Mar 23, 2026

Your retatrutide is losing potency right now. Maybe not dramatically. Maybe not visibly. But if your storage conditions are even slightly off, every single day chips away at the compound you paid good money for. The frustrating part is that degradation does not announce itself. A vial stored at room temperature for three weeks can look identical to one kept properly in the refrigerator, yet contain only 60 percent of the original active compound. That is not a minor detail when you are relying on precise dosing for your retatrutide protocol.
Retatrutide, the triple-agonist GLP-1/GIP/glucagon receptor peptide, presents unique storage challenges that go beyond what most researchers expect. Its molecular structure is sensitive to temperature fluctuations, light exposure, and moisture, with even small environmental changes creating measurable potency loss.
And because retatrutide cost is not trivial, every percentage point of degradation is money wasted.
This guide covers everything. Lyophilized storage. Reconstituted solution handling. Temperature thresholds that matter. Travel protocols that actually work. Signs your peptide has degraded beyond usability. Whether you just received your first vial or you are an experienced researcher managing multiple retatrutide vials, the information here will protect your investment and ensure consistent results throughout your entire dosing schedule. SeekPeptides members already know that proper storage is the difference between a protocol that works and one that quietly fails.
Why retatrutide storage matters more than you think
Peptides are not like most medications sitting in your medicine cabinet. They are chains of amino acids held together by bonds that break under specific environmental conditions. Heat provides the kinetic energy that accelerates every potential degradation pathway. Light triggers photodegradation. Moisture causes hydrolysis, literally breaking peptide bonds apart.
Retatrutide is particularly vulnerable because of its size and complexity. As a triple-receptor agonist, its molecular structure must maintain a precise three-dimensional shape to bind effectively to GLP-1, GIP, and glucagon receptors. Even minor structural changes from improper storage can reduce or eliminate binding affinity at one or more receptor sites, which directly impacts the compound effectiveness for fat loss, appetite suppression, and metabolic regulation.
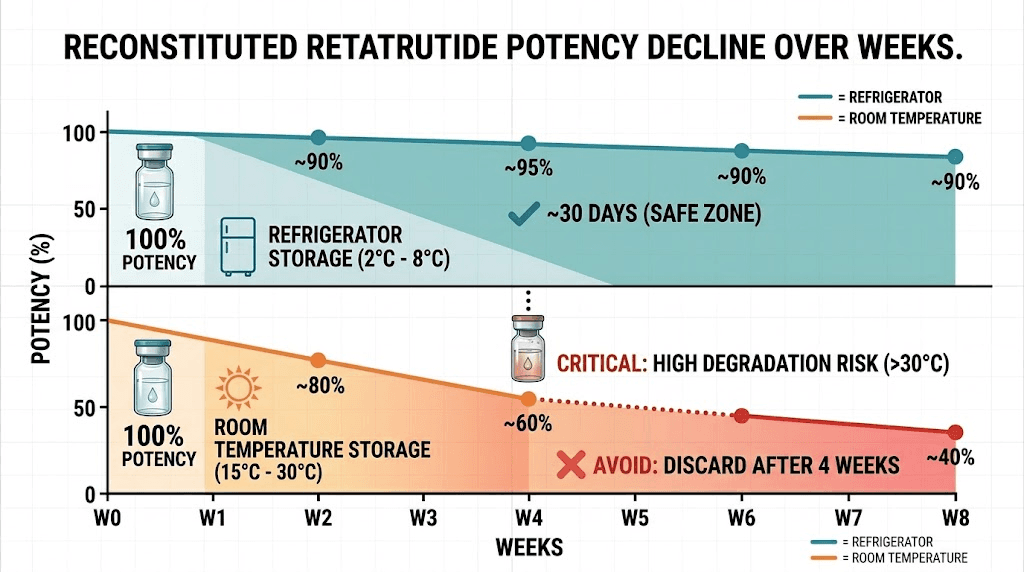
Here is what the data shows. Retatrutide stored at room temperature loses potency dramatically. Within the first week, potency drops to approximately 75 percent. By day 14, you are looking at roughly 55 percent. And by day 21, only about 35 percent of the original active compound remains. Compare that to properly refrigerated storage, where potency remains above 95 percent for months.
That gap is enormous.
It means the difference between a dosing protocol that delivers consistent results and one where you are effectively taking a different dose every week without knowing it. If you have ever wondered how long retatrutide takes to work and felt like progress stalled midway through a cycle, storage problems should be the first thing you investigate.
Understanding the two forms of retatrutide
Before diving into specific storage protocols, you need to understand that retatrutide exists in two distinct forms, and each requires a completely different storage approach.
Lyophilized (freeze-dried) powder
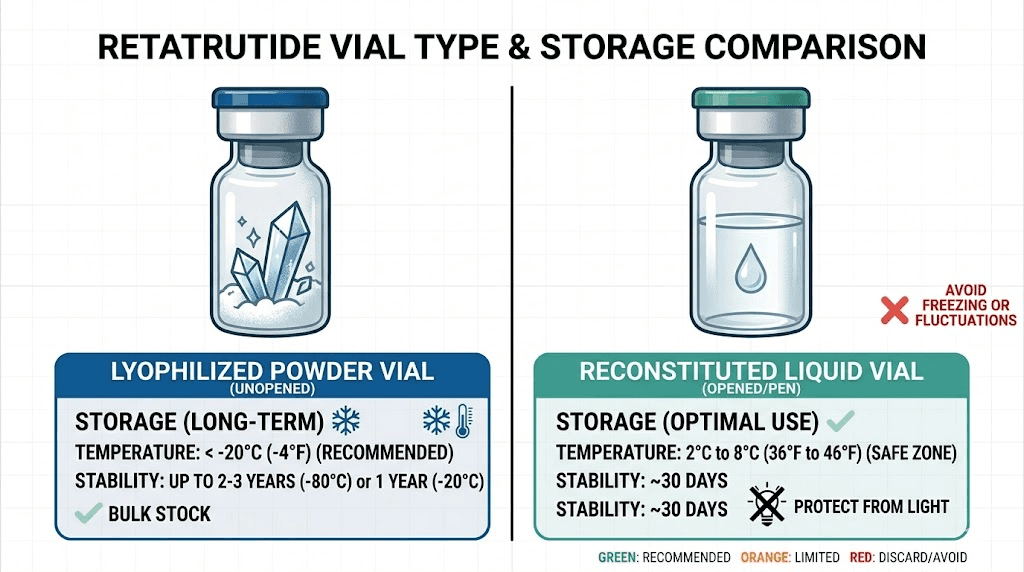
This is how retatrutide arrives from most suppliers. The lyophilization process removes water from the peptide solution through sublimation, leaving behind a dry powder or cake inside the vial. This form is inherently more stable than liquid because water, the primary catalyst for hydrolysis and microbial growth, has been removed.
