Mar 20, 2026

You did everything right. You found a compounding pharmacy, got your prescription, started your injections on schedule. Weeks pass. Nothing. No appetite changes. No scale movement. No sign that this medication is doing anything at all. Meanwhile, your friend on brand-name Wegovy dropped twelve pounds in her first month. You are taking the same active ingredient. So why is her semaglutide working and yours is not?
This is not an uncommon story. And the answer is almost never "semaglutide does not work for you." The answer, far more often, lives in the compounding itself. In the pharmacy that made it. In how it was stored, shipped, and formulated. In whether the active ingredient in your vial is actually what the label says it is. These are problems that general semaglutide troubleshooting guides never address, because they assume your medication is what it claims to be. When you are using compounded semaglutide, that assumption can be dangerously wrong.
This guide focuses exclusively on compounding-specific failures. Not the diet and exercise factors. Not the patience-required timeline. The actual pharmaceutical and quality-control reasons that your compounded semaglutide might contain less active peptide than advertised, or the wrong form entirely, or a degraded product that lost potency before it ever reached your refrigerator.
How compounded semaglutide differs from brand-name versions
Before diagnosing why your compounded semaglutide is not working, you need to understand what makes it fundamentally different from FDA-approved products like Ozempic and Wegovy. These are not minor differences. They affect every aspect of what you are injecting.
Brand-name semaglutide goes through years of clinical trials. The FDA verifies potency, purity, sterility, and stability. Every batch must meet identical specifications. The manufacturing process uses validated equipment in facilities inspected regularly by federal regulators. When you inject Wegovy, you can trust that each 0.25 mg dose contains exactly 0.25 mg of active semaglutide base.
Compounded semaglutide operates in a different regulatory universe. 503A and 503B compounding pharmacies create these preparations under state pharmacy board oversight, not direct FDA approval. The raw semaglutide powder typically comes from third-party suppliers, often overseas. The pharmacy reconstitutes it, determines concentration, adds preservatives, and ships it to you. Every step introduces potential for error that simply does not exist with manufactured products.
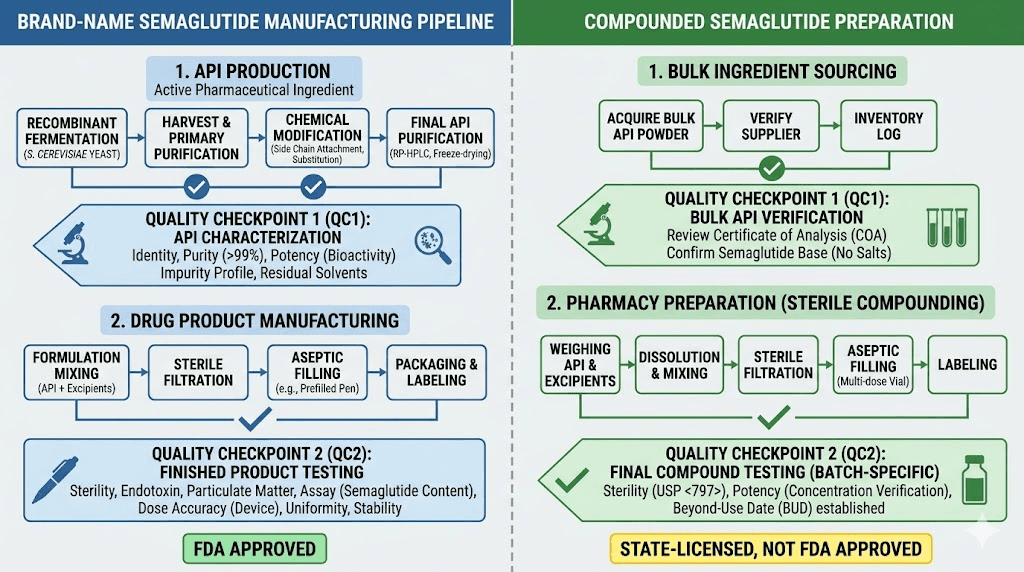
The clinical evidence gap is enormous. FDA-approved semaglutide has data from trials involving tens of thousands of patients showing approximately 15% body weight loss over 68 weeks. Compounded semaglutide has zero clinical trials. None. The assumption that it works identically is exactly that, an assumption, not a proven fact.
This does not mean compounded semaglutide cannot work. Many people achieve excellent results. But when it fails, the list of potential causes is much longer than with brand-name, because you are adding an entire layer of pharmaceutical variables that manufactured products have already controlled for.
Twelve compounding-specific reasons your semaglutide is not working
If your compounded semaglutide is failing, the cause almost always falls into one of these categories. Some are within your control. Others depend entirely on the pharmacy that made your medication. Understanding each one helps you narrow down the problem and find a solution.
1. Your vial contains less active peptide than labeled
This is the single most common reason compounded semaglutide fails. And it is the hardest to detect.
