Mar 25, 2026

What if the problem is not the peptide itself, but the way you are handling the 5mg vial sitting in your refrigerator right now? Researchers new to cagrilintide face a steep learning curve. The reconstitution math feels unfamiliar.
The dosing schedule looks complicated. And most guides online skip the practical details that actually matter, like how many weeks one vial lasts or what happens when you accidentally add too much bacteriostatic water.
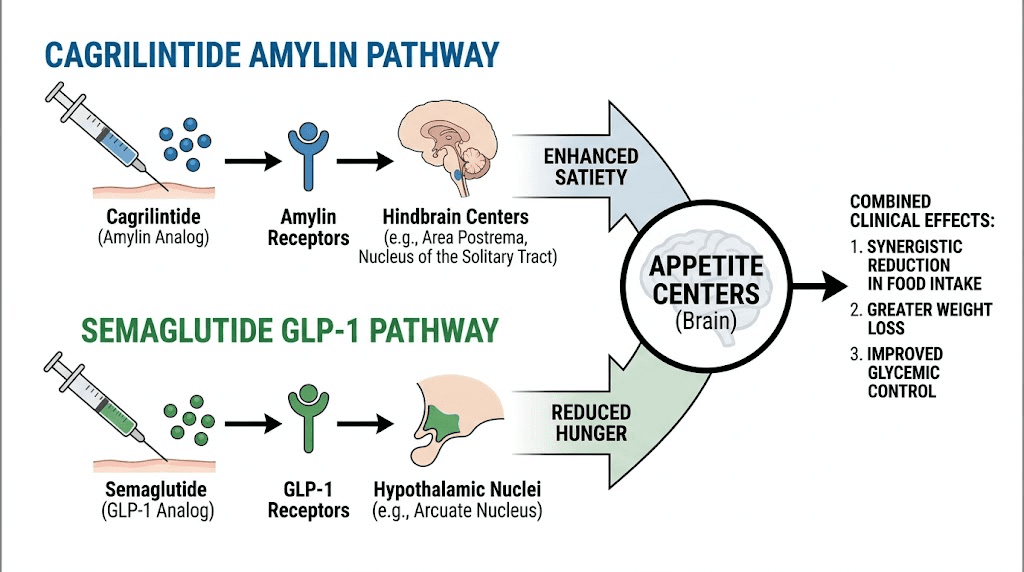
Cagrilintide is not just another weight management peptide. It is a long-acting amylin analog, a completely different mechanism from the GLP-1 receptor agonists that dominate the conversation right now. Where semaglutide and tirzepatide work through incretin pathways, cagrilintide targets amylin receptors in the brain to reduce appetite and slow gastric emptying through an entirely separate signaling cascade. That distinction matters for researchers exploring dual-pathway approaches to metabolic regulation.
The 5mg vial is the most common format available for research purposes. Getting the reconstitution right, calculating accurate doses on an insulin syringe, following the proper titration schedule, and storing the solution correctly are the fundamentals that determine whether a research protocol produces meaningful data or wasted peptide. This guide covers every practical detail for working with cagrilintide 5mg vials, from the moment you open the package to the final injection of a completed protocol. No guesswork. No vague advice. Just the specific numbers, calculations, and procedures you need to run a proper cagrilintide protocol from start to finish.
What is cagrilintide and why does the 5mg vial matter
Cagrilintide is a synthetic analog of amylin, a 37-amino acid peptide hormone co-secreted with insulin from pancreatic beta cells. Novo Nordisk developed it as a long-acting version of the naturally occurring hormone, engineered with specific amino acid substitutions that extend its duration of action from minutes to days. The result is a peptide with a half-life of 159 to 195 hours, roughly 7 to 8 days, making once-weekly administration possible.
That is a massive improvement over pramlintide, the only other amylin analog that reached commercial use. Pramlintide requires three injections per day. Cagrilintide requires one per week.
The 5mg vial is the standard research format for several reasons. It provides enough peptide for approximately 2 to 10 weeks of dosing depending on your protocol stage, it reconstitutes cleanly with standard bacteriostatic water volumes, and it produces concentrations that translate well to measurable units on a standard U-100 insulin syringe. For researchers running their first cagrilintide protocol, the 5mg vial offers the best balance between cost efficiency and practical dosing flexibility. Larger vials exist, but the 5mg format reduces waste if a protocol needs to be adjusted mid-course, and it keeps reconstituted solution within its optimal stability window before a new vial is needed.
How cagrilintide works at the cellular level
Understanding the mechanism helps you appreciate why cagrilintide produces different effects than GLP-1 agonists like semaglutide. The two pathways are complementary, not redundant. That is the entire basis for the CagriSema combination therapy that has generated so much excitement in clinical trials.
Natural amylin acts on three receptor subtypes in the brain. AMY1R, AMY2R, and AMY3R. These receptors sit in the area postrema and the nucleus of the solitary tract, regions that regulate satiety, gastric emptying, and food intake. When amylin binds to these receptors, it triggers a cascade that makes you feel full sooner, slows the rate at which food leaves your stomach, and reduces the hedonic drive to eat, the pleasure-seeking component that makes high-calorie foods irresistible.
Cagrilintide was specifically engineered to retain strong affinity for AMY1R and AMY3R while avoiding the fibrillation problems that plague natural amylin. The engineering involves three key modifications. First, mutations at positions 14 and 17 stabilize the central helix through a salt bridge. Second, proline substitutions at positions 25, 28, and 29 reduce the tendency to form beta-sheet structures that lead to aggregation. Third, a C20 fatty acid chain attached to the N-terminus promotes albumin binding in the bloodstream, which is the modification responsible for the dramatically extended half-life.
