Mar 31, 2026

You just got your first vial of GLP-1 medication. It arrived in a cold pack. The label says refrigerate. But then you read something online that says room temperature is fine. Someone else says freezing is okay. A forum post claims their compounded semaglutide sat on the counter for a week and still worked perfectly.
Who do you believe?
Here is the problem. GLP-1 medications are peptides, and peptides are fragile biological molecules that start degrading the moment they leave controlled storage conditions. The wrong temperature for the wrong duration does not just reduce effectiveness. It can render your medication completely useless, waste hundreds of dollars, and potentially introduce safety risks from bacterial contamination or degraded compounds. Every major GLP-1 receptor agonist on the market, from semaglutide to tirzepatide to retatrutide, has specific storage requirements that most guides oversimplify or get wrong entirely. This guide covers every GLP-1 medication on the market, the exact temperature ranges for each one, what happens when storage goes wrong, how to travel safely with your medication, and the critical differences between brand-name and compounded formulations that change everything about how long your vial or pen actually lasts.
The short answer: yes, GLP-1 medications need refrigeration
Every GLP-1 receptor agonist requires refrigeration. No exceptions. The standard storage temperature across all formulations is 36 to 46 degrees Fahrenheit, which translates to 2 to 8 degrees Celsius. This is the temperature range of a properly functioning household refrigerator.
But the full picture is more nuanced than that.
Different medications have different tolerance windows for room temperature exposure. Some can handle weeks outside the fridge after first use. Others need to stay cold at all times. Brand-name pen injectors follow different rules than compounded vials. And the state of the medication, whether it is an unopened pen, an opened vial, a lyophilized powder, or a reconstituted solution, dramatically changes the storage requirements.
Understanding these differences is not optional. It is the difference between a medication that works as intended and one that has quietly lost potency while sitting in your bathroom cabinet. SeekPeptides members consistently report that proper storage is one of the most overlooked factors in getting consistent results from their GLP-1 protocols, and the data backs them up.
Why refrigeration matters for GLP-1 peptides
GLP-1 receptor agonists are peptide-based medications. That means they are built from chains of amino acids folded into precise three-dimensional shapes. These shapes are what allow the medication to bind to GLP-1 receptors in your body and produce the appetite suppression, blood sugar regulation, and fat loss effects that make these drugs so effective.
Heat breaks those shapes.
When peptides are exposed to temperatures above their recommended storage range, the amino acid chains begin to unfold. This process is called denaturation, and it is irreversible. Once a peptide molecule unfolds, returning it to the correct temperature does not refold it. The damage is permanent. Research shows that semaglutide stored at 86 degrees Fahrenheit for just four weeks can lose up to 10 percent of its active ingredient. That might not sound catastrophic, but when you are on a carefully calibrated dosing protocol, a 10 percent reduction means your 0.5 mg dose is really delivering 0.45 mg. Over weeks, that gap compounds. Results slow. Appetite suppression weakens. And most people blame the medication rather than the storage.
Temperature is not the only threat. Light exposure accelerates degradation, which is why many compounded vials come in amber glass. Moisture causes hydrolysis, literally breaking the bonds between amino acids. And bacterial contamination becomes a risk once preservatives in the solution begin to degrade, which happens faster at higher temperatures.
Freezing is equally destructive. When a peptide solution freezes, ice crystals form within the liquid and physically tear apart the molecular structures. A frozen and thawed vial of tirzepatide or semaglutide should be discarded immediately. It cannot be salvaged.
Storage requirements for every major GLP-1 medication
Not all GLP-1 medications follow the same rules. Each formulation has been tested for stability under specific conditions, and the manufacturer or compounding pharmacy assigns storage guidelines based on those tests. Here is what you need to know about each one.
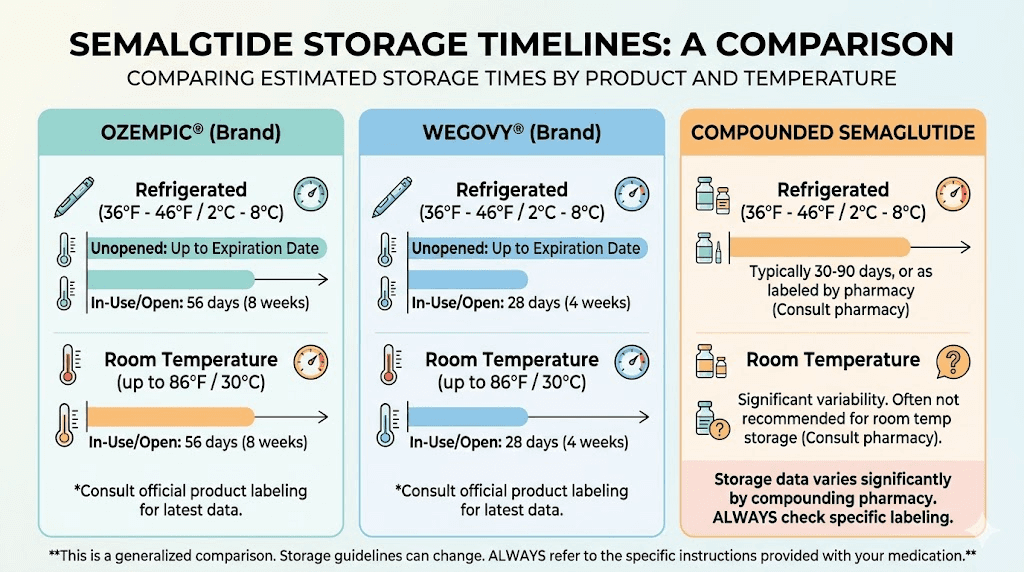
Semaglutide: Ozempic, Wegovy, and compounded formulations
Semaglutide is the most widely prescribed GLP-1 receptor agonist, available in both brand-name and compounded forms. Storage requirements vary significantly depending on which version you are using.
