disease (NAFLD) or its more serious form, non-alcoholic steatohepatitis (NASH). Weight loss driven by tirzepatide compounds this benefit, as even modest weight loss reduces liver fat. Studies show maximum ALT improvement typically occurs around 30 weeks of treatment.
The concerning scenario is rare but real. Acute liver injury has been reported with GLP-1 receptor agonists, though establishing direct causation is difficult because many users have pre-existing liver conditions that could independently cause enzyme elevations. If your ALT or AST rise above 3 times the upper limit of normal after starting tirzepatide, that requires immediate evaluation. Stop the medication and consult your healthcare provider.
For researchers tracking liver health, here are the reference ranges to know:
ALT: Normal is 7-56 U/L for most labs. Mild elevation (1-3x upper limit) is common during the first few weeks and often self-resolves.
AST: Normal is 10-40 U/L. Follows similar patterns to ALT.
ALP: Normal is 44-147 U/L. Elevation suggests biliary rather than hepatocellular issues.
GGT: Normal is 0-45 U/L for women, 0-65 U/L for men. Particularly useful for distinguishing liver from bone sources of ALP elevation.
If you consume alcohol while on tirzepatide, monitoring liver enzymes becomes even more important. Alcohol and reduced food intake together create additional hepatic stress that tirzepatide alone would not cause. Consider the type and quantity of alcohol carefully if your liver enzymes are borderline.
Kidney function and tirzepatide: the dehydration factor
Kidney monitoring on tirzepatide is less about the medication harming your kidneys and more about the side effects creating conditions that stress them. Tirzepatide itself has not been shown to cause direct nephrotoxicity. But the gastrointestinal side effects it produces, particularly nausea, vomiting, and diarrhea, can cause dehydration severe enough to impact kidney function temporarily.
Dehydration reduces blood flow to the kidneys, causing a pre-renal acute kidney injury. Creatinine rises. BUN rises. eGFR drops. These changes are usually reversible with adequate hydration, but they can look alarming on paper. If your kidney markers spike after a particularly rough week of GI side effects, rehydrate aggressively and retest in 1-2 weeks before drawing conclusions.
The weight loss-creatinine paradox is another important consideration. As mentioned earlier, creatinine is produced by muscles. Lose significant muscle mass during weight loss, and creatinine drops. Lower creatinine makes eGFR look higher, creating a false sense of improved kidney function. For accurate kidney assessment during weight loss on tirzepatide, request cystatin C-based eGFR instead of, or in addition to, the standard creatinine-based calculation.
Researchers with pre-existing kidney disease, including those with diabetes-related nephropathy, should follow a tighter monitoring schedule.
Check kidney markers at baseline, 4 weeks, 12 weeks, and every 3 months thereafter. Report any dark urine, dramatically decreased urine output, or lower back pain immediately, as these could signal acute kidney stress that needs prompt intervention.
One positive note: tirzepatide has actually shown renal protective effects in some studies, particularly in patients with type 2 diabetes. Improved blood sugar control reduces the metabolic stress on kidneys over time. The SURPASS trials demonstrated stable or improved kidney function in the tirzepatide groups compared to placebo, suggesting long-term kidney benefits that outweigh the short-term risks from GI-related dehydration.
Thyroid markers and tirzepatide: the calcitonin question
Every tirzepatide label carries a black box warning about medullary thyroid carcinoma (MTC). In rodent studies, GLP-1 receptor agonists caused thyroid C-cell tumors. Whether this translates to human risk remains uncertain, but the warning exists and thyroid monitoring deserves attention.
The key marker is calcitonin. Calcitonin is a hormone produced by thyroid C-cells, and elevated levels can indicate C-cell hyperplasia or medullary thyroid carcinoma. Normal serum calcitonin is below 10 pg/mL in most people. If calcitonin exceeds 50 ng/L (50 pg/mL), further diagnostic workup for MTC is warranted, including thyroid ultrasound and potentially fine-needle aspiration biopsy.
Here is where it gets complicated. Routine calcitonin monitoring for everyone on tirzepatide is not universally recommended. The FDA labeling states that the utility of routine serum calcitonin monitoring or thyroid ultrasound for early detection of MTC in patients treated with tirzepatide is uncertain. Such monitoring may increase the risk of unnecessary procedures, given the low absolute risk of MTC.
