Mar 24, 2026

What if the burning sensation you noticed last week has nothing to do with semaglutide itself, and everything to do with how your body is responding to treatment? That question matters more than most people realize. Urinary tract infections affect millions of people every year, and when you start a new medication, every new symptom feels connected. Every ache becomes suspicious. Every bathroom visit gets scrutinized.
But here is the reality. The relationship between semaglutide and urinary tract infections is not what most online sources suggest. Clinical trial data from the SUSTAIN and STEP programs, which enrolled thousands of participants across multiple countries, did not identify UTIs as a significant adverse event. The FDA prescribing information for Ozempic, Wegovy, and Rybelsus does not list urinary tract infections among common side effects. Yet people keep asking the question. They keep searching. And they deserve a complete, honest answer that goes beyond a simple yes or no.
This guide breaks down every mechanism, every risk factor, and every prevention strategy that connects semaglutide treatment to urinary health. Because while semaglutide may not directly cause UTIs, the changes it creates in your body can shift the balance in ways that matter.
Understanding those shifts, and knowing exactly what to do about them, is the difference between a smooth treatment experience and months of unnecessary discomfort. SeekPeptides has helped thousands of researchers navigate exactly these kinds of questions, and the answers are more nuanced than most sources admit.
What the clinical evidence actually shows
Start with the data. That is always the right move.
The SUSTAIN clinical trial program evaluated semaglutide across multiple phase 3 trials involving over 8,000 participants with type 2 diabetes. The STEP program added thousands more participants focused specifically on weight management. Across both programs, researchers tracked adverse events meticulously, documenting everything from nausea to injection site reactions. UTIs did not emerge as a statistically significant side effect in any of these trials.
This matters enormously. When a medication goes through phase 3 clinical trials with that many participants, genuine side effects tend to surface. Semaglutide showed clear associations with gastrointestinal symptoms like nausea, vomiting, diarrhea, and constipation. Those connections were unmistakable in the data. But UTIs? The numbers simply did not support a direct causal link.
A network meta-analysis examining 105 randomized controlled trials with over 219,000 participants found no significant association between GLP-1 receptor agonists and increased infection risk, including urinary tract infections. In fact, one recent propensity-matched analysis of non-diabetic patients found that GLP-1 use was associated with a lower incidence of UTI, 8.8% compared to 13.3% in the control group.
That finding deserves emphasis. Some evidence suggests GLP-1 therapy might actually reduce UTI risk rather than increase it. The anti-inflammatory properties of GLP-1 receptor agonists could play a protective role, though researchers are still investigating the precise mechanisms. Understanding how semaglutide works in the body helps explain why this protective effect might exist.
So why do some people experience UTIs during treatment?
Because correlation is not causation. People starting semaglutide are often managing multiple health conditions. They may have diabetes, which independently increases UTI risk. They may be overweight, which can affect pelvic floor function and bladder emptying. They may be taking other medications that influence urinary health. Attributing every new symptom to the most recent medication change is human nature, but it is not always accurate.
The indirect pathways: how semaglutide might contribute to UTI risk
Direct causation is off the table. The data is clear on that. But indirect mechanisms? Those deserve serious attention. Because while semaglutide does not cause UTIs the way SGLT-2 inhibitors do, it creates several physiological changes that can shift urinary health in subtle but meaningful ways.
The dehydration connection
This is the biggest indirect risk factor. Full stop.
Semaglutide gastrointestinal side effects are well documented. Nausea affects up to 44% of users at higher doses. Vomiting occurs in approximately 24% of participants. Diarrhea hits around 30%. These symptoms are most intense during dose escalation, particularly in the first 4-8 weeks of treatment, and they all share one critical consequence.
They pull water from your body.
Every episode of vomiting depletes fluids. Every bout of diarrhea strips electrolytes and water from the intestinal tract. Even persistent nausea, which does not directly cause fluid loss, leads to reduced water intake because the last thing a nauseated person wants to do is drink. The result is a body that is consistently running behind on hydration, sometimes without the person even realizing it.
Dehydration concentrates urine. Concentrated urine is more acidic, more irritating to the bladder lining, and creates a more favorable environment for bacterial colonization. The urinary tract relies on regular flushing to sweep bacteria away before they can establish an infection. When urine volume drops and frequency decreases, bacteria get more time to adhere to the urethral and bladder walls, multiply, and trigger inflammation.
Researchers studying semaglutide dosing protocols have noted that most gastrointestinal side effects peak during the initial dose escalation period, which is exactly when dehydration risk is highest. If you are timing your semaglutide doses without accounting for hydration needs, you may be creating conditions that favor UTI development without realizing the connection.
Constipation and bladder pressure
Constipation is one of the most common semaglutide side effects, affecting roughly 12-20% of users depending on the dose. And its connection to UTIs is more direct than most people realize.
A full, distended colon sits right next to the bladder. When stool accumulates and the colon expands, it physically presses against the bladder wall. This pressure does two things. First, it reduces the bladder effective capacity, meaning you feel the urge to urinate more frequently but with smaller volumes. Second, and more importantly, it can prevent complete bladder emptying.
Incomplete bladder emptying is one of the strongest risk factors for recurrent UTIs. Residual urine acts as a standing pool where bacteria can thrive. Each time you urinate without fully emptying, you leave behind a reservoir of warm, nutrient-rich fluid that bacteria love. Over days and weeks of chronic constipation, this creates a perfect breeding ground for infection.
The constipation treatment strategies that work for semaglutide users go beyond simple fiber supplements. They address the underlying mechanism, semaglutide slowing gastric motility, which in turn slows colonic transit time. Managing this effectively requires understanding the complete gastrointestinal picture, not just treating the symptom in isolation.
Additionally, chronic constipation can lead to inflammation of the colon, which sits in close proximity to the urinary tract. This inflammation can create a localized immune response that affects neighboring tissues, potentially making the bladder and urethra more vulnerable to bacterial invasion. Research from infectious disease specialists has documented cases where chronic constipation during GLP-1 therapy preceded the onset of recurrent UTI patterns.
Gut microbiome shifts
This is the mechanism that gets the least attention but may matter the most over time.