Ozempic (injectable pen): Store in the refrigerator at 36 to 46 degrees Fahrenheit before first use. After the first injection, Ozempic can be kept at room temperature below 86 degrees Fahrenheit or returned to the refrigerator. Either way, it must be used within 56 days (8 weeks) of first use. Do not store the pen with the needle attached. Keep the pen cap on when not in use to protect from light. Never store Ozempic directly next to the refrigerator cooling element, as contact with the cold wall can freeze the medication.
Wegovy (injectable pen): Refrigerate at 36 to 46 degrees Fahrenheit before first use. If needed, an unopened Wegovy pen can be kept at temperatures between 46 and 86 degrees Fahrenheit for up to 28 days. Once that 28-day window expires, the pen must be discarded even if it has not been used. This is a shorter room-temperature window than Ozempic, which matters for travel planning.
Compounded semaglutide (vials): This is where storage gets more complicated. Compounded formulations do not have an expiration date in the traditional sense. Instead, the compounding pharmacy assigns a Beyond-Use Date (BUD) based on stability testing of their specific formulation. The BUD typically ranges from 30 to 90 days from the compounding date when stored properly in the refrigerator. Once the vial is first punctured, most pharmacies recommend use within 28 days. Some pharmacies that conduct rigorous stability testing may assign BUDs of up to 120 days. Others using less rigorous methods default to 14 to 28 days. Always check the label on your specific vial.
Unlike brand-name pens, compounded semaglutide should remain refrigerated at all times unless your pharmacy explicitly states otherwise. The preservative systems in compounded formulations are typically less robust than those in manufactured products, meaning room temperature exposure degrades the medication faster. If you are using compounded semaglutide and want to understand the specifics of your formulation, our guide on how long compounded semaglutide lasts in the fridge covers every scenario in detail.
Tirzepatide: Mounjaro, Zepbound, and compounded formulations
Tirzepatide is technically a dual GIP/GLP-1 receptor agonist, but it follows similar storage principles. The temperature sensitivity of the tirzepatide molecule means that proper storage is just as critical.
Mounjaro (injectable pen): Store in the refrigerator at 36 to 46 degrees Fahrenheit before first use. An unopened pen can be stored at room temperature below 86 degrees Fahrenheit for up to 21 days. This is shorter than both Ozempic and Wegovy, making Mounjaro the most temperature-sensitive of the major brand-name options. After 21 days at room temperature, the pen must be discarded. Once opened, continue storing in the refrigerator or at room temperature below 86 degrees, but always use within the 21-day window.
Zepbound (injectable pen): Zepbound contains the same tirzepatide molecule as Mounjaro and follows identical storage guidelines. Refrigerate before first use, up to 21 days at room temperature below 86 degrees Fahrenheit.
Compounded tirzepatide (vials): Like compounded semaglutide, compounded tirzepatide must be refrigerated at all times. The BUD is typically 28 to 60 days from the compounding date, with many pharmacies defaulting to 28 days after first puncture for multi-dose vials. Compounded tirzepatide should never be stored at room temperature unless your specific pharmacy provides written instructions allowing it. For detailed information on compounded formulation shelf life, see our guide on compounded tirzepatide expiration dates.
One important note about tirzepatide vials: many compounding pharmacies use amber-colored glass to protect the medication from light degradation. Even with amber glass, keep the vial in its box or in a dark section of the refrigerator. Every layer of protection extends the effective life of your medication. If you are curious about whether tirzepatide lasts as long as the label suggests, proper storage is the single biggest factor.
Retatrutide storage requirements
Retatrutide is a triple-agonist peptide targeting GLP-1, GIP, and glucagon receptors simultaneously. As a research peptide without FDA approval, storage guidelines come from stability studies and manufacturer recommendations rather than official prescribing information.
Reconstituted retatrutide solutions should be refrigerated at 36 to 46 degrees Fahrenheit and used within 4 to 6 weeks. The peptide is particularly sensitive to heat, and exposure to temperatures above normal refrigeration accelerates degradation significantly. Our complete guide on how to store retatrutide covers every detail of proper handling.
