Mar 25, 2026
Search the internet for tirzepatide side effects and you will find a long list of concerns. Nausea. Fatigue. Digestive upset. But urinary tract infections? That one keeps showing up in forums and comment threads, and it has left thousands of people worried about a connection that may not actually exist. The fear is understandable. Nobody wants to trade weight loss progress for painful, burning trips to the bathroom. And when you are injecting a relatively new medication, every unusual symptom feels like it could be connected.
Here is the reality. A meta-analysis covering 15 randomized controlled trials and more than 14,000 patients found no statistically significant increase in UTI risk among tirzepatide users compared to placebo.
Not at the 5mg dose. Not at 10mg. Not at 15mg. The data is remarkably consistent across every major clinical trial program, and yet the myth persists.
The confusion likely stems from a different class of diabetes medication, one that genuinely does increase UTI risk through a completely different mechanism. People hear "diabetes drug" and assume the risks are interchangeable. They are not. And understanding that distinction matters for anyone making informed decisions about their treatment protocol. SeekPeptides has compiled the most thorough breakdown of this topic available, drawing from clinical trial data, pharmacological mechanisms, and practical prevention strategies that every tirzepatide user should know.
This guide covers everything from the hard clinical numbers to the indirect ways tirzepatide might affect urinary health, along with proven prevention strategies and clear guidance on when to seek medical attention. Whether you are currently taking tirzepatide, considering it, or simply trying to separate fact from fiction, the evidence tells a clear story.
What the clinical research actually says about tirzepatide and UTIs
Clinical evidence is the foundation of any honest answer about medication side effects. Opinions vary. Forum posts contradict each other. But controlled trials with thousands of participants provide the kind of clarity that anecdotal reports simply cannot match. When it comes to tirzepatide and urinary tract infections, the research tells a consistent and reassuring story.
Before examining the data, it helps to understand the scale of evidence available. Tirzepatide has been studied in two massive clinical trial programs, SURPASS for type 2 diabetes and SURMOUNT for weight management, collectively enrolling tens of thousands of participants across multiple countries and treatment durations. This is not a medication where we are guessing about side effects based on small samples. The data is robust, and the conclusions are remarkably uniform. Understanding tirzepatide results across clinical settings provides important context for evaluating safety data.
The meta-analysis that settled the debate
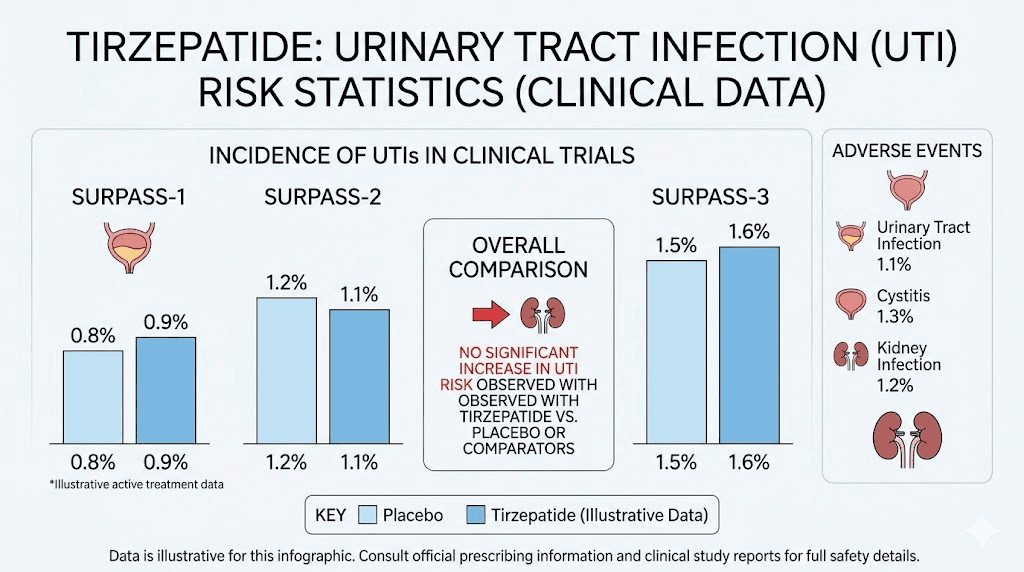
A comprehensive meta-analysis examined 15 randomized controlled trials involving 14,471 patients. This was not a small study or a single trial. It pooled data from every major tirzepatide investigation available, creating the most complete picture of UTI risk that currently exists.
The numbers speak for themselves.
Among tirzepatide users across all doses, 28 patients developed urinary tract infections. In the placebo groups, 20 patients developed UTIs. The relative risk came out to 0.76, with a confidence interval of 0.42 to 1.36. In plain language, tirzepatide users actually had a slightly lower rate of UTIs than people taking a placebo, though this difference was not statistically significant. The medication neither increased nor decreased UTI risk in any meaningful way. This mirrors what researchers have found regarding other tirzepatide and semaglutide side effect comparisons, where urinary complications do not emerge as a pattern.
What makes this finding so reliable is the consistency across doses. Whether patients took the lowest 5mg dose or the highest 15mg dose, the UTI rates remained statistically indistinguishable from placebo. If tirzepatide were truly causing urinary tract infections, you would expect to see a dose-dependent relationship, more medication leading to more infections. That pattern simply does not exist in the data. People tracking their tirzepatide dosing for weight loss can take some reassurance from this dose-independence finding.
SURPASS and SURMOUNT trial findings
The SURPASS clinical trial program enrolled more than 6,000 patients with type 2 diabetes across five major trials. These studies compared tirzepatide against placebo, insulin glargine, insulin degludec, and semaglutide. Across all SURPASS trials, UTIs were not identified as a significant adverse event associated with tirzepatide use. The safety monitoring committees, whose entire job is to flag concerning side effect patterns, did not raise urinary tract infections as a signal worth investigating further.
The SURMOUNT program focused on weight management in people without diabetes. These trials matter because they remove the confounding variable of high blood sugar, which independently increases UTI risk in diabetic populations.
Even in this cleaner population, tirzepatide showed no increase in urinary tract infections. People wondering how long tirzepatide takes to work can focus on efficacy outcomes rather than worrying about UTI complications.
The SURMOUNT findings are particularly important for people using tirzepatide primarily for weight loss rather than blood sugar control. They confirm that the lack of UTI risk is not merely a byproduct of better glucose management. The medication itself does not create conditions favorable to urinary infections, regardless of why you are taking it.
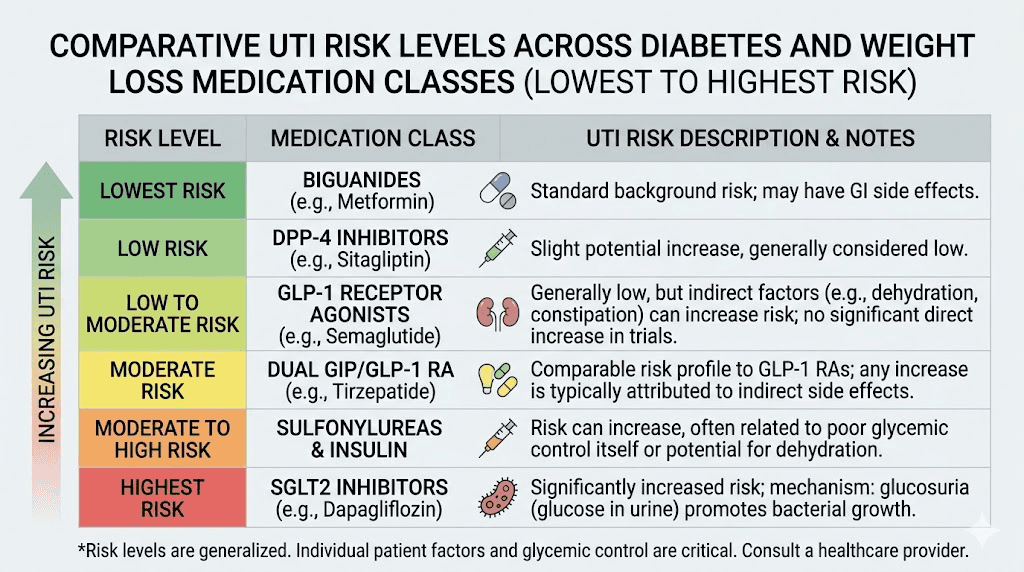
How tirzepatide compares to other diabetes medications
Context matters when evaluating medication safety. Tirzepatide does not exist in a vacuum. It belongs to a treatment landscape that includes multiple drug classes, each with distinct safety profiles. When researchers compared tirzepatide directly against other medications, the UTI picture remained favorable.