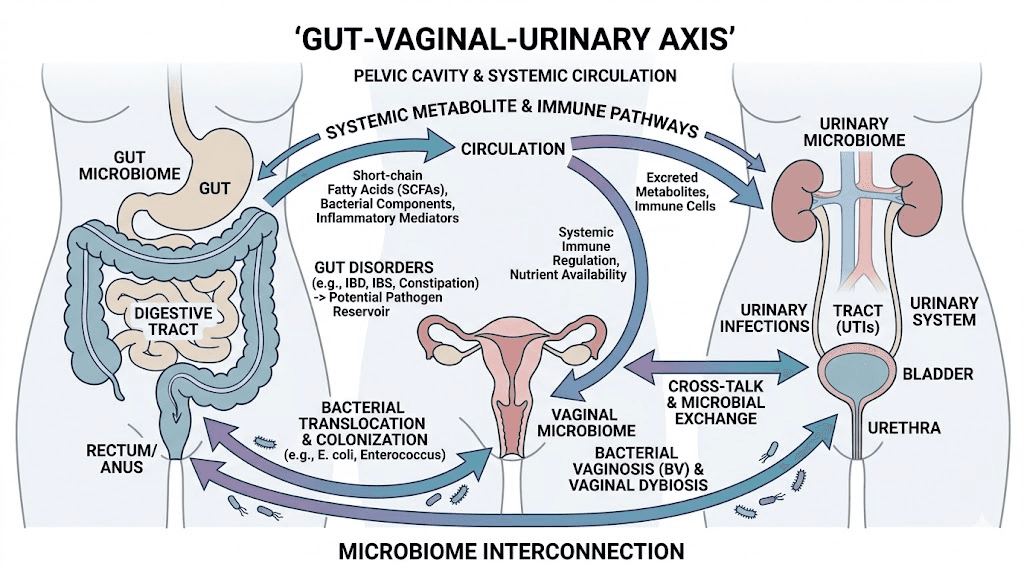
Semaglutide fundamentally alters the gastrointestinal environment. By slowing gastric emptying and changing motility patterns throughout the digestive tract, it shifts the conditions that determine which bacterial populations thrive. Emerging research suggests these changes can extend beyond the gut itself, affecting the vaginal and urinary microbiomes through what scientists call the gut-vaginal-urinary axis.
The connection works like this. Beneficial bacteria in the gut, particularly Lactobacillus species, play a protective role throughout the body. They help maintain acidic pH in the vaginal tract, which prevents colonization by pathogenic bacteria like E. coli, the organism responsible for approximately 80-90% of all UTIs. When gut microbiome composition shifts, it can reduce the protective bacterial populations that serve as the first line of defense against urinary pathogens.
Understanding how semaglutide interacts with other compounds becomes important in this context. Some formulations include additional ingredients like methylcobalamin or B12, which may influence the microbiome differently than semaglutide alone. Probiotic supplementation, particularly with strains like those found in GLP-1 specific probiotic formulations, is gaining attention as a strategy to maintain microbiome balance during treatment.
Reduced food and fluid intake
Semaglutide works, in part, by suppressing appetite. That is the whole point. But appetite suppression comes with an underappreciated side effect that relates directly to urinary health.
When people eat less, they often drink less too. Many people get a significant portion of their daily fluid intake from food, particularly fruits, vegetables, soups, and other water-rich items. A person following a semaglutide diet plan who reduces overall food volume by 30-40% may unknowingly cut their fluid intake by a similar percentage. Add in the nausea-driven avoidance of drinking, and you have a recipe for chronic mild dehydration that compounds over weeks.
This is why the foods you choose while on semaglutide matter for more than just nutrition. Water-rich foods serve double duty, providing nutrients while maintaining hydration levels that protect urinary health. People who focus exclusively on protein-dense, low-volume foods may optimize their macronutrients while inadvertently compromising their hydration status.
Semaglutide vs SGLT-2 inhibitors: an important distinction
This section exists because confusion between these two medication classes is rampant online, and the confusion creates unnecessary anxiety for semaglutide users.
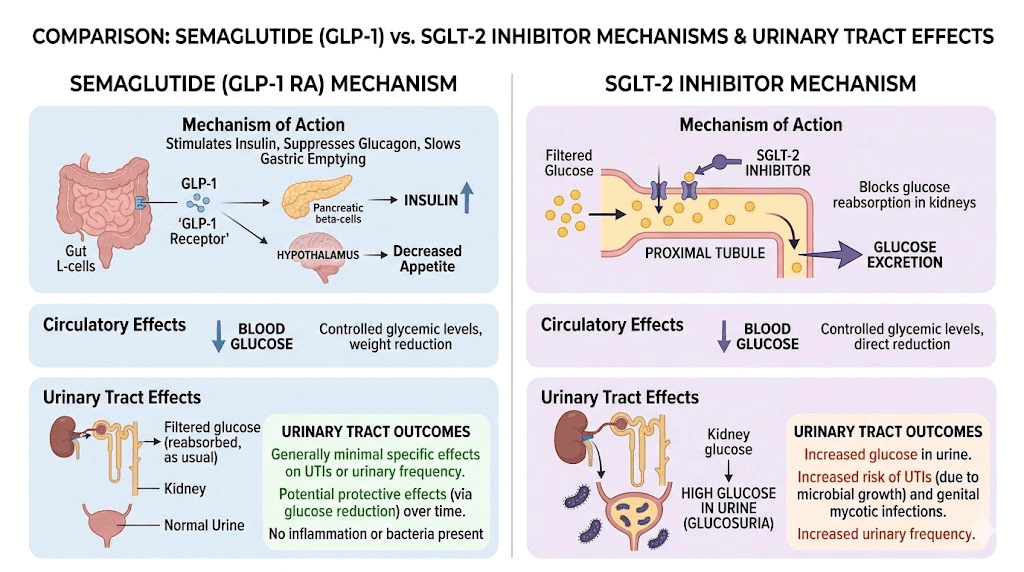
SGLT-2 inhibitors, medications like empagliflozin, dapagliflozin, and canagliflozin, work by blocking glucose reabsorption in the kidneys. This means excess glucose gets excreted in the urine, which is how these medications lower blood sugar. But glucose-rich urine creates a literal feast for bacteria. The warm, sugar-laden environment in the urinary tract becomes an ideal growth medium for organisms like E. coli and Candida species.
