Mar 23, 2026

At the molecular level, retatrutide does something no other weight loss peptide can do. It activates three receptors simultaneously. GLP-1. GIP. And glucagon. That triple mechanism produced up to 28.7% body weight reduction in Phase 3 trials, numbers that stunned even the researchers who designed the study. But here is the question nobody expected people to ask: can you deliver this molecule through your nose instead of a needle?
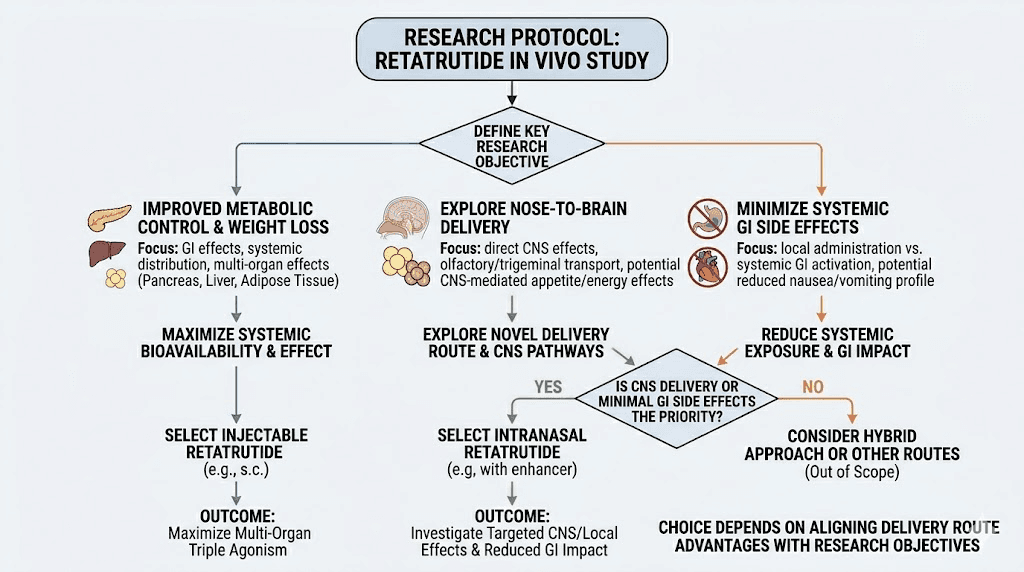
The answer is more complicated than a simple yes or no. Intranasal retatrutide represents an emerging area of peptide delivery research that sits at the intersection of convenience and biological reality. The nasal mucosa offers a direct pathway to systemic circulation, bypassing first-pass metabolism entirely. That sounds promising. It is promising. But the retatrutide molecule weighs approximately 4,895 daltons, and that molecular weight creates absorption challenges that every researcher needs to understand before pursuing this route. SeekPeptides members have been asking about nasal delivery with increasing frequency, and this guide addresses every aspect of intranasal retatrutide, from the pharmacokinetics to the practical preparation, from the bioavailability limitations to the strategies researchers use to overcome them.
How retatrutide works as a triple-agonist peptide
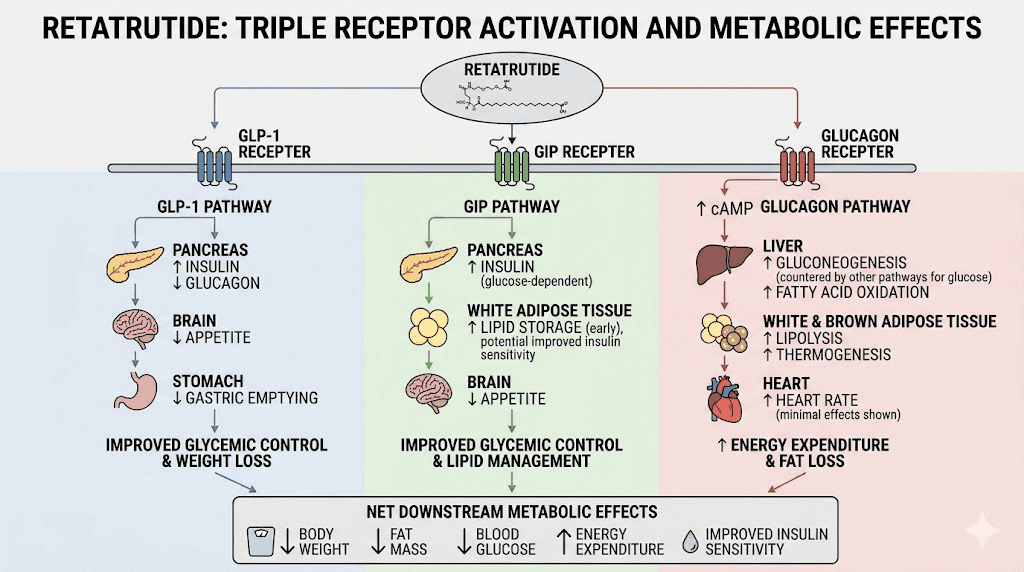
Understanding why intranasal delivery matters requires understanding what retatrutide actually does inside the body. Most GLP-1 receptor agonists like semaglutide target a single receptor. Tirzepatide advanced the field by targeting two receptors, GLP-1 and GIP. Retatrutide targets all three.
The GLP-1 receptor activation slows gastric emptying, reduces appetite, and improves insulin secretion. The GIP receptor activation enhances fat metabolism and works synergistically with GLP-1 signaling. The glucagon receptor activation, the truly novel component, directly increases energy expenditure and promotes fat oxidation in the liver. This is the mechanism that separates retatrutide from everything else on the market.
In the Phase 3 TRIUMPH-4 trial, participants taking 12mg of retatrutide lost an average of 28.7% of their body weight over 68 weeks. The 9mg group lost 26.4%. Those numbers compare to just 2.1% with placebo. Nearly 40% of participants on the highest dose achieved 30% or greater weight loss. These results make retatrutide the most potent weight loss peptide ever tested in Phase 3 clinical trials.
The molecule itself is a synthetic peptide of approximately 34 amino acids with a molecular formula of C228H350N48O66.
A C18 fatty diacid moiety is conjugated at Lys30 through a chemical linker, which extends the half-life and enables once-weekly dosing. That fatty acid tail is critical for subcutaneous injection efficacy. It is also one of the factors that complicates nasal delivery.