Against insulin, tirzepatide showed a relative risk of 0.54 for UTIs, with a confidence interval of 0.07 to 4.19. While not statistically significant due to wide confidence intervals, the trend actually favored tirzepatide. Against other GLP-1 receptor agonists, the relative risk was 1.08, essentially identical, with a confidence interval of 0.12 to 9.72. Neither comparison produced any evidence that tirzepatide increases UTI risk relative to established treatments.
Perhaps most telling is the comparison with GLP-1 receptor agonists as a class. Large observational studies have found that GLP-1 receptor agonists are associated with a lower risk of non-candida urinary tract infections compared to SGLT2 inhibitors, with a hazard ratio of 0.91 or lower and statistical significance below 0.001. Since tirzepatide acts partly through the GLP-1 pathway, this broader class data further supports its favorable UTI profile. People exploring the differences between semaglutide and tirzepatide will find that neither medication carries meaningful UTI risk.
The comparison with SGLT2 inhibitors deserves special attention, and that brings us to the root cause of most tirzepatide UTI confusion.
Why people confuse tirzepatide with UTI-causing medications
Misinformation rarely appears out of thin air. There is usually a grain of truth buried somewhere beneath the misunderstanding, and the tirzepatide UTI myth is a perfect example. The confusion has a specific, traceable origin, and understanding it helps explain why so many people believe something the clinical data does not support.
The SGLT2 inhibitor confusion
SGLT2 inhibitors are a class of diabetes medications that includes empagliflozin, dapagliflozin, and canagliflozin. These drugs work by blocking the reabsorption of glucose in the kidneys, causing excess sugar to spill into the urine. This mechanism is effective for lowering blood sugar, but it creates a significant side effect. Glucose in the urine provides a rich food source for bacteria, particularly the organisms responsible for urinary tract infections.
This is not theoretical. SGLT2 inhibitors carry FDA-recognized warnings about increased UTI and genital infection risk. The mechanism is well-established, thoroughly documented, and essentially unavoidable given how these drugs work. When glucose concentrations rise in the urinary tract, bacterial growth accelerates. The connection is direct and pharmacologically predictable.
The problem arises when people group all diabetes medications together. Someone reads that "diabetes drugs cause UTIs," starts tirzepatide, and assumes the risk applies to their medication too. Online forums amplify this confusion. A person on an SGLT2 inhibitor reports a UTI, someone else on tirzepatide reads that post, and suddenly the association feels real even though the medications work through entirely different mechanisms. People who experience increased thirst on tirzepatide sometimes worry this indicates a urinary problem, when it is typically a normal physiological response to the medication.
How tirzepatide actually works differently
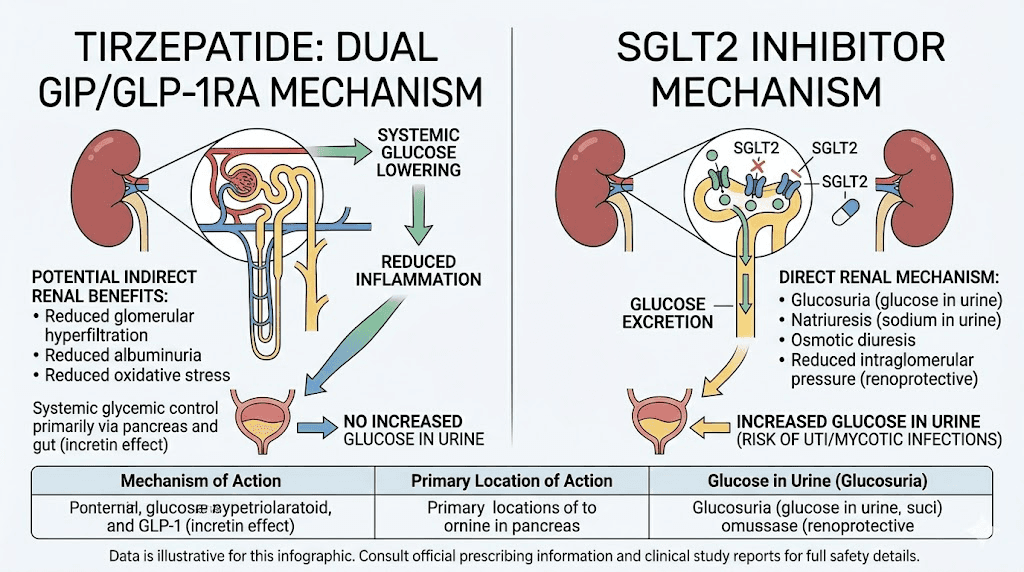
Tirzepatide is a dual GIP/GLP-1 receptor agonist. It mimics two naturally occurring hormones, glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1. These hormones regulate blood sugar by enhancing insulin secretion, suppressing glucagon release, and slowing gastric emptying. None of these mechanisms involve the kidneys filtering extra glucose into the urine.
This distinction is critical. Tirzepatide lowers blood sugar by helping your body use insulin more effectively. SGLT2 inhibitors lower blood sugar by dumping glucose into your urine. One approach creates no urinary risk. The other creates substantial urinary risk. They could not be more different in this regard.
In fact, tirzepatide may indirectly protect against UTIs in people with diabetes. By improving blood sugar control without causing glycosuria, tirzepatide reduces the overall glucose burden in the body, including glucose levels in the urinary tract, without creating the bacterial growth medium that SGLT2 inhibitors produce. Lower systemic glucose means a less hospitable environment for the bacteria that cause urinary infections. Understanding how tirzepatide affects metabolism helps clarify why its mechanism does not create UTI risk.
For people taking both tirzepatide and an SGLT2 inhibitor, which some diabetes treatment plans do include, any UTI risk comes from the SGLT2 inhibitor, not the tirzepatide. If you are on combination therapy and experiencing recurrent UTIs, that conversation with your healthcare provider should focus on the SGLT2 component. The interaction between tirzepatide and other diabetes medications is worth understanding in this context.
How tirzepatide can indirectly affect urinary health
While tirzepatide does not directly cause urinary tract infections, honesty requires acknowledging that the medication can create conditions that might increase susceptibility in some people. These are indirect effects, secondary consequences of the gastrointestinal side effects that tirzepatide is known for. Understanding these pathways helps you prevent problems before they start.
