Mar 3, 2026

You started semaglutide expecting weight loss. Not this. Not the itching, the burning, the uncomfortable discharge that showed up seemingly out of nowhere. And now you are wondering whether your semaglutide protocol caused it, or if something else entirely is going on.
You are not alone. Thousands of women on semaglutide have reported yeast infections during treatment. Forums are full of these stories. Reddit threads overflow with questions. And yet, when you check the official side effects list for Ozempic or Wegovy, yeast infections are nowhere to be found.
That disconnect between what women experience and what the clinical data shows is exactly why this topic deserves a thorough, honest examination. The relationship between semaglutide and yeast infections is not as simple as cause and effect. It involves blood sugar fluctuations, hormonal shifts from rapid weight loss, gut microbiome disruptions, and a fascinating biological connection between your digestive tract and vaginal health that most guides completely ignore. This article covers all of it, from the mechanisms driving these infections to the specific protocols that help prevent them, so you can continue your GLP-1 therapy without this unwelcome complication derailing your progress.
Does semaglutide actually cause yeast infections?
The short answer is no. Not directly.
Semaglutide is a GLP-1 receptor agonist that mimics the incretin hormone GLP-1. It slows gastric emptying, reduces appetite, and helps regulate blood sugar. None of these mechanisms involve increasing glucose in the urine, altering vaginal pH directly, or suppressing immune function in ways that would cause fungal overgrowth.
This matters because there IS a class of diabetes medication that directly causes yeast infections. SGLT2 inhibitors, drugs like Farxiga, Invokana, and Jardiance, work by forcing excess glucose out through urine. That sugar-rich environment in the urinary tract creates a feast for Candida albicans, the fungus responsible for most yeast infections. Clinical data shows yeast infection rates of 10 to 15 percent in women taking SGLT2 inhibitors and 3 to 7 percent in men. It is a well-documented, well-understood side effect.
Semaglutide does not work this way. The GLP-1 pathway does not increase glucosuria. It does not dump sugar into your urinary tract. And in clinical trials for Ozempic and Wegovy, yeast infection rates were not elevated compared to placebo groups.
So if semaglutide does not directly cause yeast infections, why are so many women experiencing them?
Because the story is more complicated than direct pharmacological causation. The indirect pathways, the ones that involve everything semaglutide does to your body beyond appetite suppression, are where the real answers live.
The indirect mechanisms that connect semaglutide to yeast infections
Five distinct mechanisms explain why women on semaglutide experience higher rates of yeast infections, even though the drug itself does not cause them. Understanding each one is critical for prevention.
Blood sugar fluctuations during treatment adjustment
This is the most straightforward pathway. Candida albicans thrives on glucose. Higher blood sugar means more available fuel for fungal growth. Research consistently shows that poor diabetic control, defined as HbA1c levels above 9 percent, is associated with significantly higher rates of Candida infections compared to well-controlled blood sugar levels below HbA1c of 6 percent.
Here is where it gets nuanced. When you start semaglutide, your blood sugar does not simply drop in a smooth, linear fashion. There are adjustment periods. Fluctuations. Days where blood sugar runs higher than expected, followed by days where it drops lower than your body is accustomed to. These swings create windows of vulnerability where Candida can establish itself.
For people with type 2 diabetes starting semaglutide, this transitional period is particularly risky. Your body has been accustomed to elevated glucose levels. The semaglutide dosing protocol starts low and escalates gradually, which means weeks or months of blood sugar adjustment. During that window, existing Candida colonies may flare before better glucose control eventually reduces their food supply.
For non-diabetic individuals using semaglutide for weight loss, this mechanism matters less. But it still plays a role if you have any degree of insulin resistance, which many people with obesity do.
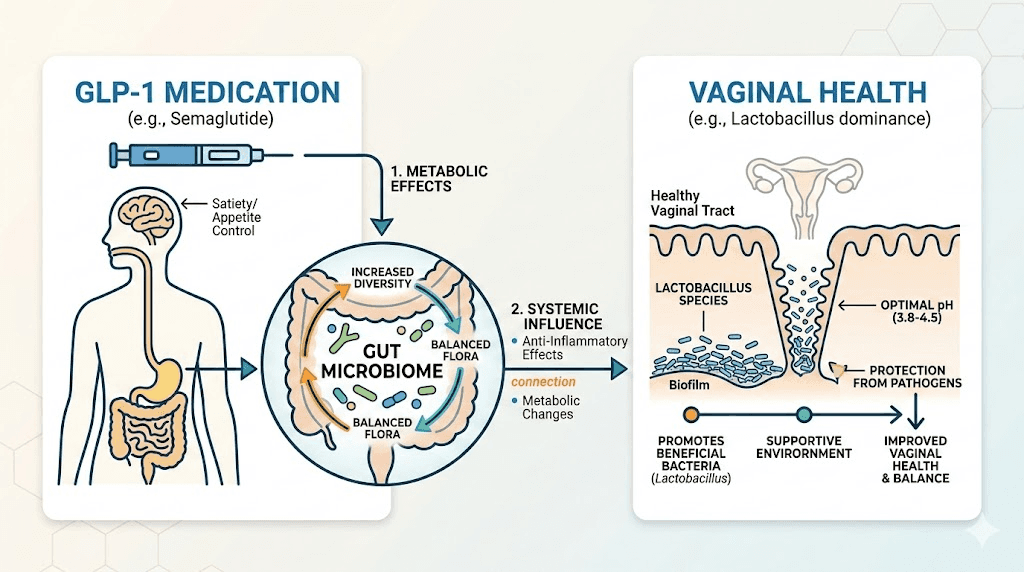
Gut microbiome disruption and the gut-vagina axis
This is the mechanism most people miss. And it is arguably the most important one.
Your gut and your vaginal microbiome are not separate systems. They communicate through something researchers call the gut-vagina axis. The bacterial populations in your digestive tract directly influence the bacterial populations in your vaginal tract. When one gets disrupted, the other often follows.