This mechanism is why SGLT-2 inhibitors carry FDA warnings about increased UTI and genital infection risk. The connection is direct, well-documented, and mechanistically clear. Meta-analyses of SGLT-2 inhibitor trials consistently show elevated rates of both urinary tract infections and genital mycotic infections.
Semaglutide does not do any of this.
As a GLP-1 receptor agonist, semaglutide works through completely different pathways. It does not alter urinary glucose levels. It does not change the composition of urine in ways that promote bacterial growth. It does not affect kidney glucose handling at all. The mechanism of action is fundamentally different, targeting incretin receptors in the pancreas, brain, and gut rather than the kidneys.
When you search online and find articles linking semaglutide to UTIs, check whether the source is conflating GLP-1 agonists with SGLT-2 inhibitors. Many general health websites lump all diabetes medications together, creating the false impression that UTI risk applies equally across all treatment classes. It does not. The distinction matters enormously for anyone trying to make informed decisions about their long-term semaglutide treatment.
If you are taking both semaglutide and an SGLT-2 inhibitor, which some diabetes management protocols include, the UTI risk from the SGLT-2 component deserves attention. But attributing that risk to semaglutide would be inaccurate. Understanding which medication carries which risk profile allows you to take targeted prevention measures rather than worrying about risks that do not actually apply to your semaglutide protocol.
Risk factors that increase UTI susceptibility during treatment
Not everyone on semaglutide faces the same UTI risk. Certain factors amplify vulnerability, and knowing which ones apply to you allows for targeted prevention rather than general worry.
Biological factors
Women experience UTIs at dramatically higher rates than men, primarily due to anatomy. The female urethra is shorter, roughly 4 centimeters compared to approximately 20 centimeters in men, giving bacteria a much shorter path to the bladder. This baseline anatomical vulnerability means that any additional risk factors, including dehydration from semaglutide side effects, compound an already elevated baseline risk.
Age matters too. Postmenopausal women face particularly high UTI risk because declining estrogen levels thin the vaginal and urethral tissues, reduce protective Lactobacillus populations, and alter urinary pH. If you are a postmenopausal woman starting semaglutide, your baseline UTI risk is already elevated, and the dehydration and constipation effects of treatment deserve extra attention.
People with diabetes carry additional risk regardless of their semaglutide use. Elevated blood glucose levels impair immune function, particularly the ability of white blood cells to identify and destroy bacteria. Diabetic neuropathy can reduce bladder sensation, leading to incomplete emptying and urinary retention. These factors exist independently of semaglutide and would be present whether or not GLP-1 therapy was part of the treatment plan. Tracking your semaglutide results should include monitoring for any urinary symptoms alongside weight and glucose metrics.
Medication interactions
Several common medications can increase UTI susceptibility, and people on semaglutide often take one or more of them concurrently.
Diuretics increase urine output, which should theoretically reduce UTI risk by flushing bacteria. However, if fluid intake does not keep pace with increased urinary output, the net effect can be dehydration, the exact opposite of what urinary health requires. People taking diuretics alongside semaglutide face a compounded dehydration risk that demands vigilant hydration monitoring.
Immunosuppressive medications, including corticosteroids, reduce the immune system ability to fight bacterial invasion. Combined with the metabolic changes from semaglutide, immunosuppression can create a window of vulnerability that targeted prevention strategies need to address.
Some people combine semaglutide with other weight management medications. Understanding how phentermine and semaglutide interact or how these medications compare is important not just for efficacy but for managing combined side effect profiles that might affect urinary health.
Lifestyle factors
Sedentary behavior, which can increase during the initial weeks of semaglutide treatment when nausea and fatigue are most prominent, independently raises UTI risk. Physical inactivity reduces pelvic blood flow, weakens pelvic floor muscles, and can contribute to urinary stasis. Even mild activity like walking promotes better bladder function and reduces UTI susceptibility.
Tight, non-breathable clothing creates a warm, moist environment around the urogenital area that bacteria thrive in. While this has nothing to do with semaglutide directly, people who are actively losing weight sometimes continue wearing tight-fitting garments that no longer fit properly, inadvertently creating conditions that promote bacterial growth.
Sexual activity remains one of the strongest risk factors for UTIs in women, and this risk does not change during semaglutide treatment. Post-coital urination and proper hygiene practices remain essential prevention strategies regardless of medication status.
A complete prevention protocol for semaglutide users
Prevention is always easier than treatment. Here is a systematic approach to protecting urinary health during semaglutide therapy, organized by priority and ease of implementation.
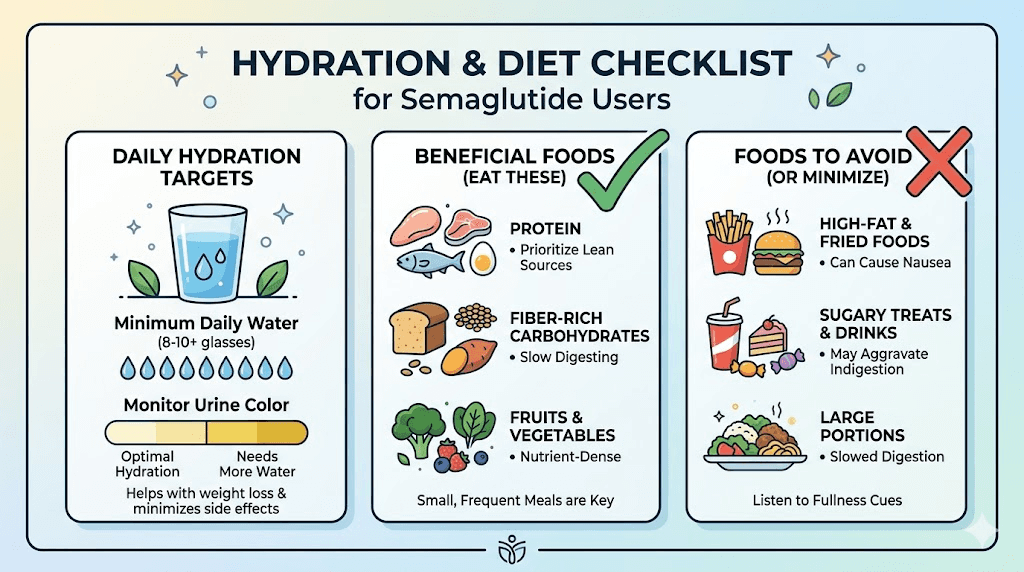
Hydration protocol
This is the single most impactful prevention strategy. Nothing else comes close.