Without FDA manufacturing standards, potency can vary significantly between batches. A vial labeled as 5 mg/mL might contain 3.8 mg/mL. Or 4.2. Or 2.1. You have no way to know without laboratory testing. A study of compounded GLP-1 preparations found that some contained as little as 60% of the labeled active ingredient. That means if you think you are injecting 0.5 mg, you might actually be getting 0.3 mg, a dose too low to produce meaningful appetite suppression or weight loss.
The raw semaglutide powder itself matters too. Pharmaceutical-grade semaglutide peptide has purity specifications above 95%. Cheaper raw material from unverified suppliers may have lower purity, meaning the pharmacy starts with less active ingredient before compounding even begins.
2. You received a salt form instead of semaglutide base
This is a critical issue the FDA has specifically warned about. Some compounding pharmacies use semaglutide sodium or semaglutide acetate instead of semaglutide base. These are chemically different molecules.
Why does this matter? Because all the clinical trial data, all the dosing guidelines, all the efficacy numbers come from semaglutide base. The salt forms have never been tested in clinical trials. The FDA considers them different active ingredients entirely. Semaglutide sodium has a different molecular weight than semaglutide base, which means the same number of milligrams contains a different amount of the active peptide. If your pharmacy used a salt form and dosed it as if it were base semaglutide, you could be getting substantially less active drug than intended.
How do you know which form you received? Ask your pharmacy directly. Request a certificate of analysis (COA) for the raw material. If they cannot or will not provide one, that itself is a red flag. The COA should specify "semaglutide" or "semaglutide base," not "semaglutide sodium" or "semaglutide acetate."
3. The medication degraded during shipping
Semaglutide is a peptide. Peptides are fragile molecules that break down when exposed to heat, light, or agitation. Brand-name products ship in temperature-controlled packaging validated through extensive stability testing. Compounded semaglutide? That depends entirely on your pharmacy.
If your vial arrived warm, sat in a hot mailbox for hours, or traveled through extreme temperatures without proper cold-chain packaging, the active peptide may have partially degraded before you ever used it. The solution might look perfectly clear and normal while containing significantly less active semaglutide. You can read more about temperature exposure risks in our guide on what happens if semaglutide gets warm and what to do when semaglutide arrives hot.
Most potency loss from heat exposure is invisible. The peptide chain unfolds and fragments. You cannot see it, smell it, or taste it. But your body absolutely notices the difference.
4. Improper storage after you received it
Even if your compounded semaglutide arrived in perfect condition, how you store it at home determines whether it stays effective. Compounded preparations are generally less stable than brand-name products because they lack the same proprietary stabilizer systems.
The rules are straightforward but unforgiving. Keep it refrigerated at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Never freeze it, as freezing damages the peptide structure permanently. Do not store it in the refrigerator door where temperature fluctuates every time you open it. Keep it away from light. For detailed storage guidance, check our articles on how long compounded semaglutide lasts in the fridge and refrigeration requirements for compounded semaglutide.
Leaving compounded semaglutide out of the fridge for extended periods accelerates degradation. A few hours at room temperature is usually fine. A full day in a warm kitchen? That could cost you meaningful potency. If you accidentally left your semaglutide out overnight, the damage depends on the ambient temperature, but some degradation is virtually guaranteed.
5. The beyond-use date has passed
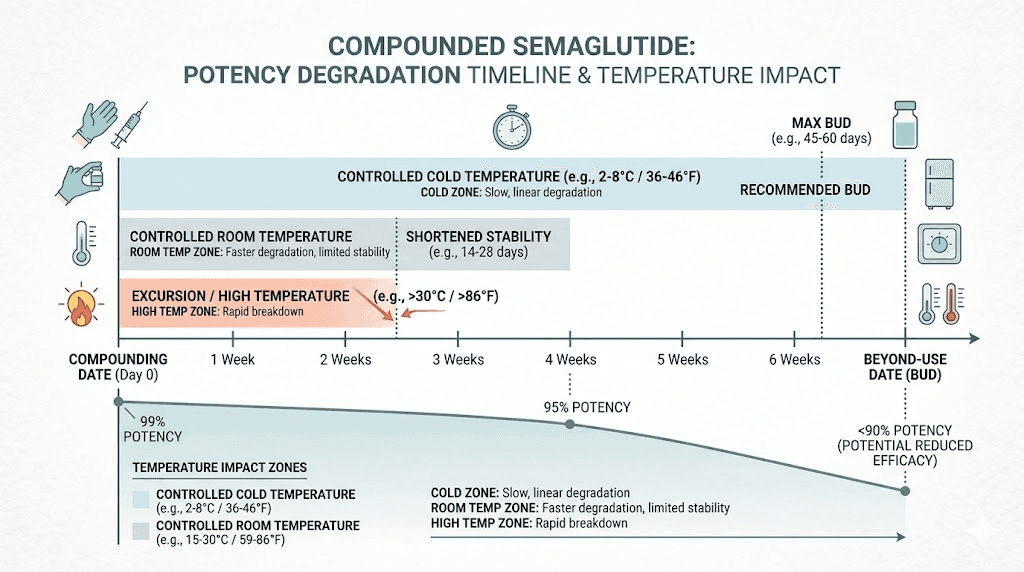
Compounded medications have beyond-use dates (BUDs), not expiration dates. The distinction matters. Expiration dates on brand-name drugs are backed by extensive stability testing, sometimes showing the drug remains effective for years. Beyond-use dates on compounded preparations are more conservative because the stability data simply does not exist.
