Mar 31, 2026

Hundreds of women in online support groups report the same unexpected story. They tried for years to conceive. Nothing worked. Then they started semaglutide for weight loss, and within months, they were pregnant. The phenomenon has a name now. People call them "Ozempic babies," and fertility specialists are paying attention. But the question everyone asks, does semaglutide actually make you more fertile, does not have a simple yes or no answer.
The reality involves weight loss, hormones, ovulation, and mechanisms that researchers are still working to fully understand.
Here is what we know so far. Semaglutide is not a fertility drug. The FDA has never approved it for reproductive purposes, and how semaglutide works has nothing to do with directly stimulating egg production or improving sperm quality. Yet the anecdotal evidence is overwhelming. Women who could not conceive for years suddenly find themselves pregnant after starting GLP-1 medications. Men with obesity-related fertility problems see improvements in testosterone and sperm parameters. Something is happening, and the research is beginning to explain what.
This guide covers every angle of semaglutide and fertility. The weight loss connection. The hormonal cascade. The PCOS link. Male fertility data. Birth control interactions that catch people off guard. The mandatory washout period before pregnancy. And the critical safety information that anyone considering semaglutide while planning a family absolutely needs to understand. SeekPeptides built this resource because the topic sits at a complicated intersection of weight management, reproductive health, and medication safety, and getting it wrong carries real consequences.
How semaglutide indirectly affects fertility
Semaglutide does not flip a fertility switch. It does not directly act on your ovaries or testes to produce more eggs or better sperm. What it does, and this is the critical distinction, is create metabolic conditions in the body that allow natural fertility to return. Think of it this way. Obesity disrupts fertility through multiple interconnected pathways. Semaglutide as a GLP-1 receptor agonist addresses the obesity, and the fertility improvements follow. The peptide-based mechanism of semaglutide works by mimicking a natural gut hormone called GLP-1, which regulates appetite, blood sugar, and metabolic function at a fundamental level.
The mechanism works through several channels simultaneously.
First, weight loss itself restores hormonal balance. Excess adipose tissue produces estrogen through a process called aromatization. Fat cells convert androgens into estrogen, creating an imbalance that disrupts the hypothalamic-pituitary-gonadal axis. This axis controls everything about reproduction, from the signals your brain sends to your ovaries to the hormones that trigger ovulation. When you carry significant excess weight, this entire signaling system gets thrown off. The weight loss achieved on GLP-1 medications helps reset this system.
Second, insulin resistance plays a massive role. Obesity almost always involves some degree of insulin resistance, and insulin resistance directly impairs reproductive function. In women, high insulin levels stimulate the ovaries to produce excess androgens like testosterone, which suppresses ovulation. In men, insulin resistance is associated with lower testosterone production and impaired spermatogenesis. Semaglutide improves insulin sensitivity independent of weight loss, meaning it addresses this pathway from two directions at once.
Third, inflammation decreases. Chronic low-grade inflammation accompanies obesity and directly harms reproductive tissue. Inflammatory cytokines damage egg quality, impair implantation, and reduce sperm motility. Semaglutide reduces systemic inflammation as a downstream effect of the weight loss it produces and possibly through direct anti-inflammatory properties of GLP-1 receptor activation itself.
The combined effect of these three mechanisms, reduced excess estrogen, improved insulin sensitivity, and lower inflammation, creates an environment where the reproductive system can function normally again. For many people with obesity-related fertility problems, this is all that was needed. Their bodies were always capable of reproduction. The excess weight was simply getting in the way.
There is also growing evidence that GLP-1 receptors exist in reproductive tissues themselves, including the ovaries, uterus, and testes. This raises the intriguing possibility that semaglutide may have direct effects on reproductive organs beyond what weight loss alone provides. Researchers have found GLP-1 receptor expression in granulosa cells, which surround and nourish developing eggs, and in placental tissue. What these direct effects mean for fertility is still under investigation, but their existence suggests the relationship between GLP-1 receptor activation and reproductive function may be more direct than originally assumed. The fat loss properties of peptide-based therapies like semaglutide create downstream hormonal effects that extend far beyond simple calorie deficit.
The Ozempic babies phenomenon explained
It started on social media. Women posting in weight loss forums about unexpected pregnancies. Not one or two stories. Hundreds. A Facebook group called "I got pregnant on Ozempic" grew to over 500 members. News outlets picked up the trend. Fertility clinics noticed an uptick in patients reporting surprise conceptions after starting GLP-1 medications. The term "Ozempic babies" entered the cultural vocabulary.
But what is actually happening here?
The most likely explanation is straightforward. Many women with obesity do not ovulate regularly. They may have gone years without a normal menstrual cycle, assuming they were infertile or had very low fertility. When semaglutide produced significant weight loss, ovulation returned. And because these women believed they could not get pregnant, many were not using reliable contraception. Surprise pregnancy followed.
Dr. Daniel Kort, a reproductive endocrinologist at Neway Fertility in New York, has explained that even modest weight loss of 5 to 10 percent of body weight can restore ovulation in women who were not ovulating. Semaglutide typically produces weight loss of 10 to 15 percent or more over several months, well beyond this threshold. The results people see on semaglutide often begin within the first few months of treatment, and hormonal changes can precede visible weight loss.
This creates a window of heightened fertility that catches people off guard. A woman who has not had a regular period in years suddenly starts ovulating again. If she is sexually active without contraception, pregnancy becomes a real possibility. And because the effects of semaglutide on menstrual cycles can be unpredictable early in treatment, she may not even realize ovulation has returned before conception occurs.
There is also a second, more concerning aspect of the Ozempic babies phenomenon. Some women became pregnant while actively taking semaglutide, which is not recommended. The FDA classifies semaglutide as not safe during pregnancy due to animal study findings, and the recommendation is to stop semaglutide before trying to conceive. These unexpected pregnancies meant that developing embryos were exposed to the medication during the critical early weeks of development, before the women even knew they were pregnant. The timing of semaglutide doses and the medication long half-life meant that even women who missed a dose were still carrying significant drug levels in their bloodstream during those early weeks.
A large observational study published in 2023 involving over 50,000 pregnant women with Type 2 diabetes found no statistically significant increase in major birth defects among the more than 900 women who were taking GLP-1 receptor agonists when they discovered their pregnancies. This is reassuring but not definitive. The study was observational, not a randomized controlled trial, and the numbers remain relatively small for drawing firm conclusions about rare outcomes.
Semaglutide and PCOS fertility
Polycystic ovary syndrome affects roughly 10 percent of women of reproductive age. It is the most common cause of anovulatory infertility, meaning infertility caused by the absence of ovulation. And semaglutide may be particularly effective at improving fertility in women with PCOS, though it is not FDA-approved for this purpose.
The connection makes biological sense. PCOS is fundamentally a metabolic and hormonal disorder. Its hallmarks include insulin resistance, elevated androgens, irregular or absent menstrual cycles, and often obesity. These are precisely the factors that semaglutide addresses most effectively.
Clinical studies are emerging that support this connection with specific numbers.
A prospective study examining combined metformin and semaglutide therapy in overweight and obese women with PCOS found that 60 percent of treated patients achieved pregnancy during the follow-up period. The mechanism appeared to work through regulatory effects on the hypothalamic-pituitary-ovarian axis, reducing insulin resistance and inflammation, which promoted ovulation and enhanced pregnancy potential.
Another pilot study found that semaglutide combined with metformin demonstrated significant efficacy for both weight reduction and fertility improvement in patients with obesity, prediabetes, and PCOS. The improvements in ovulation rates were substantial enough that researchers described the results as encouraging for fertility applications.