The practical implication for researchers is significant. Because cagrilintide works through amylin receptors rather than GLP-1 receptors, it produces appetite suppression through a mechanism that does not overlap with semaglutide or tirzepatide. The brain receives satiety signals from two completely independent pathways. This is why the combination of cagrilintide with semaglutide produced 22.7% body weight reduction in trials, significantly more than either peptide alone.
For researchers focused on fat loss applications, understanding this dual mechanism explains why cagrilintide alone produces meaningful but moderate weight loss (around 11.8% in phase 3 data), while the combination pushes results dramatically higher. The amylin pathway adds a layer of appetite regulation that GLP-1 agonists simply cannot reach on their own.
Reconstituting your cagrilintide 5mg vial step by step
Reconstitution is where most protocol errors begin. Add too much water and your doses become impractically large volumes. Add too little and small measurement errors translate to significant dosing inaccuracies. The goal is a concentration that allows precise dosing with standard insulin syringes across the entire titration range.
What you need before starting
Gather everything before you begin. You will need your cagrilintide 5mg lyophilized vial, a vial of bacteriostatic water (not sterile water, not saline), alcohol swabs, a mixing syringe (1mL or 3mL), and your dosing syringes (U-100 insulin syringes with 29-31 gauge needles). The peptide reconstitution calculator at SeekPeptides can help verify your math before you start.
Use bacteriostatic water specifically. It contains 0.9% benzyl alcohol as a preservative, which inhibits bacterial growth and keeps your reconstituted solution safe for the 3 to 4 weeks you will be drawing from the vial. Sterile water contains no preservative and should only be used for single-draw applications.
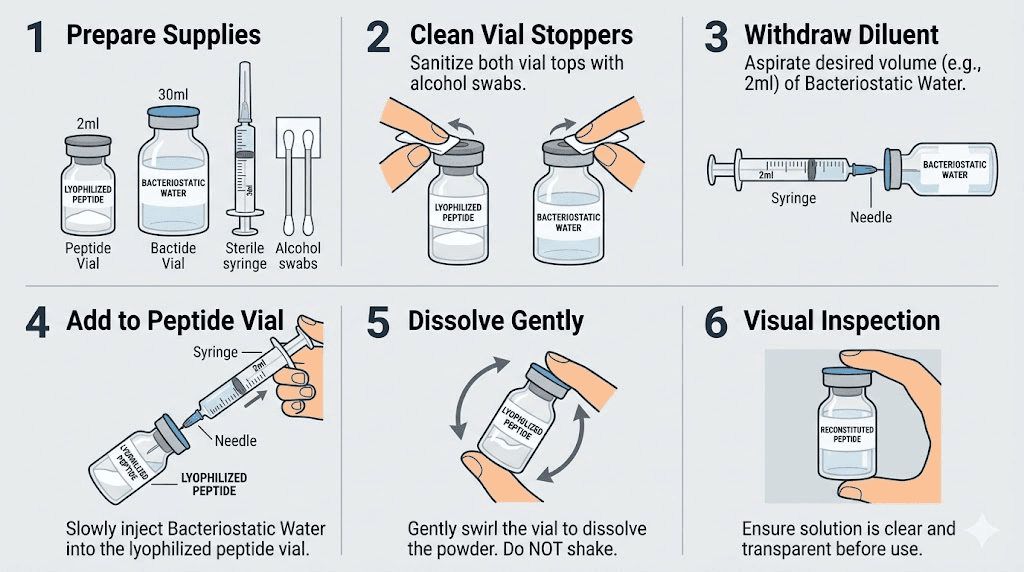
The reconstitution process
Step one. Wipe the top of both the cagrilintide vial and the bacteriostatic water vial with alcohol swabs. Let them dry for 10 seconds.
Step two. Draw 2.0 mL of bacteriostatic water into your mixing syringe. This is the recommended volume for a 5mg vial because it produces a concentration of 2.5 mg/mL, which translates to clean, whole-number dosing on a U-100 syringe.
Some guides recommend 3.0 mL, which gives you 1.67 mg/mL. Both work. But 2.0 mL produces simpler math and smaller injection volumes, which matters during the higher-dose maintenance phase. The tradeoff is slightly less precision at the lowest titration doses, though the difference is clinically negligible.
Step three. Insert the needle through the rubber stopper of the cagrilintide vial. Angle it so the water runs down the inside wall of the glass. Do not blast it directly onto the lyophilized cake. Gentle is the word here. The peptide powder is delicate, and forceful reconstitution can damage the molecular structure.
Step four. Once all the water is in the vial, remove the syringe. Gently swirl the vial in small circles. Do not shake it. Shaking creates foam and can denature the peptide. Swirl for 30 to 60 seconds. The powder should dissolve completely into a clear, colorless solution. If it looks cloudy or has visible particles after 2 minutes of gentle swirling, something went wrong. Do not use that vial.
Step five. Label the vial with the date of reconstitution and the concentration (2.5 mg/mL or 1.67 mg/mL depending on your water volume). This sounds obvious. But three weeks into a protocol, you will be glad you did it.
Concentration reference table
Bacteriostatic water added | Concentration | 0.25mg dose | 0.5mg dose | 1.0mg dose | 2.4mg dose |
|---|---|---|---|---|---|
1.0 mL | 5.0 mg/mL | 5 units | 10 units | 20 units | 48 units |
2.0 mL | 2.5 mg/mL | 10 units | 20 units | 40 units | 96 units |
2.5 mL | 2.0 mg/mL | 12.5 units | 25 units | 50 units | 120 units (exceeds syringe) |
3.0 mL | 1.67 mg/mL | 15 units | 30 units | 60 units | 144 units (exceeds syringe) |
Notice how the 2.0 mL reconstitution keeps all doses within a standard 100-unit insulin syringe range, even at the 2.4mg maintenance dose. At 3.0 mL, the higher doses exceed the syringe capacity and require either a different syringe type or splitting the dose into two injections. For simplicity and accuracy, 2.0 mL is the optimal choice for most cagrilintide 5mg protocols. If you need help with similar calculations for other peptides, the peptide dosage calculator handles the math automatically.