The science behind intranasal peptide delivery
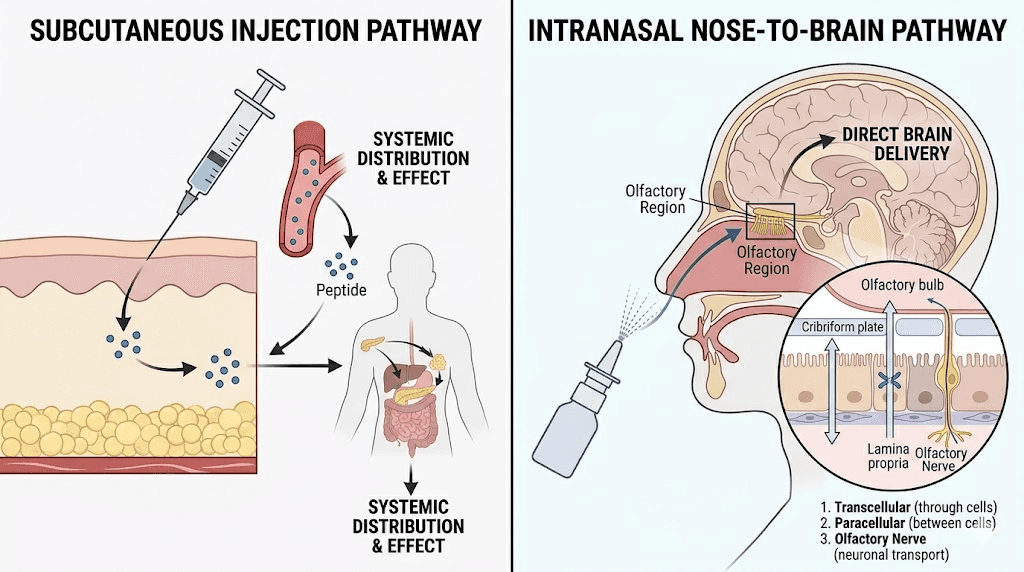
Nasal delivery is not new. Researchers have been exploring intranasal routes for peptide administration for decades, and several successful products exist. Desmopressin nasal spray for diabetes insipidus. Calcitonin nasal spray for osteoporosis. Oxytocin nasal spray for research applications. The nasal mucosa provides a rich blood supply, a large surface area of approximately 150 square centimeters, and direct access to systemic circulation without hepatic first-pass metabolism.
But not all peptides are created equal when it comes to nasal absorption.
The nasal epithelium acts as a selective barrier. Small molecules pass through relatively easily. Peptides under 1,000 daltons achieve reasonable bioavailability through the nasal route. Between 1,000 and 6,000 daltons, absorption becomes increasingly challenging but remains possible with enhancement strategies. Above 6,000 daltons, nasal delivery becomes extremely difficult without advanced formulation technologies.
Retatrutide sits at approximately 4,895 daltons. That places it in the challenging but theoretically achievable range. For context, semaglutide has a molecular weight of approximately 4,114 daltons, and researchers have explored intranasal routes for GLP-1 class peptides with mixed but instructive results.
The nasal absorption pathway
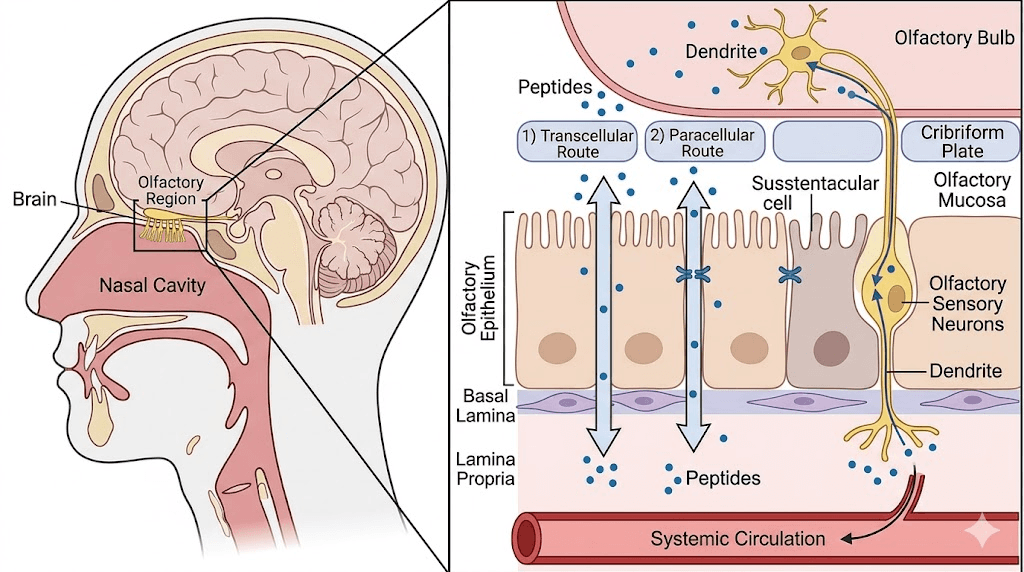
When you spray a peptide solution into the nasal cavity, the molecule encounters several barriers before reaching systemic circulation. The mucus layer comes first. This gel-like coating traps particles and contains enzymes that can degrade peptides before they ever reach the epithelial cells beneath. Mucociliary clearance constantly sweeps material toward the throat, giving the peptide a limited window of approximately 15-20 minutes for absorption.
Beneath the mucus sits the nasal epithelium. Peptides can cross this barrier through two primary routes. The transcellular pathway involves the molecule passing directly through epithelial cells, which requires either small molecular size or active transport mechanisms. The paracellular pathway involves passage between cells through tight junctions, which normally restrict molecules larger than approximately 1,000 daltons.
There is also a third pathway that makes nasal delivery uniquely interesting for certain applications. The olfactory and trigeminal nerve pathways provide a direct nose-to-brain route that bypasses the blood-brain barrier entirely. This pathway is particularly relevant for GLP-1 agonists because GLP-1 receptors in the brain play a crucial role in appetite suppression and metabolic regulation. A 2024 review in Expert Opinion on Drug Delivery specifically highlighted intranasal GLP-1 delivery to the brain as a promising approach for obesity treatment.
Bioavailability: the critical limitation
Here is where the conversation gets real.
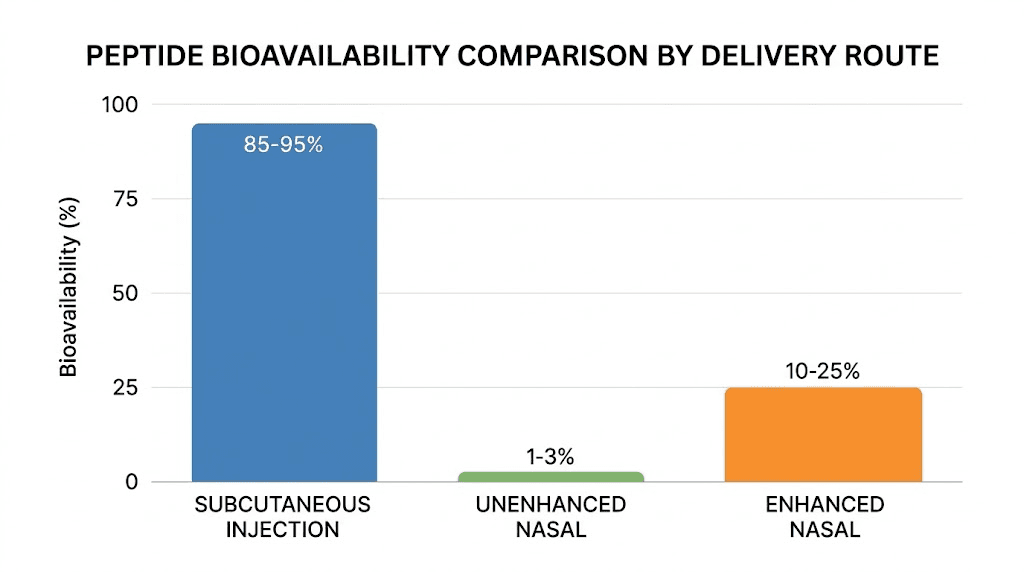
In a landmark clinical trial published in Diabetes Care, Japanese researchers administered intranasal GLP-1 to 26 patients with type 2 diabetes. The bioavailability was 2.7% compared to intravenous administration. That means for every 100 units of peptide sprayed into the nose, only 2.7 units reached systemic circulation in active form. The time to maximum plasma concentration was 8.1 minutes, which is significantly faster than subcutaneous injection but at a dramatically lower total absorption.