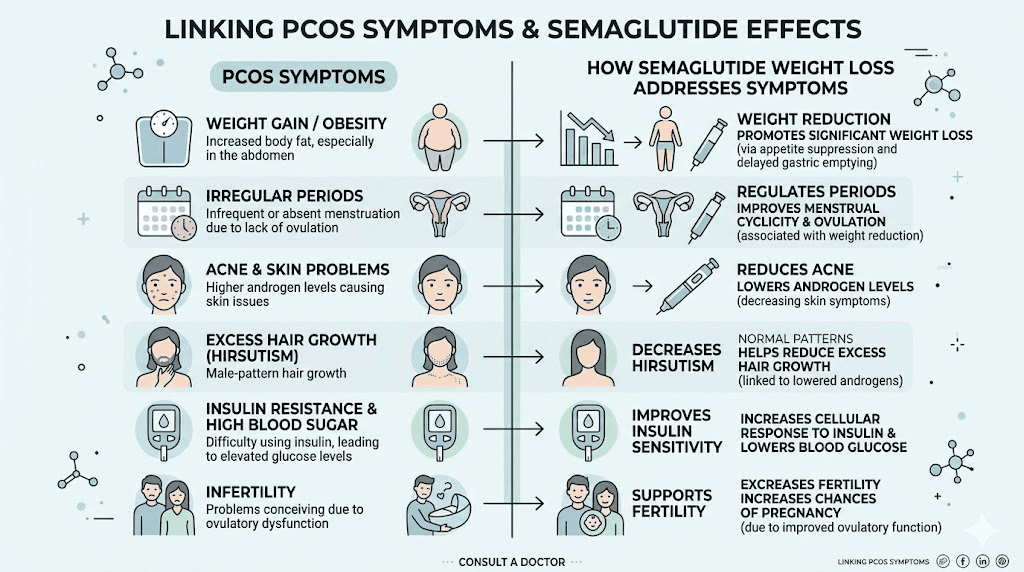
Clinical research shows restoration of ovulation in 60 to 80 percent of women within 6 months of treatment, along with significant reductions in testosterone levels and improvement in fertility markers. These numbers are remarkable for a medication that was not designed as a fertility treatment. They exceed what many dedicated fertility interventions achieve in the same timeframe.
The practical implications for women with PCOS are significant. Traditional first-line treatments for PCOS-related infertility include lifestyle modifications, metformin, and ovulation-inducing medications like clomiphene or letrozole. Semaglutide may represent an additional tool in this arsenal, particularly for women whose PCOS is strongly associated with obesity and insulin resistance. Some practitioners are already prescribing semaglutide off-label for PCOS management, recognizing its potential to address multiple aspects of the condition simultaneously.
However, this creates a paradox that women with PCOS need to understand. Semaglutide may improve your fertility significantly, but you should not be taking it when you actually try to conceive. The strategy that fertility specialists recommend involves using semaglutide for metabolic optimization and weight loss first, then stopping the medication with an adequate washout period before attempting conception. The fertility benefits of the weight loss and metabolic improvement can persist even after discontinuing the drug. Women who experience slower progress in the initial weeks should not be discouraged, as the metabolic improvements that support fertility often precede visible weight changes on the scale.
The connection between PCOS and semaglutide extends to several specific symptoms that affect quality of life beyond fertility. Hair loss and thinning caused by elevated androgens in PCOS often improves as testosterone levels normalize through semaglutide treatment. The fatigue that many PCOS patients experience also tends to improve as insulin resistance decreases. These quality-of-life improvements matter because they affect stress levels, sleep quality, and overall wellbeing, all of which have indirect effects on fertility outcomes. The energy improvements from semaglutide can help patients engage in the physical activity that further supports metabolic health and reproductive function.
How weight loss restores ovulation and reproductive function
The relationship between body weight and fertility is not linear. You do not need to reach a "normal" BMI to see fertility improvements. Research consistently shows that losing just 5 to 10 percent of body weight can restore ovulation in women who were not ovulating due to excess weight. For a 200-pound woman, that means losing 10 to 20 pounds could be enough to restart regular menstrual cycles.
This happens through a cascade of hormonal changes.
As fat tissue decreases, estrogen production from aromatization drops. This allows the hypothalamus and pituitary gland to resume normal signaling. Follicle-stimulating hormone and luteinizing hormone begin cycling properly again. The ovaries respond by developing and releasing mature eggs. Menstrual cycles become more regular and predictable.
Simultaneously, insulin levels drop. Lower insulin means the ovaries produce fewer androgens. Testosterone levels in women decrease, which further supports normal follicular development. The environment inside the ovary shifts from one that suppresses egg maturation to one that supports it.
Semaglutide produces this degree of weight loss reliably. In the SELECT trial, 44 percent of patients using semaglutide lost more than 10 percent of their body weight within two years. Eleven percent lost more than 20 percent. These are substantial numbers, and they translate directly to reproductive benefit for those with weight-related fertility issues. The transformations people experience on semaglutide often include not just physical changes but hormonal normalization that ripples through every system in the body.
The timeline for semaglutide to produce meaningful weight loss typically begins within the first month of treatment, with progressive results over 3 to 6 months. Hormonal changes can precede significant scale changes, meaning ovulation might return before a woman has lost enough weight to notice a major difference in her body. This is one reason the Ozempic babies phenomenon catches so many people by surprise. The internal metabolic shift happens faster than the external physical transformation.
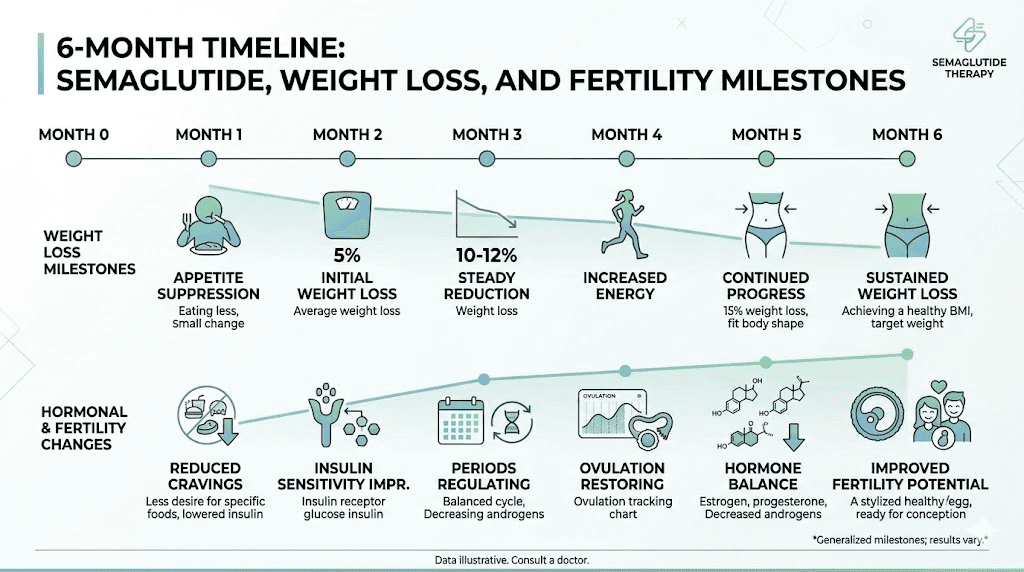
There is also evidence that weight loss improves egg quality, not just ovulation frequency. Obese women undergoing IVF produce eggs with higher rates of chromosomal abnormalities compared to women at healthy weights. Weight loss reduces oxidative stress in the ovarian environment, which may improve the genetic integrity of developing eggs.
While this specific benefit has not been studied directly with semaglutide, the weight loss it produces would logically confer the same advantages seen with weight loss through other methods.