Lyophilized retatrutide looks like a white to off-white powder or a small disc at the bottom of the vial. Some vials contain a fluffy cake that fills more of the space, while others have a compact puck. Both are normal. What matters is that the powder remains dry and free of visible contamination. If you are comparing this to other peptides you may have used, the principle is identical to how lyophilized tirzepatide is stored.
Reconstituted solution
Once you add bacteriostatic water to your retatrutide vial, everything changes. The peptide is now in solution, suspended in liquid, and the clock starts ticking much faster. Water reintroduces the possibility of hydrolysis, oxidation, and microbial contamination. The reconstituted form requires stricter temperature control, more careful handling, and has a significantly shorter usable lifespan.
Think of it this way. Lyophilized retatrutide is like dried pasta in a sealed box. Stable for years under reasonable conditions. Reconstituted retatrutide is like cooked pasta. Still perfectly good if refrigerated and used within a reasonable timeframe, but it will not last anywhere near as long and must be handled with more care.
Understanding which form you are working with at any given time is the foundation of proper storage. Mistakes happen when researchers apply reconstituted storage rules to lyophilized powder (unnecessarily restrictive) or, worse, apply lyophilized storage assumptions to reconstituted solution (dangerously lax).
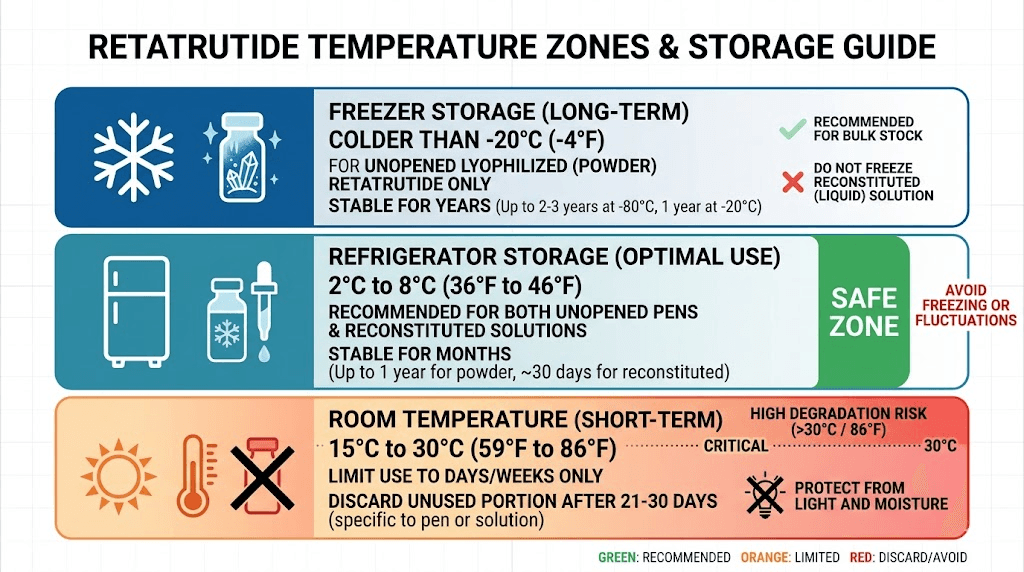
Storing lyophilized retatrutide: the complete protocol
Lyophilized retatrutide is remarkably stable when stored correctly. The key variables are temperature, light exposure, and moisture protection. Get all three right, and your powder will maintain potency for years.
Temperature guidelines for powder storage
The ideal storage temperatures, ranked from best to acceptable:
Freezer storage at -20 degrees Celsius or below is the gold standard. At this temperature, lyophilized retatrutide remains stable for up to 48 months, that is four full years. Research data shows that samples stored at minus 20 degrees Celsius retained 98 percent potency after 12 months. Some facilities use ultra-low freezers at minus 80 degrees Celsius for maximum stability, but this is unnecessary for most researchers. A standard home freezer set to minus 20 Celsius works perfectly.
This is the approach you should take if you buy retatrutide in bulk or plan to store vials for more than a few months before use.
Refrigerator storage at 2 to 8 degrees Celsius is the next best option. Lyophilized powder remains stable for 12 to 24 months at standard refrigerator temperature. This is perfectly acceptable for vials you plan to reconstitute within the next year. Most researchers store their active supply in the refrigerator and any long-term backup supply in the freezer.
If you keep your retatrutide dosing protocol on a consistent schedule, a single vial rarely sits in the fridge for more than a few weeks before reconstitution, making this temperature range more than adequate.
Room temperature storage is acceptable only for very short periods. Lyophilized retatrutide at room temperature (below 25 degrees Celsius) degrades significantly faster, with studies showing approximately 40 percent potency loss in just 30 days. Only use room temperature storage for transit periods of a few days at most.
Light protection
Retatrutide is photosensitive. Exposure to light, particularly UV light, triggers photodegradation that breaks down the peptide structure. This matters more than most researchers realize.
Store vials in their original packaging whenever possible. If the vial is clear glass, wrap it in aluminum foil or place it inside an opaque container. Keep vials in light-protected compartments of your refrigerator or freezer. The back of a refrigerator shelf, behind other items, provides natural light protection whenever the door opens.
Never leave vials on a countertop near windows. Even indirect sunlight over a few hours can initiate photodegradation. This applies equally to other GLP-1 peptides, which is why proper storage guidance for tirzepatide in the fridge and semaglutide in the fridge emphasizes the same light protection principles.
Moisture protection
Lyophilized peptides are hygroscopic, meaning they readily absorb moisture from the air. This is a bigger problem than most people realize. Even brief exposure to humid air can trigger partial breakdown as the peptide begins to rehydrate unevenly.