Lyophilized (freeze-dried) retatrutide powder is far more stable than reconstituted solutions. In powder form, retatrutide can be stored at minus 20 degrees Celsius (minus 4 degrees Fahrenheit) for extended periods, making it suitable for bulk purchasing. At refrigerator temperatures, lyophilized powder remains stable for several months. Just keep it sealed, dry, and away from light.
Retatrutide is also sensitive to light and moisture. Store vials in their original packaging whenever possible. If you are mixing your own solutions using bacteriostatic water, work in a clean environment and return the reconstituted vial to the refrigerator immediately after use. The retatrutide reconstitution chart walks you through the exact process.
Other GLP-1 and related medications
Liraglutide (Saxenda, Victoza): Refrigerate before first use at 36 to 46 degrees Fahrenheit. After first use, store at room temperature below 86 degrees or in the refrigerator. Use within 30 days of first injection. Liraglutide differs from semaglutide in several ways, but storage requirements are broadly similar.
Cagrilintide: As a research peptide, cagrilintide should be refrigerated at 2 to 8 degrees Celsius at all times. Reconstituted solutions are generally stable for 4 to 6 weeks under proper refrigeration. Lyophilized powder can be stored frozen for longer periods. The cagrilintide dosing guide includes storage recommendations specific to common vial sizes.
Survodutide: Still in clinical trials, survodutide follows the same general peptide storage principles. Refrigerate at 2 to 8 degrees Celsius, protect from light, and never freeze reconstituted solutions.
Mazdutide: Similar storage requirements to other GLP-1 peptides. Keep refrigerated, protect from light, use reconstituted solutions within the recommended timeframe. Our comparison of mazdutide versus tirzepatide and mazdutide versus retatrutide includes storage notes for each.
Compounded versus brand-name: the storage differences that matter
This distinction trips up more people than any other aspect of GLP-1 storage. Brand-name medications and compounded formulations are not interchangeable when it comes to how long they last and how they should be stored.
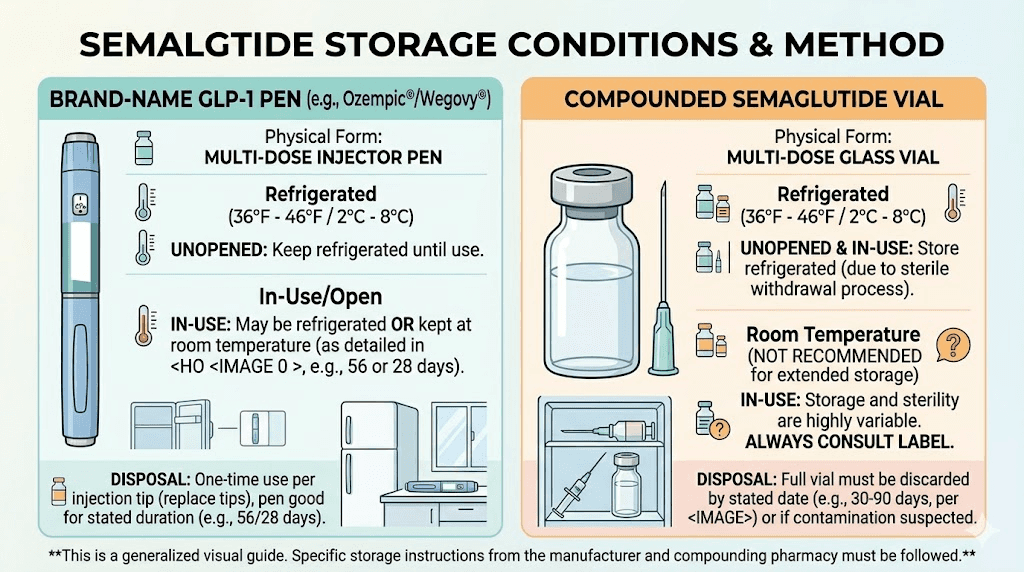
Brand-name pens (Ozempic, Wegovy, Mounjaro, Zepbound) undergo extensive stability testing by the manufacturer. These tests determine exactly how long the medication remains safe and effective under various conditions. The results allow for specific room-temperature windows, like the 56-day room temperature tolerance of Ozempic. The preservative systems, delivery mechanisms, and packaging are all optimized for maximum stability.
Compounded formulations are made by compounding pharmacies under different conditions. The stability testing may be less extensive, the preservative systems may differ, and the packaging may provide less protection. This is why compounded formulations typically have shorter Beyond-Use Dates and stricter storage requirements.
Here is a practical comparison:
Factor | Brand-name pens | Compounded vials |
|---|---|---|
Before first use | Refrigerate (2-8 C) | Refrigerate (2-8 C) |
Room temp tolerance | 21-56 days depending on product | Minimal, keep refrigerated |
After opening | Room temp or fridge for set period | Refrigerate at all times |
Shelf life | Up to 2 years unopened | 30-120 days (BUD) |
Light protection | Built into pen design | Amber glass or keep in box |
Preservative system | Robust, manufacturer-tested | Varies by pharmacy |
The key takeaway: if you are using compounded semaglutide or tirzepatide, treat your vial as more fragile than a brand-name pen. Keep it in the refrigerator. Always. Do not assume the same room-temperature rules apply.