For men, the weight loss story is equally compelling though less discussed. Obesity in men is associated with lower testosterone, higher estrogen, reduced sperm concentration, decreased motility, and increased DNA fragmentation in sperm. Weight loss through any means tends to improve all of these parameters. The metabolic improvements from semaglutide may further support testosterone production and spermatogenesis beyond what weight loss alone would predict. Couples where both partners are overweight should consider that addressing metabolic health in both individuals simultaneously may produce synergistic fertility benefits. While the woman focuses on restoring ovulation, the man can be simultaneously improving sperm quality and achieving meaningful weight loss that supports his hormonal profile.
Semaglutide and male fertility
The conversation about semaglutide and fertility focuses overwhelmingly on women. But male factor infertility contributes to roughly half of all infertility cases, and emerging research suggests semaglutide may benefit male reproductive health as well.
The data is early but promising.
A randomized clinical trial compared semaglutide to testosterone replacement therapy in obese men with type 2 diabetes and functional hypogonadism. The results were surprising. Semaglutide showed a significant increase in morphologically normal sperm, from 2 percent at baseline to 4 percent at study end. Meanwhile, the testosterone replacement therapy group saw decreases in sperm concentration and total sperm count, which is expected since exogenous testosterone suppresses the body natural sperm production.
This finding is clinically meaningful. Many obese men with low testosterone face a difficult choice. Testosterone replacement therapy improves their symptoms but damages their fertility. Semaglutide improved both. It raised testosterone levels comparably to testosterone replacement while simultaneously improving sperm quality rather than harming it. For men who want to address low testosterone without sacrificing their fertility, this represents a potential paradigm shift.
The mechanisms are becoming clearer. GLP-1 receptors have been identified directly in testicular tissue, including Leydig cells that produce testosterone, Sertoli cells that support sperm development, and even on sperm cells themselves. This means semaglutide may have direct effects on testicular function beyond what weight loss alone provides. Research in animal models has shown that GLP-1 receptor activation can improve testicular blood flow, reduce oxidative stress in the testes, and support the signaling pathways that drive spermatogenesis.
Decreased insulin and leptin signaling from obesity disrupts the hypothalamic-pituitary-gonadal axis in men, resulting in low testosterone production and impaired spermatogenesis. Semaglutide addresses this at the root by improving insulin sensitivity and reducing leptin resistance, allowing the hormonal axis to normalize. Understanding how peptides interact with hormonal balance provides additional context for why GLP-1 medications have such broad effects on reproductive health.
However, important caveats apply. The research on semaglutide and male fertility remains limited. Most studies are small, short-term, and focused on specific populations like obese men with diabetes. Large, long-term studies specifically designed to evaluate fertility outcomes in men taking semaglutide do not yet exist. The positive signals are encouraging, but they require confirmation in broader populations before definitive conclusions can be drawn.
Men considering semaglutide with fertility in mind should also be aware of the same washout period recommendations that apply to women. While the concern is less about direct harm to sperm and more about ensuring the medication is cleared before conception occurs through their partner, the conservative approach is to follow the same two-month discontinuation window. Men who also experience changes in sexual function during treatment, whether positive or negative, should discuss these openly with their healthcare provider, as sexual function and fertility are closely linked. The injection technique and site selection for semaglutide do not affect its reproductive impacts, as the medication works systemically regardless of where it is administered.
Birth control interactions you need to know about
This section matters more than most people realize. If you are taking semaglutide and do not want to become pregnant, understanding how the medication interacts with your birth control is essential.
The good news first. A clinical pharmacokinetic study published in the Journal of Clinical Pharmacology found that semaglutide does not reduce the bioavailability of ethinylestradiol or levonorgestrel, two common components of oral contraceptive pills. This means that semaglutide itself does not chemically interfere with how your body absorbs birth control hormones. The same appears true for other GLP-1 medications like liraglutide, exenatide, and dulaglutide.
Now the caveat that changes everything.
While semaglutide does not directly reduce birth control effectiveness, its gastrointestinal side effects absolutely can. Digestive side effects from semaglutide are common, especially during dose escalation. Nausea, vomiting, and diarrhea affect a significant percentage of users. If you vomit within a few hours of taking an oral contraceptive pill, the medication may not have been fully absorbed. If you experience significant diarrhea, absorption can also be impaired. The pill only works if your body absorbs it, and GI disturbance can prevent that.
This indirect interaction is the likely explanation for many of the surprise "Ozempic baby" pregnancies reported in women who believed they were protected by oral contraceptives. They were technically taking their birth control as prescribed, but the digestive side effects of semaglutide were reducing its effectiveness without them realizing it.
There is an additional concern specific to other GLP-1 medications like tirzepatide. Unlike semaglutide, tirzepatide has been shown to directly reduce the absorption of oral contraceptives by approximately 20 percent. The prescribing information for tirzepatide recommends using a backup birth control method for four weeks after starting the medication or increasing the dose.
Practical recommendations for women taking semaglutide who do not want to become pregnant include the following considerations. If you experience frequent nausea, vomiting, or diarrhea, consider switching to a non-oral contraceptive method. IUDs, implants, injections, and patches are not affected by GI disturbances because they bypass the digestive system entirely. If you prefer to stay on oral contraceptives, be vigilant about timing. If you vomit within two hours of taking your pill, take another one as soon as you can keep it down. And during the dose escalation phase when side effects are most common, using a backup method like condoms provides an extra layer of protection.
The combination of restored ovulation from weight loss plus potentially compromised oral contraception creates a perfect storm for unintended pregnancy. Being aware of both factors allows you to make informed decisions about your contraceptive strategy while on semaglutide. Whether you are on compounded semaglutide or brand-name formulations, the same contraceptive considerations apply. The oral formulations of semaglutide may carry slightly different GI side effect profiles than injectable versions, but the fundamental fertility and birth control considerations remain identical.
The two-month washout rule before pregnancy
If you are planning a pregnancy, the timing of when you stop semaglutide matters enormously. The FDA and Novo Nordisk, the manufacturer of both Ozempic and Wegovy, recommend discontinuing semaglutide at least two months before a planned pregnancy. This is not a suggestion. It is a safety-driven recommendation based on the pharmacokinetics of the drug and the findings from animal reproduction studies.
Here is why two months.
Semaglutide has a half-life of approximately one week. This means that after your last injection, it takes about one week for the concentration in your blood to drop by half. After two weeks, it drops by half again. Following this pattern, it takes roughly five to seven weeks for the medication to be essentially eliminated from your body. The two-month recommendation builds in a safety buffer beyond this clearance period, ensuring that semaglutide levels are negligible before conception occurs.
The concern driving this recommendation comes primarily from animal studies. In pregnant rats administered semaglutide during organogenesis, the critical period when organs form, researchers observed embryofetal mortality, structural abnormalities, and alterations to growth. These effects occurred at doses comparable to human therapeutic doses on a body surface area basis, which is why the FDA takes them seriously.
It is important to put this in context. Animal studies do not always predict human outcomes. Many medications that cause problems in rats are perfectly safe in humans, and vice versa. But the standard approach in reproductive medicine is to err on the side of extreme caution. When data is limited, as it is with semaglutide in human pregnancy, the ethical approach is to avoid exposure rather than assume safety.
The MotherToBaby organization, which provides evidence-based information about medication safety during pregnancy, notes that there are limited data with semaglutide use in pregnant women. They cannot rule out a risk to a developing pregnancy, which is the current FDA position as well. This does not mean semaglutide is definitely harmful during pregnancy. It means we do not have enough human data to know for certain, and that uncertainty alone is reason for the conservative two-month washout.
For women who become pregnant unexpectedly while taking semaglutide, the recommendation is to stop the medication immediately and contact a healthcare provider. The reassuring observational data from studies of women who were taking GLP-1 medications early in pregnancy suggests that first-trimester exposure does not appear to dramatically increase the risk of major birth defects, but this data is preliminary and should not be interpreted as an endorsement of using semaglutide during pregnancy.