The complete cagrilintide dosing protocol
Cagrilintide follows a slow titration schedule. This is not optional. Jumping to the maintenance dose immediately leads to significant gastrointestinal side effects that can derail an entire protocol. The gradual approach allows the body to adapt to the amylin receptor activation, and clinical trial data shows that nausea rates drop substantially with proper dose escalation.
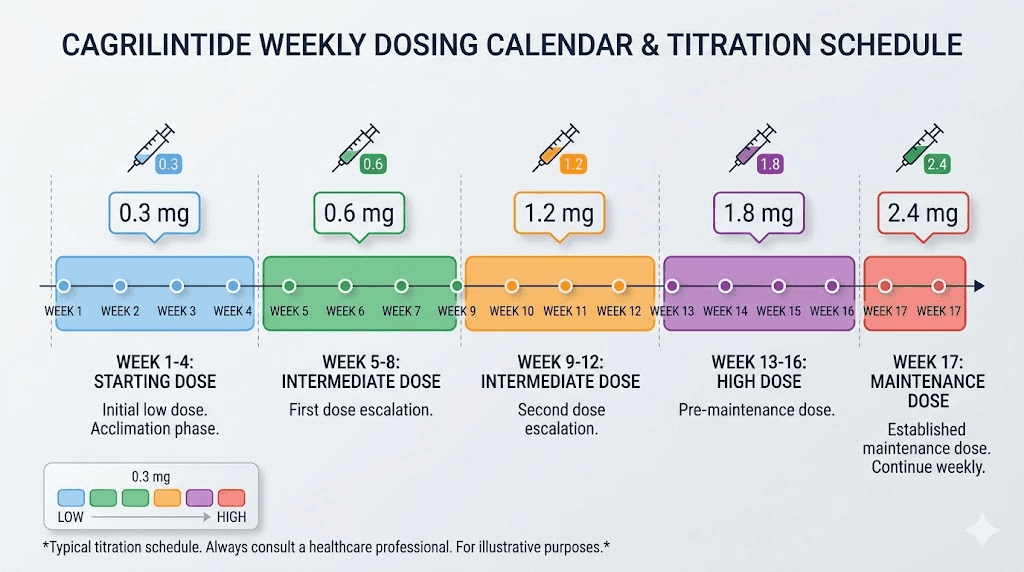
Standard titration schedule
The protocol used in phase 2 and phase 3 clinical trials follows this pattern:
Period | Weekly dose | Units per injection (at 2.5 mg/mL) | Purpose |
|---|---|---|---|
Weeks 1-4 | 0.25 mg | 10 units | Initial tolerance assessment |
Weeks 5-8 | 0.50 mg | 20 units | Early dose escalation |
Weeks 9-12 | 1.0 mg | 40 units | Mid-range escalation |
Weeks 13-16 | 1.7 mg | 68 units | Advanced escalation |
Week 17 onward | 2.4 mg | 96 units | Maintenance dose |
Each dose step lasts four weeks. That gives the gastrointestinal system time to adapt before increasing the amylin receptor stimulation. Researchers who try to accelerate this schedule almost always regret it. The nausea at higher doses without adequate adaptation can be severe enough to force protocol abandonment entirely.
The 2.4 mg weekly maintenance dose is where the phase 3 monotherapy trials showed the strongest results, with an average body weight reduction of 11.8% over 68 weeks. Some research protocols have explored doses up to 4.5 mg weekly, which showed slightly greater efficacy in the phase 2 dose-finding trial but also increased side effect burden. For most research purposes, 2.4 mg represents the optimal balance between efficacy and tolerability.
Injection day and timing
Choose one day per week and stick with it. Consistency matters with long-acting peptides. The half-life of 159 to 195 hours means there is some flexibility, a day early or late will not ruin the protocol, but regular weekly timing produces the most stable plasma levels. The best time to take your weekly injection is whenever you will remember it consistently, though many researchers prefer mornings.
Unlike some peptides that have optimal timing windows, cagrilintide does not appear to show significant differences based on time of day. Morning, afternoon, or evening all produce comparable pharmacokinetic profiles. Pick whatever fits your schedule.
What if you miss a dose
If you miss your scheduled injection day by 1 to 2 days, take it as soon as you remember and resume your regular schedule the following week. If more than 3 days have passed since the missed dose, skip it entirely and take the next scheduled dose on your regular day. Do not double up. The long half-life means you still have circulating cagrilintide even several days after a missed dose, though at reduced levels.
Calculating doses on a syringe from a 5mg vial
Syringe math intimidates new researchers, but it follows a simple formula. Once you understand the logic, every calculation becomes automatic. The dosage calculator tools can verify your work, but knowing the underlying math protects against errors.
The core formula
Units to draw = (desired dose in mg / concentration in mg per mL) x 100
For a 5mg vial reconstituted with 2.0 mL of bacteriostatic water, the concentration is 2.5 mg/mL. Every unit on a U-100 insulin syringe equals 0.01 mL. So each unit contains 0.025 mg (25 mcg) of cagrilintide.
Quick math for common doses at 2.5 mg/mL:
0.25 mg: 0.25 / 2.5 x 100 = 10 units
0.50 mg: 0.50 / 2.5 x 100 = 20 units
1.0 mg: 1.0 / 2.5 x 100 = 40 units
1.7 mg: 1.7 / 2.5 x 100 = 68 units
2.4 mg: 2.4 / 2.5 x 100 = 96 units
If you reconstituted with 3.0 mL instead (1.67 mg/mL), the same doses become 15, 30, 60, 102, and 144 units respectively. See the problem? At 1.67 mg/mL, the maintenance dose of 2.4 mg requires 144 units, which exceeds a standard 100-unit syringe. You would need a 1mL syringe or need to split the dose across two injections. That is why the 2.0 mL reconstitution is recommended for 5mg vials.