Semaglutide profoundly affects your gut microbiome. Research published in 2024 showed that semaglutide alters the composition, richness, and diversity of gut microbiota. It increases beneficial bacteria like Bifidobacterium and Bacteroidota while reducing potentially harmful genera like Klebsiella. In many ways, these changes are positive. They represent a healthier gut environment.
But the transition period matters. When semaglutide slows gastric emptying, the entire digestive environment changes. Food moves through your system differently. The pH of different gut segments shifts. Some bacterial populations that previously thrived in your old digestive environment decline, while others expand into the newly available territory.
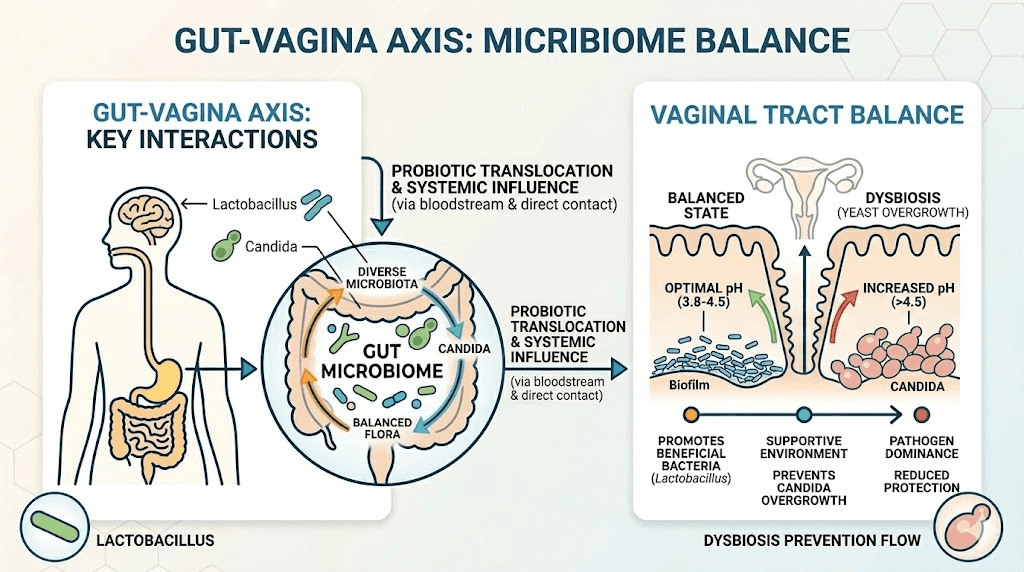
During this rebalancing, the gut-vagina axis can transmit disruption. When protective Lactobacillus levels in the gut fluctuate, the vaginal microbiome often loses Lactobacillus populations as well. And Lactobacillus is the primary defender against vaginal Candida. These bacteria produce lactic acid that maintains vaginal pH between 3.8 and 4.5, an environment hostile to yeast. When Lactobacillus levels drop, vaginal pH rises, and Candida seizes the opportunity.
The gastrointestinal side effects of semaglutide, nausea in 15 to 44 percent of users, vomiting, diarrhea, and bloating, further destabilize the gut microbiome. Frequent vomiting or diarrhea flushes beneficial bacteria from the system. Reduced food intake means less prebiotic fiber reaching the lower gut where those bacteria live. Every one of these factors cascades through the gut-vagina axis.
Hormonal shifts from rapid weight loss
Fat tissue is not passive storage. It is an active endocrine organ that produces estrogen through a process called aromatization, where fat cells convert testosterone into estradiol. When you lose body fat rapidly on semaglutide, you lose estrogen production capacity along with it.
Estrogen performs critical functions in vaginal health. It thickens vaginal tissue. It stimulates the production of glycogen, which Lactobacillus bacteria feed on to produce the lactic acid that keeps vaginal pH acidic. It supports blood flow to vaginal tissues, maintaining moisture and immune function.
A rapid decline in estrogen does several things simultaneously. Vaginal tissue becomes thinner and more fragile. Glycogen production drops, starving the Lactobacillus that depend on it. Vaginal pH rises toward neutral, creating conditions where Candida can flourish. And vaginal moisture decreases, which reduces the flushing mechanism that naturally clears away fungal cells.
This is why the phenomenon some people call "Ozempic vagina" or "Ozempic vulva" exists. It is not the semaglutide directly affecting vaginal tissue. It is the weight loss that semaglutide produces, specifically rapid weight loss, that triggers hormonal shifts affecting vaginal health.
The effect is dose-dependent and speed-dependent. Slower, more gradual weight loss gives the body time to adjust estrogen production from other sources. Rapid weight loss, the kind semaglutide is particularly good at producing, does not allow that adjustment period. Clinical trials showed participants losing up to 15 to 20 percent of body weight on semaglutide, and that magnitude of fat loss can significantly impact circulating estrogen levels.
Women in perimenopause or menopause face compounded risk here. Their estrogen production is already declining naturally. Adding rapid fat loss on top of age-related hormonal decline can push vaginal health past a tipping point where infections become likely.
Dehydration from gastrointestinal side effects
This mechanism is surprisingly important and rarely discussed.
Semaglutide side effects like nausea, vomiting, and diarrhea cause fluid loss. Many women on semaglutide also report reduced thirst, making it harder to maintain adequate hydration even without active fluid loss. The result is systemic dehydration that affects every mucosal surface in the body, including vaginal tissue.
Vaginal moisture serves a protective function beyond comfort. The fluid that keeps vaginal tissue moist contains antimicrobial compounds. It provides a physical barrier against pathogen adhesion. It maintains the slightly acidic environment that Lactobacillus requires. When hydration drops, all of these protective mechanisms weaken.
Dehydration also concentrates urine, which can irritate vulvar tissue and create micro-abrasions that Candida exploits as entry points. Women who experience dizziness alongside their semaglutide treatment are often significantly dehydrated, and that dehydration may be contributing to recurrent vaginal infections they attribute to the medication itself.