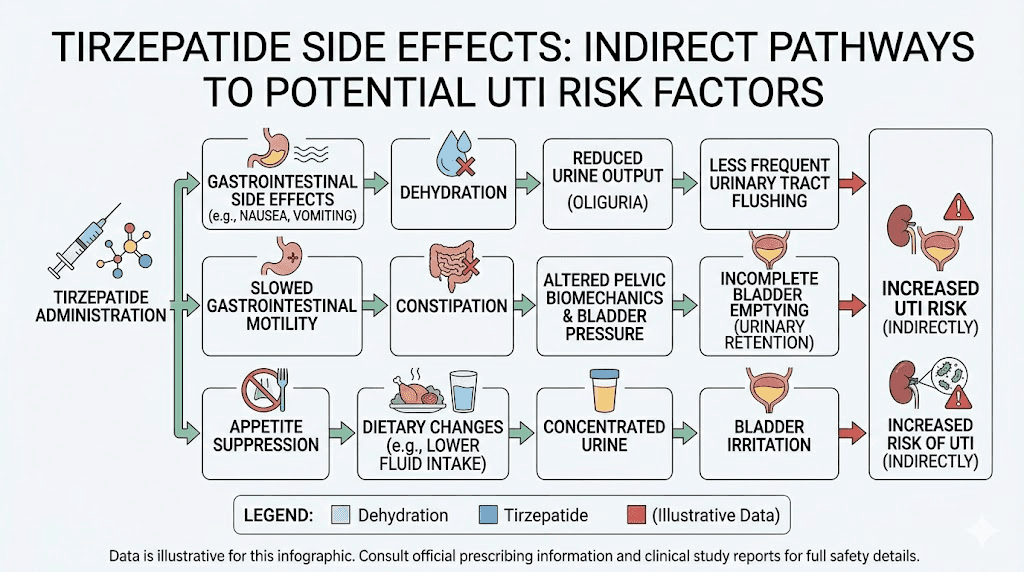
Dehydration from gastrointestinal side effects
Nausea. Vomiting. Diarrhea. These are the most commonly reported side effects of tirzepatide, particularly during the initial weeks and following dose increases. While they typically improve over time, their impact on hydration status can be significant. People dealing with tirzepatide-related diarrhea are at particular risk for fluid loss that affects the entire urinary system.
When you lose fluid through vomiting or diarrhea, your body has less water available for all its functions, including urine production. Concentrated urine means less frequent voiding, and less frequent voiding means bacteria have more time to establish themselves in the urinary tract. The normal flushing mechanism that helps prevent UTIs becomes less effective when urine output drops.
This is not unique to tirzepatide. Any condition or medication that causes fluid loss can increase UTI susceptibility through dehydration. The connection is between hydration status and UTI risk, not between tirzepatide and UTI risk. The distinction matters because the solution is straightforward: maintain adequate hydration, especially during periods of gastrointestinal distress. People who experience bloating on tirzepatide sometimes reduce their fluid intake to manage discomfort, which can compound the dehydration problem.
Some people reduce fluid intake because nausea makes drinking uncomfortable. This is understandable but counterproductive. Sipping small amounts frequently, choosing room temperature or slightly cool water, and adding electrolytes can help maintain hydration even when nausea is present. The dry mouth that sometimes accompanies tirzepatide use can actually serve as a useful reminder to drink more water.
Constipation and bladder function
Constipation is another common tirzepatide side effect, and its connection to urinary health is more direct than many people realize. The rectum and bladder share close anatomical quarters in the pelvis. When the rectum becomes distended with stool, it can press against the bladder, reducing its capacity and preventing complete emptying.
Incomplete bladder emptying is a well-established risk factor for urinary tract infections. Residual urine sitting in the bladder creates a stagnant environment where bacteria can multiply. The body designed the urinary system to flush itself regularly, and anything that interferes with complete voiding disrupts that protective mechanism. Managing tirzepatide-related constipation is not just about digestive comfort. It has real implications for urinary health as well.
The relationship between constipation and UTIs is particularly relevant for women, who already face higher baseline UTI risk due to anatomical factors. A woman dealing with tirzepatide-related constipation and reduced fluid intake from nausea has compounding risk factors that deserve attention. The broader strategies for managing GLP-1 related constipation apply here and can meaningfully reduce secondary UTI risk.
Changes in urination patterns during weight loss
Significant weight loss, which tirzepatide frequently produces, can change urination patterns in several ways. As fat cells shrink, they release stored water, temporarily increasing urine output in many people. This phase can actually be protective against UTIs due to increased flushing. However, as the body adjusts, urination patterns normalize, and people may not maintain the same fluid intake they had during the initial weight loss phase.
Rapid weight loss can also affect pelvic floor function. Excess abdominal weight places chronic pressure on the pelvic floor muscles, and as that weight decreases, the muscles may need time to readjust. For some people, this transition period involves changes in bladder function, including altered urinary frequency or incomplete emptying. The tirzepatide weight loss timeline helps set realistic expectations for how body changes unfold during treatment.
Diet changes compound these effects. People taking tirzepatide typically eat less, and dietary choices while on tirzepatide significantly affect hydration status. High-protein, low-carbohydrate diets, which many tirzepatide users follow, can increase urinary concentration. Following a structured tirzepatide diet plan that accounts for fluid intake helps prevent this issue. Certain foods to avoid while on tirzepatide can also contribute to urinary irritation, making dietary awareness doubly important.
None of these factors mean tirzepatide causes UTIs. They mean that the metabolic and behavioral changes associated with tirzepatide treatment can, in some circumstances, create environments where UTIs are more likely if preventive measures are not taken. The medication itself remains neutral regarding UTI risk.
Recognizing UTI symptoms while on tirzepatide
Whether or not tirzepatide contributes to your UTI risk, knowing how to recognize urinary tract infection symptoms early can prevent a minor inconvenience from becoming a serious medical event. Early detection leads to faster treatment, fewer complications, and less disruption to your tirzepatide protocol. Some UTI symptoms can overlap with normal tirzepatide side effects, making awareness especially important.
Common UTI warning signs
Dysuria, the medical term for painful urination, is the hallmark symptom of a urinary tract infection. It presents as burning, stinging, or sharp discomfort during or immediately after urinating. This symptom is distinct from any known tirzepatide side effect and should never be dismissed as medication-related.
Increased urinary frequency and urgency are classic UTI indicators. You might feel the need to urinate far more often than usual, and when the urge strikes, it feels impossible to wait. The frustrating part is that each trip to the bathroom often produces only a small amount of urine. This frequency-urgency pattern differs from the temporary changes in urination that can accompany weight loss or dietary adjustments. People monitoring their experience after starting tirzepatide should note that painful urination is not an expected medication effect.
Suprapubic pain, discomfort in the lower abdomen just above the pubic bone, often accompanies bladder infections. This can feel like pressure, cramping, or a dull ache. It differs from the gastrointestinal discomfort that tirzepatide sometimes causes, which tends to be more diffuse and centered around the stomach or intestines rather than the lower pelvis.
Changes in urine appearance provide visual clues. Cloudy urine suggests the presence of white blood cells fighting an infection. Foul-smelling urine, particularly a strong ammonia-like or unusually unpleasant odor, indicates bacterial activity. Pink or red-tinged urine, known as hematuria, signals that the infection has caused bleeding somewhere in the urinary tract. Any of these changes warrant prompt medical attention.
When to seek emergency care
Most urinary tract infections are uncomplicated and respond well to outpatient antibiotic treatment. However, certain symptoms indicate the infection has spread beyond the bladder to the kidneys or bloodstream, creating a medical emergency.
Fever above 100.4 degrees Fahrenheit combined with UTI symptoms suggests pyelonephritis, a kidney infection that requires aggressive treatment. Flank pain, particularly on one side, radiating from the lower back toward the groin, is another indicator of kidney involvement. Rigors, the severe shaking chills that accompany high fevers, suggest the infection may be entering the bloodstream.