These calculations follow the same principles used for semaglutide unit conversions and tirzepatide syringe dosing. If you are already familiar with those peptides, the math is identical. Only the concentration values change.
Reading the syringe correctly
U-100 insulin syringes come in three common sizes: 30-unit (0.3 mL), 50-unit (0.5 mL), and 100-unit (1.0 mL). For the titration phase (weeks 1-8), a 30-unit or 50-unit syringe provides the finest graduation marks and best precision. For the maintenance phase at 96 units, you will need a 100-unit syringe.
Each small line on a 100-unit syringe represents 2 units. Each small line on a 50-unit syringe represents 1 unit. The finer graduations on smaller syringes allow more precise measurements during the low-dose titration phase. Researchers working with syringe-based dosing for other peptides will recognize this principle. Precision at low volumes matters.
A practical tip. Draw slightly past your target mark, then push the plunger back down to the exact line. This eliminates air bubbles at the needle hub and ensures you are injecting the full measured volume. The same technique applies to semaglutide syringe injections and any other subcutaneous peptide protocol.
How long one 5mg vial lasts at each dose
Budget planning matters. Knowing exactly how many vials you need for a complete protocol prevents interruptions and helps you compare costs accurately against other weight management peptides. The peptide cost calculator can help with detailed cost projections.
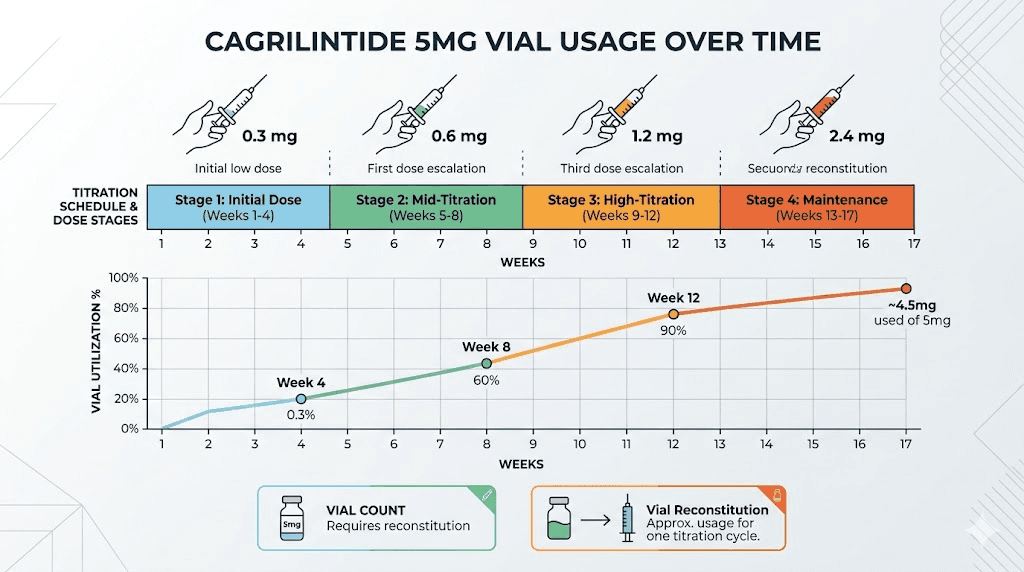
Dose stage | Weekly dose | Doses per 5mg vial | Weeks per vial |
|---|---|---|---|
Titration stage 1 | 0.25 mg | 20 | 20 weeks |
Titration stage 2 | 0.50 mg | 10 | 10 weeks |
Titration stage 3 | 1.0 mg | 5 | 5 weeks |
Titration stage 4 | 1.7 mg | 2.9 | About 3 weeks |
Maintenance | 2.4 mg | 2.08 | About 2 weeks |
Here is the important detail most guides miss. A reconstituted vial is only stable for about 30 days in the refrigerator. During the low-dose titration stages, you will use far less than 5mg in 30 days. At the 0.25 mg starting dose, a single vial contains 20 weeks of doses, but the reconstituted solution will degrade before you use it all.
The practical solution: use a smaller reconstitution volume (1.0 mL) during the early titration phase so the total solution volume is smaller and you draw more per dose (relatively), or accept that you will waste some peptide during the titration stages. Many researchers split a 5mg vial with a training partner or use the stacking calculator to plan their supply needs across multiple peptides.
For a complete 17-week titration protocol to reach maintenance, you will need approximately 24.35 mg total. That is roughly 5 vials. For ongoing maintenance at 2.4 mg per week, you need about 2.5 vials per month. These numbers help you plan supply procurement and purchasing decisions before starting a protocol.
Storage and stability after reconstitution
Proper storage is the difference between a peptide that works and expensive water. Cagrilintide follows the same general storage principles as other lyophilized peptides, with a few specific considerations that matter.
Before reconstitution
Lyophilized (freeze-dried) cagrilintide should be stored at -20 degrees Celsius for long-term storage. If you plan to use it within a few weeks, refrigerator temperatures of 2 to 8 degrees Celsius are acceptable. Keep it away from light and moisture. The lyophilized form is relatively stable, but heat and humidity are its enemies.
Never store unreconstituted peptides at room temperature for extended periods. A day or two during shipping is fine. Weeks on a shelf is not. This applies equally to cagrilintide, compounded semaglutide, tirzepatide, and every other research peptide in your inventory.
After reconstitution
Once reconstituted with bacteriostatic water, store the vial in the refrigerator at 2 to 8 degrees Celsius. Use it within 30 days. After 30 days, discard any remaining solution regardless of how much is left. The preservative in bacteriostatic water slows bacterial growth but does not stop it indefinitely, and the peptide itself gradually degrades in solution.