SeekPeptides emphasizes that timing your semaglutide dosing protocol around pregnancy planning requires careful coordination with your healthcare provider. The ideal approach for many women is to use semaglutide for metabolic optimization and weight loss, achieve your target improvements, then stop the medication at least two months before actively trying to conceive. The metabolic benefits of the weight loss can persist well beyond discontinuation, meaning the fertility advantages you gained do not disappear the moment you stop taking the drug. For those who need to travel while taking semaglutide during this pre-conception phase, maintaining proper medication storage and dosing consistency is important for achieving the metabolic targets that will support your fertility goals.
The washout period also provides an important opportunity to address any digestive issues from semaglutide and restore normal gastrointestinal function before pregnancy. Pregnancy itself commonly causes nausea, constipation, and other GI symptoms, and entering pregnancy with a GI system already stressed by semaglutide could compound these discomforts. Allowing two full months for your digestive system to normalize ensures you start pregnancy from the strongest possible baseline. The dizziness that some users experience on semaglutide also typically resolves during the washout period, which is relevant since early pregnancy can cause its own balance and lightheadedness issues.
Using semaglutide strategically before IVF
For couples pursuing in vitro fertilization, semaglutide presents both an opportunity and a timing challenge. Obesity reduces IVF success rates through multiple mechanisms. It impairs egg quality, reduces response to ovarian stimulation medications, complicates egg retrieval procedures, and lowers implantation rates. Addressing obesity before an IVF cycle can meaningfully improve outcomes.
Research shows that overweight patients who lose just 5 percent of their body weight can see improvements in ovulation rates, applying to both patients with PCOS and those without. For IVF specifically, this weight loss translates to better response to stimulation medications, higher quality eggs, and improved embryo development. The dietary changes that complement semaglutide further support the metabolic optimization that enhances IVF outcomes.
The timing protocol that fertility specialists recommend typically follows this structure. Start semaglutide 4 to 6 months before your planned IVF cycle. Use this time to achieve meaningful weight loss and metabolic improvement while following a nutrition plan optimized for semaglutide. Stop semaglutide at least 8 weeks, and ideally closer to 10 weeks, before planned oocyte retrieval. This provides adequate time for the medication to clear while preserving the metabolic benefits.
Some fertility clinics are beginning to incorporate GLP-1 medications into their pre-IVF optimization protocols for patients with elevated BMI. This is an off-label use, but the logic is sound. The weight loss and metabolic improvements from semaglutide address many of the factors that reduce IVF success in obese patients. Improved insulin sensitivity alone can enhance ovarian response to stimulation medications, potentially resulting in more and better quality eggs.
However, semaglutide does not directly improve egg quality, embryo development, or IVF success rates in and of itself.
The benefits come entirely from the metabolic and weight loss improvements it produces. A woman who achieves the same degree of weight loss through diet and exercise alone would see similar IVF benefits. Semaglutide simply makes achieving that weight loss more reliable and predictable for many patients.
The cost-benefit calculation for using semaglutide before IVF is worth considering. IVF cycles are expensive, often costing $15,000 to $25,000 or more per attempt. If a few months on semaglutide improves the success rate of that cycle meaningfully, the investment in pre-cycle optimization can pay for itself many times over by reducing the number of cycles needed. For couples exploring different GLP-1 medication options for pre-IVF optimization, the choice between semaglutide, tirzepatide, and other agents should be guided by the magnitude of weight loss needed, the timeline before the planned IVF cycle, and individual tolerance profiles. Your fertility specialist and prescribing provider should collaborate on this decision.
Timeline of fertility changes while taking semaglutide
Understanding when fertility changes occur on semaglutide helps with both planning and precaution. The timeline is not identical for everyone, but general patterns have emerged from clinical experience and research.
Weeks 1 through 4
During the initial dose escalation phase, the first weeks on semaglutide primarily involve gastrointestinal adjustment. Weight loss may begin, but hormonal changes at this stage are typically minimal. Insulin sensitivity starts improving almost immediately, which is one of the earliest metabolic changes. For most women, menstrual cycles remain unchanged during this period. However, if you experience significant GI side effects like vomiting, the appetite suppression from semaglutide may already be affecting your eating patterns enough to shift body composition.
Weeks 4 through 8
This is when meaningful weight loss typically begins to accumulate. Most patients have reached a therapeutic dose or are approaching it. Insulin levels are dropping. For women with obesity-related anovulation, this may be the earliest window where ovulation could potentially resume. Hormonal shifts including estrogen level changes can begin during this period as fat mass decreases. If you are not planning a pregnancy, this is the time to ensure your contraceptive method is reliable.
Months 2 through 4
Significant weight loss is now occurring for most patients. Hormonal profiles are shifting meaningfully. Women who were not menstruating may begin having periods again. Those with irregular cycles may notice increased regularity. Testosterone levels in women with PCOS are typically declining, which supports follicular development. For men, testosterone levels may begin rising as insulin resistance improves and body fat decreases. This window represents the period of highest unexpected pregnancy risk because fertility may be returning while patients are still actively taking the medication.
Months 4 through 6
By this point, the metabolic and hormonal improvements are well established. Women who are going to regain ovulation from the weight loss have typically done so. Some patients may be approaching a weight loss plateau, but hormonal benefits continue to consolidate. This is often the ideal window for those planning to stop semaglutide in preparation for conception, as the metabolic improvements are substantial and can be maintained with proper nutrition and lifestyle habits after discontinuation.
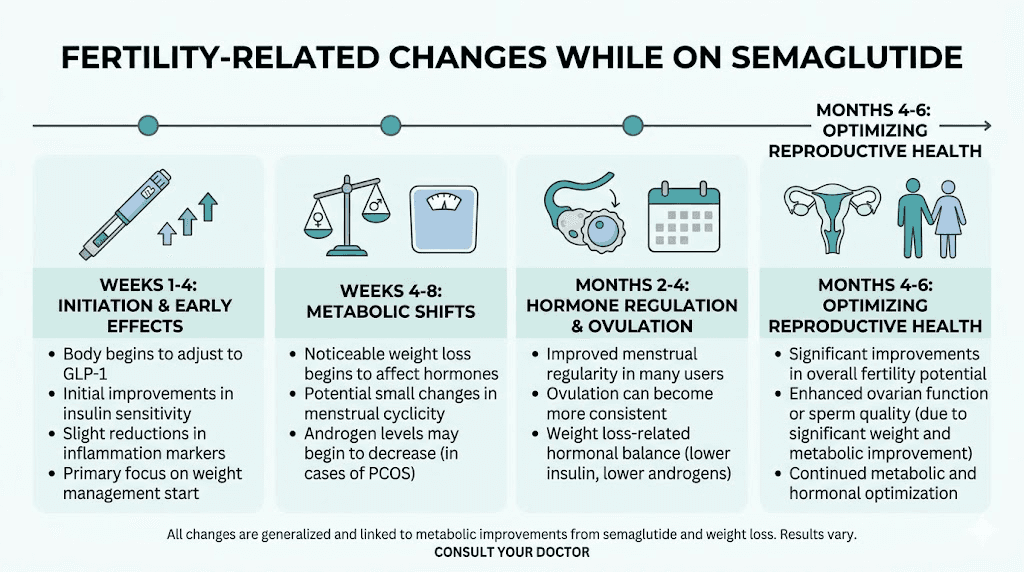
After discontinuation
The fertility benefits from weight loss and metabolic improvement do not vanish when you stop taking semaglutide. As long as you maintain the weight loss through dietary strategies and exercise, the hormonal improvements persist. Ovulation continues.
Insulin sensitivity remains improved. The two-month washout period allows the drug to clear while these benefits remain intact. This is the key insight that makes the sequential approach, use semaglutide for optimization then stop before conception, viable as a fertility strategy.