Dietary changes and reduced nutrient intake
Semaglutide dramatically reduces appetite. Most people on the medication eat significantly less than they did before starting treatment. And the foods they do eat often shift toward simpler, easier-to-digest options, especially during the early weeks when nausea is most pronounced.
This dietary shift has consequences for vaginal health. Reduced fiber intake means less prebiotic fuel for gut Lactobacillus, which cascades through the gut-vagina axis. Reduced overall caloric intake can impair immune function, weakening the body's ability to keep Candida populations in check. And many of the micronutrients that support vaginal immune defense, zinc, vitamin A, vitamin D, iron, may be consumed in insufficient quantities when total food intake drops significantly.
Women following a semaglutide diet plan that emphasizes protein and limits carbohydrates may inadvertently reduce their intake of prebiotic-rich foods like whole grains, legumes, and certain vegetables that feed beneficial bacteria.
The best foods to eat on semaglutide should include probiotic and prebiotic sources specifically to counter this effect, but many people on GLP-1 medications are not aware of this connection.
Who is most at risk for yeast infections on semaglutide?
Not everyone on semaglutide will develop yeast infections. Certain risk factors make some women significantly more vulnerable than others. Understanding these factors helps you assess your personal risk and take targeted preventive action.
Women with type 2 diabetes
If you are taking semaglutide for diabetes management rather than weight loss alone, your risk is inherently higher. Years of elevated blood sugar may have already created an environment where Candida is established. The transition period as semaglutide brings your glucose levels down creates the fluctuations discussed earlier. And if you are also taking an SGLT2 inhibitor alongside semaglutide, the compounded risk is substantial.
Research using standard semaglutide protocols for diabetes showed that participants with HbA1c above 9 had significantly higher Candida colonization rates than those with better glucose control. The transition from poor to good control does not immediately eliminate existing Candida colonies, and they can flare during the adjustment period.
Women experiencing rapid weight loss
The faster you lose weight, the more dramatic the hormonal shifts. Women who respond particularly well to semaglutide, losing 15 percent or more of body weight within the first six months, face greater estrogen disruption than those losing weight more gradually.
If you are on a higher semaglutide dose and losing weight quickly, your risk goes up. This does not mean you should slow your progress intentionally, but it does mean you should be proactive about vaginal health support.
Women in perimenopause or menopause
Estrogen levels are already declining. Fat loss compounds this decline. The combination of age-related hormonal changes and rapid weight loss can create severe vaginal dryness, thinning, and pH disruption that makes yeast infections almost inevitable without intervention.
Women over 40 on semaglutide should consider discussing vaginal estrogen therapy with their healthcare provider as a preventive measure, not just a reactive one.
Women with a history of recurrent yeast infections
If you have experienced three or more yeast infections in a single year at any point in your life, you carry a higher baseline susceptibility. Starting semaglutide adds multiple additional risk factors on top of that existing vulnerability. Proactive prevention is essential rather than optional for this group.
Women taking concurrent antibiotics
Antibiotics kill bacteria indiscriminately. They wipe out the protective Lactobacillus alongside the pathogenic bacteria they target. If you need antibiotics while on semaglutide, the combined assault on your vaginal Lactobacillus population makes yeast infection almost certain without probiotic intervention.
Ask your doctor about prophylactic antifungal treatment or aggressive probiotic supplementation whenever antibiotics are prescribed during your semaglutide treatment course.
The SGLT2 confusion: why people mix up the drug classes
A significant part of the confusion around semaglutide and yeast infections comes from mixing up GLP-1 agonists with SGLT2 inhibitors. Both are used for type 2 diabetes. Both are sometimes prescribed for weight management. And many patients take both simultaneously. But their relationship to yeast infections is completely different.
How SGLT2 inhibitors directly cause yeast infections
SGLT2 inhibitors block the sodium-glucose cotransporter 2 in the kidneys. Normally, this transporter reabsorbs glucose from urine back into the bloodstream. When blocked, glucose stays in the urine and gets excreted. This lowers blood sugar, which is the therapeutic goal. But the glucose-rich urine creates an ideal growth medium for Candida around the vulvar and perineal area.
The incidence is not subtle. Clinical trials show yeast infection rates of 10 to 15 percent in women taking SGLT2 inhibitors. The FDA requires this risk to be prominently disclosed. It is a known, expected, well-documented side effect.
SGLT2 inhibitors linked to yeast infections include Farxiga (dapagliflozin), Invokana (canagliflozin), Jardiance (empagliflozin), and Steglatro (ertugliflozin).
How GLP-1 agonists do not directly cause yeast infections
GLP-1 agonists like semaglutide work through completely different mechanisms. They mimic the GLP-1 hormone, stimulating insulin release when blood sugar is elevated, suppressing glucagon, slowing gastric emptying, and reducing appetite. None of these mechanisms increase glucose in urine or directly create conditions favorable for yeast growth.
In clinical trials for semaglutide, yeast infections were not listed as a common adverse event. They appeared at rates comparable to placebo groups. This is why the FDA does not require yeast infection warnings on semaglutide labels.
Why the confusion persists
Many diabetes patients take both drug classes simultaneously. An SGLT2 inhibitor alongside semaglutide is a common combination therapy. When a patient develops a yeast infection while taking both medications, they may attribute it to semaglutide rather than the SGLT2 inhibitor. Online discussions then perpetuate this attribution.
Additionally, the indirect mechanisms discussed above, blood sugar fluctuations, gut microbiome changes, hormonal shifts from weight loss, can produce real yeast infections in semaglutide users. These infections are real. The women experiencing them are not imagining symptoms. But the cause is indirect, not the direct pharmacological action of semaglutide.
Understanding this distinction matters for treatment and prevention. If your yeast infections are caused by an SGLT2 inhibitor you take alongside semaglutide, the solution might involve switching the SGLT2 inhibitor. If they are caused by the indirect effects of semaglutide, the solution involves addressing those specific mechanisms.
Symptoms of a yeast infection to watch for during semaglutide treatment
Knowing what to look for helps you catch infections early, when they are easiest to treat. Some symptoms overlap with normal adjustment effects from semaglutide, which makes awareness particularly important.