Nausea and vomiting in the context of UTI symptoms can indicate kidney infection rather than a tirzepatide side effect. This distinction matters. If you are on tirzepatide and experience nausea alongside urinary symptoms, do not assume the nausea is medication-related. The combination of urinary and systemic symptoms demands evaluation. People who are already managing headaches from tirzepatide need to differentiate between known side effects and symptoms that signal a new medical problem like a UTI.
Do not wait.
Kidney infections and urosepsis can escalate quickly. If you have fever, flank pain, rigors, or confusion alongside urinary symptoms, go to an emergency department or urgent care facility immediately. Delayed treatment for complicated UTIs leads to significantly worse outcomes.
UTI symptoms vs normal tirzepatide side effects
The overlap between certain UTI symptoms and tirzepatide side effects creates a diagnostic challenge that deserves careful attention. Knowing what belongs to which category helps you respond appropriately.
Nausea is common with tirzepatide, especially during dose escalation. It is also common with kidney infections. The differentiator is context. Tirzepatide nausea typically correlates with meals, appears within the first few days after injection, and improves as your body adjusts. UTI-related nausea comes with urinary symptoms and often with fever. If nausea appears alongside painful urination, treat it as a potential UTI sign rather than a medication side effect. The strategies for managing tirzepatide side effects should not be applied to symptoms that might indicate infection.
Abdominal discomfort from tirzepatide tends to be epigastric, centered in the upper abdomen around the stomach. UTI-related pain tends to be suprapubic, focused in the lower abdomen above the bladder. Location matters here, and paying attention to exactly where your discomfort centers can help distinguish between the two causes.
Fatigue accompanies both tirzepatide use and urinary tract infections. Tirzepatide-related fatigue typically improves with adequate rest, proper nutrition, and time on the medication. UTI-related fatigue tends to worsen progressively, especially if the infection spreads. If fatigue suddenly worsens and coincides with any urinary symptoms, consider infection as the cause. The same pattern applies to body aches sometimes reported with tirzepatide, which can mimic the systemic symptoms of a more advanced UTI.
When in doubt, get tested. A simple urinalysis and urine culture can definitively diagnose or rule out a UTI within minutes to hours. There is no reason to guess when definitive testing is quick, inexpensive, and widely available.
Preventing UTIs while taking tirzepatide
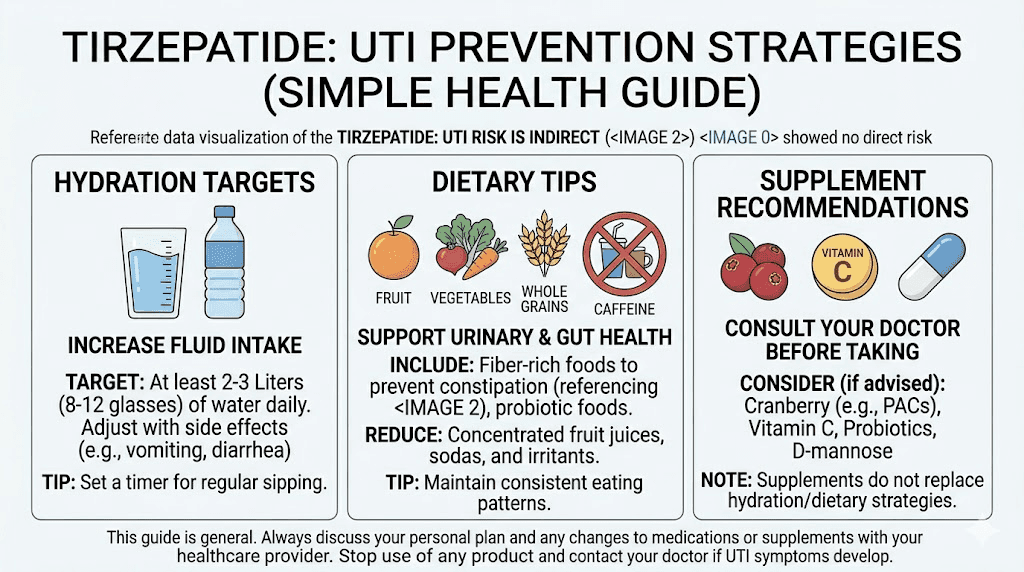
Prevention is always preferable to treatment. While tirzepatide does not directly cause UTIs, the practical realities of being on the medication, reduced appetite, gastrointestinal side effects, and shifting dietary patterns, create opportunities for prevention that are worth taking seriously. The strategies below are evidence-based and relevant for anyone, whether or not they are taking tirzepatide.
Hydration strategies that actually work
The single most effective UTI prevention measure is adequate hydration. Water dilutes urine and promotes regular voiding, both of which flush bacteria from the urinary tract before they can establish an infection. For tirzepatide users, maintaining hydration can be challenging when nausea or reduced appetite suppresses the desire to drink.
Aim for 2 to 3 liters of fluid daily as a baseline. This is a general recommendation, and individual needs vary based on body size, activity level, climate, and the severity of any gastrointestinal side effects. If you are experiencing diarrhea from tirzepatide, your fluid needs increase beyond this baseline to compensate for losses.
Monitor your urine color. Pale yellow to light straw indicates adequate hydration. Dark yellow, amber, or honey-colored urine means you are not drinking enough. This simple visual check takes no effort and provides reliable real-time feedback about your hydration status. People who have noticed increased thirst on tirzepatide should listen to that signal rather than ignoring it.
Timing matters. Drinking large amounts at once can worsen nausea. Instead, sip consistently throughout the day. Keep a water bottle within reach at all times. Set hourly reminders if needed. Room temperature or slightly warm water often sits better on a sensitive stomach than ice cold water. Some tirzepatide users find that adding a small amount of lemon, cucumber, or electrolyte powder makes water more palatable when nausea is present.
Do not count caffeinated beverages as your primary hydration source. While they do contribute to fluid intake, caffeine has mild diuretic properties and can irritate the bladder lining, which is counterproductive for UTI prevention. Water, herbal teas, and electrolyte solutions are better choices. The supplement strategies for tirzepatide users often include electrolyte support that serves double duty for hydration and UTI prevention.
Evidence-based prevention methods
Beyond hydration, several UTI prevention strategies have strong evidence supporting their effectiveness. These apply regardless of medication status, but they carry extra relevance for people navigating the physiological changes that tirzepatide produces.
Urinate when you feel the urge. Holding urine allows bacteria to multiply in the bladder. This sounds simple, but tirzepatide users who are managing their intake carefully sometimes fall into patterns of infrequent voiding, particularly if they are drinking less due to nausea. Make regular bathroom trips a priority rather than an afterthought.
Urinate after sexual intercourse. This is one of the most well-supported UTI prevention measures for women. Intercourse can introduce bacteria into the urethra, and voiding afterward flushes them out before they reach the bladder. This recommendation applies whether or not you are taking any medication. People who have questions about how tirzepatide affects reproductive health should discuss UTI prevention with their healthcare provider as part of that broader conversation.
Wipe front to back after using the bathroom. This prevents the transfer of bacteria from the anal region to the urethra. The anatomy of the female urogenital tract makes this particularly important for women, but it is sound advice for everyone.
Avoid irritating products. Douches, feminine sprays, and scented products in the genital area can disrupt the natural bacterial balance that helps prevent infections. Stick with gentle, unscented cleansers for intimate hygiene. The same principle applies to laundry detergent and fabric softener used on undergarments.
Wear breathable cotton underwear. Synthetic fabrics trap moisture, creating a warm, humid environment that bacteria love. Cotton allows air circulation and moisture evaporation. This is a minor change that can reduce infection risk over time.
Supplements and natural approaches
Several supplements have evidence supporting their role in UTI prevention, and some of them complement tirzepatide use well.