Most compounded semaglutide carries a BUD of 30 to 90 days from the date of compounding. Not from the date you received it, from the date it was made. If your pharmacy compounded it three weeks before shipping, your effective window is already shorter than you think. Using compounded semaglutide past its BUD means you could be injecting a product with substantially reduced potency. See our complete guide on compounded semaglutide expiration and semaglutide shelf life for specific timelines.
6. Dosing errors from vial-and-syringe administration
Brand-name Wegovy and Ozempic use pre-filled injection pens with fixed doses. You click, inject, done. The dose is mechanically precise every time.
Compounded semaglutide comes in multi-dose vials. You draw it up yourself with an insulin syringe. This introduces multiple error points that can dramatically affect your dose.
First, you need to know the concentration. If your vial is 5 mg/mL and you want 0.25 mg, you need to draw exactly 5 units (0.05 mL). If your vial is 2.5 mg/mL, you need 10 units for the same dose. Confusing concentrations is one of the most common medication errors reported to poison control centers, with some patients accidentally administering ten times their intended dose. Our semaglutide dosage calculator can help you determine exact volumes, and our guides on semaglutide dosage in units and semaglutide unit-to-mg conversions walk through the math step by step.
Second, reading an insulin syringe accurately requires good technique. Air bubbles reduce the actual volume delivered. Drawing past the mark and pushing back introduces waste. These small errors compound over weeks, potentially delivering 10 to 20% less medication than intended. That gap can mean the difference between effective and ineffective treatment.
7. Incorrect reconstitution destroyed the peptide
If you received lyophilized (freeze-dried) semaglutide powder that requires reconstitution, the preparation step itself can damage the medication.
The most common reconstitution errors include using the wrong diluent, injecting bacteriostatic water directly onto the powder cake instead of letting it run down the vial wall, shaking the vial vigorously instead of swirling gently, and using the wrong volume of diluent. Any of these can partially denature the peptide, reducing potency without any visible sign of damage. Our semaglutide reconstitution guide covers proper technique in detail, and our bacteriostatic water mixing guides provide exact volume calculations for 5 mg and 10 mg vials.
Temperature matters during reconstitution too. Let the lyophilized powder and the bacteriostatic water reach the same temperature before mixing. Combining cold powder with room-temperature water (or vice versa) can stress the peptide.
8. Contamination or impurities in the formulation
The FDA has issued multiple warnings about compounded semaglutide products containing impurities, including bacterial endotoxins, particulate matter, and unknown degradation products. These contaminants may not affect potency directly, but they can cause side effects that mask or complicate the medication response.
Some users report unusual side effects from compounded semaglutide that they never experienced with brand-name versions. Injection site reactions, unusual fatigue, dizziness, and gastrointestinal symptoms beyond what the drug typically causes could indicate contamination. If your semaglutide looks discolored, cloudy, or contains visible particles, do not use it. But even clear solutions can contain harmful impurities at levels invisible to the naked eye.
9. The pharmacy is not legitimate
This is harder to accept but essential to consider. The FDA has identified fraudulent compounded semaglutide products with labels listing pharmacy names that either do not exist or did not actually compound the product. Some operations sell repackaged research chemicals as pharmaceutical compounds.
Red flags include prices significantly below market rate (under $100 per month for injectable semaglutide should raise immediate suspicion), pharmacies that do not require a valid prescription, vendors that ship without temperature-controlled packaging, and operations with no verifiable state pharmacy license. Our guide on cheap compounded semaglutide covers pricing realities, and the grey market GLP-1 guide explains the risks of unverified sources.
10. Your dose is too low for your body weight
This is not strictly a compounding issue, but it is far more common with compounded semaglutide because many online telehealth clinics prescribe conservatively. Some intentionally keep patients on lower doses to reduce their cost per patient.
The FDA-approved titration schedule for Wegovy starts at 0.25 mg weekly for four weeks, then increases to 0.5 mg, then 1.0 mg, then 1.7 mg, and finally the maintenance dose of 2.4 mg. Many compounded semaglutide prescribers never titrate patients to the higher doses where the medication produces its strongest effects. If you have been sitting at 0.25 mg or 0.5 mg for months without titration, your dose may simply be too low to produce meaningful results. Check your dose against our semaglutide dosage chart and compounded semaglutide dose chart.
Some patients who spent four weeks on semaglutide with no weight loss simply needed a higher dose. The starting dose exists for tolerability, not efficacy. The medication truly starts working for most people at 1.0 mg and above.
11. The added ingredients are interfering
Unlike brand-name semaglutide, which contains a standardized set of inactive ingredients, compounded versions often include additional compounds. Vitamin B12, glycine, niacinamide, L-carnitine, and pyridoxine (vitamin B6) are popular additions.