What happens when GLP-1 medications get warm
Maybe your medication sat on the kitchen counter while you were at work. Perhaps the delivery arrived on a hot day and the cold pack was warm by the time you opened the box. Or your travel cooler ran out of ice on a long road trip.
What actually happens to GLP-1 peptides when they get warm?
The degradation process follows a predictable pattern. First, the peptide molecules begin to unfold from their precise three-dimensional configurations. This reduces binding affinity to GLP-1 receptors, meaning each molecule is less effective at triggering the biological response you need. At the same time, the unfolded peptides can aggregate, forming clumps of misfolded protein. These aggregates are not only ineffective but can potentially trigger immune reactions at the injection site.
If your semaglutide gets warm, the severity of the damage depends on two factors: how hot it got and how long it stayed hot. A few hours at room temperature (68 to 77 degrees) is unlikely to cause significant degradation for most formulations. A full day at 86 degrees is a different story. And anything above 86 degrees, even for short periods, should raise serious concerns.
The same applies if tirzepatide gets warm. The tirzepatide molecule is actually slightly more sensitive to temperature fluctuations than semaglutide, which is why Mounjaro has a shorter room-temperature window (21 days) compared to Ozempic (56 days).
Critical point: returning a warm medication to the refrigerator does not undo the damage. The degradation that occurred during the warm period is permanent. And each warm exposure is cumulative. If your vial sat out for four hours on Monday and six hours on Thursday, you have used 10 hours of your room-temperature budget. These exposures add up.
If your medication arrived hot, our guide on what to do when semaglutide arrives hot covers the exact steps to determine whether the medication is still usable.
Signs your GLP-1 has gone bad
Sometimes you cannot be certain whether storage conditions were maintained. Maybe the power went out overnight. Maybe someone moved your vial to the wrong shelf. In these situations, visual inspection becomes your first line of defense.
Color changes: Most GLP-1 formulations are clear and colorless. Any yellowing, browning, or cloudiness indicates degradation. Semaglutide solutions should be clear. Normal semaglutide has a slight to no color. If yours has turned pink or red, discard it immediately. Similarly, tirzepatide color changes signal degradation.
Particles or floaters: Hold the vial up to a light source and look carefully. Any visible particles, fibers, or floating matter means the solution has been contaminated or the peptide has aggregated. Do not use it.
Cloudiness or haziness: A solution that was once clear but now appears milky, hazy, or cloudy has degraded beyond safe use. This cloudiness comes from peptide aggregation, and it means a significant portion of the active ingredient is no longer functional.
Unusual odor: Open the vial carefully and smell it. Peptide solutions should have minimal to no odor. A foul, sour, or unusual smell indicates bacterial contamination, which can develop when preservatives break down at elevated temperatures.
Reduced effectiveness: This is the subtlest sign and the hardest to confirm. If you have been experiencing consistent appetite suppression and weight loss and suddenly notice your medication seems less effective despite no changes to your dose or diet plan, degraded medication could be the cause. It is not the only possible explanation, as plateaus happen, but storage should be one of the first things you investigate.
When in doubt, discard it. The cost of a replacement vial is always less than the cost of injecting degraded or contaminated medication. If you have been using expired semaglutide or suspect your medication has degraded, stop using it and contact your provider.