Keep vials sealed until you are ready to reconstitute. If you need to open a vial for partial use, reseal it immediately and return it to cold storage. Adding desiccant packs to the storage container provides an extra layer of protection, especially in humid climates.
For researchers who purchase retatrutide in bulk from overseas suppliers, check the seal integrity on arrival. Shipping damage that compromises the vial seal means the peptide has been exposed to ambient humidity for the entire transit period.
Lyophilized storage summary table
Storage condition | Temperature | Expected stability | Best for |
|---|---|---|---|
Ultra-low freezer | -80 degrees C | 48+ months | Long-term research supply |
Standard freezer | -20 degrees C | Up to 48 months | Bulk storage, backup vials |
Refrigerator | 2-8 degrees C | 12-24 months | Active supply, near-term use |
Room temperature | Below 25 degrees C | Days to weeks only | Transit only, not recommended |
The takeaway is simple. Colder is better. Dry is essential. Dark is non-negotiable. Nail all three, and your retatrutide peptide will maintain its potency for as long as you need it.
Storing reconstituted retatrutide: what changes after mixing
The moment you add bacteriostatic water to your lyophilized retatrutide, the storage equation shifts dramatically. You have reintroduced water, which means hydrolysis becomes an active threat, microbial contamination becomes possible, and the compound shelf life shrinks from years to weeks.
Refrigeration is mandatory after reconstitution
Store reconstituted retatrutide at 2 to 8 degrees Celsius immediately after mixing. There is no exception to this rule. The refrigerator is not optional for reconstituted peptides, it is the only acceptable storage location.
Under proper refrigeration with sterile handling, reconstituted retatrutide remains usable for approximately 30 days. Some compounding facilities and experienced researchers report safe usage for 6 to 8 weeks under strict aseptic conditions, but the conservative recommendation is to use it within 4 weeks for optimal potency.
This timeline is comparable to how long compounded tirzepatide lasts and compounded semaglutide shelf life after reconstitution. The biochemistry is similar across GLP-1 receptor agonist peptides.
The reconstitution process matters for storage
How you reconstitute directly affects how long the solution remains stable. Contamination introduced during mixing accelerates degradation and creates safety risks. Use the retatrutide reconstitution chart and follow these critical handling practices:
Sterile technique is non-negotiable. Wipe the vial stopper with an alcohol swab before inserting the needle. Use a fresh, sterile syringe for each reconstitution. Do not touch the needle or allow it to contact any non-sterile surface.
Use bacteriostatic water, not sterile water. Bacteriostatic water contains 0.9 percent benzyl alcohol, which inhibits microbial growth and extends the solution usable life. Sterile water lacks this preservative, meaning any contamination introduced during mixing will multiply unchecked. If you use sterile water, the solution should be used within 24 to 48 hours and stored in the refrigerator continuously.
Inject the water slowly along the vial wall. Do not spray it directly onto the powder. Aggressive mixing can damage the peptide structure through physical shearing forces. Let the solution sit for a few minutes after adding water, then swirl gently. Never shake the vial.
These same reconstitution principles apply whether you are working with retatrutide, reconstituting tirzepatide, or reconstituting semaglutide. The peptide chemistry is different, but the handling requirements are universal.
Never freeze reconstituted retatrutide
This is one of the most common storage mistakes. Researchers sometimes assume that if freezing is good for lyophilized powder, freezing must also be good for the reconstituted solution. It is not. In fact, freezing reconstituted retatrutide can render it completely useless.
Here is why. When an aqueous peptide solution freezes, ice crystals form within the liquid. These crystals create physical shearing forces that can unfold or fragment the peptide chains. The freeze-thaw cycle is particularly destructive, as repeated freezing and thawing compounds the damage with each cycle.
A reconstituted vial that has been frozen and thawed even once may have suffered significant structural damage that is invisible to the naked eye. The solution can still look perfectly clear and colorless while containing substantially degraded peptide. This is the same reason you should never freeze reconstituted tirzepatide or other GLP-1 peptides after mixing.
If you accidentally freeze your reconstituted retatrutide, the safest approach is to discard it and reconstitute a fresh vial.
The cost of wasting one vial is far less than the cost of running an unreliable protocol with degraded compound.
Reconstituted storage summary
Factor | Guideline | Why it matters |
|---|---|---|
Temperature | 2-8 degrees C (refrigerator) | Slows hydrolysis and microbial growth |
Shelf life (bac water) | Up to 30 days | Benzyl alcohol inhibits bacteria |
Shelf life (sterile water) | 24-48 hours | No preservative, contamination risk |
Freezing | Never freeze after reconstitution | Ice crystals damage peptide structure |
Light | Keep in dark/opaque container | Prevents photodegradation |
Handling | Sterile technique always | Prevents contamination |
Temperature danger zones: what happens when storage goes wrong
Understanding exactly what happens at different temperatures helps you make informed decisions when storage conditions are not perfect. Because they will not always be perfect. Shipments arrive warm. Power outages happen. You forget to return a vial to the fridge.
The degradation curve at room temperature
Room temperature exposure is the most common storage mistake. Here is the data on what happens to reconstituted retatrutide left at approximately 25 degrees Celsius:
Day 1 to 3: Minimal measurable degradation, perhaps 1 to 3 percent potency loss. The peptide is still well within usable range.
Day 7: Potency drops to approximately 75 percent of original. This is significant, it means a planned 1mg dose is effectively delivering 0.75mg. Your retatrutide dose is no longer what you intended.
Day 14: Only about 55 percent potency remains. At this point, the compound is delivering roughly half the intended dose. Results will be noticeably compromised.
Day 21: Approximately 35 percent potency. The peptide is functionally degraded beyond useful application for most protocols.
These numbers assume consistent room temperature around 25 degrees Celsius. Higher temperatures accelerate degradation further. A vial left in a hot car (which can reach 50 to 60 degrees Celsius) may suffer catastrophic degradation in hours, not days.
What about brief temperature excursions?
Brief periods outside refrigeration are not always catastrophic. The key word is brief.
A single overnight on the counter, roughly 8 to 12 hours at room temperature, will reduce potency by an estimated 2 to 5 percent depending on the ambient temperature. This is annoying but recoverable. Return the vial to the refrigerator immediately and use it sooner than you otherwise would.