Classic yeast infection symptoms
Vaginal itching is usually the first sign. It starts mild and escalates. The itching localizes to the vulvar area and vaginal opening, and it tends to worsen at night or after showering.
Burning comes next. It is most noticeable during urination, when acidic urine contacts inflamed tissue, and during sexual activity. The burning is distinct from the general discomfort some women feel from vaginal dryness on semaglutide. Yeast-related burning has an acute, irritation quality rather than the dull dryness sensation.
Discharge changes are the third major symptom. Yeast infections produce a characteristic thick, white, cottage-cheese-like discharge. It is typically odorless or has a mild, bread-like smell. This differs from bacterial vaginosis, which produces a thin, grayish discharge with a distinct fishy odor.
Redness and swelling of the vulvar tissue accompany most yeast infections. The labia may appear visibly inflamed, and the skin around the vaginal opening may look raw or irritated.
How yeast infection symptoms differ from semaglutide side effects
Some women mistake early vaginal changes from semaglutide-related hormonal shifts for yeast infections, or vice versa. Here is how to tell the difference.
Vaginal dryness from weight loss produces a generalized dryness without itching, usually without unusual discharge, and tends to worsen gradually over weeks to months as weight loss accumulates. It does not come with swelling or redness.
Yeast infections produce localized itching with discharge, come on relatively quickly over days, and involve visible inflammation. They do not resolve on their own the way hormonal dryness might stabilize.
If you are unsure, do not self-diagnose. See your healthcare provider for a simple swab test that can definitively identify whether Candida is present. This is especially important if it is your first suspected yeast infection while on semaglutide, because starting antifungal treatment for a non-fungal issue wastes time and money while the actual problem goes unaddressed.
When to see a doctor immediately
Certain symptoms warrant urgent medical attention rather than self-treatment. See your provider if you experience symptoms for the first time and are not certain it is a yeast infection. Go in if you have had three or more infections in a single year. Seek care if over-the-counter treatments have not resolved symptoms after 7 days. Contact your provider immediately if you develop fever, chills, or pelvic pain alongside vaginal symptoms, as these could indicate a more serious infection.
Severe redness, swelling, or ulcerations also require professional evaluation. And if your symptoms keep returning despite treatment, your provider may need to evaluate whether an underlying condition, medication interaction, or resistant Candida strain is involved.
Prevention strategies that actually work
Prevention is significantly more effective than treatment for yeast infections during semaglutide therapy. The following strategies address each of the indirect mechanisms discussed above and, used together, can dramatically reduce your risk.
Strategy 1: Aggressive probiotic supplementation
This is the single most impactful intervention for preventing yeast infections during semaglutide treatment. Probiotics directly replenish the Lactobacillus populations that semaglutide-related gut changes can deplete.
Not all probiotics are equal for this purpose. The strains with the strongest evidence for vaginal health include Lactobacillus rhamnosus GR-1, Lactobacillus reuteri RC-14, Lactobacillus crispatus, and Lactobacillus acidophilus. Look for products that specifically list these strains.
Clinical data supports their effectiveness. In one trial, 79 percent of women taking probiotics alongside antifungal treatment were free of yeast infections at day 90, compared to only 43 percent taking antifungals alone. That is nearly double the cure rate from adding probiotics.
Timing matters. Start probiotics before you begin semaglutide if possible, or at the same time. Do not wait until a yeast infection develops. The goal is to maintain Lactobacillus populations through the gut microbiome disruption that semaglutide causes, not to rebuild them after they have already collapsed.
Both oral and vaginal probiotics can be beneficial. Oral probiotics address the gut-vagina axis from the source. Vaginal probiotic suppositories deliver Lactobacillus directly where it is needed. Using both simultaneously provides the most comprehensive protection.
Strategy 2: Maintain hydration aggressively
Dehydration contributes to yeast infections through multiple pathways. It concentrates urine, irritating vulvar tissue. It reduces vaginal moisture, weakening physical barriers against Candida. And it impairs overall immune function.
During semaglutide treatment, especially in the early weeks when nausea and reduced appetite make eating and drinking difficult, intentional hydration becomes non-negotiable. Aim for at least 2 to 3 liters of water daily. More if you are experiencing vomiting or diarrhea.
Water is best. Avoid sugary beverages, which feed Candida. Herbal teas and electrolyte solutions without added sugar are acceptable alternatives. Some women find that adding a small amount of lemon juice to water helps with semaglutide-related nausea while supporting hydration.
Strategy 3: Optimize your diet for microbiome support
What you eat while on semaglutide directly affects your yeast infection risk. Even though your appetite is reduced, the foods you choose matter enormously.
Prioritize prebiotic fiber. Foods like garlic, onions, leeks, asparagus, bananas, oats, and flaxseed feed beneficial gut bacteria. Even small amounts make a difference when total food intake is reduced. A semaglutide-friendly food plan should include these daily.
Include fermented foods. Yogurt with live cultures, kefir, sauerkraut, kimchi, and miso all provide live beneficial bacteria that support both gut and vaginal microbiome health. Aim for at least one serving daily.
Limit added sugar. Candida feeds on sugar. While semaglutide itself reduces blood sugar, consuming concentrated sources of sugar still gives yeast a growth advantage. This does not mean eliminating all carbohydrates, complex carbohydrates from whole grains and vegetables are important. It means avoiding candy, sugary drinks, pastries, and other concentrated sugar sources.
Ensure adequate protein. Your protein intake on GLP-1 therapy supports immune function. The immune system needs amino acids to produce the antibodies and white blood cells that keep Candida in check. Many women on semaglutide do not eat enough protein, which can weaken immune defense over time.
Strategy 4: Practice targeted hygiene
Basic hygiene practices make a meaningful difference in yeast infection prevention, but some common practices actually increase risk.
Do: Wear breathable cotton underwear. Keep the vulvar area clean and dry. Change out of wet swimsuits or sweaty workout clothes promptly. Wipe front to back after using the toilet. Shower after exercise.