That said, specific populations should absolutely monitor thyroid markers. Anyone with a personal or family history of MTC. Anyone with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Anyone with pre-existing thyroid nodules or goiter. For these individuals, baseline calcitonin and thyroid ultrasound before starting tirzepatide, with periodic follow-up, is prudent.
TSH and Free T4 should be checked at baseline and annually for everyone on tirzepatide. Weight loss itself can alter thyroid function, and some autoimmune thyroid conditions can be unmasked or affected by the metabolic changes tirzepatide produces. If you notice new fatigue, cold intolerance, hair thinning, or unexplained weight regain while on a stable dose, thyroid function testing should be a first step.
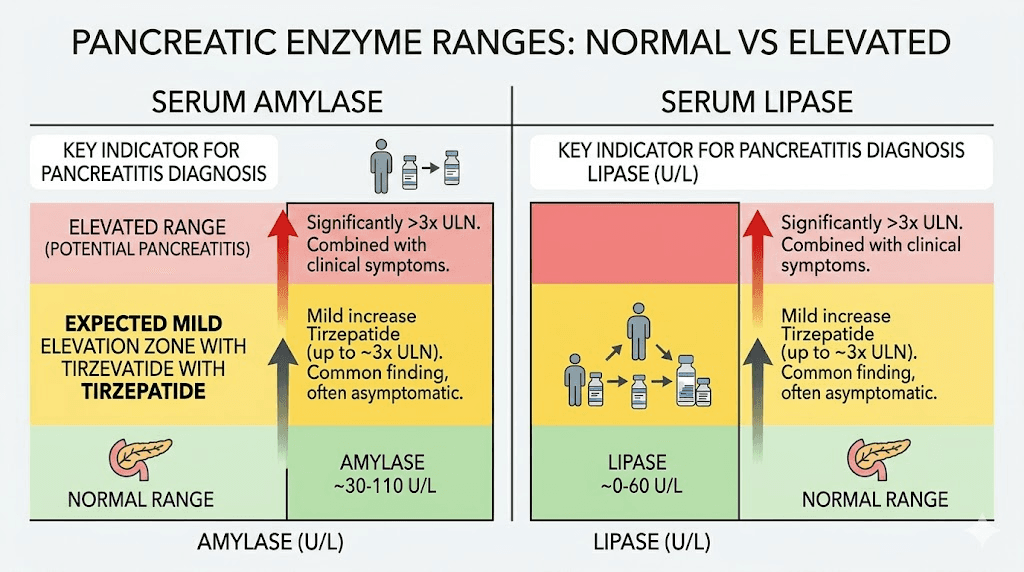
Pancreatic enzymes: amylase and lipase on tirzepatide
This is one of the most misunderstood aspects of tirzepatide blood work. The medication raises pancreatic enzyme levels. That fact alone causes unnecessary panic in many users who see elevated lipase or amylase on their lab results. Context matters enormously here.
Clinical data from the SURPASS trials shows that tirzepatide at 15 mg increases amylase by approximately 23.7% and lipase by approximately 32.6% compared to placebo. These are statistically significant elevations. They are also, in most cases, clinically insignificant.
The critical distinction is between asymptomatic enzyme elevation and pancreatitis. Elevated lipase alone, without abdominal pain, nausea, or vomiting, does not indicate pancreatitis. Many medications raise pancreatic enzymes without causing pancreatic inflammation. Tirzepatide appears to be one of them. A systematic review and meta-analysis found that despite enzyme elevations, tirzepatide carried identical pancreatitis risk compared to placebo across follow-up periods of 12-72 weeks.
When should you worry? If lipase exceeds 3 times the upper limit of normal AND you have symptoms. Severe, persistent upper abdominal pain that radiates to the back. Nausea and vomiting that do not resolve. Abdominal tenderness. These symptoms combined with dramatically elevated enzymes constitute a medical emergency. Seek immediate care.