Those numbers are sobering. But they tell only part of the story.
The trial used unmodified GLP-1 without absorption enhancers, without nanocarrier formulations, without any of the technologies that modern intranasal peptide research employs to improve bioavailability. When researchers added cyclodextrin absorption enhancers, specifically dimethyl-beta-cyclodextrin at concentrations of just 2% w/v, nasal peptide bioavailability in animal models jumped to 70-100%. Glucagon formulations with 2% cyclodextrin enhancers achieved bioavailability between 42.78% and 44.61%.
The gap between unenhanced and enhanced nasal delivery is enormous. It suggests that the route itself is viable, but the formulation determines everything.
Intranasal retatrutide: current research status
Retatrutide is currently being developed by Eli Lilly as a subcutaneous injection. The Phase 3 clinical trial program includes seven additional studies beyond TRIUMPH-4, all evaluating the injectable formulation. There are no published clinical trials specifically evaluating intranasal retatrutide in humans.
That does not mean intranasal delivery is impossible or unexplored.
Research suppliers have developed nasal spray formulations of retatrutide for investigational use. These formulations are intended for research purposes and are not approved for human consumption. However, the existence of these products reflects genuine scientific interest in alternative delivery routes for triple-agonist peptides.
The theoretical basis for intranasal retatrutide delivery rests on several factors. The molecule falls within the upper range of peptides that can be absorbed nasally with enhancement. The nose-to-brain pathway could potentially deliver retatrutide directly to central appetite-regulating centers. And the avoidance of injection could improve adherence in long-term treatment protocols.
Why researchers are interested in nasal delivery
Needle fatigue is real. In the TRIUMPH-4 trial, discontinuation rates due to adverse events ranged from 12.2% to 18.2% with retatrutide. While gastrointestinal side effects drove most discontinuations, the requirement for weekly injections contributes to long-term compliance challenges. A nasal spray that delivered even a fraction of the injectable dose efficacy could represent a meaningful alternative for certain research applications.
The injection route also presents practical challenges. Cold chain storage requirements, syringe handling, injection site rotation, and the discomfort of subcutaneous administration all add friction to research protocols. Nasal delivery eliminates several of these barriers simultaneously.
There is also the brain delivery angle. When retatrutide is injected subcutaneously, it must cross the blood-brain barrier to reach central GLP-1 receptors. Only a small percentage of the circulating peptide accomplishes this crossing. Intranasal delivery through the olfactory pathway could theoretically deliver retatrutide directly to the brain, potentially achieving central nervous system effects at lower systemic doses. This is speculation based on the broader GLP-1 nasal delivery literature, not confirmed data for retatrutide specifically.
Bioavailability challenges specific to retatrutide
Retatrutide presents unique challenges for nasal delivery that go beyond its molecular weight.
The fatty acid tail problem
The C18 octadecanedioic acid moiety that gives retatrutide its extended half-life through albumin binding also makes the molecule more lipophilic than unmodified peptides. This lipophilicity affects how the molecule interacts with the aqueous mucus layer in the nasal cavity. Lipophilic compounds can become trapped in the mucus rather than diffusing through to the epithelium, reducing absorption efficiency.
The fatty acid conjugation is essential for the once-weekly dosing that makes retatrutide dosing schedules practical. Without it, the peptide would be degraded rapidly and require multiple daily administrations. But for nasal delivery, this structural feature becomes a liability that must be overcome through formulation strategies.
Enzymatic degradation in the nasal cavity
The nasal mucosa contains proteases, enzymes that break down peptide bonds. Aminopeptidases, carboxypeptidases, and endopeptidases all reside in the nasal tissue and actively degrade peptide drugs as they attempt to cross the epithelial barrier. Retatrutide, as a 34-amino-acid peptide, is susceptible to this enzymatic attack at multiple sites along its chain.
The degradation begins immediately upon contact with the nasal mucosa and continues throughout the absorption process. Studies on other peptides of similar size suggest that enzymatic degradation can reduce bioavailability by 50-80% even before accounting for barrier permeability limitations. This is why protease inhibitors are sometimes included in nasal peptide formulations.
Mucociliary clearance timing
The nasal cavity clears deposited material at a rate of approximately 5-6 mm per minute through mucociliary transport. This gives a typical peptide formulation a residence time of 15-20 minutes before it is swept to the nasopharynx and swallowed. For a large peptide like retatrutide that absorbs slowly, this limited window is a significant constraint.
Mucoadhesive formulations can extend this residence time. Chitosan-based formulations, for example, can increase nasal residence time to 60 minutes or longer by adhering to the mucosal surface. Thermogelling polymers that transition from liquid to gel at nasal temperature offer another approach. These strategies are well-established in the nasal drug delivery literature and applicable to retatrutide formulations.
Absorption enhancement strategies for intranasal retatrutide
The gap between unenhanced nasal delivery and injectable delivery is large. But researchers have developed multiple strategies to narrow that gap significantly. Each approach targets a different barrier to absorption, and combining strategies can produce synergistic improvements.
Cyclodextrin complexation
Cyclodextrins are the most extensively studied absorption enhancers for nasal peptide delivery. These cyclic oligosaccharides form inclusion complexes with peptide molecules, temporarily increasing their aqueous solubility and facilitating transport across the nasal epithelium. Dimethyl-beta-cyclodextrin and randomly methylated beta-cyclodextrin are the most effective variants, active at concentrations as low as 2-5% w/v.
In rat studies, dimethyl-beta-cyclodextrin improved nasal peptide absorption to bioavailability levels of 70-100%. Human studies show more modest improvements, reflecting important species differences in nasal permeability. But even in human applications, cyclodextrin-enhanced formulations consistently outperform unenhanced nasal delivery by 5-15 fold.
The safety profile is favorable. Toxicological studies of cyclodextrin effects on the nasal mucosa confirm that dimethyl-beta-cyclodextrin and randomly methylated beta-cyclodextrin are considered safe for nasal use at therapeutic concentrations. This is important because some absorption enhancers achieve their effects through mucosal damage, which is not acceptable for repeated administration.
Cell-penetrating peptides
Cell-penetrating peptides represent a more recent approach to nasal delivery enhancement. These short peptide sequences, typically 5-30 amino acids, facilitate the transport of larger cargo molecules across biological membranes through direct translocation or endocytosis-mediated pathways.
Research published in Frontiers in Pharmacology demonstrated that TCTP-PTD variants and penetratin analogs significantly improved the nasal absorption of exendin-4 and GLP-1 in both normal and diabetic mouse models. These findings are directly relevant to retatrutide because the molecule shares structural similarities with the GLP-1 agonists tested.