Do not: Douche. Ever. Douching disrupts the vaginal microbiome and increases, not decreases, infection risk. Do not use scented soaps, body washes, or feminine hygiene sprays on the vulvar area. Avoid tight-fitting synthetic underwear that traps moisture. Do not use antibacterial soap on the vulvar area, as it kills protective Lactobacillus along with everything else.
Clean warm water is sufficient for vulvar cleansing. If you prefer soap, use a mild, unscented, pH-balanced cleanser designed for intimate use. Nothing else should contact this area.
Strategy 5: Monitor and manage your dose escalation carefully
The risk of gut microbiome disruption, and by extension yeast infections, correlates with the severity of gastrointestinal side effects. Slower dose escalation produces less gut disruption.
If you are following a standard semaglutide escalation protocol and experiencing significant GI side effects, discuss extending the time at each dose level with your provider. The standard escalation can be slowed without reducing the medication's long-term effectiveness. Many prescribing clinicians now recommend extended escalation periods specifically to improve tolerability and reduce secondary complications.
Some women find that splitting their semaglutide dose across two injections per week rather than one reduces GI symptoms and, by extension, gut disruption. Discuss this option with your provider if single weekly injections produce severe nausea or diarrhea.
Strategy 6: Consider vaginal estrogen therapy if appropriate
For women in perimenopause or menopause, or for premenopausal women experiencing significant vaginal dryness from rapid weight loss on semaglutide, topical vaginal estrogen can be transformative.
Vaginal estrogen comes in creams, rings, and suppositories. It restores vaginal tissue thickness, stimulates glycogen production that feeds Lactobacillus, restores vaginal pH to its protective acidic range, and improves vaginal moisture. Unlike systemic hormone therapy, vaginal estrogen delivers extremely low levels of hormone with minimal systemic absorption.
This is not something to self-prescribe. Discuss it with your healthcare provider, who can evaluate whether vaginal estrogen is appropriate based on your medical history, age, and risk factors. But do not dismiss it as unnecessary. For many women on semaglutide, vaginal estrogen is the single most effective intervention for preventing both yeast infections and the broader vaginal health changes associated with rapid weight loss.
Treatment options when prevention falls short
Even with the best prevention strategies, some women will develop yeast infections during semaglutide treatment. Prompt, effective treatment prevents infections from becoming recurrent and minimizes the impact on your GLP-1 therapy.
Over-the-counter antifungal treatments
For straightforward, non-recurrent yeast infections, over-the-counter treatments are usually the first line of defense. These include miconazole (Monistat), clotrimazole (Gyne-Lotrimin), and tioconazole (Vagistat-1).
These come as vaginal creams, suppositories, or ointments. Treatment courses range from 1 to 7 days depending on the product. Shorter courses contain higher concentrations of antifungal medication. Longer courses use lower concentrations and tend to cause less irritation.
For women on semaglutide, the 3-day or 7-day treatment courses generally work better than single-dose options. The indirect mechanisms that caused the infection, gut microbiome disruption, hormonal changes, are ongoing, which means the yeast has a more favorable growth environment than it would in someone not taking GLP-1 medication. A longer treatment course gives the antifungal more time to clear the infection thoroughly.
Prescription antifungal treatment
Fluconazole (Diflucan) is the standard prescription treatment for yeast infections. It is an oral antifungal taken as a single 150 mg dose for simple infections. For recurrent infections, the protocol typically involves three doses taken on days 1, 4, and 7, followed by weekly maintenance doses for six months.
Fluconazole has no known interactions with semaglutide. You can safely take both simultaneously. If you are experiencing recurrent yeast infections on semaglutide, discuss a fluconazole maintenance protocol with your provider rather than repeatedly treating individual episodes.
The probiotic boost during treatment
Taking probiotics alongside antifungal treatment significantly improves outcomes. Clinical data shows that combining probiotics with antifungals nearly doubles the infection-free rate at 90 days compared to antifungals alone.
During active treatment, take oral probiotics containing Lactobacillus strains twice daily. You can also use vaginal probiotic suppositories after completing the antifungal course to repopulate the vaginal microbiome with protective bacteria.
When switching medications might be necessary
In rare cases, recurrent yeast infections may become severe enough to warrant reconsidering your GLP-1 medication choice. Some women experience fewer GI side effects on tirzepatide compared to semaglutide, which may reduce gut microbiome disruption and, consequently, yeast infection risk.
However, switching should be a last resort after all prevention strategies have been implemented and given adequate time to work. The decision involves weighing the benefits of your current semaglutide therapy against the burden of recurrent infections, and this is a conversation best had with your healthcare provider who understands your complete medical picture.
If you are considering switching between GLP-1 medications, understand that the indirect mechanisms causing yeast infections, weight loss, gut changes, dietary shifts, apply to all drugs in this class. A switch might reduce symptom severity but is unlikely to eliminate the risk entirely.
The gut-vagina axis: understanding the deeper connection
The relationship between your digestive system and vaginal health deserves deeper exploration because it explains so many of the symptoms women experience on GLP-1 medications beyond just yeast infections.
How the gut-vagina axis works
The gut microbiome contains trillions of bacteria from hundreds of species. These bacteria do not stay confined to your digestive tract. They migrate, colonize, and communicate with other mucosal surfaces throughout your body, including the vaginal tract.
Lactobacillus species are the key connectors. They dominate both a healthy vaginal microbiome and portions of the gut microbiome. When gut Lactobacillus populations decline, vaginal Lactobacillus populations often follow within weeks. The reverse is also true, vaginal Lactobacillus depletion from antibiotic use or douching often correlates with shifts in gut Lactobacillus.
This axis operates through several pathways. Direct bacterial translocation from the gut to the vaginal tract occurs through the perineal area. Immune signaling molecules produced by gut bacteria influence vaginal immune responses. Metabolites produced by gut bacteria, particularly short-chain fatty acids, enter the bloodstream and affect vaginal tissue health.