Baseline target: 2.5 to 3.5 liters of water daily. This is higher than the standard recommendation of 2 liters because semaglutide users face additional fluid losses from gastrointestinal side effects. During active dose escalation, when nausea and vomiting are most common, aim for the higher end of this range.
Timing strategy: Do not try to drink large volumes at once, especially if you are experiencing nausea. Instead, take small sips consistently throughout the day. Keep a water bottle within arm reach at all times. Set hourly reminders if needed. Some people find that room-temperature water is easier to tolerate than cold water during periods of nausea.
Hydration indicators: Urine color is your most reliable real-time hydration monitor. Pale yellow to nearly clear urine indicates adequate hydration. Dark yellow or amber-colored urine signals dehydration and increased UTI risk. If your urine is consistently dark, you are not drinking enough, regardless of what the volume targets say.
Electrolyte balance: Plain water is not always sufficient, especially if you are experiencing vomiting or diarrhea. Electrolyte drinks or supplements help maintain the mineral balance that keeps your kidneys functioning optimally. Avoid sugary sports drinks, which can feed bacteria. Look for low-sugar or sugar-free electrolyte options.
For people using compounded semaglutide, the hydration protocol remains the same as for brand-name formulations. The active ingredient affects the body identically regardless of the source, and the dehydration risk does not change based on whether you are using pharmacy-sourced or compounding pharmacy formulations.
Dietary strategies
What you eat while on semaglutide affects urinary health more than most people realize.
Water-rich foods: Cucumbers, watermelon, strawberries, lettuce, celery, oranges, and tomatoes all contribute significantly to daily fluid intake. Including these in your semaglutide meal planning serves double duty, providing nutrition while supporting hydration and urinary flushing.
Cranberry products: The evidence on cranberry for UTI prevention is mixed but trending positive. Proanthocyanidins in cranberries can prevent E. coli from adhering to the bladder wall, which is the critical first step in infection establishment. Unsweetened cranberry juice or cranberry supplements (at least 36mg proanthocyanidins daily) offer the most consistent evidence of benefit. Avoid cranberry cocktails loaded with sugar.
Probiotic-rich foods: Yogurt, kefir, sauerkraut, kimchi, and other fermented foods support the microbiome populations that protect against UTIs. This is particularly important given the potential microbiome shifts that semaglutide can cause. Some researchers specifically recommend probiotics formulated for GLP-1 users to address the unique microbiome changes associated with this medication class.
Foods to limit: Caffeine and alcohol both have diuretic effects that can worsen dehydration. Understanding alcohol interactions with semaglutide matters for multiple reasons, including urinary health. Spicy foods and artificial sweeteners can irritate the bladder lining, making it more susceptible to bacterial invasion. During dose escalation, when your body is most vulnerable, minimizing these bladder irritants provides an additional layer of protection.
Protein considerations: Adequate protein intake is essential during semaglutide treatment to preserve lean mass, but very high protein diets can make urine more acidic and concentrated. Finding the right balance means consuming enough protein to support your body composition goals, typically 1.2 to 1.6 grams per kilogram of body weight, while maintaining adequate hydration to dilute the metabolic byproducts of protein metabolism. Using tools like those available through SeekPeptides can help you calculate appropriate intake levels for your specific situation.
Bathroom habits that matter
Simple habits make a measurable difference in UTI prevention. These apply to everyone, but they become especially important when semaglutide side effects create additional vulnerability.
Never delay urination. When you feel the urge, go. Holding urine gives bacteria more time to multiply and adhere to the bladder wall. Semaglutide users who are managing nausea may unconsciously avoid getting up to use the bathroom. This seemingly minor behavior change can have meaningful consequences for urinary health over time.
Ensure complete emptying. Take your time. Do not rush. After you think you are finished, wait 10-15 seconds and try again. The double-void technique, urinating, waiting briefly, then urinating again, is one of the most effective strategies for reducing residual urine volume, particularly for people experiencing constipation-related bladder pressure.
Post-sexual activity urination. Urinate within 15-30 minutes after sexual intercourse. This flushes bacteria that may have been introduced to the urethral opening during activity. This recommendation applies regardless of medication status, but adherence becomes more important when other risk factors are present.
Wiping direction. Front to back, always. This prevents fecal bacteria from contaminating the urethral opening. This is especially relevant during periods of diarrhea, a common semaglutide side effect, when bacterial load in the perianal area is elevated.
Managing constipation proactively
Because constipation is both a common semaglutide side effect and a significant UTI risk factor, addressing it proactively is a two-for-one prevention strategy.
Fiber intake: Aim for 25-35 grams of fiber daily from a mix of soluble and insoluble sources. Increase gradually to avoid gas and bloating, which are already common during semaglutide dose escalation. Psyllium husk, ground flaxseed, and chia seeds provide gentle, effective fiber supplementation. Check our detailed semaglutide constipation treatment guide for specific protocols.
Magnesium supplementation: Magnesium citrate or magnesium oxide at 200-400mg daily can improve colonic motility without harsh stimulant effects. Many semaglutide users find this provides consistent, gentle relief that keeps the colon moving and reduces bladder compression.
Regular movement: Even 20-30 minutes of walking daily stimulates colonic contractions and promotes regular bowel movements. Exercise also benefits pelvic floor function and blood flow, providing indirect protection against UTIs. If semaglutide fatigue is limiting your activity, even gentle movement is better than none.
Recognizing UTI symptoms while on semaglutide
Early detection leads to faster treatment and fewer complications. But recognizing UTI symptoms can be tricky when you are already experiencing semaglutide side effects that create overlapping sensations.
Classic UTI symptoms
The textbook presentation includes a burning sensation during urination, increased frequency and urgency, cloudy or strong-smelling urine, pelvic pain or pressure, and sometimes blood in the urine. These symptoms develop over hours to days and typically worsen without treatment.
But here is where it gets complicated for semaglutide users.