How long can GLP-1 medications be out of the fridge
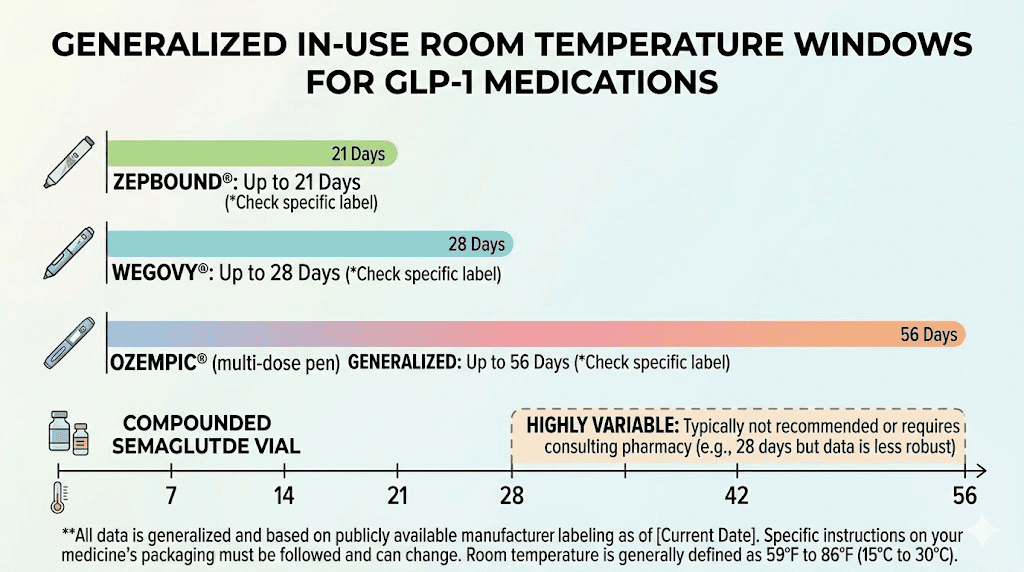
This is the question everyone asks, and the answer depends entirely on which medication you are using. Here is the complete breakdown:
Medication | Form | Room temp window | Max temp | Notes |
|---|---|---|---|---|
Ozempic | Brand pen | 56 days after first use | 86 F (30 C) | Most forgiving brand-name option |
Wegovy | Brand pen | 28 days before cap removal | 86 F (30 C) | Shorter than Ozempic |
Mounjaro | Brand pen | 21 days | 86 F (30 C) | Most temperature-sensitive brand |
Zepbound | Brand pen | 21 days | 86 F (30 C) | Same as Mounjaro |
Compounded semaglutide | Vial | Minimal (check pharmacy) | Keep refrigerated | Some allow brief room temp |
Compounded tirzepatide | Vial | Minimal (check pharmacy) | Keep refrigerated | Stricter than brand pens |
Retatrutide | Vial | Not recommended | Keep refrigerated | Very temperature sensitive |
Cagrilintide | Vial | Not recommended | Keep refrigerated | Refrigerate at all times |
A few critical points about these numbers. First, the room temperature windows assume the medication stays below 86 degrees Fahrenheit at all times. If it gets warmer than that, even briefly, the clock accelerates dramatically. Second, these windows represent maximum allowed time, not optimal time. Your medication will maintain higher potency if you minimize room temperature exposure even within the allowed window. Third, for compounded formulations, always defer to your specific pharmacy label. The numbers in this table are general guidelines. Your pharmacy BUD overrides everything.
Detailed guides on specific medications include how long compounded semaglutide can be out of the fridge, how long compounded semaglutide can be unrefrigerated, how long compounded tirzepatide can be out of the fridge, and how long tirzepatide lasts in the fridge.
Traveling with GLP-1 medications
Travel is where good storage habits get tested. Whether you are flying across the country or driving to a weekend trip, your GLP-1 medication needs a plan.
Flying with GLP-1 medications
Rule number one: never put your medication in checked baggage. The cargo hold of an airplane is not temperature controlled. Temperatures can drop below freezing at altitude or climb above safe ranges on the tarmac. Your medication goes in your carry-on bag. Period.
TSA allows injectable medications through security. You can bring your GLP-1 syringes, pens, vials, and needles in your carry-on. Keep the medication in its original packaging if possible. Carrying a copy of your prescription is a good idea, though it is rarely requested. Declare injectable medications at the security checkpoint if you want to avoid any confusion.
For keeping your medication cold during air travel, use a medical-grade insulated cooler or a dedicated GLP-1 travel case. These cases use gel ice packs or phase-change materials that maintain the correct temperature range for 12 to 24 hours. Important: never place your medication directly against an ice pack. Wrap the ice pack in a cloth or towel to prevent freezing damage. Direct contact with a frozen surface is just as dangerous as heat exposure.
Road trips and car travel
Cars are one of the most dangerous environments for GLP-1 medications. Interior temperatures can exceed 150 degrees Fahrenheit on a hot day, even with windows cracked. Never leave your medication in the car. Not in the glove box. Not in the center console. Not in the trunk. Bring it inside with you wherever you go.
For road trips, use an insulated cooler with ice packs. Check the temperature inside the cooler periodically if you have a thermometer. Replace ice packs as needed. If you are traveling for more than 24 hours, plan stops where you can refrigerate your medication overnight, such as hotel rooms with mini-fridges.
International travel
If you are traveling internationally, research the customs regulations for your destination regarding injectable medications. Most countries allow personal quantities of prescription medications. Carry your prescription documentation, and consider having a letter from your provider explaining the medication and its storage requirements.
Time zone changes can also affect your injection schedule. If you normally inject at 8 AM and you travel across multiple time zones, you may need to adjust your timing. Our guide on traveling with semaglutide and traveling with tirzepatide covers these scenarios in detail.
Storage mistakes that waste your GLP-1
Even people who know the basics of refrigeration make mistakes that silently degrade their medication. Here are the most common ones.