These added ingredients create two potential problems. First, their interaction with semaglutide peptide stability has not been studied in controlled conditions. Some compounds may accelerate peptide degradation. Second, the pH adjustments required to incorporate additional ingredients can affect semaglutide stability. Peptides are highly sensitive to pH changes, and even small shifts can alter folding and bioactivity.
If your compound contains additives and is not working, consider requesting plain semaglutide without extras. The semaglutide glycine B12 blend and semaglutide methylcobalamin combinations are popular but unproven from a stability standpoint.
12. Injection technique is reducing bioavailability
How you inject affects how much medication your body actually absorbs. Subcutaneous injection delivers semaglutide into the fat layer beneath the skin, where it absorbs slowly and consistently. Errors in technique can reduce bioavailability significantly.
Common injection technique problems include injecting too shallow (intradermal, causing irritation and poor absorption), injecting too deep (intramuscular, causing faster absorption and shorter duration), not rotating injection sites (leading to lipodystrophy that impairs absorption), and injecting into areas with minimal subcutaneous fat. Read our guides on best injection sites for semaglutide and proper semaglutide injection technique for step-by-step instructions.
If you accidentally injected semaglutide into muscle, the medication will absorb faster and clear your system sooner, reducing the sustained appetite suppression that makes weekly dosing effective.
How to verify your compounding pharmacy
Not all compounding pharmacies are equal. The difference between a reputable operation and a questionable one can be the difference between effective medication and expensive water. Here is how to evaluate yours.
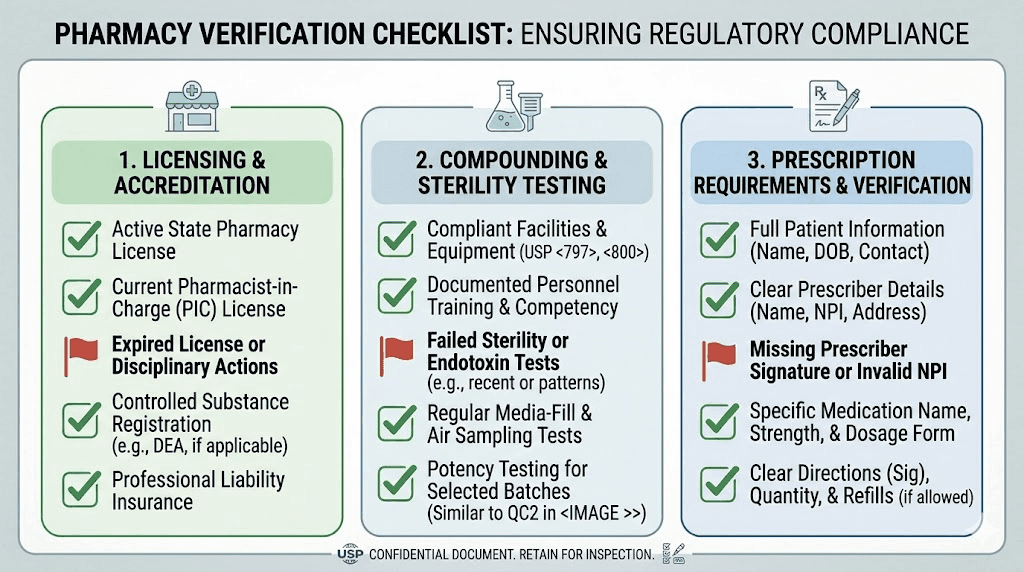
Check licensing and accreditation
Every legitimate compounding pharmacy must hold a state pharmacy license. You can verify this through your state board of pharmacy website. Beyond state licensing, look for PCAB (Pharmacy Compounding Accreditation Board) accreditation or USP 797/800 compliance. These voluntary standards indicate a pharmacy that exceeds minimum requirements.
The distinction between 503A and 503B pharmacies matters too. 503A pharmacies compound individual prescriptions and are regulated by state boards. 503B outsourcing facilities can compound in larger batches and face more FDA oversight, including regular inspections. Generally, 503B facilities offer more consistent quality because they operate under stricter regulatory requirements.
Request third-party testing
Ask your pharmacy if they conduct third-party potency and purity testing on their semaglutide batches. A reputable pharmacy will provide certificates of analysis (COAs) showing the actual measured potency of the raw material and the finished product. If a pharmacy refuses to provide testing documentation or says they do not conduct independent testing, consider that a significant warning sign.
The COA should show potency within 90 to 110% of the labeled concentration, sterility test results, endotoxin testing results, and pH measurements. These are standard pharmaceutical quality metrics that any legitimate compounding operation should track.
Evaluate the prescription process
A legitimate compounding pharmacy requires a valid prescription from a licensed provider. They should verify your medical history, confirm the indication, and ensure proper dosing. Operations that prescribe through a brief online questionnaire without meaningful medical evaluation, or that do not require a prescription at all, are cutting corners that extend beyond just the paperwork.