Symptom overlap with semaglutide side effects
Semaglutide can cause dizziness and general malaise, which can mask the subtle early signs of UTI. Abdominal discomfort from slowed gastric emptying can overlap with pelvic pressure from a bladder infection. Gastrointestinal symptoms like burping and nausea can dominate attention, causing people to overlook urinary symptoms that would normally prompt immediate action.
Changes in urination frequency can also be confusing. Semaglutide users who are drinking more water to prevent dehydration will naturally urinate more frequently. Distinguishing between appropriate frequency from increased fluid intake and pathological frequency from bladder inflammation requires paying attention to accompanying symptoms, particularly burning, urgency, and changes in urine appearance or smell.
When symptoms demand attention
Three symptoms should always prompt immediate medical evaluation, regardless of whether you think they might be related to semaglutide.
Blood in the urine. This is never normal and always requires evaluation. While UTIs commonly cause microscopic or visible blood in urine, other conditions need to be ruled out.
Fever with urinary symptoms. A UTI that causes fever has likely progressed beyond the bladder to involve the kidneys. Kidney infections, pyelonephritis, are serious and can require hospitalization, intravenous antibiotics, and temporary discontinuation of semaglutide to manage dehydration risk.
Flank pain. Pain in the lower back or sides, particularly on one side, combined with urinary symptoms suggests kidney involvement. This requires urgent medical attention and should not be managed with over-the-counter remedies or watchful waiting.
Understanding what to expect during treatment helps you distinguish between normal adjustment symptoms and genuine warning signs. Our guide on the first weeks on semaglutide provides context for what typical side effects look like versus symptoms that require additional investigation.
Managing a UTI during semaglutide treatment
If you develop a UTI while taking semaglutide, the treatment approach involves managing the infection while accounting for the unique considerations that GLP-1 therapy creates.
Antibiotic treatment considerations
Standard UTI antibiotics, including nitrofurantoin, trimethoprim-sulfamethoxazole, and fosfomycin, remain effective regardless of semaglutide use. There are no significant drug interactions between semaglutide and common UTI antibiotics. However, some antibiotics can cause gastrointestinal side effects of their own, which can compound the nausea and stomach discomfort from semaglutide.
Tell your healthcare provider that you are taking semaglutide. This information helps them choose antibiotics that are less likely to worsen your existing gastrointestinal symptoms. Nitrofurantoin, for example, should be taken with food but may cause nausea in some people. A provider who knows you are already managing semaglutide-related nausea might choose a different first-line option.
Hydration during infection
Increase your fluid intake beyond your normal semaglutide hydration protocol during an active UTI. The goal is to flush bacteria from the urinary tract as aggressively as possible. Aim for at least 3 liters daily, more if you can tolerate it. Yes, this means more frequent bathroom trips. That is exactly the point.
If nausea from semaglutide makes drinking difficult, consider small, frequent sips of clear broths, herbal teas, and electrolyte solutions rather than trying to drink large glasses of water. Popsicles made from diluted juice or electrolyte drinks can also provide fluid in a more tolerable format. Whatever it takes to keep fluids moving through your system.
Should you pause semaglutide during a UTI?
For uncomplicated lower urinary tract infections, there is generally no medical reason to pause semaglutide. The infection is localized to the bladder, antibiotics will resolve it, and semaglutide does not interfere with the treatment process.
For complicated infections involving the kidneys, fever, or severe dehydration, the calculus changes. Severe vomiting from either the infection or semaglutide can worsen dehydration to dangerous levels. In these situations, your healthcare provider may recommend temporarily pausing semaglutide until the infection is fully resolved and you can maintain adequate hydration. Understanding what happens when you pause semaglutide helps set realistic expectations for the temporary interruption.
The key principle is that managing the acute infection takes priority. Semaglutide can always be restarted. A kidney infection that progresses due to inadequate hydration creates much bigger problems than a brief treatment interruption.
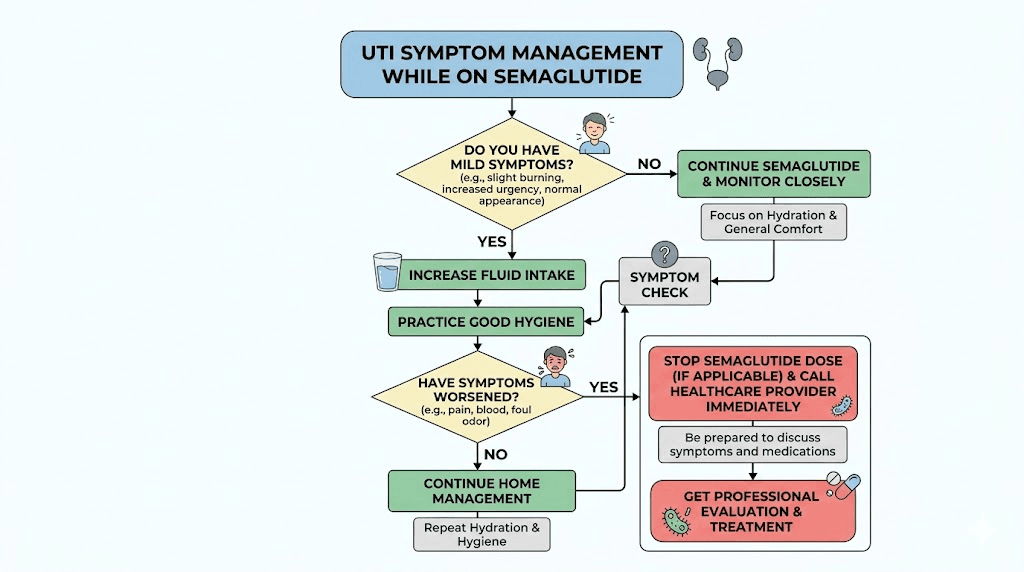
Other urinary concerns with semaglutide
UTIs are not the only urinary issue that semaglutide users report. Several other urinary symptoms have been associated with GLP-1 therapy, and understanding the full picture helps you identify what is happening and respond appropriately.
Frequent urination
Increased urinary frequency is commonly reported during semaglutide treatment, but the mechanism is usually benign. Most often, it simply reflects increased water intake, which is exactly what you should be doing. If you are following the hydration protocol outlined above and drinking 2.5-3.5 liters daily, you will urinate more frequently than before starting treatment. That is normal and healthy.