The advantage of cell-penetrating peptides over chemical enhancers is specificity. Rather than broadly disrupting the mucosal barrier, they actively shuttle the therapeutic peptide across the epithelium through targeted mechanisms. This could theoretically allow higher retatrutide doses to reach systemic circulation without causing nasal irritation.
Nanocarrier formulations
Nanoparticles, liposomes, and nanoemulsions can encapsulate retatrutide and protect it from enzymatic degradation while facilitating epithelial transport. Chitosan nanoparticles are particularly popular for nasal peptide delivery because chitosan is mucoadhesive (extending residence time) and opens tight junctions between epithelial cells (enhancing paracellular transport).
PLGA nanoparticles offer sustained release characteristics that could compensate for the lower bioavailability of nasal delivery by providing a continuous supply of peptide to the absorption site.
Solid lipid nanoparticles combine the biocompatibility of lipids with the structural advantages of nanoparticle formulations.
The complexity of nanocarrier formulations is both their strength and their weakness. They can dramatically improve bioavailability, but they require sophisticated preparation techniques that may be beyond standard research laboratory capabilities. For most researchers exploring intranasal retatrutide, simpler enhancement strategies offer a more practical starting point.
Focused ultrasound enhancement
An emerging approach uses low-intensity focused ultrasound to temporarily increase blood-brain barrier permeability in conjunction with intranasal administration. This technique, highlighted in the 2024 Expert Opinion on Drug Delivery review, could enhance the nose-to-brain delivery of GLP-1 agonists for central appetite suppression effects.
This is cutting-edge research with limited practical application currently. But it illustrates the direction the field is moving, toward multimodal enhancement strategies that combine physical and chemical approaches to maximize nasal peptide delivery.
How intranasal retatrutide compares to injection
The comparison between nasal and injectable retatrutide requires honest assessment of trade-offs. Neither route is universally superior. Each has distinct advantages that matter differently depending on research objectives.
Head-to-head comparison
Factor | Subcutaneous injection | Intranasal spray |
|---|---|---|
Bioavailability | ~100% (reference standard) | 2-5% unenhanced, 15-45% enhanced |
Onset of action | Hours (depot absorption) | Minutes (rapid mucosal absorption) |
Brain delivery | Limited BBB crossing | Direct nose-to-brain pathway available |
Duration of effect | 7 days (fatty acid tail binding) | Hours (no depot effect) |
Administration ease | Requires syringe, alcohol swab, technique | Simple spray, no needles |
Storage requirements | Refrigeration required | Refrigeration required |
GI side effects | Common (38-43% nausea) | Potentially reduced (lower systemic exposure) |
Dosing frequency | Once weekly | Likely daily or more frequent |
Research maturity | Phase 3 clinical trials complete | Preclinical and investigational only |
Cost efficiency | Higher per-dose utilization | Lower per-dose utilization (more peptide needed) |
The most important difference is bioavailability. Subcutaneous injection delivers nearly 100% of the administered dose to systemic circulation. Unenhanced nasal delivery achieves approximately 2-5%. Even with the best enhancement strategies currently available, nasal bioavailability for peptides in this molecular weight range typically reaches 15-45% in optimized animal models, with human bioavailability often lower.
This bioavailability gap has direct implications for dosing. If only 5% of a nasal dose reaches circulation, you would need to administer 20 times more peptide nasally than by injection to achieve equivalent systemic levels. At the current cost of retatrutide, this makes unenhanced nasal delivery economically impractical for sustained use.
But the speed advantage is real. The Japanese clinical trial showed peak GLP-1 levels at 8.1 minutes with nasal delivery. Subcutaneous injection typically produces peak levels hours later. For research applications where rapid onset matters more than sustained effect, nasal delivery offers a genuinely useful characteristic.
The brain delivery advantage
This is where intranasal retatrutide becomes genuinely interesting rather than merely a convenience play.
The nose-to-brain pathway bypasses the blood-brain barrier through direct transport along olfactory and trigeminal nerve fibers. This pathway is well-documented for several peptides, including insulin, oxytocin, and various neuropeptides. GLP-1 receptors are abundant in brain regions controlling appetite, reward, and metabolic regulation, including the hypothalamus, brainstem, and nucleus tractus solitarius.
When retatrutide is injected subcutaneously, it must cross the blood-brain barrier to access these central receptors. The BBB restricts passage of molecules above approximately 500 daltons, meaning only a tiny fraction of circulating retatrutide reaches the brain. This is adequate for peripheral metabolic effects like appetite suppression and insulin sensitization, but it may limit the full potential of central GLP-1 receptor activation.
Intranasal delivery could theoretically achieve higher brain concentrations of retatrutide at lower systemic doses. This is not proven for retatrutide specifically, but the principle is well-established for other peptides of comparable size. If confirmed, this would mean intranasal retatrutide could produce stronger appetite suppression with fewer peripheral side effects like diarrhea and fatigue.
Preparation and reconstitution for nasal delivery
Preparing retatrutide for nasal delivery differs from standard reconstitution for injection in several important ways. The solvent choice, concentration, pH, and additives all require adjustment for nasal application.
Solvent selection
Bacteriostatic water is the recommended reconstitution solvent for nasal peptide sprays. The bacteriostatic agent, typically 0.9% benzyl alcohol, prevents microbial growth during the multi-use period of a nasal spray device. Standard sterile water lacks preservatives and creates contamination risk with repeated spray actuations.
Normal saline (0.9% sodium chloride) is sometimes used because it matches the tonicity of nasal mucosal fluids, reducing irritation. However, standard saline solutions lack preservatives, making them unsuitable for multi-use nasal sprays without additional antimicrobial agents. Some researchers use isotonic bacteriostatic saline, which combines the tonicity benefits with preservation.
The reconstitution process follows standard peptide handling protocols. Using a sterile syringe, slowly inject the chosen solvent into the lyophilized retatrutide vial, directing the stream against the vial wall at a 45-degree angle. Do not shake. Allow 15-30 minutes for complete dissolution. The solution should be clear and free of particulates before transferring to a nasal spray device.
Concentration considerations
Nasal spray devices typically deliver 100 microliters (0.1 mL) per actuation. This small volume limits how much peptide can be delivered per spray. The concentration of the reconstituted solution must be high enough to deliver a meaningful dose in one or two actuations, but not so high that the peptide precipitates out of solution.
For a hypothetical research protocol targeting 1mg of retatrutide per nasal administration, you would need a concentration of 10mg/mL to deliver 1mg in a single 100-microliter spray. This is achievable but approaches the solubility limits for some peptides.
Using the retatrutide dosage calculator can help researchers determine exact reconstitution volumes for their target concentrations.
Nasal spray device selection
Not all spray devices are equal for peptide delivery. The ideal nasal spray device for peptide research produces a plume with droplet sizes between 10-50 micrometers. Smaller droplets risk inhalation into the lungs. Larger droplets deposit primarily in the anterior nasal cavity where absorption is less efficient.
Metered-dose spray pumps with crimp-on actuators provide the most consistent dosing. Glass vials with spray pump tops are preferable to plastic for peptide compatibility. The device should be primed before first use according to manufacturer instructions, and stored upright to maintain proper function.
Storage after reconstitution
Reconstituted intranasal retatrutide requires refrigeration at 2-8 degrees Celsius, similar to injectable retatrutide storage requirements. The reconstituted nasal solution may have a shorter stability window than injectable preparations due to the absence of depot-forming conditions. Most researchers limit nasal peptide solution use to 14-28 days after reconstitution, depending on the preservative system used.