How semaglutide specifically affects this axis
Semaglutide alters the gut environment in ways that inevitably affect the gut-vagina axis. Delayed gastric emptying changes the pH and oxygen levels throughout the digestive tract, shifting which bacterial species can thrive. Reduced food intake decreases the volume of prebiotic substrate reaching the lower gut. GI side effects like nausea and constipation change transit time, affecting bacterial distribution.
Research on semaglutide's gut microbiome effects shows it increases Bifidobacterium and Bacteroidota populations while reducing Klebsiella. These are generally positive changes for overall health. But the transitional disruption as one microbial community gives way to another creates a temporary vulnerability in the gut-vagina axis.
Think of it like renovating a house. The end result is better than what you started with. But during construction, things are messy, systems do not work properly, and problems can arise that would not exist in either the old or new version of the house. The gut microbiome remodeling that semaglutide triggers is similar, the destination is healthier, but the journey creates temporary vulnerabilities.
Supporting the axis during treatment
Understanding the gut-vagina axis informs more effective prevention strategies. Rather than treating gut health and vaginal health as separate concerns, address them as connected systems.
Oral probiotics support the gut side of the axis. Vaginal probiotics support the vaginal side. Prebiotic foods feed beneficial bacteria throughout both systems. Adequate hydration maintains the mucosal environment in both the gut and vaginal tract. And avoiding disruptions, unnecessary antibiotics, douching, and harsh soaps, protects both ecosystems simultaneously.
Members of SeekPeptides access detailed protocols for supporting overall health during peptide and GLP-1 therapy, including gut health optimization strategies that address the gut-vagina axis specifically.
Semaglutide and yeast infections in men
While yeast infections are more commonly associated with women, men on semaglutide can develop them as well. Male yeast infections, also called balanitis when affecting the head of the penis, present differently but arise from similar indirect mechanisms.
How men develop yeast infections on semaglutide
Men with type 2 diabetes on semaglutide face elevated risk through blood sugar fluctuations during treatment adjustment. Candida can colonize the penile skin, particularly under the foreskin in uncircumcised men, when glucose levels create a favorable growth environment.
The gut microbiome disruption mechanism applies to men as well. While there is no gut-vagina axis equivalent in men, gut bacteria influence systemic immune function, which affects the body's ability to suppress Candida overgrowth at any location.
Men who take SGLT2 inhibitors alongside semaglutide face the same direct yeast infection risk as women, with clinical rates of 3 to 7 percent. The glucose-rich urine irritates penile tissue and provides a growth medium for Candida.
Symptoms in men
Male yeast infections produce redness, itching, and burning of the penile head (glans). White, cottage-cheese-like discharge may accumulate under the foreskin. The area may appear swollen or develop small red bumps. Some men experience pain during urination or sexual activity.
Treatment for men
Topical antifungal creams, typically clotrimazole or miconazole applied twice daily for 7 to 14 days, resolve most male yeast infections. Oral fluconazole is an alternative for infections that do not respond to topical treatment. Men should also keep the genital area clean and dry, avoid tight underwear, and practice thorough drying after bathing.
The tirzepatide comparison: does the drug class matter?
Many women wonder whether switching from semaglutide to tirzepatide might reduce their yeast infection risk. The answer is nuanced.
Tirzepatide is a dual GIP/GLP-1 receptor agonist. It works through similar mechanisms as semaglutide but with an additional incretin pathway. Like semaglutide, it slows gastric emptying, reduces appetite, and produces significant weight loss.
The indirect mechanisms that connect semaglutide to yeast infections, blood sugar fluctuations, gut microbiome disruption, hormonal shifts from weight loss, dehydration from GI effects, and dietary changes, all apply to tirzepatide as well. In fact, tirzepatide often produces even greater weight loss than semaglutide, which means the hormonal shifts from fat loss may be more pronounced.
However, some women report fewer GI side effects on tirzepatide compared to semaglutide, which could translate to less gut microbiome disruption and potentially fewer yeast infections through that specific pathway. But this varies significantly between individuals. Other women find tirzepatide produces worse GI symptoms.
The same prevention strategies apply regardless of which GLP-1 medication you take. Probiotics, hydration, dietary optimization, proper hygiene, and vaginal estrogen when appropriate all reduce yeast infection risk on both medications. Switching GLP-1 drugs specifically to avoid yeast infections is unlikely to solve the problem without also implementing these foundational strategies.
If you are evaluating whether to switch, the semaglutide to tirzepatide conversion chart can help your provider determine equivalent dosing, and our complete dosage comparison covers the practical details of transitioning between these medications.
Long-term management: preventing recurrent infections
For women who experience more than occasional yeast infections during long-term semaglutide therapy, a systematic approach to prevention becomes necessary.
The maintenance probiotic protocol
Consistent, daily probiotic supplementation is the foundation of long-term prevention. Choose a product with Lactobacillus rhamnosus GR-1 and Lactobacillus reuteri RC-14, the two strains with the strongest evidence for vaginal health.
Take probiotics at the same time daily. Many women find that taking them before bed gives the bacteria the best chance to colonize during the overnight fast, when competition from food-borne bacteria is lowest. Continue probiotics for as long as you remain on semaglutide. Stopping probiotic supplementation while continuing GLP-1 therapy often results in yeast infections returning within weeks.
The quarterly check-in approach
Every three months during semaglutide treatment, take stock of your vaginal health. Are you experiencing any symptoms? Has your discharge changed? Is your vaginal pH normal?
Over-the-counter vaginal pH test strips can serve as a simple screening tool. Normal vaginal pH ranges from 3.8 to 4.5. If your pH is consistently above 4.5, Lactobacillus populations may be insufficient, and you should intensify your probiotic protocol before a yeast infection develops.
Seasonal adjustments
Yeast infection risk is not constant throughout the year. Summer heat and humidity increase risk. Travel, especially to warm climates, disrupts routines and increases risk. Holiday periods often involve more sugar consumption and less consistent probiotic use.