Routine monitoring of pancreatic enzymes in asymptomatic patients is not currently recommended by most guidelines. If your doctor orders them and they come back mildly elevated (1-2 times upper normal), this is expected and does not require stopping the medication. Document the values for trending purposes, but do not panic. If you have risk factors for pancreatitis, including gallbladder disease, heavy alcohol use, very high triglycerides, or history of pancreatitis, discuss more frequent monitoring with your healthcare provider.
Nutritional markers: the hidden deficiency risk
Tirzepatide reduces appetite. That is the point. But reduced appetite means reduced food intake, and reduced food intake can mean reduced nutrient absorption. Over months of treatment, nutritional deficiencies can develop silently and only become apparent through blood work or symptoms.
Vitamin B12 deserves the most attention. GLP-1 receptor agonists slow gastric emptying, which can impair B12 absorption from food. This is why many compounded tirzepatide formulations include B12. Check B12 levels at baseline and every 6 months. Symptoms of deficiency include fatigue, numbness or tingling in hands and feet, cognitive fog, and anemia. If levels drop below 300 pg/mL, supplementation is warranted even without symptoms.
Vitamin D status can deteriorate with reduced dietary intake, particularly in people who were already borderline deficient. Given that vitamin D plays critical roles in bone health, immune function, and mood regulation, monitoring every 6 months is reasonable. Target levels above 30 ng/mL, ideally between 40-60 ng/mL.
Iron is another concern, especially for menstruating women. Reduced food intake combined with menstrual blood loss can push iron stores dangerously low. Check ferritin, serum iron, TIBC, and transferrin saturation at baseline and every 6 months. Ferritin below 30 ng/mL warrants supplementation even if hemoglobin remains normal.
Other nutrients to track include magnesium (especially if you experience muscle cramps or body aches), zinc, folate, and potassium. A comprehensive metabolic panel captures potassium, but the others require separate orders. The right supplementation strategy can prevent these deficiencies from developing, but blood work confirmation is always better than guessing.
How tirzepatide changes blood work results over time
Blood work on tirzepatide tells a story. Understanding the typical timeline of changes helps you know what to expect and when to raise concerns.
Weeks 1-4 (2.5 mg starting dose)
Fasting glucose begins dropping within the first week. Mild changes in electrolytes are possible, particularly if GI side effects cause fluid loss. Liver enzymes may fluctuate slightly. A1c will not have changed meaningfully yet because it reflects a 2-3 month average. This is your adjustment period, and dramatic lab shifts are uncommon.
Weeks 4-12 (dose titration phase)
This is when changes accelerate. Fasting glucose drops more significantly as doses increase from 5 to 7.5 to 10 mg. Lipids begin improving, with triglycerides often the first to show notable reductions. A1c starts reflecting the treatment effect by week 8-12. Pancreatic enzymes may elevate as the dose increases. Weight loss becomes measurable, and related metabolic improvements follow.
Weeks 12-24 (optimization phase)
A1c shows its most dramatic improvement during this period. Lipid panels typically reach their best values. Liver enzymes, if they were elevated at baseline from fatty liver, often normalize. Inflammatory markers decrease. Insulin resistance improves as reflected by lower fasting insulin and improved HOMA-IR. This is the period where blood work looks most dramatically different from baseline.
Months 6-12 and beyond (maintenance phase)
Metabolic improvements plateau and stabilize. A1c settles at its new set point. Lipids maintain their improved levels. This is also when nutritional deficiencies may first appear if dietary intake has been significantly restricted. Long-term metabolic adaptation occurs, and your body establishes a new metabolic baseline that blood work reflects.
What to tell your doctor before blood work
Transparency with healthcare providers is not just ethical. It is practical. Withholding information about tirzepatide use leads to misinterpretation of lab results, unnecessary follow-up testing, and potentially inappropriate medical decisions.
Tell your doctor:
That you are taking tirzepatide, including the specific dose, how long you have been on it, and when your last injection was. This context is essential for interpreting glucose, A1c, lipid, and liver enzyme results.
Whether it is prescription or compounded. Compounded tirzepatide may contain additional ingredients like glycine, niacinamide, methylcobalamin, or levocarnitine that could independently affect lab values.
Any GI side effects you have experienced. Nausea, vomiting, diarrhea, and constipation all affect hydration status, which directly impacts kidney markers and electrolytes.