However, if frequent urination is accompanied by burning, urgency, or changes in urine appearance, it may indicate infection rather than simple fluid balance. The distinction matters because one requires treatment while the other just requires patience and perhaps slightly adjusted fluid timing.
For people using semaglutide for diabetes management, frequent urination could also indicate inadequate blood glucose control. Elevated blood sugar causes osmotic diuresis, where excess glucose pulls water into the urine. If your blood sugar is not well controlled, frequent urination may reflect glycemic issues rather than hydration effects. Checking blood glucose when urinary frequency increases unexpectedly can help identify the true cause.
Urinary incontinence
Some semaglutide users report stress incontinence, involuntary urine leakage during physical activities like coughing, sneezing, laughing, or exercising. The connection is not fully understood, but several potential mechanisms exist.
Rapid weight loss can change abdominal pressure dynamics. The pelvic floor muscles that support the urinary sphincter may need time to adapt to a changing body composition. As fat tissue decreases, the support structures around the bladder and urethra adjust, and this transition period can temporarily weaken continence control.
Pelvic floor exercises, commonly known as Kegel exercises, can significantly improve stress incontinence during the weight loss phase. Strengthening these muscles provides better urethral support and improves the closure mechanism that prevents leakage. Consistency matters more than intensity, aim for three sets of 10-15 contractions daily.
If incontinence persists or worsens, a pelvic floor physical therapist can provide targeted assessment and treatment. This is not a symptom to simply endure. Effective interventions exist, and addressing incontinence early prevents the compensatory behaviors, like limiting fluid intake, that can actually increase UTI risk.
Kidney considerations
Semaglutide has been associated with rare cases of acute kidney injury, primarily in the context of severe dehydration from gastrointestinal side effects. While this is uncommon, it underscores the critical importance of maintaining hydration during treatment.
The kidneys filter approximately 150 liters of blood daily and produce 1-2 liters of urine. When dehydration reduces blood volume, kidney filtration decreases, urine becomes concentrated, and the risk of both kidney injury and urinary infection increases simultaneously. The prevention strategy for both conditions is identical: adequate hydration.
If you are taking semaglutide and notice a significant decrease in urine output, dark-colored urine, or swelling in the legs and feet, contact your healthcare provider promptly. These signs can indicate kidney stress that needs immediate evaluation. Regular monitoring of kidney function through simple blood tests is advisable for anyone on long-term semaglutide therapy, particularly those with pre-existing kidney conditions or diabetes.
Monitoring your semaglutide storage and quality also matters for overall safety. Properly stored medication at the correct temperature maintains its efficacy and safety profile. Using properly refrigerated semaglutide ensures you are getting the intended therapeutic effect without unexpected complications.
Special populations and elevated risk
Certain groups need to be particularly vigilant about urinary health during semaglutide treatment.
Women over 50
Postmenopausal women face the highest UTI risk of any demographic group, and adding semaglutide side effects to this baseline vulnerability requires proactive management. Estrogen decline thins the vaginal and urethral tissues, reduces protective bacterial populations, and alters urinary pH. These changes create an environment where bacteria can establish infections more easily.
Topical vaginal estrogen, available by prescription, can significantly reduce UTI recurrence in postmenopausal women by restoring tissue thickness and supporting protective bacteria. If you are a postmenopausal woman starting semaglutide and have any history of UTIs, discuss vaginal estrogen with your healthcare provider before UTIs become a recurring problem.
Understanding how semaglutide affects hormonal health provides additional context for women navigating both weight management and the hormonal shifts that come with aging. These interactions deserve attention from both you and your healthcare provider.
People with diabetes
Diabetes independently increases UTI risk through multiple mechanisms. Elevated glucose levels impair white blood cell function, reduce bacterial killing capacity, and in some cases create glucose-rich environments that promote bacterial growth. Diabetic neuropathy can reduce bladder sensation, leading to incomplete emptying and urinary retention.
For diabetic patients using semaglutide, the drug itself may actually help reduce UTI risk over time by improving blood glucose control. Better glycemic management restores immune function and reduces the glucose-mediated bacterial growth that drives infection risk. This is one area where the long-term benefits of semaglutide may actually outweigh the short-term indirect risks from side effects.
Regular monitoring of both blood glucose and urinary symptoms provides the most comprehensive picture. Many diabetic patients are already accustomed to self-monitoring, and adding a brief urinary health check to this routine requires minimal additional effort.
People with recurrent UTI history
If you have a history of three or more UTIs in the past year, or two or more in the past six months, you are classified as having recurrent UTIs. Starting semaglutide in this context requires a more aggressive prevention strategy from day one.
Consider discussing prophylactic measures with your healthcare provider before beginning semaglutide. Options include low-dose prophylactic antibiotics, D-mannose supplementation (which can prevent E. coli adhesion to the bladder wall), and more intensive probiotic therapy targeting urogenital health. Some providers recommend methenamine hippurate as a non-antibiotic prophylactic option for patients who want to avoid long-term antibiotic use.
Keeping a symptom diary during the initial months of semaglutide treatment helps identify patterns and triggers specific to your situation. Note your fluid intake, bowel habits, urinary symptoms, and semaglutide side effects daily. This data gives your healthcare provider concrete information to work with if UTIs do occur.
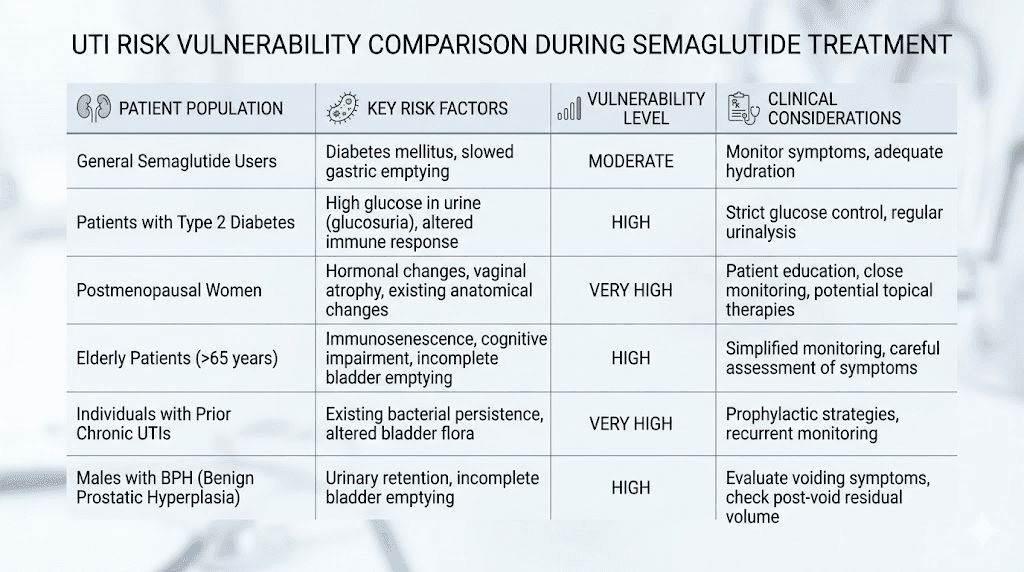
The emerging research: what scientists are studying now
The relationship between GLP-1 therapy and urinary health is an active area of investigation, and several research threads deserve attention.