If your retatrutide was left out for less than 24 hours, it is generally still usable. Refrigerate it immediately and plan to finish that vial within the next week or two rather than stretching it to the full 30-day window.
However, repeated temperature excursions are the real problem. One overnight on the counter is recoverable. Three or four cycles between room temperature and refrigerator cause cumulative damage that compounds with each cycle. If you find yourself repeatedly forgetting to return vials to the fridge, invest in a dedicated medication refrigerator or a small cooler that stays in your workspace.
The same principles apply to semaglutide left out overnight and tirzepatide that gets warm. All GLP-1 receptor agonist peptides share similar temperature sensitivity profiles.
Heat exposure above 30 degrees Celsius
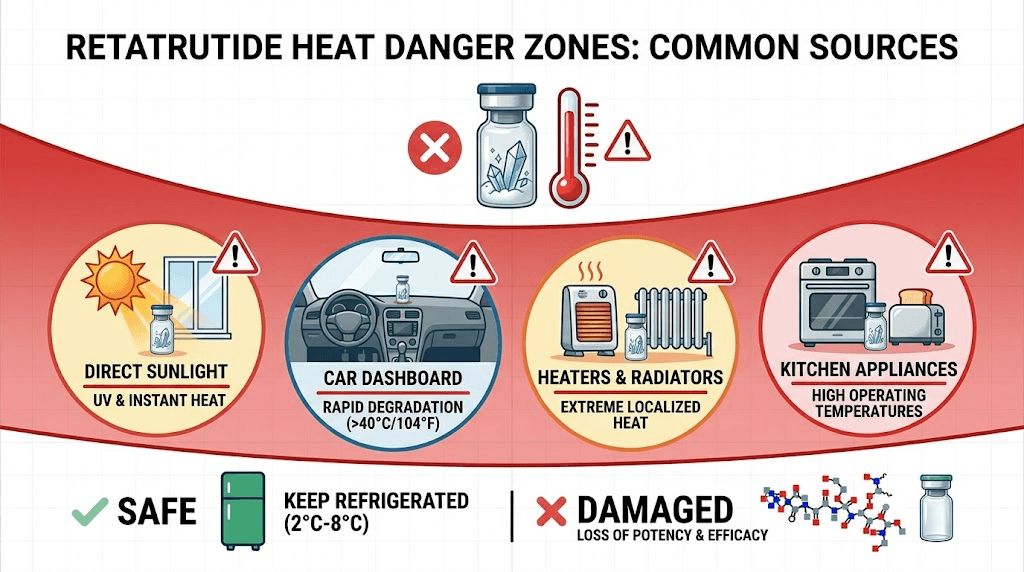
Temperatures at or above 30 degrees Celsius (86 degrees Fahrenheit) cause rapid, often irreversible degradation. This is the critical threshold you must never exceed for any extended period.
Exposure to direct sunlight, hot car interiors, radiators, or heated surfaces can easily push temperatures well beyond this threshold. Even leaving a vial next to a warm laptop or near a kitchen appliance can create a localized heat zone that damages the peptide.
If your vial has been exposed to temperatures above 30 degrees Celsius for more than a few hours, assume significant potency loss. If above 40 degrees, the compound may be functionally destroyed regardless of how it looks visually.
How to tell if your retatrutide has degraded
This is the tricky part. Degraded retatrutide often looks exactly like fresh retatrutide. The most dangerous scenario is not the obviously cloudy vial you would never use. It is the perfectly clear solution that has silently lost 40 percent of its potency.
Visual signs you can detect
Cloudiness or haziness in a reconstituted solution is a clear danger sign. Fresh retatrutide solution should be completely clear and colorless. Any cloudiness indicates peptide aggregation (where degraded molecules clump together) or bacterial contamination. Discard immediately.
Visible particles floating in the solution mean something has gone wrong. Particles can be degraded peptide fragments, bacterial colonies, or contaminants introduced during handling. Do not use.
Color changes in either lyophilized powder or reconstituted solution suggest chemical degradation. The powder should be white to off-white. The solution should be clear and colorless. Any yellowing, browning, or other discoloration is a red flag. If you are uncertain about what your peptide should look like, understanding peptide color and normal semaglutide appearance can help calibrate your expectations.
Unusual odor from the vial suggests bacterial contamination or significant chemical breakdown. Properly stored retatrutide in bacteriostatic water should have at most a very faint alcohol smell from the benzyl alcohol preservative. Anything else warrants immediate disposal.
The silent degradation problem
Here is the uncomfortable truth. A vial can pass every visual inspection and still be significantly degraded. Gradual potency loss from temperature exposure does not change the solution appearance. It does not create particles. It does not cause cloudiness.
The most reliable non-laboratory indicator of degradation is reduced effectiveness. If your retatrutide seems less effective than it was, if appetite suppression has weakened, or if results have stalled despite consistent dosing, potency loss from improper storage should be one of the first things you investigate.
Ask yourself these questions:
Has the vial been out of the refrigerator for extended periods?
Has the vial gone through any freeze-thaw cycles?
How long ago was this vial reconstituted?
Was sterile technique maintained during every draw?
If the answer to any of these raises concerns, a fresh vial may be warranted. The same troubleshooting logic applies when you are not losing weight on tirzepatide or wondering why compounded semaglutide is not working. Storage is often the hidden culprit behind inconsistent results.