Storage mistakes that silently destroy potency
You might be doing everything else right and still have ineffective medication because of storage errors. Compounded semaglutide is more sensitive to environmental conditions than most people realize. And the tricky part? Degraded semaglutide looks exactly the same as fresh semaglutide.
Temperature is everything
The ideal storage range is 36 to 46 degrees Fahrenheit. That is the main compartment of your refrigerator, not the door. Refrigerator door temperatures can swing 5 to 10 degrees every time you open it. Over weeks of daily door-opening, that cumulative temperature instability degrades peptides faster than constant refrigeration.
Freezing is worse than warmth. Ice crystal formation physically damages the peptide structure, and this damage is permanent. Even brief freezing, like placing the vial against the back wall of an overly cold refrigerator, can cause irreversible degradation. If you suspect your vial froze, do not use it.
Room temperature exposure follows a rough rule: potency loss is minimal for a few hours, measurable after 24 hours, and potentially significant after several days. How long your specific vial can tolerate room temperature depends on the formulation, preservatives used, and ambient conditions. See how long compounded semaglutide can be unrefrigerated for detailed timelines.
Light exposure accelerates breakdown
UV and visible light both accelerate peptide degradation through photolysis. Brand-name semaglutide pens are designed with light-protective casings. Your compounded vial is clear glass. Every minute it sits on the counter under kitchen lights, or in a car with sunlight streaming through the window, the peptide degrades slightly. Store your vial in its original box, inside the refrigerator, away from the light.
Track your vial from day one
Write the date you received your vial and the beyond-use date on the box with a marker. Do not rely on memory. After two months, even perfectly stored compounded semaglutide may have lost enough potency to reduce effectiveness. If your semaglutide vial has been in your fridge for longer than the labeled BUD, it is time for a fresh one, even if there is medication remaining. See our vial shelf life guide for specific benchmarks.
The dosage accuracy problem: why vials fail where pens succeed
Pre-filled pens eliminate human error. Vials introduce it at every step. This is not a criticism of compounded semaglutide as a concept, but a reality of the delivery system that most patients underestimate.
Concentration confusion
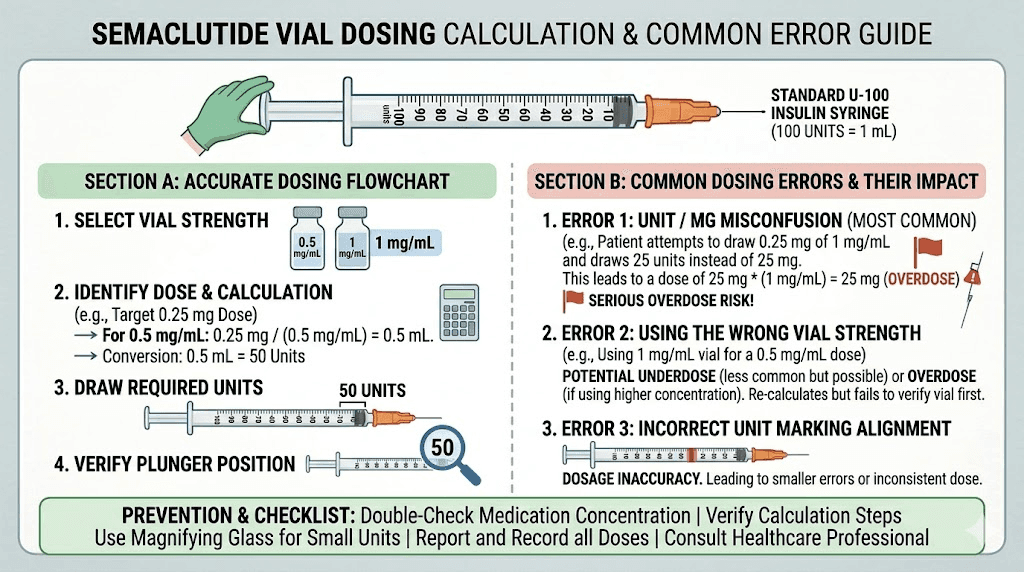
Compounded semaglutide comes in various concentrations: 1 mg/mL, 2 mg/mL, 2.5 mg/mL, 5 mg/mL, and sometimes higher. If your provider tells you to take "0.25 mg" and your vial concentration is 5 mg/mL, you need exactly 5 units on an insulin syringe. But if you previously had a 2.5 mg/mL vial and are used to drawing 10 units for the same dose, and you continue drawing 10 units with the new concentration, you just doubled your dose.
This exact scenario, drawing the same number of units without adjusting for a new concentration, has been reported to poison control centers as a serious medication error. Our conversion guides for 0.25 mg, 0.5 mg, 1.0 mg, and 1.7 mg of semaglutide can help you calculate the correct volume for any concentration.
Syringe selection matters
Not all insulin syringes are the same. A 100-unit (1 mL) syringe has different graduation marks than a 50-unit (0.5 mL) syringe or a 30-unit (0.3 mL) syringe. For small doses like 0.25 mg from a 5 mg/mL vial (5 units), a 30-unit syringe provides the most accurate measurement because each graduation mark represents 0.5 units instead of 1 or 2 units. Read our GLP-1 syringe selection guide for detailed recommendations.