Anti-inflammatory properties of GLP-1 agonists
GLP-1 receptors exist throughout the body, not just in the pancreas and brain. Emerging research has identified GLP-1 receptor expression in kidney tissue and the urinary tract, suggesting that semaglutide may have direct effects on urinary tract biology that we are only beginning to understand.
Preclinical studies suggest that GLP-1 receptor activation can reduce inflammatory cytokine production and modulate immune responses in ways that could be protective against infection. If these findings translate to clinical practice, semaglutide might eventually be recognized as having a net protective effect on urinary health, not a harmful one.
Microbiome research
The gut-urinary axis is a rapidly expanding field of study. Researchers are mapping how changes in gut bacterial composition, including those caused by GLP-1 agonists, ripple outward to affect urinary tract colonization patterns. This research may eventually lead to specific probiotic recommendations tailored to GLP-1 users, optimizing both gut and urinary health simultaneously.
Weight loss and UTI risk reduction
Obesity is an independent risk factor for UTIs. Excess abdominal weight increases intra-abdominal pressure, which can impair bladder emptying and promote urinary stasis. By facilitating significant weight loss, semaglutide may actually reduce long-term UTI risk even as short-term side effects create temporary vulnerability.
This framing matters. The short-term risks from dehydration and constipation are manageable with the prevention strategies outlined in this guide. The long-term benefits of weight loss on urinary health are substantial and lasting. The calculus overwhelmingly favors continuing treatment with appropriate prevention measures rather than avoiding semaglutide out of UTI concerns.
For researchers tracking their progress, comparing semaglutide before and after results should include improvements in urinary health alongside weight and metabolic markers. Many people find that as they lose weight and their gastrointestinal side effects stabilize, urinary concerns resolve completely.
Practical troubleshooting: common scenarios and solutions
Real situations require real solutions. Here are the scenarios semaglutide users encounter most frequently regarding urinary health, with specific guidance for each.
Scenario 1: UTI during dose escalation
You have just increased your semaglutide dose, nausea has intensified, and now you are noticing urinary burning. This is the most common timing for UTI development during semaglutide treatment.
Action plan: See your healthcare provider for urine culture and appropriate antibiotic treatment. Increase fluid intake to maximum tolerable levels. Consider requesting a slower dose escalation schedule to reduce gastrointestinal side effects and associated dehydration. Many practitioners will adjust the semaglutide dosing schedule based on side effect tolerance.
Scenario 2: Recurrent UTIs after starting semaglutide
You have had two or more UTIs since beginning semaglutide, and you rarely had them before.
Action plan: Document the timeline carefully, including dose changes, side effect severity, and hydration patterns. Bring this information to your healthcare provider. Request urine culture with sensitivity testing rather than empiric antibiotic treatment to ensure you are getting the right antibiotic. Implement the full prevention protocol described above. Consider additional prophylactic measures like D-mannose or cranberry supplements. Your provider may also evaluate for other contributing factors that coincided with semaglutide initiation.
Scenario 3: Urinary symptoms without confirmed infection
You have urinary burning, frequency, or urgency, but urine culture comes back negative for bacteria.
Action plan: This is more common than people realize, and it often relates to concentrated urine irritating the bladder rather than actual infection. Increase hydration aggressively. Eliminate bladder irritants, including caffeine, alcohol, citrus, and spicy foods, for two weeks to see if symptoms resolve. If symptoms persist, your provider may evaluate for interstitial cystitis or other non-infectious urinary conditions.
Scenario 4: You are afraid to drink water because of nausea
Nausea from semaglutide makes every sip of water feel like a battle, and you know you are not drinking enough.
Action plan: Try alternative fluid sources. Herbal tea, particularly ginger or peppermint, can settle nausea while providing fluids. Watermelon, cucumber slices, and frozen fruit provide water in a more tolerable form. Ice chips are often easier to manage than liquid water. Electrolyte popsicles offer fluid, minerals, and a cooling sensation that can reduce nausea. Small sips every 5-10 minutes add up faster than you think. If nausea is severe enough to prevent any fluid intake, contact your healthcare provider, this level of intolerance may require antiemetic medication or a dose adjustment. Resources like our guide on semaglutide and energy levels provide additional strategies for managing side effects that affect daily functioning.
Comparing UTI risk across GLP-1 medications
Not all GLP-1 medications are identical, and understanding how UTI-related risk factors compare can inform treatment decisions.
Semaglutide, tirzepatide, liraglutide, and dulaglutide all share the GLP-1 receptor agonist mechanism, but they differ in potency, side effect profiles, and dosing schedules. Tirzepatide, which targets both GLP-1 and GIP receptors, tends to produce similar gastrointestinal side effects but may have different impacts on gut motility patterns. Our comparison of semaglutide versus tirzepatide side effects covers these differences in detail.
For people considering a switch between medications, understanding how semaglutide to tirzepatide conversion works helps plan for the transition period when side effects may temporarily intensify. Switching between medications can reset the gastrointestinal adjustment process, creating a new window of elevated dehydration risk that needs proactive management.
Triple agonists like retatrutide are emerging as next-generation options with potentially different side effect profiles. Early data suggests that while retatrutide produces more dramatic weight loss, the gastrointestinal side effect patterns may differ from those seen with semaglutide alone. Understanding the retatrutide dosing protocols and their side effect implications is important for anyone considering future treatment options.