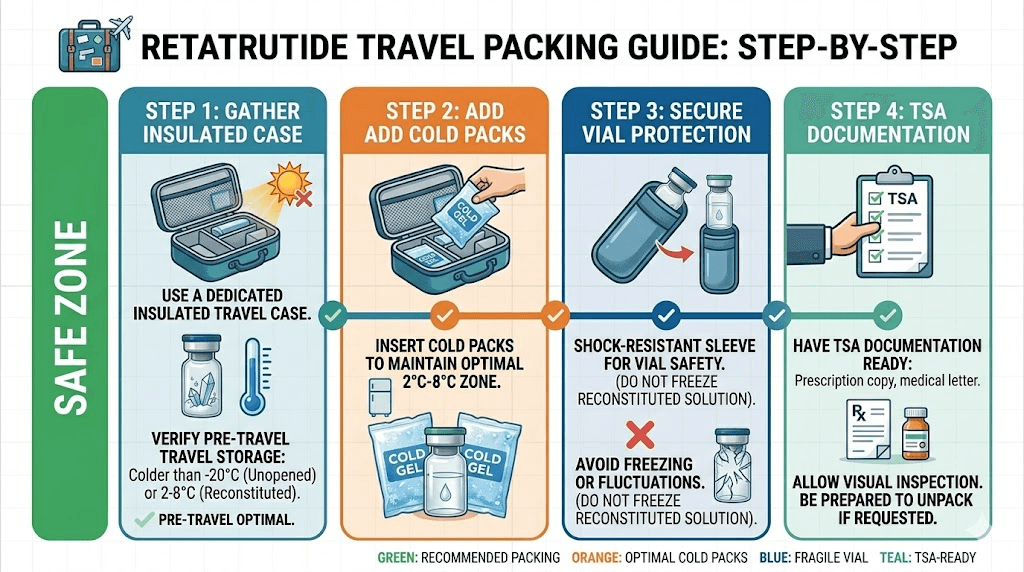
Traveling with retatrutide: maintaining the cold chain
Travel is where most storage protocols break down. You cannot bring your refrigerator on a plane, but you still need to maintain the cold chain for your retatrutide dosing schedule.
Short trips (1 to 3 days)
For weekend trips or brief travel, an insulated medication cooler with a cold pack is sufficient. These portable coolers maintain temperatures between 2 and 8 degrees Celsius for 12 to 24 hours depending on the ambient temperature and insulation quality.
Key tips for short trips:
Pre-chill the cooler and cold packs before departure
Wrap the vial in a cloth or foam sleeve to prevent direct contact with the cold pack (direct contact can freeze the solution)
Keep the cooler in the coolest part of your vehicle, not the trunk on a hot day
Refrigerate the vial immediately upon arrival at your destination
For context on managing injectable medications during travel, the complete guide to traveling with semaglutide and tirzepatide travel guide cover many of the same principles in detail. A dedicated GLP-1 travel case makes the entire process much simpler.
Longer trips (4+ days)
For extended travel, you need a more robust cold chain solution. Options include:
Medical-grade travel coolers with phase-change technology can maintain proper temperatures for 48 to 72 hours or more. These are purpose-built for transporting temperature-sensitive medications and represent the most reliable option for longer trips.
Hotel room mini-fridges work in a pinch, but their temperature control is often inconsistent. Many run warmer than a standard refrigerator. If using a hotel fridge, place the vial toward the back where temperature is most stable and coldest.
Requesting a full-size refrigerator from the hotel is another option. Many hotels accommodate medical needs. Mentioning that you need refrigeration for medication usually gets a prompt response.
Air travel considerations
Always carry your retatrutide in your carry-on bag, never in checked luggage. Checked baggage compartments experience extreme temperature variations and physical impacts that can damage vials. The cabin environment, while not ideal, is far more temperature-controlled.
TSA rules allow medically necessary liquids, including injectable medications, to exceed the standard 3.4-ounce limit when accompanied by a prescription or medical documentation. Keep your medication clearly labeled and accessible for security screening.
An insulated pouch keeps the vial cool during the flight and the time between leaving your home refrigerator and reaching your destination. For longer flights, a cooler with cold packs provides extended temperature protection. Just be aware that gel ice packs must be frozen solid to pass through TSA security checkpoints.
What if the cold chain breaks during travel?
If your retatrutide has been at room temperature for less than 24 hours during travel, return it to refrigeration immediately. The solution is still usable but should be finished sooner.
If it has been warm (above 25 degrees Celsius) for more than 24 hours, assess the situation carefully. Consider how warm it got, how long it was warm, and whether you are early or late in the vial life. A mostly-full vial that was warm for 30 hours may still have acceptable potency for a few more doses, but do not rely on it for the remaining weeks of the expected shelf life.
When in doubt, start a fresh vial. The cost of a replacement vial is always less than the cost of running an unreliable protocol.
Storage comparison: retatrutide vs other GLP-1 peptides
If you have experience storing other peptides, understanding how retatrutide compares helps you calibrate your expectations and apply existing knowledge. The fundamental storage principles are similar across GLP-1 receptor agonists, but some nuances exist.
Storage factor | Retatrutide | Tirzepatide | Semaglutide |
|---|---|---|---|
Lyophilized at -20C | Up to 48 months | Up to 36 months | Up to 36 months |
Lyophilized at 2-8C | 12-24 months | 12-24 months | 12-24 months |
Reconstituted at 2-8C | Up to 30 days | Up to 28 days | Up to 28 days |
Room temp tolerance | Up to 21 days below 30C | Up to 21 days below 30C | Up to 28 days below 30C |
Light sensitivity | High | High | Moderate to high |
Freeze after reconstitution | Never | Never | Never |
The key differences are subtle. Retatrutide lyophilized powder may have slightly longer freezer stability due to its molecular structure, but reconstituted shelf life is comparable across all three compounds. For detailed storage guidance on the others, see the tirzepatide shelf life guide and semaglutide shelf life guide.
For researchers who switch from tirzepatide to retatrutide or are comparing all three compounds, the storage protocols transfer almost directly. If you can properly store one GLP-1 peptide, you can store them all.
Building the perfect retatrutide storage setup
Whether you are a first-time researcher setting up your storage or an experienced user optimizing an existing setup, here is how to build a storage system that protects your retatrutide investment.
Essential equipment
A dedicated medication refrigerator or a section of your home refrigerator. The ideal location is a shelf in the back of the main compartment, away from the door. Door shelves experience the most temperature fluctuation every time you open the fridge. Back shelves maintain the most consistent temperature. If you use a shared refrigerator, a small opaque container keeps your vials dark, protected, and organized.
A refrigerator thermometer. Your refrigerator built-in thermometer, if it even has one, is often inaccurate. A standalone digital thermometer with a probe gives you precise readings. Verify that your refrigerator maintains 2 to 8 degrees Celsius consistently. Many home refrigerators run slightly warm, especially if frequently opened.
Alcohol swabs. Essential for maintaining sterile technique when drawing from reconstituted vials. Wipe the stopper before every draw without exception.
Opaque storage container. A small box, bag, or case that blocks light. Some researchers use amber-tinted vial holders, others simply use a small opaque bag. The goal is zero light exposure during storage.
Desiccant packs for lyophilized storage. Place a silica gel desiccant pack in the same container as your sealed, unreconstituted vials. This absorbs ambient moisture and provides extra protection, especially in humid environments.