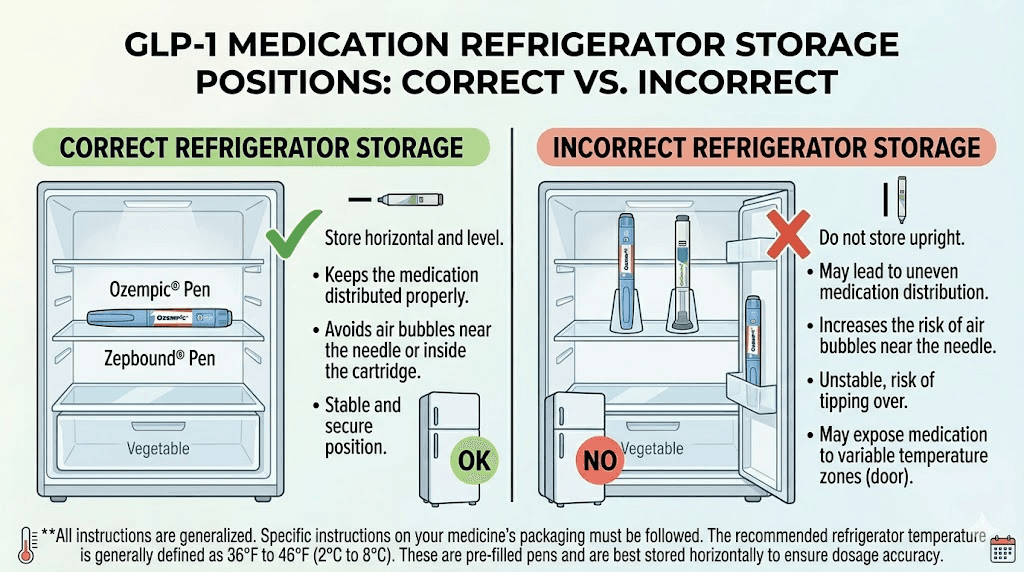
Storing vials in the refrigerator door
The refrigerator door is the warmest spot in your fridge. Every time you open the door, the temperature swings by several degrees. For medications that need consistent 36 to 46 degree storage, these fluctuations matter. Store your GLP-1 medication on a middle shelf toward the back of the refrigerator, where temperatures remain most stable.
Placing vials directly against the cooling element
The back wall or cooling element of a refrigerator can drop below freezing, especially in older models. If your vial or pen touches this surface, it can partially freeze. As discussed earlier, freezing destroys peptide structures. Keep at least a few inches between your medication and the back wall. Some people use a small plastic container on a middle shelf to keep their vials organized and protected from both the door and the cooling element. If you have ever accidentally left semaglutide out overnight, you know how stressful these mistakes can be.
Forgetting to refrigerate after injection
You wake up, draw your dose, inject, and then get busy with your morning routine. The vial sits on the counter. You remember it four hours later. For brand-name pens with room-temperature windows, this is usually fine. For compounded vials, every minute on the counter counts. Build a habit: inject, recap, refrigerate. Three steps. Every time.
Using medication past the BUD or open-vial window
The Beyond-Use Date on your compounded vial is not a suggestion. It is the date after which the pharmacy can no longer guarantee the medication is safe and effective. Similarly, the 28-day or 56-day windows for brand-name products are hard limits. Using medication past these dates means you are injecting a substance with unknown potency and potentially degraded preservatives. The risk of bacterial contamination increases with every day past the recommended use period. Check the dates on semaglutide expiration and tirzepatide expiration to understand the specifics for your medication.
Exposing medication to direct sunlight
UV radiation from sunlight accelerates peptide degradation significantly. Even indirect bright light can cause damage over time. Store your medication in its original box or in a dark area of the refrigerator. If you are using a travel cooler, make sure it is opaque and not sitting in direct sunlight.
Beyond-use dates and expiration dates explained
These two terms sound similar but mean very different things, and confusing them is one of the most common mistakes in GLP-1 storage.
Expiration date: Assigned by the manufacturer of FDA-approved medications. Based on extensive stability testing under controlled conditions. Tells you how long the unopened, properly stored product remains safe and effective. Ozempic, Wegovy, Mounjaro, and Zepbound all have manufacturer expiration dates, typically 18 to 24 months from manufacturing.
Beyond-Use Date (BUD): Assigned by compounding pharmacies for compounded formulations. Based on the pharmacy own stability testing or USP <797> guidelines for sterile preparations. The BUD is almost always much shorter than a manufacturer expiration date, typically 30 to 90 days for compounded GLP-1 medications.
USP <797> is the regulatory standard that governs how compounding pharmacies determine BUDs. The standard categorizes compounded sterile preparations based on the environment where they were made and the level of testing conducted. Category 1 preparations, made in less controlled environments, get shorter BUDs. Category 2 preparations, made in full cleanroom suites, can receive longer BUDs. Category 3, with the most rigorous facilities and testing, can assign BUDs up to 180 days.
The practical implication: if your compounded semaglutide has a 28-day BUD and your friend at a different pharmacy has a 90-day BUD for the same concentration, neither label is wrong. The difference reflects the testing and facilities of each pharmacy. Always follow your own label. For a deeper understanding of compounded formulation timelines, our guides on compounded semaglutide expiration and semaglutide shelf life provide the full picture.