Some weight regain is common after stopping semaglutide, and discontinuation can come with challenges. Working with a healthcare provider to develop a maintenance plan that preserves your metabolic gains during the washout period and early pregnancy is important for sustaining the fertility benefits you worked to achieve. Some patients find that transitioning to other weight management support during the washout helps maintain results, though any medication used during the conception window must be pregnancy-safe. The criteria used to qualify patients for semaglutide often overlap with the BMI thresholds where obesity most significantly impairs fertility, making the medication a natural fit for this pre-conception optimization role.
What the animal studies actually showed
The animal data behind the semaglutide pregnancy warnings deserves a closer look. Too many summaries simply state "animal studies showed problems" without explaining what was found, at what doses, and what that means for humans. Understanding the details helps you have more informed conversations with your healthcare provider.
In the primary animal reproduction studies, pregnant rats were given semaglutide during organogenesis. At doses of 0.03 mg/kg per day and higher, researchers observed increased embryofetal mortality, structural abnormalities, and growth alterations. The structural abnormalities included skeletal and visceral malformations. These findings were dose-dependent, meaning higher doses produced more severe effects.
To put the doses in context, the human therapeutic dose for weight management with Wegovy reaches 2.4 mg per week, which works out to roughly 0.34 mg per week for a 70 kg person, or about 0.05 mg/kg/week. The rat studies used daily dosing at levels that, when adjusted for body surface area differences between species, are considered relevant to human exposure. This is why the FDA takes the findings seriously, the exposure levels are not dramatically different from what humans experience therapeutically.
An additional finding in the animal studies was an increase in estrous cycle length and a small reduction in the number of corpora lutea. Corpora lutea are the structures that form in the ovary after ovulation and produce progesterone to support early pregnancy. A reduction in corpora lutea numbers suggests that semaglutide may affect ovarian function at a fundamental level, which has implications for both fertility and early pregnancy maintenance.
In rabbit studies, semaglutide at doses approximately 4 times the maximum recommended human dose based on body surface area resulted in increased early pregnancy losses. Similar adverse effects were seen in cynomolgus monkeys at higher exposure levels.
However, several important caveats apply when interpreting these results. Rats and rabbits have different reproductive physiology from humans. The metabolic demands placed on pregnant rodents by semaglutide, including significant weight loss and reduced food intake, may themselves have contributed to adverse outcomes independent of any direct toxic effect on the embryo. In other words, some of the observed harm may have been from malnutrition-related effects of the drug extreme appetite suppression rather than from direct developmental toxicity.
This distinction matters because it changes how we think about the risk. If the primary danger comes from severe caloric restriction during pregnancy, that is a risk that can be managed by stopping the medication before conception. If the risk comes from a direct toxic effect of the semaglutide molecule on developing tissues, the washout period becomes even more critical to ensure complete clearance.
The current scientific consensus treats both possibilities as real and supports the conservative approach of complete drug clearance before conception. The two-month washout period addresses both scenarios by ensuring the medication is gone and normal appetite has returned before pregnancy begins. The shelf life and stability of semaglutide are separate considerations, but understanding the medication pharmacokinetics helps explain why the clearance period is measured in weeks rather than days. Visual changes in the medication can indicate degradation, and using degraded product could affect the consistency of treatment outcomes during the optimization phase before planned pregnancy.
When semaglutide helps fertility and when it does not
Not everyone who takes semaglutide will experience fertility improvements. The medication is most likely to help in specific situations and much less likely to benefit in others. Understanding where you fall on this spectrum can set realistic expectations.
Semaglutide is most likely to improve fertility when
Obesity is the primary driver of your infertility. If you carry significant excess weight and have irregular or absent menstrual cycles, weight loss from semaglutide has a strong chance of restoring ovulation. The more directly your fertility issues are linked to excess body fat, insulin resistance, and the hormonal disruptions they cause, the more likely semaglutide is to help.
You have PCOS with insulin resistance. This is perhaps the population most likely to benefit. PCOS involves a cluster of metabolic abnormalities that semaglutide directly addresses. Women with PCOS who achieve meaningful weight loss on semaglutide frequently see improvements in menstrual regularity, androgen levels, and ovulation rates.
Your male partner has obesity-related low testosterone. As discussed earlier, semaglutide can improve testosterone levels and sperm quality in obese men while preserving, and even enhancing, spermatogenesis. This is a significant advantage over testosterone replacement therapy, which improves hormone levels but suppresses sperm production.
You are preparing for IVF and need to optimize metabolic health first. Using semaglutide for pre-cycle weight loss can improve your response to stimulation medications and potentially increase the success rate of your IVF cycle.
Semaglutide is unlikely to improve fertility when
Your infertility has a structural cause. Blocked fallopian tubes, uterine abnormalities, severe endometriosis, or absent vas deferens are not going to respond to weight loss or improved insulin sensitivity. These require surgical or procedural interventions that semaglutide cannot replace.
You are already at a healthy weight. If your BMI is in the normal range and you do not have insulin resistance, the mechanisms through which semaglutide improves fertility are largely absent. Weight loss in already lean individuals can actually harm fertility by disrupting hormonal function in the opposite direction, potentially causing hypothalamic amenorrhea.
Age-related egg quality decline is the primary issue. A woman at age 41 with diminished ovarian reserve is unlikely to see meaningful improvements from semaglutide. Egg quantity and chromosomal quality decline with age through mechanisms that are independent of weight and metabolic health. While optimizing weight may provide marginal benefits, it cannot reverse the fundamental biology of ovarian aging.
Your partner has a genetic or structural cause of male infertility.
Conditions like Klinefelter syndrome, Y-chromosome microdeletions, or azoospermia from testicular failure will not respond to semaglutide. These require specialized reproductive medicine evaluation and treatment.
The bottom line is that semaglutide improves fertility when the problem is metabolic. It does not when the problem is structural, genetic, or age-related. Most fertility issues are multifactorial, and for many couples, metabolic optimization through semaglutide addresses one important piece of a larger puzzle.
Semaglutide and estrogen, testosterone, and other reproductive hormones
The hormonal effects of semaglutide extend well beyond simple weight loss. Understanding these hormonal shifts provides insight into how and why fertility changes occur.
Estrogen
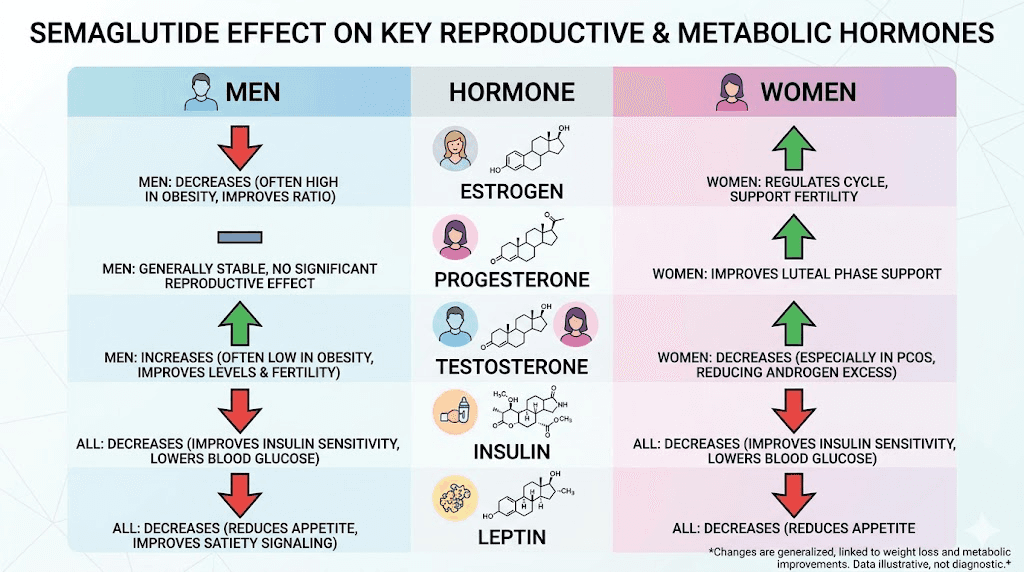
Adipose tissue is an active endocrine organ that produces estrogen through the aromatase enzyme. In obese women, this peripheral estrogen production can significantly elevate total estrogen levels, creating a state of relative hyperestrogenism. This excess estrogen suppresses FSH production through negative feedback, which impairs follicular development and ovulation. As semaglutide reduces body fat, estrogen levels shift toward a more normal range, restoring the delicate hormonal balance needed for regular ovulation.