Storage organization by form
For unreconstituted (lyophilized) vials:
Long-term supply (months out): freezer at -20C or below, in opaque container with desiccant
Next vial to be reconstituted: can be moved to refrigerator a day or two before mixing
Label each vial with the date received and the lot number if available
For reconstituted vials:
Always in the refrigerator at 2-8C
Label with the reconstitution date and the planned discard date (30 days from mixing)
Store in an opaque container or wrap in foil
Keep away from the door and away from any food items with strong odors
This organizational approach is the same system that works for managing tirzepatide vial lifespan and semaglutide vial shelf life. The principles are universal across injectable peptide research.
The labeling system that prevents mistakes
Every vial in your storage should have a label with these three pieces of information:
Date received (for lyophilized vials)
Date reconstituted (for mixed vials)
Discard by date (30 days after reconstitution for bac water, 24-48 hours for sterile water)
A simple piece of tape and a pen is all you need. This tiny habit prevents the most common storage mistake, which is losing track of how long a vial has been reconstituted and using it well past its reliable shelf life.
Common storage mistakes and how to avoid them
Even experienced researchers make storage errors. Here are the most frequent mistakes and their fixes.
Mistake 1: storing reconstituted vials in the refrigerator door
The door is the warmest, most temperature-variable part of any refrigerator. Every time you open the fridge, the door contents experience a temperature spike. Over weeks of regular use, these repeated fluctuations accelerate degradation.
Fix: Always store vials on a back shelf, preferably in the middle or lower section where temperature stays most consistent.
Mistake 2: using sterile water instead of bacteriostatic water
Sterile water does not contain any preservative. Any bacteria introduced during reconstitution or subsequent draws will multiply freely. Beyond the contamination risk, the lack of preservative means the solution degrades faster.
Fix: Always use bacteriostatic water for multi-use vials. The bacteriostatic water guide covers the proper ratios and techniques.
Mistake 3: drawing from the vial with the same syringe
Reusing syringes introduces bacteria from your skin, the injection site, and the air into the vial. Each draw with a used syringe increases the contamination load in the remaining solution.
Fix: Always use a fresh, sterile syringe for every draw from the vial. The GLP-1 syringe guide covers proper selection and technique.
Mistake 4: shaking the vial to mix
Vigorous shaking creates foam and physical shearing forces that can denature the peptide. Denaturation unfolds the protein structure, potentially destroying the binding sites that make retatrutide effective at its three target receptors.
Fix: Always swirl gently. If the powder does not dissolve immediately, let the vial sit at room temperature for a few minutes and try gentle swirling again. Most lyophilized peptides dissolve within 5 to 10 minutes of gentle handling.
Mistake 5: not tracking reconstitution dates
Without a date label, it is impossible to know how long ago a vial was reconstituted. This leads to researchers using solutions well beyond their safe shelf life, with degraded potency they cannot detect visually.
Fix: Label every vial immediately upon reconstitution. No exceptions.
Mistake 6: storing all supply as reconstituted solution
Some researchers reconstitute their entire supply at once for convenience. This means every vial starts its 30-day countdown simultaneously. By the time you reach the last vial, it may be months old in reconstituted form.
Fix: Only reconstitute one vial at a time. Keep the rest in lyophilized form in the freezer or refrigerator. Reconstitute the next vial when the current one is nearly finished.
Handling shipping and receiving retatrutide
Storage begins the moment your package arrives, and often the most vulnerable period is the one you have the least control over: shipping.
Evaluating your shipment on arrival
When your retatrutide arrives, assess the shipping conditions immediately:
Check the cold pack status. If the package included cold packs or dry ice, are they still cold? Completely thawed cold packs suggest the shipment spent significant time above optimal temperature.
Feel the vial temperature. Is it still cool to the touch, or has it reached room temperature?
Inspect the vial. Is the seal intact? Is the powder still dry and compact, or has it shifted or clumped (suggesting moisture exposure)?
Note the transit time. A next-day delivery that arrived warm is less concerning than a 5-day shipment that arrived warm.
For lyophilized powder that arrived at room temperature but the seal is intact and the powder looks normal, refrigerate or freeze immediately. The stability of lyophilized peptides means short transit periods at room temperature cause minimal degradation. This is one of the advantages of ordering lyophilized versus pre-mixed.
If you are comparing suppliers, Peptide Sciences retatrutide and other online retatrutide suppliers vary significantly in their shipping practices. Suppliers who use insulated packaging with cold packs demonstrate more care for product integrity than those who ship vials in padded envelopes.
What to do with a warm shipment
If your lyophilized retatrutide arrived warm but the seal is intact:
Refrigerate or freeze immediately
The peptide is most likely fine for lyophilized powder
Use it within the standard timeframe after reconstitution
Monitor effectiveness closely during the first week of use
The situation with semaglutide arriving hot is directly comparable. Lyophilized peptides tolerate shipping conditions far better than reconstituted ones.
If your shipment is a pre-mixed (already reconstituted) solution that arrived warm, the risk is much higher. Contact the supplier about a replacement.
Advanced storage considerations
Power outages and refrigerator failure
A power outage is essentially an uncontrolled temperature excursion for everything in your refrigerator. Here is how to handle it:
Keep the refrigerator door closed. An unopened refrigerator maintains safe temperatures for approximately 4 hours during a power outage. Every time you open the door, that window shortens significantly.
If the outage lasts longer than 4 hours, move your vials to an insulated cooler with ice packs. Prioritize reconstituted vials over lyophilized ones, as the reconstituted form is more vulnerable.
After power returns, check your refrigerator thermometer. If the internal temperature stayed below 10 degrees Celsius throughout the outage, your peptides are fine. If it rose significantly above that, assess each vial individually based on its form (lyophilized vs. reconstituted) and the estimated time at elevated temperature.
Aliquoting for extended storage
Some researchers aliquot their reconstituted solution into single-use portions to minimize the number of times they pierce the vial stopper. Each needle insertion is an opportunity for contamination, and reducing the total number of draws from a single vial extends its usable life.