Using the wrong syringe can introduce 10 to 40% dosing error at small volumes. For semaglutide, where the difference between a therapeutic dose and a subtherapeutic dose can be fractions of a milligram, syringe accuracy is not optional.
Air bubbles steal your dose
Air bubbles in the syringe displace medication. A bubble occupying 2 units of space in a syringe meant to deliver 5 units means you are actually injecting only 3 units of medication, a 40% dose reduction. Tap the syringe firmly with your fingernail, flick it upward, and push the plunger gently to expel air before each injection. This simple step can make a meaningful difference in dose accuracy.
Salt forms explained: why the chemistry matters
The salt form issue deserves its own section because it is both widespread and poorly understood. If your compounded semaglutide contains a salt form, the dosing math changes, and not in your favor.
Semaglutide base versus semaglutide sodium
Semaglutide base is the form used in Ozempic and Wegovy. It has a molecular weight of approximately 4,113 daltons. Semaglutide sodium includes a sodium counterion that increases the molecular weight. This means that 1 mg of semaglutide sodium contains less active semaglutide peptide than 1 mg of semaglutide base.
The practical impact: if your pharmacy compounds using semaglutide sodium but doses as if it were semaglutide base, you are receiving somewhere between 5% and 15% less active drug per injection, depending on the specific salt form and how many sodium atoms are incorporated. Over a weekly injection schedule, this underdosing compounds, potentially keeping you below the therapeutic threshold.
Semaglutide acetate
Similar issue, different counterion. Semaglutide acetate has been found in some compounded preparations. Like sodium, the acetate salt adds molecular weight without adding therapeutic activity. The FDA has explicitly stated that these salt forms are different active ingredients from semaglutide base, meaning pharmacies technically need separate approval to compound with them.
How to find out what you are taking
Contact your pharmacy and ask two specific questions. What form of semaglutide do they use (base, sodium, or acetate)? Can they provide a certificate of analysis from their raw material supplier? If the COA lists anything other than "semaglutide" or "semaglutide base," you may have found your problem.
Some pharmacies genuinely may not know which form they are using, especially smaller 503A operations that purchase from intermediate suppliers. This lack of supply chain transparency is itself concerning.
When the problem is not the compound
Not every failure of compounded semaglutide is a compounding problem. After ruling out the pharmaceutical issues above, consider whether biological factors are limiting your response. SeekPeptides provides comprehensive resources to help you troubleshoot from every angle.
Your body may need more time
Semaglutide reaches steady-state blood levels after four to five weeks of consistent weekly injections. Before that point, the drug has not fully accumulated in your system. If you are in your first few weeks, the medication may simply need more time. Check our semaglutide results week by week guide and timeline for semaglutide to work for realistic expectations.
Patience is difficult when you are paying out of pocket for compounded medication. But switching pharmacies or abandoning semaglutide before reaching steady state and adequate dose may mean you never gave it a fair trial.
Medical conditions that reduce response
Several conditions can blunt semaglutide effectiveness regardless of whether it is compounded or brand-name:
Hypothyroidism, especially untreated or undertreated, slows metabolism and counteracts weight loss
Insulin resistance and type 2 diabetes can require higher doses for the same effect
PCOS creates hormonal resistance to weight loss interventions
Cushing syndrome and other cortisol disorders override appetite suppression mechanisms
Certain medications, including some antidepressants, antipsychotics, corticosteroids, and beta-blockers, promote weight gain through independent pathways
If you have any of these conditions, semaglutide may still work but could require higher doses or additional interventions. Discuss these factors with your prescribing provider.
Diet and lifestyle still matter
Semaglutide reduces appetite. It does not eliminate caloric intake. If your diet remains extremely calorie-dense despite reduced hunger, weight loss will be minimal. Our guides on best foods to eat on semaglutide, foods to avoid, and creating a semaglutide diet plan can help you optimize nutrition alongside medication.
Exercise is not strictly required, and some patients lose weight on semaglutide without exercise, but physical activity improves outcomes, preserves muscle mass, and enhances the metabolic benefits of GLP-1 therapy.