Avoid freezing reconstituted nasal solutions. Freeze-thaw cycles can cause peptide aggregation and loss of potency. Protect from light exposure. And never use a nasal spray solution that appears cloudy, discolored, or contains visible particles.
Dosing considerations for intranasal retatrutide
Dosing intranasal retatrutide is fundamentally different from dosing the injectable form. The bioavailability gap means that nasal doses cannot simply mirror standard retatrutide dosing protocols. Researchers must account for absorption losses and adjust accordingly.
The bioavailability adjustment
If nasal bioavailability is 3% (unenhanced), a researcher targeting systemic exposure equivalent to a 4mg subcutaneous dose would theoretically need approximately 133mg delivered nasally. This is clearly impractical for several reasons: cost, nasal cavity volume limitations, and peptide solubility constraints.
With enhancement strategies, the math becomes more reasonable. At 20% bioavailability (achievable with optimized cyclodextrin formulations in human studies), the equivalent nasal dose drops to approximately 20mg. At 40% (the upper range of what enhanced nasal delivery achieves), it drops to 10mg. These amounts are still significantly more than injectable doses but fall within the realm of technical feasibility.
However, dose equivalence may not be the right framework. If intranasal retatrutide achieves preferential brain delivery, lower systemic doses could potentially produce central appetite suppression effects comparable to higher subcutaneous doses.
This hypothesis has not been tested for retatrutide but is supported by data from other nasally delivered GLP-1 agonists.
Frequency adjustments
Injectable retatrutide is designed for once-weekly administration. The fatty acid tail enables prolonged albumin binding and sustained receptor activation. Nasal delivery eliminates the depot effect, meaning the peptide clears from circulation much faster.
Based on pharmacokinetic principles and data from intranasal GLP-1 studies, intranasal retatrutide would likely require daily or even twice-daily administration to maintain meaningful receptor activation. The Japanese GLP-1 trial administered the nasal formulation before every meal, three times daily. This frequency may be necessary for intranasal retatrutide as well.
More frequent dosing is not necessarily a disadvantage. Some researchers argue that pulsatile peptide delivery, with peaks and troughs in plasma concentration, may produce different receptor signaling patterns than the sustained exposure from weekly injections. Whether these different signaling patterns translate to different outcomes is an open question in peptide pharmacology.
Starting dose suggestions for research
No established dosing protocols exist for intranasal retatrutide. Researchers exploring this route typically begin with conservative doses and titrate based on observed effects. A reasonable starting framework, based on extrapolation from GLP-1 nasal delivery literature, might look like this:
Conservative approach: Begin with 0.5-1mg per nasal actuation, administered once daily. Assess tolerance and effects over 7-14 days before adjusting. This accounts for the uncertainty in bioavailability and individual variation in nasal absorption.
Moderate approach: Start with 1-2mg per actuation, administered twice daily (morning and evening). This mirrors the twice-daily pattern used in some GLP-1 nasal research protocols and provides more consistent receptor activation throughout the day.
Enhanced formulation approach: When using cyclodextrin or other absorption enhancers, lower doses of 0.25-0.5mg per actuation may be sufficient due to improved bioavailability. Start low and increase gradually while monitoring for nasal irritation from the enhancer components.
These are theoretical frameworks, not validated protocols. Researchers should exercise caution and document all parameters carefully. The starting dose principles that apply to injectable retatrutide, beginning low and titrating slowly, are even more important with the nasal route where bioavailability is less predictable.
Side effects and safety considerations
Intranasal retatrutide presents a different side effect profile than injectable administration. Some systemic side effects may be reduced. But nasal-specific adverse effects must be considered.
Potential reduction in GI side effects
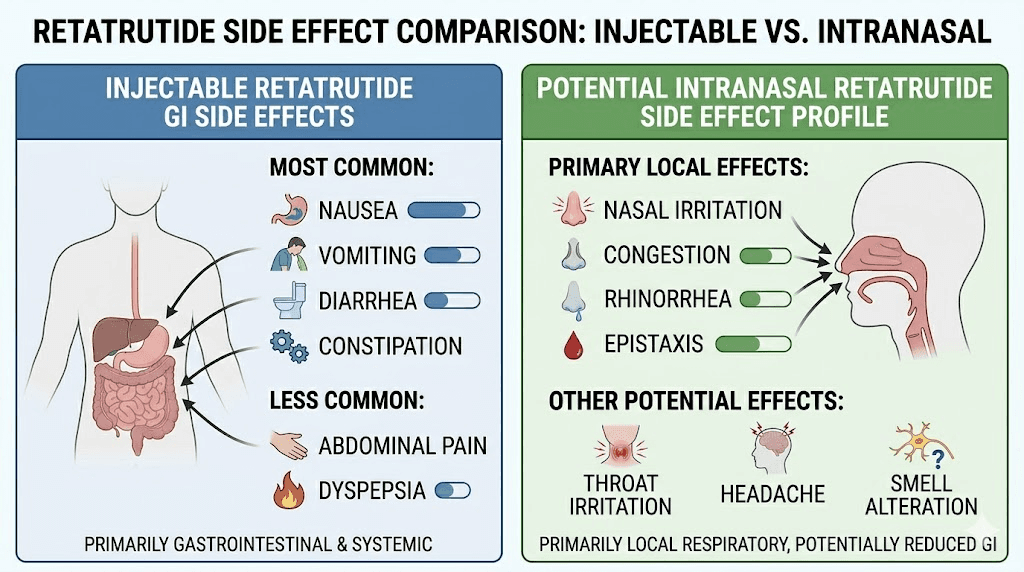
The most common side effects of injectable retatrutide in Phase 3 trials were gastrointestinal. Nausea affected 38-43% of participants. Diarrhea occurred in 33-35%. Constipation in 22-25%. Vomiting in 20-21%.
Lower systemic bioavailability from nasal delivery would be expected to reduce the incidence and severity of these GI effects. The Japanese intranasal GLP-1 trial specifically noted low adverse events, especially nausea and vomiting, compared to injectable exenatide and liraglutide. If this pattern holds for retatrutide, nasal delivery could offer a GI-tolerability advantage for individuals who respond well to the central appetite effects but struggle with peripheral GI symptoms.
However, this assumes that nasal delivery produces meaningful systemic levels. At very low bioavailability, GI effects may be reduced simply because the peptide is not reaching effective concentrations anywhere in the body, including the brain.
Nasal-specific side effects
Repeated nasal peptide administration can cause local irritation. Symptoms may include nasal dryness, burning sensation, rhinorrhea (runny nose), epistaxis (nosebleeds), and mucosal erosion with long-term use. These effects are generally mild with well-formulated preparations but can become significant with certain absorption enhancers.
Some absorption enhancers, particularly surfactant-based compounds, achieve their effects by partially disrupting the mucosal barrier. While this increases peptide absorption, it can also cause cumulative mucosal damage with repeated use. Cyclodextrins are generally considered safer in this regard, with toxicological studies confirming acceptable nasal tolerability at therapeutic concentrations.