In obese men, the same aromatization process converts testosterone to estrogen, creating both low testosterone and elevated estrogen simultaneously. This double hormonal insult suppresses the HPG axis and impairs spermatogenesis. Weight loss from semaglutide reduces this estrogen excess while allowing testosterone to rise.
Testosterone
In women with PCOS, semaglutide-driven weight loss typically reduces testosterone levels, which is beneficial because excess androgens are a primary driver of anovulation and other PCOS symptoms. The reduction in insulin levels plays a direct role here, as insulin stimulates ovarian androgen production through its action on theca cells.
In men, the effect is opposite and equally beneficial. Semaglutide increases testosterone, particularly in obese men with functional hypogonadism. GLP-1 receptor agonist therapy has been associated with significant increases in total testosterone concentrations, with the improvements being most pronounced in obese men and individuals with metabolic dysfunction. Understanding how GLP-1 medications interact with testosterone provides broader context for these effects across different medication types.
Progesterone
Progesterone production depends on ovulation. Without ovulation, the corpus luteum does not form, and progesterone levels remain low. By restoring ovulation, semaglutide indirectly normalizes progesterone production in women who were previously anovulatory. Adequate progesterone is essential for endometrial preparation and early pregnancy maintenance, making this an important downstream effect of restored ovulation.
Insulin
While not a sex hormone, insulin plays a central role in reproductive function. Semaglutide reduces both fasting and postprandial insulin levels through improved insulin sensitivity. Lower insulin reduces ovarian androgen production in women, supports hypothalamic GnRH pulsatility, and improves testicular Leydig cell function in men. The insulin-lowering effect of semaglutide may be one of its most important mechanisms for fertility improvement, potentially more significant than the weight loss itself in some cases.
Leptin
Leptin, produced by fat cells, signals the brain about energy stores. In obesity, leptin resistance develops, disrupting the reproductive axis. High leptin levels interfere with GnRH pulsatility and directly impair gonadal function. As semaglutide reduces body fat and improves metabolic signaling, leptin sensitivity improves, which helps restore normal reproductive hormone cycling.
Safety considerations for semaglutide and reproduction
Beyond the washout period and animal study findings already discussed, several additional safety considerations deserve attention for anyone thinking about semaglutide in the context of fertility and reproduction.
Nutritional adequacy
Semaglutide significantly reduces appetite, and many users eat substantially less while on the medication. This caloric restriction, while beneficial for weight loss, raises concerns about nutritional adequacy in the months leading up to conception. Folate, iron, vitamin D, omega-3 fatty acids, and other nutrients critical for fertility and early fetal development may be insufficient if food intake drops too dramatically. The appetite suppression timeline varies between individuals, but most users experience significant reductions in food intake within the first month, which is when nutritional monitoring should begin.
Women planning to conceive after a period of semaglutide use should ensure they are taking a comprehensive prenatal vitamin for at least three months before attempting conception. Strategic supplementation while on GLP-1 medications can help prevent nutritional deficiencies that could impair fertility or early pregnancy outcomes. B12 supplementation with semaglutide deserves particular attention, as both GI changes and reduced food intake can affect B12 status.
Rapid weight loss and pregnancy
Very rapid weight loss releases stored toxins from fat tissue, including fat-soluble environmental chemicals and endocrine disruptors. There is theoretical concern that this mobilization of stored toxins could be harmful during early pregnancy. This is another reason the washout period is valuable. It allows the rapid weight loss phase to conclude and toxin mobilization to stabilize before conception.
Gallbladder issues
Rapid weight loss from any cause increases the risk of gallstone formation. Semaglutide is no exception. Gallstone complications during pregnancy can be serious and may require surgical intervention. Women who have experienced significant weight loss on semaglutide should discuss gallbladder screening with their healthcare provider before conceiving.
Mental health considerations
The hormonal and body composition changes from semaglutide can affect mood and emotional wellbeing. Combined with the hormonal shifts of early pregnancy, this creates a period of significant physiological adjustment. Sleep disturbances that some users experience with semaglutide can compound these effects. Having mental health support in place during the transition from semaglutide use to pregnancy planning is a wise precaution.
Breastfeeding
Semaglutide is not recommended during breastfeeding. It is present in the milk of lactating rats, and there is no data on its presence in human breast milk. Women who want to breastfeed should not resume semaglutide until after weaning. Understanding the guidelines for GLP-1 medications and breastfeeding is essential for postpartum planning. The same consideration applies to tirzepatide and breastfeeding, as all GLP-1 receptor agonists share similar safety profiles regarding lactation. Women who used semaglutide before pregnancy and plan to resume after breastfeeding should discuss this timeline with their provider well in advance.
Medication interactions
Women pursuing fertility treatment often take multiple medications simultaneously, including ovulation induction drugs, progesterone supplements, and prenatal vitamins. While semaglutide should be stopped before active fertility treatment begins, understanding potential interactions during the overlap period matters. The L-carnitine combinations with semaglutide and other supplement stacks should be reviewed with a healthcare provider who understands both the weight management and fertility aspects of your treatment plan. Similarly, hormone replacement therapy and GLP-1 interactions deserve careful evaluation for women who are using hormonal support alongside semaglutide.
What fertility specialists are saying
The medical community response to semaglutide and fertility has evolved rapidly. Initially, most reproductive endocrinologists focused solely on the safety concerns, advising patients to avoid GLP-1 medications entirely around conception. Now, a more nuanced view is emerging.
Fertility specialists at major academic centers are increasingly recognizing the potential benefits of semaglutide for pre-conception metabolic optimization, particularly in patients with obesity and PCOS. The approach most recommend involves what researchers call "sequential therapy." Use semaglutide for metabolic improvement and weight loss first, then transition off the medication with an adequate washout before attempting conception or starting fertility treatment.
The American Society for Reproductive Medicine has not yet issued formal guidelines specifically addressing GLP-1 medications and fertility treatment.
But individual practitioners and some fertility clinics have developed protocols that incorporate semaglutide into pre-treatment optimization plans.
These protocols typically recommend 3 to 6 months of semaglutide therapy followed by a minimum 2-month washout before ovulation induction, intrauterine insemination, or IVF.
The key message from most fertility specialists is that semaglutide should be viewed as a tool for improving the conditions for fertility, not as a fertility treatment itself. It addresses the metabolic barriers to conception. It does not directly enhance reproductive capacity. This distinction matters for setting realistic expectations and for avoiding the misconception that semaglutide is a shortcut to pregnancy.
SeekPeptides members gain access to comprehensive protocols and expert-reviewed guides that help navigate complex topics like the intersection of weight management medications and reproductive health. Understanding the nuances of how long to stay on semaglutide and when to transition off requires reliable information that goes beyond surface-level advice.
Practical protocol for using semaglutide with fertility goals
For women and couples who want to leverage semaglutide for metabolic optimization before trying to conceive, a structured approach maximizes benefits while minimizing risks.