Oral formulations of GLP-1 medications, including oral semaglutide drops and sublingual semaglutide, may produce different gastrointestinal side effect patterns compared to injectable formulations. Oral delivery changes the absorption kinetics and can affect how GI side effects manifest. For people who experience severe nausea with injectable semaglutide leading to dehydration and UTI risk, oral formulations might offer a way to continue GLP-1 therapy with a more tolerable side effect profile.
Long-term urinary health on GLP-1 therapy
Most discussions about semaglutide and UTIs focus on the short-term risks. But what about people who take semaglutide for months or years? The long-term picture is actually more reassuring than the short-term one.
Gastrointestinal side effects typically improve significantly within the first 8-16 weeks of treatment. As your body adjusts to semaglutide, nausea decreases, vomiting becomes rare, and bowel habits normalize. This means the primary indirect risk factors for UTIs, dehydration and constipation, diminish over time even as treatment continues.
Simultaneously, the benefits of weight loss accumulate. Reduced abdominal pressure improves bladder function. Better glycemic control restores immune competence. Improved mobility increases pelvic blood flow and muscle tone. These changes create a net positive effect on urinary health that grows stronger the longer treatment continues.
For people committed to long-term semaglutide use, the message is clear. The initial weeks require extra vigilance around hydration and urinary health. But the trajectory is toward improvement, not worsening. The prevention strategies in this guide are most critical during the dose escalation phase and become less burdensome as treatment stabilizes.
SeekPeptides members access comprehensive guides that cover long-term management strategies, including protocols for maintaining urinary health throughout extended GLP-1 therapy. Understanding these long-term dynamics helps you plan proactively rather than reacting to problems as they arise.
The bottom line on semaglutide and urinary tract infections
Semaglutide does not cause UTIs. The clinical evidence is clear on this point, and no amount of anecdotal reports changes the trial data. But semaglutide does create conditions, primarily through dehydration from gastrointestinal side effects and constipation-related bladder pressure, that can indirectly increase UTI vulnerability in susceptible individuals.
The good news is that these indirect risks are entirely manageable. Adequate hydration, constipation prevention, proper hygiene, and awareness of symptoms provide robust protection against UTIs during semaglutide treatment. These are not complicated or expensive interventions. They are simple habits that become second nature within a few weeks.
The even better news is that the long-term trajectory favors improved urinary health. As side effects diminish and weight loss benefits accumulate, the indirect risk factors that drive UTI vulnerability decrease while protective factors increase. For most people, the initial adjustment period represents the highest-risk window, and it passes relatively quickly with proper management.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Understanding the complete picture, not just the headline, is what separates informed users from anxious ones.
Frequently asked questions
Can semaglutide directly cause a urinary tract infection?
No. Clinical trials including the SUSTAIN and STEP programs did not identify UTIs as a significant adverse event associated with semaglutide. Unlike SGLT-2 inhibitors, semaglutide does not alter urinary glucose levels or create conditions that directly promote bacterial growth in the urinary tract. However, indirect factors like dehydration from gastrointestinal side effects can increase susceptibility in some individuals.
Should I stop taking semaglutide if I get a UTI?
For uncomplicated lower urinary tract infections, there is generally no reason to stop semaglutide. Standard antibiotics work normally alongside GLP-1 therapy with no significant interactions. For complicated infections involving the kidneys, fever, or severe dehydration, your healthcare provider may recommend a temporary pause until the infection resolves. See our guide on semaglutide treatment interruptions for more details.
How much water should I drink on semaglutide to prevent UTIs?
Aim for 2.5 to 3.5 liters daily, with the higher end recommended during dose escalation when gastrointestinal side effects are most intense. Monitor urine color as a real-time hydration indicator. Pale yellow to nearly clear urine suggests adequate hydration. Dark yellow or amber urine indicates you need to increase fluid intake immediately.
Does the type of semaglutide formulation affect UTI risk?
The active ingredient works identically regardless of formulation. Compounded semaglutide, brand-name injections, and oral formulations all carry the same indirect risk profile related to gastrointestinal side effects and dehydration. However, oral formulations may produce different side effect patterns that could affect hydration status differently than injectable versions.
Can cranberry juice prevent UTIs while on semaglutide?
Unsweetened cranberry juice or cranberry supplements containing at least 36mg of proanthocyanidins daily show modest evidence for UTI prevention by preventing E. coli adhesion to the bladder wall. Avoid sweetened cranberry cocktails, which contain excessive sugar. Cranberry products work best as part of a comprehensive prevention strategy that includes adequate hydration, not as a standalone solution.
Is the UTI risk higher during semaglutide dose escalation?
Yes. Dose escalation is when gastrointestinal side effects peak, creating the highest risk of dehydration and associated UTI vulnerability. The standard dose escalation schedule gradually increases doses to minimize side effects, but the first 8-16 weeks typically require the most vigilant hydration and prevention efforts.
Can probiotics help prevent UTIs during semaglutide treatment?
Probiotics containing Lactobacillus strains, particularly L. rhamnosus and L. reuteri, show evidence for UTI prevention by supporting the vaginal and urinary microbiomes. Given that semaglutide can alter gut microbiome composition, probiotic supplementation may provide additional protection. GLP-1 specific probiotics are designed to address the unique microbiome changes associated with this medication class.
Does semaglutide interact with UTI antibiotics?
There are no clinically significant drug interactions between semaglutide and common UTI antibiotics including nitrofurantoin, trimethoprim-sulfamethoxazole, and fosfomycin. However, some antibiotics can cause gastrointestinal side effects that may compound semaglutide-related nausea. Inform your prescriber that you are taking semaglutide so they can choose the most tolerable antibiotic option for your situation.
External resources
PMC - Saving muscle while losing weight: strategies for GLP-1 related drugs
American Urological Association - Semaglutide and urinary tract implications
Annals of Internal Medicine - SGLT-2 inhibitors UTI comparison with GLP-1 agonists
Diabetes Obesity Metabolism - Changes in lean body mass with GLP-1 therapies
In case I do not see you, good afternoon, good evening, and good night. May your hydration stay consistent, your urinary health stay strong, and your semaglutide results stay on track.