Cranberry products have the strongest natural evidence base. Proanthocyanidins (PACs) in cranberries prevent E. coli, the most common UTI-causing bacterium, from adhering to the bladder wall. Research shows that cranberry products providing at least 36mg of PACs daily reduce UTI risk by approximately 26%. Cranberry capsules or tablets are preferable to juice, which contains high amounts of sugar that can interfere with blood sugar management and conflict with your tirzepatide diet plan.
D-mannose is a natural sugar that works similarly to cranberry PACs by preventing bacterial adhesion in the urinary tract. Some research suggests it may be as effective as low-dose antibiotics for preventing recurrent UTIs in susceptible individuals. A typical dose is 2 grams daily for prevention. It does not significantly affect blood sugar despite being a sugar molecule, making it compatible with tirzepatide therapy.
Probiotics containing specific Lactobacillus strains may help maintain a healthy urogenital microbiome. Lactobacillus rhamnosus and Lactobacillus reuteri have the most evidence for UTI prevention. These can be taken orally and work by promoting a healthy bacterial balance that crowds out pathogenic organisms. Given that tirzepatide can alter gut flora through its effects on digestion and gastric motility, probiotic support makes particular sense during treatment.
Vitamin C may help by acidifying urine, creating a less hospitable environment for bacterial growth. However, high doses can cause gastrointestinal distress, which is the last thing you need when tirzepatide is already challenging your digestive system. If you choose to supplement with vitamin C, start with modest doses of 500mg to 1000mg daily and monitor for any worsening of GI symptoms. The broader supplement guide for tirzepatide users covers additional considerations for combining supplements with this medication.
Managing a UTI without stopping tirzepatide
Developing a UTI while on tirzepatide raises an immediate practical question. Do you need to stop your medication? For most people, the answer provides welcome relief. Understanding how to manage both the infection and your ongoing treatment simultaneously helps prevent unnecessary disruption to your weight loss or diabetes management progress.
Do you need to pause your dose
For uncomplicated lower urinary tract infections, there is generally no medical reason to stop or pause tirzepatide. The medication does not interfere with UTI treatment, does not worsen the infection, and does not interact with standard UTI antibiotics in any clinically significant way. Your prescribing physician should make the final call, but the pharmacological evidence does not support pausing tirzepatide for a straightforward bladder infection.
Complicated UTIs, including kidney infections with systemic symptoms, present a different scenario. Severe infections that cause vomiting may impair your ability to maintain the hydration needed during tirzepatide treatment. If a kidney infection requires hospitalization or intravenous antibiotics, your medical team may recommend temporarily pausing tirzepatide until you are eating and drinking normally again. This decision relates to the practical challenges of severe illness rather than any interaction between tirzepatide and the infection itself.
If you are in the early weeks of starting your tirzepatide dose and already managing gastrointestinal side effects, adding a UTI to the mix can feel overwhelming. In these cases, talk to your prescribing physician about whether to continue your scheduled dose increase or hold at the current dose until the UTI resolves. Staying at a stable dose while treating an infection is often more comfortable than escalating during an illness. The tirzepatide dose chart can help you track where you are in your escalation schedule.
Antibiotic considerations
Standard UTI antibiotics, including nitrofurantoin, trimethoprim-sulfamethoxazole, and fosfomycin, do not have known interactions with tirzepatide. These medications are absorbed and metabolized through pathways that do not overlap with tirzepatide pharmacokinetics.
You can take them concurrently without concern about reduced effectiveness of either medication.
One practical consideration deserves attention. Tirzepatide slows gastric emptying, which means oral medications may be absorbed differently than they would without the drug. In most cases, this does not meaningfully affect antibiotic effectiveness for UTI treatment. However, if your healthcare provider prescribes an antibiotic that requires specific timing relative to meals, follow those instructions carefully. Taking antibiotics on an empty stomach, when recommended, helps ensure adequate absorption even with slowed gastric motility. People who are also taking metformin alongside tirzepatide should mention all their medications when an antibiotic is prescribed so potential interactions can be reviewed comprehensively.
Complete the full course of antibiotics. This advice applies universally, but it bears repeating for tirzepatide users who may already be dealing with nausea and digestive discomfort. Adding antibiotics to an already sensitive stomach can increase GI symptoms temporarily. Taking probiotics two hours apart from your antibiotic doses can help mitigate antibiotic-related digestive distress without reducing antibiotic effectiveness.
Fluoroquinolone antibiotics like ciprofloxacin are sometimes prescribed for complicated UTIs. These medications carry their own side effect profile, including tendon issues and neurological effects. If you are prescribed a fluoroquinolone while on tirzepatide and experience joint pain or unusual anxiety, discuss with your doctor whether the antibiotic rather than the tirzepatide might be responsible.
Recovery timeline expectations
Uncomplicated UTIs typically respond to antibiotics within 24 to 48 hours, with symptom improvement beginning within the first day of treatment. Full resolution usually occurs within 3 to 7 days, depending on the antibiotic used and the severity of the infection. This timeline is the same whether or not you are taking tirzepatide.
During recovery, increase your fluid intake beyond your usual target. Aim for an additional liter beyond your normal 2 to 3 liters daily. This helps flush bacteria and antibiotic metabolites from the urinary tract and supports faster healing. If nausea from tirzepatide makes this challenging, electrolyte solutions and clear broths count toward your fluid goals. Many of the foods to avoid on tirzepatide are also worth avoiding during UTI recovery, particularly caffeine, alcohol, and spicy foods that can irritate the bladder.
If symptoms do not improve within 48 hours of starting antibiotics, contact your healthcare provider. Persistent symptoms may indicate antibiotic resistance, an alternative diagnosis, or a complicated infection that requires different treatment. Do not assume that tirzepatide is interfering with your recovery. In virtually all cases, treatment failure relates to the infection itself, not the concurrent medication.
Tirzepatide vs other weight loss medications and UTI risk
Understanding where tirzepatide stands in the broader landscape of weight loss and diabetes medications helps put UTI risk into proper perspective. Not all medications carry the same risk profile, and the differences are rooted in their mechanisms of action rather than arbitrary variation.
Medication comparison for UTI risk
The following comparison covers the major medication classes that people considering tirzepatide are likely to encounter.
Tirzepatide, as a dual GIP/GLP-1 receptor agonist, shows no increased UTI risk in clinical trials. The mechanism does not involve the kidneys dumping glucose into urine. Gastrointestinal side effects can indirectly affect hydration, but the direct UTI risk is equivalent to placebo. People comparing phentermine and tirzepatide should note that neither medication carries direct UTI risk, though they differ substantially in other side effect profiles.
Semaglutide, a pure GLP-1 receptor agonist, shares the same favorable UTI profile as tirzepatide. Clinical trials of semaglutide for both diabetes and weight loss have not identified increased UTI risk. The evidence regarding semaglutide and UTIs mirrors the tirzepatide data closely. The broader semaglutide UTI analysis reaches the same reassuring conclusions. This makes sense given the shared mechanism. Neither drug causes glycosuria, and neither creates favorable conditions for bacterial growth in the urinary tract.
SGLT2 inhibitors, including empagliflozin, dapagliflozin, and canagliflozin, carry the highest UTI risk of any diabetes medication class. By causing glycosuria as their primary mechanism of action, they create a persistent bacterial growth medium in the urinary tract. This risk is dose-dependent and well-documented in clinical trials and post-marketing surveillance data.
Metformin, the most widely prescribed diabetes medication, does not directly increase UTI risk. However, it can cause gastrointestinal side effects similar to tirzepatide, including diarrhea that may lead to dehydration. People taking metformin and tirzepatide together may experience compounded GI effects that require extra attention to hydration.