Taste disturbance is another common complaint. Peptide solutions that drain from the nasal cavity to the oropharynx can produce a bitter or unpleasant taste. This is cosmetically bothersome but not medically significant. Proper spray technique, directing the spray toward the lateral nasal wall rather than straight back, minimizes posterior drainage.
Systemic safety considerations
Retatrutide carries several safety considerations regardless of administration route. The TRIUMPH-4 trial reported that cardiovascular risk markers were reduced, including non-HDL cholesterol, triglycerides, and high-sensitivity C-reactive protein. Systolic blood pressure decreased by 14 mmHg at the highest dose.
Whether intranasal delivery replicates these cardiovascular benefits depends on achieving sufficient systemic exposure.
Lower bioavailability could mean reduced cardiovascular benefit but also reduced risk of blood pressure drops that might cause dizziness or lightheadedness.
Researchers using intranasal retatrutide should monitor the same parameters as those using the injectable form: body weight, appetite changes, gastrointestinal symptoms, blood pressure, and heart rate. Additional monitoring of nasal mucosal integrity through periodic visual inspection is advisable for long-term nasal delivery protocols.
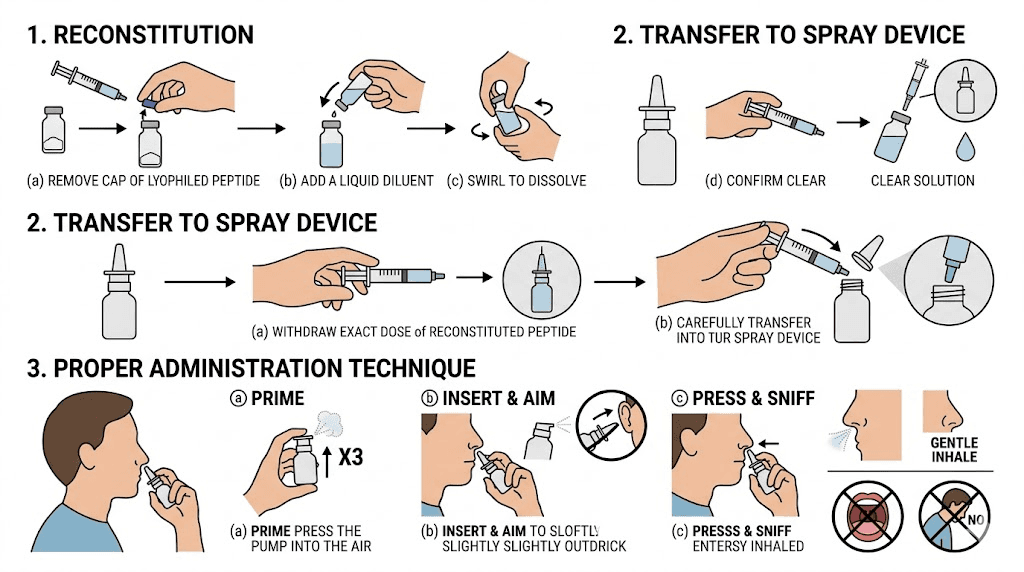
Practical administration technique
Proper nasal spray technique significantly affects peptide absorption. Poor technique can reduce bioavailability by 50% or more compared to optimal administration. Every researcher using intranasal retatrutide should understand and practice correct technique.
Step-by-step administration
Step 1: Prepare the nasal passages. Gently blow the nose to clear excess mucus. Congested or inflamed nasal passages reduce absorption dramatically. If nasal congestion is present, consider using a saline rinse 10-15 minutes before peptide administration and allowing the passages to dry.
Step 2: Prime the spray device. If using a metered-dose spray pump for the first time, actuate the pump 3-5 times pointed away from the face until a fine mist appears. This ensures the pump mechanism is primed and the first delivered dose is accurate.
Step 3: Position correctly. Tilt the head slightly forward, approximately 10-15 degrees. Insert the spray nozzle into the nostril, angling the tip toward the lateral nasal wall (the outer wall of the nose), not toward the septum (center wall). This directs the spray toward the turbinate area where blood supply is richest and absorption is most efficient.
Step 4: Administer the spray. Close the opposite nostril with a finger. Breathe in gently through the nose while simultaneously actuating the spray pump. The inhalation should be gentle, not a forceful sniff. Forceful inhalation deposits the spray too deeply, potentially sending it into the sinuses or throat where absorption is poor.
Step 5: Hold and avoid sniffing. After spraying, keep the head slightly forward for 30-60 seconds.
Do not sniff, blow the nose, or tilt the head back. A slight sniff immediately after spraying is the most common technique error, as it moves the peptide solution away from the optimal absorption zone.
Step 6: Repeat if bilateral dosing is required. If the protocol calls for administration in both nostrils, wait 2-3 minutes between sides. Alternating nostrils between doses can help reduce localized irritation.
Timing considerations
Based on the GLP-1 nasal delivery literature, administering intranasal retatrutide before meals may optimize the appetite suppression effect. The rapid onset of nasal delivery, with peak levels at approximately 8 minutes, means the peptide reaches relevant brain and peripheral receptors by the time eating begins.
For timing optimization, consider administering the nasal spray 10-15 minutes before meals. This allows the peptide to reach peak absorption and begin receptor activation before food intake. Avoid administration immediately after eating, as postprandial nasal congestion from increased blood flow can reduce absorption.
Comparing intranasal retatrutide to other nasal peptides
Intranasal retatrutide is not the first peptide to be explored through nasal delivery. Understanding how other nasal peptide products perform provides useful context for setting realistic expectations.
Established nasal peptide products
Desmopressin (DDAVP nasal spray): This is the gold standard for nasal peptide delivery. Desmopressin weighs only 1,069 daltons, well within the easy absorption range. Nasal bioavailability is approximately 3-5%, which is sufficient because the peptide is extremely potent. Approved for diabetes insipidus and nocturnal enuresis.
Calcitonin (Miacalcin nasal spray): Calcitonin weighs 3,432 daltons, closer to the retatrutide range. Nasal bioavailability is approximately 3%. Despite this low bioavailability, the product is clinically effective for osteoporosis because regular nasal administration maintains sufficient calcitonin levels for bone metabolism effects.
Oxytocin nasal spray: Oxytocin weighs 1,007 daltons. Nasal bioavailability for systemic effects is low, but the nose-to-brain pathway delivers oxytocin directly to central receptors with good efficiency. This is the closest model to what intranasal retatrutide brain delivery might achieve.
Insulin nasal spray: Insulin weighs approximately 5,800 daltons, larger than retatrutide. Nasal insulin delivery has been extensively studied for both diabetes management and Alzheimer disease research (brain insulin delivery). Bioavailability is typically 10-20% with absorption enhancers.
Clinical development has been challenging due to variable absorption.
What these comparisons tell us
Several patterns emerge from the nasal peptide landscape. Low bioavailability does not necessarily prevent clinical utility, as demonstrated by desmopressin and calcitonin. Brain delivery can be achieved even when systemic bioavailability is low, as shown by oxytocin. Peptides in the 4,000-6,000 dalton range can be delivered nasally with enhancement, as demonstrated by insulin research. And variability in absorption remains a significant challenge for all nasal peptide products.