Other medications and supplements. Whether you take metformin, phentermine, berberine, or other substances that affect blood sugar and metabolism.
Fasting status. Standard lipid panels and glucose tests require fasting. Tirzepatide delays gastric emptying, which means "fasting" might need to be longer than the typical 8-12 hours. Consider fasting 12-14 hours before blood work to ensure accurate results.
If you are concerned about privacy, know that blood test results are part of your medical record but are protected by HIPAA. Your employer does not have access to your medical records or lab results. Your doctor cannot disclose your tirzepatide use to anyone without your consent, except in very specific legal circumstances.
Common blood work changes and what they mean
Here is a quick-reference table of the most common lab changes on tirzepatide and whether they require action.
Lab Marker | Expected Change | Concerning Level | Action Required |
|---|---|---|---|
Fasting glucose | Decreased 40-70 mg/dL | Below 60 mg/dL (hypoglycemia) | Reduce dose if symptomatic, check for sulfonylurea or insulin use |
HbA1c | Decreased 1.0-2.5% | Below 5.0% if on other diabetes meds | Evaluate for hypoglycemia risk, adjust other medications |
Triglycerides | Decreased 19-25% | Increase or no change despite weight loss | Evaluate dietary factors, consider genetic causes |
LDL cholesterol | Modest decrease | Significant increase | Dietary evaluation, consider statin discussion |
ALT/AST | Stable or decreased | Above 3x upper normal limit | Stop tirzepatide, urgent liver evaluation |
Creatinine | Decreased (muscle loss) or increased (dehydration) | Rapid increase with symptoms | Hydrate, retest in 1-2 weeks, evaluate kidney function |
eGFR | May appear improved (artifact of lower creatinine) | Significant decline from baseline | Verify with cystatin C-based eGFR |
TSH | Usually stable | Outside 0.4-4.0 mIU/L range | Full thyroid panel, evaluate symptoms |
Lipase | Increased 20-33% | Above 3x upper normal with symptoms | Symptom-based: if abdominal pain, seek emergency care |
Vitamin B12 | May decrease over months | Below 300 pg/mL | Supplement with B12, consider compounded formulation with B12 |
Potassium | May decrease with GI losses | Below 3.5 mEq/L | Electrolyte replacement, evaluate hydration |
Use this table as a reference guide, not a replacement for professional medical interpretation. Every person metabolic context is unique, and lab values must be interpreted within the full clinical picture. SeekPeptides provides detailed monitoring guides for members navigating these decisions.
Troubleshooting abnormal results on tirzepatide
Abnormal lab results on tirzepatide do not always mean something is wrong. Often, they reflect expected medication effects or transient physiological changes. Here is how to approach the most common scenarios.
Blood sugar too low
If fasting glucose drops below 70 mg/dL, evaluate two things. First, are you also taking insulin, sulfonylureas, or meglitinides? These medications combined with tirzepatide create significant hypoglycemia risk. Second, when did you last eat? Tirzepatide suppresses appetite so effectively that some people inadvertently fast for extended periods, driving blood sugar dangerously low. The fix is usually adjusting companion diabetes medications or ensuring consistent meal timing, even when appetite is minimal.
Liver enzymes elevated
Mild elevations (1-2x upper normal) during the first month are common and typically self-resolve. Retest in 4 weeks. If enzymes are 2-3x upper normal, reduce alcohol intake if applicable, review all medications and supplements for hepatotoxic potential, and retest in 2 weeks. If above 3x, stop tirzepatide and seek urgent evaluation. The medication should not be restarted until the cause is identified and resolved.
Kidney markers worsening
First question: have you been dehydrated? Check hydration status, rehydrate, and retest in 1-2 weeks. If you have experienced significant vomiting or diarrhea, that is likely the culprit. Second question: how much weight have you lost? Rapid weight loss with muscle wasting can distort creatinine-based kidney markers. Request cystatin C. If kidney function is genuinely declining independent of dehydration and muscle changes, nephrology referral is appropriate.