Increase your vigilance and probiotic intake during high-risk periods. Add vaginal probiotic suppositories during travel or summer months. Pack antifungal treatment when traveling with semaglutide so you can treat infections immediately if they develop, rather than waiting to access a pharmacy.
When to consider prophylactic antifungals
For women with truly recurrent infections, defined as four or more episodes per year, prophylactic fluconazole may be appropriate. The standard maintenance protocol involves a single 150 mg fluconazole dose weekly for six months. Studies show this reduces recurrence rates significantly.
This is a prescription decision your provider should make. Discuss it proactively rather than waiting until you have suffered through multiple infections. Prophylactic treatment is more effective and less disruptive than repeated acute treatment courses.
Common myths about semaglutide and yeast infections
Misinformation about this topic is widespread. Let us correct the most common myths.
Myth: Semaglutide directly causes yeast infections
Reality: Semaglutide does not directly cause yeast infections. It does not increase glucose in urine, directly alter vaginal pH, or suppress vaginal immune function. The connection is indirect, through gut microbiome changes, hormonal shifts from weight loss, dehydration, and dietary changes. This distinction matters because it changes how you prevent and treat the problem.
Myth: Yeast infections mean you should stop semaglutide
Reality: Yeast infections are treatable and manageable conditions. They do not indicate that semaglutide is harmful to you. Stopping a medication that is producing meaningful weight loss and metabolic benefits because of a treatable secondary issue is rarely the right decision. Address the yeast infections with the prevention and treatment strategies discussed above while continuing your GLP-1 therapy.
Myth: Eating less sugar on semaglutide prevents yeast infections
Reality: Reducing dietary sugar helps, but it is only one piece of the puzzle. The gut microbiome disruption, hormonal changes, and dehydration pathways are independent of sugar intake. A woman who eats no sugar at all can still develop yeast infections on semaglutide through these other mechanisms. Comprehensive prevention addresses all pathways, not just diet.
Myth: Cranberry supplements prevent yeast infections
Reality: Cranberry supplements have evidence for preventing urinary tract infections, not yeast infections. They are two entirely different types of infection caused by different organisms. Taking cranberry supplements for yeast prevention is ineffective. Direct your investment toward probiotics with documented Lactobacillus strains instead.
Myth: Home remedies like garlic or tea tree oil cure yeast infections
Reality: There is no clinical evidence that garlic cloves, tea tree oil, apple cider vinegar, boric acid, or other home remedies reliably cure yeast infections. Some of these substances can irritate vaginal tissue and make symptoms worse. Stick with evidence-based treatments, OTC antifungals or prescription fluconazole, and save the home remedies for conditions where they actually work.
Myth: All probiotics are equally effective for yeast prevention
Reality: Most commercial probiotics are formulated for general gut health, not vaginal health. The strains that protect against yeast infections, Lactobacillus rhamnosus GR-1, Lactobacillus reuteri RC-14, Lactobacillus crispatus, are not found in most general-purpose probiotic products. Read labels carefully and choose products specifically formulated for vaginal health.
When semaglutide yeast infections signal a bigger problem
Most yeast infections during semaglutide treatment are straightforward and respond to standard treatment. But sometimes recurrent infections indicate an underlying issue that needs investigation.
Undiagnosed or poorly controlled diabetes
If you are taking semaglutide for weight loss and experiencing frequent yeast infections, consider having your blood sugar and HbA1c tested. You may have undiagnosed prediabetes or type 2 diabetes. The elevated blood sugar would explain both the weight issues that led you to semaglutide and the yeast infections you are experiencing on it.
Immune system dysfunction
Recurrent Candida infections can sometimes signal immune system problems. If you are experiencing yeast infections alongside frequent upper respiratory infections, slow wound healing, or persistent fatigue beyond what semaglutide explains, discuss immune function testing with your provider.
Resistant Candida strains
Some Candida species, particularly Candida glabrata and Candida krusei, are naturally resistant to fluconazole and some OTC antifungals. If your infections do not respond to standard treatment, your provider should culture the organism to identify the species and its antifungal sensitivity profile. Non-albicans Candida infections require different treatment approaches.
Medication interactions
If you take multiple medications alongside semaglutide, some of them may contribute to yeast infection risk. Corticosteroids suppress immune function. Hormonal contraceptives alter vaginal pH. Certain immunosuppressants increase fungal infection susceptibility. Review your complete medication list with your provider if yeast infections become recurrent.
The research perspective: what studies actually show
Understanding what scientific literature says about GLP-1 medications and vaginal health helps separate anecdote from evidence.
What clinical trials show
The STEP trials for semaglutide (Wegovy) and the SURMOUNT trials for tirzepatide (Zepbound) did not identify yeast infections as a statistically significant adverse event compared to placebo. This does not mean no participants developed yeast infections. It means the rate in treatment groups was not meaningfully higher than in control groups.
This makes sense given what we know about the mechanisms. The indirect pathways, gut microbiome changes, hormonal shifts, dehydration, take time to manifest. Clinical trial durations and reporting structures may not capture these delayed, indirect effects as effectively as they capture direct pharmacological side effects like nausea or injection site reactions.
What post-market surveillance shows
Post-market reports, collected through the FDA Adverse Event Reporting System (FAERS), do include yeast infections in association with GLP-1 medications. However, these reports cannot establish causation. They show correlation only, a woman took semaglutide and developed a yeast infection, but cannot determine whether the semaglutide caused the infection or whether other factors were responsible.
What microbiome research shows
Studies published in 2024 and into the following years confirm that semaglutide significantly alters gut microbiome composition. The research shows increased Bifidobacterium and Bacteroidota populations with decreased Klebsiella. These changes correlate with improved metabolic health but also represent a significant disruption to established microbial communities.
The gut-vagina axis research establishes that gut microbiome disruption does affect vaginal microbiome health. This biological pathway provides a plausible mechanism for semaglutide-associated yeast infections, even without direct pharmacological causation.