Do not freeze reconstituted cagrilintide. Freezing and thawing can cause the peptide to aggregate and lose potency. This is a different situation from the lyophilized form. Once it is in solution, freezing is harmful. The same principle applies to reconstituted semaglutide and reconstituted tirzepatide.
What if you accidentally leave it out? Brief temperature excursions of a few hours are unlikely to cause significant degradation. Overnight at room temperature is concerning but probably not catastrophic, similar to the situation researchers face when they accidentally leave semaglutide out. Extended room temperature exposure of 24 hours or more means you should discard the vial. When in doubt, start a fresh vial. The cost of a replacement is always less than the cost of running a protocol with degraded peptide.
Storage checklist
Lyophilized vials: -20 degrees C long-term, 2-8 degrees C short-term
Reconstituted vials: 2-8 degrees C only, use within 30 days
Never freeze reconstituted solution
Protect from light at all stages
Label every vial with reconstitution date and concentration
Discard any vial exposed to room temperature for more than 12 hours
Injection technique and site selection
Cagrilintide is administered subcutaneously, meaning the needle goes into the fat layer just beneath the skin. Not into muscle. The injection technique is identical to what you would use for semaglutide injections or tirzepatide stomach injections.
Preferred injection sites
The abdomen is the most common site, specifically the area around the navel but at least 2 inches away from it. The front of the thighs and the back of the upper arms are also suitable. Rotate between these sites weekly to minimize injection site reactions and prevent lipodystrophy (changes in the fat tissue at the injection site).
For GLP-1 and amylin analog injections, the abdomen generally provides the most consistent absorption. But individual variation exists. Some researchers find that thigh injections produce fewer injection site reactions, while others prefer the abdomen for its reliability. The key is consistency within your rotation pattern.
The injection process
Clean the injection site with an alcohol swab. Let it dry completely, this takes about 10 seconds. Pinch a fold of skin between your thumb and index finger. Insert the needle at a 45 to 90 degree angle (90 degrees for most people with adequate subcutaneous tissue, 45 degrees for leaner individuals). Push the plunger slowly and steadily. Hold for 5 to 10 seconds after the plunger is fully depressed. Withdraw the needle and release the skin fold. Do not rub the injection site.
If you see a small drop of blood at the injection site, that is normal. You nicked a capillary. Apply gentle pressure with a cotton ball for 30 seconds. If the injection stings more than usual, you may have gone slightly too shallow or hit a nerve ending. Adjusting the angle by a few degrees on your next injection usually resolves this.
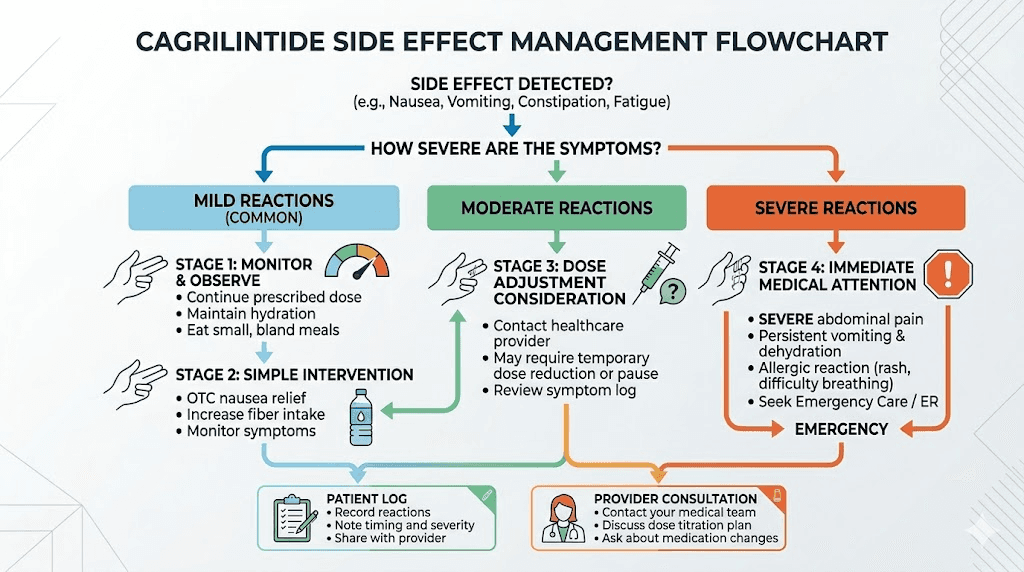
Side effects and how to manage them
Every peptide has side effects. Cagrilintide is no exception. But the profile is predictable, manageable, and for most researchers, temporary. Understanding what to expect and how to respond keeps protocols on track rather than abandoned prematurely.
Gastrointestinal effects
Nausea is the most common side effect, affecting roughly 41% of participants in the REDEFINE 1 trial. But context matters. Most of that nausea is mild to moderate and concentrated in the first few weeks of each dose escalation. It typically resolves within 3 to 7 days as the body adapts.
Constipation affects about 18% of users. This is a predictable consequence of slowed gastric emptying, one of the mechanisms through which cagrilintide reduces food intake. Constipation management strategies that work for GLP-1 users, fiber supplementation, adequate hydration, and mild osmotic laxatives, apply equally to cagrilintide protocols. The same approaches used for tirzepatide-related constipation are effective here.
Diarrhea occurs in about 23% of cases. Vomiting in about 20%. These sound alarming, but they are almost always transient and mild.
The slow titration schedule exists specifically to minimize these effects.
Researchers who skip titration steps or escalate doses too quickly experience dramatically worse GI symptoms.
Injection site reactions
Mild redness, swelling, or itching at the injection site occurs in a subset of users. These reactions are typically self-limiting and resolve within 24 to 48 hours. Rotating injection sites is the primary prevention strategy. Applying ice for 5 minutes before the injection can reduce discomfort, and ensuring proper injection technique minimizes tissue irritation.