Thyroid markers shifting
New thyroid abnormalities on tirzepatide warrant attention but not panic. Weight loss itself can alter thyroid hormone metabolism, and TSH may shift as your body adapts to a new metabolic rate. If TSH moves outside the normal range, repeat it in 6-8 weeks. If it remains abnormal, add Free T3 and thyroid antibodies to the workup. Fatigue and hair loss during treatment should prompt thyroid testing even if not otherwise scheduled.
A1c not improving
If A1c has not changed after 12 weeks on a therapeutic dose, several factors could be at play. Dose may be too low. Dietary carbohydrate intake may be offsetting the medication effect. Medication potency could be compromised if storage conditions were suboptimal. Confirm that the reconstitution was done correctly, that storage is proper, and that the dosing is accurate.
Blood work timing: when to schedule relative to your injection
The timing of your blood draw relative to your weekly injection matters more than most people realize. Tirzepatide levels fluctuate throughout the week, and some blood markers are more affected by these fluctuations than others.
For the most representative results, schedule blood work 3-4 days after your last injection. This puts you near the middle of your dosing cycle, when tirzepatide levels are steady and not at their peak or trough. Drawing blood the day after your injection captures peak levels, which may exaggerate glucose-lowering effects. Drawing blood right before your next injection captures trough levels, which may underrepresent the medication effectiveness.
Fasting matters. Plan for a 12-14 hour fast before your draw, not the standard 8-12 hours. Tirzepatide slows gastric emptying, which means food from your previous meal may still be processing even 8-10 hours later. A longer fast ensures fasting glucose and lipid values are truly fasting numbers rather than postprandial values masquerading as fasting ones.
Time of day can also influence results. Morning draws typically produce the most consistent and interpretable results. Cortisol, glucose, and other markers have natural diurnal variation, and early morning values provide the most standardized comparison between draws. Try to schedule blood work at approximately the same time of day for each draw to minimize variability.
If you are on a split dosing schedule, the timing becomes more complex. Aim for blood work at the midpoint between your two doses for the most representative snapshot. Document when both doses were administered so your healthcare provider can interpret results with full context.
Comparing blood work changes: tirzepatide vs semaglutide
Since many researchers switch between or compare these two medications, understanding how their blood work effects differ is valuable. The tirzepatide vs semaglutide comparison extends beyond weight loss and appetite suppression into measurable lab differences.
Tirzepatide generally produces larger A1c reductions than semaglutide at comparable doses. Head-to-head data from SURPASS-2 showed tirzepatide 15 mg reducing A1c by approximately 2.5% compared to semaglutide 1 mg at approximately 1.9%. This difference reflects the additional GIP receptor activation that semaglutide lacks.
Lipid improvements are also more pronounced with tirzepatide. Triglyceride reductions are 5-10% greater, and the overall lipid profile improvement is more comprehensive. This matters for cardiovascular risk assessment and is one reason some researchers switch between medications based on their specific metabolic goals and side effect tolerance.
Liver enzyme improvements appear similar between the two medications, as both reduce hepatic fat accumulation effectively. Kidney marker changes follow similar patterns, driven primarily by GI side effects and weight loss rather than direct drug effects. Pancreatic enzyme elevations may be slightly more pronounced with tirzepatide due to the dual receptor mechanism, though clinically significant pancreatitis remains equally rare with both.
If you are considering converting from one to the other, baseline labs before the switch and follow-up labs at 4 and 12 weeks help establish how your body responds to the new medication specifically.
Special populations: additional blood work considerations
Certain groups need modified monitoring approaches when using tirzepatide.
People with type 2 diabetes
The most important addition: fructosamine testing. While A1c reflects 2-3 month averages, fructosamine reflects 2-3 week averages. During rapid glucose improvement, fructosamine captures changes faster than A1c. Also critical: self-monitoring of blood glucose or continuous glucose monitoring (CGM) to catch hypoglycemia that lab work might miss between draws. Adjust insulin and sulfonylurea doses proactively as tirzepatide takes effect, since hypoglycemia risk compounds with multiple glucose-lowering agents.
People with kidney disease
More frequent kidney monitoring, every 4-6 weeks during dose titration. Always include cystatin C-based eGFR. Monitor urine albumin-to-creatinine ratio (UACR) to track whether diabetic nephropathy is improving or worsening. Report any signs of acute kidney injury promptly, including sudden decreases in urine output, swelling, or flank pain.