What hormone research shows
Research on weight loss and hormonal changes confirms that rapid fat loss reduces circulating estrogen levels. Studies in postmenopausal women demonstrate that vaginal estrogen levels directly correlate with vaginal Lactobacillus populations, vaginal pH, and resistance to Candida colonization. The chain of evidence from semaglutide to weight loss to reduced estrogen to reduced Lactobacillus to increased Candida risk is well-supported by existing research, even though no single study has traced this complete pathway specifically in semaglutide users.
This is an area where more research is needed. Prospective studies tracking vaginal microbiome changes in women starting semaglutide would provide definitive evidence for or against the indirect mechanisms discussed in this article. Until that research is conducted, the prevention strategies outlined above remain the most evidence-based approach to managing this risk.
For researchers focused on optimizing their peptide research protocols, SeekPeptides provides access to comprehensive research guides, evidence-based protocols, and a community of thousands who navigate these exact questions daily.
Your complete action plan
If you are currently on semaglutide or planning to start, here is your step-by-step plan for minimizing yeast infection risk.
Before starting semaglutide
Begin a vaginal-health-specific probiotic containing Lactobacillus rhamnosus GR-1 and Lactobacillus reuteri RC-14 at least two weeks before your first injection. Stock your kitchen with prebiotic and probiotic foods. Purchase a water bottle and commit to carrying it everywhere. Have OTC antifungal treatment on hand so you can respond immediately if symptoms develop.
If you are in perimenopause or menopause, discuss prophylactic vaginal estrogen with your provider before beginning semaglutide. Establishing vaginal health support before adding the stress of GLP-1 therapy is far more effective than trying to catch up after problems develop.
During the first month of treatment
The first four weeks involve the most dramatic gut microbiome changes as your body adjusts to semaglutide. Take probiotics daily without exception. Drink at least 2 liters of water daily. Eat prebiotic-rich foods even if your appetite is reduced. Monitor for any vaginal symptoms and treat immediately if they appear.
Keep a brief daily log of GI symptoms and vaginal comfort. This helps you identify patterns. Many women notice that yeast infection symptoms correlate with their worst nausea days, supporting the gut-vagina axis mechanism.
During dose escalation
Each dose increase restarts some degree of gut microbiome disruption. When you move from one semaglutide dose level to the next, your GI symptoms may temporarily worsen, and your yeast infection risk may temporarily increase.
Consider adding vaginal probiotic suppositories during the week of each dose increase. Increase water intake by 500 ml during dose adjustment weeks. Be especially diligent about prebiotic food intake during these periods.
During maintenance therapy
Once you are stable on your maintenance dose and GI symptoms have resolved, you can simplify your prevention protocol. Continue daily oral probiotics. Maintain hydration. Include prebiotic and fermented foods regularly. Perform quarterly vaginal health check-ins using pH test strips.
If you have gone six months without a yeast infection on maintenance therapy, your microbiome has likely reached a new equilibrium. Continue basic prevention but know that your risk has decreased significantly from the initial adjustment period.
Frequently asked questions
Can semaglutide cause yeast infections in men?
Yes, though less commonly than in women. Men with type 2 diabetes on semaglutide face elevated risk through blood sugar fluctuations. Men taking SGLT2 inhibitors alongside semaglutide face direct risk from glucose-rich urine. Male yeast infections present as redness, itching, and discharge on the penile head and respond to topical antifungal treatment.
Should I stop semaglutide if I get a yeast infection?
No. Yeast infections are common, treatable conditions that do not indicate semaglutide is unsafe for you. Treat the infection with appropriate antifungals, implement the prevention strategies discussed above, and continue your semaglutide therapy. Only consider discontinuation if infections become severely recurrent despite comprehensive prevention efforts, and only after discussing alternatives with your provider.
Are yeast infections more common at higher semaglutide doses?
Indirectly, yes. Higher doses produce more weight loss, more GI side effects, and potentially greater gut microbiome disruption. All of these increase yeast infection risk through the indirect mechanisms discussed. The higher semaglutide doses also tend to suppress appetite more aggressively, which can lead to reduced nutrient and prebiotic intake.
How long after starting semaglutide do yeast infections typically develop?
Most women who develop yeast infections on semaglutide report them occurring within the first 3 months of treatment, coinciding with the period of greatest gut microbiome disruption and dose escalation. Some experience them specifically during dose increases. Women who implement prevention strategies from the start significantly reduce their risk during this vulnerable period.
Can probiotics interfere with semaglutide effectiveness?
No. Probiotics do not interfere with semaglutide absorption, metabolism, or effectiveness. Semaglutide is injected subcutaneously and enters the bloodstream directly, bypassing the gut entirely. Oral probiotics pass through the digestive tract. The two do not interact pharmacologically. You can and should take probiotics concurrently with semaglutide injections.
Is tirzepatide less likely to cause yeast infections than semaglutide?
There is no clinical evidence that tirzepatide produces fewer yeast infections than semaglutide. Both medications work through similar mechanisms and produce similar indirect effects on gut microbiome, weight loss hormonal changes, and dietary patterns. Some women tolerate tirzepatide with fewer GI side effects, which could theoretically reduce gut-mediated yeast infection risk, but this varies individually. The same prevention strategies apply to both medications.
Do compounded semaglutide formulations with B6 or B12 reduce yeast infection risk?
There is no evidence that compounded semaglutide with B12, pyridoxine, or glycine reduces yeast infection risk specifically. These additives may reduce nausea (pyridoxine) or support gut comfort (glycine), which could indirectly reduce gut microbiome disruption. But the primary mechanisms driving yeast infections, hormonal changes and microbiome shifts, are not addressed by these compounds.
Should I take antifungals preventatively when starting semaglutide?
Routine prophylactic antifungal treatment is not recommended for all women starting semaglutide. However, women with a history of recurrent yeast infections should discuss prophylactic fluconazole with their provider. The risk-benefit calculation favors prevention in women with established susceptibility.
External resources
For researchers serious about optimizing their GLP-1 protocols while managing side effects effectively, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your Lactobacillus stay plentiful, your protocols stay comfortable, and your progress stay uninterrupted.