For retatrutide at 4,895 daltons, these comparisons suggest that nasal delivery is feasible but will require optimization, that brain delivery effects may be achievable at doses lower than needed for full systemic effect, and that absorption variability will be higher than with injection.
Intranasal retatrutide versus other delivery alternatives
Nasal delivery is not the only alternative to subcutaneous injection being explored for GLP-1 class peptides. Understanding the full landscape of delivery options helps researchers evaluate where intranasal retatrutide fits.
Oral delivery
Oral tirzepatide is in clinical development and represents a major advance in non-injectable peptide delivery. Oral semaglutide (Rybelsus) is already approved and marketed. However, oral delivery requires high doses due to gastric degradation and low intestinal bioavailability (typically less than 1%). Oral retatrutide has not been publicly studied.
Compared to oral delivery, nasal delivery typically achieves higher bioavailability for peptides, faster onset, and avoids the requirement for fasting that oral peptide formulations demand. The trade-off is that nasal delivery is limited by the volume that can be administered per spray (100 microliters) while oral delivery can accommodate larger doses.
Sublingual and buccal delivery
Sublingual semaglutide and tirzepatide drops represent another non-injectable approach. The sublingual mucosa has a rich blood supply and avoids first-pass metabolism, similar to nasal delivery. Bioavailability for peptides through the sublingual route is generally in the 3-10% range, comparable to nasal delivery.
The sublingual route may be simpler to prepare and administer than nasal spray but has its own challenges, including peptide degradation by salivary enzymes and the difficulty of keeping the solution under the tongue long enough for absorption.
Transdermal patches
GLP-1 patches deliver peptides through the skin using microneedle arrays or iontophoresis. These devices can provide sustained delivery over hours or days but require sophisticated technology. Bioavailability varies widely depending on the device but can exceed nasal delivery for some formulations.
The advantage of patches is sustained, consistent delivery without repeated administration. The disadvantage is cost, complexity, and the current lack of retatrutide-specific patch formulations.
Where intranasal fits in the landscape
Intranasal retatrutide occupies a specific niche. It is the best option for researchers who prioritize rapid onset, brain delivery, or needle-free administration and are willing to accept lower bioavailability and more frequent dosing. It is not the best option for those seeking the highest possible bioavailability or once-weekly convenience. And it remains investigational, without the clinical validation that injectable retatrutide has achieved.
The nose-to-brain pathway: deeper implications for retatrutide
The potential for direct brain delivery deserves deeper exploration because it could fundamentally change how we think about retatrutide dosing strategies.
Central versus peripheral effects
Retatrutide produces weight loss through both central and peripheral mechanisms. Centrally, GLP-1 receptor activation in the hypothalamus and brainstem suppresses appetite and reduces food intake. Peripherally, GLP-1 slows gastric emptying, GIP modulates fat metabolism, and glucagon receptor activation increases hepatic energy expenditure.
With subcutaneous injection, these central and peripheral effects occur simultaneously because the peptide distributes throughout the body via systemic circulation. The GI side effects, nausea, vomiting, diarrhea, are primarily peripheral effects related to GLP-1 receptor activation in the gut.
Intranasal delivery through the nose-to-brain pathway could theoretically decouple these effects. By delivering retatrutide preferentially to the brain, researchers might achieve appetite suppression and metabolic regulation through central mechanisms while minimizing peripheral GI receptor activation. This could mean effective appetite control with fewer side effects.
Implications for the glucagon receptor component
Retatrutide is unique because of its glucagon receptor agonism. Glucagon receptors in the liver drive glycogenolysis and hepatic fat oxidation, contributing significantly to the weight loss effect. These are peripheral effects that depend on systemic circulating levels of the peptide.
If intranasal delivery achieves preferential brain delivery at the expense of systemic exposure, the hepatic glucagon receptor activation would be reduced. This could diminish the unique advantage retatrutide holds over dual-agonists like tirzepatide. Researchers must weigh whether the brain delivery benefits outweigh the potential loss of peripheral glucagon signaling.
Combining routes for optimal effect
Some researchers have theorized about combining intranasal and subcutaneous administration. A reduced-dose weekly injection for sustained peripheral effects, supplemented by intranasal administration before meals for acute appetite suppression. This dual-route approach is entirely speculative but illustrates the kind of creative protocol design that intranasal delivery enables.
The concept has precedent in other therapeutic areas. Insulin therapy, for example, uses both long-acting basal injections and rapid-acting mealtime doses. A similar basal-bolus approach with injectable and intranasal retatrutide could theoretically optimize both peripheral metabolic effects and central appetite regulation.
Stability and formulation challenges
Creating a stable nasal formulation of retatrutide presents challenges beyond simple reconstitution. Long-term stability, spray device compatibility, and preservation all require attention.
pH optimization
Peptide stability and nasal mucosal compatibility both depend on solution pH. The nasal mucosa has a pH of approximately 5.5-6.5. Solutions that deviate significantly from this range cause irritation and may trigger excessive mucus secretion that impairs absorption. Most nasal peptide formulations target a pH of 4.5-6.5, balancing peptide stability with mucosal compatibility.
Retatrutide stability at various pH levels has not been extensively characterized in the public literature. Researchers preparing nasal formulations should test stability at the target pH over the intended use period and monitor for degradation products through appropriate analytical methods.
Preservative compatibility
Benzyl alcohol (0.9% in bacteriostatic water) is the most common preservative for nasal peptide solutions. It is generally compatible with peptides in this molecular weight range and provides adequate antimicrobial protection for multi-use devices. However, some peptides are sensitive to benzyl alcohol, and stability should be confirmed.
Alternative preservatives include phenoxyethanol, potassium sorbate, and methylparaben. Each has different compatibility profiles with peptide formulations. The choice of preservative can affect both peptide stability and nasal tolerability.
Spray device material compatibility
Peptides can adsorb onto container surfaces, reducing the delivered dose over time. Glass containers minimize adsorption and are generally preferred for peptide formulations. If plastic spray devices are used, the material should be tested for peptide adsorption. Polypropylene and high-density polyethylene typically show lower peptide adsorption than polystyrene or PVC.
Research applications and protocols
Intranasal retatrutide serves specific research applications where its unique characteristics provide advantages over injectable delivery.
Acute appetite studies
The rapid onset of nasal peptide delivery makes it particularly useful for acute feeding studies. Researchers can administer intranasal retatrutide minutes before a test meal and measure the immediate impact on food intake, satiety ratings, and gut hormone responses. This is difficult to accomplish with weekly subcutaneous injection, where the peptide is always present at relatively steady-state levels.
CNS receptor occupancy research
Studying the central effects of GLP-1 receptor agonists requires brain delivery. Intranasal administration through the olfactory pathway could enable researchers to investigate retatrutide effects on brain GLP-1, GIP, and glucagon receptors without the confounding variable of high peripheral peptide levels. This application is relevant to understanding the mechanisms behind appetite suppression and food reward processing.
Tolerability screening
The lower systemic exposure from nasal delivery could serve as a tolerability screen before full-dose subcutaneous injection. Researchers could assess individual sensitivity to retatrutide GI effects at low systemic doses before committing to the higher exposure from injection. This is not standard practice but represents a logical application of the pharmacokinetic differences between routes.