Phentermine, a sympathomimetic appetite suppressant, does not carry UTI risk. Its main concerns involve cardiovascular effects, insomnia, and potential for dependence. For people weighing their options, the phentermine vs tirzepatide comparison involves many factors beyond UTI risk.
Newer dual and triple agonists in development, such as mazdutide, are expected to share the favorable UTI profile of the GLP-1 class given their similar mechanisms. Early clinical data supports this expectation, though longer-term surveillance will be needed as these medications reach broader populations.
Why GLP-1 agonists have a better safety profile for urinary health
The fundamental reason GLP-1 receptor agonists, including tirzepatide, do not cause UTIs comes down to how they lower blood sugar. They work by enhancing insulin secretion in a glucose-dependent manner, meaning they help your body produce more insulin when blood sugar is high and back off when blood sugar is normal. This approach keeps glucose in the bloodstream and tissues where it belongs rather than diverting it into the urine.
Large observational studies comparing GLP-1 agonists to SGLT2 inhibitors have consistently shown lower UTI rates in the GLP-1 group. One major study found a hazard ratio of 0.91 or lower for non-candida UTIs in GLP-1 users compared to SGLT2 users, with statistical significance below 0.001. This is not a marginal difference. It represents a meaningful and consistent protective effect.
The protective mechanism likely involves improved glycemic control without glycosuria. Better blood sugar management reduces glucose availability throughout the body, including in the urinary tract. Lower glucose levels in bladder urine mean fewer nutrients for bacteria. Tirzepatide accomplishes this without the urinary glucose spillage that defines SGLT2 inhibitor therapy. Understanding how tirzepatide affects blood work markers helps illustrate its glucose-lowering mechanism and why that mechanism is inherently safer for urinary health.
For people with diabetes who are choosing between medication classes, UTI risk is one factor among many. But it is a legitimate consideration, especially for individuals with a history of recurrent UTIs. In those cases, GLP-1 based therapies like tirzepatide and semaglutide offer clear advantages over SGLT2 inhibitors regarding urinary infection risk.
Special considerations for women on tirzepatide
Women account for the vast majority of UTI cases, with some estimates suggesting that 50 to 60 percent of women will experience at least one UTI during their lifetime. This baseline vulnerability makes UTI prevention especially relevant for the large number of women using tirzepatide for weight management or diabetes control. Several factors specific to women merit additional discussion.
Why women face higher UTI risk
Anatomy drives much of the gender disparity in UTI rates. The female urethra is significantly shorter than the male urethra, approximately 4 centimeters compared to 20 centimeters. This shorter distance means bacteria have a much shorter path to travel from the external environment to the bladder. The proximity of the urethral opening to both the vaginal opening and the anus further increases exposure to potentially pathogenic bacteria.
These anatomical factors exist regardless of medication use. Tirzepatide does not change urethral anatomy or increase bacterial exposure. However, the dehydration risk associated with tirzepatide GI side effects becomes more consequential when baseline UTI susceptibility is already elevated. A man who becomes mildly dehydrated from tirzepatide nausea faces minimal UTI risk. A woman with recurrent UTI history who becomes mildly dehydrated faces meaningfully higher risk. The prevention strategies discussed earlier become proportionally more important.
Sexual activity is a major UTI trigger for women, and it does not interact with tirzepatide in any direct way. However, women who experience changes in libido or sexual patterns during weight loss may alter behaviors that previously protected them. For instance, someone who was less sexually active before starting tirzepatide and becomes more active as they lose weight and gain confidence may encounter UTI risk factors they had not previously needed to manage. Understanding how tirzepatide can affect menstrual and reproductive health provides broader context for these changes.
Hormonal changes during weight loss
Significant weight loss affects hormone levels in ways that can influence urinary health. Fat tissue produces and stores estrogen. As fat cells shrink during tirzepatide-assisted weight loss, estrogen levels may shift, particularly in postmenopausal women whose fat tissue is a primary estrogen source.
Estrogen plays an important role in maintaining the health of the vaginal and urethral tissues. It supports the thickness and elasticity of the vaginal mucosa and helps maintain the acidic pH that keeps pathogenic bacteria in check. Reduced estrogen can thin these tissues, alter vaginal pH, and reduce the population of protective Lactobacillus bacteria, all of which increase UTI susceptibility.
This is not a tirzepatide-specific effect. Any form of significant weight loss can produce similar hormonal shifts. But the magnitude and speed of weight loss that tirzepatide often produces may make these changes more pronounced than with slower weight loss methods. Women who notice vaginal dryness, irritation, or recurrent infections during rapid weight loss should discuss hormonal evaluation with their healthcare provider. The relationship between HRT and GLP-1 therapy is increasingly relevant for women managing both weight and hormonal health.
Vaginal estrogen therapy, which delivers estrogen locally without significant systemic absorption, is one of the most effective treatments for recurrent UTIs in postmenopausal women. It can be used safely alongside tirzepatide and addresses the root cause of hormonal UTI susceptibility.
Pregnancy and breastfeeding considerations
Tirzepatide is not recommended during pregnancy or breastfeeding, and the medication should be discontinued before planned conception. However, the so-called "fertility rebound" effect means that some women become pregnant unexpectedly while using tirzepatide or shortly after stopping. Women who have become pregnant on tirzepatide know this reality firsthand.
UTIs during pregnancy carry elevated risks, including preterm labor and low birth weight. If you are trying to conceive while on tirzepatide, or if you discover you are pregnant during treatment, UTI prevention becomes even more critical. Discuss a transition plan with your healthcare provider that includes both tirzepatide discontinuation and UTI prevention strategies.
Women who are considering tirzepatide while breastfeeding should know that hydration demands increase significantly during lactation. The fluid requirements for milk production, combined with any residual GI effects from recent tirzepatide use, can create dehydration risk that elevates UTI susceptibility. Maintaining fluid intake of 3 liters or more daily during this period is essential for both milk production and urinary health.
The role of blood sugar control in UTI prevention
The relationship between blood sugar and urinary tract infections is well-established in medical literature, and it provides yet another angle from which to understand why tirzepatide does not increase UTI risk. In fact, for people with diabetes or prediabetes, tirzepatide may indirectly reduce UTI risk through its primary therapeutic effect.
Elevated blood sugar impairs immune function in several ways. It reduces the effectiveness of white blood cells, impairs the complement system, and weakens the mucosal barriers that serve as the first line of defense against urinary pathogens. People with poorly controlled diabetes experience UTIs at significantly higher rates than those with normal blood sugar, and the infections tend to be more severe and more likely to involve the kidneys.
Tirzepatide achieves substantial blood sugar reductions. In clinical trials, it lowered HbA1c by 1.5 to 2.5 percentage points depending on dose and baseline levels. This degree of improvement restores immune function, strengthens mucosal defenses, and reduces the glucose available for bacterial metabolism throughout the body, including the urinary tract. The benefits of tirzepatide beyond weight loss include this meaningful improvement in immune-related outcomes.
For people with type 2 diabetes who are switching from an SGLT2 inhibitor to tirzepatide, the UTI benefit can be particularly noticeable. They are simultaneously eliminating the glycosuria that fed urinary bacteria while improving overall glycemic control through a mechanism that does not compromise urinary health. This switch represents a meaningful reduction in infection risk for the right patient population.
People without diabetes who use tirzepatide for weight management also benefit from improved metabolic health. Even without a diabetes diagnosis, many people who qualify for tirzepatide have insulin resistance and blood sugar levels that are higher than optimal. By improving insulin sensitivity and reducing fasting glucose, tirzepatide creates a metabolic environment that is less hospitable to the bacteria responsible for UTIs. Tracking how tirzepatide affects blood work results can confirm these metabolic improvements over time.