Reconstituted versus lyophilized storage
If you are working with research peptides like retatrutide or cagrilintide, you likely need to reconstitute lyophilized powder before use. The storage rules for powder and solution are dramatically different.
Lyophilized (freeze-dried) powder:
Room temperature storage: stable for weeks to months depending on peptide
Refrigerator storage (2-8 C): stable for months to over a year
Freezer storage (-20 C): stable for years in some cases
Keep sealed, dry, and away from light
Moisture is the primary enemy of lyophilized peptides
Reconstituted solution:
Must be refrigerated at 2-8 C at all times
Typical stability: 4-6 weeks for most peptides
Never freeze reconstituted solutions
Use bacteriostatic water (not sterile water) for multi-use vials
Each needle puncture introduces contamination risk
The difference in stability is dramatic. A vial of lyophilized retatrutide stored in the freezer might remain potent for over a year. Once you add bacteriostatic water and reconstitute it, that same peptide has 4 to 6 weeks of useful life in the refrigerator. This is why many researchers reconstitute only what they need for the near term and keep remaining powder in long-term storage. Our peptide reconstitution guide covers the exact process.
For semaglutide specifically, the semaglutide reconstitution process and tirzepatide reconstitution process each have specific water-to-peptide ratios that affect both dosing accuracy and solution stability. Using the correct amount of bacteriostatic water matters for both. Check our semaglutide reconstitution chart or tirzepatide reconstitution chart for the exact calculations.
Seasonal considerations and power outages
Summer heat and winter storms both present unique storage challenges that catch people off guard.
Summer storage tips
During hot months, pay extra attention to delivery timing. Schedule medication deliveries for days when you will be home to receive the package immediately. If your medication arrives via mail order, check the cold pack upon arrival. If the gel pack is warm or fully thawed, the medication may have been exposed to unsafe temperatures during transit.
If you live in a hot climate and your home loses air conditioning, your refrigerator will continue working, but monitor the internal temperature with a thermometer. Most refrigerators maintain safe temperatures for 4 to 6 hours without power, as long as you keep the door closed.
Winter considerations
Freezing is the bigger risk in winter. If your medication is shipped during cold weather and the package sits on an unheated porch, the medication can freeze. Check the solution for ice crystals or unusual cloudiness upon arrival. If tirzepatide has frozen, do not use it. The same applies to semaglutide and all other GLP-1 formulations.
Power outages
A modern refrigerator keeps food safe for about 4 hours during a power outage, as long as the door stays closed. GLP-1 medications in the refrigerator will remain within safe temperature range for that same window. If the outage extends beyond 4 hours, consider transferring your medication to a cooler with ice packs. Do not open the refrigerator door to check on the medication, as this lets warm air in and reduces the cold reserve.
For people in areas prone to extended power outages, keeping a backup plan ready makes sense: an insulated cooler, a supply of reusable ice packs in the freezer, and a thermometer to monitor conditions.
Long-term storage and bulk purchasing
Many researchers buy peptides in bulk to save money. If you are purchasing multiple vials of retatrutide, semaglutide, or cagrilintide, long-term storage strategy matters.
For lyophilized powders: Store unopened vials in the freezer at minus 20 degrees Celsius. Ensure vials are sealed against moisture. Label each vial with the date received. Use the oldest vials first (first in, first out). Under these conditions, most peptides maintain potency for 12 to 24 months.
For pre-mixed compounded vials: Long-term bulk storage is not practical because the BUD limits shelf life to 30 to 90 days. Order only what you will use within the BUD window. If your compounded pharmacy offers volume discounts, calculate whether you can realistically use the extra vials before they expire.
For brand-name pens: Manufacturer expiration dates allow longer storage, typically 18 to 24 months from manufacturing. Check expiration dates before purchasing. Some pharmacies or online retailers may ship pens that are closer to expiration, which limits your buffer. Store unopened pens in the refrigerator until you are ready to start each one.
The complete peptide storage guide covers storage protocols for every form factor and peptide type, including information on storing peptides after reconstitution and how long peptides last in the fridge by type.
Temperature monitoring tools
If you are serious about maintaining your GLP-1 medication potency, a refrigerator thermometer is a worthwhile investment. Standard kitchen thermometers work, but digital models with high and low temperature recording give you the most useful data. These record the warmest and coldest temperatures reached since the last reset, which means you can check whether your refrigerator stayed in range overnight or during a power outage.
For travel, there are compact digital thermometers designed for medication coolers that provide real-time temperature readings. Some even have Bluetooth connectivity and will alert your phone if the temperature goes outside the safe range.
SeekPeptides members who use home monitoring tools report better consistency in their results, which makes sense. When you know your medication has been stored correctly, you can trust that any changes in effectiveness are due to biological factors rather than degraded medication. It removes one of the biggest variables from your protocol.
Special considerations for oral and alternative GLP-1 formulations
Not all GLP-1 medications are injectable. The storage requirements for alternative delivery methods differ from standard vials and pens.