Fatigue and energy changes
Some researchers report fatigue, particularly during the first 2 weeks at a new dose level. This parallels the fatigue reported with GLP-1 agonists and semaglutide-specific tiredness. The mechanism is likely related to reduced caloric intake and metabolic adjustment rather than a direct drug effect. Adequate protein intake and appropriate supplementation can help manage energy levels during adaptation.
Headaches
Headaches are reported occasionally, similar in frequency and character to the headaches associated with GLP-1 therapy. Adequate hydration is the first-line response. Dehydration from reduced food and fluid intake is often the actual culprit rather than the peptide itself.
When to pause or stop
Most side effects resolve with time and do not require protocol changes. However, severe persistent vomiting (more than 3 episodes per day for more than 2 days), signs of pancreatitis (severe abdominal pain radiating to the back), or allergic reactions (hives, difficulty breathing, facial swelling) warrant immediate protocol cessation and medical attention.
If nausea is persistent but not severe, dropping back one titration step for an additional 2 to 4 weeks before attempting the increase again often resolves the issue. The body may simply need more time to adapt at that particular dose level. This is a strategy that also works well for managing tirzepatide tolerance and navigating semaglutide adjustments.
Cagrilintide compared to other weight management peptides
Researchers choosing between peptides need clear, honest comparisons. Cagrilintide occupies a unique position in the weight management landscape because it works through a mechanism that no other commonly available peptide targets. But unique mechanism does not automatically mean superior results. Context determines the best choice.
Cagrilintide vs semaglutide
Semaglutide is the dominant GLP-1 receptor agonist, with extensive clinical data showing approximately 15-17% body weight reduction at the 2.4 mg weekly dose. Cagrilintide monotherapy produces about 11.8% reduction at 2.4 mg weekly. On paper, semaglutide wins the head-to-head comparison for monotherapy weight loss.
But that is not the whole story. Cagrilintide targets amylin receptors, producing satiety through a different neural pathway. Some researchers who experience inadequate appetite suppression with semaglutide alone or who are not losing weight on GLP-1 monotherapy find that the amylin pathway provides the additional appetite regulation they need. The mechanisms are complementary, not competitive.
Factor | Cagrilintide 2.4mg | Semaglutide 2.4mg | Tirzepatide 15mg |
|---|---|---|---|
Mechanism | Amylin receptor agonist | GLP-1 receptor agonist | GLP-1 + GIP dual agonist |
Weight loss (monotherapy) | ~11.8% | ~15-17% | ~22.5% |
Frequency | Once weekly | Once weekly | Once weekly |
Titration period | 16 weeks | 16 weeks | 20 weeks |
Primary side effects | Nausea, GI issues | Nausea, GI issues | Nausea, GI issues |
Combination potential | High (different pathway) | High (pairs with amylin) | Moderate |
Cagrilintide vs tirzepatide
Tirzepatide is a dual GIP/GLP-1 agonist that produces the highest monotherapy weight loss results currently available, approximately 22.5% at the maximum dose. It targets two incretin pathways simultaneously. Compared to cagrilintide monotherapy, tirzepatide delivers roughly twice the weight loss percentage.
However, cagrilintide and tirzepatide work through entirely different receptor systems. A researcher who has already maximized their tirzepatide dosing and hit a plateau might theoretically benefit from adding amylin pathway stimulation. This combination has not been studied in clinical trials, so any such approach would be highly experimental. The studied combination is cagrilintide plus semaglutide, not cagrilintide plus tirzepatide.
Cagrilintide vs retatrutide
Retatrutide is a triple agonist targeting GLP-1, GIP, and glucagon receptors simultaneously. Early trial data shows weight loss exceeding 24% at higher doses. Like tirzepatide, it works through incretin pathways, meaning it occupies different receptor territory than cagrilintide.
For researchers tracking the availability of retatrutide and comparing options, the key distinction remains the same. Cagrilintide is the only available amylin pathway option. Everything else, semaglutide, mazdutide, survodutide, tirzepatide, retatrutide, works through some combination of incretin receptors. If your research interest is specifically in amylin receptor activation, cagrilintide is currently the only viable long-acting option.
Stacking cagrilintide with semaglutide
The combination of cagrilintide and semaglutide, known as CagriSema in Novo Nordisk clinical trials, represents the most exciting development in peptide weight management research. The rationale is straightforward. Two different satiety pathways, activated simultaneously, produce greater appetite suppression than either pathway alone.
What the CagriSema trials showed
The REDEFINE 1 trial published in the New England Journal of Medicine delivered remarkable numbers. At 68 weeks, participants receiving the cagrilintide-semaglutide combination achieved a mean body weight reduction of 22.7% under the trial product estimand. That compared to 16.1% for semaglutide alone, 11.8% for cagrilintide alone, and 2.3% for placebo.
Read those numbers again. The combination did not just add the individual effects together. The 22.7% result exceeded what simple addition would predict (11.8 + 16.1 = 27.9, minus baseline placebo effect). But the clinical significance is that 60% of combination participants achieved at least 20% weight loss, and roughly 23% achieved 30% or more. Those are transformative results.
The REDEFINE 2 trial examined the combination in adults with type 2 diabetes, where weight loss is typically harder to achieve due to insulin resistance and medication effects. Even in this more challenging population, the combination produced 13.7% weight reduction versus 3.4% for placebo, along with dramatic improvements in glycemic control. 73.5% of combination participants achieved a glycated hemoglobin level of 6.5% or less.
Practical stacking considerations
Researchers interested in replicating a CagriSema-like protocol need to manage two separate peptides with their own reconstitution, storage, and dosing requirements. The semaglutide reconstitution process follows similar principles to cagrilintide, though the concentrations and volumes differ.