What to do if you experience recurrent UTIs on tirzepatide
While tirzepatide does not cause UTIs, some people may coincidentally develop recurrent infections during treatment. Recurrent UTIs, defined as two or more infections in six months or three or more in twelve months, warrant thorough investigation regardless of medication status. SeekPeptides emphasizes the importance of not attributing all health changes to a single medication when multiple factors may be at play.
The first step is a thorough evaluation to identify contributing factors. These might include anatomical abnormalities, incomplete bladder emptying, kidney stones, immunosuppression, or undiagnosed conditions that predispose to infection. A urologist can perform cystoscopy, post-void residual measurements, and other diagnostic tests to identify treatable causes.
Review your hydration habits honestly. Many tirzepatide users drink less than they think they do, particularly when appetite suppression reduces their overall consumption of both food and beverages. Track your actual fluid intake for a week using a measuring container or tracking app. The results often reveal a significant gap between perceived and actual hydration levels.
Evaluate whether tirzepatide side effects are contributing to dehydration. If persistent diarrhea or ongoing vomiting is making it impossible to maintain adequate hydration despite your best efforts, discuss dose adjustment with your prescribing physician. Sometimes splitting the tirzepatide dose or microdosing tirzepatide can reduce GI side effects while maintaining therapeutic benefit. The microdosing tirzepatide chart provides guidance on alternative dosing approaches.
Consider whether constipation is contributing to incomplete bladder emptying. If you are struggling with constipation on tirzepatide, addressing it may simultaneously improve your urinary health. Adequate fiber, magnesium supplementation, and proper hydration address both issues at once.
For women with recurrent UTIs, prophylactic strategies may include post-coital antibiotics, low-dose daily antibiotics, or vaginal estrogen therapy. These are established medical interventions with strong evidence behind them, and they work independently of and alongside tirzepatide without any conflict. Discuss these options with your healthcare provider if prevention strategies alone are not sufficient.
Practical hydration protocol for tirzepatide users
Because hydration is the single most controllable factor in UTI prevention for tirzepatide users, a structured approach to fluid intake deserves its own dedicated discussion. This protocol accounts for the specific challenges that tirzepatide creates, including reduced appetite, nausea, and altered GI function.
Upon waking, drink 500ml of room temperature water before doing anything else. Your body dehydrates overnight, and this morning bolus restores baseline hydration and initiates the flushing mechanism that clears any bacteria that may have colonized the urethra during sleep. Add a pinch of salt and a squeeze of lemon if plain water triggers nausea.
Between breakfast and lunch, sip an additional 500 to 750ml. Do not try to drink this all at once. Small, frequent sips are better tolerated when bloating or nausea is present. Herbal teas, particularly ginger tea, serve double duty by contributing to hydration while helping manage nausea symptoms.
During the afternoon, target another 500 to 750ml. If you are exercising, add an additional 500ml for every hour of moderate activity. Electrolyte solutions are preferable to plain water during and after exercise, as they replace sodium, potassium, and magnesium lost through sweat while supporting cellular hydration.
In the evening, consume 500ml before dinner. Taper fluid intake as bedtime approaches to avoid disrupting sleep with frequent trips to the bathroom. However, do not cut off fluids entirely. A small glass of water before bed and another if you wake during the night keeps the urinary tract from becoming excessively concentrated overnight.
On days when you administer your tirzepatide injection, pay particular attention to hydration. Many users report that side effects peak 24 to 48 hours after injection, and this is when dehydration risk is highest. Pre-loading fluids on injection day and maintaining aggressive hydration for the following two days can prevent the dehydration-related UTI risk that comes from GI side effects. Knowing the best time to take your tirzepatide shot allows you to plan your hydration strategy around your injection schedule. Similarly, understanding proper injection technique and injection site selection can minimize side effects that contribute to dehydration.
Track your fluid intake for at least the first month of tirzepatide treatment. Use a marked water bottle, a tracking app, or a simple tally system. What gets measured gets managed, and most people discover they are drinking significantly less than they estimated before tracking. This simple habit protects against both dehydration and the UTI risk that follows from it.
Understanding tirzepatide side effects in proper context
The UTI question sits within a broader pattern of concern about tirzepatide side effects. Some of these concerns are well-founded, based on clinical data and pharmacological mechanisms. Others arise from misunderstanding, anecdotal reports, or confusion with different medications. Placing the UTI question in the context of what tirzepatide actually does and does not cause helps build an accurate picture of the medication risk profile.
The genuinely common side effects of tirzepatide are predominantly gastrointestinal. Nausea affects a substantial percentage of users, particularly during dose escalation. Diarrhea, constipation, bloating, and reduced appetite are all well-documented in clinical trials and confirmed through post-marketing surveillance. These are real effects that deserve real management strategies. Fatigue and headaches also appear with enough frequency to warrant attention.
Less common but documented effects include injection site reactions, which can range from mild redness to more significant itching and inflammation. Gallbladder events have been reported at higher rates in tirzepatide users, likely related to rapid weight loss rather than a direct drug effect. Feeling cold during weight loss is common and relates to reduced insulating fat and metabolic adjustments.
Some reported effects remain uncertain in their relationship to tirzepatide. Sleep disturbances, anxiety, muscle pain, and joint pain appear in user reports but are not consistently identified in controlled trials as occurring above placebo rates. These may represent individual susceptibility, coincidental timing, or the general effects of significant physiological change during rapid weight loss.
UTIs fall into the category of effects that are not associated with tirzepatide in clinical data. Like bone loss concerns and erectile dysfunction questions, the UTI association represents a worry that the evidence does not substantiate. This does not mean the concern is silly or invalid. It means that the specific mechanism of tirzepatide does not create the conditions required for this particular side effect to occur.
Keeping a symptom journal during tirzepatide treatment helps distinguish between medication effects, coincidental illness, and indirect consequences of the metabolic changes the drug produces. Record symptoms along with their timing, severity, and relationship to doses, meals, and hydration status. This information is invaluable for your healthcare provider and helps prevent both over-attribution and under-attribution of symptoms to the medication.
Tirzepatide delivery methods and UTI considerations
Tirzepatide is available in several formulations, and the delivery method does not change the UTI risk profile. However, different formulations may have varying side effect intensities that indirectly affect hydration and, by extension, UTI susceptibility.
Standard subcutaneous injection remains the most common delivery method. Proper injection technique ensures consistent medication absorption and predictable side effect timing. The thigh injection site and abdominal sites produce reliable absorption, and understanding how to manage injection site reactions keeps the focus on systemic effects rather than local complications.
Oral tirzepatide is an emerging option that eliminates the injection requirement entirely. Orally disintegrating tablets and liquid drop formulations may have different gastrointestinal side effect profiles compared to injectable versions. Some users report fewer GI issues with oral formulations, which could indirectly benefit hydration status and UTI prevention. However, clinical data comparing UTI rates between formulations is not yet available, and the fundamental pharmacological mechanism remains the same regardless of how the medication enters the body.
Compounded tirzepatide offers another option, with the compounded dosage chart allowing for more flexible dose adjustments. The ability to fine-tune dosing may help users find the minimum effective dose that produces the fewest GI side effects, thereby reducing dehydration risk. Reconstituting tirzepatide properly ensures accurate dosing. Use the peptide calculator and reconstitution calculator to verify your measurements. The syringe dosage guide provides additional measurement accuracy support.
Regardless of formulation, the UTI risk assessment remains the same. The medication does not cause glycosuria. It does not alter urinary tract physiology. It does not impair local immune function in the urogenital tract. The only UTI-relevant consideration across all delivery methods is the potential for GI side effects to affect hydration status.