Oral semaglutide (Rybelsus): Tablet form semaglutide does not require refrigeration. Store at room temperature between 68 and 77 degrees Fahrenheit. Keep in the original blister packaging until use. Protect from moisture. This is one of the few GLP-1 formulations that genuinely does not need a refrigerator, making it the most travel-friendly option. For more on oral options, see our guide on oral semaglutide drops.
Sublingual formulations: Sublingual GLP-1 preparations typically need refrigeration, though some stable formulations may allow short-term room temperature storage. Always follow the pharmacy instructions for your specific preparation. Sublingual tirzepatide and semaglutide troches generally need refrigeration, which our guides on tirzepatide ODT and semaglutide troches cover in detail.
GLP-1 patches: Newer delivery methods like GLP-1 patches have their own storage requirements. Generally, patches should be stored at room temperature in their sealed packaging until use. Once removed from packaging, apply immediately.
Nasal sprays: Research peptide nasal sprays like intranasal retatrutide should be refrigerated after reconstitution and used within the recommended timeframe.
How storage affects your dosing accuracy
Storage is not just about safety. It directly affects whether the dose you measure is the dose you actually receive.
When semaglutide or tirzepatide degrades, the active compound breaks down into inactive fragments. Your syringe still draws the same volume, but a percentage of the molecules in that volume are no longer functional. If your medication has degraded by 15 percent, your 2.5 mg dose is really delivering about 2.1 mg of active compound.
This matters enormously for people on titration protocols. If you are titrating tirzepatide for weight loss and your medication has quietly degraded, you might increase your dose prematurely because you are not getting the expected response at the current level. Or you might conclude that a particular medication does not work for you, when in reality the medication was fine but the degraded vial was not delivering the full dose.
This is one reason why some people report that compounded semaglutide is not working or that tirzepatide stopped working. Before adjusting your protocol, check your storage conditions and verify the medication has not passed its BUD or expiration date. Use our semaglutide dosage calculator or tirzepatide dosage calculator to verify your dosing is correct, and then assess whether storage might be the variable.
Frequently asked questions
Can I store GLP-1 medication in a mini-fridge?
Yes, but with caution. Mini-fridges often have less stable temperature control than full-size refrigerators, with wider temperature swings between cooling cycles. If you use a mini-fridge, place a thermometer inside to verify the temperature stays consistently between 36 and 46 degrees Fahrenheit. Avoid placing your medication against the back wall where freezing can occur.
What should I do if I accidentally froze my GLP-1 medication?
Discard it. Freezing irreversibly damages the peptide structure of semaglutide, tirzepatide, and all other GLP-1 receptor agonists. Even if the solution looks clear after thawing, the molecular damage is invisible and permanent. Contact your pharmacy for a replacement.
Is it safe to use GLP-1 medication that was left out overnight?
It depends on the specific medication and how warm your room was. For brand-name pens with room-temperature windows (Ozempic, Mounjaro), a single overnight at room temperature below 86 degrees Fahrenheit is generally safe and counts against the product room-temperature allowance. For compounded formulations, consult your pharmacy. The risk increases with each exposure. Our detailed guide on the topic of semaglutide left out overnight covers specific scenarios.
Can I pre-fill syringes and store them in the fridge?
This is generally not recommended. Pre-filling syringes exposes the medication to additional contamination risk and removes it from the preservative environment of the vial. If you are considering this for convenience, our guide on pre-filling semaglutide syringes discusses the risks and best practices in detail.
How do I know if my compounding pharmacy has good stability testing?
Ask your pharmacy directly about their stability testing procedures. Pharmacies that conduct their own stability studies can typically assign longer BUDs (60-120 days). Pharmacies that rely on published literature or default USP guidelines usually assign shorter BUDs (14-28 days). A longer BUD from a reputable pharmacy generally indicates more rigorous testing, not a less safe product.
Does the needle size or injection site affect how I should store my medication?
No. Storage requirements are about the medication itself, not how you administer it. Whether you inject in the abdomen, thigh, or arm, the storage rules remain the same.
Should I store different GLP-1 medications separately in the fridge?
There is no need to physically separate different GLP-1 medications in the refrigerator. However, clearly label each vial with the medication name, concentration, date opened, and BUD. If you are stacking peptides like cagrilintide with retatrutide or using semaglutide and tirzepatide together, keeping them organized prevents mix-ups.
External resources
USP General Chapter 797: Pharmaceutical Compounding, Sterile Preparations
CDC Vaccine Storage and Handling Guidelines (applicable principles)
For researchers serious about optimizing their GLP-1 protocols and ensuring every dose delivers maximum potency, SeekPeptides provides comprehensive storage guides, stability databases, and handling protocols that take the guesswork out of medication management. Members access detailed protocol builders, weight-based calculators like our semaglutide dosage calculator and peptide reconstitution calculator, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your refrigerators stay calibrated, your peptides stay potent, and your storage habits stay consistent.