To aliquot safely:
Work in a clean environment, ideally with minimal air movement
Use sterile technique throughout
Prepare labeled sterile vials or containers in advance
Draw from the master vial and dispense into individual containers
Refrigerate all aliquots immediately
The trade-off is that aliquoting introduces more handling steps where contamination can occur. It is only worth doing if your sterile technique is excellent and you are working with larger vials over longer timeframes.
Storing alongside other peptides
Many researchers run multiple protocols simultaneously. Storing retatrutide alongside cagrilintide, tesamorelin, or other peptides is common and perfectly safe as long as each vial is clearly labeled and stored at the proper temperature.
The key risk with multi-peptide storage is confusion. When you have five similar-looking vials in the same container, grabbing the wrong one is easy. Color-coded labels, a consistent organization system, and the habit of reading the label before every draw prevent dosing errors.
Seasonal storage adjustments
Your storage needs shift with the seasons. Summer heat and winter cold both present unique challenges.
Summer considerations
Higher ambient temperatures mean your refrigerator works harder, and any time a vial spends outside the fridge is more dangerous. Room temperature in a non-air-conditioned space can easily reach 30 degrees Celsius or higher during summer months.
During summer:
Minimize the time vials spend out of the refrigerator during draws
Be extra careful about leaving vials on countertops
Check your refrigerator temperature more frequently, as the unit is working harder
Be especially cautious with shipping, as transit temperatures are higher
Consider insulated shipping options even for lyophilized orders
Winter considerations
Cold weather introduces the opposite risk: accidental freezing. Vials stored in unheated spaces, near windows, or in garages during winter can freeze.
For lyophilized powder, accidental freezing is not a concern, it is actually how you want to store long-term supply. But for reconstituted solution, freezing is destructive. Make sure your storage location does not drop below 2 degrees Celsius. This is particularly relevant if your refrigerator is in an unheated garage or basement.
How storage affects your retatrutide protocol results
Storage is not just a preservation concern. It directly impacts your ability to run a consistent, reliable protocol with predictable outcomes.
Dosing accuracy depends on consistent potency
When you calculate your dose based on the retatrutide dosage chart, you are assuming the compound in your vial matches the labeled concentration. If degradation has reduced potency by 20 percent, your actual dose is 20 percent lower than intended. This creates unpredictable results, inconsistent appetite suppression, and a timeline that does not match what the research suggests.
For researchers following a careful starting dose titration, degraded compound is particularly problematic. You may interpret a low response as needing a higher dose when the real issue is that you are not receiving the full dose you intended. This can lead to unnecessary dose escalation that causes problems when you eventually switch to a fresh, full-potency vial.
Why some protocols fail and storage is the hidden cause
If your retatrutide results are not matching expectations, work through this storage troubleshooting checklist before changing your protocol:
When was the vial reconstituted? If more than 4 weeks ago with bac water, potency may be compromised
Has the vial been out of the fridge for extended periods?
Did the vial arrive warm from shipping?
Is your refrigerator maintaining 2-8C consistently?
Have you been using sterile technique for every draw?
Was the reconstitution done with bacteriostatic water or sterile water?
Addressing storage issues before adjusting dosing prevents the cascade of problems that come from compensating for degraded compound. SeekPeptides members frequently report that fixing storage issues alone resolved what seemed like a compound efficacy problem.
The economics of proper storage
Proper storage is not just about maintaining potency. It is about protecting a financial investment.
Consider the math. If improper storage causes 30 percent degradation in a vial, you are effectively throwing away 30 percent of what you paid. Across a multi-month protocol, that waste adds up significantly, especially given the cost of retatrutide.
A refrigerator thermometer costs a few dollars. Alcohol swabs cost pennies per use. Bacteriostatic water is inexpensive. Opaque storage containers and desiccant packs are minimal investments. The total cost of a proper storage setup is negligible compared to the cost of a single wasted vial.
The researchers who get the best results and the lowest cost per effective dose are the ones who treat storage as a critical part of their protocol, not an afterthought. The same logic applies whether you are using retatrutide, looking for affordable retatrutide sources, or managing affordable tirzepatide or cheap compounded semaglutide. Every dollar you save on purchase price is wasted if improper storage degrades the compound before you use it.
Special scenarios and troubleshooting
Vial was frozen after reconstitution by accident
Discard it. Reconstitute a fresh vial. The freeze-thaw damage to the peptide structure cannot be reversed, and the solution may look perfectly fine while being significantly degraded. This is not a situation where you want to take chances.
The powder looks different than expected
Lyophilized retatrutide can vary in appearance between batches and suppliers. It may look like a compact disc, a fluffy cake, or loose powder. All of these are normal variations in the lyophilization process. What matters is that it dissolves cleanly when reconstituted.
However, if the powder looks yellow, brown, or has visible moisture (clumpy or wet-looking), there may be a quality issue. Contact the supplier before proceeding.
The reconstituted solution has tiny bubbles
Small bubbles after reconstitution are normal. They will dissipate on their own if you let the vial sit upright in the refrigerator for 30 minutes to an hour. Do not shake the vial to try to remove them, as shaking creates more bubbles and can damage the peptide.
Large, persistent foam on the surface of the solution is a different matter. Excessive foaming can indicate denaturation (structural damage to the peptide) and may have been caused by too-aggressive mixing. If the foam persists after an hour, the peptide may be compromised.
Refrigerator was accidentally set too cold
If your refrigerator temperature dropped below 0 degrees Celsius and the reconstituted solution partially froze, treat it as a freeze event and discard. If the temperature dropped to 0-2C but the solution did not freeze, it is fine, simply adjust the refrigerator setting.
Lyophilized vials that experienced sub-zero temperatures in the refrigerator are not a concern at all. The freezer is where they ideally should be anyway.