Phase 1: metabolic optimization (months 1 through 6)
Begin semaglutide at the standard starting dose and titrate up as tolerated. Focus simultaneously on nutrition quality, not just calorie reduction. Ensure adequate intake of fertility-critical nutrients including folate, iron, DHA, vitamin D, zinc, and coenzyme Q10. Begin a prenatal vitamin immediately if you are not already taking one. Track menstrual cycles to monitor for changes in regularity and signs of ovulation return. Your semaglutide dosage should be managed in consultation with your prescribing provider throughout this phase. Understanding proper injection technique ensures consistent dosing throughout the optimization period. If you experience significant side effects that limit food intake, glycine-enhanced formulations or niacinamide combinations may help reduce gastrointestinal discomfort.
Phase 2: consolidation (months 4 through 6)
By this point, significant weight loss and metabolic improvement should have occurred. If menstrual cycles have normalized, this is a positive sign that ovulation has returned. Begin establishing the dietary and exercise habits that will maintain your weight after discontinuing semaglutide. A sustainable nutrition plan becomes critical during this transition. Discuss your conception timeline with your healthcare provider and reproductive specialist if applicable.
Phase 3: washout (months 6 through 8)
Stop semaglutide at least 8 to 10 weeks before planned conception attempts. Continue the nutritional and lifestyle strategies from Phase 2 to maintain your metabolic gains. Monitor your weight closely. Some regain is normal, but the goal is to minimize it. Understanding what happens when you stop semaglutide helps set realistic expectations for this period. Continue prenatal vitamins. Get comprehensive fertility testing during this window if you have not already. Be aware that alcohol consumption patterns should also be evaluated during this phase, as both the washout period and pre-conception period benefit from reduced or eliminated alcohol intake. Consider whether supplements like berberine might help maintain metabolic improvements during the transition off semaglutide, though all supplements should be cleared with your provider when pregnancy is planned.
Phase 4: conception
After the washout period, begin actively trying to conceive. Your body should now be in an optimized metabolic state with normal ovulation, improved insulin sensitivity, and balanced reproductive hormones. If you are pursuing fertility treatment like IUI or IVF, your clinic will have specific protocols for timing.
This phased approach treats semaglutide as a preparatory tool rather than an ongoing fertility intervention. The benefits persist beyond the medication because the weight loss, improved insulin sensitivity, and hormonal normalization are maintained through the lifestyle changes established during treatment.
Comparing semaglutide to other fertility-adjacent weight loss approaches
Semaglutide is not the only path to the weight loss that can improve fertility. Understanding how it compares to alternatives helps couples make informed decisions about their pre-conception strategy.
Semaglutide versus lifestyle modification alone
Diet and exercise remain the foundational recommendation for weight loss in the fertility context. Lifestyle modification produces the same metabolic and hormonal benefits as pharmaceutical weight loss. The difference is magnitude and reliability. Most people lose 3 to 5 percent of body weight through lifestyle changes alone, while semaglutide typically produces 10 to 15 percent weight loss. For fertility purposes, the additional weight loss from semaglutide may cross critical thresholds that lifestyle changes alone do not reach. Whether exercise is combined with semaglutide or not, the medication consistently produces greater weight loss than lifestyle changes alone, which translates to more reliable fertility improvements in the obesity-related infertility population.
Semaglutide versus metformin
Metformin has been used for decades in the fertility context, primarily for women with PCOS. It improves insulin sensitivity and can promote ovulation. However, metformin produces relatively modest weight loss compared to semaglutide. The combination of metformin and semaglutide may be synergistic, as shown in PCOS studies. Importantly, metformin has a much longer safety track record in pregnancy and is sometimes continued into early pregnancy for women with PCOS or gestational diabetes risk.
Semaglutide versus tirzepatide
Tirzepatide compared to semaglutide produces greater average weight loss in head-to-head trials. For fertility optimization, the additional weight loss from tirzepatide might provide greater metabolic benefits. However, tirzepatide has a direct interaction with oral contraceptives that semaglutide does not, reducing absorption by about 20 percent. The unexpected pregnancy risk with tirzepatide may be higher for this reason. Both medications require the same two-month washout before planned conception.
Semaglutide versus bariatric surgery
Bariatric surgery produces the most dramatic weight loss and metabolic improvement of any intervention. It also produces the most significant fertility improvements, particularly in severely obese patients. However, surgery carries its own risks, requires a longer recovery period before conception is safe, typically 12 to 18 months, and creates unique nutritional challenges during pregnancy. Semaglutide offers a less invasive alternative for patients who do not meet surgical criteria or prefer a non-surgical approach. The simplicity of semaglutide preparation and use compared to the complexity of surgical recovery makes it an attractive option for couples who want to move to conception on a shorter timeline.
Semaglutide versus other GLP-1 medications
Beyond tirzepatide, other GLP-1 receptor agonists like liraglutide have been studied in the fertility context, particularly for PCOS. Liraglutide was one of the first GLP-1 medications investigated for its effects on reproductive hormones in women with PCOS, and the results were positive. However, semaglutide has largely superseded liraglutide due to its greater efficacy for weight loss and its once-weekly dosing convenience. Older weight loss medications like phentermine have their own risks during pregnancy and are generally not recommended in the pre-conception period. The newer sublingual formulations of semaglutide offer additional dosing flexibility, though the fertility and pregnancy safety considerations remain identical regardless of administration route.
Common myths about semaglutide and fertility
Misinformation about semaglutide and fertility is widespread. Clarifying the most common myths helps ensure your decisions are based on accurate information.
Myth: semaglutide is a fertility drug
It is not. Semaglutide has never been approved, studied, or marketed as a fertility treatment. Any fertility benefits are secondary to its effects on weight, metabolism, and insulin sensitivity. Using it specifically as a fertility intervention without medical supervision is not recommended.
Myth: you cannot get pregnant on semaglutide
You absolutely can. Many women have become pregnant while actively taking semaglutide. The Ozempic babies phenomenon demonstrates this clearly. If you are taking semaglutide and are sexually active, you can get pregnant, and you may be at increased risk of unintended pregnancy if your weight loss has restored ovulation. Even women who have been diagnosed with infertility and have struggled with weight loss for years have reported unexpected conceptions once semaglutide produced meaningful metabolic changes.
Myth: semaglutide causes birth defects
This is not established. Animal studies showed adverse effects on embryo development, but human data has not confirmed these findings. The large observational study of women who took GLP-1 medications in early pregnancy did not find a statistically significant increase in major birth defects. The FDA position is that risk cannot be ruled out, which is different from saying risk has been confirmed.
Myth: the fertility effects disappear when you stop semaglutide
The metabolic and hormonal improvements from weight loss persist after stopping semaglutide, as long as the weight loss is maintained. Ovulation does not cease the moment you discontinue the medication. The fertility benefits are driven by the physiological changes in your body, not by the ongoing presence of the drug. The duration of semaglutide use matters less than the degree of metabolic improvement achieved. Understanding what happens when restarting semaglutide after pregnancy and breastfeeding is also relevant for long-term planning, as many women will want to resume the medication postpartum for continued weight management.
Myth: men do not need to worry about semaglutide and fertility
Male fertility is affected by the same metabolic factors that semaglutide addresses. Obese men on semaglutide should be aware of both the potential benefits to their sperm quality and testosterone levels and the same safety considerations regarding timing and washout periods before their partner attempts conception.
Myth: if you lost weight on semaglutide, any weight loss method would have given the same fertility benefit
This is partially true but incomplete. The weight loss itself is the primary driver of fertility improvement, and equivalent weight loss from other methods would produce similar benefits. However, semaglutide may have additional effects through direct GLP-1 receptor activation in reproductive tissues that go beyond what weight loss alone provides. This direct effect hypothesis is still under investigation, but early evidence suggests it may be real.
Questions to ask your healthcare provider
If you are considering semaglutide and have fertility concerns, having a prepared set of questions for your doctor can make your appointment more productive. Here are the conversations that matter most.