People with liver disease
Baseline liver imaging (ultrasound) in addition to enzyme levels. Monthly liver enzyme checks during the first 3 months, then every 3 months ongoing. The potential for liver improvement is higher in this population, but so is the risk of complications if things go wrong.
Women of reproductive age
Tirzepatide increases fertility by improving metabolic function and ovulation regularity. If you do not wish to become pregnant, blood work should include a pregnancy test before starting and periodic testing thereafter. Unintended pregnancies on tirzepatide have been reported, and breastfeeding on tirzepatide raises separate considerations. Monitor menstrual regularity and discuss contraception with your healthcare provider.
People over 65
Age-related decline in kidney function makes monitoring more critical. Add bone density markers (calcium, vitamin D, alkaline phosphatase) because rapid weight loss in older adults accelerates bone density loss. Nutritional monitoring should be more frequent, every 3 months, since reduced appetite combined with age-related absorption changes increases deficiency risk.
Frequently asked questions
Will my doctor know I am taking tirzepatide from blood work alone?
Not definitively. A healthcare provider cannot identify tirzepatide specifically from standard blood work. However, a pattern of dramatically improved glucose, A1c, lipids, and liver enzymes combined with significant weight loss may raise clinical suspicion that a GLP-1 receptor agonist or similar metabolic medication is being used. Experienced clinicians recognize this pattern.
Does tirzepatide show up on a urine test?
No. Standard urine drug screens do not test for tirzepatide or any GLP-1 receptor agonist. Specialized research assays can detect tirzepatide metabolites in urine, but these are not available through standard clinical or occupational testing laboratories.
Should I stop tirzepatide before blood work?
Generally, no. Your blood work should reflect your actual medicated state so your healthcare provider can make informed decisions. The exception is if your doctor specifically wants to see unmedicated baseline values, in which case you would need to stop tirzepatide for at least 4-5 weeks for complete clearance. Discuss this with your provider before making changes to your dosing schedule.
How long after starting tirzepatide will blood work show changes?
Fasting glucose can change within the first week. Lipid changes become measurable by 4-8 weeks. A1c reflects changes after 8-12 weeks because it measures a 2-3 month average. Liver enzyme improvements typically peak around 30 weeks. The full metabolic picture takes about 6 months to stabilize.
Can tirzepatide cause a false positive on any blood test?
Tirzepatide does not cause false positives on drug screens or standard diagnostic tests. However, the metabolic changes it produces can sometimes obscure other conditions. For example, dramatically improved glucose could mask an underlying insulin resistance pattern that resurfaces if the medication is stopped. Similarly, improved liver enzymes could mask concurrent liver disease that the weight loss temporarily improves.
Do I need different blood work for compounded vs brand tirzepatide?
The core monitoring panel is the same. However, compounded formulations often contain additional ingredients. If your compound includes B12, monitoring serum B12 helps confirm adequate absorption. If it contains niacinamide, liver function monitoring is particularly important since niacinamide can independently affect liver enzymes at higher doses. Levocarnitine-containing compounds may warrant carnitine level monitoring in people with kidney impairment.
What if my doctor refuses to order the tests I need?
Direct-to-consumer lab testing is available through services like Quest Direct, Ulta Lab Tests, and similar platforms. You can order comprehensive metabolic panels, lipid panels, thyroid tests, and most other monitoring labs without a doctor order. Results are delivered directly to you. While this does not replace medical interpretation, it ensures you have the data you need to monitor your health. SeekPeptides members have access to detailed interpretation guides for self-ordered lab results.
Does tirzepatide affect blood pressure readings?
Blood pressure is not a blood test, but it is worth addressing. Yes, tirzepatide often reduces blood pressure, typically by 5-10 mmHg systolic and 3-5 mmHg diastolic. This is primarily driven by weight loss and improved insulin sensitivity. If you are on blood pressure medications, monitor closely and discuss dose adjustments with your provider as weight loss progresses.
External resources
For researchers serious about optimizing their tirzepatide protocols and monitoring, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven monitoring frameworks, and a community of thousands who have navigated these exact questions.