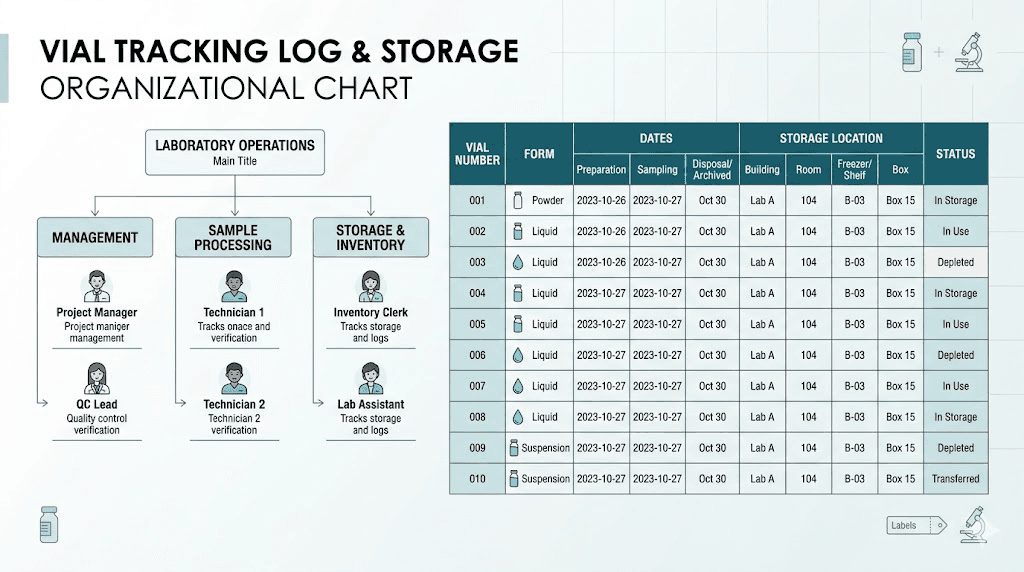
Multiple vials at different stages
When managing multiple vials, keep a simple log:
Vial | Form | Date received | Date reconstituted | Discard by | Storage location |
|---|---|---|---|---|---|
Vial 1 | Reconstituted | March 1 | March 10 | April 9 | Fridge, back shelf |
Vial 2 | Lyophilized | March 1 | Not yet | N/A | Freezer |
Vial 3 | Lyophilized | March 15 | Not yet | N/A | Freezer |
This simple tracking system prevents confusion and ensures you always use the oldest reconstituted vial first.
Storage best practices from experienced researchers
After analyzing hundreds of retatrutide protocols and the storage practices behind them, certain patterns emerge among researchers who get the most consistent results.
They only reconstitute what they need. One vial at a time. The rest stays lyophilized in the freezer. This maximizes the total usable life of their supply and ensures every dose comes from a fresh or recently-mixed vial.
They check refrigerator temperature weekly. A 30-second check with a thermometer catches drift before it becomes a problem. Refrigerators age, seals wear, and settings can be accidentally changed. Catching a temperature problem early prevents silent degradation.
They use a consistent draw schedule. Rather than drawing at random times, experienced researchers draw at the same time on the same days. This consistency means the vial spends a predictable, minimal amount of time outside the refrigerator during each use.
They invest in quality bacteriostatic water. Not all bacteriostatic water is created equal. Sourcing from a reputable supplier ensures the benzyl alcohol concentration is correct and the water is genuinely sterile.
They track everything. Reconstitution dates, lot numbers, supplier information, storage conditions. When something goes wrong, this data helps them identify the cause quickly rather than guessing. This is the level of protocol discipline that SeekPeptides members bring to their research, and it shows in their outcomes.
Frequently asked questions
Does retatrutide need to be refrigerated?
Yes. Reconstituted retatrutide must be refrigerated at 2-8 degrees Celsius at all times. Lyophilized (powder) retatrutide should be refrigerated for short-term storage or frozen at -20C for long-term storage. Room temperature storage causes significant potency loss within days for reconstituted solution and within weeks for powder.
How long does reconstituted retatrutide last in the fridge?
When reconstituted with bacteriostatic water and stored at 2-8 degrees Celsius, retatrutide remains usable for approximately 30 days. Some researchers report stability up to 6-8 weeks under strict sterile conditions, but the conservative recommendation is 4 weeks. This is comparable to tirzepatide fridge life and semaglutide fridge life.
Can I freeze reconstituted retatrutide?
No. Never freeze reconstituted retatrutide. Ice crystal formation during freezing causes physical damage to the peptide structure that cannot be reversed. The solution may look normal after thawing but can be significantly degraded. The same rule applies to tirzepatide freezing.
What happens if I leave retatrutide out overnight?
A single overnight at room temperature (8-12 hours) causes approximately 2-5 percent potency loss. This is not ideal but is recoverable. Return the vial to the refrigerator immediately and plan to use it sooner than originally planned. Repeated temperature excursions are far more damaging than a single incident. For comparison, see what happens when semaglutide is left out overnight.
How can I tell if my retatrutide has gone bad?
Visual signs include cloudiness, particles, discoloration, or unusual odor. However, significant potency loss can occur without any visible changes. The most reliable non-laboratory indicator is reduced effectiveness, weaker appetite suppression or slower results despite consistent dosing. If you suspect degradation, starting a fresh vial is the safest approach.
Does lyophilized retatrutide expire?
Lyophilized retatrutide stored at -20C remains stable for up to 48 months (4 years). At refrigerator temperature (2-8C), it remains stable for 12-24 months. At room temperature, degradation becomes significant within 30 days. Always check for visual changes before reconstituting a stored vial.
What temperature destroys retatrutide?
Temperatures above 30 degrees Celsius (86F) cause rapid degradation. Exposure to 40C or above can destroy the peptide within hours. Car interiors, direct sunlight, and proximity to heat sources are the most common causes of heat damage during storage and transport.
Should I use bacteriostatic water or sterile water?
Bacteriostatic water is strongly recommended for any vial that will be used over multiple days. It contains 0.9% benzyl alcohol, which inhibits bacterial growth and extends usable shelf life to approximately 30 days. Sterile water should only be used for single-use situations, as the solution must be used within 24-48 hours. See the complete bac water mixing guide for retatrutide.
Can I store retatrutide with other peptides?
Yes. Storing multiple peptides in the same refrigerator or freezer is perfectly safe as long as each vial is clearly labeled. Use a consistent organization system to prevent accidentally drawing from the wrong vial. Many researchers store cagrilintide alongside retatrutide or other peptides without any cross-contamination concerns.
How do I store retatrutide when traveling?
Use an insulated medication cooler with cold packs for short trips. For longer travel, medical-grade coolers with phase-change technology maintain temperature for 48-72 hours. Always carry medications in your carry-on for air travel. See the GLP-1 travel case guide for specific product recommendations and packing strategies.
External resources
For researchers serious about optimizing their retatrutide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your vials stay cold, your potency stay high, and your protocols stay consistent.