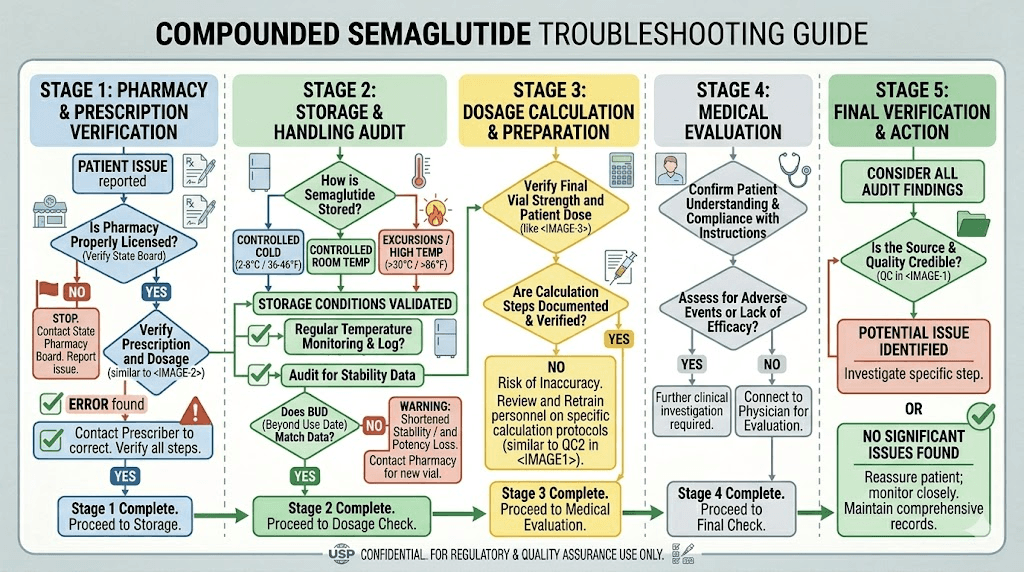
Complete troubleshooting checklist
Work through this checklist systematically. Start with the compounding-specific factors first, then move to general factors if needed.
Step 1: Verify your pharmacy
Confirm state pharmacy license is active and in good standing
Check for PCAB accreditation or USP compliance
Request certificate of analysis for the semaglutide raw material
Confirm they use semaglutide base (not sodium or acetate)
Ask about third-party potency testing on finished product
Step 2: Audit your storage
Check refrigerator temperature with a thermometer (should read 36 to 46 degrees F)
Move vial away from freezer compartment and door
Verify vial is within the beyond-use date
Confirm vial was never frozen
Check if vial was ever left out for extended periods
Verify the solution is clear, colorless, and free of particles, and review what discolored semaglutide means
Step 3: Verify your dose
Confirm the concentration on your vial label
Calculate exact units for your prescribed dose using our semaglutide dosage calculator
Use the smallest syringe that accommodates your dose volume
Eliminate air bubbles before every injection
Verify you are injecting the full amount subcutaneously
Confirm your prescribed dose has been titrated appropriately
Step 4: Evaluate your timeline
Have you been on a stable dose for at least four to five weeks?
Are you on a dose of 1.0 mg or higher? Lower doses often produce minimal appetite suppression
Have you been consistent with weekly timing? See our guide on best time of day to take semaglutide
Step 5: Rule out medical factors
Get thyroid levels checked (TSH, free T3, free T4)
Check for insulin resistance markers (fasting insulin, HOMA-IR)
Review current medications for weight-promoting side effects
Assess for conditions that impair weight loss response
When to switch medications or sources
Sometimes the best answer is not to fix your current compounded semaglutide but to change course entirely. Here are the decision points.
Switch pharmacies, not medications
If you suspect a quality issue with your current pharmacy, try a different compounding source before abandoning semaglutide. The medication itself works, the evidence is overwhelming. The question is whether YOUR specific vial contains effective medication. A switch to a reputable pharmacy like Empower, Olympia, or Belmar with documented quality testing may solve the problem completely.
Try brand-name semaglutide
If your insurance covers it or you can afford the out-of-pocket cost, switching to brand-name Wegovy or Ozempic eliminates every compounding-specific variable on this list. The potency is guaranteed. The dose is mechanically precise. The stability is validated. If you respond well to brand-name after failing on compounded, you have your answer: the compound was the problem, not the drug.
Consider switching to tirzepatide
If semaglutide, whether compounded or brand-name, is genuinely not producing results after adequate dosing and time, tirzepatide may be worth considering. As a dual GIP/GLP-1 receptor agonist, tirzepatide works through additional pathways that some patients respond to more strongly. Clinical trials show tirzepatide produces greater average weight loss than semaglutide at comparable doses.
Our semaglutide to tirzepatide conversion chart can help with the transition, and our semaglutide vs tirzepatide side effects comparison covers what to expect. The complete semaglutide vs tirzepatide comparison examines effectiveness, dosing, and practical differences in depth.
Explore alternative delivery methods
If injection compliance is an issue, oral semaglutide drops, sublingual semaglutide, and semaglutide troches offer non-injection alternatives. These have their own absorption considerations, but some patients find them easier to use consistently. However, be aware that oral bioavailability is significantly lower than injectable, so doses and expectations differ substantially.
What the FDA says about compounded semaglutide
Understanding the regulatory landscape helps put compounded semaglutide in proper context. The FDA has been increasingly vocal about concerns.
During the semaglutide shortage, compounding pharmacies were legally permitted to produce copies under Section 503A and 503B of the Federal Food, Drug, and Cosmetic Act. However, when the FDA declared the semaglutide shortage resolved, the legal basis for most compounding operations changed significantly.
The FDA has issued warnings about adverse events associated with compounded semaglutide, including hospitalizations from dosing errors, contaminated products, and preparations containing unapproved salt forms. The agency has also identified outright fraudulent products, vials with labels listing pharmacies that either do not exist or did not actually make the product.