Ask about your specific fertility diagnosis and whether metabolic optimization through semaglutide could address any of the contributing factors. Not all infertility has a metabolic component, and understanding your specific situation determines whether semaglutide could help. Inquire about the recommended timeline. How long should you take semaglutide, what target weight or metabolic markers should you aim for, and when should you stop relative to conception attempts?
Discuss contraception during semaglutide treatment. If you are not ready to conceive, your doctor should ensure your contraceptive method is not vulnerable to GI side effects. Ask about nutritional monitoring. Should you have blood work checked for fertility-critical nutrients during treatment? What supplements should you take? Raise the question of coordination with a reproductive endocrinologist if you are also pursuing fertility treatment. Your weight management provider and fertility specialist should be communicating about your case.
Finally, ask about the plan for if you become pregnant unexpectedly while on semaglutide. Knowing what to do before it happens is far better than scrambling afterward. The immediate steps are straightforward, stop the medication and contact your provider, but discussing it in advance reduces anxiety if the situation arises. You should also ask about medication storage and expiration to ensure you are always using properly stored product during your treatment, as degraded medication may produce inconsistent results that complicate your fertility timeline. If your provider prescribes compounded semaglutide that requires reconstitution, proper mixing technique directly affects dosing accuracy, which matters when you are trying to achieve specific metabolic targets before transitioning to your conception phase.
The role of diet and lifestyle alongside semaglutide for fertility
Semaglutide creates a powerful metabolic shift, but it works best when supported by intentional dietary and lifestyle choices. For fertility optimization specifically, certain nutritional strategies become even more important than they are for weight loss alone.
Protein intake deserves primary attention. Many semaglutide users under-eat protein because the appetite suppression from the medication makes all food less appealing. But protein is essential for egg quality, hormonal production, and maintaining lean muscle mass during weight loss. Aiming for at least 0.8 grams of protein per pound of goal body weight helps preserve the metabolic tissue that supports reproductive function. Choosing nutrient-dense foods becomes critical when total food volume decreases.
Fertility-specific nutrients that require special attention during semaglutide treatment include folate, which is essential for preventing neural tube defects and should be supplemented as methylfolate rather than synthetic folic acid for optimal absorption. Iron status should be monitored, particularly in women who have been restricting calories.
Vitamin D plays a direct role in ovarian function and implantation, and many people with obesity are deficient. Omega-3 fatty acids from fish or supplements support egg and sperm quality.
CoQ10 has been shown to improve egg quality in women over 35 and may complement the metabolic benefits of semaglutide.
Exercise during semaglutide treatment for fertility should emphasize moderate-intensity activity rather than extreme training. Excessive exercise combined with semaglutide caloric restriction can create an energy deficit severe enough to suppress the reproductive axis, which is the opposite of what you want. Walking, moderate resistance training, yoga, and swimming are excellent choices that support both weight loss and reproductive health without overwhelming the system. The fatigue that some GLP-1 users experience can make intense exercise difficult anyway, and listening to your body during this phase is more important than pushing for maximum caloric burn.
Sleep quality profoundly affects both fertility and semaglutide effectiveness. Poor sleep disrupts the hormonal rhythms that control ovulation, reduces insulin sensitivity, and increases cortisol, all of which work against fertility goals. Prioritizing 7 to 9 hours of quality sleep per night supports the metabolic improvements that semaglutide produces and creates a hormonal environment more conducive to conception. Addressing sleep disturbances from semaglutide early in treatment prevents them from undermining the broader fertility benefits of the medication.
Stress management rounds out the lifestyle picture. Chronic stress elevates cortisol, which directly suppresses reproductive hormone production and can prevent ovulation even when all other metabolic factors are optimized. The emotional stress of infertility itself can become a self-reinforcing cycle that semaglutide cannot address pharmacologically. Mind-body practices, therapy, support groups, and deliberate stress reduction strategies complement the metabolic optimization from semaglutide and create the holistic conditions most supportive of conception.
Frequently asked questions
Does semaglutide directly increase fertility?
No. Semaglutide is not a fertility drug and does not directly stimulate ovulation or improve egg quality. However, the weight loss and metabolic improvements it produces can indirectly restore fertility in people whose reproductive function was impaired by obesity, insulin resistance, or related hormonal imbalances. Studies show the weight loss achieved on GLP-1 medications can restore ovulation in 60 to 80 percent of anovulatory women within 6 months. The peptide dosage calculators on SeekPeptides can help you understand dosing fundamentals for various peptide-based therapies.
How long before trying to conceive should I stop semaglutide?
The FDA and the manufacturer recommend stopping semaglutide at least two months before a planned pregnancy. This allows the medication, which has a one-week half-life, to fully clear from your body. Some fertility specialists recommend a slightly longer washout of 8 to 10 weeks for additional safety margin. Our complete guide to semaglutide and pregnancy planning covers this timeline in detail.
Can semaglutide make my birth control less effective?
Semaglutide itself does not directly reduce oral contraceptive effectiveness. However, gastrointestinal side effects like nausea, vomiting, and diarrhea can reduce the absorption of oral birth control pills. Additionally, semaglutide-driven weight loss can restore ovulation in women who were previously not ovulating, increasing the chance of pregnancy even with contraception. Consider using a non-oral contraceptive method while on semaglutide if you experience GI side effects.
Is it safe to take semaglutide during pregnancy?
No. Semaglutide is not recommended during pregnancy. Animal studies showed adverse effects on embryo development at therapeutic doses, and there is insufficient human data to confirm safety. If you become pregnant while taking semaglutide, stop the medication immediately and contact your healthcare provider. The medication remains in your system for several weeks after your last dose due to its long half-life.
Can semaglutide help with PCOS-related infertility?
Emerging research suggests yes. Clinical studies show that semaglutide, alone or combined with metformin, can restore ovulation, reduce androgen levels, and improve pregnancy rates in women with PCOS and obesity. One study found that 60 percent of PCOS patients treated with combined semaglutide and metformin achieved pregnancy. However, semaglutide is not FDA-approved for PCOS, and this remains an off-label use.
Does semaglutide affect male fertility?
Preliminary research shows positive effects. A clinical trial found that semaglutide improved sperm morphology and increased testosterone levels in obese men with type 2 diabetes and hypogonadism, while preserving sperm production. This contrasts with testosterone replacement therapy, which improves hormone levels but suppresses sperm production. However, research on GLP-1 medications and male reproductive health is still in early stages.
What are Ozempic babies?
Ozempic babies is the popular term for unexpected pregnancies that occur in women taking semaglutide or similar GLP-1 medications for weight loss. These surprise pregnancies typically happen because semaglutide-driven weight loss restores ovulation in women who believed they were infertile, and many were not using reliable contraception. The phenomenon has been widely reported in media and online support groups, with hundreds of women sharing similar stories. Similar reports have emerged with tirzepatide and unexpected pregnancies, suggesting this is a class effect of GLP-1 medications rather than specific to semaglutide alone.
Will my fertility benefits disappear after stopping semaglutide?
The fertility improvements from semaglutide are primarily driven by weight loss and metabolic changes, not by the ongoing presence of the drug. As long as you maintain the weight loss through proper nutrition and exercise after discontinuing, the hormonal improvements and ovulation regularity should persist. However, significant weight regain after stopping can reverse these benefits.
External resources
For researchers serious about understanding how GLP-1 medications interact with reproductive health, SeekPeptides provides the most comprehensive resource available. Members access evidence-based guides, expert-reviewed protocols, and a community of thousands who have navigated these exact questions about semaglutide, tirzepatide, and other peptide-based therapies.
In case I do not see you, good afternoon, good evening, and good night. May your hormones stay balanced, your cycles stay regular, and your family planning stay informed.