Key considerations for a dual protocol:
Titrate each peptide separately. Start cagrilintide first, reach at least the 0.5 mg dose, then begin semaglutide titration. This isolates side effects and allows you to identify which peptide causes any issues.
Different injection sites. Inject each peptide in a different location on the same day. Do not mix them in the same syringe.
Same day dosing. Both peptides are once-weekly. Take them on the same day for simplicity. There is no pharmacokinetic reason to separate the injection days.
Monitor GI effects carefully. The combination increases gastrointestinal side effects. In the REDEFINE 1 trial, 79.6% of combination participants experienced GI events versus 39.9% on placebo. Slower titration may be necessary.
For researchers already running a semaglutide protocol, adding cagrilintide involves a separate titration curve starting from the 0.25 mg dose. You do not need to restart your semaglutide from scratch. The peptide stacking calculator can help plan supply needs for dual protocols, and SeekPeptides members access detailed combination protocol guidance that accounts for individual factors most generic guides ignore.
What the clinical trials actually show
Claims without data are worthless. Here is what the published research demonstrates about cagrilintide, with specific numbers from specific studies.
Phase 2 dose-finding trial
Published in The Lancet in 2021, this multicenter, randomized, double-blind trial tested cagrilintide doses ranging from 0.3 mg to 4.5 mg weekly in people with overweight or obesity. The results showed clear dose-dependent weight loss. The 0.3 mg dose produced 6.0% reduction. The 4.5 mg dose produced 10.8% reduction (11.5 kg average). For comparison, liraglutide 3.0 mg (the active comparator) produced 9.0% reduction.
The highest dose outperformed liraglutide, which was notable because liraglutide was, at the time, a leading GLP-1 obesity treatment. This trial established proof of concept that amylin receptor agonism alone could produce clinically meaningful weight loss.
Phase 3 monotherapy results
A post-hoc analysis of the phase 3 data showed cagrilintide 2.4 mg monotherapy produced 11.8% average body weight reduction versus 2.3% for placebo over 68 weeks. 31.6% of cagrilintide participants achieved greater than 15% weight loss, compared to just 4.7% on placebo. These results position cagrilintide monotherapy as less effective than semaglutide for raw weight loss but still clinically significant and far superior to placebo.
REDEFINE 1 (combination, no diabetes)
The landmark trial. 3,417 adults with overweight or obesity but without type 2 diabetes. Cagrilintide 2.4 mg plus semaglutide 2.4 mg versus each component alone versus placebo. The combination achieved 20.4% mean body weight reduction from baseline to week 68 (22.7% under the trial product estimand that accounts for discontinuation). 60% of combination participants reached the 20% or more weight loss threshold. 23% reached 30% or more.
These numbers rival and in some analyses exceed the results seen with tirzepatide weight loss in the SURMOUNT trials. The combination approach of targeting two separate pathways produces results competitive with even the most effective single-agent therapies currently available.
REDEFINE 2 (combination, with type 2 diabetes)
In adults with type 2 diabetes, the combination achieved 13.7% weight reduction versus 3.4% for placebo. The glycemic improvements were equally impressive, with 73.5% of combination participants achieving HbA1c levels at or below 6.5%. For context, that level of glycemic control often allows patients to reduce or eliminate other diabetes medications. Researchers tracking metabolic effects of peptide therapy should note these results carefully.
Safety across all trials
The side effect profile is consistent across studies. GI effects are the primary concern, primarily nausea, vomiting, diarrhea, and constipation. These are predominantly mild to moderate and transient. Serious adverse events were low and comparable to placebo in most analyses. No clinically relevant QTc prolongation was observed in the thorough QT study. The profile is broadly comparable to, though not identical to, the side effect profiles of semaglutide and tirzepatide.
Diet and lifestyle considerations during a cagrilintide protocol
A peptide is not a substitute for proper nutrition. It is a tool that makes proper nutrition easier by reducing appetite and slowing gastric emptying. But what you eat while your appetite is suppressed matters enormously for body composition, energy levels, and long-term outcomes.
Protein is non-negotiable
When appetite is significantly reduced, protein intake often drops first. This is a problem. Inadequate protein during a caloric deficit leads to muscle loss rather than fat loss, defeats the purpose of the protocol, and creates the metabolic slowdown that causes weight loss plateaus.
Target a minimum of 0.7 to 1.0 grams of protein per pound of body weight per day. Yes, even when you are not hungry. Protein shakes, Greek yogurt, lean meats, and eggs become staples during any aggressive weight management protocol. The semaglutide diet planning principles apply directly to cagrilintide protocols. So do the food recommendations and foods to avoid during GLP-1 and amylin therapy.
Hydration
Reduced food intake means reduced water intake from food sources. Deliberately increase fluid consumption to compensate. Dehydration exacerbates nausea, causes headaches (often misattributed to the peptide itself), and can lead to dizziness and lightheadedness. Aim for at least 2 to 3 liters of water daily.
Micronutrient supplementation
Caloric restriction inevitably reduces micronutrient intake. A quality multivitamin covers the basics. Beyond that, consider B12 supplementation (particularly relevant for researchers also using B12-enhanced semaglutide compounds or tirzepatide-B12 formulations), magnesium for GI regularity, and electrolytes to prevent the mineral imbalances that come with reduced food volume.
Exercise during the protocol
Resistance training is more important than cardio during a caloric deficit supported by appetite-suppressing peptides. Weight training preserves muscle mass, maintains metabolic rate, and improves body composition beyond what the scale shows. Research on whether you can lose weight on peptides without exercise shows that while weight loss occurs regardless, body composition outcomes are dramatically better with resistance training included.
For researchers serious about maximizing their protocol results, SeekPeptides provides detailed diet frameworks, supplementation guides, and exercise recommendations tailored specifically to peptide-supported weight management. The combination of proper nutrition with an optimized peptide protocol is what separates transformative results from mediocre ones.