This does not mean all compounded semaglutide is dangerous or ineffective. But it does mean the risk profile is fundamentally different from FDA-approved products, and patients need to take active responsibility for verifying the quality of what they are injecting.
How to maximize your chances of success with compounded semaglutide
If you choose to continue with compounded semaglutide, these practices significantly improve your odds of getting effective medication.
Choose a 503B outsourcing facility over a 503A pharmacy when possible. The additional FDA oversight and batch testing requirements provide meaningful quality assurance. Ask for and review certificates of analysis before your first injection. Confirm the pharmacy uses semaglutide base, not a salt form.
Invest in a refrigerator thermometer. Place your vial in the back-center of the main compartment, inside its box. Write the beyond-use date on the box. Never use medication past this date, regardless of how much remains.
Use the smallest insulin syringe practical for your dose. A 30-unit syringe for doses under 15 units. A 50-unit syringe for doses between 15 and 40 units. Master the technique of drawing medication without air bubbles. Our syringe injection technique guide walks through the process visually.
Follow the proper titration schedule. Start at 0.25 mg for four weeks, then increase monthly through 0.5, 1.0, 1.7, and 2.4 mg. Do not skip steps, and do not stay at low doses indefinitely. If your prescriber resists titration despite lack of results, advocate for yourself or seek a second opinion.
Track your results objectively. Weigh yourself weekly under consistent conditions (same time, same clothing, same scale). Track waist measurements monthly. Monitor appetite changes in a simple journal. These data points help you and your provider determine whether the medication is working before reaching for a dose change or medication switch.
For researchers serious about getting the most from their GLP-1 protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Members access detailed optimization strategies, dosing calculators, and expert-reviewed troubleshooting resources that go far beyond what you will find in generic online guides.

Frequently asked questions
Is compounded semaglutide as effective as Wegovy or Ozempic?
When properly compounded with pharmaceutical-grade semaglutide base at the correct concentration, stored properly, and dosed accurately, compounded semaglutide should produce similar results. The problem is that "properly compounded" involves many variables that are not guaranteed without FDA oversight. For a full breakdown, see our complete compounded semaglutide guide.
How do I know if my compounded semaglutide has lost potency?
You cannot tell by looking at it. Degraded semaglutide looks identical to fresh semaglutide, clear and colorless. However, visible changes like cloudiness, particles, color changes, or unusual texture definitely indicate a problem. The most reliable indicator of potency loss is reduced effectiveness, specifically, diminished appetite suppression at a dose that previously worked.
Should I switch from compounded to brand-name semaglutide?
If you have tried multiple compounding pharmacies, verified storage and dosing, and still see no results, switching to brand-name eliminates all compounding variables. If brand-name works but compounded did not, the compound quality was the issue. If neither works, the problem lies elsewhere. See how long semaglutide takes to work for realistic timelines.
Can I test my compounded semaglutide for potency at home?
No. Potency testing requires specialized laboratory equipment including HPLC (high-performance liquid chromatography). There are no consumer-grade tests for semaglutide concentration. Your best protection is choosing a pharmacy that conducts and shares third-party testing results.
What if my compounded semaglutide was working but stopped?
This pattern often indicates a batch-to-batch quality variation, which is a known issue with compounded preparations. It could also mean your current vial has degraded due to age or storage conditions. Request a fresh vial, ideally from a recently compounded batch, and verify storage conditions. If the pattern continues, consider whether you have hit a semaglutide plateau that requires dose adjustment rather than a quality issue.
Is it worth paying more for a better compounding pharmacy?
Absolutely. The cheapest compounded semaglutide is often the most expensive in the long run if it does not work. A $150/month product that works is dramatically cheaper than a $99/month product that you take for three months with zero results before switching. Quality compounding pharmacies invest in raw material testing, proper equipment, trained staff, and temperature-controlled shipping. Those costs show up in the price. See our guide on affordable compounded semaglutide for realistic pricing expectations.
Will my compounded semaglutide still work if it was left out overnight?
Probably, but with some potency reduction depending on ambient temperature. A single overnight at normal room temperature (68 to 72 degrees F) typically causes minimal degradation. Warmer environments or repeated temperature excursions cause cumulative damage. Our complete guide on semaglutide left out overnight covers specific scenarios.
Can I switch from semaglutide to tirzepatide if compounded semaglutide is not working?
Yes, many patients who do not respond well to semaglutide find success with tirzepatide. The conversion process is straightforward, though you will restart at a low dose and titrate up. Before switching, make sure you have given properly compounded semaglutide at an adequate dose sufficient time to work. Our guide on whether tirzepatide works when semaglutide fails covers this decision in detail.
External resources
For those navigating the complexities of compounded peptide protocols, SeekPeptides provides evidence-based guides, reconstitution calculators, and a knowledgeable community that has helped thousands troubleshoot exactly these problems.
In case I do not see you, good afternoon, good evening, and good night. May your compounds stay potent, your syringes stay accurate, and your results stay consistent.