Combining tirzepatide with other treatments and UTI implications
Many tirzepatide users take additional supplements or medications as part of their overall health protocol. Some of these combinations have implications for hydration, urinary health, or both. Understanding these interactions helps maintain comprehensive UTI prevention.
Vitamin B12 supplementation is common among tirzepatide users, as the medication can affect B12 absorption over time. B12 does not affect UTI risk in any direction. Glycine supplementation and niacinamide addition are also popular combinations that do not interact with urinary health.
Berberine combined with tirzepatide deserves consideration because berberine itself has antimicrobial properties and may offer mild protective effects against urinary pathogens. However, berberine can also cause GI side effects, potentially compounding dehydration risk when taken alongside tirzepatide. Balance the potential benefits against the practical reality of managing multiple sources of gastrointestinal disturbance.
Creatine supplementation alongside tirzepatide requires increased water intake to prevent kidney strain and support creatine transport into muscles. This additional hydration demand actually benefits UTI prevention as a side effect, provided you actually follow through with the increased fluid intake that creatine requires.
NAD+ supplementation with tirzepatide and testosterone therapy alongside tirzepatide are combinations that some users pursue for broader metabolic benefits. Neither directly affects UTI risk, but both require monitoring that should include kidney function tests, which can incidentally identify early urinary tract issues.
Combining phentermine with tirzepatide can increase the risk of dehydration through additive appetite suppression. When you are eating and drinking even less than with either medication alone, UTI prevention through conscious hydration becomes even more important.
Alcohol consumption while on tirzepatide compounds dehydration risk significantly. Alcohol is a diuretic that increases fluid loss, and when combined with the reduced fluid intake that tirzepatide often produces, the resulting dehydration can meaningfully increase UTI susceptibility. Limiting alcohol intake and matching every alcoholic drink with an equal volume of water helps mitigate this risk.
Long-term tirzepatide use and urinary health outlook
As tirzepatide moves from a relatively new medication to an established long-term treatment option, understanding its effects on urinary health over extended periods becomes increasingly relevant. The current clinical trial data covers treatment periods of up to two years, and the reassuring UTI safety profile has remained consistent throughout.
Long-term GI side effects tend to decrease as the body adapts to the medication. Most users report that nausea, diarrhea, and other gastrointestinal symptoms peak during the first few months of treatment and dose escalation, then gradually diminish. This means the indirect UTI risk factors related to dehydration from GI symptoms also decrease over time. The longer you are on tirzepatide, the less likely GI-mediated dehydration becomes a significant concern.
Sustained weight loss produces lasting metabolic improvements that favor urinary health. Improved insulin sensitivity, lower fasting glucose, reduced inflammation, and better immune function all contribute to a urogenital environment that is less hospitable to pathogenic bacteria. For people with diabetes, the sustained HbA1c improvements that tirzepatide produces over years of use represent ongoing protection against the UTI risk that poorly controlled blood sugar creates.
If you are considering weaning off tirzepatide after achieving your weight loss goals, be aware that weight regain can reverse the metabolic improvements that protected against UTIs. Maintaining weight loss after tirzepatide through diet, exercise, and potentially a lower GLP-1 maintenance dose helps preserve these protective effects. The withdrawal considerations for GLP-1 medications are worth understanding in this broader health context.
People who experience a plateau on tirzepatide or find themselves not losing weight despite treatment sometimes consider switching from tirzepatide to semaglutide or vice versa. From a UTI perspective, this switch is neutral. Both medications share the same favorable urinary safety profile, and neither introduces new UTI risk factors that the other does not carry.
Regular medical monitoring during long-term tirzepatide use should include periodic urinalysis as part of comprehensive metabolic panels. This catches any urinary issues early, whether related to the medication, weight loss, dietary changes, or unconnected medical conditions. Proactive monitoring is always better than reactive treatment.
Frequently asked questions
Does tirzepatide directly cause urinary tract infections?
No. A meta-analysis of 15 randomized controlled trials involving 14,471 patients found no statistically significant increase in UTI risk among tirzepatide users compared to placebo at any dose (5mg, 10mg, or 15mg). The relative risk was 0.76, actually trending slightly lower than placebo, though not statistically significant. The side effect profiles of both major GLP-1 medications confirm this finding.
Why do some people report UTIs while taking tirzepatide?
UTIs are common in the general population, especially among women, and coincidental timing with tirzepatide use can create a false association. Additionally, tirzepatide side effects like diarrhea and nausea can cause dehydration, which indirectly increases UTI susceptibility. The UTI is caused by dehydration and reduced fluid intake, not by tirzepatide itself.
Should I stop tirzepatide if I get a UTI?
For uncomplicated bladder infections, there is generally no reason to stop tirzepatide. Standard UTI antibiotics do not interact with tirzepatide in clinically significant ways. For complicated infections with fever or kidney involvement, discuss with your healthcare provider whether a temporary pause is appropriate. Review the dose chart if you need to adjust your schedule.
Is tirzepatide safer than SGLT2 inhibitors for UTI risk?
Yes. SGLT2 inhibitors (empagliflozin, dapagliflozin, canagliflozin) cause glycosuria, meaning they dump glucose into the urine, which provides a growth medium for bacteria. Tirzepatide does not cause glycosuria. Clinical data consistently shows lower UTI rates with GLP-1 based therapies compared to SGLT2 inhibitors.
How much water should I drink on tirzepatide to prevent UTIs?
Aim for 2 to 3 liters of fluid daily as a baseline, with additional intake during periods of GI side effects, exercise, or hot weather. Monitor urine color as a real-time hydration gauge. Pale yellow indicates adequate hydration. The supplement strategies for tirzepatide section covers electrolyte options that support hydration.
Do cranberry supplements help prevent UTIs while on tirzepatide?
Research supports cranberry products containing at least 36mg of proanthocyanidins (PACs) daily for UTI prevention, reducing risk by approximately 26%. Cranberry capsules are preferable to juice when on tirzepatide, as they avoid the sugar content that can interfere with blood sugar management and dietary goals.
Can dehydration from tirzepatide side effects cause recurrent UTIs?
Chronic dehydration from ongoing GI side effects can contribute to recurrent UTIs by reducing urinary flushing and concentrating urine. If you are unable to maintain adequate hydration despite your best efforts, discuss dose adjustment strategies with your healthcare provider. Addressing the dehydration source is more effective than treating recurrent infections individually.
Does semaglutide have the same UTI safety profile as tirzepatide?
Yes. Semaglutide and UTI risk has been studied extensively, and the findings mirror tirzepatide data. Neither GLP-1 receptor agonist increases UTI risk compared to placebo. The comprehensive semaglutide UTI analysis reaches the same conclusions. Both medications share the mechanism of improving blood sugar without glycosuria.
External resources
FDA Drug Safety and Availability - Official safety communications and labeling updates for tirzepatide
CDC Urinary Tract Infection Information - Evidence-based UTI prevention and treatment guidance
American Urological Association Guidelines - Clinical guidelines for recurrent UTI management
ClinicalTrials.gov - Complete database of SURPASS and SURMOUNT tirzepatide trial results
National Institute of Diabetes and Digestive and Kidney Diseases - Comprehensive UTI resource from NIH
For people serious about understanding how their medications affect every aspect of their health, SeekPeptides offers the most comprehensive resource available for evidence-based peptide and medication guidance. Members access detailed safety profiles, interaction databases, and a community of thousands who have navigated these exact questions. Whether you are optimizing your tirzepatide protocol, managing side effects, or simply trying to separate fact from fiction, SeekPeptides provides the tools and support to make informed decisions with confidence.
In case I do not see you, good afternoon, good evening, and good night. May your urinary health stay clear, your hydration stay consistent, and your tirzepatide protocol stay on track.