Combination research
Intranasal retatrutide could be combined with other GLP-1 protocols in ways that injectable forms cannot. For example, researchers could use intranasal retatrutide as a supplemental appetite suppressant in individuals already receiving a different GLP-1 agonist by injection. The lower systemic exposure from nasal delivery would reduce the risk of excessive GLP-1 receptor activation.
Cost analysis: nasal versus injectable retatrutide
Cost is a practical consideration that often determines research feasibility. The bioavailability difference between nasal and injectable delivery directly impacts cost per effective dose.
Peptide cost per effective milligram
At 3% nasal bioavailability (unenhanced), delivering 1mg of systemically available retatrutide requires approximately 33mg of peptide sprayed nasally. At current retatrutide pricing, this makes unenhanced nasal delivery approximately 33 times more expensive per effective dose than injection.
With cyclodextrin enhancement improving bioavailability to approximately 20%, the cost multiplier drops to 5 times. At 40% bioavailability with optimized formulations, it drops to 2.5 times. These enhanced formulations also add the cost of the enhancer itself, though cyclodextrins are relatively inexpensive.
The cost equation changes if brain delivery is the primary objective. If nasal delivery achieves effective central appetite suppression at lower total systemic doses than injection, the cost per therapeutic outcome could potentially favor the nasal route despite lower bioavailability. This is speculative but economically significant.
Equipment and consumables
Nasal spray devices cost $2-15 each for standard metered-dose pump bottles. Bacteriostatic water or saline adds minimal cost. Cyclodextrin enhancers are available from chemical suppliers at reasonable prices for research quantities. Overall, the equipment cost for nasal delivery is lower than for injection, which requires syringes, needles, and alcohol swabs for each administration.
The net cost comparison depends heavily on bioavailability. For researchers using enhanced formulations that achieve reasonable bioavailability, the total cost of nasal delivery may be comparable to injection when equipment savings offset the additional peptide required. For those using unenhanced formulations, injection remains significantly more cost-effective.
Future directions for intranasal retatrutide research
The field of intranasal peptide delivery is advancing rapidly. Several developments could significantly impact the viability of intranasal retatrutide in the coming years.
Advanced nanoformulations
Next-generation nanocarrier systems are being designed specifically for large peptide nasal delivery. Lipid nanoparticles similar to those used in mRNA vaccines could potentially encapsulate retatrutide and protect it from enzymatic degradation while facilitating mucosal transport. If these formulations achieve the 50-70% bioavailability seen in some preclinical studies, intranasal delivery could become a practical alternative to injection for a wider range of applications.
Device technology improvements
Precision nasal delivery devices that target the olfactory region specifically, rather than depositing peptide across the entire nasal cavity, could improve brain delivery efficiency by 3-5 fold according to preliminary studies. These devices use computational fluid dynamics modeling to optimize spray characteristics for upper nasal deposition where olfactory nerve endings are concentrated.
Formulation optimization by Eli Lilly
Eli Lilly is investing heavily in non-injectable delivery for its GLP-1 class peptides. While no public announcement has been made regarding intranasal retatrutide specifically, the company has explored oral and alternative delivery routes for other molecules in its pipeline. If clinical data from other delivery route studies prove encouraging, intranasal formulations could receive more attention and investment.
Regulatory pathway
For intranasal retatrutide to move beyond research applications, it would need to demonstrate bioequivalence or clinical equivalence to the injectable form in human trials. The regulatory pathway for nasal peptide delivery is well-established through precedents like desmopressin and calcitonin nasal sprays. The challenge is demonstrating consistent, predictable absorption that meets regulatory standards for variability.
Frequently asked questions
Does intranasal retatrutide work as well as injection?
Current evidence suggests intranasal delivery achieves significantly lower systemic bioavailability than injection, typically 2-5% without enhancement and potentially 15-45% with optimized formulations. The clinical efficacy has not been directly compared in human trials. However, the potential for direct brain delivery through the olfactory pathway could provide unique appetite suppression benefits not achievable with injection alone.
How do you prepare retatrutide for nasal spray use?
Reconstitute lyophilized retatrutide with bacteriostatic water, directing the solvent against the vial wall at a 45-degree angle. Allow 15-30 minutes for complete dissolution. Transfer the clear solution to a sterile metered-dose nasal spray device. Store refrigerated at 2-8 degrees Celsius and use within 14-28 days. See the reconstitution guide for detailed bacteriostatic water calculations.
What is the right dosage for intranasal retatrutide?
No established dosing protocol exists for intranasal retatrutide. Research doses are typically calculated by adjusting the standard injectable dose upward to account for reduced nasal bioavailability. Most researchers start conservatively at 0.5-1mg per actuation and titrate based on observed effects. The optimal dose depends on the formulation, enhancement strategy used, and research objectives.
Is intranasal retatrutide safer than injection?
Intranasal delivery may produce fewer gastrointestinal side effects due to lower systemic exposure, consistent with findings from intranasal GLP-1 clinical trials. However, it introduces nasal-specific risks including mucosal irritation, dryness, and potential epistaxis with long-term use. Neither route has been directly compared for safety in clinical trials.
Can you use regular bacteriostatic water for nasal spray?
Yes, bacteriostatic water containing 0.9% benzyl alcohol is the recommended reconstitution solvent for nasal peptide sprays. The preservative protects against bacterial contamination during repeated spray actuations. Standard sterile water without preservatives is not recommended for multi-use nasal sprays due to contamination risk.
How often do you need to use intranasal retatrutide compared to injection?
Injectable retatrutide is designed for once-weekly dosing due to its fatty acid tail that enables sustained albumin binding. Intranasal delivery eliminates this depot effect, meaning the peptide clears much faster. Based on GLP-1 nasal delivery research, intranasal retatrutide would likely require daily or multiple-times-daily administration.
Does intranasal delivery bypass the blood-brain barrier?
Yes, the olfactory and trigeminal nerve pathways provide a direct nose-to-brain route that bypasses the blood-brain barrier. This is well-documented for several peptides including insulin and oxytocin. For GLP-1 agonists like retatrutide, this pathway could enable direct activation of brain appetite and metabolic centers, potentially at lower systemic doses than injection requires.
What absorption enhancers work best for nasal peptide delivery?
Cyclodextrins, particularly dimethyl-beta-cyclodextrin and randomly methylated beta-cyclodextrin, are the most extensively validated enhancers with good safety profiles. Cell-penetrating peptides like penetratin analogs show promise for GLP-1 class molecules. Chitosan-based mucoadhesive formulations extend nasal residence time. The optimal approach depends on the specific peptide, target bioavailability, and tolerance for formulation complexity.
External resources
Exploratory trial of intranasal GLP-1 administration in type 2 diabetes (Diabetes Care)
Evaluation of recent intranasal drug delivery systems to the CNS (PMC)
Versatile nasal application of cyclodextrins: excipients and actives (PMC)
For researchers serious about exploring alternative retatrutide delivery routes, SeekPeptides offers comprehensive protocol databases, dosing calculators, and a community of experienced researchers who have navigated these exact questions. Members access detailed retatrutide protocols, weight loss dosing guides, and expert guidance for optimizing research outcomes across all administration routes.
In case I do not see you, good afternoon, good evening, and good night. May your nasal passages stay clear, your bioavailability stay optimized, and your research protocols stay rigorous.