Understanding purity and quality for research peptides
Not all cagrilintide is created equal. The research peptide market operates with minimal regulatory oversight, which means quality varies dramatically between suppliers. Researchers who purchase cagrilintide without understanding quality indicators are gambling with their protocol outcomes.
What to look for in a certificate of analysis
Every reputable supplier provides a Certificate of Analysis (COA) with each batch. The COA should include HPLC purity data (target: 98% or higher), mass spectrometry confirmation of molecular identity, and ideally, endotoxin testing results. Independent third-party testing from laboratories like Janoshik or ACS Labs carries more credibility than in-house testing alone.
A critical finding from independent peptide testing: GLP-1 receptor agonists and related peptides show the highest discrepancy rates of any peptide category, with only 27% of supplier-provided COAs exactly matching independent laboratory results. That means roughly 3 out of 4 COAs contain some degree of inaccuracy. This does not necessarily mean the peptide is dangerous, but it means stated purity numbers should be taken with a grain of salt unless verified independently. The same quality considerations apply to compounded semaglutide and research versus pharmaceutical grade peptides generally.
Red flags to watch for
No COA available. Walk away. Every legitimate supplier tests their products.
COA without HPLC chromatogram. A purity number without the supporting chromatogram is just a claim, not evidence.
Unusually low prices. Cagrilintide synthesis is complex and expensive. If the price seems too good to be true, the quality likely reflects the cost savings.
No batch-specific testing. A single COA reused across multiple batches means they are not testing each production run independently.
Salt form discrepancies. Cagrilintide should be provided as the base form or clearly labeled if provided as a salt (acetate, sodium). Different salt forms have different effective potencies, and unlabeled salt forms introduce dosing uncertainty.
Testing your own peptides through services like ACS Peptide Testing Labs or Peptide Test costs between $50 and $150 per sample, and is worth doing at least once when establishing a new supplier relationship. Consider it insurance for your entire protocol investment.
Frequently asked questions
How much bacteriostatic water do I add to a cagrilintide 5mg vial?
The recommended volume is 2.0 mL, which creates a 2.5 mg/mL concentration. This keeps all titration and maintenance doses within the range of a standard 100-unit insulin syringe. Using 3.0 mL (1.67 mg/mL) is also acceptable but requires larger syringes at higher doses. Use the reconstitution calculator to verify your specific setup.
How many weeks does one cagrilintide 5mg vial last?
It depends entirely on your dose stage. At the starting dose of 0.25 mg weekly, one vial contains 20 doses (20 weeks). At the maintenance dose of 2.4 mg weekly, one vial lasts approximately 2 weeks. However, reconstituted solution should be used within 30 days regardless of remaining volume.
Can I use cagrilintide and semaglutide at the same time?
Yes, this combination has been extensively studied in clinical trials under the name CagriSema. The REDEFINE trials showed the combination produces approximately 22.7% body weight reduction, significantly more than either peptide alone. They work through different receptor pathways (amylin and GLP-1 respectively) and are injected separately at different sites on the same day. See the CagriSema comparison guide for detailed combination protocol information.
What is the difference between cagrilintide and semaglutide?
Semaglutide is a GLP-1 receptor agonist. Cagrilintide is an amylin receptor agonist. They target completely different receptor systems in the brain to reduce appetite through separate neural pathways. Semaglutide produces greater weight loss as monotherapy (15-17% vs 11.8%), but the two peptides can be combined for additive effects. Read the comparison guides for more context on how different weight management peptides relate to each other.
Do I need to refrigerate cagrilintide after reconstitution?
Yes, absolutely. Reconstituted cagrilintide must be stored at 2-8 degrees Celsius (standard refrigerator temperature) and used within 30 days. Never freeze reconstituted solution. This is consistent with storage requirements for tirzepatide and semaglutide after reconstitution.
What are the most common cagrilintide side effects?
Gastrointestinal effects dominate the side effect profile. Nausea (41%), diarrhea (23%), vomiting (20%), and constipation (18%) are the most frequently reported. Injection site reactions occur less commonly. Most side effects are mild to moderate and improve with continued use. Following the standard titration schedule significantly reduces severity. See the complete cagrilintide side effects guide for detailed management strategies.
How long does it take for cagrilintide to start working?
Appetite suppression typically becomes noticeable within the first 1 to 2 weeks of dosing. Meaningful weight loss generally requires reaching at least the 1.0 mg weekly dose, which takes about 8 weeks with standard titration. Full results in clinical trials were measured at 68 weeks. Unlike semaglutide appetite suppression, which some researchers notice within days, cagrilintide onset tends to be more gradual.
Is cagrilintide FDA approved?
As of early 2026, cagrilintide is not FDA approved as a standalone medication. The CagriSema combination (cagrilintide plus semaglutide) is under FDA review following the positive REDEFINE trial results. Cagrilintide is currently available through research peptide suppliers for investigational use only.
External resources
Phase 2 trial: Once-weekly cagrilintide for weight management (PubMed)
REDEFINE 1: Coadministered cagrilintide and semaglutide in adults with overweight or obesity (NEJM)
Development of cagrilintide, a long-acting amylin analogue (Journal of Medicinal Chemistry)
For researchers serious about building evidence-based peptide protocols, SeekPeptides offers the most comprehensive resource available. Members access detailed protocol builders, dosing calculators, combination therapy guidance, and a community of thousands of experienced researchers who have navigated these exact questions. Whether you are running your first cagrilintide protocol or optimizing a complex multi-peptide stack, SeekPeptides provides the tools and knowledge to do it right.
In case I do not see you, good afternoon, good evening, and good night. May your reconstitutions stay precise, your titrations stay patient, and your protocols stay productive.